Transcriptomic and Proteomic Landscape of Sugarcane Response to Biotic and Abiotic Stressors
Abstract
:1. Introduction
2. Sugarcane Transcriptomic Analysis
2.1. Transcriptomics of Sugarcane Response to Biotic Stress
2.1.1. Sugarcane Diseases
2.1.2. Plant Growth-Promoting Rhizobacteria
2.2. Transcriptomics of Sugarcane Response to Abiotic Stress
2.2.1. Nutrient Deficiency
2.2.2. Aluminum Toxicity
2.2.3. Drought Stress
2.2.4. Extreme Temperatures
2.3. Transcriptomics of Sugarcane Response to Exogenous Phytohormone Application
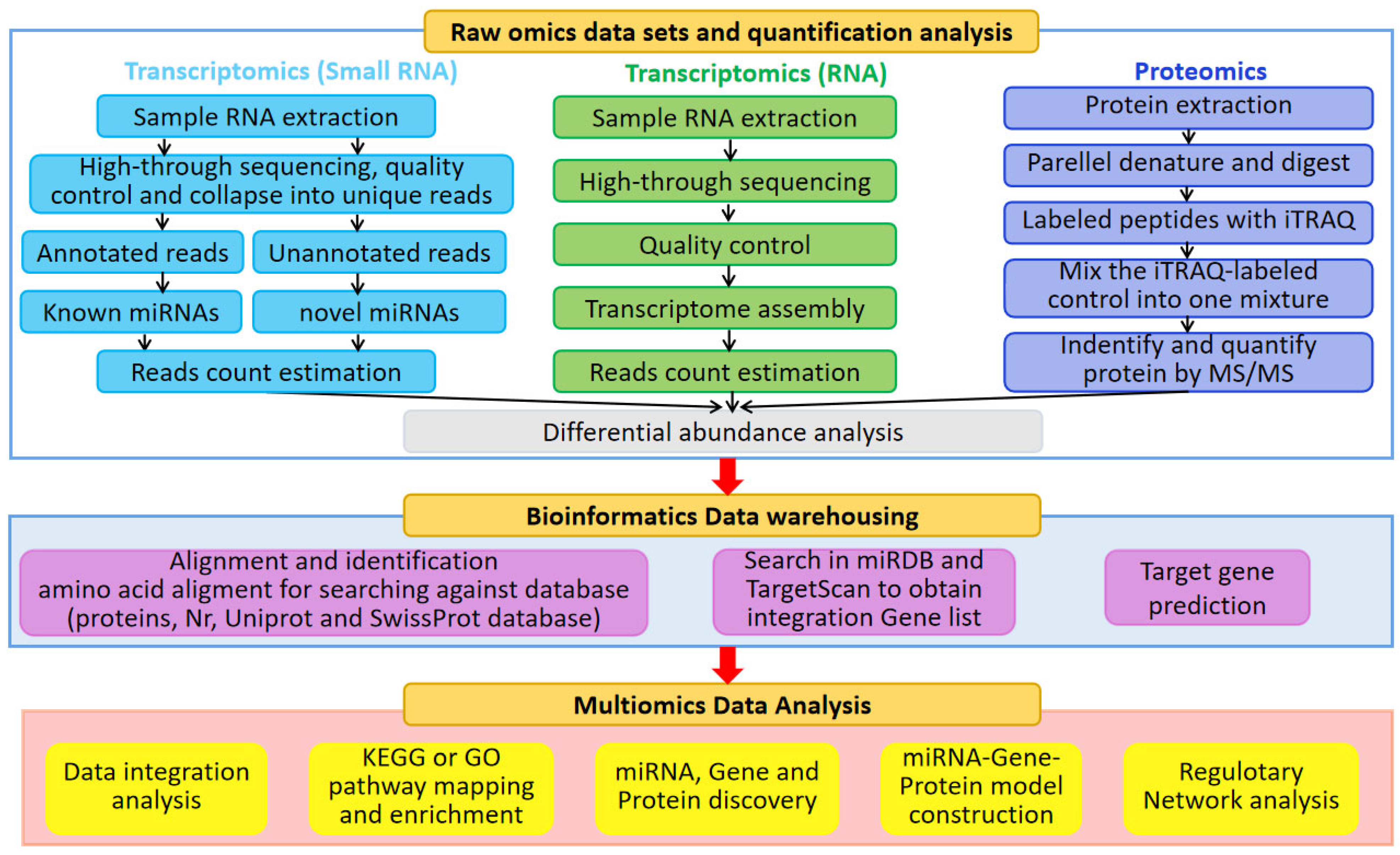
3. Role of Transcriptomics in Studies of Environmentally Stressed Sugarcane
4. Sugarcane Proteomic Analysis
4.1. Proteomic Analyses of Sugarcane Subjected to Biotic and Abiotic Stressors
4.1.1. Sugarcane Diseases
4.1.2. Drought and Salt Stress
4.1.3. Ethylene Treatment
4.1.4. Putrescine Treatment
5. Role of Proteomics in Studies of Environmentally Stressed Sugarcane
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mahajan, S.; Tuteja, N. Cold, salinity and drought stresses: An overview. Arch. Biochem. Biophys. 2005, 444, 139–158. [Google Scholar] [CrossRef]
- Augustine, S.M.; Narayan, J.A.; Syamaladevi, D.P.; Appunu, C.; Chakravarthi, M.; Ravichandran, V.; Tuteja, N.; Subramonian, N. Overexpression of EaDREB2 and pyramiding of EaDREB2 with the pea DNA helicase gene (PDH45) enhance drought and salinity tolerance in sugarcane (Saccharum spp. hybrid). Plant Cell Rep. 2015, 34, 247–263. [Google Scholar] [CrossRef]
- Verma, K.K.; Singh, P.; Song, X.P.; Malviya, M.K.; Singh, R.K.; Chen, G.L.; Solomon, S.; Li, Y.R. Mitigating climate change for sugarcane improvement: Role of silicon in alleviating abiotic stresses. Sugar Tech 2020, 22, 741–749. [Google Scholar] [CrossRef]
- Verma, K.K.; Liu, X.H.; Wu, K.C.; Singh, R.K.; Song, Q.Q.; Malviya, M.K.; Song, X.P.; Singh, P.; Verma, C.L.; Li, Y.R. The impact of silicon on photosynthetic and biochemical responses of sugarcane under different soil moisture levels. Silicon 2020, 12, 1355–1367. [Google Scholar] [CrossRef]
- Neumann, P.M. Coping mechanisms for crop plants in drought-prone environments. Ann. Bot. 2008, 101, 901–907. [Google Scholar] [CrossRef]
- Verma, K.K.; Song, X.P.; Verma, C.L.; Malviya, M.K.; Guo, D.J.; Rajput, V.D.; Sharma, A.; Wei, K.J.; Chen, G.L.; Solomon, S.; et al. Predication of photosynthetic leaf gas exchange of sugarcane (Saccharum spp.) leaves in response to leaf positions to foliar spray of potassium salt of active phosphorus under limited water irrigation. ACS Omega 2021, 6, 2396–2409. [Google Scholar] [CrossRef]
- Cho, E.K.; Cho, Y.J. A nuclear-localized HSP70 confers thermo protective activity and drought-stress tolerance on plants. Biotechnol. Lett. 2009, 31, 597–606. [Google Scholar] [CrossRef]
- Smith, A.; Stitt, M. Coordination of carbon supply and plant growth. Plant Cell Environ. 2007, 30, 1126–1149. [Google Scholar] [CrossRef]
- De Souza, A.P.; Grandis, A.; Arenque-Musa, B.C.; Buckeridge, M.S. Diurnal variation in gas exchange and nonstructural carbohydrates throughout sugarcane development. Funct. Plant Biol. 2018, 45, 865–876. [Google Scholar] [CrossRef]
- Chen, G.-L.; Zheng, F.-J.; Lin, B.; Lao, S.-B.; He, J.; Huang, Z.; Zeng, Y.; Sun, J.; Verma, K.K. Phenolic and volatile compounds in the production of sugarcane vinegar. ACS Omega 2020, 5, 30587. [Google Scholar] [CrossRef]
- Waclawovsky, A.J.; Sato, P.M.; Lembke, C.G.; Moore, P.H.; Souza, G.M. Sugarcane for bioenergy production: An assessment of yield and regulation of sucrose content. Plant Biotechnol. J. 2010, 8, 263–276. [Google Scholar] [CrossRef]
- Ali, A.; Pan, Y.-B.; Wang, Q.-N.; Wang, J.-D.; Chen, J.-L.; Gao, S.-J. Genetic diversity and population structure analysis of Saccharum and Erianthus genera using microsatellite (SSR) markers. Sci. Rep. 2019, 9, 395. [Google Scholar] [CrossRef]
- De Souza, A.P.; Grandis, A.; Leite, D.C.C.; Buckeridge, M.S. Sugarcane as a bioenergy source: History, performance, and perspectives for second-generation bioethanol. BioEnergy Res. 2014, 7, 24–35. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, X.; Tang, H.; Zhang, Q.; Hua, X.; Ma, X.; Zhu, F.; Jones, T.; Zhu, X.; Bowers, J. Allele-defined genome of the autopolyploid sugarcane Saccharum spontaneum L. Nat. Genet. 2018, 50, 1565. [Google Scholar] [CrossRef]
- Garsmeur, O.; Droc, G.; Antonise, R.; Grimwood, J.; Potier, B.; Aitken, K.; Jenkins, J.; Martin, G.; Charron, C.; Hervouet, C. A mosaic monoploid reference sequence for the highly complex genome of sugarcane. Nat. Commun. 2018, 9, 2638. [Google Scholar] [CrossRef]
- Mustafa, G.; Joyia, F.A.; Anwar, S.; Parvaiz, A.; Khan, M.S. Biotechnological interventions for the improvement of sugarcane crop and sugar production. In Sugarcane-Technology and Research; IntechOpen: London, UK, 2018; pp. 113–138. [Google Scholar]
- Barnabas, L.; Ashwin, N.M.; Kaverinathan, K.; Trentin, A.R.; Pivato, M.; Sundar, A.R.; Malathi, P.; Viswanathan, R.; Rosana, O.B.; Neethukrishna, K.; et al. Proteomic analysis of a compatible interaction between sugarcane and Sporisorium scitamineum. Proteomics 2016, 16, 1111–1122. [Google Scholar] [CrossRef]
- Kaur, L.; Dharshini, S.; Ram, B.; Appunu, C. Sugarcane Genomics and Transcriptomics. In Sugarcane Biotechnology: Challenges and Prospects; Springer: Cham, Switzerland, 2017; pp. 13–32. [Google Scholar]
- Gong, Z.; Xiong, L.; Shi, H.; Yang, S.; Herrera-Estrella, L.R.; Xu, G.; Chao, D.Y.; Li, J.; Wang, P.Y.; Qin, F.; et al. Plant abiotic stress response and nutrient use efficiency. Sci. China Life Sci. 2020, 63, 635–674. [Google Scholar] [CrossRef]
- Morton, M.J.L.; Awlia, M.; Al-Tamimi, N.; Saade, S.; Pailles, Y.; Negrão, S.; Tester, M. Salt stress under the scalpel-dissecting the genetics of salt tolerance. Plant J. 2019, 97, 148–163. [Google Scholar] [CrossRef]
- Farooq, M.A.; Ali, S.; Hameed, A.; Bharwana, S.A.; Rizwan, M.; Ishaque, W.; Farid, M.; Mahmood, K.; Iqbal, Z. Cadmium stress in cotton seedlings: Physiological, photosynthesis and oxidative damages alleviated by glycinebetaine. S. Afr. J. Bot. 2016, 104, 61–68. [Google Scholar] [CrossRef]
- Eleftheriou, E.P.; Adamakis, I.D.S.; Panteris, E.; Fatsiou, M. Chromium-induced ultrastructural changes and oxidative stress in roots of Arabidopsis thaliana. Int. J. Med. Sci. 2015, 16, 15852–15871. [Google Scholar] [CrossRef]
- Chu, N.; Zhou, J.R.; Fu, H.Y.; Huang, M.T.; Zhang, H.L.; Gao, S.J. Global gene responses of resistant and susceptible sugarcane cultivars to Acidovorax avenae subsp. avenae identified using comparative transcriptome analysis. Microorganisms 2019, 8, 10. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Y.; Li, C.; Song, X.; Lei, J.; Gao, Y.; Liang, Q. Comparative transcriptome profiling of resistant and susceptible sugarcane genotypes in response to the airborne pathogen Fusarium verticillioides. Mol. Biol. Rep. 2019, 46, 3777–3789. [Google Scholar] [CrossRef]
- Ntambo, M.S.; Meng, J.Y.; Rott, P.C.; Henry, R.J.; Zhang, H.L.; Gao, S.J. Comparative transcriptome profiling of resistant and susceptible sugarcane cultivars in response to infection by Xanthomonas albilineans. Int. J. Mol. Sci. 2019, 20, 6138. [Google Scholar] [CrossRef]
- Rajput, M.A.; Rajput, N.A.; Syed, R.N.; Lodhi, A.M.; Que, Y.X. Sugarcane smut: Current knowledge and the way forward for management. J. Fungi 2021, 7, 1095. [Google Scholar] [CrossRef]
- Marguerat, S.; Bähler, J. RNA-seq: From technology to biology. Cell Mol. Life Sci. 2010, 67, 569–579. [Google Scholar] [CrossRef]
- Huang, D.-L.; Gao, Y.-J.; Gui, Y.-Y.; Chen, Z.-L.; Qin, C.-X.; Wang, M.; Liao, Q.; Yang, L.-T.; Li, Y.-R. Transcriptome of high sucrose sugarcane variety GT35. Sugar Tech 2016, 18, 520–528. [Google Scholar] [CrossRef]
- Debat, H.J.; Ducasse, D.A. Plant microRNAs: Recent advances and future challenges. Plant Mol. Biol. Rep. 2014, 32, 1257–1269. [Google Scholar] [CrossRef]
- Nogoy, F.M.; Niño, M.C.; Song, J.Y.; Jung, Y.J.; Kang, K.K.; Nou, I.; Cho, Y.G. Plant microRNAs in molecular breeding. Plant Biotechnol. Rep. 2018, 12, 15–25. [Google Scholar] [CrossRef]
- Wu, Q.; Xu, L.; Guo, J.; Su, Y.; Que, Y. Transcriptome profile analysis of sugarcane responses to Sporisorium scitaminea infection using Solexa sequencing technology. Biomed. Res. Int. 2013, 2013, 298920. [Google Scholar] [CrossRef]
- Que, Y.X.; Lin, J.W.; Song, X.X.; Xu, L.P.; Chen, R.K. Differential gene expression in sugarcane in response to challenge by fungal pathogen Ustilago scitaminea revealed by cDNA-AFLP. J. Biomed. Biotechnol. 2011, 2011, 160934. [Google Scholar]
- Que, Y.X.; Su, Y.C.; Guo, J.L.; Wu, Q.B.; Xu, L.P. A global view of transcriptome dynamics during Sporisorium scitamineum challenge in sugarcane by RNA-seq. PLoS ONE 2014, 9, e106476. [Google Scholar] [CrossRef] [PubMed]
- Que, Y.X.; Xu, L.P.; Wu, Q.B.; Liu, Y.F.; Ling, H.; Liu, Y.H.; Zhang, Y.Y.; Guo, J.L.; Su, Y.C.; Chen, J.B.; et al. Genome sequencing of Sporisorium scitamineum provides insights into the pathogenic mechanisms of sugarcane smut. BMC Genom. 2014, 15, 996. [Google Scholar]
- Huang, N.; Ling, H.; Su, Y.; Liu, F.; Xu, L.; Su, W.; Wu, Q.; Guo, J.; Gao, S.; Que, Y. Transcriptional analysis identifies major pathways as response components to Sporisorium scitamineum stress in sugarcane. Gene 2018, 678, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Ling, H.; Fu, X.Q.; Huang, N.; Zhong, Z.F.; Su, W.H.; Lin, W.X.; Cui, H.T.; Que, Y.X. A sugarcane smut fungus effector simulates the host endogenous elicitor peptide to suppress plant immunity. New Phytol. 2022, 233, 919–933. [Google Scholar] [CrossRef]
- Wu, Q.B.; Pan, Y.-B.; Su, Y.C.; Zou, W.H.; Xu, F.; Sun, T.T.; Grisham, M.P.; Yang, S.L.; Xu, L.P.; Que, Y.X. WGCNA identifies a comprehensive and dynamic gene co-expression network that associates with smut resistance in sugarcane. Int. J. Mol. Sci. 2022, 23, 10770. [Google Scholar] [CrossRef]
- Wu, Q.B.; Su, Y.C.; Pan, Y.-B.; Xu, F.; Zou, W.T.; Que, B.B.; Lin, P.X.; Sun, T.T.; Grisham, M.P.; Xu, L.P.; et al. Genetic identification of SNP markers and candidate genes associated with sugarcane smut resistance using BSR-Seq. Front. Plant Sci. 2022, 13, 1035266. [Google Scholar] [CrossRef]
- Su, Y.C.; Zhang, Y.Y.; Huang, N.; Liu, F.; Su, W.H.; Xu, L.P.; Ahmad, W.; Wu, Q.B.; Guo, J.L.; Que, Y.X. Small RNA sequencing reveals a role for sugarcane miRNAs and their targets in response to Sporisorium scitamineum infection. BMC Genom. 2017, 18, 325. [Google Scholar]
- Su, Y.; Xiao, X.; Ling, H.; Huang, N.; Liu, F.; Su, W.; Zhang, Y.; Xu, L.; Muhammad, K.; Que, Y. A dynamic degradome landscape on miRNAs and their predicted targets in sugarcane caused by Sporisorium scitamineum stress. BMC Genom. 2019, 20, 57. [Google Scholar] [CrossRef]
- Santa Brigida, A.B.; Rojas, C.A.; Grativol, C.; de Armas, E.M.; Entenza, J.O.; Thiebaut, F.; Lima, M.F.; Farrinelli, L.; Hemerly, A.S.; Lifschitz, S.; et al. Sugarcane transcriptome analysis in response to infection caused by Acidovorax avenae subsp. avenae. PLoS ONE 2016, 11, e0166473. [Google Scholar] [CrossRef]
- Su, Y.C.; Peng, Q.; Ling, H.; You, C.H.; Wu, Q.B.; Xu, L.P.; Que, Y.X. Systematic identification of miRNA-regulatory networks unveils their potential roles in sugarcane response to Sorghum mosaic virus infection. BMC Plant Biol. 2022, 22, 247. [Google Scholar] [CrossRef]
- Ling, H.; Huang, N.; Wu, Q.B.; Su, Y.C.; Peng, Q.; Ahmad, W.; Gao, S.W.; Su, W.H.; Que, Y.X.; Xu, L.P. Transcriptional insights into the sugarcane-Sorghum mosaic virus interaction. Trop. Plant Biol. 2018, 11, 163–176. [Google Scholar] [CrossRef]
- Malviya, M.K.; Li, C.N.; Solanki, M.K.; Singh, R.K.; Htun, R.; Singh, P.; Verma, K.K.; Yang, L.T.; Li, Y.R. Comparative analysis of sugarcane root transcriptome in response to the plant growth-promoting Burkholderia anthina MYSP113. PLoS ONE 2020, 15, e0231206. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Ling, Q.; Fan, L.; Li, Y.; Hu, F.; Chen, J.; Huang, Z.; Deng, H.; Li, Q.; Qi, Y. Transcriptome profiling of sugarcane roots in response to low potassium stress. PLoS ONE 2015, 10, e0126306. [Google Scholar] [CrossRef] [PubMed]
- Rosa-Santos, T.M.; Silva, R.G.D.; Kumar, P.; Kottapalli, P.; Crasto, C.; Kottapalli, K.R.; França, S.C.; Zingaretti, S.M. Molecular mechanisms underlying sugarcane response to aluminum stress by RNA-Seq. Int. J. Mol. Sci. 2020, 21, 7934. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.G.D.; Rosa-Santos, T.M.; França, S.C.; Kottapalli, P.; Kottapalli, K.R.; Zingaretti, S.M. Microtranscriptome analysis of sugarcane cultivars in response to aluminum stress. PLoS ONE 2019, 14, e0217806. [Google Scholar] [CrossRef]
- Li, M.; Liang, Z.; Zeng, Y.; Jing, Y.; Wu, K.; Liang, J.; He, S.; Wang, G.; Mo, Z.; Tan, F. De novo analysis of transcriptome reveals genes associated with leaf abscission in sugarcane (Saccharum officinarum L.). BMC Genom. 2016, 17, 195. [Google Scholar] [CrossRef]
- Belesini, A.; Carvalho, F.; Telles, B.; De Castro, G.; Giachetto, P.; Vantini, J.; Carlin, S.; Cazetta, J.; Pinheiro, D.; Ferro, M. De novo transcriptome assembly of sugarcane leaves submitted to prolonged water-deficit stress. Genet. Mol. Res. 2017, 16, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Selvi, A.; Devi, K.; Manimekalai, R.; Prathima, P.T. Comparative analysis of drought-responsive transcriptomes of sugarcane genotypes with differential tolerance to drought. 3 Biotech 2020, 10, 236. [Google Scholar] [CrossRef]
- Nawae, W.; Shearman, J.R.; Tangphatsornruang, S.; Punpee, P.; Yoocha, T.; Sangsrakru, D.; Naktang, C.; Sonthirod, C.; Wirojsirasak, W.; Ukoskit, K.; et al. Differential expression between drought-tolerant and drought-sensitive sugarcane under mild and moderate water stress as revealed by a comparative analysis of leaf transcriptome. Peer J. 2020, 8, e9608. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, R.; Ou, H.; Gui, Y.; Wei, J.; Zhou, H.; Tan, H.; Li, Y. Comprehensive transcriptome analysis reveals genes in response to water deficit in the leaves of Saccharum narenga (Nees ex Steud.) hack. BMC Plant Biol. 2018, 18, 250. [Google Scholar] [CrossRef]
- Li, C.; Wang, Z.; Nong, Q.; Lin, L.; Xie, J.; Mo, Z.; Huang, X.; Song, X.; Malviya, M.K.; Solanki, M.K.; et al. Physiological changes and transcriptome profiling in Saccharum spontaneum L. leaf under water stress and re-watering conditions. Sci. Rep. 2021, 11, 5525. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, T.H.; Gentile, A.; Vilela, R.D.; Costa, G.G.; Dias, L.I.; Endres, L.; Menossi, M. microRNAs associated with drought response in the bioenergy crop sugarcane (Saccharum spp.). PLoS ONE 2012, 7, e46703. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Chen, T.; Qin, X.; Wu, H.; Khan, M.A.; Lin, W. Identification of microRNA families expressed in sugarcane leaves subjected to drought stress and targets thereof. Pak. J. Agric. Sci. 2014, 51, 925–934. [Google Scholar]
- Raju, G.; Shanmugam, K.; Kasirajan, L. High-throughput sequencing reveals genes associated with high-temperature stress tolerance in sugarcane. 3 Biotech 2020, 10, 198. [Google Scholar] [CrossRef]
- Chen, Z.; Qin, C.; Wang, M.; Liao, F.; Liao, Q.; Liu, X.; Li, Y.; Lakshmanan, P.; Long, M.; Huang, D. Ethylene-mediated improvement in sucrose accumulation in ripening sugarcane involves increased sink strength. BMC Plant Biol. 2019, 19, 285. [Google Scholar] [CrossRef]
- Chandra, A.; Roopendra, K.; Verma, I. Transcriptome analysis of the effect of GA(3) in sugarcane culm. 3 Biotech 2019, 9, 376. [Google Scholar] [CrossRef]
- Li, A.; Lakshmanan, P.; He, W.; Tan, H.; Liu, L.; Liu, H.; Liu, J.; Huang, D.; Chen, Z. Transcriptome profiling provides molecular insights into auxin-induced adventitious root formation in sugarcane (Saccharum spp. interspecific hybrids) microshoots. Plants 2020, 9, 931. [Google Scholar]
- Tripathi, P.; Chandra, A.; Prakash, J. Physio-biochemical assessment and expression analysis of genes associated with drought tolerance in sugarcane (Saccharum spp. hybrids) exposed toGA3 at grand growth stage. Plant Biol. 2019, 21, 45–53. [Google Scholar] [CrossRef]
- Cardoso-Silva, C.B.; Costa, E.A.; Mancini, M.C.; Balsalobre, T.W.A.; Canesin, L.E.C.; Pinto, L.R.; Carneiro, M.S.; Garcia, A.A.F.; de Souza, A.P.; Vicentini, R. De novo assembly and transcriptome analysis of contrasting sugarcane varieties. PLoS ONE 2014, 9, e88462. [Google Scholar] [CrossRef]
- Xu, S.; Wang, J.; Shang, H.; Huang, Y.; Yao, W.; Chen, B.; Zhang, M. Transcriptomic characterization and potential marker development of contrasting sugarcane cultivars. Sci. Rep. 2018, 8, 1683. [Google Scholar] [CrossRef]
- Wang, J.; Roe, B.; Macmil, S.; Yu, Q.; Murray, J.E.; Tang, H.; Chen, C.; Najar, F.; Wiley, G.; Bowers, J. Microcollinearity between autopolyploid sugarcane and diploid sorghum genomes. BMC Genom. 2010, 11, 261. [Google Scholar] [CrossRef] [PubMed]
- Grivet, L.; D’Hont, A.; Dufour, P.; Hamon, P.; Roques, D.; Glaszmann, J.-C. Comparative genome mapping of sugar cane with other species within the Andropogoneae tribe. Heredity 1994, 73, 500. [Google Scholar] [CrossRef]
- Song, S.; Qi, T.; Huang, H.; Ren, Q.; Wu, D.; Chang, C.; Peng, W.; Liu, Y.; Peng, J.; Xie, D. The jasmonate-ZIM domain proteins interact with the R2R3-MYB transcription factors MYB21 and MYB24 to affect jasmonate-regulated stamen development in Arabidopsis. Plant Cell 2011, 23, 1000–1013. [Google Scholar] [CrossRef] [PubMed]
- Sakuraba, Y.; Kim, Y.-S.; Han, S.-H.; Lee, B.-D.; Paek, N.-C. The Arabidopsis transcription factor NAC016 promotes drought stress responses by repressing AREB1 transcription through a trifurcate feed-forward regulatory loop involving NAP. Plant Cell 2015, 27, 1771–1787. [Google Scholar] [CrossRef]
- Guo, J.; Ling, H.; Ma, J.; Chen, Y.; Su, Y.; Lin, Q.; Gao, S.; Wang, H.; Que, Y.; Xu, L. A sugarcane R2R3-MYB transcription factor gene is alternatively spliced during drought stress. Sci. Rep. 2017, 7, 41922. [Google Scholar] [CrossRef]
- Da Silva, M.D.; de Oliveira Silva, R.L.; Neto, J.R.C.F.; Benko-Iseppon, A.M.; Kido, E.A. Genotype-dependent regulation of drought-responsive genes in tolerant and sensitive sugarcane cultivars. Gene 2017, 633, 17–27. [Google Scholar] [CrossRef]
- Sreenivasulu, N.; Sopory, S.K.; Kavi Kishor, P.B. Deciphering the regulatory mechanisms of abiotic stress tolerance in plants by genomic approaches. Gene 2007, 388, 1–13. [Google Scholar] [CrossRef]
- Selvarajan, D.; Mohan, C.; Dhandapani, V.; Nerkar, G.; Jayanarayanan, A.N.; Mohanan, M.V.; Murugan, N.; Kaur, L.; Chennapp, M.; Kumar, R. Differential gene expression profiling through transcriptome approach of Saccharum spontaneum L. under low temperature stress reveals genes potentially involved in cold acclimation. 3 Biotech 2018, 8, 195. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Gao, S.; Su, Y.; Lin, Z.; Guo, J.; Li, M.; Wang, Z.; Que, Y.; Xu, L. Transcripts and low nitrogen tolerance: Regulatory and metabolic pathways in sugarcane under low nitrogen stress. Environ. Exp. Bot. 2019, 163, 97–111. [Google Scholar] [CrossRef]
- Yang, Y.T.; Yu, Q.; Yang, Y.Y.; Su, Y.C.; Ahmad, W.; Guo, J.L.; Gao, S.W.; Xu, L.P.; Que, Y.X. Identification of cold-related miRNAs in sugarcane by small RNA sequencing and functional analysis of a cold inducible ScmiR393 to cold stress. Environ. Exp. Bot. 2018, 155, 464–476. [Google Scholar] [CrossRef]
- Xu, P.; Huecksteadt, T.P.; Hoidal, J.R. Molecular cloning and characterization of the human xanthine dehydrogenase gene (XDH). Genomics 1996, 34, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, F.T.S.; De Rosa, V.E., Jr.; Menossi, M.; Ulian, E.C.; Arruda, P. RNA Expression profiles and data mining of sugarcane response to low temperature. Plant Physiol. 2003, 132, 1811–1824. [Google Scholar] [CrossRef]
- Azevedo, R.A.; Carvalho, R.F.; Cia, M.C.; Gratao, P.L. Sugarcane under pressure: An overview of biochemical and physiological studies of abiotic stress. Trop. Plant Biol. 2011, 4, 42–51. [Google Scholar] [CrossRef]
- Rodrigues, F.A.; Laia, M.L.; Zingaretti, S.M. Analysis of gene expression profiles under water stress in tolerant and sensitive sugarcane plants. Plant Sci. 2009, 176, 286–302. [Google Scholar] [CrossRef]
- Nishiyama, M.Y., Jr.; Ferreira, S.S.; Tang, P.-Z.; Becker, S.; Poertner-Taliana, A.; Souza, G.M. Full-length enriched cDNA libraries and ORFeome analysis of sugarcane hybrid and ancestor genotypes. PLoS ONE 2014, 9, e107351. [Google Scholar] [CrossRef]
- Manners, J.M.; Casu, R.E. Transcriptome analysis and functional genomics of sugarcane. Trop. Plant Biol. 2011, 4, 9–21. [Google Scholar] [CrossRef]
- Mattiello, L.; Riaño-Pachón, D.M.; Martins, M.C.M.; da Cruz, L.P.; Bassi, D.; Marchiori, P.E.R.; Ribeiro, R.V.; Labate, M.T.V.; Labate, C.A.; Menossi, M. Physiological and transcriptional analyses of developmental stages along sugarcane leaf. BMC Plant Biol. 2015, 15, 300. [Google Scholar] [CrossRef]
- Kasirajan, L.; Hoang, N.V.; Furtado, A.; Botha, F.C.; Henry, R.J. Transcriptome analysis highlights key differentially expressed genes involved in cellulose and lignin biosynthesis of sugarcane genotypes varying in fiber content. Sci. Rep. 2018, 8, 11612. [Google Scholar] [CrossRef]
- Li, C.; Nong, Q.; Solanki, M.K.; Liang, Q.; Xie, J.; Liu, X.; Li, Y.; Wang, W.; Yang, L.; Li, Y.R. Differential expression profiles and pathways of genes in sugarcane leaf at elongation stage in response to drought stress. Sci. Rep. 2016, 6, 25698. [Google Scholar] [CrossRef]
- Aslam, B.; Basit, M.; Nisar, M.A.; Khurshid, M.; Rasool, M.H. Proteomics: Technologies and their applications. J. Chromatogr. Sci. 2017, 55, 182–196. [Google Scholar] [CrossRef]
- Su, Y.; Xu, L.; Wang, Z.; Peng, Q.; Yang, Y.; Chen, Y.; Que, Y. Comparative proteomics reveals that central metabolism changes are associated with resistance against Sporisorium scitamineum in sugarcane. BMC Genom. 2016, 17, 800. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Song, Q.Q.; Singh, R.K.; Li, H.B.; Solanki, M.K.; Malviya, M.K.; Verma, K.K.; Yang, L.T.; Li, Y.R. Proteomic analysis of the resistance mechanisms in sugarcane during Sporisorium scitamineum infection. Int. J. Mol. Sci. 2019, 20, 569. [Google Scholar] [CrossRef] [PubMed]
- Que, Y.X.; Xu, L.P.; Lin, J.W.; Ruan, M.H.; Zhang, M.Q.; Chen, R.K. Differential protein expression in sugarcane during sugarcane-Sporisorium scitamineum interaction revealed by 2-DE and MALDI-TOF-TOF/MS. Comp. Funct. Genom. 2011, 2011, 989016. [Google Scholar] [CrossRef]
- Meng, J.Y.; Ntambo, M.S.; Rott, P.C.; Fu, H.Y.; Huang, M.T.; Zhang, H.L.; Gao, S.J. Identification of differentially expressed proteins in sugarcane in response to infection by Xanthomonas albilineans using iTRAQ quantitative proteomics. Microorganisms 2020, 8, 76. [Google Scholar] [CrossRef]
- Salvato, F.; Loziuk, P.; Kiyota, E.; Daneluzzi, G.S.; Araújo, P.; Muddiman, D.C.; Mazzafera, P. Label-free quantitative proteomics of enriched nuclei from sugarcane (Saccharum spp.) stems in response to drought stress. Proteomics 2019, 19, e1900004. [Google Scholar]
- Passamani, L.Z.; Barbosa, R.R.; Reis, R.S.; Heringer, A.S.; Rangel, P.L.; Santa-Catarina, C.; Grativol, C.; Veiga, C.F.M.; Souza-Filho, G.A.; Silveira, V. Salt stress induces changes in the proteomic profile of micropropagated sugarcane shoots. PLoS ONE 2017, 12, e0176076. [Google Scholar] [CrossRef]
- Chiconato, D.A.; de Santana Costa, M.G.; Balbuena, T.S.; Munns, R.; Dos Santos, D.M.M. Proteomic analysis of young sugarcane plants with contrasting salt tolerance. Funct. Plant Biol. 2021, 48, 588–596. [Google Scholar] [CrossRef]
- Qin, C.-X.; Chen, Z.-L.; Wang, M.; Li, A.-M.; Liao, F.; Li, Y.-R.; Wang, M.-Q.; Long, M.-H.; Lakshmanan, P.; Huang, D.-L. Identification of proteins and metabolic networks associated with sucrose accumulation in sugarcane (Saccharum spp. interspecific hybrids). J. Plant Interact. 2021, 16, 166–178. [Google Scholar] [CrossRef]
- Heringer, A.S.; Reis, R.S.; Passamani, L.Z.; de Souza-Filho, G.A.; Santa-Catarina, C.; Silveira, V. Comparative proteomics analysis of the eect of combined red and blue lights on sugarcane somatic embryogenesis. Acta Physiol. Plant 2017, 39, 52. [Google Scholar] [CrossRef]
- Singh, N.P.; Shami, V. Abiotic stress tolerance in sugarcane using genomics and proteomics techniques. In Biotechnology to Enhance Sugarcane Productivity and Stress Tolerance; CRC Press: Boca Raton, FL, USA, 2018; pp. 97–114. [Google Scholar]
- Barnabas, L.; Ramadas, A.; Amalraj, R.S.; Palaniyandi, M.; Rasappa, V. Sugarcane proteomics: An update on current status, challenges, and future prospects. Proteomics 2015, 15, 1658–1670. [Google Scholar] [CrossRef]
- Velez-Bermúdez, I.C.; Wen, T.-N.; Lan, P.; Schmidt, W. Isobaric tag for relative and absolute quantitation (iTRAQ)-based protein profiling in plants. In Plant Proteostasis; Springer: New York, NY, USA, 2016; pp. 213–221. [Google Scholar]
- Khueychai, S.; Jangpromma, N.; Daduang, S.; Jaisil, P.; Lomthaisong, K.; Dhiravisit, A.; Klaynongsruang, S. Comparative proteomic analysis of leaves, leaf sheaths, and roots of drought-contrasting sugarcane cultivars in response to drought stress. Acta Physiol. Plant 2015, 37, 88. [Google Scholar] [CrossRef]
- Ngamhui, N.O.; Akkasaeng, C.; Zhu, Y.J.; Tantisuwichwong, N.; Roytrakul, S.; Sansayawichai, T. Differentially expressed proteins in sugarcane leaves in response to water deficit stress. Plant Omics 2012, 5, 365. [Google Scholar]
- Murad, A.M.; Molinari, H.B.C.; Magalhães, B.S.; Franco, A.C.; Takahashi, F.S.C.; Franco, O.L.; Quirino, B.F. Physiological and proteomic analyses of Saccharum spp. grown under salt stress. PLoS ONE 2014, 9, e98463. [Google Scholar] [CrossRef] [PubMed]
- Augustine, S.M.; Cherian, A.v.; Syamaladevi, D.P.; Subramonian, N. Erianthus arundinaceus HSP70 (EaHSP70) Acts as a key regulator in the formation of anisotropic interdigitation in sugarcane (Saccharum spp. hybrid) in response to drought stress. Plant Cell Physiol. 2015, 56, 2368–2380. [Google Scholar] [CrossRef]
- Zhao, D.; Li, Y.-R. Climate change and sugarcane production: Potential impact and mitigation strategies. Int. J. Agron. 2015, 2015, 547386. [Google Scholar] [CrossRef]
- Fonseca, J.G.; Calderaneran Pases, M.J.; de Moraes, F.E.; Cataldi, T.R.; Jamet, E.; Labate, C.A. Cell wall proteome of sugarcane young and mature leaves and stems. Proteomics 2018, 18, 1700129. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, A.-M.; Liao, F.; Wang, M.; Chen, Z.-L.; Qin, C.-X.; Huang, R.-Q.; Verma, K.K.; Li, Y.-R.; Que, Y.-X.; Pan, Y.-Q.; et al. Transcriptomic and Proteomic Landscape of Sugarcane Response to Biotic and Abiotic Stressors. Int. J. Mol. Sci. 2023, 24, 8913. https://doi.org/10.3390/ijms24108913
Li A-M, Liao F, Wang M, Chen Z-L, Qin C-X, Huang R-Q, Verma KK, Li Y-R, Que Y-X, Pan Y-Q, et al. Transcriptomic and Proteomic Landscape of Sugarcane Response to Biotic and Abiotic Stressors. International Journal of Molecular Sciences. 2023; 24(10):8913. https://doi.org/10.3390/ijms24108913
Chicago/Turabian StyleLi, Ao-Mei, Fen Liao, Miao Wang, Zhong-Liang Chen, Cui-Xian Qin, Ruo-Qi Huang, Krishan K. Verma, Yang-Rui Li, You-Xiong Que, You-Qiang Pan, and et al. 2023. "Transcriptomic and Proteomic Landscape of Sugarcane Response to Biotic and Abiotic Stressors" International Journal of Molecular Sciences 24, no. 10: 8913. https://doi.org/10.3390/ijms24108913






