Current Treatments for Diabetic Macular Edema
Abstract
1. Introduction
2. Pathogenesis of Diabetic Retinopathy and DME
3. Treatments for Diabetic Macular Edema
3.1. Anti-VEGF Treatment
3.1.1. Ranibizumab
3.1.2. Aflibercept
3.1.3. Bevacizumab
3.1.4. Faricimab
3.1.5. Brolucizumab
3.2. Topical Corticosteroid Treatment
3.2.1. Intravitreal Injection of Triamcinolone Acetonide (IVTA)
3.2.2. Subtenon Triamcinolone Acetonide Injection (STTA)
3.2.3. Intravitreal Sustained-Release Steroid Implants
3.2.4. Switching to Steroid Treatments from Anti-VEGF Treatments
3.2.5. STTA or IVTA during Cataract Surgery for Eyes with DME
3.3. Laser Photocoagulation
3.3.1. Focal/Grid Laser
3.3.2. Subthreshold Laser Treatments
PASCAL and Endpoint Management (EpM) Algorithm
Micropulse Diode Laser
3.3.3. Navigated Laser
3.3.4. Selective Retina Therapy
3.4. Combination Therapy with Laser Photocoagulation and Anti-VEGF Agents
3.4.1. Grid Laser + Anti-VEGF Combined Treatments
3.4.2. Focal Laser + Anti-VEGF Treatments
3.4.3. Combined Navigated Laser + Anti-VEGF
3.4.4. Subthreshold Laser + Anti-VEGF
3.4.5. Targeted Retinal Photocoagulation (TRP) + Anti-VEGF
3.5. Pars Plana Vitrectomy
3.5.1. Efficacy of Vitrectomy for DME Refractory to Anti-VEGF Treatment
3.5.2. New Surgical Procedures
3.6. Agents for Systemic Treatments
3.6.1. SGLT2 Inhibitor
3.6.2. Fenofibrate
3.6.3. Metformin
3.7. Recommended Treatment for DME
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Sun, H.; Saeedi, P.; Karuranga, S.; Pinkepank, M.; Ogurtsova, K.; Duncan, B.B.; Stein, C.; Basit, A.; Chan, J.C.N.; Mbanya, J.C.; et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res. Clin. Pract. 2022, 183, 109119. [Google Scholar] [CrossRef] [PubMed]
- Ogurtsova, K.; da Rocha Fernandes, J.D.; Huang, Y.; Linnenkamp, U.; Guariguata, L.; Cho, N.H.; Cavan, D.; Shaw, J.E.; Makaroff, L.E. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 2017, 128, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Yau, J.W.; Rogers, S.L.; Kawasaki, R.; Lamoureux, E.L.; Kowalski, J.W.; Bek, T.; Chen, S.J.; Dekker, J.M.; Fletcher, A.; Grauslund, J.; et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 2012, 35, 556–564. [Google Scholar] [CrossRef]
- O’Doherty, M.; Dooley, I.; Hickey-Dwyer, M. Interventions for diabetic macular oedema: A systematic review of the literature. Br. J. Ophthalmol. 2008, 92, 1581–1590. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.M.; Schmidt-Erfurth, U.; Do, D.V.; Holz, F.G.; Boyer, D.S.; Midena, E.; Heier, J.S.; Terasaki, H.; Kaiser, P.K.; Marcus, D.M.; et al. Intravitreal Aflibercept for Diabetic Macular Edema: 100-Week Results from the VISTA and VIVID Studies. Ophthalmology 2015, 122, 2044–2052. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, T.; Li, X.; Koh, A.; Lai, T.Y.; Lee, F.L.; Lee, W.K.; Ma, Z.; Ohji, M.; Tan, N.; Cha, S.B.; et al. The REVEAL Study: Ranibizumab Monotherapy or Combined with Laser versus Laser Monotherapy in Asian Patients with Diabetic Macular Edema. Ophthalmology 2015, 122, 1402–1415. [Google Scholar] [CrossRef]
- Wells, J.A.; Glassman, A.R.; Ayala, A.R.; Jampol, L.M.; Bressler, N.M.; Bressler, S.B.; Brucker, A.J.; Ferris, F.L.; Hampton, G.R.; Jhaveri, C.; et al. Aflibercept, Bevacizumab, or Ranibizumab for Diabetic Macular Edema: Two-Year Results from a Comparative Effectiveness Randomized Clinical Trial. Ophthalmology 2016, 123, 1351–1359. [Google Scholar] [CrossRef]
- Bressler, N.M.; Beaulieu, W.T.; Glassman, A.R.; Blinder, K.J.; Bressler, S.B.; Jampol, L.M.; Melia, M.; Wells, J.A., 3rd. Diabetic Retinopathy Clinical Research Network. Persistent Macular Thickening Following Intravitreous Aflibercept, Bevacizumab, or Ranibizumab for Central-Involved Diabetic Macular Edema with Vision Impairment: A Secondary Analysis of a Randomized Clinical Trial. JAMA Ophthalmol. 2018, 136, 257–269. [Google Scholar] [CrossRef]
- Antonetti, D.A.; Klein, R.; Gardner, T.W. Diabetic retinopathy. N. Engl. J. Med. 2012, 366, 1227–1239. [Google Scholar] [CrossRef]
- Barber, A.J.; Lieth, E.; Khin, S.A.; Antonetti, D.A.; Buchanan, A.G.; Gardner, T.W. Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J. Clin. Investig. 1998, 102, 783–791. [Google Scholar] [CrossRef]
- Oshitari, T.; Bikbova, G.; Yamamoto, S. Increased expression of phosphorylated c-Jun and phosphorylated c-Jun N-terminal kinase associated with neuronal cell death in diabetic and high glucose exposed rat retinas. Brain Res. Bull. 2014, 101, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.M.; Roon, P.; Van Ells, T.K.; Ganapathy, V.; Smith, S.B. Death of retinal neurons in streptozotocin-induced diabetic mice. Investig. Ophthalmol. Vis. Sci. 2004, 45, 3330–3336. [Google Scholar] [CrossRef]
- Oshitari, T.; Roy, S. Diabetes: A potential enhancer of retinal injury in rat retinas. Neurosci. Lett. 2005, 390, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Oshitari, T.; Yoshida-Hata, N.; Yamamoto, S. Effect of neurotrophic factors on neuronal apoptosis and neurite regeneration in cultured rat retinas exposed to high glucose. Brain Res. 2010, 1346, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Asnaghi, V.; Gerhardinger, C.; Hoehn, T.; Adeboje, A.; Lorenzi, M. A role for the polyol pathway in the early neuroretinal apoptosis and glial changes induced by diabetes in the rat. Diabetes 2003, 52, 506–511. [Google Scholar] [CrossRef]
- Sasaki, M.; Ozawa, Y.; Kurihara, T.; Kubota, S.; Yuki, K.; Noda, K.; Kobayashi, S.; Ishida, S.; Tsubota, K. Neurodegenerative influence of oxidative stress in the retina of a murine model of diabetes. Diabetologia 2010, 53, 971–979. [Google Scholar] [CrossRef]
- Suzumura, A.; Kaneko, H.; Funahashi, Y.; Takayama, K.; Nagaya, M.; Ito, S.; Okuno, T.; Hirakata, T.; Nonobe, N.; Kataoka, K.; et al. n-3 Fatty Acid and Its Metabolite 18-HEPE Ameliorate Retinal Neuronal Cell Dysfunction by Enhancing Müller BDNF in Diabetic Retinopathy. Diabetes 2020, 69, 724–735. [Google Scholar] [CrossRef]
- Sohn, E.H.; van Dijk, H.W.; Jiao, C.; Kok, P.H.; Jeong, W.; Demirkaya, N.; Garmager, A.; Wit, F.; Kucukevcilioglu, M.; van Velthoven, M.E.; et al. Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proc. Natl. Acad. Sci. USA 2016, 113, E2655–E2664. [Google Scholar] [CrossRef] [PubMed]
- van Dijk, H.W.; Verbraak, F.D.; Kok, P.H.; Stehouwer, M.; Garvin, M.K.; Sonka, M.; DeVries, J.H.; Schlingemann, R.O.; Abràmoff, M.D. Early neurodegeneration in the retina of type 2 diabetic patients. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2715–2719. [Google Scholar] [CrossRef]
- Chen, X.; Nie, C.; Gong, Y.; Zhang, Y.; Jin, X.; Wei, S.; Zhang, M. Peripapillary retinal nerve fiber layer changes in preclinical diabetic retinopathy: A meta-analysis. PLoS ONE 2015, 10, e0125919. [Google Scholar] [CrossRef]
- Ng, J.S.; Bearse, M.A., Jr.; Schneck, M.E.; Barez, S.; Adams, A.J. Local diabetic retinopathy prediction by multifocal ERG delays over 3 years. Investig. Ophthalmol. Vis. Sci. 2008, 49, 1622–1628. [Google Scholar] [CrossRef] [PubMed]
- Harrison, W.W.; Bearse, M.A., Jr.; Ng, J.S.; Jewell, N.P.; Barez, S.; Burger, D.; Schneck, M.E.; Adams, A.J. Multifocal electroretinograms predict onset of diabetic retinopathy in adult patients with diabetes. Investig. Ophthalmol. Vis. Sci. 2011, 52, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Simó, R.; Hernández, C. Neurodegeneration is an early event in diabetic retinopathy: Therapeutic implications. Br. J. Ophthalmol. 2012, 96, 1285–1290. [Google Scholar] [CrossRef]
- Suciu, C.I.; Suciu, V.I.; Nicoara, S.D. Optical Coherence Tomography (Angiography) Biomarkers in the Assessment and Monitoring of Diabetic Macular Edema. J. Diabetes Res. 2020, 2020, 6655021. [Google Scholar] [CrossRef] [PubMed]
- Murakami, T.; Nishijima, K.; Sakamoto, A.; Ota, M.; Horii, T.; Yoshimura, N. Association of pathomorphology, photoreceptor status, and retinal thickness with visual acuity in diabetic retinopathy. Am. J. Ophthalmol. 2011, 151, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Otani, T.; Yamaguchi, Y.; Kishi, S. Correlation between visual acuity and foveal microstructural changes in diabetic macular edema. Retina 2010, 30, 774–780. [Google Scholar] [CrossRef]
- Zur, D.; Iglicki, M.; Busch, C.; Invernizzi, A.; Mariussi, M.; Loewenstein, A. OCT Biomarkers as Functional Outcome Predictors in Diabetic Macular Edema Treated with Dexamethasone Implant. Ophthalmology 2018, 125, 267–275. [Google Scholar] [CrossRef]
- Yohannan, J.; Bittencourt, M.; Sepah, Y.J.; Hatef, E.; Sophie, R.; Moradi, A.; Liu, H.; Ibrahim, M.; Do, D.V.; Coulantuoni, E.; et al. Association of retinal sensitivity to integrity of photoreceptor inner/outer segment junction in patients with diabetic macular edema. Ophthalmology 2013, 120, 1254–1261. [Google Scholar] [CrossRef]
- Maheshwary, A.S.; Oster, S.F.; Yuson, R.M.; Cheng, L.; Mojana, F.; Freeman, W.R. The association between percent disruption of the photoreceptor inner segment-outer segment junction and visual acuity in diabetic macular edema. Am. J. Ophthalmol. 2010, 150, 63–67.e61. [Google Scholar] [CrossRef]
- Shin, H.J.; Lee, S.H.; Chung, H.; Kim, H.C. Association between photoreceptor integrity and visual outcome in diabetic macular edema. Graefes Arch. Clin. Exp. Ophthalmol. 2012, 250, 61–70. [Google Scholar] [CrossRef]
- Reznicek, L.; Cserhati, S.; Seidensticker, F.; Liegl, R.; Kampik, A.; Ulbig, M.; Neubauer, A.S.; Kernt, M. Functional and morphological changes in diabetic macular edema over the course of anti-vascular endothelial growth factor treatment. Acta Ophthalmol. 2013, 91, e529–e536. [Google Scholar] [CrossRef]
- Lujan, B.J.; Roorda, A.; Croskrey, J.A.; Dubis, A.M.; Cooper, R.F.; Bayabo, J.K.; Duncan, J.L.; Antony, B.J.; Carroll, J. Directional Optical Coherence Tomography Provides Accurate Outer Nuclear Layer and Henle Fiber Layer Measurements. Retina 2015, 35, 1511–1520. [Google Scholar] [CrossRef]
- Nguyen, Q.D.; Das, A.; Do, D.V.; Dugel, P.U.; Gomes, A.; Holz, F.G.; Koh, A.; Pan, C.K.; Sepah, Y.J.; Patel, N.; et al. Brolucizumab: Evolution through Preclinical and Clinical Studies and the Implications for the Management of Neovascular Age-Related Macular Degeneration. Ophthalmology 2020, 127, 963–976. [Google Scholar] [CrossRef]
- Tadayoni, R.; Sararols, L.; Weissgerber, G.; Verma, R.; Clemens, A.; Holz, F.G. Brolucizumab: A Newly Developed Anti-VEGF Molecule for the Treatment of Neovascular Age-Related Macular Degeneration. Ophthalmologica 2021, 244, 93–101. [Google Scholar] [CrossRef]
- Liberski, S.; Wichrowska, M.; Kociecki, J. Aflibercept versus Faricimab in the Treatment of Neovascular Age-Related Macular Degeneration and Diabetic Macular Edema: A Review. Int. J. Mol. Sci. 2022, 23, 9424. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N.; Damico, L.; Shams, N.; Lowman, H.; Kim, R. Development of ranibizumab, an anti-vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina 2006, 26, 859–870. [Google Scholar] [CrossRef]
- Mordenti, J.; Cuthbertson, R.A.; Ferrara, N.; Thomsen, K.; Berleau, L.; Licko, V.; Allen, P.C.; Valverde, C.R.; Meng, Y.G.; Fei, D.T.; et al. Comparisons of the intraocular tissue distribution, pharmacokinetics, and safety of 125I-labeled full-length and Fab antibodies in rhesus monkeys following intravitreal administration. Toxicol. Pathol. 1999, 27, 536–544. [Google Scholar] [CrossRef]
- Nguyen, Q.D.; Brown, D.M.; Marcus, D.M.; Boyer, D.S.; Patel, S.; Feiner, L.; Gibson, A.; Sy, J.; Rundle, A.C.; Hopkins, J.J.; et al. Ranibizumab for diabetic macular edema: Results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology 2012, 119, 789–801. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Toriyama, Y.; Iesato, Y.; Imai, A.; Murata, T. Changes in Plasma Vascular Endothelial Growth Factor Level after Intravitreal Injection of Bevacizumab, Aflibercept, or Ranibizumab for Diabetic Macular Edema. Retina 2018, 38, 1801–1808. [Google Scholar] [CrossRef] [PubMed]
- Diabetic Retinopathy Clinical Research Network; Wells, J.A.; Glassman, A.R.; Ayala, A.R.; Jampol, L.M.; Aiello, L.P.; Antoszyk, A.N.; Arnold-Bush, B.; Baker, C.W.; Bressler, N.M.; et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. N. Engl. J. Med. 2015, 372, 1193–1203. [Google Scholar] [CrossRef]
- Papadopoulos, N.; Martin, J.; Ruan, Q.; Rafique, A.; Rosconi, M.P.; Shi, E.; Pyles, E.A.; Yancopoulos, G.D.; Stahl, N.; Wiegand, S.J. Binding and neutralization of vascular endothelial growth factor (VEGF) and related ligands by VEGF Trap, ranibizumab and bevacizumab. Angiogenesis 2012, 15, 171–185. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.; Loewenstein, A. Therapeutic Monoclonal Antibodies and Fragments: Bevacizumab. Dev. Ophthalmol. 2016, 55, 232–245. [Google Scholar] [CrossRef] [PubMed]
- Ross, E.L.; Hutton, D.W.; Stein, J.D.; Bressler, N.M.; Jampol, L.M.; Glassman, A.R.; Diabetic Retinopathy Clinical Research Network. Cost-effectiveness of Aflibercept, Bevacizumab, and Ranibizumab for Diabetic Macular Edema Treatment: Analysis from the Diabetic Retinopathy Clinical Research Network Comparative Effectiveness Trial. JAMA Ophthalmol. 2016, 134, 888–896. [Google Scholar] [CrossRef] [PubMed]
- Regula, J.T.; Lundh von Leithner, P.; Foxton, R.; Barathi, V.A.; Cheung, C.M.; Bo Tun, S.B.; Wey, Y.S.; Iwata, D.; Dostalek, M.; Moelleken, J.; et al. Targeting key angiogenic pathways with a bispecific CrossMAb optimized for neovascular eye diseases. EMBO Mol. Med. 2016, 8, 1265–1288. [Google Scholar] [CrossRef]
- Wykoff, C.C.; Abreu, F.; Adamis, A.P.; Basu, K.; Eichenbaum, D.A.; Haskova, Z.; Lin, H.; Loewenstein, A.; Mohan, S.; Pearce, I.A.; et al. Efficacy, durability, and safety of intravitreal faricimab with extended dosing up to every 16 weeks in patients with diabetic macular oedema (YOSEMITE and RHINE): Two randomised, double-masked, phase 3 trials. Lancet 2022, 399, 741–755. [Google Scholar] [CrossRef]
- Joussen, A.M.; Ricci, F.; Paris, L.P.; Korn, C.; Quezada-Ruiz, C.; Zarbin, M. Angiopoietin/Tie2 signalling and its role in retinal and choroidal vascular diseases: A review of preclinical data. Eye 2021, 35, 1305–1316. [Google Scholar] [CrossRef]
- Schlottmann, P.; Wells, J.A.; Haskova, Z.; Mohan, S.; Silverman, D.; Tang, Y.; Lin, H. Efficacy, Durability and Safety of Faricimab in Diabetic Macular Edema: 2-Year Results from the Phase 3 YOSEMITE and RHINE Trials. Investig. Ophthalmol. Vis. Sci. 2022, 63, 3850. [Google Scholar]
- Gül, N.; van Egmond, M. Antibody-Dependent Phagocytosis of Tumor Cells by Macrophages: A Potent Effector Mechanism of Monoclonal Antibody Therapy of Cancer. Cancer Res. 2015, 75, 5008–5013. [Google Scholar] [CrossRef]
- Roopenian, D.C.; Akilesh, S. FcRn: The neonatal Fc receptor comes of age. Nat. Rev. Immunol. 2007, 7, 715–725. [Google Scholar] [CrossRef]
- Gaudreault, J.; Fei, D.; Rusit, J.; Suboc, P.; Shiu, V. Preclinical pharmacokinetics of Ranibizumab (rhuFabV2) after a single intravitreal administration. Investig. Ophthalmol. Vis. Sci. 2005, 46, 726–733. [Google Scholar] [CrossRef]
- Park, S.J.; Choi, Y.; Na, Y.M.; Hong, H.K.; Park, J.Y.; Park, K.H.; Chung, J.Y.; Woo, S.J. Intraocular Pharmacokinetics of Intravitreal Aflibercept (Eylea) in a Rabbit Model. Investig. Ophthalmol. Vis. Sci. 2016, 57, 2612–2617. [Google Scholar] [CrossRef]
- Brown, D.M.; Emanuelli, A.; Bandello, F.; Barranco, J.J.E.; Figueira, J.; Souied, E.; Wolf, S.; Gupta, V.; Ngah, N.F.; Liew, G.; et al. KESTREL and KITE: 52-Week Results from Two Phase III Pivotal Trials of Brolucizumab for Diabetic Macular Edema. Am. J. Ophthalmol. 2022, 238, 157–172. [Google Scholar] [CrossRef] [PubMed]
- Mones, J.; Srivastava, S.K.; Jaffe, G.J.; Tadayoni, R.; Albini, T.A.; Kaiser, P.K.; Holz, F.G.; Korobelnik, J.F.; Kim, I.K.; Pruente, C.; et al. Risk of Inflammation, Retinal Vasculitis, and Retinal Occlusion-Related Events with Brolucizumab: Post Hoc Review of HAWK and HARRIER. Ophthalmology 2021, 128, 1050–1059. [Google Scholar] [CrossRef]
- Diabetic Retinopathy Clinical Research Network; Elman, M.J.; Aiello, L.P.; Beck, R.W.; Bressler, N.M.; Bressler, S.B.; Edwards, A.R.; Ferris, F.L., 3rd; Friedman, S.M.; Glassman, A.R.; et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology 2010, 117, 1064–1077.e35. [Google Scholar] [CrossRef]
- Campochiaro, P.A.; Brown, D.M.; Pearson, A.; Ciulla, T.; Boyer, D.; Holz, F.G.; Tolentino, M.; Gupta, A.; Duarte, L.; Madreperla, S.; et al. Long-term benefit of sustained-delivery fluocinolone acetonide vitreous inserts for diabetic macular edema. Ophthalmology 2011, 118, 626–635.e2. [Google Scholar] [CrossRef]
- Boyer, D.S.; Yoon, Y.H.; Belfort, R., Jr.; Bandello, F.; Maturi, R.K.; Augustin, A.J.; Li, X.Y.; Cui, H.; Hashad, Y.; Whitcup, S.M. Three-year, randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with diabetic macular edema. Ophthalmology 2014, 121, 1904–1914. [Google Scholar] [CrossRef]
- Sutter, F.K.; Simpson, J.M.; Gillies, M.C. Intravitreal triamcinolone for diabetic macular edema that persists after laser treatment: Three-month efficacy and safety results of a prospective, randomized, double-masked, placebo-controlled clinical trial. Ophthalmology 2004, 111, 2044–2049. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.K.; Mohamed, S.; Lee, V.Y.; Lai, T.Y.; Shanmugam, M.P.; Lam, D.S. Intravitreal dexamethasone for diabetic macular edema: A pilot study. Ophthalmic Surg. Lasers Imaging 2010, 41, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Bakri, S.J.; Kaiser, P.K. Posterior subtenon triamcinolone acetonide for refractory diabetic macular edema. Am. J. Ophthalmol. 2005, 139, 290–294. [Google Scholar] [CrossRef]
- Bucolo, C.; Gozzo, L.; Longo, L.; Mansueto, S.; Vitale, D.C.; Drago, F. Long-term efficacy and safety profile of multiple injections of intravitreal dexamethasone implant to manage diabetic macular edema: A systematic review of real-world studies. J. Pharmacol. Sci. 2018, 138, 219–232. [Google Scholar] [CrossRef]
- Avitabile, T.; Longo, A.; Reibaldi, A. Intravitreal triamcinolone compared with macular laser grid photocoagulation for the treatment of cystoid macular edema. Am. J. Ophthalmol. 2005, 140, 695–702. [Google Scholar] [CrossRef] [PubMed]
- Diabetic Retinopathy Clinical Research Network. A randomized trial comparing intravitreal triamcinolone acetonide and focal/grid photocoagulation for diabetic macular edema. Ophthalmology 2008, 115, 1447–1459. [Google Scholar] [CrossRef] [PubMed]
- Ockrim, Z.K.; Sivaprasad, S.; Falk, S.; Roghani, S.; Bunce, C.; Gregor, Z.; Hykin, P. Intravitreal triamcinolone versus laser photocoagulation for persistent diabetic macular oedema. Br. J. Ophthalmol. 2008, 92, 795–799. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.W.; Lee, H.K.; Shin, M.C. Comparison of intravitreal bevacizumab alone or combined with triamcinolone versus triamcinolone in diabetic macular edema: A randomized clinical trial. Ophthalmologica 2012, 227, 100–106. [Google Scholar] [CrossRef]
- Gillies, M.C.; Lim, L.L.; Campain, A.; Quin, G.J.; Salem, W.; Li, J.; Goodwin, S.; Aroney, C.; McAllister, I.L.; Fraser-Bell, S. A randomized clinical trial of intravitreal bevacizumab versus intravitreal dexamethasone for diabetic macular edema: The BEVORDEX study. Ophthalmology 2014, 121, 2473–2481. [Google Scholar] [CrossRef] [PubMed]
- Kriechbaum, K.; Prager, S.; Mylonas, G.; Scholda, C.; Rainer, G.; Funk, M.; Kundi, M.; Schmidt-Erfurth, U. Intravitreal bevacizumab (Avastin) versus triamcinolone (Volon A) for treatment of diabetic macular edema: One-year results. Eye 2014, 28, 9–16. [Google Scholar] [CrossRef]
- Callanan, D.G.; Loewenstein, A.; Patel, S.S.; Massin, P.; Corcóstegui, B.; Li, X.Y.; Jiao, J.; Hashad, Y.; Whitcup, S.M. A multicenter, 12-month randomized study comparing dexamethasone intravitreal implant with ranibizumab in patients with diabetic macular edema. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 463–473. [Google Scholar] [CrossRef]
- Audren, F.; Erginay, A.; Haouchine, B.; Benosman, R.; Conrath, J.; Bergmann, J.F.; Gaudric, A.; Massin, P. Intravitreal triamcinolone acetonide for diffuse diabetic macular oedema: 6-month results of a prospective controlled trial. Acta Ophthalmol. Scand. 2006, 84, 624–630. [Google Scholar] [CrossRef]
- Gillies, M.C.; Sutter, F.K.; Simpson, J.M.; Larsson, J.; Ali, H.; Zhu, M. Intravitreal triamcinolone for refractory diabetic macular edema: Two-year results of a double-masked, placebo-controlled, randomized clinical trial. Ophthalmology 2006, 113, 1533–1538. [Google Scholar] [CrossRef]
- Jonas, J.B.; Kamppeter, B.A.; Harder, B.; Vossmerbaeumer, U.; Sauder, G.; Spandau, U.H. Intravitreal triamcinolone acetonide for diabetic macular edema: A prospective, randomized study. J. Ocul. Pharmacol. Ther. 2006, 22, 200–207. [Google Scholar] [CrossRef]
- Dehghan, M.H.; Ahmadieh, H.; Ramezani, A.; Entezari, M.; Anisian, A. A randomized, placebo-controlled clinical trial of intravitreal triamcinolone for refractory diabetic macular edema. Int. Ophthalmol. 2008, 28, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Larsson, J.; Kifley, A.; Zhu, M.; Wang, J.J.; Mitchell, P.; Sutter, F.K.; Gillies, M.C. Rapid reduction of hard exudates in eyes with diabetic retinopathy after intravitreal triamcinolone: Data from a randomized, placebo-controlled, clinical trial. Acta Ophthalmol. 2009, 87, 275–280. [Google Scholar] [CrossRef]
- Pearson, P.A.; Comstock, T.L.; Ip, M.; Callanan, D.; Morse, L.S.; Ashton, P.; Levy, B.; Mann, E.S.; Eliott, D. Fluocinolone acetonide intravitreal implant for diabetic macular edema: A 3-year multicenter, randomized, controlled clinical trial. Ophthalmology 2011, 118, 1580–1587. [Google Scholar] [CrossRef] [PubMed]
- Rittiphairoj, T.; Mir, T.A.; Li, T.; Virgili, G. Intravitreal steroids for macular edema in diabetes. Cochrane Database Syst. Rev. 2020, 11, Cd005656. [Google Scholar] [CrossRef]
- Gao, L.; Zhao, X.; Jiao, L.; Tang, L. Intravitreal corticosteroids for diabetic macular edema: A network meta-analysis of randomized controlled trials. Eye Vis. 2021, 8, 35. [Google Scholar] [CrossRef]
- Solomon, S.D.; Chew, E.; Duh, E.J.; Sobrin, L.; Sun, J.K.; VanderBeek, B.L.; Wykoff, C.C.; Gardner, T.W. Diabetic Retinopathy: A Position Statement by the American Diabetes Association. Diabetes Care 2017, 40, 412–418. [Google Scholar] [CrossRef]
- Schmidt-Erfurth, U.; Garcia-Arumi, J.; Bandello, F.; Berg, K.; Chakravarthy, U.; Gerendas, B.S.; Jonas, J.; Larsen, M.; Tadayoni, R.; Loewenstein, A. Guidelines for the Management of Diabetic Macular Edema by the European Society of Retina Specialists (EURETINA). Ophthalmologica 2017, 237, 185–222. [Google Scholar] [CrossRef]
- Terasaki, H.; Ogura, Y.; Kitano, S.; Sakamoto, T.; Murata, T.; Hirakata, A.; Ishibashi, T. Management of diabetic macular edema in Japan: A review and expert opinion. Jpn. J. Ophthalmol. 2018, 62, 1–23. [Google Scholar] [CrossRef]
- Yoshida, S.; Murakami, T.; Nozaki, M.; Suzuma, K.; Baba, T.; Hirano, T.; Sawada, O.; Sugimoto, M.; Takamura, Y.; Tsuiki, E. Review of clinical studies and recommendation for a therapeutic flow chart for diabetic macular edema. Graefe’s Arch. Clin. Exp. Ophthalmol. 2020, 259, 815–836. [Google Scholar] [CrossRef]
- Shimura, M.; Yasuda, K.; Shiono, T. Posterior sub-Tenon’s capsule injection of triamcinolone acetonide prevents panretinal photocoagulation-induced visual dysfunction in patients with severe diabetic retinopathy and good vision. Ophthalmology 2006, 113, 381–387. [Google Scholar] [CrossRef]
- Early Treatment Diabetic Retinopathy Study Research Group. Treatment Techniques and Clinical Guidelines for Photocoagulation of Diabetic Macular Edema: Early Treatment Diabetic Retinopathy Study Report Number 2. Ophthalmology 1987, 94, 761–774. [Google Scholar] [CrossRef]
- Martidis, A.; Duker, J.S.; Greenberg, P.B.; Rogers, A.H.; Puliafito, C.A.; Reichel, E.; Baumal, C. Intravitreal triamcinolone for refractory diabetic macular edema. Ophthalmology 2002, 109, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Spandau, U.H.; Derse, M.; Schmitz-Valckenberg, P.; Papoulis, C.; Jonas, J.B. Dosage dependency of intravitreal triamcinolone acetonide as treatment for diabetic macular oedema. Br. J. Ophthalmol. 2005, 89, 999–1003. [Google Scholar] [CrossRef]
- Audren, F.; Lecleire-Collet, A.; Erginay, A.; Haouchine, B.; Benosman, R.; Bergmann, J.F.; Gaudric, A.; Massin, P. Intravitreal triamcinolone acetonide for diffuse diabetic macular edema: Phase 2 trial comparing 4 mg vs. 2 mg. Am. J. Ophthalmol. 2006, 142, 794–799. [Google Scholar] [CrossRef]
- Lam, D.S.; Chan, C.K.; Mohamed, S.; Lai, T.Y.; Li, K.K.; Li, P.S.; Tsang, C.W.; Chan, W.M.; Shanmugam, M.P. A prospective randomised trial of different doses of intravitreal triamcinolone for diabetic macular oedema. Br. J. Ophthalmol. 2007, 91, 199–203. [Google Scholar] [CrossRef][Green Version]
- Hauser, D.; Bukelman, A.; Pokroy, R.; Katz, H.; Len, A.; Thein, R.; Parness-Yossifon, R.; Pollack, A. Intravitreal triamcinolone for diabetic macular edema: Comparison of 1, 2, and 4 mg. Retina 2008, 28, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Pollack, J.S.; Miller, D.G.; Mittra, R.A.; Spaide, R.F. ISIS-DME: A prospective, randomized, dose-escalation intravitreal steroid injection study for refractory diabetic macular edema. Retina 2008, 28, 735–740. [Google Scholar] [CrossRef]
- Qi, H.-P.; Bi, S.; Wei, S.-Q.; Cui, H.; Zhao, J.-B. Intravitreal versus Subtenon Triamcinolone Acetonide Injection for Diabetic Macular Edema: A Systematic Review and Meta-analysis. Curr. Eye Res. 2012, 37, 1136–1147. [Google Scholar] [CrossRef]
- Maeda, Y.; Ishikawa, H.; Nishikawa, H.; Shimizu, M.; Kinoshita, T.; Ogihara, R.; Kitano, S.; Yamanaka, C.; Mitamura, Y.; Sugimoto, M.; et al. Intraocular pressure elevation after subtenon triamcinolone acetonide injection; Multicentre retrospective cohort study in Japan. PLoS ONE 2019, 14, e0226118. [Google Scholar] [CrossRef]
- Hirano, Y.; Ito, T.; Nozaki, M.; Yasukawa, T.; Sakurai, E.; Yoshida, M.; Ogura, Y. Intraocular pressure elevation following triamcinolone acetonide administration as related to administration routes. Jpn. J. Ophthalmol. 2009, 53, 519–522. [Google Scholar] [CrossRef]
- Campochiaro, P.A.; Hafiz, G.; Shah, S.M.; Bloom, S.; Brown, D.M.; Busquets, M.; Ciulla, T.; Feiner, L.; Sabates, N.; Billman, K.; et al. Sustained ocular delivery of fluocinolone acetonide by an intravitreal insert. Ophthalmology 2010, 117, 1393–1399.e1393. [Google Scholar] [CrossRef] [PubMed]
- Campochiaro, P.A.; Brown, D.M.; Pearson, A.; Chen, S.; Boyer, D.; Ruiz-Moreno, J.; Garretson, B.; Gupta, A.; Hariprasad, S.M.; Bailey, C.; et al. Sustained delivery fluocinolone acetonide vitreous inserts provide benefit for at least 3 years in patients with diabetic macular edema. Ophthalmology 2012, 119, 2125–2132. [Google Scholar] [CrossRef] [PubMed]
- Hong, I.H.; Choi, W.; Han, J.R. The effects of intravitreal triamcinolone acetonide in diabetic macular edema refractory to anti-VEGF treatment. Jpn. J. Ophthalmol. 2020, 64, 196–202. [Google Scholar] [CrossRef]
- Tatsumi, T.; Oshitari, T.; Baba, T.; Takatsuna, Y.; Yamamoto, S. Effects of Switching from Anti-VEGF Treatment to Triamcinolone Acetonide in Eyes with Refractory Macular Edema Associated with Diabetic Retinopathy or Retinal Vein Occlusion. BioMed Res. Int. 2020, 2020, 4529850. [Google Scholar] [CrossRef] [PubMed]
- Busch, C.; Zur, D.; Fraser-Bell, S.; Laíns, I.; Santos, A.R.; Lupidi, M.; Cagini, C.; Gabrielle, P.H.; Couturier, A.; Mané-Tauty, V.; et al. Shall we stay, or shall we switch? Continued anti-VEGF therapy versus early switch to dexamethasone implant in refractory diabetic macular edema. Acta Diabetol. 2018, 55, 789–796. [Google Scholar] [CrossRef]
- Klein, B.E.; Klein, R.; Moss, S.E. Prevalence of cataracts in a population-based study of persons with diabetes mellitus. Ophthalmology 1985, 92, 1191–1196. [Google Scholar] [CrossRef] [PubMed]
- Dong, N.; Xu, B.; Wang, B.; Chu, L.; Tang, X. Aqueous cytokines as predictors of macular edema in patients with diabetes following uncomplicated phacoemulsification cataract surgery. BioMed Res. Int. 2015, 2015, 126984. [Google Scholar] [CrossRef]
- Lim, L.L.; Morrison, J.L.; Constantinou, M.; Rogers, S.; Sandhu, S.S.; Wickremasinghe, S.S.; Kawasaki, R.; Al-Qureshi, S. Diabetic Macular Edema at the time of Cataract Surgery trial: A prospective, randomized clinical trial of intravitreous bevacizumab versus triamcinolone in patients with diabetic macular oedema at the time of cataract surgery—Preliminary 6 month results. Clin. Exp. Ophthalmol. 2016, 44, 233–242. [Google Scholar] [CrossRef]
- Sohn, H.J.; Han, D.H.; Kim, I.T.; Oh, I.K.; Kim, K.H.; Lee, D.Y.; Nam, D.H. Changes in aqueous concentrations of various cytokines after intravitreal triamcinolone versus bevacizumab for diabetic macular edema. Am. J. Ophthalmol. 2011, 152, 686–694. [Google Scholar] [CrossRef]
- Tatsumi, T.; Oshitari, T.; Ando, T.; Takatsuna, Y.; Arai, M.; Baba, T.; Sato, E.; Yamamoto, S. Comparison of the Efficacy of Sub-Tenon versus Intravitreal Triamcinolone Acetonide Injection during Cataract Surgery for Diabetic Macular Edema. Ophthalmologica 2019, 241, 17–23. [Google Scholar] [CrossRef]
- Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch. Ophthalmol. 1985, 103, 1796–1806. [Google Scholar] [CrossRef]
- Lewis, H.; Schachat, A.P.; Haimann, M.H.; Haller, J.A.; Quinlan, P.; von Fricken, M.A.; Fine, S.L.; Murphy, R.P. Choroidal neovascularization after laser photocoagulation for diabetic macular edema. Ophthalmology 1990, 97, 503–510; discussion 510–501. [Google Scholar] [CrossRef]
- Schatz, H.; Madeira, D.; McDonald, H.R.; Johnson, R.N. Progressive enlargement of laser scars following grid laser photocoagulation for diffuse diabetic macular edema. Arch. Ophthalmol. 1991, 109, 1549–1551. [Google Scholar] [CrossRef]
- Guyer, D.R.; D’Amico, D.J.; Smith, C.W. Subretinal fibrosis after laser photocoagulation for diabetic macular edema. Am. J. Ophthalmol. 1992, 113, 652–656. [Google Scholar] [CrossRef]
- Hirano, T.; Toriyama, Y.; Iesato, Y.; Imai, A.; Hirabayashi, K.; Nagaoka, T.; Takamura, Y.; Sugimoto, M.; Murata, T. Effect of leaking perifoveal microaneurysms on resolution of diabetic macular edema treated by combination therapy using anti-vascular endothelial growth factor and short pulse focal/grid laser photocoagulation. Jpn. J. Ophthalmol. 2017, 61, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Takamura, Y.; Matsumura, T.; Gozawa, M.; Morioka, M.; Inatani, M. Regional Variety of Reduction in Retinal Thickness of Diabetic Macular Edema after Anti-VEGF Treatment. Medicina 2022, 58, 933. [Google Scholar] [CrossRef]
- Pankratov, M. Pulsed Delivery of Laser Energy in Experimental Thermal Retinal Photocoagulation; SPIE: Bellingham, WA, USA, 1990; Volume 1202. [Google Scholar]
- Lavinsky, D.; Sramek, C.; Wang, J.; Huie, P.; Dalal, R.; Mandel, Y.; Palanker, D. Subvisible retinal laser therapy: Titration algorithm and tissue response. Retina 2014, 34, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Sliney, D.H. Tissue specific damage to the retinal pigment epithelium: Mecanisms and therapeutic implications. Lasers Light Ophthalmol. 1992, 5, 17–28. [Google Scholar]
- Sivaprasad, S.; Dorin, G. Subthreshold diode laser micropulse photocoagulation for the treatment of diabetic macular edema. Expert Rev. Med. Devices 2012, 9, 189–197. [Google Scholar] [CrossRef]
- Inagaki, K.; Shuo, T.; Katakura, K.; Ebihara, N.; Murakami, A.; Ohkoshi, K. Sublethal Photothermal Stimulation with a Micropulse Laser Induces Heat Shock Protein Expression in ARPE-19 Cells. J. Ophthalmol. 2015, 2015, 729792. [Google Scholar] [CrossRef]
- Pei-Pei, W.; Shi-Zhou, H.; Zhen, T.; Lin, L.; Ying, L.; Jiexiong, O.; Wen-Bo, Z.; Chen-Jin, J. Randomised clinical trial evaluating best-corrected visual acuity and central macular thickness after 532-nm subthreshold laser grid photocoagulation treatment in diabetic macular oedema. Eye 2015, 29, 313–322. [Google Scholar] [CrossRef]
- Hamada, M.; Ohkoshi, K.; Inagaki, K.; Ebihara, N.; Murakami, A. Subthreshold Photocoagulation Using Endpoint Management in the PASCAL® System for Diffuse Diabetic Macular Edema. J. Ophthalmol. 2018, 2018, 7465794. [Google Scholar] [CrossRef]
- Karasu, B.; Akbas, Y.B.; Aykut, A.; Çelebi, A.R.C. Subthreshold Photocoagulation, Laser Endpoint Management Based on Optical Coherence Tomography Angiography in Cases of Diabetic Macular Edema Refractory to Anti-VEGF. Klin. Monbl. Augenheilkd. 2022. [Google Scholar] [CrossRef] [PubMed]
- Friberg, T.R.; Karatza, E.C. The Treatment of Macular Disease Using a Micropulsed and Continuous Wave 810-nm Diode Laser. Ophthalmology 1997, 104, 2030–2038. [Google Scholar] [CrossRef] [PubMed]
- Moorman, C.M.; Hamilton, A.M. Clinical applications of the MicroPulse diode laser. Eye 1999, 13 Pt 2, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Ohkoshi, K.; Yamaguchi, T. Subthreshold micropulse diode laser photocoagulation for diabetic macular edema in Japanese patients. Am. J. Ophthalmol. 2010, 149, 133–139. [Google Scholar] [CrossRef]
- Takatsuna, Y.; Yamamoto, S.; Nakamura, Y.; Tatsumi, T.; Arai, M.; Mitamura, Y. Long-term therapeutic efficacy of the subthreshold micropulse diode laser photocoagulation for diabetic macular edema. Jpn. J. Ophthalmol. 2011, 55, 365–369. [Google Scholar] [CrossRef]
- Inagaki, K.; Ohkoshi, K.; Ohde, S.; Deshpande, G.A.; Ebihara, N.; Murakami, A. Comparative efficacy of pure yellow (577-nm) and 810-nm subthreshold micropulse laser photocoagulation combined with yellow (561–577-nm) direct photocoagulation for diabetic macular edema. Jpn. J. Ophthalmol. 2015, 59, 21–28. [Google Scholar] [CrossRef]
- Figueira, J.; Khan, J.; Nunes, S.; Sivaprasad, S.; Rosa, A.; de Abreu, J.F.; Cunha-Vaz, J.G.; Chong, N.V. Prospective randomised controlled trial comparing sub-threshold micropulse diode laser photocoagulation and conventional green laser for clinically significant diabetic macular oedema. Br. J. Ophthalmol. 2009, 93, 1341–1344. [Google Scholar] [CrossRef]
- Vujosevic, S.; Bottega, E.; Casciano, M.; Pilotto, E.; Convento, E.; Midena, E. Microperimetry and fundus autofluorescence in diabetic macular edema: Subthreshold micropulse diode laser versus modified early treatment diabetic retinopathy study laser photocoagulation. Retina 2010, 30, 908–916. [Google Scholar] [CrossRef]
- Lavinsky, D.; Cardillo, J.A.; Melo, L.A., Jr.; Dare, A.; Farah, M.E.; Belfort, R., Jr. Randomized clinical trial evaluating mETDRS versus normal or high-density micropulse photocoagulation for diabetic macular edema. Investig. Ophthalmol. Vis. Sci. 2011, 52, 4314–4323. [Google Scholar] [CrossRef] [PubMed]
- Lois, N.; Campbell, C.; Waugh, N.; Azuara-Blanco, A.; Maredza, M.; Mistry, H.; McAuley, D.; Acharya, N.; Aslam, T.M.; Bailey, C.; et al. Diabetic Macular Edema and Diode Subthreshold Micropulse Laser: A Randomized Double-Masked Noninferiority Clinical Trial. Ophthalmology 2023, 130, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Kozak, I.; Oster, S.F.; Cortes, M.A.; Dowell, D.; Hartmann, K.; Kim, J.S.; Freeman, W.R. Clinical evaluation and treatment accuracy in diabetic macular edema using navigated laser photocoagulator NAVILAS. Ophthalmology 2011, 118, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Neubauer, A.S.; Langer, J.; Liegl, R.; Haritoglou, C.; Wolf, A.; Kozak, I.; Seidensticker, F.; Ulbig, M.; Freeman, W.R.; Kampik, A.; et al. Navigated macular laser decreases retreatment rate for diabetic macular edema: A comparison with conventional macular laser. Clin. Ophthalmol. 2013, 7, 121–128. [Google Scholar] [CrossRef][Green Version]
- Kato, F.; Nozaki, M.; Kato, A.; Hasegawa, N.; Morita, H.; Yoshida, M.; Ogura, Y. Evaluation of Navigated Laser Photocoagulation (Navilas 577+) for the Treatment of Refractory Diabetic Macular Edema. J. Ophthalmol. 2018, 2018, 3978514. [Google Scholar] [CrossRef]
- Nozaki, M.; Kato, A.; Yasukawa, T.; Suzuki, K.; Yoshida, M.; Ogura, Y. Indocyanine green angiography-guided focal navigated laser photocoagulation for diabetic macular edema. Jpn. J. Ophthalmol. 2019, 63, 243–254. [Google Scholar] [CrossRef]
- Roider, J.; Michaud, N.A.; Flotte, T.J.; Birngruber, R. Response of the retinal pigment epithelium to selective photocoagulation. Arch. Ophthalmol. 1992, 110, 1786–1792. [Google Scholar] [CrossRef]
- Brinkmann, R.; Huttmann, G.; Rogener, J.; Roider, J.; Birngruber, R.; Lin, C.P. Origin of retinal pigment epithelium cell damage by pulsed laser irradiance in the nanosecond to microsecond time regimen. Lasers Surg. Med. 2000, 27, 451–464. [Google Scholar] [CrossRef]
- Yamamoto, M.; Miura, Y.; Hirayama, K.; Kohno, T.; Kabata, D.; Theisen-Kunde, D.; Brinkmann, R.; Honda, S. Predictive factors of outcome of selective retina therapy for diabetic macular edema. Int. Ophthalmol. 2020, 40, 1221–1232. [Google Scholar] [CrossRef]
- Roider, J.; Liew, S.H.; Klatt, C.; Elsner, H.; Poerksen, E.; Hillenkamp, J.; Brinkmann, R.; Birngruber, R. Selective retina therapy (SRT) for clinically significant diabetic macular edema. Graefes Arch. Clin. Exp. Ophthalmol. 2010, 248, 1263–1272. [Google Scholar] [CrossRef]
- Mitchell, P.; Bandello, F.; Schmidt-Erfurth, U.; Lang, G.E.; Massin, P.; Schlingemann, R.O.; Sutter, F.; Simader, C.; Burian, G.; Gerstner, O.; et al. The RESTORE study: Ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology 2011, 118, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Korobelnik, J.F.; Do, D.V.; Schmidt-Erfurth, U.; Boyer, D.S.; Holz, F.G.; Heier, J.S.; Midena, E.; Kaiser, P.K.; Terasaki, H.; Marcus, D.M.; et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology 2014, 121, 2247–2254. [Google Scholar] [CrossRef] [PubMed]
- Liegl, R.; Langer, J.; Seidensticker, F.; Reznicek, L.; Haritoglou, C.; Ulbig, M.W.; Neubauer, A.S.; Kampik, A.; Kernt, M. Comparative evaluation of combined navigated laser photocoagulation and intravitreal ranibizumab in the treatment of diabetic macular edema. PLoS ONE 2014, 9, e113981. [Google Scholar] [CrossRef] [PubMed]
- Payne, J.F.; Wykoff, C.C.; Clark, W.L.; Bruce, B.B.; Boyer, D.S.; Brown, D.M.; Group, T.-D.S. Randomized Trial of Treat and Extend Ranibizumab with and without Navigated Laser for Diabetic Macular Edema: TREX-DME 1 Year Outcomes. Ophthalmology 2017, 124, 74–81. [Google Scholar] [CrossRef]
- Moisseiev, E.; Abbassi, S.; Thinda, S.; Yoon, J.; Yiu, G.; Morse, L.S. Subthreshold micropulse laser reduces anti-VEGF injection burden in patients with diabetic macular edema. Eur. J. Ophthalmol. 2018, 28, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Altinel, M.G.; Acikalin, B.; Alis, M.G.; Demir, G.; Mutibayraktaroglu, K.M.; Totuk, O.M.G.; Ardagil, A. Comparison of the efficacy and safety of anti-VEGF monotherapy versus anti-VEGF therapy combined with subthreshold micropulse laser therapy for diabetic macular edema. Lasers Med. Sci. 2021, 36, 1545–1553. [Google Scholar] [CrossRef]
- El Matri, L.; Chebil, A.; El Matri, K.; Falfoul, Y.; Chebbi, Z. Subthreshold micropulse laser adjuvant to bevacizumab versus bevacizumab monotherapy in treating diabetic macular edema: One- year- follow-up. Ther. Adv. Ophthalmol. 2021, 13, 25158414211040887. [Google Scholar] [CrossRef]
- Khattab, A.M.; Hagras, S.M.; AbdElhamid, A.; Torky, M.A.; Awad, E.A.; Abdelhameed, A.G. Aflibercept with adjuvant micropulsed yellow laser versus aflibercept monotherapy in diabetic macular edema. Graefes Arch. Clin. Exp. Ophthalmol. 2019, 257, 1373–1380. [Google Scholar] [CrossRef]
- Abouhussein, M.A.; Gomaa, A.R. Aflibercept plus micropulse laser versus aflibercept monotherapy for diabetic macular edema: 1-year results of a randomized clinical trial. Int. Ophthalmol. 2020, 40, 1147–1154. [Google Scholar] [CrossRef]
- Kanar, H.S.; Arsan, A.; Altun, A.; Aki, S.F.; Hacisalihoglu, A. Can subthreshold micropulse yellow laser treatment change the anti-vascular endothelial growth factor algorithm in diabetic macular edema? A randomized clinical trial. Indian J. Ophthalmol. 2020, 68, 145–151. [Google Scholar] [CrossRef]
- Koushan, K.; Eshtiaghi, A.; Fung, P.; Berger, A.R.; Chow, D.R. Treatment of Diabetic Macular Edema with Aflibercept and Micropulse Laser (DAM Study). Clin. Ophthalmol. 2022, 16, 1109–1115. [Google Scholar] [CrossRef]
- Tatsumi, T.; Takatsuna, Y.; Oshitari, T.; Kaiho, T.; Kawasaki, Y.; Shiko, Y.; Sugawara, T.; Baba, T.; Yamamoto, S. Randomized clinical trial comparing intravitreal aflibercept combined with subthreshold laser to intravitreal aflibercept monotherapy for diabetic macular edema. Sci. Rep. 2022, 12, 10672. [Google Scholar] [CrossRef] [PubMed]
- Takamura, Y.; Tomomatsu, T.; Matsumura, T.; Arimura, S.; Gozawa, M.; Takihara, Y.; Inatani, M. The effect of photocoagulation in ischemic areas to prevent recurrence of diabetic macular edema after intravitreal bevacizumab injection. Investig. Ophthalmol. Vis. Sci. 2014, 55, 4741–4746. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.M.; Ou, W.C.; Wong, T.P.; Kim, R.Y.; Croft, D.E.; Wykoff, C.C.; Group, D.S. Targeted Retinal Photocoagulation for Diabetic Macular Edema with Peripheral Retinal Nonperfusion: Three-Year Randomized DAVE Trial. Ophthalmology 2018, 125, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Lewis, H.; Abrams, G.W.; Blumenkranz, M.S.; Campo, R.V. Vitrectomy for diabetic macular traction and edema associated with posterior hyaloidal traction. Ophthalmology 1992, 99, 753–759. [Google Scholar] [CrossRef] [PubMed]
- La Heij, E.C.; Hendrikse, F.; Kessels, A.G.; Derhaag, P.J. Vitrectomy results in diabetic macular oedema without evident vitreomacular traction. Graefes Arch. Clin. Exp. Ophthalmol. 2001, 239, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, K.; Furukawa, M.; Ogino, N.; Larson, E.; Iwaki, M.; Tachi, N. Long-term follow-up of vitrectomy for diffuse nontractional diabetic macular edema. Retina 2009, 29, 464–472. [Google Scholar] [CrossRef]
- Gandorfer, A.; Messmer, E.M.; Ulbig, M.W.; Kampik, A. Resolution of diabetic macular edema after surgical removal of the posterior hyaloid and the inner limiting membrane. Retina 2000, 20, 126–133. [Google Scholar] [CrossRef]
- Flaxel, C.J.; Edwards, A.R.; Aiello, L.P.; Arrigg, P.G.; Beck, R.W.; Bressler, N.M.; Bressler, S.B.; Ferris, F.L., 3rd; Gupta, S.K.; Haller, J.A.; et al. Factors associated with visual acuity outcomes after vitrectomy for diabetic macular edema: Diabetic retinopathy clinical research network. Retina 2010, 30, 1488–1495. [Google Scholar] [CrossRef]
- Elkayal, H.; Bedda, A.M.; El-Goweini, H.; Souka, A.A.; Gomaa, A.R. Pars Plana Vitrectomy versus Intravitreal Injection of Ranibizumab in the Treatment of Diabetic Macular Edema Associated with Vitreomacular Interface Abnormalities. J. Ophthalmol. 2021, 2021, 6699668. [Google Scholar] [CrossRef]
- Hwang, S.; Kang, S.W.; Kim, K.T.; Noh, H.; Kim, S.J. Three-year outcomes of vitrectomy combined with intraoperative dexamethasone implantation for non-tractional refractory diabetic macular edema. Sci. Rep. 2021, 11, 1292. [Google Scholar] [CrossRef] [PubMed]
- Mukai, R.; Matsumoto, H.; Akiyama, H. Surgical outcomes of vitrectomy for intractable diabetic macular edema. Graefes Arch. Clin. Exp. Ophthalmol. 2021, 259, 363–368. [Google Scholar] [CrossRef]
- Vikas, S.J.; Agarwal, D.; Seth, S.; Kumar, A.; Kumar, A. Comparison of anatomical and functional outcomes of vitrectomy with internal limiting membrane peeling in recalcitrant diabetic macular edema with and without traction in Indian patients. Indian J. Ophthalmol. 2021, 69, 3297–3301. [Google Scholar] [CrossRef]
- Tachi, N.; Hashimoto, Y.; Ogino, N. Cystotomy for diabetic cystoid macular edema. Doc. Ophthalmol. 1999, 97, 459–463. [Google Scholar] [CrossRef]
- Singh, R.P.; Margolis, R.; Kaiser, P.K. Cystoid puncture for chronic cystoid macular oedema. Br. J. Ophthalmol. 2007, 91, 1062–1064. [Google Scholar] [CrossRef] [PubMed]
- Asahina, Y.; Tachi, N.; Asahina, Y.; Yoshimura, K.; Ueta, Y.; Hashimoto, Y. Six-month postoperative outcomes of intraoperative OCT-guided surgical cystotomy for refractory cystoid macular edema in diabetic eyes. Clin. Ophthalmol. 2017, 11, 2099–2105. [Google Scholar] [CrossRef]
- Imai, H.; Otsuka, K.; Tetsumoto, A.; Miki, A.; Nakamura, M. Effectiveness of En Bloc Removal of Fibrinogen-Rich Component of Cystoid Lesion for the Treatment of Cystoid Macular Edema. Retina 2020, 40, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Morizane, Y.; Kimura, S.; Hosokawa, M.; Shiode, Y.; Hirano, M.; Doi, S.; Hosogi, M.; Fujiwara, A.; Inoue, Y.; Shiraga, F. Planned foveal detachment technique for the resolution of diffuse diabetic macular edema. Jpn. J. Ophthalmol. 2015, 59, 279–287. [Google Scholar] [CrossRef] [PubMed]
- El-Baha, S.M.; Abdel Hadi, A.M.; Abouhussein, M.A. Submacular Injection of Ranibizumab as a New Surgical Treatment for Refractory Diabetic Macular Edema. J. Ophthalmol. 2019, 2019, 6274209. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, K.; Ogino, N.; Fukami, M.; Furukawa, M. Removal of foveal hard exudates by subretinal balanced salt solution injection using 38-gauge needle in diabetic patients. Graefes Arch. Clin. Exp. Ophthalmol. 2020, 258, 1893–1899. [Google Scholar] [CrossRef]
- Ryan, E.H., Jr.; Han, D.P.; Ramsay, R.C.; Cantrill, H.L.; Bennett, S.R.; Dev, S.; Williams, D.F. Diabetic macular edema associated with glitazone use. Retina 2006, 26, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Oshitari, T.; Asaumi, N.; Watanabe, M.; Kumagai, K.; Mitamura, Y. Severe macular edema induced by pioglitazone in a patient with diabetic retinopathy: A case study. Vasc. Health Risk Manag. 2008, 4, 1137–1140. [Google Scholar] [CrossRef] [PubMed]
- Fong, D.S.; Contreras, R. Glitazone use associated with diabetic macular edema. Am. J. Ophthalmol. 2009, 147, 583–586.e581. [Google Scholar] [CrossRef]
- Zhang, J.; Ma, J.; Zhou, N.; Zhang, B.; An, J. Insulin use and risk of diabetic macular edema in diabetes mellitus: A systemic review and meta-analysis of observational studies. Med. Sci. Monit. 2015, 21, 929–936. [Google Scholar] [CrossRef]
- Gower, E.W.; Lovato, J.F.; Ambrosius, W.T.; Chew, E.Y.; Danis, R.P.; Davis, M.D.; Goff, D.C., Jr.; Greven, C.M. Lack of Longitudinal Association Between Thiazolidinediones and Incidence and Progression of Diabetic Eye Disease: The ACCORD Eye Study. Am. J. Ophthalmol. 2018, 187, 138–147. [Google Scholar] [CrossRef]
- Dicembrini, I.; Nreu, B.; Scatena, A.; Andreozzi, F.; Sesti, G.; Mannucci, E.; Monami, M. Microvascular effects of glucagon-like peptide-1 receptor agonists in type 2 diabetes: A meta-analysis of randomized controlled trials. Acta Diabetol. 2017, 54, 933–941. [Google Scholar] [CrossRef]
- Nangaku, M.; Kondo, K.; Kokado, Y.; Ueta, K.; Kaneko, G.; Tandai, T.; Kawaguchi, Y.; Komatsu, Y. Phase 3 Randomized Study Comparing Vadadustat with Darbepoetin Alfa for Anemia in Japanese Patients with Nondialysis-Dependent CKD. J. Am. Soc. Nephrol. 2021, 32, 1779–1790. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.J. Renal glucose reabsorption inhibitors to treat diabetes. Trends Pharmacol. Sci. 2011, 32, 63–71. [Google Scholar] [CrossRef]
- Packer, M.; Anker, S.D.; Butler, J.; Filippatos, G.; Pocock, S.J.; Carson, P.; Januzzi, J.; Verma, S.; Tsutsui, H.; Brueckmann, M.; et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N. Engl. J. Med. 2020, 383, 1413–1424. [Google Scholar] [CrossRef]
- Wanner, C.; Inzucchi, S.E.; Lachin, J.M.; Fitchett, D.; von Eynatten, M.; Mattheus, M.; Johansen, O.E.; Woerle, H.J.; Broedl, U.C.; Zinman, B. Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 323–334. [Google Scholar] [CrossRef]
- Hanaguri, J.; Yokota, H.; Kushiyama, A.; Kushiyama, S.; Watanabe, M.; Yamagami, S.; Nagaoka, T. The Effect of Sodium-Dependent Glucose Cotransporter 2 Inhibitor Tofogliflozin on Neurovascular Coupling in the Retina in Type 2 Diabetic Mice. Int. J. Mol. Sci. 2022, 23, 1362. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Xu, Q.; Li, H.; Meng, Z.; Hao, M.; Ma, X.; Lin, W.; Kuang, H. Dapagliflozin Reduces Apoptosis of Diabetic Retina and Human Retinal Microvascular Endothelial Cells Through ERK1/2/cPLA2/AA/ROS Pathway Independent of Hypoglycemic. Front. Pharmacol. 2022, 13, 827896. [Google Scholar] [CrossRef]
- Matthews, J.; Herat, L.; Rooney, J.; Rakoczy, E.; Schlaich, M.; Matthews, V.B. Determining the role of SGLT2 inhibition with Empagliflozin in the development of diabetic retinopathy. Biosci. Rep. 2022, 42, BSR20212209. [Google Scholar] [CrossRef]
- Mieno, H.; Yoneda, K.; Yamazaki, M.; Sakai, R.; Sotozono, C.; Fukui, M. The Efficacy of Sodium-Glucose Cotransporter 2 (SGLT2) inhibitors for the treatment of chronic diabetic macular oedema in vitrectomised eyes: A retrospective study. BMJ Open Ophthalmol. 2018, 3, e000130. [Google Scholar] [CrossRef]
- Yoshizumi, H.; Ejima, T.; Nagao, T.; Wakisaka, M. Recovery from Diabetic Macular Edema in a Diabetic Patient After Minimal Dose of a Sodium Glucose Co-Transporter 2 Inhibitor. Am. J. Case Rep. 2018, 19, 462–466. [Google Scholar] [CrossRef]
- Takatsuna, Y.; Ishibashi, R.; Tatsumi, T.; Koshizaka, M.; Baba, T.; Yamamoto, S.; Yokote, K. Sodium-Glucose Cotransporter 2 Inhibitors Improve Chronic Diabetic Macular Edema. Case Rep. Ophthalmol. Med. 2020, 2020, 8867079. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.C.; Shao, S.C.; Lai, E.C.; Lee, C.N.; Hung, M.J.; Lai, C.C.; Hsu, S.M.; Hung, J.H. Risk of diabetic macular oedema with sodium-glucose cotransporter-2 inhibitors in type 2 diabetes patients: A multi-institutional cohort study in Taiwan. Diabetes Obes. Metab. 2021, 23, 2067–2076. [Google Scholar] [CrossRef] [PubMed]
- Tatsumi, T.; Oshitari, T.; Takatsuna, Y.; Ishibashi, R.; Koshizaka, M.; Shiko, Y.; Baba, T.; Yokote, K.; Yamamoto, S. Sodium-Glucose Co-Transporter 2 Inhibitors Reduce Macular Edema in Patients with Diabetes mellitus. Life 2022, 12, 692. [Google Scholar] [CrossRef]
- Chung, Y.R.; Ha, K.H.; Lee, K.; Kim, D.J. Effects of sodium-glucose cotransporter-2 inhibitors and dipeptidyl peptidase-4 inhibitors on diabetic retinopathy and its progression: A real-world Korean study. PLoS ONE 2019, 14, e0224549. [Google Scholar] [CrossRef]
- Wakisaka, M.; Yoshinari, M.; Yamamoto, M.; Nakamura, S.; Asano, T.; Himeno, T.; Ichikawa, K.; Doi, Y.; Fujishima, M. Na+-dependent glucose uptake and collagen synthesis by cultured bovine retinal pericytes. Biochim. Biophys. Acta 1997, 1362, 87–96. [Google Scholar] [CrossRef]
- Wakisaka, M.; Kitazono, T.; Kato, M.; Nakamura, U.; Yoshioka, M.; Uchizono, Y.; Yoshinari, M. Sodium-coupled glucose transporter as a functional glucose sensor of retinal microvascular circulation. Circ. Res. 2001, 88, 1183–1188. [Google Scholar] [CrossRef] [PubMed]
- Najib, J. Fenofibrate in the treatment of dyslipidemia: A review of the data as they relate to the new suprabioavailable tablet formulation. Clin. Ther. 2002, 24, 2022–2050. [Google Scholar] [CrossRef] [PubMed]
- Tomita, Y.; Lee, D.; Tsubota, K.; Kurihara, T. PPARalpha Agonist Oral Therapy in Diabetic Retinopathy. Biomedicines 2020, 8, 433. [Google Scholar] [CrossRef] [PubMed]
- Group, A.S.; Group, A.E.S.; Chew, E.Y.; Ambrosius, W.T.; Davis, M.D.; Danis, R.P.; Gangaputra, S.; Greven, C.M.; Hubbard, L.; Esser, B.A.; et al. Effects of medical therapies on retinopathy progression in type 2 diabetes. N. Engl. J. Med. 2010, 363, 233–244. [Google Scholar] [CrossRef]
- Keech, A.C.; Mitchell, P.; Summanen, P.A.; O’Day, J.; Davis, T.M.; Moffitt, M.S.; Taskinen, M.R.; Simes, R.J.; Tse, D.; Williamson, E.; et al. Effect of fenofibrate on the need for laser treatment for diabetic retinopathy (FIELD study): A randomised controlled trial. Lancet 2007, 370, 1687–1697. [Google Scholar] [CrossRef]
- Fujita, Y.; Inagaki, N. Metformin: New Preparations and Nonglycemic Benefits. Curr. Diab. Rep. 2017, 17, 5. [Google Scholar] [CrossRef]
- Yi, Q.Y.; Deng, G.; Chen, N.; Bai, Z.S.; Yuan, J.S.; Wu, G.H.; Wang, Y.W.; Wu, S.J. Metformin inhibits development of diabetic retinopathy through inducing alternative splicing of VEGF-A. Am. J. Transl. Res. 2016, 8, 3947–3954. [Google Scholar]
- Zhang, Y.; Chen, F.; Wang, L. Metformin inhibits development of diabetic retinopathy through microRNA-497a-5p. Am. J. Transl. Res. 2017, 9, 5558–5566. [Google Scholar]
- Xu, L.; Kong, L.; Wang, J.; Ash, J.D. Stimulation of AMPK prevents degeneration of photoreceptors and the retinal pigment epithelium. Proc. Natl. Acad. Sci. USA 2018, 115, 10475–10480. [Google Scholar] [CrossRef]
- Uwimana, A.; Ma, C.; Chen, S.; Ma, X. Metformin therapy as a strategy to compensate anti-VEGF resistance in patients with diabetic macular edema. Medicine 2022, 101, e31266. [Google Scholar] [CrossRef]
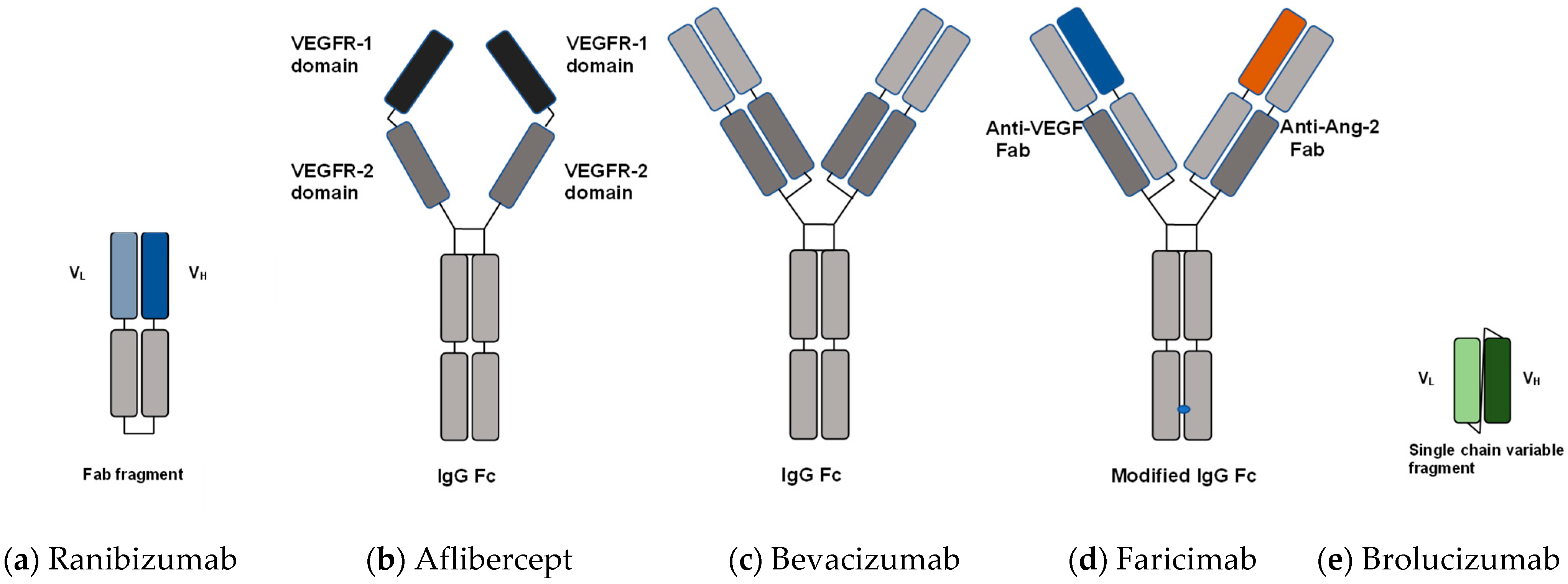
| Anti-VEGF Agents | Ranibizumab | Aflibercept | Bevacizumab | Faricimab | Brolucizumab |
|---|---|---|---|---|---|
| Molecular format | Fab fragment | VEGFR1/2-Fc fusion protein | Full antibody (lgG1) | anti-VEGF/anti-Ang-2 humanized bispecific monoclonal antibody | Single-chain variable fragment |
| Molecular weight | ~48 × 103 | 97–115 × 103 | ~149 × 103 | 150 × 103 | 26 × 103 |
| Binding molecule | VEGF-A | VEGF-A, VEGF-B, PlGF-1, PlGF-2 | VEGF-A | VEGF-A, Ang-2 | VEGF-A |
| Clinical dose | 0.50 mg | 2.0 mg | 1.25 mg | 6.0 mg | 6.0 mg |
| Relative number of molecules per injection | 0.5–0.6 | 1.0 | 0.4–0.5 | 1.15 | 11.2–13.3 |
| Author/Journal/Year | Material and Protocol Study Period | Retreatment Criteria | Study Design Follow-Up Periods | Patient Inclusion Criteria of CRT and BCVA | CRT (µm) Baseline ⇒ Final * Difference | BCVA (logMAR) Baseline ⇒ Final * Difference | No. of Injections | † p |
|---|---|---|---|---|---|---|---|---|
| Moisseiev E et al. Eur J Ophthalmol 2018 [136] | IVR + SML vs. IVR Jan 2013–Jun 2015 | Retrospective 12 months | n = 38 (19 vs. matched control 19) All patients had no more than 3 prior IVR | IVR 408.4 ⇒ 335.9 72.5 | 0.41 ⇒ 0.39 0.02 | 5.6/12 months | <0.001 | |
| IVR + SML 316.8 ⇒ 282.6 34.2 | 0.29 ⇒ 0.24 0.05 | 1.7/12 months | ||||||
| Altınel MG, et al. Lasers Med Sci. 2021 [137] | IVB + SML vs. IVB Sep 2017–Mar 2020 | Retrospective 15 months | n = 80 (40 vs. 40) Excluded intravitreal injections within the preceding 6 months, CRT > 250 µm | IVB 384.7 ⇒ 325.8 58.9 | 0.39 ⇒ 0.32 0.07 | 8.65/15 months | <0.05 | |
| IVB + SML 379.2 ⇒ 292.6 86.6 | 0.38 ⇒ 0.25 0.13 | 7.38/15 months | ||||||
| El Matri L, et al. Ther Adv Ophthalmol. 2021 [138] | IVB + SML vs. IVB 3 + PRN Jan 2015–Jan 2019 | BCVA ⩽ 20/25 Presence of IRF and/or SRF | Retrospective 12 months | n = 98 eyes (49 vs. 49) (63 patients) Treatment naïve for DME CRT ⩽ 500 µm, BCVA ⩾ 20/400 | IVB 359.9 ⇒ 305.9 54.0 | 0.60 ⇒ 0.49 0.11 | 7.2/12 months | <0.005 |
| IVB + SML 479.1 ⇒ 289.6 189.5 | 0.69 ⇒ 0.50 0.19 | 4.1/12 months | ||||||
| Khattab AM et al. Graefes Arch Clin Exp Ophthalmol 2019 [139] | IVA + SML vs. IVA 3 + PRN Feb 2017–Dec 2018 | CRT > 250 µm | Prospective 18 months | n = 54 eyes (27 vs. 27) (51 patients) Excluded intravitreal injections within the preceding 6 months, CRT > 250 µm, BCVA: 20/400–20/40 | IVA 462.0 ⇒ 249.5 212.5 | ‡ 31.7 ⇒ 50.6 18.9 (0.378) | 7.3/18 months | <0.005 |
| IVA + SML 457.1 ⇒ 244.6 212.5 | ‡ 35.0 ⇒ 54.8 19.8 (0.396) | 4.1/18 months | ||||||
| Kanar HS et al. Ind J Ophthalmol 2020 [141] | IVA + SML vs. IVA 3 + PRN Apr 2015–Nov 2017 | 20% increase in CRT 1-line decrease at BCVA | Prospective 12 months | n = 56 (28 vs. 28) Treatment naïve for DME CRT ≧ 300 µm, BCVA: 0.2–0.9 | IVA 451.28 ⇒ 328.8 122.5 | 0.38 ⇒ 0.20 0.18 | 5.39/12 months | <0.001 |
| IVA + SML 466.07 ⇒ 312.0 154.1 | 0.40 ⇒ 0.17 0.23 | 3.21/12 months | ||||||
| Abouhussein MA et al. Int Ophthalmol 2020 [140] | IVA + SML vs. IVA 3 + PRN period: not stated | CRT ≧ 300 µm | Prospective 15 months | n = 40 (20 vs. 20) Treatment naïve for DME CRT ≧ 300 µm, BCVA > 3/60 | IVA 457.9 ⇒ 290.5 167.4 | 0.70 ⇒ 0.24 0.46 | 8.4/15 months | 0.029 |
| IVA + SML 469.6 ⇒ 288.5 181.1 | 0.76 ⇒ 0.20 0.56 | 7.5/15 months | ||||||
| Koushan K et al. Clin Ophthalmol 2022 [142] | IVA + SML vs. IVA 1 (Continue until ME resolves) + PRN Mar 2017–Oct 2018 | 10% change in CRT 1-Snellen-line change at BCVA | Prospective 12 months | n = 30 (15 vs. 15) Excluded intravitreal injections within the preceding 120 days, CRT > 310 µm, BCVA: 20/400–20/30 | IVA 433.4 ⇒ 288.3 145.1 | 0.38 ⇒ 0.32 0.06 | 8.5/12 months | 0.61 |
| IVA + SL 457.8 ⇒ 289.5 168.3 | 0.36 ⇒ 0.22 0.14 | 7.9/12 months | ||||||
| Tatsumi T et al. Sci Rep 2022 [143] | IVA + SL vs. IVA 3 + PRN Sep 2016–Sep 2020 | 100 µm increase in CRT 2-line decrease at BCVA | Prospective 24 months | n = 51 (25 vs. 26) Excluded intravitreal injections within the preceding 90 days, CRT > 300 µm, BCVA: 0.05–0.7 | IVA 442.8 ⇒ 319.5 123.3 | 0.37 ⇒ 0.32 0.05 | 5.86/24 months | 0.86 |
| IVA + SL 472.8 ⇒ 329.5 143.3 | 0.48 ⇒ 0.28 0.20 | 6.05/24 months |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tatsumi, T. Current Treatments for Diabetic Macular Edema. Int. J. Mol. Sci. 2023, 24, 9591. https://doi.org/10.3390/ijms24119591
Tatsumi T. Current Treatments for Diabetic Macular Edema. International Journal of Molecular Sciences. 2023; 24(11):9591. https://doi.org/10.3390/ijms24119591
Chicago/Turabian StyleTatsumi, Tomoaki. 2023. "Current Treatments for Diabetic Macular Edema" International Journal of Molecular Sciences 24, no. 11: 9591. https://doi.org/10.3390/ijms24119591
APA StyleTatsumi, T. (2023). Current Treatments for Diabetic Macular Edema. International Journal of Molecular Sciences, 24(11), 9591. https://doi.org/10.3390/ijms24119591






