Exploring the Interplay between Polyphenols and Lysyl Oxidase Enzymes for Maintaining Extracellular Matrix Homeostasis
Abstract
:1. Introduction
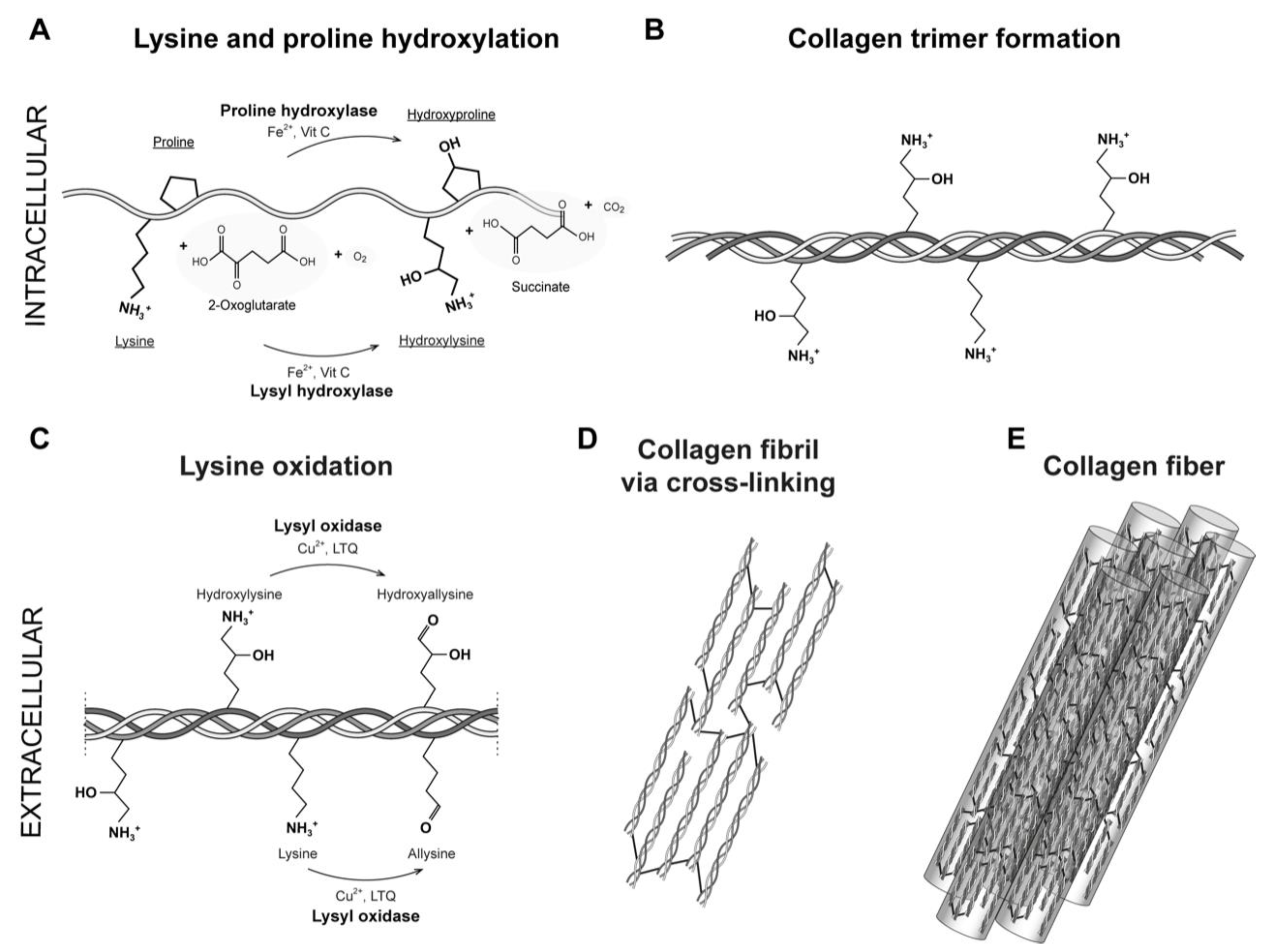
2. Fibrillar Type I Collagen
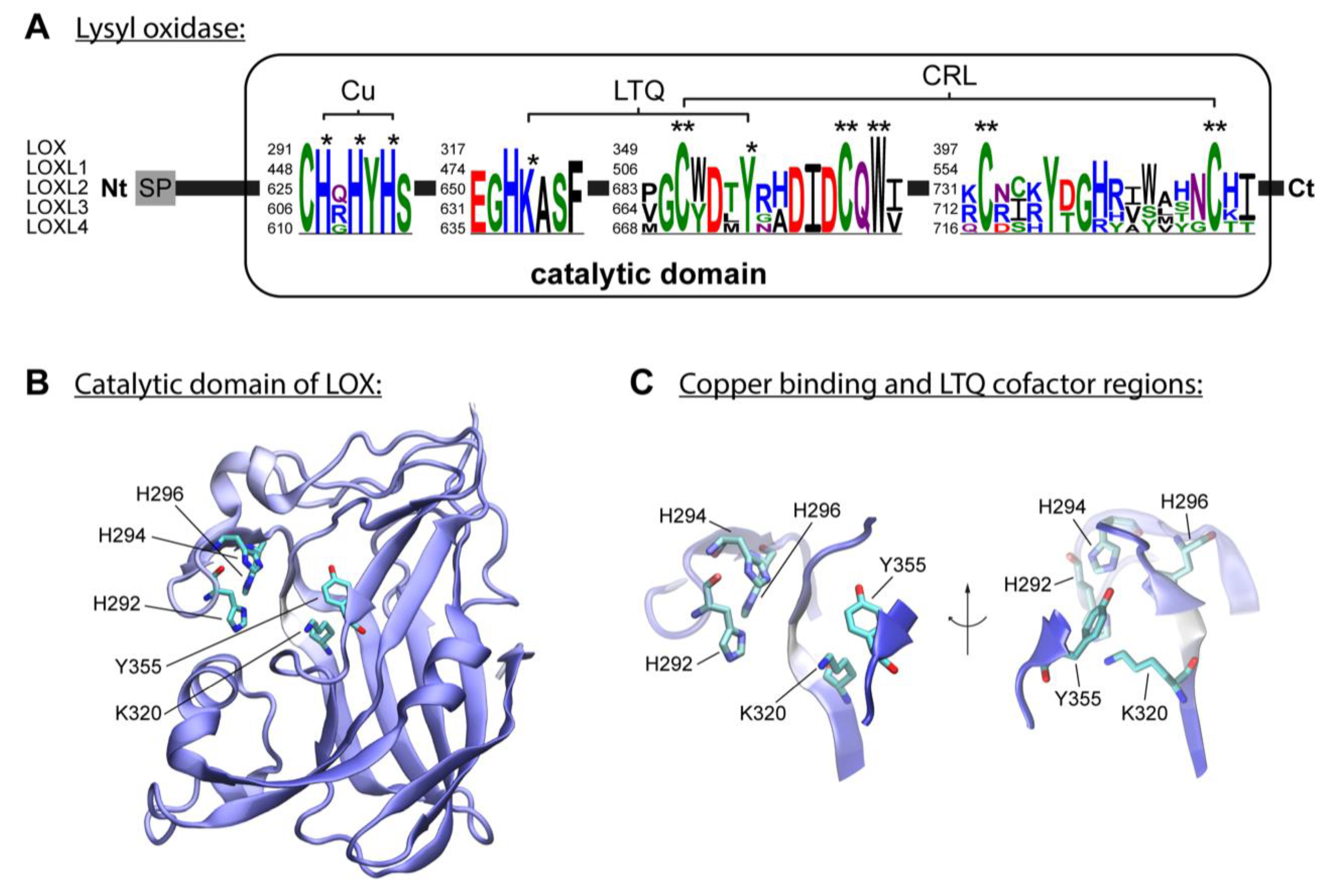
3. Enzymatic Collagen Cross-Linking Mediated by Lysyl Oxidases
4. Lysine-Derived Cross-Links and ECM Proteins
5. Lysine-Derived Cross-Link Analysis Methods
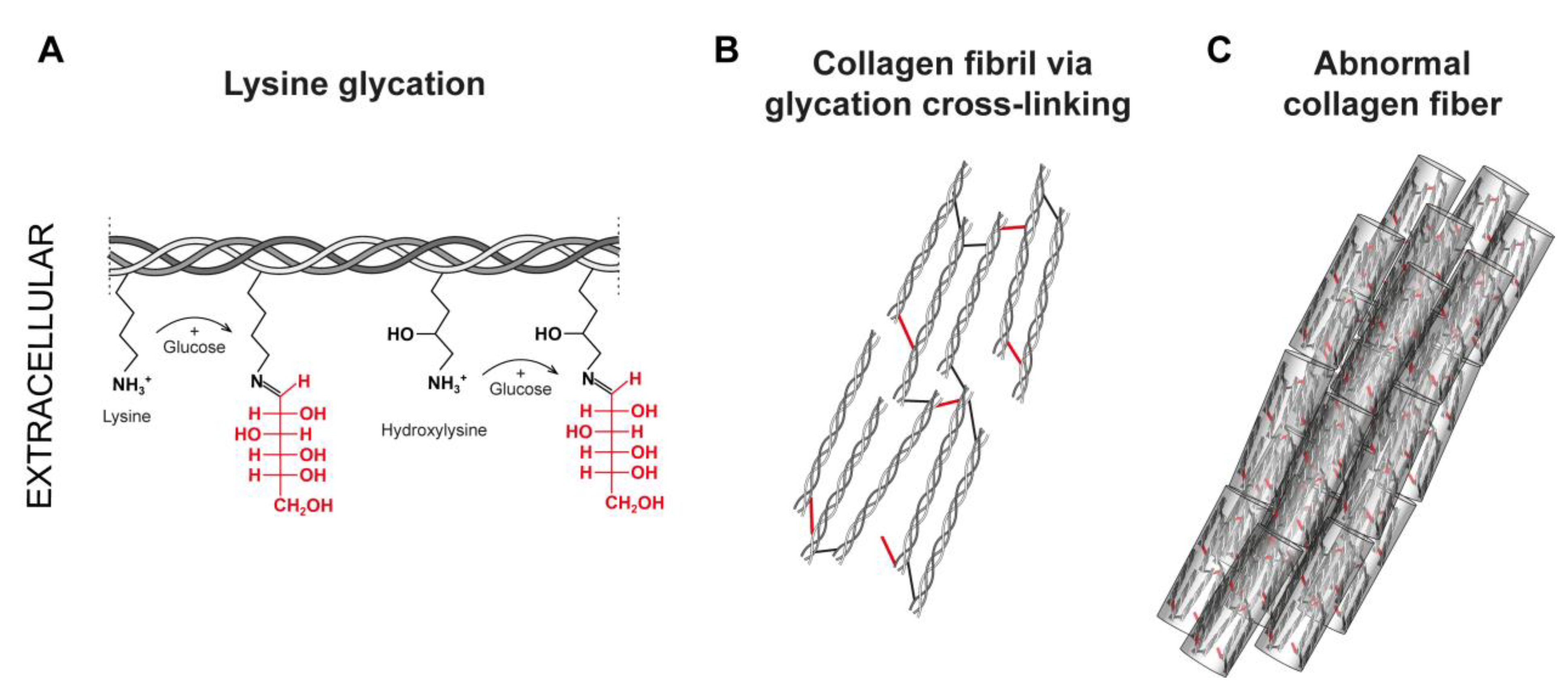
6. Non-Enzymatic Collagen Glycation
7. Lysyl Oxidase Activity of Polyphenols
8. Anti-Glycating Activity of Polyphenols
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gaar, J.; Naffa, R.; Brimble, M. Enzymatic and non-enzymatic crosslinks found in collagen and elastin and their chemical synthesis. Org. Chem. Front. 2020, 7, 2789–2814. [Google Scholar] [CrossRef]
- Robins, S. Biochemistry and functional significance of collagen cross-linking. Biochem. Soc. Trans. 2007, 35, 849–852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heikkinen, J.; Risteli, M.; Wang, C.; Latvala, J.; Rossi, M.; Valtavaara, M.; Myllylä, R. Lysyl Hydroxylase 3 Is a Multifunctional Protein Possessing Collagen Glucosyltransferase Activity. J. Biol. Chem. 2000, 275, 36158–36163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirchner, M.; Deng, H.; Xu, Y. Heterogeneity in proline hydroxylation of fibrillar collagens observed by mass spectrometry. PLoS ONE 2021, 16, e0250544. [Google Scholar] [CrossRef]
- Yamauchi, M.; Sricholpech, M. Lysine post-translational modifications of collagen. Essays Biochem. 2012, 52, 113–133. [Google Scholar] [CrossRef] [Green Version]
- Lucero, H.A.; Kagan, H.M. Lysyl oxidase: An oxidative enzyme and effector of cell function. Cell Mol. Life Sci. 2006, 63, 2304–2316. [Google Scholar] [CrossRef]
- Ricard-Blum, S. The collagen family. Cold Spring Harb. Perspect. Biol. 2011, 3, a004978. [Google Scholar] [CrossRef] [Green Version]
- Siegel, R.C. Biosynthesis of Collagen Crosslinks: Increased Activity of Purified Lysyl Oxidase with Reconstituted Collagen Fibrils. Proc. Natl. Acad. Sci. USA 1974, 71, 4826–4830. [Google Scholar] [CrossRef] [PubMed]
- Trackman, P.C. Enzymatic and non-enzymatic functions of the lysyl oxidase family in bone. Matrix Biol. 2016, 52–54, 7–18. [Google Scholar] [CrossRef] [Green Version]
- Van der Slot-Verhoeven, A.J.; van Dura, E.A.; Attema, J.; Blauw, B.; DeGroot, J.; Huizinga, T.W.; Zuurmond, A.M.; Bank, R.A. The type of collagen cross-link determines the reversibility of experimental skin fibrosis. Biochim. Biophys. Acta 2005, 1740, 60–67. [Google Scholar] [CrossRef] [Green Version]
- Nagan, N.; Kagan, H. Modulation of lysyl oxidase activity toward peptidyl lysine by vicinal dicarboxylic amino acid residues. Implications for collagen cross-linking. J. Biol. Chem. 1994, 269, 22366–22371. [Google Scholar] [CrossRef] [PubMed]
- Finney, J.; Moon, H.J.; Ronnebaum, T.; Lantz, M.; Mure, M. Human copper-dependent amine oxidases. Arch. Biochem. Biophys. 2014, 546, 19–32. [Google Scholar] [CrossRef]
- Hudson, D.M.; Archer, M.; Rai, J.; Weis, M.; Fernandes, R.J.; Eyre, D.R. Age-related type I collagen modifications reveal tissue-defining differences between ligament and tendon. Matrix Biol. Plus 2021, 12, 100070. [Google Scholar] [CrossRef]
- Hudson, D.M.; Archer, M.; King, K.B.; Eyre, D.R. Glycation of type I collagen selectively targets the same helical domain lysine sites as lysyl oxidase–mediated cross-linking. J. Biol. Chem. 2018, 293, 15620–15627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.-Y.; Zhang, J.-Q.; Li, L.; Guo, M.-M.; He, Y.-F.; Dong, Y.-M.; Meng, H.; Yi, F. Advanced Glycation End Products in the Skin: Molecular Mechanisms, Methods of Measurement, and Inhibitory Pathways. Front. Med. 2022, 9, 837222. [Google Scholar] [CrossRef]
- Imran, M.; Rauf, A.; Imran, A.; Nadeem, M.; Ahmad, Z.; Atif, M.; Awais, M.; Sami, M.; Fatima, Z.; Waqar, B.A. Health Benefits of Grapes Polyphenols. J. Environ. Agric. Sci. 2017, 10, 40–51. [Google Scholar]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Polyphenols and Glycemic Control. Nutrients 2016, 8, 17. [Google Scholar] [CrossRef] [Green Version]
- Stagos, D. Antioxidant Activity of Polyphenolic Plant Extracts. Antioxidants 2020, 9, 19. [Google Scholar] [CrossRef] [Green Version]
- Shishido, S.; Miyano, R.; Nakashima, T.; Matsuo, H.; Iwatsuki, M.; Nakamura, K.; Kanno, T.; Egusa, H.; Niwano, Y. A novel pathway for the photooxidation of catechin in relation to its prooxidative activity. Sci. Rep. 2018, 8, 12888. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Itakura, M.; Kitazawa, R.; Lim, S.-Y.; Nagata, K.; Shibata, T.; Akagawa, M.; Uchida, K. Oxidative deamination of lysine residues by polyphenols generates an equilibrium of aldehyde and 2-piperidinol products. J. Biol. Chem. 2021, 297, 101035. [Google Scholar] [CrossRef]
- Akagawa, M.; Suyama, K. Amine oxidase-like activity of polyphenols. JBIC J. Biol. Inorg. Chem. 2001, 268, 1953–1963. [Google Scholar] [CrossRef]
- Akagawa, M.; Shigemitsu, T.; Suyama, K. Oxidative Deamination of Benzylamine and Lysine Residue in Bovine Serum Albumin by Green Tea, Black Tea, and Coffee. J. Agric. Food Chem. 2005, 53, 8019–8024. [Google Scholar] [CrossRef]
- Bravo, L. Polyphenols: Chemistry, Dietary Sources, Metabolism, and Nutritional Significance. Nutr. Rev. 2009, 56, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Prochazkova, D.; Bousova, I.; Wilhelmova, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef]
- Añazco, C.; Rojas, A.; Gonzalez, I.; Castro, M.A.; Robert, P.; Oyarzun-Ampuero, F. Dermal Collagen Stabilization by Polyphenols and Spray Drying as an Encapsulation Strategy. Curr. Top. Med. Chem. 2018, 18, 1259–1268. [Google Scholar] [CrossRef] [PubMed]
- Hanauske-Abel, H.M.; Tschank, G.; Günzler, V.; Baader, E.; Gallop, P. Pyrroloquinoline quinone and molecules mimicking its functional domains Modulators of connective tissue formation? FEBS Lett. 1987, 214, 236–243. [Google Scholar] [CrossRef] [Green Version]
- DiSilvestro, R.A.; Harris, E.D. Evaluation of (+)-catechin action on lysyl oxidase activity in aortic tissue. Biochem. Pharmacol. 1983, 32, 343–346. [Google Scholar] [CrossRef] [PubMed]
- González, I.; Morales, M.A.; Rojas, A. Polyphenols and AGEs/RAGE axis. Trends and challenges. Food Res. Int. 2020, 129, 108843. [Google Scholar] [CrossRef]
- Odjakova, M.; Popova, E.; Al Sharif, M.; Mironova, R. Plant-Derived Agents with Anti-Glycation Activity. Glycosylation 2012, 10, 48186. [Google Scholar] [CrossRef] [Green Version]
- Theocharis, A.D.; Skandalis, S.S.; Gialeli, C.; Karamanos, N.K. Extracellular matrix structure. Adv. Drug Delivery Rev. 2016, 97, 4–27. [Google Scholar] [CrossRef]
- Karamanos, N.K.; Theocharis, A.D.; Piperigkou, Z.; Manou, D.; Passi, A.; Skandalis, S.S.; Vynios, D.H.; Orian-Rousseau, V.; Ricard-Blum, S.; Schmelzer, C.E.; et al. A guide to the composition and functions of the extracellular matrix. FEBS J. 2021, 288, 6850–6912. [Google Scholar] [CrossRef]
- Gauza-Włodarczyk, M.; Kubisz, L.; Włodarczyk, D. Amino acid composition in determination of collagen origin and assessment of physical factors effects. Int. J. Biol. Macromol. 2017, 104, 987–991. [Google Scholar] [CrossRef] [PubMed]
- Gordon, M.K.; Hahn, R.A. Collagens. Cell Tissue Res. 2010, 339, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Fidler, A.L.; Boudko, S.P.; Rokas, A.; Hudson, B.G. The triple helix of collagens—An ancient protein structure that enabled animal multicellularity and tissue evolution. J. Cell Sci. 2018, 131, jcs203950. [Google Scholar] [CrossRef] [Green Version]
- Ramachandran, G.; Kartha, G. Structure of collagen. Nature 1955, 176, 593–595. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xu, S.; Shen, L.; Li, G. Factors affecting thermal stability of collagen from the aspects of extraction, processing and modification. J. Leather Sci. Eng. 2020, 2, 1–29. [Google Scholar] [CrossRef]
- Reiser, K.M.; Amigable, M.; Last, J.A. Nonenzymatic glycation of type I collagen. The effects of aging on preferential glycation sites. J. Biol. Chem. 1992, 267, 24207–24216. [Google Scholar] [CrossRef] [PubMed]
- Gautieri, A.; Redaelli, A.; Buehler, M.J.; Vesentini, S. Age-and diabetes-related nonenzymatic crosslinks in collagen fibrils: Candidate amino acids involved in Advanced Glycation End-products. Matrix Biol. 2014, 34, 89–95. [Google Scholar] [CrossRef]
- Eekhoff, J.D.; Fang, F.; Lake, S.P. Multiscale mechanical effects of native collagen cross-linking in tendon. Connect. Tissue Res. 2018, 59, 410–422. [Google Scholar] [CrossRef]
- Cabral-Pacheco, G.; Garza-Veloz, I.; La Rosa, C.C.-D.; Ramirez-Acuña, J.M.; Perez-Romero, B.; Guerrero-Rodriguez, J.F.; Martinez-Avila, N.; Martinez-Fierro, M.L. The Roles of Matrix Metalloproteinases and Their Inhibitors in Human Diseases. Int. J. Mol. Sci. 2020, 21, 9739. [Google Scholar] [CrossRef]
- Shoulders, M.D.; Raines, R.T. Collagen Structure and Stability. Annu. Rev. Biochem. 2009, 78, 929–958. [Google Scholar] [CrossRef] [Green Version]
- Makris, E.A.; MacBarb, R.F.; Responte, D.J.; Hu, J.C.; Athanasiou, K.A. A copper sulfate and hydroxylysine treatment regimen for enhancing collagen cross-linking and biomechanical properties in engineered neocartilage. FASEB J. 2013, 27, 2421–2430. [Google Scholar] [CrossRef] [Green Version]
- Knott, L.; Bailey, A. Collagen cross-links in mineralizing tissues: A review of their chemistry, function, and clinical relevance. Bone 1998, 22, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Reiser, K.; McCormick, R.J.; Rucker, R.B. Enzymatic and nonenzymatic cross-linking of collagen and elastin. FASEB J. 1992, 6, 2439–2449. [Google Scholar] [CrossRef] [PubMed]
- Molnar, J.; Fong, K.; He, Q.; Hayashi, K.; Kim, Y.; Fong, S.; Fogelgren, B.; Szauter, K.M.; Mink, M.; Csiszar, K. Structural and functional diversity of lysyl oxidase and the LOX-like proteins. Biochim. Biophys. Acta Proteins Proteom. 2003, 1647, 220–224. [Google Scholar] [CrossRef]
- Trackman, P.C.; Kagan, H.M. Nonpeptidyl amine inhibitors are substrates of lysyl oxidase. J. Biol. Chem. 1979, 254, 7831–7836. [Google Scholar] [CrossRef] [PubMed]
- Vallet, S.D.; Ricard-Blum, S. Lysyl oxidases: From enzyme activity to extracellular matrix cross-links. Essays Biochem. 2019, 63, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Kagan, H.M.; Li, W. Lysyl oxidase: Properties, specificity, and biological roles inside and outside of the cell. J. Cell Biochem. 2003, 88, 660–672. [Google Scholar] [CrossRef]
- Liburkin-Dan, T.; Toledano, S.; Neufeld, G. Lysyl Oxidase Family Enzymes and Their Role in Tumor Progression. Int. J. Mol. Sci. 2022, 23, 6249. [Google Scholar] [CrossRef]
- Csiszar, K. Lysyl oxidases: A novel multifunctional amine oxidase family. Prog. Nucleic Acid Res. Mol. Biol. 2001, 70, 1–32. [Google Scholar] [CrossRef]
- Gacheru, S.N.; Trackman, P.C.; Shah, M.; O’Gara, C.Y.; Spacciapoli, P.; Greenaway, F.T.; Kagan, H.M. Structural and catalytic properties of copper in lysyl oxidase. J. Biol. Chem. 1990, 265, 19022–19027. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Poe, A.; Yus, M.M.; Pak, L.; Nandakumar, K.; Santhanam, L. Lysyl oxidase-like 2 processing by factor Xa modulates its activity and substrate preference. Commun. Biol. 2023, 6, 375. [Google Scholar] [CrossRef] [PubMed]
- Añazco, C.; Lopez-Jimenez, A.J.; Rafi, M.; Vega-Montoto, L.; Zhang, M.Z.; Hudson, B.G.; Vanacore, R.M. Lysyl Oxidase-like-2 Cross-links Collagen IV of Glomerular Basement Membrane. J. Biol. Chem. 2016, 291, 25999–26012. [Google Scholar] [CrossRef] [Green Version]
- Bignon, M.; Pichol-Thievend, C.; Hardouin, J.; Malbouyres, M.; Bréchot, N.; Nasciutti, L.; Barret, A.; Teillon, J.; Guillon, E.; Etienne, E.; et al. Lysyl oxidase-like protein-2 regulates sprouting angiogenesis and type IV collagen assembly in the endothelial basement membrane. Blood J. Am. Soc. Hematol. 2011, 118, 3979–3989. [Google Scholar] [CrossRef] [PubMed]
- Busnadiego, O.; González-Santamaría, J.; Lagares, D.; Guinea-Viniegra, J.; Pichol-Thievend, C.; Muller, L.; Rodríguez-Pascual, F. LOXL4 Is Induced by Transforming Growth Factor β1 through Smad and JunB/Fra2 and Contributes to Vascular Matrix Remodeling. Mol. Cell Biol. 2013, 33, 2388–2401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aronoff, M.R.; Hiebert, P.; Hentzen, N.B.; Werner, S.; Wennemers, H. Imaging and targeting LOX-mediated tissue remodeling with a reactive collagen peptide. Nat. Chem. Biol. 2021, 17, 865–871. [Google Scholar] [CrossRef]
- Moon, H.-J.; Finney, J.; Xu, L.; Moore, D.; Welch, D.; Mure, M. MCF-7 Cells Expressing Nuclear Associated Lysyl Oxidase-like 2 (LOXL2) Exhibit an Epithelial-to-Mesenchymal Transition (EMT) Phenotype and Are Highly Invasive in Vitro. J. Biol. Chem. 2013, 288, 30000–30008. [Google Scholar] [CrossRef] [Green Version]
- Añazco, C.; Delgado-López, F.; Araya, P.; González, I.; Morales, E.; Pérez-Castro, R.; Romero, J.; Rojas, A. Lysyl oxidase isoforms in gastric cancer. Biomarkers Med. 2016, 10, 987–998. [Google Scholar] [CrossRef]
- Añazco, C.; Cerro, S.; Pereira, N.; Rojas, C.; Torres, A.; Vidal-Beltrán, I. Dysregulation of Lysyl Oxidases Expression in Diabetic Nephropathy and Renal Cell Carcinoma. Curr. Drug Targets 2021, 22, 1916–1925. [Google Scholar] [CrossRef]
- Miner, J.H. The glomerular basement membrane. Exp. Cell Res. 2012, 318, 973–978. [Google Scholar] [CrossRef] [Green Version]
- Di Donato, A.; Ghiggeri, G.M.; Di Duca, M.; Jivotenko, E.; Acinni, R.; Campolo, J.; Ginevri, F.; Gusmano, R. Lysyl Oxidase Expression and Collagen Cross-Linking during Chronic Adriamycin Nephropathy. Nephron 1997, 76, 192–200. [Google Scholar] [CrossRef]
- Neusser, M.A.; Lindenmeyer, M.T.; Moll, A.G.; Segerer, S.; Edenhofer, I.; Sen, K.; Stiehl, D.P.; Kretzler, M.; Gröne, H.-J.; Schlöndorff, D.; et al. Human Nephrosclerosis Triggers a Hypoxia-Related Glomerulopathy. Am. J. Pathol. 2010, 176, 594–607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, H.-Y.; Li, Q.; Wong, W.R.; N’diaye, E.-N.; Caplazi, P.; Bender, H.; Huang, Z.; Arlantico, A.; Jeet, S.; Wong, A.; et al. LOXL4, but not LOXL2, is the critical determinant of pathological collagen cross-linking and fibrosis in the lung. Sci. Adv. 2023, 9, eadf0133. [Google Scholar] [CrossRef] [PubMed]
- Setargew, Y.F.; Wyllie, K.; Grant, R.D.; Chitty, J.L.; Cox, T.R. Targeting Lysyl Oxidase Family Meditated Matrix Cross-Linking as an Anti-Stromal Therapy in Solid Tumours. Cancers 2021, 13, 491. [Google Scholar] [CrossRef]
- Grau-Bové, X.; Ruiz-Trillo, I.; Rodriguez-Pascual, F. Origin and evolution of lysyl oxidases. Sci. Rep. 2015, 5, 10568. [Google Scholar] [CrossRef] [Green Version]
- Bollinger, J.A.; Brown, D.E.; Dooley, D.M. The Formation of Lysine Tyrosylquinone (LTQ) Is a Self-Processing Reaction. Expression and Characterization of a Drosophila Lysyl Oxidase. Biochemistry 2005, 44, 11708–11714. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, Q.; Wu, J.; Wang, J.; Shi, Y.; Liu, M. Crystal structure of human lysyl oxidase-like 2 (hLOXL2) in a precursor state. Proc. Natl. Acad. Sci. USA 2018, 115, 3828–3833. [Google Scholar] [CrossRef] [Green Version]
- Shanbhag, V.; Jasmer-McDonald, K.; Zhu, S.; Martin, A.L.; Gudekar, N.; Khan, A.; Ladomersky, E.; Singh, K.; Weisman, G.A.; Petris, M.J. ATP7A delivers copper to the lysyl oxidase family of enzymes and promotes tumorigenesis and metastasis. Proc. Natl. Acad. Sci. USA 2019, 116, 6836–6841. [Google Scholar] [CrossRef] [Green Version]
- Vallet, S.D.; Guéroult, M.; Belloy, N.; Dauchez, M.; Ricard-Blum, S. A Three-Dimensional Model of Human Lysyl Oxidase, a Cross-Linking Enzyme. ACS Omega 2019, 4, 8495–8505. [Google Scholar] [CrossRef] [Green Version]
- Meier, A.A.; Kuczera, K.; Mure, M. A 3D–Predicted Structure of the Amine Oxidase Domain of Lysyl Oxidase–Like 2. Int. J. Mol. Sci. 2022, 23, 13385. [Google Scholar] [CrossRef]
- Meier, A.A.; Moon, H.-J.; Sabuncu, S.; Singh, P.; Ronnebaum, T.A.; Ou, S.; Douglas, J.T.; Jackson, T.A.; Moënne-Loccoz, P.; Mure, M. Insight into the Spatial Arrangement of the Lysine Tyrosylquinone and Cu2+ in the Active Site of Lysyl Oxidase-like 2. Int. J. Mol. Sci. 2022, 23, 13966. [Google Scholar] [CrossRef] [PubMed]
- Mure, M. Tyrosine-Derived Quinone Cofactors. Accounts Chem. Res. 2004, 37, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with Alpha Fold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Varadi, M.; Anyango, S.; Deshpande, M.; Nair, S.; Natassia, C.; Yordanova, G.; Yuan, D.; Stroe, O.; Wood, G.; Laydon, A.; et al. AlphaFold Protein Structure Database: Massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. 2022, 50, D439–D444. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Moon, H.-J.; Finney, J.; Meier, A.; Mure, M. Extracellular Processing of Lysyl Oxidase-like 2 and Its Effect on Amine Oxidase Activity. Biochemistry 2018, 57, 6973–6983. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Go, E.P.; Finney, J.; Moon, H.; Lantz, M.; Rebecchi, K.; Desaire, H.; Mure, M. Post-translational Modifications of Recombinant Human Lysyl Oxidase-like 2 (rhLOXL2) Secreted from Drosophila S2 Cells*. J. Biol. Chem. 2013, 288, 5357–5363. [Google Scholar] [CrossRef] [Green Version]
- Sell, D.R.; Monnier, V.M. Structure Elucidation of a Senescence Cross-Link from Human Extracellular Matrix. Implication of pentoses in the aging process. J. Biol. Chem. 1989, 264, 21597–21602. [Google Scholar] [CrossRef]
- Eyre, D.R.; Weis, M.A.; Wu, J.-J. Maturation of Collagen Ketoimine Cross-links by an Alternative Mechanism to Pyridinoline Formation in Cartilage. J. Biol. Chem. 2010, 285, 16675–16682. [Google Scholar] [CrossRef] [Green Version]
- Hanson, D.A.; Eyre, D.R. Molecular Site Specificity of Pyridinoline and Pyrrole Cross-links in Type I Collagen of Human Bone. J. Biol. Chem. 1996, 271, 26508–26516. [Google Scholar] [CrossRef] [Green Version]
- Eyre, D.R.; Weis, M.; Rai, J. Analyses of lysine aldehyde cross-linking in collagen reveal that the mature cross-link histidinohydroxylysinonorleucine is an artifact. J. Biol. Chem. 2019, 294, 6578–6590. [Google Scholar] [CrossRef] [PubMed]
- Bailey, A.J. The chemistry of natural enzyme-induced cross-links of proteins. Amino Acids 1991, 1, 293–306. [Google Scholar] [CrossRef]
- Fraser, D.; Tenner, A. Directing an appropriate immune response: The role of defense collagens and other soluble pattern recognition molecules. Curr. Drug Targets 2008, 9, 113–122. [Google Scholar] [CrossRef]
- Maertens, B.; Hopkins, D.; Franzke, C.-W.; Keene, D.R.; Bruckner-Tuderman, L.; Greenspan, D.S.; Koch, M. Cleavage and Oligomerization of Gliomedin, a Transmembrane Collagen Required for Node of Ranvier Formation. J. Biol. Chem. 2007, 282, 10647–10659. [Google Scholar] [CrossRef] [Green Version]
- Teuscher, A.C.; Statzer, C.; Pantasis, S.; Bordoli, M.R.; Ewald, C.Y. Assessing Collagen Deposition during Aging in Mammalian Tissue and in Caenorhabditis Elegans; Springer: Berlin/Heidelberg, Germany, 2019; pp. 169–188. [Google Scholar] [CrossRef]
- Fenske, N.A.; Lober, C.W. Structural and functional changes of normal aging skin. J. Am. Acad. Dermatol. 1986, 15, 571–585. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Quan, T.; Purohit, T.; Shao, Y.; Cho, M.K.; He, T.; Varani, J.; Kang, S.; Voorhees, J.J. Collagen Fragmentation Promotes Oxidative Stress and Elevates Matrix Metalloproteinase-1 in Fibroblasts in Aged Human Skin. Am. J. Pathol. 2009, 174, 101–114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reiser, K.M. Nonenzymatic glycation of collagen in aging and diabetes. Proc. Soc. Exp. Biol. Med. 1998, 218, 23–37. [Google Scholar] [CrossRef]
- Varani, J.; Dame, M.K.; Rittie, L.; Fligiel, S.E.; Kang, S.; Fisher, G.J.; Voorhees, J.J. Decreased Collagen Production in Chronologically Aged Skin: Roles of Age-Dependent Alteration in Fibroblast Function and Defective Mechanical Stimulation. Am. J. Pathol. 2006, 168, 1861–1868. [Google Scholar] [CrossRef] [Green Version]
- Eyre, D.R.; Weis, M.A.; Wu, J.-J. Advances in collagen cross-link analysis. Methods 2008, 45, 65–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalamajski, S.; Liu, C.; Tillgren, V.; Rubin, K.; Oldberg, Å.; Rai, J.; Weis, M.; Eyre, D.R. Increased C-telopeptide Cross-linking of Tendon Type I Collagen in Fibromodulin-deficient Mice. J. Biol. Chem. 2014, 289, 18873–18879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalamajski, S.; Bihan, D.; Bonna, A.; Rubin, K.; Farndale, R.W. Fibromodulin Interacts with Collagen Cross-linking Sites and Activates Lysyl Oxidase. J. Biol. Chem. 2016, 291, 7951–7960. [Google Scholar] [CrossRef] [Green Version]
- Herum, K.M.; Lunde, I.G.; Skrbic, B.; Louch, W.E.; Hasic, A.; Boye, S.; Unger, A.; Brorson, S.-H.; Sjaastad, I.; Tønnessen, T.; et al. Syndecan-4 is a key determinant of collagen cross-linking and passive myocardial stiffness in the pressure-overloaded heart. Cardiovasc. Res. 2015, 106, 217–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmelzer, C.E.H.; Heinz, A.; Troilo, H.; Lockhart-Cairns, M.P.; Jowitt, T.A.; Marchand, M.F.; Bidault, L.; Bignon, M.; Hedtke, T.; Barret, A.; et al. Lysyl oxidase-like 2 (LOXL2)-mediated cross-linking of tropoelastin. FASEB J. 2019, 33, 5468–5481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomassin, L.; Werneck, C.C.; Broekelmann, T.J.; Gleyzal, C.; Hornstra, I.K.; Mecham, R.P.; Sommer, P. The Pro-regions of Lysyl Oxidase and Lysyl Oxidase-like 1 Are Required for Deposition onto Elastic Fibers. J. Biol. Chem. 2005, 280, 42848–42855. [Google Scholar] [CrossRef] [Green Version]
- Semkova, M.E.; Hsuan, J.J. TGFβ-1 Induced Cross-Linking of the Extracellular Matrix of Primary Human Dermal Fibroblasts. Int. J. Mol. Sci. 2021, 22, 984. [Google Scholar] [CrossRef]
- Vallet, S.D.; Miele, A.E.; Uciechowska-Kaczmarzyk, U.; Liwo, A.; Duclos, B.; Samsonov, S.A.; Ricard-Blum, S. Insights into the structure and dynamics of lysyl oxidase propeptide, a flexible protein with numerous partners. Sci. Rep. 2018, 8, 11768. [Google Scholar] [CrossRef] [Green Version]
- Brady, J.D.; Robins, S.P. Structural Characterization of Pyrrolic Cross-links in Collagen Using a Biotinylated Ehrlich’s Reagent*. J. Biol. Chem. 2001, 276, 18812–18818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuentes-Lemus, E.; Hägglund, P.; López-Alarcón, C.; Davies, M.J. Oxidative Crosslinking of Peptides and Proteins: Mechanisms of Formation, Detection, Characterization and Quantification. Molecules 2021, 27, 15. [Google Scholar] [CrossRef]
- Bucala, R.; Cerami, A. Advanced Glycosylation: Chemistry, Biology, and Implications for Diabetes and Aging. Adv. Pharmacol. 1992, 23, 13. [Google Scholar] [CrossRef]
- Rojas, A.; Morales, M.A. Advanced glycation and endothelial functions: A link towards vascular complications in diabetes. Life Sci. 2004, 76, 715–730. [Google Scholar] [CrossRef]
- Fournet, M.; Bonté, F.; Desmoulière, A. Glycation Damage: A Possible Hub for Major Pathophysiological Disorders and Aging. Aging Dis. 2018, 9, 880–900. [Google Scholar] [CrossRef] [Green Version]
- Brings, S.; Fleming, T.; Freichel, M.; Muckenthaler, M.U.; Herzig, S.; Nawroth, P.P. Dicarbonyls and Advanced Glycation End-Products in the Development of Diabetic Complications and Targets for Intervention. Int. J. Mol. Sci. 2017, 18, 984. [Google Scholar] [CrossRef] [Green Version]
- Meade, S.J.; Miller, A.G.; Gerrard, J. The role of dicarbonyl compounds in non-enzymatic crosslinking: A structure–activity study. Bioorganic Med. Chem. 2003, 11, 853–862. [Google Scholar] [CrossRef] [PubMed]
- Sell, D.R.; Monnier, V.M. Aging of long-lived proteins: Extracellular matrix (collagens, elastins, proteoglycans) and lens crystallins. Compr. Physiol. 2010, 235–305. [Google Scholar] [CrossRef]
- Wautier, J.-L.; Guillausseau, P.-J. Diabetes, advanced glycation endproducts and vascular disease. Vasc. Med. 1998, 3, 131–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nash, A.; Notou, M.; Lopez-Clavijo, A.F.; Bozec, L.; de Leeuw, N.H.; Birch, H.L. Glucosepane is associated with changes to structural and physical properties of collagen fibrils. Matrix Biol. Plus 2019, 4, 100013. [Google Scholar] [CrossRef] [PubMed]
- Bansode, S.; Bashtanova, U.; Li, R.; Clark, J.; Müller, K.H.; Puszkarska, A.; Goldberga, I.; Chetwood, H.H.; Reid, D.G.; Colwell, L.J.; et al. Glycation changes molecular organization and charge distribution in type I collagen fibrils. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fessel, G.; Li, Y.; Diederich, V.; Guizar-Sicairos, M.; Schneider, P.; Sell, D.R.; Monnier, V.M.; Snedeker, J.G. Advanced Glycation End-Products Reduce Collagen Molecular Sliding to Affect Collagen Fibril Damage Mechanisms but Not Stiffness. PLoS ONE 2014, 9, e110948. [Google Scholar] [CrossRef]
- Rojas, A.; Anazco, C.; González, I.; Araya, P. Extracellular matrix glycation and receptor for advanced glycation end-products activation: A missing piece in the puzzle of the association between diabetes and cancer. Carcinogenesis 2018, 39, 515–521. [Google Scholar] [CrossRef]
- Sell, D.R.; Biemel, K.M.; Reihl, O.; Lederer, M.O.; Strauch, C.M.; Monnier, V.M. Glucosepane is a major protein cross-link of the senescent human extracellular matrix: Relationship with diabetes. J. Biol. Chem. 2005, 280, 12310–12315. [Google Scholar] [CrossRef] [Green Version]
- Vaez, M.; Asgari, M.; Hirvonen, L.; Bakir, G.; Khattignavong, E.; Ezzo, M.; Aguayo, S.; Schuh, C.M.; Gough, K.; Bozec, L. Modulation of the biophysical and biochemical properties of collagen by glycation for tissue engineering applications. Acta Biomater. 2023, 155, 182–198. [Google Scholar] [CrossRef]
- Monnier, V.M.; Sell, D.R.; Strauch, C.; Sun, W.; Lachin, J.M.; Cleary, P.A.; Genuth, S. The association between skin collagen glucosepane and past progression of microvascular and neuropathic complications in type 1 diabetes. J. Diabetes its Complicat. 2012, 27, 141–149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lucarini, M.; Sciubba, F.; Capitani, D.; Di Cocco, M.E.; D’evoli, L.; Durazzo, A.; Delfini, M.; Boccia, G.L. Role of catechin on collagen type I stability upon oxidation: A NMR approach. Nat. Prod. Res. 2019, 34, 53–62. [Google Scholar] [CrossRef]
- Porto, I.C.C.M.; Nascimento, T.G.; Oliveira, J.M.S.; Freitas, P.H.; Haimeur, A.; França, R. Use of polyphenols as a strategy to prevent bond degradation in the dentin-resin interface. Eur. J. Oral Sci. 2018, 126, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Nazaruk, J.; Galicka, A. The influence of selected flavonoids from the leaves of Cirsium palustre (L.) Scop. on collagen expression in human skin fibroblasts. Phytother. Res. 2014, 28, 1399–1405. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, J.; Cheng, X.; Yi, B.; Zhang, X.; Li, Q. Apigenin induces dermal collagen synthesis via smad2/3 signaling pathway. Eur. J. Histochem. 2015, 59, 2467. [Google Scholar] [CrossRef]
- Bae, J.-Y.; Lim, S.S.; Kim, S.J.; Choi, J.-S.; Park, J.; Ju, S.M.; Han, S.J.; Kang, I.-J.; Kang, Y.-H. Bog blueberry anthocyanins alleviate photoaging in ultraviolet-B irradiation-induced human dermal fibroblasts. Mol. Nutr. Food Res. 2009, 53, 726–738. [Google Scholar] [CrossRef]
- Wittenauer, J.; Mäckle, S.; Sußmann, D.; Schweiggert-Weisz, U.; Carle, R. Inhibitory effects of polyphenols from grape pomace extract on collagenase and elastase activity. Fitoterapia 2015, 101, 179–187. [Google Scholar] [CrossRef]
- Furuhashi, M.; Hatasa, Y.; Kawamura, S.; Shibata, T.; Akagawa, M.; Uchida, K. Identification of Polyphenol-Specific Innate Epitopes That Originated from a Resveratrol Analogue. Biochemistry 2017, 56, 4701–4712. [Google Scholar] [CrossRef]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef]
- Stirban, A.; Gawlowski, T.; Roden, M. Vascular effects of advanced glycation endproducts: Clinical effects and molecular mechanisms. Mol. Metab. 2013, 3, 94–108. [Google Scholar] [CrossRef] [PubMed]
- Twarda-Clapa, A.; Olczak, A.; Białkowska, A.M.; Koziołkiewicz, M. Advanced Glycation End-Products (AGEs): Formation, Chemistry, Classification, Receptors, and Diseases Related to AGEs. Cells 2022, 11, 1312. [Google Scholar] [CrossRef]
- Bierhaus, A.; Humpert, P.M.; Morcos, M.; Wendt, T.; Chavakis, T.; Arnold, B.; Stern, D.M.; Nawroth, P.P. Understanding RAGE, the receptor for advanced glycation end products. J. Mol. Med. 2005, 83, 876–886. [Google Scholar] [CrossRef]
- Verzijl, N.; DeGroot, J.; Ben Zaken, C.; Braun-Benjamin, O.; Maroudas, A.; Bank, R.A.; Mizrahi, J.; Schalkwijk, C.G.; Thorpe, S.R.; Baynes, J.W.; et al. Crosslinking by advanced glycation end products increases the stiffness of the collagen network in human articular cartilage: A possible mechanism through which age is a risk factor for osteoarthritis. Arthritis Rheum. 2002, 46, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, M.N.I.; Mitsuhashi, S.; Sigetomi, K.; Ubukata, M. Quercetin inhibits advanced glycation end product formation via chelating metal ions, trapping methylglyoxal, and trapping reactive oxygen species. Biosci. Biotechnol. Biochem. 2017, 81, 882–890. [Google Scholar] [CrossRef] [Green Version]
- Hwang, S.H.; Kim, H.Y.; Zuo, G.; Wang, Z.; Lee, J.-Y.; Lim, S.S. Anti-glycation, Carbonyl Trapping and Anti-inflammatory Activities of Chrysin Derivatives. Molecules 2018, 23, 1752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lv, L.; Shao, X.; Chen, H.; Ho, C.-T.; Sang, S. Genistein Inhibits Advanced Glycation End Product Formation by Trapping Methylglyoxal. Chem. Res. Toxicol. 2011, 24, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Sang, S.; Shao, X.; Bai, N.; Lo, C.-Y.; Yang, C.S.; Ho, C.-T. Tea Polyphenol (−)-Epigallocatechin-3-Gallate: A New Trapping Agent of Reactive Dicarbonyl Species. Chem. Res. Toxicol. 2007, 20, 1862–1870. [Google Scholar] [CrossRef]
- Intagliata, S.; Spadaro, A.; Lorenti, M.; Panico, A.; Siciliano, E.A.; Barbagallo, S.; Macaluso, B.; Kamble, S.H.; Modica, M.N.; Montenegro, L. In Vitro Antioxidant and Anti-Glycation Activity of Resveratrol and Its Novel Triester with Trolox. Antioxidants 2020, 10, 12. [Google Scholar] [CrossRef]
- Lee, S.M.; Zheng, L.W.; Jung, Y.; Hwang, G.-S.; Kim, Y.-S. Effects of hydroxycinnamic acids on the reduction of furan and α-dicarbonyl compounds. Food Chem. 2019, 312, 126085. [Google Scholar] [CrossRef]
- Li, J.; Zhang, H.; Yang, X.; Zhu, L.; Wu, G.; Qi, X.; Zhang, H. Trapping of reactive carbonyl species by fiber-bound polyphenols from whole grains under simulated physiological conditions. Food Res. Int. 2022, 156, 111142. [Google Scholar] [CrossRef] [PubMed]
- Mienaltowski, M.J.; Gonzales, N.L.; Beall, J.M.; Pechanec, M.Y. Basic Structure, Physiology, and Biochemistry of Connective Tissues and Extracellular Matrix Collagens. Adv. Exp. Med. Biol. 2021, 1348, 5–43. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Shao, H.; Fang, Z.; Zhao, Y.; Cao, C.Y.; Li, Q. Mechanism and Effects of Polyphenol Derivatives for Modifying Collagen. ACS Biomater. Sci. Eng. 2019, 5, 4272–4284. [Google Scholar] [CrossRef] [PubMed]

| Immature Cross-Links | Mature Cross-Links | Glycation-Derived Cross-Links |
|---|---|---|
Aldimine cross-links
| Pyrrole-containing cross-links
| Alpha-dicarbonyl compounds (α-DC)
|
Ketoamine cross-links
| Histidine-containing cross-links
| Fluorescent cross-linking AGEs
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Añazco, C.; Riedelsberger, J.; Vega-Montoto, L.; Rojas, A. Exploring the Interplay between Polyphenols and Lysyl Oxidase Enzymes for Maintaining Extracellular Matrix Homeostasis. Int. J. Mol. Sci. 2023, 24, 10985. https://doi.org/10.3390/ijms241310985
Añazco C, Riedelsberger J, Vega-Montoto L, Rojas A. Exploring the Interplay between Polyphenols and Lysyl Oxidase Enzymes for Maintaining Extracellular Matrix Homeostasis. International Journal of Molecular Sciences. 2023; 24(13):10985. https://doi.org/10.3390/ijms241310985
Chicago/Turabian StyleAñazco, Carolina, Janin Riedelsberger, Lorenzo Vega-Montoto, and Armando Rojas. 2023. "Exploring the Interplay between Polyphenols and Lysyl Oxidase Enzymes for Maintaining Extracellular Matrix Homeostasis" International Journal of Molecular Sciences 24, no. 13: 10985. https://doi.org/10.3390/ijms241310985
APA StyleAñazco, C., Riedelsberger, J., Vega-Montoto, L., & Rojas, A. (2023). Exploring the Interplay between Polyphenols and Lysyl Oxidase Enzymes for Maintaining Extracellular Matrix Homeostasis. International Journal of Molecular Sciences, 24(13), 10985. https://doi.org/10.3390/ijms241310985





