Epigenetic Modulation of Inflammatory Pathways in Myometrial Stem Cells and Risk of Uterine Fibroids
Abstract
1. Introduction
2. Results
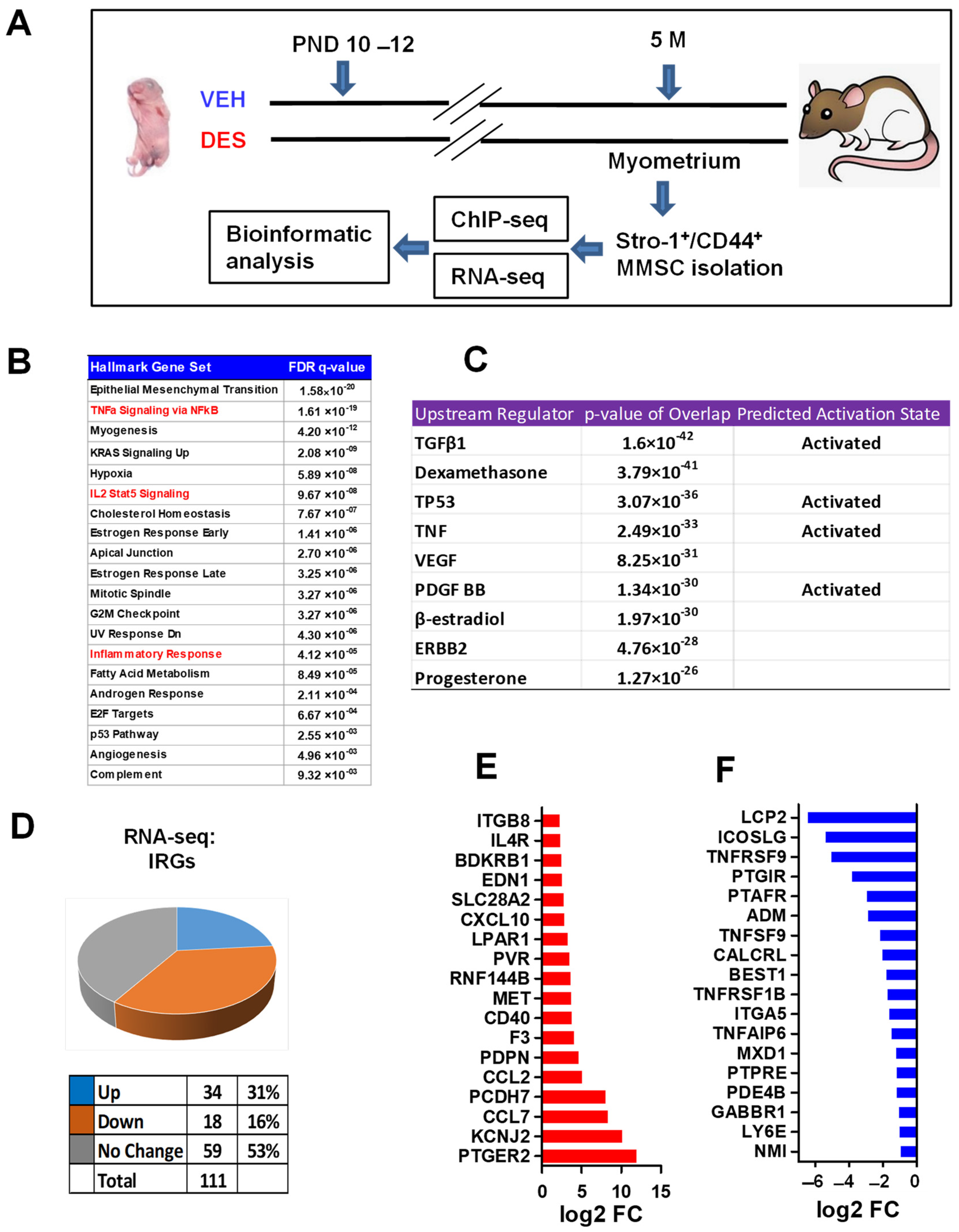
2.1. Identification of Distinct Inflammatory Responsive Genes in DES- vs. VEH-Exposed MMSCs of Eker Rats
2.2. DES Exposure Reprograms the Inflammatory Responsive Genes in MMSCs
2.3. Reprogrammed MMSCs Activates Inflammatory Pathway in Myometrium
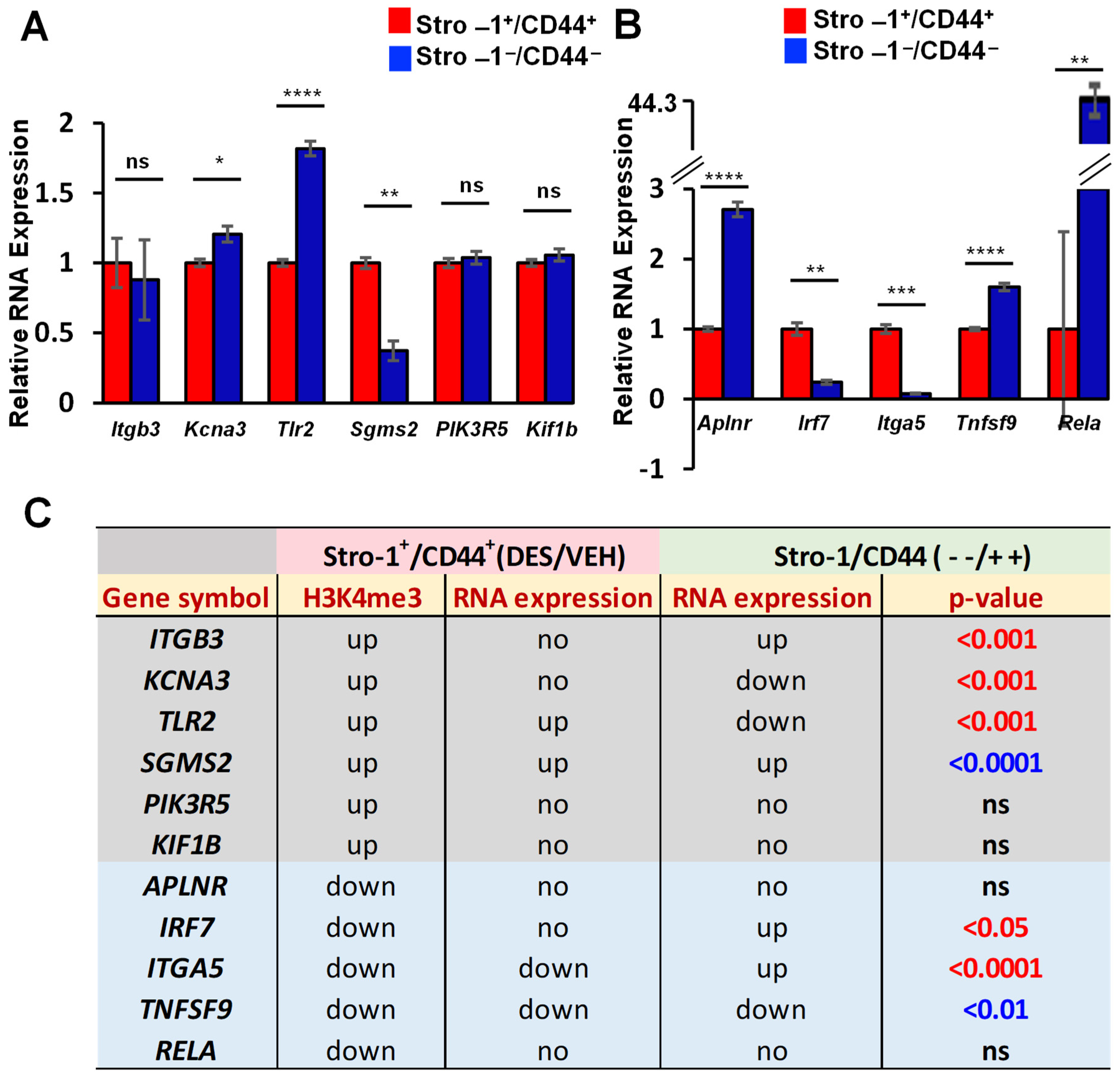
2.4. EDC Exposure Reprograms IRGs via H3K4me3 Mechanism
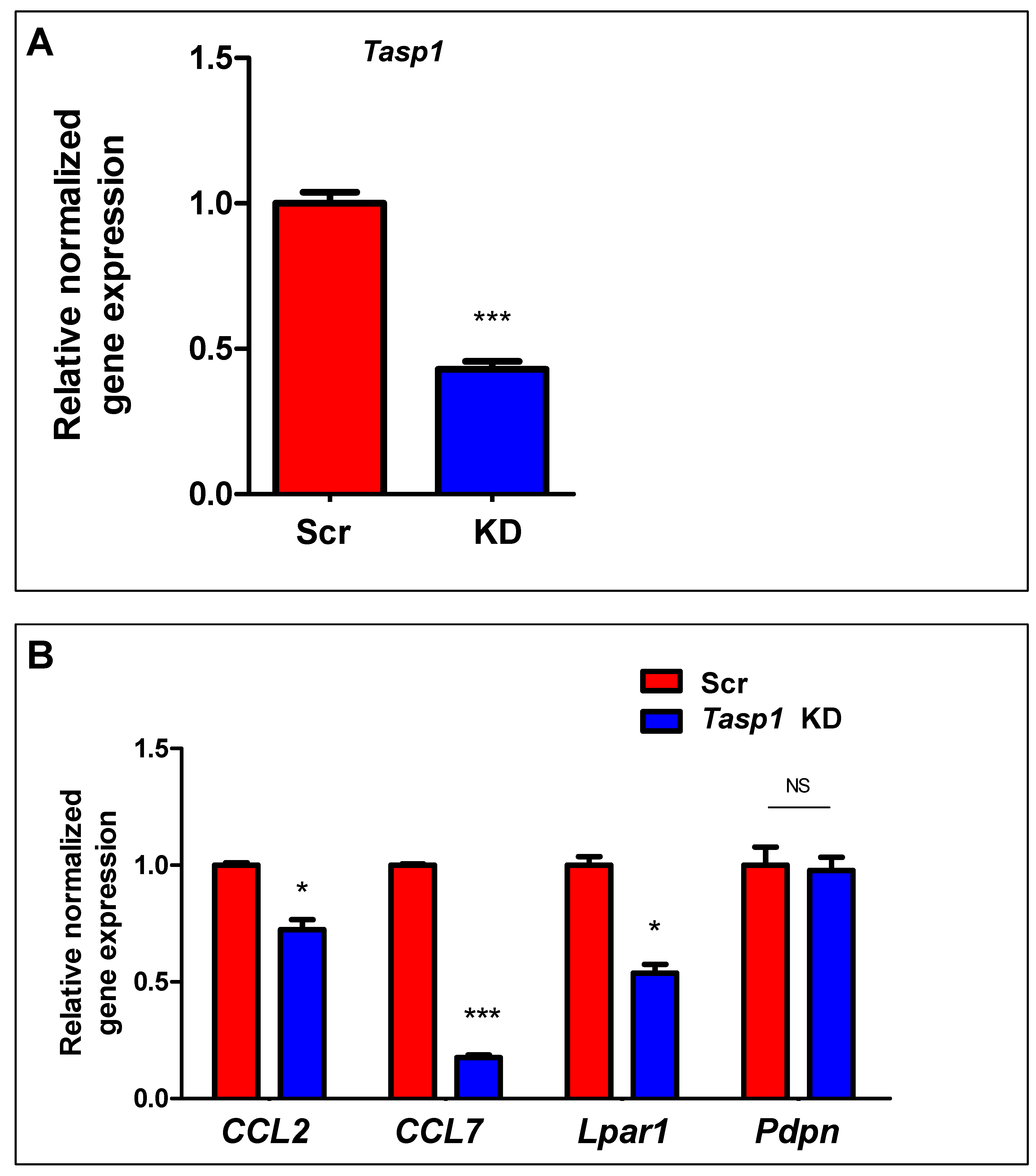
2.5. Inhibition of HDACs Reversed the DES-Induced Aberrant Expression of IRGs in MMSCs
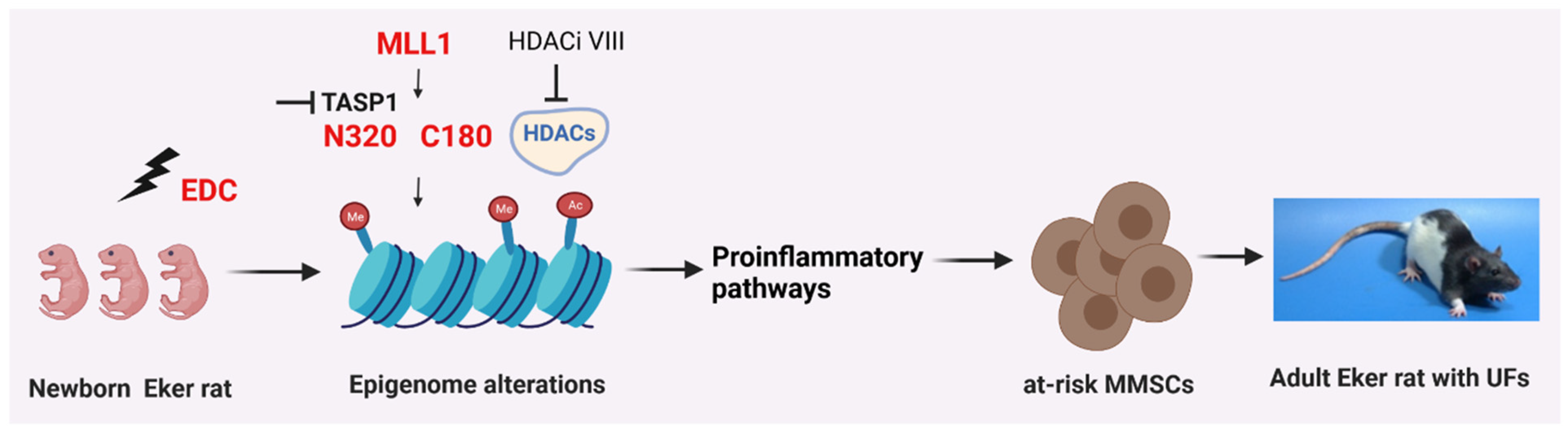
3. Discussion
4. Materials and Methods
4.1. Postnatal Exposure to the Endocrine Disruptor DES
4.2. Isolation and Characterization of Myometrial Stro-1+/CD44+ Cells from Eker Rats Exposed to DES and VEH
4.3. Whole-Transcriptome RNA Sequencing (RNA-seq) and Quantitative Real-Time RT-PCR (RT-qPCR)
4.4. Chromatin Immunoprecipitation Sequencing (ChIP-seq)
4.5. Knockdown of Tasp1 in MMSCs from Eker Rats
4.6. HDAC Inhibition in MMSCs from Eker Rats
4.7. The Effect of MMSC Secretome on the Expression of Proinflammatory Factors in DMCs
4.8. Statistical Analysis
4.9. Bioinformatics Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DEG | differentially expressed gene |
| UF | uterine fibroids |
| EDC | endocrine-disrupting chemicals |
| DES | diethylstilbestrol |
| MMSC | myometrial stem cell |
| IRG | Inflammatory responsive genes |
| MLL1 | mixed-lineage leukemia protein 1 |
References
- Yang, Q.; Ciebiera, M.; Bariani, M.V.; Ali, M.; Elkafas, H.; Boyer, T.G.; Al-Hendy, A. Comprehensive Review of Uterine Fibroids: Developmental Origin, Pathogenesis, and Treatment. Endocr. Rev. 2022, 43, 678–719. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E. Uterine fibroids. N. Engl. J. Med. 2013, 369, 1344–1355. [Google Scholar] [CrossRef] [PubMed]
- Stewart, E.A.; Laughlin-Tommaso, S.K.; Catherino, W.H.; Lalitkumar, S.; Gupta, D.; Vollenhoven, B. Uterine fibroids. Nat. Rev. Dis. Primers 2016, 2, 16043. [Google Scholar] [CrossRef] [PubMed]
- Segars, J.H.; Parrott, E.C.; Nagel, J.D.; Guo, X.C.; Gao, X.; Birnbaum, L.S.; Pinn, V.W.; Dixon, D. Proceedings from the Third National Institutes of Health International Congress on Advances in Uterine Leiomyoma Research: Comprehensive review, conference summary and future recommendations. Hum. Reprod. Update 2014, 20, 309–333. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Diamond, M.P.; Al-Hendy, A. Early Life Adverse Environmental Exposures Increase the Risk of Uterine Fibroid Development: Role of Epigenetic Regulation. Front. Pharmacol. 2016, 7, 40. [Google Scholar] [CrossRef] [PubMed]
- Mas, A.; Stone, L.; O’Connor, P.M.; Yang, Q.; Kleven, D.; Simon, C.; Walker, C.L.; Al-Hendy, A. Developmental Exposure to Endocrine Disruptors Expands Murine Myometrial Stem Cell Compartment as a Prerequisite to Leiomyoma Tumorigenesis. Stem Cells 2017, 35, 666–678. [Google Scholar] [CrossRef] [PubMed]
- Bredfeldt, T.G.; Greathouse, K.L.; Safe, S.H.; Hung, M.C.; Bedford, M.T.; Walker, C.L. Xenoestrogen-induced regulation of EZH2 and histone methylation via estrogen receptor signaling to PI3K/AKT. Mol. Endocrinol. 2010, 24, 993–1006. [Google Scholar] [CrossRef] [PubMed]
- Greathouse, K.L.; Bredfeldt, T.; Everitt, J.I.; Lin, K.; Berry, T.; Kannan, K.; Mittelstadt, M.L.; Ho, S.M.; Walker, C.L. Environmental estrogens differentially engage the histone methyltransferase EZH2 to increase risk of uterine tumorigenesis. Mol. Cancer Res. 2012, 10, 546–557. [Google Scholar] [CrossRef] [PubMed]
- Baird, D.D.; Newbold, R. Prenatal diethylstilbestrol (DES) exposure is associated with uterine leiomyoma development. Reprod. Toxicol. 2005, 20, 81–84. [Google Scholar] [CrossRef] [PubMed]
- Bariani, M.V.; Rangaswamy, R.; Siblini, H.; Yang, Q.; Al-Hendy, A.; Zota, A.R. The role of endocrine-disrupting chemicals in uterine fibroid pathogenesis. Curr. Opin. Endocrinol. Diabetes Obes. 2020, 27, 380–387. [Google Scholar] [CrossRef] [PubMed]
- D’Aloisio, A.A.; Baird, D.D.; DeRoo, L.A.; Sandler, D.P. Association of intrauterine and early-life exposures with diagnosis of uterine leiomyomata by 35 years of age in the Sister Study. Environ. Health Perspect. 2010, 118, 375–381. [Google Scholar] [CrossRef] [PubMed]
- D’Aloisio, A.A.; Baird, D.D.; DeRoo, L.A.; Sandler, D.P. Early-life exposures and early-onset uterine leiomyomata in black women in the Sister Study. Environ. Health Perspect. 2012, 120, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Maruyama, T.; Masuda, H.; Kajitani, T.; Nagashima, T.; Arase, T.; Ito, M.; Ohta, K.; Uchida, H.; Asada, H.; et al. Side population in human uterine myometrium displays phenotypic and functional characteristics of myometrial stem cells. Proc. Natl. Acad. Sci. USA 2007, 104, 18700–18705. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.L.; Senaratne, T.N.; Zhang, L.; Szotek, P.P.; Stewart, E.; Dombkowski, D.; Preffer, F.; Donahoe, P.K.; Teixeira, J. Uterine leiomyomas exhibit fewer stem/progenitor cell characteristics when compared with corresponding normal myometrium. Reprod. Sci. 2010, 17, 158–167. [Google Scholar] [CrossRef]
- Mas, A.; Cervello, I.; Gil-Sanchis, C.; Faus, A.; Ferro, J.; Pellicer, A.; Simon, C. Identification and characterization of the human leiomyoma side population as putative tumor-initiating cells. Fertil. Steril. 2012, 98, 741–751.e6. [Google Scholar] [CrossRef]
- Ono, M.; Qiang, W.; Serna, V.A.; Yin, P.; Coon, J.S.T.; Navarro, A.; Monsivais, D.; Kakinuma, T.; Dyson, M.; Druschitz, S.; et al. Role of stem cells in human uterine leiomyoma growth. PLoS ONE 2012, 7, e36935. [Google Scholar] [CrossRef]
- Elkafas, H.; Ali, M.; Elmorsy, E.; Kamel, R.; Thompson, W.E.; Badary, O.; Al-Hendy, A.; Yang, Q. Vitamin D3 Ameliorates DNA Damage Caused by Developmental Exposure to Endocrine Disruptors in the Uterine Myometrial Stem Cells of Eker Rats. Cells 2020, 9, 1459. [Google Scholar] [CrossRef]
- Prusinski Fernung, L.E.; Yang, Q.; Sakamuro, D.; Kumari, A.; Mas, A.; Al-Hendy, A. Endocrine disruptor exposure during development increases incidence of uterine fibroids by altering DNA repair in myometrial stem cells. Biol. Reprod. 2018, 99, 735–748. [Google Scholar] [CrossRef]
- Ley, D.; Desseyn, J.L.; Mischke, M.; Knol, J.; Turck, D.; Gottrand, F. Early-life origin of intestinal inflammatory disorders. Nutr. Rev. 2017, 75, 175–187. [Google Scholar] [CrossRef]
- Cook, J.D.; Davis, B.J.; Cai, S.L.; Barrett, J.C.; Conti, C.J.; Walker, C.L. Interaction between genetic susceptibility and early-life environmental exposure determines tumor-suppressor-gene penetrance. Proc. Natl. Acad. Sci. USA 2005, 102, 8644–8649. [Google Scholar] [CrossRef]
- Wang, Q.; Trevino, L.S.; Wong, R.L.; Medvedovic, M.; Chen, J.; Ho, S.M.; Shen, J.; Foulds, C.E.; Coarfa, C.; O’Malley, B.W.; et al. Reprogramming of the Epigenome by MLL1 Links Early-Life Environmental Exposures to Prostate Cancer Risk. Mol. Endocrinol. 2016, 30, 856–871. [Google Scholar] [CrossRef]
- Wang, Z.; Song, J.; Milne, T.A.; Wang, G.G.; Li, H.; Allis, C.D.; Patel, D.J. Pro isomerization in MLL1 PHD3-bromo cassette connects H3K4me readout to CyP33 and HDAC-mediated repression. Cell 2010, 141, 1183–1194. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Shahin, S.M.; Sabri, N.A.; Al-Hendy, A.; Yang, Q. Activation of Beta-Catenin Signaling and Its Crosstalk with Estrogen and Histone Deacetylases in Human Uterine Fibroids. J. Clin. Endocrinol. Metab. 2020, 105, e1517–e1535. [Google Scholar] [CrossRef] [PubMed]
- Hayward, A.D.; Rigby, F.L.; Lummaa, V. Early-life disease exposure and associations with adult survival, cause of death, and reproductive success in preindustrial humans. Proc. Natl. Acad. Sci. USA 2016, 113, 8951–8956. [Google Scholar] [CrossRef] [PubMed]
- Trevino, L.S.; Dong, J.; Kaushal, A.; Katz, T.A.; Jangid, R.K.; Robertson, M.J.; Grimm, S.L.; Ambati, C.S.R.; Putluri, V.; Cox, A.R.; et al. Epigenome environment interactions accelerate epigenomic aging and unlock metabolically restricted epigenetic reprogramming in adulthood. Nat. Commun. 2020, 11, 2316. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.L.; Ho, S.M. Developmental reprogramming of cancer susceptibility. Nat. Rev. Cancer 2012, 12, 479–486. [Google Scholar] [CrossRef]
- Katz, T.A.; Yang, Q.; Trevino, L.S.; Walker, C.L.; Al-Hendy, A. Endocrine-disrupting chemicals and uterine fibroids. Fertil. Steril. 2016, 106, 967–977. [Google Scholar] [CrossRef]
- El Andaloussi, A.; Chaudhry, Z.; Al-Hendy, A.; Ismail, N. Uterine Fibroids: Bridging Genomic Defects and Chronic Inflammation. Semin. Reprod. Med. 2017, 35, 494–498. [Google Scholar] [CrossRef]
- Islam, M.S.; Akhtar, M.M.; Ciavattini, A.; Giannubilo, S.R.; Protic, O.; Janjusevic, M.; Procopio, A.D.; Segars, J.H.; Castellucci, M.; Ciarmela, P. Use of dietary phytochemicals to target inflammation, fibrosis, proliferation, and angiogenesis in uterine tissues: Promising options for prevention and treatment of uterine fibroids? Mol. Nutr. Food Res. 2014, 58, 1667–1684. [Google Scholar] [CrossRef]
- Ciebiera, M.; Wlodarczyk, M.; Wrzosek, M.; Wojtyla, C.; Blazej, M.; Nowicka, G.; Lukaszuk, K.; Jakiel, G. TNF-alpha serum levels are elevated in women with clinically symptomatic uterine fibroids. Int. J. Immunopathol. Pharmacol. 2018, 32, 2058738418779461. [Google Scholar] [CrossRef]
- Zannotti, A.; Greco, S.; Pellegrino, P.; Giantomassi, F.; Delli Carpini, G.; Goteri, G.; Ciavattini, A.; Ciarmela, P. Macrophages and Immune Responses in Uterine Fibroids. Cells 2021, 10, 982. [Google Scholar] [CrossRef]
- Faraji, A.; Shamsadinimoghadam, R.; Jahromi, M.A.; Namazi, N. TGF-beta1 role in uterine leiomyoma and endometrial polyp: An insight to drug-based treatment instead of surgical techniques. Obstet. Gynecol. Sci. 2021, 64, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.S.; Nowak, R.A. Human leiomyoma smooth muscle cells show increased expression of transforming growth factor-beta 3 (TGF beta 3) and altered responses to the antiproliferative effects of TGF beta. J. Clin. Endocrinol. Metab. 2001, 86, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Orciani, M.; Caffarini, M.; Biagini, A.; Lucarini, G.; Delli Carpini, G.; Berretta, A.; Di Primio, R.; Ciavattini, A. Chronic Inflammation May Enhance Leiomyoma Development by the Involvement of Progenitor Cells. Stem Cells Int. 2018, 2018, 1716246. [Google Scholar] [CrossRef] [PubMed]
- Kidane, D.; Chae, W.J.; Czochor, J.; Eckert, K.A.; Glazer, P.M.; Bothwell, A.L.; Sweasy, J.B. Interplay between DNA repair and inflammation, and the link to cancer. Crit. Rev. Biochem. Mol. Biol. 2014, 49, 116–139. [Google Scholar] [CrossRef]
- Kay, J.; Thadhani, E.; Samson, L.; Engelward, B. Inflammation-induced DNA damage, mutations and cancer. DNA Repair 2019, 83, 102673. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, K.; Li, S.; Liao, Y.; Du, R.; Zhang, X.; Shu, H.B.; Guo, A.Y.; Li, L.; Wu, M. MLL1, a H3K4 methyltransferase, regulates the TNFalpha-stimulated activation of genes downstream of NF-kappaB. J. Cell Sci. 2012, 125, 4058–4066. [Google Scholar] [CrossRef]
- Bariani, M.V.; Cui, Y.H.; Ali, M.; Bai, T.; Grimm, S.L.; Coarfa, C.; Walker, C.L.; He, Y.Y.; Yang, Q.; Al-Hendy, A. TGFbeta signaling links early-life endocrine-disrupting chemicals exposure to suppression of nucleotide excision repair in rat myometrial stem cells. Res. Sq. 2023. [Google Scholar] [CrossRef]
- Czarny, P.; Wigner, P.; Galecki, P.; Sliwinski, T. The interplay between inflammation, oxidative stress, DNA damage, DNA repair and mitochondrial dysfunction in depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 80, 309–321. [Google Scholar] [CrossRef]
- Tudek, B.; Swoboda, M.; Kowalczyk, P.; Olinski, R. Modulation of oxidative DNA damage repair by the diet, inflammation and neoplastic transformation. J. Physiol. Pharmacol. 2006, 57 (Suppl. 7), 33–49. [Google Scholar]
- Budworth, H.; Snijders, A.M.; Marchetti, F.; Mannion, B.; Bhatnagar, S.; Kwoh, E.; Tan, Y.; Wang, S.X.; Blakely, W.F.; Coleman, M.; et al. DNA repair and cell cycle biomarkers of radiation exposure and inflammation stress in human blood. PLoS ONE 2012, 7, e48619. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.A.; Witherspoon, M.; Wang, K.; Afrasiabi, K.; Pham, T.; Birnbaumer, L.; Lipkin, S.M. Epigenetic repression of DNA mismatch repair by inflammation and hypoxia in inflammatory bowel disease-associated colorectal cancer. Cancer Res. 2009, 69, 6423–6429. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.T.; Chen, C.; Chen, J.W. CCL7 as a novel inflammatory mediator in cardiovascular disease, diabetes mellitus, and kidney disease. Cardiovasc. Diabetol. 2022, 21, 185. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, H.; Kouadir, M.; Song, H.; Shi, F. Recent advances in the mechanisms of NLRP3 inflammasome activation and its inhibitors. Cell Death Dis. 2019, 10, 128. [Google Scholar] [CrossRef] [PubMed]
- Irandoost, E.; Najibi, S.; Talebbeigi, S.; Nassiri, S. Focus on the role of NLRP3 inflammasome in the pathology of endometriosis: A review on molecular mechanisms and possible medical applications. Naunyn Schmiedebergs Arch. Pharmacol. 2023, 396, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Arif Ansari, M.; Choudhary, K.; Singh, S. NLRP3 inflammasome in depression: A review. Int. Immunopharmacol. 2023, 117, 109916. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Zhao, S.; Zhou, J.; Yan, J.; Wang, L.; Du, X.; Li, H.; Chen, Y.; Cai, W.; Wu, J. Curcumin alleviates DSS-induced colitis via inhibiting NLRP3 inflammsome activation and IL-1beta production. Mol. Immunol. 2018, 104, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.L. Epigenomic reprogramming of the developing reproductive tract and disease susceptibility in adulthood. Birth Defects Res. A Clin. Mol. Teratol. 2011, 91, 666–671. [Google Scholar] [CrossRef]
- Lassiter, T.L.; Ryde, I.T.; Mackillop, E.A.; Brown, K.K.; Levin, E.D.; Seidler, F.J.; Slotkin, T.A. Exposure of neonatal rats to parathion elicits sex-selective reprogramming of metabolism and alters the response to a high-fat diet in adulthood. Environ. Health Perspect. 2008, 116, 1456–1462. [Google Scholar] [CrossRef]
- de Picoli Souza, K.; Silva, F.G.; Nunes, M.T. Effect of neonatal hyperthyroidism on GH gene expression reprogramming and physiological repercussions in rat adulthood. J. Endocrinol. 2006, 190, 407–414. [Google Scholar] [CrossRef]
- Mas, A.; Prusinski, L.; Yang, Q.; Diaz-Gimeno, P.; Stone, L.; Diamond, M.P.; Simon, C.; Al-Hendy, A. Role of Stro1+/CD44+ stem cells in myometrial physiology and uterine remodeling during pregnancy. Biol. Reprod. 2017, 96, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, T.; Meyer, C.A.; Eeckhoute, J.; Johnson, D.S.; Bernstein, B.E.; Nusbaum, C.; Myers, R.M.; Brown, M.; Li, W.; et al. Model-based analysis of ChIP-Seq (MACS). Genome Biol. 2008, 9, R137. [Google Scholar] [CrossRef]
- Yang, Q.W.; Liu, S.; Tian, Y.; Salwen, H.R.; Chlenski, A.; Weinstein, J.; Cohn, S.L. Methylation-associated silencing of the thrombospondin-1 gene in human neuroblastoma. Cancer Res. 2003, 63, 6299–6310. [Google Scholar] [PubMed]
- Yang, Q.; Laknaur, A.; Elam, L.; Ismail, N.; Gavrilova-Jordan, L.; Lue, J.; Diamond, M.P.; Al-Hendy, A. Identification of Polycomb Group Protein EZH2-Mediated DNA Mismatch Repair Gene MSH2 in Human Uterine Fibroids. Reprod. Sci. 2016, 23, 1314–1325. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Q.; Ali, M.; Treviño, L.S.; Mas, A.; Ismail, N.; Al-Hendy, A. Epigenetic Modulation of Inflammatory Pathways in Myometrial Stem Cells and Risk of Uterine Fibroids. Int. J. Mol. Sci. 2023, 24, 11641. https://doi.org/10.3390/ijms241411641
Yang Q, Ali M, Treviño LS, Mas A, Ismail N, Al-Hendy A. Epigenetic Modulation of Inflammatory Pathways in Myometrial Stem Cells and Risk of Uterine Fibroids. International Journal of Molecular Sciences. 2023; 24(14):11641. https://doi.org/10.3390/ijms241411641
Chicago/Turabian StyleYang, Qiwei, Mohamed Ali, Lindsey S. Treviño, Aymara Mas, Nahed Ismail, and Ayman Al-Hendy. 2023. "Epigenetic Modulation of Inflammatory Pathways in Myometrial Stem Cells and Risk of Uterine Fibroids" International Journal of Molecular Sciences 24, no. 14: 11641. https://doi.org/10.3390/ijms241411641
APA StyleYang, Q., Ali, M., Treviño, L. S., Mas, A., Ismail, N., & Al-Hendy, A. (2023). Epigenetic Modulation of Inflammatory Pathways in Myometrial Stem Cells and Risk of Uterine Fibroids. International Journal of Molecular Sciences, 24(14), 11641. https://doi.org/10.3390/ijms241411641









