Gene–Smoking Interaction Analysis for the Identification of Novel Asthma-Associated Genetic Factors
Abstract
1. Introduction
2. Results
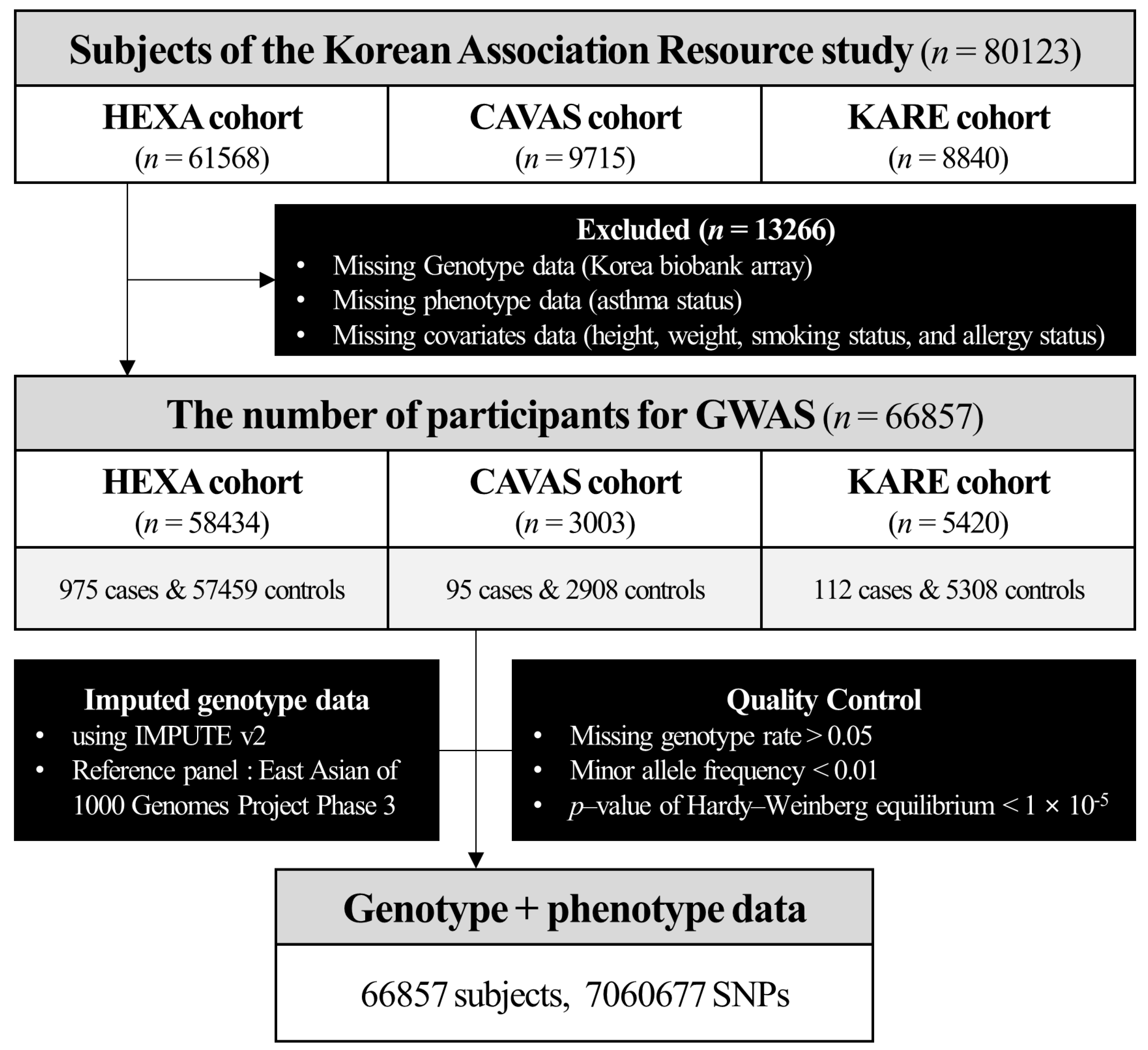
2.1. General Characteristics of Study Participants
2.2. GWAS Results
2.3. Gene Analysis and Gene-Set Analysis
2.4. Functional Annotation
3. Discussion
4. Materials and Methods
4.1. Study Participants
4.2. Quality Control
4.3. Genome-Wide Association Analysis
4.4. Gene Analysis and Gene-Set Analysis
4.5. Functional Annotation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Asher, I.; Pearce, N. Global burden of asthma among children. Int. J. Tuberc. Lung Dis. 2014, 18, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- Mizgerd, J.P. Lung infection--a public health priority. PLoS Med. 2006, 3, e76. [Google Scholar] [CrossRef] [PubMed]
- GBD 2015 Chronic Respiratory Disease Collaborators. Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990-2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Respir. Med. 2017, 5, 691–706. [Google Scholar] [CrossRef]
- Enilari, O.; Sinha, S. The Global Impact of Asthma in Adult Populations. Ann. Glob. Health 2019, 85, 2. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Ryu, J.; Nam, E.; Chung, S.J.; Yeo, Y.; Park, D.W.; Park, T.S.; Moon, J.-Y.; Kim, T.-H.; Sohn, J.W.; et al. Increased mortality in patients with corticosteroid-dependent asthma: A nationwide population-based study. Eur. Respir. J. 2019, 54, 1900804. [Google Scholar] [CrossRef]
- Seth, D.; Saini, S.; Poowuttikul, P. Pediatric Inner-City Asthma. Pediatr. Clin. N. Am. 2019, 66, 967–979. [Google Scholar] [CrossRef]
- Simpson, C.B.; Amin, M.R. Chronic cough: State-of-the-art review. Otolaryngol. Head Neck Surg. 2006, 134, 693–700. [Google Scholar] [CrossRef]
- Rank, M.A.; Shah, N.D. Multiple Chronic Conditions and Asthma: Implications for Practice and Research. J. Allergy Clin. Immunol. Pr. 2014, 2, 518–524. [Google Scholar] [CrossRef]
- Boulet, L.-P. Influence of comorbid conditions on asthma. Eur. Respir. J. 2009, 33, 897–906. [Google Scholar] [CrossRef]
- Kauppi, P.; Linna, M.; Jantunen, J.; Martikainen, J.E.; Haahtela, T.; Pelkonen, A.; Mäkelä, M. Chronic Comorbidities Contribute to the Burden and Costs of Persistent Asthma. Mediat. Inflamm. 2015, 2015, 819194. [Google Scholar] [CrossRef]
- Barnes, P.J. Immunology of asthma and chronic obstructive pulmonary disease. Nat. Rev. Immunol. 2008, 8, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Ober, C.; Vercelli, D. Gene-environment interactions in human disease: Nuisance or opportunity? Trends Genet. 2011, 27, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Ober, C.; Yao, T.-C. The genetics of asthma and allergic disease: A 21st century perspective. Immunol. Rev. 2011, 242, 10–30. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, S.F.; Van Der Sluis, S.; Kyvik, K.O.; Skytthe, A.; Backer, V. Estimates of asthma heritability in a large twin sample. Clin. Exp. Allergy 2010, 40, 1054–1061. [Google Scholar] [CrossRef]
- Hernandez-Pacheco, N.; Pino-Yanes, M.; Flores, C. Genomic Predictors of Asthma Phenotypes and Treatment Response. Front. Pediatr. 2019, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Moffatt, M.F.; Kabesch, M.; Liang, L.; Dixon, A.L.; Strachan, D.; Heath, S.; Depner, M.; von Berg, A.; Bufe, A.; Rietschel, E.; et al. Genetic variants regulating ORMDL3 expression contribute to the risk of childhood asthma. Nature 2007, 448, 470–473. [Google Scholar] [CrossRef] [PubMed]
- Willis-Owen, S.A.; Cookson, W.O.; Moffatt, M.F. The Genetics and Genomics of Asthma. Annu. Rev. Genom. Hum. Genet. 2018, 19, 223–246. [Google Scholar] [CrossRef]
- Vicente, C.T.; Revez, J.A.; Ferreira, M.A.R. Lessons from ten years of genome-wide association studies of asthma. Clin. Transl. Immunol. 2017, 6, e165. [Google Scholar] [CrossRef]
- Kim, K.W.; Ober, C. Lessons Learned from GWAS of Asthma. Allergy Asthma Immunol. Res. 2019, 11, 170–187. [Google Scholar] [CrossRef]
- Shrine, N.; Portelli, M.A.; John, C.; Artigas, M.S.; Bennett, N.; Hall, R.; Lewis, J.; Henry, A.P.; Billington, C.K.; Ahmad, A.; et al. Moderate-to-severe asthma in individuals of European ancestry: A genome-wide association study. Lancet Respir. Med. 2019, 7, 20–34. [Google Scholar] [CrossRef]
- Daya, M.; Rafaels, N.; Brunetti, T.M.; Chavan, S.; Levin, A.M.; Shetty, A.; Gignoux, C.R.; Boorgula, M.P.; Wojcik, G.; Campbell, M.; et al. Association study in African-admixed populations across the Americas recapitulates asthma risk loci in non-African populations. Nat. Commun. 2019, 10, 880. [Google Scholar] [CrossRef]
- Ferreira, M.A.; Mathur, R.; Vonk, J.M.; Szwajda, A.; Brumpton, B.; Granell, R.; Brew, B.K.; Ullemar, V.; Lu, Y.; Jiang, Y.; et al. Genetic Architectures of Childhood- and Adult-Onset Asthma Are Partly Distinct. Am. J. Hum. Genet. 2019, 104, 665–684. [Google Scholar] [CrossRef] [PubMed]
- Pividori, M.; Schoettler, N.; Nicolae, D.L.; Ober, C.; Im, H.K. Shared and distinct genetic risk factors for childhood-onset and adult-onset asthma: Genome-wide and transcriptome-wide studies. Lancet Respir. Med. 2019, 7, 509–522. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A.; Rask-Andersen, M.; Karlsson, T.; Ek, W.E. Genome-wide association analysis of 350 000 Caucasians from the UK Biobank identifies novel loci for asthma, hay fever and eczema. Hum. Mol. Genet. 2019, 28, 4022–4041. [Google Scholar] [CrossRef] [PubMed]
- Olafsdottir, T.A.; Theodors, F.; Bjarnadottir, K.; Bjornsdottir, U.S.; Agustsdottir, A.B.; Stefansson, O.A.; Ivarsdottir, E.V.; Sigurdsson, J.K.; Benonisdottir, S.; Eyjolfsson, G.I.; et al. Eighty-eight variants highlight the role of T cell regulation and airway remodeling in asthma pathogenesis. Nat. Commun. 2020, 11, 393. [Google Scholar] [CrossRef]
- Maher, B. Personal genomes: The case of the missing heritability. Nature 2008, 456, 18–21. [Google Scholar] [CrossRef]
- Manolio, T.A.; Collins, F.S.; Cox, N.J.; Goldstein, D.B.; Hindorff, L.A.; Hunter, D.J.; McCarthy, M.I.; Ramos, E.M.; Cardon, L.R.; Chakravarti, A.; et al. Finding the missing heritability of complex diseases. Nature 2009, 461, 747–753. [Google Scholar] [CrossRef]
- Igartua, C.; Myers, R.A.; Mathias, R.A.; Pino-Yanes, M.; Eng, C.; Graves, P.E.; Levin, A.M.; Del-Rio-Navarro, B.E.; Jackson, D.J.; Livne, O.E.; et al. Ethnic-specific associations of rare and low-frequency DNA sequence variants with asthma. Nat. Commun. 2015, 6, 5965. [Google Scholar] [CrossRef]
- Ionita-Laza, I.; Perry, G.H.; Raby, B.A.; Klanderman, B.; Lee, C.; Laird, N.M.; Weiss, S.T.; Lange, C. On the analysis of copy-number variations in genome-wide association studies: A translation of the family-based association test. Genet. Epidemiol. 2008, 32, 273–284. [Google Scholar] [CrossRef]
- Walsh, K.M.; Bracken, M.B.; Murk, W.K.; Hoh, J.; Dewan, A.T. Association between reduced copy-number at T-cell receptor gamma (TCRgamma) and childhood allergic asthma: A possible role for somatic mosaicism. Mutat. Res. 2010, 690, 89–94. [Google Scholar] [CrossRef][Green Version]
- Cooper, G.M.; Zerr, T.; Kidd, J.M.; Eichler, E.E.; Nickerson, D.A. Systematic assessment of copy number variant detection via genome-wide SNP genotyping. Nat. Genet. 2008, 40, 1199–1203. [Google Scholar] [CrossRef]
- Dominiczak, A.F.; McBride, M.W. Genetics of common polygenic stroke. Nat. Genet. 2003, 35, 116–117. [Google Scholar] [CrossRef] [PubMed]
- Heng, H.H.Q. Missing heritability and stochastic genome alterations. Nat. Rev. Genet. 2010, 11, 812. [Google Scholar] [CrossRef] [PubMed]
- Rava, M.; Smit, L.A.; Nadif, R. Gene–environment interactions in the study of asthma in the postgenomewide association studies era. Curr. Opin. Allergy Clin. Immunol. 2015, 15, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.; Mannino, D.; Brown, C.; Crocker, D.; Twum-Baah, N.; Holguin, F. Body mass index and asthma severity in the National Asthma Survey. Thorax 2008, 63, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Curjuric, I.; Imboden, M.; Nadif, R.; Kumar, A.; Schindler, C.; Haun, M.; Kronenberg, F.; Künzli, N.; Phuleria, H.; Postma, D.S.; et al. Different Genes Interact with Particulate Matter and Tobacco Smoke Exposure in Affecting Lung Function Decline in the General Population. PLoS ONE 2012, 7, e40175. [Google Scholar] [CrossRef]
- Hedman, L.; Bjerg, A.; Sundberg, S.; Forsberg, B.; Rönmark, E. Both environmental tobacco smoke and personal smoking is related to asthma and wheeze in teenagers. Thorax 2010, 66, 20–25. [Google Scholar] [CrossRef]
- Guibas, G.V.; Mathioudakis, A.G.; Tsoumani, M.; Tsabouri, S. Relationship of Allergy with Asthma: There Are More Than the Allergy “Eggs” in the Asthma “Basket”. Front. Pediatr. 2017, 5, 92. [Google Scholar] [CrossRef]
- Rodgman, A.; Smith, C.J.; Perfetti, T.A. The composition of cigarette smoke: A retrospective, with emphasis on polycyclic components. Hum. Exp. Toxicol. 2000, 19, 573–595. [Google Scholar] [CrossRef]
- Çalışkan, M.; Bochkov, Y.A.; Kreiner-Møller, E.; Bønnelykke, K.; Stein, M.M.; Du, G.; Bisgaard, H.; Jackson, D.J.; Gern, J.E.; Lemanske, R.F.; et al. Rhinovirus Wheezing Illness and Genetic Risk of Childhood-Onset Asthma. N. Engl. J. Med. 2013, 368, 1398–1407. [Google Scholar] [CrossRef]
- Smit, L.A.M.; Bouzigon, E.; Pin, I.; Siroux, V.; Monier, F.; Aschard, H.; Bousquet, J.; Gormand, F.; Just, J.; Le Moual, N.; et al. 17q21 variants modify the association between early respiratory infections and asthma. Eur. Respir. J. 2009, 36, 57–64. [Google Scholar] [CrossRef]
- Bukvic, B.K.; Blekic, M.; Simpson, A.; Marinho, S.; Curtin, J.A.; Hankinson, J.; Aberle, N.; Custovic, A. Asthma severity, polymorphisms in 20p13 and their interaction with tobacco smoke exposure. Pediatr. Allergy Immunol. 2013, 24, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Van Eerdewegh, P.; Little, R.D.; Dupuis, J.; Del Mastro, R.G.; Falls, K.; Simon, J.; Torrey, D.; Pandit, S.; McKenny, J.; Braunschweiger, K.; et al. Association of the ADAM33 gene with asthma and bronchial hyperresponsiveness. Nature 2002, 418, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Hancock, D.B.; Artigas, M.S.; Gharib, S.A.; Henry, A.; Manichaikul, A.; Ramasamy, A.; Loth, D.W.; Imboden, M.; Koch, B.; McArdle, W.L.; et al. Genome-Wide Joint Meta-Analysis of SNP and SNP-by-Smoking Interaction Identifies Novel Loci for Pulmonary Function. PLOS Genet. 2012, 8, e1003098. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Tanaka, K.; Arakawa, M. IL3rs40401 Polymorphism and Interaction with Smoking in Risk of Asthma in Japanese Women: The Kyushu Okinawa Maternal and Child Health Study. Scand. J. Immunol. 2014, 79, 410–414. [Google Scholar] [CrossRef]
- Wu, J.; Hankinson, J.; Kopec-Harding, K.; Custovic, A.; Simpson, A. Interaction between glutathione S-transferase variants, maternal smoking and childhood wheezing changes with age. Pediatr. Allergy Immunol. 2013, 24, 501–508. [Google Scholar] [CrossRef]
- Dempfle, A.; Scherag, A.; Hein, R.; Beckmann, L.; Chang-Claude, J.; Schafer, H. Gene-environment interactions for complex traits: Definitions, methodological requirements and challenges. Eur. J. Hum. Genet. 2008, 16, 1164–1172. [Google Scholar] [CrossRef]
- Kim, Y.; Han, B.G.; KoGES Group. Cohort Profile: The Korean Genome and Epidemiology Study (KoGES) Consortium. Int. J. Epidemiol. 2017, 46, e20. [Google Scholar] [CrossRef]
- Rentzsch, P.; Witten, D.; Cooper, G.M.; Shendure, J.; Kircher, M. CADD: Predicting the deleteriousness of variants throughout the human genome. Nucleic Acids Res. 2018, 47, D886–D894. [Google Scholar] [CrossRef]
- Quang, D.; Chen, Y.; Xie, X. DANN: A deep learning approach for annotating the pathogenicity of genetic variants. Bioinformatics 2015, 31, 761–763. [Google Scholar] [CrossRef]
- Boyle, A.P.; Hong, E.L.; Hariharan, M.; Cheng, Y.; Schaub, M.A.; Kasowski, M.; Karczewski, K.J.; Park, J.; Hitz, B.C.; Weng, S.; et al. Annotation of functional variation in personal genomes using RegulomeDB. Genome Res. 2012, 22, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.; Daly, M.J.; et al. PLINK: A tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef] [PubMed]
- Devlin, B.; Roeder, K. Genomic control for association studies. Biometrics 1999, 55, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- D’antona, S.; Castiglioni, I.; Porro, D.; Cava, C. Consequences of exposure to pollutants on respiratory health: From genetic correlations to causal relationships. PLoS ONE 2022, 17, e0277235. [Google Scholar] [CrossRef] [PubMed]
- Kidd, C.D.A.; Thompson, P.J.; Barrett, L.; Baltic, S. Histone Modifications and Asthma. The Interface of the Epigenetic and Genetic Landscapes. Am. J. Respir. Cell Mol. Biol. 2016, 54, 3–12. [Google Scholar] [CrossRef]
- Moffatt, M.F.; Gut, I.G.; Demenais, F.; Strachan, D.P.; Bouzigon, E.; Heath, S.; von Mutius, E.; Farrall, M.; Lathrop, M.; Cookson, W.; et al. A large-scale, consortium-based genomewide association study of asthma. N. Engl. J. Med. 2010, 363, 1211–1221. [Google Scholar] [CrossRef]
- Soini, Y.; Kosma, V.-M.; Pirinen, R. KDM4A, KDM4B and KDM4C in non-small cell lung cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 12922–12928. [Google Scholar]
- Greenlee, K.J.; Werb, Z.; Kheradmand, F.; Hardy, E.; Hardy-Sosa, A.; Fernandez-Patron, C.; Sun, W.; Hu, Q.; Ji, W.; Wright, G.; et al. Matrix Metalloproteinases in Lung: Multiple, Multifarious, and Multifaceted. Physiol. Rev. 2007, 87, 69–98. [Google Scholar] [CrossRef]
- Liu, X.; Zhao, D.; Shan, Y.; Cui, W.; Xie, Q.; Jiang, J.; Peng, W.; Zhang, C.; Duan, C. Development and validation of a novel immune-related prognostic signature in lung squamous cell carcinoma patients. Sci. Rep. 2022, 12, 20737. [Google Scholar] [CrossRef]
- Mok, P.L.; Anandasayanam, A.N.K.; David, H.M.O.; Tong, J.; Farhana, A.; Khan, M.S.A.; Sivaprakasam, G.; Koh, A.E.-H.; Alzahrani, B. Lung development, repair and cancer: A study on the role of MMP20 gene in adenocarcinoma. PLoS ONE 2021, 16, e0250552. [Google Scholar] [CrossRef]
- de Leeuw, C.A.; Mooij, J.M.; Heskes, T.; Posthuma, D. MAGMA: Generalized Gene-Set Analysis of GWAS Data. PLoS Comput. Biol. 2015, 11, e1004219. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Taskesen, E.; van Bochoven, A.; Posthuma, D. Functional mapping and annotation of genetic associations with FUMA. Nat. Commun. 2017, 8, 1826. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.; Yoon, H.-Y.; Park, J.-Y.; Kim, Y.-J.; Hwang, H.-S.; Yee, J.; Gwak, H.-S. Association between ADCY9 Gene Polymorphisms and Ritodrine Treatment Outcomes in Patients with Preterm Labor. Pharmaceutics 2021, 13, 1653. [Google Scholar] [CrossRef]
- Teixeira, H.M.; Alcantara-Neves, N.M.; Barreto, M.; Figueiredo, C.A.; Costa, R.S. Adenylyl cyclase type 9 gene polymorphisms are associated with asthma and allergy in Brazilian children. Mol. Immunol. 2017, 82, 137–145. [Google Scholar] [CrossRef]
- Ferreira, C.M.; Chen, J.L.; Li, J.; Shimomura, K.; Yang, X.; Lussier, Y.A.; Pinto, L.H.; Solway, J. Genetic Interactions between Chromosomes 11 and 18 Contribute to Airway Hyperresponsiveness in Mice. PLoS ONE 2012, 7, e29579. [Google Scholar] [CrossRef]
- Jiang, J.; Qin, T.; Zhang, L.; Liu, Q.; Wu, J.; Dai, R.; Zhou, L.; Zhao, Q.; Luo, X.; Wang, H.; et al. IL-21 Rescues the Defect of IL-10-Producing Regulatory B Cells and Improves Allergic Asthma in DOCK8 Deficient Mice. Front. Immunol. 2021, 12, 695596. [Google Scholar] [CrossRef] [PubMed]
- Freeman, A.F.; Olivier, K.N. Hyper-IgE Syndromes and the Lung. Clin. Chest Med. 2016, 37, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhang, S.; Qin, T.; Jiang, J.; Liu, Q.; Zhang, L.; Zhao, X.; Dai, J. IL-21 alleviates allergic asthma in DOCK8-knockout mice. Biochem. Biophys. Res. Commun. 2018, 501, 92–99. [Google Scholar] [CrossRef]
- Liberzon, A. A Description of the Molecular Signatures Database (MSigDB) Web Site. Methods Mol. Biol. 2014, 1150, 153–160. [Google Scholar] [CrossRef]
- Liberzon, A.; Birger, C.; Thorvaldsdottir, H.; Ghandi, M.; Mesirov, J.P.; Tamayo, P. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst. 2015, 1, 417–425. [Google Scholar] [CrossRef]
- Liberzon, A.; Subramanian, A.; Pinchback, R.; Thorvaldsdóttir, H.; Tamayo, P.; Mesirov, J.P. Molecular signatures database (MSigDB) 3.0. Bioinformatics 2011, 27, 1739–1740. [Google Scholar] [CrossRef] [PubMed]
- Nicetto, D.; Zaret, K.S. Role of H3K9me3 heterochromatin in cell identity establishment and maintenance. Curr. Opin. Genet. Dev. 2019, 55, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Th2 cytokines and asthma: An introduction. Respir. Res. 2001, 2, 64–65. [Google Scholar] [CrossRef]
- Nguyen, M.L.T.; Jones, S.A.; Prier, J.E.; Russ, B.E. Transcriptional Enhancers in the Regulation of T Cell Differentiation. Front. Immunol. 2015, 6, 462. [Google Scholar] [CrossRef] [PubMed]
- Brightling, C.E.; Bradding, P.; Pavord, I.D.; Wardlaw, A.J. New insights into the role of the mast cell in asthma. Clin. Exp. Allergy 2003, 33, 550–556. [Google Scholar] [CrossRef]
- Casale, T.B.; Marom, Z. Mast cells and asthma. The role of mast cell mediators in the pathogenesis of allergic asthma. Ann. Allergy 1983, 51, 2–6. [Google Scholar]
- Stephens, N.L.; Jiang, H. Relaxation of smooth muscle. Can. J. Physiol. Pharmacol. 1994, 72, 1345–1350. [Google Scholar] [CrossRef]
- Rawat, K.; Syeda, S.; Shrivastava, A. Neutrophil-derived granule cargoes: Paving the way for tumor growth and progression. Cancer Metastasis Rev. 2021, 40, 221–244. [Google Scholar] [CrossRef]
- Lauredo, I.T.; Forteza, R.M.; Botvinnikova, Y.; Abraham, W.M.; Magnen, M.; Gueugnon, F.; Petit-Courty, A.; Baranek, T.; Sizaret, D.; Brewah, Y.A.; et al. Leukocytic cell sources of airway tissue kallikrein. Am. J. Physiol. Cell. Mol. Physiol. 2004, 286, L734–L740. [Google Scholar] [CrossRef][Green Version]
- Chai, O.H.; Han, E.-H.; Lee, H.-K.; Song, C.H. Mast cells play a key role in Th2 cytokine-dependent asthma model through production of adhesion molecules by liberation of TNF-α. Exp. Mol. Med. 2011, 43, 35–43. [Google Scholar] [CrossRef]
- Shoemark, A.; Dixon, M.; Beales, P.L.; Hogg, C.L. Bardet Biedl syndrome: Motile ciliary phenotype. Chest 2015, 147, 764–770. [Google Scholar] [CrossRef] [PubMed]
- Tanizaki, Y.; Hosokawa, M.; Akagi, K.; Matsuka, Y.; Shioda, Y.; Sato, T.; Tada, S.; Takahashi, K.; Kimura, I. Function of blood monocytes in bronchial asthma--numerical changes and beta-galactosidase activity. Nihon Kyobu Shikkan Gakkai Zasshi 1984, 22, 208–213. [Google Scholar] [PubMed]
- Chen, T.; Yan, Y.E.; Liu, S.; Liu, H.X.; Yan, H.Y.; Hou, L.F.; Qu, W.; Ping, J. Increased Fetal Thymocytes Apoptosis Contributes to Prenatal Nicotine Exposure-induced Th1/Th2 Imbalance in Male Offspring Mice. Sci. Rep. 2016, 6, 39013. [Google Scholar] [CrossRef]
- Middlebrook, A.J.; Martina, C.; Chang, Y.; Lukas, R.J.; DeLuca, D. Effects of Nicotine Exposure on T Cell Development in Fetal Thymus Organ Culture: Arrest of T Cell Maturation. J. Immunol. 2002, 169, 2915–2924. [Google Scholar] [CrossRef]
- Qu, W.; Zhao, W.-H.; Wen, X.; Yan, H.-Y.; Liu, H.-X.; Hou, L.-F.; Ping, J. Prenatal nicotine exposure induces thymic hypoplasia in mice offspring from neonatal to adulthood. Toxicol. Lett. 2018, 304, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Verma, S.; Burra, U.; Murthy, N.S.; Mohanty, N.K.; Saxena, S. Flow Cytometric analysis of Th1 and Th2 cytokines in PBMCs as a parameter of immunological dysfunction in patients of Superficial Transitional cell carcinoma of bladder. Cancer Immunol. Immunother. 2005, 55, 734–743. [Google Scholar] [CrossRef]
- Karpuzoglu-Sahin, E.; Hissong, B.D.; Ansar Ahmed, S. Interferon-gamma levels are upregulated by 17-beta-estradiol and diethylstilbestrol. J. Reprod. Immunol. 2001, 52, 113–127. [Google Scholar] [CrossRef]
- Stromnes, I.M.; Cerretti, L.M.; Liggitt, D.; Harris, R.A.; Goverman, J.M. Differential regulation of central nervous system autoimmunity by T(H)1 and T(H)17 cells. Nat. Med. 2008, 14, 337–342. [Google Scholar] [CrossRef]
- Guerra-Álvarez, M.; Moreno-Ortega, A.J.; Navarro, E.; Fernández-Morales, J.C.; Egea, J.; López, M.G.; Cano-Abad, M.F. Positive allosteric modulation of alpha-7 nicotinic receptors promotes cell death by inducing Ca2+ release from the endoplasmic reticulum. J. Neurochem. 2015, 133, 309–319. [Google Scholar] [CrossRef]
- Vivekanandarajah, A.; Chan, Y.L.; Chen, H.; Machaalani, R. Prenatal cigarette smoke exposure effects on apoptotic and nicotinic acetylcholine receptor expression in the infant mouse brainstem. Neurotoxicology 2016, 53, 53–63. [Google Scholar] [CrossRef]
- Liu, H.-X.; Liu, S.; Qu, W.; Yang, H.-Y.; Wen, X.; Chen, T.; Hou, L.-F.; Ping, J. α7 nAChR mediated Fas demethylation contributes to prenatal nicotine exposure-induced programmed thymocyte apoptosis in mice. Oncotarget 2017, 8, 93741–93756. [Google Scholar] [CrossRef] [PubMed]
- McManus, J.M.; Gaston, B.; Zein, J.; Sharifi, N. Association Between Asthma and Reduced Androgen Receptor Expression in Airways. J. Endocr. Soc. 2022, 6, bvac047. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.; Wei, J.; Hao, Y.; Tao, J.; Li, Y.; Liu, M.; Xu, B.; Li, B. Consistent Biomarkers and Related Pathogenesis Underlying Asthma Revealed by Systems Biology Approach. Int. J. Mol. Sci. 2019, 20, 4037. [Google Scholar] [CrossRef]
- Park, S.-W.; Verhaeghe, C.; Nguyenvu, L.T.; Barbeau, R.; Eisley, C.J.; Nakagami, Y.; Huang, X.; Woodruff, P.G.; Fahy, J.V.; Erle, D.J. Distinct Roles of FOXA2 and FOXA3 in Allergic Airway Disease and Asthma. Am. J. Respir. Crit. Care Med. 2009, 180, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; An, L.; Ran, D.; Lizarraga, R.; Bondy, C.; Zhou, X.; Harper, R.W.; Liao, S.-Y.; Chen, Y. The Club Cell Marker SCGB1A1 Downstream of FOXA2 is Reduced in Asthma. Am. J. Respir. Cell Mol. Biol. 2019, 60, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Caramori, G.; Lim, S.; Ito, K.; Tomita, K.; Oates, T.; Jazrawi, E.; Chung, K.; Barnes, P.; Adcock, I. Expression of GATA family of transcription factors in T-cells, monocytes and bronchial biopsies. Eur. Respir. J. 2001, 18, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Fang, P.; Shi, H.Y.; Wu, X.M.; Zhang, Y.H.; Zhong, Y.J.; Deng, W.J.; Zhang, Y.P.; Xie, M. Targeted inhibition of GATA-6 attenuates airway inflammation and remodeling by regulating caveolin-1 through TLR2/MyD88/NF-κB in murine model of asthma. Mol. Immunol. 2016, 75, 144–150. [Google Scholar] [CrossRef]
- Tumes, D.; Hirahara, K.; Papadopoulos, M.; Shinoda, K.; Onodera, A.; Kumagai, J.; Yip, K.H.; Pant, H.; Kokubo, K.; Kiuchi, M.; et al. Ezh2 controls development of natural killer T cells, which cause spontaneous asthma-like pathology. J. Allergy Clin. Immunol. 2019, 144, 549–560.e10. [Google Scholar] [CrossRef]
- Kozmus, C.E.; Potočnik, U. Reference genes for real-time qPCR in leukocytes from asthmatic patients before and after anti-asthma treatment. Gene 2015, 570, 71–77. [Google Scholar] [CrossRef]
- Jolliffe, D.A.; Kilpin, K.; MacLaughlin, B.D.; Greiller, C.L.; Hooper, R.L.; Barnes, N.C.; Timms, P.M.; Rajakulasingam, R.K.; Bhowmik, A.; Choudhury, A.B.; et al. Prevalence, determinants and clinical correlates of vitamin D deficiency in adults with inhaled corticosteroid-treated asthma in London, UK. J. Steroid Biochem. Mol. Biol. 2018, 175, 88–96. [Google Scholar] [CrossRef]
- Diez, D.; Goto, S.; Fahy, J.V.; Erle, D.J.; Woodruff, P.G.; Wheelock, M.; Wheelock, C.E. Network analysis identifies a putative role for the PPAR and type 1 interferon pathways in glucocorticoid actions in asthmatics. BMC Med. Genom. 2012, 5, 27. [Google Scholar] [CrossRef]
- Pniewska, E.; Sokolowska, M.; Kupryś-Lipińska, I.; Kacprzak, D.; Kuna, P.; Pawliczak, R. Exacerbating Factors Induce Different Gene Expression Profiles in Peripheral Blood Mononuclear Cells from Asthmatics, Patients with Chronic Obstructive Pulmonary Disease and Healthy Subjects. Int. Arch. Allergy Immunol. 2014, 165, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Pazdrak, K.; Moon, Y.; Straub, C.; Stafford, S.; Kurosky, A. Eosinophil resistance to glucocorticoid-induced apoptosis is mediated by the transcription factor NFIL3. Apoptosis 2016, 21, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Rothman, P.B. The transcriptional regulator NFIL3 controls IgE production. Trans. Am. Clin. Clim. Assoc. 2010, 121, 156–171. [Google Scholar]
- Nowak, J.K.; Dworacka, M.; Gubaj, N.; Dossimov, A.; Dossimov, Z.; Walkowiak, J. Expression profiling of ileal mucosa in asthma reveals upregulation of innate immunity and genes characteristic of Paneth and goblet cells. Allergy Asthma Clin. Immunol. 2021, 17, 82. [Google Scholar] [CrossRef]
- Song, Y.; Wang, Z.; Jiang, J.; Piao, Y.; Li, L.; Xu, C.; Piao, H.; Li, L.; Yan, G. DEK-targeting aptamer DTA-64 attenuates bronchial EMT-mediated airway remodelling by suppressing TGF-β1/Smad, MAPK and PI3K signalling pathway in asthma. J. Cell Mol. Med. 2020, 24, 13739–13750. [Google Scholar] [CrossRef]
- Lin, L.; Wang, Y.; Sun, B.; Liu, L.; Ying, W.; Wang, W.; Zhou, Q.; Hou, J.; Yao, H.; Hu, L.; et al. The clinical, immunological and genetic features of 12 Chinese patients with STAT3 mutations. Allergy Asthma Clin. Immunol. 2020, 16, 65. [Google Scholar] [CrossRef]
- Frey-Jakobs, S.; Hartberger, J.M.; Fliegauf, M.; Bossen, C.; Wehmeyer, M.L.; Neubauer, J.C.; Bulashevska, A.; Proietti, M.; Fröbel, P.; Nöltner, C.; et al. ZNF341 controls STAT3 expression and thereby immunocompetence. Sci. Immunol. 2018, 3, eaat4941. [Google Scholar] [CrossRef]
- Schlag, K.; Steinhilber, D.; Karas, M.; Sorg, B.L. Analysis of proximal ALOX5 promoter binding proteins by quantitative proteomics. FEBS J. 2020, 287, 4481–4499. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, Y.; Chen, W.-X.; Xu, Z.-M. sj-xlsx-1-imr-10.1177_03000605211029521—Supplemental material for Identification of key genes in allergic rhinitis by bioinformatics analysis. J. Int. Med. Res. 2021, 49, 3000605211029521. [Google Scholar] [CrossRef]
- Jargosch, M.; Kröger, S.; Gralinska, E.; Klotz, U.; Fang, Z.; Chen, W.; Leser, U.; Selbig, J.; Groth, D.; Baumgrass, R. Data integration for identification of important transcription factors of STAT6-mediated cell fate decisions. Genet. Mol. Res. 2016, 15, 4238. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, M.; Henderson, W.R.; Clark, A.E.; Simmons, R.M.; Ye, X.; Smith, K.D.; Aderem, A. Activating transcription factor 3 is a negative regulator of allergic pulmonary inflammation. J. Exp. Med. 2008, 205, 2349–2357. [Google Scholar] [CrossRef]
- Ogasawara, T.; Hatano, M.; Satake, H.; Ikari, J.; Taniguchi, T.; Tsuruoka, N.; Watanabe-Takano, H.; Fujimura, L.; Sakamoto, A.; Hirata, H.; et al. Development of chronic allergic responses by dampening Bcl6-mediated suppressor activity in memory T helper 2 cells. Proc. Natl. Acad. Sci. USA 2017, 114, E741–E750. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Hettinga, A.; Kale, S.L.; Hu, S.; Xie, M.M.; Dent, A.L.; Ray, A.; Poholek, A.C. Blimp-1 is essential for allergen-induced asthma and Th2 cell development in the lung. J. Exp. Med. 2020, 217, e20190742. [Google Scholar] [CrossRef] [PubMed]
- Golebski, K.; Luiten, S.; van Egmond, D.; de Groot, E.; Röschmann, K.I.; Fokkens, W.J.; van Drunen, C.M. High degree of overlap between responses to a virus and to the house dust mite allergen in airway epithelial cells. PLoS ONE 2014, 9, e87768. [Google Scholar] [CrossRef]
- Jiang, Y.; Yan, Q.; Liu, C.-X.; Peng, C.-W.; Zheng, W.-J.; Zhuang, H.-F.; Huang, H.-T.; Liu, Q.; Liao, H.-L.; Zhan, S.-F.; et al. Insights into potential mechanisms of asthma patients with COVID-19: A study based on the gene expression profiling of bronchoalveolar lavage fluid. Comput. Biol. Med. 2022, 146, 105601. [Google Scholar] [CrossRef]
- Wu, J.; Duan, R.; Cao, H.; Field, D.; Newnham, C.M.; Koehler, D.R.; Zamel, N.; Pritchard, M.A.; Hertzog, P.; Post, M.; et al. Regulation of epithelium-specific Ets-like factors ESE-1 and ESE-3 in airway epithelial cells: Potential roles in airway inflammation. Cell Res. 2008, 18, 649–663. [Google Scholar] [CrossRef]
- Lee, S.-Y.; Won, H.-K.; Kim, B.-K.; Kim, S.-H.; Chang, Y.-S.; Cho, S.-H.; Kelly, H.W.; Tantisira, K.G.; Park, H.-W. Identification of a key gene module associated with glucocorticoid- induced derangement in bone mineral density in patients with asthma. Sci. Rep. 2019, 9, 20133. [Google Scholar] [CrossRef]
- Gunawardhana, L.P.; Gibson, P.G.; Simpson, J.L.; Powell, H.; Baines, K.J. Activity and expression of histone acetylases and deacetylases in inflammatory phenotypes of asthma. Clin. Exp. Allergy 2014, 44, 47–57. [Google Scholar] [CrossRef]
- Chen, G.; Korfhagen, T.R.; Karp, C.L.; Impey, S.; Xu, Y.; Randell, S.H.; Kitzmiller, J.; Maeda, Y.; Haitchi, H.M.; Sridharan, A.; et al. Foxa3 Induces Goblet Cell Metaplasia and Inhibits Innate Antiviral Immunity. Am. J. Respir. Crit. Care Med. 2014, 189, 301–313. [Google Scholar] [CrossRef]
- Xia, M.; Xu, H.; Dai, W.; Zhu, C.; Wu, L.; Yan, S.; Ge, X.; Zhou, W.; Chen, C.; Dai, Y. The role of HDAC2 in cigarette smoke–induced airway inflammation in a murine model of asthma and the effect of intervention with roxithromycin. J. Asthma 2018, 55, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Chaturvedi, R.; Hashim, Z.; Nath, A.; Khan, A.; Gupta, M.; Singh, H.; Agarwal, V. Role of P-gp and HDAC2 and their Reciprocal Relationship in Uncontrolled Asthma. Curr. Pharm. Biotechnol. 2021, 22, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Das, S.; Agrawal, A.; Mukhopadhyay, I.; Ghosh, B. Genetic association of key Th1/Th2 pathway candidate genes, IRF2, IL6, IFNGR2, STAT4 and IL4RA, with atopic asthma in the Indian population. J. Hum. Genet. 2015, 60, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Verma, M.; Michalec, L.; Sripada, A.; McKay, J.; Sirohi, K.; Verma, D.; Sheth, D.; Martin, R.; Dyjack, N.; Seibold, M.A.; et al. The molecular and epigenetic mechanisms of innate lymphoid cell (ILC) memory and its relevance for asthma. J. Exp. Med. 2021, 218, e20201354. [Google Scholar] [CrossRef] [PubMed]
- Díaz, L.A.C.; Ortega, A.G.; Álvarez, R.D.C.C.; Félix, J.S.V.; de Oca, E.P.M. Regulatory SNP rs5743417 impairs constitutive expression of human β-defensin 1 and has high frequency in Africans and Afro-Americans. Int. J. Immunogenet. 2020, 47, 332–341. [Google Scholar] [CrossRef]
- Bosch, T.v.D.; Leus, N.G.; Wapenaar, H.; Boichenko, A.; Hermans, J.; Bischoff, R.; Haisma, H.J.; Dekker, F.J. A 6-alkylsalicylate histone acetyltransferase inhibitor inhibits histone acetylation and pro-inflammatory gene expression in murine precision-cut lung slices. Pulm. Pharmacol. Ther. 2017, 44, 88–95. [Google Scholar] [CrossRef]
- Liu, Y.; Du, J.; Liu, X.; Wang, L.; Han, Y.; Huang, C.; Liang, R.; Zheng, F.; Shi, G.; Li, B. MG149 inhibits histone acetyltransferase KAT8-mediated IL-33 acetylation to alleviate allergic airway inflammation and airway hyperresponsiveness. Signal Transduct. Target. Ther. 2021, 6, 321. [Google Scholar] [CrossRef]
- van der Plaat, D.A.; Vonk, J.M.; Lahousse, L.; de Jong, K.; Faiz, A.; Nedeljkovic, I.; Amin, N.; van Diemen, C.C.; Brusselle, G.G.; Bossé, Y.; et al. Limited overlap in significant hits between genome-wide association studies on two airflow obstruction definitions in the same population. BMC Pulm. Med. 2019, 19, 58. [Google Scholar] [CrossRef]
- Fox, A.; Harland, K.L.; Kedzierska, K.; Kelso, A. Exposure of Human CD8+ T Cells to Type-2 Cytokines Impairs Division and Differentiation and Induces Limited Polarization. Front. Immunol. 2018, 9, 1141. [Google Scholar] [CrossRef]
- Wang, S.-R.; Hu, R.-D.; Ma, M.; You, X.; Cui, H.; He, Y.; Xu, D.; Zhao, Z.-B.; Selmi, C.; Gershwin, M.E.; et al. FoxO1 suppresses IL-10 producing B cell differentiation via negatively regulating Blimp-1 expression and contributes to allergic asthma progression. Mucosal Immunol. 2022, 15, 459–470. [Google Scholar] [CrossRef]
- Zhang, X.; Myers, J.M.B.; Burleson, J.; Ulm, A.; Bryan, K.S.; Chen, X.; Weirauch, M.T.; Baker, T.A.; Kovacic, M.S.B.; Ji, H. Nasal DNA methylation is associated with childhood asthma. Epigenomics 2018, 10, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Aneas, I.; Decker, D.C.; Howard, C.L.; Sobreira, D.R.; Sakabe, N.J.; Blaine, K.M.; Stein, M.M.; Hrusch, C.L.; Montefiori, L.E.; Tena, J.; et al. Asthma-associated genetic variants induce IL33 differential expression through an enhancer-blocking regulatory region. Nat. Commun. 2021, 12, 6115. [Google Scholar] [CrossRef] [PubMed]
- Amo, G.; Martí, M.; García-Menaya, J.M.; Cordobés, C.; Cornejo-García, J.A.; Blanca-López, N.; Canto, G.; Doña, I.; Blanca, M.; Torres, M.J.; et al. Identification of Novel Biomarkers for Drug Hypersensitivity After Sequencing of the Promoter Area in 16 Genes of the Vitamin D Pathway and the High-Affinity IgE Receptor. Front. Genet. 2019, 10, 582. [Google Scholar] [CrossRef] [PubMed]
- Mougey, E.; Lang, J.E.; Allayee, H.; Teague, W.G.; Dozor, A.J.; Wise, R.A.; Lima, J.J. ALOX5 Polymorphism associates with increased leukotriene production and reduced lung function and asthma control in children with poorly controlled asthma. Clin. Exp. Allergy 2013, 43, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Scaparrotta, A.; Franzago, M.; Marcovecchio, M.L.; Di Pillo, S.; Chiarelli, F.; Mohn, A.; Stuppia, L. Role of THRB, ARG1, and ADRB2 Genetic Variants on Bronchodilators Response in Asthmatic Children. J. Aerosol. Med. Pulm. Drug Deliv. 2019, 32, 164–173. [Google Scholar] [CrossRef]
- García-Menaya, J.M.; Cordobés-Durán, C.; García-Martín, E.; Agúndez, J.A.G. Pharmacogenetic Factors Affecting Asthma Treatment Response. Potential Implications for Drug Therapy. Front. Pharmacol. 2019, 10, 520. [Google Scholar] [CrossRef]
- Pulleyn, L.J.; Newton, R.; Adcock, I.M.; Barnes, P.J. TGFbeta1 allele association with asthma severity. Hum. Genet. 2001, 109, 623–627. [Google Scholar] [CrossRef]
- Hwang, S.S.; Kim, Y.U.; Lee, S.; Jang, S.W.; Kim, M.K.; Koh, B.H.; Lee, W.; Kim, J.; Souabni, A.; Busslinger, M.; et al. Transcription factor YY1 is essential for regulation of the Th2 cytokine locus and for Th2 cell differentiation. Proc. Natl. Acad. Sci. USA 2013, 110, 276–281. [Google Scholar] [CrossRef]
- Lee, M.; Lim, S.; Kim, Y.S.; Khalmuratova, R.; Shin, S.-H.; Kim, I.; Kim, H.-J.; Kim, D.-Y.; Rhee, C.-S.; Park, J.-W.; et al. DEP-induced ZEB2 promotes nasal polyp formation via epithelial-to-mesenchymal transition. J. Allergy Clin. Immunol. 2022, 149, 340–357. [Google Scholar] [CrossRef]
- Sakai, H.; Hirahara, M.; Chiba, Y.; Misawa, M. Antigen challenge influences various transcription factors of rat bronchus: Protein/DNA array study. Int. Immunopharmacol. 2011, 11, 1133–1136. [Google Scholar] [CrossRef]
- Popa, S.C.; Shin, J.A. The Intrinsically Disordered Loop in the USF1 bHLHZ Domain Modulates Its DNA-Binding Sequence Specificity in Hereditary Asthma. J. Phys. Chem. B 2019, 123, 9862–9871. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Shen, W.; Li, Z.; Zhang, W. CCCTC-binding factor transcriptionally regulates Galectin-7 and activates the JNK/STAT3 axis to aggravate bronchial epithelial cell injury. Pediatr. Pulmonol. 2022, 57, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Schmiedel, B.J.; Seumois, G.; Samaniego-Castruita, D.; Cayford, J.; Schulten, V.; Chavez, L.; Ay, F.; Sette, A.; Peters, B.; Vijayanand, P. 17q21 asthma-risk variants switch CTCF binding and regulate IL-2 production by T cells. Nat. Commun. 2016, 7, 13426. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Lu, Y.; Zou, F.; Fan, X.; Li, X.; Zhang, H.; Chen, H.; Sun, X.; Liu, Y. PTEN participates in airway remodeling of asthma by regulating CD38/Ca(2+)/CREB signaling. Aging 2020, 12, 16326–16340. [Google Scholar] [CrossRef] [PubMed]
- Bartel, S.; Schulz, N.; Alessandrini, F.; Schamberger, A.C.; Pagel, P.; Theis, F.J.; Milger, K.; Noessner, E.; Stick, S.M.; Kicic, A.; et al. Pulmonary microRNA profiles identify involvement of Creb1 and Sec14l3 in bronchial epithelial changes in allergic asthma. Sci. Rep. 2017, 7, srep46026. [Google Scholar] [CrossRef]
- Adoue, V.; Binet, B.; Malbec, A.; Fourquet, J.; Romagnoli, P.; van Meerwijk, J.; Amigorena, S.; Joffre, O.P. The Histone Methyltransferase SETDB1 Controls T Helper Cell Lineage Integrity by Repressing Endogenous Retroviruses. Immunity 2019, 50, 629–644.e8. [Google Scholar] [CrossRef]
- Barnes, P.J. Targeting cytokines to treat asthma and chronic obstructive pulmonary disease. Nat. Rev. Immunol. 2018, 18, 454–466. [Google Scholar] [CrossRef]
- Yuan, Y.L.; Zhang, X.; Liu, L.; Wang, G.; Chen-Yu Hsu, A.; Huang, D.; Wang, G.; Oliver, B.G. Total IgE Variability Is Associated with Future Asthma Exacerbations: A 1-Year Prospective Cohort Study. J. Allergy Clin. Immunol. Pr. 2021, 9, 2812–2824. [Google Scholar] [CrossRef]
- Han, S.; Zhen, W.; Guo, T.; Zou, J.; Li, F. SETDB1 promotes glioblastoma growth via CSF-1-dependent macrophage recruitment by activating the AKT/mTOR signaling pathway. J. Exp. Clin. Cancer Res. 2020, 39, 218. [Google Scholar] [CrossRef]
- Rot, A. Endothelial cell binding of NAP-1/IL-8: Role in neutrophil emigration. Immunol. Today 1992, 13, 291–294. [Google Scholar] [CrossRef]
- Koch, A.E.; Kunkel, S.L.; Pearce, W.H.; Shah, M.R.; Parikh, D.; Evanoff, H.L.; Haines, G.K.; Burdick, M.D.; Strieter, R.M. Enhanced production of the chemotactic cytokines interleukin-8 and monocyte chemoattractant protein-1 in human abdominal aortic aneurysms. Am. J. Pathol. 1993, 142, 1423–1431. [Google Scholar]
- Ribeiro, R.A.; Flores, C.A.; Cunha, F.Q.; Ferreira, S.H. IL-8 causes in vivo neutrophil migration by a cell-dependent mechanism. Immunology 1991, 73, 472–477. [Google Scholar] [PubMed]
- Muñoz, J.; Akhavan, N.S.; Mullins, A.P.; Arjmandi, B.H. Macrophage Polarization and Osteoporosis: A Review. Nutrients 2020, 12, 2999. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.F. Cytokines in chronic obstructive pulmonary disease. Eur. Respir. J. Suppl. 2001, 34, 50s–59s. [Google Scholar] [CrossRef]
- Wills-Karp, M.; Luyimbazi, J.; Xu, X.; Schofield, B.; Neben, T.Y.; Karp, C.L.; Donaldson, D.D. Interleukin-13: Central Mediator of Allergic Asthma. Science 1998, 282, 2258–2261. [Google Scholar] [CrossRef] [PubMed]
- Rose, C.E., Jr.; Sung, S.S.; Fu, S.M. Significant involvement of CCL2 (MCP-1) in inflammatory disorders of the lung. Microcirculation 2003, 10, 273–288. [Google Scholar] [CrossRef] [PubMed]
- Torrano, J.; Al Emran, A.; Hammerlindl, H.; Schaider, H. Emerging roles of H3K9me3, SETDB1 and SETDB2 in therapy-induced cellular reprogramming. Clin. Epigenetics 2019, 11, 43. [Google Scholar] [CrossRef]
- Verlaan, D.J.; Berlivet, S.; Hunninghake, G.M.; Madore, A.M.; Larivière, M.; Moussette, S.; Grundberg, E.; Kwan, T.; Ouimet, M.; Ge, B.; et al. Allele-specific chromatin remodeling in the ZPBP2/GSDMB/ORMDL3 locus associated with the risk of asthma and autoimmune disease. Am. J. Hum. Genet. 2009, 85, 377–393. [Google Scholar] [CrossRef]
- Chase, K.A.; Sharma, R.P. Nicotine induces chromatin remodelling through decreases in the methyltransferases GLP, G9a, Setdb1 and levels of H3K9me2. Int. J. Neuropsychopharmacol. 2013, 16, 1129–1138. [Google Scholar] [CrossRef]
- Lafuente-Sanchis, A.; Zúñiga, Á.; Galbis, J.M.; Cremades, A.; Estors, M.; Martínez-Hernández, N.J.; Carretero, J. Prognostic value of ERCC1, RRM1, BRCA1 and SETDB1 in early stage of non-small cell lung cancer. Clin. Transl. Oncol. 2016, 18, 798–804. [Google Scholar] [CrossRef]
- Hirano, T.; Matsunaga, K. Late-onset asthma: Current perspectives. J. Asthma Allergy 2018, 11, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Baptist, A.P.; Ross, J.A.; Clark, N.M. Older adults with asthma: Does age of asthma onset make a difference? J. Asthma 2013, 50, 836–841. [Google Scholar] [CrossRef]
- Moore, W.C.; Meyers, D.A.; Wenzel, S.E.; Teague, W.G.; Li, H.; Li, X.; D’Agostino, R., Jr.; Castro, M.; Curran-Everett, D.; Fitzpatrick, A.M.; et al. Identification of asthma phenotypes using cluster analysis in the Severe Asthma Research Program. Am. J. Respir. Crit. Care Med. 2010, 181, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Bleecker, E.; Moore, W.; Busse, W.W.; Castro, M.; Chung, K.F.; Calhoun, W.J.; Erzurum, S.; Gaston, B.; Israel, E.; et al. Unsupervised phenotyping of Severe Asthma Research Program participants using expanded lung data. J. Allergy Clin. Immunol. 2014, 133, 1280–1288. [Google Scholar] [CrossRef] [PubMed]
- McDonald, V.M.; Gibson, P.G. Exacerbations of severe asthma. Clin. Exp. Allergy 2012, 42, 670–677. [Google Scholar] [CrossRef]
- Jiao, K.; Zhou, Y.; Hogan, B.L.M. Identification of mZnf8, a Mouse Krüpel-Like Transcriptional Repressor, as a Novel Nuclear Interaction Partner of Smad1. Mol. Cell. Biol. 2002, 22, 7633–7644. [Google Scholar] [CrossRef]
- Baldwin, L.; Roche, W.R. Does remodelling of the airway wall precede asthma? Paediatr. Respir. Rev. 2002, 3, 315–320. [Google Scholar] [CrossRef]
- Grainge, C.L.; Lau, L.C.; Ward, J.A.; Dulay, V.; Lahiff, G.; Wilson, S.; Holgate, S.; Davies, D.E.; Howarth, P.H. Effect of bronchoconstriction on airway remodeling in asthma. N. Engl. J. Med. 2011, 364, 2006–2015. [Google Scholar] [CrossRef]
- Tagaya, E.; Tamaoki, J. Mechanisms of Airway Remodeling in Asthma. Allergol. Int. 2007, 56, 331–340. [Google Scholar] [CrossRef]
- Wnuk, D.; Paw, M.; Ryczek, K.; Bochenek, G.; Sładek, K.; Madeja, Z.; Michalik, M. Enhanced asthma-related fibroblast to myofibroblast transition is the result of profibrotic TGF-β/Smad2/3 pathway intensification and antifibrotic TGF-β/Smad1/5/(8)9 pathway impairment. Sci. Rep. 2020, 10, 16492. [Google Scholar] [CrossRef]
- Minshall, E.M.; Leung, D.Y.; Martin, R.J.; Song, Y.L.; Cameron, L.; Ernst, P.; Hamid, Q. Eosinophil-associated TGF-beta1 mRNA expression and airways fibrosis in bronchial asthma. Am. J. Respir. Cell Mol. Biol. 1997, 17, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Halwani, R.; Al-Muhsen, S.; Al-Jahdali, H.; Hamid, Q. Role of transforming growth factor-β in airway remodeling in asthma. Am. J. Respir. Cell Mol. Biol. 2011, 44, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Vignola, A.M.; Chanez, P.; Chiappara, G.; Merendino, A.; Pace, E.; Rizzo, A.; la Rocca, A.M.; Bellia, V.; Bonsignore, G.; Bousquet, J. Transforming growth factor-beta expression in mucosal biopsies in asthma and chronic bronchitis. Am. J. Respir. Crit. Care Med. 1997, 156 Pt 1, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Raez-Villanueva, S.; Debnath, A.; Hardy, D.B.; Holloway, A.C. Prenatal nicotine exposure leads to decreased histone H3 lysine 9 (H3K9) methylation and increased p66shc expression in the neonatal pancreas. J. Dev. Orig. Health Dis. 2021, 13, 156–160. [Google Scholar] [CrossRef]
- Demenais, F.; Margaritte-Jeannin, P.; Barnes, K.C.; Cookson, W.O.C.; Altmüller, J.; Ang, W.; Barr, R.G.; Beaty, T.H.; Becker, A.B.; Beilby, J.; et al. Multiancestry association study identifies new asthma risk loci that colocalize with immune-cell enhancer marks. Nat. Genet. 2018, 50, 42–53. [Google Scholar] [CrossRef]
- Sudlow, C.; Gallacher, J.; Allen, N.; Beral, V.; Burton, P.; Danesh, J.; Downey, P.; Elliott, P.; Green, J.; Landray, M.; et al. UK Biobank: An Open Access Resource for Identifying the Causes of a Wide Range of Complex Diseases of Middle and Old Age. PLoS Med. 2015, 12, e1001779. [Google Scholar] [CrossRef]
- Owen, A.B. Infinitely imbalanced logistic regression. J. Mach. Learn. Res. 2007, 8, 761–773. [Google Scholar]
- Cepeda, M.S.; Boston, R.; Farrar, J.T.; Strom, B.L. Comparison of Logistic Regression versus Propensity Score When the Number of Events Is Low and There Are Multiple Confounders. Am. J. Epidemiol. 2003, 158, 280–287. [Google Scholar] [CrossRef]
- King, G.; Zeng, L. Logistic regression in rare events data. Political Anal. 2001, 9, 137–163. [Google Scholar] [CrossRef]
- He, H.B.; Garcia, E.A. Learning from Imbalanced Data. IEEE Trans. Knowl. Data Eng. 2009, 21, 1263–1284. [Google Scholar]
- Soda, P. A multi-objective optimisation approach for class imbalance learning. Pattern Recognit. 2011, 44, 1801–1810. [Google Scholar] [CrossRef]
- Chawla, N.V.; Bowyer, K.W.; Hall, L.O.; Kegelmeyer, W.P. SMOTE: Synthetic Minority Over-sampling Technique. J. Artif. Intell. Res. 2002, 16, 321–357. [Google Scholar] [CrossRef]
- Elkan, C. The Foundations of Cost-Sensitive Learning, International Joint Conference on Artificial Intelligence; Lawrence Erlbaum Associates Ltd.: Mahwah, NJ, USA, 2001; pp. 973–978. [Google Scholar]
- Lee, J.-E.; Kim, J.-H.; Hong, E.-J.; Yoo, H.S.; Nam, H.-Y.; Park, O. National Biobank of Korea: Quality control Programs of Collected-human Biospecimens. Osong Public Health Res. Perspect. 2012, 3, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.; Kim, Y.J.; Han, S.; Hwang, M.Y.; Shin, D.M.; Park, M.Y.; Lu, Y.; Yoon, K.; Jang, H.-M.; Kim, Y.K.; et al. The Korea Biobank Array: Design and Identification of Coding Variants Associated with Blood Biochemical Traits. Sci. Rep. 2019, 9, 1382. [Google Scholar] [CrossRef] [PubMed]
- Howie, B.N.; Donnelly, P.; Marchini, J. A Flexible and Accurate Genotype Imputation Method for the Next Generation of Genome-Wide Association Studies. PLOS Genet. 2009, 5, e1000529. [Google Scholar] [CrossRef]
- Solovieva, E.; Sakai, H. PSReliP: An integrated pipeline for analysis and visualization of population structure and relatedness based on genome-wide genetic variant data. BMC Bioinform. 2023, 24, 135. [Google Scholar] [CrossRef]
- Massy, W.F. Principal Components Regression in Exploratory Statistical Research. J. Am. Stat. Assoc. 1965, 60, 234–256. [Google Scholar] [CrossRef]
- Barrett, J.C.; Fry, B.; Maller, J.; Daly, M.J. Haploview: Analysis and visualization of LD and haplotype maps. Bioinformatics 2004, 21, 263–265. [Google Scholar] [CrossRef]
- Hu, Y.; Bien, S.A.; Nishimura, K.K.; Haessler, J.; Hodonsky, C.J.; Baldassari, A.R.; Highland, H.M.; Wang, Z.; Preuss, M.; Sitlani, C.M.; et al. Multi-ethnic genome-wide association analyses of white blood cell and platelet traits in the Population Architecture using Genomics and Epidemiology (PAGE) study. BMC Genom. 2021, 22, 432. [Google Scholar]
- Masoodi, T.A.; Banaganapalli, B.; Vaidyanathan, V.; Talluri, V.R.; Shaik, N.A. Computational Analysis of Breast Cancer GWAS Loci Identifies the Putative Deleterious Effect of STXBP4 and ZNF404 Gene Variants. J. Cell. Biochem. 2017, 118, 4296–4307. [Google Scholar] [CrossRef]
- Schaid, D.J.; Chen, W.; Larson, N.B. From genome-wide associations to candidate causal variants by statistical fine-mapping. Nat. Rev. Genet. 2018, 19, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, M.; Hakonarson, H. ANNOVAR: Functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010, 38, e164. [Google Scholar] [CrossRef] [PubMed]
- Kopanos, C.; Tsiolkas, V.; Kouris, A.; Chapple, C.E.; Aguilera, M.A.; Meyer, R.; Massouras, A. VarSome: The human genomic variant search engine. Bioinformatics 2019, 35, 1978–1980. [Google Scholar] [CrossRef] [PubMed]



| 9 | HEXA | p-Value | CAVAS | p-Value | KARE | p-Value | |||
|---|---|---|---|---|---|---|---|---|---|
| Case (n = 975) | Control (n = 57,459) | Case (n = 95) | Control (n = 2908) | Case (n = 112) | Control (n = 5308) | ||||
| SEX | <0.0001 | 0.8325 | 0.0048 | ||||||
| Male | 283 (29.0%) | 19,924 (34.7%) | 37 (38.9%) | 1164 (40.0%) | 39 (34.8%) | 2563 (48.3%) | |||
| Female | 692 (71.0%) | 37,535 (65.3%) | 58 (61.1%) | 1744 (60.0%) | 73 (65.2%) | 2745 (51.7%) | |||
| AGE (years) a | 55.4 ± 8.4 | 53.8 ± 8.0 | <0.0001 | 57.9 ±7.8 | 55.4 ± 7.8 | 0.0025 | 53.3 ± 7.9 | 51.5 ± 8.5 | 0.0026 |
| BMI (kg/m2) b | 24.3 ± 3.2 | 23.9 ± 2.9 | 0.0002 | 25.5 ±3.4 | 24.5 ± 3.0 | 0.0002 | 25.0 ± 3.5 | 24.6 ± 3.0 | 0.1536 |
| ALLER status c | <0.0001 | <0.0001 | <0.0001 | ||||||
| Non-ALLER | 727 (74.6%) | 53,642 (93.4%) | 74 (77.9%) | 2695 (92.7%) | 86 (76.8%) | 5015 (94.5%) | |||
| ALLER | 248 (25.4%) | 3817 (6.6%) | 21 (22.1%) | 213 (7.3%) | 26 (23.2%) | 293 (5.5%) | |||
| Smoking status d, e | 0.0003 | 0.6627 | 0.0129 | ||||||
| Non-smokers | 721 (73.9%) | 42,070 (73.2%) | 72 (75.8%) | 2123 (73.0%) | 71 (63.4%) | 3173 (59.8%) | |||
| Smokers | 254 (26.1%) | 15,389 (26.8%) | 23 (24.2%) | 785 (27.0%) | 41 (37.6%) | 2135 (40.2%) | |||
| CHR a | rsID b | Alt c | MAF d | OR | 95% CI | STAT | p-Value | Location | Nearest Gene |
|---|---|---|---|---|---|---|---|---|---|
| 8 | rs77079226 | C | 0.034 | 3.083 | 1.985–4.788 | 5.012 | 5.37 × 10–7 | Downstream transcript | TMEM74 |
| 10 | rs17153428 | A | 0.297 | 1.618 | 1.329–1.970 | 4.791 | 1.66 × 10–6 | Intron | RP11-383C5.4 |
| 22 | rs4823536 | G | 0.103 | 2.013 | 1.511–2.682 | 4.781 | 1.74 × 10–6 | Intergenic | MIR3201 |
| 2 | rs7563259 | A | 0.382 | 1.566 | 1.299–1.889 | 4.692 | 2.70 × 10–6 | Intergenic | RNU5E-7P |
| 10 | rs74743572 | G | 0.292 | 1.605 | 1.317–1.957 | 4.689 | 2.74 × 10–6 | Intron | RP11-383C5.4 |
| 12 | rs17837082 | A | 0.124 | 1.882 | 1.444–2.453 | 4.679 | 2.88 × 10–6 | Intron | RP11-121E16.1 |
| 2 | rs7602146 | G | 0.382 | 1.563 | 1.296–1.886 | 4.673 | 2.97 × 10–6 | Intergenic | RNU5E-7P |
| 5 | rs1469393 | T | 0.031 | 3.020 | 1.899–4.802 | 4.671 | 3.00 × 10–6 | Upstream transcript | FAM81B |
| 2 | rs6725714 | G | 0.382 | 1.563 | 1.296–1.885 | 4.670 | 3.01 × 10–6 | Intergenic | RNU5E-7P |
| 10 | rs79219793 | G | 0.334 | 1.576 | 1.301–1.910 | 4.652 | 3.29 × 10–6 | Intron | RP11-383C5.4 |
| 2 | rs10929394 | C | 0.383 | 1.557 | 1.291–1.877 | 4.630 | 3.67 × 10–6 | Intergenic | RNU5E-7P |
| 10 | rs57227860 | G | 0.464 | 0.644 | 0.533–0.778 | –4.568 | 4.93 × 10–6 | Upstream transcript | CDH23 |
| 19 | rs8104061 | A | 0.174 | 1.704 | 1.355–2.143 | 4.561 | 5.10 × 10–6 | Intron | ZNF8 |
| 9 | rs2765969 | T | 0.337 | 1.552 | 1.285–1.875 | 4.557 | 5.18 × 10–6 | Intergenic | RPL4P5 |
| 19 | rs112884174 | C | 0.174 | 1.703 | 1.355–2.141 | 4.557 | 5.19 × 10–6 | Intron | ZNF8 |
| 19 | rs10421600 | A | 0.174 | 1.702 | 1.354–2.140 | 4.552 | 5.30 × 10–6 | Intron | ZNF8 |
| 19 | rs10420807 | C | 0.174 | 1.702 | 1.354–2.140 | 4.552 | 5.30 × 10–6 | Intron | ZNF8 |
| 19 | rs7253055 | A | 0.174 | 1.700 | 1.352–2.138 | 4.543 | 5.56 × 10–6 | Intron | ZNF8 |
| 3 | rs144605956 | G | 0.016 | 5.094 | 2.522–10.29 | 4.539 | 5.64 × 10–6 | Intergenic | RP11-654C22.2 |
| 12 | rs74497942 | A | 0.131 | 1.836 | 1.412–2.387 | 4.538 | 5.68 × 10–6 | Intron | RP11-121E16.1 |
| 9 | rs2986259 | C | 0.350 | 1.543 | 1.278–1.862 | 4.508 | 6.55 × 10–6 | Intergenic | KDM4C |
| 11 | rs2292731 | T | 0.385 | 0.637 | 0.523–0.775 | –4.504 | 6.68 × 10–6 | Intergenic | MMP20 |
| 19 | rs57831195 | C | 0.070 | 2.130 | 1.532–2.961 | 4.498 | 6.85 × 10–6 | Intergenic | RFPL4A |
| 12 | rs61432389 | C | 0.131 | 1.820 | 1.402–2.362 | 4.495 | 6.97 × 10–6 | Intron | RP11-121E16.1 |
| 2 | rs6754791 | T | 0.381 | 1.536 | 1.273–1.853 | 4.486 | 7.26 × 10–6 | Intergenic | RNU5E-7P |
| 2 | rs6726202 | G | 0.381 | 1.536 | 1.273–1.852 | 4.484 | 7.32 × 10–6 | Intergenic | RNU5E-7P |
| 2 | rs4669006 | A | 0.374 | 1.538 | 1.274–1.857 | 4.480 | 7.46 × 10–6 | Intergenic | RNU5E-7P |
| 12 | rs17837077 | A | 0.130 | 1.823 | 1.402–2.371 | 4.477 | 7.56 × 10–6 | Intron | RP11-121E16.1 |
| 2 | rs4566338 | C | 0.372 | 1.536 | 1.273–1.855 | 4.469 | 7.86 × 10–6 | Intergenic | RNU5E-7P |
| 1 | rs34207591 | G | 0.090 | 1.990 | 1.471–2.692 | 4.466 | 7.97 × 10–6 | Upstream transcript | SETDB1 |
| 1 | rs71624514 | T | 0.090 | 1.990 | 1.471–2.692 | 4.466 | 7.97 × 10–6 | Upstream transcript | SETDB1 |
| 12 | rs7488720 | G | 0.131 | 1.815 | 1.397–2.359 | 4.461 | 8.17 × 10–6 | Intron | RP11-121E16.1 |
| 12 | rs75289086 | C | 0.133 | 1.805 | 1.392–2.341 | 4.452 | 8.52 × 10–6 | Intron | RP11-121E16.1 |
| 9 | rs2997570 | T | 0.335 | 1.537 | 1.272–1.858 | 4.446 | 8.76 × 10–6 | Intergenic | KDM4C |
| 12 | rs78145337 | C | 0.130 | 1.812 | 1.394–2.356 | 4.436 | 9.16 × 10–6 | Intron | RP11-121E16.1 |
| 10 | rs17153422 | C | 0.286 | 1.568 | 1.285–1.912 | 4.431 | 9.40 × 10–6 | Intron | RP11-383C5.3 |
| 9 | rs2997577 | A | 0.343 | 1.532 | 1.268–1.850 | 4.428 | 9.49 × 10–6 | Intergenic | KDM4C |
| 16 | rs12928443 | A | 0.187 | 1.677 | 1.334–2.109 | 4.426 | 9.58 × 10–6 | Intergenic | CTD-2535I10.1 |
| 12 | rs111721834 | C | 0.131 | 1.805 | 1.389–2.345 | 4.419 | 9.90 × 10–6 | Intron | RP11-121E16.1 |
| 9 | rs2997572 | T | 0.336 | 1.533 | 1.268–1.853 | 4.418 | 9.96 × 10–6 | Intergenic | KDM4C |
| CHR | SNP | CADD a | DANN b | Transcription Factors c | Rank Score d | Gene |
|---|---|---|---|---|---|---|
| 1 | rs139189121 | 11.940 | 0.792 | AR [92] | 5 | SETDB1 |
| 1 | rs75406390 | 12.280 | 0.836 | AR [92] | 5 | SETDB1 |
| 1 | rs59024312 | 8.982 | 0.833 | FOXA1 [93], GATA3 [94,95], GATA6 [96,97] | 4 | SETDB1 |
| 19 | rs8104061 | 0.751 | 0.718 | EZH2 [98] | 2b | ZNF8 |
| 19 | rs112884174 | 3.736 | 0.812 | EZH2 [98] | 4 | ZNF8 |
| 19 | rs260498 | 10.310 | 0.895 | POLR2A [99] | 4 | ZNF8 |
| 19 | rs11671486 | 11.590 | 0.886 | POLR2A [99] | 1f | ZNF8 |
| 11 | rs2245803 | 17.260 | 0.985 | RXRA [100,101], FOXP1 [102], NFIL3 [103,104], RARA [105], SMAD4 [106] | 3a | MMP20 |
| 16 | rs2532008 | 0.206 | 0.558 | RXRA [100,101], ZNF341 [107,108], PRDM10 [109,110] | 2b | ADCY9 |
| 16 | rs2532007 | 0.350 | 0.382 | ATF3 [111,112], BCL6 [113,114], EGR1 [115,116], ELF3 [117], EP300 [118,119], FOXA1 [93], FOXA2 [94,95], FOXA3 [94,120], HDAC2 [121,122], IRF2 [123], JUND [124,125], KAT8 [126,127], NFIL3 [103,104], NFYC [128], PRDM1 [129,130], RAD21 [131,132], RARA [105], RXRA [100,101], RXRB [133], SMAD4 [106], SP1 [134], THRB [135,136], YY1 [137,138] ZEB2 [139], and USF1 [140,141] | 2b | ADCY9 |
| 16 | rs384067 | 2.631 | 0.677 | CTCF [142,143], CREB1 [144,145] | 2b | ADCY9 |
| 9 | rs10814023 | 16.070 | 0.540 | POLR2A [99], CTCF [142,143] | 3a | DOCK8 |
| 9 | rs10758219 | 13.540 | 0.851 | POLR2A [99], CTCF [142,143] | 3a | DOCK8 |
| RegulomeDB Category | Category Description |
|---|---|
| Likely to affect binding and linked to expression of a gene target | |
| 1a | eQTL a + TF b binding + matched TF motif + matched DNase c footprint + DNase peak |
| 1b | eQTL + TF binding + any motif + DNase footprint + DNase peak |
| 1c | eQTL + TF binding + matched TF motif + DNase peak |
| 1d | eQTL + TF binding + any motif + DNase peak |
| 1e | eQTL + TF binding + matched TF motif |
| 1f | eQTL + TF binding/DNase peak |
| Likely to affect binding | |
| 2a | TF binding + matched TF motif + matched DNase footprint + DNase peak |
| 2b | TF binding + any motif + DNase footprint + DNase peak |
| 2c | TF binding + matched TF motif + DNase peak |
| Less likely to affect binding | |
| 3a | TF binding + any motif + DNase peak |
| 3b | TF binding + matched TF motif |
| Minimal binding evidence | |
| 4 | TF binding + DNase peak |
| 5 | TF binding or DNase peak |
| 6 | Motif hit |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cha, J.; Choi, S. Gene–Smoking Interaction Analysis for the Identification of Novel Asthma-Associated Genetic Factors. Int. J. Mol. Sci. 2023, 24, 12266. https://doi.org/10.3390/ijms241512266
Cha J, Choi S. Gene–Smoking Interaction Analysis for the Identification of Novel Asthma-Associated Genetic Factors. International Journal of Molecular Sciences. 2023; 24(15):12266. https://doi.org/10.3390/ijms241512266
Chicago/Turabian StyleCha, Junho, and Sungkyoung Choi. 2023. "Gene–Smoking Interaction Analysis for the Identification of Novel Asthma-Associated Genetic Factors" International Journal of Molecular Sciences 24, no. 15: 12266. https://doi.org/10.3390/ijms241512266
APA StyleCha, J., & Choi, S. (2023). Gene–Smoking Interaction Analysis for the Identification of Novel Asthma-Associated Genetic Factors. International Journal of Molecular Sciences, 24(15), 12266. https://doi.org/10.3390/ijms241512266






