Abstract
Uveal melanoma (UM) is rare, but it is the most common primary intraocular malignancy among adults. This review represents the molecular, genetic, and immunobiological mechanisms involved in UM carcinogenesis and progression, as well as data about the association of chromosomal changes, genetic mutations, selective proteins, and biochemical biomarkers with the clinical implications of UM. Genetic analysis has the potential to identify patients with a high risk of UM metastasis, enabling management that is more effective and allowing for the follow-up of patients. Advancements in molecular characterization of UM offer opportunities to develop targeted therapeutic strategies by focusing on relevant signaling pathways. Changes in miRNA expression could be useful in the diagnosis and prognosis of UM, due to unique miRNA profiles in melanoma cells or tissue and its association with metastasis. Although liver function tests do not provide enough data on the prognosis of UM, due to the high frequency of liver metastasis, liver function tests (LFTs) might be useful indicators; however, the absence of rising LFT values cannot lead to the exclusion of liver metastases. Molecular analysis of tumor tissue will allow us to identify patients with the added benefit of new therapeutic agents and provide a better insight into melanoma pathogenesis and its biological behavior.
1. Introduction
Uveal melanoma (UM) is rare, yet it is the most common primary intraocular malignancy among adults. It represents 80% of ocular melanomas and 3 to 5% of all melanoma cases [1,2]. The largest proportion of cases are derived from the choroid (90%) with other sites of tumor origin being the ciliary body (7%) and the iris (3%) [3]. UM incidence is from five to six cases per million in the United States and Europe and seven per million in Australia, respectively, which has remained unchanged over the past 30 years [1,4,5,6,7]. On the other hand, the incidence in Asia and Africa is only 0.2 to 0.3 cases per million per year [8]. Predisposing factors for UM development are age, fair skin, sensitivity to sunburn, light eye color, cutaneous, choroidal, or iris nevi. Other factors include a family history of cutaneous and uveal melanoma, dysplastic nevus syndrome, ocular or oculodermal melanocytosis—namely Nevus of Ota—and mutation of breast cancer 1 (BRCA1) associated protein 1 (BAP1) [3,6,9,10,11,12,13,14,15,16]. Risk factors for metastases also include large tumor size, epithelioid cell type, extra-scleral extension of primary tumor, chromosome 3, and loss of amplification of chromosome 8q [15,16,17].
Primary tumor treatment includes various forms and combinations of laser therapy, radiation therapy, and surgery which include local resection and enucleation of large tumors [17,18,19]. The aim of local treatment is to preserve useful vision and prevent metastatic spread; however, despite significant improvement in nearly 50% of all patients, metastatic disease will develop. When the primary tumor is diagnosed, metastases are found in less than 2% of patients, yet they can develop even over 30 years after initial diagnosis and treatment [20,21].
The 5-year survival rate of UM patients is between 50 and 70% and has remained unchanged over the past decades [2,5,9,22,23]. In the presence of metastatic disease, the median overall survival rate is 6–12 months [24,25], where 8% of patients survive for more than two years [16,17].
UM mainly spreads hematogenously, showing a tendency to metastasize in the liver (89%). Other sites include the lungs (29%) and bones (17%) [6,21,22,26]. For patients with metastatic disease, there is currently no standardized method of treatment. Potential treatment options for metastatic UM are chemotherapy, immunotherapy, and targeted therapies; however, these usually show poor outcomes [15,16,27]. Methods of treatment for patients with metastases solely in the liver include liver-directed therapies such as radiofrequency ablation, isolated hepatic perfusion (IHP), or percutan IHP [15,27]. Irrespective of the treatment options used in patients with liver metastases, the mortality rate after 2 years is nearly 90%, and approximately 1% of patients survive over 5 years. Patients with metastases in other organs or when the liver is not the primary site of metastases have a better survival rate [22,25,28]. Therefore, recognizing patients with a high risk for metastatic disease is of great value.
In recent years, several novel treatment modalities aimed at improving tumor control and reducing unfavorable outcomes have emerged [15,21,22,26,28,29,30]. Compared to other types of melanomas, metastatic UMs have different genetic characteristics, resulting in reduced survival when traditional melanoma treatment methods are applied. Changes in the approach to treatment occurred at the beginning of 2022 when the US Food and Drug Administration (FDA) approved the use of tebentafusp for the treatment of HLA-A*02:01-positive adult patients with metastatic or inoperable UM, a bispecific glycoprotein 100 (gp100) peptide-human leukocyte antigen (HLA)-directed CD3 T-cell activator. Even though this form of treatment has not yielded the anticipated results, it does offer hope for successful targeted therapy in the near future [29,30].
More recently, the molecular, genetic, and immunobiological mechanisms involved in carcinogenesis and the progression of UM are gaining more importance [31]. A better insight into the molecular and genetic profile of UM could aid in identifying reliable prognostic and diagnostic biomarkers, modifying existing as well as developing novel therapeutic approaches specifically designed for UM patients.
2. Genetic Basis of Uveal Melanoma
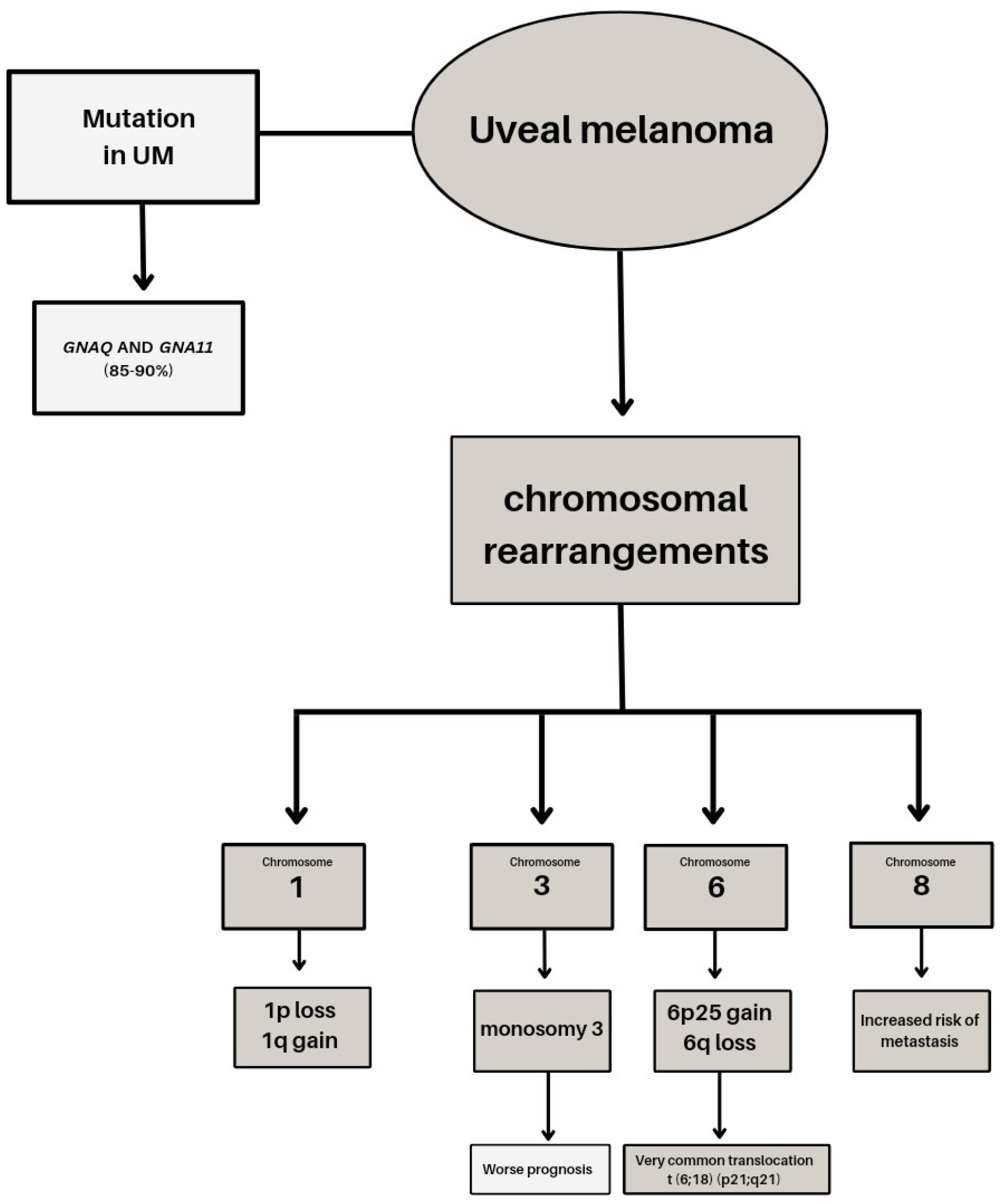
Genetic studies conducted over the years have provided valuable information about the changes observed in primary posterior uveal melanoma (UM) affecting the ciliary body and choroid. Metastases show disparate mutations than primary tumors [32]. Uveal melanomas are generally characterized by a low mutation burden and resistance to immunotherapy. UM and cutaneous melanoma are distinct genetic entities, differing not only in chromosomal changes but also in mutational signatures (Figure 1).
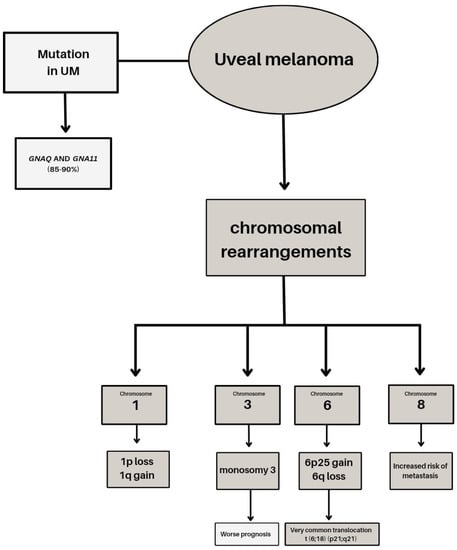
Figure 1.
Genetic basis of uveal melanoma.
Cytogenetic studies have highlighted the role of genetic factors such as chromosomal aberrations and mutations in predicting patient survival. Chromosomal rearrangements in UM, particularly on chromosomes 1, 3, 6, and 8, have been extensively studied. Monosomy 3 is a frequent early event occurring in 50–60% of tumors and is often accompanied by isochromosome 8q, which leads to abnormal segregation during mitosis and high levels of 8q gain tumors with monosomy 3 or gain of 8q are associated with poor prognosis and increased risk of metastasis [33]. Other chromosomal alterations, such as gain of 6p and loss of chromosomes 1p and 8, also have significant prognostic value [34,35]. Molecular genetic analyses using DNA sequencing techniques have identified several alterations implicated in metastatic UM. Mutations in GNAQ and GNA11 genes, primarily targeting codon 209, are found in up to 90% of posterior UM cases [36,37]. These mutations result in an amino acid change and are mutually exclusive. Less common mutations affecting codon 183 occur in the absence of codon 209 mutations [37]. GNAQ and GNA11 genes encode the heterotrimeric Gq-proteins involved in coupling transmembrane receptors to intracellular pathways. The activation of these genes is considered an early event in UM progression [38,39]. The BAP1 gene, located on chromosome 3, is a tumor-suppressor gene involved in cell growth and cancer pathogenesis. Inactivation or mutations of BAP1 are present in over 80% of metastatic UMs and are associated with decreased disease-free survival rates [40]. Mutations in the BAP1 gene lead to the premature termination of the BAP1 protein and can also impact the ubiquitin carboxyl-terminal hydrolase domain, thereby altering its deubiquitinase activity [40]. BAP1 interacts with various proteins, including the tumor suppressor gene BRCA1, and plays a crucial role in maintaining genome stability, regulating transcription, and responding to DNA damage [41]. Decreased disease-free survival rates are observed in tumors exhibiting monosomy 3 and BAP1 mutations [42]. Detecting a germline mutation of BAP1 not only indicates an increased risk of uveal melanoma (UM), but also suggests a potential association with other types of tumors. In such cases, a more comprehensive approach may be considered, involving genetic counseling and screening of family members [43]. Approximately 10–20% of uveal melanoma (UM) cases exhibit mutations in the splicing factor 3B subunit 1 (SF3B1) gene [44]. The SF3B1 gene is located at chromosome 2q33 and is responsible for encoding a subunit of the spliceosome, a large complex involved in processing precursor mRNA. SF3B1 plays a crucial role in maintaining precise splicing by retaining pre-mRNA and defining the splicing site [45]. EIF1AX, located on chromosome 10 encodes for the X-linked Eukaryotic Translation Initiation Factor 1A protein (Eif1A), which plays a crucial role in regulating the initiation of protein translation. Mutations in EIF1AX can result in the mis-selection of start sites, leading to suppressed translation of canonical transcripts or potentially upregulating oncogenes [46]. The presence of mutant EIF1AX has been linked to abnormal translation processes [47]. The telomerase reverse transcriptase (TERT) gene, located on chromosome 5p15, has been extensively implicated in various cancers [48]. However, UM exhibits a low mutation frequency of only 1% [49]. TERT plays a multifunctional role that encompasses the maintenance of telomere length, cell-cycle control, and DNA damage response mechanisms [50].
The MBD4 (Methyl-CpG Binding Domain 4, DNA Glycosylase) gene encodes an enzyme that plays a crucial role in DNA repair and acts as a tumor-suppressor gene. When MBD4 is inactivated, it results in the accumulation of a specific type of mutation, like those observed during the aging process. Germline mutations in MBD4 are associated with a relative risk: individuals carrying this mutation have a 10 times higher risk of developing uveal melanoma compared to those without the mutation [51].
Epigenetic mechanisms play a significant role in UM, involving processes such as DNA methylation, chromatin remodeling, histone modification, and non-coding RNAs (miRNAs). Methylation events in UM can affect tumor suppressor genes, including RAS association domain family 1 isoform A (RASSF1A) and cyclin-dependent kinase inhibitor 2A (p16INK4a), which have been extensively studied in this context [52].
BAP1 methylation has been recognized as a significant prognostic indicator for metastasis in UM. Robertson et al. identified four distinct UM subtypes based on the status of chromosome 3 and BAP1 methylation: two groups associated with poor prognosis characterized by monosomy 3, and two groups with a better prognosis characterized by disomy 3. The DNA methylation pattern of BAP1 in the first group differed from that observed in the second group [53]. In their study, Jurmeister et al. investigated the potential of DNA methylation profiling to establish a relationship between melanomas and their respective primary sites. Their findings revealed that only uveal melanomas exhibited distinct global DNA methylation profiles, displaying unique epigenetic signatures. Consequently, DNA methylation analysis allows for differentiation between uveal melanomas and melanomas originating from other primary sites [54].
Uveal melanomas can be categorized into prognostic groups by evaluating the expression of a specific set of genes. Initially consisting of 26 genes, this set was later refined to 15 genes. The expression pattern of these genes enables the classification of UM into low-risk (class 1) and high-risk (class 2) groups [55]. Based on the provided data set, patients with class 1 uveal melanoma (UM) exhibit a survival rate of approximately 95% at around 7 years, whereas class 2 patients have a significantly lower survival rate of approximately 30%. Further stratification within these subgroups can be achieved by considering the expression of preferentially expressed antigens in melanoma (PRAME) [56]. PRAME status has emerged as an independent prognostic biomarker for UM, specifically identifying a higher risk of metastasis in patients with Class 1 tumors. As a result, the classification of UM using genome-wide expression profiling has been revised to incorporate PRAME status. Research has indicated that when combined with a 12-gene expression panel, PRAME expression can predict a five-year metastatic rate of 0% in Class 1/PRAME− tumors, 38% in Class 1/PRAME+ tumors, and 71% in Class 2 tumors. Additionally, PRAME expression has been found to be positively associated with larger tumor diameter, and SF3B1 mutations, as well as gains of 1q, 6p, 8q, and 9q, and losses of 6q and 11q [56].
There has been a notable disparity in the number of genetic studies conducted on iris melanomas compared to posterior uveal melanomas. Iris melanomas share certain genetic alterations with posterior uveal melanomas, such as deletions of 1p and alterations in chromosome 6 (both p and q). Evidence suggests that iris melanomas also exhibit abnormalities in other chromosomes, which are less commonly affected in posterior uveal melanomas. These include changes in chromosome 9p, which is more frequently associated with cutaneous melanoma, as well as rearrangements involving chromosome 18 [57]. Studies have indicated that iris melanomas exhibit mutations shared with both posterior UMs and cutaneous melanomas (CM) [58]. Specifically, mutations in genes GNAQ/GNA11 and BRAF have been identified in iris melanomas [58,59] with a surprisingly high frequency of EIF1AX mutations [60]. An observation revealed a higher likelihood of tumor recurrence being associated with BRAF mutations [59].
Genetic studies have significantly contributed to enhancing our understanding of UM by identifying chromosomal changes and genetic mutations that can serve as valuable biomarkers for prognostic prediction. The advancements in molecular characterization of UM offer opportunities to develop targeted therapeutic strategies by focusing on relevant signaling pathways. Genetic analysis has the potential to identify patients at a high risk of metastasis, enabling more effective management and follow-up of patients.
3. miRNA and Uveal Melanoma
miRNAs are crucial factors that can alter the expression of genes in the regulatory mechanisms of carcinogenic processes. miRNAs are small molecules (17 - 22-nucleotides) that participate in the regulation of 60% of all protein-encoding genes and more than 400 different mRNAs [61,62]. In the development of UM, miRNAs serve as tumor suppressors or as oncogenes. Table 1 shows the literature data that includes the association between miRNAs, uveal melanoma, and their target genes. The majority of miRNAs are inhibitors of tumor development, because they participate in oncogene downregulation.
Several studies have investigated the role of miR-34a in UM. Yan et al. [63] reported that the transfection of miR-34a into UM cells leads to a decrease in cell growth and migration. This affects the downregulation of C-Met protein expression, a protein that is essential for cellular processes, and therefore indicates that miR-34a plays a role as a tumor suppressor. Furthermore, a recent study on human UM cell lines investigated the influence of miR-34a on leucine-rich repeat-containing G protein-coupled receptor 4 (LGR4), a novel target of miR-34a first discovered in retinal pigment epithelial cells. The study showed that LGR4 was upregulated in UM cells and that miRNA-34a transfection leads to reduced expression of LGR4 and causes inhibition of migration and invasion of the affected cell [64]. Previously, Dond and Lou found that two miRNAs from the same miR-34 family, miR-34b and miR-34c have also been associated with UM as suppressors of cell proliferation and migration by multiple targets. It was shown that miR-34b and miR34c inhibit cell growth and migration by blocking the cell cycle at the G1 phase and by decreasing tyrosine-kinase receptor (c-Met), cyclin-dependent kinase (CDK) 4, and CDK6 levels [65]. Downregulation of c-Met in UM cell lines was also the result of the overexpression of miR-122 and miR-144, which leads to impaired cell proliferation and migration [66]. Analyses of the miRNA in UM-cell lines and melanocytes showed significantly lower expression of miR-137 in UM cells, while its transfection into the same cells decreased cell growth and affected the downregulation of CDK6 and oncogene microphthalmia-associated transcription factor (MITF) [67]. MITF was also downregulated by miR-182 [68].
Additionally, miR-124a also acts as a tumor suppressor of UM, as it inhibits cell growth, migration, and invasion by affecting multiple targets such as CDK4, CDK6, cyclin D2 and enhancer of zeste homolog 2 (EZH2) [69]. It is known that CDK4 and cyclin D2 are essential for cell cycle G1 phase [70,71], while EZH2 is a protein that serves as a histone lysine methyltransferase and was previously found to be associated with the progression of skin melanoma [72]. Li et al. in a recent study reported lower expression of miR-145 and miR-205 in clinical samples taken from patients with UM than in normal tissue samples [73]. Furthermore, overexpression of those two miRNAs decreases the level of cell division control protein 42 (CDC42), which is associated with many cancers in humans, such as cervical squamous cell carcinoma, cutaneous melanoma, sarcoma, uterine carcinosarcoma, and others [74]. The list of miRNAs that act as tumor suppressors in UM additionally includes miR-9 [75], miR-17-3p [76], miR-142-3p [77], miR-216a-5p [78], and miR-224-5p [79]. miR-216a-5p inhibits hexokinase-2 (HK2) expression and therefore reduces glycolysis, glucose uptake, ATP production, extracellular acidification rate (ECAR), and an increase in oxygen consumption rate (OCR) [78].
In addition to acting as tumor suppressors, miRNAs in UM can also play the role of oncogenes and therefore promote disease progression and initiation. It was noted in the literature that miRNA-21, which could be a potential biomarker as well as a therapeutic target in colon cancer, was the most investigated [80]. Overexpression of miR-21 in UM cell lines promotes cell proliferation, inhibits the expression of the p53 gene, and increases LIM and SH3 protein 1 (LASP1) [81].
A study conducted on tumor samples obtained from patients with choroidal UM reported upregulation of miR-155 in both melanoma cells and tissues [82]. Additionally, transfection of miR-155 into the cells causes an increase in cell growth and inhibits the Nedd4-family interacting protein 1 (NDFIP1). This suggests that miR-155 might serve as a potential therapeutic target in UM [82]. Cell cycle progression in UM was also induced by upregulation of miR-181, especially miR-181b which inhibits expression of CTD small phosphatase-like tumor suppressor (CTDSPL), an activator of the downstream effector E2F, which leads to the disruption of cell cycle control [83]. Previous studies have reported that expression of miR-367 and miR-454 decreases levels of phosphatase and the tensin homolog (PTEN) [84,85]. PTEN is involved in the phosphatidylinositol-3-kinase (PI3K) and serine/threonine-protein kinase (AKT) pathways. Low expression of PTEN is associated with the progression of disease and metastasis formation [86]. A recent study reported higher expression of miR-652 in UM cell lines in comparison to melanocyte cells and non-tumor tissues [87]. The target gene of miR-652 is oncogene homeobox A9 (HOXA9), and downregulation of miR-652 increased HOXA9 expression in cells [87].
A study conducted by Souri et al. compared the expressions of HLA and miRNA in the same tumors using mRNA expression with four unlined HLA Class I probes [88]. The study showed that one cluster of miRNAs was positively matched with HLA Class I and infiltrating leukocytes, while the other showed the opposite effect. The study also confirmed the relationship between both miRNA expression patterns and chromosome 3 (BAP1) status in UM. This finding could be the starting point to consider miRNAs as regulators of inflammation in UM, regulated by BAP1 [88].

Table 1.
The role of miRNAs found to be associated with uveal melanoma (UM) in humans.
Table 1.
The role of miRNAs found to be associated with uveal melanoma (UM) in humans.
| miRNAs | Sample Type | Role | Regulation Action | Target | Reference |
|---|---|---|---|---|---|
| miR-9 | cells with highly and poorly invasive potential | tumor suppressor | suppresses cell migration and invasion | downregulation NF-κB1 | [75] |
| miR-17-3p | UM patients tissues and cell lines | tumor suppressor | inhibiting cell proliferation, migration and invasion | decreased MDM2 expression | [76] |
| miR-20a | cells and melanocytes, and tumor tissue samples for patients | oncogenic | increases cell growth, migration and invasion activities | not validated | [89] |
| miR-21 | cell lines and tissue samples | oncogenic | promoted proliferation, migration, and invasion | inhibited expression p53 | [81] |
| miR-34a | cells lines and melanocytes | tumor suppressor | inhibiting cell proliferation, migration and invasion | downregulation c-Met; decreased LGR4 | [63,64] |
| miR-34b/c | cells and melanocytes from patients, the uveal stromal tissues | tumor suppressor | reduction in cell growth and migration | downregulation c-Met | [65] |
| miR-122 | cell lines and patients’ tissues | tumor suppressor | impaired cell proliferation and migration | reduced expression ADAM10 and c-Met | [66] |
| miR-124a | cell culture and tumor specimens | tumor suppressor | inhibited cell growth, migration and invasion | downregulation CDK4, CDK6, cyclin D2 and EZH2 | [69] |
| miR-137 | cells lines from patients | tumor suppressor | decrease cell growth | downregulated MITF and CDK6 | [67] |
| miR-142-3p | cells and uveal melanocytes from patients | tumor suppressor | inhibited cell proliferation, migration and invasiveness | reduced CDC25C, TGFβR1, GNAQ, WASL, and RAC1 | [77] |
| miR-144 | cell lines and patients tissues | tumor suppressor | impaired cell proliferation and migration | reduced expression ADAM10 and c-Met | [66] |
| miR-145 | cells with highly and low invasive potential and patients’ tissues | tumor suppressor | reduce proliferation, migration and invasion | downregulation CDC42 | [73] |
| miR-155 | cell culture and tumor specimens | oncogenic | increase cell growth and invasion | inhibited NDFIP1 | [82] |
| miR-181 | cell lines | oncogenic | promoted cell cycle progression | inhibited CTDSPL | [83] |
| miR-182 | cells lines from patients | tumor suppressor | decrease cell growth, migration, and invasiveness | downregulation MITF, BCL2 and cyclin D2 | [68] |
| miR-205 | cells with highly and low invasive potential and patients’ tissues | tumor suppressor | reduce proliferation, migration and invasion | downregulation CDC42 | [73] |
| miR-216a-5p | cell lines and human embryonic kidney cell line | tumor suppressor | reduce cell proliferation | inhibit HK2 expression | [78] |
| miR-222 | cell lines | oncogenic | increases proliferation and migration | decreased PI3K, Akt, MMP-9 | [87] |
| miR-224-5p | cell lines and human embryonic kidney cell line and patients’ tissues | tumor suppressor | inhibited capacities of proliferation, invasion and migration | decreased PIK3R3 and AKT3 | [79] |
| miR-296-3-p | choroidal tissues | tumor suppressor | inhibiting cell proliferation, cell cycle progression, migration | targeting of MMP-2 and MMP-9 in combination with FOXCUT | [90] |
| miR-367 | tissue specimens, cell culture | oncogenic | promote cell proliferation and migration | reduced PTEN | [84] |
| miR-454 | tumor samples from patients | oncogenic | promote cell proliferation, colony formation, invasion and induction | reduced PTEN | [85] |
| miR-652 | cell lines and tissues samples from patients | oncogenic | increases proliferation and migration | decrease HOXA9 | [91] |
NF-κB1: nuclear factor kappa B cells 1; MDM2: murine double minute clone 2 oncoprotein; LGR4: leucine-rich repeat-containing G protein-coupled receptor 4; ADAM10: disintegrin metalloproteinase 10; c-Met: tyrosine-kinase receptor; CDK4: cyclin dependent kinase 4; CDK6: cyclin dependent kinase 6; cyclin D2: protein coding gene; EZH2: enhancer of zeste homolog 2; MITF: microphthalmia associated transcription factor; CDC25C: cell division cycle 25 homolog c; TGFβR1: transforming growth factor beta receptor 1; GNAQ: guanine nucleotide-binding protein alpha-Q; WASL: Wiskott–Aldrich syndrome like; RAC1: ras-related c3 botulinum toxin substrate 1; CDC42: cell division control protein 42; HK2: hexokinase-2; PI3K: phosphatidylinositol-3-kinase; AKT: serine/threonine-protein kinases; PIK3R3: subunit of phosphatidylinositol 3-kinase; p53: tumor protein; NDFIP1: nedd4-family interacting protein 1; BCL2: B-cell lymphoma 2; MMP-9: matrix metallopeptidase-9; PTEN: phosphatase and tensin homolog; HOXA9: Homeobox A9; FOXCUT: FOXC1 promoter upstream transcript.
miRNAs as Biomarkers
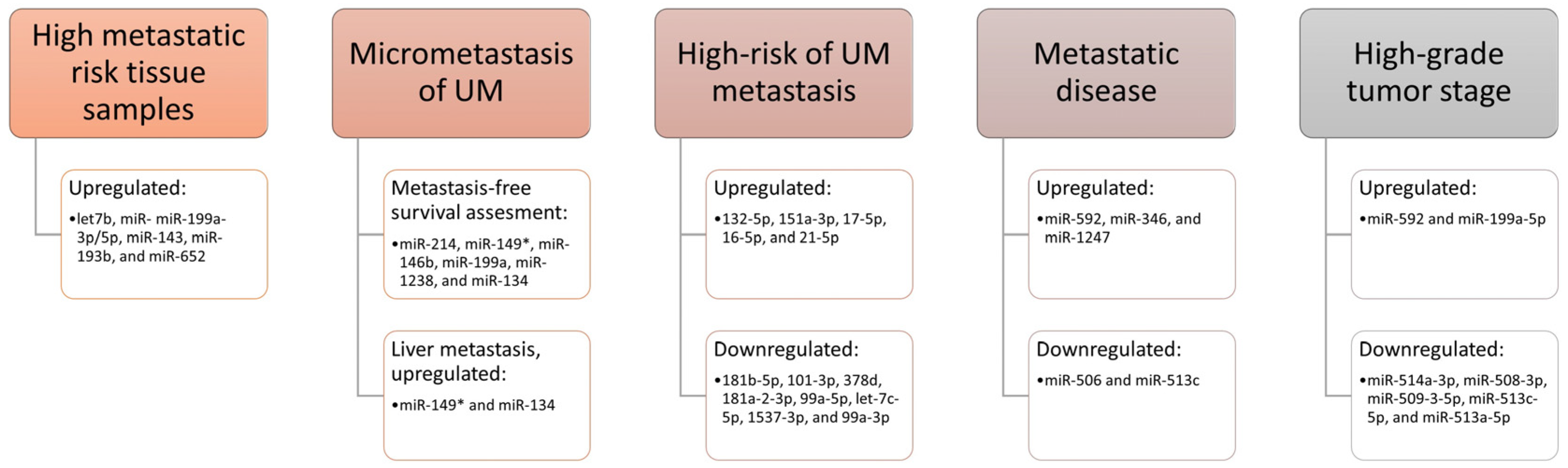
A study conducted on fresh frozen UM tissue reported the association between miRNA expression and melanoma metastasis [92]. The authors used a multiplexed microarray-based platform to identify miRNA profiles in two classes of tumor tissues, low (class 1) and high metastatic risk (class 2). They found six miRNAs (let7b, miR-miR-199a-3p/5p, miR-143, miR-193b, and miR-652) to be upregulated in high metastatic risk tissue samples, while two of them, let-7b and miR-199a, had the strongest association, which makes them more accurate predictors of metastasis [92].
Venkatesan and co-authors [93] studied the involvement of miRNAs in the micrometastasis of UM. They found that miR-214, miR-149*, miR-146b, miR-199a, miR-1238, and miR-134 may be used to assess metastasis-free survival in patients with UM, while miR-149* and miR-134 have a significant association with liver metastasis. A recent study used a next-generation sequencing approach and identified thirteen miRNAs with expressions that were different in the high-risk group of UM in comparison to the low- and intermediate-risk groups [94]. Five of them (132-5p, 151a-3p, 17-5p, 16-5p, and 21-5p) were upregulated, while eight (181b-5p, 101-3p, 378d, 181a-2-3p, 99a-5p, let-7c-5p, 1537-3p, and 99a-3p) were downregulated in the group at high risk for UM-metastasis, indicating that some miRNAs have a potential role in future treatment. Furthermore, studies conducted on patients with primary or metastatic UM tumors reported increased expression of miR-592, miR-346, and miR-1247, and decreased expression of miR-506 and miR-513c in patients with metastatic disease [95]. These findings showed that selected miRNAs may serve as early biomarkers of disease progression. miRNAs as a risk assessment for the development of metastases are summarized in Figure 2.
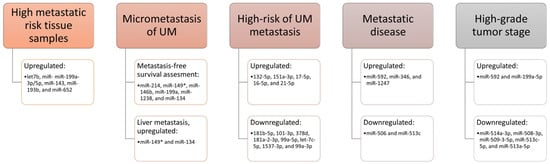
Figure 2.
miRNA as a risk assessment for the development of metastases (studies with different designs) [92,93,94,95,96].
In addition, some authors used the data on miRNA expression contained in the public database ‘The Cancer Genome Atlas’ (TCGA) to identify miRNAs that can be used as biomarkers in clinical practice. They found that miR-514a-3p, miR-508-3p, miR-509-3-5p, miR-513c-5p, and miR-513a-5p were downregulated, while miR-592 and miR-199a-5p were upregulated in the high-grade tumor stage compared to the low-grade tumor stage and in the deceased group vs. the alive group regarding vital status [96]. On the other hand, Xin and co-authors used a linear prognostic model of nine miRNAs (miR-195, miR-224, miR-365a, miR-365b, miR-452, miR-4709, miR-7702, miR-513c, and miR-873) to divide patients with UM into a high- and a low-risk group [97]. The overall survival was shorter in the high-risk group than in the low-risk group. They identified 418 genes as target genes of these 9 miRNAs, which were involved in the activity of protein binding and phosphorylation and the transforming growth factor beta (TGF-β) pathway [97].
The association of miRNA expression, metastasis, and survival in patients with UM using the TCGA dataset was also investigated by Vashishtha et al. [98]. They reported 22 miRNAs, 3 upregulated (miR-199a-5p, miR-708-5p, and miR-592), and 9 downregulated (miR-508-3p, miR-509-3p, miR-508-5p, miR-514a-3p, miR-506-3p, miR-509-3-5p, miR-513c-5p, miR-513a-5p, and miR-513b-5p) in patients with vs. those without metastatic UM. The overall survival in patients with UM was associated with 15 miRNAs, 11 miRNAs with a hazard ratio <0.10, and 4 miRNAs with a hazard ratio >10. Furthermore, tumor specimens obtained from formalin-fixed paraffin-embedded tissue of 52 cutaneous melanomas and 41 UMs had lower expression of miR-15a, miR-185, and miR-221 and higher levels of IL-10Rα than normal skin specimens [99]. Expression of IL-10Rα in melanoma cells is regulated by miR-15a, miR-185, and miR-211, and the knockdown of these miRNAs supported proliferation in the cell lines.
Circulating miRNAs were investigated as diagnostic and prognostic biomarkers due to their stability in the serum, high specificity and selectivity, and non-invasive sampling method. Triozzi et al. [100] investigated miRNA expression in the plasma of UM patients during 33 weeks of therapy with dacarbazine and interferon-alfa-2b, and in follow-up patients after 6 months from their last therapy. They observed downregulation of miR-126 and miR-199a and upregulation of miR-16 and miR-106a after therapy only with interferon-alfa-2b. The limitation of this study was the lack of risk data for metastasis during follow-up. The same group of authors also investigated miRNA profiles in the plasma of 10 patients with monosomy-3 and 10 without it [101]. They found that 15 miRNAs had different expression between patients with monosomy-3 and controls, 11 miRNAs (miR-191, miR-93, miR-221, miR-342-3p, miR-19b, miR-199a-5p, miR-25, miR-27a, miR-23a, miR-15b, and miR-223) were upregulated, and 4 (miR-1227, miR-663, miR-654-5p, and miR-1238) were downregulated. Furthermore, in this study, they investigated differences in miRNA expression in patients with and without metastatic disease during follow-up. Metastatic patients had seven downregulated miRNAs (miR-509-3-5p, miR-508-3p, miR-506, miR-513a-5p, miR-509-3p, miR-513b, miR-935) and one upregulated miRNA (miR-624) when compared to non-metastatic patients. Stark et al. analyzed [102] miRNA expression in the serum of participants with uveal nevi and patients with localized and metastatic UM. The expression of six miRNAs (miR-16, miR-145, miR-146a, miR-204, miR-211, and miR-363-3p) was significantly different among those three groups, whereas miR-211 was shown to be a discriminator between metastatic disease and localized UM. Downregulation of six miRNAs (miR-19a, miR-30d, miR-127, miR-451, miR-518f, miR-1274b) and upregulation of two miRNAs (miRNA-146a, miR-523) were found in the serum of patients with UM vs. controls [103]. After singular validation of miR-146a and miR-523 as potential markers for disease, only miR-146a was overexpressed in patients’ serum and UM cells compared to controls. On the other hand, patients with UM had higher expression of miR-20a, 125b, 146a, 155, 181a, and 223 in plasma samples than controls. Also, these miRNAs increased with metastasis, with the exception of miR-181a, which decreased.
In summary, for a better understanding of the role in UM, using miRNAs as predictive markers or potential biomarkers requires further studies on a larger number of samples/patients, adequate bioinformatics tools for predictive analysis, clinical validation of findings such as the comparison of early and late metastasis. However, miRNAs have a future in the diagnosis and prognosis of UM, due to unique miRNA profiles in melanoma cells or tissue and their association with metastasis.
4. Uveal Melanoma and DNA Methylation
DNA methylation, one of the most prevalent epigenetic forms, controls the expression of genes in two different ways: either by inhibiting the activity of transcriptional proteins, which prevents their binding to the gene, or by methylated DNA binding methyl-CpG-binding domain proteins, resulting in a buildup of inactive chromatin [104].
According to a study by Bakhoum, the levels of BAP1 protein, BAP1 mutations, and BAP1 genomic copy loss are all correlated with the methylation of BAP1 at a single genomic locus. They concluded that this offers helpful prognostic information, even in tumors where whole-exome sequencing failed to identify any BAP1 mutations, and that UM metastasis is associated with BAP1 deletion in the primary tumor [105]. In the bone, skin, and liver, UM metastasis was found to be caused by aberrant methylation of BAP1 and SF3B1. The study demonstrated a significant degree of patient variability and that epigenetic alterations in metastasized UM lead to the altering of many tumor-related genes [106]. Genes linked to early metastasis and a poor prognosis were discovered using integrated differential DNA methylation and gene expression study samples. While TMEM200C, RGS10, ADAM12, and PAM are hypomethylated and candidate oncogenes connected to early metastasis, RNF13, ZNF217, and HYAL1 are hypermethylated and candidate tumor suppressors [107]. As a result of their down-regulated expression, the tumor suppressor genes encoding p16INK4a (cyclin-dependent kinase inhibitor 2A), RASSF1A (RAS association domain family 1 isoform A), and p16INK4b (RAS association domain family 1 isoform B) may play a significant role in tumorigenesis. The tumor suppressor gene is less frequently hypomethylated than it is hypermethylated. However, it was discovered that the DSS1 gene and preferentially expressed antigen in melanoma (PRAME), which is a reliable indicator of metastasis in uveal melanoma, are overexpressed in UM as a result of hypomethylation [52].
A recent study using UM tissue from 68 UM patients in Slovakia revealed that high-risk tumors had a total of 7810 CpGs that were hypomethylated and 16,588 that were hypermethylated. Three genes with hypomethylation or upregulation, HTR2B (FC 191.4), AHNAK2 (FC 12.6), and CALHM2 (FC 7.8), as well as six genes with hypermethylation or downregulation, SLC25A38 (FC-4.6), EDNRB (FC-4.7), TLR1 (FC-8.6), RNF43 (FC-10.8), IL12RB2 (FC-18.1), and MEGF10 (FC-25) were selected for validation and the number of CpGs inversely correlated with gene expression [108].
All of the studies mentioned above demonstrated the important part that DNA methylation has in UM development, but more research is needed to determine the diagnostic utility and potential future applications of this sort of epigenetic analysis.
5. Uveal Melanoma Biomarkers in Body Fluids
Many studies have implicated the interpretation of different serum biomarkers in uveal melanoma. This included the determination of liver function tests (LFT), tumor markers, growth factors, and other proteins. The results showed high levels of heterogeneity.
5.1. Liver Function Tests as Uveal Melanoma Biomarkers
Due to very frequent metastases in the liver, LFT measurements are routine diagnostics during check-ups of patients with UM. The diagnostic and prognostic value of LFTs showed some contradictions among different studies. A study conducted on 88 Canadian patients from Quebec who developed metastases detected during a semiannual follow-up showed that overall LFT sensitivity ranged between 12.5% and 58.0%, while the positive predictive value (PPV) ranged between 9.4% and 38.6%. This implies that LFT might be useless in the detection of early metastasis [109]. A total of 95 Finnish patients with systemic metastases participated in an annual review that included LFTs (AST, ALT, AP, and LDH), a chest radiogram, and liver imaging. The results showed that AP in serum was higher than the upper reference limit in 30%, LDH in 63%, AST in 36%, and ALT in 36% of patients [110]. A US study performed on 505 medical records of patients with UM selected 76 patients with liver metastasis according to a study protocol. In 69% of patients, at least one of the LFTs was increased: 55% showed an increased value of AST, ALT, or both, 43% AP, and 33% had an increased concentration of bilirubin [111]. A prospective study on 102 French patients with UM did not show sufficient accuracy of LFT screening in the detection of liver metastases [112].
We can conclude that liver function tests do not provide enough data for the prognosis of uveal melanoma. Indeed, due to the high frequency of liver metastases, LFTs might be useful indicators, but the absence of rising LFT values cannot lead to the exclusion of liver metastases. Therefore, all alternative and more selective and accurate diagnostic methods must be performed during a patient’s follow-up.
5.2. UM-Serum Biomarkers
Investigation of serum tumor markers in UM mostly reflects their capacity to improve the prognosis of uveal UM metastases. S-100β, melanoma-inhibitory activity (MIA), and osteopontin (OPN) serum concentrations, as well as concentrations of some growth factors and cytokines, might serve as important prognostic tools in UM.
A study conducted on serum samples from the Ocular Oncology Serum Bank at Hadassah University Hospital showed that the concentrations of osteopontin OPN, MIA, and S-100β significantly correlate with liver metastases of UM. The combination of these markers proved to be very sensitive and indicative of the detection of liver metastases [113]. The same group of authors conducted a study that included 43 patients without UM disease (DF) for at least 10 years, 32 patients with metastatic UM, and 53 healthy subjects. Their findings show that concentrations of the tumor markers OPN, S-100β, MIA, and tissue-specific polypeptide antigen (TPS) increased before confirmation of metastases by imaging techniques [114]. This indicates that routine measurements of these biomarkers in serum could be important for the early detection of liver metastases and an early therapeutic approach. OPN and MIA were found to be significant tumor markers in German patients with metastatic UM, as both markers were markedly higher in patients with metastases than in those without metastases [115]. A case-control study performed on patients from the Netherlands showed that mean serum concentrations of S-100B and MIA were significantly higher in patients with metastases compared to melanoma patients without metastases [116]. Early detection of tumor progression was also studied in serum exosomes as a carrier of different tumor progression biomarkers. A study involving 20 Polish patients with primary or metastatic UM and 20 healthy donors showed that the concentrations of several molecules associated with inflammation, such as interferon-gamma, IL-2, IL-22, IL-12, Pentraxin-3, TNFSF13B, and TNFSF8, were significantly higher in exosomes from subjects with metastatic UM than in exosomes from healthy donors [117]. Serum concentrations of IGF-1 in patients from Israel indicated that this biomarker could be useful for predicting UM metastases, as IGF-1 was significantly lower in subjects with metastatic tumors compared to subjects who recovered from the disease 10 or more years ago (or even more) than in control healthy subjects [118]. A study of 45 patients with immune-related adverse events (irAEs) in patients with metastatic UM treated with an immune checkpoint inhibitor (ICI) showed that serum CRP and IL-6 concentrations may be useful markers for the early detection of irAEs [119].
Through a comparison of studies that investigate LFTs and some tumor biomarkers in serum, we can conclude that LFTs have an indicative impact on the prognosis of metastatic events in UM patients. Furthermore, additional biomarkers with higher sensitivity must be determined during patient follow-ups. OPN, MIA, and S-100β seem to be the markers of choice, but some other serum biomarkers like them need to be further investigated because the etiology of UM is still unclear. This also includes prognostic biomarkers that can be used in the diagnosis and prognosis of disease progression.
5.3. Ocular Fluid Proteins as Biomarkers in Uveal Melanoma
Aqueous humor (liquid fluid) and vitreous humor (gel-like fluid) are fluids localized in front of and behind the lens, respectively [120]. The composition of aqueous humor includes organic and inorganic ions, glutathione, carbohydrates, amino acids, carbon dioxide, oxygen, and water [121]. Depending on the various eye disorders, the testing of several types of cytokines, chemokines, growth factors, and other proteins can play an important role in the diagnosis of various eye diseases. Those proteins might serve as biomarkers in the diagnosis, prognosis, and treatment of uveal melanoma (UM). Several studies investigated the difference in protein composition of the aqueous humor between patients who suffered from uveal melanoma and subjects with different benign ocular disorders.
Expression of many angiogenic, chemotactic, and inflammatory cytokines was significantly different between Chinese patients with UM and control subjects with cataracts. This included interleukins IL-6 and IL-8, interferon-inducible protein-10 (IP-10), placental growth factor-1 (PLGF-1), monocyte chemoattractant protein-1 (MCP-1), nerve growth factor-b (NGF-β), epidermal GF (EGF), b-fibroblast GF (b-FGF), vascular endothelial GF-A (VEGF-A), and regulated upon activation, normal T cell expression, and secreted chemokine (RANTES) [122]. Similar studies were performed on 35 Italian subjects with UM and 35 controls with cataracts. The results showed higher concentrations of IL-6, IL-8, EGF, bFGF, macrophage inhibitory factor (MIF), and MCP-1 in the aqueous humor of patients with UM compared to subjects with cataracts [123].
The protein composition of patients with UM was also investigated in vitreous humor. Comparison of Dutch patients with UM and controls revealed results that demonstrated higher vitreous fluid concentrations of IL-6, IL-8, IP-10, MCP-1, macrophage inflammatory protein 1α (MIP-1α), MIP-1β, TNF-α, and RANTES. It was also shown that IL-6, IL-8, IP-10, MCP-1, MIP-1α, MIP-1β, TNF-α, RANTES, GCSF, IFN-γ, and VEGF concentrations positively correlate with tumor size [124]. A comparison of aqueous humor composition between Japanese subjects with UM and some benign eye tumors showed significantly higher concentrations of IL-8, MCP-1, and angiogenin. These findings indicate that IL-8, MCP-1, and angiogenin might serve as potential biomarkers for differentiation between malign UM and benign intraocular tumors [125].
The measurements of biomarkers in vitreous and aqueous humor of Austrian patients with UM showed higher concentrations of Flt-3 ligand, IL-6, IL-8, (IP)-10, MCP-1, MIP-1α, platelet-derived growth factor AA (PDGF-AA), and VEGF when compared with subjects with a cataract [126]. Eotaxin was significantly higher in the aqueous, and IL-7 in the vitreous humor of patients with UM. Tumor dimensions showed positive correlations with IP-10, and MIP-1 (in aqueous and vitreous humor), while Fms-related tyrosine kinase 3 ligand (FLT3LG), IL-6, IL-8, and MCP-1 were positively correlated in vitreous humor. Tumor infiltration in Bruch’s membrane, an extracellular matrix between the retinal pigment epithelium and the choroid, showed a positive correlation with FLT3LG and MCP-1 (in aqueous and vitreous humor), and IL-8, IP-10, MIP-1α, and PDGF-AA (in vitreous humor) [126].
Soluble human leukocyte antigens (sHLA) were investigated in Dutch patients with UM. In patients with sHLA-positive aqueous humor, the frequency of metastases was higher, and they showed significantly worse survival [127].
Overall, we can conclude that different case-control studies involving UM and benign eye disorders have revealed the role of angiogenic, inflammatory, and chemotaxic biomarkers as significant factors supporting the association between inflammation and tumorigenesis.
In addition to this diagnostic feature of cytokines and growth factors, biomedical research results in the measurement of biomarkers that can be useful for determining treatment effect and prognostic value.
A study involving 18 Chinese patients who underwent iodine-125 plaque therapy showed a significantly strong positive correlation of aqueous humor VEGF-A and PLGF levels with tumor thickness, and the correlation remained the same after radiation therapy [128]. Additionally, the expression of both growth factors increased after the surgery. IFN-γ, IL-2, and IL-10 concentrations were also observed to be significantly different between subgroups with elevated and non-elevated VEGF-A and PLGF, suggesting their potential role in inflammation and angiogenesis after radiotherapy [128]. The effect of combined Ru-106 brachytherapy and transpupillary thermotherapy (TTT) was investigated in 20 South Korean patients. An increase in the expression of IL-6, IL-8, and IL-1β was observed. This could serve as a useful tool for detecting radiation-related side effects [129]. Analysis of aqueous humor from 83 Dutch patients who underwent primary enucleation for UM showed a positive association between high ANG-2 expression, the development of metastases, and the presence of monosomy-3 [130]. Therefore, ANG-2 may be useful as an effective target for the potential treatment of UM.
The concentrations of different cystatins were investigated in different body fluids of Russian patients with UM, intraocular fluid, tears, and sera, and the results showed the diagnostic potential of cystatins [131]. Cystatins, as reversible inhibitors of cysteine proteases, have an important role in tumor progression.
The most frequently investigated biomarkers and their significance in the diagnosis, prognosis, and treatment of UM are listed in Table 2.

Table 2.
The roles of different body fluid biomarkers in diagnosis prognosis and treatment of uveal melanoma.
6. Current Challenges and Clinical Implications
UM is a serious and rare disease and irrespective of the modality of treatment administered over 50% of patients will develop fatal metastasis within ten years after initial diagnosis, most commonly in the liver [132]. Given that the occurrence of systemic disease is associated with increased mortality and adverse outcomes, it is important to identify patients at high risk. Once metastases develop, life expectancy is reduced, as existing treatment methods are still relatively ineffective [16,25,133].
6.1. Metastatic Risk Assessment
To date, treatment of metastatic UM has not given satisfactory results and as such remains a challenging problem for specialists dealing with ocular oncology [9,15,17,22,23,25,26,27,28,29,30,134]. In spite of long-term research, the only established method for controlling the spread of the disease is early treatment aimed at removing liver metastases, which has provided benefits for some patients [15]. Therefore, a better insight into molecular, genetic, and immunobiological features, early detection, and new effective treatment strategies are essential. Research in this area may aid in identifying valuable biomarkers for diagnostic and prognostic purposes and new treatment methods with the aim of selecting patients who would achieve the most benefit. Assessment of the risk for metastases could be achieved by genetic and epigenetic profiling of UM tumor tissue [21,24,31,135,136,137]. Future research should take into consideration how the tumor cells evade the immune system in order to develop new therapies that would affect these pathways.
Understanding the processes involved in drug resistance and side effects could also significantly improve treatment outcomes in patients with metastatic tumors and setting guidelines for novel treatment options [15,31,138,139,140]. Research should therefore be aimed towards applying the available knowledge regarding molecular, genetic, and immunobiological features of UM and directing them into effective forms of treatment targeting UM-specific molecular variations.
6.2. Tumor Tissue Analysis
Biopsy of primary UM is a valuable method that provides us with data pertaining to the morphology of tumor tissue, its immunological, cytological, biochemical, histopathological, and genetic features. The analysis of the samples obtained at the time of diagnosis and treatment of the primary tumor is essential for the assessment of its malignant and metastatic potential as well as the selection of the most effective treatment approach [3,9,21,25,31,55,132,133,138,139]. Fine-needle aspiration biopsy (FNAB) is the most commonly used method for biopsy of the primary UM, while other methods include incisional biopsy, with or without vitrectomy, and excisional biopsy. These are invasive techniques with a possible risk of tumor spread eye complications or insufficient sampling. However, if performed by an experienced eye surgeon, biopsy is a safe procedure whose benefits achieved by the progress in diagnostic and prognostic analysis of UM specimens outweighs the potential risks [3,9,135].
A new approach that uses liquid biopsies to analyze blood and other body fluids, such as vitreous and aqueous humor samples, may represent an additional less invasive method of analysis. In the future, it could be useful in diagnosis, monitoring tumor progression, determination of diagnostic and prognostic biomarkers, early detection of metastatic disease, local tumor recurrence, and response to treatment [21,135,137]. Nevertheless, histopathological, cytological, biochemical, and genetic analyses of tumor samples obtained by biopsy are still necessary to assess the malignant and metastatic potential of UMs [3,21,135]. Current evidence suggests that liquid biopsies may be a source of biomarkers, but in the absence of standardized methodology and analysis, further research is needed to justify the use of liquid biopsy methods in the diagnosis and follow-up of patients with UM [21,135,141].
Genetic and epigenetic features of tumors have attracted increasing attention in recent years due to their significant role in the process of carcinogenesis and the possibility of a better understanding of tumor behavior. This could enable the development of reliable biomarkers and a new therapeutic approach, resulting in new discoveries in the treatment of UM, better survival, and giving hope to patients who develop metastases [31,32,139].
6.3. Novel Treatment Options for Metastatic Uveal Melanoma
The treatment options for metastatic UM, albeit with some limitations, have improved with the use of tebentafusp, an immunotherapeutic agent consisting of an engineered T-cell receptor aimed at the gp100 epitope presented by HLA A*02:01 cells, fused to an anti-CD3 single-chain variable fragment. However, its action is limited to patients who have an HLA-A*02:01 allele with an additional drawback being the need for intensive monitoring and a weekly dosing schedule [142]. Further progress in the treatment of metastatic UM is represented by studies on the combined use of tebentafusp together with immune checkpoint inhibitors. Future trials could evaluate its role in the adjuvant therapy for patients with high-risk UM and molecular, genetic, and immunobiological analysis could give a significant contribution in this area [29,30,142].
7. Conclusions
UM is a life-threatening and relatively rare cancer that exhibits markedly distinctive biological behavior when compared to other types of melanomas and therefore necessitates particular treatment approaches. Despite its rarity, it presents a problem since the outcome continues to be unfavorable in a significant proportion of cases. To date, regardless of advancements in the diagnosis and treatment of primary tumors, metastatic disease continues to pose a challenge due to the lack of available successful therapeutic treatment options.
Molecular research has given a better understanding of the genetic and epigenetic mechanisms involved in the biological behavior of UM. In order to improve the treatment of these patients, emphasis needs to be aimed on researching diagnostic, prognostic, and therapeutic biomarkers, tumor immunogenicity, and the underlying mechanisms of tumor formation and spread. Better knowledge regarding the molecular changes underlying the development of UM promises a new perspective for improved and targeted therapeutic agents that influence certain phases of tumor development with a personalized approach. This will undoubtedly lead to advances in the systemic treatment of patients with metastatic disease as well as the prevention of spread in those having tumors with high metastatic potential. Molecular analysis of tumor tissue specimens will allow us to identify patients who would benefit from new therapeutic agents and provide a better insight into melanoma pathogenesis and its biological behavior.
Author Contributions
Conceptualization, D.P. and S.K.; methodology, D.P., T.N.-M. and A.S.; software, A.S.; validation, D.P. and S.K.; formal analysis, D.P. and T.N.-M.; investigation, D.P., T.N.-M., A.S. and S.K.; resources, S.K.; data curation, D.P. and T.N.-M.; writing—original draft preparation, D.P., T.N.-M., A.S. and S.K.; writing—review and editing, D.P. and S.K.; visualization, S.K.; supervision, D.P.; project administration, S.K.; funding acquisition, S.K. All authors have read and agreed to the published version of the manuscript.
Funding
University support 2022, No.10106-22-3067.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
Special thanks to Makso Herman for proofreading and editing the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chang, A.E.; Karnell, L.H.; Menck, H.R. The National Cancer Data Base Report on cutaneous and noncutaneous melanoma: A summary of 84,836 cases from the past decade. Cancer 1998, 83, 1664–1678. [Google Scholar] [CrossRef]
- Shields, C.L.; Kaliki, S.; Furuta, M.; Mashayekhi, A.; Shields, J.A. Clinical spectrum and prognosis of uveal melanoma based on age at presentation in 8033 cases. Retina 2012, 32, 1363–1372. [Google Scholar] [CrossRef]
- Kaštelan, S.; Antunica, A.G.; Beketić-Orešković, L.; Bakija, I.; Bogadi, M. Uveal mealnoma: Clinical features and diagnostic procedures. Libr. Oncol. 2017, 45, 81–88. [Google Scholar]
- Damato, E.M.; Damato, B.E. Detection and time to treatment of uveal melanoma in the United Kingdom: An evaluation of 2384 patients. Ophthalmology 2012, 119, 1582–1589. [Google Scholar] [CrossRef]
- Andreoli, M.T.; Mieler, W.F.; Leiderman, Y.I. Epidemiological trends in uveal melanoma. Br. J. Ophthalmol. 2015, 99, 1550–1553. [Google Scholar] [CrossRef] [PubMed]
- Virgili, G.; Gatta, G.; Ciccolallo, L.; Capocaccia, R.; Biggeri, A.; Crocetti, E.; Lutz, J.M.; Paci, E. Incidence of uveal melanoma in Europe. Ophthalmology 2007, 114, 2309–2315. [Google Scholar] [CrossRef]
- Vajdic, C.M.; Kricker, A.; Giblin, M.; McKenzie, J.; Aitken, J.; Giles, G.G.; Armstrong, B.K. Incidence of ocular melanoma in Australia from 1990 to 1998. Int. J. Cancer 2003, 105, 117–122. [Google Scholar] [CrossRef]
- Margo, C.E.; Mulla, Z.; Billiris, K. Incidence of surgically treated uveal melanoma by race and ethnicity. Ophthalmology 1998, 105, 1087–1090. [Google Scholar] [CrossRef] [PubMed]
- Tarlan, B.; Kıratlı, H. Uveal melanoma: Current trends in diagnosis and management. Turk Oftalmoloiji Derg. 2016, 46, 123–137. [Google Scholar] [CrossRef]
- Nayman, T.; Bostan, C.; Logan, P.; Burnier, M.N. Uveal melanoma risk factors: A systematic review of meta-analyses. Curr. Eye Res. 2017, 42, 1085–1093. [Google Scholar] [CrossRef]
- Hammer, H.; Oláh, J.; Tóth-Molnár, E. Dysplastic nevi are a risk factor for uveal melanoma. Eur. J. Ophthalmol. 1996, 6, 472–474. [Google Scholar] [CrossRef]
- Shields, C.L.; Kaliki, S.; Livesey, M.; Walker, B.; Garoon, R.; Bucci, M.; Feinstein, E.; Pesch, A.; Gonzalez, C.; Lally, S.E.; et al. Association of ocular and oculodermal melanocytosis with the rate of uveal melanoma metastasis analysis of 7872 consecutive eyes. JAMA Ophthalmol. 2013, 131, 993–1003. [Google Scholar] [CrossRef] [PubMed]
- Weis, E.; Shah, C.P.; Lajous, M.; Shields, J.; Shields, C. The association between host susceptibility factors and uveal melanoma. Arch. Ophthalmol. 2006, 124, 54. [Google Scholar] [CrossRef]
- Harbour, J.W.; Onken, M.D.; Roberson, E.D.O.; Duan, S.; Cao, L.; Worley, L.A.; Council, M.L.; Matatall, K.A.; Helms, C.; Bowcock, A.M. Frequent mutation of BAP1 in metastasizing uveal melanomas. Science 2010, 330, 1410–1413. [Google Scholar] [CrossRef] [PubMed]
- Kastelan, S.; Zimak, D.M.; Ivankovic, M.; Markovic, I.; Antunica, A.G. Liver metastasis in uveal melanoma-treatment options and clinical outcome. Front. Biosci. Landmark 2022, 27, 72. [Google Scholar] [CrossRef]
- Damato, B.; Eleuteri, A.; Taktak, A.F.G.; Coupland, S.E. Estimating prognosis for survival after treatment of choroidal melanoma. Prog. Retin. Eye Res. 2011, 30, 285–295. [Google Scholar] [CrossRef]
- Rodriguez-Vidal, C.; Fernandez-Diaz, D.; Fernandez-Marta, B.; Lago-Baameiro, N.; Pardo, M.; Silva, P.; Paniagua, L.; Blanco-Teijeiro, M.J.; Piñeiro, A.; Bande, M. Treatment of metastatic uveal melanoma: Systematic review. Cancers 2020, 12, 2557. [Google Scholar] [CrossRef]
- Damato, B. Treatment of primary intraocular melanoma. Expert Rev. Anticancer Ther. 2006, 6, 493–506. [Google Scholar] [CrossRef]
- Kaštelan, S.; Gverović Antunica, A.; Beketić Oresković, L.; Kasun, B.; Hat, K. Uveal melanoma: An overview of management and prognosis. Libr. Oncol. 2018, 46, 95–104. [Google Scholar] [CrossRef]
- Garg, G.; Finger, P.T.; Kivelä, T.T.; Simpson, R.; Gallie, B.L.; Saakyan, S.; Amiryan, A.G.; Valskiy, V.; Chin, K.J.; Semenova, K.; et al. Patients presenting with metastases: Stage IV uveal melanoma, an international study. Br. J. Ophthalmol. 2022, 106, 510–517. [Google Scholar] [CrossRef]
- Lamas, N.J.; Martel, A.; Nahon-Estève, S.; Goffinet, S.; Macocco, A.; Bertolotto, C.; Lassalle, S.; Hofman, P. Prognostic biomarkers in uveal melanoma: The status quo, recent advances and future directions. Cancers 2022, 14, 96. [Google Scholar] [CrossRef]
- Kaštelan, S.; Antunica, A.G.; Oresković, L.B.; Pelčić, G.; Kasun, E.; Hat, K. Immunotherapy for uveal melanoma—Current knowledge and perspectives. Curr. Med. Chem. 2020, 27, 1350–1366. [Google Scholar] [CrossRef]
- Sussman, T.A.; Funchain, P.; Singh, A. clinical trials in metastatic uveal melanoma: Current status. Ocul. Oncol. Pathol. 2020, 6, 381–387. [Google Scholar] [CrossRef]
- Barbagallo, C.; Stella, M.; Broggi, G.; Russo, A.; Caltabiano, R.; Ragusa, M. Genetics and RNA regulation of uveal melanoma. Cancers 2023, 15, 775. [Google Scholar] [CrossRef] [PubMed]
- Kaliki, S.; Shields, C.L.; Shields, J.A. Uveal melanoma: Estimating prognosis. Indian J. Ophthalmol. 2015, 63, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Oliva, M.; Rullan, A.J.; Piulats, J.M. Uveal melanoma as a target for immune-therapy. Ann. Transl. Med. 2016, 4, 172. [Google Scholar] [CrossRef]
- Rowcroft, A.; Loveday, B.P.T.; Thomson, B.N.J.; Banting, S.; Knowles, B. Systematic review of liver directed therapy for uveal melanoma hepatic metastases. Hpb 2020, 22, 497–505. [Google Scholar] [CrossRef]
- Buder, K.; Gesierich, A.; Gelbrich, G.; Goebeler, M. Systemic treatment of metastatic uveal melanoma: Review of literature and future perspectives. Cancer Med. 2013, 2, 674–686. [Google Scholar] [CrossRef]
- Wang, Z.; Xie, Y.; Wang, J.; Cheng, Y.; Fleishman, J.; Chen, Z.; Chen, Y. Tebentafusp: A novel drug for the treatment of metastatic uveal melanoma. Drugs Today 2023, 59, 179–193. [Google Scholar] [CrossRef]
- Montazeri, K.; Pattanayak, V.; Sullivan, R.J. Tebentafusp in the treatment of metastatic uveal melanoma: Patient selection and special considerations. Drug Des. Devel. Ther. 2023, 17, 333–339. [Google Scholar] [CrossRef]
- Fallico, M.; Raciti, G.; Longo, A.; Reibaldi, M.; Bonfiglio, V.; Russo, A.; Caltabiano, R.; Gattuso, G.; Falzone, L.; Avitabile, T. Current molecular and clinical insights into uveal melanoma (review). Int. J. Oncol. 2021, 58, 1–22. [Google Scholar] [CrossRef]
- Shain, A.H.; Bagger, M.M.; Yu, R.; Chang, D.; Liu, S.; Vemula, S.; Weier, J.F.; Wadt, K.; Heegaard, S.; Bastian, B.C.; et al. The Genetic evolution of metastatic uveal melanoma. Nat. Genet. 2019, 51, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Kilic, E.; Van Gils, W.; Lodder, E.; Beverloo, H.B.; Van Til, M.E.; Mooy, C.M.; Paridaens, D.; De Klein, A.; Luyten, G.P.M. Clinical and cytogenetic analyses in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2006, 47, 3703–3707. [Google Scholar] [CrossRef]
- Harbour, J.W. Molecular prognostic testing and individualized patient care in uveal melanoma. Am. J. Ophthalmol. 2009, 148, 823–829.e1. [Google Scholar] [CrossRef]
- Mudhar, H.S.; Parsons, M.A.; Sisley, K.; Rundle, P.; Singh, A.; Rennie, I.G. A Critical appraisal of the prognostic and predictive factors for uveal malignant melanoma. Histopathology 2004, 45, 1–12. [Google Scholar] [CrossRef]
- Sisley, K.; Doherty, R.; Cross, N.A. What hope for the future? GNAQ and uveal melanoma. Br. J. Ophthalmol. 2011, 95, 620–623. [Google Scholar] [CrossRef]
- Van Raamsdonk, C.D.; Griewank, K.G.; Crosby, M.B.; Garrido, M.C.; Vemula, S.; Wiesner, T.; Obenauf, A.C.; Wackernagel, W.; Green, G.; Bouvier, N.; et al. Mutations in GNA11 in uveal melanoma. N. Engl. J. Med. 2010, 363, 2191–2199. [Google Scholar] [CrossRef]
- Onken, M.D.; Worley, L.A.; Long, M.D.; Duan, S.; Council, M.L.; Bowcock, A.M.; Harbour, J.W. Oncogenic Mutations in GNAQ occur early in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2008, 49, 5230–5234. [Google Scholar] [CrossRef]
- Bauer, J.; Kilic, E.; Vaarwater, J.; Bastian, B.C.; Garbe, C.; De Klein, A. oncogenic GNAQ mutations are not correlated with disease-free survival in uveal melanoma. Br. J. Cancer 2009, 101, 813–815. [Google Scholar] [CrossRef] [PubMed]
- Koopmans, A.E.; Verdijk, R.M.; Brouwer, R.W.W.; Van Den Bosch, T.P.P.; Van Den Berg, M.M.P.; Vaarwater, J.; Kockx, C.E.M.; Paridaens, D.; Naus, N.C.; Nellist, M.; et al. Clinical significance of immunohistochemistry for detection of BAP1 mutations in uveal melanoma. Mod. Pathol. 2014, 27, 1321–1330. [Google Scholar] [CrossRef]
- Jensen, D.E.; Proctor, M.; Marquis, S.T.; Gardner, H.P.; Ha, S.I.; Chodosh, L.A.; Ishov, A.M.; Tommerup, N.; Vissing, H.; Sekido, Y.; et al. BAP1: A novel ubiquitin hydrolase which binds to the BRCA1 RING finger and enhances BRCA1-mediated cell growth suppression. Oncogene 1998, 16, 1097–1112. [Google Scholar] [CrossRef]
- Karlsson, J.; Nilsson, L.M.; Mitra, S.; Alsén, S.; Shelke, G.V.; Sah, V.R.; Forsberg, E.M.V.; Stierner, U.; All-Eriksson, C.; Einarsdottir, B.; et al. Molecular profiling of driver events in metastatic uveal melanoma. Nat. Commun. 2020, 11, 1894. [Google Scholar] [CrossRef]
- Riechardt, A.I.; Kilic, E.; Joussen, A.M. The genetics of uveal melanoma: Overview and clinical relevance. Klin. Monbl. Augenheilkd. 2021, 238, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Harbour, J.W.; Roberson, E.D.O.; Anbunathan, H.; Onken, M.D.; Worley, L.A.; Bowcock, A.M. Recurrent mutations at codon 625 of the splicing factor SF3B1 in uveal melanoma. Nat. Genet. 2013, 45, 133–135. [Google Scholar] [CrossRef] [PubMed]
- Alsafadi, S.; Houy, A.; Battistella, A.; Popova, T.; Wassef, M.; Henry, E.; Tirode, F.; Constantinou, A.; Piperno-Neumann, S.; Roman-Roman, S.; et al. Cancer-associated SF3B1 mutations affect alternative splicing by promoting alternative branchpoint usage. Nat. Commun. 2016, 7, 10615. [Google Scholar] [CrossRef]
- Li, Y.; Shi, J.; Yang, J.; Ge, S.; Zhang, J.; Jia, R.; Fan, X. Uveal melanoma: Progress in molecular biology and therapeutics. Ther. Adv. Med. Oncol. 2020, 12, 175883592096585. [Google Scholar] [CrossRef]
- Johnson, C.P.; Kim, I.K.; Esmaeli, B.; Amin-Mansour, A.; Treacy, D.J.; Carter, S.L.; Hodis, E.; Wagle, N.; Seepo, S.; Yu, X.; et al. Systematic Genomic and translational efficiency studies of uveal melanoma. PLoS ONE 2017, 12, e0178189. [Google Scholar] [CrossRef]
- Koopmans, A.E.; Ober, K.; Dubbink, H.J.; Paridaens, D.; Naus, N.C.; Belunek, S.; Krist, B.; Post, E.; Zwarthoff, E.C.; De Klein, A.; et al. Prevalence and implications of TERT Promoter mutation in uveal and conjunctival melanoma and in benign and premalignant conjunctival melanocytic lesions. Investig. Ophthalmol. Vis. Sci. 2014, 55, 6024–6030. [Google Scholar] [CrossRef]
- Huang, F.W.; Hodis, E.; Xu, M.J.; Kryukov, G.V.; Chin, L.; Garraway, L.A. Highly recurrent TERT promoter mutations in human melanoma. Science 2013, 339, 957–959. [Google Scholar] [CrossRef]
- Wu, X.; Huang, C.; He, X.; Tian, Y.; Zhou, D.; He, Y.; Liu, X.; Li, J. Feedback regulation of telomerase reverse transcriptase: New insight into the evolving field of telomerase in cancer. Cell. Signal. 2013, 25, 2462–2468. [Google Scholar] [CrossRef]
- Derrien, A.C.; Rodrigues, M.; Eeckhoutte, A.; Dayot, S.; Houy, A.; Mobuchon, L.; Gardrat, S.; Lequin, D.; Ballet, S.; Pierron, G.; et al. Germline MBD4 mutations and predisposition to uveal melanoma. J. Natl. Cancer Inst. 2021, 113, 80–87. [Google Scholar] [CrossRef]
- Li, Y.; Jia, R.; Ge, S. Role of epigenetics in uveal melanoma. Int. J. Biol. Sci. 2017, 13, 426–433. [Google Scholar] [CrossRef]
- Robertson, A.G.; Shih, J.; Yau, C.; Gibb, E.A.; Oba, J.; Mungall, K.L.; Hess, J.M.; Uzunangelov, V.; Walter, V.; Danilova, L.; et al. Integrative analysis identifies four molecular and clinical subsets in uveal melanoma. Cancer Cell 2017, 32, 204–220.e15. [Google Scholar] [CrossRef] [PubMed]
- Jurmeister, P.; Wrede, N.; Hoffmann, I.; Vollbrecht, C.; Heim, D.; Hummel, M.; Wolkenstein, P.; Koch, I.; Heynol, V.; Schmitt, W.D.; et al. Mucosal melanomas of different anatomic sites share a common global DNA methylation profile with cutaneous melanoma but show location-dependent patterns of genetic and epigenetic alterations. J. Pathol. 2022, 256, 61–70. [Google Scholar] [CrossRef]
- Harbour, J.W. A prognostic test to predict the risk of metastasis in uveal melanoma based on a 15-gene expression profile. Methods Mol. Biol. 2014, 1102, 427–440. [Google Scholar] [PubMed]
- Field, M.G.; Decatur, C.L.; Kurtenbach, S.; Gezgin, G.; van der Velden, P.A.; Jager, M.J.; Kozak, K.N.; Harbour, J.W. PRAME as an independent biomarker for metastasis in uveal melanoma. Clin. Cancer Res. 2016, 22, 1234–1242. [Google Scholar] [CrossRef]
- Fabian, I.D.; Thaung, C.; AlHarby, L.; Sisley, K.; Mudhar, H.S.; Doherty, R.E.; Stacey, A.W.; Arora, A.K.; Cohen, V.M.L.; Sagoo, M.S. Late solitary extraocular recurrence from previously resected iris melanoma. Am. J. Ophthalmol. 2017, 181, 97–105. [Google Scholar] [CrossRef]
- Mensink, H.W.; Vaarwater, J.; de Keizer, R.J.W.; de Wolff-Rouendaal, D.; Mooy, C.M.; de Klein, A.; Paridaens, D. Chromosomal aberrations in iris melanomas. Br. J. Ophthalmol. 2011, 95, 424–428. [Google Scholar] [CrossRef][Green Version]
- Henriquez, F.; Janssen, C.; Kemp, E.G.; Roberts, F. The T1799A BRAF mutation is present in iris melanoma. Investig. Ophthalmol. Vis. Sci. 2007, 48, 4897–4900. [Google Scholar] [CrossRef] [PubMed]
- Scholz, S.L.; Möller, I.; Reis, H.; Süßkind, D.; Van De Nes, J.A.P.; Leonardelli, S.; Schilling, B.; Livingstone, E.; Schimming, T.; Paschen, A.; et al. Frequent GNAQ, GNA11, and EIF1AX mutations in iris melanoma. Investig. Ophthalmol. Vis. Sci. 2017, 58, 3464–3470. [Google Scholar] [CrossRef]
- Friedman, R.C.; Farh, K.K.H.; Burge, C.B.; Bartel, D.P. Most mammalian MRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef]
- Peng, Y.; Croce, C.M. The role of microRNAs in human cancer. Signal Transduct. Target. Ther. 2016, 1, 15004. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Zhou, X.; Chen, X.; Hu, D.N.; Da Dong, X.; Wang, J.; Lu, F.; Tu, L.L.; Qu, J. MicroRNA-34a inhibits uveal melanoma cell proliferation and migration through downregulation of c-Met. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1559–1565. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Han, S.; Yang, L.; Chen, S.; Chen, J.; Ma, N.; Tang, J.; Chen, X.; Chen, F.; Da, X.; et al. The interplay of microRNA-34a, LGR4, EMT-associated factors, and MMP2 in regulating uveal melanoma cells. Invest. Ophthalmol. Vis. Sci. 2019, 60, 4503–4510. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Lou, D. MicroRNA-34b/c suppresses uveal melanoma cell proliferation and migration through multiple targets. Mol. Vis. 2012, 18, 537–546. [Google Scholar]
- Amaro, A.; Croce, M.; Ferrini, S.; Barisione, G.; Gualco, M.; Perri, P.; Pfe, U.; Jager, M.J.; Coupland, S.E.; Mosci, C.; et al. Potential onco-suppressive role of miR122 and miR144 in uveal melanoma through ADAM10 and C-Met inhibition. Cancers 2020, 12, 1468. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, J.; Shen, H.; Lu, J.; Li, C.; Hu, D.N.; da Dong, X.; Yan, D.; Tu, L. Epigenetics, microRNAs, and carcinogenesis: Functional role of microRNA-137 in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2011, 52, 1193–1199. [Google Scholar] [CrossRef]
- Yan, D.; da Dong, X.; Chen, X.; Yao, S.; Wang, L.; Wang, J.; Wang, C.; Hu, D.N.; Qu, J.; Tu, L.L. Role of microRNA-182 in posterior uveal melanoma: Regulation of tumor development through MITF, BCL2 and cyclin D2. PLoS ONE 2012, 7, e40967. [Google Scholar] [CrossRef]
- Chen, X.; He, D.; Da Dong, X.; Dong, F.; Wang, J.; Wang, L.; Tang, J.; Hu, D.N.; Yan, D.; Tu, L.L. MicroRNA-124a is epigenetically regulated and acts as a tumor suppressor by controlling multiple targets in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2013, 54, 2248–2256. [Google Scholar] [CrossRef]
- Ewen, M.E.; Sluss, H.K.; Sherr, C.J.; Matsushime, H.; Kato, J.; Livingston, D.M. Functional interactions of the retinoblastoma protein with mammalian D-type cyclins. Cell 1993, 73, 487–497. [Google Scholar] [CrossRef]
- Harbour, J.W.; Luo, R.X.; Dei Santi, A.; Postigo, A.A.; Dean, D.C. Cdk phosphorylation triggers sequential intramolecular interactions that progressively Block Rb Functions as Cells Move through G1. Cell 1999, 98, 859–869. [Google Scholar] [CrossRef] [PubMed]
- Bachmann, I.M.; Halvorsen, O.J.; Collett, K.; Stefansson, I.M.; Straume, O.; Haukaas, S.A.; Salvesen, H.B.; Otte, A.P.; Akslen, L.A. EZH2 expression is associated with high proliferation rate and aggressive tumor subgroups in cutaneous melanoma and cancers of the endometrium, Prostate, and Breast. J. Clin. Oncol. 2006, 24, 268–273. [Google Scholar] [CrossRef]
- Li, Y.; Luo, J.T.; Liu, Y.M.; Wei, W. Bin miRNA-145/miRNA-205 inhibits proliferation and invasion of uveal melanoma cells by targeting NPR1/CDC42. Int. J. Ophthalmol. 2020, 13, 718–724. [Google Scholar] [CrossRef]
- Del Mar Maldonado, M.; Dharmawardhane, S. Targeting Rac and Cdc42 GT pases in cancer. Cancer Res. 2018, 78, 3101–3111. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Sun, Q.; Chen, J.; Li, J.; Zeng, Y.; Zhai, S.; Li, P.; Wang, B.; Wang, X. MicroRNA-9 suppresses uveal melanoma cell migration and invasion through the NF-ΚB1 pathway. Oncol. Rep. 2012, 28, 961–968. [Google Scholar] [CrossRef]
- Wu, S.; Chen, H.; Han, N.; Zhang, C.; Yan, H. Long noncoding RNA PVT1 silencing prevents the development of uveal melanoma by impairing microRNA-17-3p–dependent MDM2 upregulation. Investig. Ophthalmol. Vis. Sci. 2019, 60, 4904–4914. [Google Scholar] [CrossRef] [PubMed]
- Peng, D.; Dong, J.; Zhao, Y.; Peng, X.; Tang, J.; Chen, X.; Wang, L.; Hu, D.N.; Reinach, P.S.; Qu, J.; et al. MiR-142-3p Suppresses uveal melanoma by targeting CDC25C, TGFβR1, GNAQ, WASL, and RAC1. Cancer Manag. Res. 2019, 11, 4729–4742. [Google Scholar] [CrossRef]
- Liu, Y.; Huo, Y.; Wang, D.; Tai, Y.; Li, J.; Pang, D.; Zhang, Y.; Zhao, W.; Du, N.; Huang, Y. MiR-216a-5p/Hexokinase 2 axis regulates uveal melanoma growth through modulation of warburg effect. Biochem. Biophys. Res. Commun. 2018, 501, 885–892. [Google Scholar] [CrossRef]
- Li, J.; Liu, X.; Li, C.; Wang, W. MiR-224-5p inhibits proliferation, migration, and invasion by targeting PIK3R3/AKT3 in uveal melanoma. J. Cell. Biochem. 2019, 120, 12412–12421. [Google Scholar] [CrossRef]
- Okayama, H.; Schetter, A.J.; Harris, C.C. MicroRNAs and inflammation in the pathogenesis and progression of colon cancer. Dig. Dis. 2012, 30, 9–15. [Google Scholar] [CrossRef]
- Wang, Y.C.; Yang, X.; Bin Wei, W.; Xu, X.L. Role of microRNA-21 in uveal melanoma cell invasion and metastasis by regulating P53 and its downstream protein. Int. J. Ophthalmol. 2018, 11, 1258–1268. [Google Scholar] [CrossRef]
- Peng, J.; Liu, H.; Liu, C. MiR-155 promotes uveal melanoma cell proliferation and invasion by regulating NDFIP1 expression. Technol. Cancer Res. Treat. 2017, 16, 1160–1167. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; He, X.; Li, F.; Pan, H.; Huang, X.; Wen, X.; Zhang, H.; Li, B.; Ge, S.; Xu, X.; et al. The miR-181 family promotes cell cycle by targeting CTDSPL, a phosphatase-like tumor suppressor in uveal melanoma. J. Exp. Clin. Cancer Res. 2018, 37, 1–13. [Google Scholar] [CrossRef]
- Ling, J.W.; Lu, P.R.; Zhang, Y.B.; Jiang, S.; Zhang, Z.C. MiR-367 promotes uveal melanoma cell proliferation and migration by regulating PTEN. Genet. Mol. Res. 2017, 16, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wang, Q.; Gao, X.; Shi, D.; Mi, S.; Han, Q. MicroRNA-454 functions as an oncogene by regulating pten in uveal melanoma. FEBS Lett. 2015, 589, 2791–2796. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.H.; Yang, Y.; Zhou, X.P.; Craig, E.L.; Davidorf, F.H.; Eng, C. High frequency of submicroscopic hemizygous deletion is a major mechanism of loss of expression of PTEN in uveal melanoma. J. Clin. Oncol. 2006, 24, 288–295. [Google Scholar] [CrossRef]
- Cheng, Y.; Cheng, T.; Zhao, Y.; Qu, Y. HMGA1 exacerbates tumor progression by activating miR-222 through PI3K/Akt/MMP-9 signaling pathway in uveal melanoma. Cell. Signal. 2019, 63, 109386. [Google Scholar] [CrossRef]
- Souri, Z.; Wierenga, A.P.A.; Kiliç, E.; Brosens, E.; Böhringer, S.; Kroes, W.G.M.; Verdijk, R.M.; van der Velden, P.A.; Luyten, G.P.M.; Jager, M.J. MiRNAs correlate with HLA expression in uveal melanoma: Both up-and Downregulation Are Related to Monosomy 3. Cancers 2021, 13, 4020. [Google Scholar] [CrossRef]
- Zhou, J.; Jiang, J.; Wang, S.; Xia, X. Oncogenic role of microrNA-20a in human uveal melanoma. Mol. Med. Rep. 2016, 14, 1560–1566. [Google Scholar] [CrossRef]
- Wang, X.; Hu, Y.; Cui, J.; Zhou, Y.; Chen, L. Coordinated targeting of MMP-2/MMP-9 by miR-296-3p/FOXCUT exerts tumor-suppressing effects in choroidal malignant melanoma. Mol. Cell. Biochem. 2018, 445, 25–33. [Google Scholar] [CrossRef]
- Xia, Z.; Yang, C.; Yang, X.; Wu, S.; Feng, Z.; Qu, L.; Chen, X.; Liu, L.; Ma, Y. MiR-652 promotes proliferation and migration of uveal melanoma cells by targeting HOXA9. Med. Sci. Monit. 2019, 25, 8722–8732. [Google Scholar] [CrossRef]
- Worley, L.A.; Long, M.D.; Onken, M.D.; Harbour, J.W. Micro-RNAs associated with metastasis in uveal melanoma identified by multiplexed microarray profiling. Melanoma Res. 2008, 18, 184–190. [Google Scholar] [CrossRef]
- Venkatesan, N.; Kanwar, J.; Deepa, P.R.; Khetan, V.; Crowley, T.M.; Raguraman, R.; Sugneswari, G.; Rishi, P.; Natarajan, V.; Biswas, J.; et al. Clinico-pathological association of delineated miRNAs in uveal melanoma with monosomy 3/disomy 3 chromosomal aberrations. PLoS ONE 2016, 11, e0146128. [Google Scholar] [CrossRef]
- Smit, K.N.; Chang, J.; Derks, K.; Vaarwater, J.; Brands, T.; Verdijk, R.M.; Wiemer, E.A.C.; Mensink, H.W.; Pothof, J.; de Klein, A.; et al. Aberrant microRNA expression and its implications for uveal melanoma metastasis. Cancers 2019, 11, 815. [Google Scholar] [CrossRef]
- Wróblewska, J.P.; Lach, M.S.; Ustaszewski, A.; Kulcenty, K.; Ibbs, M.; Jagiełło, I.; Suchorska, W.M.; Marszałek, A. The Potential role of selected miRNA in uveal melanoma primary tumors as early biomarkers of disease progression. Genes 2020, 11, 271. [Google Scholar] [CrossRef] [PubMed]
- Falzone, L.; Romano, G.L.; Salemi, R.; Bucolo, C.; Tomasello, B.; Lupo, G.; Anfuso, C.D.; Spandidos, D.A.; Libra, M.; Candido, S. Prognostic significance of deregulated microRNAs in uveal melanomas. Mol. Med. Rep. 2019, 19, 2599–2610. [Google Scholar] [CrossRef] [PubMed]
- Xin, X.; Zhang, Y.; Ling, F.; Wang, L.; Sheng, X.; Qin, L.; Zhao, X. Identification of a nine-miRNA signature for the prognosis of uveal melanoma. Exp. Eye Res. 2019, 180, 242–249. [Google Scholar] [CrossRef]
- Vashishtha, A.; Lee, T.J.; Sharma, A.; Wallbillich, J.J. Research paper changes in microRNA expression associated with metastasis and survival in patients with uveal melanoma. Oncotarget 2020, 11, 1435–1447. [Google Scholar] [CrossRef][Green Version]
- Venza, I.; Visalli, M.; Beninati, C.; Benfatto, S.; Teti, D.; Venza, M. IL-10Rα expression is post-transcriptionally regulated by miR-15a, miR-185, and miR-211 in melanoma. BMC Med. Genomics 2015, 8, 81. [Google Scholar] [CrossRef]
- Triozzi, P.L.; Achberger, S.; Aldrich, W.; Singh, A.D.; Grane, R.; Borden, E.C. The association of blood angioregulatory microRNA levels with circulating endothelial cells and angiogenic proteins in patients receiving dacarbazine and interferon. J. Transl. Med. 2012, 10, 241. [Google Scholar] [CrossRef] [PubMed]
- Triozzi, P.L.; Achberger, S.; Aldrich, W.; Crabb, J.W.; Saunthararajah, Y.; Singh, A.D. Association of tumor and plasma microRNA expression with tumor monosomy-3 in patients with uveal melanoma. Clin. Epigenetics 2016, 8, 35. [Google Scholar] [CrossRef]
- Stark, M.S.; Gray, E.S.; Isaacs, T.; Chen, F.K.; Millward, M.; McEvoy, A.; Zaenker, P.; Ziman, M.; Soyer, H.P.; Glasson, W.J.; et al. A panel of circulating micrornas detects uveal melanoma with high precision. Transl. Vis. Sci. Technol. 2019, 8, 12. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Caltabiano, R.; Longo, A.; Avitabile, T.; Franco, L.M.; Bonfiglio, V.; Puzzo, L.; Reibaldi, M. Increased levels of mirNA-146a in serum and histologic samples of patients with uveal melanoma. Front. Pharmacol. 2016, 7, 424. [Google Scholar] [CrossRef]
- Yang, Z.K.; Yang, J.Y.; Xu, Z.Z.; Yu, W.H. DNA methylation and uveal melanoma. Chin. Med. J. 2018, 131, 845–851. [Google Scholar] [CrossRef]
- Bakhoum, M.F.; Curtis, E.J.; Goldbaum, M.H.; Mischel, P.S. BAP1 methylation: A prognostic marker of uveal melanoma metastasis. NPJ Precis. Onc. 2021, 5, 89. [Google Scholar] [CrossRef]
- Smit, K.N.; Boers, R.; Vaarwater, J.; Boers, J.; Brands, T.; Mensink, H.; Verdijk, R.M.; van IJcken, W.F.J.; Gribnau, J.; de Klein, A.; et al. Genome-wide aberrant methylation in primary metastatic UM and their matched metastases. Sci. Rep. 2022, 12, 42. [Google Scholar] [CrossRef]
- Ness, C.; Katta, K.; Garred, Ø.; Kumar, T.; Olstad, O.K.; Petrovski, G.; Moe, M.C.; Noer, A. Integrated differential DNA methylation and gene expression of formalin-fixed paraffin-embedded uveal melanoma specimens identifies genes associated with early metastasis and poor prognosis. Exp. Eye Res. 2021, 203, 108426. [Google Scholar] [CrossRef]
- Soltysova, A.; Dvorska, D.; Kajabova, V.H.; Pecimonova, M.; Cepcekova, K.; Ficek, A.; Demkova, L.; Buocikova, V.; Babal, P.; Juras, I.; et al. DNA methylation aberrancy is a reliable prognostic tool in uveal melanoma. Res. Square 2023. [Google Scholar] [CrossRef]
- Mouriaux, F.; Diorio, C.; Bergeron, D.; Berchi, C.; Rousseau, A. Liver function testing is not helpful for early diagnosis of metastatic uveal melanoma. Ophthalmology 2012, 119, 1590–1595. [Google Scholar] [CrossRef] [PubMed]
- Eskelin, S.; Pyrhönen, S.; Hahka-Kemppinen, M.; Tuomaala, S.; Kivelä, T. A prognostic model and staging for metastatic uveal melanoma. Cancer 2003, 97, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Patel, M. Characterization of computed tomography scan abnormalities in patients with biopsy-proven hepatic metastases from uveal melanoma. Arch. Ophthalmol. 2011, 129, 1576. [Google Scholar] [CrossRef][Green Version]
- Eskelin, S.; Pyrhonen, S.; Summanen, P.; Hahka-Kemppinen, M.; Kivelä, T.; Schachat, A.P. Tumor doubling times in metastatic malignant melanoma of the uvea: Tumor progression before and after treatment. Evid.-Based Eye Care 2001, 2, 36–37. [Google Scholar] [CrossRef]
- Barak, V.; Frenkel, S.; Kalickman, I.; Maniotis, A.; Folberg, R.; Pe’Er, J. Serum markers to detect metastatic uveal melanoma. J. Anticancer Res 2007, 27, 1897–1900. [Google Scholar]
- Barak, V.; Kaiserman, I.; Frenkel, S.; Hendler, K.; Kalickman, I.; Pe’er, J. The dynamics of serum tumor markers in predicting metastatic uveal melanoma (part 1). Anticancer Res. 2011, 31, 345–349. [Google Scholar] [PubMed]
- Haritoglou, I.; Wolf, A.; Maier, T.; Haritoglou, C.; Hein, R.; Schaller, U.C. Osteopontin and “melanoma inhibitory activity”: Comparison of two serological tumor markers in metastatic uveal melanoma patients. Ophthalmologica 2009, 223, 239–243. [Google Scholar] [CrossRef]
- Missotten, G.S.; Korse, C.M.; Van Dehn, C.; Linders, T.C.; Keunen, J.E.; Jager, M.J.; Bonfrer, J.M. S-100B Protein and melanoma inhibitory activity protein in uveal melanoma screening: A comparison with liver function tests. Tumor Biol. 2007, 28, 63–69. [Google Scholar] [CrossRef]
- Wróblewska, J.P.; Lach, M.S.; Kulcenty, K.; Galus, Ł.; Suchorska, W.M.; Rösel, D.; Brábek, J.; Marszałek, A. The analysis of inflammation-related proteins in a cargo of exosomes derived from the serum of uveal melanoma patients reveals potential biomarkers of disease progression. Cancers 2021, 13, 3334. [Google Scholar] [CrossRef]
- Frenkel, S.; Zloto, O.; Pe’er, J.; Barak, V. Insulin-like growth factor-1 as a predictive biomarker for metastatic uveal melanoma in humans. Investig. Ophthalmol. Vis. Sci. 2013, 54, 490–493. [Google Scholar] [CrossRef] [PubMed]
- Husain, B.; Kirchberger, M.C.; Erdmann, M.; Schüpferling, S.; Abolhassani, A.R.; Fröhlich, W.; Berking, C.; Heinzerling, L. Inflammatory markers in autoimmunity induced by checkpoint inhibitors. J. Cancer Res. Clin. Oncol. 2021, 147, 1623–1630. [Google Scholar] [CrossRef]
- Sunderland, D.; Sapra, A. Physiology, Aqueous Humor Circulation. In Study Guide StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK553209/ (accessed on 1 May 2023).
- Goel, M. Aqueous humor dynamics: A review. Open Ophthalmol. J. 2010, 4, 52–59. [Google Scholar] [CrossRef]
- Cheng, Y.; Feng, J.; Zhu, X.; Liang, J. Cytokines Concentrations in aqueous humor of eyes with uveal melanoma. Medicine 2019, 98, e14030. [Google Scholar] [CrossRef] [PubMed]
- Midena, E.; Parrozzani, R.; Midena, G.; Trainiti, S.; Marchione, G.; Cosmo, E.; Londei, D.; Frizziero, L. In vivo intraocular biomarkers: Changes of aqueous humor cytokines and chemokines in patients affected by uveal melanoma. Medicine 2020, 99, e22091. [Google Scholar] [CrossRef]
- Nagarkatti-Gude, N.; Bronkhorst, I.H.G.; van Duinen, S.G.; Luyten, G.P.M.; Jager, M.J. Cytokines and Chemokines in the vitreous fluid of eyes with uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2012, 53, 6748–6755. [Google Scholar] [CrossRef] [PubMed]
- Usui, Y.; Tsubota, K.; Agawa, T.; Ueda, S.; Umazume, K.; Okunuki, Y.; Kezuka, T.; Yamakawa, N.; Goto, H. Aqueous immune mediators in malignant uveal melanomas in comparison to benign pigmented intraocular tumors. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 255, 393–399. [Google Scholar] [CrossRef]
- Dunavoelgyi, R.; Funk, M.; Sacu, S.; Georgopoulos, M.; Zlabinger, G.; Zehetmayer, M.; Schmidt-Erfurth, U. Intraocular activation of angiogenic and inflammatory pathways in uveal melanoma. Retina 2012, 32, 1373–1384. [Google Scholar] [CrossRef]
- Wierenga, A.P.A.; Gezgin, G.; van Beelen, E.; Eikmans, M.; Spruyt-Gerritse, M.; Brouwer, N.J.; Versluis, M.; Verdijk, R.M.; van Duinen, S.G.; Marinkovic, M.; et al. Soluble HLA in the aqueous humour of uveal melanoma is associated with unfavourable tumour characteristics. Cancers 2019, 11, 1202. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.X.; Liu, Y.M.; Yang, L.; Xuan, Y.; Wei, W. Bin elevated VEGF-A & PLGF concentration in aqueous humor of patients with uveal melanoma following iodine-125 plaque radiotherapy. Int. J. Ophthalmol. 2020, 13, 599–605. [Google Scholar] [CrossRef]
- Lee, C.S.; Jun, I.H.; Kim, T.I.; Byeon, S.H.; Koh, H.J.; Lee, S.C. Expression of 12 cytokines in aqueous humour of uveal melanoma before and after combined ruthenium-106 brachytherapy and transpupillary thermotherapy. Acta Ophthalmol. 2012, 90, 314–320. [Google Scholar] [CrossRef]
- Ten Voorde, A.M.W.; Wierenga, A.P.A.; Nell, R.J.; van der Velden, P.A.; Luyten, G.P.M.; Verdijk, R.M.; Jager, M.J. In uveal melanoma, angiopoietin-2 but not angiopoietin-1 is increased in high-risk tumors, providing a potential druggable target. Cancers 2021, 13, 3986. [Google Scholar] [CrossRef]
- Dikovskaya, M.A.; Russkikh, G.S.; Loktev, K.V.; Johnston, T.P.; Gevorgyan, M.M.; Voronina, N.P.; Chernykh, V.V.; Trunov, A.N.; Korolenko, T.A. Cystatin C and cystatin SN as possible soluble tumor markers in malignant uveal melanoma. Radiol. Oncol. 2021, 56, 83–91. [Google Scholar] [CrossRef]
- Kujala, E.; Mäkitie, T.; Kivelä, T. Very long-term prognosis of patients with malignant uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2003, 44, 4651–4659. [Google Scholar] [CrossRef]
- Bol, K.F.; Donia, M.; Heegaard, S.; Kiilgaard, J.F.; Svane, I.M. Genetic biomarkers in melanoma of the ocular region: What the medical oncologist should know. Int. J. Mol. Sci. 2020, 21, 5231. [Google Scholar] [CrossRef]
- Schank, T.E.; Hassel, J.C. Immunotherapies for the treatment of uveal melanoma—History and future. Cancers 2019, 11, 1048. [Google Scholar] [CrossRef]
- Beasley, A.B.; Chen, F.K.; Isaacs, T.W.; Gray, E.S. Future perspectives of uveal melanoma blood based biomarkers. Br. J. Cancer 2022, 126, 1511–1528. [Google Scholar] [CrossRef]
- Gallenga, C.E.; Franco, E.; Adamo, G.G.; Violanti, S.S.; Tassinari, P.; Tognon, M.; Perri, P. Genetic basis and molecular mechanisms of uveal melanoma metastasis: A focus on prognosis. Front. Oncol. 2022, 12, 828112. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, M.F.B.; Marta, B.F.; Baameiro, N.L.; Santiago-Varela, M.; Silva-Rodríguez, P.; Blanco-Teijeiro, M.J.; Perez, M.P.; Ces, A.P. Blood biomarkers of uveal melanoma: Current perspectives. Clin. Ophthalmol. 2020, 14, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.Y.; Lai, T.T.; Liao, W.T.; Li, C.J. Clinicopathological and prognostic significance and molecular mechanisms governing uveal melanoma. Ther. Adv. Med. Oncol. 2020, 12, 17566. [Google Scholar] [CrossRef] [PubMed]
- Ortega, M.A.; Fraile-Martínez, O.; García-Honduvilla, N.; Coca, S.; Álvarez-Mon, M.; Buján, J.; Teus, M.A. Update on uveal melanoma: Translational research from biology to clinical practice (review). Int. J. Oncol. 2020, 57, 1262–1279. [Google Scholar] [CrossRef]
- Seedor, R.S.; Orloff, M.; Sato, T. Genetic landscape and emerging therapies in uveal melanoma. Cancers 2021, 13, 5503. [Google Scholar] [CrossRef]
- Jin, E.; Burnier, J.V. Liquid biopsy in uveal melanoma: Are we there yet? Ocul. Oncol. Pathol. 2021, 7, 1–16. [Google Scholar] [CrossRef]
- Howlett, S.; Carter, T.J.; Shaw, H.M.; Nathan, P.D. Tebentafusp: A first-in-class treatment for metastatic uveal melanoma. Ther. Adv. Med. Oncol. 2023, 15, 175883592311601. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).