The Pleiotropic Effects of YBX1 on HTLV-1 Transcription
Abstract
:1. Introduction
2. Results
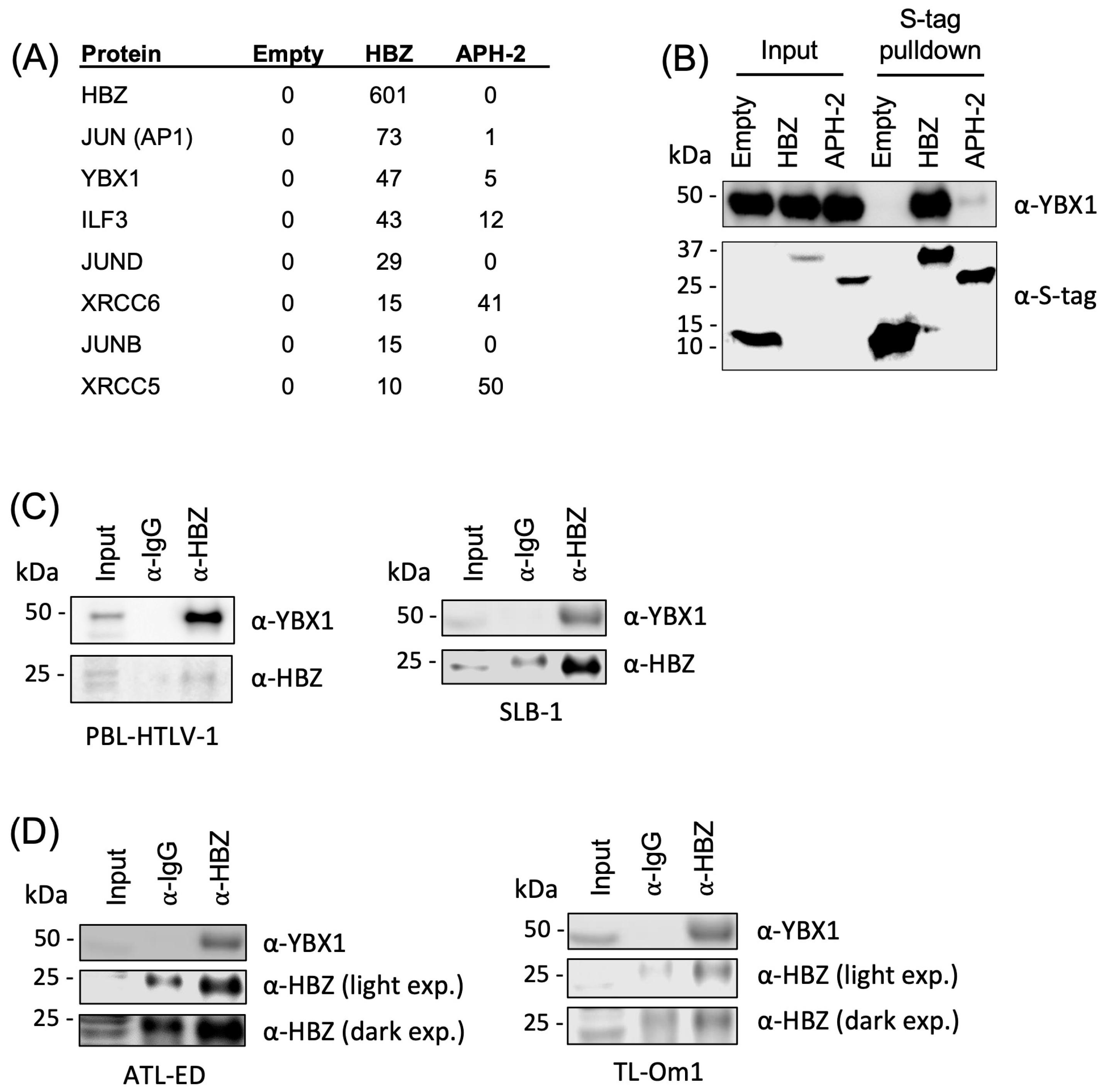
2.1. HBZ Interacts with the Cellular Factor YBX1
2.2. YBX1 Activates Viral Transcription
2.3. YBX1 Interacts with Tax
2.4. YBX1 Activates Sense and Antisense Viral Transcription
2.5. YBX1 Associates with the 5′ and 3′ Viral LTR
2.6. YBX1 Enhances Tax Binding and HBZ Represses YBX1 Transcriptional Activation
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Plasmids
4.3. S-Tag-Affinity Pulldown Assay
4.4. Mass Spectrometry and Proteomic Analysis
4.5. Immunoprecipitation
4.6. Transfections, Luciferase Reporter Assays, and p19 Gag ELISA
4.7. Immunoblotting
4.8. Lentiviral Production and Cell Transduction
4.9. Chromatin Immunoprecipitation (ChIP)
4.10. Quantitative RT-PCR (qRT-PCR)
4.11. Statistics
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gessain, A.; Cassar, O. Epidemiological Aspects and World Distribution of HTLV-1 Infection. Front. Microbiol. 2012, 3, 388. [Google Scholar] [CrossRef] [PubMed]
- Lairmore, M.D.; Franchini, G. Human T-cell leukemia virus types 1 and 2. In Fields Virology, 5th ed.; Lairmore, M.D., Franchini, G., Fields, B., Knipe, D.M., Howley, P.M., Eds.; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; pp. 2071–2106. [Google Scholar]
- Yoshida, M.; Miyoshi, I.; Hinuma, Y. Isolation and characterization of retrovirus from cell lines of human adult T-cell leukemia and its implication in the disease. Proc. Natl. Acad. Sci. USA 1982, 79, 2031–2035. [Google Scholar] [CrossRef]
- Poiesz, B.J.; Ruscetti, F.W.; Gazdar, A.F.; Bunn, P.A.; Minna, J.D.; Gallo, R.C. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc. Natl. Acad. Sci. USA 1980, 77, 7415–7419. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, T.; Yodoi, J.; Sagawa, K.; Takatsuki, K.; Uchino, H. Adult T-cell leukemia: Clinical and hematologic features of 16 cases. Blood 1977, 50, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Iwanaga, M.; Watanabe, T.; Yamaguchi, K. Adult T-Cell Leukemia: A Review of Epidemiological Evidence. Front. Microbiol. 2012, 3, 322. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.E.; Osame, M.; Kubota, H.; Igata, A.; Nishitani, H.; Maeda, Y.; Khabbaz, R.F.; Janssen, R.S. The risk of development of HTLV-I-associated myelopathy/tropical spastic paraparesis among persons infected with, HTLV-I. J. Acquir. Immune Defic. Syndr. 1990, 3, 1096–1101. [Google Scholar] [PubMed]
- Matsuoka, M.; Jeang, K.T. Human T-cell leukemia virus type 1 (HTLV-1) and leukemic transformation: Viral infectivity, Tax, HBZ and therapy. Oncogene 2011, 30, 1379–1389. [Google Scholar] [CrossRef]
- Zhao, L.J.; Giam, C.Z. Human T-cell lymphotropic virus type I (HTLV-I) transcriptional activator, Tax, enhances CREB binding to HTLV-I 21-base-pair repeats by protein-protein interaction. Proc. Natl. Acad. Sci. USA 1992, 89, 7070–7074. [Google Scholar] [CrossRef]
- Yin, M.J.; Gaynor, R.B. HTLV-1 21 bp repeat sequences facilitate stable association between Tax and CREB to increase CREB binding affinity. J. Mol. Biol. 1996, 264, 20–31. [Google Scholar] [CrossRef]
- Harrod, R.; Kuo, Y.L.; Tang, Y.; Yao, Y.; Vassilev, A.; Nakatani, Y.; Giam, C.Z. p300 and p300/cAMP-responsive element-binding protein associated factor interact with human T-cell lymphotropic virus type-1 Tax in a multi-histone acetyltransferase/activator-enhancer complex. J. Biol. Chem. 2000, 275, 11852–11857. [Google Scholar] [CrossRef]
- Harrod, R.; Tang, Y.; Nicot, C.; Lu, H.S.; Vassilev, A.; Nakatani, Y.; Giam, C.Z. An exposed KID-like domain in human T-cell lymphotropic virus type 1 Tax is responsible for the recruitment of coactivators CBP/p300. Mol. Cell. Biol. 1998, 18, 5052–5061. [Google Scholar] [CrossRef]
- Satou, Y.; Yasunaga, J.; Yoshida, M.; Matsuoka, M. HTLV-I basic leucine zipper factor gene mRNA supports proliferation of adult T cell leukemia cells. Proc. Natl. Acad. Sci. USA 2006, 103, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Arnold, J.; Yamamoto, B.; Li, M.; Phipps, A.J.; Younis, I.; Lairmore, M.D.; Green, P.L. Enhancement of infectivity and persistence in vivo by HBZ, a natural antisense coded protein of HTLV-1. Blood 2006, 107, 3976–3982. [Google Scholar] [CrossRef] [PubMed]
- Gaudray, G.; Gachon, F.; Basbous, J.; Biard-Piechaczyk, M.; Devaux, C.; Mesnard, J.-M. The complementary strand of the human T-cell leukemia virus type 1 RNA genome encodes a bZIP transcription factor that down-regulates viral transcription. J. Virol. 2002, 76, 12813–12822. [Google Scholar] [CrossRef]
- Lemasson, I.; Lewis, M.R.; Polakowski, N.; Hivin, P.; Cavanagh, M.H.; Thebault, S.; Barbeau, B.; Nyborg, J.K.; Mesnard, J.-M. Human T-cell leukemia virus type 1 (HTLV-1) bZIP protein interacts with the cellular transcription factor CREB to inhibit HTLV-1 transcription. J. Virol. 2007, 81, 1543–1553. [Google Scholar] [CrossRef] [PubMed]
- Clerc, I.; Polakowski, N.; Andre-Arpin, C.; Cook, P.; Barbeau, B.; Mesnard, J.-M.; Lemasson, I. An interaction between the human T cell leukemia virus type 1 basic leucine zipper factor, (HBZ) and the KIX domain of p300/CBP contributes to the down-regulation of tax-dependent viral transcription by, HBZ. J. Biol. Chem. 2008, 283, 23903–23913. [Google Scholar] [CrossRef]
- Zhao, T.; Yasunaga, J.; Satou, Y.; Nakao, M.; Takahashi, M.; Fujii, M.; Matsuoka, M. Human T-cell leukemia virus type 1 bZIP factor selectively suppresses the classical pathway of, NF-κB. Blood 2009, 113, 2755–2764. [Google Scholar] [CrossRef]
- Arnold, J.; Zimmerman, B.; Li, M.; Lairmore, M.D.; Green, P.L. Human T-cell leukemia virus type-1 antisense-encoded gene, Hbz, promotes T-lymphocyte proliferation. Blood 2008, 112, 3788–3797. [Google Scholar] [CrossRef]
- Maksimova, V.; Wilkie, T.; Smith, S.; Phelps, C.; Melvin, C.; Yu, L.; Niewiesk, S.; Green, P.L.; Panfil, A.R. HTLV-1 Hbz protein, but not hbz mRNA secondary structure, is critical for viral persistence and disease development. PLoS Pathog. 2023, 19, e1011459. [Google Scholar] [CrossRef]
- Katsuya, H.; Islam, S.; Tan, B.J.Y.; Ito, J.; Miyazato, P.; Matsuo, M.; Inada, Y.; Iwase, S.C.; Uchiyama, Y.; Hata, H.; et al. The Nature of the HTLV-1 Provirus in Naturally Infected Individuals Analyzed by the Viral DNA-Capture-Seq Approach. Cell Rep. 2019, 29, 724–735 e4. [Google Scholar] [CrossRef]
- Takeda, S.; Maeda, M.; Morikawa, S.; Taniguchi, Y.; Yasunaga, J.; Nosaka, K.; Tanaka, Y.; Matsuoka, M. Genetic and epigenetic inactivation of tax gene in adult T-cell leukemia cells. Int. J. Cancer 2004, 109, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Cavanagh, M.H.; Landry, S.; Audet, B.; Arpin-Andre, C.; Hivin, P.; Pare, M.E.; Thête, J.; Wattel, É.; Marriott, S.J.; Mesnard, J.-M.; et al. HTLV-I antisense transcripts initiating in the 3′LTR are alternatively spliced and polyadenylated. Retrovirology 2006, 3, 15. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Satou, Y.; Yasunaga, J.; Fujisawa, J.; Matsuoka, M. Transcriptional control of spliced and unspliced human T-cell leukemia virus type 1 bZIP factor (HBZ) gene. J. Virol. 2008, 82, 9359–9368. [Google Scholar] [CrossRef] [PubMed]
- Gazon, H.; Lemasson, I.; Polakowski, N.; Césaire, R.; Matsuoka, M.; Barbeau, B.; Mesnard, J.-M.; Peloponese, J.-M. Human T-cell leukemia virus type 1 (HTLV-1) bZIP factor requires cellular transcription factor JunD to upregulate HTLV-1 antisense transcription from the 3′ long terminal repeat. J. Virol. 2012, 86, 9070–9078. [Google Scholar] [CrossRef]
- Arpin-André, C.; Laverdure, S.; Barbeau, B.; Gross, A.; Mesnard, J.-M. Construction of a reporter vector for analysis of bidirectional transcriptional activity of retrovirus LTR. Plasmid 2014, 74, 45–51. [Google Scholar] [CrossRef]
- Madugula, K.K.; Joseph, J.; DeMarino, C.; Ginwala, R.; Teixeira, V.; Khan, Z.K.; Sales, D.; Wilson, S.; Kashanchi, F.; Rushing, A.W.; et al. Regulation of Human T-cell Leukemia Virus type 1 antisense promoter by Myocyte Enhancer Factor-2C in the context of Adult T-cell leukemia and lymphoma. Haematologica 2022, 107, 2928–2943. [Google Scholar] [CrossRef]
- Satou, Y.; Miyazato, P.; Ishihara, K.; Yaguchi, H.; Melamed, A.; Miura, M.; Fukuda, A.; Nosaka, K.; Watanabe, T.; Rowan, A.G.; et al. The retrovirus HTLV-1 inserts an ectopic CTCF-binding site into the human genome. Proc. Natl. Acad. Sci. USA 2016, 113, 3054–3059. [Google Scholar] [CrossRef]
- Matsuo, M.; Ueno, T.; Monde, K.; Sugata, K.; Tan, B.J.Y.; Rahman, A.; Miyazato, P.; Uchiyama, K.; Islam, S.; Katsuya, H.; et al. Identification and characterization of a novel enhancer in the HTLV-1 proviral genome. Nat. Commun. 2022, 13, 2405. [Google Scholar] [CrossRef]
- Maksimova, V.; Smith, S.; Seth, J.; Phelps, C.; Niewiesk, S.; Satou, Y.; Green, P.L.; Panfil, A.R. HTLV-1 intragenic viral enhancer influences immortalization phenotype in vitro, but is dispensable for persistence and disease development in animal models. Front. Immunol. 2022, 13, 954077. [Google Scholar] [CrossRef]
- Koiwa, T.; Hamano-Usami, A.; Ishida, T.; Okayama, A.; Yamaguchi, K.; Kamihira, S.; Watanabe, T. 5′-long terminal repeat-selective CpG methylation of latent human T-cell leukemia virus type 1 provirus in vitro and in vivo. J. Virol. 2002, 76, 9389–9397. [Google Scholar] [CrossRef]
- Taniguchi, Y.; Nosaka, K.; Yasunaga, J.; Maeda, M.; Mueller, N.; Okayama, A.; Matsuoka, M. Silencing of human T-cell leukemia virus type I gene transcription by epigenetic mechanisms. Retrovirology 2005, 2, 64. [Google Scholar] [CrossRef] [PubMed]
- Kannagi, M. Immunologic control of human T-cell leukemia virus type I and adult T-cell leukemia. Int. J. Hematol. 2007, 86, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Laverdure, S.; Polakowski, N.; Hoang, K.; Lemasson, I. Permissive Sense and Antisense Transcription from the 5′ and 3′ Long Terminal Repeats of Human T-Cell Leukemia Virus Type 1. J. Virol. 2016, 90, 3600–3610. [Google Scholar] [CrossRef] [PubMed]
- Sangermano, F.; Delicato, A.; Calabro, V. Y box binding protein 1 (YB-1) oncoprotein at the hub of DNA proliferation, damage and cancer progression. Biochimie 2020, 179, 205–216. [Google Scholar] [CrossRef]
- Lyabin, D.N.; Eliseeva, I.A.; Ovchinnikov, L.P. YB-1 protein: Functions and regulation. Wiley Interdiscip. Rev. RNA 2014, 5, 95–110. [Google Scholar] [CrossRef]
- Kuwano, M.; Shibata, T.; Watari, K.; Ono, M. Oncogenic Y-box binding protein-1 as an effective therapeutic target in drug-resistant cancer. Cancer Sci. 2019, 110, 1536–1543. [Google Scholar] [CrossRef]
- Gieseler-Halbach, S.; Meltendorf, S.; Pierau, M.; Weinert, S.; Heidel, F.H.; Fischer, T.; Handschuh, J.; Braun-Dullaeus, R.C.; Schrappe, M.; Lindquist, J.A.; et al. RSK-mediated nuclear accumulation of the cold-shock Y-box protein-1 controls proliferation of T cells and T-ALL blasts. Cell Death Differ. 2017, 24, 371–383. [Google Scholar] [CrossRef]
- Shah, A.; Plaza-Sirvent, C.; Weinert, S.; Buchbinder, J.H.; Lavrik, I.N.; Mertens, P.R.; Schmitz, I.; Lindquist, J.A. YB-1 Mediates TNF-Induced Pro-Survival Signaling by Regulating NF-κB Activation. Cancers 2020, 12, 2188. [Google Scholar] [CrossRef]
- Weydert, C.; van Heertum, B.; Dirix, L.; De Houwer, S.; De Wit, F.; Mast, J.; Husson, S.J.; Busschots, K.; König, R.; Gijsbers, R.; et al. Y-box-binding protein 1 supports the early and late steps of HIV replication. PLoS ONE 2018, 13, e0200080. [Google Scholar] [CrossRef]
- Mu, X.; Li, W.; Wang, X.; Gao, G. YB-1 stabilizes HIV-1 genomic RNA and enhances viral production. Protein Cell 2013, 4, 591–597. [Google Scholar] [CrossRef]
- Patzina, C.; Botting, C.H.; Garcia-Sastre, A.; Randall, R.E.; Hale, B.G. Human interactome of the influenza B virus NS1 protein. J. Gen. Virol. 2017, 98, 2267–2273. [Google Scholar] [CrossRef] [PubMed]
- Diosa-Toro, M.; Kennedy, D.R.; Chuo, V.; Popov, V.L.; Pompon, J.; Garcia-Blanco, M.A. Y-Box Binding Protein 1 Interacts with Dengue Virus Nucleocapsid and Mediates Viral Assembly. mBio 2022, 13, e0019622. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, X.; Gao, G. Expression of YB-1 enhances production of murine leukemia virus vectors by stabilizing genomic viral RNA. Protein Cell 2012, 3, 943–949. [Google Scholar] [CrossRef] [PubMed]
- Chatel-Chaix, L.; Germain, M.A.; Motorina, A.; Bonneil, E.; Thibault, P.; Baril, M.; Lamarre, D. A host YB-1 ribonucleoprotein complex is hijacked by hepatitis C virus for the control of NS3-dependent particle production. J. Virol. 2013, 87, 11704–11720. [Google Scholar] [CrossRef]
- Ansari, S.A.; Safak, M.; Gallia, G.L.; Sawaya, B.E.; Amini, S.; Khalili, K. Interaction of YB-1 with human immunodeficiency virus type 1 Tat and TAR RNA modulates viral promoter activity. J. Gen. Virol. 1999, 80 Pt 10, 2629–2938. [Google Scholar] [CrossRef]
- Bann, D.V.; Beyer, A.R.; Parent, L.J. A murine retrovirus co-Opts YB-1, a translational regulator and stress granule-associated protein, to facilitate virus assembly. J. Virol. 2014, 88, 4434–4450. [Google Scholar] [CrossRef]
- Kashanchi, F.; Duvall, J.F.; Dittmer, J.; Mireskandari, A.; Reid, R.L.; Gitlin, S.D.; Brady, J.N. Involvement of transcription factor YB-1 in human T-cell lymphotropic virus type I basal gene expression. J. Virol. 1994, 68, 561–565. [Google Scholar] [CrossRef]
- Martinez, M.P.; Al-Saleem, J.; Green, P.L. Comparative virology of HTLV-1 and HTLV-2. Retrovirology 2019, 16, 21. [Google Scholar] [CrossRef]
- Panfil, A.R.; Dissinger, N.J.; Howard, C.M.; Murphy, B.M.; Landes, K.; Fernandez, S.A.; Green, P.L. Functional Comparison of HBZ and the Related APH-2 Protein Provides Insight into Human T-Cell Leukemia Virus Type 1 Pathogenesis. J. Virol. 2016, 90, 3760–3772. [Google Scholar] [CrossRef]
- Hivin, P.; Frederic, M.; Arpin-Andre, C.; Basbous, J.; Gay, B.; Thebault, S.; Mesnard, J.-M. Nuclear localization of HTLV-I bZIP factor (HBZ) is mediated by three distinct motifs. J. Cell Sci. 2005, 118, 1355–1362. [Google Scholar] [CrossRef]
- Kloks, C.P.; Spronk, C.A.; Lasonder, E.; Hoffmann, A.; Vuister, G.W.; Grzesiek, S.; Hilbers, C.W. The solution structure and DNA-binding properties of the cold-shock domain of the human Y-box protein YB-1. J. Mol. Biol. 2002, 316, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Nagata, K.; Ohtani, K.; Nakamura, M.; Sugamura, K. Activation of endogenous c-fos proto-oncogene expression by human T-cell leukemia virus type I-encoded p40tax protein in the human T-cell line, Jurkat. J. Virol. 1989, 63, 3220–3226. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, K.; Tsujimoto, A.; Tsukahara, T.; Numata, N.; Miura, S.; Sugamura, K.; Nakamura, M. Molecular mechanisms of promoter regulation of the gp34 gene that is trans-activated by an oncoprotein Tax of human T cell leukemia virus type I. J. Biol. Chem. 1998, 273, 14119–14129. [Google Scholar] [CrossRef] [PubMed]
- Giam, C.Z.; Semmes, O.J. HTLV-1 Infection Adult T-Cell Leukemia/Lymphoma-ATale of Two Proteins: Tax, HBZ. Viruses 2016, 8, 161. [Google Scholar] [CrossRef] [PubMed]
- Sodroski, J.G.; Rosen, C.A.; Haseltine, W.A. Trans-acting transcriptional activation of the long terminal repeat of human T lymphotropic viruses in infected cells. Science 1984, 225, 381–385. [Google Scholar] [CrossRef]
- Chen, I.S.; Slamon, D.J.; Rosenblatt, J.D.; Shah, N.P.; Quan, S.G.; Wachsman, W. The x gene is essential for HTLV replication. Science 1985, 229, 54–58. [Google Scholar] [CrossRef]
- Felber, B.K.; Paskalis, H.; Kleinman-Ewing, C.; Wong-Staal, F.; Pavlakis, G.N. The pX protein of HTLV-I is a transcriptional activator of its long terminal repeats. Science 1985, 229, 675–679. [Google Scholar] [CrossRef]
- Fujisawa, J.; Seiki, M.; Kiyokawa, T.; Yoshida, M. Functional activation of the long terminal repeat of human T-cell leukemia virus type I by a trans-acting factor. Proc. Natl. Acad. Sci. USA 1985, 82, 2277–2281. [Google Scholar] [CrossRef]
- Geleziunas, R.; Ferrell, S.; Lin, X.; Mu, Y.; Cunningham, E.T., Jr.; Grant, M.; Connelly, M.A.; Hambor, J.E.; Marcu, K.B.; Greene, W.C. Human T-cell leukemia virus type 1 Tax induction of NF-κB involves activation of the IκB kinase α (IKKα) and IKKβ cellular kinases. Mol. Cell. Biol. 1998, 18, 5157–5165. [Google Scholar] [CrossRef]
- Chu, Z.L.; Shin, Y.A.; Yang, J.M.; DiDonato, J.A.; Ballard, D.W. IKKgamma mediates the interaction of cellular IkappaB kinases with the tax transforming protein of human T cell leukemia virus type 1. J. Biol. Chem. 1999, 274, 15297–15300. [Google Scholar] [CrossRef]
- Arima, N.; Tei, C. HTLV-I Tax related dysfunction of cell cycle regulators and oncogenesis of adult T cell leukemia. Leuk. Lymphoma 2001, 40, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.P.; Irvine, J.; Blyth, K.; Cameron, E.R.; Onions, D.E.; Campbell, M.E. Tumours derived from HTLV-I tax transgenic mice are characterized by enhanced levels of apoptosis and oncogene expression. J. Pathol. 1998, 186, 209–214. [Google Scholar] [CrossRef]
- Rivera-Walsh, I.; Waterfield, M.; Xiao, G.; Fong, A.; Sun, S.C. NF-κB signaling pathway governs TRAIL gene expression and human T-cell leukemia virus-I Tax-induced T-cell death. J. Biol. Chem. 2001, 276, 40385–40388. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Higuchi, M.; Makokha, G.N.; Matsuki, H.; Yoshita, M.; Tanaka, Y.; Fujii, M. HTLV-1 Tax oncoprotein stimulates ROS production and apoptosis in T cells by interacting with USP10. Blood 2013, 122, 715–725. [Google Scholar] [CrossRef] [PubMed]
- Chlichlia, K.; Moldenhauer, G.; Daniel, P.T.; Busslinger, M.; Gazzolo, L.; Schirrmacher, V.; Khazaie, K. Immediate effects of reversible HTLV-1 tax function: T-cell activation and apoptosis. Oncogene 1995, 10, 269–277. [Google Scholar]
- Zhi, H.; Yang, L.; Kuo, Y.L.; Ho, Y.K.; Shih, H.M.; Giam, C.Z. NF-κB hyper-activation by HTLV-1 tax induces cellular senescence, but can be alleviated by the viral anti-sense protein HBZ. PLoS Pathog. 2011, 7, e1002025. [Google Scholar] [CrossRef]
- Mahgoub, M.; Yasunaga, J.I.; Iwami, S.; Nakaoka, S.; Koizumi, Y.; Shimura, K.; Matsuoka, M. Sporadic on/off switching of HTLV-1 Tax expression is crucial to maintain the whole population of virus-induced leukemic cells. Proc. Natl. Acad. Sci. USA 2018, 115, E1269–E1278. [Google Scholar] [CrossRef]
- Kawatsuki, A.; Yasunaga, J.I.; Mitobe, Y.; Green, P.L.; Matsuoka, M. HTLV-1 bZIP factor protein targets the Rb/E2F-1 pathway to promote proliferation and apoptosis of primary CD4(+) T cells. Oncogene 2016, 35, 4509–4517. [Google Scholar] [CrossRef]
- Ashizuka, M.; Fukuda, T.; Nakamura, T.; Shirasuna, K.; Iwai, K.; Izumi, H.; Kohno, K.; Kuwano, M.; Uchiumi, T. Novel translational control through an iron-responsive element by interaction of multifunctional protein YB-1 and IRP2. Mol. Cell. Biol. 2002, 22, 6375–6383. [Google Scholar] [CrossRef]
- Matsumoto, K.; Tanaka, K.J.; Tsujimoto, M. An acidic protein, YBAP1, mediates the release of YB-1 from mRNA and relieves the translational repression activity of YB-1. Mol. Cell. Biol. 2005, 25, 1779–1792. [Google Scholar] [CrossRef]
- Shnyreva, M.; Schullery, D.S.; Suzuki, H.; Higaki, Y.; Bomsztyk, K. Interaction of two multifunctional proteins. Heterogeneous nuclear ribonucleoprotein K and Y-box-binding protein. J. Biol. Chem. 2000, 275, 15498–15503. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, D.; Fu, Q.; Wang, S.; Wang, Z.; Zhang, X.; Chen, X.; Zhu, X.; An, N.; Chen, Y.; et al. YBX1 as an oncogenic factor in T-cell acute lymphoblastic leukemia. Blood Adv. 2023. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Kannian, P.; Dissinger, N.; Haines, R.; Niewiesk, S.; Green, P.L. Human T-cell leukemia virus type 2 antisense viral protein 2 is dispensable for in vitro immortalization but functions to repress early virus replication in vivo. J. Virol. 2012, 86, 8412–8421. [Google Scholar] [CrossRef]
- Panfil, A.R.; Al-Saleem, J.; Howard, C.M.; Shkriabai, N.; Kvaratskhelia, M.; Green, P.L. Stability of the HTLV-1 Antisense-Derived Protein, HBZ, Is Regulated by the E3 Ubiquitin-Protein Ligase, UBR5. Front. Microbiol. 2018, 9, 80. [Google Scholar] [CrossRef] [PubMed]
- Landthaler, M.; Gaidatzis, D.; Rothballer, A.; Chen, P.Y.; Soll, S.J.; Dinic, L.; Ojo, T.; Hafner, M.; Zavolan, M.; Tuschl, T. Molecular characterization of human Argonaute-containing ribonucleoprotein complexes and their bound target mRNAs. RNA 2008, 14, 2580–2596. [Google Scholar] [CrossRef]
- Anderson, M.D.; Ye, J.; Xie, L.; Green, P.L. Transformation studies with a human T-cell leukemia virus type 1 molecular clone. J. Virol. Methods 2004, 116, 195–202. [Google Scholar] [CrossRef]
- Younis, I.; Khair, L.; Dundr, M.; Lairmore, M.D.; Franchini, G.; Green, P.L. Repression of human T-cell leukemia virus type 1 and type 2 replication by a viral mRNA-encoded posttranscriptional regulator. J. Virol. 2004, 78, 11077–11083. [Google Scholar] [CrossRef]
- Al-Saleem, J.; Dirksen, W.P.; Martinez, M.P.; Shkriabai, N.; Kvaratskhelia, M.; Ratner, L.; Green, P.L. HTLV-1 Tax-1 interacts with SNX27 to regulate cellular localization of the HTLV-1 receptor molecule, GLUT1. PLoS ONE 2019, 14, e0214059. [Google Scholar] [CrossRef]
- Jain, P.; Lavorgna, A.; Sehgal, M.; Gao, L.; Ginwala, R.; Sagar, D.; Harhaj, E.W.; Khan, Z.K. Myocyte enhancer factor (MEF)-2 plays essential roles in T-cell transformation associated with HTLV-1 infection by stabilizing complex between Tax, CREB. Retrovirology 2015, 12, 23. [Google Scholar] [CrossRef]
- Panfil, A.R.; Al-Saleem, J.; Howard, C.M.; Mates, J.M.; Kwiek, J.J.; Baiocchi, R.A.; Green, P.L. PRMT5 Is Upregulated in HTLV-1-Mediated T-Cell Transformation and Selective Inhibition Alters Viral Gene Expression and Infected Cell Survival. Viruses 2015, 8, 7. [Google Scholar] [CrossRef]
- Subbaiah, V.K.; Zhang, Y.; Rajagopalan, D.; Abdullah, L.N.; Yeo-Teh, N.S.; Tomaić, V.; Banks, L.; Myers, M.P.; Chow, E.K.; Jha, S. E3 ligase EDD1/UBR5 is utilized by the HPV E6 oncogene to destabilize tumor suppressor TIP60. Oncogene 2016, 35, 2062–2074. [Google Scholar] [CrossRef] [PubMed]








| Deletion Mutant | Primer Sequence |
|---|---|
| ∆A/P (∆1–55) | Forward primer: 5′-GCAACGAAGGTTTTGGGAACAGTAAAATG-3′ Reverse primer: 5′-CAAAACCTTCGTTGCGGTTCCCATGGTGG-3′ |
| ∆CSD (∆56–128) | Forward primer: 5′-GGTGGTGTTCCAGTTCAAGGCAGTAAATATG-3′ Reverse primer: 5′-GAACTGGAACACCACCGATGACCTTCTTGTC-3′ |
| ∆CTD (∆129–324) | Forward primer: 5′-CAAATGTTACAGGTCCTTAAGCGGCCGCACTC-3′ Reverse primer: 5′-GGACCTGTAACATTTGCTGCCTCCGCAC-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, S.; Seth, J.; Midkiff, A.; Stahl, R.; Syu, Y.-C.; Shkriabai, N.; Kvaratskhelia, M.; Musier-Forsyth, K.; Jain, P.; Green, P.L.; et al. The Pleiotropic Effects of YBX1 on HTLV-1 Transcription. Int. J. Mol. Sci. 2023, 24, 13119. https://doi.org/10.3390/ijms241713119
Smith S, Seth J, Midkiff A, Stahl R, Syu Y-C, Shkriabai N, Kvaratskhelia M, Musier-Forsyth K, Jain P, Green PL, et al. The Pleiotropic Effects of YBX1 on HTLV-1 Transcription. International Journal of Molecular Sciences. 2023; 24(17):13119. https://doi.org/10.3390/ijms241713119
Chicago/Turabian StyleSmith, Susan, Jaideep Seth, Amanda Midkiff, Rachel Stahl, Yu-Ci Syu, Nikoloz Shkriabai, Mamuka Kvaratskhelia, Karin Musier-Forsyth, Pooja Jain, Patrick L. Green, and et al. 2023. "The Pleiotropic Effects of YBX1 on HTLV-1 Transcription" International Journal of Molecular Sciences 24, no. 17: 13119. https://doi.org/10.3390/ijms241713119





