Role of Soluble ST2 Biomarker in Predicting Recurrence of Atrial Fibrillation after Electrical Cardioversion or Pulmonary Vein Isolation
Abstract
:1. Introduction
2. Results
2.1. Baseline Characteristics
2.2. Characteristics of Patients with Atrial Fibrillation vs. Those of Control Participants
2.3. Characteristics of Patients According to the Procedure Performed
2.4. Recurrence of AF and ST2S Biomarker
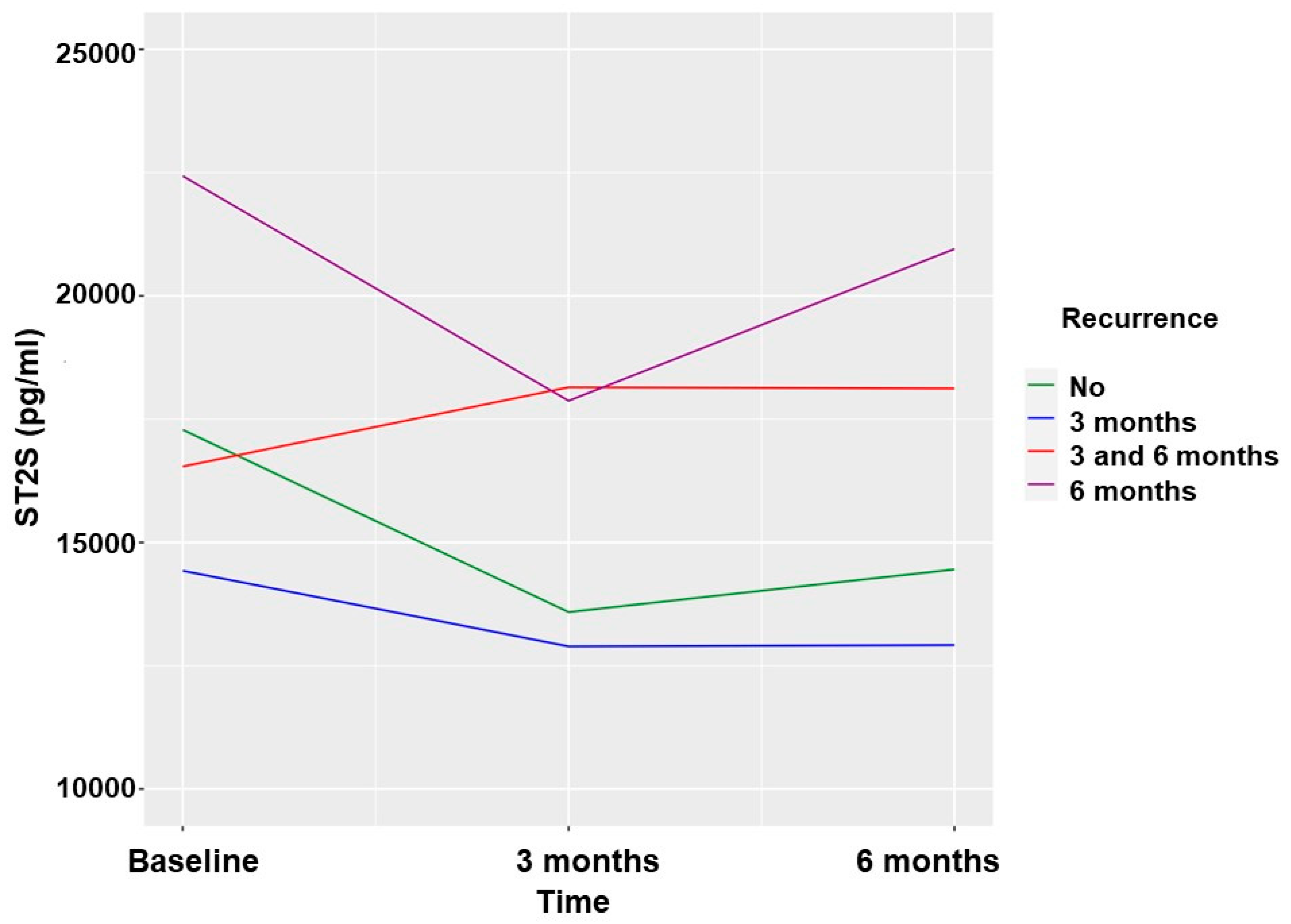
2.5. Recurrence of AF and sST2 Biomarker Patients Undergoing CVE
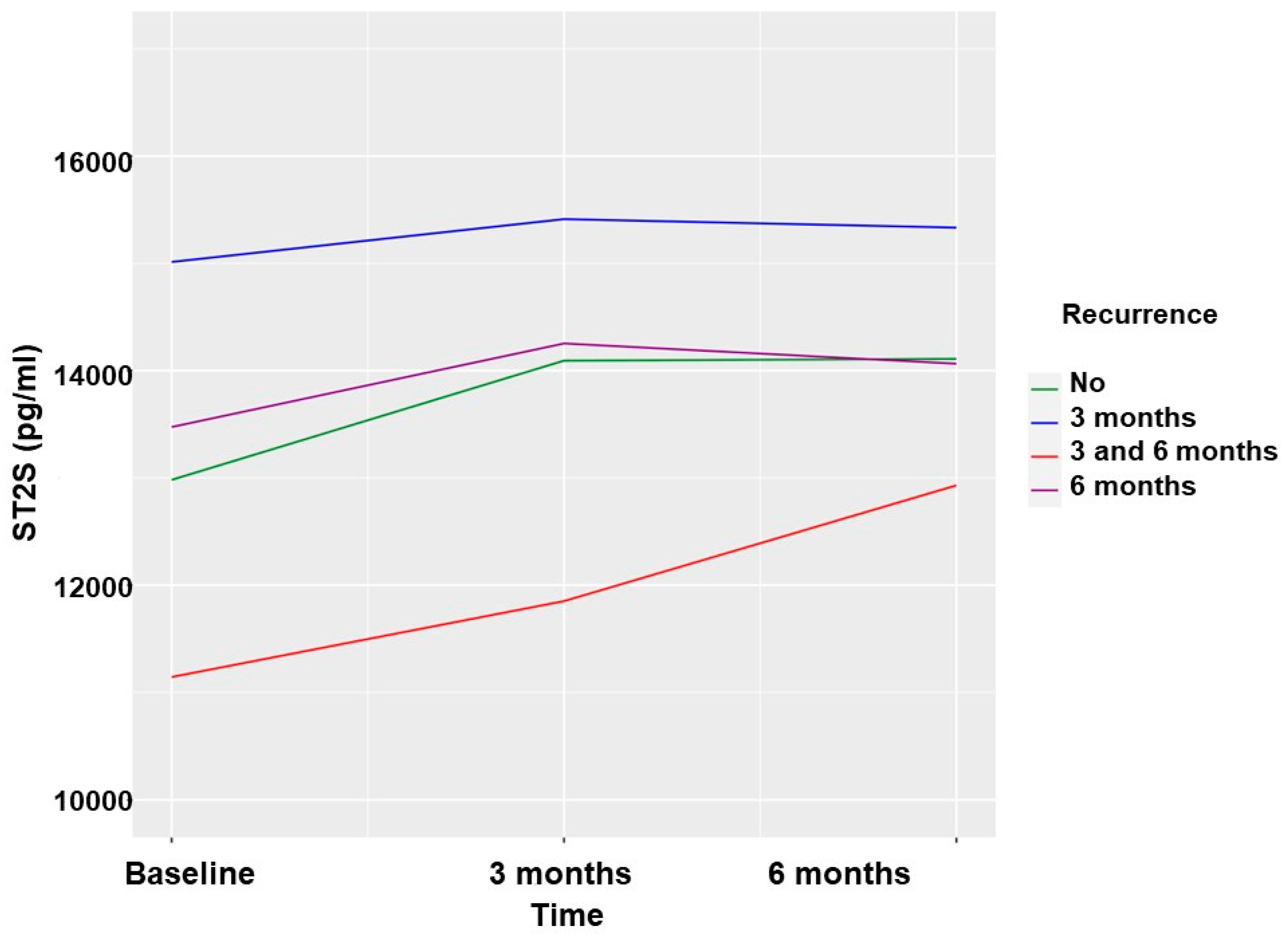
2.6. Recurrence of AF and sST2 in Patients Undergoing PVI
2.7. Recurrence of AF and sST2 Biomarker in the Global Cohort Excluding Patients Who Underwent a Procedure during Follow-Up
2.8. Predictive Capacity of the sST2 Biomarker for AF Recurrence
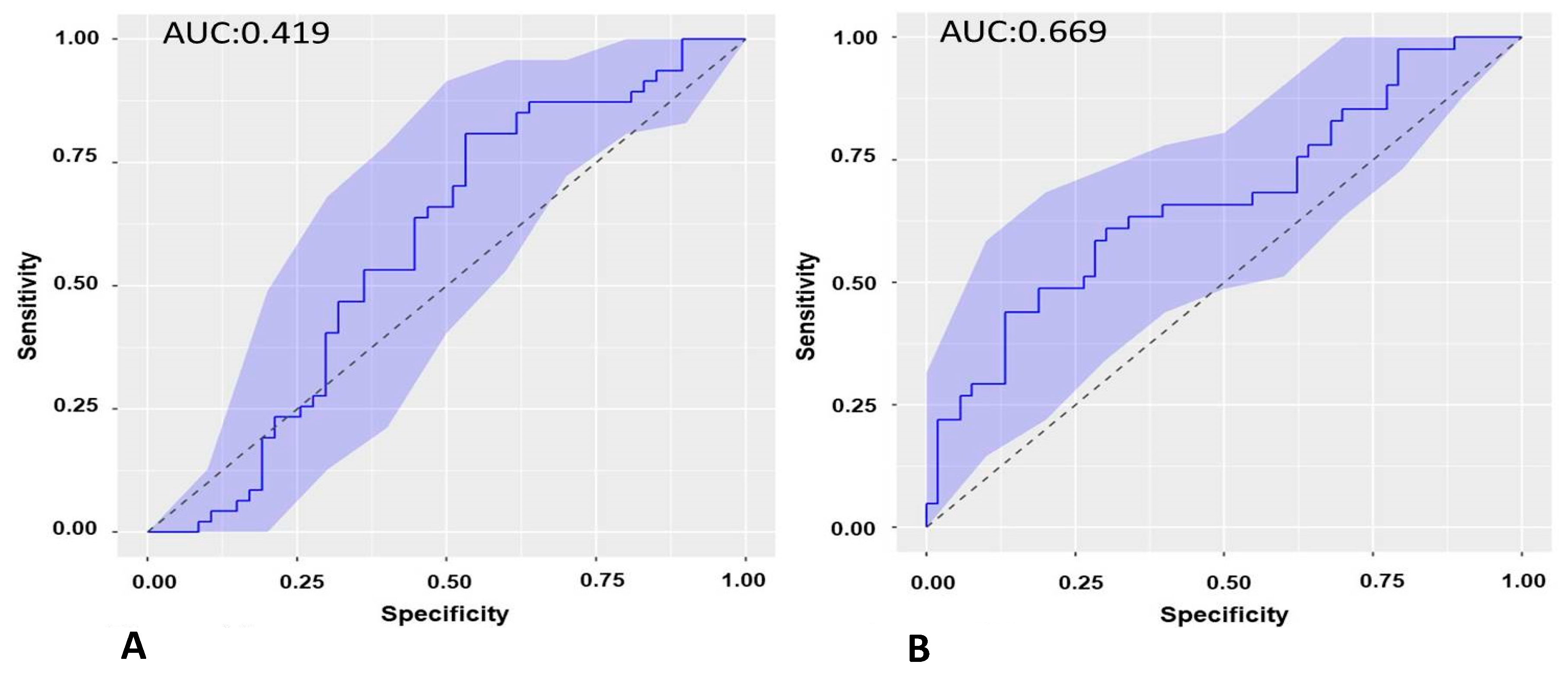
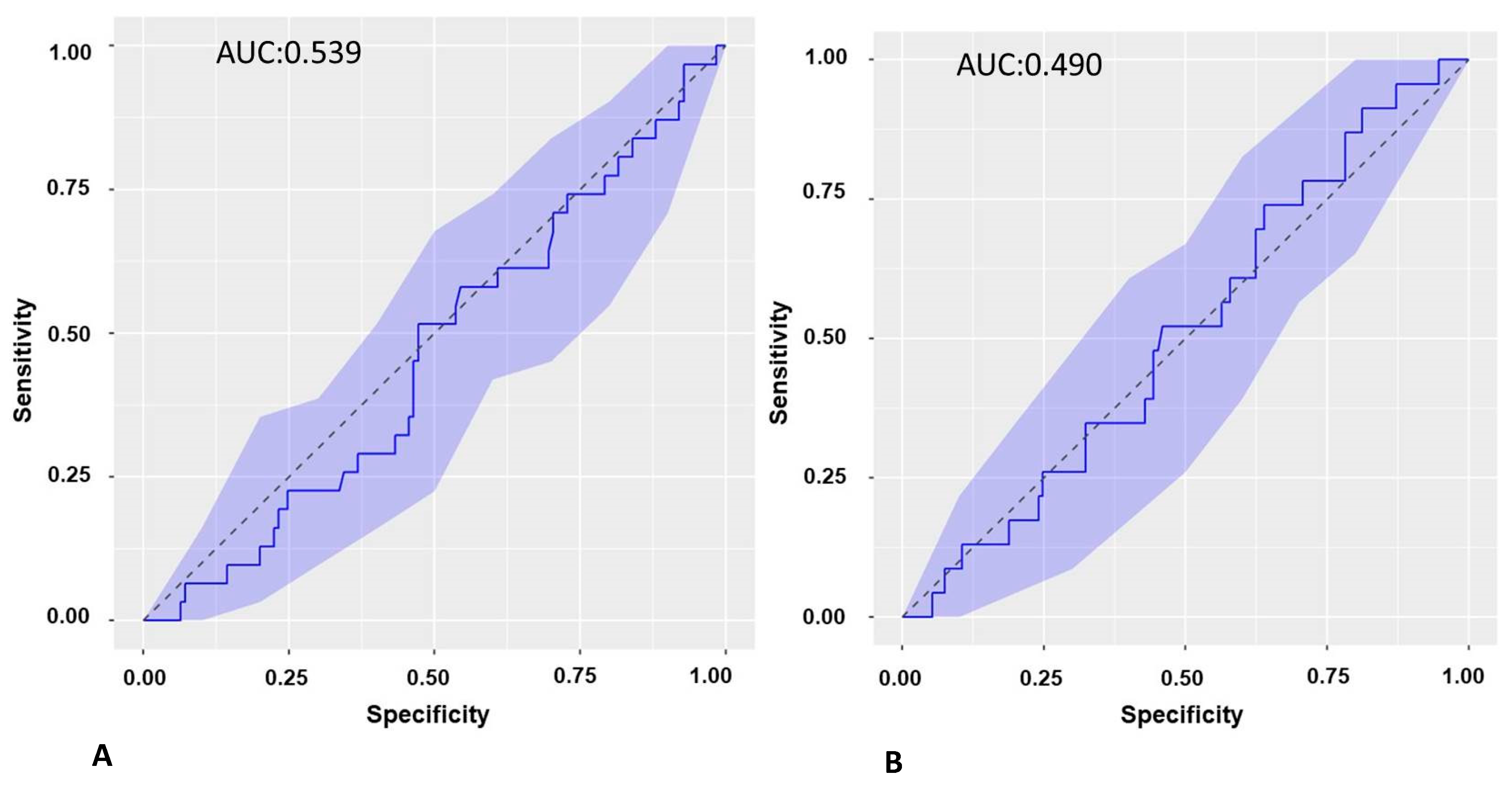
2.8.1. ROC Curve of the Baseline Biomarker (sST2.0) in the Cohort of Patients Undergoing ECV and Recurrence at 3 Months
2.8.2. ROC Curve of the Biomarker sST2 at 3-Month Follow-Up (sST2.1) in the Cohort of Patients Undergoing ECV and Recurrence from 3 to 6 Months
2.8.3. ROC Curve of the Biomarker sST2.0 in the Cohort of Patients Undergoing PVI and Recurrence at 3 Months
2.8.4. ROC Curve of the Biomarker ST2S.1 in the Cohort of Patients Undergoing PVI and Recurrence from 3 to 6 Months
2.9. Logistic Regression Models to Predict AF Recurrence
2.9.1. Logistic Regression Models in Patients Undergoing ECV
2.9.2. Logistic Regression Models in Patients Undergoing PVI
2.10. sST2 Biomarker and LA Low-Voltage Areas
3. Discussion
3.1. Baseline sST2 in AF Patients and Controls
3.2. Baseline sST2 and AF Recurrence
3.3. sST2 at 3 Months of Follow-Up and AF Recurrence
3.4. sST2 in PVI Patients and Fibrosis
3.5. Limitations
4. Methods and Materials
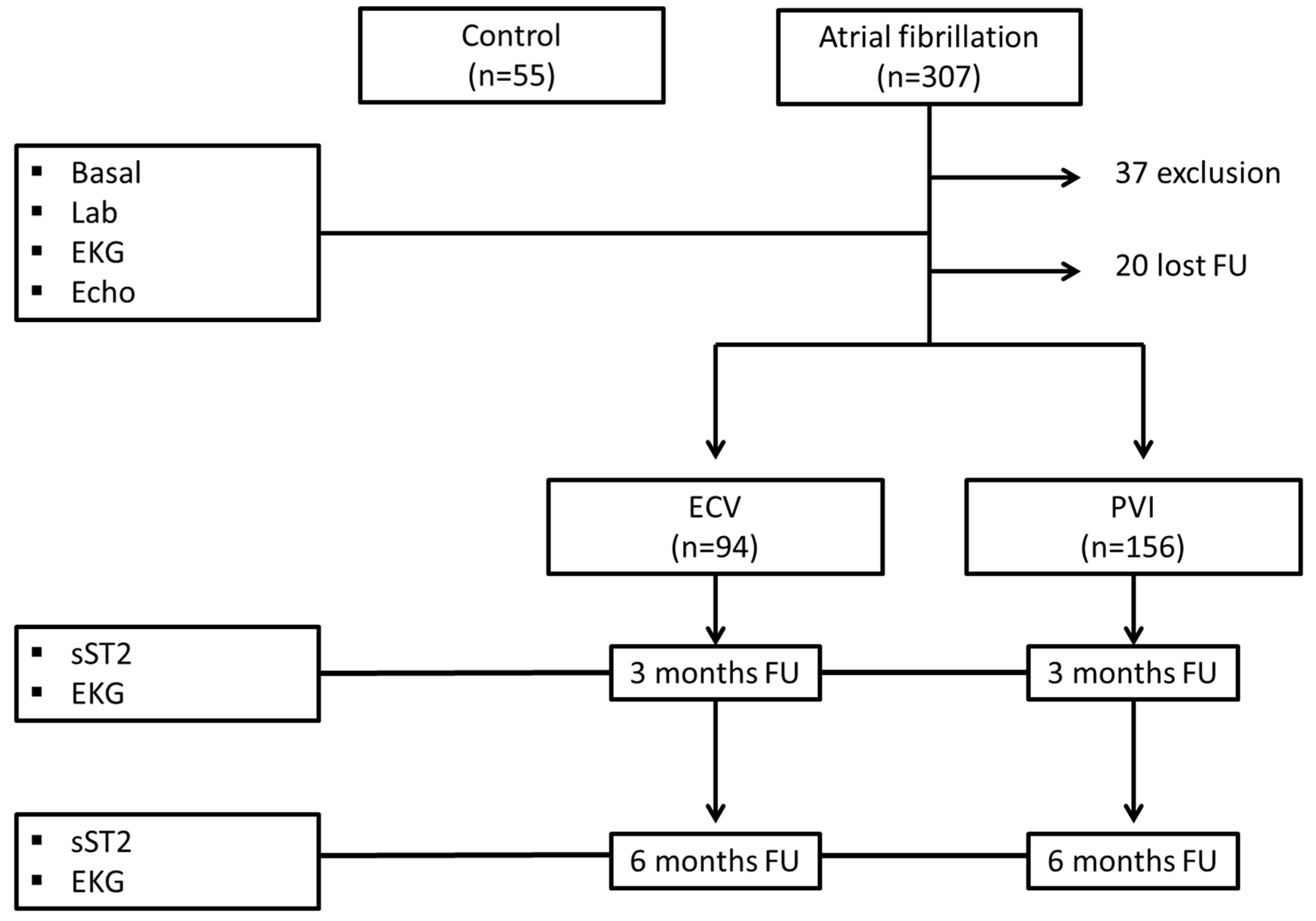
4.1. Study Design
4.2. Study Population
4.2.1. Inclusion Criteria
4.2.2. Exclusion Criteria
4.3. sST2 Measurement
4.4. AF Ablation/Electrical Cardioversion
4.5. Follow-Up
4.6. Statistical Analysis
4.7. Ethical Considerations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| sST2 | soluble suppression of tumorigenicity 2 |
| AF | atrial fibrillation |
| ECV | electrical cardioversion |
| PVI | pulmonary vein ablation |
| HF | heart failure |
| LVEF | left ventricular ejection fraction |
References
- Li, H.; Tago, K.; Io, K.; Kuroiwa, K.; Arai, T.; Iwahana, H.; Tominaga, S.-I.; Yanagisawa, K. The Cloning and Nucleotide Sequence of Human ST2L cDNA. Genomics 2000, 67, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, S. A putative protein of a growth specific cDNA from BALB/c-3T3 cells is highly similar to the extracellular portion of mouse interleukin 1 receptor. FEBS Lett. 1989, 258, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Demyanets, S.; Kaun, C.; Pentz, R.; Krychtiuk, K.A.; Rauscher, S.; Pfaffenberger, S.; Zuckermann, A.; Aliabadi, A.; Gröger, M.; Maurer, G.; et al. Components of the interleukin-33/ST2 system 246 are differentially expressed and regulated in human cardiac cells and in cells of the cardiac vasculature. J. Mol. Cell Cardiol. 2013, 60, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Figal, D.A.; Manzano-Fernández, S.; Boronat, M.; Casas, T.; Garrido, I.P.; Bonaque, J.C.; Pastor-Perez, F.; Valdés, M.; Januzzi, J.L. Soluble ST2, high-sensitivity troponin T- and N-terminal pro-B-type natriuretic peptide: Complementary role for risk stratification in acutely decompensated heart failure. Eur. J. Heart Fail. 2011, 13, 718–725. [Google Scholar] [CrossRef]
- Brown, A.M.; Wu, A.H.; Clopton, P.; Robey, J.L.; Hollander, J.E. ST2 in emergency department chest pain patients with potential acute coronary syndromes. Ann. Emerg. Med. 2007, 50, 153–158.e1. [Google Scholar] [CrossRef]
- Demyanets, S.; Speidl, W.; Tentzeris, I.; Jarai, R.; Katsaros, K.; Farhan, S.; Krychtiuk, K.; Anna Wonnerth, A.; Weiss, T.; Huber, K.; et al. Soluble ST2 and Interleukin-33 Levels in Coronary Artery Disease: Relation to Disease Activity and Adverse Outcome. PLoS ONE 2014, 9, e95055. [Google Scholar] [CrossRef]
- Bayes, A.; Antonio, M.; Vila, J.; Peñafiel, J.; Galán, A.; Barallat, J.; Zamora, E.; Urrutia, A.; Lupón, J. Head-to-head comparison of 2 myocardial fibrosis biomarkers for long-term heart failure risk stratification. ST2 versus Galectin-3. J. Am. Coll. Cardiol. 2014, 63, 158–166. [Google Scholar] [CrossRef]
- Nortamo, S.; Ukkola, O.; Lepojärvii, S.; Kenttä, T.; Kiviniemi, A.; Junttila, J.; Huikuri, H.; Perkiömäki, J. Association of sST2 and hs-CRP leveles with new onset of atrial fibrillation in coronary artery disease. Int. J. Cardiol. 2017, 248, 173–178. [Google Scholar] [CrossRef]
- Bartunek, J.; Delrue, L.; Van Durme, F.; Muller, O.; Casselman, F.; De Wiest, B.; Croes, R.; Verstreken, S.; Goethals, M.; de Raedt, H.; et al. Nonmyocardial production of ST2 protein in human hypertrophy and failure is related to diastolic load. J. Am. Coll. Cardiol. 2008, 52, 2166–2174. [Google Scholar] [CrossRef]
- See, V.; Dickfeld, T.; Shorofsky, S.; Saliaris, A.; Remo, B.; Klein, T.; Zohlman, A.; Christenson, R.; DeFilippi, C. Soluble ST2 and high sensitivity cardiac troponin-T levels predict atrial fibrillation ablation outcome. J. Am. Coll. Cardiol. 2016, 67, 840. [Google Scholar] [CrossRef]
- Alkhiary Abdalaal, M.; El-Sabbagh, A. Serum Concentrations of interleukin-33 and its soluble receptor sST2 in patients with persistent atrial fibrillation. J. Mol. Biomark. Diagn. 2015, 6, 3. [Google Scholar]
- Chen CQu, X.; Gao, Z.; Zheng, G.; Wang, Y.; Chen, X.; Li, H.; Huang, W.; Zhou, H. Soluble ST2s in patients with non valvular atrial fibrillation and prediction of heart failure. Int. Heart J. 2018, 59, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Merino-Merino, A.; Saez-Maleta, R.; Salgado-Aranda, R.; Alkassam-Martinez, D.; Pascual-Tejerina, V.; Martin-Gonzalez, J.; Garcia-Fernandez, J.; Perez-Rivera, J.A. When should we measure biomarkers in patients with atrial fibrillation to predict recurrences? Am. J. Emerg. Med. 2021, 39, 248–249. [Google Scholar] [CrossRef]
- Okar, S.; Kaypakli, O.; Yildiray, D.; Koç, M. Fibrosis marker soluble ST2 predicts atrial fibrillation recurrence after cryoballon catheter ablation of nonvalvular paroxysmal atrial fibrillation. Korean Circ. J. 2018, 48, 920–929. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, K.; Lin, Y.; Liang, X.; Zhao, S.; Li, M.; Chen, M. Role of sST2 in predicting recurrence of atrial fibrillation after radiofrequency catheter ablation. Pacing Clin. Electrophysiol. 2020, 43, 1235–1241. [Google Scholar] [CrossRef] [PubMed]
- Kottkamp, H.; Schreiber, D.; Moser, F.; Rieger, A. Therapeutic approaches to atrial fibrillation ablation targeting atrial fibrosis. JACC Clin. Electrophysiol. 2017, 3, 643–653. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, R.; Lu, C.; Chen, Q.; Xu, T.; Li, D. Effects of the angiotensin receptor neprilysin inhibitor on cardiac reverse remodeling: Meta-analysis. J. Am. Heart Assoc. 2019, 8, e012272. [Google Scholar] [CrossRef]
- O’Meara, E.; Prescott, M.; Claggett, B.; Rouleau, J.; Chiang, L.; Solomon, S.; Packer, M.; McMurray, J.J.; Zile, M.R. Independent prognostic value of serum soluble ST2 measurements in patients with heart failure and a reduced ejection fraction in the PARADIGM-HF Trial (Prospective Comparison of ARNI with ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure). Circ. Heart Fail. 2018, 11, e004446. [Google Scholar]
- Arrieta, V.; Jover, E.; Navarro, A.; Martín-Núñez, E.; Garaikoetxea, M.; Matilla, L.; García-Peña, A.; Fernández-Celis, A.; Gainza, A.; Álvarez, V.; et al. Soluble ST2 levels are related to replacement myocardial fibrosis in severe aortic stenosis. Rev. Esp. Cardiol. 2023, 76, 679–689. [Google Scholar] [CrossRef]
- Maesen, B.; Verheule, S.; Zeemering, S.; La Meir, M.; Nijs, J.; Lumeij, S.; Lau, D.H.; Granier, M.; Crijns, H.J.; Maessen, J.G.; et al. Endomysial fibrosis, rather than overall connective tissue content, is the main determinant of conduction disturbances in human atrial fibrillation. Europace 2022, 24, 1015–1024. [Google Scholar] [CrossRef]
- Vyas, V.; Hunter, R.J.; Longhi, M.P.; Finlay, M.C. Inflammation and adiposity: New frontiers in atrial fibrillation. Europace 2020, 22, 1609–1618. [Google Scholar] [CrossRef] [PubMed]
- Yalcin, M.; Gurses, K.; Kocyigit, D.; Canpinar, H.; Canpolat, U.; Evranos, B.; Yorgun, H.; Sahiner, M.; Kaya, E.; Hazirolan, T.; et al. The association of serum galectin-3 levels with atrial electrical and structural remodeling. J. Cardiovasc. Electrophysiol. 2015, 26, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Schultz, C.; Dang, J.; Alasady, M.; Lau, D.; Brooks, A.; Wong, C.; Roberts-Thomson, K.; Young, G.; Worthley, M.; et al. Time course of inflammation, myocardial injury, and prothrombotic response after radiofrequency catheter ablation for atrial fibrillation. Circ. Arrhythm. Electrophysiol. 2014, 7, 83–89. [Google Scholar] [CrossRef]
- Begg, G.; Karim, R.; Oesterlein, T.; Graham, L.; Hogarth, A.; Page, S.; Pepper, C.B.; Rhode, K.; Lip, G.Y.H.; Holden, A.V.; et al. Left atrial voltage, circulating biomarkers of fibrosis, and atrial fibrillation ablation. A prospective cohort study. PLoS ONE 2018, 13, e0189936. [Google Scholar] [CrossRef] [PubMed]
- Boos, C.J.; Anderson, R.A.; Lip, G.Y. Is atrial fibrillation an inflammatory disorder? Eur. Heart J. 2006, 27, 136–149. [Google Scholar] [CrossRef]
- Hastie, T.; Tibshirani, R. Generalized Additive Models; Chapman and Hall/CRC Monographs on Statistics and Applied Probability: Boca Raton, FL, USA, 1990. [Google Scholar]
- Wood, S. Generalized Additive Models: An Introduction with R; Chapman and Hall/CRC Texts in Statistical Science: Boca Raton, FL, USA, 2017. [Google Scholar]
- Wood, S.N. Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. J. R. Stat. Soc. B 2011, 73, 3–36. [Google Scholar] [CrossRef]
| ECV (n = 94) | PVI (n = 156) | P1 | Control (n = 40) | P2 | |
|---|---|---|---|---|---|
| Age (years) | 61.1 ± 9.1 | 56.9 ± 10.7 | 0.001 * | 55.8 ± 11.3 | 0.068 |
| Men (n, %) | 74 (78.7) | 110 (70.5) | 0.154 | 25 (62.5) | 0.146 |
| Weight (kg) | 86.5 ± 13.1 | 86.5 ± 14.7 | 0.494 | 74.6 ± 12.1 | <0.000 * |
| Height | 1.69 ± 0.09 | 1.69 ± 0.09 | 0.378 | 1.67 ± 0.09 | 0.094 |
| BMI (kg/m2) | 30.2 ± 4.3 | 30.3 ± 4.4 | 0.423 | 26.6 ± 2.89 | <0.000 * |
| HT (n, %) | 50 (53.2) | 68 (43.6) | 0.141 | 3 (7.5) | <0.000 * |
| DM (n, %) | 10 (10.6) | 13 (8.3) | 0.541 | 2 (5) | 0.379 |
| Smoking (n, %) | 22 (23.4) | 47 (30.1) | 0.249 | 11 (27.5) | 0.989 |
| COPD (n, %) | 6 (6.4) | 8 (5.1) | 0.676 | 2 (5) | 0.877 |
| OSA (n, %) | 5 (5.3) | 9 (5.8) | 0.881 | 0 | 0.230 |
| CKD (n, %) | 1 (1.1) | 5 (3.2) | 0.284 | 0 | 1 |
| Obesity (n, %) | 36 (38.3) | 76 (48.7) | 0.108 | 7 (17.5) | 0.001 * |
| Tachycardiomyopathy (n, %) | 12 (12.8) | 19 (12.2) | 0.892 | 0 | 0.011 * |
| Time from first AF diagnosis (months) | 24.1 (32.2) | 58.2 (70.1) | <0.000 * | ||
| AF pattern (n, %) -Paroxysmal (n = 68) -Persistent (n = 182) | |||||
| 0 (0) | 68 (100) | <0.000 | |||
| 94 (51.6) | 88 (48.4) | 0.234 | |||
| RA Area (cm2) | 15.4 ± 4.2 | 13.6 ± 3.9 | 0.000 * | 12.8 ± 3.1 | 0.001 * |
| LA AP Diameter (mm) | 45.2 ± 5.5 | 41.2 ± 6.2 | 0.078383 | 23 ± 3.9 | <0.000 * |
| LA Area (cm2) | 24.2 ± 5 | 21.5 ± 5.4 | 0.090 | 13 ± 3.2 | <0.000 * |
| LA Volume (mL) | 96.2 ± 47.5 | 94.4 ± 54.3 | 0.397 | 53 ± 30.9 | <0.000 * |
| LA Index Volume (mL/m2) | 48.3 ± 22.5 | 47.9 ± 27.8 | 0.446 | 28.7 ± 16.8 | <0.000 * |
| LVEF (%) | 61.4 ± 7.2 | 62.6 ± 7.2 | 0.103 | 63 ± 6 | 0.176 |
| LVEDV (mL) | 60.4 ± 23.2 | 56.4 ± 25.6 | 0.106 | 59 ± 21.8 | 0.382 |
| LVESV (mL) | 23.2 ± 10.4 | 21.3 ± 10.8 | 0.092 | 22 ± 8.5 | 0.408 |
| LVEDd (mm) | 39.3 ± 7.1 | 38.9 ± 8.7 | 0.374 | 38 ± 7.1 | 0.349 |
| LVESd (mm) | 27.2 ± 4.3 | 26.9 ± 6.1 | 0.338 | 27 ± 4.8 | 0.415 |
| RVEDd (mm) | 26.7 ± 4.8 | 26.7 ± 4.7 | 0.496 | 25 ± 4.5 | 0.027 * |
| Glucose (mg/dL) | 105.1 ± 23.3 | 98.3 ± 18 | 0.041 * | ||
| Urea (mg/dL) | 45.6 ± 14.5 | 39.2 ± 10.8 | 0.005 * | ||
| Creatinine (mg/dL) | 0.98 ± 0.30 | 0.80 ± 0.16 | 0.001 * | ||
| Hb1Ac (%) | 6.9 ± 1.2 | 5.3 ± 0.7 | 0.001 * | ||
| TSH (mU/L) | 2.9 ± 2.4 | 3.1 ± 5.7 | 0.232 | ||
| AAD (n, %) -Amiodarone -Flecainide -Propafenone -Betablockers -Calcium antagonists | 92 (97.8) | 152 (97.4) | 0.542 | 11(27.5) | 0.001 * |
| 44 (46.8) | 48 (30.8) | ||||
| 46 (48.9) | 101 (64.7) | ||||
| 2 (2.1) | 3 (1.9) | ||||
| 88 (93.6) | 138 (88.5) | ||||
| 2 (2.1) | 8 (5.1) | ||||
| sST2.0 (pg/mL) | 17,163 ± 9147 | 13,178 ± 7223 | 0.000 * | 11,016 ± 5618 | 0.007 * |
| sST2.0 and AF type (pg/mL) -Paroxysmal -Persistent -Persistent vs. paroxysmal | |||||
| 12,113 ± 6278 | 11,016 ± 5618 | 0.234 | |||
| 15,634 ± 8651 | 11,016 ± 5618 | 0.011 * | |||
| 15,634 ± 8651 vs. 12,113 ± 6278 | 0.002 * | ||||
| sST2.0 and initial rhythm (pg/mL) -Sinus rhythm, n = 89 -AF, n = 161 | 0.003 * | ||||
| 12,626 ± 6483 | |||||
| 15,810 ± 8846 | |||||
| sST2.0 and rate (pg/mL) <100 bpm, n = 221 ≥100 bpm, n = 29 | 0.065 | ||||
| 14,330 ± 7641 | |||||
| 17,321 ± 11,534 | |||||
| sST2.0 and rate in persistent AF (pg/mL) <100 bpm, n = 159 ≥100 bpm, n = 23 | 0.032 * | ||||
| 15,113 ± 7941 | |||||
| 19,237 ± 12,136 | |||||
| Recurrence from 0 to 3 Months FU | Recurrence from 3 to 6 Months FU | Recurrence from 0 to 6 Months FU | |||||
|---|---|---|---|---|---|---|---|
| NO n = 172 | Yes n = 78 | NO n = 186 | Yes n = 64 | NO n = 146 | Yes n = 104 | ||
| Paroxysmal | ECV | - | - | - | - | - | - |
| PVI | 60 | 8 | 60 | 8 | 54 | 14 | |
| Persistent | ECV | 47 | 47 | 53 | 41 | 36 | 58 |
| PVI | 65 | 23 | 73 | 15 | 56 | 32 | |
| sST2.0 | sST2.1 | p | ||
| 3-month FU | No recurrence FU(n = 47) | 18,598 ± 10,916 | 14,680 ± 7561 | 0.002 * |
| Recurrence (n = 47) | 15,729 ± 6768 | 16,133 ± 7218 | 0.618 | |
| sST2.0 | sST2.2 | p | ||
| 6-month FU | No recurrence (n = 53) | 16,313 ± 10,114 | 13,931 ± 7211 | 0.019 * |
| Recurrence FU2 (n = 41) | 18,263 ± 7611 | 18,950 ± 9402 | 0.559 | |
| sST2.1 | sST2.2 | p | ||
| 3- vs. 6-month FU | No recurrence (n = 53) | 13,350 ± 5941 | 13,931 ± 7211 | 0.286 |
| Recurrence (n = 41) | 18,066 ± 8256 | 18,950 ± 9402 | 0.277 |
| sST2.0 | sST2.1 | p | ||
| 3-month FU | No recurrence (n = 125) | 13,033 ± 7408 | 14,109 ± 8108 | 0.05 |
| Recurrence (n = 31) | 13,765 ± 6501 | 14,263 ± 6430 | 0.565 | |
| sST2.0 | sST2.2 | p | ||
| 6-month FU | No recurrence (n = 133) | 13,302 ± 7431 | 14,302 ± 6945 | 0.022 * |
| Recurrence (n = 23) | 12,461 ± 5837 | 13,571 ± 4181 | 0.451 | |
| sST2.1 | sST2.2 | p | ||
| 3- vs. 6-month FU | No recurrence (n = 133) | 14,301 ± 8130 | 14,302 ± 6945 | 0.997 |
| Recurrence (n = 23) | 13,208 ± 5405 | 13,571 ± 4181 | 0.728 |
| (A) | ||||
| OR | CI 95% | p | ||
| Age (years) | 0.980 | 0.927 | 1.035 | 0.223 |
| Gender—male | 0.443 | 0.123 | 1.503 | 0.165 |
| LA index volume (mL/m2) | 1.028 | 1.007 | 1.051 | 0.006 * |
| BMI (kg/m2) | 1.008 | 0.911 | 1.121 | 0.674 |
| Rate (bpm) | 1.006 | 0.981 | 1.030 | 0.145 |
| sST2.0 (pg/mL) | 0.999 | 0.999 | 1.000 | 0.357 |
| (B) | ||||
| OR | CI 95% | p | ||
| Age (years) | 1.054 | 0.991 | 1.127 | 0.203 |
| Gender—male | 2.168 | 0.863 | 3.530 | 0.751 |
| LA index volume (mL/m2) | 0.986 | 1.009 | 1.034 | 0.122 |
| BMI (kg/m2) | 0.940 | 1.009 | 1.034 | 0.417 |
| Rate (bpm) | 0.997 | 0.963 | 1.033 | 0.809 |
| Rhythm at 3 months: AF | 7.403 | 2.188 | 28.192 | 0.001 * |
| sST2.1 (pg/mL) | 1.345 | 1.085 | 1.823 | 0.001 * |
| (A) | ||||
| OR | CI 95% | p | ||
| Age (years) | 0.974 | 0.924 | 1.026 | 0.330 |
| Gender—male | 0.261 | 0.065 | 0.974 | 0.049 * |
| BMI (kg/m2) | 0.916 | 0.802 | 1.033 | 0.281 |
| Rate (bpm) | 0.956 | 0.918 | 0.989 | 0.019 * |
| Rhythm at basal: AF | 5.702 | 1.289 | 6.497 | 0.023 * |
| Type AF (persistent) | 2.122 | 0.505 | 9.248 | 0.302 |
| sST2.0 | 1.000 | 0.999 | 1.000 | 0.353 |
| LA index volume (mL/m2) | 1.027 | 1.009 | 1.047 | 0.047 * |
| Low-voltage area (%) | 1.008 | 0.986 | 1.032 | 0.376 |
| (B) | ||||
| OR | CI 95% | p | ||
| Age (years) | 1.017 | 0.962 | 1.081 | 0.582 |
| Gender—male | 1.166 | 0.305 | 4.868 | 0.799 |
| BMI (kg/m2) | 1.036 | 0.898 | 1.188 | 0.625 |
| Rate (bpm) | 0.969 | 0.915 | 1.023 | 0.171 |
| Rhythm at 3 months: AF | 3.001 | 2.919 | 4.262 | 0.004 * |
| Type of AF (persistent) | 2.247 | 0.164 | 6.290 | 0.625 |
| sST2.1 (pg/mL) | 0.999 | 0.999 | 0.9999 | 0.390 |
| LA index volume (mL/m2) | 0.999 | 0.999 | 1.000 | 0.209 |
| Low-voltage area (%) | 1.018 | 0.989 | 1.047 | 0.288 |
| Low-Voltage Area (%) | ST2S.0 (pg/mL) |
|---|---|
| <5 (n = 49) | 11,324 ± 5698 |
| 5 to 35 (n = 62) | 14,294 ± 7621 |
| >35 (n = 17) | 10,983 ± 6679 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Seara, J.; González Melchor, L.; Rodríguez García, J.; Gude, F.; Martínez Sande, J.L.; Rodríguez Mañero, M.; Fernández López, X.A.; Minguito Carazo, C.; González Ferrero, T.; Eiras, S.; et al. Role of Soluble ST2 Biomarker in Predicting Recurrence of Atrial Fibrillation after Electrical Cardioversion or Pulmonary Vein Isolation. Int. J. Mol. Sci. 2023, 24, 14045. https://doi.org/10.3390/ijms241814045
García-Seara J, González Melchor L, Rodríguez García J, Gude F, Martínez Sande JL, Rodríguez Mañero M, Fernández López XA, Minguito Carazo C, González Ferrero T, Eiras S, et al. Role of Soluble ST2 Biomarker in Predicting Recurrence of Atrial Fibrillation after Electrical Cardioversion or Pulmonary Vein Isolation. International Journal of Molecular Sciences. 2023; 24(18):14045. https://doi.org/10.3390/ijms241814045
Chicago/Turabian StyleGarcía-Seara, Javier, Laila González Melchor, Javier Rodríguez García, Francisco Gude, José Luis Martínez Sande, Moisés Rodríguez Mañero, Xesús Alberte Fernández López, Carlos Minguito Carazo, Teba González Ferrero, Sonia Eiras, and et al. 2023. "Role of Soluble ST2 Biomarker in Predicting Recurrence of Atrial Fibrillation after Electrical Cardioversion or Pulmonary Vein Isolation" International Journal of Molecular Sciences 24, no. 18: 14045. https://doi.org/10.3390/ijms241814045






