How Nitric Oxide Hindered the Search for Hemoglobin-Based Oxygen Carriers as Human Blood Substitutes
Abstract
:1. Introduction
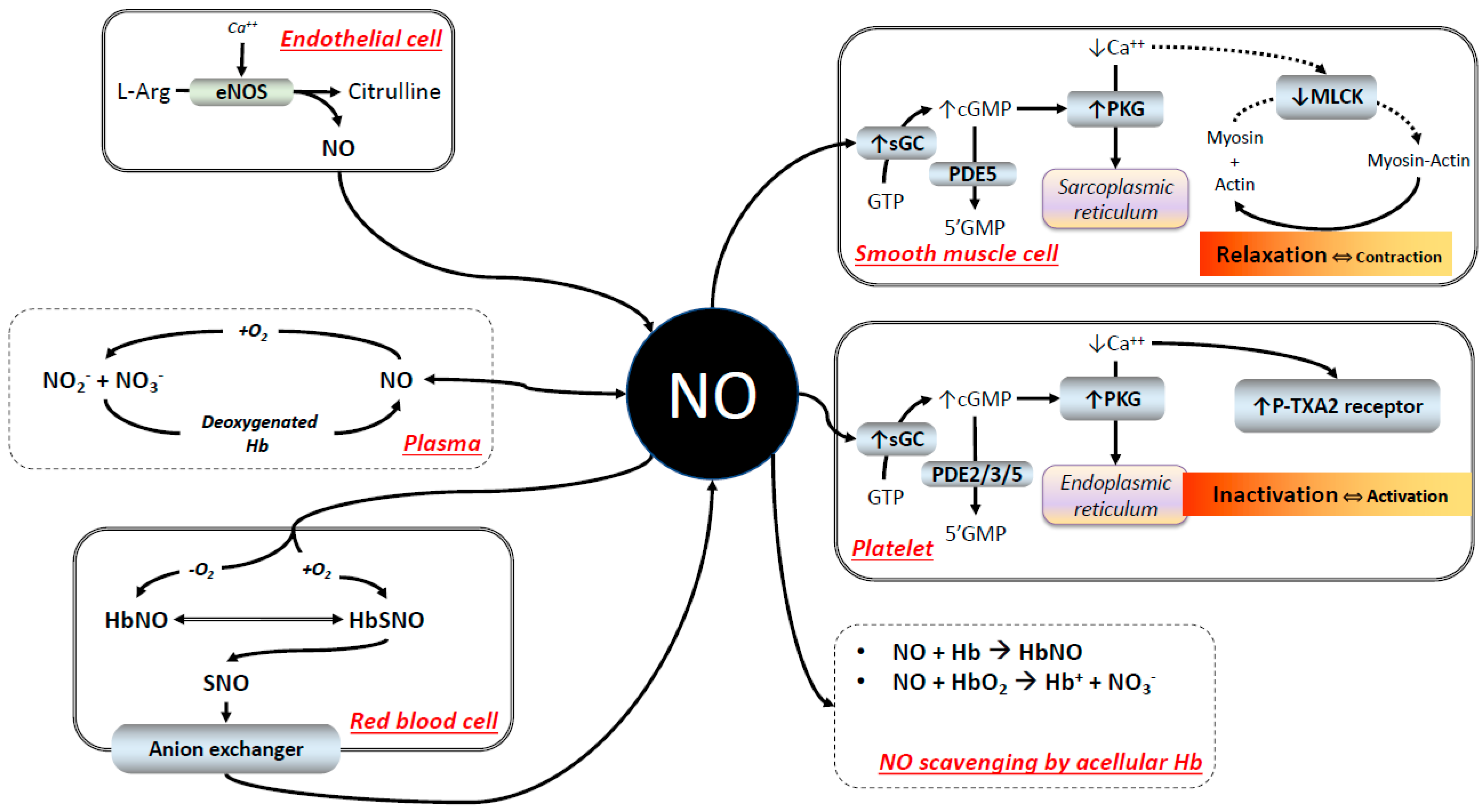
2. NO Scavenging: The Difficult Co-Existence of NO with Hb
2.1. The Reaction of Hb with NO
2.2. RBC Hb and NO Scavenging
2.3. NO Signaling
2.4. NO Signaling in the Presence of HBOCs
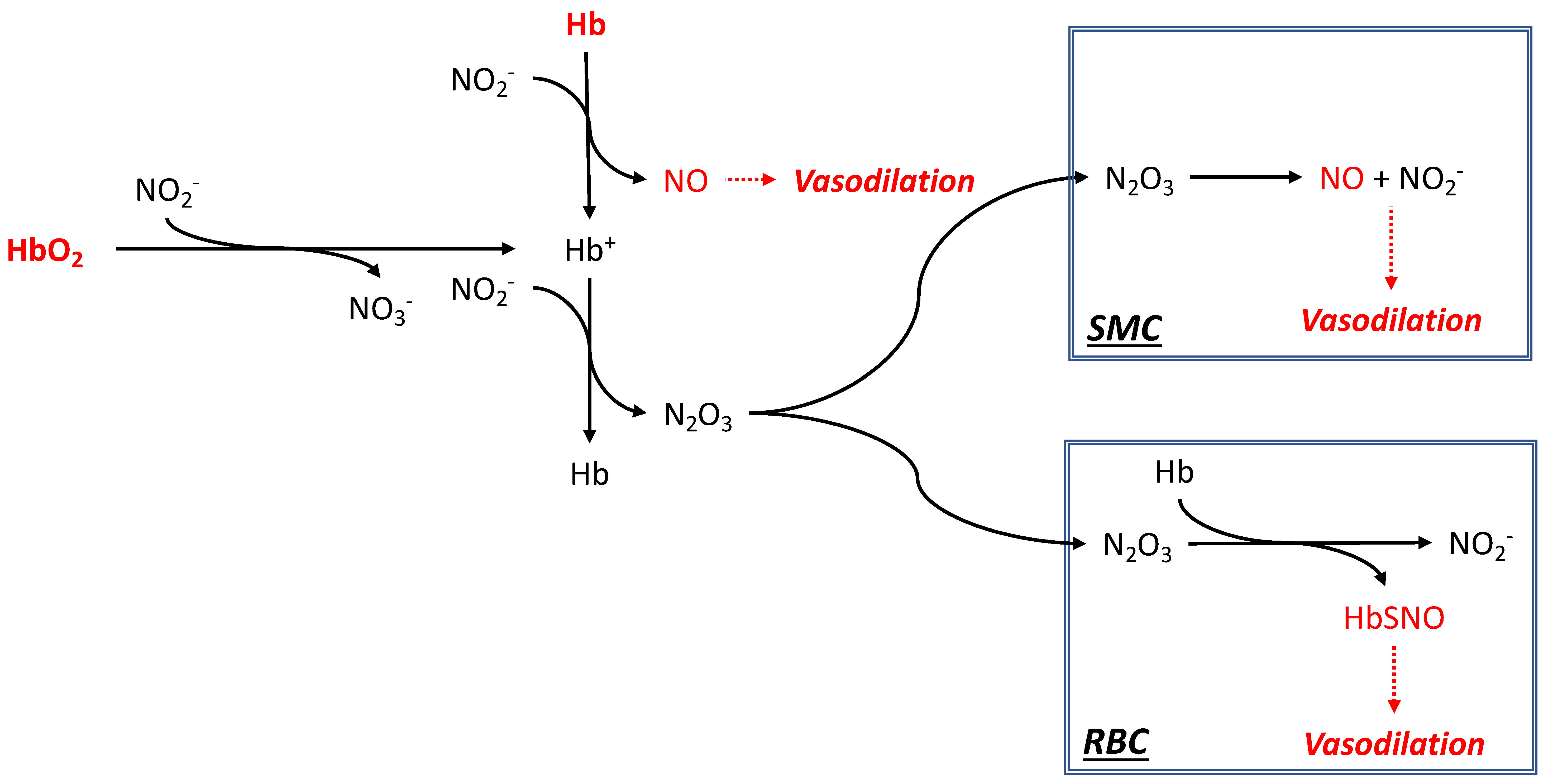
2.5. Nitrite Reductase Activity as a Mechanism of Vascular Control
3. Main Problems Associated with HBOC Infusion: A View from NO
3.1. Systemic Effects
3.2. Inflammation
3.3. Coagulopathy
3.4. Oxidative Stress
3.5. O2 Carrying Function
4. Protein Engineering
4.1. Stabilizing the Hb Tetramers
4.2. Hb Polymerization
4.3. Hb Conjugation
4.4. Recombinant Hb
4.5. Polynitroxylated Hb
4.6. Microencapsulation
4.7. Earthworms and Marine Worm Erythrocruorin
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviation
References
- Palmer, A.F.; Intaglietta, M. Blood substitutes. Annu. Rev. Biomed. Eng. 2014, 16, 77–101. [Google Scholar] [CrossRef] [PubMed]
- Keipert, P.E. Oxygen therapeutics (“blood substitutes”) where are they, and what can we expect? Adv. Exp. Med. Biol. 2003, 540, 207–213. [Google Scholar] [PubMed]
- Thom, C.S.; Chou, S.T.; French, D.L. Mechanistic and Translational Advances Using iPSC-Derived Blood Cells. J. Exp. Pathol. 2020, 1, 36–44. [Google Scholar]
- Jahr, J.S. Blood substitutes: Basic science, translational studies and clinical trials. Front. Med. Technol. 2022, 4, 989829. [Google Scholar] [CrossRef]
- Moradi, S.; Jahanian-Najafabadi, A.; Roudkenar, M.H. Artificial Blood Substitutes: First Steps on the Long Route to Clinical Utility. Clin. Med. Insights Blood Disord. 2016, 9, 33–41. [Google Scholar] [PubMed]
- Khan, F.; Singh, K.; Friedman, M.T. Artificial Blood: The History and Current Perspectives of Blood Substitutes. Discoveries 2020, 8, e104. [Google Scholar] [CrossRef] [PubMed]
- Winslow, R.M. (Ed.) Blood Substitutes; Elsevier: Amsterdam, the Netherlands, 2006. [Google Scholar]
- Chang, T.M.S.; Bulow, L.; Jahr, J.S.; Sakai, H.; Yang, C. Nanobiotherapeutic Based Blood Substitutes; World Scientific Publishing Company: Singapore, 2021. [Google Scholar]
- Jahr, J.S.; Guinn, N.R.; Lowery, D.R.; Shore-Lesserson, L.; Shander, A. Blood Substitutes and Oxygen Therapeutics: A Review. Anesth. Analg. 2021, 132, 119–129. [Google Scholar]
- Ignarro, L.; Buga, G.; Wood, K. Endothelium-derived relaxing factor produced and released from artery and vein is nitric oxide. Proc. Natl. Acad. Sci. USA 1987, 84, 9265–9269. [Google Scholar] [CrossRef]
- Mancardi, D.; Ottolenghi, S.; Attanasio, U.; Tocchetti, C.G.; Paroni, R.; Pagliaro, P.; Samaja, M. Janus, or the Inevitable Battle Between Too Much and Too Little Oxygen. Antioxid. Redox Signal. 2022, 37, 972–989. [Google Scholar] [CrossRef]
- Liu, X.; Miller, M.J.; Joshi, M.S.; Sadowska-Krowicka, H.; Clark, D.A.; Lancaster, J.R., Jr. Diffusion-limited reaction of free nitric oxide with erythrocytes. J. Biol. Chem. 1998, 273, 18709–18713. [Google Scholar]
- Stamler, J.S.; Simon, D.I.; Osborne, J.A.; Mullins, M.E.; Jaraki, O.; Michel, T.; Singel, D.J.; Loscalzo, J. S-nitrosylation of proteins with nitric oxide: Synthesis and characterization of biologically active compounds. Proc. Natl. Acad. Sci. USA 1992, 89, 444–448. [Google Scholar] [CrossRef]
- Gladwin, M.T.; Crawford, J.H.; Patel, R.P. The biochemistry of nitric oxide, nitrite, and hemoglobin: Role in blood flow regulation. Free Radic. Biol. Med. 2004, 36, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Gibson, Q. The kinetics of reactions between hemoglobin and gases. Prog. Biophys. Biophys. Chem. 1959, 9, 1–53. [Google Scholar] [CrossRef]
- Eich, R.F.; Li, T.; Lemon, D.D.; Doherty, D.H.; Curry, S.R.; Aitken, J.F.; Mathews, A.J.; Johnson, K.A.; Smith, R.D.; Phillips, G.N., Jr.; et al. Mechanism of NO-induced oxidation of myoglobin and hemoglobin. Biochemistry 1996, 35, 6976–6983. [Google Scholar] [CrossRef] [PubMed]
- Rohlfs, R.; Bruner, E.; Chiu, A.; Gonzales, A.; Gonzales, M.; Magde, D.; Magde, M.; Vandegriff, K.; Winslow, R. Arterial blood pressure responses to cell-free hemoglobin solutions and the reaction with nitric oxide. J. Biol. Chem. 1998, 273, 12128–12134. [Google Scholar] [CrossRef]
- Vandegriff, K.D.; Bellelli, A.; Samaja, M.; Malavalli, A.; Brunori, M.; Winslow, R.M. Kinetics of NO and O2 binding to a maleimide poly(ethylene glycol)-conjugated human haemoglobin. Biochem. J. 2004, 382, 183–189. [Google Scholar] [CrossRef]
- Herold, S.; Exner, M.; Nauser, T. Kinetic and mechanistic studies of the NO*-mediated oxidation of oxymyoglobin and oxyhemoglobin. Biochemistry 2001, 40, 3385–3395. [Google Scholar] [CrossRef]
- Herold, S.; Rehmann, F.J. Kinetics of the reactions of nitrogen monoxide and nitrite with ferryl hemoglobin. Free Radic. Biol. Med. 2003, 34, 531–545. [Google Scholar] [CrossRef]
- Chan, N.L.; Kavanaugh, J.S.; Rogers, P.H.; Arnone, A. Crystallographic analysis of the interaction of nitric oxide with quaternary-T human hemoglobin. Biochemistry 2004, 43, 118–132. [Google Scholar] [CrossRef]
- Premont, R.T.; Reynolds, J.D.; Zhang, R.; Stamler, J.S. Red Blood Cell-Mediated S-Nitrosohemoglobin-Dependent Vasodilation: Lessons Learned from a beta-Globin Cys93 Knock-In Mouse. Antioxid. Redox Signal. 2021, 34, 936–961. [Google Scholar] [CrossRef]
- Sun, C.W.; Yang, J.; Kleschyov, A.L.; Zhuge, Z.; Carlstrom, M.; Pernow, J.; Wajih, N.; Isbell, T.S.; Oh, J.Y.; Cabrales, P.; et al. Hemoglobin beta93 Cysteine Is Not Required for Export of Nitric Oxide Bioactivity From the Red Blood Cell. Circulation 2019, 139, 2654–2663. [Google Scholar] [CrossRef]
- Isbell, T.S.; Sun, C.W.; Wu, L.C.; Teng, X.; Vitturi, D.A.; Branch, B.G.; Kevil, C.G.; Peng, N.; Wyss, J.M.; Ambalavanan, N.; et al. SNO-hemoglobin is not essential for red blood cell-dependent hypoxic vasodilation. Nat. Med. 2008, 14, 773–777. [Google Scholar] [CrossRef] [PubMed]
- Vitturi, D.A.; Sun, C.W.; Harper, V.M.; Thrash-Williams, B.; Cantu-Medellin, N.; Chacko, B.K.; Peng, N.; Dai, Y.; Wyss, J.M.; Townes, T.; et al. Antioxidant functions for the hemoglobin beta93 cysteine residue in erythrocytes and in the vascular compartment in vivo. Free Radic. Biol. Med. 2013, 55, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Herold, S.; Rock, G. Reactions of deoxy-, oxy-, and methemoglobin with nitrogen monoxide. Mechanistic studies of the S-nitrosothiol formation under different mixing conditions. J. Biol. Chem. 2003, 278, 6623–6634. [Google Scholar] [CrossRef]
- Huang, Z.; Louderback, J.G.; Goyal, M.; Azizi, F.; King, S.B.; Kim-Shapiro, D.B. Nitric oxide binding to oxygenated hemoglobin under physiological conditions. Biochim. Biophys. Acta 2001, 1568, 252–260. [Google Scholar] [CrossRef]
- Huang, Z.; Ucer, K.B.; Murphy, T.; Williams, R.T.; King, S.B.; Kim-Shapiro, D.B. Kinetics of nitric oxide binding to R-state hemoglobin. Biochem. Biophys. Res. Commun. 2002, 292, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.P.; Hogg, N.; Spencer, N.Y.; Kalyanaraman, B.; Matalon, S.; Darley-Usmar, V.M. Biochemical characterization of human S-nitrosohemoglobin. Effects on oxygen binding and transnitrosation. J. Biol. Chem. 1999, 274, 15487–15492. [Google Scholar] [CrossRef] [PubMed]
- Herold, S. Interaction of nitrogen monoxide with hemoglobin and the artefactual production of S-nitroso-hemoglobin. Comptes Rendus Biol. 2003, 326, 533–541. [Google Scholar] [CrossRef]
- Herold, S.; Röck, G. Mechanistic studies of S-nitrosothiol formation by NO*/O2 and by NO*/methemoglobin. Arch. Biochem. Biophys. 2005, 436, 386–396. [Google Scholar] [CrossRef]
- Azarov, I.; Liu, C.; Reynolds, H.; Tsekouras, Z.; Lee, J.S.; Gladwin, M.T.; Kim-Shapiro, D.B. Mechanisms of slower nitric oxide uptake by red blood cells and other hemoglobin-containing vesicles. J. Biol. Chem. 2011, 286, 33567–33579. [Google Scholar] [CrossRef]
- Chen, K.; Piknova, B.; Pittman, R.N.; Schechter, A.N.; Popel, A.S. Nitric oxide from nitrite reduction by hemoglobin in the plasma and erythrocytes. Nitric Oxide 2008, 18, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Donadee, C.; Raat, N.J.; Kanias, T.; Tejero, J.; Lee, J.S.; Kelley, E.E.; Zhao, X.; Liu, C.; Reynolds, H.; Azarov, I.; et al. Nitric oxide scavenging by red blood cell microparticles and cell-free hemoglobin as a mechanism for the red cell storage lesion. Circulation 2011, 124, 465–476. [Google Scholar] [CrossRef]
- Loscalzo, J. Nitric oxide binding and the adverse effects of cell-free hemoglobins: What makes us different from earthworms. J. Lab. Clin. Med. 1997, 129, 580–583. [Google Scholar] [CrossRef] [PubMed]
- Lui, F.E.; Kluger, R. Reviving artificial blood: Meeting the challenge of dealing with NO scavenging by hemoglobin. Chembiochem 2010, 11, 1816–1824. [Google Scholar] [CrossRef] [PubMed]
- Gladwin, M.T.; Shelhamer, J.H.; Schechter, A.N.; Pease-Fye, M.E.; Waclawiw, M.A.; Panza, J.A.; Ognibene, F.P.; Cannon, R.O., 3rd. Role of circulating nitrite and S-nitrosohemoglobin in the regulation of regional blood flow in humans. Proc. Natl. Acad. Sci. USA 2000, 97, 11482–11487. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Bonaventura, C.; Bonaventura, J.; Stamler, J. S-nitrosohemoglobin: A dynamic activity of blood involved in vascular control. Nature 1996, 380, 221–226. [Google Scholar] [CrossRef]
- Gladwin, M.; Ognibene, F.; Pannell, L.; Nichols, J.; Pease-Fye, M.; Shelhamer, J.; Schechter, A. Relative role of heme nitrosylation and beta-cysteine 93 nitrosation in the transport and metabolism of nitric oxide by hemoglobin in the human circulation. Proc. Natl. Acad. Sci. USA 2000, 97, 9943–9948. [Google Scholar] [CrossRef]
- Tsoukias, N.M.; Popel, A.S. Erythrocyte consumption of nitric oxide in presence and absence of plasma-based hemoglobin. Am. J. Physiol. Heart Circ. Physiol. 2002, 282, H2265–H2277. [Google Scholar] [CrossRef]
- Kavdia, M.; Tsoukias, N.M.; Popel, A.S. Model of nitric oxide diffusion in an arteriole: Impact of hemoglobin-based blood substitutes. Am. J. Physiol. Heart Circ. Physiol. 2002, 282, H2245–H2253. [Google Scholar] [CrossRef]
- Caretti, A.; Fantacci, M.; Caccia, D.; Perrella, M.; Lowe, K.C.; Samaja, M. Modulation of the NO/cGMP pathway reduces the vasoconstriction induced by acellular and PEGylated haemoglobin. Biochim. Biophys. Acta-Proteins Proteom. 2008, 1784, 1428–1434. [Google Scholar] [CrossRef]
- Ehler, E.; Karlhuber, G.; Bauer, H.C.; Draeger, A. Heterogeneity of smooth muscle-associated proteins in mammalian brain microvasculature. Cell Tissue Res. 1995, 279, 393–403. [Google Scholar] [CrossRef]
- Hill, R.A.; Tong, L.; Yuan, P.; Murikinati, S.; Gupta, S.; Grutzendler, J. Regional Blood Flow in the Normal and Ischemic Brain Is Controlled by Arteriolar Smooth Muscle Cell Contractility and Not by Capillary Pericytes. Neuron 2015, 87, 95–110. [Google Scholar] [CrossRef]
- Alarcon-Martinez, L.; Yilmaz-Ozcan, S.; Yemisci, M.; Schallek, J.; Kilic, K.; Can, A.; Di Polo, A.; Dalkara, T. Capillary pericytes express alpha-smooth muscle actin, which requires prevention of filamentous-actin depolymerization for detection. Elife 2018, 7, e34861. [Google Scholar] [CrossRef] [PubMed]
- Borysova, L.; Burdyga, T. Evidence that NO/cGMP/PKG signalling cascade mediates endothelium dependent inhibition of IP(3)R mediated Ca(2)(+) oscillations in myocytes and pericytes of ureteric microvascular network in situ. Cell Calcium 2015, 58, 535–540. [Google Scholar] [CrossRef]
- Palacios-Callender, M.; Quintero, M.; Hollis, V.S.; Springett, R.J.; Moncada, S. Endogenous NO regulates superoxide production at low oxygen concentrations by modifying the redox state of cytochrome c oxidase. Proc. Natl. Acad. Sci. USA 2004, 101, 7630–7635. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.C. Regulation of mitochondrial respiration by nitric oxide inhibition of cytochrome c oxidase. Biochim. Biophys. Acta 2001, 1504, 46–57. [Google Scholar] [CrossRef]
- Sarti, P.; Forte, E.; Mastronicola, D.; Giuffre, A.; Arese, M. Cytochrome c oxidase and nitric oxide in action: Molecular mechanisms and pathophysiological implications. Biochim. Biophys. Acta 2012, 1817, 610–619. [Google Scholar] [CrossRef] [PubMed]
- Carlström, M. Nitric oxide signalling in kidney regulation and cardiometabolic health. Nat. Rev. Nephrol. 2021, 17, 575–590. [Google Scholar] [CrossRef]
- Ghashghaeinia, M.; Köberle, M.; Mrowietz, U.; Bernhardt, I. Proliferating tumor cells mimick glucose metabolism of mature human erythrocytes. Cell Cycle 2019, 18, 1316–1334. [Google Scholar] [CrossRef]
- Caneba, C.A.; Yang, L.; Baddour, J.; Curtis, R.; Win, J.; Hartig, S.; Marini, J.; Nagrath, D. Nitric oxide is a positive regulator of the Warburg effect in ovarian cancer cells. Cell Death Dis. 2014, 5, e1302. [Google Scholar] [CrossRef]
- Riera Leal, A.; Ortiz-Lazareno, P.C.; Jave-Suárez, L.F.; Ramírez De Arellano, A.; Aguilar-Lemarroy, A.; Ortiz-García, Y.M.; Barrón-Gallardo, C.A.; Solís-Martínez, R.; Luquin De Anda, S.; Muñoz-Valle, J.F.; et al. 17β-estradiol-induced mitochondrial dysfunction and Warburg effect in cervical cancer cells allow cell survival under metabolic stress. Int. J. Oncol. 2020, 56, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Barros, L.F.; Ruminot, I.; San Martín, A.; Lerchundi, R.; Fernández-Moncada, I.; Baeza-Lehnert, F. Aerobic Glycolysis in the Brain: Warburg and Crabtree Contra Pasteur. Neurochem. Res. 2021, 46, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Riou, M.; Enache, I.; Sauer, F.; Charles, A.L.; Geny, B. Targeting Mitochondrial Metabolic Dysfunction in Pulmonary Hypertension: Toward New Therapeutic Approaches? Int. J. Mol. Sci. 2023, 24, 9572. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.Y.; Mauro, C. Similarities in the Metabolic Reprogramming of Immune System and Endothelium. Front. Immunol. 2017, 8, 837. [Google Scholar] [CrossRef]
- Liao, S.T.; Han, C.; Xu, D.Q.; Fu, X.W.; Wang, J.S.; Kong, L.Y. 4-Octyl itaconate inhibits aerobic glycolysis by targeting GAPDH to exert anti-inflammatory effects. Nat. Commun. 2019, 10, 5091. [Google Scholar] [CrossRef]
- Yu, B.; Zapol, W.M. Hemoglobin-Based Oxygen Carriers and Inhaled Nitric Oxide. In Nanobiotheraputic Based Blood Substitues; Chang, T.M.S., Bulow, L., Jahr, J.S., Sakai, H., Yang, C., Eds.; World Scientific: Singapore, 2021; pp. 267–277. [Google Scholar]
- Yu, B.; Raher, M.J.; Volpato, G.P.; Bloch, K.D.; Ichinose, F.; Zapol, W.M. Inhaled nitric oxide enables artificial blood transfusion without hypertension. Circulation 2008, 117, 1982–1990. [Google Scholar] [CrossRef]
- Yu, B.; Bloch, K.D.; Zapol, W.M. Hemoglobin-based red blood cell substitutes and nitric oxide. Trends Cardiovasc. Med. 2009, 19, 103–107. [Google Scholar] [CrossRef]
- Marrazzo, F.; Larson, G.; Sherpa Lama, T.T.; Teggia Droghi, M.; Joyce, M.; Ichinose, F.; Watkins, M.T.; Stowell, C.; Crowley, J.; Berra, L. Inhaled nitric oxide prevents systemic and pulmonary vasoconstriction due to hemoglobin-based oxygen carrier infusion: A case report. J. Crit. Care 2019, 51, 213–216. [Google Scholar] [CrossRef]
- Tsai, A.G.; Cabrales, P.; Manjula, B.N.; Acharya, S.A.; Winslow, R.M.; Intaglietta, M. Dissociation of local nitric oxide concentration and vasoconstriction in the presence of cell-free hemoglobin oxygen carriers. Blood 2006, 108, 3603–3610. [Google Scholar] [CrossRef]
- Richmond, K.N.; Shonat, R.D.; Lynch, R.M.; Johnson, P.C. Critical PO(2) of skeletal muscle in vivo. Am. J. Physiol. 1999, 277, H1831–H1840. [Google Scholar]
- Watson, N.A.; Beards, S.C.; Altaf, N.; Kassner, A.; Jackson, A. The effect of hyperoxia on cerebral blood flow: A study in healthy volunteers using magnetic resonance phase-contrast angiography. Eur. J. Anaesthesiol.|EJA 2000, 17, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, H.; Weatherall, M.; Wijesinghe, M.; Perrin, K.; Ranchord, A.; Simmonds, M.; Beasley, R. Systematic review of studies of the effect of hyperoxia on coronary blood flow. Am. Heart J. 2009, 158, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Crawford, P.; Good, P.A.; Gutierrez, E.; Feinberg, J.H.; Boehmer, J.P.; Silber, D.H.; Sinoway, L.I. Effects of supplemental oxygen on forearm vasodilation in humans. J. Appl. Physiol. 1997, 82, 1601–1606. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, A.; Bak, Z.; Janerot-Sjoberg, B.; Sjoberg, F. Acute hyperoxaemia-induced effects on regional blood flow, oxygen consumption and central circulation in man. Acta Physiol. Scand. 2005, 183, 231–240. [Google Scholar] [CrossRef]
- Sharkey, R.A.; Mulloy, E.M.; O’Neill, S.J. Acute effects of hypoxaemia, hyperoxaemia and hypercapnia on renal blood flow in normal and renal transplant subjects. Eur. Respir. J. 1998, 12, 653–657. [Google Scholar] [CrossRef] [PubMed]
- Lodato, R.F. Decreased O2 consumption and cardiac output during normobaric hyperoxia in conscious dogs. J. Appl. Physiol. 1989, 67, 1551–1559. [Google Scholar] [CrossRef]
- Intaglietta, M.; Johnson, P.; Winslow, R. Microvascular and tissue oxygen distribution. Cardiovasc. Res. 1996, 32, 632–643. [Google Scholar] [CrossRef]
- Gladwin, M.T.; Kim-Shapiro, D.B. The functional nitrite reductase activity of the heme-globins. Blood 2008, 112, 2636–2647. [Google Scholar] [CrossRef]
- Grubina, R.; Huang, Z.; Shiva, S.; Joshi, M.S.; Azarov, I.; Basu, S.; Ringwood, L.A.; Jiang, A.; Hogg, N.; Kim-Shapiro, D.B.; et al. Concerted nitric oxide formation and release from the simultaneous reactions of nitrite with deoxy- and oxy-hemoglobin. J. Biol. Chem. 2007, 282, 12916–12927. [Google Scholar] [CrossRef]
- Basu, S.; Grubina, R.; Huang, J.; Conradie, J.; Huang, Z.; Jeffers, A.; Jiang, A.; He, X.; Azarov, I.; Seibert, R.; et al. Catalytic generation of N2O3 by the concerted nitrite reductase and anhydrase activity of hemoglobin. Nat. Chem. Biol. 2007, 3, 785–794. [Google Scholar] [CrossRef]
- Alayash, A.I. Mechanisms of Toxicity and Modulation of Hemoglobin-based Oxygen Carriers. Shock. 2019, 52, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.Y.; Scerbo, M.; Kramer, G. A review of blood substitutes: Examining the history, clinical trial results, and ethics of hemoglobin-based oxygen carriers. Clinics 2009, 64, 803–813. [Google Scholar] [CrossRef] [PubMed]
- Natanson, C.; Kern, S.J.; Lurie, P.; Banks, S.M.; Wolfe, S.M. Cell-free hemoglobin-based blood substitutes and risk of myocardial infarction and death: A meta-analysis. JAMA 2008, 299, 2304–2312. [Google Scholar] [CrossRef] [PubMed]
- Mongan, P.D.; Moon-Massat, P.F.; Rentko, V.; Mihok, S.; Dragovich, A.; Sharma, P. Regional blood flow after serial normovolemic exchange transfusion with HBOC-201 (Hemopure) in anesthetized swine. J. Trauma. 2009, 67, 51–60. [Google Scholar] [CrossRef]
- Ulatowski, J.A.; Bucci, E.; Razynska, A.; Traystman, R.J.; Koehler, R.C. Cerebral blood flow during hypoxic hypoxia with plasma-based hemoglobin at reduced hematocrit. Am. J. Physiol. 1998, 274, H1933–H1942. [Google Scholar] [CrossRef]
- Mintun, M.A.; Lundstrom, B.N.; Snyder, A.Z.; Vlassenko, A.G.; Shulman, G.L.; Raichle, M.E. Blood flow and oxygen delivery to human brain during functional activity: Theoretical modeling and experimental data. Proc. Natl. Acad. Sci. USA 2001, 98, 6859–6864. [Google Scholar] [CrossRef]
- Vandegriff, K.D.; Malavalli, A.; Lohman, J.; Young, M.A.; Terraneo, L.; Virgili, E.; Bianciardi, P.; Caretti, A.; Samaja, M. Impact of acellular hemoglobin-based oxygen carriers on brain apoptosis in rats. Transfusion 2014, 54, 2045–2054. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.; Malavalli, A.; Vandegriff, K. Correlation between hemoglobin oxygen affinity and nitrite reductase activity. Faseb J. 2012, 26, 573.4. [Google Scholar] [CrossRef]
- Minneci, P.C.; Deans, K.J.; Shiva, S.; Zhi, H.; Banks, S.M.; Kern, S.; Natanson, C.; Solomon, S.B.; Gladwin, M.T. Nitrite reductase activity of hemoglobin as a systemic nitric oxide generator mechanism to detoxify plasma hemoglobin produced during hemolysis. Am. J. Physiol. Heart Circ. Physiol. 2008, 295, H743–H754. [Google Scholar] [CrossRef]
- Terraneo, L.; Bianciardi, P.; Malavalli, A.; Mkrtchyan, G.; Spann, S.N.; Lohman, J.; Samaja, M.; Vandegriff, K.D. Hemoglobin extravasation in the brain of rats exchange-transfused with hemoglobin-based oxygen carriers. Artif. Cells Nanomed. Biotechnol. 2017, 45, 710–716. [Google Scholar] [CrossRef]
- Forstermann, U. Nitric oxide and oxidative stress in vascular disease. Pflug. Arch. 2010, 459, 923–939. [Google Scholar] [CrossRef] [PubMed]
- Guzik, T.J.; Korbut, R.; Adamek-Guzik, T. Nitric oxide and superoxide in inflammation and immune regulation. J. Physiol. Pharmacol. 2003, 54, 469–487. [Google Scholar] [PubMed]
- Tai, S.C.; Robb, G.B.; Marsden, P.A. Endothelial nitric oxide synthase: A new paradigm for gene regulation in the injured blood vessel. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 405–412. [Google Scholar] [CrossRef]
- Arnal, J.F.; Dinh-Xuan, A.T.; Pueyo, M.; Darblade, B.; Rami, J. Endothelium-derived nitric oxide and vascular physiology and pathology. Cell. Mol. Life Sci. 1999, 55, 1078–1087. [Google Scholar] [CrossRef] [PubMed]
- Caswell, J.E.; Strange, M.B.; Rimmer, D.M., 3rd; Gibson, M.F.; Cole, P.; Lefer, D.J. A novel hemoglobin-based blood substitute protects against myocardial reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 2005, 288, H1796–H1801. [Google Scholar] [CrossRef]
- Damle, S.S.; Moore, E.E.; Babu, A.N.; Meng, X.; Fullerton, D.A.; Banerjee, A. Hemoglobin-based oxygen carrier induces heme oxygenase-1 in the heart and lung but not brain. J. Am. Coll. Surg. 2009, 208, 592–598. [Google Scholar] [CrossRef]
- Babu, A.N.; Damle, S.S.; Moore, E.E.; Ao, L.; Song, Y.; Johnson, J.L.; Weyant, M.; Banerjee, A.; Meng, X.; Fullerton, D.A. Hemoglobin-based oxygen carrier induces hepatic heme oxygenase 1 expression in Kupffer cells. Surgery 2007, 142, 289–294. [Google Scholar] [CrossRef]
- Saetzler, R.K.; Arfors, K.E.; Tuma, R.F.; Vasthare, U.; Ma, L.; Hsia, C.J.; Lehr, H.A. Polynitroxylated hemoglobin-based oxygen carrier: Inhibition of free radical-induced microcirculatory dysfunction. Free Radic. Biol. Med. 1999, 27, 1–6. [Google Scholar] [CrossRef]
- Wang, G.R.; Zhu, Y.; Halushka, P.V.; Lincoln, T.M.; Mendelsohn, M.E. Mechanism of platelet inhibition by nitric oxide: In vivo phosphorylation of thromboxane receptor by cyclic GMP-dependent protein kinase. Proc. Natl. Acad. Sci. USA 1998, 95, 4888–4893. [Google Scholar] [CrossRef]
- Malfertheiner, M.V.; Garrett, A.; Passmore, M.; Haymet, A.B.; Webb, R.I.; Von Bahr, V.; Millar, J.E.; Schneider, B.A.; Obonyo, N.G.; Black, D.; et al. The effects of nitric oxide on coagulation and inflammation in ex vivo models of extracorporeal membrane oxygenation and cardiopulmonary bypass. Artif. Organs 2023. Online ahead of print. [Google Scholar] [CrossRef]
- Park, J.W.; Piknova, B.; Nghiem, K.; Lozier, J.N.; Schechter, A.N. Inhibitory effect of nitrite on coagulation processes demonstrated by thrombelastography. Nitric Oxide 2014, 40, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Bardossy, L.; Peterson, N.; Blajchman, M.A. o-raffinose cross-linked hemoglobin improves the hemostatic defect associated with anemia and thrombocytopenia in rabbits. Blood 2000, 96, 3630–3636. [Google Scholar] [CrossRef]
- Kim, H.W.; Stubdal, H.; Greenburg, A.G. Coagulation dynamics after hemodilution with polyhemoglobin. Surg. Gynecol. Obstet. 1992, 175, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, F.; Handrigan, M.; Hammett, M.; Philbin, N.; Rice, J.; Dong, F.; Pearce, L.B.; McCarron, R.; Freilich, D. Coagulation patterns following haemoglobin-based oxygen carrier resuscitation in severe uncontrolled haemorrhagic shock in swine. Transfus. Med. 2006, 16, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, F.; Hammett, M.; Asher, L.; Philbin, N.; Rice, J.; Dong, F.; Pearce, B.; Flournoy, W.S.; Nicholson, C.; McCarron, R.; et al. Effects of bovine polymerized hemoglobin on coagulation in controlled hemorrhagic shock in swine. Shock 2005, 24, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Jahr, J.S.; Liu, H.; Albert, O.K.; Gull, A.; Moallempour, M.; Lim, J.; Gosselin, R. Does HBOC-201 (Hemopure) affect platelet function in orthopedic surgery: A single-site analysis from a multicenter study. Am. J. Ther. 2010, 17, 140–147. [Google Scholar] [CrossRef] [PubMed]
- James, M.F.; Potgieter, H.E.; Ellis, P. The effect of hemopure on coagulation in clinically relevant concentrations. Anesth. Analg. 2004, 99, 1593–1597. [Google Scholar] [CrossRef] [PubMed]
- Leytin, V.; Mazer, D.; Mody, M.; Garvey, B.; Freedman, J. Hemolink, an o-raffinose cross-linked haemoglobin-based oxygen carrier, does not affect activation and function of human platelets in whole blood in vitro. Br. J. Haematol. 2003, 120, 535–541. [Google Scholar] [CrossRef]
- Jahr, J.S.; Mackenzie, C.; Pearce, L.B.; Pitman, A.; Greenburg, A.G. HBOC-201 as an alternative to blood transfusion: Efficacy and safety evaluation in a multicenter phase III trial in elective orthopedic surgery. J. Trauma. 2008, 64, 1484–1497. [Google Scholar] [CrossRef]
- Roghani, K.; Holtby, R.J.; Jahr, J.S. Effects of hemoglobin-based oxygen carriers on blood coagulation. J. Funct. Biomater. 2014, 5, 288–295. [Google Scholar] [CrossRef]
- Radi, R. Oxygen radicals, nitric oxide, and peroxynitrite: Redox pathways in molecular medicine. Proc. Natl. Acad. Sci. USA 2018, 115, 5839–5848. [Google Scholar] [CrossRef] [PubMed]
- Moon-Massat, P.; Scultetus, A.; Arnaud, F.; Brown, A.; Haque, A.; Saha, B.; Kim, B.; Sagini, E.; McGwin, G., Jr.; Auker, C.; et al. The effect HBOC-201 and sodium nitrite resuscitation after uncontrolled haemorrhagic shock in swine. Injury 2012, 43, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Zhang, X.; Williams, M.C.; Hicks, W.; Buehler, P.W.; D’Agnillo, F. Sodium nitrite potentiates renal oxidative stress and injury in hemoglobin exposed guinea pigs. Toxicology 2015, 333, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Chintagari, N.R.; Jana, S.; Alayash, A.I. Oxidized Ferric and Ferryl Forms of Hemoglobin Trigger Mitochondrial Dysfunction and Injury in Alveolar Type I Cells. Am. J. Respir. Cell Mol. Biol. 2016, 55, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.E.; Schaer, D.J.; Buehler, P.W.; Wilson, M.T.; Reeder, B.J.; Silkstone, G.; Svistunenko, D.A.; Bulow, L.; Alayash, A.I. Haptoglobin Binding Stabilizes Hemoglobin Ferryl Iron and the Globin Radical on Tyrosine beta145. Antioxid. Redox Signal. 2013, 18, 2264–2273. [Google Scholar] [CrossRef]
- Benderro, G.F.; Sun, X.; Kuang, Y.; Lamanna, J.C. Decreased VEGF expression and microvascular density, but increased HIF-1 and 2alpha accumulation and EPO expression in chronic moderate hyperoxia in the mouse brain. Brain Res. 2012, 1471, 46–55. [Google Scholar] [CrossRef]
- Lee, E.S.; Smith, W.E.; Quach, H.T.; Jones, B.D.; Santilli, S.M.; Vatassery, G.T. Moderate hyperoxia (40%) increases antioxidant levels in mouse tissue. J. Surg. Res. 2005, 127, 80–84. [Google Scholar] [CrossRef]
- Sorce, S.; Krause, K.H. NOX enzymes in the central nervous system: From signaling to disease. Antioxid. Redox Signal. 2009, 11, 2481–2504. [Google Scholar] [CrossRef]
- Nanduri, J.; Vaddi, D.R.; Khan, S.A.; Wang, N.; Makarenko, V.; Semenza, G.L.; Prabhakar, N.R. HIF-1α activation by intermittent hypoxia requires NADPH oxidase stimulation by xanthine oxidase. PLoS ONE 2015, 10, e0119762. [Google Scholar] [CrossRef]
- Sethy, N.K.; Singh, M.; Kumar, R.; Ilavazhagan, G.; Bhargava, K. Upregulation of transcription factor NRF2-mediated oxidative stress response pathway in rat brain under short-term chronic hypobaric hypoxia. Funct. Integr. Genom. 2011, 11, 119–137. [Google Scholar] [CrossRef]
- Ma, Q. Role of nrf2 in oxidative stress and toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Ren, C.; Chen, X.; Shen, J. From rapid to delayed and remote postconditioning: The evolving concept of ischemic postconditioning in brain ischemia. Curr. Drug Targets 2012, 13, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Chen, Z.; Chen, J.; Chen, H. Isoflurane preconditioning protects rat brain from ischemia reperfusion injury via up-regulating the HIF-1alpha expression through Akt/mTOR/s6K activation. Cell. Mol. Biol. 2016, 62, 38–44. [Google Scholar] [PubMed]
- Terraneo, L.; Paroni, R.; Bianciardi, P.; Giallongo, T.; Carelli, S.; Gorio, A.; Samaja, M. Brain adaptation to hypoxia and hyperoxia in mice. Redox Biol. 2017, 11, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Mejía-García, T.A.; Portugal, C.C.; Encarnação, T.G.; Prado, M.A.; Paes-de-Carvalho, R. Nitric oxide regulates AKT phosphorylation and nuclear translocation in cultured retinal cells. Cell. Signal. 2013, 25, 2424–2439. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.C. Nitric oxide and neuronal death. Nitric Oxide 2010, 23, 153–165. [Google Scholar] [CrossRef]
- Samaja, M.; Crespi, T.; Guazzi, M.; Vandegriff, K.D. Oxygen transport in blood at high altitude: Role of the hemoglobin-oxygen affinity and impact of the phenomena related to hemoglobin allosterism and red cell function. Eur. J. Appl. Physiol. 2003, 90, 351–359. [Google Scholar] [CrossRef]
- Sehgal, L.; Rosen, A.; Gould, S.; Sehgal, H.; Dalton, L.; Mayoral, M.; Moss, G. In vitro and in vivo characteristics of polymerized pyridoxalated hemoglobin solutions. Fed. Proc. 1980, 39, 2383. [Google Scholar]
- Eike, J.H.; Palmer, A.F. Effect of Cl− and H+ on the oxygen binding properties of glutaraldehyde-polymerized bovine hemoglobin-based blood substitutes. Biotechnol. Prog. 2004, 20, 1543–1549. [Google Scholar] [CrossRef]
- Vandegriff, K.D.; Malavalli, A.; Minn, C.; Jiang, E.; Lohman, J.; Young, M.A.; Samaja, M.; Winslow, R.M. Oxidation and haem loss kinetics of poly(ethylene glycol)-conjugated haemoglobin (MP4): Dissociation between in vitro and in vivo oxidation rates. Biochem. J. 2006, 399, 463–471. [Google Scholar] [CrossRef]
- Samaja, M.; Rovida, E.; Niggeler, M.; Perrella, M.; Rossi Bernardi, L. The dissociation of carbon monoxide from hemoglobin intermediates. J. Biol. Chem. 1987, 262, 4528–4533. [Google Scholar] [CrossRef] [PubMed]
- Chu, A.H.; Ackers, G.K. Mutual effects of protons, NaCl, and oxygen on the dimer-tetramer assembly of human hemoglobin. The dimer Bohr effect. J. Biol. Chem. 1981, 256, 1199–1205. [Google Scholar] [CrossRef] [PubMed]
- Samaja, M.; Mosca, A.; Perrella, M.; Rovida, E.; Niggeler, M.; Rossi Bernardi, L. Respiratory function of blood and the mechanism of oxygen transport. Med. Sport. Sci. 1985, 19, 39–47. [Google Scholar]
- Buehler, P.W.; Abraham, B.; Vallelian, F.; Linnemayr, C.; Pereira, C.P.; Cipollo, J.F.; Jia, Y.; Mikolajczyk, M.; Boretti, F.S.; Schoedon, G.; et al. Haptoglobin preserves the CD163 hemoglobin scavenger pathway by shielding hemoglobin from peroxidative modification. Blood 2009, 113, 2578–2586. [Google Scholar] [CrossRef]
- Roche, C.J.; Dantsker, D.; Alayash, A.I.; Friedman, J.M. Enhanced nitrite reductase activity associated with the haptoglobin complexed hemoglobin dimer: Functional and antioxidative implications. Nitric Oxide 2012, 27, 32–39. [Google Scholar] [CrossRef]
- Boretti, F.S.; Buehler, P.W.; D’Agnillo, F.; Kluge, K.; Glaus, T.; Butt, O.I.; Jia, Y.; Goede, J.; Pereira, C.P.; Maggiorini, M.; et al. Sequestration of extracellular hemoglobin within a haptoglobin complex decreases its hypertensive and oxidative effects in dogs and guinea pigs. J. Clin. Investig. 2009, 119, 2271–2280. [Google Scholar] [CrossRef] [PubMed]
- Snyder, S.R.; Welty, E.V.; Walder, R.Y.; Williams, L.A.; Walder, J.A. HbXL99 alpha: A hemoglobin derivative that is cross-linked between the alpha subunits is useful as a blood substitute. Proc. Natl. Acad. Sci. USA 1987, 84, 7280–7284. [Google Scholar] [CrossRef]
- Walder, J.A.; Zaugg, R.H.; Walder, R.Y.; Steele, J.M.; Klotz, I.M. Diaspirins that cross-link beta chains of hemoglobin: Bis(3,5-dibromosalicyl) succinate and bis(3,5-dibromosalicyl) fumarate. Biochemistry 1979, 18, 4265–4270. [Google Scholar] [CrossRef]
- Chatterjee, R.; Welty, E.V.; Walder, R.Y.; Pruitt, S.L.; Rogers, P.H.; Arnone, A.; Walder, J.A. Isolation and characterization of a new hemoglobin derivative cross-linked between the alpha chains (lysine 99 alpha 1----lysine 99 alpha 2). J. Biol. Chem. 1986, 261, 9929–9937. [Google Scholar] [CrossRef]
- Gulati, A.; Sen, A.P. Dose-dependent effect of diaspirin cross-linked hemoglobin on regional blood circulation of severely hemorrhaged rats. Shock 1998, 9, 65–73. [Google Scholar] [CrossRef]
- Schultz, S.C.; Hamilton, I.N., Jr.; Malcolm, D.S. Use of base deficit to compare resuscitation with lactated Ringer’s solution, Haemaccel, whole blood, and diaspirin cross-linked hemoglobin following hemorrhage in rats. J. Trauma. 1993, 35, 619–625; discussion 625-616. [Google Scholar] [CrossRef]
- Sloan, E.P.; Koenigsberg, M.; Gens, D.; Cipolle, M.; Runge, J.; Mallory, M.N.; Rodman, G., Jr. Diaspirin cross-linked hemoglobin (DCLHb) in the treatment of severe traumatic hemorrhagic shock: A randomized controlled efficacy trial. JAMA 1999, 282, 1857–1864. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.; Kluger, R. Functional cross-linked hemoglobin bis-tetramers: Geometry and cooperativity. Biochemistry 2008, 47, 12551–12561. [Google Scholar] [CrossRef] [PubMed]
- Kuang, L.; Zhu, Y.; Wu, Y.; Tian, K.; Peng, X.; Xue, M.; Xiang, X.; Lau, B.; Tzang, F.C.; Liu, L.; et al. A Novel Cross-Linked Hemoglobin-Based Oxygen Carrier, YQ23, Extended the Golden Hour for Uncontrolled Hemorrhagic Shock in Rats and Miniature Pigs. Front. Pharmacol. 2021, 12, 652716. [Google Scholar] [CrossRef]
- Chang, T.M. Semipermeable Microcapsules. Science 1964, 146, 524–525. [Google Scholar] [CrossRef]
- Chang, T.M. Stabilisation of enzymes by microencapsulation with a concentrated protein solution or by microencapsulation followed by cross-linking with glutaraldehyde. Biochem. Biophys. Res. Commun. 1971, 44, 1531–1536. [Google Scholar] [CrossRef]
- Moore, E.E.; Moore, F.A.; Fabian, T.C.; Bernard, A.C.; Fulda, G.J.; Hoyt, D.B.; Duane, T.M.; Weireter, L.J., Jr.; Gomez, G.A.; Cipolle, M.D.; et al. Human polymerized hemoglobin for the treatment of hemorrhagic shock when blood is unavailable: The USA multicenter trial. J. Am. Coll. Surg. 2009, 208, 1–13. [Google Scholar] [CrossRef]
- D’Agnillo, F.; Chang, T.M. Polyhemoglobin-superoxide dismutase-catalase as a blood substitute with antioxidant properties. Nat. Biotechnol. 1998, 16, 667–671. [Google Scholar] [CrossRef] [PubMed]
- Powanda, D.D.; Chang, T.M. Cross-linked polyhemoglobin-superoxide dismutase-catalase supplies oxygen without causing blood-brain barrier disruption or brain edema in a rat model of transient global brain ischemia-reperfusion. Artif. Cells Blood Substit. Immobil. Biotechnol. 2002, 30, 23–37. [Google Scholar] [CrossRef] [PubMed]
- Alayash, A.I. Oxygen therapeutics: Can we tame haemoglobin? Nat. Rev. Drug Discov. 2004, 3, 152–159. [Google Scholar] [CrossRef]
- Cabrales, P.; Intaglietta, M. Blood substitutes: Evolution from noncarrying to oxygen- and gas-carrying fluids. ASAIO J. 2013, 59, 337–354. [Google Scholar] [CrossRef]
- Williams, A.T.; Muller, C.R.; Eaker, A.M.; Belcher, D.A.; Bolden-Rush, C.; Palmer, A.F.; Cabrales, P. Polymerized Hemoglobin With Increased Molecular Size Reduces Toxicity in Healthy Guinea Pigs. ACS Appl. Bio Mater. 2020, 3, 2976–2985. [Google Scholar] [CrossRef] [PubMed]
- Manjula, B.N.; Tsai, A.; Upadhya, R.; Perumalsamy, K.; Smith, P.K.; Malavalli, A.; Vandegriff, K.; Winslow, R.M.; Intaglietta, M.; Prabhakaran, M.; et al. Site-specific PEGylation of hemoglobin at Cys-93(beta): Correlation between the colligative properties of the PEGylated protein and the length of the conjugated PEG chain. Bioconjug Chem. 2003, 14, 464–472. [Google Scholar] [CrossRef]
- Vandegriff, K.D.; Malavalli, A.; Wooldridge, J.; Lohman, J.; Winslow, R.M. MP4, a new nonvasoactive PEG-Hb conjugate. Transfusion 2003, 43, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Juszczak, L.J.; Manjula, B.; Bonaventura, C.; Acharya, S.A.; Friedman, J.M. UV resonance Raman study of beta93-modified hemoglobin A: Chemical modifier-specific effects and added influences of attached poly(ethylene glycol) chains. Biochemistry 2002, 41, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Manjula, B.N.; Tsai, A.G.; Intaglietta, M.; Tsai, C.H.; Ho, C.; Smith, P.K.; Perumalsamy, K.; Kanika, N.D.; Friedman, J.M.; Acharya, S.A. Conjugation of multiple copies of polyethylene glycol to hemoglobin facilitated through thiolation: Influence on hemoglobin structure and function. Protein J. 2005, 24, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, L.; Yu, W.; Gao, D.; You, G.; Li, P.; Zhang, S.; Zhang, J.; Hu, T.; Zhao, L.; et al. A PEGylated bovine hemoglobin as a potent hemoglobin-based oxygen carrier. Biotechnol. Prog. 2017, 33, 252–260. [Google Scholar] [CrossRef]
- Sakai, H.; Hara, H.; Yuasa, M.; Tsai, A.; Takeoka, S.; Tsuchida, E.; Intaglietta, M. Molecular dimensions of Hb—Based oxygen carriers determine constriction of resistance arteries and hypertension. Am. J. Physiol. 2000, 279, H908–H915. [Google Scholar]
- Song, B.K.; Light, W.R.; Vandegriff, K.D.; Tucker, J.; Nugent, W.H. Systemic and microvascular comparison of Lactated Ringer’s solution, VIR-HBOC, and alpha-alpha crosslinked haemoglobin-based oxygen carrier in a rat 10% topload model. Artif. Cells Nanomed. Biotechnol. 2020, 48, 1079–1088. [Google Scholar] [CrossRef]
- Cooper, C.E.; Silkstone, G.G.A.; Simons, M.; Gretton, S.; Rajagopal, B.S.; Allen-Baume, V.; Syrett, N.; Shaik, T.; Popa, G.; Sheng, X.; et al. Engineering hemoglobin to enable homogenous PEGylation without modifying protein functionality. Biomater. Sci. 2020, 8, 3896–3906. [Google Scholar] [CrossRef]
- Ishchuk, O.P.; Frost, A.T.; Muñiz-Paredes, F.; Matsumoto, S.; Laforge, N.; Eriksson, N.L.; Martínez, J.L.; Petranovic, D. Improved production of human hemoglobin in yeast by engineering hemoglobin degradation. Metab. Eng. 2021, 66, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Shi, Z.; Gao, N.; Zhou, Y.; Ni, X.; Chen, J.; Liu, J.; Zhou, W.; Guo, X.; Xin, B.; et al. Sustainable and high-level microbial production of plant hemoglobin in Corynebacterium glutamicum. Biotechnol. Biofuels Bioprod. 2023, 16, 80. [Google Scholar] [CrossRef] [PubMed]
- Frost, A.T.; Jacobsen, I.H.; Worberg, A.; Martinez, J.L. How Synthetic Biology and Metabolic Engineering Can Boost the Generation of Artificial Blood Using Microbial Production Hosts. Front. Bioeng. Biotechnol. 2018, 6, 186. [Google Scholar] [CrossRef]
- Bulow, L.; Alayash, A.I. Redox Chemistry of Hemoglobin-Associated Disorders. Antioxid. Redox Signal. 2017, 26, 745–747. [Google Scholar] [CrossRef]
- Strader, M.B.; Alayash, A.I. Exploring Oxidative Reactions in Hemoglobin Variants Using Mass Spectrometry: Lessons for Engineering Oxidatively Stable Oxygen Therapeutics. Antioxid. Redox Signal. 2017, 26, 777–793. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, J.; Bonaventura, C.; Sullivan, B.; Ferruzzi, G.; McCurdy, P.R.; Fox, J.; Moo-Penn, W.F. Hemoglobin providence. Functional consequences of two alterations of the 2,3-diphosphoglycerate binding site at position beta 82. J. Biol. Chem. 1976, 251, 7563–7571. [Google Scholar] [CrossRef] [PubMed]
- Strader, M.B.; Bangle, R.; Parker Siburt, C.J.; Varnado, C.L.; Soman, J.; Benitez Cardenas, A.S.; Samuel, P.P.; Singleton, E.W.; Crumbliss, A.L.; Olson, J.S.; et al. Engineering oxidative stability in human hemoglobin based on the Hb providence (βK82D) mutation and genetic cross-linking. Biochem. J. 2017, 474, 4171–4192. [Google Scholar] [CrossRef] [PubMed]
- Jana, S.; Strader, M.B.; Alayash, A.I. The Providence Mutation (βK82D) in Human Hemoglobin Substantially Reduces βCysteine 93 Oxidation and Oxidative Stress in Endothelial Cells. Int. J. Mol. Sci. 2020, 21, 9453. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.; Gretton, S.; Silkstone, G.G.A.; Rajagopal, B.S.; Allen-Baume, V.; Syrett, N.; Shaik, T.; Leiva-Eriksson, N.; Ronda, L.; Mozzarelli, A.; et al. Comparison of the oxidative reactivity of recombinant fetal and adult human hemoglobin: Implications for the design of hemoglobin-based oxygen carriers. Biosci. Rep. 2018, 38, BSR20180370. [Google Scholar] [CrossRef]
- Kettisen, K.; Strader, M.B.; Wood, F.; Alayash, A.I.; Bülow, L. Site-directed mutagenesis of cysteine residues alters oxidative stability of fetal hemoglobin. Redox Biol. 2018, 19, 218–225. [Google Scholar] [CrossRef]
- Prapan, A.; Suwannasom, N.; Kloypan, C.; Chaiwaree, S.; Steffen, A.; Xiong, Y.; Kao, I.; Pruß, A.; Georgieva, R.; Bäumler, H. Surface Modification of Hemoglobin-Based Oxygen Carriers Reduces Recognition by Haptoglobin, Immunoglobulin, and Hemoglobin Antibodies. Coatings 2019, 9, 454. [Google Scholar] [CrossRef]
- Alayash, A.I. βCysteine 93 in human hemoglobin: A gateway to oxidative stability in health and disease. Lab. Investig. 2021, 101, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Looker, D.; Mathews, A.J.; Neway, J.O.; Stetler, G.L. Expression of recombinant human hemoglobin in Escherichia coli. Methods Enzymol. 1994, 231, 364–374. [Google Scholar]
- Bobofchak, K.M.; Mito, T.; Texel, S.J.; Bellelli, A.; Nemoto, M.; Traystman, R.J.; Koehler, R.C.; Brinigar, W.S.; Fronticelli, C. A recombinant polymeric hemoglobin with conformational, functional, and physiological characteristics of an in vivo O2 transporter. Am. J. Physiol. Heart Circ. Physiol. 2003, 285, H549–H561. [Google Scholar] [CrossRef] [PubMed]
- Fablet, C.; Marden, M.C.; Green, B.N.; Ho, C.; Pagnier, J.; Baudin-Creuza, V. Stable octameric structure of recombinant hemoglobin alpha(2)beta(2)83 Gly-->Cys. Protein Sci. 2003, 12, 690–695. [Google Scholar] [CrossRef] [PubMed]
- Varnado, C.L.; Mollan, T.L.; Birukou, I.; Smith, B.J.; Henderson, D.P.; Olson, J.S. Development of recombinant hemoglobin-based oxygen carriers. Antioxid. Redox Signal. 2013, 18, 2314–2328. [Google Scholar] [CrossRef] [PubMed]
- DuMond, J.F.; King, S.B. The chemistry of nitroxyl-releasing compounds. Antioxid. Redox Signal. 2011, 14, 1637–1648. [Google Scholar] [CrossRef]
- Hsia, C.J.; Ma, L. A hemoglobin-based multifunctional therapeutic: Polynitroxylated pegylated hemoglobin. Artif. Organs 2012, 36, 215–220. [Google Scholar] [CrossRef]
- Brockman, E.C.; Bayır, H.; Blasiole, B.; Shein, S.L.; Fink, E.L.; Dixon, C.; Clark, R.S.; Vagni, V.A.; Ma, L.; Hsia, C.J.; et al. Polynitroxylated-pegylated hemoglobin attenuates fluid requirements and brain edema in combined traumatic brain injury plus hemorrhagic shock in mice. J. Cereb. Blood Flow. Metab. 2013, 33, 1457–1464. [Google Scholar] [CrossRef]
- Brockman, E.C.; Jackson, T.C.; Dixon, C.E.; Bayɪr, H.; Clark, R.S.; Vagni, V.; Feldman, K.; Byrd, C.; Ma, L.; Hsia, C.; et al. Polynitroxylated Pegylated Hemoglobin-A Novel, Small Volume Therapeutic for Traumatic Brain Injury Resuscitation: Comparison to Whole Blood and Dose Response Evaluation. J. Neurotrauma 2017, 34, 1337–1350. [Google Scholar] [CrossRef]
- Shellington, D.K.; Du, L.; Wu, X.; Exo, J.; Vagni, V.; Ma, L.; Janesko-Feldman, K.; Clark, R.S.; Bayir, H.; Dixon, C.E.; et al. Polynitroxylated pegylated hemoglobin: A novel neuroprotective hemoglobin for acute volume-limited fluid resuscitation after combined traumatic brain injury and hemorrhagic hypotension in mice. Crit. Care Med. 2011, 39, 494–505. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Zhang, J.; Ma, L.; Hsia, C.J.C.; Koehler, R.C. Transfusion of Polynitroxylated Pegylated Hemoglobin Stabilizes Pial Arterial Dilation and Decreases Infarct Volume After Transient Middle Cerebral Artery Occlusion. J. Am. Heart Assoc. 2017, 6, e006505. [Google Scholar] [CrossRef] [PubMed]
- Fagone, P.; Mazzon, E.; Bramanti, P.; Bendtzen, K.; Nicoletti, F. Gasotransmitters and the immune system: Mode of action and novel therapeutic targets. Eur. J. Pharmacol. 2018, 834, 92–102. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, D.; Fu, X.; Yu, L.; Lu, Z.; Gao, Y.; Liu, X.; Man, J.; Li, S.; Li, N.; et al. Carbon monoxide-releasing molecule-3 protects against ischemic stroke by suppressing neuroinflammation and alleviating blood-brain barrier disruption. J. Neuroinflamm. 2018, 15, 188. [Google Scholar] [CrossRef]
- Wang, J.; Shi, Y.; Cao, S.; Liu, X.; Martin, L.J.; Simoni, J.; Soltys, B.J.; Hsia, C.J.C.; Koehler, R.C. Polynitroxylated PEGylated hemoglobin protects pig brain neocortical gray and white matter after traumatic brain injury and hemorrhagic shock. Front. Med. Technol. 2023, 5, 1074643. [Google Scholar] [CrossRef] [PubMed]
- Krishna, M.C.; Russo, A.; Mitchell, J.B.; Goldstein, S.; Dafni, H.; Samuni, A. Do nitroxide antioxidants act as scavengers of O2−. or as SOD mimics? J. Biol. Chem. 1996, 271, 26026–26031. [Google Scholar] [CrossRef] [PubMed]
- Buehler, P.W.; Haney, C.R.; Gulati, A.; Ma, L.; Hsia, C.J. Polynitroxyl hemoglobin: A pharmacokinetic study of covalently bound nitroxides to hemoglobin platforms. Free Radic. Biol. Med. 2004, 37, 124–135. [Google Scholar] [CrossRef]
- Okayama, N.; Park, J.H.; Coe, L.; Granger, D.N.; Ma, L.; Hisa, C.J.; Alexander, J.S. Polynitroxyl alphaalpha-hemoglobin (PNH) inhibits peroxide and superoxide-mediated neutrophil adherence to human endothelial cells. Free Radic. Res. 1999, 31, 53–58. [Google Scholar] [CrossRef]
- Stoyanovsky, D.A.; Kapralov, A.; Huang, Z.; Maeda, A.; Osipov, A.; Hsia, C.J.; Ma, L.; Kochanek, P.M.; Bayr, H.; Kagan, V.E. Unusual peroxidase activity of polynitroxylated pegylated hemoglobin: Elimination of H2O2 coupled with intramolecular oxidation of nitroxides. Biochem. Biophys. Res. Commun. 2010, 399, 139–143. [Google Scholar] [CrossRef]
- Seno, S.; Wang, J.; Cao, S.; Saraswati, M.; Park, S.; Simoni, J.; Ma, L.; Soltys, B.; Hsia, C.J.C.; Koehler, R.C.; et al. Resuscitation with macromolecular superoxide dismutase/catalase mimetic polynitroxylated PEGylated hemoglobin offers neuroprotection in guinea pigs after traumatic brain injury combined with hemorrhage shock. BMC Neurosci. 2020, 21, 22. [Google Scholar] [CrossRef]
- Chang, T.M. Therapeutic applications of polymeric artificial cells. Nat. Rev. Drug Discov. 2005, 4, 221–235. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.C.; Chang, T.M. Long-term effects on the histology and function of livers and spleens in rats after 33% toploading of PEG-PLA-nano artificial red blood cells. Artif. Cells Blood Substit. Immobil. Biotechnol. 2008, 36, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.C.; Chang, T.M. Effects of PEG-PLA-nano artificial cells containing hemoglobin on kidney function and renal histology in rats. Artif. Cells Blood Substit. Immobil. Biotechnol. 2008, 36, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Sakai, H.; Sato, A.; Masuda, K.; Takeoka, S.; Tsuchida, E. Encapsulation of concentrated hemoglobin solution in phospholipid vesicles retards the reaction with NO, but not CO, by intracellular diffusion barrier. J. Biol. Chem. 2008, 283, 1508–1517. [Google Scholar] [CrossRef] [PubMed]
- Azarov, I.; Huang, K.T.; Basu, S.; Gladwin, M.T.; Hogg, N.; Kim-Shapiro, D.B. Nitric oxide scavenging by red blood cells as a function of hematocrit and oxygenation. J. Biol. Chem. 2005, 280, 39024–39032. [Google Scholar] [CrossRef]
- Huang, K.T.; Huang, Z.; Kim-Shapiro, D.B. Nitric oxide red blood cell membrane permeability at high and low oxygen tension. Nitric Oxide 2007, 16, 209–216. [Google Scholar] [CrossRef]
- Bian, Y.; Chang, T.M.S. Nanobiotechnological basis of an oxygen carrier with enhanced carbonic anhydrase for CO2 transport and enhanced catalase and superoxide dismutase for antioxidant function. Front. Bioeng. Biotechnol. 2023, 11, 1188399. [Google Scholar] [CrossRef]
- Guo, C.; Chang, T.M.S. Long term safety and immunological effects of a nanobiotherapeutic, bovine poly-[hemoglobin-catalase-superoxide dismutase-carbonic anhydrase], after four weekly 5% blood volume top-loading followed by a challenge of 30% exchange transfusion. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1349–1363. [Google Scholar] [CrossRef]
- Liochev, S.I.; Kuchumov, A.R.; Vinogradov, S.N.; Fridovich, I. Superoxide dismutase activity in the giant hemoglobin of the earthworm, Lumbricus terrestris. Arch. Biochem. Biophys. 1996, 330, 281–284. [Google Scholar] [CrossRef]
- Royer, W.E., Jr.; Sharma, H.; Strand, K.; Knapp, J.E.; Bhyravbhatla, B. Lumbricus erythrocruorin at 3.5 A resolution: Architecture of a megadalton respiratory complex. Structure 2006, 14, 1167–1177. [Google Scholar] [CrossRef]
- Kruczkowska, W.; Kciuk, M.; Pasieka, Z.; Kłosiński, K.; Płuciennik, E.; Elmer, J.; Waszczykowska, K.; Kołat, D.; Kałuzińska-Kołat, Ż. The artificial oxygen carrier erythrocruorin-characteristics and potential significance in medicine. J. Mol. Med. 2023, 101, 961–972. [Google Scholar] [CrossRef] [PubMed]
- Batool, F.; Delpy, E.; Zal, F.; Leize-Zal, E.; Huck, O. Therapeutic Potential of Hemoglobin Derived from the Marine Worm Arenicola marina (M101): A Literature Review of a Breakthrough Innovation. Mar. Drugs 2021, 19, 376. [Google Scholar] [CrossRef] [PubMed]
- Rousselot, M.; Delpy, E.; Drieu La Rochelle, C.; Lagente, V.; Pirow, R.; Rees, J.F.; Hagege, A.; Le Guen, D.; Hourdez, S.; Zal, F. Arenicola marina extracellular hemoglobin: A new promising blood substitute. Biotechnol. J. 2006, 1, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Elmer, J.; Zorc, K.; Rameez, S.; Zhou, Y.; Cabrales, P.; Palmer, A.F. Hypervolemic infusion of Lumbricus terrestris erythrocruorin purified by tangential-flow filtration. Transfusion 2012, 52, 1729–1740. [Google Scholar] [CrossRef]
- Strand, K.; Knapp, J.E.; Bhyravbhatla, B.; Royer, W.E., Jr. Crystal structure of the hemoglobin dodecamer from Lumbricus erythrocruorin: Allosteric core of giant annelid respiratory complexes. J. Mol. Biol. 2004, 344, 119–134. [Google Scholar] [CrossRef] [PubMed]
- Roche, C.J.; Talwar, A.; Palmer, A.F.; Cabrales, P.; Gerfen, G.; Friedman, J.M. Evaluating the capacity to generate and preserve nitric oxide bioactivity in highly purified earthworm erythrocruorin: A giant polymeric hemoglobin with potential blood substitute properties. J. Biol. Chem. 2015, 290, 99–117. [Google Scholar] [CrossRef]
- Tsai, A.G.; Intaglietta, M.; Sakai, H.; Delpy, E.; La Rochelle, C.D.; Rousselot, M.; Zal, F. Microcirculation and NO-CO studies of a natural extracellular hemoglobin developed for an oxygen therapeutic carrier. Curr. Drug Discov. Technol. 2012, 9, 166–172. [Google Scholar] [CrossRef]
- Cabrales, P.; Friedman, J.M. HBOC Vasoactivity: Interplay Between Nitric Oxide Scavenging and Capacity to Generate Bioactive Nitric Oxide Species. Antioxid. Redox Signal. 2013, 18, 2284–2297. [Google Scholar] [CrossRef]
- Le Meur, Y.; Delpy, E.; Renard, F.; Hauet, T.; Badet, L.; Rerolle, J.P.; Thierry, A.; Buchler, M.; Zal, F.; Barrou, B. HEMO(2) life(R) improves renal function independent of cold ischemia time in kidney recipients: A comparison with a large multicenter prospective cohort study. Artif. Organs 2022, 46, 597–605. [Google Scholar] [CrossRef]
- Asong-Fontem, N.; Panisello-Rosello, A.; Lopez, A.; Imai, K.; Zal, F.; Delpy, E.; Rosello-Catafau, J.; Adam, R. A Novel Oxygen Carrier (M101) Attenuates Ischemia-Reperfusion Injuries during Static Cold Storage in Steatotic Livers. Int. J. Mol. Sci. 2021, 22, 8542. [Google Scholar] [CrossRef]
- Ali, A.; Watanabe, Y.; Galasso, M.; Watanabe, T.; Chen, M.; Fan, E.; Brochard, L.; Ramadan, K.; Ribeiro, R.V.P.; Stansfield, W.; et al. An extracellular oxygen carrier during prolonged pulmonary preservation improves post-transplant lung function. J. Heart Lung Transplant. 2020, 39, 595–603. [Google Scholar] [CrossRef]
- Alix, P.; Val-Laillet, D.; Turlin, B.; Ben Mosbah, I.; Burel, A.; Bobillier, E.; Bendavid, C.; Delpy, E.; Zal, F.; Corlu, A.; et al. Adding the oxygen carrier M101 to a cold-storage solution could be an alternative to HOPE for liver graft preservation. JHEP Rep. 2020, 2, 100119. [Google Scholar] [CrossRef] [PubMed]
- Batool, F.; Stutz, C.; Petit, C.; Benkirane-Jessel, N.; Delpy, E.; Zal, F.; Leize-Zal, E.; Huck, O. A therapeutic oxygen carrier isolated from Arenicola marina decreased P. gingivalis induced inflammation and tissue destruction. Sci. Rep. 2020, 10, 14745. [Google Scholar] [CrossRef] [PubMed]
- Batool, F.; Petit, C.; Stutz, C.; Ozcelik, H.; Gegout, P.Y.; Benkirane-Jessel, N.; Delpy, E.; Zal, F.; Leize-Zal, E.; Huck, O. M101, a therapeutic oxygen carrier derived from Arenicola marina, decreased Porphyromonas gingivalis-induced hypoxia and improved periodontal healing. J. Periodontol. 2022, 93, 1712–1724. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samaja, M.; Malavalli, A.; Vandegriff, K.D. How Nitric Oxide Hindered the Search for Hemoglobin-Based Oxygen Carriers as Human Blood Substitutes. Int. J. Mol. Sci. 2023, 24, 14902. https://doi.org/10.3390/ijms241914902
Samaja M, Malavalli A, Vandegriff KD. How Nitric Oxide Hindered the Search for Hemoglobin-Based Oxygen Carriers as Human Blood Substitutes. International Journal of Molecular Sciences. 2023; 24(19):14902. https://doi.org/10.3390/ijms241914902
Chicago/Turabian StyleSamaja, Michele, Ashok Malavalli, and Kim D. Vandegriff. 2023. "How Nitric Oxide Hindered the Search for Hemoglobin-Based Oxygen Carriers as Human Blood Substitutes" International Journal of Molecular Sciences 24, no. 19: 14902. https://doi.org/10.3390/ijms241914902





