Unexpected Crosslinking Effects of a Human Thyroid Stimulating Monoclonal Autoantibody, M22, with IGF1 on Adipogenesis in 3T3L-1 Cells
Abstract
:1. Introduction
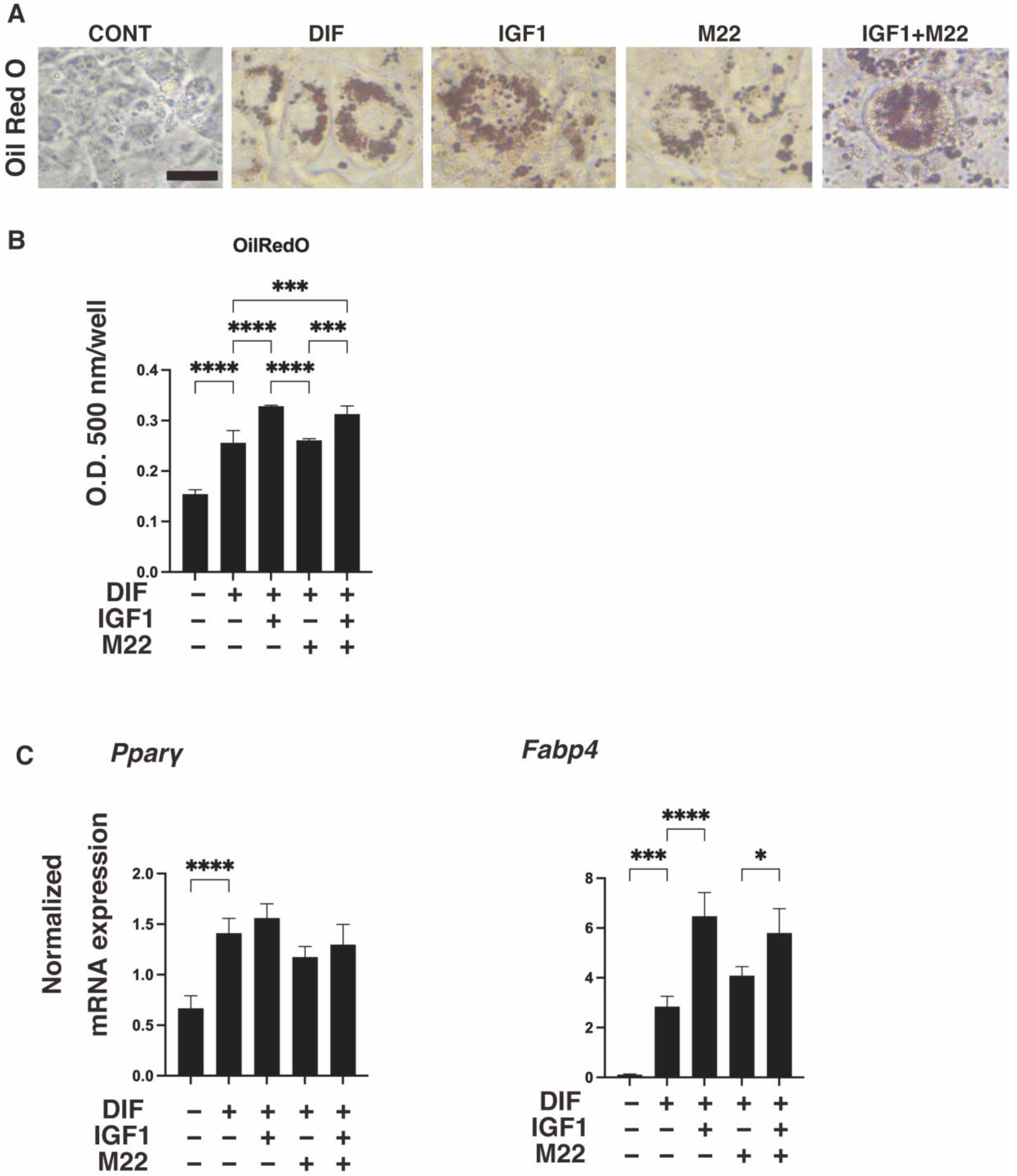
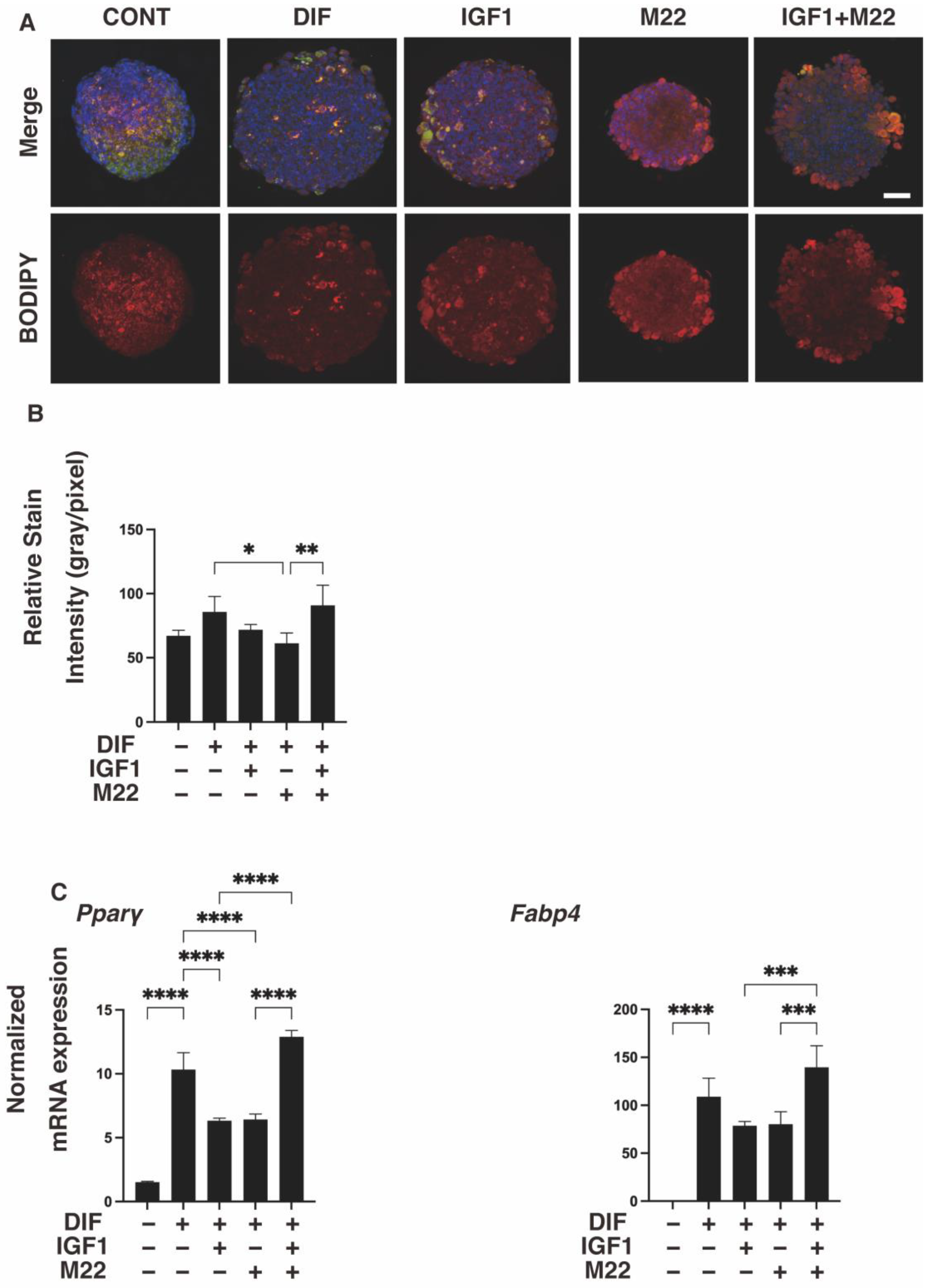
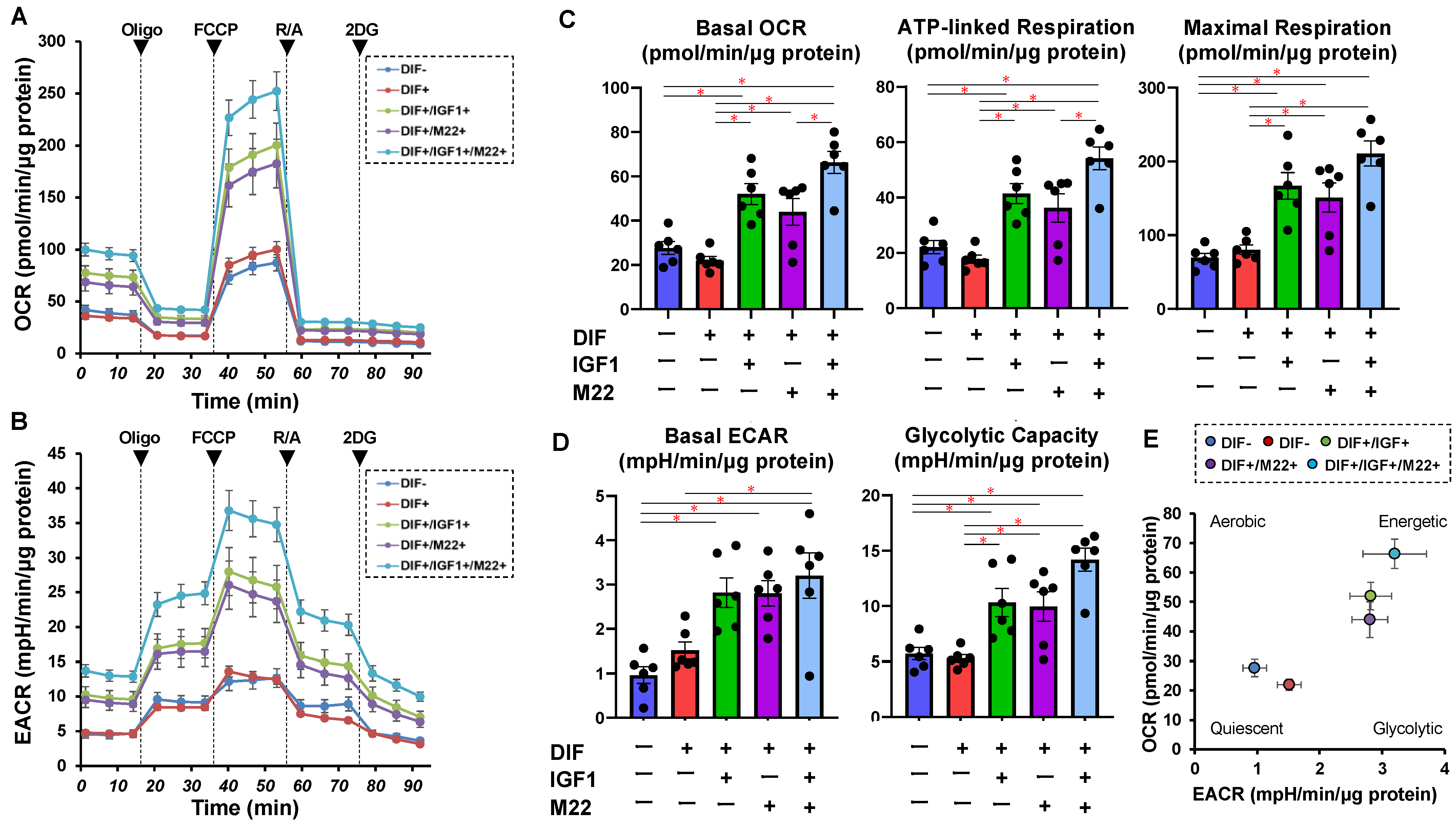
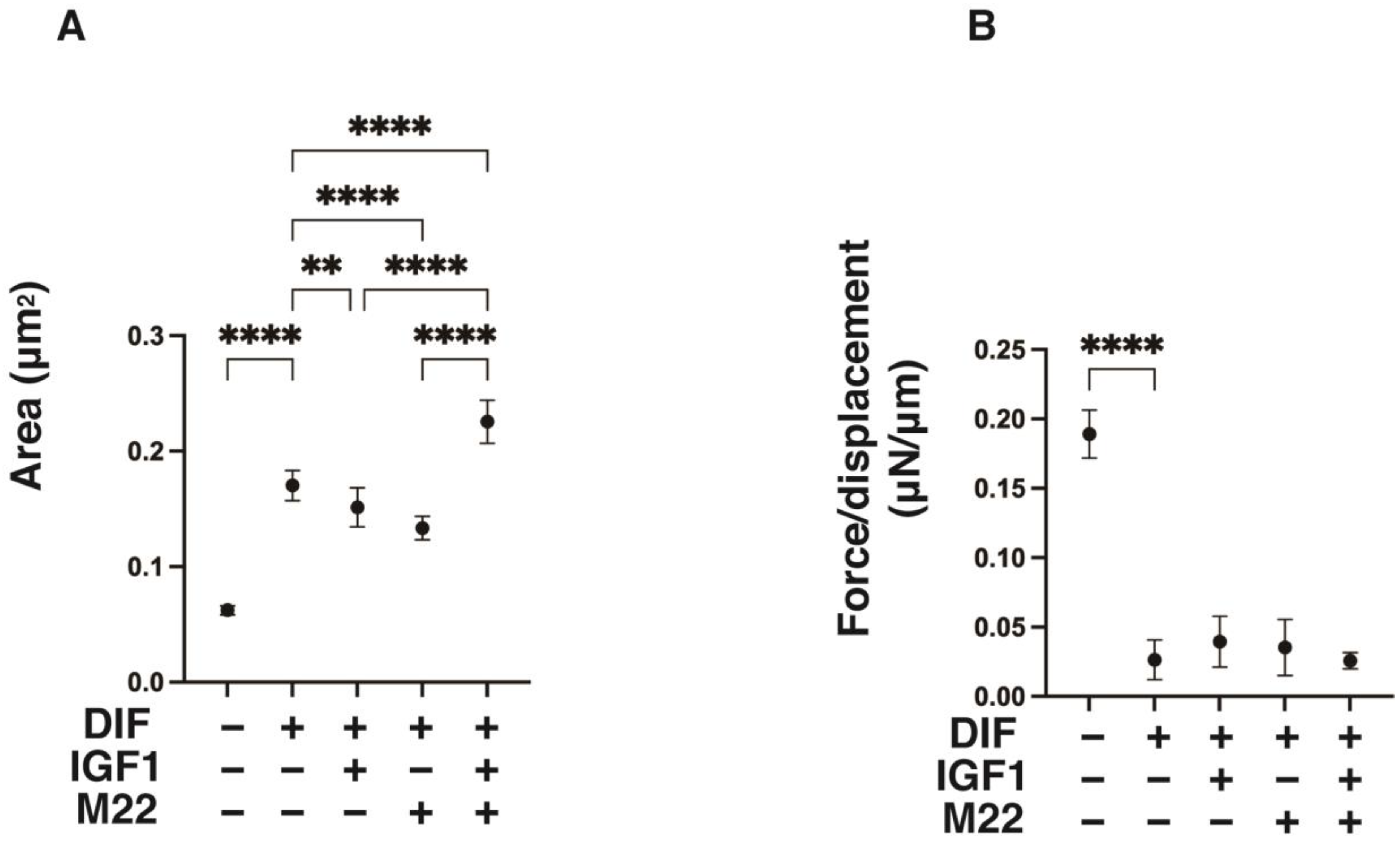
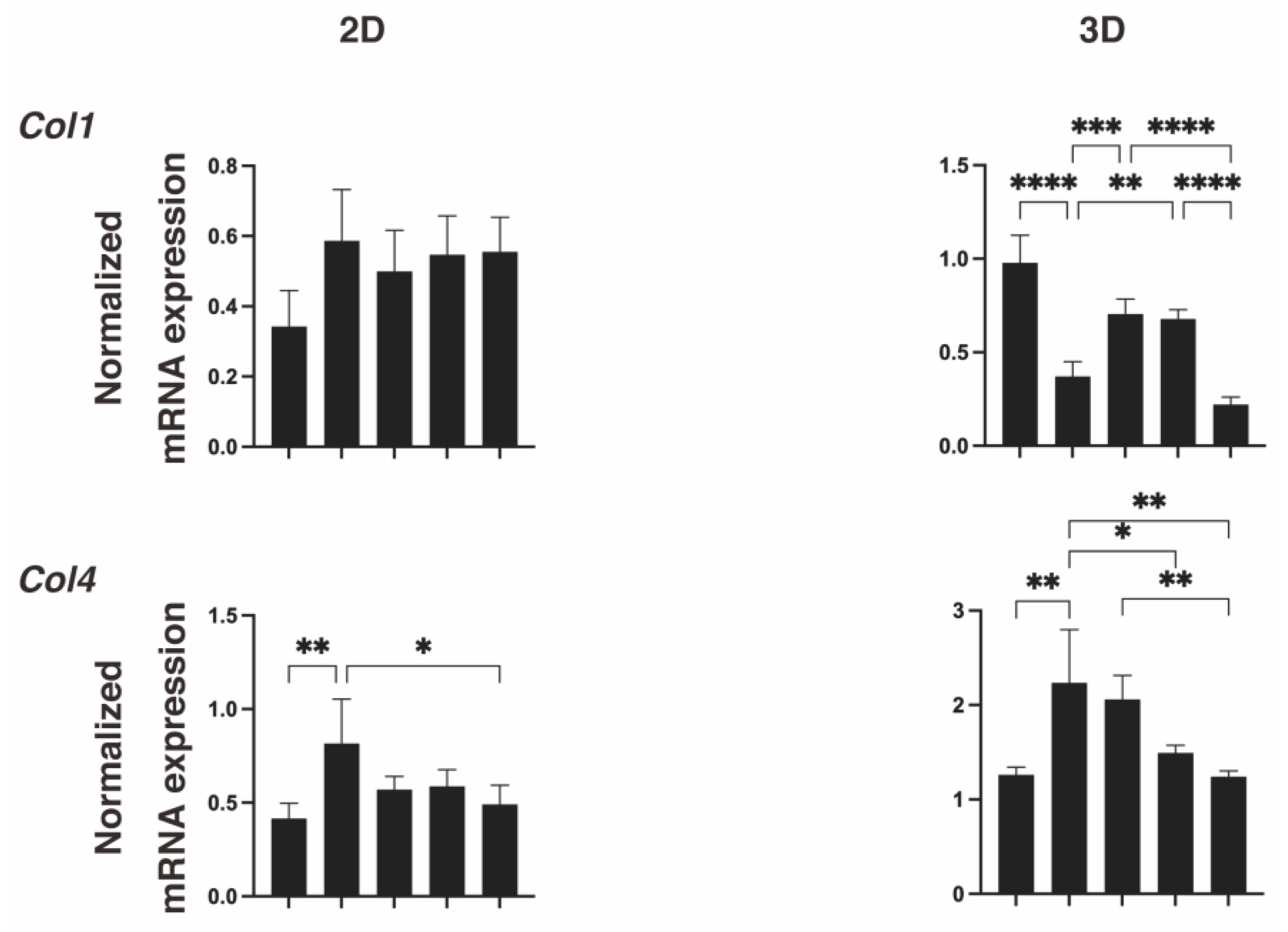
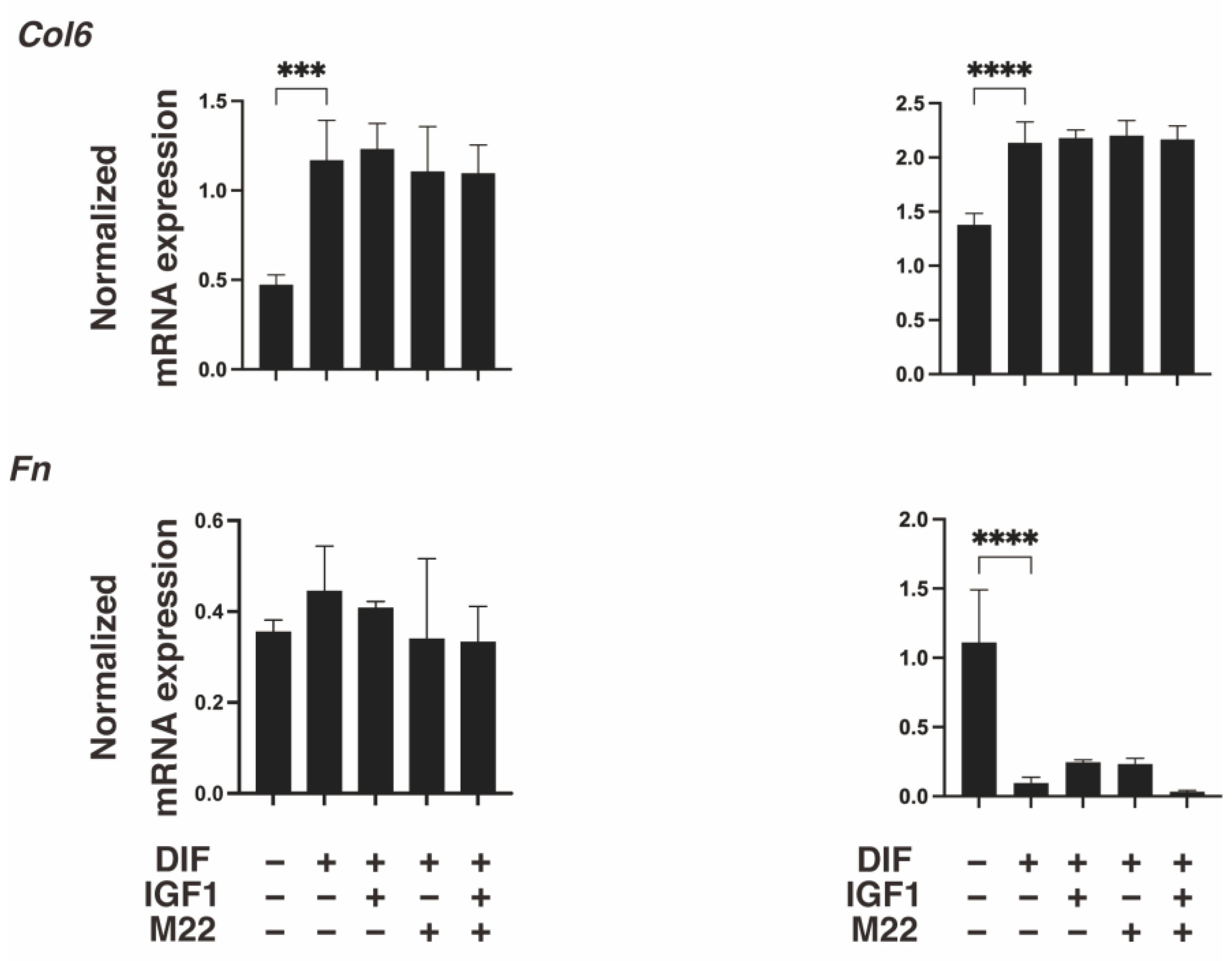
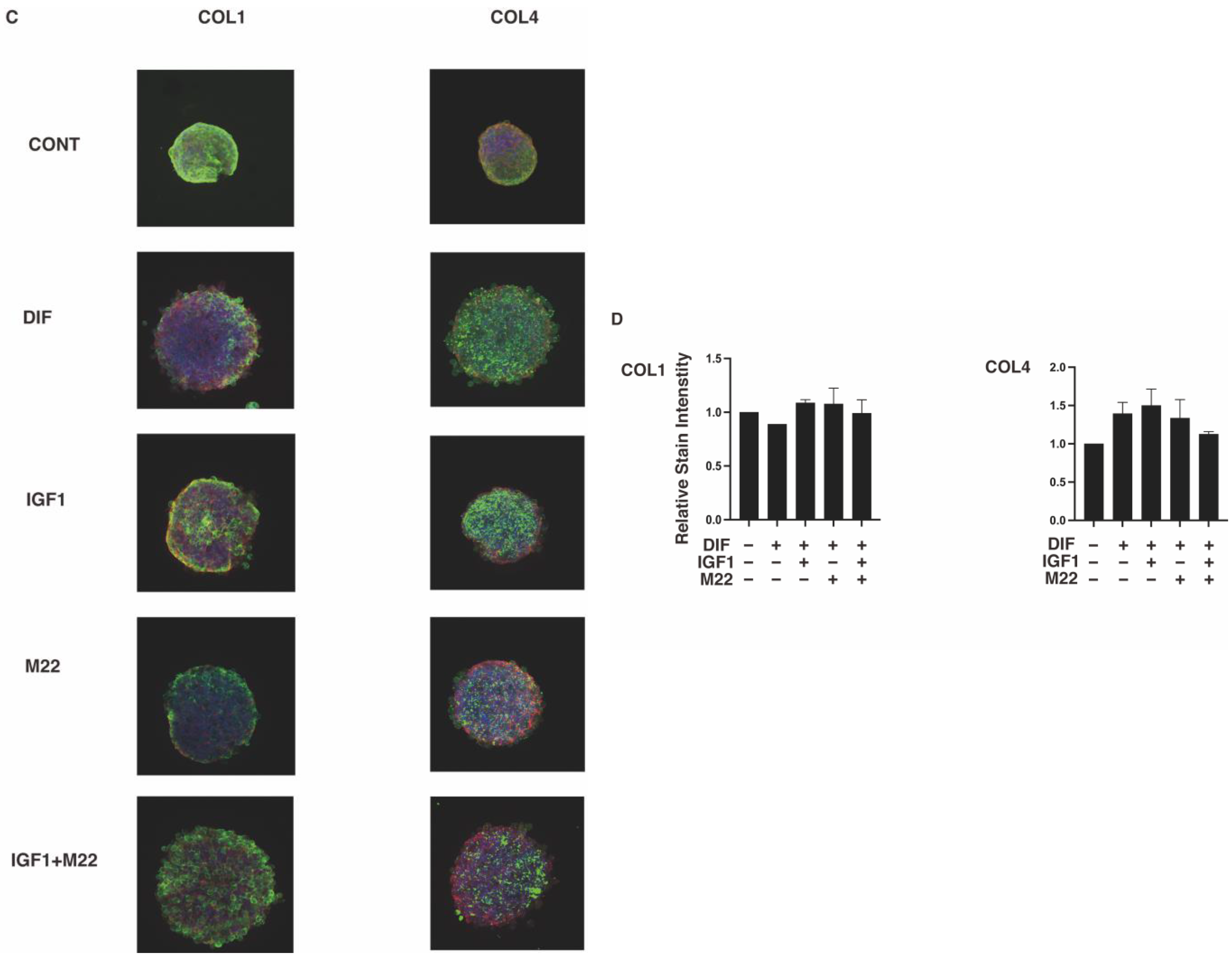
2. Results
3. Discussion
4. Materials and Methods
4.1. Measurement of Real-Time Cellular Metabolic Functions of 2D-Cultured 3T3-L1 Cells
4.2. Characterization of the 3D 3T3-L1 Spheroids
4.3. Other Analytical Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Smith, T.J.; Hegedüs, L. Graves′ Disease. New Engl. J. Med. 2016, 375, 1552–1565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahn, R.S. Graves′ ophthalmopathy. New Engl. J. Med. 2010, 362, 726–738. [Google Scholar] [CrossRef] [Green Version]
- Turcu, A.F.; Kumar, S.; Neumann, S.; Coenen, M.; Iyer, S.; Chiriboga, P.; Gershengorn, M.C.; Bahn, R.S. A small molecule antagonist inhibits thyrotropin receptor antibody-induced orbital fibroblast functions involved in the pathogenesis of Graves ophthalmopathy. J. Clin. Endocrinol. Metab. 2013, 98, 2153–2159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, T.J. TSH-receptor-expressing fibrocytes and thyroid-associated ophthalmopathy. Nat. Reviews. Endocrinol. 2015, 11, 171–181. [Google Scholar] [CrossRef] [Green Version]
- Sanders, J.; Evans, M.; Premawardhana, L.D.; Depraetere, H.; Jeffreys, J.; Richards, T.; Furmaniak, J.; Rees Smith, B. Human monoclonal thyroid stimulating autoantibody. Lancet 2003, 362, 126–128. [Google Scholar] [CrossRef]
- Kumar, S.; Schiefer, R.; Coenen, M.J.; Bahn, R.S. A stimulatory thyrotropin receptor antibody (M22) and thyrotropin increase interleukin-6 expression and secretion in Graves′ orbital preadipocyte fibroblasts. Thyroid Off. J. Am. Thyroid Assoc. 2010, 20, 59–65. [Google Scholar] [CrossRef] [Green Version]
- Delcourt, N.; Bockaert, J.; Marin, P. GPCR-jacking: From a new route in RTK signalling to a new concept in GPCR activation. Trends Pharmacol. Sci. 2007, 28, 602–607. [Google Scholar] [CrossRef]
- Pyne, N.J.; Pyne, S. Receptor tyrosine kinase-G-protein-coupled receptor signalling platforms: Out of the shadow? Trends Pharmacol. Sci. 2011, 32, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Santisteban, P.; Kohn, L.D.; Di Lauro, R. Thyroglobulin gene expression is regulated by insulin and insulin-like growth factor I, as well as thyrotropin, in FRTL-5 thyroid cells. J. Biol. Chem. 1987, 262, 4048–4052. [Google Scholar] [CrossRef]
- Tsui, S.; Naik, V.; Hoa, N.; Hwang, C.J.; Afifiyan, N.F.; Sinha Hikim, A.; Gianoukakis, A.G.; Douglas, R.S.; Smith, T.J. Evidence for an association between thyroid-stimulating hormone and insulin-like growth factor 1 receptors: A tale of two antigens implicated in Graves′ disease. J. Immunol. 2008, 181, 4397–4405. [Google Scholar] [CrossRef]
- Zhang, L.; Grennan-Jones, F.; Draman, M.S.; Lane, C.; Morris, D.; Dayan, C.M.; Tee, A.R.; Ludgate, M. Possible targets for nonimmunosuppressive therapy of Graves′ orbitopathy. J. Clin. Endocrinol. Metab. 2014, 99, E1183-90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rapoport, B.; McLachlan, S.M. The thyrotropin receptor in Graves′ disease. Thyroid Off. J. Am. Thyroid Assoc. 2007, 17, 911–922. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Hegedüs, L.; Douglas, R.S. Role of insulin-like growth factor-1 (IGF-1) pathway in the pathogenesis of Graves′ orbitopathy. Best Pract. Res. Clin. Endocrinol. Metab. 2012, 26, 291–302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, T.J.; Hoa, N. Immunoglobulins from patients with Graves′ disease induce hyaluronan synthesis in their orbital fibroblasts through the self-antigen, insulin-like growth factor-I receptor. J. Clin. Endocrinol. Metab. 2004, 89, 5076–5080. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.R.; Pichurin, P.; Nagayama, Y.; Latrofa, F.; Rapoport, B.; McLachlan, S.M. The thyrotropin receptor autoantigen in Graves disease is the culprit as well as the victim. J. Clin. Investig. 2003, 111, 1897–1904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanders, J.; Chirgadze, D.Y.; Sanders, P.; Baker, S.; Sullivan, A.; Bhardwaja, A.; Bolton, J.; Reeve, M.; Nakatake, N.; Evans, M.; et al. Crystal structure of the TSH receptor in complex with a thyroid-stimulating autoantibody. Thyroid Off. J. Am. Thyroid Assoc. 2007, 17, 395–410. [Google Scholar] [CrossRef] [PubMed]
- Chazenbalk, G.D.; Wang, Y.; Guo, J.; Hutchison, J.S.; Segal, D.; Jaume, J.C.; McLachlan, S.M.; Rapoport, B. A mouse monoclonal antibody to a thyrotropin receptor ectodomain variant provides insight into the exquisite antigenic conformational requirement, epitopes and in vivo concentration of human autoantibodies. J. Clin. Endocrinol. Metab. 1999, 84, 702–710. [Google Scholar] [PubMed] [Green Version]
- Chazenbalk, G.D.; Pichurin, P.; Chen, C.R.; Latrofa, F.; Johnstone, A.P.; McLachlan, S.M.; Rapoport, B. Thyroid-stimulating autoantibodies in Graves disease preferentially recognize the free A subunit, not the thyrotropin holoreceptor. J. Clin. Investig. 2002, 110, 209–217. [Google Scholar] [CrossRef]
- Sanders, P.; Young, S.; Sanders, J.; Kabelis, K.; Baker, S.; Sullivan, A.; Evans, M.; Clark, J.; Wilmot, J.; Hu, X.; et al. Crystal structure of the TSH receptor (TSHR) bound to a blocking-type TSHR autoantibody. J. Mol. Endocrinol. 2011, 46, 81–99. [Google Scholar] [CrossRef] [Green Version]
- Neumann, S.; Nir, E.A.; Eliseeva, E.; Huang, W.; Marugan, J.; Xiao, J.; Dulcey, A.E.; Gershengorn, M.C. A selective TSH receptor antagonist inhibits stimulation of thyroid function in female mice. Endocrinology 2014, 155, 310–314. [Google Scholar] [CrossRef]
- Huh, D.; Hamilton, G.A.; Ingber, D.E. From 3D cell culture to organs-on-chips. Trends Cell Biol. 2011, 21, 745–754. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Weaver, V.M.; Petersen, O.W.; Larabell, C.A.; Dedhar, S.; Briand, P.; Lupu, R.; Bissell, M.J. Reciprocal interactions between beta1-integrin and epidermal growth factor receptor in three-dimensional basement membrane breast cultures: A different perspective in epithelial biology. Proc. Natl. Acad. Sci. USA 1998, 95, 14821–14826. [Google Scholar] [CrossRef]
- Ohguro, H.; Ida, Y.; Hikage, F.; Umetsu, A.; Ichioka, H.; Watanabe, M.; Furuhashi, M. STAT3 Is the Master Regulator for the Forming of 3D Spheroids of 3T3-L1 Preadipocytes. Cells 2022, 11, 300. [Google Scholar] [CrossRef] [PubMed]
- Ida, Y.; Hikage, F.; Itoh, K.; Ida, H.; Ohguro, H. Prostaglandin F2α agonist-induced suppression of 3T3-L1 cell adipogenesis affects spatial formation of extra-cellular matrix. Sci Rep 2020, 10, 7958. [Google Scholar] [CrossRef] [PubMed]
- Ida, Y.; Hikage, F.; Ohguro, H. ROCK inhibitors enhance the production of large lipid-enriched 3D organoids of 3T3-L1 cells. Sci. Rep. 2021, 11, 5479. [Google Scholar] [CrossRef]
- Ida, Y.; Hikage, F.; Umetsu, A.; Ida, H.; Ohguro, H. Omidenepag, a non-prostanoid EP2 receptor agonist, induces enlargement of the 3D organoid of 3T3-L1 cells. Sci. Rep. 2020, 10, 16018. [Google Scholar] [CrossRef]
- Ida, Y.; Watanabe, M.; Ohguro, H.; Hikage, F. Simultaneous Use of ROCK Inhibitors and EP2 Agonists Induces Unexpected Effects on Adipogenesis and the Physical Properties of 3T3-L1 Preadipocytes. Int. J. Mol. Sci. 2021, 22, 4648. [Google Scholar] [CrossRef]
- Ida, Y.; Watanabe, M.; Umetsu, A.; Ohguro, H.; Hikage, F. Addition of EP2 agonists to an FP agonist additively and synergistically modulates adipogenesis and the physical properties of 3D 3T3-L1 sphenoids. Prostaglandins Leukot. Essent. Fat. Acids 2021, 171, 102315. [Google Scholar] [CrossRef] [PubMed]
- Hikage, F.; Atkins, S.; Kahana, A.; Smith, T.J.; Chun, T.H. HIF2A-LOX Pathway Promotes Fibrotic Tissue Remodeling in Thyroid-Associated Orbitopathy. Endocrinology 2019, 160, 20–35. [Google Scholar] [CrossRef] [Green Version]
- Ida, Y.; Ichioka, H.; Furuhashi, M.; Hikage, F.; Watanabe, M.; Umetsu, A.; Ohguro, H. Reactivities of a Prostanoid EP2 Agonist, Omidenepag, Are Useful for Distinguishing between 3D Spheroids of Human Orbital Fibroblasts without or with Graves′ Orbitopathy. Cells 2021, 10, 3196. [Google Scholar] [CrossRef]
- Itoh, K.; Hikage, F.; Ida, Y.; Ohguro, H. Prostaglandin F2α Agonists Negatively Modulate the Size of 3D Organoids from Primary Human Orbital Fibroblasts. Investig. Ophthalmol. Vis. Sci. 2020, 61, 13. [Google Scholar] [CrossRef] [PubMed]
- Ida, Y.; Umetsu, A.; Furuhashi, M.; Watanabe, M.; Tsugeno, Y.; Suzuki, S.; Hikage, F.; Ohguro, H. ROCK 1 and 2 affect the spatial architecture of 3D spheroids derived from human corneal stromal fibroblasts in different manners. Sci. Rep. 2022, 12, 7419. [Google Scholar] [CrossRef]
- Watanabe, M.; Ida, Y.; Ohguro, H.; Ota, C.; Hikage, F. Establishment of appropriate glaucoma models using dexamethasone or TGFβ2 treated three-dimension (3D) cultured human trabecular meshwork (HTM) cells. Sci. Rep. 2021, 11, 19369. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Ida, Y.; Ohguro, H.; Ota, C.; Hikage, F. Diverse effects of pan-ROCK and ROCK2 inhibitors on 2 D and 3D cultured human trabecular meshwork (HTM) cells treated with TGFβ2. Sci. Rep. 2021, 11, 15286. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Ida, Y.; Furuhashi, M.; Tsugeno, Y.; Ohguro, H.; Hikage, F. Screening of the Drug-Induced Effects of Prostaglandin EP2 and FP Agonists on 3D Cultures of Dexamethasone-Treated Human Trabecular Meshwork Cells. Biomedicines 2021, 9, 930. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Ida, Y.; Furuhashi, M.; Tsugeno, Y.; Hikage, F.; Ohguro, H. Pan-ROCK and ROCK2 Inhibitors Affect Dexamethasone-Treated 2D- and 3D-Cultured Human Trabecular Meshwork (HTM) Cells in Opposite Manners. Molecules 2021, 26, 6382. [Google Scholar] [CrossRef]
- Oouchi, Y.; Watanabe, M.; Ida, Y.; Ohguro, H.; Hikage, F. Rosiglitasone and ROCK Inhibitors Modulate Fibrogenetic Changes in TGF-β2 Treated Human Conjunctival Fibroblasts (HconF) in Different Manners. Int. J. Mol. Sci. 2021, 22, 7335. [Google Scholar] [CrossRef]
- Tsugeno, Y.; Furuhashi, M.; Sato, T.; Watanabe, M.; Umetsu, A.; Suzuki, S.; Ida, Y.; Hikage, F.; Ohguro, H. FGF-2 enhances fibrogenetic changes in TGF-β2 treated human conjunctival fibroblasts. Sci. Rep. 2022, 12, 16006. [Google Scholar] [CrossRef]
- Tsugeno, Y.; Sato, T.; Watanabe, M.; Higashide, M.; Furuhashi, M.; Umetsu, A.; Suzuki, S.; Ida, Y.; Hikage, F.; Ohguro, H. All Trans-Retinoic Acids Facilitate the Remodeling of 2D and 3D Cultured Human Conjunctival Fibroblasts. Bioengineering 2022, 9, 463. [Google Scholar] [CrossRef] [PubMed]
- Tsugeno, Y.; Sato, T.; Watanabe, M.; Furuhashi, M.; Umetsu, A.; Ida, Y.; Hikage, F.; Ohguro, H. Benzalkonium Chloride, Even at Low Concentrations, Deteriorates Intracellular Metabolic Capacity in Human Conjunctival Fibroblasts. Biomedicines 2022, 10, 2315. [Google Scholar] [CrossRef]
- Sanders, J.; Jeffreys, J.; Depraetere, H.; Evans, M.; Richards, T.; Kiddie, A.; Brereton, K.; Premawardhana, L.D.; Chirgadze, D.Y.; Núñez Miguel, R.; et al. Characteristics of a human monoclonal autoantibody to the thyrotropin receptor: Sequence structure and function. Thyroid Off. J. Am. Thyroid Assoc. 2004, 14, 560–570. [Google Scholar] [CrossRef]
- Kumar, S.; Nadeem, S.; Stan, M.N.; Coenen, M.; Bahn, R.S. A stimulatory TSH receptor antibody enhances adipogenesis via phosphoinositide 3-kinase activation in orbital preadipocytes from patients with Graves’ ophthalmopathy. J. Mol. Endocrinol. 2011, 46, 155–163. [Google Scholar] [CrossRef] [Green Version]
- Lu, M.; Lin, R.Y. TSH stimulates adipogenesis in mouse embryonic stem cells. J. Endocrinol. 2008, 196, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Valyasevi, R.W.; Erickson, D.Z.; Harteneck, D.A.; Dutton, C.M.; Heufelder, A.E.; Jyonouchi, S.C.; Bahn, R.S. Differentiation of human orbital preadipocyte fibroblasts induces expression of functional thyrotropin receptor. J. Clin. Endocrinol. Metab. 1999, 84, 2557–2562. [Google Scholar] [CrossRef]
- Crisp, M.; Starkey, K.J.; Lane, C.; Ham, J.; Ludgate, M. Adipogenesis in thyroid eye disease. Investig. Ophthalmol. Vis. Sci. 2000, 41, 3249–3255. [Google Scholar]
- Krieger, C.C.; Boutin, A.; Jang, D.; Morgan, S.J.; Banga, J.P.; Kahaly, G.J.; Klubo-Gwiezdzinska, J.; Neumann, S.; Gershengorn, M.C. Arrestin-β-1 Physically Scaffolds TSH and IGF1 Receptors to Enable Crosstalk. Endocrinology 2019, 160, 1468–1479. [Google Scholar] [CrossRef]
- Bech, K.; Larsen, J.H.; Hansen, J.M.; Nerup, J. Letter: Yersinia enterocolitica infection and thyroid disorders. Lancet 1974, 2, 951–952. [Google Scholar] [CrossRef]
- Lidman, K.; Eriksson, U.; Fagraeus, A.; Norberg, R. Letter: Antibodies against thyroid cells in Yersinia enterocolitica infection. Lancet 1974, 2, 1449. [Google Scholar] [CrossRef] [PubMed]
- Shenkman, L.; Bottone, E.J. Antibodies to Yersinia enterocolitica in thyroid disease. Ann. Intern. Med. 1976, 85, 735–739. [Google Scholar] [CrossRef]
- Hargreaves, C.E.; Grasso, M.; Hampe, C.S.; Stenkova, A.; Atkinson, S.; Joshua, G.W.; Wren, B.W.; Buckle, A.M.; Dunn-Walters, D.; Banga, J.P. Yersinia enterocolitica provides the link between thyroid-stimulating antibodies and their germline counterparts in Graves′ disease. J. Immunol. 2013, 190, 5373–5381. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Tian, L.M.; Han, Y.; Ma, H.Y.; Wang, L.C.; Guo, J.; Gao, L.; Zhao, J.J. Presence of thyrotropin receptor in hepatocytes: Not a case of illegitimate transcription. J. Cell Mol. Med. 2009, 13, 4636–4642. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, K.; Shimura, H.; Lin, L.; Endo, T.; Onaya, T. Differentiation of rat preadipocytes is accompanied by expression of thyrotropin receptors. Endocrinology 1996, 137, 3200–3205. [Google Scholar] [CrossRef] [Green Version]
- Sorisky, A.; Bell, A.; Gagnon, A. TSH receptor in adipose cells. Horm. Metab. Res. = Horm. Stoffwechs. = Horm. Metab. 2000, 32, 468–474. [Google Scholar] [CrossRef]
- Lu, S.; Guan, Q.; Liu, Y.; Wang, H.; Xu, W.; Li, X.; Fu, Y.; Gao, L.; Zhao, J.; Wang, X. Role of extrathyroidal TSHR expression in adipocyte differentiation and its association with obesity. Lipids. Health Dis. 2012, 11, 17. [Google Scholar] [CrossRef] [Green Version]
- Valyasevi, R.W.; Harteneck, D.A.; Dutton, C.M.; Bahn, R.S. Stimulation of adipogenesis, peroxisome proliferator-activated receptor-gamma (PPARgamma), and thyrotropin receptor by PPARgamma agonist in human orbital preadipocyte fibroblasts. J. Clin. Endocrinol. Metab. 2002, 87, 2352–2358. [Google Scholar] [PubMed]
- Zhang, L.; Baker, G.; Janus, D.; Paddon, C.A.; Fuhrer, D.; Ludgate, M. Biological effects of thyrotropin receptor activation on human orbital preadipocytes. Investig. Ophthalmol. Vis. Sci. 2006, 47, 5197–5203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kobe, B.; Deisenhofer, J. The leucine-rich repeat: A versatile binding motif. Trends Biochem. Sci. 1994, 19, 415–421. [Google Scholar] [CrossRef]
- Martin-Zanca, D.; Oskam, R.; Mitra, G.; Copeland, T.; Barbacid, M. Molecular and biochemical characterization of the human trk proto-oncogene. Mol. Cell. Biol. 1989, 9, 24–33. [Google Scholar]
- Tan, F.; Weerasinghe, D.K.; Skidgel, R.A.; Tamei, H.; Kaul, R.K.; Roninson, I.B.; Schilling, J.W.; Erdös, E.G. The deduced protein sequence of the human carboxypeptidase N high molecular weight subunit reveals the presence of leucine-rich tandem repeats. J. Biol. Chem. 1990, 265, 13–19. [Google Scholar] [CrossRef]
- Hashimoto, C.; Hudson, K.L.; Anderson, K.V. The Toll gene of Drosophila, required for dorsal-ventral embryonic polarity, appears to encode a transmembrane protein. Cell 1988, 52, 269–279. [Google Scholar] [CrossRef]
- Krusius, T.; Ruoslahti, E. Primary structure of an extracellular matrix proteoglycan core protein deduced from cloned cDNA. Proc. Natl. Acad. Sci. USA 1986, 83, 7683–7687. [Google Scholar] [CrossRef] [Green Version]
- Roth, N.S.; Lefkowitz, R.J.; Caron, M.G. Structure and function of the adrenergic receptor family. Adv. Exp. Med. Biol. 1991, 308, 223–238. [Google Scholar] [PubMed]
- Strader, C.D.; Fong, T.M.; Tota, M.R.; Underwood, D.; Dixon, R.A. Structure and function of G protein-coupled receptors. Annu. Rev. Biochem. 1994, 63, 101–132. [Google Scholar] [CrossRef] [PubMed]
- McFarland, K.C.; Sprengel, R.; Phillips, H.S.; Köhler, M.; Rosemblit, N.; Nikolics, K.; Segaloff, D.L.; Seeburg, P.H. Lutropin-choriogonadotropin receptor: An unusual member of the G protein-coupled receptor family. Science 1989, 245, 494–499. [Google Scholar] [CrossRef]
- Vassart, G.; Dumont, J.E. The thyrotropin receptor and the regulation of thyrocyte function and growth. Endocr. Rev. 1992, 13, 596–611. [Google Scholar] [PubMed]
- Sprengel, R.; Braun, T.; Nikolics, K.; Segaloff, D.L.; Seeburg, P.H. The testicular receptor for follicle stimulating hormone: Structure and functional expression of cloned cDNA. Mol. Endocrinol. 1990, 4, 525–530. [Google Scholar] [CrossRef] [Green Version]
- Braun, T.; Schofield, P.R.; Sprengel, R. Amino-terminal leucine-rich repeats in gonadotropin receptors determine hormone selectivity. EMBO J. 1991, 10, 1885–1890. [Google Scholar] [CrossRef]
- Ji, T.H.; Ryu, K.S.; Gilchrist, R.; Ji, I. Interaction, signal generation, signal divergence, and signal transduction of LH/CG and the receptor. Recent Prog. Horm. Res. 1997, 52, 431–453, discussion 454. [Google Scholar]
- Choi, J.S.; Choi, S.S.; Kim, E.S.; Seo, Y.K.; Seo, J.K.; Kim, E.K.; Suh, P.G.; Choi, J.H. Flightless-1, a novel transcriptional modulator of PPARγ through competing with RXRα. Cell. Signal. 2015, 27, 614–620. [Google Scholar] [CrossRef]
- Hikage, F.; Ichioka, H.; Watanabe, M.; Umetsu, A.; Ohguro, H.; Ida, Y. Addition of ROCK inhibitors to prostaglandin derivative (PG) synergistically affects adipogenesis of the 3D spheroids of human orbital fibroblasts (HOFs). Hum. Cell 2022, 35, 125–132. [Google Scholar] [CrossRef]
- Hikage, F.; Ichioka, H.; Watanabe, M.; Umetsu, A.; Ohguro, H.; Ida, Y. ROCK inhibitors modulate the physical properties and adipogenesis of 3D spheroids of human orbital fibroblasts in different manners. FASEB Bioadvances 2021, 3, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Ichioka, H.; Ida, Y.; Watanabe, M.; Ohguro, H.; Hikage, F. Prostaglandin F2α and EP2 agonists, and a ROCK inhibitor modulate the formation of 3D organoids of Grave′s orbitopathy related human orbital fibroblasts. Exp. Eye Res. 2021, 205, 108489. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Chang, H.C.; Bayeva, M.; Shapiro, J.S.; Ramos-Alonso, L.; Kouzu, H.; Jiang, X.; Liu, T.; Yar, S.; Sawicki, K.T.; et al. mRNA-binding protein tristetraprolin is essential for cardiac response to iron deficiency by regulating mitochondrial function. Proc. Natl. Acad. Sci. USA 2018, 115, e6291–e6300. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Ichise, N.; Kobayashi, T.; Fusagawa, H.; Yamazaki, H.; Kudo, T.; Tohse, N. Enhanced glucose metabolism through activation of HIF-1α covers the energy demand in a rat embryonic heart primordium after heartbeat initiation. Sci. Rep. 2022, 12, 74. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umetsu, A.; Sato, T.; Watanabe, M.; Ida, Y.; Furuhashi, M.; Tsugeno, Y.; Ohguro, H. Unexpected Crosslinking Effects of a Human Thyroid Stimulating Monoclonal Autoantibody, M22, with IGF1 on Adipogenesis in 3T3L-1 Cells. Int. J. Mol. Sci. 2023, 24, 1110. https://doi.org/10.3390/ijms24021110
Umetsu A, Sato T, Watanabe M, Ida Y, Furuhashi M, Tsugeno Y, Ohguro H. Unexpected Crosslinking Effects of a Human Thyroid Stimulating Monoclonal Autoantibody, M22, with IGF1 on Adipogenesis in 3T3L-1 Cells. International Journal of Molecular Sciences. 2023; 24(2):1110. https://doi.org/10.3390/ijms24021110
Chicago/Turabian StyleUmetsu, Araya, Tatsuya Sato, Megumi Watanabe, Yosuke Ida, Masato Furuhashi, Yuri Tsugeno, and Hiroshi Ohguro. 2023. "Unexpected Crosslinking Effects of a Human Thyroid Stimulating Monoclonal Autoantibody, M22, with IGF1 on Adipogenesis in 3T3L-1 Cells" International Journal of Molecular Sciences 24, no. 2: 1110. https://doi.org/10.3390/ijms24021110






