The Bright Side of Psychedelics: Latest Advances and Challenges in Neuropharmacology
Abstract
:1. Introduction
2. Tabernanthe Iboga
2.1. Ethnobotany
2.2. Central Nervous System Pathways
2.3. Structural and Computational Studies

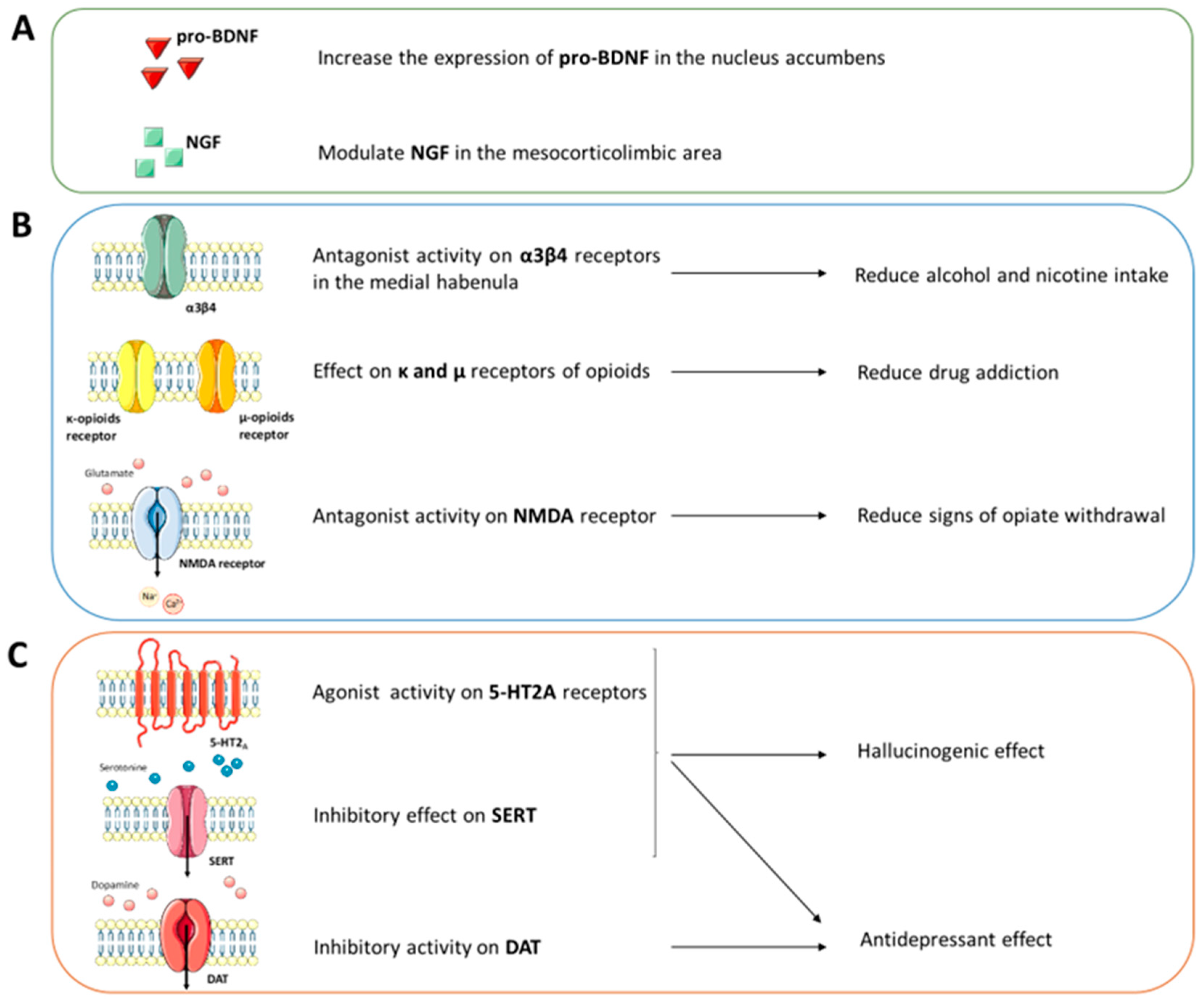
2.4. Therapeutic Hypotheses
3. Echinocactus williamsii
3.1. Ethnobotany
3.2. Central Nervous System Pathways
3.3. Structural and Computational Studies
3.4. Therapeutic Hypotheses
4. Psychotria Viridis
4.1. Ethnobotany
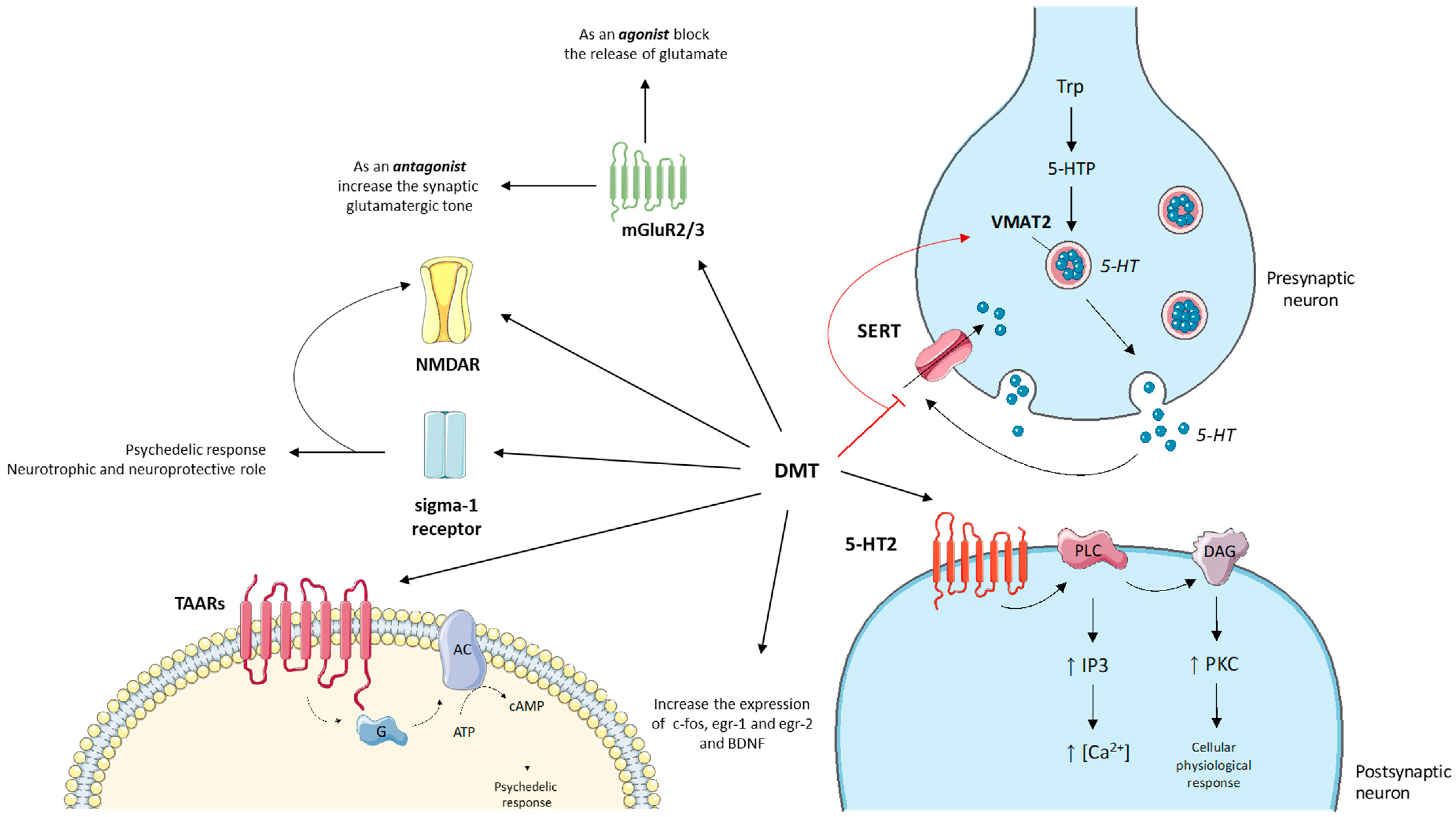
4.2. Central Nervous System Pathways
4.3. Structural and Computational Studies
4.4. Therapeutic Hypotheses
5. Psilocybe Cubensis
5.1. Ethnobotany
5.2. Central Nervous System Pathways
5.3. Structural and Computational Studies
5.4. Therapeutic Hypotheses
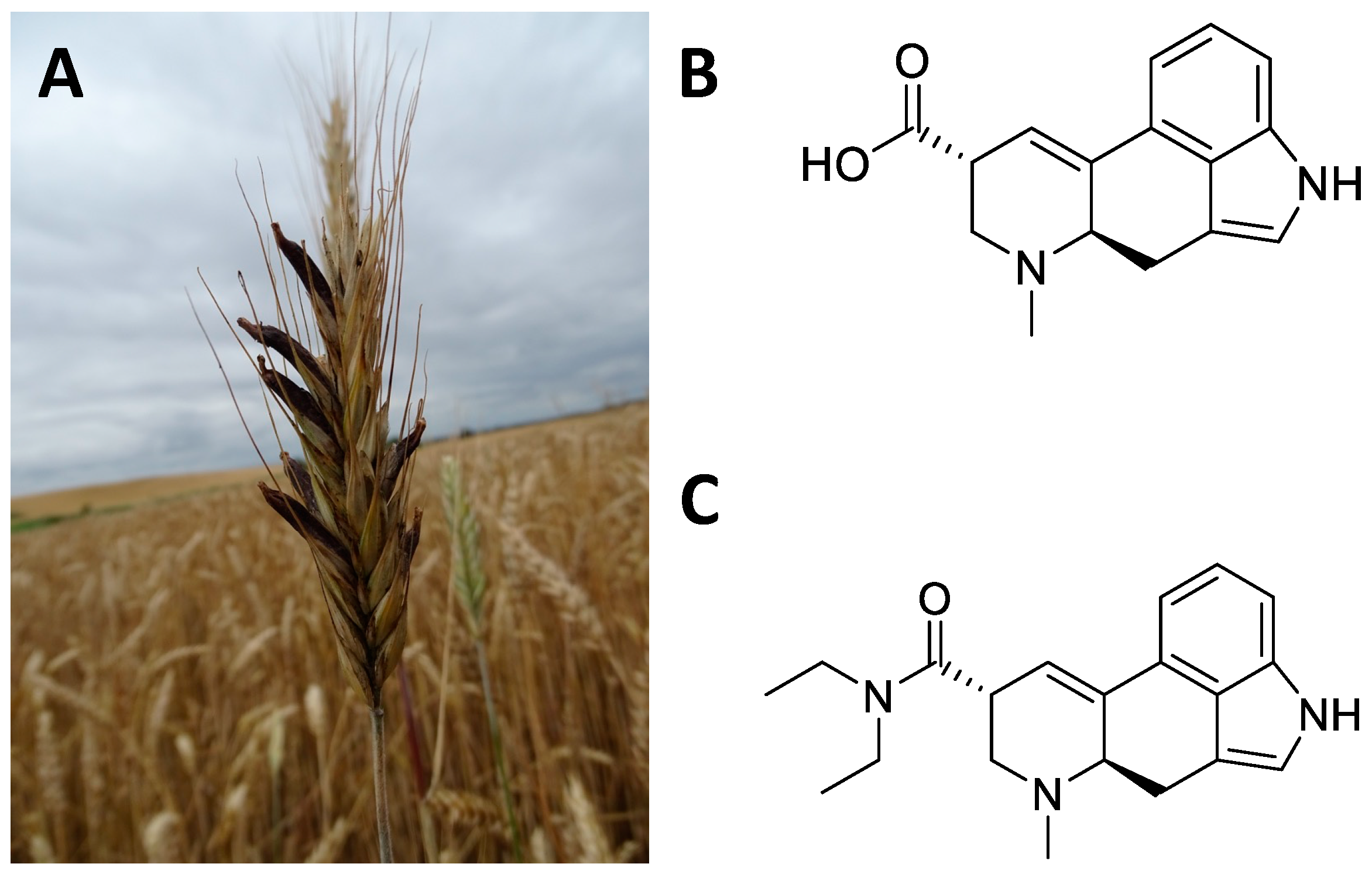
6. Claviceps Purpurea
6.1. Ethnobotany
6.2. Central Nervous System Pathways
6.3. Structural and Computational Studies
6.4. Therapeutic Hypotheses
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| ADHD | attention deficit hyperactivity disorder |
| ASD | autism spectrum disorders |
| AUC | area under the curve |
| AUD | alcohol use disorder |
| BDNF | brain-derived neurotrophic factor |
| BPD | bipolar disorder |
| CBD | cannabidiol |
| CNS | central nervous system |
| DMT | N,N-dimethyltryptamine |
| DSM-V | Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition |
| FDA | Food and Drug Administration |
| fMRI | functional magnetic resonance imaging |
| FRA | flexible docking mode |
| GDNF | glial cell line-derived neurotrophic factor |
| 5-HT | serotonin |
| IND | investigational new drug |
| iPSC | induced pluripotent stem cells |
| LSD | D-lysergic acid diethylamide |
| MADRS | Montgomery–Åsberg Depression Rating Scale |
| mGluR | metabotropic glutamate receptor |
| mTOR | mammalian target rapamycin receptor |
| NIH | National Institutes of Health |
| NGF | neural growth factor |
| NMDA | N-methyl-D-aspartate |
| NSSs | neurotransmitter sodium symporters |
| 6-OHDA | 6-hydroxydopamine |
| OUD | opioid use disorder |
| PAP | assisted psychotherapy |
| PD | Parkinson’s disease |
| PDB | protein data bank |
| PET | positron emission tomography |
| PG | prostaglandin |
| PTSD | post-traumatic stress disorder |
| RMSF | root-mean-square-fluctuation |
| RRA | rigid docking mode |
| SERT | serotonin transporter |
| STAI | state-trait anxiety inventory |
| SV2A | synaptic vesicle glycoprotein 2A |
| TAARs | trace amine associated receptors |
| TrkB | tropomyosin kinase B |
| VMAT2 | vesicular monoamine transporter 2 |
| VTA | ventral tegmental area |
References
- Lansky, E.S.; Lansky, S.; Paavilainen, H.M. Entheogenesis and Entheogenic Employment of Harmal. Tradit. Herb. Med. Mod. 2017, 20, 55–68. [Google Scholar] [CrossRef]
- Tangarife-Puerta, H.F.; Ceballos-Ceballos, L.A.; Rodriguez-Osorio, J.E. Shamanism, Enthogen and Contemporary Art. Cult. Drog. 2018, 25, 106–136. [Google Scholar] [CrossRef]
- Pothier, B. Potential identification of an entheogenic plant species on the Chi Silk Manuscript. Time Mind 2021, 14, 111–134. [Google Scholar] [CrossRef]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef] [PubMed]
- Berlowitz, I.; O’Shaughnessy, D.M.; Heinrich, M.; Wolf, U.; Maake, C.; Martin-Soelch, C. Teacher plants—Indigenous Peruvian-Amazonian dietary practices as a method for using psychoactives. J. Ethnopharmacol. 2022, 286, 114910. [Google Scholar] [CrossRef]
- Nemu, D. Getting high with the most high: Entheogens in the Old Testament. J. Psychedelic Stud. 2019, 3, 117–132. [Google Scholar] [CrossRef]
- Carvalhoa, I.C.D.; Steila, C.A.; Gonzaga, F.A. Learning from a more-than-human perspective. Plants as teachers J. Env. Educ 2021, 52, 205. [Google Scholar] [CrossRef]
- Sayin, H.U. The Consumption of Psychoactive Plants During Religious Rituals: The Roots of Common Symbols and Figures in Religions and Myths. Neuroquantology 2014, 12, 276–296. [Google Scholar] [CrossRef] [Green Version]
- Apud, I. Ayahuasca from Peru to Uruguay: Ritual Design and Redesign through a Distributed Cognition Approach. Anthr. Conscious 2015, 26, 1–27. [Google Scholar] [CrossRef]
- Prue, R.; Voss, R.W. Indigenous Healing Practice: Ayahuasca. Opening a Discussion. J. Pastor. Care Couns. 2014, 68, 1–13. [Google Scholar] [CrossRef]
- Crockford, S. Shamanisms and the Authenticity of Religious Experience. Pomegranate 2010, 12, 139–158. [Google Scholar] [CrossRef]
- Ona, G.; Berrada, A.; Bouso, J.C. Communalistic use of psychoactive plants as a bridge between traditional healing practices and Western medicine: A new path for the Global Mental Health movement. Transcult. Psychiatry 2022, 59, 638–651. [Google Scholar] [CrossRef]
- Majic, T.; Sauter, M.; Bermpohl, F.; Schmidt, T.T. Connected to the spirit of the frog: An Internet-based survey on Kambo, the secretion of the Amazonian Giant Maki Frog (Phyllomedusa bicolor): Motivations for use, settings and subjective experiences. J. Psychopharmacol. 2021, 35, 421–436. [Google Scholar] [CrossRef]
- Hartogsohn, I. Modalities of the psychedelic experience: Microclimates of set and setting in hallucinogen research and culture. Transcult. Psychiatry 2022, 59, 579–591. [Google Scholar] [CrossRef]
- Jones, M.T. The creativity of Crumb: Research on the effects of psychedelic drugs on the comic art of Robert Crumb. J. Psychoact. Drugs 2007, 39, 283–291. [Google Scholar] [CrossRef]
- ten Berge, J. Jekyll and Hyde revisited: Paradoxes in the appreciation of drug experiences and their effects on creativity. J. Psychoact. Drugs 2002, 34, 249–262. [Google Scholar] [CrossRef]
- Janiger, O.; Dobkin de Rios, M. LSD and creativity. J. Psychoact. Drugs 1989, 21, 129–134. [Google Scholar] [CrossRef]
- Winkelman, M.J. The Mechanisms of Psychedelic Visionary Experiences: Hypotheses from Evolutionary Psychology. Front. Neurosci. 2017, 11, 539. [Google Scholar] [CrossRef] [Green Version]
- Belouin, S.J.; Henningfield, J.E. Psychedelics: Where we are now, why we got here, what we must do. Neuropharmacology 2018, 142, 7–19. [Google Scholar] [CrossRef]
- Fischman, L.G. Knowing and being known: Psychedelic-assisted psychotherapy and the sense of authenticity. Front. Psychiatry 2022, 13, 933495. [Google Scholar] [CrossRef]
- Ross, S.; Agrawal, M.; Griffiths, R.R.; Grob, C.; Berger, A.; Henningfield, J.E. Psychedelic-assisted psychotherapy to treat psychiatric and existential distress in life-threatening medical illnesses and palliative care. Neuropharmacology 2022, 216, 109174. [Google Scholar] [CrossRef] [PubMed]
- King, F. Psychedelic-Assisted Psychotherapy: Great Promise and Great Challenges. Psychiat. Ann. 2022, 52, 352. [Google Scholar] [CrossRef]
- Cavarra, M.; Falzone, A.; Ramaekers, J.G.; Kuypers, K.P.C.; Mento, C. Psychedelic-Assisted Psychotherapy-A Systematic Review of Associated Psychological Interventions. Front. Psychol. 2022, 13, 2996. [Google Scholar] [CrossRef] [PubMed]
- Puspanathan, P.; Sekula, A.D.; Starke, J. Psychedelic-Assisted Psychotherapy: The Past, the Present, the Future. Aust. Nz. J. Psychiat. 2022, 56, 56–57. [Google Scholar]
- Bogenschutz, M.P.; Johnson, M.W. Classic hallucinogens in the treatment of addictions. Prog. Neuro-Psychoph. 2016, 64, 250–258. [Google Scholar] [CrossRef]
- Calleja-Conde, J.; Morales-García, J.A.; Echeverry-Alzate, V.; Bühler, K.M.; Giné, E.; López-Moreno, J.A. Classic psychedelics and alcohol use disorders: A systematic review of human and animal studies. Addict. Biol. 2022, 27, e13229. [Google Scholar] [CrossRef]
- Siegel, A.N.; Meshkat, S.; Benitah, K.; Lipsitz, O.; Gill, H.; Lui, L.M.W.; Teopiz, K.M.; McIntyre, R.S.; Rosenblat, J.D. Registered clinical studies investigating psychedelic drugs for psychiatric disorders. J. Psychiatr. Res. 2021, 139, 71–81. [Google Scholar] [CrossRef]
- Brown, T.K.; Alper, K. Treatment of opioid use disorder with ibogaine: Detoxification and drug use outcomes. Am. J. Drug Alcohol Abus. 2017, 44, 24–36. [Google Scholar] [CrossRef] [Green Version]
- Markopoulos, A.; Inserra, A.; De Gregorio, D.; Gobbi, G. Evaluating the Potential Use of Serotonergic Psychedelics in Autism Spectrum Disorder. Front. Pharmacol. 2022, 12, 3341. [Google Scholar] [CrossRef]
- Johnston, C.B.; Mangini, M.; Grob, C.; Anderson, B. The Safety and Efficacy of Psychedelic-Assisted Therapies for Older Adults: Knowns and Unknowns. Am. J. Geriatr. Psychiatry 2023, 31, 44–53. [Google Scholar] [CrossRef]
- Vann Jones, S.A.; O’Kelly, A. Psychedelics as a Treatment for Alzheimer’s Disease Dementia. Front. Synaptic Neurosci. 2020, 12, 34. [Google Scholar] [CrossRef]
- Carhart-Harris, R.; Giribaldi, B.; Watts, R.; Baker-Jones, M.; Murphy-Beiner, A.; Murphy, R.; Martell, J.; Blemings, A.; Erritzoe, D.; Nutt, D.J. Trial of Psilocybin versus Escitalopram for Depression. N. Engl. J. Med. 2021, 384, 1402–1411. [Google Scholar] [CrossRef]
- De Gregorio, D.; Popic, J.; Enns, J.P.; Inserra, A.; Skalecka, A.; Markopoulos, A.; Posa, L.; Lopez-Canul, M.; Qianzi, H.; Lafferty, C.K.; et al. Lysergic acid diethylamide (LSD) promotes social behavior through mTORC1 in the excitatory neurotransmission. Proc. Natl. Acad. Sci. USA 2021, 118, e2020705118. [Google Scholar] [CrossRef]
- Hall, W. Why was early therapeutic research on psychedelic drugs abandoned? Psychol. Med. 2021, 52, 26–31. [Google Scholar] [CrossRef]
- Lavaud, C.; Massiot, G. The Iboga Alkaloids. Prog. Chem. Org. Nat. Pract. 2017, 105, 89–136. [Google Scholar] [CrossRef]
- Kohek, M.; Ohren, M.; Hornby, P.; Alcázar-Córcoles, M.Á.; Bouso, J.C. The Ibogaine Experience: A Qualitative Study on the Acute Subjective Effects of Ibogaine. Anthr. Conscious 2020, 31, 91–119. [Google Scholar] [CrossRef]
- Paskulin, R.; Jamnik, P.; Danevcic, T.; Kozelj, G.; Krasovec, R.; Krstic-Milosevic, D.; Blagojevic, D.; Strukelj, B. Metabolic plasticity and the energy economizing effect of ibogaine, the principal alkaloid of Tabernanthe iboga. J. Ethnopharmacol. 2012, 143, 319–324. [Google Scholar] [CrossRef]
- Heink, A.; Katsikas, S.; Lange-Altman, T. Examination of the Phenomenology of the Ibogaine Treatment Experience: Role of Altered States of Consciousness and Psychedelic Experiences. J. Psychoact. Drugs 2017, 49, 201–208. [Google Scholar] [CrossRef]
- Litjens, R.P.W.; Brunt, T.M. How toxic is ibogaine? Clin. Toxicol. 2016, 54, 297–302. [Google Scholar] [CrossRef]
- Cameron, L.P.; Tombari, R.J.; Lu, J.; Pell, A.J.; Hurley, Z.Q.; Ehinger, Y.; Vargas, M.V.; McCarroll, M.N.; Taylor, J.C.; Myers-Turnbull, D.; et al. A non-hallucinogenic psychedelic analogue with therapeutic potential. Nature 2020, 589, 474–479. [Google Scholar] [CrossRef]
- Koenig, X.; Hilber, K. The Anti-Addiction Drug Ibogaine and the Heart: A Delicate Relation. Molecules 2015, 20, 2208–2228. [Google Scholar] [CrossRef] [PubMed]
- He, D.Y.; McGough, N.N.; Ravindranathan, A.; Jeanblanc, J.; Logrip, M.L.; Phamluong, K.; Janak, P.H.; Ron, D. Glial cell line-derived neurotrophic factor mediates the desirable actions of the anti-addiction drug ibogaine against alcohol consumption. J. Neurosci. Off. J. Soc. Neurosci. 2005, 25, 619–628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belgers, M.; Leenaars, M.; Homberg, J.R.; Ritskes-Hoitinga, M.; Schellekens, A.F.A.; Hooijmans, C.R. Ibogaine and addiction in the animal model, a systematic review and meta-analysis. Transl. Psychiatry 2016, 6, e826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marton, S.; González, B.; Rodríguez-Bottero, S.; Miquel, E.; Martínez-Palma, L.; Pazos, M.; Prieto, J.P.; Rodríguez, P.; Sames, D.; Seoane, G.; et al. Ibogaine Administration Modifies GDNF and BDNF Expression in Brain Regions Involved in Mesocorticolimbic and Nigral Dopaminergic Circuits. Front. Pharmacol. 2019, 10, 193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chatterjee, S.; Steensland, P.; Simms, J.A.; Holgate, J.; Coe, J.W.; Hurst, R.S.; Shaffer, C.L.; Lowe, J.; Rollema, H.; Bartlett, S.E. Partial Agonists of the α3β4* Neuronal Nicotinic Acetylcholine Receptor Reduce Ethanol Consumption and Seeking in Rats. Neuropsychopharmacology 2010, 36, 603–615. [Google Scholar] [CrossRef] [Green Version]
- Glick, S.D.; Maisonneuve, I.M.; Kitchen, B.A.; Fleck, M.W. Antagonism of alpha 3 beta 4 nicotinic receptors as a strategy to reduce opioid and stimulant self-administration. Eur. J. Pharm. 2002, 438, 99–105. [Google Scholar] [CrossRef]
- Daly, J.W. Nicotinic agonists, antagonists, and modulators from natural sources. Cell Mol. Neurobiol. 2005, 25, 513–552. [Google Scholar] [CrossRef]
- Mah, S.J.; Tang, Y.M.; Liauw, P.E.; Nagel, J.E.; Schneider, A.S. Ibogaine acts at the nicotinic acetylcholine receptor to inhibit catecholamine release. Brain Res. 1998, 797, 173–180. [Google Scholar] [CrossRef]
- Sershen, H.; Hashim, A.; Lajtha, A. Gender differences in kappa-opioid modulation of cocaine-induced behavior and NMDA-evoked dopamine release. Brain Res. 1998, 801, 67–71. [Google Scholar] [CrossRef]
- Henriques, G.M.; Anjos-Santos, A.; Rodrigues, I.R.S.; Nascimento-Rocha, V.; Reis, H.S.; Libarino-Santos, M.; Barros-Santos, T.; Yokoyama, T.S.; Bertagna, N.B.; Favoretto, C.A.; et al. Ibogaine Blocks Cue- and Drug-Induced Reinstatement of Conditioned Place Preference to Ethanol in Male Mice. Front. Pharmacol. 2021, 12, 2579. [Google Scholar] [CrossRef]
- Joshi, D.C.; Zhang, C.L.; Babujee, L.; Vevea, J.D.; August, B.K.; Sheng, Z.H.; Chapman, E.R.; Gomez, T.M.; Chiu, S.Y. Inappropriate Intrusion of an Axonal Mitochondrial Anchor into Dendrites Causes Neurodegeneration. Cell Rep. 2019, 29, 685–696.e5. [Google Scholar] [CrossRef]
- Pace, C.J.; Glick, S.D.; Maisonneuve, I.M.; He, L.W.; Jokiel, P.A.; Kuehne, M.E.; Fleck, M.W. Novel iboga alkaloid congeners block nicotinic receptors and reduce drug self-administration. Eur. J. Pharm. 2004, 492, 159–167. [Google Scholar] [CrossRef]
- Leal, M.B.; Michelin, K.; Souza, D.O.; Elisabetsky, E. Lbogaine attenuation of morphine withdrawal in mice: Role of glutamate N-methyl-D-aspartate receptors. Prog. Neuro-Psychoph. 2003, 27, 781–785. [Google Scholar] [CrossRef]
- Helsley, S. Behavioral and Biochemical Evidence for a Nonessential 5-HT2A Component of the Ibogaine-Induced Discriminative Stimulus. Pharmacol. Biochem. Behav. 1998, 59, 419–425. [Google Scholar] [CrossRef]
- Wei, D.; Maisonneuve, I.M.; Kuehne, M.E.; Glick, S.D. Acute iboga alkaloid effects on extracellular serotonin (5-HT) levels in nucleus accumbens and striatum in rats. Brain Res. 1998, 800, 260–268. [Google Scholar] [CrossRef]
- Chan, M.C.; Selvam, B.; Young, H.J.; Procko, E.; Shukla, D. The substrate import mechanism of the human serotonin transporter. Biophys. J. 2022, 121, 715–730. [Google Scholar] [CrossRef]
- Coleman, J.A.; Yang, D.; Zhao, Z.; Wen, P.-C.; Yoshioka, C.; Tajkhorshid, E.; Gouaux, E. Serotonin transporter–ibogaine complexes illuminate mechanisms of inhibition and transport. Nature 2019, 569, 141–145. [Google Scholar] [CrossRef]
- Möller, I.R.; Slivacka, M.; Nielsen, A.K.; Rasmussen, S.G.F.; Gether, U.; Loland, C.J.; Rand, K.D. Conformational dynamics of the human serotonin transporter during substrate and drug binding. Nat. Commun. 2019, 10, 1687. [Google Scholar] [CrossRef] [Green Version]
- Sutton, C.; Williams, E.Q.; Homsi, H.; Beerepoot, P.; Nazari, R.; Han, D.; Ramsey, A.J.; Mash, D.C.; Olson, D.E.; Blough, B.; et al. Structure-Activity Relationships of Dopamine Transporter Pharmacological Chaperones. Front. Cell. Neurosci. 2022, 16, 832536. [Google Scholar] [CrossRef]
- Glue, P.; Lockhart, M.; Lam, F.; Hung, N.; Hung, C.T.; Friedhoff, L. Ascending-Dose Study of Noribogaine in Healthy Volunteers: Pharmacokinetics, Pharmacodynamics, Safety, and Tolerability. J. Clin. Pharm. 2015, 55, 189–194. [Google Scholar] [CrossRef]
- Torres, G.E.; Gainetdinov, R.R.; Caron, M.G. Plasma membrane monoamine transporters: Structure, regulation and function. Nat. Rev. Neurosci. 2003, 4, 13–25. [Google Scholar] [CrossRef]
- Soares, C.M.; Hellsberg, E.; Ecker, G.F.; Stary-Weinzinger, A.; Forrest, L.R. A structural model of the human serotonin transporter in an outward-occluded state. PLoS ONE 2019, 14, e0217377. [Google Scholar] [CrossRef] [Green Version]
- Jacobs, M.T.; Zhang, Y.-W.; Campbell, S.D.; Rudnick, G. Ibogaine, a Noncompetitive Inhibitor of Serotonin Transport, Acts by Stabilizing the Cytoplasm-facing State of the Transporter. J. Biol. Chem. 2007, 282, 29441–29447. [Google Scholar] [CrossRef] [Green Version]
- Coleman, J.A.; Navratna, V.; Antermite, D.; Yang, D.; Bull, J.A.; Gouaux, E. Chemical and structural investigation of the paroxetine-human serotonin transporter complex. eLife 2020, 9, e56427. [Google Scholar] [CrossRef]
- Berman, H.; Henrick, K.; Nakamura, H. Announcing the worldwide Protein Data Bank. Nat. Struct. Mol. Biol. 2003, 10, 980. [Google Scholar] [CrossRef]
- Blat, Y. Non-Competitive Inhibition by Active Site Binders. Chem. Biol. Drug Des. 2010, 75, 535–540. [Google Scholar] [CrossRef]
- Coleman, J.A.; Green, E.M.; Gouaux, E. X-ray structures and mechanism of the human serotonin transporter. Nature 2016, 532, 334–339. [Google Scholar] [CrossRef] [Green Version]
- Chen, F.; Larsen, M.B.; Neubauer, H.A.; Sanchez, C.; Plenge, P.; Wiborg, O. Characterization of an allosteric citalopram-binding site at the serotonin transporter. J. Neurochem. 2005, 92, 21–28. [Google Scholar] [CrossRef]
- van Veen, H.W.; Navratna, V.; Tosh, D.K.; Jacobson, K.A.; Gouaux, E. Thermostabilization and purification of the human dopamine transporter (hDAT) in an inhibitor and allosteric ligand bound conformation. PLoS ONE 2018, 13, e0200085. [Google Scholar] [CrossRef] [Green Version]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera?A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [Green Version]
- Mash, D.C.; Kovera, C.A.; Pablo, J.; Tyndale, R.; Ervin, F.R.; Kamlet, J.D.; Hearn, W.L. Ibogaine in the treatment of heroin withdrawal. Alkaloids. Chem. Biol. 2001, 56, 155–171. [Google Scholar] [CrossRef]
- Mash, D.C.; Duque, L.; Page, B.; Allen-Ferdinand, K. Ibogaine Detoxification Transitions Opioid and Cocaine Abusers Between Dependence and Abstinence: Clinical Observations and Treatment Outcomes. Front. Pharmacol. 2018, 9, 529. [Google Scholar] [CrossRef] [Green Version]
- Szumlinski, K.K.; Haskew, R.E.; Balogun, M.Y.; Maisonneuve, I.M.; Glick, S.D. Iboga compounds reverse the behavioural disinhibiting and corticosterone effects of acute methamphetamine: Implications for their antiaddictive properties. Pharmacol. Biochem. Behav. 2001, 69, 485–491. [Google Scholar] [CrossRef]
- Schenberg, E.E.; Comis, M.A.D.; Chaves, B.R.; da Silveira, D.X. Treating drug dependence with the aid of ibogaine: A retrospective study. J. Psychopharmacol. 2014, 28, 993–1000. [Google Scholar] [CrossRef]
- Noller, G.E.; Frampton, C.M.; Yazar-Klosinski, B. Ibogaine treatment outcomes for opioid dependence from a twelve-month follow-up observational study. Am. J. Drug Alcohol Abus. 2017, 44, 37–46. [Google Scholar] [CrossRef] [Green Version]
- Malcolm, B.J.; Polanco, M.; Barsuglia, J.P. Changes in Withdrawal and Craving Scores in Participants Undergoing Opioid Detoxification Utilizing Ibogaine. J. Psychoact. Drugs 2018, 50, 256–265. [Google Scholar] [CrossRef]
- Wilson, C.; Millar, T.; Matieschyn, Z. Novel treatment of opioid use disorder using ibogaine and iboga in two adults. J. Psychedelic Stud. 2021, 4, 149–155. [Google Scholar] [CrossRef]
- Fernandes-Nascimento, M.H.; Viana-Ferreira, K.; Chaves, B.D.R.; Negrao, A.B.; Wang, Y.P. Ibogaine microdosing in a patient with bipolar depression: A case report. Braz. J. Psychiatry 2022, 44, 462–463. [Google Scholar] [CrossRef]
- Lea, T.; Almada, N.; Jungaberle, H.; Schecke, H.; Klein, M. Microdosing psychedelics: Motivations, subjective effects and harm reduction. Int. J. Drug Policy 2020, 75, 102600. [Google Scholar] [CrossRef]
- Polito, V.; Stevenson, R.J. A systematic study of microdosing psychedelics. PLoS ONE 2019, 14, e0211023. [Google Scholar] [CrossRef] [Green Version]
- Rodriguez, P.; Urbanavicius, J.; Prieto, J.P.; Fabius, S.; Reyes, A.L.; Havel, V.; Sames, D.; Scorza, C.; Carrera, I. A Single Administration of the Atypical Psychedelic Ibogaine or Its Metabolite Noribogaine Induces an Antidepressant-Like Effect in Rats. ACS Chem. Neurosci. 2020, 11, 1661–1672. [Google Scholar] [CrossRef]
- Nuzhyna, N.; Baglay, K.; Golubenko, A.; Lushchak, O. Anatomically distinct representatives of Cactaceae Juss. family have different response to acute heat shock stress. Flora 2018, 242, 137–145. [Google Scholar] [CrossRef]
- Rodriguez, S.M.M.; Rosas, H.G.; de los Santos, G.G.; Garcia-Moya, E.; Espinosa-Hernandez, V.; Torres, T.C.; Robledo-Paz, A. Viability and germination of seeds of four endangered species of cacti. Caldasia 2022, 44, 209–220. [Google Scholar] [CrossRef]
- Hofer, A. (2816) Proposal to conserve the name Echinocactus williamsii Lem. ex Salm-Dyck (Lophophora williamsii) against E. williamsianus Lem. (Cactaceae). Taxon 2021, 70, 676. [Google Scholar] [CrossRef]
- Cassels, B.K.; Saez-Briones, P. Dark Classics in Chemical Neuroscience: Mescaline. ACS Chem. Neurosci. 2018, 9, 2448–2458. [Google Scholar] [CrossRef]
- Carod-Artal, F.J. Hallucinogenic drugs in pre-Columbian Mesoamerican cultures. Neurologia 2015, 30, 42–49. [Google Scholar] [CrossRef]
- Carod-Artal, F.J.; Vazquez-Cabrera, C.B. Mescaline and the San Pedro cactus ritual: Archaeological and ethnographic evidence in northern Peru. Rev. Neurol. 2006, 42, 489–498. [Google Scholar] [CrossRef]
- Castro-Klaren, S. Chavin: How Do We Understand Thee? An Inquiry into the Jaguar and the San Pedro Cactus. Rev. Estud. Hisp. 2021, 55, 515–535. [Google Scholar] [CrossRef]
- Diaz, J.L. Sacred plants and visionary consciousness. Phenomenol. Cogn. Sci. 2010, 9, 159–170. [Google Scholar] [CrossRef]
- Vamvakopoulou, I.A.; Narine, K.A.D.; Campbell, I.; Dyck, J.R.B.; Nutt, D.J. Mescaline: The forgotten psychedelic. Neuropharmacology 2023, 222, 109294. [Google Scholar] [CrossRef]
- Dinis-Oliveira, R.J.; Pereira, C.L.; da Silva, D.D. Pharmacokinetic and Pharmacodynamic Aspects of Peyote and Mescaline: Clinical and Forensic Repercussions. Curr. Mol. Pharm. 2019, 12, 184–194. [Google Scholar] [CrossRef]
- Dell’Erba, S.; Brown, D.J.; Proulx, M.J. Synesthetic hallucinations induced by psychedelic drugs in a congenitally blind man. Conscious Cogn. 2018, 60, 127–132. [Google Scholar] [CrossRef]
- Kolaczynska, K.E.; Luethi, D.; Trachsel, D.; Hoener, M.C.; Liechti, M.E. Receptor Interaction Profiles of 4-Alkoxy-3,5-Dimethoxy-Phenethylamines (Mescaline Derivatives) and Related Amphetamines. Front. Pharmacol. 2022, 12, 794254. [Google Scholar] [CrossRef]
- Cumming, P.; Scheidegger, M.; Dornbierer, D.; Palner, M.; Quednow, B.B.; Martin-Soelch, C. Molecular and Functional Imaging Studies of Psychedelic Drug Action in Animals and Humans. Molecules 2021, 26, 2451. [Google Scholar] [CrossRef]
- Lopez-Gimenez, J.F.; Gonzalez-Maeso, J. Hallucinogens and Serotonin 5-HT2A Receptor-Mediated Signaling Pathways. Behav. Neurobiol. Psychedelic Drugs 2018, 36, 45–73. [Google Scholar] [CrossRef]
- Battal, D.; Barnes, A.J.; Castaneto, M.S.; Martin, T.M.; Klette, K.L.; Huestis, M.A. Urine Mescaline Screening With a Biochip Array Immunoassay and Quantification by Gas Chromatography-Mass Spectrometry. Drug Monit. 2015, 37, 805–811. [Google Scholar] [CrossRef]
- Halberstadt, A.L.; Chatha, M.; Chapman, S.J.; Brandt, S.D. Comparison of the behavioral effects of mescaline analogs using the head twitch response in mice. J. Psychopharmacol. 2019, 33, 406–414. [Google Scholar] [CrossRef]
- Thomann, J.; Ley, L.; Klaiber, A.; Liechti, M.E.; Duthaler, U. Development and validation of an LC-MS/MS method for the quantification of mescaline and major metabolites in human plasma. J. Pharm. Biomed. Anal. 2022, 220, 114980. [Google Scholar] [CrossRef]
- Brogaard, B.; Gatzia, D.E. Psilocybin, Lysergic Acid Diethylamide, Mescaline, and Drug-Induced Synesthesia. In Neuropathology of Drug Addictions and Substance Misuse; Academic Press: Cambridge, MA, USA, 2016; pp. 890–905. [Google Scholar] [CrossRef]
- Papaseit, E.; Olesti, E.; Pérez-Mañá, C.; Torrens, M.; Grifell, M.; Ventura, M.; Pozo, O.; de Sousa Fernandes Perna, E.B.; Ramaekers, J.G.; De La Torre, R.; et al. Acute Effects of 2C-E in Humans: An Observational Study. Front. Pharmacol. 2020, 11, 233. [Google Scholar] [CrossRef] [Green Version]
- McLean, T.H.; Chambers, J.J.; Parrish, J.C.; Braden, M.R.; Marona-Lewicka, D.; Kurrasch-Orbaugh, D.; Nichols, D.E. C-(4,5,6-Trimethoxyindan-1-yl)methanamine: A Mescaline Analogue Designed Using a Homology Model of the 5-HT2A Receptor. J. Med. Chem. 2006, 49, 4269–4274. [Google Scholar] [CrossRef]
- Yamamoto, T.; Ueki, S. Behavioral-Effects of 2,5-Dimethoxy-4-Methylamphetamine (Dom) in Rats and Mice. Eur. J. Pharm. 1975, 32, 156–162. [Google Scholar] [CrossRef]
- Halberstadt, A.L.; Geyer, M.A. Effect of Hallucinogens on Unconditioned Behavior. Curr. Top Behav. Neuro 2018, 36, 159–199. [Google Scholar] [CrossRef]
- Halberstadt, A.L.; Powell, S.B.; Geyer, M.A. Role of the 5-HT2A receptor in the locomotor hyperactivity produced by phenylalkylamine hallucinogens in mice. Neuropharmacology 2013, 70, 218–227. [Google Scholar] [CrossRef] [Green Version]
- Geyer, M.A.; Petersen, L.R.; Rose, G.J.; Horwitt, D.D.; Light, R.K.; Adams, L.M.; Zook, J.A.; Hawkins, R.L.; Mandell, A.J. The effects of lysergic acid diethylamide and mescaline-derived hallucinogens on sensory-integrative function: Tactile startle. J. Pharmacol. Exp. Ther. 1978, 207, 837–847. [Google Scholar]
- Agin-Liebes, G.; Haas, T.F.; Lancelotta, R.; Uthaug, M.V.; Ramaekers, J.G.; Davis, A.K. Naturalistic Use of Mescaline Is Associated with Self-Reported Psychiatric Improvements and Enduring Positive Life Changes. ACS Pharm. Transl. 2021, 4, 543–552. [Google Scholar] [CrossRef]
- Uthaug, M.V.; Davis, A.K.; Haas, T.F.; Davis, D.; Dolan, S.B.; Lancelotta, R.; Timmermann, C.; Ramaekers, J.G. The epidemiology of mescaline use: Pattern of use, motivations for consumption, and perceived consequences, benefits, and acute and enduring subjective effects. J. Psychopharmacol. 2022, 36, 309–320. [Google Scholar] [CrossRef]
- Trevizan, R.; Ludtke, R.; Cardoso, J.C.F.; Oliveira, P.E.; Streher, N.S. Is distyly in subtropical Psychotria brachyceras (Rubiaceae) similar to the general trends observed for the genus? Acta Bot. Bras. 2021, 35, 627–637. [Google Scholar] [CrossRef]
- Lachenaud, O. Diversity and endemism of Psychotria in West and Central Africa. Opera Bot Belg 2019, 17, 71–84. [Google Scholar]
- Lachenaud, O. Revision of the genus Psychotria (Rubiaceae) in West and Central Africa General context of the study. Opera Bot Belg 2019, 17, 15. [Google Scholar]
- Lachenaud, O. Morphological characteristics of the genus. Opera Bot Belg 2019, 17, 33–59. [Google Scholar]
- Lachenaud, O. Taxonomic revision of the genus. Opera Bot Belg 2019, 17, 95–399. [Google Scholar]
- Lachenaud, O. Ecology. Opera Bot Belg 2019, 17, 61–70. [Google Scholar]
- Lachenaud, O. Revision du Genre Psychotria (Rubiaceae) en Afrique Occidentale et Centrale, Tomes 1&2. Opera Bot Belg 2019, 17, 1–909. [Google Scholar]
- Lachenaud, O. Local names and uses. Opera Bot Belg 2019, 17, 87–93. [Google Scholar]
- Soares, D.B.S.; Duarte, L.P.; Cavalcanti, A.D.; Silva, F.C.; Braga, A.D.; Lopes, M.T.P.; Takahashi, J.A.; Vieira, S.A. Psychotria viridis: Chemical constituents from leaves and biological properties. Acad. Bras. Cienc. 2017, 89, 927–938. [Google Scholar] [CrossRef] [Green Version]
- Barker, S.A. N, N-Dimethyltryptamine (DMT), an Endogenous Hallucinogen: Past, Present, and Future Research to Determine Its Role and Function. Front. Neurosci. 2018, 12, 536. [Google Scholar] [CrossRef] [Green Version]
- Pic-Taylor, A.; da Motta, L.G.; de Morais, J.A.; Melo, W.; Santos, A.D.A.; Campos, L.A.; Mortari, M.R.; von Zuben, M.V.; Caldas, E.D. Behavioural and neurotoxic effects of ayahuasca infusion (Banisteriopsis caapi and Psychotria viridis) in female Wistar rat. Behav. Process. 2015, 118, 102–110. [Google Scholar] [CrossRef]
- James, E.; Keppler, J.; Robertshaw, T.L.; Sessa, B. N,N-dimethyltryptamine and Amazonian ayahuasca plant medicine. Hum. Psychopharmacol. Clin. Exp. 2022, 37, e2835. [Google Scholar] [CrossRef]
- Serra, Y.A.; Barros-Santos, T.; Anjos-Santos, A.; Kisaki, N.D.; Jovita-Farias, C.; Leite, J.P.C.; Santana, M.C.E.; Coimbra, J.P.S.A.; de Jesus, N.M.S.; Sulima, A.; et al. Role of 5-HT2A receptors in the effects of ayahuasca on ethanol self-administration using a two-bottle choice paradigm in male mice. Psychopharmacology 2022, 239, 1679–1687. [Google Scholar] [CrossRef]
- Gerwe, H.; He, F.; Pottie, E.; Stove, C.; Decker, M. Enlightening the “Spirit Molecule”: Photomodulation of the 5-HT2A Receptor by a Light-Controllable N,N-Dimethyltryptamine Derivative. Angew. Chem. Int. Ed. 2022, 61, e202203034. [Google Scholar] [CrossRef]
- Rossi, G.N.; Guerra, L.T.L.; Baker, G.B.; Dursun, S.M.; Saiz, J.C.B.; Hallak, J.E.C.; dos Santos, R.G. Molecular Pathways of the Therapeutic Effects of Ayahuasca, a Botanical Psychedelic and Potential Rapid-Acting Antidepressant. Biomolecules 2022, 12, 1618. [Google Scholar] [CrossRef]
- Brito-da-Costa, A.M.; Dias-da-Silva, D.; Gomes, N.G.M.; Dinis-Oliveira, R.J.; Madureira-Carvalho, A. Toxicokinetics and Toxicodynamics of Ayahuasca Alkaloids N,N-Dimethyltryptamine (DMT), Harmine, Harmaline and Tetrahydroharmine: Clinical and Forensic Impact. Pharm.-Base 2020, 13, 334. [Google Scholar] [CrossRef]
- Cozzi, N.V.; Gopalakrishnan, A.; Anderson, L.L.; Feih, J.T.; Shulgin, A.T.; Daley, P.F.; Ruoho, A.E. Dimethyltryptamine and other hallucinogenic tryptamines exhibit substrate behavior at the serotonin uptake transporter and the vesicle monoamine transporter. J. Neural. Transm. 2009, 116, 1591–1599. [Google Scholar] [CrossRef]
- Carbonaro, T.M.; Gatch, M.B. Neuropharmacology of N,N-dimethyltryptamine. Brain Res. Bull. 2016, 126, 74–88. [Google Scholar] [CrossRef] [Green Version]
- Aleksandrova, L.R.; Phillips, A.G. Neuroplasticity as a convergent mechanism of ketamine and classical psychedelics. Trends Pharmacol. Sci. 2021, 42, 929–942. [Google Scholar] [CrossRef]
- Carbonaro, T.M.; Eshleman, A.J.; Forster, M.J.; Cheng, K.; Rice, K.C.; Gatch, M. The role of 5-HT2A, 5-HT2C and mGlu2 receptors in the behavioral effects of tryptamine hallucinogens N,N-dimethyltryptamine and N,N-diisopropyltryptamine in rats and mice. Psychopharmacology 2014, 232, 275–284. [Google Scholar] [CrossRef]
- Gouzoulis-Mayfrank, E.; Heekeren, K.; Neukirch, A.; Stoll, M.; Stock, C.; Obradovic, M.; Kovar, K.A. Psychological effects of (S)-ketamine and N,N-dimethyltryptamine (DMT): A double-blind, cross-over study in healthy volunteers. Pharmacopsychiatry 2005, 38, 301–311. [Google Scholar] [CrossRef]
- Fontanilla, D.; Johannessen, M.; Hajipour, A.R.; Cozzi, N.V.; Jackson, M.B.; Ruoho, A.E. The hallucinogen N,N-dimethyltryptamine (DMT) is an endogenous sigma-1 receptor regulator. Science 2009, 323, 934–937. [Google Scholar] [CrossRef]
- Inserra, A. Hypothesis: The Psychedelic Ayahuasca Heals Traumatic Memories via a Sigma 1 Receptor-Mediated Epigenetic-Mnemonic Process. Front. Pharm. 2018, 9, 330. [Google Scholar] [CrossRef] [Green Version]
- Nemes, B.; Peto, K.; Nemeth, N.; Mester, A.; Magyar, Z.; Ghanem, S.; Sogor, V.; Tanczos, B.; Deak, A.; Kallay, M.; et al. N,N-dimethyltryptamine Prevents Renal Ischemia-Reperfusion Injury in a Rat Model. Transplant. Proc. 2019, 51, 1268–1275. [Google Scholar] [CrossRef]
- Su, T.P.; Hayashi, T.; Vaupel, D.B. When the endogenous hallucinogenic trace amine N,N-dimethyltryptamine meets the sigma-1 receptor. Sci. Signal. 2009, 2, pe12. [Google Scholar] [CrossRef] [Green Version]
- Abatematteo, F.S.; Niso, M.; Contino, M.; Leopoldo, M.; Abate, C. Multi-Target Directed Ligands (MTDLs) Binding the sigma(1) Receptor as Promising Therapeutics: State of the Art and Perspectives. Int. J. Mol. Sci. 2021, 22, 6359. [Google Scholar] [CrossRef]
- Penke, B.; Fulop, L.; Szucs, M.; Frecska, E. The Role of Sigma-1 Receptor, an Intracellular Chaperone in Neurodegenerative Diseases. Curr. Neuropharmacol. 2018, 16, 97–116. [Google Scholar] [CrossRef]
- Ryskamp, D.A.; Korban, S.; Zhemkov, V.; Kraskovskaya, N.; Bezprozvanny, I. Neuronal Sigma-1 Receptors: Signaling Functions and Protective Roles in Neurodegenerative Diseases. Front. Neurosci. 2019, 13, 862. [Google Scholar] [CrossRef] [Green Version]
- Szabo, I.; Varga, V.E.; Dvoracsko, S.; Farkas, A.E.; Kormoczi, T.; Berkecz, R.; Kecskes, S.; Menyhart, A.; Frank, R.; Hantosi, D.; et al. N,N-Dimethyltryptamine attenuates spreading depolarization and restrains neurodegeneration by sigma-1 receptor activation in the ischemic rat brain. Neuropharmacology 2021, 192, 108612. [Google Scholar] [CrossRef]
- Lindemann, L.; Ebeling, M.; Kratochwil, N.A.; Bunzow, J.R.; Grandy, D.K.; Hoener, M.C. Trace amine-associated receptors form structurally and functionally distinct subfamilies of novel G protein-coupled receptors. Genomics 2005, 85, 372–385. [Google Scholar] [CrossRef]
- Simão, A.Y.; Gonçalves, J.; Duarte, A.P.; Barroso, M.; Cristóvão, A.C.; Gallardo, E. Toxicological Aspects and Determination of the Main Components of Ayahuasca: A Critical Review. Medicines 2019, 6, 106. [Google Scholar] [CrossRef] [Green Version]
- de Vos, C.M.H.; Mason, N.L.; Kuypers, K.P.C. Psychedelics and Neuroplasticity: A Systematic Review Unraveling the Biological Underpinnings of Psychedelics. Front. Psychiatry 2021, 12, 724606. [Google Scholar] [CrossRef]
- Yin, W.; Zhou, X.E.; Yang, D.; de Waal, P.W.; Wang, M.; Dai, A.; Cai, X.; Huang, C.-Y.; Liu, P.; Wang, X.; et al. Crystal structure of the human 5-HT1B serotonin receptor bound to an inverse agonist. Cell Discov. 2018, 4, 1–13. [Google Scholar] [CrossRef]
- Navarro, P.P.; Stahlberg, H.; Castano-Diez, D. Protocols for Subtomogram Averaging of Membrane Proteins in the Dynamo Software Package. Front. Mol. Biosci. 2018, 5, 82. [Google Scholar] [CrossRef]
- Contreras, N.; Alvíz-Amador, A.; Manzur-Villalobos, I. In silico study of dimethyltryptamine analogues against 5-HT1B receptor: Molecular docking, dynamic simulations and ADMET prediction. J. Herbmed Pharmacol. 2022, 11, 204–212. [Google Scholar] [CrossRef]
- Barbano, A.C.; der Mei, W.F.; deRoon-Cassini, T.A.; Grauer, E.; Lowe, S.R.; Matsuoka, Y.J.; O’Donnell, M.; Olff, M.; Qi, W.; Ratanatharathorn, A.; et al. Differentiating PTSD from anxiety and depression: Lessons from the ICD-11 PTSD diagnostic criteria. Depress. Anxiety 2019, 36, 490–498. [Google Scholar] [CrossRef]
- Wang, Y.-q.; Lin, W.-w.; Wu, N.; Wang, S.-y.; Chen, M.-z.; Lin, Z.-h.; Xie, X.-Q.; Feng, Z.-w. Structural insight into the serotonin (5-HT) receptor family by molecular docking, molecular dynamics simulation and systems pharmacology analysis. Acta Pharmacol. Sin. 2019, 40, 1138–1156. [Google Scholar] [CrossRef]
- Frecska, E.; Szabo, A.; Winkelman, M.J.; Luna, L.E.; McKenna, D.J. A possibly sigma-1 receptor mediated role of dimethyltryptamine in tissue protection, regeneration, and immunity. J. Neural. Transm. 2013, 120, 1295–1303. [Google Scholar] [CrossRef]
- Szabo, A.; Kovacs, A.; Riba, J.; Djurovic, S.; Rajnavolgyi, E.; Frecska, E. The Endogenous Hallucinogen and Trace Amine N,N-Dimethyltryptamine (DMT) Displays Potent Protective Effects against Hypoxia via Sigma-1 Receptor Activation in Human Primary iPSC-Derived Cortical Neurons and Microglia-Like Immune Cells. Front. Neurosci. 2016, 10, 423. [Google Scholar] [CrossRef] [Green Version]
- Nardai, S.; László, M.; Szabó, A.; Alpár, A.; Hanics, J.; Zahola, P.; Merkely, B.; Frecska, E.; Nagy, Z. N,N-dimethyltryptamine reduces infarct size and improves functional recovery following transient focal brain ischemia in rats. Exp. Neurol. 2020, 327, 113245. [Google Scholar] [CrossRef]
- Morales-Garcia, J.A.; Calleja-Conde, J.; Lopez-Moreno, J.A.; Alonso-Gil, S.; Sanz-SanCristobal, M.; Riba, J.; Perez-Castillo, A. N,N-dimethyltryptamine compound found in the hallucinogenic tea ayahuasca, regulates adult neurogenesis in vitro and in vivo. Transl. Psychiatry 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Lie, D.C.; Song, H.J.; Colamarino, S.A.; Ming, G.L.; Gage, F.H. Neurogenesis in the adult brain: New strategies for central nervous system diseases. Annu. Rev. Pharm. 2004, 44, 399–421. [Google Scholar] [CrossRef]
- Cummings, J. Disease modification and Neuroprotection in neurodegenerative disorders. Transl. Neurodegener. 2017, 6, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Lauterbach, E.C.; Victoroff, J.; Coburn, K.L.; Shillcutt, S.D.; Doonan, S.M.; Mendez, M.F. Psychopharmacological Neuroprotection in Neurodegenerative Disease: Assessing the Preclinical Data. J. Neuropsychiatry Clin. Neurosci. 2010, 22, 8–18. [Google Scholar] [CrossRef]
- Apple, D.M.; Fonseca, R.S.; Kokovay, E. The role of adult neurogenesis in psychiatric and cognitive disorders. Brain Res. 2017, 1655, 270–276. [Google Scholar] [CrossRef] [Green Version]
- Katchborian-Neto, A.; Santos, W.T.; Nicácio, K.J.; Corrêa, J.O.; Murgu, M.; Martins, T.M.; Gomes, D.A.; Goes, A.M.; Soares, M.G.; Dias, D.F.; et al. Neuroprotective potential of Ayahuasca and untargeted metabolomics analyses: Applicability to Parkinson’s disease. J. Ethnopharmacol. 2020, 255, 112743. [Google Scholar] [CrossRef]
- Strassman, R.J.; Qualls, C.R.; Uhlenhuth, E.H.; Kellner, R. Dose-response study of N,N-dimethyltryptamine in humans. II. Subjective effects and preliminary results of a new rating scale. Arch. Gen. Psychiatry 1994, 51, 98–108. [Google Scholar] [CrossRef]
- Grob, C.S.; McKenna, D.J.; Callaway, J.C.; Brito, G.S.; Neves, E.S.; Oberlaender, G.; Saide, O.L.; Labigalini, E.; Tacla, C.; Miranda, C.T.; et al. Human psychopharmacology of hoasca, a plant hallucinogen used in ritual context in Brazil. J. Nerv. Ment. Dis. 1996, 184, 86–94. [Google Scholar] [CrossRef] [Green Version]
- Osorio Fde, L.; Sanches, R.F.; Macedo, L.R.; Santos, R.G.; Maia-de-Oliveira, J.P.; Wichert-Ana, L.; Araujo, D.B.; Riba, J.; Crippa, J.A.; Hallak, J.E. Antidepressant effects of a single dose of ayahuasca in patients with recurrent depression: A preliminary report. Rev. Bras. De Psiquiatr. 2015, 37, 13–20. [Google Scholar] [CrossRef] [Green Version]
- Sanches, R.F.; Osorio, F.D.; dos Santos, R.G.; Macedo, L.R.H.; Maia-de-Oliveira, J.P.; Wichert-Ana, L.; de Araujo, D.B.; Riba, J.; Crippa, J.A.S.; Hallak, J.E.C. Antidepressant Effects of a Single Dose of Ayahuasca in Patients With Recurrent Depression A SPECT Study. J. Clin. Psychopharm. 2016, 36, 77–81. [Google Scholar] [CrossRef] [Green Version]
- Palhano-Fontes, F.; Barreto, D.; Onias, H.; Andrade, K.C.; Novaes, M.M.; Pessoa, J.A.; Mota-Rolim, S.A.; Osorio, F.L.; Sanches, R.; dos Santos, R.G.; et al. Rapid antidepressant effects of the psychedelic ayahuasca in treatment-resistant depression: A randomized placebo-controlled trial. Psychol. Med. 2019, 49, 655–663. [Google Scholar] [CrossRef] [Green Version]
- D’Souza, D.C.; Syed, S.A.; Flynn, L.T.; Safi-Aghdam, H.; Cozzi, N.V.; Ranganathan, M. Exploratory study of the dose-related safety, tolerability, and efficacy of dimethyltryptamine (DMT) in healthy volunteers and major depressive disorder. Neuropsychopharmacology 2022, 47, 1854–1862. [Google Scholar] [CrossRef]
- de Almeida, R.N.; Galvão, A.C.D.M.; da Silva, F.S.; Silva, E.A.D.S.; Palhano-Fontes, F.; Maia-De-Oliveira, J.P.; de Araújo, L.-S.B.; Lobão-Soares, B.; Galvão-Coelho, N.L. Modulation of Serum Brain-Derived Neurotrophic Factor by a Single Dose of Ayahuasca: Observation from a Randomized Controlled Trial. Front. Psychol. 2019, 10, 1234. [Google Scholar] [CrossRef] [Green Version]
- Wilkinson, S.T.; Kiselycznyk, C.; Banasr, M.; Webler, R.D.; Haile, C.; Mathew, S.J. Serum and plasma brain-derived neurotrophic factor and response in a randomized controlled trial of riluzole for treatment resistant depression. J. Affect Disord. 2018, 241, 514–518. [Google Scholar] [CrossRef]
- Molendijk, M.L.; Bus, B.A.A.; Spinhoven, P.; Penninx, B.W.J.H.; Kenis, G.; Prickaerts, J.; Voshaar, R.C.O.; Elzinga, B.M. Serum levels of brain-derived neurotrophic factor in major depressive disorder: State-trait issues, clinical features and pharmacological treatment. Mol. Psychiatry 2011, 16, 1088–1095. [Google Scholar] [CrossRef] [Green Version]
- Davis, A.K.; So, S.; Lancelotta, R.; Barsuglia, J.P.; Griffiths, R.R. 5-methoxy-N,N-dimethyltryptamine (5-MeO-DMT) used in a naturalistic group setting is associated with unintended improvements in depression and anxiety. Am. J. Drug Alcohol Abuse 2019, 45, 161–169. [Google Scholar] [CrossRef] [PubMed]
- thaug, M.V.; Lancelotta, R.; van Oorsouw, K.; Kuypers, K.P.C.; Mason, N.; Rak, J.; Šuláková, A.; Jurok, R.; Maryška, M.; Kuchař, M.; et al. A single inhalation of vapor from dried toad secretion containing 5-methoxy-N,N-dimethyltryptamine (5-MeO-DMT) in a naturalistic setting is related to sustained enhancement of satisfaction with life, mindfulness-related capacities, and a decrement of psychopathological symptoms. Psychopharmacology 2019, 236, 2653–2666. [Google Scholar] [CrossRef] [Green Version]
- Michael, P.; Luke, D.; Robinson, O. An Encounter With the Other: A Thematic and Content Analysis of DMT Experiences From a Naturalistic Field Study. Front. Psychol. 2021, 12, 720717. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Garrido, D.F.; Gomez-Sousa, M.; Ona, G.; Dos Santos, R.G.; Hallak, J.E.C.; Alcazar-Corcoles, M.A.; Bouso, J.C. Effects of ayahuasca on mental health and quality of life in naive users: A longitudinal and cross-sectional study combination. Sci. Rep. 2020, 10, 4075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cameron, L.P.; Benson, C.J.; DeFelice, B.C.; Fiehn, O.; Olson, D.E. Chronic, Intermittent Microdoses of the Psychedelic N,N-Dimethyltryptamine (DMT) Produce Positive Effects on Mood and Anxiety in Rodents. ACS Chem. Neurosci. 2019, 10, 3261–3270. [Google Scholar] [CrossRef] [Green Version]
- Shomrat, T.; Nesher, N. Updated View on the Relation of the Pineal Gland to Autism Spectrum Disorders. Front. Endocrinol. 2019, 10, 37. [Google Scholar] [CrossRef]
- Martinez-Cerdeno, V. Dendrite and Spine Modifications in Autism and Related Neurodevelopmental Disorders in Patients and Animal Models. Dev. Neurobiol. 2017, 77, 393–404. [Google Scholar] [CrossRef]
- Ly, C.; Greb, A.C.; Cameron, L.P.; Wong, J.M.; Barragan, E.V.; Wilson, P.C.; Burbach, K.F.; Zarandi, S.S.; Sood, A.; Paddy, M.R.; et al. Psychedelics Promote Structural and Functional Neural Plasticity. Cell Rep. 2018, 23, 3170–3182. [Google Scholar] [CrossRef]
- Lee, J.W.; Park, M.S.; Park, J.-H.; Cho, Y.; Kim, C.; Kim, C.S.; Jo, J.W.; Lim, Y.W. Taxonomic Study of the Genus Pholiota (Strophariaceae, Basidiomycota) in Korea. Mycobiology 2020, 48, 476–483. [Google Scholar] [CrossRef]
- Dorner, S.; Rogge, K.; Fricke, J.; Schafer, T.; Wurlitzer, J.M.; Gressler, M.; Pham, D.N.K.; Manke, D.R.; Chadeayne, A.R.; Hoffmeister, D. Genetic Survey of Psilocybe Natural Products. Chembiochem 2022, 23, e202200249. [Google Scholar] [CrossRef]
- Van Court, R.C.; Wiseman, M.S.; Meyer, K.W.; Ballhorn, D.J.; Amses, K.R.; Slot, J.C.; Dentinger, B.T.M.; Garibay-Orijel, R.; Uehling, J.K. Diversity, biology, and history of psilocybin-containing fungi: Suggestions for research and technological development. Fungal Biol. 2022, 126, 308–319. [Google Scholar] [CrossRef]
- Akers, B.P.; Ruiz, J.F.; Piper, A.; Ruck, C.A.P. A Prehistoric Mural in Spain Depicting Neurotropic Psilocybe Mushrooms? 1. Econ. Bot. 2011, 65, 121–128. [Google Scholar] [CrossRef]
- Winkelman, M. Introduction: Evidence for entheogen use in prehistory and world religions. J. Psychedelic Stud. 2019, 3, 43–62. [Google Scholar] [CrossRef]
- Guzmán, G. Hallucinogenic Mushrooms in Mexico: An Overview. Econ. Bot. 2008, 62, 404–412. [Google Scholar] [CrossRef]
- Zhuk, O.; Jasicka-Misiak, I.; Poliwoda, A.; Kazakova, A.; Godovan, V.V.; Halama, M.; Wieczorek, P.P. Research on Acute Toxicity and the Behavioral Effects of Methanolic Extract from Psilocybin Mushrooms and Psilocin in Mice. Toxins 2015, 7, 1018–1029. [Google Scholar] [CrossRef] [Green Version]
- Badham, E.R. Ethnobotany of psilocybin mushrooms, especially Psilocybe cubensis. J. Ethnopharmacol. 1984, 10, 249–254. [Google Scholar] [CrossRef]
- Christiansen, A.; Rasmussen, K.; Høiland, K. Detection of psilocybin and psilocin in Norwegian species of Pluteus and Conocybe. Planta Med. 2007, 50, 341–343. [Google Scholar] [CrossRef]
- Repke, D.B.; Leslie, D.T.; Guzman, G. Baeocystin in psilocybe, conocybe and panaeolus. Lloydia 1977, 40, 566–578. [Google Scholar]
- Moncalvo, J.-M.; Vilgalys, R.; Redhead, S.A.; Johnson, J.E.; James, T.Y.; Catherine Aime, M.; Hofstetter, V.; Verduin, S.J.W.; Larsson, E.; Baroni, T.J.; et al. One hundred and seventeen clades of euagarics. Mol. Phylogenetics Evol. 2002, 23, 357–400. [Google Scholar] [CrossRef]
- Klein, A.K.; Chatha, M.; Laskowski, L.J.; Anderson, E.I.; Brandt, S.D.; Chapman, S.J.; McCorvy, J.D.; Halberstadt, A.L. Investigation of the Structure-Activity Relationships of Psilocybin Analogues. Acs Pharm. Transl 2021, 4, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Kolaczynska, K.E.; Liechti, M.E.; Duthaler, U. Development and validation of an LC-MS/MS method for the bioanalysis of psilocybin’s main metabolites, psilocin and 4-hydroxyindole-3-acetic acid, in human plasma. J. Chromatogr. B 2020, 1164, 122486. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.T.; Nicholas, C.R.; Cozzi, N.V.; Gassman, M.C.; Cooper, K.M.; Muller, D.; Thomas, C.D.; Hetzel, S.J.; Henriquez, K.M.; Ribaudo, A.S.; et al. Pharmacokinetics of Escalating Doses of Oral Psilocybin in Healthy Adults. Clin. Pharm. 2017, 56, 1543–1554. [Google Scholar] [CrossRef] [PubMed]
- Erkizia-Santamaria, I.; Alles-Pascual, R.; Horrillo, I.; Meana, J.J.; Ortega, J.E. Serotonin 5-HT2A, 5-HT2c and 5-HT1A receptor involvement in the acute effects of psilocybin in mice. In vitro pharmacological profile and modulation of thermoregulation and head-twich response. Biomed Pharm. 2022, 154, 113612. [Google Scholar] [CrossRef] [PubMed]
- Odland, A.U.; Kristensen, J.L.; Andreasen, J.T. Investigating the role of 5-HT2A and 5-HT2C receptor activation in the effects of psilocybin, DOI, and citalopram on marble burying in mice. Behav. Brain Res. 2020, 401, 113093. [Google Scholar] [CrossRef]
- Raval, N.R.; Johansen, A.; Donovan, L.L.; Ros, N.F.; Ozenne, B.; Hansen, H.D.; Knudsen, G.M. A Single Dose of Psilocybin Increases Synaptic Density and Decreases 5-HT2A Receptor Density in the Pig Brain. Int. J. Mol. Sci. 2021, 22, 835. [Google Scholar] [CrossRef]
- Glatfelter, G.C.; Pottie, E.; Partilla, J.S.; Sherwood, A.M.; Kaylo, K.; Pham, D.N.K.; Naeem, M.; Sammeta, V.R.; DeBoer, S.; Golen, J.A.; et al. Structure–Activity Relationships for Psilocybin, Baeocystin, Aeruginascin, and Related Analogues to Produce Pharmacological Effects in Mice. ACS Pharm. Transl. 2022, 5, 1181–1196. [Google Scholar] [CrossRef]
- Lowe, H.; Toyang, N.; Steele, B.; Valentine, H.; Grant, J.; Ali, A.; Ngwa, W.; Gordon, L. The Therapeutic Potential of Psilocybin. Molecules 2021, 26, 2948. [Google Scholar] [CrossRef]
- Mason, N.L.; Kuypers, K.P.C.; Müller, F.; Reckweg, J.; Tse, D.H.Y.; Toennes, S.W.; Hutten, N.R.P.W.; Jansen, J.F.A.; Stiers, P.; Feilding, A.; et al. Me, myself, bye: Regional alterations in glutamate and the experience of ego dissolution with psilocybin. Neuropsychopharmacology 2020, 45, 2003–2011. [Google Scholar] [CrossRef]
- Sakashita, Y.; Abe, K.; Katagiri, N.; Kambe, T.; Saitoh, T.; Utsunomiya, I.; Horiguchi, Y.; Taguchi, K. Effect of psilocin on extracellular dopamine and serotonin levels in the mesoaccumbens and mesocortical pathway in awake rats. Biol. Pharm. Bull. 2015, 38, 134–138. [Google Scholar] [CrossRef] [Green Version]
- Wojtas, A.; Bysiek, A.; Wawrzczak-Bargiela, A.; Szych, Z.; Majcher-Maślanka, I.; Herian, M.; Maćkowiak, M.; Gołembiowska, K. Effect of Psilocybin and Ketamine on Brain Neurotransmitters, Glutamate Receptors, DNA and Rat Behavior. Int. J. Mol. Sci. 2022, 23, 6713. [Google Scholar] [CrossRef]
- Abate, G.; Vezzoli, M.; Polito, L.; Guaita, A.; Albani, D.; Marizzoni, M.; Garrafa, E.; Marengoni, A.; Forloni, G.; Frisoni, G.B.; et al. A Conformation Variant of p53 Combined with Machine Learning Identifies Alzheimer Disease in Preclinical and Prodromal Stages. J. Pers. Med. 2021, 11, 14. [Google Scholar] [CrossRef]
- Cao, C.; Barros-Álvarez, X.; Zhang, S.; Kim, K.; Dämgen, M.A.; Panova, O.; Suomivuori, C.-M.; Fay, J.F.; Zhong, X.; Krumm, B.E.; et al. Signaling snapshots of a serotonin receptor activated by the prototypical psychedelic LSD. Neuron 2022, 110, 3154–3167.e3157. [Google Scholar] [CrossRef]
- Cao, D.; Yu, J.; Wang, H.; Luo, Z.; Liu, X.; He, L.; Qi, J.; Fan, L.; Tang, L.; Chen, Z.; et al. Structure-based discovery of nonhallucinogenic psychedelic analogs. Science 2022, 375, 403–411. [Google Scholar] [CrossRef]
- Gumpper, R.H.; Fay, J.F.; Roth, B.L. Molecular insights into the regulation of constitutive activity by RNA editing of 5HT2C serotonin receptors. Cell Rep. 2022, 40, 111211. [Google Scholar] [CrossRef]
- Studerus, E.; Kometer, M.; Hasler, F.; Vollenweider, F.X. Acute, subacute and long-term subjective effects of psilocybin in healthy humans: A pooled analysis of experimental studies. J. Psychopharmacol. 2011, 25, 1434–1452. [Google Scholar] [CrossRef]
- Gukasyan, N.; Davis, A.K.; Barrett, F.S.; Cosimano, M.P.; Sepeda, N.D.; Johnson, M.W.; Griffiths, R.R. Efficacy and safety of psilocybin-assisted treatment for major depressive disorder: Prospective 12-month follow-up. J. Psychopharmacol. 2022, 36, 151–158. [Google Scholar] [CrossRef]
- Goodwin, G.M.; Aaronson, S.T.; Alvarez, O.; Arden, P.C.; Baker, A.; Bennett, J.C.; Bird, C.; Blom, R.E.; Brennan, C.; Brusch, D.; et al. Single-Dose Psilocybin for a Treatment-Resistant Episode of Major Depression. N. Engl. J. Med. 2022, 387, 1637–1648. [Google Scholar] [CrossRef]
- Zeiss, R.; Gahr, M.; Graf, H. Rediscovering Psilocybin as an Antidepressive Treatment Strategy. Pharmaceuticals 2021, 14, 985. [Google Scholar] [CrossRef]
- Abate, G.; Uberti, D.; Tambaro, S. Potential and Limits of Cannabinoids in Alzheimer’s Disease Therapy. Biology 2021, 10, 542. [Google Scholar] [CrossRef]
- Daws, R.E.; Timmermann, C.; Giribaldi, B.; Sexton, J.D.; Wall, M.B.; Erritzoe, D.; Roseman, L.; Nutt, D.; Carhart-Harris, R. Increased global integration in the brain after psilocybin therapy for depression. Nat. Med. 2022, 28, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Romeu, A.; Darcy, S.; Jackson, H.; White, T.; Rosenberg, P. Psychedelics as Novel Therapeutics in Alzheimer’s Disease: Rationale and Potential Mechanisms. Curr. Top. Behav. Neurosci. 2022, 56, 287–317. [Google Scholar] [CrossRef]
- Foldi, C.J.; Liknaitzky, P.; Williams, M.; Oldfield, B.J. Rethinking Therapeutic Strategies for Anorexia Nervosa: Insights From Psychedelic Medicine and Animal Models. Front. Neurosci. 2020, 14, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spriggs, M.J.; Douglass, H.M.; Park, R.J.; Read, T.; Danby, J.L.; de Magalhaes, F.J.C.; Alderton, K.L.; Williams, T.M.; Blemings, A.; Lafrance, A.; et al. Study Protocol for “Psilocybin as a Treatment for Anorexia Nervosa: A Pilot Study”. Front. Psychiatry 2021, 12, 735523. [Google Scholar] [CrossRef] [PubMed]
- Nielson, E.M.; May, D.G.; Forcehimes, A.A.; Bogenschutz, M.P. The Psychedelic Debriefing in Alcohol Dependence Treatment: Illustrating Key Change Phenomena through Qualitative Content Analysis of Clinical Sessions. Front. Pharm. 2018, 9, 132. [Google Scholar] [CrossRef] [Green Version]
- Johnson, M.W.; Garcia-Romeu, A.; Griffiths, R.R. Long-term follow-up of psilocybin-facilitated smoking cessation. Am. J. Drug Alcohol Abus. 2017, 43, 55–60. [Google Scholar] [CrossRef] [Green Version]
- de Veen, B.T.; Schellekens, A.F.; Verheij, M.M.; Homberg, J.R. Psilocybin for treating substance use disorders? Expert Rev. Neurother. 2017, 17, 203–212. [Google Scholar] [CrossRef]
- Oberti, H.; Dalla Rizza, M.; Reyno, R.; Murchio, S.; Altier, N.; Abreo, E. Diversity of Claviceps paspali reveals unknown lineages and unique alkaloid genotypes. Mycologia 2020, 112, 230–243. [Google Scholar] [CrossRef]
- Hradilov, M.; Vrabka, J.; Vankov, V.; Galuszka, P. Ergot alkaloid production in Claviceps purpurea is regulated by tryptophan related genes. New Biotechnol. 2018, 44, S115. [Google Scholar] [CrossRef]
- Tasker, N.R.; Wipf, P. Biosynthesis, total synthesis, and biological profiles of Ergot alkaloids. Alkaloids. Chem. Biol. 2021, 85, 1–112. [Google Scholar] [CrossRef]
- Hofmann, A. History of the Discovery of Lsd. 50 Years Lsd 1994, 7–16. [Google Scholar]
- Holloway, K. The Secret Black History of LSD. Nation 2022, 314, 16–19. [Google Scholar]
- Bayne, T.; Carter, O. Dimensions of consciousness and the psychedelic state. Neurosci Conscious 2018, 4, niy008. [Google Scholar] [CrossRef] [Green Version]
- Juszczak, G.R. Disrupted integration of sensory stimuli with information about the movement of the body as a mechanism explaining LSD-induced experience. Med. Hypotheses 2017, 100, 94–97. [Google Scholar] [CrossRef]
- Preller, K.H.; Vollenweider, F.X. Phenomenology, Structure, and Dynamic of Psychedelic States. Curr. Top. Behav. Neuro. 2018, 36, 221–256. [Google Scholar] [CrossRef]
- Timmermann, C.; Spriggs, M.J.; Kaelen, M.; Leech, R.; Nutt, D.J.; Moran, R.J.; Carhart-Harris, R.L.; Muthukumaraswamy, S.D. LSD modulates effective connectivity and neural adaptation mechanisms in an auditory oddball paradigm. Neuropharmacology 2018, 142, 251–262. [Google Scholar] [CrossRef]
- Family, N.; Hendricks, P.S.; Williams, L.T.J.; Luke, D.; Krediet, E.; Maillet, E.L.; Raz, S. Safety, tolerability, pharmacokinetics, and subjective effects of 50, 75, and 100 mu g LSD in healthy participants within a novel intervention paradigm: A proof-of-concept study. J. Psychopharmacol. 2022, 36, 321–336. [Google Scholar] [CrossRef]
- Family, N.; Maillet, E.L.; Williams, L.T.J.; Krediet, E.; Carhart-Harris, R.L.; Williams, T.M.; Nichols, C.D.; Goble, D.J.; Raz, S. Safety, tolerability, pharmacokinetics, and pharmacodynamics of low dose lysergic acid diethylamide (LSD) in healthy older volunteers. Psychopharmacology 2020, 237, 841–853. [Google Scholar] [CrossRef] [Green Version]
- Grumann, C.; Henkel, K.; Brandt, S.D.; Stratford, A.; Passie, T.; Auwarter, V. Pharmacokinetics and subjective effects of 1P-LSD in humans after oral and intravenous administration. Drug Test Anal. 2020, 12, 1144–1153. [Google Scholar] [CrossRef]
- Holze, F.; Duthaler, U.; Vizeli, P.; Liechti, M.E. Pharmacokinetics of an oral lysergic acid diethylamide (LSD) solution in healthy subjects. Clin. Toxicol. 2019, 57, 531. [Google Scholar]
- Liechti, M.E.; Schmid, Y.; Rentsch, K.M.; Hammann, F.; Dolder, P.C. Pharmacokinetics and pharmacodynamics of two doses of oral LSD in healthy subjects. Clin. Toxicol. 2017, 55, 475. [Google Scholar]
- Libânio Osório Marta, R.F. Metabolism of lysergic acid diethylamide (LSD): An update. Drug Metab. Rev. 2019, 51, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Passie, T.; Halpern, J.H.; Stichtenoth, D.O.; Emrich, H.M.; Hintzen, A. The pharmacology of lysergic acid diethylamide: A review. CNS Neurosci. Ther. 2008, 14, 295–314. [Google Scholar] [CrossRef] [PubMed]
- Barnes, N.M.; Sharp, T. A review of central 5-HT receptors and their function. Neuropharmacology 1999, 38, 1083–1152. [Google Scholar] [CrossRef] [PubMed]
- Nichols, D.E. Hallucinogens. Pharmacol. Ther. 2004, 101, 131–181. [Google Scholar] [CrossRef]
- González-Maeso, J.; Weisstaub, N.V.; Zhou, M.; Chan, P.; Ivic, L.; Ang, R.; Lira, A.; Bradley-Moore, M.; Ge, Y.; Zhou, Q.; et al. Hallucinogens Recruit Specific Cortical 5-HT2A Receptor-Mediated Signaling Pathways to Affect Behavior. Neuron 2007, 53, 439–452. [Google Scholar] [CrossRef] [Green Version]
- Reissig, C.J.; Eckler, J.R.; Rabin, R.A.; Winter, J.C. The 5-HT1A receptor and the stimulus effects of LSD in the rat. Psychopharmacology 2005, 182, 197–204. [Google Scholar] [CrossRef]
- Moreno, J.L.; Gonzalez-Maeso, J. Crosstalk Between 5-HT2A and mGlu2 Receptors: Implications in Schizophrenia and Its Treatment. Recept. Ser. 2018, 32, 147–189. [Google Scholar] [CrossRef]
- Moreno, J.L.; Holloway, T.; Albizu, L.; Sealfon, S.C.; Gonzalez-Maeso, J. Metabotropic glutamate mGlu2 receptor is necessary for the pharmacological and behavioral effects induced by hallucinogenic 5-HT2A receptor agonists. Neurosci. Lett. 2011, 493, 76–79. [Google Scholar] [CrossRef] [Green Version]
- Ham, S.; Kim, T.K.; Chung, S.; Im, H.I. Drug Abuse and Psychosis: New Insights into Drug-induced Psychosis. Exp. Neurobiol. 2017, 26, 11–24. [Google Scholar] [CrossRef] [Green Version]
- Spigset, O.; Mjorndal, T. Serotonin 5-HT2A receptor binding in platelets from healthy subjects as studied by [H-3]-lysergic acid diethylamide ([H-3]-LSD): Intra- and interindividual variability. Neuropsychopharmacology 1997, 16, 285–293. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.; Che, T.; Panova, O.; DiBerto, J.F.; Lyu, J.; Krumm, B.E.; Wacker, D.; Robertson, M.J.; Seven, A.B.; Nichols, D.E.; et al. Structure of a Hallucinogen-Activated Gq-Coupled 5-HT2A Serotonin Receptor. Cell 2020, 182, 1574–1588.e1519. [Google Scholar] [CrossRef]
- Wacker, D.; Wang, S.; McCorvy, J.D.; Betz, R.M.; Venkatakrishnan, A.J.; Levit, A.; Lansu, K.; Schools, Z.L.; Che, T.; Nichols, D.E.; et al. Crystal Structure of an LSD-Bound Human Serotonin Receptor. Cell 2017, 168, 377–389.e312. [Google Scholar] [CrossRef]
- Wang, C.; Jiang, Y.; Ma, J.; Wu, H.; Wacker, D.; Katritch, V.; Han, G.W.; Liu, W.; Huang, X.-P.; Vardy, E.; et al. Structural Basis for Molecular Recognition at Serotonin Receptors. Science 2013, 340, 610–614. [Google Scholar] [CrossRef] [Green Version]
- Holze, F.; Avedisian, I.; Varghese, N.; Eckert, A.; Liechti, M.E. Role of the 5-HT2A Receptor in Acute Effects of LSD on Empathy and Circulating Oxytocin. Front. Pharmacol. 2021, 12, 711255. [Google Scholar] [CrossRef]
- Lee, Y.; Warne, T.; Nehmé, R.; Pandey, S.; Dwivedi-Agnihotri, H.; Chaturvedi, M.; Edwards, P.C.; García-Nafría, J.; Leslie, A.G.W.; Shukla, A.K.; et al. Molecular basis of β-arrestin coupling to formoterol-bound β1-adrenoceptor. Nature 2020, 583, 862–866. [Google Scholar] [CrossRef]
- Hibicke, M.; Landry, A.N.; Kramer, H.M.; Talman, Z.K.; Nichols, C.D. Psychedelics, but Not Ketamine, Produce Persistent Antidepressant-like Effects in a Rodent Experimental System for the Study of Depression. ACS Chem. Neurosci. 2020, 11, 864–871. [Google Scholar] [CrossRef] [Green Version]
- Inserra, A.; Billard, E.; Grant, E.; Markopoulous, A.; Pilegggi, M.; Haque, M.; Oveisi, A.; Singer, J.; De Gregorio, D.; Hébert, T.; et al. P.0835 Synergistic effects of lysergic acid diethylamide (LSD) and cannabidiol (CBD). Eur. Neuropsychopharmacol. 2021, 53, S610. [Google Scholar] [CrossRef]
- Markopoulos, A.; Inserra, A.; De Gregorio, D.; Gobbi, G. Lysergic acid diethylamide (LSD) promotes social behaviour through 5-HT2A and ampa in the medial prefrontal cortex (MPFC). Eur. Psychiatry 2021, 64, S416–S417. [Google Scholar] [CrossRef]
- Inserra, A.; Giorgini, G.; Lacroix, S.; Bertazzo, A.; Choo, J.; Markopolous, A.; Grant, E.; Abolghasemi, A.; De Gregorio, D.; Flamand, N.; et al. Effects of Repeated Lysergic Acid Diethylamide (LSD) on the Mouse Brain Endocannabinoidome and Gut Microbiome. Br. J. Pharmacol. 2022. [Google Scholar] [CrossRef]
- dos Santos, R.G.; Osório, F.L.; Crippa, J.A.S.; Riba, J.; Zuardi, A.W.; Hallak, J.E.C. Antidepressive, anxiolytic, and antiaddictive effects of ayahuasca, psilocybin and lysergic acid diethylamide (LSD): A systematic review of clinical trials published in the last 25 years. Ther. Adv. Psychopharmacol. 2016, 6, 193–213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muttoni, S.; Ardissino, M.; John, C. Classical psychedelics for the treatment of depression and anxiety: A systematic review. J. Affect Disord. 2019, 258, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Krebs, T.S.; Johansen, P.-Ø. Lysergic acid diethylamide (LSD) for alcoholism: Meta-analysis of randomized controlled trials. J. Psychopharmacol. 2012, 26, 994–1002. [Google Scholar] [CrossRef] [PubMed]
- Gasser, P.; Holstein, D.; Michel, Y.; Doblin, R.; Yazar-Klosinski, B.; Passie, T.; Brenneisen, R. Safety and Efficacy of Lysergic Acid Diethylamide-Assisted Psychotherapy for Anxiety Associated With Life-threatening Diseases. J. Nerv. Ment. Dis. 2014, 202, 513–520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gasser, P.; Kirchner, K.; Passie, T. LSD-assisted psychotherapy for anxiety associated with a life-threatening disease: A qualitative study of acute and sustained subjective effects. J. Psychopharmacol. 2015, 29, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Hutten, N.R.P.W.; Mason, N.L.; Dolder, P.C.; Theunissen, E.L.; Holze, F.; Liechti, M.E.; Feilding, A.; Ramaekers, J.G.; Kuypers, K.P.C. Mood and cognition after administration of low LSD doses in healthy volunteers: A placebo controlled dose-effect finding study. Eur. Neuropsychopharmacol. 2020, 41, 81–91. [Google Scholar] [CrossRef]
- Gupta, A.K.; Dhua, S.; Sahu, P.P.; Abate, G.; Mishra, P.; Mastinu, A. Variation in Phytochemical, Antioxidant and Volatile Composition of Pomelo Fruit (Citrus grandis (L.) Osbeck) during Seasonal Growth and Development. Plants 2021, 10, 1941. [Google Scholar] [CrossRef]
- Kumar, A.; Memo, M.; Mastinu, A. Plant behaviour: An evolutionary response to the environment? Plant Biol. 2020, 22, 961–970. [Google Scholar] [CrossRef]
- Kumar, A.; Premoli, M.; Aria, F.; Bonini, S.A.; Maccarinelli, G.; Gianoncelli, A.; Memo, M.; Mastinu, A. Cannabimimetic plants: Are they new cannabinoidergic modulators? Planta 2019, 249, 1681–1694. [Google Scholar] [CrossRef]
- Manca, I.; Mastinu, A.; Olimpieri, F.; Falzoi, M.; Sani, M.; Ruiu, S.; Loriga, G.; Volonterio, A.; Tambaro, S.; Bottazzi, M.E.H.; et al. Novel pyrazole derivatives as neutral CB1 antagonists with significant activity towards food intake. Eur. J. Med. Chem. 2013, 62, 256–269. [Google Scholar] [CrossRef]
- Mastinu, A.; Ascrizzi, R.; Ribaudo, G.; Bonini, S.A.; Premoli, M.; Aria, F.; Maccarinelli, G.; Gianoncelli, A.; Flamini, G.; Pistelli, L.; et al. Prosocial Effects of Nonpsychotropic Cannabis sativa in Mice. Cannabis Cannabinoid 2022, 7, 170–178. [Google Scholar] [CrossRef]
- Mastinu, A.; Pira, M.; Pani, L.; Pinna, G.A.; Lazzari, P. NESS038C6, a novel selective CB1 antagonist agent with anti-obesity activity and improved molecular profile. Behav Brain Res 2012, 234, 192–204. [Google Scholar] [CrossRef]
- Pucci, M.; Mandrone, M.; Chiocchio, I.; Mac Sweeney, E.; Tirelli, E.; Uberti, D.; Memo, M.; Poli, F.; Mastinu, A.; Abate, G. Different Seasonal Collections of Ficus carica L. Leaves Diversely Modulate Lipid Metabolism and Adipogenesis in 3T3-L1 Adipocytes. Nutrients 2022, 14, 2833. [Google Scholar] [CrossRef]
- Ribaudo, G.; Landucci, E.; Giannangeli, M.; Mazzantini, C.; Maccarinelli, G.; Mastinu, A.; Bonini, S.A.; Memo, M.; Pellegrini-Giampietro, D.E.; Gianoncelli, A. Virtual Screening and In Vitro Experiments Highlight Cannabidiol as a Drug-like Phosphodiesterase 9 Inhibitor. Eur. J. Neurosci. 2022. [Google Scholar] [CrossRef]














Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mastinu, A.; Anyanwu, M.; Carone, M.; Abate, G.; Bonini, S.A.; Peron, G.; Tirelli, E.; Pucci, M.; Ribaudo, G.; Oselladore, E.; et al. The Bright Side of Psychedelics: Latest Advances and Challenges in Neuropharmacology. Int. J. Mol. Sci. 2023, 24, 1329. https://doi.org/10.3390/ijms24021329
Mastinu A, Anyanwu M, Carone M, Abate G, Bonini SA, Peron G, Tirelli E, Pucci M, Ribaudo G, Oselladore E, et al. The Bright Side of Psychedelics: Latest Advances and Challenges in Neuropharmacology. International Journal of Molecular Sciences. 2023; 24(2):1329. https://doi.org/10.3390/ijms24021329
Chicago/Turabian StyleMastinu, Andrea, Margrate Anyanwu, Marinella Carone, Giulia Abate, Sara Anna Bonini, Gregorio Peron, Emanuela Tirelli, Mariachiara Pucci, Giovanni Ribaudo, Erika Oselladore, and et al. 2023. "The Bright Side of Psychedelics: Latest Advances and Challenges in Neuropharmacology" International Journal of Molecular Sciences 24, no. 2: 1329. https://doi.org/10.3390/ijms24021329






