Studying the Association of TKS4 and CD2AP Scaffold Proteins and Their Implications in the Partial Epithelial–Mesenchymal Transition (EMT) Process
Abstract
:1. Introduction
2. Results
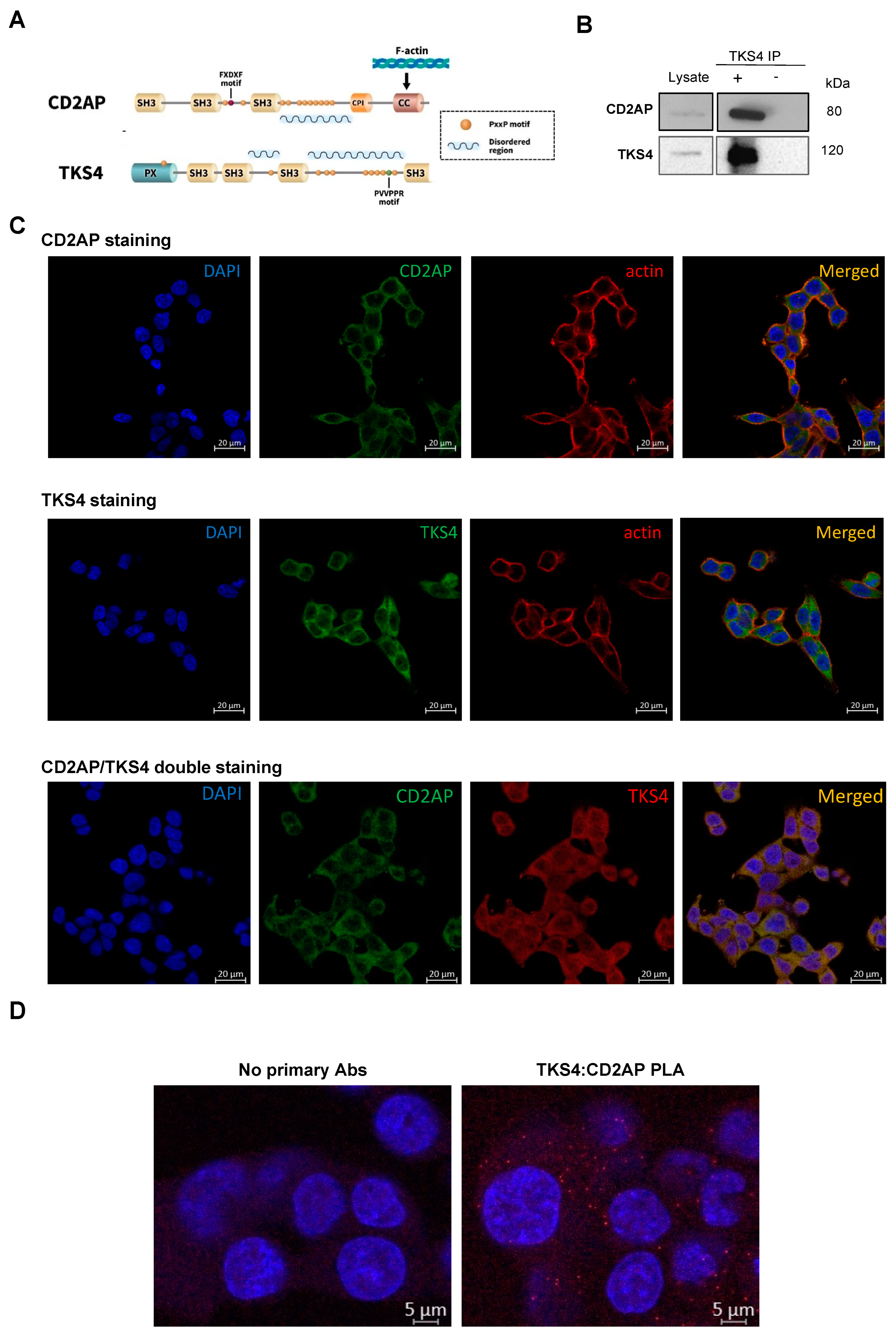
2.1. CD2AP and TKS4 Form a Scaffolding Protein Complex
2.2. Absence of CD2AP and TKS4 Promotes Colon Cancer Cell Migration Separately and Simultaneously via a Partial Epithelial–Mesenchymal Transition Process
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. siRNA and DNA Plasmid Transfection
4.3. RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
4.4. Western Blotting and Immunoprecipitation
4.5. Mass Spectrometry Analyses
4.6. In-Gel Protein Digestion
4.7. Liquid Chromatography-Mass Spectrometry Analysis
4.8. Protein Identification
4.9. Cell Proliferation Assay
4.10. Wound Healing Assays
4.11. Immunocytochemistry
4.12. DuoLink Proximity Ligation Assays
4.13. Peptide Array
4.14. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- László, L.; Kurilla, A.; Takács, T.; Kudlik, G.; Koprivanacz, K.; Buday, L.; Vas, V. Recent Updates on the Significance of KRAS Mutations in Colorectal Cancer Biology. Cells 2021, 10, 667. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Weinberg, R.A. HHS Public Access. Trends Cell Biol. 2015, 25, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Antin, P.; Berx, G.; Blanpain, C.; Brabletz, T.; Bronner, M.; Campbell, K.; Cano, A.; Casanova, J.; Christofori, G.; et al. Guidelines and Definitions for Research on Epithelial–Mesenchymal Transition. Nat. Rev. Mol. Cell Biol. 2020, 21, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Pastushenko, I.; Brisebarre, A.; Sifrim, A.; Fioramonti, M.; Revenco, T.; Boumahdi, S.; Van Keymeulen, A.; Brown, D.; Moers, V.; Lemaire, S.; et al. Identification of the Tumour Transition States Occurring during EMT. Nature 2018, 556, 463–468. [Google Scholar] [CrossRef]
- Saitoh, M. JB Special Review-Cellular Plasticity in Epithelial Homeostasis and Diseases: Involvement of Partial EMT in Cancer Progression. J. Biochem. 2018, 164, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Yagasaki, R.; Noguchi, M.; Minami, M.; Earashi, M. Clinical Significance of E-Cadherin and Vimentin Co-Expression in Breast Cancer. Int. J. Oncol. 1996, 9, 755–761. [Google Scholar] [CrossRef]
- Aggarwal, V.; Montoya, C.A.; Donnenberg, V.S.; Sant, S. Interplay between Tumor Microenvironment and Partial EMT as the Driver of Tumor Progression. iScience 2021, 24, 102113. [Google Scholar] [CrossRef]
- Buschman, M.D.; Bromann, P.A.; Cejudo-Martin, P.; Wen, F.; Pass, I.; Courtneidge, S.A. The Novel Adaptor Protein Tks4 (SH3PXD2B) Is Required for Functional Podosome Formation. Mol. Biol. Cell 2009, 20, 1302–1311. [Google Scholar] [CrossRef]
- Iizuka, S.; Abdullah, C.; Buschman, M.D.; Diaz, B.; Courtneidge, S.A. The Role of Tks Adaptor Proteins in Invadopodia Formation, Growth and Metastasis of Melanoma. Oncotarget 2016, 7, 78473–78486. [Google Scholar] [CrossRef]
- Dülk, M.; Kudlik, G.; Fekete, A.; Ernszt, D.; Kvell, K.; Pongrácz, J.E.; Mero, B.L.; Szeder, B.; Radnai, L.; Geiszt, M.; et al. The Scaffold Protein Tks4 Is Required for the Differentiation of Mesenchymal Stromal Cells (MSCs) into Adipogenic and Osteogenic Lineages. Sci. Rep. 2016, 6, 34280. [Google Scholar] [CrossRef]
- Vas, V.; Háhner, T.; Kudlik, G.; Ernszt, D.; Kvell, K.; Kuti, D.; Kovács, K.J.; Tóvári, J.; Trexler, M.; Merő, B.L.; et al. Analysis of Tks4 Knockout Mice Suggests a Role for Tks4 in Adipose Tissue Homeostasis in the Context of Beigeing. Cells 2019, 8, 831. [Google Scholar] [CrossRef]
- Vas, V.; Kovács, T.; Körmendi, S.; Bródy, A.; Kudlik, G.; Szeder, B.; Mező, D.; Kállai, D.; Koprivanacz, K.; Merő, B.L.; et al. Significance of the Tks4 Scaffold Protein in Bone Tissue Homeostasis. Sci. Rep. 2019, 9, 5781. [Google Scholar] [CrossRef] [PubMed]
- Maziveyi, M.; Dong, S.; Baranwal, S.; Alahari, S.K. Nischarin Regulates Focal Adhesion and Invadopodia Formation in Breast Cancer Cells. Mol. Cancer 2018, 17, 21. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-H.; Liu, Y.; Gao, S.-Y. Correlation between IL-7 Genomic Protein Methylation Level and Acute Myeloid Leukemia. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 1196–1202. [Google Scholar] [CrossRef] [PubMed]
- Kui, X.; Wang, Y.; Zhang, C.; Li, H.; Li, Q.; Ke, Y.; Wang, L. Prognostic Value of SH3PXD2B (Tks4) in Human Hepatocellular Carcinoma: A Combined Multi-Omics and Experimental Study. BMC Med. Genomics 2021, 14, 115. [Google Scholar] [CrossRef]
- Caires-dos-Santos, L.; da Silva, S.V.; Smuczek, B.; de Siqueira, A.S.; Cruz, K.S.P.; Barbuto, J.A.M.; Augusto, T.M.; Freitas, V.M.; Carvalho, H.F.; Jaeger, R.G. Laminin-Derived Peptide C16 Regulates Tks Expression and Reactive Oxygen Species Generation in Human Prostate Cancer Cells. J. Cell. Physiol. 2020, 235, 587–598. [Google Scholar] [CrossRef]
- Szeder, B.; Tárnoki-Zách, J.; Lakatos, D.; Vas, V.; Kudlik, G.; Merő, B.; Koprivanacz, K.; Bányai, L.; Hámori, L.; Róna, G.; et al. Absence of the Tks4 Scaffold Protein Induces Epithelial-Mesenchymal Transition-Like Changes in Human Colon Cancer Cells. Cells 2019, 8, 1343. [Google Scholar] [CrossRef]
- Bazalii, A.V.; Samoylenko, A.A.; Petukhov, D.M.; Rynditch, A.V.; Redowicz, M.J.; Drobot, L.B. Interaction between Adaptor Proteins Ruk/CIN85 and Tks4 in Normal and Tumor Cells of Different Tissue Origins. Biopolym. Cell 2015, 30, 37–41. [Google Scholar] [CrossRef]
- Kowanetz, K.; Szymkiewicz, I.; Haglund, K.; Kowanetz, M.; Husnjak, K.; Taylor, J.D.; Soubeyran, P.; Engstrom, U.; Ladbury, J.E.; Dikic, I. Identification of a Novel Proline-Arginine Motif Involved in CIN85-Dependent Clustering of Cbl and Down-Regulation of Epidermal Growth Factor Receptors. J. Biol. Chem. 2003, 278, 39735–39746. [Google Scholar] [CrossRef]
- Rouka, E.; Simister, P.C.; Janning, M.; Kumbrink, J.; Konstantinou, T.; Muniz, J.R.C.; Joshi, D.; O’Reilly, N.; Volkmer, R.; Ritter, B.; et al. Differential Recognition Preferences of the Three Src Homology 3 (SH3) Domains from the Adaptor CD2-Associated Protein (CD2AP) and Direct Association with Ras and Rab Interactor 3 (RIN3). J. Biol. Chem. 2015, 290, 25275–25292. [Google Scholar] [CrossRef]
- Lehtonen, S.; Zhao, F.; Lehtonen, E. CD2-Associated Protein Directly Interacts with the Actin Cytoskeleton. Am. J. Physiol.-Ren. Physiol. 2002, 283, 734–743. [Google Scholar] [CrossRef] [PubMed]
- Lynch, D.K.; Winata, S.C.; Lyons, R.J.; Hughes, W.E.; Lehrbach, G.M.; Wasinger, V.; Corthals, G.; Cordwell, S.; Daly, R.J. A Cortactin-CD2-Associated Protein (CD2AP) Complex Provides a Novel Link between Epidermal Growth Factor Receptor Endocytosis and the Actin Cytoskeleton. J. Biol. Chem. 2003, 278, 21805–21813. [Google Scholar] [CrossRef] [PubMed]
- Tang, V.W.; Brieher, W.M. FSGS3/CD2AP Is a Barbed-End Capping Protein That Stabilizes Actin and Strengthens Adherens Junctions. J. Cell Biol. 2013, 203, 815–833. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Chen, C.; Han, Z.; Huang, J.; Liu, X.; Chen, H.; Zhang, T.; Chen, S.; Chen, C.; Lu, M.; et al. CD2AP Inhibits Metastasis in Gastric Cancer by Promoting Cellular Adhesion and Cytoskeleton Assembly. Mol. Carcinog. 2020, 59, 339–352. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Chen, J.; Ding, Y.; Kong, H.; You, H.; Zhao, Y.; Wei, H.; Liu, Y. MiR-188-5p Promotes Tumor Growth by Targeting CD2AP through PI3K/AKT/MTOR Signaling in Children with Acute Promyelocytic Leukemia. Onco. Targets. Ther. 2020, 13, 6681–6697. [Google Scholar] [CrossRef]
- Kurakin, A.V.; Wu, S.; Bredesen, D.E. Atypical Recognition Consensus of CIN85/SETA/Ruk SH3 Domains Revealed by Target-Assisted Iterative Screening. J. Biol. Chem. 2003, 278, 34102–34109. [Google Scholar] [CrossRef]
- Haglund, K.; Schmidt, M.H.H.; Wong, E.S.M.; Guy, G.R.; Dikic, I. Sprouty2 Acts at the Cbl/CIN85 Interface to Inhibit Epidermal Growth Factor Receptor Downregulation. EMBO Rep. 2005, 6, 635–641. [Google Scholar] [CrossRef]
- Jozic, D.; Cárdenes, N.; Deribe, Y.L.; Moncalián, G.; Hoeller, D.; Groemping, Y.; Dikic, I.; Rittinger, K.; Bravo, J. Cbl Promotes Clustering of Endocytic Adaptor Proteins. Nat. Struct. Mol. Biol. 2005, 12, 972–979. [Google Scholar] [CrossRef]
- Peiró, S.; Escrivà, M.; Puig, I.; Barberà, M.J.; Dave, N.; Herranz, N.; Larriba, M.J.; Takkunen, M.; Francí, C.; Muñoz, A.; et al. Snail1 Transcriptional Repressor Binds to Its Own Promoter and Controls Its Expression. Nucleic Acids Res. 2006, 34, 2077–2084. [Google Scholar] [CrossRef]
- Yablonski, D. Bridging the Gap: Modulatory Roles of the Grb2-Family Adaptor, Gads, in Cellular and Allergic Immune Responses. Front. Immunol. 2019, 10, 1704. [Google Scholar] [CrossRef]
- Buday, L.; Khwaja, A.; Sipeki, S.; Faragó, A.; Downward, J. Interactions of Cbl with Two Adaptor Proteins, Grb2 and Crk, upon T Cell Activation. J. Biol. Chem. 1996, 271, 6159–6163. [Google Scholar] [CrossRef] [PubMed]
- Lányi, Á.; Baráth, M.; Péterfi, Z.; Bőgel, G.; Orient, A.; Simon, T.; Petrovszki, E.; Kis-Tóth, K.; Sirokmány, G.; Rajnavölgyi, É.; et al. The Homolog of the Five SH3-Domain Protein (HOFI/SH3PXD2B) Regulates Lamellipodia Formation and Cell Spreading. PLoS ONE 2011, 6, e23653. [Google Scholar] [CrossRef] [PubMed]
- Bögel, G.; Gujdár, A.; Geiszt, M.; Lányi, Á.; Fekete, A.; Sipeki, S.; Downward, J.; Buday, L. Frank-Ter Haar Syndrome Protein Tks4 Regulates Epidermal Growth Factor-Dependent Cell Migration. J. Biol. Chem. 2012, 287, 31321–31329. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Bruck, S.; Cemerski, S.; Zhang, L.; Butler, B.; Dani, A.; Cooper, J.A.; Shaw, A.S. CD2AP Links Cortactin and Capping Protein at the Cell Periphery To Facilitate Formation of Lamellipodia. Mol. Cell. Biol. 2013, 33, 38–47. [Google Scholar] [CrossRef]
- Ni, Q.F.; Yu, J.W.; Qian, F.; Sun, N.Z.; Xiao, J.J.; Zhu, J.W. Cortactin Promotes Colon Cancer Progression by Regulating ERK Pathway. Int. J. Oncol. 2015, 47, 1034–1042. [Google Scholar] [CrossRef]
- Ji, R.; Zhu, X.J.; Wang, Z.R.; Huang, L.Q. Cortactin in Epithelial–Mesenchymal Transition. Front. Cell Dev. Biol. 2020, 8, 585619. [Google Scholar] [CrossRef]
- Feldker, N.; Ferrazzi, F.; Schuhwerk, H.; Widholz, S.A.; Guenther, K.; Frisch, I.; Jakob, K.; Kleemann, J.; Riegel, D.; Bönisch, U.; et al. Genome-wide Cooperation of EMT Transcription Factor ZEB 1 with YAP and AP-1 in Breast Cancer. EMBO J. 2020, 39, e103209. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 Years of Image Analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Cox, J.; Mann, M. MaxQuant Enables High Peptide Identification Rates, Individualized p.p.b.-Range Mass Accuracies and Proteome-Wide Protein Quantification. Nat. Biotechnol. 2008, 26, 1367–1372. [Google Scholar] [CrossRef]
- Stauffer, W.; Sheng, H.; Lim, H.N. EzColocalization: An ImageJ Plugin for Visualizing and Measuring Colocalization in Cells and Organisms. Sci. Rep. 2018, 8, 15764. [Google Scholar] [CrossRef]
- Dülk, M.; Szeder, B.; Glatz, G.; Merő, B.L.; Koprivanacz, K.; Kudlik, G.; Vas, V.; Sipeki, S.; Cserkaszky, A.; Radnai, L.; et al. EGF Regulates the Interaction of Tks4 with Src through Its SH2 and SH3 Domains. Biochemistry 2018, 57, 4186–4196. [Google Scholar] [CrossRef] [PubMed]
- Harnoš, J.; Ryneš, J.; Víšková, P.; Foldynová-Trantírková, S.; Bajard-Ešner, L.; Trantírek, L.; Bryja, V. Analysis of Binding Interfaces of the Human Scaffold Protein AXIN1 by Peptide Microarrays. J. Biol. Chem. 2018, 293, 16337–16347. [Google Scholar] [CrossRef] [PubMed]
- Kabsch, W.; Sander, C. Dictionary of Protein Secondary Structure: Pattern Recognition of Hydrogen-Bonded and Geometrical Features. Biopolymers 1983, 22, 2577–2637. [Google Scholar] [CrossRef]
- Tunyasuvunakool, K.; Adler, J.; Wu, Z.; Green, T.; Zielinski, M.; Žídek, A.; Bridgland, A.; Cowie, A.; Meyer, C.; Laydon, A.; et al. Highly Accurate Protein Structure Prediction for the Human Proteome. Nature 2021, 596, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Tien, M.Z.; Meyer, A.G.; Sydykova, D.K.; Spielman, S.J.; Wilke, C.O. Maximum Allowed Solvent Accessibilites of Residues in Proteins. PLoS ONE 2013, 8, e80635. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing . J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kurilla, A.; László, L.; Takács, T.; Tilajka, Á.; Lukács, L.; Novák, J.; Pancsa, R.; Buday, L.; Vas, V. Studying the Association of TKS4 and CD2AP Scaffold Proteins and Their Implications in the Partial Epithelial–Mesenchymal Transition (EMT) Process. Int. J. Mol. Sci. 2023, 24, 15136. https://doi.org/10.3390/ijms242015136
Kurilla A, László L, Takács T, Tilajka Á, Lukács L, Novák J, Pancsa R, Buday L, Vas V. Studying the Association of TKS4 and CD2AP Scaffold Proteins and Their Implications in the Partial Epithelial–Mesenchymal Transition (EMT) Process. International Journal of Molecular Sciences. 2023; 24(20):15136. https://doi.org/10.3390/ijms242015136
Chicago/Turabian StyleKurilla, Anita, Loretta László, Tamás Takács, Álmos Tilajka, Laura Lukács, Julianna Novák, Rita Pancsa, László Buday, and Virág Vas. 2023. "Studying the Association of TKS4 and CD2AP Scaffold Proteins and Their Implications in the Partial Epithelial–Mesenchymal Transition (EMT) Process" International Journal of Molecular Sciences 24, no. 20: 15136. https://doi.org/10.3390/ijms242015136







