Molecular and Cellular Insights: A Focus on Glycans and the HNK1 Epitope in Autism Spectrum Disorder
Abstract
:1. Introduction
2. Insights into Transcription Patterns and Gene Expression in Normal and Pathological Neurodevelopment
2.1. miRNAs
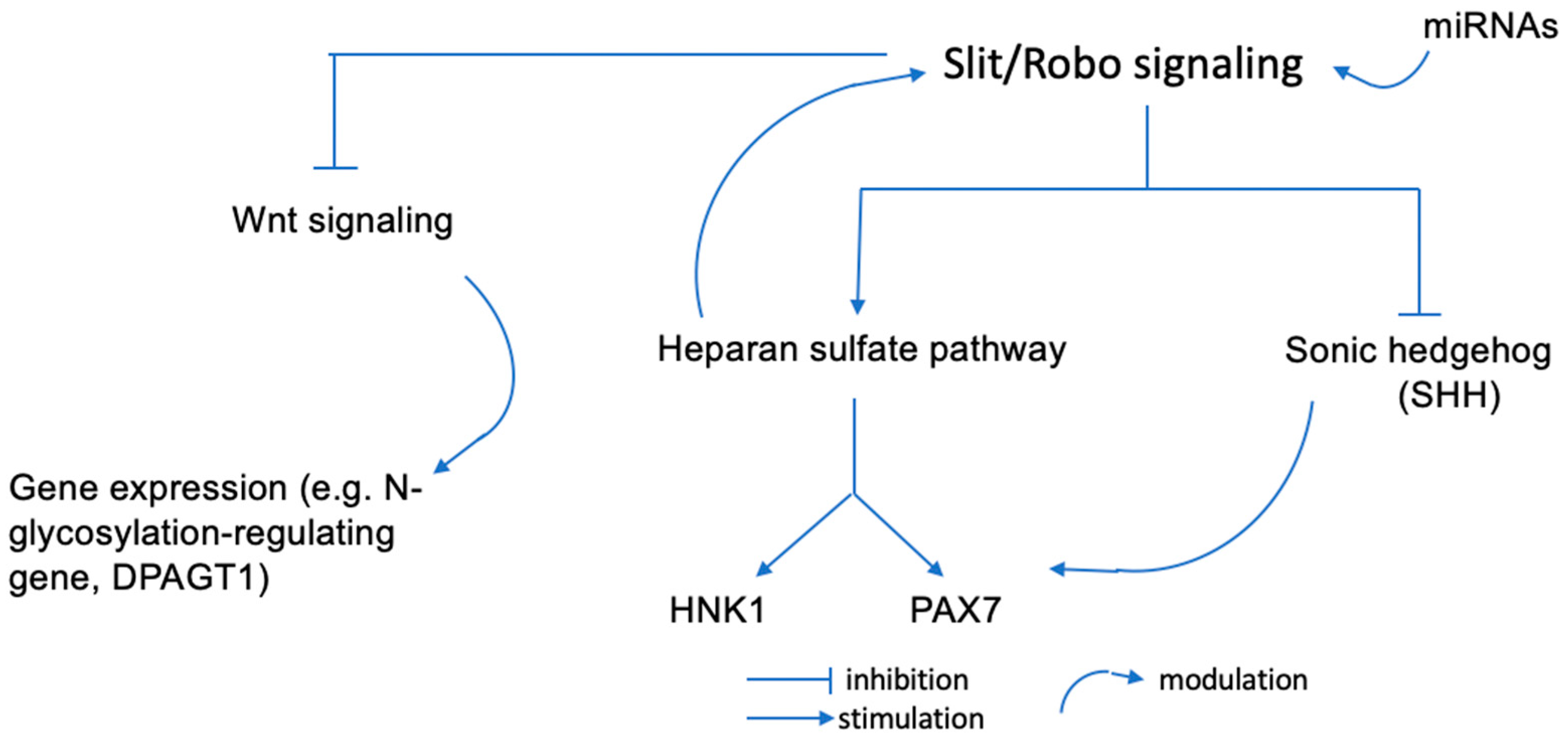
2.2. Slit/Robo Signaling and HNK1
2.3. Mecanisms of Genetic Expression Regulation Implicated in Normal and Pathological Neurodevelopment
2.4. GlycoRNA
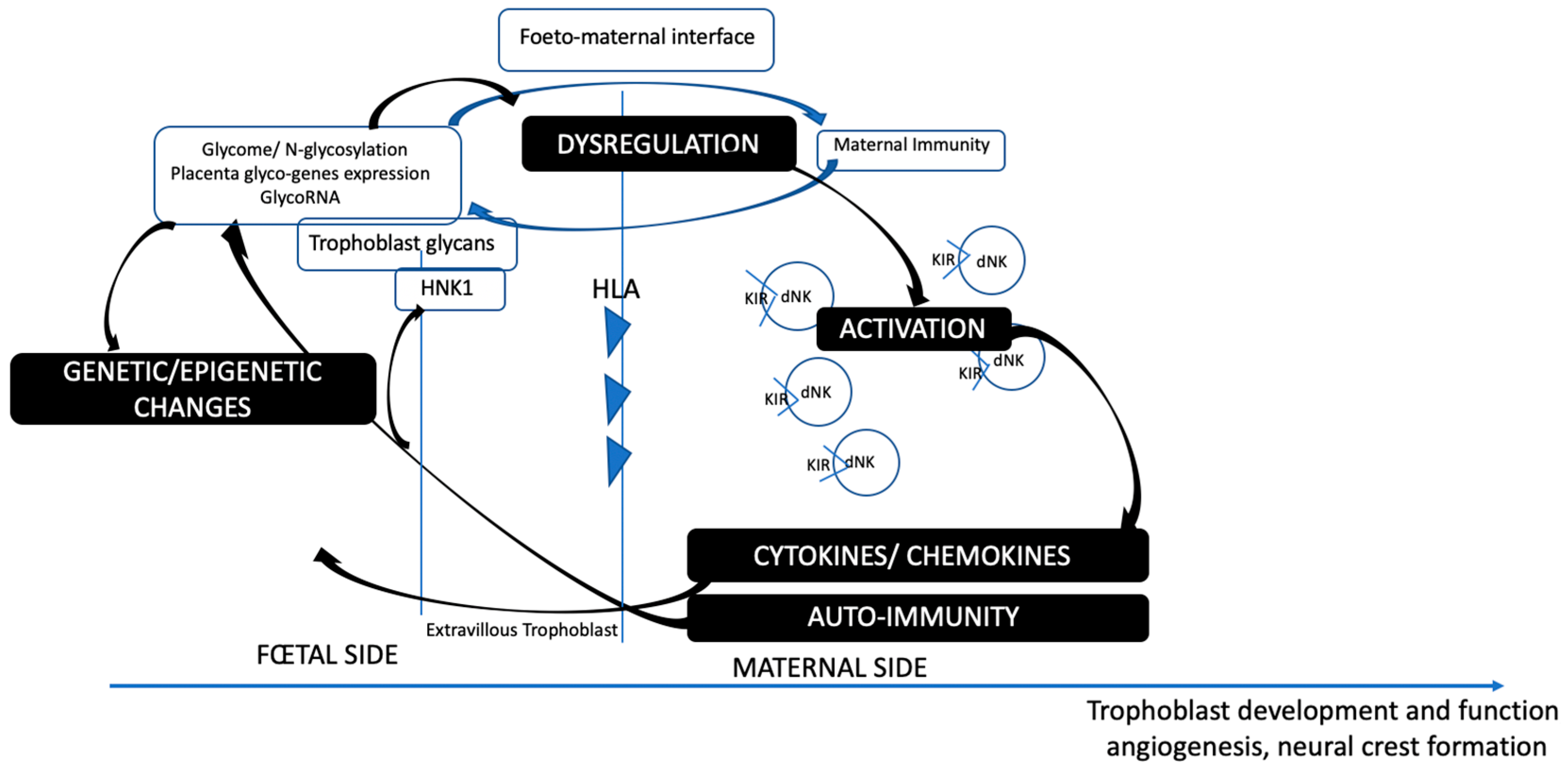
3. Insights into the Maternofetal Interface: HNK1 and Placental Glycosylation Processes
3.1. Slit/Robo in the Uterus and Placenta
3.2. Involvement of Other Glycan Epitopes That Bind to the Siglec Family: CD22, CD24, CD33, and CD45
3.3. Autoimmunity and Antibodies at the Maternofetal Interface
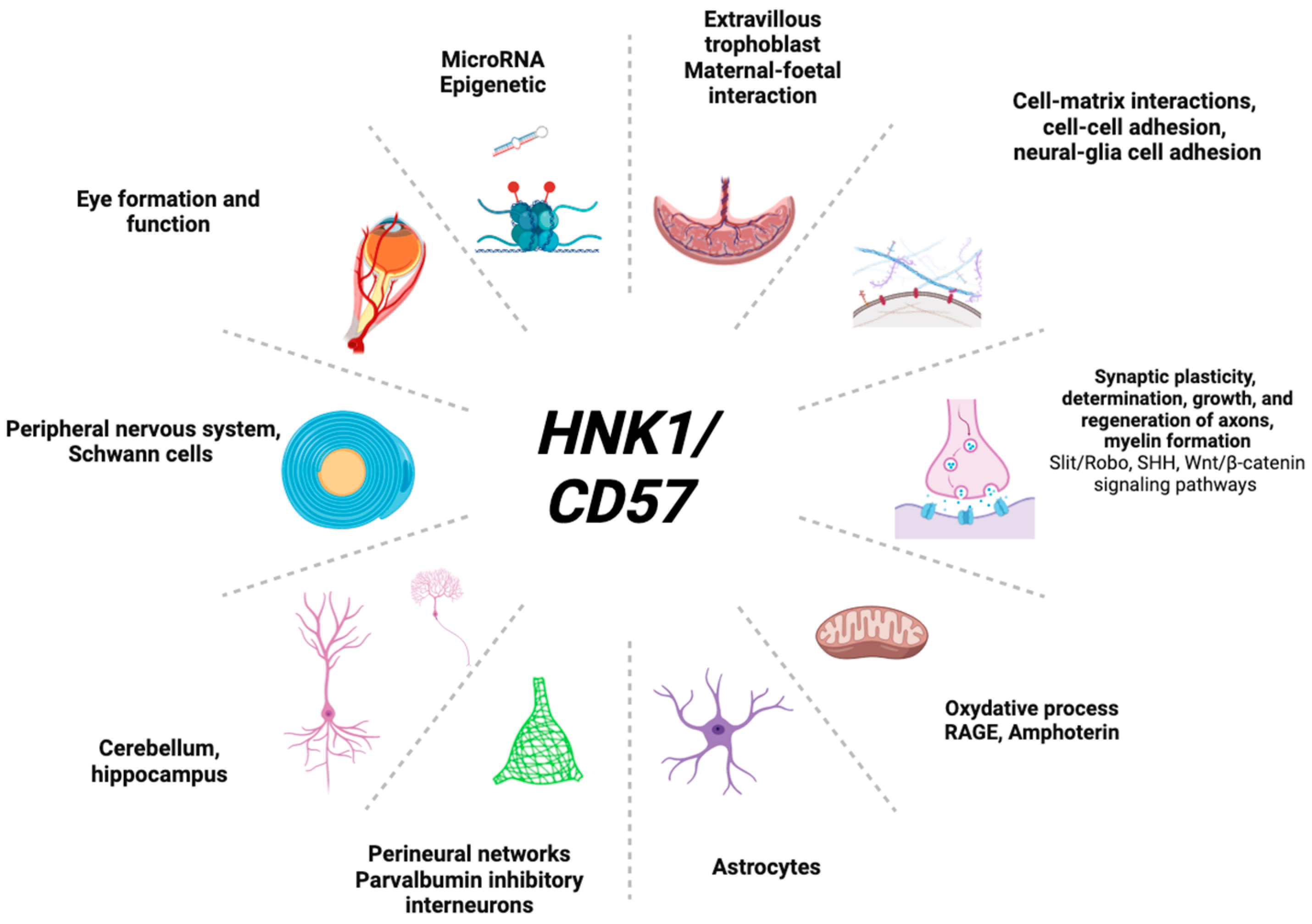
4. Insights on HNK1 Expression in Organs
4.1. In Eye Formation
4.2. In the Brain
4.2.1. RPTP Signaling
4.2.2. SHANK and Ankyrin Protein
4.2.3. NMDA Signaling
4.2.4. Slit/Robo Signaling
4.2.5. Astrocytes
4.3. In Other Organs
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alves, I.; Fernandes, Â.; Santos-Pereira, B.; Azevedo, C.M.; Pinho, S.S. Glycans as a key factor in self and nonself discrimination: Impact on the breach of immune tolerance. FEBS Lett. 2022, 596, 1485–1502. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.S.; Alves, I.; Vicente, M.; Campar, A.; Silva, M.C.; Padrão, N.A.; Pinto, V.; Fernandes, Â.; Dias, A.M.; Pinho, S.S. Glycans as Key Checkpoints of T Cell Activity and Function. Front. Immunol. 2018, 9, 2754. [Google Scholar] [CrossRef]
- Sakuda, K.; Kizuka, Y.; Yamaguchi, Y.; Tanaka, K.; Ogiwara, K.; Segawa, T.; Hagiwara, Y.; Matsuo, I.; Ogawa, H.; Taniguchi, N.; et al. Reactivity of anti-HNK-1 antibodies to branched O-mannose glycans associated with demyelination. Biochem. Biophys. Res. Commun. 2017, 487, 450–456. [Google Scholar] [CrossRef]
- Higuero, A.M.; Díez-Revuelta, N.; Abad-Rodríguez, J. The sugar code in neuronal physiology. Histochem. Cell Biol. 2017, 147, 257–267. [Google Scholar] [CrossRef]
- Huang, Z.; Lai, P.F.; Cocker, A.T.H.; Haslam, S.M.; Dell, A.; Brady, H.J.M.; Johnson, M.R. Roles of N-linked glycosylation and glycan-binding proteins in placentation: Trophoblast infiltration, immunomodulation, angiogenesis, and pathophysiology. Biochem. Soc. Trans. 2023, 51, 639–653. [Google Scholar] [CrossRef]
- McDonald, A.J.; Hamilton, P.G.; Barnstable, C.J. Perineuronal nets labeled by monoclonal antibody VC1.1 ensheath interneurons expressing parvalbumin and calbindin in the rat amygdala. Brain Struct. Funct. 2018, 223, 1133–1148. [Google Scholar] [CrossRef]
- Yabuno, K.; Morise, J.; Kizuka, Y.; Hashii, N.; Kawasaki, N.; Takahashi, S.; Miyata, S.; Izumikawa, T.; Kitagawa, H.; Takematsu, H.; et al. A Sulfated Glycosaminoglycan Linkage Region is a Novel Type of Human Natural Killer-1 (HNK-1) Epitope Expressed on Aggrecan in Perineuronal Nets. PLoS ONE 2015, 10, e0144560. [Google Scholar] [CrossRef] [PubMed]
- Kosaka, T.; Isogai, K.; Barnstable, C.J.; Heizmann, C.W. Monoclonal antibody HNK-1 selectively stains a subpopulation of GABAergic neurons containing the calcium-binding protein parvalbumin in the rat cerebral cortex. Exp. Brain Res. 1990, 82, 566–574. [Google Scholar] [CrossRef]
- Sytnyk, V.; Leshchyns’ka, I.; Schachner, M. Neural glycomics: The sweet side of nervous system functions. Cell Mol. Life Sci. 2021, 78, 93–116. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, T.; Sugihara, K.; Kizuka, Y.; Oka, S.; Asano, M. Learning/memory impairment and reduced expression of the HNK-1 carbohydrate in beta4-galactosyltransferase-II-deficient mice. J. Biol. Chem. 2009, 284, 12550–12561. [Google Scholar] [CrossRef] [PubMed]
- Freedman, A.N.; Eaves, L.A.; Rager, J.E.; Gavino-Lopez, N.; Smeester, L.; Bangma, J.; Santos, H.P.; Joseph, R.M.; Kuban, K.C.; O’Shea, T.M.; et al. The placenta epigenome-brain axis: Placental epigenomic and transcriptomic responses that preprogram cognitive impairment. Epigenomics 2022, 14, 897–911. [Google Scholar] [CrossRef]
- Fetit, R.; Hillary, R.F.; Price, D.J.; Lawrie, S.M. The neuropathology of autism: A systematic review of post-mortem studies of autism and related disorders. Neurosci. Biobehav. Rev. 2021, 129, 35–62. [Google Scholar] [CrossRef]
- Rajgor, D.; Hanley, J.G. The Ins and Outs of miRNA-Mediated Gene Silencing during Neuronal Synaptic Plasticity. Noncoding RNA 2016, 2, 1. [Google Scholar] [CrossRef]
- Vaccaro, T.D.S.; Sorrentino, J.M.; Salvador, S.; Veit, T.; Souza, D.O.; de Almeida, R.F. Alterations in the MicroRNA of the Blood of Autism Spectrum Disorder Patients: Effects on Epigenetic Regulation and Potential Biomarkers. Behav. Sci. 2018, 8, 75. [Google Scholar] [CrossRef]
- Noroozi, R.; Dinger, M.E.; Fatehi, R.; Taheri, M.; Ghafouri-Fard, S. Identification of miRNA-mRNA Network in Autism Spectrum Disorder Using a Bioinformatics Method. J. Mol. Neurosci. 2021, 71, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Gonda, Y.; Namba, T.; Hanashima, C. Beyond Axon Guidance: Roles of Slit-Robo Signaling in Neocortical Formation. Front. Cell Dev. Biol. 2020, 8, 607415. [Google Scholar] [CrossRef] [PubMed]
- Small, E.M.; Sutherland, L.B.; Rajagopalan, K.N.; Wang, S.; Olson, E.N. MicroRNA-218 regulates vascular patterning by modulation of Slit-Robo signaling. Circ. Res. 2010, 107, 1336–1344. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Li, Y.; Wang, X.Y.; Han, Z.; Chuai, M.; Wang, L.J.; Ho Lee, K.K.; Geng, J.G.; Yang, X. Slit/Robo1 signaling regulates neural tube development by balancing neuroepithelial cell proliferation and differentiation. Exp. Cell Res. 2013, 319, 1083–1093. [Google Scholar] [CrossRef]
- Lallier, T.; Leblanc, G.; Artinger, K.B.; Bronner-Fraser, M. Cranial and trunk neural crest cells use different mechanisms for attachment to extracellular matrices. Development 1992, 116, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Bai, X.; Sarkar, A.K.; Esko, J.D. Formation of HNK-1 determinants and the glycosaminoglycan tetrasaccharide linkage region by UDP-GlcUA:Galactose beta1, 3-glucuronosyltransferases. J. Biol. Chem. 1999, 274, 7857–7864. [Google Scholar] [CrossRef]
- Pérez, C.; Sawmiller, D.; Tan, J. The role of heparan sulfate deficiency in autistic phenotype: Potential involvement of Slit/Robo/srGAPs-mediated dendritic spine formation. Neural Dev. 2016, 11, 11. [Google Scholar] [CrossRef] [PubMed]
- Vargas, D.A.; Sun, M.; Sadykov, K.; Kukuruzinska, M.A.; Zaman, M.H. The Integrated Role of Wnt/β-Catenin, N-Glycosylation, and E-Cadherin-Mediated Adhesion in Network Dynamics. PLoS Comput. Biol. 2016, 12, e1005007. [Google Scholar] [CrossRef]
- Markenscoff-Papadimitriou, E.; Binyameen, F.; Whalen, S.; Price, J.; Lim, K.; Ypsilanti, A.R.; Catta-Preta, R.; Pai, E.L.; Mu, X.; Xu, D.; et al. Autism risk gene POGZ promotes chromatin accessibility and expression of clustered synaptic genes. Cell Rep. 2021, 37, 110089. [Google Scholar] [CrossRef]
- Flynn, R.A.; Pedram, K.; Malaker, S.A.; Batista, P.J.; Smith, B.A.H.; Johnson, A.G.; George, B.M.; Majzoub, K.; Villalta, P.W.; Carette, J.E.; et al. Small RNAs are modified with N-glycans and displayed on the surface of living cells. Cell 2021, 184, 3109–3124.e22. [Google Scholar] [CrossRef] [PubMed]
- Ueda, M.; Tsuchiya, K.J.; Yaguchi, C.; Furuta-Isomura, N.; Horikoshi, Y.; Matsumoto, M.; Suzuki, M.; Oda, T.; Kawai, K.; Itoh, T.; et al. Placental pathology predicts infantile neurodevelopment. Sci. Rep. 2022, 12, 2578. [Google Scholar] [CrossRef]
- Liao, W.X.; Wing, D.A.; Geng, J.G.; Chen, D.B. Perspectives of SLIT/ROBO signaling in placental angiogenesis. Histol. Histopathol. 2010, 25, 1181–1190. [Google Scholar] [CrossRef]
- Liao, W.X.; Laurent, L.C.; Agent, S.; Hodges, J.; Chen, D.B. Human placental expression of SLIT/ROBO signaling cues: Effects of preeclampsia and hypoxia. Biol. Reprod. 2012, 86, 111. [Google Scholar] [CrossRef]
- Laufer, B.I.; Neier, K.; Valenzuela, A.E.; Yasui, D.H.; Schmidt, R.J.; Lein, P.J.; LaSalle, J.M. Placenta and fetal brain share a neurodevelopmental disorder DNA methylation profile in a mouse model of prenatal PCB exposure. Cell Rep. 2022, 38, 110442. [Google Scholar] [CrossRef]
- Tiensuu, H.; Haapalainen, A.M.; Karjalainen, M.K.; Pasanen, A.; Huusko, J.M.; Marttila, R.; Ojaniemi, M.; Muglia, L.J.; Hallman, M.; Rämet, M. Risk of spontaneous preterm birth and fetal growth associates with fetal SLIT2. PLoS Genet. 2019, 15, e1008107. [Google Scholar] [CrossRef] [PubMed]
- Tiensuu, H.; Haapalainen, A.M.; Tissarinen, P.; Pasanen, A.; Määttä, T.A.; Huusko, J.M.; Ohlmeier, S.; Bergmann, U.; Ojaniemi, M.; Muglia, L.J.; et al. Human placental proteomics and exon variant studies link AAT/SERPINA1 with spontaneous preterm birth. BMC Med. 2022, 20, 141. [Google Scholar] [CrossRef]
- Lim, R.; Lappas, M. Slit2 exerts anti-inflammatory actions in human placenta and is decreased with maternal obesity. Am. J. Reprod. Immunol. 2015, 73, 66–78. [Google Scholar] [CrossRef]
- Liao, Q.P.; Xu, Q.; Yan, J.Y. SHH expression in placental tissues and trophoblast cell oxidative stress injury during preeclampsia. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 6026–6034. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, X.; Zhang, L.; Shi, Y.; Wang, J.; Yan, H. Wnt/β-catenin signaling pathway in trophoblasts and abnormal activation in preeclampsia (Review). Mol. Med. Rep. 2017, 16, 1007–1013. [Google Scholar] [CrossRef]
- Rizzuto, G.; Brooks, J.F.; Tuomivaara, S.T.; McIntyre, T.I.; Ma, S.; Rideaux, D.; Zikherman, J.; Fisher, S.J.; Erlebacher, A. Establishment of fetomaternal tolerance through glycan-mediated B cell suppression. Nature 2022, 603, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.L.; Fox, C.H.; Fauci, A.S.; Kehrl, J.H. cDNA cloning of the B cell membrane protein CD22: A mediator of B-B cell interactions. J. Exp. Med. 1991, 173, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Sammar, M.; Siwetz, M.; Meiri, H.; Fleming, V.; Altevogt, P.; Huppertz, B. Expression of CD24 and Siglec-10 in first trimester placenta: Implications for immune tolerance at the fetal-maternal interface. Histochem. Cell Biol. 2017, 147, 565–574. [Google Scholar] [CrossRef]
- Bleckmann, C.; Geyer, H.; Lieberoth, A.; Splittstoesser, F.; Liu, Y.; Feizi, T.; Schachner, M.; Kleene, R.; Reinhold, V.; Geyer, R. O-glycosylation pattern of CD24 from mouse brain. Biol. Chem. 2009, 390, 627–645. [Google Scholar] [CrossRef]
- Brinkman-Van der Linden, E.C.; Hurtado-Ziola, N.; Hayakawa, T.; Wiggleton, L.; Benirschke, K.; Varki, A.; Varki, N. Human-specific expression of Siglec-6 in the placenta. Glycobiology 2007, 17, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Bartmann, C.; Junker, M.; Segerer, S.E.; Häusler, S.F.; Krockenberger, M.; Kämmerer, U. CD33(+)/HLA-DR(neg) and CD33(+) /HLA-DR(+/-) Cells: Rare Populations in the Human Decidua with Characteristics of MDSC. Am. J. Reprod. Immunol. 2016, 75, 539–556. [Google Scholar] [CrossRef]
- Earl, L.A.; Baum, L.G. CD45 glycosylation controls T-cell life and death. Immunol. Cell Biol. 2008, 86, 608–615. [Google Scholar] [CrossRef]
- Earl, L.A.; Bi, S.; Baum, L.G. N- and O-glycans modulate galectin-1 binding, CD45 signaling, and T cell death. J. Biol. Chem. 2010, 285, 2232–2244. [Google Scholar] [CrossRef]
- Rheinländer, A.; Schraven, B.; Bommhardt, U. CD45 in human physiology and clinical medicine. Immunol. Lett. 2018, 196, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.F.; Ansari, M.A.; Nadeem, A.; Bakheet, S.A.; Al-Ayadhi, L.Y.; Alasmari, A.F.; Alanazi, M.M.; Al-Mazroua, H.A.; Attia, S.M. Involvement of CD45 cells in the development of Autism Spectrum Disorder through dysregulation of granulocyte-macrophage colony-stimulating factor, key inflammatory cytokines, and transcription factors. Int. Immunopharmacol. 2020, 83, 106466. [Google Scholar] [CrossRef]
- Torres, A.R.; Westover, J.B.; Rosenspire, A.J. HLA Immune Function Genes in Autism. Autism Res. Treat. 2012, 2012, 959073. [Google Scholar] [CrossRef]
- Ebrahimi Meimand, S.; Rostam-Abadi, Y.; Rezaei, N. Autism Spectrum Disorders and natural killer cells: A review on pathogenesis and treatment. Expert Rev. Clin. Immunol. 2021, 17, 27–35. [Google Scholar] [CrossRef]
- Fox-Edmiston, E.; Van de Water, J. Maternal Anti-Fetal Brain IgG Autoantibodies and Autism Spectrum Disorder: Current Knowledge and its Implications for Potential Therapeutics. CNS Drugs 2015, 29, 715–724. [Google Scholar] [CrossRef] [PubMed]
- Konova, E. The role of NK cells in the autoimmune thyroid disease-associated pregnancy loss. Clin. Rev. Allergy Immunol. 2010, 39, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Kucuksezer, U.C.; Aktas Cetin, E.; Esen, F.; Tahrali, I.; Akdeniz, N.; Gelmez, M.Y.; Deniz, G. The Role of Natural Killer Cells in Autoimmune Diseases. Front. Immunol. 2021, 12, 622306. [Google Scholar] [CrossRef]
- Gianchecchi, E.; Delfino, D.V.; Fierabracci, A. Natural Killer Cells: Potential Biomarkers and Therapeutic Target in Autoimmune Diseases? Front. Immunol. 2021, 12, 616853. [Google Scholar] [CrossRef]
- Leveque, L.; Khosrotehrani, K. Feto-maternal allo-immunity, regulatory T cells and predisposition to auto-immunity. Does it all start in utero? Chimerism 2014, 5, 59–62. [Google Scholar] [CrossRef]
- Jennewein, M.F.; Goldfarb, I.; Dolatshahi, S.; Cosgrove, C.; Noelette, F.J.; Krykbaeva, M.; Das, J.; Sarkar, A.; Gorman, M.J.; Fischinger, S.; et al. Fc Glycan-Mediated Regulation of Placental Antibody Transfer. Cell 2019, 178, 202–215.e14. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Lei, Y.; Huang, Y.; Zhao, Y.; Li, J.; Huang, T.; Zhang, J.; Wang, J.; Deng, X.; Chen, Z.; et al. Fab fragment glycosylated IgG may play a central role in placental immune evasion. Hum. Reprod. 2015, 30, 380–391. [Google Scholar] [CrossRef] [PubMed]
- Ziganshina, M.M.; Shilova, N.V.; Khasbiullina, N.R.; Terentyeva, A.V.; Dolgopolova, E.L.; Nokel, A.Y.; Yarotskaya, E.L.; Shmakov, R.G.; Bovin, N.V.; Sukhikh, G.T. Repertoire of glycan-binding placenta-associated antibodies in healthy pregnancy and in pre-eclampsia. Scand J. Immunol. 2022, 95, e13157. [Google Scholar] [CrossRef] [PubMed]
- Chou, D.K.; Henion, T.R.; Jungalwala, F.B. Regulation of expression of sulfoglucuronyl carbohydrate (HNK-1), Amphoterin and RAGE in retinoic acid-differentiated P19 embryonal carcinoma cells. J. Neurochem. 2003, 86, 917–931. [Google Scholar] [CrossRef]
- Jungalwala, F.B. Expression and biological functions of sulfoglucuronyl glycolipids (SGGLs) in the nervous system—A review. Neurochem. Res. 1994, 19, 945–957. [Google Scholar] [CrossRef]
- Mercier, F.; Cho Kwon, Y.; Kodama, R. Meningeal/vascular alterations and loss of extracellular matrix in the neurogenic zone of adult BTBR T+ tf/J mice, animal model for autism. Neurosci. Lett. 2011, 498, 173–178. [Google Scholar] [CrossRef]
- Reynolds, M.; Culican, S.M. Visual Autism. Children 2023, 10, 606. [Google Scholar] [CrossRef]
- Uusitalo, M.; Kivelä, T. The HNK-1 carbohydrate epitope in the eye: Basic science and functional implications. Prog. Retin. Eye Res. 2001, 20, 1–28. [Google Scholar] [CrossRef]
- Uusitalo, M.; Schlötzer-Schrehardt, U.; Kivelä, T. Ultrastructural localization of the HNK-1 carbohydrate epitope to glial and neuronal cells of the human retina. Investig. Ophthalmol. Vis. Sci. 2003, 44, 961–964. [Google Scholar] [CrossRef]
- Klausmeyer, A.; Garwood, J.; Faissner, A. Differential expression of phosphacan/RPTPbeta isoforms in the developing mouse visual system. J. Comp. Neurol. 2007, 504, 659–679. [Google Scholar] [CrossRef]
- Araki, M.; Takano, T.; Uemonsa, T.; Nakane, Y.; Tsudzuki, M.; Kaneko, T. Epithelia-mesenchyme interaction plays an essential role in transdifferentiation of retinal pigment epithelium of silver mutant quail: Localization of FGF and related molecules and aberrant migration pattern of neural crest cells during eye rudiment formation. Dev. Biol. 2002, 244, 358–371. [Google Scholar] [CrossRef]
- Chung, S.; Rho, S.; Kim, G.; Kim, S.R.; Baek, K.H.; Kang, M.; Lew, H. Human umbilical cord blood mononuclear cells and chorionic plate-derived mesenchymal stem cells promote axon survival in a rat model of optic nerve crush injury. Int. J. Mol. Med. 2016, 37, 1170–1180. [Google Scholar] [CrossRef]
- Domowicz, M.S.; Mueller, M.M.; Novak, T.E.; Schwartz, L.E.; Schwartz, N.B. Developmental expression of the HNK-1 carbohydrate epitope on aggrecan during chondrogenesis. Dev. Dyn. 2003, 226, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Melrose, J. Keratan sulfate (KS)-proteoglycans and neuronal regulation in health and disease: The importance of KS-glycodynamics and interactive capability with neuroregulatory ligands. J. Neurochem. 2019, 149, 170–194. [Google Scholar] [CrossRef]
- Ye, K.; Yu, J.; Li, L.; Wang, H.; Tang, B.; Ni, W.; Zhou, J.; Ling, Y.; Lu, X.; Niu, D.; et al. Microvesicles from Schwann-Like Cells as a New Biomaterial Promote Axonal Growth. J. Biomed. Nanotechnol. 2021, 17, 291–302. [Google Scholar] [CrossRef]
- Dino, M.R.; Harroch, S.; Hockfield, S.; Matthews, R.T. Monoclonal antibody Cat-315 detects a glycoform of receptor protein tyrosine phosphatase beta/phosphacan early in CNS development that localizes to extrasynaptic sites prior to synapse formation. Neuroscience 2006, 142, 1055–1069. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, C.A.; Katoh, T.; Tiemeyer, M.; Matthews, R.T. Neurons and glia modify receptor protein-tyrosine phosphatase ζ (RPTPζ)/phosphacan with cell-specific O-mannosyl glycans in the developing brain. J. Biol. Chem. 2015, 290, 10256–10273. [Google Scholar] [CrossRef] [PubMed]
- Ishioka, N.; Sato, J.; Kurioka, S. A soluble neurite outgrowth molecule in bovine brain. Biochem. Biophys. Res. Commun. 1994, 199, 1174–1180. [Google Scholar] [CrossRef]
- Heise, C.; Preuss, J.M.; Schroeder, J.C.; Battaglia, C.R.; Kolibius, J.; Schmid, R.; Kreutz, M.R.; Kas, M.J.H.; Burbach, J.P.H.; Boeckers, T.M. Heterogeneity of Cell Surface Glutamate and GABA Receptor Expression in Shank and CNTN4 Autism Mouse Models. Front. Mol. Neurosci. 2018, 11, 212. [Google Scholar] [CrossRef]
- Yamada, J.; Ohgomori, T.; Jinno, S. Alterations in expression of Cat-315 epitope of perineuronal nets during normal ageing, and its modulation by an open-channel NMDA receptor blocker, memantine. J. Comp. Neurol. 2017, 525, 2035–2049. [Google Scholar] [CrossRef]
- Deutsch, S.I.; Luyo, Z.N.M.; Burket, J.A. Targeted NMDA Receptor Interventions for Autism: Developmentally Determined Expression of GluN2B and GluN2A-Containing Receptors and Balanced Allosteric Modulatory Approaches. Biomolecules 2022, 12, 181. [Google Scholar] [CrossRef] [PubMed]
- Mascio, G.; Bucci, D.; Notartomaso, S.; Liberatore, F.; Antenucci, N.; Scarselli, P.; Imbriglio, T.; Caruso, S.; Gradini, R.; Cannella, M.; et al. Perineuronal nets are under the control of type-5 metabotropic glutamate receptors in the developing somatosensory cortex. Transl. Psychiatry 2021, 11, 109. [Google Scholar] [CrossRef]
- Kizuka, Y.; Kanekiyo, K.; Kitazume, S.; Taniguchi, N. Use of glycan-targeted antibodies/lectins to study the expression/function of glycosyltransferases in the nervous system. Adv. Neurobiol. 2014, 9, 117–127. [Google Scholar] [CrossRef]
- Takahashi, K.; Kanekiyo, K.; Sakuda, K.; Muto, Y.; Iguchi, M.; Matsuda, N.; Hashimoto, Y.; Kanai, K.; Ogawa, H.; Hirase, H.; et al. Brain-specific glycosylation of protein tyrosine phosphatase receptor type Z (PTPRZ) marks a demyelination-associated astrocyte subtype. J. Neurochem 2023. epub ahead of print. [Google Scholar] [CrossRef]
- Ye, H.; Hernandez, M.R. Heterogeneity of astrocytes in human optic nerve head. J. Comp. Neurol. 1995, 362, 441–452. [Google Scholar] [CrossRef]
- Ishizuka, S.; Jin, Z.W.; Yamamoto, M.; Murakami, G.; Takayama, T.; Hayashi, K.; Abe, S.I. CD57 (Leu-7, HNK-1) immunoreactivity seen in thin arteries in the human fetal lung. Anat. Cell Biol. 2018, 51, 105–112. [Google Scholar] [CrossRef]
- Chou, H.C.; Chen, C.M. Effects of uteroplacental insufficiency on cardiac development in growth-restricted newborn rats. J. Dev. Orig. Health Dis. 2023, 14, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Tagawa, H.; Kizuka, Y.; Ikeda, T.; Itoh, S.; Kawasaki, N.; Kurihara, H.; Onozato, M.L.; Tojo, A.; Sakai, T.; Kawasaki, T.; et al. A non-sulfated form of the HNK-1 carbohydrate is expressed in mouse kidney. J. Biol. Chem. 2005, 280, 23876–23883. [Google Scholar] [CrossRef] [PubMed]
- Radovani, B.; Gudelj, I. N-Glycosylation and Inflammation; the Not-So-Sweet Relation. Front. Immunol. 2022, 13, 893365. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hours, C.M.; Gil, S.; Gressens, P. Molecular and Cellular Insights: A Focus on Glycans and the HNK1 Epitope in Autism Spectrum Disorder. Int. J. Mol. Sci. 2023, 24, 15139. https://doi.org/10.3390/ijms242015139
Hours CM, Gil S, Gressens P. Molecular and Cellular Insights: A Focus on Glycans and the HNK1 Epitope in Autism Spectrum Disorder. International Journal of Molecular Sciences. 2023; 24(20):15139. https://doi.org/10.3390/ijms242015139
Chicago/Turabian StyleHours, Camille M., Sophie Gil, and Pierre Gressens. 2023. "Molecular and Cellular Insights: A Focus on Glycans and the HNK1 Epitope in Autism Spectrum Disorder" International Journal of Molecular Sciences 24, no. 20: 15139. https://doi.org/10.3390/ijms242015139





