Abstract
To aid the possible prevention of multidrug resistance in tumors and cause lower toxicity, a set of sixteen novel dihydropyridine carboxylic acids derivatives 3a–p were produced; thus, the activation of various ynones with triflic anhydride was performed, involving a nucleophilic addition of several bis(trimethylsilyl) ketene acetals, achieving good yields requiring easy workup. The target molecules were unequivocally characterized by common spectroscopic methods. In addition, two of the tested compounds (3a, and 3b) were selected to perform in silico studies due to the highest cytotoxic activity towards the HCT-15 cell line (7.94 ± 1.6 μM and 9.24 ± 0.9 μM, respectively). Employing theoretical calculations with density functional theory (DFT) using the B3LYP/6-311++G(d,p) showed that the molecular parameters correlate adequately with the experimental results. In contrast, predictions employing Osiris Property Explorer showed that compounds 3a and 3b present physicochemical characteristics that would likely make it an orally active drug. Moreover, the performance of Docking studies with proteins related to the apoptosis pathway allowed a proposal of which compounds could interact with PARP-1 protein. Pondering the obtained results (synthesis, in silico, and cytotoxic activity) of the target compounds, they can be judged as suitable antineoplastic agent candidates.
1. Introduction
The group of α,β-acetylenic ketones, more commonly known as ynones, are molecules of great interest due to their importance as building blocks in the synthesis of many heterocycles; several of them are secondary metabolites, bearing biological activities and pharmaceutical applications [1,2,3,4,5,6]. This kind of compounds is highly reactive due to the presence of a carbonyl and an alkyne group [7,8]; it should also be noted that they are highly reactive Michael systems [9].
The pyridine nucleus is a versatile building block in organic synthesis [10,11] that is susceptible to activation by different agents; for example, it reacts with triflic anhydride (Tf2O) [12]. Furthermore, it is important to note that bis(trimethylsilyl) ketene acetals are nucleophiles that react with pyridinium salts supplying functionalized dihydropyridines [13,14,15]. Moreover, many pyridine derivatives such as 1,4-dihydropyridines are of special interest in drug design since they display interesting applications, for example: antibacterial [16], antifungal [17], anti-HIV [18], antihypertensive [19], and anticancer activities, among others, highlighting that any modification within the structure of dihydropyridine can be associated with excellent medicinal effects, decrease tumor growth, cell migration, and principally to avoid resistance of cancer cells against anticancer drugs [20]. As a part of our research interest, some members of our group have appropriately employed bis(trimethylsilyl) ketene acetals in combination with aza-aromatic substrates activated with triflic anhydride as an electrophilic activating agent to produce dihydropyridine carboxylic acids [21].
Cancer is a serious health problem, placed as the second leading cause of death worldwide. It is most manifested in the prostate, lungs, bronchi, uterine corpus, breasts, colon, liver, stomach, and blood [22]. The most frequently used cancer treatments are surgery, radiation, chemotherapy, hormonal therapy, and targeted therapy. However, many treatments are expensive, and in the treatment of malignant tumors, neoplastic cells can generate multidrug resistance (MDR) with different structures and functions. Additionally, current anticancer drugs are associated with several severe side effects [23]. Also, in anticancer drugs based on metal, the metal can bind to sites that should be occupied by other metals [24]. Consequently, the development of new molecules that avoid MDR in tumors, have a better pharmacological profile, and have lower toxicity for healthy cells is necessary. It is known that 1,4-dihydropyrine derivatives overcome MDR [25]. However, their clinical use is limited due to their strong vasodilator activity, and new chemotherapeutics that do not present this drawback are needed to overcome MDR in cancer treatment [26].
On the other hand, DFT calculations are favored nowadays for replications of experimental facts such as molecular geometry, vibrational frequencies, and chemical shifts, including other properties of organic molecules [27]. In this sense, calculated frequencies and chemical shifts are scaled to compensate for the estimated handling of electron correlation, basis set deficiencies, and anharmonicity [28,29], creating good approximations for organic molecules [27,30].
Considering the commentary above, the goal of this work involves the use of several bis(trimethylsilyl) ketene acetals to produce sixteen novel carboxylic acid derivatives and their corresponding cytotoxic evaluation in several cancer cell lines (U251, PC-3, K-562, HCT-15, MCF-7, and SKLU-1) and normal cell (COS7). Complementary, physicochemical, and toxicological predictions; in silico studies (docking) to predict the affinity to proteins related to apoptosis induction; and quantum chemistry for the determination of molecular properties were performed.
2. Results and Discussion
2.1. Synthesis
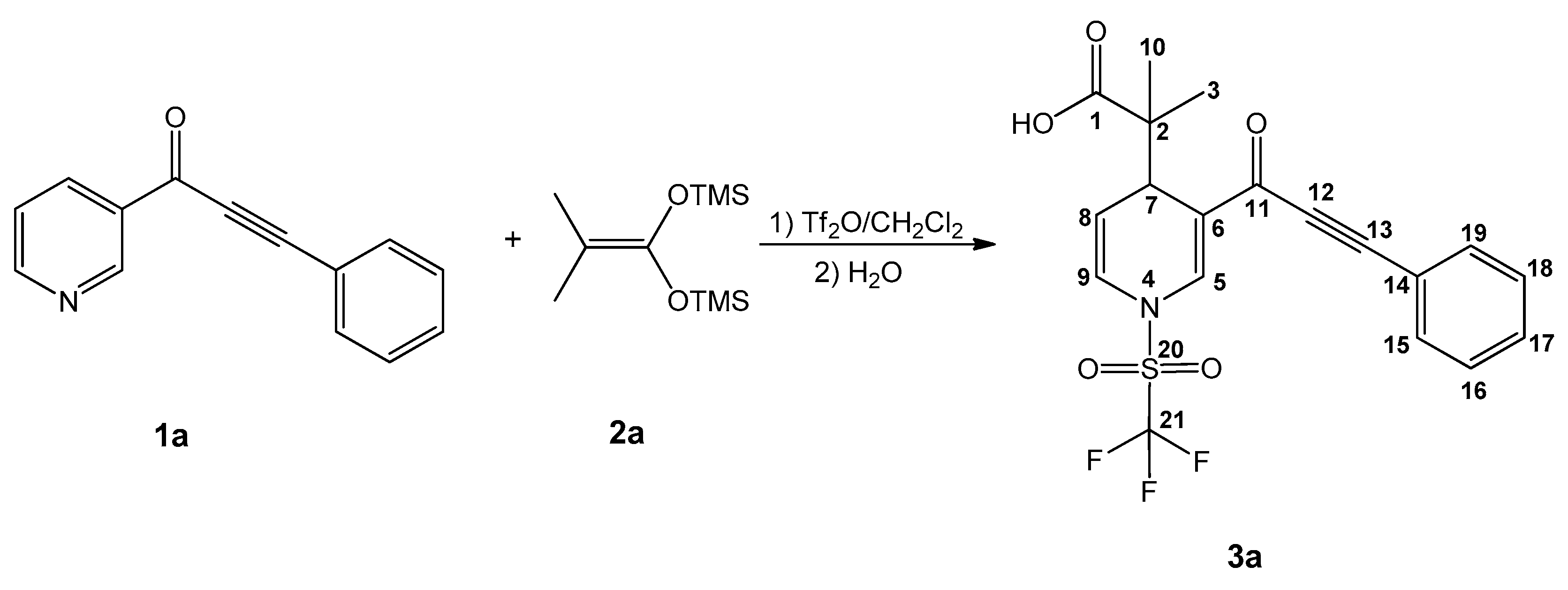
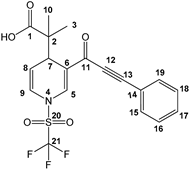
Dihydropyridine carboxylic acid 3a was produced (Scheme 1) by fine-tuning a procedure from previous research [31]: the ynone 1a was activated with a slight excess (1.2 equiv) of triflic anhydride for 3h, followed by a nucleophilic addition (at C6) of 1.2 equiv of the bis(trimethylsilyl) ketene acetal (2a) in 15 mL of anhydrous CH2Cl2 (under an inert atmosphere at −78 °C), and stirring continued at −78 °C for another 8 h. To generalize the procedure (Table 1), a set of sixteen (3a–p) new molecules were synthesized, using different ynones as substrates, in the presence of several bis(trimethylsilyl) ketene acetals. In general, the reaction yields ranged from regular to good, even when comparing the reaction with imidazoles as substrates as observed in previous research [31]. The procedure resulted in easy workup, achieving sixteen new products; it will be demonstrated that several of the products are good antineoplasic candidates.
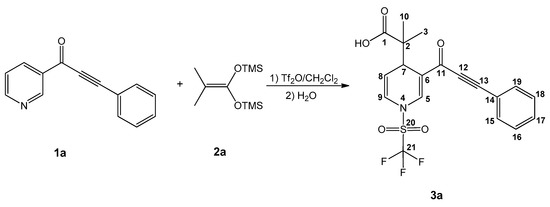
Scheme 1.
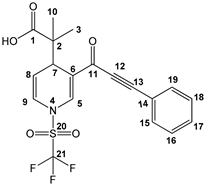
Production and assigned numeration of the dihydropyridine carboxylic acid 3a.

Table 1.
Synthesized dihydropyridine carboxylic acids 3a–p 1.
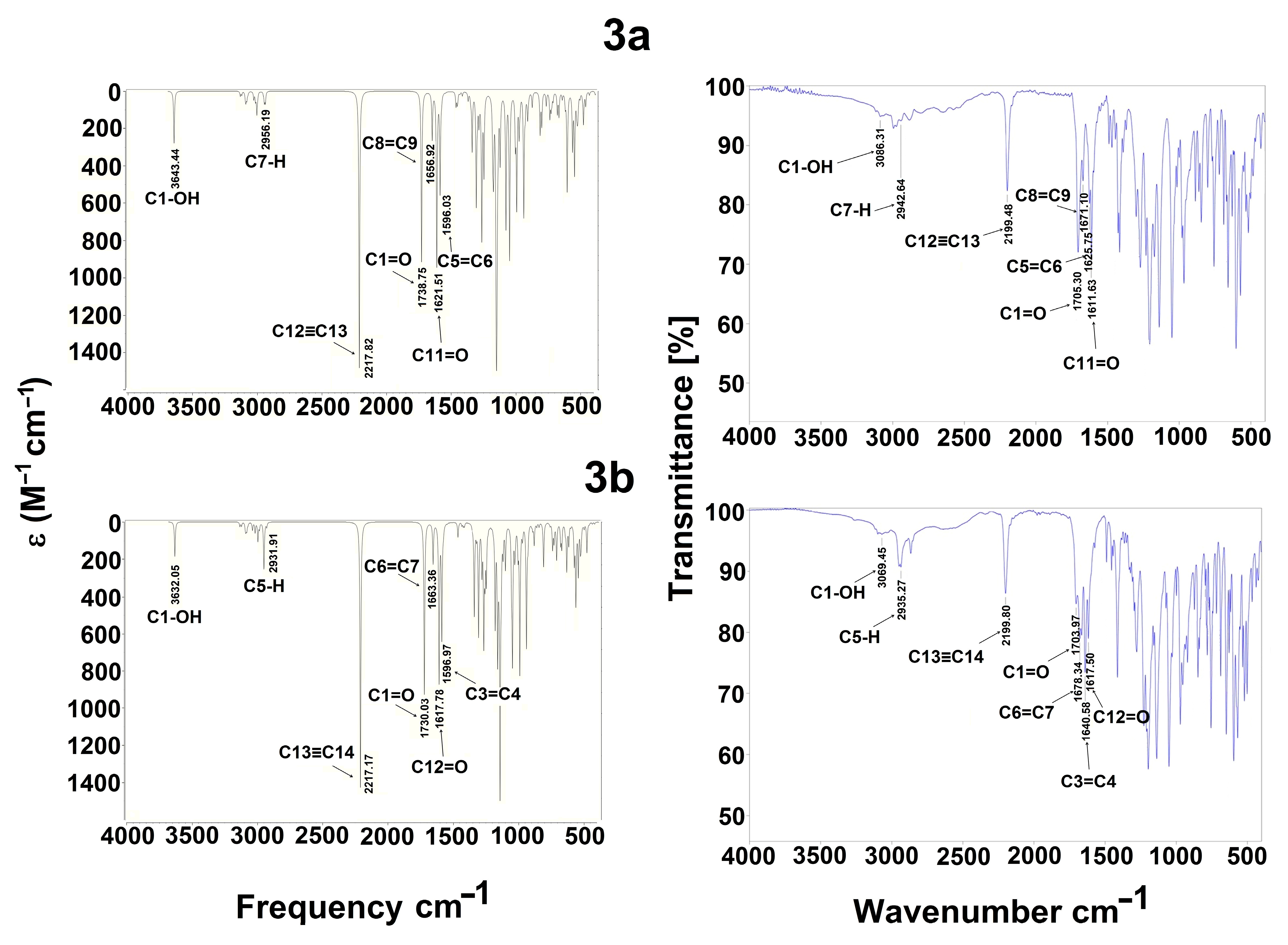
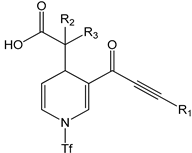
For the structural attribution of the obtained molecules, the target 3a is offered as an example: it is a white solid and obtained with a 91% yield, displaying an mp of 122–124 °C; implying a pure compound, the infrared spectrophotometric data (cm−1) showed a broad band centered at 3086 cm−1, which was assigned to the OH of the carboxylic group; a sharp medium band was observed at 2199 cm−1, which unequivocally belongs to the C≡C system; as expected, two intense bands were present at 1705 and 1625 cm−1, which were assignable to the C=O group and to the system C=C, respectively. The corresponding 1H NMR spectrum displayed a broad singlet at 11.28 ppm, which was unequivocally assigned to the proton of the carboxylic moiety; a simple signal at 8.18 ppm was assigned to H5, denoting a displacement to high frequency due to the electron-withdrawing influence of the α,β-unsaturated ketone system; at 6.77, 5.55, and 4.16 ppm, a double, a double of double and another double signals were presented. These signals are assigned, respectively, to H9, H8, and H7, which is in agreement with the corresponding coupling data (J9–8 = 5.4 Hz, J8–7 = 1.8 Hz); a double and multiple signals were observed in the range of 7.63–7.45 ppm, which is conveniently assigned to the phenyl group. Related to the 13C NMR spectrum, the main signals that agree appropriately with the 3a structure were assigned to the carbonyl groups at 182.3 and 176.1 ppm of the carboxylic moiety and ynone, respectively. Likewise, the carbons corresponding to the triple bond are highlighted at 92.5 and 84.6 ppm. Associated to the HRMS, acquired by the DART+ mode (19 eV), an elemental composition C19H17F3NO5S was stated, in agreement with a fragment ion [M+1]+; this fragment was unequivocally correlated (error of −1.37 ppm) with an exact value of 428.0779 Daltons and a precise value of 428.0773 Daltons, complementarily the provided unsaturation data (11.5) was in agreement with the structure. Additionally, it is important to comment that the spectroscopic spectra for all compounds, Figures S1–S64, is offered in Supplementary Materials.
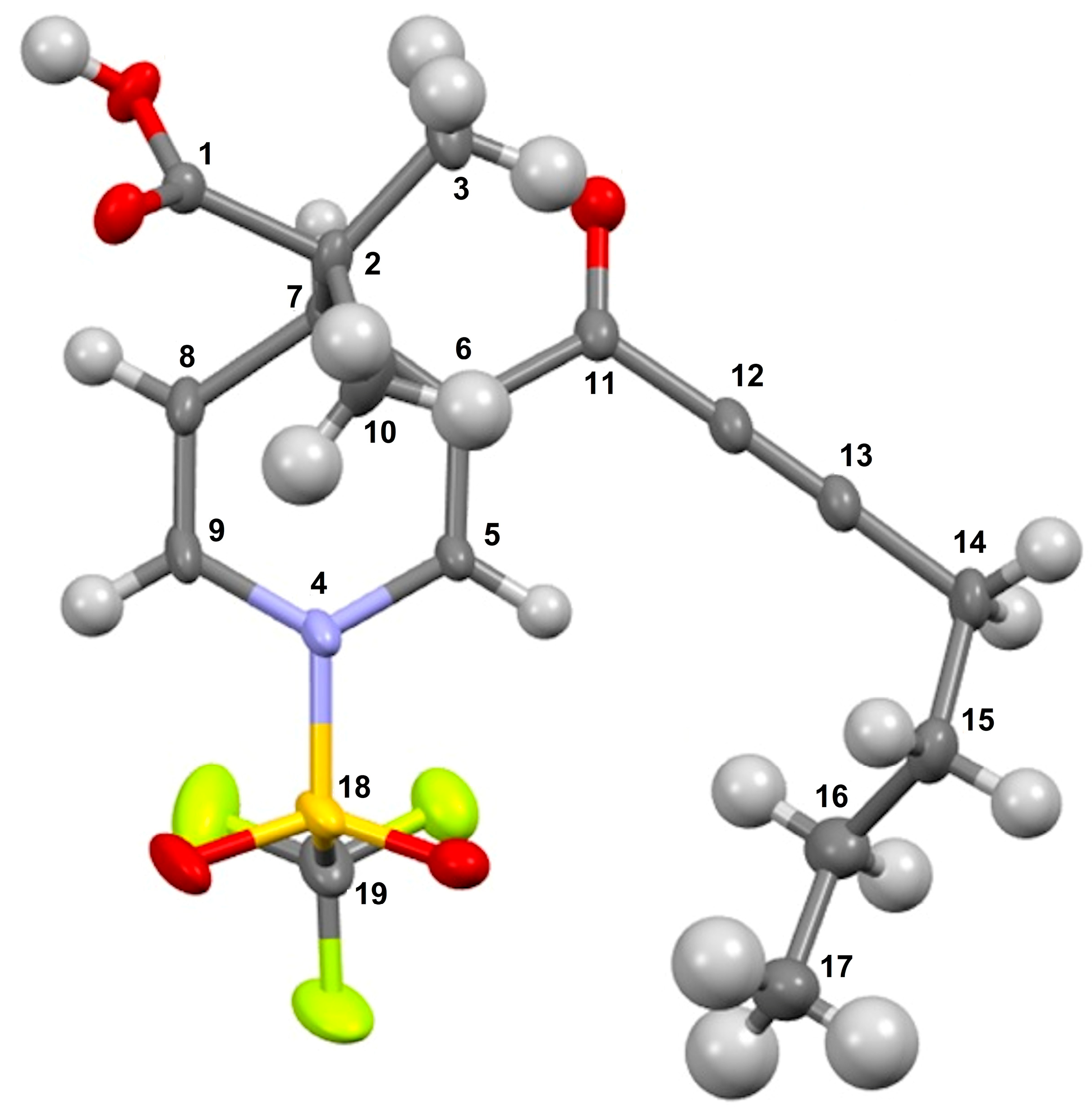
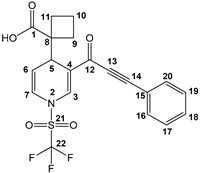
It is convenient to highlight that the molecular structure of the title molecules was unequivocally confirmed, correlated through an X-ray diffraction analysis; molecule 3f is shown as an example in Figure 1.
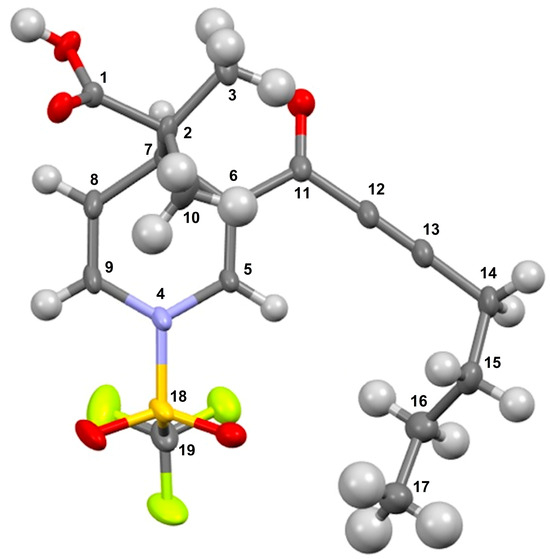
Figure 1.
ORTEP view and assigned numeration of compound 3f. Thermal ellipsoids are shown at the 50% probability level.
In this sense, suitable X-ray quality crystals of 3f were grown by slow evaporation from an n-hexane/CH2Cl2 solution at 4 °C. Details of the data collected and the structure refinement parameters employed are summarized in Table 2. The dihydropyridine carboxylic 3f crystallizes in monoclinic (space group P21/c) with the cell measurements of a = 5.8706(1) Å, b = 15.1627(2) Å, c = 20.9890(3) Å, and α = 90°, β = 93.576°, and γ = 90°, with volume of 1864.68 Å3 and Z of 4 at 25 °C. The complete refined crystal structure specification using SHELXS-2013 is summarized in Table 2.

Table 2.
Crystal data and structure refinement for 3f.
2.2. Cytotoxic Assay
The compounds were tested in vitro for cytotoxic activity (Table 3) and were determined by using the protein-binding dye at 25 μM for 3a–p in six tumor cells: U251 (human glioblastoma), PC-3 (human prostatic adenocarcinoma), K-562 (human chronic myelogenous leukemia), HCT- 15 (human colorectal adenocarcinoma), MCF-7 (human mammary adenocarcinoma), and SKLU-1 (human lung adenocarcinoma) were determined by using the protein-binding dye sulforhodamine B (SRB) assay in microculture to determine cell growth [32].

Table 3.
Inhibition of the growth (%) of human tumor cell lines for 3a–p at 25 μM in DMSO.
The initial cytotoxicity screening data listed in Table 3 show moderate inhibition to tumor cell lines. However, only nine compounds (3a, 3d, 3e, 3i, 3j, 3m, 3n, 3o, and 3p) showed an inhibition percentage greater than 50% in the HCT-15 cell line; however, it is important to mention that several of these compounds display cytotoxic effect against normal cells (COS7); consequently, these compounds were discarded for further studies. Also, it is important to mention that the compound 3b was used to further studies due to displaying an inhibition of 42.7%, but does not show cytotoxicity against COS7. Finally, we determined the IC50 of the most active compounds over HCT-15 (Table 4).

Table 4.
Cytotoxic activity of dihydropyridine carboxylic acids against HCT-15.
Analyzing these results, we observe that only the compounds 3a and 3b display the lower IC50 values in comparison with both references Cisplatin and Gefitinib, which indicates their good activity against the evaluated tumor cell line.
2.3. In Silico Evaluation of Physicochemical Properties
According to the above results, the physicochemical characteristics of compound 3a and 3b were analyzed by employing the Osiris Property Explorer. As can be seen in Table 5, both compounds show physicochemical properties that are in accordance with Lipinski’s rule of five (molecular weight < 500 g/mol, Log p < 5, hydrogen bond donator < 5, and hydrogen bond acceptor < 10). This rule describes molecular properties important for a drug’s pharmacokinetics in the human body, including absorption, distribution, and excretion (named ADME), as these physicochemical properties may represent the difference between failure and the development of a successful oral drug [33].

Table 5.
Computational analysis of physicochemical characteristics according to Lipinski’s rule of compound 3a and 3b.
In addition, the topological polar surface area (TPSA), which is defined as the surface total over all polar atoms, including oxygen, nitrogen, etc., and their bonding hydrogen atoms, were calculated for compounds 3a and 3b. TPSA is a commonly used medicinal chemistry metric for the optimization of a drug’s ability to permeate cells [34]. Molecules with a TPSA value higher than 140 angstroms squared (Å2) tend to be poor at permeating cell membranes. As shown in Table 5, the TPSA values for studied compounds are lower than 140 Å2.
The other biological properties evaluated were mutagenicity, teratogenicity, irritant and reproductive effects to computational assessment of studied compounds. Only compound 3a showed low risk for reproductive effects, while compound 3b was shown to be safe according to the computational evaluation. Therefore, all estimates indicate that there is a high possibility that the designed molecules should be able to pass through lipid membranes when they are experimentally evaluated in animal or human models [35].
2.4. Docking Studies
Docking studies represent a computational tool that are highly employed to propose appropriately, at an atomic level, interesting protein–ligand interactions [36]. It is worth noting that numerous docking studies with proteins involved in apoptosis pathway have been described elsewhere [37]. The averages of ΔG values obtained by docking studies with proteins related to apoptosis cascade and cell cycle regulators are summarized in Table 6. As it can be seen, for proteins related to apoptosis regulation, compounds 3a and 3b exhibit more affinity to PARP-1 protein than the rest of the studied proteins, evidenced by the lowest values of ΔG (−9.8 and −10.6 kcal/mol, respectively). The binding mode and the interactions established with PARP-1 in the complex with the lowest affinity and the highest frequency are shown in Figure 2 and described below.

Table 6.
ΔG values obtained by docking studies between 3a, 3b, and proteins related to apoptosis.
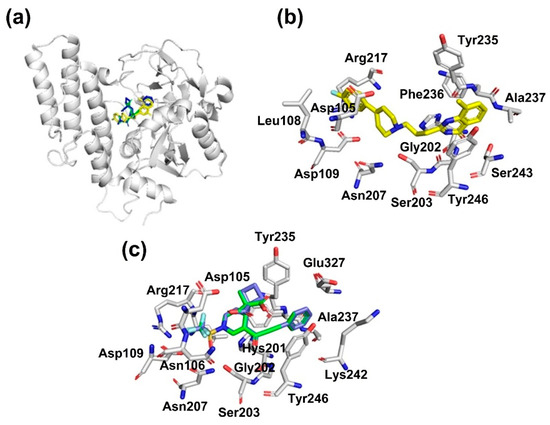
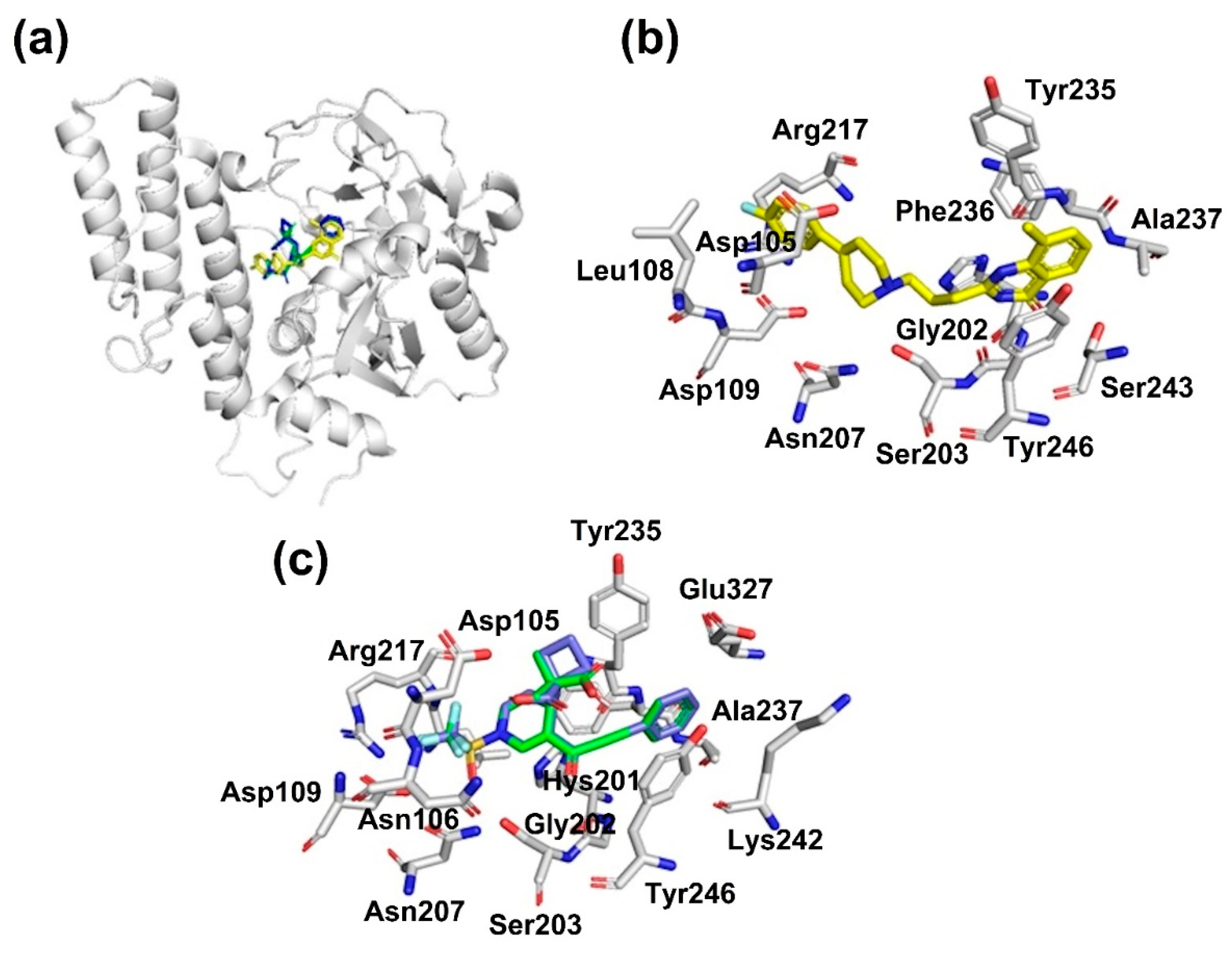
Figure 2.
Binding mode of compounds 3a and 3b with PARP-1, obtained by docking studies. (a) Binding mode of PARP-1 inhibitor reference (FR257517, yellow color), 3a (green color), and 3b (blue color). (b) Amino acid residues which drive the interaction between PARP-1 and know inhibitor (FR257517). (c) Amino residues which drive the interaction between PARP-1 and compounds 3a (green color), and 3b (blue color).
PARP-1 is an enzyme which plays a pivotal role in cellular biological processes, including DNA repair. Interestingly, PARP-1 has been found to be overexpressed in various carcinomas, including colorectal cancer [39]. These findings highlight the clinical potential of PARP-1 inhibitors as a therapeutic target of human cancer. Additionally, multiple preclinical trials demonstrate that the inhibition of PARP-1 can repress tumor growth and metastasis [40]. As shown in Figure 2a, compounds 3a and 3b bind to the catalytic site of PARP-1. Figure 2b shows the interaction of a known inhibitor (FR257517) with PARP-1 by stablishing three hydrogen bonds with Ser243, Ser203, and Gly202; a π–π interaction with Tyr246; in addition to a set of van der Waals contacts with Arg217, Leu108, Asp109, Asp105, Asn207, Gly202, Phe236, Ala 237, and Tyr235, as was reported previously [38]. Likewise, the analysis of the conformation with the lowest ΔG and the highest frequency obtained by docking studies showed that the studied compound interacts with PARP-1 protein through a π–π interaction with Tyr246; and through hydrogen bonds with Asp105, Arg217, Asp109, and Ser203; in addition to a set of van der Waals contacts with Asn106, Asn207, Tyr235, Hys201, Gly202, Glu327, Ala237, and Lys242, Figure 2c. In general, the high affinity of studied compounds to PARP-1 could be explained by two important characteristics: a small pocket, where compounds 3a and 3b can fit during the interaction; and the catalytic site of the protein is formed by polar amino acids allowing it to target compounds to establish hydrogen bond interactions that stabilize the binding mode. Thus, the interaction of compounds 3a and 3b could explain their cytotoxic effect in the HTC-15 cell line.
2.5. Quantum Chemistry Studies
Taking into account the aforementioned comments, the molecular properties and the experimental characterization of 3a and 3b were contrasted with their corresponding theoretical data obtained from DFT calculations, using the B3LYP with 6-311++G(d,p) basis set. The theoretical calculations were related to the chemical shifts, in ppm, with nuclear magnetic resonance (NMR); for the vibrational modes, they were related, in cm−1, with infrared spectrophotometry; these analyses were performed for the most stable conformer of 3a and 3b molecules in the gas phase.
2.5.1. Geometrical Parameters for 3a and 3b
The most stable structures of 3a and 3b, Figure 3, were computed with optimized energies of −116.867 × 103 kcal/mol and −119.257 × 103 kcal/mol. Selected geometric parameters (bond lengths, bond angles, dihedral angles) of compounds 3a and 3b are shown for comparative analysis with XRD-3f values in Table 7. Most of the experimental values fit with DFT data calculated in the gas phase while XRD-3f is in a solid phase. The sp3, sp2, and sp carbon–carbon bond lengths in 3a and 3b range from 1.198 to 1.573 Å (XRD-3f) and from 1.211 to 1.597 Å (B3LYP). The carbon–oxygen bond lengths for 3a and 3b range from 1.218 to 1.317 Å (XRD-3f) and from 1.207 to 1.355 Å (B3LYP). Some lengths showed variations between 0.003 and 0.119 Å, for molecules 3a and 3b with respect to XRD-3f [41]. The C2–C7 and C1–C2 bond lengths of the 3a molecule showed deviations of 0.021 Å and 0.012 Å with respect to 3b, respectively. Moreover, the bond angles of C7–C2–C1 of 3b showed a slightly increased deviation, 2.3°, with respect to 3a. Some bond angles showed variations between 0.01 and 4.9° with respect to XRD-3f. The dihedral angles in Table 5 for 3a and 3b are similar; the variation between them is 0.5 and 6.6°. The type of substituent at N3 does not contribute to the planarity of a certain part of the molecules 3a and 3b. Therefore, the C5–C6–C7–C2, C9–C8–C7–C2, and C11–C6–C7–C2 dihedral angles showed some differences between them, 5.1, 5.9, and 6.6°, respectively.
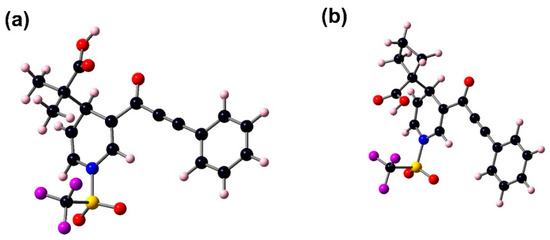
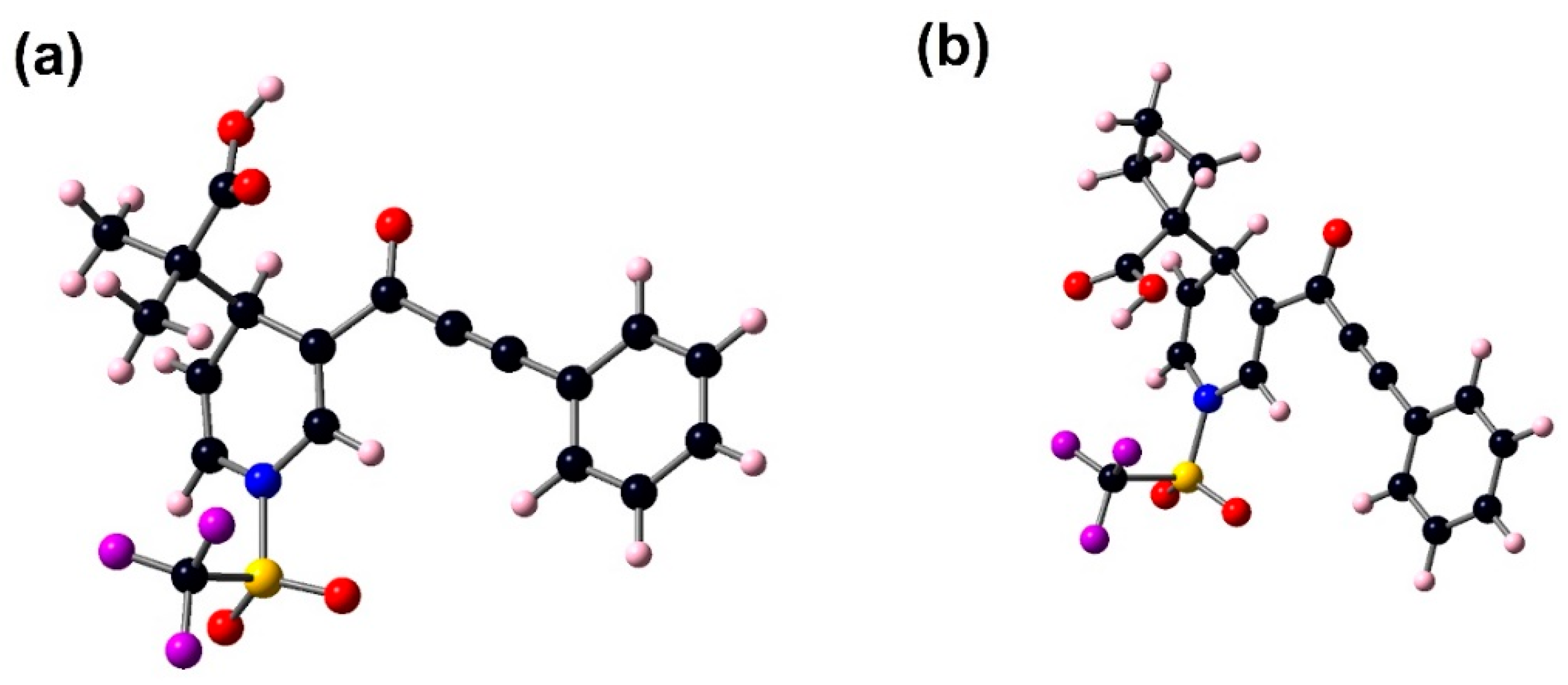
Figure 3.
Optimized structures of 3a (a) and 3b (b) at the B3LYP/6–311+G(d,p) in gas phase.

Table 7.
The selected geometric parameters of compounds 3a and 3b.
2.5.2. Frontier Molecular Orbital (FMO)-Reactivity Parameters of 3a and 3b
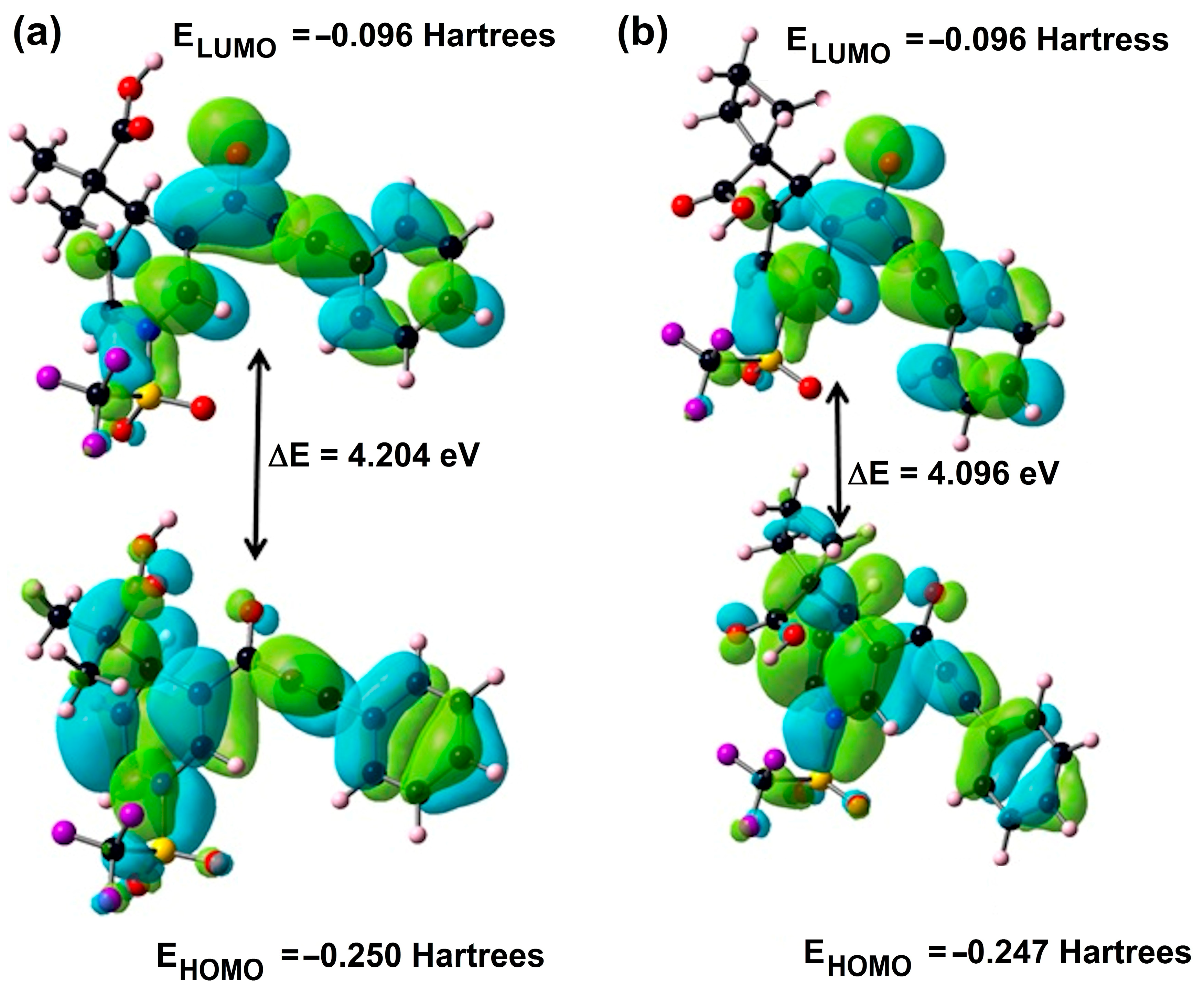
The electron that donates the outermost molecular orbital packed with electrons is identified as the highest occupied molecular orbital (HOMO), while the electron that accepts the lowest empty molecular orbital is the lowest unoccupied molecular orbital (LUMO). The DFT approach reveals that molecules 3a and 3b have 752 and 774 molecular orbitals including 110 and 113 orbitals filled by electrons and 642 and 661 empty orbitals, respectively. In the present computational study of the title of compounds 3a and 3b by the DFT method, the representation of molecular orbitals HOMO (orbital number: 110 and 113) and LUMO (orbital number: 111 and 114) with their corresponding energies (in eV) are illustrated in Figure 4. The energies of HOMO and LUMO, for 3a and 3b, are −6.806 eV and −6.718, and −2.602 and −2–622 eV, respectively.
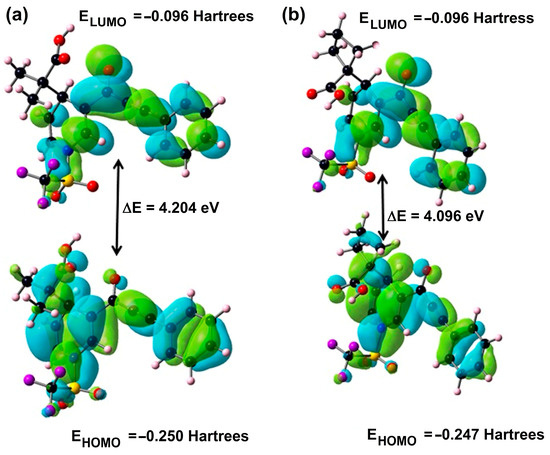
Figure 4.
HOMO and LUMO of 3a and 3b.
The difference in energy between HOMO and LUMO (frontier molecular orbital analysis) can be employed to predict the bioactive potency (reactivity) and chemical stability of the molecule [42,43]. In this sense, quantum molecular reactivity parameters like electronegativity (χ), chemical potential (μ), electrophilicity index (ω), global hardness (η), and charge transfer capability (Δ) were calculated by HOMO and LUMO energies [44] for compounds 3a and 3b, Table 8.

Table 8.
Chemical parameters, indicative of the reactive behavior of compounds 3a and 3b.
Accordingly, the ionization energy of the compounds changed as 3a (8.121 eV) > 3b (8.053 eV), and the electron affinity changed as 3b (1.217 eV) > 3a (1.190 eV) in the gas phase. As far as the energy gap values, it can be observed that intermolecular interactions for 3b are more likely than in 3a because the ΔELUMO-HOMO for the compounds is calculated as 3a (4.204 eV) > 3b (4.096 eV); it can be said that compound 3a is harder (3.466 eV) than compound 3b for the gas phase. In this study, the electrophilicity index values of the compounds support the relative acidic characterization. In Table 6, compound 3b (3.142 eV) has a more electrophilic character than compound 3a (3.127 eV). Furthermore, compound 3b (0.678 eV) has a slightly higher capability of charge transfer than compound 3a (0.672 eV).
It is well known [45,46,47] that the ΔELUMO-HOMO has been normally used to provide evidence on the kinetic stability and reactivity of related molecular systems. Thus, the nucleophilicity of compound 3a is greater than compound 3b, which can be the reason for the anticancer activity of compound 3a. The highest value of chemical potential occurs in 3a (−4.656 eV). This property may be related to the nucleophilic character of the molecule being the most reactive. The highest value of electronegativity is for 3a (4.656 eV), which indicates that 3a is capable of attracting electrons to itself.
Occupied orbitals (HOMO) of 3a and 3b have contributions of the oxygen of hydroxyl and carbonyl groups, triple bond and phenyl ring. These moieties could be considered to be nucleophilic sites that could account for the antiproliferative activity of 3a and 3b against the studied human cancer cells. The carbon of carbonyl group and phenyl ring contributed to virtual frontier molecular orbitals, pointing to them as potential electrophilic sites for 3a and 3b and derivatives’ antiproliferative activity against cancer cells.
2.5.3. Molecular Electrostatic Potential (MEP) of 3a and 3b
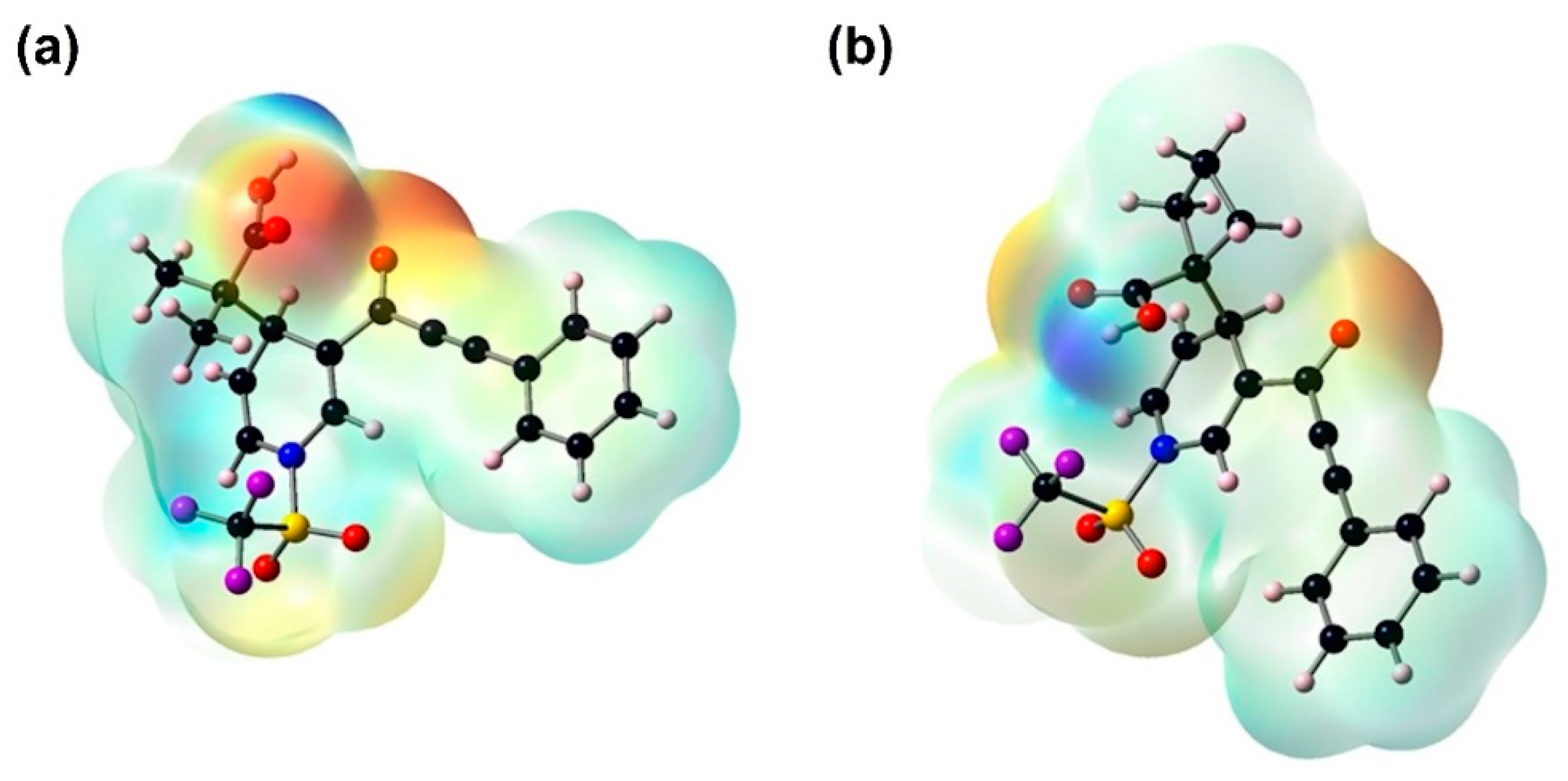
The MEP plots indicate the electrophilic and nucleophilic sites. The positive potential is definite with a blue color, which establishes the electrophilic attack center, while the negative potential is visualized in red, representing the nucleophilic attack center. In Figure 5, the MEP plot for 3a and 3b shows that the negative potential is mostly due to lone pairs of the oxygen of the carbonyl group and the hydroxyl oxygen; the strongest is that of the oxygen of 3b; its most negative value is −56.79 kcal/mol, and positive potential regions are mainly over the hydrogen atom belonging to the hydroxyl group.
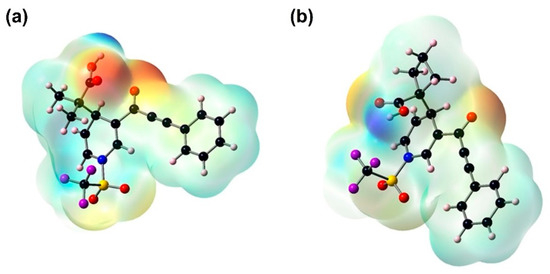
Figure 5.
Molecular electrostatic potential surfaces plotted on the van-der Waals’ surface of the molecule calculated with B3LYP/6-311++G(d,p) and a positive elemental charge in vacuum as the probe. Positive regions are shown in blue (a) 3a = +50.08 kcal/mol, and (b) 3b = +56.79 kcal/mol. The negative regions are shown in red (a) 3a = −50.08 kcal/mol, and (b) 3b = −56.79 kcal/mol.
Accordingly, molecules 3a and 3b could interact with a receptor site directly, and this interaction stabilizes the oxygen atoms of the carbonyl group of these molecules. The large positive potential in the hydrogen atom may have a contributing effect toward the association of these compounds with the receptor site, as it is well known that positively charged hydroxyl hydrogen atoms have a high affinity for negative centers.
2.5.4. Atomic Charges for 3a and 3b
The atomic charges in a compound are a key feature in numerous applications of quantum mechanical calculations to evaluate various properties of molecules [48,49]. By analyzing charge distributions in molecules, details about donor and acceptor pair formation can be obtained, which involves charge transport inside the molecule. The full atomic charges for both compounds are given in Table 9. Here, the natural atomic charge calculations revealed that the positive charge was located on the electropositive H and S atoms as expected; the net atomic charges for the O atoms of compounds 3a and 3b were calculated as −0.574 to −0.870 e-. However, the net charge for the O atom of sulfate groups was predicted between −0.860 e- and −0.870 e-. The net charges of the H atoms in compounds 3a and 3b were calculated as +0.224 to +0.487e- and +0.223 e- to +0.480, respectively. Furthermore, the charge distribution over the triple bond, carbonyl group, and C14 phenyl moiety of both compounds showed a change due to the resonance effect. The sulfur atom exhibits a positive charge due to the bonds with the oxygen and nitrogen atoms. Also, the C21 atom through the bonds with the fluorine atoms.

Table 9.
NPA charges for some atoms for the 3a and 3b, in e-, obtained using B3LYP/6–311++G(d,p).
As seen from the MEP surfaces and the charge analysis, hydrogen in the hydroxyl group for 3a and 3b is acidic (Lewis’s acid); and the carbonyl in the ketonic and carboxylic groups is more electronegative (Lewis’s base).
Consequently, the spin density derived from NPA calculations can be used to explain the electronic distribution in the molecular systems. The most positive or most negative NPA charges are indicators of the distribution of the electronic density of atoms for 3a and 3b molecules; and the availability of attracting or donating electrons atoms are indicators for the formation of intermolecular interactions, hydrogen bonds, π–π interaction, and van der Waals contacts between these molecules and amino acids residue of protein.
2.5.5. Theoretical 1H and 13C NMR Spectra of 3a and 3b
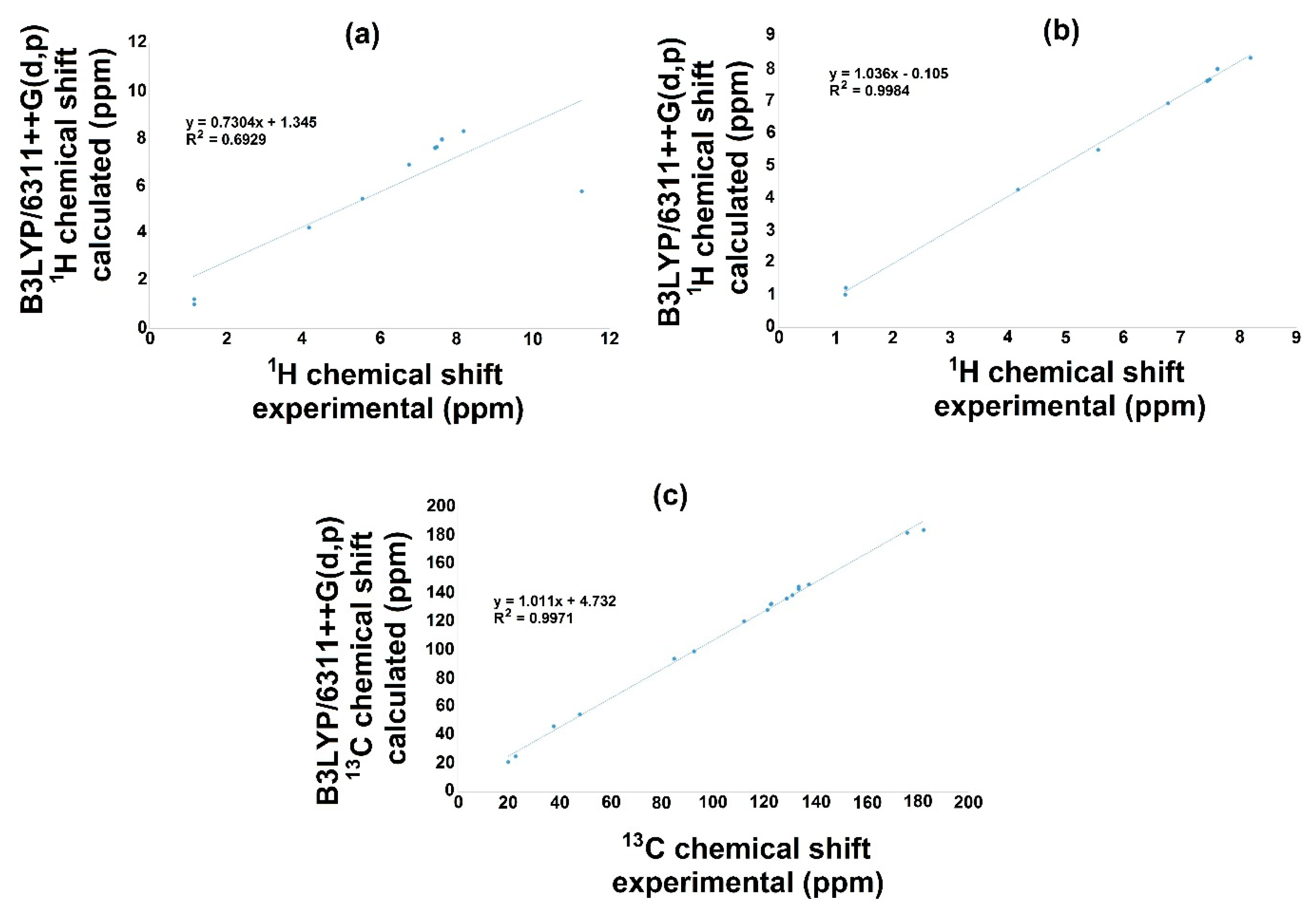
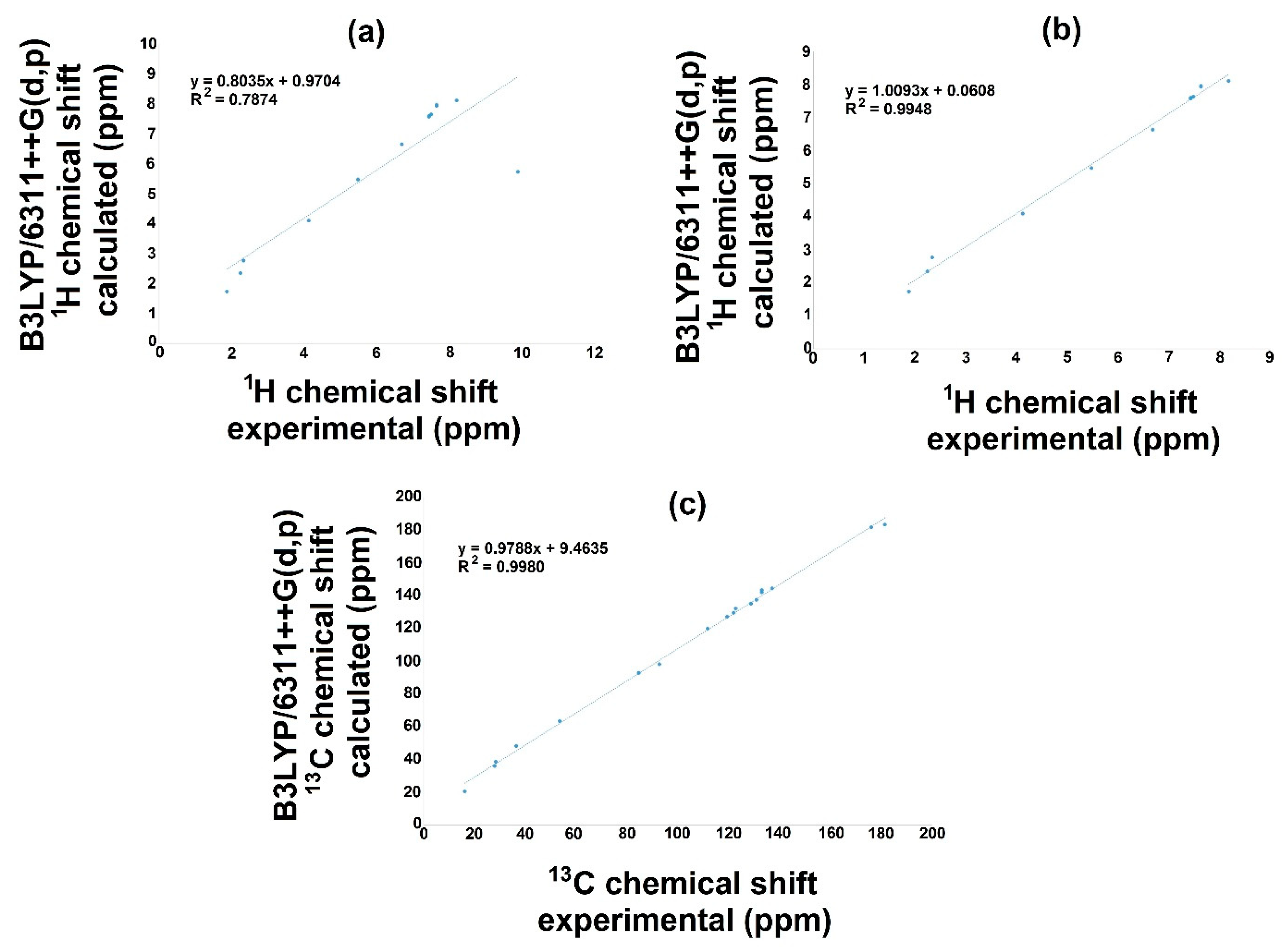
The NMR theoretical chemical shift for 1H and 13C in the gas phase for 3a and 3b were calculated using B3LYP with the 6-311++G(d,p) basis set and the GIAO method. Hence, Figure 6 and Figure 7 display the correlation employing linear regression, and Table 10 displays both 1H and 13C NMR theoretical and experimental chemical shift values. Related to the 1H NMR spectra for both molecules (3a, 3b), it is important to mention that the signal of OH influences the linearity, Figure 6a and Figure 7a. Therefore, the theoretical values for this signal show a chemical shift of 5.7737 ppm, and the experimental results presented a value of 11.28 ppm for 3a; furthermore, for 3b, the corresponding values were 5.7251 and 9.87 ppm for the theoretical and the experimental chemical shift, respectively. These differences may be explained considering that in the experimental spectrum, the OH group displays a common dissociation, producing an intramolecular hydrogen bond with the oxygen of the carbonylic group. This could not succeed in the gas phase. Thus, these hydrogen bonds could modify the electron density, consequently diminishing the regression coefficient value [50]. Additionally, the small discrepancy may be because the experimental NMR was developed in the liquid phase, and theoretical calculation was conducted in the gas phase [51,52].
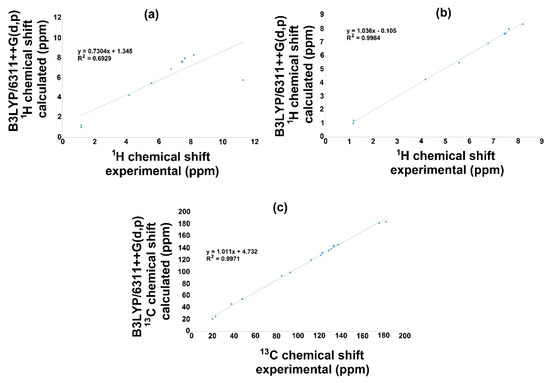
Figure 6.
1H and 13C NMR linear regression between experimental and calculated values with B3LYP/6311++G(d,p) for 3a.
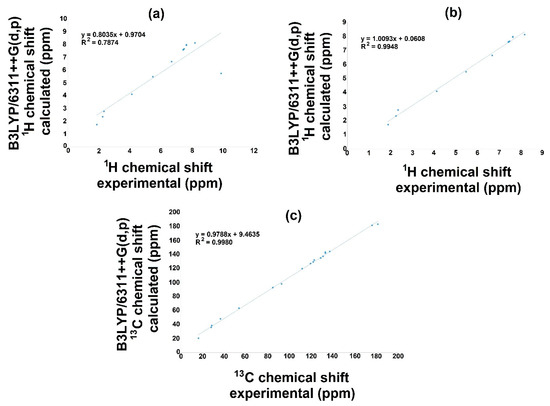
Figure 7.
1H and 13C NMR linear regression between experimental and calculated values with B3LYP/6311++G(d,p) for 3b.

Table 10.
1H and 13C NMR experimental and theoretical chemical shift (ppm) for 3a and 3b compounds.
On the other hand, removing this signal from the correlation, Figure 6b and Figure 7b, excellent regression coefficient values were obtained, R2 = 0.9984 with a corresponding standard deviation of 2.5803 and a typical error of 0.1135 ppm for 3a. In this sense, the equation to describe the fit is δCalc = 1.036δexperimental–0.105 ppm. The slope and intercept values are 1.04 and 0.11 ppm, respectively. For 3b, the value is R2 = 0.9948 with a standard deviation of 2.3787 and a typical error of 0.1847 ppm; the equation to describe the fit is δCalc = 1.0093δexperimental + 0.0608 ppm. The slope and intercept values are 1.00 and 0.06 ppm, respectively.
On behalf of the 13C NMR, Figure 6c and Figure 7c, the linear regression results show a regression coefficient of 0.9971 and 0.9980 with a standard deviations of 47.4734 and 48.2897, with typical errors of 2.66 and 2.19 ppm for 3a and 3b, respectively: the equation to describe the fit for 3a is δCalc = 1.011δexperimental + 4.732 ppm, with 1.01 and 4.73 ppm being the slope and intercept, respectively; for 3b, the equation is δCalc = 0.9788δexperimental + 9.4635 ppm, with a slope of 0.98 and an intercept of 9.46 ppm.
These results showed a good prediction of the chemical shifts, considering the regression value, indicating a good agreement between theoretical and experimental values.
2.5.6. Theoretical Infrared Spectra of 3a and 3b
The side chain on nitrogen atom does not have any important contribution to the dihydropyridine ring; therefore, this work was focused on the ring, the carbonylic groups, and the alkyne substituents. In this sense, the analyses are focused on specific vibrational modes: C5=C6, C8=C9, C7-H, C11=O, C1=O, C1-OH, and C12≡C13 for molecule 3a, and C3=C4, C6=C7, C5-H, C12=O, C1=O, C1-OH, and C13≡C14 for molecule 3b, Table 11. The 3a and 3b molecules have 45 and 46 atoms, exhibiting 129 and 132 normal active vibrational modes, respectively.

Table 11.
Selected stretching vibrational frequencies of 3a and 3b.
Experimental and Theoretical Vibrational Spectra
The experimental spectrum of 3a and 3b, Figure 8, showed a weak broad band at 3086 and 3069 cm−1 assigned to the OH of the carboxylic group, generally found as a dimer. These values agree with the reported range of 3300 to 2500 cm−1, usually centered at 3000 cm−1 [53]. Finally, the weak band is probably due to the intramolecular hydrogen bond with a carbonyl group (ketone) [54]. Additionally, the C=O stretching intense band appearing at 1705 and 1611 cm−1 and 1703 and 1640 cm−1 are assigned to the carboxylic acid and ketone groups for 3a and 3b, respectively, which is consistent with the literature value, 1870–1540 cm−1 [53,55]. The 2199 cm−1 value which appears corresponds to alkyne group as a medium band is congruent with the range previously reported, 2260–2100 cm−1 [53]. Regarding the 1,4-dihydropyridine system, two stretching medium bands were found for C=C groups at 1671 and 1625 cm−1 and 1678 and 1640 cm−1 for 3a and 3b, respectively. These results are consistent to those reported by Soliman et al. [56] at 1600 cm−1 for dipyrazolopyrimidine-1,4-dihydropyridines and Tu et al. [57] at 1652 to 1604 cm−1 for dihydropyridines. Finally, a weak stretching band at 2942.64 and 2935 cm−1 was observed for C7–H and C5–H bonds, respectively, and agrees with the literature data [53,58].
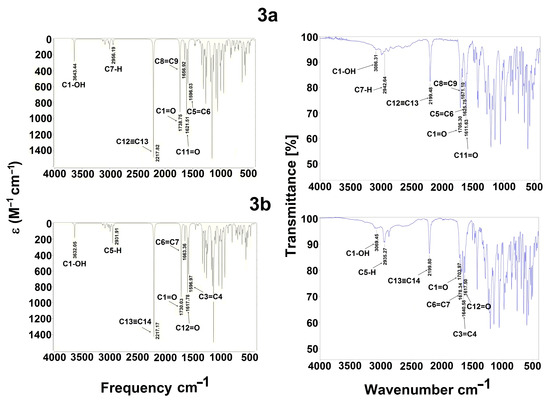
Figure 8.
The calculated vibration spectra of 3a and 3b, computed at B3LYP/6-311++G(d,p) (left) and experimental (right).
Regarding the theoretical level, Figure 8 and Table 11, it was observed that the OH for acidic moiety displays a weak band at 3643 and 3632 cm−1 for 3a and 3b, respectively; these bands probably recognize the OH of carboxylic acid as a free hydroxyl. These values are according to those reported by Devi et al. [59] at 3634 cm−1 using the B3LYP method with a 6-31G(d,p) basis set. Also, Devi et al. [60] reported the value for OH at 3545 cm−1.
The bands C7–H and C5–H at 2942 and 2935 cm−1 are presented as weak bands for 3a and 3b, respectively. In this case, Karthick et al. [61] detected values in the span of 2979–2978 cm−1 for 1,4-dihydropyridines, Kavitha and Verlaj [62] detected values between 3144 and 2900 cm−1 for this band for isoxazoles; Radder et al. [51] reported the value at 3029 cm−1 for a benzonitrile derivative; and Fekri and Nikpassand [54] reported values comprising 3077–2987 cm−1 for dihydropryridine derivatives.
Related to C=O bands, two carbonyl groups are presented: C=O for the carbonyl of the acid moiety, which is displayed at 1738 cm−1 for 3a and 1730 cm−1 for 3b as medium bands. In this sense, Devi et al. [59] detected this band at 1766 cm−1; Devi et al. [60] reported this band at 1729 cm−1. For C=O, the carbonyl of the ketone moiety is displayed as medium bands at 1621 cm−1 for 3a and 1617 cm−1 for 3b; this band is easily to identify due to higher strength and conjugation with the double bonds C5=C6 or C3=C4. Karthick et al. [61] report this band at 1697–1685 cm−1; Fekri and Nikpassand [54] report it at 1657–1643 cm−1 for 1,4-dihydropyridines and at 1652 cm−1 for a benzonitrile derivative [51].
At 1596 and 1656 cm−1 for 3a and 1596 and 1663 cm−1 for 3b, weak bands for C8=C9 and C6=C7 bonds were detected and medium bands for C5=C6 and C3=C4 bonds of the dihydropyridine ring were detected, respectively; probably, these bands are displayed as a medium band due to conjugation with the carbonyl of ketone group. Kavitha and Verlaj [62] detected theoretical values from 1601 to 1491 cm−1, obtained for isoxazoles; Radder et al. [51] also reported values ranging from 1652 to 1577 cm−1 for a benzonitrile derivative, and 1499–1643 cm−1 for dihydropryridine derivatives [54].
Finally, the alkyne group C12≡C13 and C13≡C14 is detected at 2217 cm−1 for both 3a and 3b as intense bands, agreeing with the previously reported values of 2370 cm−1 for propargyl arms containing a Schiff base [63] and 1800 cm−1 for complex between terminal alkynes with ruthenium [64].
These results demonstrate a good correlation between the experimental and theoretical data, correctly allowing for band assignments.
3. Materials and Methods
3.1. General
All reagents and solvents were of analytical grade, acquired from commercial suppliers, and used without further purification. Melting points were measured on a Melt Temp II apparatus and are uncorrected. The IR spectra were recorded on a Bruker TENSOR 27 spectrophotometer. The 1H and 13C NMR spectra were performed on a Bruker Advance III apparatus at 300 MHz and 75 MHz, respectively, in chloroform-d; the corresponding δ-chemical shifts are given in ppm, utilizing TMS as the internal reference. HRMS-DART+ (19 eV) spectra were obtained with a JEOL JMS-T100LC spectrometer.
Suitable crystals of 3f were grown in a n-hexane/CH2Cl2 system at 4 °C for more detailed scrutiny of its molecular structure with X-ray diffraction. The crystal was placed on a glass fiber at 25 °C and then put in a Bruker Smart Apex CC diffractometer equipped with Mo radiation (λMoKα 1⁄4 0.71073 Å). The decay was negligible in all cases. Systematic absences and intensity statistics were employed for space group determination. The structure was established with direct methods on the SHELXS-2013 program. The solution and refinement of structures were performed with SHELXL-2013. The refinements of the anisotropic structure were made with the least squares technique for all non-hydrogen atoms. The hydrogens were placed in idealized positions based on their hybridization with isotropic thermal parameters fixed at 1.2 times the value of the attached atom. The different acetals of bis(trimethyl) silyl ketene 2a–d were prepared according to methods reported in the literature [65]. The substrate ynones (1a–h) were prepared accordingly to a reported approach [66].
3.2. Ab Initio Calculations
Mechanic molecular [67,68,69] geometries of a conformational analysis previous, using the Spartan06 (Version 06, Wavefunction, Inc., Irvine CA, USA) [70] program, were used as initial models for optimizations for the density functional theory (DFT) [71,72,73]. The geometry of the title compound 3a and 3b was optimized by functional B3LYP [74,75] and 6-311++G(d,p) method [76,77,78] using Gaussian 16 (Version 2016.03, Gaussian, Inc., 340 Quinnipiac St., Bldg. 40, Wallingford CT 06492, UK) [79] software and visualized by GaussView [80] application. The geometries found by theoretical calculations were considered as true energy minima only when all calculated frequencies (PES) were positive. Theoretical vibrational analysis of 3a and 3b were executed with the same theory approach, and computed frequencies were assigned to respective modes of vibrations using a scale factor of 0.9679 [27,30,81,82]. Optimized geometries were also used to reproduce 1H and 13C NMR chemical shifts obtained using the Gauge-Independent Atomic Orbital (GIAO) approach [83,84] with a global scaling factor of 31.9699 and 184.1279 for 1H and 13C, respectively, with respect to TMS in gas phase. Atomic charge distribution analysis for 3a and 3b was carried out by Natural population analysis (NPA) [85], by Natural Bond Orbital (NBO) analysis [86] implemented in Gaussian 16 software. Moreover, a frontier molecular orbital (FMO) [87] determination and global reactivity descriptors [42,88] were found out using HOMO and LUMO energy values, and molecular electrostatic potential (MEP) [89] diagrams were drawn to estimate/evaluate the chemical reactive behavior/site of two compounds.
3.3. In Silico Evaluation of Physicochemical Properties
The OSIRIS property explorer (http://www.organic-chemistry.org, accessed on 3 August 2023) was employed to predict the physicochemical properties of the compounds according to Lipinski’s rules [33] to select ones with characteristics favoring absorption and distribution after oral administration.
3.4. Docking Studies
To determine the interaction between selected compounds with proteins related to apoptosis induction, the 3-dimensional (3D) structure of the following proteins was retrieved from protein data bank: B-cell CLL/lymphoma 2 (Bcl-2) PDB ID: 4IEH, B-cell lymphoma-extra-large (Bcl-XL) PDB ID: 4QVF, Mouse double minute 2 homolog (MDM2) PDB ID: 4HG7, Mammalian target of rapamycin (mTOR) PDB ID: 4JT5, Histone deacetylase 8 (HDAC-8) PDB ID: 5FCW, and Poly (ADP-ribose) polymerase 1 (PARP-1) PDB ID: 1UK0. The water molecules, co-crystalized ligands, and peptides were eliminated, conserving only the cofactors necessary to the enzymatic activity for each protein.
Autodock 4.2 software was used for docking studies (http://autodock.scripps.edu, accessed on 18 October 2023) [90]. This program has been widely used because it displays good free energy and binding pose correlation values between docking simulations and experimental data [91]. Three-dimensional structures of compound 3a and 3b were generated as previously described. In addition, to validate the docking protocol, known inhibitors for the studied proteins were included in the docking studies, Table 12. A GRID-based procedure was utilized to prepare the structural inputs and define all the binding sites. A rectangular lattice (70 × 70 × 70 Å) with points separated by 0.375 Å was centered on the active site of each protein, Table 12. All docking simulations were conducted using the hybrid Lamarckian genetic algorithm with an initial population of 100 randomly placed individuals and a maximum of 1.0 x 107 energy evaluations. All other parameters were maintained at their default settings. The resulting docked orientation within an RMSD of 0.5 Å was clustered together. The lowest energy cluster for each ligand was subjected to further free energy and binding geometry analysis, as previously reported [92]. Conformations with the lowest free energy binding (ΔG) and the highest frequency were selected employing AutoDock tools. The images were created by employing PyMol [93].

Table 12.
Regions for each protein selected and their respective know inhibitors employed to perform docking studies.
3.5. Cytotoxic Activity
The in vitro cytotoxicity of the compounds was evaluated on HCT-15 cell line (human colorectal adenocarcinoma), using the protein binding dye sulforhodamine B (SRB) in a microculture assay to measure cell growth [99]. The cell lines were grown in RPMI-1640 medium (Sigma Chemical Co., Ltd., St. Louis, MO, USA) supplemented with 10% fetal bovine serum (Invitrogen Corporation, Waltham, MA, USA), 2 mM L-glutamine, 10,000 units/mL of penicillin G, 10,000 μg/mL streptomycin, and 0.25 μg/mL of fungizone (Gibco, Amarillo, TX, USA). The cells were maintained at 37 °C in a 5% CO2 atmosphere with 95% humidity.
Preparations were made with 7.5104 cell/mL of the HCT-15 cell line, adding 100 mL of each of these suspensions to the wells of 96-well plates. Incubation was carried out for 24 h to promote cell attachment, and then, 100 μL per well of a test compound, gefitinib or cisplatin (as controls), was added. After 48 h, the adhered cell cultures were fixed in situ with a cold 50% (w/v) solution of trichloroacetic acid, followed by incubation at 4 °C for 1 h. Upon completion of this time, the supernatant was discarded, and the plates were washed and air dried. Cells cultured with TCA were stained with 100 μL of 0.4% SRB solution for 30 min. The protein-bound dye was extracted with a 10 nm unbuffered tris base, and the optical density was read at 515 nm on a Synergy HT Microplate Reader (Elx 808, BIO-TEK Instruments, Inc, Winooski, VT, USA). IC50 values were determined with the Monks protocol. Accordingly, a concentration–response curve was constructed for the dihydropyridine carboxylic acids 3a, 3b, 3f, 3i, 3o, 3p, and the values corresponding to inhibition of 50% (IC50) were calculated from non-linear regression equations. IC50 values are expressed as the mean ± standard error (SE) [100,101].
3.6. General Procedure for the Synthesis of Dihydropyridine Carboxylic Acids 3a–p
To a solution with 0.2 g (1.0 mmol) ynone derivative of pyridine dissolved in 15 mL of anhydrous CH2Cl2 (under an inert atmosphere at −78 °C), 0.19 mL (1.2 mmol) of triflic anhydride was added with a syringe, the mixture was stirred constantly during 3 h. Subsequently, 1.2 mmol of the corresponding ketene acetal was added and stirring continued at −78 °C for another 8 h. The reaction was then allowed to reach 25 °C before being transferred to a separatory funnel and washed with water (3 × 30 mL). The organic layers were combined and dried up with anhydrous Na2SO4; then, the solvent was removed by vacuum evaporation. Finally, the reaction crude was purified by column chromatography using silica gel 60 (0.063–0.200 mm, 70–230 mesh ASTM, acquired from Merck-millipore, Germany) with different n-hexane/ethyl acetate mixtures as eluents, obtaining the title pure compounds 3a–p.
Spectroscopical Characterization of Dihydropyridine Carboxylic Acids 3a–p
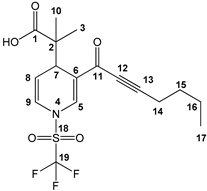
2-methyl-2-(3-(3-phenylpropioloyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-propanoic acid (3a), white solid was obtained with 91% isolated yield (0.37g); mp 122–124 °C; IR (cm−1): 3086 (COOH), 2199 (C≡C), 1705 (C=O), 1625 (C=C); 1H NMR (CDCl3) δ (ppm): 11.28 (s, 1H, OH), 8.20 (s, 1H, H5), 7.63 (d, J = 6.6 Hz, 2H, H15, H19), 7.49 (m, 1H, H17), 7.45 (d, J = 7.5 Hz, 2H, H16, H18), 6.77 (d, J = 5.4 Hz, 1H, H9), 5.55 (dd, J = 1.8 Hz, J = 5.4 Hz, 1H, H8), 4.16 (d, J = 5.4 Hz, 1H, H7), 1.17 (s, 3H, H10), 1.16 (s, 3H, H3); 13C NMR (CDCl3) δ (ppm): 182.4 (C1, C=O), 176.1 (C11, C=O), 137.5 (C5), 133.5 (C15, C19), 131.0 (C17), 128.8 (C16, C18), 122.7 (C9), 122.9 (C6), 121.3 (C14), 119.1 (q, JC-F = 321 Hz, C21, CF3), 112.2 (C8), 92.5 (C13), 84.6 (C12), 47.8 (C2), 37.5 (C7), 22.6 (C10), 19.8 (C3); HRMS-DART+ (19 eV): elemental composition C19H17F3NO5S, [M+1]+, correlation (error of −1.37 ppm) exact value 428.0779 Daltons and precise value of 428.0773 Daltons; complementarily, the provided unsaturation data (11.5) were in agreement with the structure.
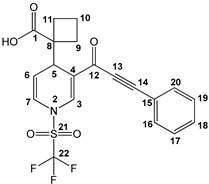
1-(3-(3-phenylpropioloyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-cyclobutane-1-carboxylic acid (3b), white solid was obtained with 82% isolated yield (0.34 g); mp 134–136 °C; IR (cm−1): 3069 (COOH), 2199 (C≡C), 1703 (C=O), 1640 (C=C); 1H NMR (CDCl3) δ(ppm): 9.87 (s, 1H, OH), 8.18 (s, 1H, H3), 7.63 (d, J= 6.9 Hz, 2H, H16, H20), 7.48 (m, 1H, H18), 7.43 (d, J= 6.9 Hz, 2H, H17, H19), 6.68 (d, J= 8.1 Hz, 1H, H7), 5.47 (dd, J= 5.4 Hz, J= 7.8 Hz, 1H, H6), 4.12 (d, J= 5.4 Hz, 1H, H5), 2.35–1.96 (m, 6H, H9, H10, H11); 13C NMR (CDCl3) δ(ppm): 181.6 (C1, C=O), 176.2 (C12, C=O), 137.2 (C3), 133.1 (C16, C20), 131.0 (C18), 128.8 (C17, C19), 122.8 (C4), 122.0 (C7), 119.5 (C15), 119.2 (q, JC-F= 321 Hz, C22, CF3), 111.8 (C6), 92.7 (C14), 84.7 (C13), 53.5 (C8), 36.4 (C5), 28.4 (C11), 28.0 (C9), 16.3 (C10); HRMS-DART+ (19 eV): elemental composition C20H17F3NO5S, [M+1]+, correlation (error of −0.17 ppm) exact value 440.0779 Daltons and precise value of 440.0778 Daltons; complementarily, the provided unsaturation data (12.5) were in agreement with the structure.
1-(3-(3-phenylpropioloyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-cyclohexane-1-carboxylic acid (3c), white solid was obtained with 93% isolated yield (0.49 g); mp 140–142 °C; IR (cm−1): 2959 (COOH), 2198 (C≡C), 1678 (C=O), 1619 (C=C); 1H NMR (CDCl3) δ (ppm): 8.17 (s, 1H, H3), 7.62 (d, J = 9.0 Hz, 2H, H18, H22), 7.50–7.40 (m, 3H, H19, H20, H21), 6.77 (d, J = 6.9 Hz, 1H, H7), 5.53 (dd, J = 6.0 Hz, J = 7.8 Hz, 1H, H6), 4.02 (d, J = 5.7 Hz, 1H, H5), 2.08–2.05 (m, 2H, H9), 1.67–1.63 (m, 4H, H10, H13), 1.33–1.24 (m, 2H, H12), 1.20–1.05 (m, 2H, H11); 13C NMR (CDCl3) δ (ppm): 178.9 (C1, C=O), 176.0 (C14, C=O), 137.1 (C3), 133.1 (C18, C22), 131.0 (C20), 128.7 (C19, C21), 122.9 (C7), 122.5 (C4), 119.5 (C17), 119.2 (q, JC-F = 321 Hz, C24, CF3), 111.6 (C6), 92.4 (C15), 84.6 (C16), 53.4 (C8), 38.6 (C5), 30.0 (C9), 29.7 (C13), 29.5 (C10), 25.4 (C12), 23.3 (C11); HRMS-DART+ (19 eV): elemental composition C22H21F3NO5S, [M+1]+, correlation (error of −2.12 ppm) exact value 468.1092 Daltons and precise value of 468.1082 Daltons; complementarily, the provided unsaturation data (12.5) were in agreement with the structure.
2-methyl-2-(3-(3-(4-(trifluoromethyl)phenyl)propioloyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-propanoic acid (3d), red solid was obtained with 82% isolated yield (0.31g); mp 118–120 °C; IR (cm−1): 2985 (COO-H), 2200 (C≡C), 1680 (C=O), 1596 (C=C); 1H NMR (CDCl3) δ (ppm): 8.13 (s, 1H, H5), 7.72–7.65 (m, 4H, H15, H16, H18, H29), 6.73 (d, J = 7.5 Hz, 1H, H9), 5.47 (dd, J = 7.8 Hz, J = 5.4 Hz, 1H, H8), 4.20 (m, 1H, H7), 1.12 (s, 3H, H3), 1.10 (s, 3H, H10); 13C NMR (CDCl3) δ (ppm): 179.9 (C1, C=O), 175.9 (C11, C=O), 167.96 (C17), 138.0 (C5), 133.3 (C15, C19), 131.02 (C16, C18), 129.9 (d, JC-F = 164 Hz, C27, CF3), 125.85 (C9), 122.9 (C6), 119.1 (q, JC-F = 321 Hz, C21, CF3), 112.2 (C8), 90.1 (C12), 85.8 (C13), 63.7 (C2), 47.5 (C7), 22.1 (C3), 20.4 (C10); HRMS-DART+ (19 eV): elemental composition C20H16F6NO5S, [M+1]+, correlation (error of −1.01 ppm) exact value 496.0653 Daltons and precise value of 496.0648 Daltons; complementarily, the provided unsaturation data (11.5) were in agreement with the structure.
2-(3-(3-(4-methoxyphenyl)propioloyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-2-methylpropanoic acid (3e), red solid was obtained with 76% isolated yield (0.27g); mp 146–148 °C; IR (cm−1): 2923 (COO-H), 2193 (C≡C), 1683 (C=O), 1600 (C=C); 1H NMR (CDCl3) δ (ppm): 9.48 (s, 1H, OH), 8.14 (s, 1H, H5), 7.54 (d, J = 9.0 Hz, 2H, H15, H19), 6.91 (d, J = 8.7 Hz, 2H, H16, H18), 6.75 (d, J = 5.4 Hz, 1H, H9), 5.48–5.46 (dd, J = 7.8 Hz, J = 5.7 Hz, 1H, H8), 4.13 (d, J = 5.7 Hz, 1H, H7), 1.24 (s, 3H, H3), 1.12 (s, 3H, H10); 13C NMR (CDCl3) δ (ppm): 181.4 (C1, C=O), 176.2 (C11, C=O), 162.0 (C17), 137.5 (C5), 135.2 (C15, C19), 122.9 (C9), 122.9 (C6), 119.1 (q, JC-F = 321 Hz, C22, CF3), 114.6 (C16, C18), 112.3 (C8), 111.3 (C14), 93.8 (C12), 84.7 (C13), 55.5 (C2), 37.5 (C7), 22.7 (C3), 19.9 (C10); HRMS-DART+ (19 eV): elemental composition C20H19F3NO5S, [M+1]+, correlation (error of −3.57 ppm) exact value 458.0885 Daltons and precise value of 458.0868 Daltons; complementarily, the provided unsaturation data (11.5) were in agreement with the structure.
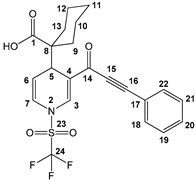
2-(3-(hept-2-ynoyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-2-methylpropanoic acid (3f), yellow solid was obtained with 77% isolated yield (0.33 g); mp 128–130 °C; IR (cm−1): 2937 (COOH), 2200 (C≡C), 1686 (C=O), 1614 (C=C); 1H NMR (CDCl3) δ (ppm): 11.2 (s, 1H, OH), 7.94 (s, 1H, H5), 6.63 (d, J = 7.8 Hz, 1H, H9), 5.42 (dd, J = 5.4 Hz, J = 7.5 Hz, 1H, H8), 3.97 (d, J = 5.7 Hz, 1H, H7), 2.36 (t, 2H, H14), 1.57–1.47 (m, 2H, H15), 1.44–1.31 (m, 2H, H16), 1.01 (s, 3H, H3), 0.99 (s, 3H, H10), 0.85 (t, 3H, H17); 13C NMR (CDCl3) δ (ppm): 182.2 (C1, C=O), 176.2 (C11, C=O), 137.1 (C5), 122.7 (C9), 119.1 (q, JC-F = 321 Hz, C19, CF3), 112.0 (C8) 95.8 (C13), 77.4 (C12), 47.6 (C2), 37.3 (C7), 22.3 (C3), 21.8 (C), 19.7 (C10), 18.5 (C14), 13.2 (C17); HRMS-DART+ (19 eV): elemental composition C17H21F3NO5S, [M+1]+, correlation (error of −2.97 ppm) exact value 408.1092 Daltons and precise value of 408.1080 Daltons; complementarily, the provided unsaturation data (7.5) were in agreement with the structure.
1-(3-(hept-2-ynoyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-cyclobutane-1-carboxylic acid (3g), yellow solid was obtained with 72% isolated yield (0.32 g); mp 116–118 °C; IR (cm−1): 2930 (COOH), 2221 (C≡C), 1720 (C=O), 1632 (C=C); 1H NMR (CDCl3) δ (ppm): 7.95 (s, 1H, H3), 6.06 (s, 1H, H7), 4.71 (s, 1H, H6), 3.8 (s, 1H, H5), 2.49–2.36 (m, 6H, H9, H11, H15), 2.13–2.10 (m, 2H, H16), 1.64–1.59 (m, 2H, H27), 1.49–1.46 (m, 2H, H10), 0.95 (t, 3H, H18); 13C NMR (CDCl3) δ (ppm): 181.7 (C1, C=O), 176.4 (C12, C=O), 136.9 (C3), 122.8 (C4), 121.9 (C7), 119.8 (q, JC-F = 321 Hz, C20, CF3), 111.7 (C6), 96.1 (C13), 77.4 (C12), 53.4 (C8), 37.8 (C5), 35.8 (C9), 29.5 (C11), 28.2 (C14), 28.0 (C16), 25.0 (C10), 21.8 (C17), 18.5 (C15), 13.2 (C18); HRMS-DART+ (19 eV): elemental composition C18H21F3NO5S, [M+1]+, correlation (error of +2.60 ppm) exact value 420.1092 Daltons and precise value of 420.1081 Daltons; complementarily, the provided unsaturation data (8.5) were in agreement with the structure.
1-(3-(hept-2-ynoyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-cyclopentane-1-carboxylic acid (3h), yellow solid was obtained with 74% isolated yield (0.34 g); mp 126–128 °C; IR (cm−1): 2937 (COOH), 2224 (C≡C), 1682 (C=O), 1613 (C=C); 1H NMR (CDCl3) δ (ppm): 8.00 (s, 1H, H3), 6.66 (d, J = 7.8 Hz, 1H, H7), 5.50 (dd, J = 5.4 Hz, J = 7.8 Hz, 1H, H6), 4.12 (d, J = 5.4 Hz, 1H, H5), 2.46 (t, 2H, H16), 2.10–2.05 (m, 2H, H9), 1.63–1.28 (m, 12H, H10, H11, H12, H17, H18), 0.96 (t, 3H, H19); 13C NMR (CDCl3) δ (ppm): 181.6 (C1, C=O), 176.4 (C13, C=O), 137.1 (C3), 123.3 (C4), 122.2 (C7), 119.1 (q, JC-F = 321 Hz, C21, CF3), 112.8 (C6), 96.4 (C15), 77.5 (C14), 60.5 (C8), 35.9 (C5), 33.1 (C9), 31.5 (C12), 29.6 (C17), 24.0 (C10), 23.9 (C11), 22.0 (C18), 18.7 (C16), 13.4 (C19); HRMS-DART+ (19 eV): elemental composition C19H23F3NO5S, [M+1]+, correlation (error of +1.85 ppm) exact value 434.1249 Daltons and precise value of 434.1257 Daltons; complementarily, the provided unsaturation data (8.5) were in agreement with the structure.
1-(3-(hept-2-ynoyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-cyclohexane-1-carboxylic acid (3i), yellow solid was obtained with 87% isolated yield (0.41 g); mp 130–132 °C; IR (cm−1): 2938 (COOH), 2223 (C≡C), 1680 (C=O), 1613 (C=C); 1H NMR (CDCl3) δ (ppm): 8.01 (s, 1H, H3), 6.73 (d, J = 7.8 Hz, 1H, H7), 5.51 (dd, J = 5.7 Hz, J = 7.8 Hz, 1H, H6), 3.94 (d, J = 5.7 Hz, 1H, H5), 2.44 (t, 2H, H17), 2.04–2.00 (m, 2H, H9), 1.66–0.92 (m, 15H, H10-H13, H18-H20); 13C NMR (CDCl3) δ (ppm): 180.5 (C1, C=O), 176.3 (C14, C=O), 137.0 (C3), 122.8 (C7), 122.7 (C4), 119.5 (q, JC-F = 321 Hz, C22, CF3), 111.7 (C6), 95.8 (C16), 77.4 (C15), 53.4 (C8), 38.6 (C5), 38.4 (C9), 29.9 (C13), 29.6 (C18), 25.4 (C10), 23.2 (C12), 21.9 (C11), 18.7 (C19), 18.6 (C17), 13.4 (C20); HRMS-DART+ (19 eV): elemental composition C20H25F3NO5S, [M+1]+, correlation (error of −1.92 ppm) exact value 448.1405 Daltons and precise value of 448.1396 Daltons; complementarily, the provided unsaturation data (8.5) were in agreement with the structure.
2-methyl-2-(3-(non-2-ynoyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-propanoic acid (3j), yellow solid was obtained with 68% isolated yield (0.37g); mp 122–124 °C; IR (cm−1): 2930 (COO-H), 2223 (C≡C), 1678 (C=O), 1613 (C=C); 1H NMR (CDCl3) δ (ppm): 10.19 (s, 1H, OH), 7.79 (s, 1H, H5), 6.47 (d, J = 7.8 Hz, 1H, H9), 5.24 (dd, J = 5.7 Hz, J = 7.8 Hz, 1H, H8), 3.82 (d, J = 5.4 Hz, 1H, 7H), 2.20 (t, 2H, H14), 1.43–1.33 (m, 2H, H15), 1.22–1.02 (m, 3H, H3, H16-H18), 0.86 (s, 3H, H10); 0.85 (s, 3H, H19); 13C NMR (CDCl3) δ (ppm): 182.2 (C1, C=O), 176.3 (C11, C=O), 137.2 (C5), 122.8 (C9), 122.6 (C6) 119.3 (q, JC-F = 321 Hz, C21 CF3), 112.1 (C8), 96.1 (C13), 77.4 (C12), 47.7 (C2), 37.3 (C7), 31.1 (C17), 28.5 (C16), 27.6 (C15), 22.5 (C3), 22.3 (C18), 19.8 (C10), 19.0 (C14), 13.9 (C19); HRMS-DART+ (19 eV): elemental composition C19H25F3NO5S, [M+1]+, correlation (error of −0.89 ppm) exact value 436.1405 Daltons and precise value of 436.1401 Daltons; complementarily, the provided unsaturation data (7.5) were in agreement with the structure.
1-(3-(non-2-ynoyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-cyclobutane-1-carboxylic acid (3k), yellow solid was obtained with 60% isolated yield (0.37g); mp 122–124 °C; IR (cm−1): 2930 (COOH), 2216 (C≡C), 1697 (C=O), 1613 (C=C); 1H NMR (CDCl3) δ (ppm): 7.99 (s, 1H, H3), 6.64 (d, J = 8.1 Hz, 1H, H7), 5.49 (dd, J = 5.4 Hz, J = 7.8 Hz, 1H, H6), 4.10 (d, J = 5.4 Hz, 1H, H5), 2.44 (t, 2H, H20), 2.07–2.03 (m, 2H, H9), 1.64–1.57 (m, 4H, H10, H11), 1.48–1.41 (m, 4H, H16, H17), 1.33–1.25 (m, 4H, H18, H19), 0.89 (t, 3H, H20); 13C NMR (CDCl3) δ (ppm): 182.1 (C1, C=O), 176.2 (C12, C=O), 137.2 (C3), 122.8 (C7), 122.6 (C4) 119.2 (q, JC-F = 321 Hz, C22, CF3), 112.1 (C6), 96.1 (C14), 77.4 (C13), 47.7 (C8), 37.3 (C5), 31.1 (C17), 28.5 (C9), 27.6 (C11), 22.5 (C16), 22.3 (C18), 19.8 (C19), 19.0 (C10), 18.3 (C15), 13.9 (C20); HRMS-DART+ (19 eV): elemental composition C20H25F3NO5S, [M+1]+, correlation (error of −3.64 ppm) exact value 448.1405 Daltons and precise value of 448.1389 Daltons; complementarily, the provided unsaturation data (8.5) were in agreement with the structure.
1-(3-(non-2-ynoyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-cyclopentane-1-carboxylic acid (3l), yellow solid was obtained with 65% isolated yield (0.37g); mp 134–136 °C; IR (cm−1): 2927 (COOH), 2223 (C≡C), 1679 (C=O), 1614 (C=C); 1H NMR (CDCl3) δ (ppm): 10.49 (s, 1H, OH), 7.99 (s, 1H, H3), 6.64 (d, J = 8.1 Hz, 1H, H7), 5.49 (dd, J = 5.4 Hz, J = 7.8 Hz, 1H, H6), 4.10 (d, J = 5.4 Hz, 1H, H5), 2.44 (t, 2H, H16), 2.07–2.03 (m, 2H, H9), 1.64–1.57 (m, 4H, H12, H17), 1.48–1.41 (m, 4H, H18, H19), 1.33–1.25 (m, 6H, H10, H11, H20), 0.89 (t, 3H, H21); 13C NMR (CDCl3) δ (ppm): 181.9 (C1, C=O), 176.5 (C13, C=O), 137.1 (C3), 123.3 (C4), 122.1 (C7), 119.3 (q, JC-F = 321 Hz, C23, CF3), 112.8 (C6), 96.1 (C15), 77.5 (C14), 60.6 (C8), 43.6 (C5), 35.9 (C9), 33.1 (C12), 31.5 (C19), 31.1 (C17), 29.9 (C18), 28.5 (C10), 27.5 (C11), 23.9 (C20), 19.8 (C16), 13.9 (C21); HRMS-DART+ (19 eV): elemental composition C21H27F3NO5S, [M+1]+, correlation (error of −3.46 ppm) exact value 462.1562 Daltons and precise value of 462.1546 Daltons; complementarily, the provided unsaturation data (8.5) were in agreement with the structure.
1-(3-(non-2-ynoyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-cyclohexane-1-carboxylic acid (3m), yellow solid was obtained with 74% isolated yield (0.37g); mp 110–112 °C; IR (cm−1): 2932 (COOH), 2217 (C≡C), 1760 (C=O), 1626 (C=C); 1H NMR (CDCl3) δ (ppm): 7.99 (s, 1H, H3), 6.71 (d, J = 7.8 Hz, 1H, H7), 5.46 (dd, J = 5.7 Hz, J = 7.8 Hz, 1H, H6), 3.92 (d, J = 5.7 Hz, 1H, H5), 2.42 (t, 2H, H17), 2.02–1.98 (m, 2H, H9), 1.65–1.56 (m, 4H, H13, H10), 1.44–1.39 (m, 4H, H12, H18), 1.31–1.24 (m, 8H, H11, H19, H20, H21), 0.88 (t, 3H, H22); 13C NMR (CDCl3) δ (ppm): 180.0 (C1, C=O), 176.3 (C14, C=O), 137.0 (C3), 122.8 (C4), 122.5 (C7), 119.3 (q, JC-F = 321, C24, CF3), 111.6 (C6), 96.0 (C16), 77.4 (C15), 53.3 (C8), 38.6 (C5), 38.4 (C20), 31.2 (C9), 29.9 (C13), 29.3 (C18), 28.6 (C19), 27.6 (C10), 25.4 (C12), 23.2 (C11), 22.4 (C21), 19.8 (C17), 13.9 (C22); HRMS-DART+ (19 eV): elemental composition C22H29F3NO5S, [M+1]+, correlation (error of −3.34 ppm) exact value 476.1718 Daltons and precise value of 476.1702 Daltons; complementarily, the provided unsaturation data (8.5) were in agreement with the structure.
2-(3-(3-cyclopentylpropioloyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-2-methylpropanoic acid (3n), yellow oil was obtained with 74% isolated yield (0.31 g); IR (cm−1): 2939 (COOH), 2206 (C≡C), 1701 (C=O), 1613 (C=C); 1H NMR (CDCl3) δ (ppm): 10.02 (s, 1H, OH), 7.97 (s, 1H, H5), 6.66 (d, J = 7.8 Hz, 1H, H9), 5.43 (dd, J = 5.7 Hz, J = 7.8 Hz, 1H, H8), 3.99 (d, J = 5.4 Hz, 1H, H7), 2.80–2.78 (m, 1H, H14), 1.97–1.66 (m, 4H, H15, H18), 1.58–1.56 (m, 4H, H16, H17), 1.04 (s, 3H, H3), 1.02 (s, 3H, H10); 13C NMR (CDCl3) δ (ppm): 182.1 (C1, C=O), 176.6 (C11, C=O), 137.4 (C5), 123.0 (C9), 122.9 (C6), 119.3 (q, JC-F = 321 Hz, C20, CF3), 112.1 (C8), 100.3 (C13), 94.5 (C12), 47.7 (C2), 37.4 (C7), 33.2 (C14), 33.2 (C15), 30.0 (C18), 25.2 (C16), 25.1 (C17), 22.4 (C3), 19.8 (C10); HRMS-DART+ (19 eV): elemental composition C18H21F3NO5S, [M+1]+, correlation (error of +4.13 ppm) exact value 420.1092 Daltons and precise value of 420.1109 Daltons; complementarily, the provided unsaturation data (8.5) were in agreement with the structure.
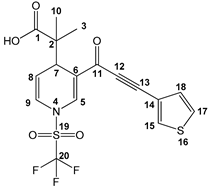
2-methyl-2-(3-(3-(thiophen-3-yl)propioloyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-propanoic acid (3o), white solid was obtained with 72% isolated yield (0.29 g); mp 156–158 °C; IR (cm−1): 2927 (COOH), 2197 (C≡C), 1758 (C=O), 1623 (C=C); 1H NMR (CDCl3) δ (ppm): 8.12 (s, 1H, H5), 7.79 (d, J = 3.0 Hz, 1H, H18), 7.36 (dd, J = 3.0 Hz, J = 5.1 Hz, 1H, H17), 7.25 (s, 1H, H16) 6.76 (d, J = 7.8 Hz, 1H, 9H), 5.50 (dd, J = 6.0 Hz, J = 8.4 Hz, 1H, H8), 4.12 (d, J = 5.7 Hz, 1H, H7), 1.12 (s, 6H, H3, H10); 13C NMR (CDCl3) δ (ppm): 181.9 (C1, C=O), 176.2 (C11, C=O), 137.4 (C5), 134.3 (C18), 130.2 (C16), 126.5 (C17), 123.0 (C9), 122.7 (C6), 119.3 (q, JC-F = 321 Hz, C20, CF3), 118.8 (C14), 112.2 (C8), 88.1 (C12), 85.0 (C13), 47.8 (C2), 37.5 (C7), 22.6 (C3), 19.8 (C10); HRMS-DART+ (19 eV): elemental composition C17H15F3NO5S2, [M+1]+, correlation (error of −2.71 ppm) exact value 434.0343 Daltons and precise value of 434.0332 Daltons; complementarily, the provided unsaturation data (11.5) were in agreement with the structure.
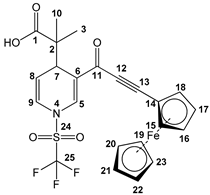
2-(3-(3-(ferrocenyl-1-yl)propioloyl)-1-((trifluoromethyl)sulfonyl)-1,4-dihydropyridin-4-yl)-propanoic acid (3p), red solid was obtained with 48% isolated yield (0.16 g); mp 148–150 °C; IR (cm−1): 2929 (COOH), 2188 (C≡C), 1725 (C=O), 1614 (C=C).;1H NMR (CDCl3) δ (ppm): 8.10 (s, 1H, H5), 6.75 (d, J = 8.1 Hz, 1H, H9), 5.50 (d, J = 6.3 Hz, 1H, H8), 4.62 (s, 2H, H16, H17, Fc), 4.41 (s, 2H, H15, H18, Fc), 4.27 (s, 5H, H19-H23, Fc) 4.12 (d, J = 5.7 Hz, 1H, H7), 1.13 (s, 6H, H3, H10); 13C NMR (CDCl3) δ (ppm): 181.1 (C1, C=O), 175.9 (C11, C=O), 136.7 (C5), 123.0 (C9), 119.3 (q, JC-F = 321 Hz, C25, CF3), 112.2 (C8), 96.1 (C13), 83.0 (C12), 73.1 (C15, C18, Fc), 73.0 (C14, Fc), 70.5 (C19-C23, Fc), 59.6 (C2), 37.6 (C7), 29.7 (C16, C18, Fc), 22.7 (C3), 20.1 (C10); HRMS-DART+ (19 eV): elemental composition C23H21F3FeNO5S, [M+1]+, correlation (error of −1.43 ppm) exact value 536.0441 Daltons and precise value of 536.0434 Daltons; complementarily, the provided unsaturation data (14.0) were in agreement with the structure.
4. Conclusions
A series of 16 dihydropyridine carboxylic acids were synthesized through a process involving the activation of pyridine derivatives ynones with triflic anhydride, followed by the nucleophilic addition of bis(trimethylsilyl) ketene acetals; the obtained molecules were unequivocally characterized by common spectroscopic modes complemented through an X-ray diffraction analysis of one of the target molecules. In general, the obtained compounds showed interesting cytotoxic activity for HCT-15 cell line; it is worth noting that two of them, 3a,b, displayed higher potency (7.94 ± 1.6 μM and 9.24 ± 0.9 μM, respectively). Consequently, it was considered important to perform theoretical calculations employing density functional theory (DFT) using the B3LYP/6–311++G(d,p). Interesting results were achieved: computational chemistry is a powerful tool used for the molecular property’s determination; it can simulate and explain how molecules interact at molecular level in the fields of chemistry, pharmacology, and drug design; additionally, complex chemical processes can also be simulated. Computational chemistry allows for precise calculations that yield highly accurate results. Moreover, this approach can eliminate the need for expensive experiments. Moreover, employing Osiris Property Explorer, it was evidenced that molecules 3a,b have physicochemical characteristics that consider them to be orally active drugs; in addition, the performed docking studies showed a high affinity to PARP-1 protein. Finally, it is very convenient to highlight that through evaluating the synthesis, in silico studies, and cytotoxic activity of two of the target compounds, new suitable antineoplasic agent candidates are now available.
Supplementary Materials
The supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms242015414/s1.
Author Contributions
Conceptualization, R.B.-I.; methodology, R.B.-I., M.I.N.-V., J.M., M.T.R.-A., A.T., M.H.-R. and E.M.J.; software, M.I.N.-V. and M.H.-R.; validation, M.I.N.-V., J.M., C.Á.-T., M.H.-R., E.M.J. and R.M.R.; formal analysis, R.B.-I., M.I.N.-V., J.M., C.Á.-T., M.H.-R., E.M.J. and R.M.R.; investigation, R.B.-I. and C.Á.-T.; resources, C.Á.-T. and R.M.R.; data curation, R.B.-I., M.I.N.-V., J.M., M.T.R.-A., C.Á.-T., A.T., M.H.-R. and E.M.J.; writing—original draft preparation, R.B.-I., M.I.N.-V., J.M., M.H.-R. and E.M.J.; writing—review and editing, R.M.R.; visualization, R.B.-I., C.Á.-T. and R.M.R.; supervision, R.M.R.; project administration, R.M.R.; funding acquisition, R.M.R. All authors have read and agreed to the published version of the manuscript.
Funding
We thank the National Autonomous University of Mexico, PAPIIT IN212421 and IN213523, in addition to Dirección General de Cómputo y de Tecnologías de Información y Comunicación (DGTIC-UNAM) for the use of the Supercomputer MIZTLI-LANCAD-UNAM-DGTIC-400. The APC was funded by CONACYT for the postdoctoral fellowship granted to R.B.-I.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data reported in this study are available upon request to mirruv@yahoo.com.mx.
Acknowledgments
The authors acknowledge to Dirección General de Cómputo y de Tecnologías de Información y Comunicación (DGTIC-UNAM) for the use of the Supercomputer MIZTLI-LANCAD-UNAM-DGTIC-400, to project PAPIIT-UNAM IN212421, and to M.C. García (Department of mass spectrometry-Instituto de Química-UNAM) for the acquisition of mass data. Additionally, thanks to Moises Hernandez Duarte and Draucin Jimenez Celi for their technical assistance. R.B.-I. gratefully acknowledge CONACYT-México for his postdoctoral fellowship 619858.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Balasubramani, A.; Gunnam, A.; Mehta, G. In situ-generated ammonia mediates deep restructuring of o-bis-ynones through a cascade process: One-pot synthesis of 2-azafluorenones. J. Org. Chem. 2022, 87, 10138–10145. [Google Scholar] [CrossRef]
- Feng, Q.; Li, S.; Li, Z.; Yan, Q.; Lin, X.; Song, L.; Zhang, X.; Wu, Y.-D.; Sun, J. Ru-catalyzed hydroboration of ynones leads to a nontraditional mode of reactivity. J. Am. Chem. Soc. 2022, 144, 14846–14855. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Chen, S.-P.; Cao, J.-H.; Li, M.; Quan, Z.-J.; Wang, X.-C.; Liang, Y.-M. Catalytic generation of remote C-N axial chirality atroposelective de novo arene construction. Org. Lett. 2022, 24, 2264–2268. [Google Scholar] [CrossRef] [PubMed]
- Nájera, C.; Sydnes, L.-K.; Yus, M. Conjugated ynones in organic synthesis. Chem. Rev. 2019, 119, 11110–11244. [Google Scholar] [CrossRef]
- Boersch, C.; Merkul, E.; Müller, T. Catalytic synthesis of N-heterocyclic ynones and ynediones by in situ activation of carboxylic acids with oxalyl chloride. Angew. Chem. Int. Ed. 2018, 50, 10448–10452. [Google Scholar] [CrossRef]
- Fraile, A.; Parra, A.; Tortosa, M.; Alemán, J. Organocatalytic transformations of alkynals, alkynones, propriolates, and related electron-deficient alkynes. Tetrahedron 2014, 70, 9145–9173. [Google Scholar] [CrossRef]
- Sengee, M.; Sydnes, L.-K. Specific conjugate addition to α, β-acetylenic ketones. Pure Appl. Chem. 2011, 83, 587–596. [Google Scholar] [CrossRef]
- Arai, T.; Ikematsu, Y.; Suemitsu, Y. Nickel-catalyzed multicomponent coupling reaction using ynones. Pure Appl. Chem. 2010, 82, 1485–1490. [Google Scholar] [CrossRef]
- Saito, Y.; Yoshida, M.; Uekusa, H.; Doi, T. Facile synthesis of pyrrolyl 4-quinone alkaloid quinolactacide by 9-aj-catalyzed tandem acyl transfer-cyclization of o-alkynoylaniline derivatives. ACS Omega 2017, 2, 4370–4381. [Google Scholar] [CrossRef]
- Schubert, U.S.; Eschbaumer, C. New synthetic strategy toward pyridine-based ligands for supramolecular chemistry utilizing 2,6-bis(trimethyltin)pyridine as the central building block. Org. Lett. 1999, 1, 1027–1029. [Google Scholar] [CrossRef]
- Gómez-García, O.; Gomez, E.; Toscano, R.A.; Salgado-Zamora, H.; Álvarez-Toledano, C. One-pot synthesis of spirotetrahydrooxino [3,4-c]pyridines and spirotetrahydrofuro[3,2-b]pyridine-2-ones via lactonization from activated pyridyldihydroozaxoles and bis(trimethylsilyl)ketene acetals. Synthesis 2016, 48, 1371–1380. [Google Scholar] [CrossRef]
- Toscano, R.A.; Rosas, R.; Hernández-Galindo, M.; Alvarez-Toledano, C.; Garcia-Mellado, O. Synthesis and structural characterization of tricarbonyl 1-azahexa-1,3,5-triene iron(0) complexes. Transit. Met. Chem. 1988, 23, 113–116. [Google Scholar] [CrossRef]
- Garduño-Alva, A.; Xu, Y.; Gualo-Soberanes, N. Synthesis of new polycyclic γ-and δ-Lactones upon activation of, and nucleophilic additions to, diazines: Influence of the activating agents. Eur. J. Org. Chem. 2008, 2008, 3714–3727. [Google Scholar] [CrossRef]
- Gualo-Soberanes, N.; Ortega-Alfaro, M.C.; López-Cortés, J.G.; Toscano, R.A.; Rudler, H.; Álvarez-Toledano, C. An expident approach to tetrahydrofuro[3,2-b]pyridine-2(3H)-ones via activation of pyridine N-oxide by triflic anhydride. Tetrahedron Lett. 2010, 51, 3186–3194. [Google Scholar] [CrossRef]
- Rivera-Hernández, A.; Chans, G.M.; Rudler, H.; López-Cortés, J.G.; Toscano, R.A.; Álvarez-Toledano, C. Reactivity of pyridines bearing EWG with bis(TMS)ketene acetals. Substituent-induced lactonization reaction. Tetrahedron 2014, 70, 1861–1874. [Google Scholar] [CrossRef]
- Malhi, D.S.; Sohal, H.S.; Singh, K.; Almarhoon, Z.M.; Bacha, A.B.; Al-Zaben, M.I. Highly efficient electrocarboxylation method to synthesize novel acid derivatives of 1,4-dihydropyridines and to study their antimicrobial activity. ACS Omega 2022, 7, 16049–16054. [Google Scholar] [CrossRef]
- Khan Saigal, M.M.; Khan, S.; Shareef, S.; Danish, M. Organocatalyzed synthesis and antifungal activity of fully substituted 1,4-dihydropyridines. ChemistrySelect 2018, 3, 6830–6835. [Google Scholar] [CrossRef]
- Hilgeroth, A.; Lilie, H. Structure-activity relationships of first bishydroxymethyl-substituted cage dimeric 4-aryl-1,4-dihydropyridines as HIV-1 protease inhibitors. Eur. J. Med. Chem. 2003, 38, 495–499. [Google Scholar] [CrossRef]
- Ozer, E.K.; Gunduz, M.G.; El-Khouly, A.; Sara, Y.; Simsek, R.; Iskit, A.B.; Safak, C. Synthesis of fused 1,4-dihydropyridines as potential calcium channel blockers. Turkish J. Biochem. 2018, 43, 578–586. [Google Scholar] [CrossRef]
- Gandomi, F.; Dilmaghani, K.A.; Massoumi, B.; Ahrabi, Y.S. Synthesis, characterization and molecular docking study of novel 1,4-dihydropyridine derivatives as antimicrobial and anticancer agents. Russ. J. Bioorg. Chem. 2023, 49, 1089–1099. [Google Scholar] [CrossRef]
- Álvarez-Toledano, C.; Penieres-Carrillo, J.-G. Reactions of bis(trimethylsilyl)ketene acetals with several activated organic substrates. Eur. J. Org. Chem. 2018, 30, 4040–4049. [Google Scholar] [CrossRef]
- Hassanpour, S.H.; Dehghani, M.J. Review of cancer from perspective of molecular. Cancer Res. Pract. 2017, 4, 127–129. [Google Scholar] [CrossRef]
- Wang, X.; Guo, Z. Targeting and delivery of platinum-based anticancer drugs. Chem. Soc. Rev. 2013, 42, 202–224. [Google Scholar] [CrossRef]
- Santini, C.; Pellei, M.; Gandin, V.; Porchia, M.; Tisato, F.; Marzano, C. Advances in copper complexes as anticancer agents. Chem. Rev. 2014, 114, 815–862. [Google Scholar] [CrossRef] [PubMed]
- Morshed, S.M.; Hashimoto, K.; Murotani, Y.; Kawase, M.; Shah, A.; Satoh, K.; Kikuchi, H.; Nishikawa, H.; Maki, J.; Sakagami, H. Tumor-specific cytotoxicity of 3,5-dibenzoyl-1,4-dihydropyridines. Anticancer Res. 2005, 25, 2033–2038. [Google Scholar] [PubMed]
- Morshed, S.M.; Hashimoto, K.; Murotani, Y.; Kawase, M.; Shah, A.; Satoh, K.; Kikuchi, H.; Nishikawa, H.; Maki, J.; Sakagami, H. Tumor-specific cytotoxicity and MDR-reversal activity of dihydropyridines. In Vivo 2006, 20, 637–644. [Google Scholar]
- Martínez, J.; Hernández-Rodríguez, M.; Miranda Ruvalcaba, R.; Escobedo-González, R.; Nicolás-Vázquez, M.I. Can (S)-stereoisomers of perezone and its derivatives show similar activity to its (R)-stereoisomers? A computational characterization and docking study. ChemistrySelect 2021, 6, 10974–10985. [Google Scholar] [CrossRef]
- Stephens, P.J.; Devlin, F.J.; Chabalowski, C.F.; Frisch, M.J. Ab initio calculation of vibrational absorption and circular spectra using density functional force fields. J. Phys. Chem. 1994, 98, 11623–11627. [Google Scholar] [CrossRef]
- Devlin, F.J.; Finley, J.W.; Stephens, P.J.; Frisch, M.J. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields: A comparison of local, nonlocal, and hybrid density functionals. J. Phys. Chem. 1995, 99, 16883–16902. [Google Scholar] [CrossRef]
- Martínez, J.; Hernández Rodríguez, M.; Escobedo-González, R.; Nicolás-Vázquez, M.I.; Saavedra-Leos, Z.; Miranda Ruvalcaba, R. Computational characterization of perezone, isoperezone and their sulfur-derivatives: Anti-inflammatory activity. ChemistrySelect 2019, 4, 13333–13346. [Google Scholar] [CrossRef]
- Rivera-Hernández, A.; López-Jimeno, I.S.; Carmona-Reyes, G.A.; Toscano, R.A.; Penieres-Carrillo, J.G.; Álvarez-Toledano, C. Facile access to imidazole derivatives: Carboxylic acids and δ-lactones. Tetrahedron Lett. 2015, 56, 4829–4832. [Google Scholar] [CrossRef]
- Noolvi, M.N.; Patel, H.M.; Kaur, M. Benzothiazoles: Search for anticancer agents. Eur. J. Med. Chem. 2012, 54, 447–462. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Prassanna, S.; Doerksen, R.J. Topological polar surface area: A useful descriptor in 2D-QSAR. Curr. Med. Chem. 2009, 16, 21–41. [Google Scholar] [CrossRef] [PubMed]
- Petit, J.; Meurice, N.; Kaiser, C.; Maggiora, G. Softening the rule of five-where to draw the line? Bioorg. Med. Chem. 2012, 20, 5343–5351. [Google Scholar] [CrossRef] [PubMed]
- Meng Zhang, X.Y.; Mezei, M.; Cui, M. Molecular docking: A powerful approach for structure-based drug discovery. Curr. Comput. Aided Drug Des. 2011, 7, 146–157. [Google Scholar] [CrossRef]
- Escobedo-González, R.; Vargas-Requena, C.L.; Moyers-Montoya, E.; Aceves-Hernández, J.M.; Nicolás-Vázquez, M.I.; Miranda-Ruvalcaba, R. In silico study of the pharmacologic properties and cytotoxicity pathways in cancer cells of various indolylquinones analogues of perezone. Molecules 2017, 22, 1060. [Google Scholar] [CrossRef]
- Kinoshita, T.; Nakanishi, I.; Warizaya, M.; Iwashita, A.; Kido, Y.; Hattori, K.; Fujii, T. Inhibitor-induced structural change of the active site of human poly(ADP-ribose) polymerase. FEBS Lett. 2004, 556, 43–46. [Google Scholar] [CrossRef]
- Puentes-Pardo, J.D.; Moreno-San Juan, S.; Casado, J.; Escudero-Feliu, J.; López-Pérez, D.; Sánchez-Uceta, P.; González-Novoa, P.; Gálvez, P.; Carazo, A.; León, J. PARP-1 expression influences cancer stem cell phenotype in colorectal cancer depending on p53. Int. J. Mol. Sci. 2023, 24, 4787. [Google Scholar] [CrossRef]
- Wang, L.; Liang, C.; Li, F.; Guan, D.; Wu, X.; Fu, X.; Lu, A.; Zhang, G. PARP-1 in carcinomas and PARP-1 inhibitors as antineoplasic drugs. Int. J. Mol. Sci. 2017, 18, 2111. [Google Scholar] [CrossRef]
- Tamer, Ö. A unique manganese (II) complex of 4-methoxy-pyridine-2-carboxylate: Synthesis, crystal structure, FT-IR and UV–vis spectra and DFT calculations. J. Mol. Struct. 2017, 1144, 370–378. [Google Scholar] [CrossRef]
- Parr, R.G.; Szentpály, L.V.; Liu, S. Electrophilicity index. J. Am. Chem. Soc. 1999, 121, 1922–1924. [Google Scholar] [CrossRef]
- Parr, R.G.; Pearson, R.G. Absolute hardness: Companion parameter to absolute electronegativity J. Am. Chem. Soc. 1983, 105, 7512–7516. [Google Scholar] [CrossRef]
- Soltani, A.; Ghari, F.; Khalaji, A.D.; Tazikeh Lemeski, E.; Fejfavora, K.; Dusek, M.; Shikhi, M. Crystal structure, spectroscopic and theoretical studies on two Schiff base compounds of 2,6-dichlorobenzylidene-2,4-dichloroaniline and 2,4- dichlorobenzylidene-2,4-dichloroaniline. Spectrochim. Acta A. Mol. Biomol. Spectrosc. 2015, 139, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Parr, R.G. Hardness, softness, and the Fukui function in the electronic theory of metals and catalysis. Proc. Natl. Acad. Sci. USA 1985, 82, 6723–6726. [Google Scholar] [CrossRef]
- Fleming, I. Frontier Orbitals and Organic Chemical Reactions; John Wiley & Sons: London, UK, 1976. [Google Scholar]
- Kim, K.H.; Han, Y.K.; Jung, J. Basis set effect on relative energies and HOMO-LUMO energy gaps of fullerene C36. Theor. Chem. Acc. 2005, 113, 233–237. [Google Scholar] [CrossRef]
- Gunasekaran, S.; Kumaresan, S.; Arunbalaji, R.; Anand, G.; Srinivasan, S. Density functional theory study of vibrational spectra, and assignment of fundamental modes of dacarbazine. J. Chem. Sci. 2008, 120, 315–324. [Google Scholar] [CrossRef]
- Sıdır, İ.; Sıdır, Y.G.; Kumalar, M.; Taşal, E. Ab initio Hartree–Fock and density functional theory investigations on the conformational stability, molecular structure, and vibrational spectra of 7-acetoxy-6-(2, 3-dibromopropyl)-4, 8-dimethylcoumarin molecule. J. Mol. Struct. 2010, 964, 134–151. [Google Scholar] [CrossRef]
- Barone, G.; Gomez-Paloma, L.; Duca, D.; Silvestri, A.; Riccio, R.; Bifulco, G. Structure of natural products by quantum-mechanical GIAO calculations of 13C NMR chemical shifts. Chem. Eur. J. 2002, 8, 3233–3239. [Google Scholar] [CrossRef]
- Radder, S.B.; Melavanki, R.; Hiremath, S.M.; Kusanur, R.; Khemalapure, S.S.; Jeyaseelan, S.C. Synthesis, spectroscopic (FT-IR, FT-Raman, NMR & UV-Vis), reactive (ELF, LOI, Fukui), drug likeness and molecular docking insights on novel 4-[3-(methoxy-phenyl)-3-oxo-propenyl]-benzonitrile by experimental and computational methods. Heliyon 2021, 7, e08429. [Google Scholar]
- Khemalapure, S.S.; Katti, V.S.; Hiremath, C.S.; Hiremath, S.M.; Basanagouda, M.; Radder, S.B. Spectroscopic (FT-IR, FT-Raman, NMR and UV-Vis), ELF, LOI, NBO, and Fukui function investigations on (5-bromo-benzofuran-3-yl)-acetic acid hydrazide (5BBAH): Experimental and theoretical approach. J. Mol. Struct. 2019, 1196, 280–290. [Google Scholar] [CrossRef]
- Silverstein, R.M.; Webster, F.X.; Klemie, D.J.; Bryce, D.L. Spectrometric Identification of Organic Compounds, 7th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2005; Chapter 2; pp. 72–110. [Google Scholar]
- Fekri, L.Z.; Nikpassand, M. Synthesis, experimental and DFT studies on the crystal structure, FTIR, 1H NMR and 13C NMR spectra of derivatives of dihydropyridines. J. Chil. Chem. Soc. 2012, 57, 1415–1421. [Google Scholar] [CrossRef]
- Ballinas-Indilí, R.; Gómez-García, O.; Treviño-Crespo, E.; Andrade-Pavón, D.; Villa-Tanaca, L.; Toscano, R.A.; Álvarez-Toledano, C. One-pot Synthesis of dihydropyridine carboxylic acids via functionalization of 3-((trimethylsilyl)ethynyl)pyridines and an unusual hydration of alkynes: Molecular docking and antifungal activity. Tetrahedron 2021, 86, 132086. [Google Scholar] [CrossRef]
- Soliman, N.N.; Tag, Y.M.; Bayoumy, N.M. Synthesis and molecular docking of some new 3,5-bis-(diazepine, pyrazolopyrimidine, pyrimidine, and pyrazole) pyridine derivatives and their in vitro and in vivo biological evaluation as potential antitumor agents. J. Heterocyclic Chem. 2021, 58, 1141–1153. [Google Scholar] [CrossRef]
- Tu, S.; Miao, C.; Fang, F.; Youjian, F.; Li, T.; Zhuang, Q.; Zhang, X.; Zhu, S.; Shi, D. New potential calcium channel modulators: Design and synthesis of compounds containing two pyridine, pyrimidine, pyridone, quinolone and acridine units under microwave irradiation. Bioorg. Med. Chem. Lett. 2004, 14, 1533–1536. [Google Scholar] [CrossRef]
- Yidirim, S.Ö.; Büyükmumcu, Z.; Butcher, R.J.; Ҫetin, G.; Şimşek, R. Synthesis and spectroscopic characterization and DFT study of benzyl 4-([1,1’-biphenyl]-4-yl)-2,6,6-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate. J. Mol. Struct. 2020, 1200, 127069. [Google Scholar] [CrossRef]
- Devi, P.; Fatma, S.; Bishnoi, A.; Srivastava, K.; Shukla, S.; Kumar, R. Synthesis, spectroscopic and DFT studies of novel 4-(morpholinomethyl)-5-oxo-1-phenylpyrrolidine-3-carboxylic acid. J. Mol. Struct. 2018, 1157, 551–559. [Google Scholar] [CrossRef]
- Devi, P.; Fatma, S.; Shukla, S.; Kumar, R.; Singh, V.; Bishnoi, A. Synthesis, spectroscopic investigation, molecular docking and DFT studies of novel (2Z,4Z)-2,4-bis(4-chlorobenzylidene)-5-oxo-1-phenylpyrrolidine-3-carboxylic acid (BCOPCA). Heliyon 2018, 4, e01009. [Google Scholar] [CrossRef]
- Karthick, R.; Verlaj, G.; Pachamuthu, M.P.; Karthikeyan, S. Synthesis, spectroscopic, DFT, and molecular docking studies on 1,4-dihydropyridine derivative compounds: A combined experimental and theoretical study. J. Mol. Mod. 2022, 28, 5. [Google Scholar] [CrossRef]
- Kavitha, T.; Verlaj, G. Structural, spectroscopic (FT-IR, FT-Raman, NMR) and computational analysis (DOS, NBO, Fukui) of 3,5-dimethylisoxazole and 4-(chloromethyl)-3,5-dimethylisoxazole: A DFT study. J. Theor. Comput. Chem. 2016, 15, 1650039. [Google Scholar] [CrossRef]
- Balakrishnan, C.; Subha, L.; Neelakantan, M.A.; Mariappan, S.S. Synthesis, spectroscopy, X-ray crystallography, DFT calculations, DNA binding and molecular docking of a propargyl arms containing Schiff base. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 150, 671–681. [Google Scholar] [CrossRef]
- Škríba, A.; Jašík, J.; Andris, E.; Roithová, J. Interaction of ruthenium(II) with terminal alkynes: Benchmarking DFT methods with spectroscopic data. Organometallics 2016, 35, 990–994. [Google Scholar] [CrossRef]
- Ainsworth, C.; Kuo, Y.J. Ketene bis (trialkylsilyl) acetals: Synthesis, pyrolysis and spectral studies. J. Organomet. Chem. 1972, 46, 73–87. [Google Scholar] [CrossRef]
- Zhao, T.; Xu, B. Palladium-catalyzed tandem amination reaction for the synthesis of 4-quinolones. Org. Lett. 2010, 12, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Halgren, T.A. Merck molecular force field. I. Basis, form, scope, parameterization, and performance of MMFF94. J. Comput. Chem. 1996, 17, 490–519. [Google Scholar] [CrossRef]
- Halgren, T.A. Merck molecular force field. V. Extension of MMFF94 using experimental data, additional computational data, and empirical rules. J. Comput. Chem. 1996, 17, 616–641. [Google Scholar] [CrossRef]
- Halgren, T.A.; Nachbar, R.B. Merck molecular force field. IV. conformational energies and geometries for MMFF94. J. Comput. Chem. 1996, 17, 587–615. [Google Scholar] [CrossRef]
- Deppmeier, B.J.; Driessen, A.J.; Hehre, T.S.; Hehre, W.J.; Johnson, J.A.; Klunzinger, P.E.; Leonard, J.M.; Pham, I.; Pietro, W.J.; Jianguo, Y.; et al. Spartan 06 for Windows; WavefunctionInc: Irvine, CA, USA, 2006. [Google Scholar]
- Parr, R.G.; Horizons of Quantum Chemistry; Fukui, K. , Pullman, B., Eds.; Académie Internationale Des Sciences Moléculaires Quantiques/International Academy of Quantum Molecular Science, Springer: Dordrecht, The Netherlands, 1980; Volume 3. [Google Scholar]
- Kohn, W.; Becke, A.D.; Parr, R.G. Density functional theory of electronic structure. J. Phys. Chem. 1996, 100, 12974–12980. [Google Scholar] [CrossRef]
- Medvedev, M.G.; Bushmarinov, I.S.; Sun, J.; Perdew, J.P.; Lyssenko, K.A. Density functional theory is straying from the path toward the exact functional. Science 2017, 355, 49–52. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Ditchfield, R.; Hehre, W.J.; Pople, J.A. Self-consistent molecular-orbital methods. IX. An extended gaussian-type basis for molecular-orbital studies of organic molecules. J. Chem. Phys. 1971, 54, 724–728. [Google Scholar] [CrossRef]
- Hariharan, P.C.; Pople, J.A. The influence of polarization functions on molecular orbital hydrogenation energies. Theor. Chim. Acta 1973, 28, 213–222. [Google Scholar] [CrossRef]
- Wiberg, K.B. Basis set effects on calculated geometries: 6–311++G** vs. aug-cc-pVDZ. J. Comput. Chem. 2004, 25, 1342–1346. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16, Revision, A.03; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Dennington, R.D., II; Keith, T.A.; Millam, J.M. (2000–2016) GaussView 6.0.16 (64-bit Windows); Semichem Inc.: Shawnee, KS, USA, 2016. [Google Scholar]
- Nicolás-Vázquez, I.; Pérez-Caballero, G.; Galano Jiménez, A.; Guzmán Rangel, G.; Miranda Ruvalcaba, R. A novel azocompounds, 2-(4-phenylazoaniline)-4-phenylphenol: Spectroscopic and quantum-chemical approach. Int. J. Quantum Chem. 2013, 113, 1107–1115. [Google Scholar] [CrossRef]
- Andersson, M.P.; Uvdal, P. New scale factors for harmonic vibrational frequencies using the B3LYP density functional method with the triple-ζ basis set 6–311+G(d,p). J. Phys. Chem. A 2005, 109, 2937–2941. [Google Scholar] [CrossRef]
- Wolinski, K.; Hilton, J.F.; Pulay, P. Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J. Am. Chem. Soc. 1990, 112, 8251–8260. [Google Scholar] [CrossRef]
- Cheeseman, J.R.; Trucks, G.W.; Keith, T.A.; Frisch, M.J. A Comparison of models for calculating nuclear magnetic resonance shielding tensors. J. Chem. Phys. 1996, 104, 5497–5509. [Google Scholar] [CrossRef]
- Reed, A.E.; Weinstock, R.B.; Weinhold, F. Natural population analysis. J. Chem. Phys. 1985, 83, 735–746. [Google Scholar] [CrossRef]
- Glendening, E.D.; Reed, A.E.; Carpenter, J.E.; Weinhold, F. NBO Version 3.1; University of Wisconsin: Madison, WI, USA, 1998; Volume 65 TCI. [Google Scholar]
- Zhang, G.; Musgrave, C.B. Comparison of DFT methods for molecular orbital eigenvalue calculations. J. Phys. Chem. A 2007, 111, 1554–1561. [Google Scholar] [CrossRef]
- Parr, R.G.; Donnelly, R.A.; Levy, M.; Palke, W.E. Electronegativity: The density functional viewpoint. J. Chem. Phys. 1978, 68, 3801–3807. [Google Scholar] [CrossRef]
- Du, Q.-S.; Huang, R.-B.; Chou, K.-C. Advances in visual representation of molecular potentials. Expert Opin. Drug Discov. 2010, 5, 513–527. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 andAutoDOck-Tools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef]
- Cosconati, S.; Forli, S.; Perryman, A.L.; Harris, R.; Goodsell, D.S.; Olson, A.J. Virtual screening with AutoDock: Theory and practice. Expert Opin. Drug Discov. 2010, 5, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Durán, L.A.; Rosales-Hernández, M.C.; Hernández-Rodríguez, M.; Mendieta-Wejebe, J.E.; Trujillo-Reffrara, J.; Correa-Basurto, J. Mapping myeloperoxidase to identify its promiscuity properties using docking and molecular dynamics simulations. Curr. Pharm. Des. 2013, 19, 2204–2215. [Google Scholar] [CrossRef]
- Schrödinger, L.; DeLano, W. PyMOL. 2020. Available online: http://www.pymol.org/pymol (accessed on 4 August 2023).
- Touré, B.B.; Miller-Moslin, K.; Yusuff, N.; Perez, L.; Doré, M.; Joud, C.; Michael, W.; DiPietro, L.; van der Plas, S.; McEwan, M.; et al. The role of the acidity of N-heteroaryl sulfonamides as inhibitors of bcl-2 family protein-protein interactions. ACS Med. Chem. Lett. 2013, 4, 186–190. [Google Scholar] [CrossRef]
- Tao, Z.F.; Wang, X.; Chen, J.; Ingram, J.P.; Jin, S.; Judge, R.A.; Kovar, P.J.; Park, C.; Sun, C.; Wakefield, B.D.; et al. Structure-based design of A-1293102, a potent and selective BCL-XL inhibitor. ACS Med. Chem. Lett. 2021, 12, 1011–1016. [Google Scholar] [CrossRef] [PubMed]
- Anil, B.; Riedinger, C.; Endicott, J.A.; Noble, M.E. The structure of an MDM2-Nutlin-3a complex solved by the use of a validated MDM2 surface-entropy reduction mutant. Acta Crystallogr. D Biol. Crystallogr. 2013, 69, 1358–1366. [Google Scholar] [CrossRef]
- Chen, K.C.; Lee, W.Y.; Chen, H.Y.; Chen, C.Y. In silico investigation of potential mTOR inhibitors from traditional Chinese medicine for treatment of Leigh syndrome. Biomed. Res. Int. 2014, 2014, 139492. [Google Scholar]
- Tabackman, A.A.; Frankson, R.; Marsan, E.S.; Perry, K.; Cole, K.E. Structure of ‘linkerless’ hydroxamic acid inhibitors-HDAC8 complex confirms the formation of an isoform-specific subpocket. J. Struct. Biol. 2016, 195, 373–378. [Google Scholar] [CrossRef]
- Lee, S.Y.; Boo, B.H. Density functional theory study of vibrational spectra of anthracene neutral and radical cation. Bull. Korean Chem. Soc. 1996, 17, 754–759. [Google Scholar]
- Gómez, E.; Contreras-Ordoñez, G.; Ramírez-Apan, T. Synthesis, characterization and in vitro cytotoxicity of pentacoordinated tin(IV) complexes derived from aminoalcohols. Chem. Pharm. Bull. 2006, 54, 54–57. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gómez-García, O.; Gómez, E.; Monzón-González, C.; Ramírez-Apan, T.; Álvarez-Toledano, C. An efficient strategy for the synthesis of 1-(trifluromethylsulfoamido)propan-2-yl esters and the evaluation of their cytotoxic activity. Chem. Pharm. Bull. 2017, 65, 248–252. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).