A Non-Coding Fc Gamma Receptor Cis-Regulatory Variant within the 1q23 Gene Cluster Is Associated with Plasmodium falciparum Infection in Children Residing in Burkina Faso
Abstract
:1. Introduction
2. Results
2.1. Prioritization and Annotation of Putative Regulatory SNPs in the Non-Coding FCGR Gene Cluster
2.2. Analysis of Allelic and Genotypic Frequencies of Non-Coding Regulatory and Coding FCGR2A/2B Variants in Burkina Faso Population
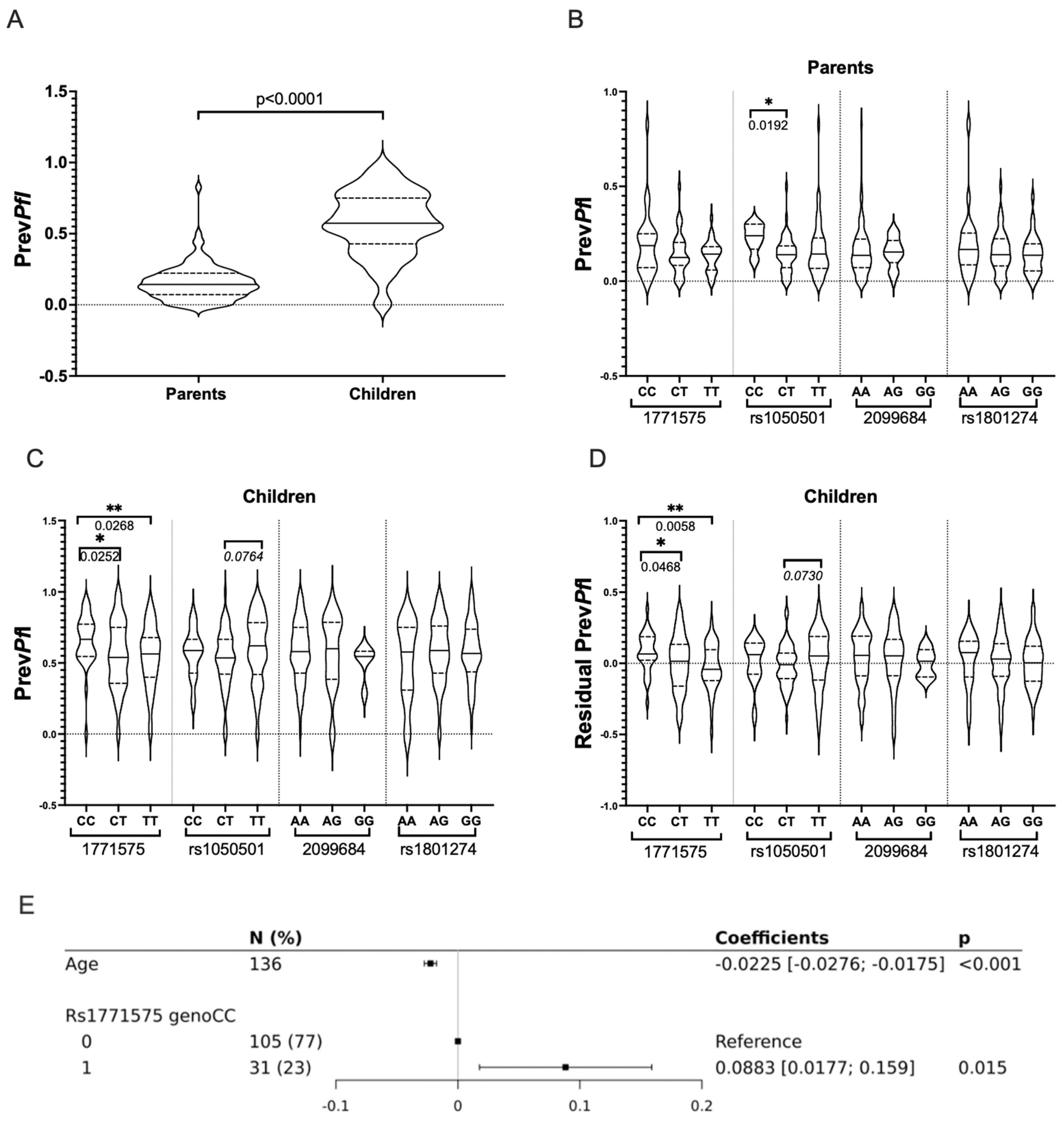
2.3. The Non-Coding rs1771575 Regulatory Variant Is Associated with Plasmodium falciparum Infection
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Phenotyping
4.3. Bioinformatic Identification of Non-Coding Variants
4.4. Genotyping of FCGR Variants
4.5. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Malaria Report. 2022. Available online: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2022 (accessed on 4 April 2023).
- Kwiatkowski, D.P. How malaria has affected the human genome and what human genetics can teach us about malaria. Am. J. Hum. Genet. 2005, 77, 171–192. [Google Scholar] [CrossRef] [PubMed]
- Esoh, K.K.; Apinjoh, T.O.; Amambua-Ngwa, A.; Nyanjom, S.G.; Chimusa, E.R.; Amenga-Etego, L.; Wonkam, A.; Achidi, E.A. Genome-wide association study identifies novel candidate malaria resistance genes in Cameroon. Hum. Mol. Genet. 2023, 32, 1946–1958. [Google Scholar] [CrossRef] [PubMed]
- Damena, D.; Agamah, F.E.; Kimathi, P.O.; Kabongo, N.E.; Girma, H.; Choga, W.T.; Golassa, L.; Chimusa, E.R. Insilico Functional Analysis of Genome-Wide Dataset from 17,000 Individuals Identifies Candidate Malaria Resistance Genes Enriched in Malaria Pathogenic Pathways. Front. Genet. 2021, 12, 676960. [Google Scholar] [CrossRef]
- Moncunill, G.; Scholzen, A.; Mpina, M.; Nhabomba, A.; Hounkpatin, A.B.; Osaba, L.; Valls, R.; Campo, J.J.; Sanz, H.; Jairoce, C.; et al. Antigen-stimulated PBMC transcriptional protective signatures for malaria immunization. Sci. Transl. Med. 2020, 12, eaay8924. [Google Scholar] [CrossRef]
- Tran, T.M.; Guha, R.; Portugal, S.; Skinner, J.; Ongoiba, A.; Bhardwaj, J.; Jones, M.; Moebius, J.; Venepally, P.; Doumbo, S.; et al. A Molecular Signature in Blood Reveals a Role for p53 in Regulating Malaria-Induced Inflammation. Immunity 2019, 51, 750–765.e10. [Google Scholar] [CrossRef]
- Antonelli, L.R.V.; Leoratti, F.M.S.; Costa, P.A.C.; Rocha, B.C.; Diniz, S.Q.; Tada, M.S.; Pereira, D.B.; Teixeira-Carvalho, A.; Golenbock, D.T.; Gonçalves, R.; et al. The CD14+CD16+ inflammatory monocyte subset displays increased mitochondrial activity and effector function during acute Plasmodium vivax malaria. PLoS Pathog. 2014, 10, e1004393. [Google Scholar] [CrossRef]
- Dechavanne, C.; Nouatin, O.; Adamou, R.; Edslev, S.; Hansen, A.; Meurisse, F.; Sadissou, I.; Gbaguidi, E.; Milet, J.; Cottrell, G.; et al. Placental Malaria is Associated with Higher LILRB2 Expression in Monocyte Subsets and Lower Anti-Malarial IgG Antibodies During Infancy. Front. Immunol. 2022, 13, 909831. [Google Scholar] [CrossRef]
- Royo, J.; Rahabi, M.; Kamaliddin, C.; Ezinmegnon, S.; Olagnier, D.; Authier, H.; Massougbodji, A.; Alao, J.; Ladipo, Y.; Deloron, P.; et al. Changes in monocyte subsets are associated with clinical outcomes in severe malarial anaemia and cerebral malaria. Sci. Rep. 2019, 9, 17545. [Google Scholar] [CrossRef]
- Nziza, N.; Tran, T.M.; DeRiso, E.A.; Dolatshahi, S.; Herman, J.D.; Lacerda, L.d.; Junqueira, C.; Lieberman, J.; Ongoiba, A.; Doumbo, S.; et al. Accumulation of Neutrophil Phagocytic Antibody Features Tracks with Naturally Acquired Immunity Against Malaria in Children. J. Infect. Dis. 2023, 228, 759–768. [Google Scholar] [CrossRef]
- Garcia-Senosiain, A.; Kana, I.H.; Singh, S.; Das, M.K.; Dziegiel, M.H.; Hertegonne, S.; Adu, B.; Theisen, M. Neutrophils dominate in opsonic phagocytosis of P. falciparum blood-stage merozoites and protect against febrile malaria. Commun. Biol. 2021, 4, 984. [Google Scholar] [CrossRef]
- Chimma, P.; Roussilhon, C.; Sratongno, P.; Ruangveerayuth, R.; Pattanapanyasat, K.; Pérignon, J.-L.; Roberts, D.J.; Druilhe, P. A distinct peripheral blood monocyte phenotype is associated with parasite inhibitory activity in acute uncomplicated Plasmodium falciparum malaria. PLoS Pathog. 2009, 5, e1000631. [Google Scholar] [CrossRef] [PubMed]
- Waisberg, M.; Molina-Cruz, A.; Mizurini, D.M.; Gera, N.; Sousa, B.C.; Ma, D.; Leal, A.C.; Gomes, T.; Kotsyfakis, M.; Ribeiro, J.M.C.; et al. Plasmodium falciparum infection induces expression of a mosquito salivary protein (Agaphelin) that targets neutrophil function and inhibits thrombosis without impairing hemostasis. PLoS Pathog. 2014, 10, e1004338. [Google Scholar] [CrossRef] [PubMed]
- Ofori, E.A.; Garcia-Senosiain, A.; Naghizadeh, M.; Kana, I.H.; Dziegiel, M.H.; Adu, B.; Singh, S.; Theisen, M. Human blood neutrophils generate ROS through FcγR-signaling to mediate protection against febrile P. falciparum malaria. Commun. Biol. 2023, 6, 743. [Google Scholar] [CrossRef] [PubMed]
- Tangteerawatana, P.; Krudsood, S.; Kanchanakhan, N.; Troye-Blomberg, M.; Khusmith, S. Low monocyte to neutrophil ratio in peripheral blood associated with disease complication in primary Plasmodium falciparum infection. Southeast Asian J. Trop. Med. Public. Health 2014, 45, 517–530. [Google Scholar]
- Feng, G.; Wines, B.D.; Kurtovic, L.; Chan, J.-A.; Boeuf, P.; Mollard, V.; Cozijnsen, A.; Drew, D.R.; Center, R.J.; Marshall, D.L.; et al. Mechanisms and targets of Fcγ-receptor mediated immunity to malaria sporozoites. Nat. Commun. 2021, 12, 1742. [Google Scholar] [CrossRef]
- Dick, J.K.; Hart, G.T. Natural Killer Cell Antibody-Dependent Cellular Cytotoxicity (ADCC) Activity Against Plasmodium falciparum-Infected Red Blood Cells. Methods Mol. Biol. 2022, 2470, 641–657. [Google Scholar] [CrossRef]
- Bouharoun-Tayoun, H.; Druilhe, P. Antibody-Dependent Cell-Mediated Inhibition (ADCI) of Plasmodium falciparum: One- and Two-Step ADCI Assays. Methods Mol. Biol. 2015, 1325, 131–144. [Google Scholar] [CrossRef]
- Pleass, R.J. Fc-receptors and immunity to malaria: From models to vaccines. Parasite. Immunol. 2009, 31, 529–538. [Google Scholar] [CrossRef]
- Tiendrebeogo, R.W.; Adu, B.; Singh, S.K.; Dziegiel, M.H.; Nébié, I.; Sirima, S.B.; Christiansen, M.; Dodoo, D.; Theisen, M. Antibody-Dependent Cellular Inhibition Is Associated with Reduced Risk against Febrile Malaria in a Longitudinal Cohort Study Involving Ghanaian Children. Open Forum. Infect. Dis. 2015, 2, ofv044. [Google Scholar] [CrossRef]
- Nagelkerke, S.Q.; Schmidt, D.E.; Haas, M.d.; Kuijpers, T.W. Genetic Variation in Low-To-Medium-Affinity Fcγ Receptors: Functional Consequences, Disease Associations, and Opportunities for Personalized Medicine. Front. Immunol. 2019, 10, 2237. [Google Scholar] [CrossRef]
- Anania, J.C.; Chenoweth, A.M.; Wines, B.D.; Hogarth, P.M. The Human FcγRII (CD32) Family of Leukocyte FcR in Health and Disease. Front. Immunol. 2019, 10, 464. [Google Scholar] [CrossRef] [PubMed]
- Gillis, C.; Gouel-Chéron, A.; Jönsson, F.; Bruhns, P. Contribution of Human FcγRs to Disease with Evidence from Human Polymorphisms and Transgenic Animal Studies. Front. Immunol. 2014, 5, 254. [Google Scholar] [CrossRef] [PubMed]
- Schuldt, K.; Esser, C.; Evans, J.; May, J.; Timmann, C.; Ehmen, C.; Loag, W.; Ansong, D.; Ziegler, A.; Agbenyega, T.; et al. FCGR2A functional genetic variant associated with susceptibility to severe malarial anaemia in Ghanaian children. J. Med. Genet. 2010, 47, 471–475. [Google Scholar] [CrossRef]
- Munde, E.O.; Okeyo, W.A.; Raballah, E.; Anyona, S.B.; Were, T.; Ong’echa, J.M.; Perkins, D.J.; Ouma, C. Association between Fcγ receptor IIA, IIIA and IIIB genetic polymorphisms and susceptibility to severe malaria anemia in children in western Kenya. BMC Infect. Dis. 2017, 17, 289. [Google Scholar] [CrossRef]
- Ouma, C.; Keller, C.C.; Opondo, D.A.; Were, T.; Otieno, R.O.; Otieno, M.F.; Orago, A.S.S.; Ong’Echa, J.M.; Vulule, J.M.; Ferrell, R.E.; et al. Association of FCgamma receptor IIA (CD32) polymorphism with malarial anemia and high-density parasitemia in infants and young children. Am. J. Trop. Med. Hyg. 2006, 74, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Mishra, S.K.; Sharma, S.; Patibandla, P.K.; Mallick, P.K.; Sharma, S.K.; Mohanty, S.; Pati, S.S.; Mishra, S.K.; Ramteke, B.K.; et al. Polymorphisms of TNF-enhancer and gene for FcgammaRIIa correlate with the severity of falciparum malaria in the ethnically diverse Indian population. Malar. J. 2008, 7, 13. [Google Scholar] [CrossRef]
- Zhao, J.; Ma, L.; Chen, S.; Xie, Y.; Xie, L.; Deng, Y.; He, Y.; Li, T.; Wang, J.; Li, S.; et al. Association between Fc-gamma receptor IIa (CD32) gene polymorphism and malaria susceptibility: A meta-analysis based on 6928 subjects. Infect. Genet. Evol. 2014, 23, 169–175. [Google Scholar] [CrossRef]
- Amiah, M.A.; Ouattara, A.; Okou, D.T.; N’Guetta, S.-P.A.; Yavo, W. Polymorphisms in Fc Gamma Receptors and Susceptibility to Malaria in an Endemic Population. Front. Immunol. 2020, 11, 561142. [Google Scholar] [CrossRef]
- Fall, A.K.D.J.; Courtin, D.; Adamou, R.; Edslev, S.; Hansen, A.; Domingo, N.; Christiansen, M.; Adu, B.; Milet, J.; Garcia, A.; et al. Fc Gamma Receptor IIIB NA1/NA2/SH Polymorphisms Are Associated with Malaria Susceptibility and Antibody Levels to P. falciparum Merozoite Antigens in Beninese Children. Int. J. Mol. Sci. 2022, 23, 14882. [Google Scholar] [CrossRef]
- Smith, K.G.C.; Clatworthy, M.R. FcgammaRIIB in autoimmunity and infection: Evolutionary and therapeutic implications. Nat. Rev. Immunol. 2010, 10, 328–343. [Google Scholar] [CrossRef]
- Fall, A.K.D.J.; Kana, I.H.; Dechavanne, C.; Garcia-Senosiain, A.; Guitard, E.; Milet, J.; Massougbodji, A.; Garcia, A.; Dugoujon, J.-M.; Migot-Nabias, F.; et al. Naturally acquired antibodies from Beninese infants promote Plasmodium falciparum merozoite-phagocytosis by human blood leukocytes: Implications for control of asymptomatic malaria infections. Malar. J. 2022, 21, 356. [Google Scholar] [CrossRef] [PubMed]
- Nasr, A.; Aljada, A.; Hamid, O.; Elsheikh, H.A.; Masuadi, E.; Al-Bawab, A.; Alenazi, T.H.; Abushouk, A.; Salah, A.M. Significant differences in FcγRIIa, FcγRIIIa and FcγRIIIb genes polymorphism and anti-malarial IgG subclass pattern are associated with severe Plasmodium falciparum malaria in Saudi children. Malar. J. 2021, 20, 376. [Google Scholar] [CrossRef] [PubMed]
- Cherif, M.K.; Sanou, G.S.; Maiga, B.; Israelsson, E.; Ouédraogo, A.L.; Bougouma, E.C.; Diarra, A.; Ouédraogo, A.; Ouattara, A.S.; Troye-Blomberg, M.; et al. FcγRIIa polymorphism and anti-malaria-specific IgG and IgG subclass responses in populations differing in susceptibility to malaria in Burkina Faso. Scand. J. Immunol. 2012, 75, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Simpson, A.P.; Roghanian, A.; Oldham, R.J.; Chan, H.T.C.; Penfold, C.A.; Kim, H.J.; Inzhelevskaya, T.; Mockridge, C.I.; Cox, K.L.; Bogdanov, Y.D.; et al. FcγRIIB controls antibody-mediated target cell depletion by ITIM-independent mechanisms. Cell Rep. 2022, 40, 111099. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Li, G.; Wang, J.; Fan, Y.; Wan, Z.; Zhang, S.; Shaheen, S.; Li, J.; Wang, L.; Yue, C.; et al. Through an ITIM-independent mechanism the FcγRIIB blocks B cell activation by disrupting the colocalized microclustering of the B cell receptor and CD19. J. Immunol. 2014, 192, 5179–5191. [Google Scholar] [CrossRef]
- Willcocks, L.C.; Smith, K.G.C.; Clatworthy, M.R. Low-affinity Fcgamma receptors, autoimmunity and infection. Expert Rev. Mol. Med. 2009, 11, e24. [Google Scholar] [CrossRef]
- Clatworthy, M.R.; Willcocks, L.; Urban, B.; Langhorne, J.; Williams, T.N.; Peshu, N.; Watkins, N.A.; Floto, R.A.; Smith, K.G.C. Systemic lupus erythematosus-associated defects in the inhibitory receptor FcgammaRIIb reduce susceptibility to malaria. Proc. Natl. Acad. Sci. USA 2007, 104, 7169–7174. [Google Scholar] [CrossRef]
- Gelabert, P.; Olalde, I.; de-Dios, T.; Civit, S.; Lalueza-Fox, C. Malaria was a weak selective force in ancient Europeans. Sci. Rep. 2017, 7, 1377. [Google Scholar] [CrossRef]
- Willcocks, L.C.; Carr, E.J.; Niederer, H.A.; Rayner, T.F.; Williams, T.N.; Yang, W.; Scott, J.A.G.; Urban, B.C.; Peshu, N.; Vyse, T.J.; et al. A defunctioning polymorphism in FCGR2B is associated with protection against malaria but susceptibility to systemic lupus erythematosus. Proc. Natl. Acad. Sci. USA 2010, 107, 7881–7885. [Google Scholar] [CrossRef]
- Ebonwu, J.; Lassaunière, R.; Paximadis, M.; Strehlau, R.; Gray, G.E.; Kuhn, L.; Tiemessen, C.T. FCGR3A gene duplication, FcγRIIb-232TT and FcγRIIIb-HNA1a associate with an increased risk of vertical acquisition of HIV-1. PLoS ONE 2022, 17, e0273933. [Google Scholar] [CrossRef]
- Hu, W.; Zhang, Y.; Sun, X.; Zhang, T.; Xu, L.; Xie, H.; Li, Z.; Liu, W.; Lou, J.; Chen, W. FcγRIIB-I232T polymorphic change allosterically suppresses ligand binding. Elife 2019, 8, e46689. [Google Scholar] [CrossRef] [PubMed]
- GTEx Consortium The Genotype-Tissue Expression (GTEx) project. Nat. Genet. 2013, 45, 580–585. [CrossRef] [PubMed]
- Lu, H.; Ma, L.; Quan, C.; Li, L.; Lu, Y.; Zhou, G.; Zhang, C. RegVar: Tissue-specific Prioritization of Noncoding Regulatory Variants. Genom. Proteom. Bioinform. 2023, 21, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Ota, M.; Nagafuchi, Y.; Hatano, H.; Ishigaki, K.; Terao, C.; Takeshima, Y.; Yanaoka, H.; Kobayashi, S.; Okubo, M.; Shirai, H.; et al. Dynamic landscape of immune cell-specific gene regulation in immune-mediated diseases. Cell 2021, 184, 3006–3021.e17. [Google Scholar] [CrossRef] [PubMed]
- Kerimov, N.; Hayhurst, J.D.; Peikova, K.; Manning, J.R.; Walter, P.; Kolberg, L.; Samoviča, M.; Sakthivel, M.P.; Kuzmin, I.; Trevanion, S.J.; et al. A compendium of uniformly processed human gene expression and splicing quantitative trait loci. Nat. Genet. 2021, 53, 1290–1299. [Google Scholar] [CrossRef] [PubMed]
- Calderon, D.; Nguyen, M.L.T.; Mezger, A.; Kathiria, A.; Müller, F.; Nguyen, V.; Lescano, N.; Wu, B.; Trombetta, J.; Ribado, J.V.; et al. Landscape of stimulation-responsive chromatin across diverse human immune cells. Nat. Genet. 2019, 51, 1494–1505. [Google Scholar] [CrossRef]
- Hammal, F.; de Langen, P.; Bergon, A.; Lopez, F.; Ballester, B. ReMap 2022: A database of Human, Mouse, Drosophila and Arabidopsis regulatory regions from an integrative analysis of DNA-binding sequencing experiments. Nucleic Acids Res. 2022, 50, D316–D325. [Google Scholar] [CrossRef]
- Sloan, C.A.; Chan, E.T.; Davidson, J.M.; Malladi, V.S.; Strattan, J.S.; Hitz, B.C.; Gabdank, I.; Narayanan, A.K.; Ho, M.; Lee, B.T.; et al. ENCODE data at the ENCODE portal. Nucleic Acids Res. 2016, 44, D726–D732. [Google Scholar] [CrossRef]
- Lassaunière, R.; Tiemessen, C.T. Variability at the FCGR locus: Characterization in Black South Africans and evidence for ethnic variation in and out of Africa. Genes. Immun. 2016, 17, 93–104. [Google Scholar] [CrossRef]
- Tai, K.Y.; Dhaliwal, J.; Balasubramaniam, V. Leveraging Mann-Whitney U test on large-scale genetic variation data for analysing malaria genetic markers. Malar. J. 2022, 21, 79. [Google Scholar] [CrossRef]
- Cherif, M.K.; Sanou, G.S.; Bougouma, E.C.; Diarra, A.; Ouédraogo, A.; Dolo, A.; Troye-Blomberg, M.; Cavanagh, D.R.; Theisen, M.; Modiano, D.; et al. Is Fc gamma receptor IIA (FcγRIIA) polymorphism associated with clinical malaria and Plasmodium falciparum specific antibody levels in children from Burkina Faso? Acta Trop. 2015, 142, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Dwomoh, D.; Adu, B.; Dodoo, D.; Theisen, M.; Iddi, S.; Gerds, T.A. Evaluating the predictive performance of malaria antibodies and FCGR3B gene polymorphisms on Plasmodium falciparum infection outcome: A prospective cohort study. Malar. J. 2020, 19, 307. [Google Scholar] [CrossRef] [PubMed]
- Adu, B.; Jepsen, M.P.G.; Gerds, T.A.; Kyei-Baafour, E.; Christiansen, M.; Dodoo, D.; Theisen, M. Fc gamma receptor 3B (FCGR3B-c.233C>A-rs5030738) polymorphism modifies the protective effect of malaria specific antibodies in Ghanaian children. J. Infect. Dis. 2014, 209, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Fall, A.K.D.J.; Dechavanne, C.; Sabbagh, A.; Garcia, A.; Courtin, D.; Migot-Nabias, F. Combined polymorphisms involving the IgG heavy chain and Fc gamma receptors among Fulani and non-Fulani in Benin: Implications for the natural protection of young Fulani against Plasmodium falciparum malaria infections. Infect. Genet. Evol. 2023, 112, 105461. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, A.; Garnier, S.; Afridi, S.; Fumoux, F.; Rihet, P. Genetic variations in genes involved in heparan sulphate biosynthesis are associated with Plasmodium falciparum parasitaemia: A familial study in Burkina Faso. Malar. J. 2012, 11, 108. [Google Scholar] [CrossRef] [PubMed]
- Rihet, P.; Abel, L.; Traoré, Y.; Traoré-Leroux, T.; Aucan, C.; Fumoux, F. Human malaria: Segregation analysis of blood infection levels in a suburban area and a rural area in Burkina Faso. Genet. Epidemiol. 1998, 15, 435–450. [Google Scholar] [CrossRef]
- Machiela, M.J.; Chanock, S.J. LDlink: A web-based application for exploring population-specific haplotype structure and linking correlated alleles of possible functional variants. Bioinformatics 2015, 31, 3555–3557. [Google Scholar] [CrossRef]
- Abecasis, G.R.; Cardon, L.R.; Cookson, W.O. A general test of association for quantitative traits in nuclear families. Am. J. Hum. Genet. 2000, 66, 279–292. [Google Scholar] [CrossRef]
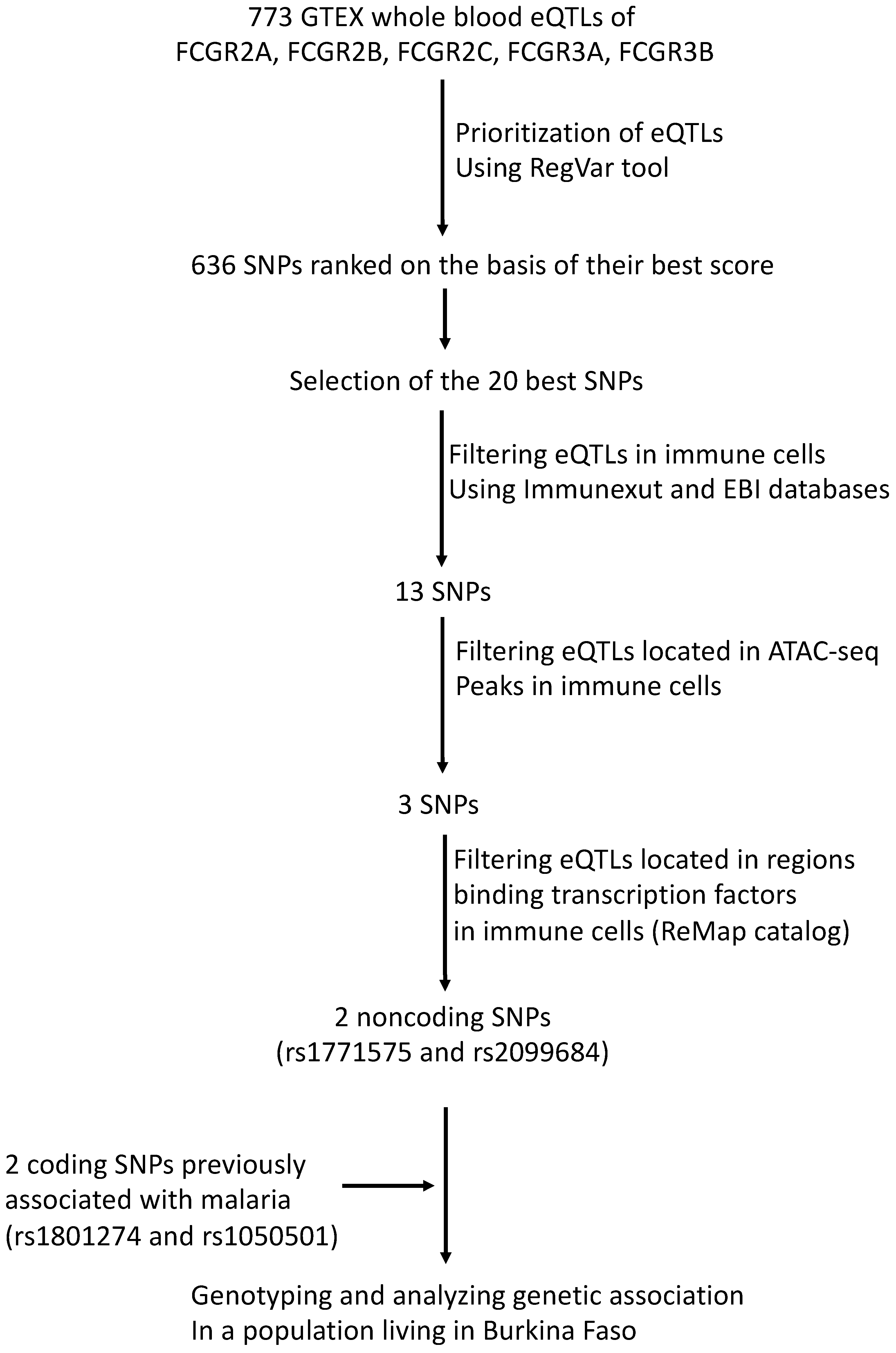

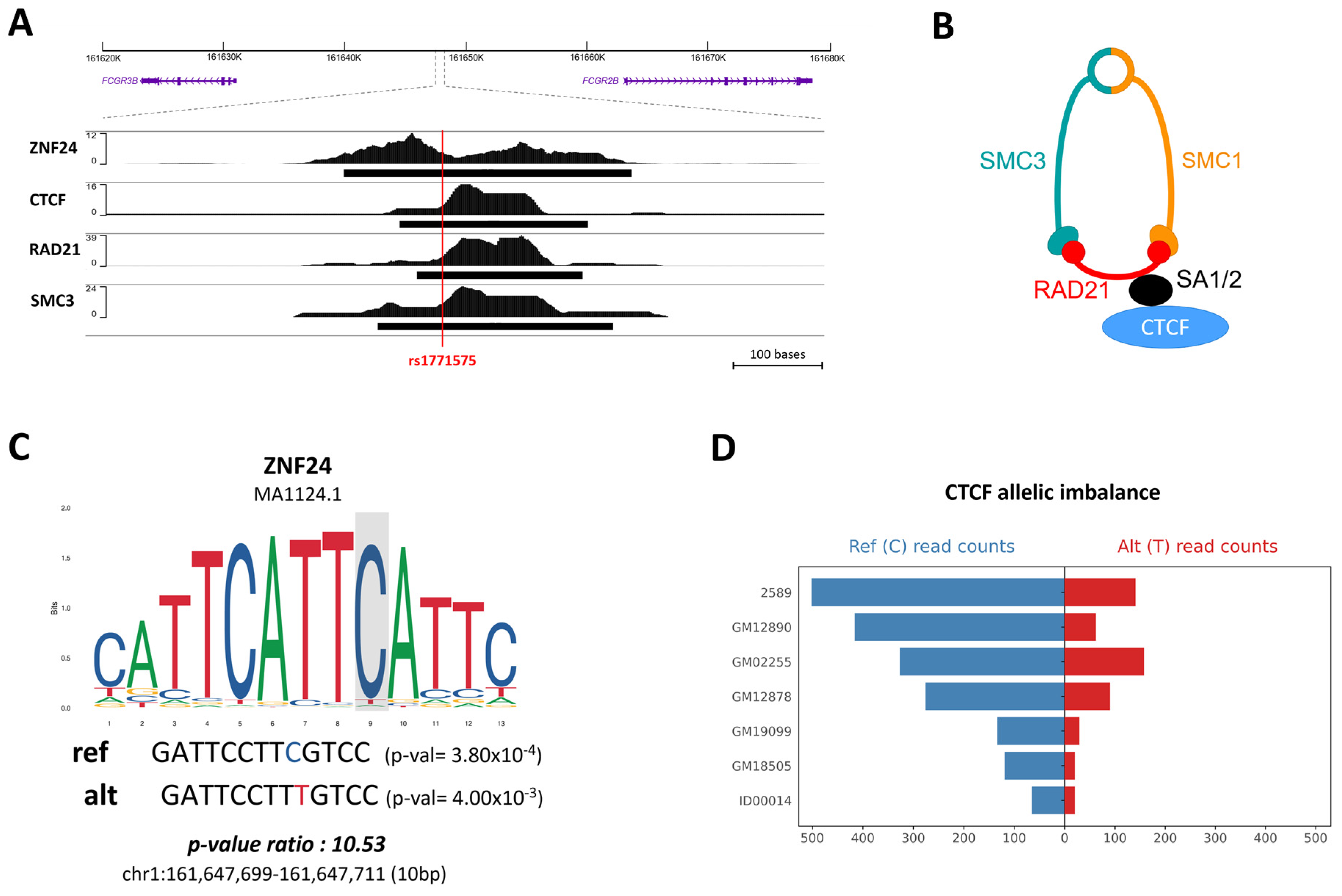

| Functional Regulatory Annotation | ||||||
|---|---|---|---|---|---|---|
| SNP | Position a | Annotation b | % Genotyped | ATAC-Seq c | ReMap c | Encode Regulatory Element |
| rs1771575 (T > C) | 161647707 | intergenic | 100.0 | DC, Mo, NK, B | Mo, Ma, N, B | E1391464 |
| rs2099684 (A > G) | 161530340 | intergenic | 96.5 | DC, NK | Mo, B | E1391397 |
| rs1801274 (G > A) | 161509955 | FCGR2A exon, missense H-R | 97.4 | - | - | - |
| rs1050501 (T > C) | 161674008 | FCGR2B exon, missense I-N | 97.5 | - | - | - |
| eQTLs from ImmuNexUT Database | eQTLs from EBI Catalog | |||||
|---|---|---|---|---|---|---|
| Target Gene | Cell Type | p Value a | Target Gene | Cell Type | p Value b | |
| rs1771575 | FCGR2B | Neutrophils | 3.10 × 10−38 | FCGR2B | Neutrophils | 1.15 × 10−10 |
| FCGR3A | Neutrophils | 6.02 × 10−26 | FCGR2B | Monocyte, naive | 9.77 × 10−7 | |
| FCGR2C | Dendritic cells | 2.77 × 10−17 | - | - | - | |
| FCGR3B | NK cells | 1.16 × 10−5 | - | - | - | |
| FCGR2A | Dendritic cells | 2.87 × 10−4 | - | - | - | |
| rs2099684 | FCGR3B | NK cells | 8.54 × 10−78 | FCGR3B | Monocyte, naive | 6.31 × 10−23 |
| FCGR2C | NK cells | 2.63 × 10−17 | FCGR3B | Monocyte, LPS | 1.35 × 10−18 | |
| FCGR2A | Neutrophils | 6.68 × 10−15 | FCGR3B | Monocyte, IAV | 3.24 × 10−18 | |
| FCGR2B | Neutrophils | 1.227 × 10−5 | FCGR3B | Monocyte | 3.55 × 10−17 | |
| - | - | - | FCGR3B | NK-cell, naive | 4.68 × 10−16 | |
| - | - | - | FCGR3B | Macrophage, Salmonella | 1.78 × 10−13 | |
| - | - | - | FCGR3B | Macrophage, naive | 5.62 × 10−13 | |
| - | - | - | FCGR3B | Macrophage, Listeria | 6.17 × 10−13 | |
| - | - | - | FCGR3B | CD16 Monocyte, naive | 1.07 × 10−11 | |
| Parents | Children | |
|---|---|---|
| (N = 98) | (N = 136) | |
| Gender | ||
| N | 98 | 136 |
| Female | 65 | 71 |
| Male | 33 | 65 |
| Ethnic group: Bobo a N | 98 | 136 |
| Age | ||
| N | 98 | 136 |
| Median | 39 | 8 |
| (25–75th percentile) | (31–45) | (6–12) |
| Number of parasitaemia measurements per subject | ||
| N | 98 | 136 |
| Median | 18 | 20 |
| (25th and 75th percentile) | (13–23) | (14–24) |
| Phenotypes related to infection levels (PrevPfI) | ||
| N | 98 | 136 |
| Median | 0.14 | 0.57 |
| (25th and 75th percentile) | (0.07–0.22) | (0.43–0.75) |
| Explanatory Variable: PrevPfI a | Univariate Analysis | Linear Multiple Regression Adjusted to Age | Family Based Linear Mixed Model | ||
|---|---|---|---|---|---|
| p | b Coefficient (IC) | p | b Coefficient (IC) | p | |
| b Recessive Model | |||||
| Children age | <0.001 * | −0.0225 [−0.0276; −0.0175] | <0.001 * | −0.023 (−0.028; −0.018) | <0.001 * |
| rs1771575-CC vs. CT/TT | 0.012 * | 0.0883 [0.0177; 0.159] | 0.015 * | 0.087 (0.017; 0.158) | 0.016 * |
| c Additive Model | |||||
| Children age | <0.001 * | −0.0227 [−0.0277; −0.0177] | <0.001 * | −0.023 (−0.028; −0.018) | <0.001 * |
| rs1771575- CC vs. -CT vs. -TT | 0.0252 * | −0.0525 [−0.0941; −0.0108] | 0.014 * | −0.052 (−0.094; −0.010) | 0.016 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cretin, J.; Adjemout, M.; Dieppois, C.; Gallardo, F.; Torres, M.; Merard, Z.; Sawadogo, S.A.; Picard, C.; Rihet, P.; Paul, P. A Non-Coding Fc Gamma Receptor Cis-Regulatory Variant within the 1q23 Gene Cluster Is Associated with Plasmodium falciparum Infection in Children Residing in Burkina Faso. Int. J. Mol. Sci. 2023, 24, 15711. https://doi.org/10.3390/ijms242115711
Cretin J, Adjemout M, Dieppois C, Gallardo F, Torres M, Merard Z, Sawadogo SA, Picard C, Rihet P, Paul P. A Non-Coding Fc Gamma Receptor Cis-Regulatory Variant within the 1q23 Gene Cluster Is Associated with Plasmodium falciparum Infection in Children Residing in Burkina Faso. International Journal of Molecular Sciences. 2023; 24(21):15711. https://doi.org/10.3390/ijms242115711
Chicago/Turabian StyleCretin, Jules, Mathieu Adjemout, Christelle Dieppois, Frederic Gallardo, Magali Torres, Zachary Merard, Serge Aimé Sawadogo, Christophe Picard, Pascal Rihet, and Pascale Paul. 2023. "A Non-Coding Fc Gamma Receptor Cis-Regulatory Variant within the 1q23 Gene Cluster Is Associated with Plasmodium falciparum Infection in Children Residing in Burkina Faso" International Journal of Molecular Sciences 24, no. 21: 15711. https://doi.org/10.3390/ijms242115711





