APOE ε4-Allele in Middle-Aged and Older Autistic Adults: Associations with Verbal Learning and Memory
Abstract
:1. Introduction
2. Results
3. Discussion
Limitations
4. Methods and Materials
4.1. Participants
4.2. Inclusion/Exclusion Criteria
4.3. Verbal Learning and Memory
4.4. APOE Genotype
4.5. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ASD | Autism spectrum disorder |
| NT | Neurotypical |
| MA+ | Middle-aged and older |
| Alz | Alzheimer’s disease |
| ADOS-2 | Autism Diagnostic Observation Schedule-2 |
| PCR | Polymerase chain reaction |
| A1 | Short-term memory |
| A1–A5 | Learning |
| A7 | Long-term memory |
| ANOVA | Analysis of Variance |
| SARRC | Southwest Autism Research & Resource Center |
| SRS-2 | Social Responsiveness Scale-2 Adult Self-Report |
| MMSE | Mini Mental State Exam |
| KBIT-2 | Kaufman Brief Intelligence Test-2 |
| DSM-5 | Diagnostic and Statistical Manual of Mental Disorders-5 |
| AVLT | Rey Auditory Verbal Learning Test |
| DNA | Deoxyribonucleic acid |
| IQ | Intelligence Quotient |
| SE | Standard error |
References
- Piven, J.; Rabins, P. Autism-in-Older Adults Working Group. Autism spectrum disorders in older adults: Toward defining a research agenda. J. Am. Geriatr. Soc. 2011, 59, 2151–2155. [Google Scholar] [CrossRef] [PubMed]
- Hodges, H.; Fealko, C.; Soares, N. Autism spectrum disorder: Definition, epidemiology, causes, and clinical evaluation. Transl. Pediatr. 2020, 9, S55–S65. [Google Scholar] [CrossRef] [PubMed]
- LaSalle, J. Epigenomic signatures reveal mechanistic clues and predictive markers for autism spectrum disorder. Mol. Psychiatry 2023, 28, 1890–1901. [Google Scholar] [CrossRef] [PubMed]
- Maenner, M.J. Prevalence and Characteristics of Autism Spectrum Disorder among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2020. MMWR Surveill Summ. 2023; Volume 72. Available online: https://wwwdev.cdc.gov/mmwr/volumes/72/ss/ss7202a1.htm (accessed on 20 September 2023).
- Braden, B.B.; Smith, C.J.; Thompson, A.; Glaspy, T.K.; Wood, E.; Vatsa, D.; Abbott, A.E.; McGee, S.C.; Baxter, L.C. Executive function and functional and structural brain differences in middle-age adults with autism spectrum disorder. Autism Res. 2017, 10, 1945–1959. [Google Scholar] [CrossRef]
- Vivanti, G.; Tao, S.; Lyall, K.; Robins, D.L.; Shea, L.L. The prevalence and incidence of early-onset dementia among adults with autism spectrum disorder. Autism Res. Off J. Int. Soc. Autism Res. 2021, 14, 2189–2199. [Google Scholar] [CrossRef]
- Croen, L.A.; Zerbo, O.; Qian, Y.; Massolo, M.L.; Rich, S.; Sidney, S.; Kripke, C. The health status of adults on the autism spectrum. Autism Int. J. Res. Pract. 2015, 19, 814–823. [Google Scholar] [CrossRef]
- Klein, C.B.; McQuaid, G.A.; Charlton, R.A.; Klinger, L.G.; Wallace, G.L. Self-reported cognitive decline among middle and older age autistic adults. Autism Res. Off J. Int. Soc. Autism Res. 2023, 16, 605–616. [Google Scholar] [CrossRef] [PubMed]
- DeTure, M.A.; Dickson, D.W. The neuropathological diagnosis of Alzheimer’s disease. Mol. Neurodegener. 2019, 14, 32. [Google Scholar] [CrossRef]
- Kumar, A.; Sidhu, J.; Goyal, A.; Tsao, J.W. Alzheimer Disease; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: http://www.ncbi.nlm.nih.gov/books/NBK499922/ (accessed on 20 September 2023).
- Hand, B.N.; Angell, A.M.; Harris, L.; Carpenter, L.A. Prevalence of physical and mental health conditions in Medicare-enrolled, autistic older adults. Autism Int. J. Res. Pract. 2020, 24, 755–764. [Google Scholar] [CrossRef]
- Nadeem, M.S.; Hosawi, S.; Alshehri, S.; Ghoneim, M.M.; Imam, S.S.; Murtaza, B.N.; Kazmi, I. Symptomatic, Genetic, and Mechanistic Overlaps between Autism and Alzheimer’s Disease. Biomolecules 2021, 11, 1635. [Google Scholar] [CrossRef] [PubMed]
- Pagni, B.A.; Walsh, M.J.M.; Ofori, E.; Chen, K.; Sullivan, G.; Alvar, J.; Monahan, L.; Guerithault, N.; Delaney, S.; Braden, B.B. Effects of age on the hippocampus and verbal memory in adults with autism spectrum disorder: Longitudinal versus cross-sectional findings. Autism Res. 2022, 15, 1810–1823. [Google Scholar] [CrossRef]
- Walsh, M.J.M.; Ofori, E.; Pagni, B.A.; Chen, K.; Sullivan, G.; Braden, B.B. Preliminary findings of accelerated visual memory decline and baseline brain correlates in middle-age and older adults with autism: The case for hippocampal free-water. Front. Aging Neurosci. 2022, 14, 1029166. [Google Scholar] [CrossRef]
- Foraker, J.; Millard, S.P.; Leong, L.; Thomson, Z.; Chen, S.; Keene, C.D.; Bekris, L.M.; Yu, C.-E. The APOE Gene is Differentially Methylated in Alzheimer’s Disease. J. Alzheimer’s Dis. 2015, 48, 745–755. [Google Scholar] [CrossRef]
- Walker, L.; Stefanis, L.; Attems, J. Clinical and neuropathological differences between Parkinson’s disease, Parkinson’s disease dementia and dementia with Lewy bodies—Current issues and future directions. J. Neurochem. 2019, 150, 467–474. [Google Scholar] [CrossRef]
- Raulin, A.-C.; Doss, S.V.; Trottier, Z.A.; Ikezu, T.C.; Bu, G.; Liu, C.-C. ApoE in Alzheimer’s disease: Pathophysiology and therapeutic strategies. Mol. Neurodegener. 2022, 17, 72. [Google Scholar] [CrossRef]
- Corder, E.H.; Saunders, A.M.; Strittmatter, W.J.; Schmechel, D.E.; Gaskell, P.C.; Small, G.W.; Roses, A.D.; Haines, J.L.; Pericak-Vance, M.A. Gene Dose of Apolipoprotein E Type 4 Allele and the Risk of Alzheimer’s Disease in Late Onset Families. Science 1993, 261, 921–923. [Google Scholar] [CrossRef] [PubMed]
- Saunders, A.M.; Strittmatter, W.J.; Schmechel, D.; George-Hyslop, P.H.S.; Pericak-Vance, M.A.; Joo, S.H.; Rosi, B.L.; Gusella, J.F.; Crapper-MacLachlan, D.R.; Alberts, M.J.; et al. Association of apolipoprotein E allele ϵ4 with late-onset familial and sporadic Alzheimer’s disease. Neurology 1993, 43, 1467. [Google Scholar] [CrossRef] [PubMed]
- Giunco, C.T.; de Oliveira, A.B.; Carvalho-Salles, A.B.; Souza, D.S.R.; Silva, A.E.; da Rocha, S.S.; Fett-Conte, A.C. Association between APOE polymorphisms and predisposition for autism. Psychiatr. Genet. 2009, 19, 338. [Google Scholar] [CrossRef] [PubMed]
- Raiford, K.L.; Shao, Y.; Allen, I.C.; Martin, E.R.; Menold, M.M.; Wright, H.H.; Abramson, R.K.; Worley, G.; DeLong, G.R.; Vance, J.M.; et al. No association between the APOE gene and autism. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2004, 125B, 57–60. [Google Scholar] [CrossRef]
- Caldwell, J.Z.K.; Berg, J.-L.; Cummings, J.L.; Banks, S.J.; Alzheimer’s Disease Neuroimaging Initiative. Moderating effects of sex on the impact of diagnosis and amyloid positivity on verbal memory and hippocampal volume. Alzheimer’s Res. Ther. 2017, 9, 72. [Google Scholar] [CrossRef]
- Emrani, S.; Arain, H.A.; DeMarshall, C.; Nuriel, T. APOE4 is associated with cognitive and pathological heterogeneity in patients with Alzheimer’s disease: A systematic review. Alzheimer’s Res. Ther. 2020, 12, 141. [Google Scholar] [CrossRef]
- McCaulley, M.E. Autism spectrum disorder and mercury toxicity: Use of genomic and epigenetic methods to solve the etiologic puzzle. Acta Neurobiol. Exp. 2019, 79, 113–125. [Google Scholar] [CrossRef]
- Sundermann, E.E.; Maki, P.M.; Rubin, L.H.; Lipton, R.B.; Landau, S.; Biegon, A.; Alzheimer’s Disease Neuroimaging Initiative. Female advantage in verbal memory: Evidence of sex-specific cognitive reserve. Neurology 2016, 87, 1916–1924. [Google Scholar] [CrossRef]
- Sundermann, E.E.; Tran, M.; Maki, P.M.; Bondi, M.W. Sex differences in the association between apolipoprotein E ε4 allele and Alzheimer’s disease markers. Alzheimer’s Dementia Diagn. Assess. Dis. Monit. 2018, 10, 438–447. [Google Scholar] [CrossRef]
- Beinhoff, U.; Tumani, H.; Brettschneider, J.; Bittner, D.; Riepe, M.W. Gender-specificities in Alzheimer’s disease and mild cognitive impairment. J. Neurol. 2008, 255, 117–122. [Google Scholar] [CrossRef]
- Sundermann, E.E.; Biegon, A.; Rubin, L.H.; Lipton, R.B.; Mowrey, W.; Landau, S.; Maki, P.M.; Alzheimer’s Disease Neuroimaging Initiative. Better verbal memory in women than men in MCI despite similar levels of hippocampal atrophy. Neurology 2016, 86, 1368–1376. [Google Scholar] [CrossRef] [PubMed]
- Brandon, J.A.; Farmer, B.C.; Williams, H.C.; Johnson, L.A. APOE and Alzheimer’s Disease: Neuroimaging of Metabolic and Cerebrovascular Dysfunction. Front. Aging Neurosci. 2018, 10, 180. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.R.; Garthwaite, P.H. Comparison of a single case to a control or normative sample in neuropsychology: Development of a Bayesian approach. Cogn. Neuropsychol. 2007, 24, 343–372. [Google Scholar] [CrossRef]
- Duarte-Guterman, P.; Albert, A.Y.; Barha, C.K.; Galea, L.A.M.; on behalf of the Alzheimer’s Disease Neuroimaging Initiative. Sex influences the effects of APOE genotype and Alzheimer’s diagnosis on neuropathology and memory. Psychoneuroendocrinology 2021, 129, 105248. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.L.; Dowell, N.G.; Prowse, F.; Tabet, N.; King, S.L.; Rusted, J.M. Mid age APOE ε4 carriers show memory-related functional diferences and disrupted structure-function relationships in hippocampal regions. Sci. Rep. 2020, 10, a3110. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Pardo, L.M.; Schuur, M.; Sanchez-Juan, P.; Isaacs, A.; Sleegers, K.; de Koning, I.; Zorkoltseva, I.V.; Axenovich, T.I.; Witteman, J.C.; et al. The apolipoprotein E gene and its age-specific effects on cognitive function. Neurobiol. Aging 2010, 31, 1831–1833. [Google Scholar] [CrossRef]
- Sauty, B.; Durrleman, S. Impact of Sex and APOE-ε4 Genotype on Patterns of Regional Brain Atrophy in Alzheimer’s Disease and Healthy Aging. Front. Neurol. 2023, 14. Available online: https://www.frontiersin.org/articles/10.3389/fneur.2023.1161527 (accessed on 26 September 2023). [CrossRef] [PubMed]
- Zokaei, N.; Giehl, K.; Sillence, A.; Neville, M.J.; Karpe, F.; Nobre, A.C.; Husain, M. Sex and APOE: A memory advantage in male APOE ε4 carriers in midlife. Cortex 2017, 88, 98–105. [Google Scholar] [CrossRef]
- Flory, J.D.; Manuck, S.B.; Ferrell, R.E.; Ryan, C.M.; Muldoon, M.F. Memory performance and the apolipoprotein E polymorphism in a community sample of middle-aged adults. Am. J. Med. Genet. 2000, 96, 707–711. [Google Scholar] [CrossRef]
- Caselli, R.J.; Dueck, A.C.; Locke, D.E.C.; Baxter, L.C.; Woodruff, B.K.; Geda, Y.E. Sex-Based Memory Advantages and Cognitive Aging: A Challenge to the Cognitive Reserve Construct? J. Int. Neuropsychol. Soc. 2015, 21, 95–104. [Google Scholar] [CrossRef]
- Altmann, A.; Tian, L.; Henderson, V.W.; Greicius, M.D.; Investigators, A.D.N.I. Sex modifies the APOE-related risk of developing Alzheimer disease. Ann. Neurol. 2014, 75, 563–573. [Google Scholar] [CrossRef]
- Williams, O.A.; An, Y.; Armstrong, N.M.; Shafer, A.T.; Helphrey, J.; Kitner-Triolo, M.; Ferrucci, L.; Resnick, S.M. Apolipoprotein E ε4 allele effects on longitudinal cognitive trajectories are sex- and age-dependent. Alzheimer’s Dement. 2019, 15, 1558–1567. [Google Scholar] [CrossRef]
- Payami, H.; Zareparsi, S.; Montee, K.R.; Sexton, G.J.; Kaye, J.A.; Bird, T.D.; Yu, C.E.; Wijsman, E.M.; Heston, L.L.; Litt, M.; et al. Gender difference in apolipoprotein E-associated risk for familial Alzheimer disease: A possible clue to the higher incidence of Alzheimer disease in women. Am. J Hum. Genet. 1996, 58, 803–811. [Google Scholar]
- Demetriou, E.A.; Pepper, K.L.; Park, S.H.; Pellicano, L.; Song, Y.J.C.; Naismith, S.L.; Hickie, I.B.; E Thomas, E.; Guastella, A.J. Autism spectrum disorder: An examination of sex differences in neuropsychological and self-report measures of executive and non-executive cognitive function. Autism Int. J. Res. Pract. 2021, 25, 2223–2237. [Google Scholar] [CrossRef] [PubMed]
- Lai, M.-C.; Lombardo, M.V.; Ruigrok, A.N.V.; Chakrabarti, B.; Wheelwright, S.J.; Auyeung, B.; Allison, C.; Baron-Cohen, S. Cognition in Males and Females with Autism: Similarities and Differences. PLoS ONE 2012, 7, e47198. [Google Scholar] [CrossRef] [PubMed]
- Riedel, B.C.; Thompson, P.M.; Brinton, R.D. Age, APOE and Sex: Triad of Risk of Alzheimer’s Disease. J. Steroid Biochem. Mol. Biol. 2016, 160, 134–147. [Google Scholar] [CrossRef]
- Baxter, L.C.; Nespodzany, A.; Walsh, M.J.M.; Wood, E.; Smith, C.J.; Braden, B.B. The influence of age and ASD on verbal fluency networks. Res. Autism Spectr. Disord. 2019, 63, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Braden, B.B.; Riecken, C. Thinning faster? Age-related cortical thickness differences in adults with autism spectrum disorder. Res. Autism Spectr. Disord. 2019, 64, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, A.S.; Kaufman, N.L. Kaufman Brief Intelligence Test|Second Edition. 2004. Available online: https://www.pearsonassessments.com/store/usassessments/en/Store/Professional-Assessments/Cognition-%26-Neuro/Non-Verbal-Ability/Kaufman-Brief-Intelligence-Test-%7C-Second-Edition/p/100000390.html (accessed on 29 July 2023).
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”: A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Lever, A.G.; Geurts, H.M. Psychiatric Co-occurring Symptoms and Disorders in Young, Middle-Aged, and Older Adults with Autism Spectrum Disorder. J. Autism Dev. Disord. 2016, 46, 1916–1930. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M. Rey Auditory Verbal Learning Test: RAVLT: A handbook; Western Psychological Services: Los Angeles, CA, USA, 1996. [Google Scholar]
- Canitano, R.; Vivanti, G. Tics and Tourette syndrome in autism spectrum disorders. Autism Int. J. Res. Pract. 2007, 11, 19–28. [Google Scholar] [CrossRef]
- Gjevik, E.; Eldevik, S.; Fjæran-Granum, T.; Sponheim, E. Kiddie-SADS Reveals High Rates of DSM-IV Disorders in Children and Adolescents with Autism Spectrum Disorders. J. Autism Dev. Disord. 2011, 41, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Joshi, G.; Petty, C.; Wozniak, J.; Henin, A.; Fried, R.; Galdo, M.; Kotarski, M.; Walls, S.; Biederman, J. The heavy burden of psychiatric comorbidity in youth with autism spectrum disorders: A large comparative study of a psychiatrically referred population. J. Autism Dev. Disord. 2010, 40, 1361–1370. [Google Scholar] [CrossRef] [PubMed]
- Simonoff, E.; Pickles, A.; Charman, T.; Chandler, S.; Loucas, T.; Baird, G. Psychiatric Disorders in Children with Autism Spectrum Disorders: Prevalence, Comorbidity, and Associated Factors in a Population-Derived Sample. J. Am. Acad. Child. Adolesc. Psychiatry 2008, 47, 921–929. [Google Scholar] [CrossRef]
- Lehnhardt, F.-G.; Falter, C.M.; Gawronski, A.; Pfeiffer, K.; Tepest, R.; Franklin, J.; Vogeley, K. Sex-Related Cognitive Profile in Autism Spectrum Disorders Diagnosed Late in Life: Implications for the Female Autistic Phenotype. J. Autism Dev. Disord. 2016, 46, 139–154. [Google Scholar] [CrossRef]
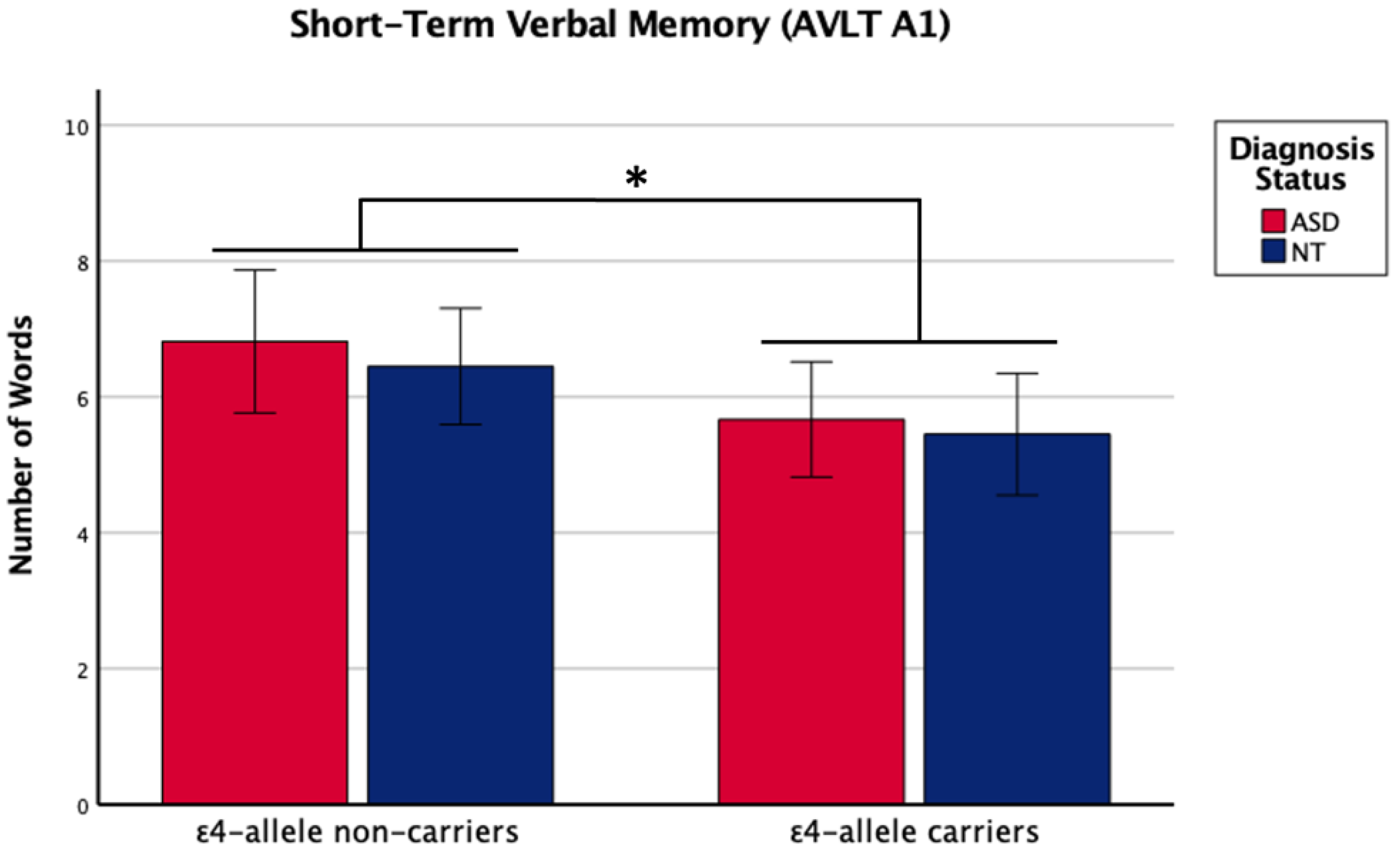


| DF | t-Value | p-Value | F | Partial Eta Squared (Effect Size) | |
|---|---|---|---|---|---|
| Short-Term Memory (AVLT A1) | |||||
| Diagnosis | 1, 71 | 0.352 | 0.526 | 0.406 | 0.006 |
| APOE ε4-allele | 1, 71 | 1.586 | 0.025 * | 5.247 | 0.069 |
| Diagnosis∗APOE ε4-allele | 1, 71 | 0.163 | 0.871 | 0.027 | n/a |
| Sex | 1, 71 | 1.37 | 0.177 | 1.863 | 0.026 |
| Total Words (AVLT A1-A5) | |||||
| Diagnosis | 1, 71 | −0.851 | 0.467 | 0.534 | 0.007 |
| APOE ε4-allele | 1, 71 | 1.783 | 0.006 * | 7.867 | 0.100 |
| Diagnosis∗APOE ε4-allele | 1, 71 | 0.420 | 0.676 | 0.176 | 0.002 |
| Sex | 1, 71 | 3.945 | <0.001 * | 15.563 | 0.180 |
| Long-Term Memory (AVLT A7) | |||||
| Diagnosis | 1, 71 | −1.342 | 0.195 | 1.710 | 0.024 |
| APOE ε4-allele | 1, 71 | 0.512 | 0.212 | 1.587 | 0.022 |
| Diagnosis∗APOE ε4-allele | 1, 71 | 0.574 | 0.571 | 0.324 | 0.005 |
| Sex | 1, 71 | 1.710 | 0.86 | 3.038 | 0.041 |
| DF | p-Value | F | Partial Eta Squared (Effect Size) | |
|---|---|---|---|---|
| ASD males | 1, 25 | 0.020 * | 6.183 | 0.198 |
| NT males | 1, 25 | 0.094 | 3.040 | 0.108 |
| NT females | 1, 12 | 0.318 | 1.087 | 0.083 |
| Bayesian Hypothesis Single Case Comparison Test for ASD Females. | ||||
| Case’s Test Score | Percentage of control population falling below case’s score | Effect Size | 95% Confidence Interval | |
| ASD Female Case 1 | 60 | 79.4571% | 0.969 | (−0.053 to 1.928) |
| ASD Female Case 2 | 37 | 6.6496% | −1.937 | (−3.321 to −0.502) |
| NT (n = 41) Mean (±SD) Range | ASD (n = 35) Mean (±SD) Range | Two-Group Comparison Statistics | NT APOE ε4 Carriers | NT APOE ε4 Non-Carriers | ASD APOE ε4 Carriers | ASD APOE ε4 Non-Carriers | Four-Group Comparison Statistics | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | 53.90 (±8.44) 40–70 | 53.06 (±8.91) 40–71 | t(74) = 0.424, p = 0.673 | 54.05 (±7.06) 41–65 | 53.76 (±9.75) 40–70 | 54.38 (±8.50) 41–71 | 51.07 (±9.44) 40–67 | t(75) = 0.234, p = 0.705 |
| Sex (M/F) | 27/14 | 27/8 | X2(1.76) = 1.170, p = 0.279 | 10/10 | 17/4 | 15/6 | 12/2 | X2(3.76) = 6.775, p = 0.079 |
| ADOS-2 a Social Affective | n/a | 10.03 (±3.12) (0–17) | n/a | n/a | n/a | 10.14 (±2.78) 7–17 | 9.86 (±3.68) 0–14 | n/a |
| Age at Diagnosis | n/a | 46.11 (±15.35) 2–67 | n/a | n/a | n/a | 48.62 (±11.74) 21–64 | 42.36 (±19.45) 2–67 | n/a |
| SRS-2 b Total t-score | 45.39 (±5.94) 37–60 | 71.64 (±11.55) 43–89 | t(45.435) = −11.854, p < 0.001 | 45.15 (±6.44) 37–59 | 45.62 (±5.57) 37–60 | 70.05 (±12.88) 43–89 | 74.08 (±9.07) 57–87 | t(73) = 26.752, p < 0.001 |
| MMSE c | 29.49 (±0.84) 26–30 | 29.06 (±1.11) 26–30 | t(62.626) = 1.775, p = 0.081 | 29.35 (±1.04) 26–30 | 29.57 (±0.598) 28–30 | 28.90 (±1.09) 27–30 | 29.29 (±1.14) 26–30 | t(75) = 0.849, p = 0.175 |
| KBIT-2 d Composite | 109.07 (±12.09) 85–141 | 108.97 (±14.52) 70–131 | t(73) = 0.033, p = 0.973 | 106.20 (±9.38) 85–124 | 111.81 (±13.88) 89–141 | 107.14 (±13.73) 70–131 | 111.92 (±15.82) 83–131 | t(74) = 0.487, p = 0.410 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harker, S.A.; Al-Hassan, L.; Huentelman, M.J.; Braden, B.B.; Lewis, C.R. APOE ε4-Allele in Middle-Aged and Older Autistic Adults: Associations with Verbal Learning and Memory. Int. J. Mol. Sci. 2023, 24, 15988. https://doi.org/10.3390/ijms242115988
Harker SA, Al-Hassan L, Huentelman MJ, Braden BB, Lewis CR. APOE ε4-Allele in Middle-Aged and Older Autistic Adults: Associations with Verbal Learning and Memory. International Journal of Molecular Sciences. 2023; 24(21):15988. https://doi.org/10.3390/ijms242115988
Chicago/Turabian StyleHarker, Samantha A., Lamees Al-Hassan, Matthew J. Huentelman, B. Blair Braden, and Candace R. Lewis. 2023. "APOE ε4-Allele in Middle-Aged and Older Autistic Adults: Associations with Verbal Learning and Memory" International Journal of Molecular Sciences 24, no. 21: 15988. https://doi.org/10.3390/ijms242115988
APA StyleHarker, S. A., Al-Hassan, L., Huentelman, M. J., Braden, B. B., & Lewis, C. R. (2023). APOE ε4-Allele in Middle-Aged and Older Autistic Adults: Associations with Verbal Learning and Memory. International Journal of Molecular Sciences, 24(21), 15988. https://doi.org/10.3390/ijms242115988








