Reduction of Osteoclastic Differentiation of Raw 264.7 Cells by EMF Exposure through TRPV4 and p-CREB Pathway
Abstract
:1. Introduction
2. Results
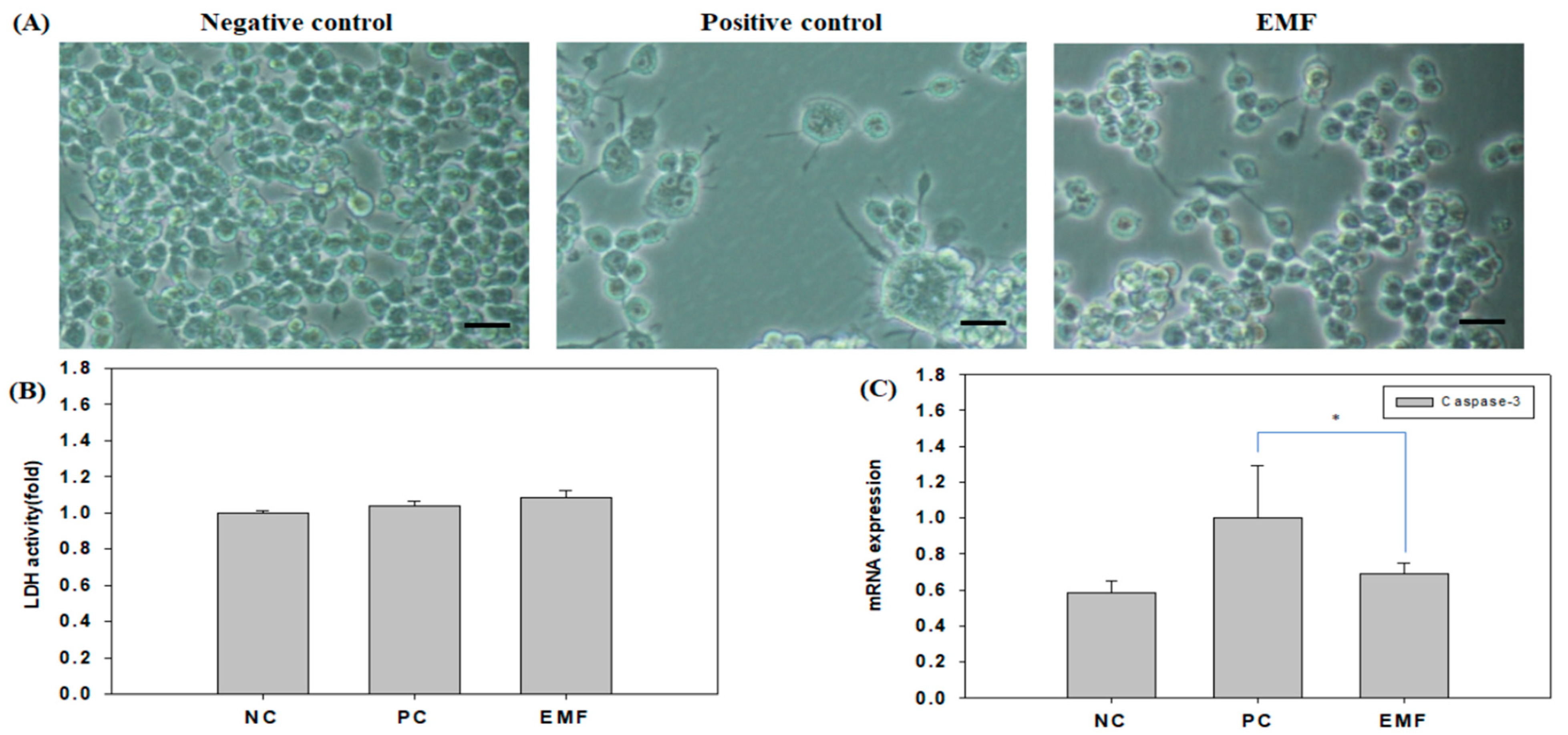
2.1. Evaluation of Cellular Stress upon EMF Exposure
2.2. Effect of EMF on Osteoclastic Differentiation and Cytoskeletal Organization
2.3. Analysis of Osteoclastic Differentiation-Related mRNA Expression
2.4. Effect of EMF Exposure on Proinflammatory and Inflammatory Cytokine Levels
2.5. TRPV4 and CREB Signaling Pathway Activity upon EMF Exposure
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. EMF Exposure
4.3. Lactate Dehydrogenase Activity Assay
4.4. Reverse Transcription Followed by Quantitative PCR
4.5. Western Blotting
4.6. Tartrate-Resistant Acid Phosphatase (TRAP) Staining
4.7. Immunofluorescence
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Klein-Nulend, J.; van Oers, R.F.; Bakker, A.D.; Bacabac, R.G. Bone cell mechanosensitivity, estrogen deficiency, and osteoporosis. J. Biomech. 2015, 48, 855–865. [Google Scholar] [CrossRef]
- Ma, J.; Fu, Q.; Wang, Z.; Zhou, P.; Qiao, S.; Wang, B.; Chen, A. Sodium hydrosulfide mitigates dexamethasone-induced osteoblast dysfunction by interfering with mitochondrial function. Biotechnol. Appl. Biochem. 2019, 66, 690–697. [Google Scholar] [CrossRef]
- Carey, J.J.; Buehring, B. Current imaging techniques in osteoporosis. Clin. Exp. Rheumatol. 2018, 36 (Suppl. 114), 115–126. [Google Scholar]
- Kang, H.K.; Park, C.Y.; Jung, S.Y.; Bin Jo, S.; Min, B.-M. A Vitronectin-Derived Peptide Restores Ovariectomy-Induced Bone Loss by Dual Regulation of Bone Remodeling. Tissue Eng. Regen. Med. 2022, 19, 1359–1376. [Google Scholar] [CrossRef]
- Rodan, G.A.; Martin, T.J. Therapeutic Approaches to Bone Diseases. Science 2000, 289, 1508–1514. [Google Scholar] [CrossRef] [PubMed]
- Teitelbaum, S.L. Bone resorption by osteoclasts. Science 2000, 289, 1504–1508. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, H.; Kido, S.; Tomomura, M.; Fujimoto, K.; Ohi, M.; Kiyomura, M.; Kanegae, H.; Inaba, A.; Sakagami, H.; Tomomura, A. Serum Calcium-decreasing Factor, Caldecrin, Inhibits Osteoclast Differentiation by Suppression of NFATc1 Activity. J. Biol. Chem. 2010, 285, 25448–25457. [Google Scholar] [CrossRef]
- Kim, M.S.; Yang, Y.-M.; Son, A.; Tian, Y.S.; Lee, S.-I.; Kang, S.W.; Muallem, S.; Shin, D.M. RANKL-mediated Reactive Oxygen Species Pathway That Induces Long Lasting Ca2+ Oscillations Essential for Osteoclastogenesis. J. Biol. Chem. 2010, 285, 6913–6921. [Google Scholar] [CrossRef]
- Clapham, D.E.; Runnels, L.W.; Strübing, C. The TRP ion channel family. Nat. Rev. Neurosci. 2001, 2, 387–396. [Google Scholar] [CrossRef]
- van der Eerden, B.C.J.; Hoenderop, J.G.J.; de Vries, T.J.; Schoenmaker, T.; Buurman, C.J.; Uitterlinden, A.G.; Pols, H.A.P.; Bindels, R.J.M.; van Leeuwen, J.P.T.M. The epithelial Ca2+ channel TRPV5 is essential for proper osteoclastic bone resorption. Proc. Natl. Acad. Sci. USA 2005, 102, 17507–17512. [Google Scholar] [CrossRef]
- Kajiya, H.; Okamoto, F.; Nemoto, T.; Kimachi, K.; Toh-Goto, K.; Nakayana, S.; Okabe, K. RANKL-induced TRPV2 expression regulates osteoclastogenesis via calcium oscillations. Cell Calcium 2010, 48, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Masuyama, R.; Mizuno, A.; Komori, H.; Kajiya, H.; Uekawa, A.; Kitaura, H.; Okabe, K.; Ohyama, K.; Komori, T. Calcium/calmodulin-signaling supports TRPV4 activation in osteoclasts and regulates bone mass. J. Bone Miner. Res. 2012, 27, 1708–1721. [Google Scholar] [CrossRef]
- Masuyama, R.; Vriens, J.; Voets, T.; Karashima, Y.; Owsianik, G.; Vennekens, R.; Lieben, L.; Torrekens, S.; Moermans, K.; Bosch, A.V. TRPV4-mediated calcium influx regulates terminal differentiation of osteoclasts. Cell Metab. 2008, 8, 257–265. [Google Scholar] [CrossRef]
- Epsley, S.; Tadros, S.; Farid, A.; Kargilis, D.; Mehta, S.; Rajapakse, C.S. The Effect of Inflammation on Bone. Front. Physiol. 2020, 11, 511799. [Google Scholar] [CrossRef]
- Boyce, B.E.; Li, P.; Yao, Z.; Zhang, Q.; Badell, I.R.; Schwarz, E.M.; O’Keefe, R.J.; Xing, L. TNFα and pathologic bone resorption. Keio J. Med. 2005, 54, 127–131. [Google Scholar] [CrossRef]
- Miyaura, C.; Inada, M.; Matsumoto, C.; Ohshiba, T.; Uozumi, N.; Shimizu, T.; Ito, A. An Essential Role of Cytosolic Phospholipase A2α in Prostaglandin E2–mediated Bone Resorption Associated with Inflammation. J. Exp. Med. 2003, 197, 1303–1310. [Google Scholar] [CrossRef]
- Tanaka, Y.; Nakayamada, S.; Okada, Y. Osteoblasts and Osteoclasts in Bone Remodeling and Inflammation. Curr. Drug Targets-Inflamm. Allergy 2005, 4, 325–328. [Google Scholar] [CrossRef]
- Griffin, X.L.; Costa, M.L.; Parsons, N.; Smith, N. Electromagnetic field stimulation for treating delayed union or non-union of long bone fractures in adults. Cochrane Database Syst. Rev. 2011, 4, 1–24. [Google Scholar] [CrossRef]
- Fredericks, D.C.; Nepola, J.V.; Baker, J.T.; Abbott, J.; Simon, B. Effects of Pulsed Electromagnetic Fields on Bone Healing in a Rabbit Tibial Osteotomy Model. J. Orthop. Trauma 2000, 14, 93–100. [Google Scholar] [CrossRef]
- Bassett, C.A.L.; Pawluk, R.J.; Pilla, A.A. Acceleration of Fracture Repair by Electromagnetic Fields. A Surgically Noninvasive Method. Ann. N. Y. Acad. Sci. 1974, 238, 242–262. [Google Scholar] [CrossRef]
- Tabrah, F.L.; Ross, P.; Hoffmeier, M.; Gilbert, F., Jr. Clinical report on long-term bone density after short-term EMF application. Bioelectromagnetics 1998, 19, 75–78. [Google Scholar] [CrossRef]
- Liu, H.-F.; Yang, L.; He, H.-C.; Zhou, J.; Liu, Y.; Wang, C.-Y.; Wu, Y.-C.; He, C.-Q. Pulsed electromagnetic fields on postmenopausal osteoporosis in southwest China: A randomized, active-controlled clinical trial. Bioelectromagnetics 2013, 34, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Bodamyali, T.; Bhatt, B.; Hughes, F.J.; Winrow, V.R.; Kanczler, J.M.; Simon, B.; Abbott, J.; Blake, D.R.; Stevens, C.R. Pulsed electromagnetic fields simultaneously induce osteogenesis and upregulate transcription of bone morphogenetic proteins 2 and 4 in rat osteoblasts in vitro. Biochem. Biophys. Res. Commun. 1998, 250, 458–461. [Google Scholar] [CrossRef]
- Jing, D.; Li, F.; Jiang, M.; Cai, J.; Wu, Y.; Xie, K.; Wu, X.; Tang, C.; Liu, J.; Guo, W.; et al. Pulsed electromagnetic fields improve bone microstructure and strength in ovariectomized rats through a Wnt/Lrp5/β-catenin signaling-associated mechanism. PLoS ONE 2013, 8, e79377. [Google Scholar] [CrossRef]
- Jing, D.; Cai, J.; Wu, Y.; Shen, G.; Li, F.; Xu, Q.; Xie, K.; Tang, C.; Liu, J.; Guo, W.; et al. Pulsed Electromagnetic Fields Partially Preserve Bone Mass, Microarchitecture, and Strength by Promoting Bone Formation in Hindlimb-Suspended Rats. J. Bone Miner. Res. 2014, 29, 2250–2261. [Google Scholar] [CrossRef]
- Zhai, M.; Jing, D.; Tong, S.; Wu, Y.; Wang, P.; Zeng, Z.; Shen, G.; Wang, X.; Xu, Q.; Luo, E. Pulsed electromagnetic fields promote in vitro osteoblastogenesis through a Wnt/β-catenin signaling-associated mechanism. Bioelectromagnetics 2016, 37, 152–162. [Google Scholar] [CrossRef]
- Huang, X.L.; Huang, L.Y.; Cheng, Y.T.; Li, F.; Zhou, Q.; Wu, C.; Shi, Q.H.; Guan, Z.Z.; Liao, J.; Hong, W. Zoledronic acid inhibits osteoclast differentiation and function through the regulation of NF-κB and JNK signalling pathways. Int. J. Mol. Med. 2019, 44, 582–592. [Google Scholar] [CrossRef]
- D’Amelio, P.; Grimaldi, A.; Cristofaro, M.A.; Ravazzoli, M.; Molinatti, P.A.; Pescarmona, G.P.; Isaia, G.C. Alendronate reduces osteoclast precursors in osteoporosis. Osteoporos. Int. 2010, 21, 1741–1750. [Google Scholar] [CrossRef]
- Kennel, K.A.; Drake, M.T. Adverse effects of bisphosphonates: Implications for osteoporosis management. Mayo Clin. Proc. 2009, 84, 632–637. [Google Scholar] [CrossRef]
- Shen, W.-W.; Zhao, J.-H. Pulsed electromagnetic fields stimulation affects BMD and local factor production of rats with disuse osteoporosis. Bioelectromagnetics 2010, 31, 113–119. [Google Scholar] [CrossRef]
- Tabrah, F.; Hoffmeier, M.; Gilbert, F., Jr.; Batkin, S.; Bassett, C.A.L. Bone density changes in osteoporosis-prone women exposed to pulsed electromagnetic fields (PEMFs). J. Bone Miner. Res. 1990, 5, 437–442. [Google Scholar] [CrossRef]
- Chang, K.; Chang, W.H.-S.; Wu, M.-L.; Shih, C. Effects of different intensities of extremely low frequency pulsed electromagnetic fields on formation of osteoclast-like cells. Bioelectromagnetics 2003, 24, 431–439. [Google Scholar] [CrossRef]
- Rubin, J.; McLeod, K.J.; Titus, L.; Nanes, M.S.; Catherwood, B.D.; Rubin, C.T. Formation of osteoclast-like cells is suppressed by low frequency, low intensity electric fields. J. Orthop. Res. 1996, 14, 7–15. [Google Scholar] [CrossRef]
- Meng, J.; Hong, J.; Zhao, C.; Zhou, C.; Hu, B.; Yang, Y.; Jiang, G.; Li, S.; Shi, Z.; Cai, X.; et al. Low-intensity pulsed ultrasound inhibits RANKL-induced osteoclast formation via modulating ERK-c-Fos-NFATc1 signaling cascades. Am. J. Transl. Res. 2018, 10, 2901–2910. [Google Scholar]
- Kim, Y.; Lim, H.; Lee, E.; Ki, G.; Seo, Y. Synergistic effect of electromagnetic fields and nanomagnetic particles on osteogenesis through calcium channels and p-ERK signaling. J. Orthop. Res. 2021, 39, 1633–1646. [Google Scholar] [CrossRef]
- Jeong, J.-W.; Choi, S.H.; Han, M.H.; Kim, G.-Y.; Park, C.; Hong, S.H.; Lee, B.-J.; Park, E.K.; Kim, S.O.; Leem, S.-H.; et al. Protective Effects of Fermented Oyster Extract against RANKL-Induced Osteoclastogenesis through Scavenging ROS Generation in RAW 264.7 Cells. Int. J. Mol. Sci. 2019, 20, 1439. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, W.; Yu, C.; Zhang, X.; Zhang, H.; Guan, Q.; Zhao, J.; Xu, J. Follicle-Stimulating Hormone Increases the Risk of Postmenopausal Osteoporosis by Stimulating Osteoclast Differentiation. PLoS ONE 2015, 10, e0134986. [Google Scholar] [CrossRef]
- Szymczyk, K.; Freeman, T.; Adams, C.; Srinivas, V.; Steinbeck, M. Active caspase-3 is required for osteoclast differentiation. J. Cell. Physiol. 2006, 209, 836–844. [Google Scholar] [CrossRef]
- Akisaka, T.; Yoshida, H.; Inoue, S.; Shimizu, K. Organization of Cytoskeletal F-Actin, G-Actin, and Gelsolin in the Adhesion Structures in Cultured Osteoclast. J. Bone Miner. Res. 2001, 16, 1248–1255. [Google Scholar] [CrossRef]
- Visagie, A.; Kasonga, A.; Deepak, V.; Moosa, S.; Marais, S.; Kruger, M.C.; Coetzee, M. Commercial Honeybush (Cyclopia spp.) Tea Extract Inhibits Osteoclast Formation and Bone Resorption in RAW264.7 Murine Macrophages-An in vitro Study. Int. J. Environ. Res. Public Health 2015, 12, 13779–13793. [Google Scholar] [CrossRef]
- Collin-Osdoby, P.; Osdoby, P. RANKL-mediated osteoclast formation from murine RAW 264.7 cells. Methods Mol. Biol. 2012, 816, 187–202. [Google Scholar] [CrossRef] [PubMed]
- Grössinger, E.M.; Kang, M.; Bouchareychas, L.; Sarin, R.; Haudenschild, D.R.; Borodinsky, L.N.; Adamopoulos, I.E. Ca2+-Dependent Regulation of NFATc1 via KCa3.1 in Inflammatory Osteoclastogenesis. J. Immunol. 2018, 200, 749–757. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Wang, H.; Zhang, K.; Yang, B.; Xie, X.; Yang, Z.; Kong, L.; Shi, P.; Zhang, Y.; Ho, Y.-P.; et al. Bisphosphonate-based hydrogel mediates biomimetic negative feedback regulation of osteoclastic activity to promote bone regeneration. Bioact. Mater. 2022, 13, 9–22. [Google Scholar] [CrossRef]
- Yoshimura, Y.; Abe, K.; Deyama, Y.; Kikuiri, T.; Hasegawa, T.; Tei, K.; Shinoda, H.; Suzuki, K.; Kitagawa, Y. Effects of bisphosphonates on osteoclastogenesis in RAW264.7 cells. Int. J. Mol. Med. 2012, 29, 1007–1015. [Google Scholar] [CrossRef]
- Dai, S.M.; Matsuno, H.; Nakamura, H.; Nishioka, K.; Yudoh, K. Interleukin-18 enhances monocyte tumor necrosis factor α and interleukin-1β production induced by direct contact with T lymphocytes: Implications in rheumatoid arthritis. Arthritis Rheum. 2004, 50, 432–443. [Google Scholar] [CrossRef]
- Katsimbri, P. The biology of normal bone remodelling. Eur. J. Cancer Care 2017, 26, 1–5. [Google Scholar] [CrossRef]
- Takeda, S.; Karsenty, G. Central control of bone formation. J. Bone Miner. Metab. 2001, 19, 195–198. [Google Scholar] [CrossRef]
- Raisz, L.G.; Seeman, E. Causes of Age-Related Bone Loss and Bone Fragility: An Alternative View. J. Bone Miner. Res. 2001, 16, 1948–1952. [Google Scholar] [CrossRef]
- Rodan, G.A. The development and function of the skeleton and bone metastases. Cancer 2003, 97 (Suppl. 3), 726–732. [Google Scholar] [CrossRef]
- Negishi-Koga, T.; Takayanagi, H. Ca2+-NFATc1 signaling is an essential axis of osteoclast differentiation. Immunol. Rev. 2009, 231, 241–256. [Google Scholar] [CrossRef]
- Sato, K.; Suematsu, A.; Nakashima, T.; Takemoto-Kimura, S.; Aoki, K.; Morishita, Y.; Asahara, H.; Ohya, K.; Yamaguchi, A.; Takai, T.; et al. Regulation of osteoclast differentiation and function by the CaMK-CREB pathway. Nat. Med. 2006, 12, 1410–1416. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.-J.; Kim, Y.-M.; Jang, H.-J.; Seo, Y.-K. Synergistic effect of rice bran extract and extremely low-frequency electromagnetic fields on dermal papilla/melanocytes in melanogenesis. Bioelectromagnetics 2018, 39, 595–603. [Google Scholar] [CrossRef] [PubMed] [Green Version]






| Gene | Forward(5′-3′) | Reverse(5′-3′) |
|---|---|---|
| Mouse β-actin | AGGCCAACCGTGAAAAGATG | TGGCGTGAGGGAGAGCATAG |
| Mouse capase3 | GGCTGAAACCACCAATCGC | CTTAGCGTACCGTTCCAAGC |
| Mouse NFATc1 | GACCGAGAGGCTCCGAAC | AGGGTCGAGGTGACACTAGG |
| Mouse MMP-9 | AGCCGACTTTTGTGGTCTTC | AGGGTTTGCCTTCTCCGTTG |
| Mouse RANK | GAACATTGAGGACAAAGGCCC | CCACACAGGTAGGCAGTGAC |
| Mouse TRAP | GAACATTGAGGACAAAGGCCC | CCACACAGGTAGGCAGTGAC |
| Mouse TNF-α | CACTCACAAACCACCAAGTG | GAGTAGACAAGGTACAACCC |
| Mouse IL-1β | TGCCACCTTTTGACAGTGATG | TGGGTGTGCCGTCTTTCATT |
| Mouse IL-23 | AAT GTG CCC CGT ATC CAG TG | GCT GCC ACT GCT GAC TAG AA |
| Mouse TRPV4 | GTG ATG GTC TTT GCC CTG GT | TGA TGC CCA AGT TCT GGT TCC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nam, M.-H.; Park, H.-J.; Seo, Y.-K. Reduction of Osteoclastic Differentiation of Raw 264.7 Cells by EMF Exposure through TRPV4 and p-CREB Pathway. Int. J. Mol. Sci. 2023, 24, 3058. https://doi.org/10.3390/ijms24043058
Nam M-H, Park H-J, Seo Y-K. Reduction of Osteoclastic Differentiation of Raw 264.7 Cells by EMF Exposure through TRPV4 and p-CREB Pathway. International Journal of Molecular Sciences. 2023; 24(4):3058. https://doi.org/10.3390/ijms24043058
Chicago/Turabian StyleNam, Myeong-Hyun, Hee-Jung Park, and Young-Kwon Seo. 2023. "Reduction of Osteoclastic Differentiation of Raw 264.7 Cells by EMF Exposure through TRPV4 and p-CREB Pathway" International Journal of Molecular Sciences 24, no. 4: 3058. https://doi.org/10.3390/ijms24043058
APA StyleNam, M. -H., Park, H. -J., & Seo, Y. -K. (2023). Reduction of Osteoclastic Differentiation of Raw 264.7 Cells by EMF Exposure through TRPV4 and p-CREB Pathway. International Journal of Molecular Sciences, 24(4), 3058. https://doi.org/10.3390/ijms24043058





