Transcriptomic Changes Following Induced De-Masculinisation of Australian Red Claw Crayfish Cherax quadricarinatus
Abstract
:1. Introduction
1.1. Aquatic Invasions
1.2. Biotechnology Solutions
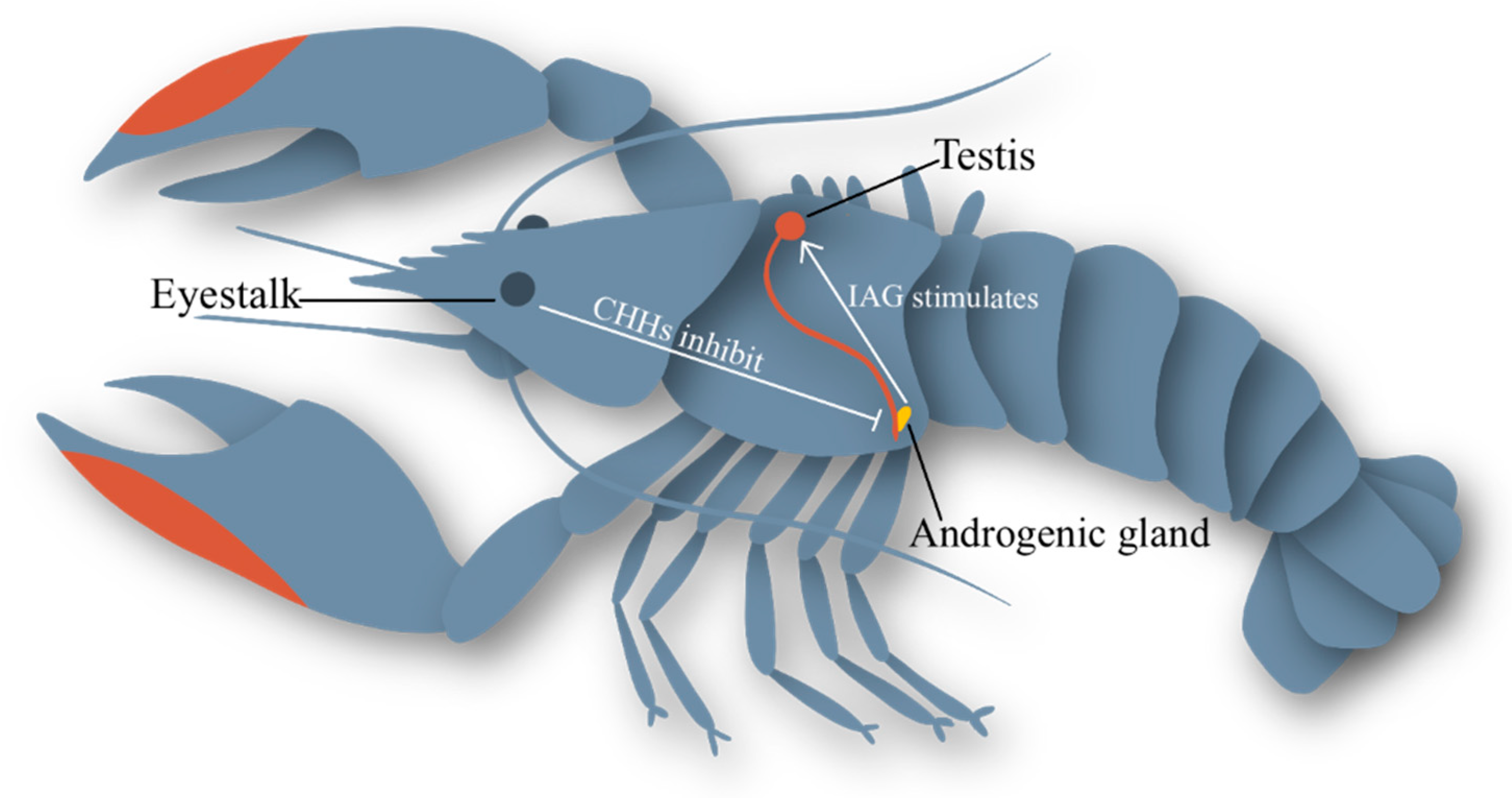
1.3. The Neuroendocrine Network
1.4. Broader Insulin Endocrinology
1.5. Research Aims
2. Results
2.1. Anatomical Measurements and Observations
2.2. RNA-Seq Results
3. Discussion
3.1. Understanding the Axis
3.2. Differential Expression in the AG
3.3. Differential Expression in the Testes
3.3.1. Upregulated Transcripts
3.3.2. Downregulated Transcripts
4. Conclusions
5. Materials and Methods
5.1. Gene Silencing and Dissection
5.2. RNA Extraction and Transcriptome Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ellender, B.R.; Weyl, O.L. A review of current knowledge, risk and ecological impacts associated with non-native freshwater fish introductions in South Africa. Aquat. Invasions 2014, 9, 117–132. [Google Scholar] [CrossRef]
- García-Vásquez, A.; Razo-Mendivil, U.; Rubio-Godoy, M. Triple trouble? Invasive poeciliid fishes carry the introduced tilapia pathogen Gyrodactylus cichlidarum in the Mexican highlands. Vet. Parasitol. 2017, 235, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Davidson, T.M.; Brenneis, V.; de Rivera, C.; Draheim, R.; Gillespi, G. Northern range expansion and coastal occurrences of the New Zealand mud snail Potamopyrgus antipodarum (Gray, 1843) in the northeast Pacific. Aquat. Invasions 2008, 3, 349–353. [Google Scholar] [CrossRef]
- Flegel, T.W. The special danger of viral pathogens in shrimp translocated for aquaculture. Sci. Asia 2006, 32, 215–221. [Google Scholar] [CrossRef]
- Hänfling, B.; Edwards, F.; Gherardi, F. Invasive alien Crustacea: Dispersal, establishment, impact and control. BioControl 2011, 56, 573–595. [Google Scholar] [CrossRef]
- Haubrock, P.J.; Oficialdegui, F.J.; Zeng, Y.; Patoka, J.; Yeo, D.C.J.; Kouba, A. The redclaw crayfish: A prominent aquaculture species with invasive potential in tropical and subtropical biodiversity hotspots. Rev. Aquac. 2021, 13, 1488–1530. [Google Scholar] [CrossRef]
- Horwitz, P. The translocation of freshwater crayfish in Australia: Potential impact, the need for control and global relevance. Biol. Conserv. 1990, 54, 291–305. [Google Scholar] [CrossRef]
- Jones, C.; Geddes, M.C. Australian freshwater crayfish: Exploitation by fishing and aquaculture. Aust. Biol. 1997, 10, 70–75. [Google Scholar]
- Holdich, D.M. A review of astaciculture: Freshwater crayfish farming. Aquat. Living Resour. 1993, 6, 307–317. [Google Scholar] [CrossRef]
- Saoud, I.P.; Ghanawi, J.; Thompson, K.R.; Webster, C.D. A Review of the Culture and Diseases of Redclaw Crayfish Cherax quadricarinatus (Von Martens 1868). J. World Aquac. Soc. 2013, 44, 1–29. [Google Scholar] [CrossRef]
- Wickins, J.F.; Lee, D.O.C. Crustacean Farming: Ranching and Culture; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Coughran, J.; McCormack, R.; Daly, G. Translocation of the Yabby Cherax destructor into eastern drainages of New South Wales, Australia. Aust. Zool. 2009, 35, 100–103. [Google Scholar] [CrossRef]
- Bortolini-Rosales, J.-L.; Alvarez, F.; Rodríguez-Almaraz, G. On the presence of the Australian redclaw crayfish, Cherax quadricarinatus, in Mexico. Biol. Invasions 2007, 9, 615–620. [Google Scholar] [CrossRef]
- Ventura, T. Monosex in Aquaculture. Results Probl. Cell Differ. 2018, 65, 91–101. [Google Scholar] [PubMed]
- Sagi, A.; Ventura, T. Insulin-Like Gene in Prawns and Uses Thereof. U.S. Patent 8440878B2, 14 May 2013. [Google Scholar]
- Ventura, T.; Rosen, O.; Sagi, A. From the discovery of the crustacean androgenic gland to the insulin-like hormone in six decades. Gen. Comp. Endocrinol. 2011, 173, 381–388. [Google Scholar] [CrossRef]
- Thompson, K.R.; Muzinic, L.A.; Yancey, D.H.; Webster, C.D.; Rouse, D.B.; Xiong, Y. Growth, Processing Measurements, Tail Meat Yield, and Tail Meat Proximate Composition of Male and Female Australian Red Claw Crayfish, Cherax quadricarinatus, Stocked into Earthen Ponds. J. Appl. Aquac. 2005, 16, 117–129. [Google Scholar] [CrossRef]
- Rodgers, L.J.; Saoud, P.; Rouse, D. The effects of monosex culture and stocking density on survival, growth and yield of redclaw crayfish (Cherax quadricarinatus) in earthen ponds. Aquaculture 2006, 259, 164–168. [Google Scholar] [CrossRef]
- Curtis, M.C.; Jones, C. Observations on Monosex Culture of Redclaw Crayfish Cherax quadricarinatus von Martens (Decapoda: Parastacidae) in Earthen Ponds. J. World Aquac. Soc. 1995, 26, 154–159. [Google Scholar] [CrossRef]
- Sagi, A.; Manor, R.; Segall, C.; DA Vis, C.; Khalaila, I. On intersexuality in the crayfish Cherax quadricarinatus: An inducible sexual plasticity model. Invertebr. Reprod. Dev. 2002, 41, 27–33. [Google Scholar] [CrossRef]
- Sagi, A.; Khalaila, I.; Barki, A.; Hulata, G.; Karplus, I. Intersex red claw crayfish, Cherax quadricarinatus (von martens): Functional males with pre-vitellogenic ovaries. Biol. Bull. 1996, 190, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Parnes, S.; Khalaila, I.; Hulata, G.; Sagi, A. Sex determination in crayfish: Are intersex Cherax quadricarinatus (Decapoda, Parastacidae) genetically females? Genet. Res. 2003, 82, 107–116. [Google Scholar] [CrossRef]
- Levy, T.; Ventura, T.; De Leo, G.; Grinshpan, N.; Abu Abayed, F.A.; Manor, R.; Savaya, A.; Sklarz, M.Y.; Chalifa-Caspi, V.; Mishmar, D.; et al. Two homogametic genotypes—One crayfish: On the consequences of intersexuality. iScience 2020, 23, 101652. [Google Scholar] [CrossRef] [PubMed]
- Rosen, O.; Weil, S.; Manor, R.; Roth, Z.; Khalaila, I.; Sagi, A. A crayfish insulin-like-binding protein: Another piece in the androgenic gland insulin-like hormone puzzle is revealed. J. Biol. Chem. 2013, 288, 22289–22298. [Google Scholar] [CrossRef]
- Chandler, J.C.; Gandhi, N.; Mancera, R.; Smith, G.; Elizur, A.; Ventura, T. Understanding Insulin Endocrinology in Decapod Crustacea: Molecular Modelling Characterization of an Insulin-Binding Protein and Insulin-Like Peptides in the Eastern Spiny Lobster, Sagmariasus verreauxi. Int. J. Mol. Sci. 2017, 18, 1832. [Google Scholar] [CrossRef]
- Chan, S.J.; Steiner, D.F. Insulin Through the Ages: Phylogeny of a Growth Promoting and Metabolic Regulatory Hormone. Am. Zool. 2000, 40, 213–222. [Google Scholar]
- Levy, T.; Sagi, A. The “IAG-Switch”—A Key Controlling Element in Decapod Crustacean Sex Differentiation. Front. Endocrinol. 2020, 11, 651. [Google Scholar] [CrossRef] [PubMed]
- Khalaila, I.; Manor, R.; Weil, S.; Granot, Y.; Keller, R.; Sagi, A. The eyestalk-androgenic gland-testis endocrine axis in the crayfish Cherax quadricarinatus. Gen. Comp. Endocrinol. 2002, 127, 147–156. [Google Scholar] [CrossRef]
- Cronin, L.E. Anatomy and histology of the male reproductive system of Callinectes sapidus Rathbun. J. Morphol. 1947, 81, 209–239. [Google Scholar] [CrossRef] [PubMed]
- Manor, R.; Weil, S.; Oren, S.; Glazer, L.; Aflalo, E.; Ventura, T.; Chalifa-Caspi, V.; Lapidot, M.; Sagi, A. Insulin and gender: An insulin-like gene expressed exclusively in the androgenic gland of the male crayfish. Gen. Comp. Endocrinol. 2007, 150, 326–336. [Google Scholar] [CrossRef]
- Rosen, O.; Manor, R.; Weil, S.; Gafni, O.; Linial, A.; Aflalo, E.D.; Ventura, T.; Sagi, A. A sexual shift induced by silencing of a single insulin-like gene in crayfish: Ovarian upregulation and testicular degeneration. PLoS ONE 2010, 5, e15281. [Google Scholar] [CrossRef]
- Barki, A.; Karplus, I.; Manor, R.; Sagi, A. Intersexuality and behavior in crayfish: The de-masculinization effects of androgenic gland ablation. Horm. Behav. 2006, 50, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Khalaila, I.; Katz, T.; Abdu, U.; Yehezkel, G.; Sagi, A. Effects of implantation of hypertrophied androgenic glands on sexual characters and physiology of the reproductive system in the female red claw crayfish, Cherax quadricarinatus. Gen. Comp. Endocrinol. 2001, 121, 242–249. [Google Scholar] [CrossRef]
- Barki, A.; Karplus, I.; Khalaila, I.; Manor, R.; Sagi, A. Male-like behavioral patterns and physiological alterations induced by androgenic gland implantation in female crayfish. J. Exp. Biol. 2003, 206, 1791–1797. [Google Scholar] [CrossRef] [Green Version]
- Manor, R.; Aflalo, E.D.; Segall, C.; Weil, S.; Azulay, D.; Ventura, T.; Sagi, A. Androgenic gland implantation promotes growth and inhibits vitellogenesis in Cherax quadricarinatus females held in individual compartments. Invertebr. Reprod. Dev. 2004, 45, 151–159. [Google Scholar] [CrossRef]
- Nguyen, T.V.; Cummins, S.F.; Elizur, A.; Ventura, T. Transcriptomic characterization and curation of candidate neuropeptides regulating reproduction in the eyestalk ganglia of the Australian crayfish, Cherax quadricarinatus. Sci. Rep. 2016, 6, 38658. [Google Scholar] [CrossRef]
- Ventura, T.; Chandler, J.C.; Nguyen, T.V.; Hyde, C.J.; Elizur, A.; FitzGibbon, Q.P.; Smith, G.G. Multi-tissue transcriptome analysis identifies key sexual development-related genes of the ornate spiny lobster (Panulirus ornatus). Genes 2020, 11, 1150. [Google Scholar] [CrossRef] [PubMed]
- Shechter, A.; Aflalo, E.D.; Davis, C.; Sagi, A. Expression of the Reproductive Female-Specific Vitellogenin Gene in Endocrinologically Induced Male and Intersex Cherax quadricarinatus Crayfish. Biol. Reprod. 2005, 73, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Bai, H.; Xiong, Y.; Fu, H.; Jiang, S.; Jiang, F.; Jin, S.; Sun, S.; Qiao, H.; Zhang, W. Molecular characterization of insulin-like androgenic gland hormone-binding protein gene from the oriental river prawn Macrobrachium nipponense and investigation of its transcriptional relationship with the insulin-like androgenic gland hormone gene. Gen. Comp. Endocrinol. 2015, 216, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Sharabi, O.; Manor, R.; Weil, S.; Aflalo, E.; Lezer, Y.; Levy, T.; Aizen, J.; Ventura, T.; Mather, P.B.; Khalaila, I.; et al. Identification and characterization of an insulin-like receptor involved in crustacean reproduction. Endocrinology 2016, 157, 928–941. [Google Scholar] [CrossRef] [PubMed]
- Aizen, J.; Chandler, J.C.; Fitzgibbon, Q.P.; Sagi, A.; Battaglene, S.C.; Elizur, A.; Ventura, T. Production of recombinant insulin-like androgenic gland hormones from three decapod species: In vitro testicular phosphorylation and activation of a newly identified tyrosine kinase receptor from the Eastern spiny lobster, Sagmariasus verreauxi. Gen. Comp. Endocrinol. 2016, 229, 8–18. [Google Scholar] [CrossRef]
- Rosen, O.; Manor, R.; Weil, S.; Aflalo, E.; Bakhrat, A.; Abdu, U.; Sagi, A. An androgenic gland membrane-anchored gene associated with the crustacean insulin-like androgenic gland hormone. J. Exp. Biol. 2013, 216, 2122–2128. [Google Scholar] [CrossRef]
- Sagi, A.; Manor, R.; Ventura, T. Gene silencing in crustaceans: From basic research to biotechnologies. Genes 2013, 4, 620–645. [Google Scholar] [CrossRef]
- Webster, S.; Keller, R.; Dirksen, H. The CHH-superfamily of multifuctional peptide hormones controlling crustacean metabolism, moulting, and reproduction. Gen. Comp. Endocrinol. 2012, 175, 217–233. [Google Scholar] [CrossRef]
- Hyde, C.J.; Fitzgibbon, Q.P.; Elizur, A.; Smith, G.G.; Ventura, T. CrustyBase: An interactive online database for crustacean transcriptomes. BMC Genom. 2020, 21, 637. [Google Scholar] [CrossRef] [PubMed]
- Chong, T.; Stary, J.M.; Wang, Y.; A Newmark, P. Molecular markers to characterize the hermaphroditic reproductive system of the planarian Schmidtea mediterranea. BMC Dev. Biol. 2011, 11, 69. [Google Scholar] [CrossRef] [PubMed]
- Palfree, R.G.E.; Bennett, H.P.J.; Bateman, A. The Evolution of the Secreted Regulatory Protein Progranulin. PLoS ONE 2015, 10, e0133749. [Google Scholar] [CrossRef] [PubMed]
- Aspatwar, A.; Tolvanen, M.E.E.; Ortutay, C.; Parkkila, S. Carbonic anhydrase related proteins: Molecular biology and evolution. Subcell Biochem. 2014, 75, 135–156. [Google Scholar] [PubMed]
- Ali, M.Y.; Pavasovic, A.; Mather, P.B.; Prentis, P.J. Analysis, characterisation and expression of gill-expressed carbonic anhydrase genes in the freshwater crayfish Cherax quadricarinatus. Gene 2015, 564, 176–187. [Google Scholar] [CrossRef]
- Sterky, F.H.; Trotter, J.H.; Lee, S.-J.; Recktenwald, C.V.; Du, X.; Zhou, B.; Zhou, P.; Schwenk, J.; Fakler, B.; Südhof, T.C. Carbonic anhydrase-related protein CA10 is an evolutionarily conserved pan-neurexin ligand. Proc. Natl. Acad. Sci. USA 2017, 114, E1253–E1262. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Gillen, C.M.; Wheatly, M.G. Molecular characterization of the sarcoplasmic calcium-binding protein (SCP) from crayfish Procambarus clarkii. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2006, 144, 478–487. [Google Scholar] [CrossRef]
- Hernández-Aguirre, L.E.; Fuentes-Sidas, Y.I.; Rivera-Rangel, L.R.; Gutiérrez-Méndez, N.; Yepiz-Plascencia, G.; Chávez-Flores, D.; de la Serna, F.J.Z.-D.; Peralta-Pérez, M.D.R.; García-Triana, A. cDNA Characterization and Expression of Selenium-Dependent CqGPx3 Isoforms in the Crayfish Cherax quadricarinatus under High Temperature and Hypoxia. Genes 2022, 13, 179. [Google Scholar] [CrossRef]
- Wang, X.; Han, Y.; Chen, F.; Wang, M.; Xiao, Y.; Wang, H.; Xu, L.; Liu, W. Glutathione Peroxidase 1 Protects Against Peroxynitrite-Induced Spiral Ganglion Neuron Damage Through Attenuating NF-κB Pathway Activation. Front. Cell. Neurosci. 2022, 16, 841731. [Google Scholar] [CrossRef] [PubMed]
- Hlaing, S.M.M.; Lou, J.; Cheng, J.; Xun, X.; Li, M.; Lu, W.; Hu, X.; Bao, Z. Tissue-Biased and Species-Specific Regulation of Glutathione Peroxidase (GPx) Genes in Scallops Exposed to Toxic Dinoflagellates. Toxins 2020, 13, 21. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.S.; Kim, M.; Youn, B.-S.; Lee, N.S.; Park, J.W.; Lee, I.K.; Lee, Y.S.; Kim, J.B.; Cho, Y.M.; Lee, H.K.; et al. Glutathione peroxidase 3 mediates the antioxidant effect of peroxisome proliferator-activated receptor gamma in human skeletal muscle cells. Mol. Cell. Biol. 2009, 29, 20–30. [Google Scholar] [CrossRef]
- Venugopal, P.; Veyssiere, H.; Couderc, J.-L.; Richard, G.; Vachias, C.; Mirouse, V. Multiple functions of the scaffold protein Discs large 5 in the control of growth, cell polarity and cell adhesion in Drosophila melanogaster. BMC Dev. Biol. 2020, 20, 10. [Google Scholar] [CrossRef] [PubMed]
- Rashid, A.; Deyholos, M. Phylogenetic relationship and in silico expression profile of PELPK1 of Arabidopsis thaliana (L.) Heynh. Intern. J. Biosci. 2015, 6, 93–99. [Google Scholar]
- Niksirat, H.; James, P.; Andersson, L.; Kouba, A.; Kozák, P. Label-free protein quantification in freshly ejaculated versus post-mating spermatophores of the noble crayfish Astacus astacus. J. Proteom. 2015, 123, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, F.; Lyon, R.C.; Chen, J. Functions of myosin light chain-2 (MYL2) in cardiac muscle and disease. Gene 2015, 569, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Rafiq, A.; Aashaq, S.; Jan, I.; Beigh, M.A. SIX1 transcription factor: A review of cellular functions and regulatory dynamics. Int. J. Biol. Macromol. 2021, 193, 1151–1164. [Google Scholar] [CrossRef]
- Ruf, R.G.; Xu, P.-X.; Silvius, D.; Otto, E.A.; Beekmann, F.; Muerb, U.T.; Kumar, S.; Neuhaus, T.J.; Kemper, M.J.; Raymond, R.M.; et al. SIX1 mutations cause branchio-oto-renal syndrome by disruption of EYA1-SIX1-DNA complexes. Proc. Natl. Acad. Sci. USA 2004, 101, 8090–8095. [Google Scholar] [CrossRef]
- Kumar, J.P. The sine oculis homeobox (SIX) family of transcription factors as regulators of development and disease. Cell. Mol. Life Sci. 2009, 66, 565–583. [Google Scholar] [CrossRef]
- Pangalos, M.N.; Neefs, J.-M.; Somers, M.; Verhasselt, P.; Bekkers, M.; van der Helm, L.; Fraiponts, E.; Ashton, D.; Gordon, R.D. Isolation and expression of novel human glutamate carboxypeptidases with N-acetylated alpha-linked acidic dipeptidase and dipeptidyl peptidase IV activity. J. Biol. Chem. 1999, 274, 8470–8483. [Google Scholar] [CrossRef]
- Umemiya, T.; Takasu, E.; Takeichi, M.; Aigaki, T.; Nose, A. Forked end: A novel transmembrane protein involved in neuromuscular specificity in drosophila identified by gain-of-function screening. J. Neurobiol. 2002, 51, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Edwards, A.S.; Scott, J.D. A-kinase anchoring proteins: Protein kinase A and beyond. Curr. Opin. Cell Biol. 2000, 12, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.V.; Tan, A.S.; Hilton, C.J. Cyclic adenosine monophosphate promotes cell survival and retards apoptosis in a factor-dependent bone marrow-derived cell line. Exp. Hematol. 1993, 21, 269–276. [Google Scholar] [PubMed]
- Laity, J.H.; Lee, B.M.; Wright, P.E. Zinc finger proteins: New insights into structural and functional diversity. Curr. Opin. Struct. Biol. 2001, 11, 39–46. [Google Scholar] [CrossRef]
- Cassandri, M.; Smirnov, A.; Novelli, F.; Pitolli, C.; Agostini, M.; Malewicz, M.; Melino, G.; Raschellà, G. Zinc-finger proteins in health and disease. Cell Death Discov. 2017, 3, 170–171. [Google Scholar] [CrossRef] [PubMed]
- Seelig, H.P.; Schranz, P.; Schröter, H.; Wiemann, C.; Renz, M. Macrogolgin—A new 376 kD Golgi complex outer membrane protein as target of antibodies in patients with rheumatic diseases and HIV infections. J. Autoimmun. 1994, 7, 67–91. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.K.L.; Fritzler, M.J. Golgins: Coiled-coil-rich proteins associated with the Golgi complex. Electron. J. Biotechnol. 1998, 1, 45–54. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing. 2022. Available online: https://www.r-project.org/ (accessed on 7 September 2022).
- Bell, T.A.; Lightner, D.V. A Handbook of Normal Penaeid Shrimp Histology; World Aquaculture Society: Baton Rouge, LA, USA, 1988. [Google Scholar]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, G.; Glendinning, S.; Ventura, T. Transcriptomic Changes Following Induced De-Masculinisation of Australian Red Claw Crayfish Cherax quadricarinatus. Int. J. Mol. Sci. 2023, 24, 3292. https://doi.org/10.3390/ijms24043292
Smith G, Glendinning S, Ventura T. Transcriptomic Changes Following Induced De-Masculinisation of Australian Red Claw Crayfish Cherax quadricarinatus. International Journal of Molecular Sciences. 2023; 24(4):3292. https://doi.org/10.3390/ijms24043292
Chicago/Turabian StyleSmith, Grace, Susan Glendinning, and Tomer Ventura. 2023. "Transcriptomic Changes Following Induced De-Masculinisation of Australian Red Claw Crayfish Cherax quadricarinatus" International Journal of Molecular Sciences 24, no. 4: 3292. https://doi.org/10.3390/ijms24043292






