Nanomicellar Formulations Loaded with Histamine and Paclitaxel as a New Strategy to Improve Chemotherapy for Breast Cancer
Abstract
:1. Introduction
2. Results
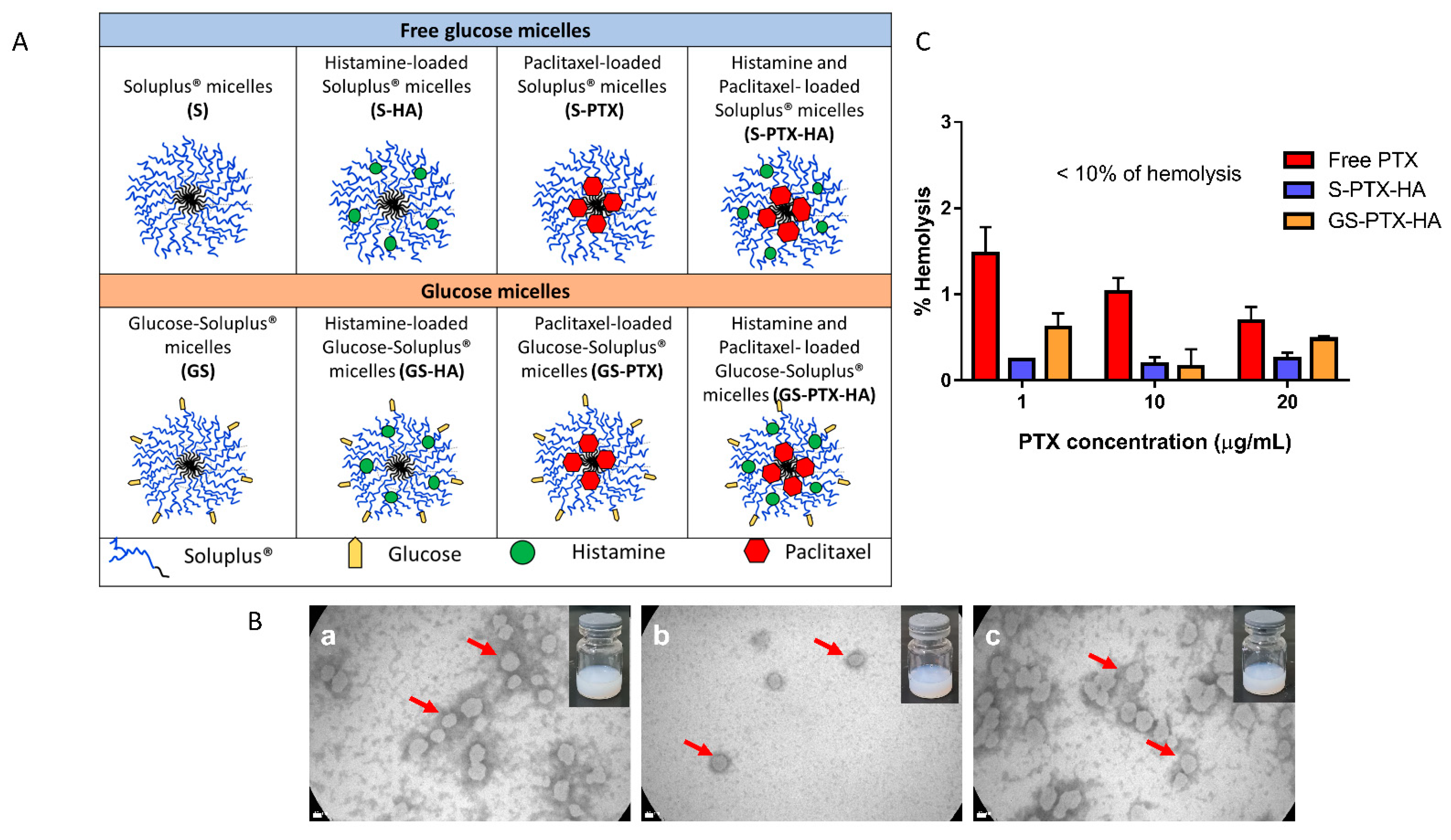
2.1. Characterization of the Nanomicellar Formulations
2.2. Stability of the Nanoformulations in Simulated Biological Fluid
2.3. In Vitro Hemolysis Assay
2.4. In Vitro Antitumor Activity of Nanomicellar Formulations in TNBC Cells
2.4.1. Cytotoxicity Assays
2.4.2. Apoptosis Assays
2.4.3. Cell Migration Assays
2.5. Cytotoxic Effect of Micellar Formulations in Murine Mammary Epithelial Cells
2.6. In Vivo Antitumor Effect of Glycosylated Nanomicellar Formulations
3. Discussion
4. Materials and Methods
4.1. Chemicals
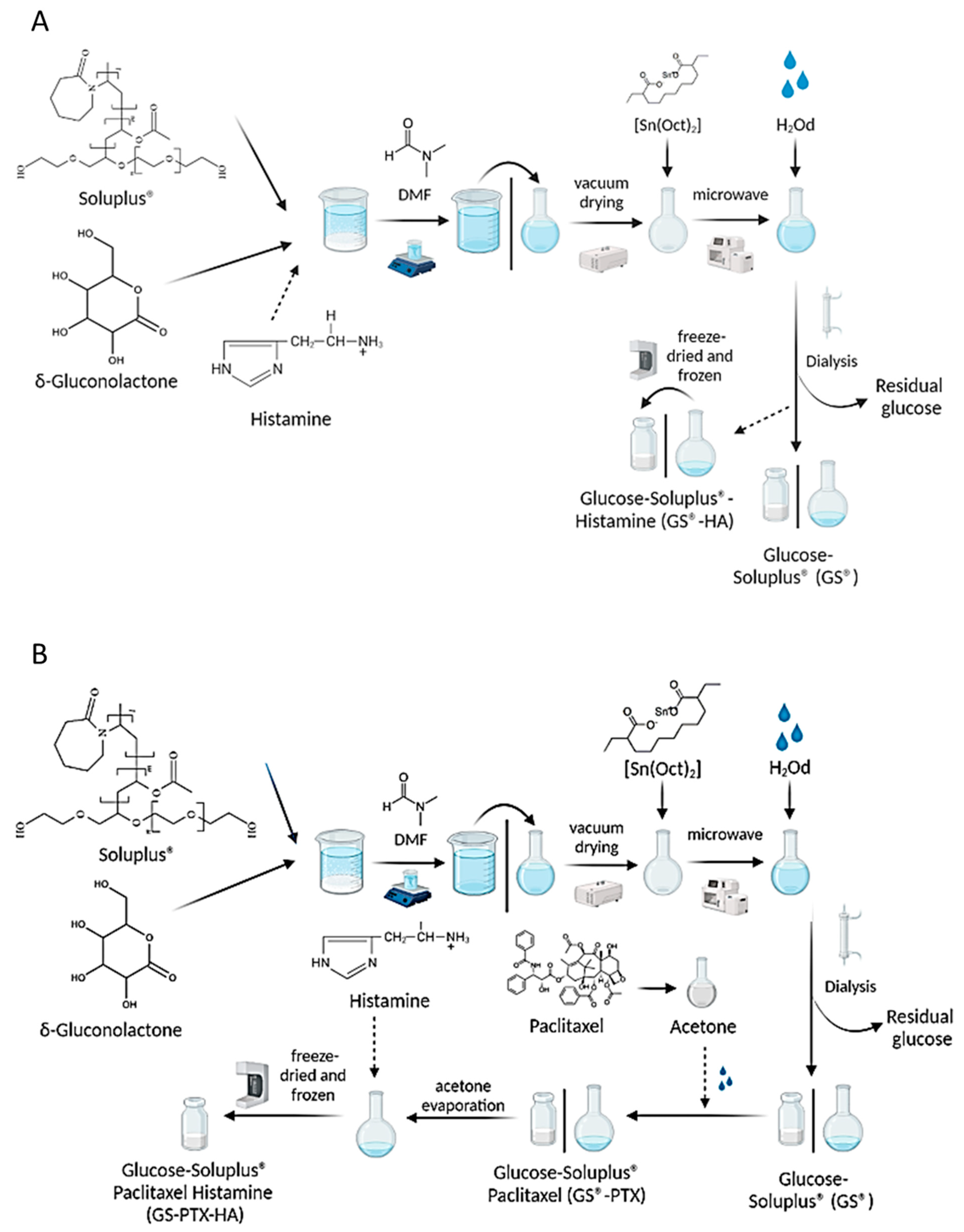
4.2. Glycosylation of Soluplus® Copolymer
4.3. Preparation of Soluplus® Micelles and Drug Encapsulation
4.4. Characterization of Soluplus® Micelles
4.5. Physicochemical Stability of the Nanoformulations in Simulated Biological Fluid
4.6. Cell Culture
4.7. Cell Proliferation Assays
4.8. Apoptosis Determinations
4.8.1. Annexin V Assay
4.8.2. TUNEL Assay
4.9. Wound Assay
4.10. Breast Cancer Model
4.11. In Vitro Hemolytic Assay
4.12. Histopathological Analysis
4.13. Flow Cytometry for Immunophenotyping
4.14. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Breast Cancer. 26 March 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/breast-cancer (accessed on 17 October 2022).
- Łukasiewicz, S.; Czeczelewski, M.; Forma, A.; Baj, J.; Sitarz, R.; Stanisławek, A. Breast Cancer-Epidemiology, Risk Factors, Classification, Prognostic Markers, and Current Treatment Strategies-An Updated Review. Cancers 2021, 13, 4287. [Google Scholar] [CrossRef] [PubMed]
- Hong, R.; Xu, B. Breast cancer: An up-to-date review and future perspectives. Cancer Commun. 2022, 42, 913–936. [Google Scholar] [CrossRef] [PubMed]
- Gierach, G.L.; Burke, A.; Anderson, W. Epidemiology of triple negative breast cancers. Breast Dis. 2011, 32, 5–24. [Google Scholar] [CrossRef]
- Almansour, N.M. Triple-Negative Breast Cancer: A Brief Review About Epidemiology, Risk Factors, Signaling Pathways, Treatment and Role of Artificial Intelligence. Front. Mol. Biosci. 2022, 9, 836417. [Google Scholar] [CrossRef]
- Ma, P.; Mumper, R. Paclitaxel Nano-Delivery Systems: A Comprehensive Review. J. Nanomed. Nanotechnol. 2013, 4, 1000164. [Google Scholar] [CrossRef] [PubMed]
- Bernabeu, E.; Gonzalez, L.; Cagel, M.; Gergic, E.P.; Moretton, M.A.; Chiappetta, D.A. Novel Soluplus(®)-TPGS mixed micelles for encapsulation of paclitaxel with enhanced in vitro cytotoxicity on breast and ovarian cancer cell lines. Colloids Surf. B Biointerfaces 2016, 140, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.L.; Cheng, M.; Lin, C. From Nanoparticles to Cancer Nanomedicine: Old Problems with New Solutions. Nanomaterials 2021, 11, 1727. [Google Scholar] [CrossRef]
- Alavi, M.; Nokhodchi, A. Micro- and nanoformulations of paclitaxel based on micelles, liposomes, cubosomes, and lipid nanoparticles: Recent advances and challenges. Drug Discov. Today 2022, 27, 576–584. [Google Scholar] [CrossRef]
- Chiappetta, D.A.; Sosnik, A. Poly(ethylene oxide)-poly(propylene oxide) block copolymer micelles as drug delivery agents: Improved hydrosolubility, stability and bioavailability of drugs. Eur. J. Pharm. Biopharm. 2007, 66, 303–317. [Google Scholar] [CrossRef]
- Trivedi, R.; Kompella, U. Nanomicellar formulations for sustained drug delivery: Strategies and underlying principles. Nanomedicine 2010, 5, 485–505. [Google Scholar] [CrossRef] [Green Version]
- Moretton, M.A.; Bernabeu, E.; Grotz, E.; Gonzalez, L.; Zubillaga, M.; Chiappetta, D.A. A glucose-targeted mixed micellar formulation outperforms Genexol in breast cancer cells. Eur. J. Pharm. Biopharm. 2017, 114, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Lei, C.; Yang, Y.; Bu, X.; Ma, H.; Gong, H.; Liu, J.; Fang, X.; Hu, Z.; Fang, Q. Abraxane, the Nanoparticle Formulation of Paclitaxel Can Induce Drug Resistance by Up-Regulation of P-gp. PLoS ONE 2015, 10, e0131429. [Google Scholar] [CrossRef] [PubMed]
- Desai, N. Nanoparticle Albumin-Bound Paclitaxel (Abraxane®). In Albumin in Medicine: Pathological and Clinical Applications; Otagiri, M., Chuang, V.T.G., Eds.; Springer: Singapore, 2016; pp. 101–119. [Google Scholar]
- Martinel Lamas, D.J.; Croci, M.; Carabajal, E.; Crescenti, E.J.; Sambuco, L.; Massari, N.A.; Bergoc, R.M.; Rivera, E.S.; Medina, V.A. Therapeutic potential of histamine H4 receptor agonists in triple-negative human breast cancer experimental model. Br. J. Pharmacol. 2013, 170, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Nicoud, M.B.; Sterle, H.A.; Massari, N.A.; Táquez Delgado, M.A.; Formoso, K.; Herrero Ducloux, M.V.; Martinel Lamas, D.; Cremaschi, G.A.; Medina, V.A. Study of the antitumour effects and the modulation of immune response by histamine in breast cancer. Br. J. Cancer 2020, 122, 348–360. [Google Scholar] [CrossRef]
- Medina, V.A.; Brenzoni, P.G.; Lamas, D.J.; Massari, N.; Mondillo, C.; Nunez, M.A.; Pignataro, O.; Rivera, E.S. Role of histamine H4 receptor in breast cancer cell proliferation. Front. Biosci. 2011, 3, 1042–1060. [Google Scholar] [CrossRef] [PubMed]
- Medina, V.; Cricco, G.; Nuñez, M.; Martín, G.; Mohamad, N.; Correa-Fiz, F.; Sanchez-Jimenez, F.; Bergoc, R.; Rivera, E.S. Histamine-mediated signaling processes in human malignant mammary cells. Cancer Biol. Ther. 2006, 5, 1462–1471. [Google Scholar] [CrossRef]
- Medina, V.; Croci, M.; Crescenti, E.; Mohamad, N.; Sanchez-Jiménez, F.; Massari, N.; Nuñez, M.; Cricco, G.; Martin, G.; Bergoc, R.; et al. The role of histamine in human mammary carcinogenesis: H3 and H4 receptors as potential therapeutic targets for breast cancer treatment. Cancer Biol. Ther. 2008, 7, 28–35. [Google Scholar] [CrossRef]
- Speisky, D.; Táquez Delgado, M.A.; Iotti, A.; Nicoud, M.B.; Ospital, I.A.; Vigovich, F.; Dezanzo, P.; Ernst, G.; Uriburu, J.L.; Medina, V.A. Histamine H4 Receptor Expression in Triple-negative Breast Cancer: An Exploratory Study. J. Histochem. Cytochem. 2022, 70, 311–322. [Google Scholar] [CrossRef]
- Massari, N.A.; Nicoud, M.; Medina, V. Histamine receptors and cancer pharmacology: An update. Br. J. Pharmacol. 2020, 177, 516–538. [Google Scholar] [CrossRef]
- Porretti, J.C.; Mohamad, N.A.; Martín, G.A.; Cricco, G.P. Fibroblasts induce epithelial to mesenchymal transition in breast tumor cells which is prevented by fibroblasts treatment with histamine in high concentration. Int. J. Biochem. Cell Biol. 2014, 51, 29–38. [Google Scholar] [CrossRef]
- Carabajal, E.; Massari, N.; Croci, M.; Martinel Lamas, D.J.; Prestifilippo, J.P.; Bergoc, R.M.; Rivera, E.S.; Medina, V.A. Radioprotective potential of histamine on rat small intestine and uterus. Eur. J. Histochem. 2012, 56, e48. [Google Scholar] [CrossRef]
- Martinel Lamas, D.J.; Nicoud, M.B.; Sterle, H.A.; Carabajal, E.; Tesan, F.; Perazzo, J.C.; Cremaschi, G.A.; Rivera, E.S.; Medina, V.A. Selective cytoprotective effect of histamine on doxorubicin-induced hepatic and cardiac toxicity in animal models. Cell Death Discov. 2015, 1, 15059. [Google Scholar] [CrossRef]
- Martinel Lamas, D.J.; Nicoud, M.B.; Sterle, H.A.; Cremaschi, G.A.; Medina, V.A. Histamine: A potential cytoprotective agent to improve cancer therapy? Cell Death Dis. 2015, 6, e2029. [Google Scholar] [CrossRef] [PubMed]
- Medina, V.A.; Croci, M.; Carabajal, E.; Bergoc, R.M.; Rivera, E.S. Histamine protects bone marrow against cellular damage induced by ionising radiation. Int. J. Radiat. Biol. 2010, 86, 283–290. [Google Scholar] [CrossRef]
- Medina, V.A.; Croci, M.; Mohamad, N.A.; Massari, N.; Garbarino, G.; Cricco, G.P.; Núñez, M.A.; Martín, G.A.; Crescenti, E.J.; Bergoc, R.M.; et al. Mechanisms underlying the radioprotective effect of histamine on small intestine. Int. J. Radiat. Biol. 2007, 83, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Medina, V.A.; Prestifilippo, J.P.; Croci, M.; Carabajal, E.; Bergoc, R.M.; Elverdin, J.C.; Rivera, E.S. Histamine prevents functional and morphological alterations of submandibular glands induced by ionising radiation. Int. J. Radiat. Biol. 2011, 87, 284–292. [Google Scholar] [CrossRef]
- Riedel, J.; Pibuel, M.; Bernabeu, E.; Poodts, D.; Díaz, M.; Allo, M.; Parola, L.; Hajos, S.; Lázaro-Martínez, J.M.; Salgueiro, M.J.; et al. Glycosylated paclitaxel mixed nanomicelles: Increasing drug brain accumulation and enhancing its in vitro antitumoral activity in glioblastoma cell lines. J. Drug Deliv. Sci. Technol. 2022, 68, 103046. [Google Scholar] [CrossRef]
- Owen, S.C.; Chan, D.; Shoichet, M. Polymeric micelle stability. Nano Today 2012, 7, 53–65. [Google Scholar] [CrossRef]
- Riches, A.C.; Sharp, J.G.; Thomas, D.B.; Smith, S.V. Blood volume determination in the mouse. J. Physiol. 1973, 228, 279–284. [Google Scholar] [CrossRef]
- Huizing, M.T.; Vermorken, J.B.; Rosing, H.; ten Bokkel Huinink, W.W.; Mandjes, I.; Pinedo, H.M.; Beijnen, J.H. Pharmacokinetics of paclitaxel and three major metabolites in patients with advanced breast carcinoma refractory to anthracycline therapy treated with a 3-hour paclitaxel infusion: A European Cancer Centre (ECC) trial. Ann. Oncol. 1995, 6, 699–704. [Google Scholar] [CrossRef]
- Amin, K.; Dannenfelser, R. In vitro hemolysis: Guidance for the pharmaceutical scientist. J. Pharm. Sci. 2006, 95, 1173–1176. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Cho, H.; Kim, D. Poly((D,L)lactic-glycolic)acid-star glucose nanoparticles for glucose transporter and hypoglycemia-mediated tumor targeting. Int. J. Nanomed. 2017, 12, 7453–7467. [Google Scholar] [CrossRef] [PubMed]
- Theerasilp, M.; Chalermpanapun, P.; Sunintaboon, P.; Sungkarat, W.; Nasongkla, N. Glucose-installed biodegradable polymeric micelles for cancer-targeted drug delivery system: Synthesis, characterization and in vitro evaluation. J. Mater. Sci. Mater. Med. 2018, 29, 177. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Pu, Y.; Peng, Y.; Fu, Q.; Guo, L.; Wu, Y.; Zheng, Y. Biotin and glucose dual-targeting, ligand-modified liposomes promote breast tumor-specific drug delivery. Bioorg. Med. Chem. Lett. 2020, 30, 127151. [Google Scholar] [CrossRef] [PubMed]
- Saha, T.; Solomon, J.; Samson, A.O.; Gil-Henn, H. Invasion and Metastasis as a Central Hallmark of Breast Cancer. J. Clin. Med. 2021, 10, 3498. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.C.; Park, A.; Guan, J. In vitro scratch assay: A convenient and inexpensive method for analysis of cell migration in vitro. Nat. Protoc. 2007, 2, 329–333. [Google Scholar] [CrossRef]
- Martinel Lamas, D.J.; Cortina, J.E.; Ventura, C.; Sterle, H.A.; Valli, E.; Balestrasse, K.B.; Blanco, H.; Cremaschi, G.A.; Rivera, E.S.; Medina, V.A. Enhancement of ionizing radiation response by histamine in vitro and in vivo in human breast cancer. Cancer Biol. Ther. 2015, 16, 137–148. [Google Scholar] [CrossRef]
- Bernabeu, E.; Cagel, M.; Lagomarsino, E.; Moretton, M.; Chiappetta, D.A. Paclitaxel: What has been done and the challenges remain ahead. Int. J. Pharm. 2017, 526, 474–495. [Google Scholar] [CrossRef]
- Shrestha, R.; Elsabahy, M.; Florez-Malaver, S.; Samarajeewa, S.; Wooley, K.L. Endosomal escape and siRNA delivery with cationic shell crosslinked knedel-like nanoparticles with tunable buffering capacities. Biomaterials 2012, 33, 8557–8568. [Google Scholar] [CrossRef]
- Yu, C.; Gao, C.; Lü, S.; Chen, C.; Yang, J.; Di, X.; Liu, M. Facile preparation of pH-sensitive micelles self-assembled from amphiphilic chondroitin sulfate-histamine conjugate for triggered intracellular drug release. Colloids Surf. B Biointerfaces 2014, 115, 331–339. [Google Scholar] [CrossRef]
- Zhang, Y.; Lundberg, P.; Diether, M.; Porsch, C.; Janson, C.; Lynd, N.A.; Ducani, C.; Malkoch, M.; Malmström, E.; Hawker, C.J.; et al. Histamine-functionalized copolymer micelles as a drug delivery system in 2D and 3D models of breast cancer. J. Mater. Chem. B 2015, 3, 2472–2486. [Google Scholar] [CrossRef]
- Yin, Q.; Shen, J.; Yu, H.; Huang, Y.; Zhang, Z.; Li, Y. Bioreducible Micelles with Endosomal Buffering and Multidrug Resistance-Reversing Function Enhance Anti-Tumor Efficacy of Doxorubicin. J. Biomed. Nanotechnol. 2015, 11, 1764–1775. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Cheng, R.; Meng, F.; Park, T.G.; Zhong, Z. Water soluble poly(histamine acrylamide) with superior buffer capacity mediates efficient and nontoxic in vitro gene transfection. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 3366–3373. [Google Scholar] [CrossRef]
- Jhaveri, A.M.; Torchilin, V. Multifunctional polymeric micelles for delivery of drugs and siRNA. Front. Pharmacol. 2014, 5, 77. [Google Scholar] [CrossRef] [PubMed]
- Bergin, A.R.T.; Loi, S. Triple-negative breast cancer: Recent treatment advances. F1000Research 2019, 8, 1342. [Google Scholar] [CrossRef]
- Kang, H.; Rho, S.; Stiles, W.R.; Hu, S.; Baek, Y.; Hwang, D.W.; Kashiwagi, S.; Kim, M.S.; Choi, H.S. Size-Dependent EPR Effect of Polymeric Nanoparticles on Tumor Targeting. Adv. Healthc. Mater. 2020, 9, e1901223. [Google Scholar] [CrossRef] [PubMed]
- Massari, N.A.; Nicoud, M.B.; Sambuco, L.; Cricco, G.P.; Martinel Lamas, D.J.; Herrero Ducloux, M.V.; Blanco, H.; Rivera, E.S.; Medina, V.A. Histamine therapeutic efficacy in metastatic melanoma: Role of histamine H4 receptor agonists and opportunity for combination with radiation. Oncotarget 2017, 8, 26471–26491. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, M.S.; Hallner, A.; Brune, M.; Nilsson, S.; Thorén, F.B.; Martner, A.; Hellstrand, K. Immunotherapy with HDC/IL-2 may be clinically efficacious in acute myeloid leukemia of normal karyotype. Hum. Vaccin. Immunother. 2020, 16, 109–111. [Google Scholar] [CrossRef] [PubMed]
- Sarasola, M.P.; Táquez Delgado, M.A.; Nicoud, M.B.; Medina, V.A. Histamine in cancer immunology and immunotherapy. Current status and new perspectives. Pharmacol. Res. Perspect. 2021, 9, e00778. [Google Scholar] [CrossRef]
- Emens, L.A.; Molinero, L.; Loi, S.; Rugo, H.S.; Schneeweiss, A.; Diéras, V.; Iwata, H.; Barrios, C.H.; Nechaeva, M.; Nguyen-Duc, A.; et al. Atezolizumab and nab-Paclitaxel in Advanced Triple-Negative Breast Cancer: Biomarker Evaluation of the IMpassion130 Study. J. Natl. Cancer Inst. 2021, 113, 1005–1016. [Google Scholar] [CrossRef]
- Bernabeu, E.; Flor, S.; Hocht, C.; Taira, C.; Chiappetta, D.; Tripodi, V.; Lucangioli, S. Development and Validation of a Highly Sensitive HPLC Method for Determination of Paclitaxel in Pharmaceutical Dosage forms and Biological Samples. Curr. Pharm. Anal. 2014, 10, 185–192. [Google Scholar] [CrossRef]
- de Figueiredo, T.C.; de Assis, D.C.; Menezes, L.D.; da Silva, G.R.; Lanza, I.P.; Heneine, L.G.; Cançado Sde, V. HPLC-UV method validation for the identification and quantification of bioactive amines in commercial eggs. Talanta 2015, 142, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Caddeo, C.; Pucci, L.; Gabriele, M.; Carbone, C.; Fernàndez-Busquets, X.; Valenti, D.; Pons, R.; Vassallo, A.; Fadda, A.M.; Manconi, M. Stability, biocompatibility and antioxidant activity of PEG-modified liposomes containing resveratrol. Int. J. Pharm. 2018, 538, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Yang, K.; Zhang, B. Pitting corrosion resistance of La added 316L stainless steel in simulated body fluids. Mater. Lett. 2007, 61, 1154–1157. [Google Scholar] [CrossRef]
- Cree, I.A. Cancer Cell Culture. In Methods and Protocols, 2nd ed.; Springer Nature: Cham, Switzerland, 2011; Volume 31, pp. 3639–3640. [Google Scholar]
- Ye, J.; Li, R.; Yang, Y.; Dong, W.; Wang, Y.; Wang, H.; Sun, T.; Li, L.; Shen, Q.; Qin, C.; et al. Comparative colloidal stability, antitumor efficacy, and immunosuppressive effect of commercial paclitaxel nanoformulations. J. Nanobiotechnology 2021, 19, 199. [Google Scholar] [CrossRef] [PubMed]
- Franco, M.S.; Roque, M.C.; de Barros, A.L.B.; de Oliveira Silva, J.; Cassali, G.D.; Oliveira, M.C. Investigation of the antitumor activity and toxicity of long-circulating and fusogenic liposomes co-encapsulating paclitaxel and doxorubicin in a murine breast cancer animal model. Biomed. Pharmacother. 2019, 109, 1728–1739. [Google Scholar] [CrossRef]
- Lamas, D.J.M.; Nicoud, M.B.; Sterle, H.; Cricco, G.P.; Martin, G.A.; Cremaschi, G.A.; Schwelberger, H.G.; Rivera, E.S.; Medina, V.A. Methodological approaches to investigate the effects of histamine receptor targeting compounds in preclinical models of breast cancer. In Histamine Receptors as Drug Targets; Springer: Berlin/Heidelberg, Germany, 2017; pp. 353–387. [Google Scholar]
- Sterle, H.A.; Nicoud, M.B.; Massari, N.A.; Táquez Delgado, M.A.; Herrero Ducloux, M.V.; Cremaschi, G.A.; Medina, V.A. Immunomodulatory role of histamine H4 receptor in breast cancer. Br. J. Cancer 2019, 120, 128–138. [Google Scholar] [CrossRef]
- He, Z.; Wan, X.; Schulz, A.; Bludau, H.; Dobrovolskaia, M.A.; Stern, S.T.; Montgomery, S.A.; Yuan, H.; Li, Z.; Alakhova, D.; et al. A high capacity polymeric micelle of paclitaxel: Implication of high dose drug therapy to safety and in vivo anti-cancer activity. Biomaterials 2016, 101, 296–309. [Google Scholar] [CrossRef]
- Song, Y.; Bugada, L.; Li, R.; Hu, H.; Zhang, L.; Li, C.; Yuan, H.; Rajanayake, K.K.; Truchan, N.A.; Wen, F.; et al. Albumin nanoparticle containing a PI3Kγ inhibitor and paclitaxel in combination with α-PD1 induces tumor remission of breast cancer in mice. Sci. Transl. Med. 2022, 14, eabl3649. [Google Scholar] [CrossRef]
- Qiu, L.; Qiao, M.; Chen, Q.; Tian, C.; Long, M.; Wang, M.; Li, Z.; Hu, W.; Li, G.; Cheng, L.; et al. Enhanced effect of pH-sensitive mixed copolymer micelles for overcoming multidrug resistance of doxorubicin. Biomaterials 2014, 35, 9877–9887. [Google Scholar] [CrossRef]







| Samples | Histamine (5 mg/mL) | Paclitaxel (4 mg/mL) | Size 1 | |
|---|---|---|---|---|
| Z-Ave (nm) (±S.D.) | PDI (±S.D.) | |||
| Soluplus® micelles | - | - | 75.5 (0.2) | 0.09 (0.01) |
| √ | - | 77.9 (0.5) | 0.09 (0.01) | |
| - | √ | 76.2 (0.9) | 0.09 (0.01) | |
| √ | √ | 78.3 (1.1) | 0.10 (0.01) | |
| Glucose-Soluplus® micelles | - | - | 75.7 (0.5) | 0.11 (0.01) |
| √ | - | 83.8 (0.6) | 0.11 (0.01) | |
| - | √ | 79.9 (1.7) | 0.19 (0.01) | |
| √ | √ | 87.2 (4.1) | 0.12 (0.01) | |
| Samples | Histamine (5 mg/mL) | Paclitaxel (4 mg/mL) | Size 1 | |
|---|---|---|---|---|
| Z-Ave (nm) (±S.D.) | PDI (±S.D.) | |||
| Soluplus® Micelles (5% w/v) | √ | - | 180.8 (2.7) | 0.06 (0.01) |
| - | √ | 138.8 (8.6) | 0.13 (0.01) | |
| √ | √ | 197.6 (4.3) | 0.13 (0.01) | |
| Glucose-Soluplus® micelles (5% w/v) | √ | - | 191.6 (1.6) | 0.19 (0.02) |
| - | √ | 155.1 (6.3) | 0.23 (0.01) | |
| √ | √ | 321.3 (3.8) | 0.35 (0.04) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicoud, M.B.; Ospital, I.A.; Táquez Delgado, M.A.; Riedel, J.; Fuentes, P.; Bernabeu, E.; Rubinstein, M.R.; Lauretta, P.; Martínez Vivot, R.; Aguilar, M.d.l.Á.; et al. Nanomicellar Formulations Loaded with Histamine and Paclitaxel as a New Strategy to Improve Chemotherapy for Breast Cancer. Int. J. Mol. Sci. 2023, 24, 3546. https://doi.org/10.3390/ijms24043546
Nicoud MB, Ospital IA, Táquez Delgado MA, Riedel J, Fuentes P, Bernabeu E, Rubinstein MR, Lauretta P, Martínez Vivot R, Aguilar MdlÁ, et al. Nanomicellar Formulations Loaded with Histamine and Paclitaxel as a New Strategy to Improve Chemotherapy for Breast Cancer. International Journal of Molecular Sciences. 2023; 24(4):3546. https://doi.org/10.3390/ijms24043546
Chicago/Turabian StyleNicoud, Melisa B., Ignacio A. Ospital, Mónica A. Táquez Delgado, Jennifer Riedel, Pedro Fuentes, Ezequiel Bernabeu, Mara R. Rubinstein, Paolo Lauretta, Rocío Martínez Vivot, María de los Ángeles Aguilar, and et al. 2023. "Nanomicellar Formulations Loaded with Histamine and Paclitaxel as a New Strategy to Improve Chemotherapy for Breast Cancer" International Journal of Molecular Sciences 24, no. 4: 3546. https://doi.org/10.3390/ijms24043546






