Activation of Endothelial Large Conductance Potassium Channels Protects against TNF-α-Induced Inflammation
Abstract
:1. Introduction
2. Results
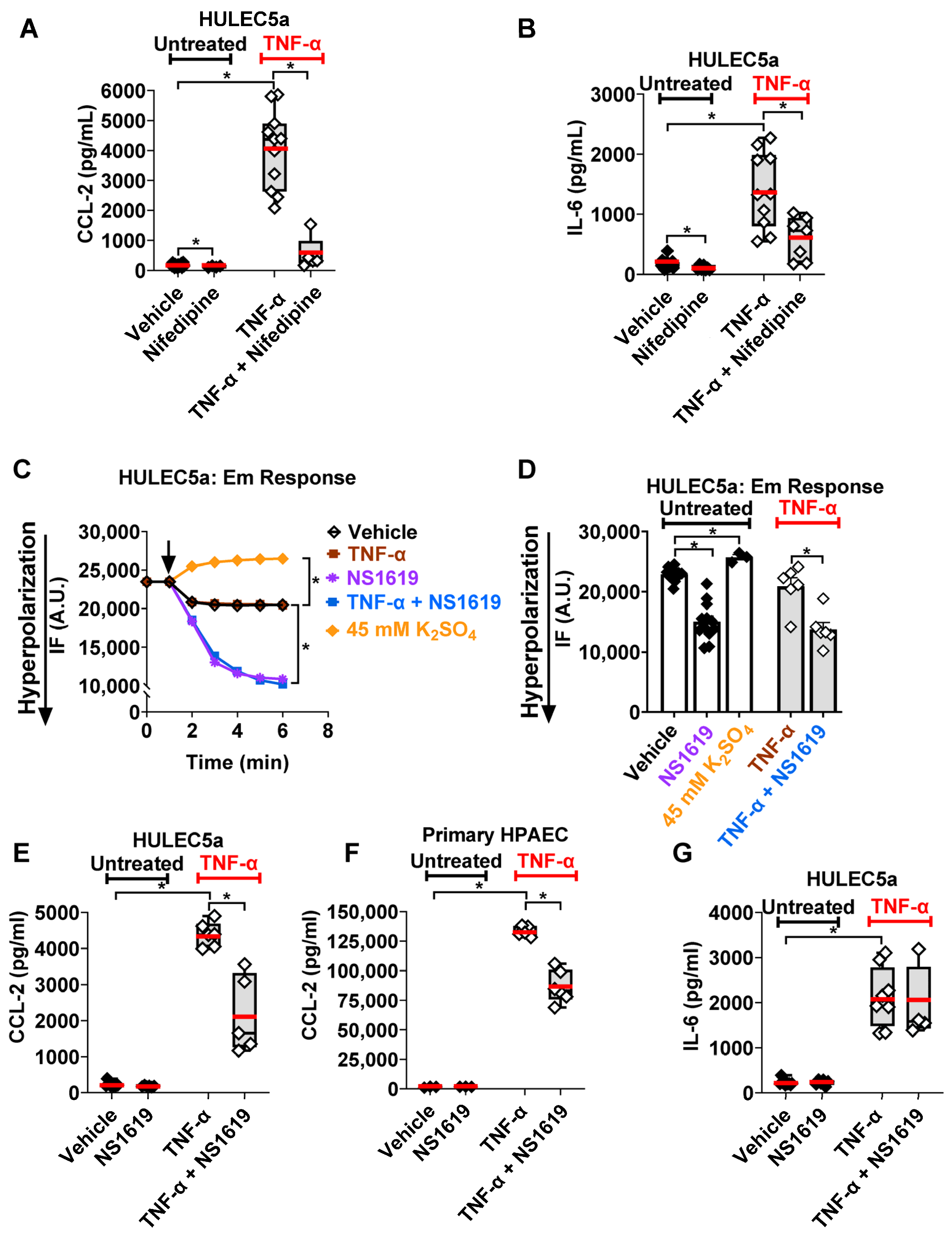
2.1. L-Type CaV Channels Regulate TNF-α-Induced Inflammatory Cytokine Secretion from Human Pulmonary Endothelial Cells
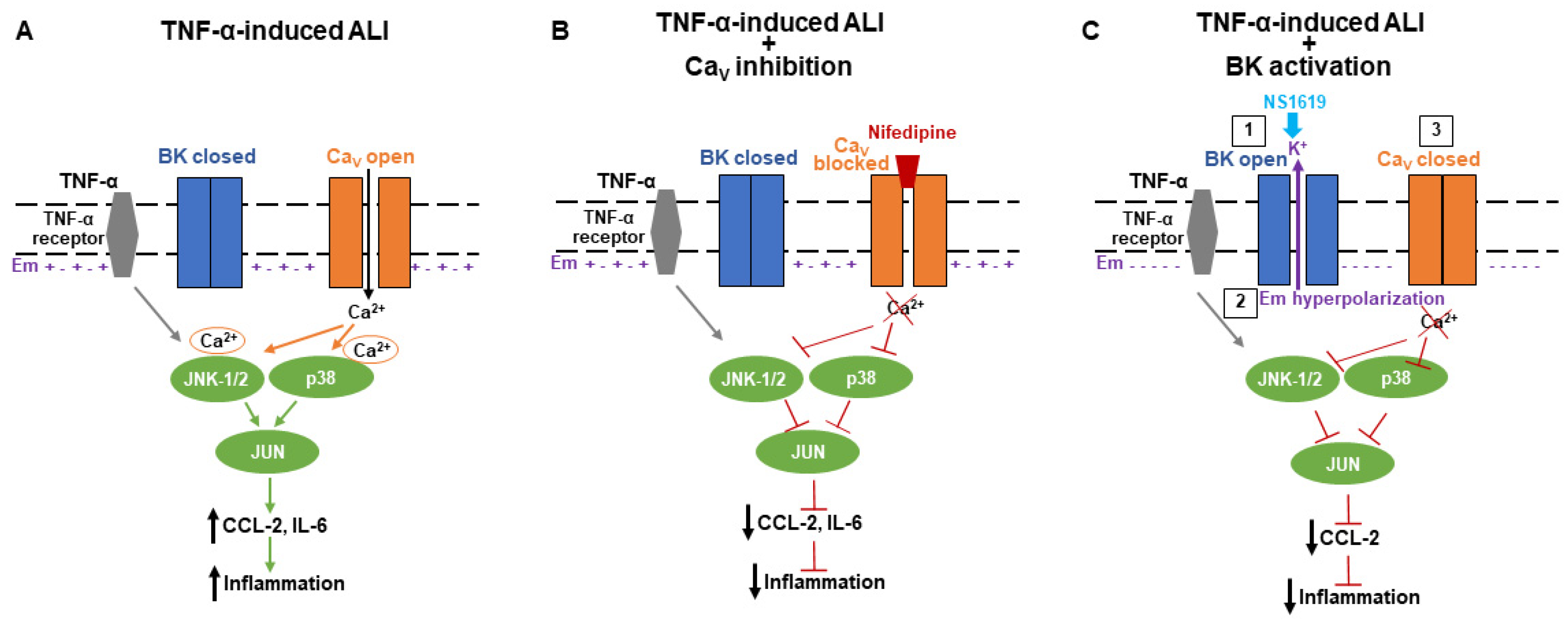
2.2. BK Channel Activation Decreases Inflammatory CCL-2 Secretion via Inhibition of Ca2+-Dependent JNK-1/2 and p38 Signaling Pathways
3. Discussion
4. Materials and Methods
4.1. Cells
4.1.1. Cell Culture Models and Cytokine Measurements
4.1.2. Em Measurements
4.1.3. Gene Expression
4.2. Prediction of Protein–Protein Interaction (PPI) Networks Using Functional Enrichment Analysis (STRING)
4.3. Experimental Validation of Predicted PPI Networks
4.4. Chemical/Drug-Protein Interaction Analysis (STITCH)
5. Significance Statement
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Patel, B.V.; Wilson, M.R.; O’Dea, K.P.; Takata, M. TNF-induced death signaling triggers alveolar epithelial dysfunction in acute lung injury. J. Immunol. 2013, 190, 4274–4282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suter, P.M.; Suter, S.; Girardin, E.; Roux-Lombard, P.; Grau, G.E.; Dayer, J.M. High bronchoalveolar levels of tumor necrosis factor and its inhibitors, interleukin-1, interferon, and elastase, in patients with adult respiratory distress syndrome after trauma, shock, or sepsis. Am. Rev. Respir. Dis. 1992, 145, 1016–1022. [Google Scholar] [CrossRef] [PubMed]
- Agostini, C.; Zambello, R.; Trentin, L.; Cerutti, A.; Enthammer, C.; Facco, M.; Milani, A.; Sancetta, R.; Garbisa, S.; Semenzato, G. Expression of TNF receptors by T cells and membrane TNF-alpha by alveolar macrophages suggests a role for TNF-alpha in the regulation of the local immune responses in the lung of HIV-1-infected patients. J. Immunol. 1995, 154, 2928–2938. [Google Scholar] [CrossRef] [PubMed]
- Lucas, R.; Verin, A.D.; Black, S.M.; Catravas, J.D. Regulators of endothelial and epithelial barrier integrity and function in acute lung injury. Biochem. Pharmacol. 2009, 77, 1763–1772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Li, Z.; Meng, Q.; Zhang, P.; Yan, P.; Zhang, Z.; Zhang, H.; Pan, J.; Zhai, Y.; Liu, Y.; et al. Chronic Calcium Channel Inhibitor Verapamil Antagonizes TNF-alpha-Mediated Inflammatory Reaction and Protects Against Inflammatory Arthritis in Mice. Inflammation 2016, 39, 1624–1634. [Google Scholar] [CrossRef]
- White, T.A.; Xue, A.; Chini, E.N.; Thompson, M.; Sieck, G.C.; Wylam, M.E. Role of transient receptor potential C3 in TNF-alpha-enhanced calcium influx in human airway myocytes. Am. J. Respir. Cell Mol. Biol. 2006, 35, 243–251. [Google Scholar] [CrossRef] [Green Version]
- Toborek, M.; Barger, S.W.; Mattson, M.P.; McClain, C.J.; Hennig, B. Role of glutathione redox cycle in TNF-alpha-mediated endothelial cell dysfunction. Atherosclerosis 1995, 117, 179–188. [Google Scholar] [CrossRef]
- Paria, B.C.; Vogel, S.M.; Ahmmed, G.U.; Alamgir, S.; Shroff, J.; Malik, A.B.; Tiruppathi, C. Tumor necrosis factor-alpha-induced TRPC1 expression amplifies store-operated Ca2+ influx and endothelial permeability. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 287, L1303–L1313. [Google Scholar] [CrossRef] [Green Version]
- Tiruppathi, C.; Minshall, R.D.; Paria, B.C.; Vogel, S.M.; Malik, A.B. Role of Ca2+ signaling in the regulation of endothelial permeability. Vascul. Pharmacol. 2002, 39, 173–185. [Google Scholar] [CrossRef]
- Yang, Y.; Delalio, L.J.; Best, A.K.; Macal, E.; Milstein, J.; Donnelly, I.; Miller, A.M.; McBride, M.; Shu, X.; Koval, M.; et al. Endothelial Pannexin 1 Channels Control Inflammation by Regulating Intracellular Calcium. J. Immunol. 2020, 204, 2995–3007. [Google Scholar] [CrossRef]
- Schwingshackl, A.; Teng, B.; Ghosh, M.; Lim, K.G.; Tigyi, G.; Narayanan, D.; Jaggar, J.H.; Waters, C.M. Regulation of interleukin-6 secretion by the two-pore-domain potassium channel Trek-1 in alveolar epithelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 2013, 304, L276–L286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stapleton, R.D.; Suratt, B.T.; Neff, M.J.; Wurfel, M.M.; Ware, L.B.; Ruzinski, J.T.; Caldwell, E.; Hallstrand, T.S.; Parsons, P.E. Bronchoalveolar fluid and plasma inflammatory biomarkers in contemporary ARDS patients. Biomarkers 2019, 24, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.C.; Lin, C.F.; Chen, C.L.; Chen, C.W.; Lin, Y.S. Prediction of outcome in patients with acute respiratory distress syndrome by bronchoalveolar lavage inflammatory mediators. Exp. Biol. Med. 2010, 235, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Takemura, Y.; Helms, M.N.; Eaton, A.F.; Self, J.; Ramosevac, S.; Jain, L.; Bao, H.F.; Eaton, D.C. Cholinergic regulation of epithelial sodium channels in rat alveolar type 2 epithelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 2013, 304, L428–L437. [Google Scholar] [CrossRef] [Green Version]
- Voets, T.; Droogmans, G.; Nilius, B. Membrane currents and the resting membrane potential in cultured bovine pulmonary artery endothelial cells. J. Physiol. 1996, 497, 95–107. [Google Scholar] [CrossRef] [Green Version]
- Kawaguchi, H.; Sawa, H.; Yasuda, H. Endothelin stimulates angiotensin I to angiotensin II conversion in cultured pulmonary artery endothelial cells. J. Mol. Cell. Cardiol. 1990, 22, 839–842. [Google Scholar] [CrossRef]
- Rethore, L.; Park, J.; Montnach, J.; Nicolas, S.; Khoury, J.; Le Seach, E.; Mabrouk, K.; De Pomyers, H.; Tricoire-Leignel, H.; Mattei, C.; et al. Pharmacological Dissection of the Crosstalk between Na(V) and Ca(V) Channels in GH3b6 Cells. Int. J. Mol. Sci. 2022, 23, 827. [Google Scholar] [CrossRef]
- Zyrianova, T.; Lopez, B.; Liao, A.; Gu, C.; Wong, L.; Ottolia, M.; Olcese, R.; Schwingshackl, A. BK Channels Regulate LPS-induced CCL-2 Release from Human Pulmonary Endothelial Cells. Am. J. Respir. Cell. Mol. Biol. 2021, 64, 224–234. [Google Scholar] [CrossRef]
- Li, D.; Ren, W.; Jiang, Z.; Zhu, L. Regulation of the NLRP3 inflammasome and macrophage pyroptosis by the p38 MAPK signaling pathway in a mouse model of acute lung injury. Mol. Med. Rep. 2018, 18, 4399–4409. [Google Scholar] [CrossRef] [Green Version]
- Gonzales, J.N.; Lucas, R.; Verin, A.D. The Acute Respiratory Distress Syndrome: Mechanisms and Perspective Therapeutic Approaches. Austin. J. Vasc. Med. 2015, 2, 1009. [Google Scholar]
- Chien, J.Y.; Hsueh, P.R.; Cheng, W.C.; Yu, C.J.; Yang, P.C. Temporal changes in cytokine/chemokine profiles and pulmonary involvement in severe acute respiratory syndrome. Respirology 2006, 11, 715–722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwingshackl, A.; Teng, B.; Ghosh, M.; Waters, C.M. Regulation of Monocyte Chemotactic Protein-1 secretion by the Two-Pore-Domain Potassium (K2P) channel TREK-1 in human alveolar epithelial cells. Am. J. Transl. Res. 2013, 5, 530–542. [Google Scholar] [PubMed]
- Villalta, P.C.; Townsley, M.I. Transient receptor potential channels and regulation of lung endothelial permeability. Pulm. Circ. 2013, 3, 802–815. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, H.N.; Zeng, B.; Zhang, Y.; Daskoulidou, N.; Fan, H.; Qu, J.M.; Xu, S.Z. Involvement of TRPC channels in lung cancer cell differentiation and the correlation analysis in human non-small cell lung cancer. PLoS ONE 2013, 8, e67637. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Proost, I.; Brouns, I.; Pintelon, I.; Timmermans, J.P.; Adriaensen, D. Pulmonary expression of voltage-gated calcium channels: Special reference to sensory airway receptors. Histochem. Cell Biol. 2007, 128, 301–316. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Long, H.; Zhao, J.; Zhong, G.; Li, J. Nifedipine inhibits oxidative stress and ameliorates osteoarthritis by activating the nuclear factor erythroid-2-related factor 2 pathway. Life Sci. 2020, 253, 117292. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, S.; Inagaki, Y.; Kikuchi, S. Nifedipine inhibits tumor necrosis factor-alpha-induced monocyte chemoattractant protein-1 overexpression by blocking NADPH oxidase-mediated reactive oxygen species generation. Drugs Exp. Clin. Res. 2003, 29, 147–152. [Google Scholar]
- Gao, X.; Iwai, M.; Inaba, S.; Tomono, Y.; Kanno, H.; Mogi, M.; Horiuchi, M. Attenuation of monocyte chemoattractant protein-1 expression via inhibition of nuclear factor-kappaB activity in inflammatory vascular injury. Am. J. Hypertens. 2007, 20, 1170–1175. [Google Scholar] [CrossRef] [Green Version]
- Filippini, A.; D’Amore, A.; D’Alessio, A. Calcium Mobilization in Endothelial Cell Functions. Int. J. Mol. Sci. 2019, 20, 4525. [Google Scholar] [CrossRef] [Green Version]
- Czeschik, J.C.; Hagenacker, T.; Schafers, M.; Busselberg, D. TNF-alpha differentially modulates ion channels of nociceptive neurons. Neurosci. Lett. 2008, 434, 293–298. [Google Scholar] [CrossRef]
- Rehn, M.; Hubschle, T.; Diener, M. TNF-alpha hyperpolarizes membrane potential and potentiates the response to nicotinic receptor stimulation in cultured rat myenteric neurones. Acta Physiol. Scand. 2004, 181, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Nietsch, H.H.; Roe, M.W.; Fiekers, J.F.; Moore, A.L.; Lidofsky, S.D. Activation of potassium and chloride channels by tumor necrosis factor alpha. Role in liver cell death. J. Biol. Chem. 2000, 275, 20556–20561. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, S.; Wang, H.N.; Xu, L.J.; Li, F.; Miao, Y.; Lei, B.; Sun, X.; Wang, Z. Soluble tumor necrosis factor-alpha-induced hyperexcitability contributes to retinal ganglion cell apoptosis by enhancing Nav1.6 in experimental glaucoma. J. Neuroinflamm. 2021, 18, 182. [Google Scholar] [CrossRef] [PubMed]
- Dopico, A.M.; Bukiya, A.N.; Jaggar, J.H. Calcium- and voltage-gated BK channels in vascular smooth muscle. Pflugers Arch. 2018, 470, 1271–1289. [Google Scholar] [CrossRef]
- Voiriot, G.; Razazi, K.; Amsellem, V.; Tran Van Nhieu, J.; Abid, S.; Adnot, S.; Mekontso Dessap, A.; Maitre, B. Interleukin-6 displays lung anti-inflammatory properties and exerts protective hemodynamic effects in a double-hit murine acute lung injury. Respir. Res. 2017, 18, 64. [Google Scholar] [CrossRef] [Green Version]
- Ward, N.S.; Waxman, A.B.; Homer, R.J.; Mantell, L.L.; Einarsson, O.; Du, Y.; Elias, J.A. Interleukin-6-induced protection in hyperoxic acute lung injury. Am. J. Respir. Cell. Mol. Biol. 2000, 22, 535–542. [Google Scholar] [CrossRef]
- Liao, L.; Liao, P. Bioinformatics analysis of the potential biomarkers for acute respiratory distress syndrome. Biosci. Rep. 2020, 40, BSR20192436. [Google Scholar] [CrossRef]
- Patel, J.K.; Clifford, R.L.; Deacon, K.; Knox, A.J. Ciclesonide inhibits TNFalpha- and IL-1beta-induced monocyte chemotactic protein-1 (MCP-1/CCL2) secretion from human airway smooth muscle cells. Am. J. Physiol. Lung Cell Mol. Physiol. 2012, 302, L785–L792. [Google Scholar] [CrossRef] [Green Version]
- Miakotina, O.L.; Snyder, J.M. TNF-alpha inhibits SP-A gene expression in lung epithelial cells via p38 MAPK. Am. J. Physiol. Lung Cell Mol. Physiol. 2002, 283, L418–L427. [Google Scholar] [CrossRef]
- Matikainen, S.; Siren, J.; Tissari, J.; Veckman, V.; Pirhonen, J.; Severa, M.; Sun, Q.; Lin, R.; Meri, S.; Uze, G.; et al. Tumor necrosis factor alpha enhances influenza A virus-induced expression of antiviral cytokines by activating RIG-I gene expression. J. Virol. 2006, 80, 3515–3522. [Google Scholar] [CrossRef] [Green Version]
- Naidu, P.S.; Velarde, V.; Kappler, C.S.; Young, R.C.; Mayfield, R.K.; Jaffa, A.A. Calcium-calmodulin mediates bradykinin-induced MAPK phosphorylation and c-fos induction in vascular cells. Am. J. Physiol. 1999, 277, H1061–H1068. [Google Scholar] [CrossRef] [PubMed]
- Yap, G.L.R.; Sachaphibulkij, K.; Foo, S.L.; Cui, J.; Fairhurst, A.M.; Lim, L.H.K. Annexin-A1 promotes RIG-I-dependent signaling and apoptosis via regulation of the IRF3-IFNAR-STAT1-IFIT1 pathway in A549 lung epithelial cells. Cell Death Dis. 2020, 11, 463. [Google Scholar] [CrossRef] [PubMed]
- Pothlichet, J.; Meunier, I.; Davis, B.K.; Ting, J.P.; Skamene, E.; von Messling, V.; Vidal, S.M. Type I IFN triggers RIG-I/TLR3/NLRP3-dependent inflammasome activation in influenza A virus infected cells. PLoS Pathog. 2013, 9, e1003256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szklarczyk, D.; Santos, A.; von Mering, C.; Jensen, L.J.; Bork, P.; Kuhn, M. STITCH 5: Augmenting protein-chemical interaction networks with tissue and affinity data. Nucleic Acids Res. 2016, 44, D380–D384. [Google Scholar] [CrossRef] [PubMed]
- Adiseshaiah, P.; Kalvakolanu, D.V.; Reddy, S.P. A JNK-independent signaling pathway regulates TNF alpha-stimulated, c-Jun-driven FRA-1 protooncogene transcription in pulmonary epithelial cells. J. Immunol. 2006, 177, 7193–7202. [Google Scholar] [CrossRef] [Green Version]
- Morel, C.; Standen, C.L.; Jung, D.Y.; Gray, S.; Ong, H.; Flavell, R.A.; Kim, J.K.; Davis, R.J. Requirement of JIP1-mediated c-Jun N-terminal kinase activation for obesity-induced insulin resistance. Mol. Cell Biol. 2010, 30, 4616–4625. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.; Zhou, Y.; Zhou, L.; Fu, Z.; Yang, C.; Zhao, L.; Li, S.; Chen, Y.; Wu, Y.; Ling, Z.; et al. TRPC6-dependent Ca(2+) signaling mediates airway inflammation in response to oxidative stress via ERK pathway. Cell Death Dis. 2020, 11, 170. [Google Scholar] [CrossRef] [Green Version]
- Xu, C.; Chen, S.; Xu, M.; Chen, X.; Wang, X.; Zhang, H.; Dong, X.; Zhang, R.; Chen, X.; Gao, W.; et al. Cadmium Impairs Autophagy Leading to Apoptosis by Ca(2+)-Dependent Activation of JNK Signaling Pathway in Neuronal Cells. Neurochem. Res. 2021, 46, 2033–2045. [Google Scholar] [CrossRef]
- Stocco, C.O.; Lau, L.F.; Gibori, G. A calcium/calmodulin-dependent activation of ERK1/2 mediates JunD phosphorylation and induction of nur77 and 20alpha-hsd genes by prostaglandin F2alpha in ovarian cells. J. Biol. Chem. 2002, 277, 3293–3302. [Google Scholar] [CrossRef] [Green Version]
- Lorigo, M.; Oliveira, N.; Cairrao, E. Clinical Importance of the Human Umbilical Artery Potassium Channels. Cells 2020, 9, 1956. [Google Scholar] [CrossRef]
- Dai, H.; Wang, M.; Patel, P.N.; Kalogeris, T.; Liu, Y.; Durante, W.; Korthuis, R.J. Preconditioning with the BKCa channel activator NS-1619 prevents ischemia-reperfusion-induced inflammation and mucosal barrier dysfunction: Roles for ROS and heme oxygenase-1. Am. J. Physiol. Heart Circ. Physiol. 2017, 313, H988–H999. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eichhorn, B.; Dobrev, D. Vascular large conductance calcium-activated potassium channels: Functional role and therapeutic potential. Naunyn Schmiedebergs Arch. Pharm. 2007, 376, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, G.; Cao, T.; Zhang, L.; Ma, Y.; Jiang, S.; Teng, X.; Sun, X. Large-conductance calcium-activated potassium channels mediate lipopolysaccharide-induced activation of murine microglia. J. Biol. Chem. 2019, 294, 12921–12932. [Google Scholar] [CrossRef] [PubMed]
- Kicinska, A.; Augustynek, B.; Kulawiak, B.; Jarmuszkiewicz, W.; Szewczyk, A.; Bednarczyk, P. A large-conductance calcium-regulated K+ channel in human dermal fibroblast mitochondria. Biochem. J. 2016, 473, 4457–4471. [Google Scholar] [CrossRef]
- Schulz, R.; Di Lisa, F. Mitochondrial potassium homeostasis: A central player in cardioprotection. Cardiovasc. Res. 2016, 110, 4–5. [Google Scholar] [CrossRef] [Green Version]
- Debska, G.; Kicinska, A.; Dobrucki, J.; Dworakowska, B.; Nurowska, E.; Skalska, J.; Dolowy, K.; Szewczyk, A. Large-conductance K+ channel openers NS1619 and NS004 as inhibitors of mitochondrial function in glioma cells. Biochem. Pharmacol. 2003, 65, 1827–1834. [Google Scholar] [CrossRef]
- Kicinska, A.; Szewczyk, A. Large-Conductance Potassium Cation Channel Opener NS1619 Inhibits Cardiac Mitochondria Respiratory Chain. Toxicol. Mech. Methods 2004, 14, 59–61. [Google Scholar] [CrossRef]
- Dubinin, M.V.; Starinets, V.S.; Belosludtseva, N.V.; Mikheeva, I.B.; Chelyadnikova, Y.A.; Igoshkina, A.D.; Vafina, A.B.; Vedernikov, A.A.; Belosludtsev, K.N. BK(Ca) Activator NS1619 Improves the Structure and Function of Skeletal Muscle Mitochondria in Duchenne Dystrophy. Pharmaceutics 2022, 14, 2336. [Google Scholar] [CrossRef]
- Soder, R.P.; Petkov, G.V. Large conductance Ca2+-activated K+ channel activation with NS1619 decreases myogenic and neurogenic contractions of rat detrusor smooth muscle. Eur. J. Pharmacol. 2011, 670, 252–259. [Google Scholar] [CrossRef] [Green Version]
- Hu, Y.; Yang, G.; Xiao, X.; Liu, L.; Li, T. Bkca opener, NS1619 pretreatment protects against shock-induced vascular hyporeactivity through PDZ-Rho GEF-RhoA-Rho kinase pathway in rats. J. Trauma Acute Care Surg. 2014, 76, 394–401. [Google Scholar] [CrossRef]
- Wang, M.; Yin, X.; Li, S.; Zhang, X.; Yi, M.; He, C.; Li, X.; Wang, W.; Zhang, S.; Liu, H. Large-Conductance Calcium-Activated Potassium Channel Opener, NS1619, Protects Against Mesenteric Artery Remodeling Induced by Agonistic Autoantibodies Against the Angiotensin II Type 1 Receptor. J. Am. Heart Assoc. 2022, 11, e024046. [Google Scholar] [CrossRef]
- Sutovska, M.; Nosalova, G.; Franova, S. The role of potassium ion channels in cough and other reflexes of the airways. J. Physiol. Pharmacol. 2007, 58, 673–683. [Google Scholar]
- Meredith, A.L.; Thorneloe, K.S.; Werner, M.E.; Nelson, M.T.; Aldrich, R.W. Overactive bladder and incontinence in the absence of the BK large conductance Ca2+-activated K+ channel. J. Biol. Chem. 2004, 279, 36746–36752. [Google Scholar] [CrossRef] [PubMed]
- Dhungel, K.U.; Kim, T.W.; Sharma, N.; Bhattarai, J.P.; Park, S.A.; Han, S.K.; Kim, C.J. Magnesium increases iberiotoxin-sensitive large conductance calcium activated potassium currents on the basilar artery smooth muscle cells in rabbits. Neurol. Res. 2012, 34, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Castranova, V.; Jones, G.S.; Miles, P.R. Transmembrane potential of isolated rat alveolar type II cells. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1983, 54, 1511–1517. [Google Scholar] [CrossRef] [PubMed]
- Bastarache, J.A.; Koyama, T.; Wickersham, N.E.; Ware, L.B. Validation of a multiplex electrochemiluminescent immunoassay platform in human and mouse samples. J. Immunol. Methods 2014, 408, 13–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zyrianova, T.; Zou, K.; Lopez, B.; Liao, A.; Gu, C.; Olcese, R.; Schwingshackl, A. Activation of Endothelial Large Conductance Potassium Channels Protects against TNF-α-Induced Inflammation. Int. J. Mol. Sci. 2023, 24, 4087. https://doi.org/10.3390/ijms24044087
Zyrianova T, Zou K, Lopez B, Liao A, Gu C, Olcese R, Schwingshackl A. Activation of Endothelial Large Conductance Potassium Channels Protects against TNF-α-Induced Inflammation. International Journal of Molecular Sciences. 2023; 24(4):4087. https://doi.org/10.3390/ijms24044087
Chicago/Turabian StyleZyrianova, Tatiana, Kathlyn Zou, Benjamin Lopez, Andy Liao, Charles Gu, Riccardo Olcese, and Andreas Schwingshackl. 2023. "Activation of Endothelial Large Conductance Potassium Channels Protects against TNF-α-Induced Inflammation" International Journal of Molecular Sciences 24, no. 4: 4087. https://doi.org/10.3390/ijms24044087





