A Postsynaptic Density Immediate Early Gene-Based Connectome Analysis of Acute NMDAR Blockade and Reversal Effect of Antipsychotic Administration
Abstract
:1. Introduction
2. Results
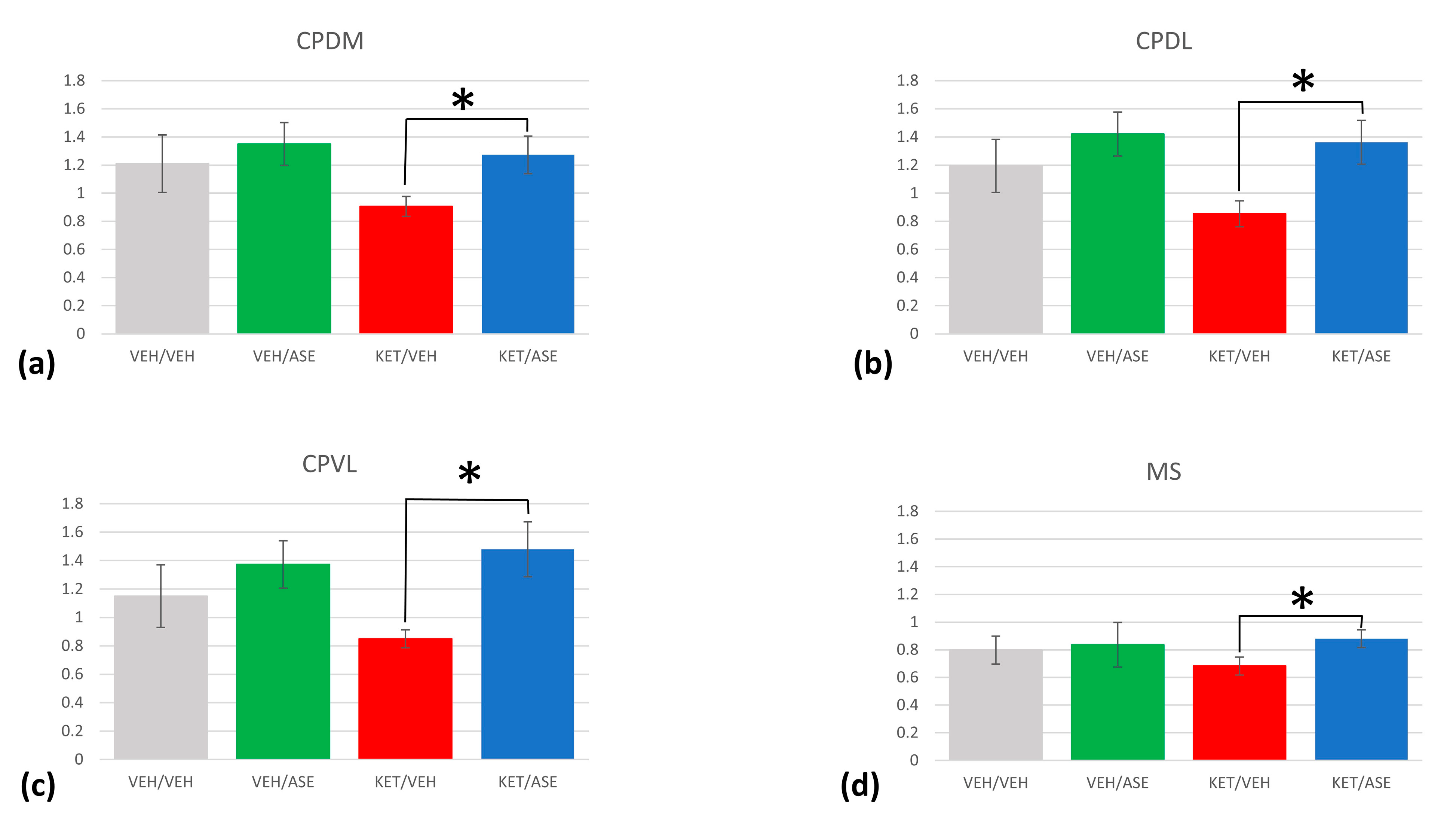
2.1. Gene Expression Analysis: Comparison between Experimental Groups in Homer1a mRNA Levels
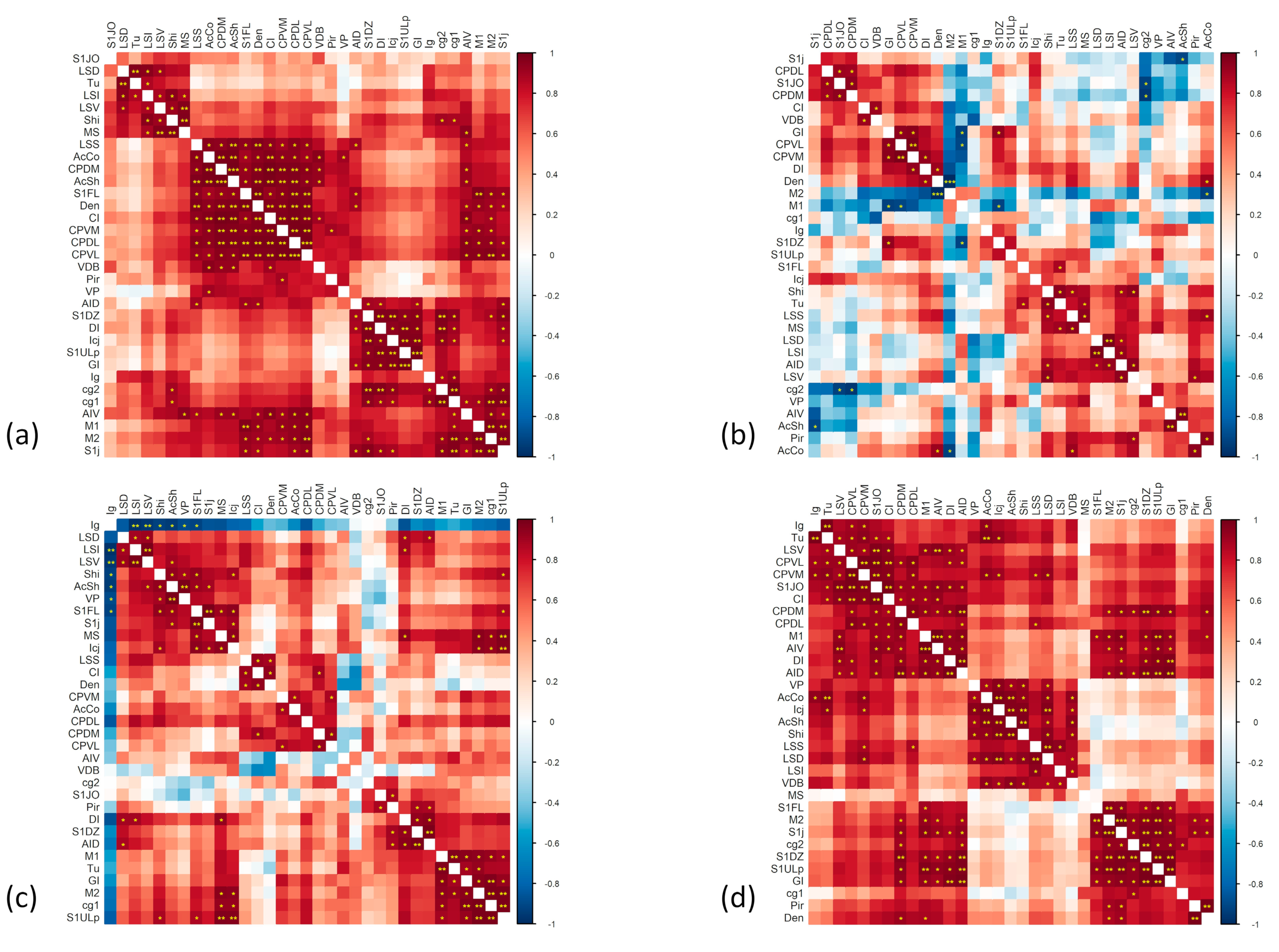
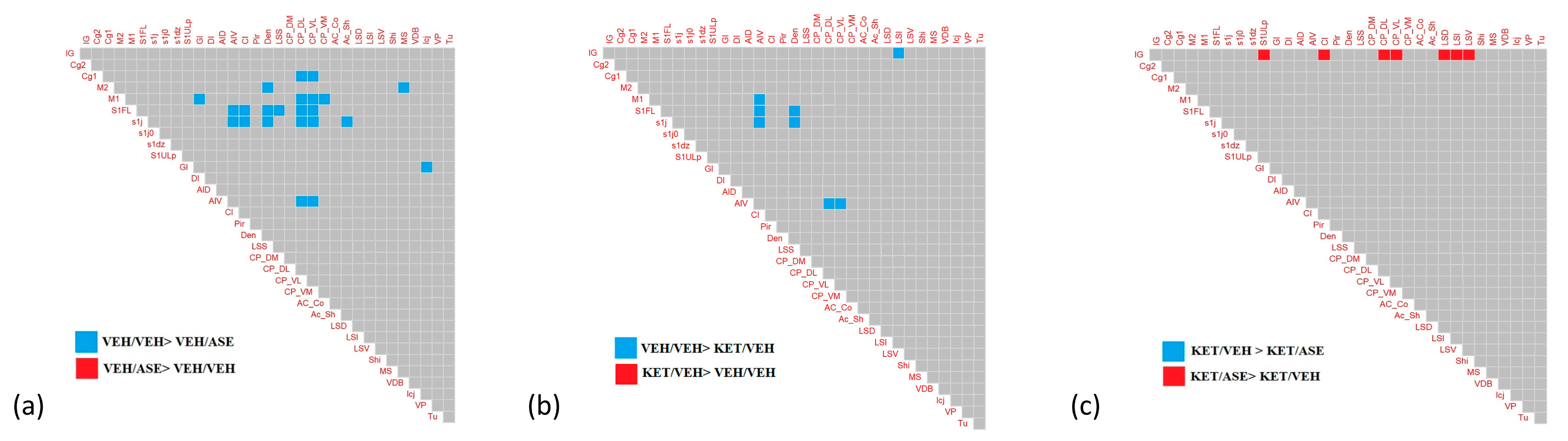
2.2. Generation and Comparison of the Correlation Matrices for the Connectivity Analysis
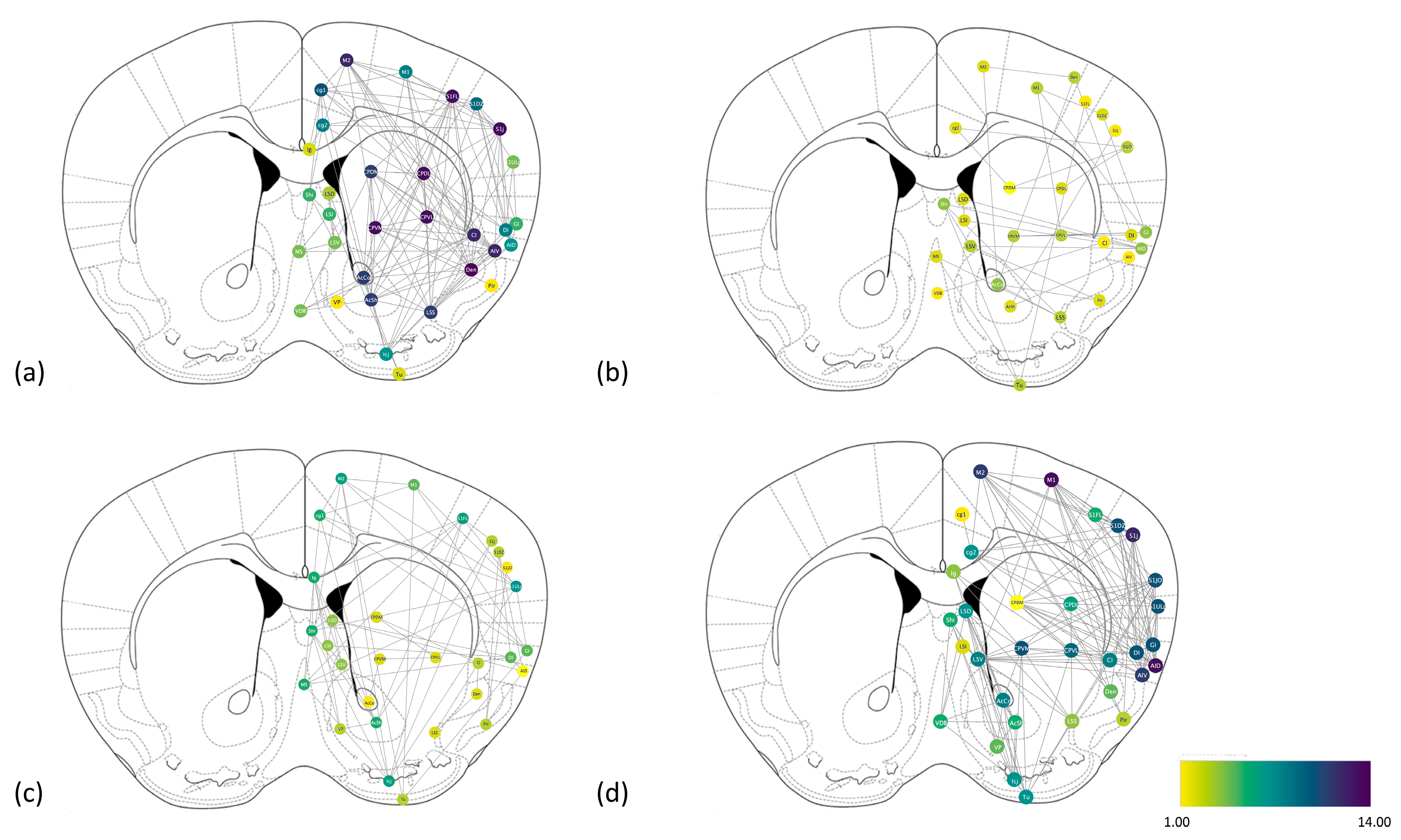
2.3. Construction and Comparison of the Brain Networks
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Drug Treatment and Study Design
4.3. In Situ Hybridization Histochemistry (ISHH)
4.4. Image Analysis
4.5. Statistical Analysis
Comparison of the Correlation Matrices and Generation of Networks
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Jones, C.A.; Watson, D.J.; Fone, K.C. Animal models of schizophrenia. Br. J. Pharmacol. 2011, 164, 1162–1194. [Google Scholar] [CrossRef] [PubMed]
- Network and Pathway Analysis Subgroup of Psychiatric Genomics Consortium. Psychiatric genome-wide association study analyses implicate neuronal, immune and histone pathways. Nat. Neurosci. 2015, 18, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Matosin, N.; Fernandez-Enright, F.; Lum, J.S.; Engel, M.; Andrews, J.L.; Gassen, N.C.; Wagner, K.V.; Schmidt, M.V.; Newell, K.A. Molecular evidence of synaptic pathology in the CA1 region in schizophrenia. NPJ Schizophr. 2016, 2, 16022. [Google Scholar] [CrossRef] [Green Version]
- Kruse, A.O.; Bustillo, J.R. Glutamatergic dysfunction in Schizophrenia. Transl. Psychiatry 2022, 12, 500. [Google Scholar] [CrossRef]
- Garey, L.J.; Ong, W.Y.; Patel, T.S.; Kanani, M.; Davis, A.; Mortimer, A.M.; Barnes, T.R.; Hirsch, S.R. Reduced dendritic spine density on cerebral cortical pyramidal neurons in schizophrenia. J. Neurol. Neurosurg. Psychiatry 1998, 65, 446–453. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, M.L.; Alhassan, J.; Newman, J.T.; Richard, M.; Gu, H.; Kelly, R.M.; Sampson, A.R.; Fish, K.N.; Penzes, P.; Wills, Z.P.; et al. Selective Loss of Smaller Spines in Schizophrenia. Am. J. Psychiatry 2017, 174, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Obi-Nagata, K.; Temma, Y.; Hayashi-Takagi, A. Synaptic functions and their disruption in schizophrenia: From clinical evidence to synaptic optogenetics in an animal model. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2019, 95, 179–197. [Google Scholar] [CrossRef] [Green Version]
- Catts, V.S.; Derminio, D.S.; Hahn, C.G.; Weickert, C.S. Postsynaptic density levels of the NMDA receptor NR1 subunit and PSD-95 protein in prefrontal cortex from people with schizophrenia. NPJ Schizophr. 2015, 1, 15037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engmann, O.; Hortobágyi, T.; Pidsley, R.; Troakes, C.; Bernstein, H.-G.; Kreutz, M.R.; Mill, J.; Nikolic, M.; Giese, K.P. Schizophrenia is associated with dysregulation of a Cdk5 activator that regulates synaptic protein expression and cognition. Brain 2011, 134, 2408–2421. [Google Scholar] [CrossRef] [Green Version]
- McNabb, C.B.; Tait, R.J.; McIlwain, M.E.; Anderson, V.M.; Suckling, J.; Kydd, R.R.; Russell, B.R. Functional network dysconnectivity as a biomarker of treatment resistance in schizophrenia. Schizophr. Res. 2018, 195, 160–167. [Google Scholar] [CrossRef] [Green Version]
- Blazer, A.; Chengappa, K.N.R.; Foran, W.; Parr, A.C.; Kahn, C.E.; Luna, B.; Sarpal, D.K. Changes in corticostriatal connectivity and striatal tissue iron associated with efficacy of clozapine for treatment-resistant schizophrenia. Psychopharmacology 2022, 239, 2503–2514. [Google Scholar] [CrossRef] [PubMed]
- Barone, A.; Signoriello, S.; Latte, G.; Vellucci, L.; Giordano, G.; Avagliano, C.; Buonaguro, E.F.; Marmo, F.; Tomasetti, C.; Iasevoli, F.; et al. Modulation of glutamatergic functional connectivity by a prototypical antipsychotic: Translational inference from a postsynaptic density immediate-early gene-based network analysis. Behav. Brain Res. 2021, 404, 113160. [Google Scholar] [CrossRef]
- Shahid, M.; Walker, G.B.; Zorn, S.H.; Wong, E.H. Asenapine: A novel psychopharmacologic agent with a unique human receptor signature. J. Psychopharmacol. 2009, 23, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xu, T.X.; Hallett, P.J.; Watanabe, M.; Grant, S.G.; Isacson, O.; Yao, W.D. PSD-95 uncouples dopamine-glutamate interaction in the D1/PSD-95/NMDA receptor complex. J. Neurosci. 2009, 29, 2948–2960. [Google Scholar] [CrossRef] [Green Version]
- Karlsgodt, K.H.; Sun, D.; Cannon, T.D. Structural and Functional Brain Abnormalities in Schizophrenia. Curr. Dir. Psychol. Sci. 2010, 19, 226–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCutcheon, R.A.; Abi-Dargham, A.; Howes, O.D. Schizophrenia, Dopamine and the Striatum: From Biology to Symptoms. Trends Neurosci. 2019, 42, 205–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwarz, A.J.; McGonigle, J. Negative edges and soft thresholding in complex network analysis of resting state functional connectivity data. NeuroImage 2011, 55, 1132–1146. [Google Scholar] [CrossRef] [PubMed]
- Zhan, L.; Jenkins, L.M.; Wolfson, O.E.; GadElkarim, J.J.; Nocito, K.; Thompson, P.M.; Ajilore, O.A.; Chung, M.K.; Leow, A.D. The significance of negative correlations in brain connectivity. J. Comp. Neurol. 2017, 525, 3251–3265. [Google Scholar] [CrossRef]
- Carmena, A.; Granado, N.; Ares Santos, S.; Alberquilla, S.; Tizabi, Y.; Moratalla, R. Methamphetamine-Induced Toxicity in Indusium Griseum of Mice is Associated with Astro- and Microgliosis. Neurotox. Res. 2015, 27, 209–216. [Google Scholar] [CrossRef] [Green Version]
- Fuzik, J.; Rehman, S.; Girach, F.; Miklosi, A.G.; Korchynska, S.; Arque, G.; Romanov, R.A.; Hanics, J.; Wagner, L.; Meletis, K.; et al. Brain-wide genetic mapping identifies the indusium griseum as a prenatal target of pharmacologically unrelated psychostimulants. Proc. Natl. Acad. Sci. USA 2019, 116, 25958–25967. [Google Scholar] [CrossRef]
- Kokkinou, M.; Ashok, A.H.; Howes, O.D. The effects of ketamine on dopaminergic function: Meta-analysis and review of the implications for neuropsychiatric disorders. Mol. Psychiatry 2018, 23, 59–69. [Google Scholar] [CrossRef] [Green Version]
- Lipska, B.K.; Weinberger, D.R. To model a psychiatric disorder in animals: Schizophrenia as a reality test. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2000, 23, 223–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krystal, J.H.; Karper, L.P.; Seibyl, J.P.; Freeman, G.K.; Delaney, R.; Bremner, J.D.; Heninger, G.R.; Bowers, M.B., Jr.; Charney, D.S. Subanesthetic Effects of the Noncompetitive NMDA Antagonist, Ketamine, in Humans: Psychotomimetic, Perceptual, Cognitive, and Neuroendocrine Responses. Arch. Gen. Psychiatry 1994, 51, 199–214. [Google Scholar] [CrossRef]
- Lahti, A.C.; Koffel, B.; LaPorte, D.; Tamminga, C.A. Subanesthetic doses of ketamine stimulate psychosis in schizophrenia. Neuropsychopharmacol 1995, 13, 9–19. [Google Scholar] [CrossRef]
- Iasevoli, F.; Polese, D.; Ambesi-Impiombato, A.; Muscettola, G.; de Bartolomeis, A. Ketamine-related expression of glutamatergic postsynaptic density genes: Possible implications in psychosis. Neurosci. Lett. 2007, 416, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Buonaguro, E.F.; Tomasetti, C.; Chiodini, P.; Marmo, F.; Latte, G.; Rossi, R.; Avvisati, L.; Iasevoli, F.; de Bartolomeis, A. Postsynaptic density protein transcripts are differentially modulated by minocycline alone or in add-on to haloperidol: Implications for treatment resistant schizophrenia. J. Psychopharmacol. 2017, 31, 406–417. [Google Scholar] [CrossRef] [PubMed]
- De Bartolomeis, A.; Iasevoli, F.; Marmo, F.; Buonaguro, E.F.; Eramo, A.; Rossi, R.; Avvisati, L.; Latte, G.; Tomasetti, C. Progressive recruitment of cortical and striatal regions by inducible postsynaptic density transcripts after increasing doses of antipsychotics with different receptor profiles: Insights for psychosis treatment. Eur. Neuropsychopharmacol. J. Eur. Coll. Neuropsychopharmacol. 2015, 25, 566–582. [Google Scholar] [CrossRef]
- Iasevoli, F.; Tomasetti, C.; Ambesi-Impiombato, A.; Muscettola, G.; de Bartolomeis, A. Dopamine receptor subtypes contribution to Homer1a induction: Insights into antipsychotic molecular action. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2009, 33, 813–821. [Google Scholar] [CrossRef]
- Wirkner, K.; Krause, T.; Köles, L.; Thümmler, S.; Al-Khrasani, M.; Illes, P. D1 but not D2 dopamine receptors or adrenoceptors mediate dopamine-induced potentiation of N-methyl-d-aspartate currents in the rat prefrontal cortex. Neurosci. Lett. 2004, 372, 89–93. [Google Scholar] [CrossRef]
- Yuen, E.Y.; Jiang, Q.; Chen, P.; Feng, J.; Yan, Z. Activation of 5-HT2A/C receptors counteracts 5-HT1A regulation of n-methyl-D-aspartate receptor channels in pyramidal neurons of prefrontal cortex. J. Biol. Chem. 2008, 283, 17194–17204. [Google Scholar] [CrossRef] [Green Version]
- Tarazi, F.I.; Moran-Gates, T.; Wong, E.H.; Henry, B.; Shahid, M. Asenapine induces differential regional effects on serotonin receptor subtypes. J. Psychopharmacol. 2010, 24, 341–348. [Google Scholar] [CrossRef]
- Hall, H.; Sedvall, G.; Magnusson, O.; Kopp, J.; Halldin, C.; Farde, L. Distribution of D1- and D2-dopamine receptors, and dopamine and its metabolites in the human brain. Neuropsychopharmacol 1994, 11, 245–256. [Google Scholar] [CrossRef]
- Duarte, I.C.; Coelho, G.; Brito-Costa, S.; Cayolla, R.; Afonso, S.; Castelo-Branco, M. Ventral Caudate and Anterior Insula Recruitment During Value Estimation of Passionate Rewarding Cues. Front. Neurosci. 2020, 14, 678. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.B.; Alloway, K.D.; Hof, P.R.; Orman, R.; Reser, D.H.; Watakabe, A.; Watson, G.D.R. The relationship between the claustrum and endopiriform nucleus: A perspective towards consensus on cross-species homology. J. Comp. Neurol. 2019, 527, 476–499. [Google Scholar] [CrossRef]
- Wylie, K.P.; Tregellas, J.R. The role of the insula in schizophrenia. Schizophr. Res. 2010, 123, 93–104. [Google Scholar] [CrossRef] [Green Version]
- Harrison, P.J. The hippocampus in schizophrenia: A review of the neuropathological evidence and its pathophysiological implications. Psychopharmacology 2004, 174, 151–162. [Google Scholar] [CrossRef]
- Laplante, F.; Mnie-Filali, O.; Sullivan, R.M. A neuroanatomical and neurochemical study of the indusium griseum and anterior hippocampal continuation: Comparison with dentate gyrus. J. Chem. Neuroanat. 2013, 50–51, 39–47. [Google Scholar] [CrossRef]
- Edmiston, E.K.; Song, Y.; Chang, M.; Yin, Z.; Zhou, Q.; Zhou, Y.; Jiang, X.; Wei, S.; Xu, K.; Tang, Y.; et al. Hippocampal Resting State Functional Connectivity in Patients with Schizophrenia and Unaffected Family Members. Front. Psychiatry 2020, 11, 278. [Google Scholar] [CrossRef]
- Margulies, D.S.; Kelly, A.M.C.; Uddin, L.Q.; Biswal, B.B.; Castellanos, F.X.; Milham, M.P. Mapping the functional connectivity of anterior cingulate cortex. NeuroImage 2007, 37, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Van den Heuvel, M.P.; Mandl, R.C.W.; Stam, C.J.; Kahn, R.S.; Hulshoff Pol, H.E. Aberrant Frontal and Temporal Complex Network Structure in Schizophrenia: A Graph Theoretical Analysis. J. Neurosci. 2010, 30, 15915. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Egerton, A.; Griffiths, K.; Casetta, C.; Deakin, B.; Drake, R.; Howes, O.D.; Kassoumeri, L.; Khan, S.; Lankshear, S.; Lees, J.; et al. Anterior cingulate glutamate metabolites as a predictor of antipsychotic response in first episode psychosis: Data from the STRATA collaboration. Neuropsychopharmacol 2023, 48, 567–575. [Google Scholar] [CrossRef]
- Besnard, A.; Leroy, F. Top-down regulation of motivated behaviors via lateral septum sub-circuits. Mol. Psychiatry 2022, 27, 3119–3128. [Google Scholar] [CrossRef]
- Tropea, D.; Molinos, I.; Petit, E.; Bellini, S.; Nagakura, I.; O’Tuathaigh, C.; Schorova, L.; Mitchell, K.J.; Waddington, J.; Sur, M.; et al. Disrupted in schizophrenia 1 (DISC1) L100P mutants have impaired activity-dependent plasticity in vivo and in vitro. Transl. Psychiatry 2016, 6, e712. [Google Scholar] [CrossRef] [Green Version]
- Ambesi-Impiombato, A.; D’Urso, G.; Muscettola, G.; de Bartolomeis, A. Method for quantitative in situ hybridization histochemistry and image analysis applied for Homer1a gene expression in rat brain. Brain Res. Brain Res. Protoc. 2003, 11, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Becker, A.; Peters, B.; Schroeder, H.; Mann, T.; Huether, G.; Grecksch, G. Ketamine-induced changes in rat behaviour: A possible animal model of schizophrenia. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2003, 27, 687–700. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Nakahara, T.; Yamada, H.; Hirano, M.; Kuroki, T.; Kanba, S. A neurotoxic dose of methamphetamine induces gene expression of Homer 1a, but not Homer 1b or 1c, in the striatum and nucleus accumbens. Neurochem. Int. 2007, 51, 227–232. [Google Scholar] [CrossRef]
- Ghasemzadeh, M.B.; Windham, L.K.; Lake, R.W.; Acker, C.J.; Kalivas, P.W. Cocaine activates Homer1 immediate early gene transcription in the mesocorticolimbic circuit: Differential regulation by dopamine and glutamate signaling. Synapse 2009, 63, 42–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iasevoli, F.; Ambesi-Impiombato, A.; Fiore, G.; Panariello, F.; Muscettola, G.; de Bartolomeis, A. Pattern of acute induction of Homer1a gene is preserved after chronic treatment with first- and second-generation antipsychotics: Effect of short-term drug discontinuation and comparison with Homer1a-interacting genes. J. Psychopharmacol. 2011, 25, 875–887. [Google Scholar] [CrossRef] [PubMed]
- Ohyama, M.; Kondo, M.; Yamauchi, M.; Imanishi, T.; Koyama, T. Asenapine reduces anxiety-related behaviours in rat conditioned fear stress model. Acta Neuropsychiatr. 2016, 28, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Majercikova, Z.; Kiss, A. Stress alters asenapine-induced Fos expression in the Meynert’s nucleus: Response of adjacent hypocretin and melanin-concentrating hormone neurons in rat. Neurol. Res. 2016, 38, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates—The New Coronal Set, 5th ed.; Academic Press: Cambridge, MA, USA, 2004. [Google Scholar]
- Tomasetti, C.; Dell’Aversano, C.; Iasevoli, F.; de Bartolomeis, A. Homer splice variants modulation within cortico-subcortical regions by dopamine D2 antagonists, a partial agonist, and an indirect agonist: Implication for glutamatergic postsynaptic density in antipsychotics action. Neuroscience 2007, 150, 144–158. [Google Scholar] [CrossRef] [PubMed]
- Schefzik, R.; Boland, L.; Hahn, B.; Kirschning, T.; Lindner, H.A.; Thiel, M.; Schneider-Lindner, V. Differential Network Testing Reveals Diverging Dynamics of Organ System Interactions for Survivors and Non-survivors in Intensive Care Medicine. Front. Physiol. 2022, 12, 801622. [Google Scholar] [CrossRef] [PubMed]
| Abbreviation | Brain Region |
|---|---|
| LSV | Lateral septal nucleus, ventral |
| Tu | Olfactory tubercle |
| LSD | Lateral septal nucleus, dorsal |
| LSI | Lateral septal nucleus, intermediate |
| Icj | Islands of Calleja |
| VP | Ventral pallidum |
| Shi | Septohippocampal nucleus |
| MS | Medial septum |
| VDB | Nucleus of the vertical limb of the diagonal band |
| IG | Indusium griseum |
| S1dz | Somatosensory 1, dysgranular zone |
| AIV | Agranular insular area, ventral |
| S1ULp | Upper lip of the primary somatosensory cortex |
| GI | Granular insular cortex |
| DI | Dysgranular insular cortex |
| AcSh | Accumbens nucleus, shell |
| Pir | Piriform cortex |
| AID | Agranular insular area, dorsal |
| LSS | Lateral stripe of striatum |
| Cg2 | Cingulate cortex, area 2 |
| CPDL | Dorsolateral caudate putamen |
| CPVL | Ventrolateral caudate putamen |
| CPVM | Ventromedial caudate putamen |
| Cg1 | Cingulate cortex, area 1 |
| M2 | Supplementary motor cortex |
| CPDM | Dorsomedial caudate putamen |
| Den | Dorsal endopiriform nucleus |
| M1 | Primary motor cortex |
| S1Fl | Somatosensory 1, forelimb region |
| AcCo | Accumbens nucleus, core |
| Cl | Claustrum |
| S1j | Somatosensory 1, jaw region |
| S1jO | Primary somatosensory cortex, jaw region, oral surface |
| ROIs | t | Degree of Freedom | p-Value | Confidence Interval | |
|---|---|---|---|---|---|
| Inferior | Superior | ||||
| Ig | 1.29 | 8 | 0.234 | −0.31 | 0.11 |
| Cg2 | −3.48 | 8 | 0.008 | −0.47 | −0.96 |
| Cg1 | −4.43 | 8 | 0.002 | −0.64 | −0.20 |
| M2 | −3.56 | 8 | 0.007 | −0.59 | −0.13 |
| M1 | −3.47 | 8 | 0.008 | −0.51 | −0.10 |
| S1FL | −3.22 | 8 | 0.012 | −0.44 | −0.73 |
| S1j | −2.59 | 8 | 0.032 | −0.38 | −0.02 |
| S1jO | 0.05 | 8 | 0.961 | −0.76 | 0.79 |
| S1DZ | −2.28 | 8 | 0.052 | −0.37 | 0.00 |
| S1ULp | −1.22 | 8 | 0.259 | −0.27 | 0.08 |
| GI | −1.03 | 8 | 0.335 | −0.35 | 0.14 |
| DI | −0.70 | 8 | 0.506 | −0.26 | 0.14 |
| AID | −0.45 | 8 | 0.662 | −0.28 | 0.19 |
| AIV | −0.16 | 8 | 0.880 | −0.15 | 0.13 |
| Cl | −0.97 | 8 | 0.363 | −0.30 | 0.12 |
| Pir | −2.07 | 8 | 0.073 | −0.44 | 0.02 |
| Den | −1.13 | 8 | 0.292 | −0.35 | 0.12 |
| LSS | −1.48 | 8 | 0.176 | −0.32 | 0.07 |
| CPDM | −3.14 | 8 | 0.014 | −0.53 | −0.08 |
| CPDL | −3.63 | 8 | 0.007 | −0.56 | −0.12 |
| CPVL | −2.93 | 8 | 0.019 | −0.53 | −0.64 |
| CPVM | −4.09 | 8 | 0.003 | −0.66 | −0.18 |
| AcCo | −2.74 | 8 | 0.026 | −0.48 | −0.04 |
| AcSh | −2.45 | 8 | 0.040 | −0.56 | −0.02 |
| LSD | −1.78 | 8 | 0.113 | −0.29 | 0.04 |
| LSI | −1.47 | 8 | 0.180 | −0.26 | 0.06 |
| LSV | −2.58 | 8 | 0.033 | −0.27 | −0.02 |
| Shi | −2.19 | 8 | 0.060 | −0.26 | 0.01 |
| MS | −2.14 | 8 | 0.065 | −0.24 | 0.01 |
| VDB | −1.311 | 8 | 0.226 | −0.29 | 0.08 |
| Icj | −3.55 | 8 | 0.007 | −0.24 | −0.05 |
| VP | −4.21 | 8 | 0.003 | −0.35 | −0.10 |
| Tu | −2.68 | 8 | 0.028 | −0.36 | −0.03 |
| ROIs | t | Degree of Freedom | p-Value | Confidence Interval | |
|---|---|---|---|---|---|
| Inferior | Superior | ||||
| Ig | 0.92 | 8 | 0.387 | −0.05 | 0.12 |
| Cg2 | −2.43 | 8 | 0.041 | −0.36 | −0.01 |
| Cg1 | −2.16 | 8 | 0.063 | −0.45 | 0.02 |
| M2 | −1.92 | 8 | 0.091 | −0.40 | 0.04 |
| M1 | −1.69 | 8 | 0.130 | −0.38 | 0.06 |
| S1FL | −1.53 | 8 | 0.164 | −0.32 | 0.07 |
| S1j | −1.28 | 8 | 0.235 | −0.30 | 0.09 |
| S1jO | 0.42 | 8 | 0.687 | −0.76 | 1.09 |
| S1DZ | −0.54 | 8 | 0.607 | −0.21 | 0.13 |
| S1ULp | −0.10 | 8 | 0.925 | −0.18 | 0.17 |
| GI | −0.43 | 8 | 0.677 | −0.31 | 0.21 |
| DI | −0.14 | 8 | 0.889 | −0.23 | 0.20 |
| AID | −0.42 | 8 | 0.686 | −0.26 | 0.18 |
| AIV | −0.88 | 8 | 0.403 | −0.20 | 0.09 |
| Cl | 1.69 | 8 | 0.129 | −0.06 | 0.39 |
| Pir | −1.77 | 8 | 0.114 | −0.38 | 0.05 |
| Den | 1.22 | 8 | 0.258 | −0.15 | 0.50 |
| LSS | 0.92 | 8 | 0.386 | −0.14 | 0.32 |
| CPDM | 1.22 | 8 | 0.256 | −0.12 | 0.40 |
| CPDL | 2.07 | 8 | 0.073 | −0.03 | 0.48 |
| CPVL | 1.81 | 8 | 0.108 | −0.06 | 0.51 |
| CPVM | 0.17 | 8 | 0.867 | −0.25 | 0.29 |
| AcCo | 0.13 | 8 | 0.901 | −0.24 | 0.27 |
| AcSh | −0.12 | 8 | 0.912 | −0.28 | 0.25 |
| LSD | −0.17 | 8 | 0.867 | −0.17 | 0.15 |
| LSI | −0.04 | 8 | 0.968 | −0.15 | 0.15 |
| LSV | −0.23 | 8 | 0.825 | −0.11 | 0.09 |
| Shi | 0.33 | 8 | 0.749 | −0.16 | 0.21 |
| MS | 0.46 | 8 | 0.657 | −0.16 | 0.24 |
| VDB | 0.75 | 8 | 0.474 | −0.12 | 0.24 |
| Icj | −0.44 | 8 | 0.669 | −0.17 | 0.12 |
| VP | −2.67 | 8 | 0.028 | −0.23 | −0.02 |
| Tu | −1.68 | 8 | 0.131 | −0.35 | 0.05 |
| ROIs | t | Degree of Freedom | p-Value | Confidence Interval | |
|---|---|---|---|---|---|
| Inferior | Superior | ||||
| Ig | −1.17 | 8 | 0.275 | −0.16 | 0.05 |
| Cg2 | −3.00 | 8 | 0.017 | −0.40 | −0.05 |
| Cg1 | −2.59 | 8 | 0.032 | −0.49 | −0.03 |
| M2 | −2.89 | 8 | 0.020 | −0.52 | −0.06 |
| M1 | −2.60 | 8 | 0.032 | −0.51 | −0.03 |
| S1FL | −3.19 | 8 | 0.013 | −0.55 | −0.09 |
| S1j | −2.32 | 8 | 0.049 | −0.53 | 0.00 |
| S1jO | −1.73 | 8 | 0.121 | −0.69 | 0.10 |
| S1DZ | −2.50 | 8 | 0.037 | −0.53 | −0.02 |
| S1ULp | −2.30 | 8 | 0.051 | −0.55 | 0.00 |
| GI | −2.52 | 8 | 0.036 | −0.61 | −0.03 |
| DI | −3.21 | 8 | 0.012 | −0.66 | −0.11 |
| AID | −3.42 | 8 | 0.009 | −0.63 | −0.12 |
| AIV | −2.83 | 8 | 0.022 | −0.54 | −0.05 |
| Cl | −2.94 | 8 | 0.019 | −0.55 | −0.07 |
| Pir | −2.66 | 8 | 0.029 | −0.50 | −0.03 |
| Den | −2.91 | 8 | 0.02 | −0.36 | −0.04 |
| LSS | −3.28 | 8 | 0.011 | −0.58 | −0.10 |
| CPDM | −5.41 | 8 | 0.001 | −0.52 | −0.21 |
| CPDL | −6.24 | 8 | <0.001 | −0.7 | −0.32 |
| CPVL | −6.93 | 8 | <0.001 | −0.84 | −0.42 |
| CPVM | −4.32 | 8 | 0.003 | −0.71 | −0.21 |
| AcCo | −3.36 | 8 | 0.01 | −0.58 | −0.11 |
| AcSh | −3.49 | 8 | 0.008 | −0.63 | −0.13 |
| LSD | −3.13 | 8 | 0.014 | −0.46 | −0.07 |
| LSI | −2.90 | 8 | 0.02 | −0.26 | −0.03 |
| LSV | −3.14 | 8 | 0.014 | −0.38 | −0.06 |
| Shi | −3.58 | 8 | 0.007 | −0.36 | −0.08 |
| MS | −4.84 | 8 | 0.001 | −0.29 | −0.1 |
| VDB | −2.61 | 8 | 0.06 | −0.37 | 0.01 |
| Icj | −2.71 | 8 | 0.027 | −0.47 | −0.04 |
| VP | −2.70 | 8 | 0.027 | −0.48 | −0.04 |
| Tu | −1.95 | 8 | 0.086 | −0.44 | 0.04 |
| Parameters | VEH/VEH | VEH/ASE | KET/VEH | KET/ASE |
|---|---|---|---|---|
| Number of nodes | 32 | 28 | 29 | 32 |
| Number of edges | 128 | 34 | 60 | 135 |
| Network density | 0.129 | 0.045 | 0.074 | 0.136 |
| Characteristic path length | 1.866 | 1.256 | 1.589 | 2.445 |
| Connected components | 1 | 4 | 2 | 1 |
| Clustering coefficient | 0.328 | 0.193 | 0.255 | 0.332 |
| Homer1a Gene Expression Comparison | |||
| Treatment Group Differences | ROIs | p-value | |
| KET/ASE > KET/VEH | CPDM | ≤0.001 | |
| KET/ASE > KET/VEH | CPDL | ≤0.001 | |
| KET/ASE > KET/VEH | CPVL | ≤0.001 | |
| KET/ASE > KET/VEH | MS | ≤0.001 | |
| Comparison of Correlation Matrices | |||
| Matrix pairs | ROIs | p-Value | |
| VEH/VEH vs. VEH/ASE | M1/GI | ≤0.05 | |
| S1j/AIV | ≤0.05 | ||
| S1FL/CPDL | ≤0.05 | ||
| S1j/CPDL | ≤0.05 | ||
| S1FL/CPVL | ≤0.05 | ||
| S1j/CPVL | ≤0.05 | ||
| S1FL/CI | ≤0.05 | ||
| S1j/CI | ≤0.05 | ||
| M2/Den | ≤0.05 | ||
| S1FL/Den | ≤0.05 | ||
| AIV/CPDL | ≤0.05 | ||
| AIV/CPVL | ≤0.05 | ||
| M1/CPVM | ≤0.05 | ||
| S1j/AcSh | ≤0.05 | ||
| S1FL/AIV | ≤0.05 | ||
| S1FL/LSS | ≤0.05 | ||
| M1/CPVL | ≤0.05 | ||
| GI/Icj | ≤0.05 | ||
| S1j/Den | ≤0.05 | ||
| Cg1/CPDL | ≤0.05 | ||
| M1/CPDL | ≤0.05 | ||
| Cg1/CPVL | ≤0.05 | ||
| M2/MS | ≤0.05 | ||
| VEH/VEH vs. KET/VEH | M1/AIV | ≤0.05 | |
| S1FL/AIV | ≤0.05 | ||
| AIV/CPDL | ≤0.05 | ||
| Ig/LSI | ≤0.05 | ||
| S1j/AIV | ≤0.05 | ||
| S1FL/Den | ≤0.05 | ||
| S1j/Den | ≤0.05 | ||
| AIV/CPVL | ≤0.05 | ||
| KET/VEH vs. KET/ASE | Ig/LSI * | ≤0.05 | |
| Ig/CPDL * | ≤0.05 | ||
| Ig/LSD * | ≤0.05 | ||
| Ig/LSV * | ≤0.05 | ||
| Ig/S1ULp * | ≤0.05 | ||
| Ig/CI * | ≤0.05 | ||
| Ig/CPVL * | ≤0.05 | ||
| Comparison of Network Measures | |||
| Matrix Pairs | Network Measures | p-Value | |
| VEH/ASE vs. VEH/VEH | Global Strength | ≤0.05 | |
| VEH/VEH vs. VEH/ASE | Node centrality | Cl | ≤0.05 |
| CPDL | ≤0.05 | ||
| CPDM | ≤0.05 | ||
| CPVL | ≤0.05 | ||
| S1FL | ≤0.05 | ||
| Den | ≤0.05 | ||
| Betweenness | Cg2 | ≤0.05 | |
| LSV | ≤0.05 | ||
| VEH/VEH vs. KET/VEH | Betweenness | M2 | ≤0.05 |
| MS | ≤0.05 | ||
| KET/VEH vs. KET/ASE | Betweenness | S1DZ | ≤0.01 |
| CPDM | ≤0.01 | ||
| LSV | ≤0.05 | ||
| Ig | ≤0.05 | ||
| Shi | ≤0.05 | ||
| Pir | ≤0.05 | ||
| Cg1 | ≤0.05 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barone, A.; De Simone, G.; Ciccarelli, M.; Buonaguro, E.F.; Tomasetti, C.; Eramo, A.; Vellucci, L.; de Bartolomeis, A. A Postsynaptic Density Immediate Early Gene-Based Connectome Analysis of Acute NMDAR Blockade and Reversal Effect of Antipsychotic Administration. Int. J. Mol. Sci. 2023, 24, 4372. https://doi.org/10.3390/ijms24054372
Barone A, De Simone G, Ciccarelli M, Buonaguro EF, Tomasetti C, Eramo A, Vellucci L, de Bartolomeis A. A Postsynaptic Density Immediate Early Gene-Based Connectome Analysis of Acute NMDAR Blockade and Reversal Effect of Antipsychotic Administration. International Journal of Molecular Sciences. 2023; 24(5):4372. https://doi.org/10.3390/ijms24054372
Chicago/Turabian StyleBarone, Annarita, Giuseppe De Simone, Mariateresa Ciccarelli, Elisabetta Filomena Buonaguro, Carmine Tomasetti, Anna Eramo, Licia Vellucci, and Andrea de Bartolomeis. 2023. "A Postsynaptic Density Immediate Early Gene-Based Connectome Analysis of Acute NMDAR Blockade and Reversal Effect of Antipsychotic Administration" International Journal of Molecular Sciences 24, no. 5: 4372. https://doi.org/10.3390/ijms24054372








