The Role of Toll-like Receptor 2 (TLR2) in the Development and Progression of Hashimoto’s Disease (HD): A Case Study on Female Patients in Poland
Abstract
:1. Introduction
2. Results
2.1. Analysis of Selected Parameters of Peripheral Blood Morphology and Thyroid Function in Patients with HD and Healthy Volunteers
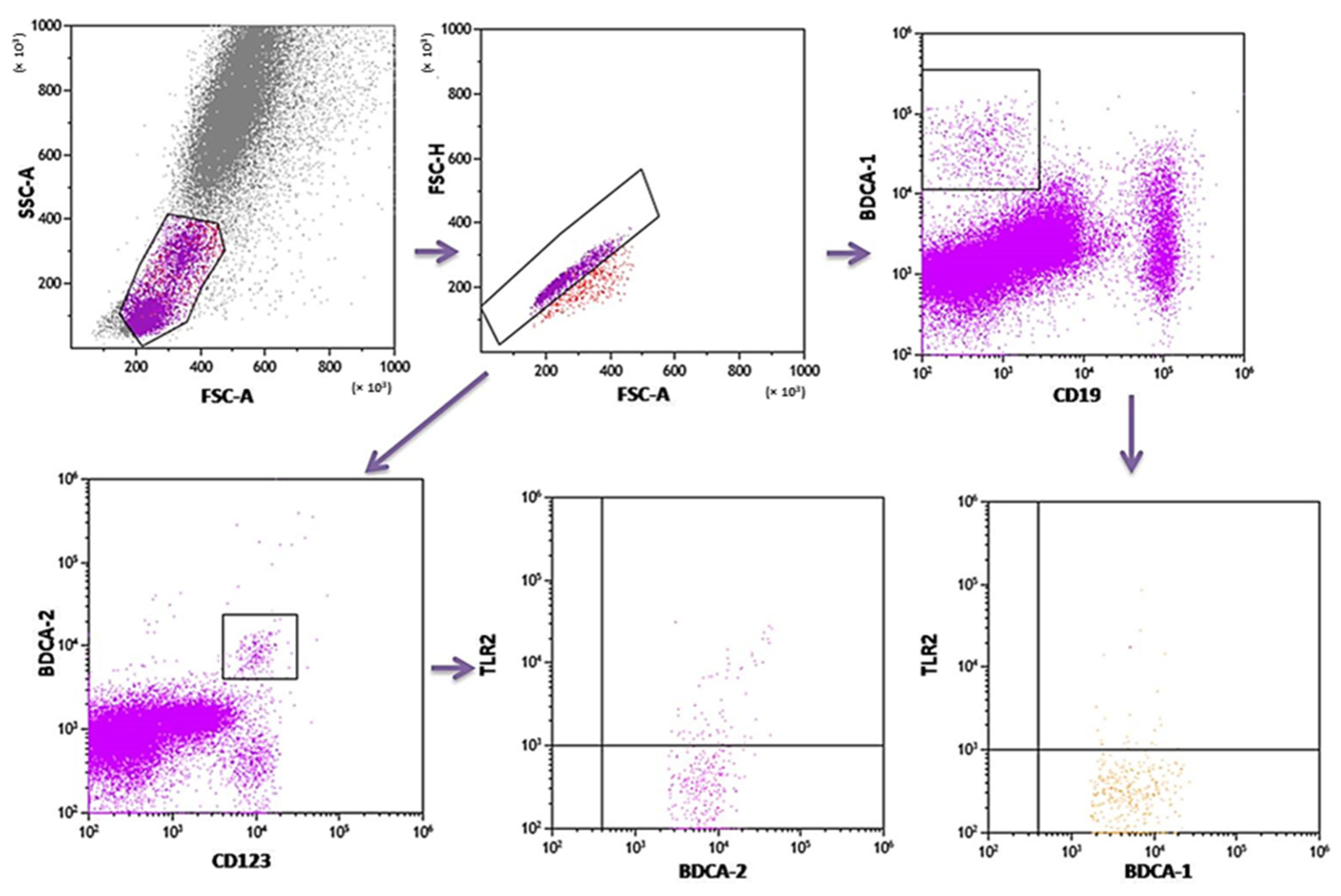
2.2. Characteristics of the Percentage of Selected Populations of Immune Cells Expressing TLR2 and Soluble Toll-like Receptor 2 (sTLR2) Concentration in the Plasma of Patients with HD in Relation to the Control Group
2.3. Correlation Analysis of the Percentage of Tested Immune System Cells Expressing TLR2 and sTLR2 Concentration in Relation to Selected Biochemical Indices Determining the Functioning of the Thyroid Gland
2.4. Analysis of the Potential of TLR2 in the Context of Diagnostic Accuracy in Differentiating Patients with HD
3. Discussion
4. Materials and Methods
4.1. Characteristics of Patients and Control Groups
4.2. The Assessment of the Population of DC and Monocytes in Peripheral Blood
4.3. Quantification of the sTLR in the Plasma of Patients by Enzyme-Linked Immunosorbent Assay (ELISA)
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ramos-Leví, A.M.; Marazuela, M. Pathogenesis of Thyroid Autoimmune Disease: The Role of Cellular Mechanisms. Endocrinol. Nutr. 2016, 63, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Villar, J.; Finol, H.J.; Torres, S.H.; Roschman-González, A. Myopathy in Patients with Hashimoto´s Disease. Investig. Clínica 2015, 56, 33–46. [Google Scholar]
- Hu, X.; Chen, Y.; Shen, Y.; Tian, R.; Sheng, Y.; Que, H. Global Prevalence and Epidemiological Trends of Hashimoto’s Thyroiditis in Adults: A Systematic Review and Meta-Analysis. Front. Public Health 2022, 10, 3858. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, F.; Fallahi, P.; Elia, G.; Gonnella, D.; Paparo, S.R.; Giusti, C.; Churilov, L.P.; Ferrari, S.M.; Antonelli, A. Hashimotos’ Thyroiditis: Epidemiology, Pathogenesis, Clinic and Therapy. Best Pract. Res. Clin. Endocrinol. Metab. 2019, 33, 101367. [Google Scholar] [CrossRef]
- Vanderpump, M.P.J. Epidemiology of Thyroid Disorders. In The Thyroid and Its Diseases: A Comprehensive Guide for the Clinician; Luster, M., Duntas, L.H., Wartofsky, L., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 75–85. ISBN 978-3-319-72102-6. [Google Scholar]
- Castell, E.C. Hashimoto’s Disease in a Cohort of 29 Children and Adolescents. Epidemiology, Clinical Course, and Comorbidities in the Short and Long Term. Arch Argent Pediatr. 2018, 116, 56–68. [Google Scholar] [CrossRef]
- Penta, L.; Cofini, M.; Lanciotti, L.; Leonardi, A.; Principi, N.; Esposito, S. Hashimoto’s Disease and Thyroid Cancer in Children: Are They Associated? Front. Endocrinol. 2018, 9, 565. [Google Scholar] [CrossRef] [Green Version]
- Tang, S.; Yang, M.; Zhang, D.; Tong, Y.; Xin, Y. Clinical Characteristics and Follow-Up of 19 Children With Hashimoto’s Thyroiditis Aged Below 3 Years: A Single-Center Retrospective Analysis. Front. Endocrinol. 2021, 12, 737527. [Google Scholar] [CrossRef]
- Vukovic, R.; Zeljkovic, A.; Bufan, B.; Spasojevic-Kalimanovska, V.; Milenkovic, T.; Vekic, J. Hashimoto Thyroiditis and Dyslipidemia in Childhood: A Review. Front. Endocrinol. 2019, 10, 868. [Google Scholar] [CrossRef] [Green Version]
- Dore, M.P.; Fanciulli, G.; Rouatbi, M.; Mereu, S.; Pes, G.M. Autoimmune Thyroid Disorders Are More Prevalent in Patients with Celiac Disease: A Retrospective Case-Control Study. J. Clin. Med. 2022, 11, 6027. [Google Scholar] [CrossRef]
- Sawicka-Gutaj, N.; Gruszczyński, D.; Zawalna, N.; Nijakowski, K.; Muller, I.; Karpiński, T.; Salvi, M.; Ruchała, M. Microbiota Alterations in Patients with Autoimmune Thyroid Diseases: A Systematic Review. Int. J. Mol. Sci. 2022, 23, 13450. [Google Scholar] [CrossRef]
- Paknys, G.; Kondrotas, A.J.; Kevelaitis, E. Risk factors and pathogenesis of Hashimoto’s thyroiditis. Medicina 2009, 45, 574–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hashimoto Thyroiditis: An Evidence-Based Guide to Etiology, Diagnosis and Treatment. Available online: https://www.mp.pl/paim/issue/article/16222 (accessed on 28 January 2023).
- Franco, J.-S.; Amaya-Amaya, J.; Anaya, J.-M. Thyroid Disease and Autoimmune Diseases; El Rosario University Press: Bogota, Colombia, 2013. [Google Scholar]
- Sawicka-Gutaj, N.; Wietrzyk, D.; Zawalna, N.; Erampamoorthy, A.; Ruchała, M. Autoimmune Disorders and Thyroid Function in Patients with Rheumatoid Arthritis after Biological Treatment. Rheumatol. Forum 2022, 8, 111–121. [Google Scholar] [CrossRef]
- Aversa, T.; Corica, D.; Zirilli, G.; Pajno, G.B.; Salzano, G.; De Luca, F.; Wasniewska, M. Phenotypic Expression of Autoimmunity in Children With Autoimmune Thyroid Disorders. Front. Endocrinol. 2019, 10, 476. [Google Scholar] [CrossRef] [Green Version]
- Štěpánek, L.; Horáková, D.; Štěpánek, L.; Janout, V.; Janoutová, J.; Bouchalová, K.; Martiník, K. Free Triiodothyronine/Free Thyroxine (FT3/FT4) Ratio Is Strongly Associated with Insulin Resistance in Euthyroid and Hypothyroid Adults: A Cross-Sectional Study. Endokrynol. Pol. 2021, 72, 8–13. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, W.; Liu, Y.; Wang, X.; Guan, Y.; Jia, L. Analysis of Basal Serum TSH, FT3, and FT4 Levels Based on Age, Sampling Time in Women with Infertility. BMC Womens Health 2021, 21, 317. [Google Scholar] [CrossRef] [PubMed]
- Lang, X.; Li, Y.; Zhang, D.; Zhang, Y.; Wu, N.; Zhang, Y. FT3/FT4 Ratio Is Correlated with All-Cause Mortality, Cardiovascular Mortality, and Cardiovascular Disease Risk: NHANES 2007–2012. Front. Endocrinol. 2022, 13, 964822. [Google Scholar] [CrossRef] [PubMed]
- Sahu, U.P.; Jha, S.; Mitra, O.; Pathak, A.; Prasad, K.N. To Study Thyroid Hormone Levels (FT3, FT4, and TSH Levels) in Critically Ill Children and Their Correlation with Disease Severity and Clinical Outcome in Rajendra Institute of Medical Sciences, Ranchi, Jharkhand. J. Fam. Med. Prim. Care 2022, 11, 6001. [Google Scholar] [CrossRef]
- Jara, E.L.; Muñoz-Durango, N.; Llanos, C.; Fardella, C.; González, P.A.; Bueno, S.M.; Kalergis, A.M.; Riedel, C.A. Modulating the Function of the Immune System by Thyroid Hormones and Thyrotropin. Immunol. Lett. 2017, 184, 76–83. [Google Scholar] [CrossRef]
- Montesinos, M.D.M.; Pellizas, C.G. Thyroid Hormone Action on Innate Immunity. Front. Endocrinol. 2019, 10, 350. [Google Scholar] [CrossRef] [Green Version]
- De Luca, R.; Davis, P.J.; Lin, H.-Y.; Gionfra, F.; Percario, Z.A.; Affabris, E.; Pedersen, J.Z.; Marchese, C.; Trivedi, P.; Anastasiadou, E.; et al. Thyroid Hormones Interaction With Immune Response, Inflammation and Non-Thyroidal Illness Syndrome. Front. Cell Dev. Biol. 2021, 8, 614030. [Google Scholar] [CrossRef]
- De Vito, P.; Incerpi, S.; Pedersen, J.Z.; Luly, P.; Davis, F.B.; Davis, P.J. Thyroid Hormones as Modulators of Immune Activities at the Cellular Level. Thyroid 2011, 21, 879–890. [Google Scholar] [CrossRef]
- Kawashima, A.; Yamazaki, K.; Hara, T.; Akama, T.; Yoshihara, A.; Sue, M.; Tanigawa, K.; Wu, H.; Ishido, Y.; Takeshita, F.; et al. Demonstration of Innate Immune Responses in the Thyroid Gland: Potential to Sense Danger and a Possible Trigger for Autoimmune Reactions. Thyroid 2013, 23, 477–487. [Google Scholar] [CrossRef] [Green Version]
- Rubingh, J.; van der Spek, A.; Fliers, E.; Boelen, A. The Role of Thyroid Hormone in the Innate and Adaptive Immune Response during Infection. Compr. Physiol. 2020, 10, 1277–1287. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, A.; Tanigawa, K.; Akama, T.; Yoshihara, A.; Ishii, N.; Suzuki, K. Innate Immune Activation and Thyroid Autoimmunity. J. Clin. Endocrinol. Metab. 2011, 96, 3661–3671. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazziotti, G.; Sorvillo, F.; Piscopo, M.; Morisco, F.; Cioffi, M.; Stornaiuolo, G.; Gaeta, G.B.; Molinari, A.M.; Lazarus, J.H.; Amato, G.; et al. Innate and Acquired Immune System in Patients Developing Interferon-α-Related Autoimmune Thyroiditis: A Prospective Study. J. Clin. Endocrinol. Metab. 2005, 90, 4138–4144. [Google Scholar] [CrossRef] [Green Version]
- Van der Spek, A.H.; Fliers, E.; Boelen, A. Thyroid Hormone and Deiodination in Innate Immune Cells. Endocrinology 2021, 162, bqaa200. [Google Scholar] [CrossRef]
- Gong, T.; Liu, L.; Jiang, W.; Zhou, R. DAMP-Sensing Receptors in Sterile Inflammation and Inflammatory Diseases. Nat. Rev. Immunol. 2020, 20, 95–112. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Kang, R.; Coyne, C.B.; Zeh, H.J.; Lotze, M.T. PAMPs and DAMPs: Signal 0s That Spur Autophagy and Immunity. Immunol. Rev. 2012, 249, 158–175. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, I.; Giroux, N.; Olson, L.; Morrison, S.A.; Llanga, T.; Akinade, T.O.; Zhu, Y.; Zhong, Y.; Bose, S.; Arvai, S.; et al. DAMPs/PAMPs Induce Monocytic TLR Activation and Tolerance in COVID-19 Patients; Nucleic Acid Binding Scavengers Can Counteract Such TLR Agonists. Biomaterials 2022, 283, 121393. [Google Scholar] [CrossRef]
- Kumar, H.; Kawai, T.; Akira, S. Toll-like Receptors and Innate Immunity. Biochem. Biophys. Res. Commun. 2009, 388, 621–625. [Google Scholar] [CrossRef]
- Kawasaki, T.; Kawai, T. Toll-Like Receptor Signaling Pathways. Front. Immunol. 2014, 5, 461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, W.; Xu, Q.; Zhang, Y.; Gao, W.; Zhang, M.; Zhai, W.; Rajkumar, R.S.; Liu, Z. Toll-like Receptor-Mediated Innate Immunity against Herpesviridae Infection: A Current Perspective on Viral Infection Signaling Pathways. Virol. J. 2020, 17, 192. [Google Scholar] [CrossRef] [PubMed]
- De Oliviera Nascimento, L.; Massari, P.; Wetzler, L. The Role of TLR2 in Infection and Immunity. Front. Immunol. 2012, 3, 79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flo, T.H.; Halaas, O.; Torp, S.; Ryan, L.; Lien, E.; Dybdahl, B.; Sundan, A.; Espevik, T. Differential Expression of Toll-like Receptor 2 in Human Cells. J. Leukoc. Biol. 2001, 69, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Karmakar, S.; Babu, S.P.S. TLR2 and TLR4 Mediated Host Immune Responses in Major Infectious Diseases: A Review. Braz. J. Infect. Dis. 2016, 20, 193–204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brandt, K.J.; Fickentscher, C.; Kruithof, E.K.O.; De Moerloose, P. TLR2 Ligands Induce NF-ΚB Activation from Endosomal Compartments of Human Monocytes. PLoS ONE 2013, 8, e80743. [Google Scholar] [CrossRef] [Green Version]
- Chistiakov, D.A.; Sobenin, I.A.; Orekhov, A.N.; Bobryshev, Y.V. Myeloid Dendritic Cells: Development, Functions, and Role in Atherosclerotic Inflammation. Immunobiology 2015, 220, 833–844. [Google Scholar] [CrossRef]
- Murray, S.M.; Zhang, Y.; Douek, D.C.; Sekaly, R.P. Myeloid Cells Enriched for a Dendritic Cell Population From People Living With HIV Have Altered Gene Expression Not Restored by Antiretroviral Therapy. Front. Immunol. 2020, 11, 261. [Google Scholar] [CrossRef] [Green Version]
- Ye, Y.; Gaugler, B.; Mohty, M.; Malard, F. Plasmacytoid Dendritic Cell Biology and Its Role in Immune-mediated Diseases. Clin. Transl. Immunol. 2020, 9, e1139. [Google Scholar] [CrossRef]
- Musumeci, A.; Lutz, K.; Winheim, E.; Krug, A.B. What Makes a PDC: Recent Advances in Understanding Plasmacytoid DC Development and Heterogeneity. Front. Immunol. 2019, 10, 1222. [Google Scholar] [CrossRef] [Green Version]
- Kapellos, T.S.; Bonaguro, L.; Gemünd, I.; Reusch, N.; Saglam, A.; Hinkley, E.R.; Schultze, J.L. Human Monocyte Subsets and Phenotypes in Major Chronic Inflammatory Diseases. Front. Immunol. 2019, 10, 2035. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cooper, G.S.; Stroehla, B.C. The Epidemiology of Autoimmune Diseases. Autoimmun. Rev. 2003, 2, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, F.-S.; Gershwin, M.E. Human Autoimmune Diseases: A Comprehensive Update. J. Intern. Med. 2015, 278, 369–395. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Parada, L. Research Round-up: Autoimmune Disease. Nature 2021, 595, S46–S47. [Google Scholar] [CrossRef]
- Chiovato, L.; Magri, F.; Carlé, A. Hypothyroidism in Context: Where We’ve Been and Where We’re Going. Adv. Ther. 2019, 36, 47–58. [Google Scholar] [CrossRef] [Green Version]
- Strikić Đula, I.; Pleić, N.; Babić Leko, M.; Gunjača, I.; Torlak, V.; Brdar, D.; Punda, A.; Polašek, O.; Hayward, C.; Zemunik, T. Epidemiology of Hypothyroidism, Hyperthyroidism and Positive Thyroid Antibodies in the Croatian Population. Biology 2022, 11, 394. [Google Scholar] [CrossRef]
- Taylor, P.N.; Albrecht, D.; Scholz, A.; Gutierrez-Buey, G.; Lazarus, J.H.; Dayan, C.M.; Okosieme, O.E. Global Epidemiology of Hyperthyroidism and Hypothyroidism. Nat. Rev. Endocrinol. 2018, 14, 301–316. [Google Scholar] [CrossRef]
- Vanderpump, M.P.J. The Epidemiology of Thyroid Disease. British Medical. Bulletin. 2011, 99, 39–51. [Google Scholar] [CrossRef] [Green Version]
- Hashimoto Thyroiditis: Practice Essentials, Background, Etiology. Available online: https://emedicine.medscape.com/article/120937-overview (accessed on 27 February 2023).
- Mincer, D.L.; Jialal, I. Hashimoto Thyroiditis; StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Mikulska, A.A.; Karaźniewicz-Łada, M.; Filipowicz, D.; Ruchała, M.; Główka, F.K. Metabolic Characteristics of Hashimoto’s Thyroiditis Patients and the Role of Microelements and Diet in the Disease Management—An Overview. Int. J. Mol. Sci. 2022, 23, 6580. [Google Scholar] [CrossRef]
- Bogusławska, J.; Godlewska, M.; Gajda, E.; Piekiełko-Witkowska, A. Cellular and Molecular Basis of Thyroid Autoimmunity. Eur. Thyroid. J. 2022, 11, e210024. [Google Scholar] [CrossRef]
- Swain, M.; Swain, T.; Mohanty, B.K. Autoimmune Thyroid Disorders-An Update. Indian J. Clin. Biochem. 2005, 20, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.H.; Fu, D.G. Autoimmune Thyroid Disease: Mechanism, Genetics and Current Knowledge. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3611–3618. [Google Scholar] [PubMed]
- Wang, B.; Shao, X.; Song, R.; Xu, D.; Zhang, J. The Emerging Role of Epigenetics in Autoimmune Thyroid Diseases. Front. Immunol. 2017, 8, 396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrari, S.M.; Ragusa, F.; Elia, G.; Paparo, S.R.; Mazzi, V.; Baldini, E.; Benvenga, S.; Antonelli, A.; Fallahi, P. Precision Medicine in Autoimmune Thyroiditis and Hypothyroidism. Front. Pharmacol. 2021, 12, 3123. [Google Scholar] [CrossRef]
- Tomer, Y.; Huber, A. The etiology of autoimmune thyroid disease: A story of genes and environment. J. Autoimmun. 2009, 32, 231–239. [Google Scholar] [CrossRef] [Green Version]
- Mikoś, H.; Mikoś, M.; Obara-Moszyńska, M.; Niedziela, M. The Role of the Immune System and Cytokines Involved in the Pathogenesis of Autoimmune Thyroid Disease (AITD). Endokrynol. Pol. 2014, 65, 150–155. [Google Scholar] [CrossRef] [Green Version]
- Ganesh, B.B.; Bhattacharya, P.; Gopisetty, A.; Prabhakar, B.S. Role of Cytokines in the Pathogenesis and Suppression of Thyroid Autoimmunity. J. Interferon Cytokine Res. 2011, 31, 721–731. [Google Scholar] [CrossRef]
- Li, Q.; Wang, B.; Mu, K.; Zhang, J.-A. The Pathogenesis of Thyroid Autoimmune Diseases: New T Lymphocytes–Cytokines Circuits beyond the Th1−Th2 Paradigm. J. Cell. Physiol. 2019, 234, 2204–2216. [Google Scholar] [CrossRef]
- Klecha, A.J.; Barreiro Arcos, M.L.; Frick, L.; Genaro, A.M.; Cremaschi, G. Immune-Endocrine Interactions in Autoimmune Thyroid Diseases. Neuroimmunomodulation 2008, 15, 68–75. [Google Scholar] [CrossRef]
- Stensland, Z.C.; Coleman, B.M.; Rihanek, M.; Baxter, R.M.; Gottlieb, P.A.; Hsieh, E.W.Y.; Sarapura, V.D.; Simmons, K.M.; Cambier, J.C.; Smith, M.J. Peripheral Immunophenotyping of AITD Subjects Reveals Alterations in Immune Cells in Pediatric vs Adult-Onset AITD. iScience 2022, 25, 103626. [Google Scholar] [CrossRef]
- Harii, N.; Lewis, C.J.; Vasko, V.; McCall, K.; Benavides-Peralta, U.; Sun, X.; Ringel, M.D.; Saji, M.; Giuliani, C.; Napolitano, G.; et al. Thyrocytes Express a Functional Toll-like Receptor 3: Overexpression Can Be Induced by Viral Infection and Reversed by Phenylmethimazole and Is Associated with Hashimoto’s Autoimmune Thyroiditis. Mol. Endocrinol. 2005, 19, 1231–1250. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Li, C.; Wang, X.; Liu, X.; Han, C.; Jin, T.; Liu, S.; Zhang, X.; Zhang, H.; He, X.; et al. Increased Toll-Like Receptors Activity and TLR Ligands in Patients with Autoimmune Thyroid Diseases. Front. Immunol. 2016, 7, 578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aktaş, T.; Celik, S.K.; Genc, G.C.; Arpaci, D.; Can, M.; Dursun, A. Higher Levels of Serum TLR2 and TLR4 in Patients with Hashimoto’s Thyroiditis. Endocr. Metab. Immune Disord. Drug Targets 2020, 20, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Dvornikova, K.A.; Bystrova, E.Y.; Platonova, O.N.; Churilov, L.P. Polymorphism of Toll-like Receptor Genes and Autoimmune Endocrine Diseases. Autoimmun. Rev. 2020, 19, 102496. [Google Scholar] [CrossRef]
- Cho, W.K.; Jang, J.-P.; Choi, E.-J.; Ahn, M.; Kim, S.H.; Cho, K.S.; Park, S.H.; Baek, I.C.; Jung, M.H.; Kim, T.-G.; et al. Association of Polymorphisms in Toll-Like Receptors 4 and 9 with Autoimmune Thyroid Disease in Korean Pediatric Patients. Int. J. Endocrinol. 2017, 2017, e2304218. [Google Scholar] [CrossRef] [Green Version]



| Parametrs | Patients with HD (n = 35) | Healthy Volunteers (n = 20) | p-Value | ||
|---|---|---|---|---|---|
| Mean ± SD | Median (Range) | Mean ± SD | Median (Range) | ||
| WBC [103/mm3] (Normal Range: 4.37–10.00) | 6.15 ± 2.05 | 6.38 (2.53–10.62) | 6.49 ± 2.01 | 6.11 (4.19–10.62) | 0.815 |
| LYM [103/mm3] (Normal Range: 1.18–3.74) | 1.68 ± 0.60 | 1.63 (0.76–3.27) | 2.42 ± 0.48 | 2.29 (1.29–3.42) | 0.000 * |
| MON [103/mm3] (Normal Range: 0.24–0.63) | 0.52 ± 0.19 | 0.49 (0.20–0.82) | 0.50 ± 0.15 | 0.44 (0.25–0.82) | 0.633 |
| NEU [103/mm3] (Normal Range: 1.56–6.13) | 3.49 ± 1.38 | 3.59 (0.91–5.47) | 3.45 ± 1.18 | 3.22 (1.59–5.47) | 0.842 |
| EOS [103/mm3] (Normal Range: 0.04–0.36) | 0.13 ± 0.07 | 0.12 (0.05–0.43) | 0.12 ± 0.09 | 0.10 (0.04–0.43) | 0.450 |
| BAS [103/mm3] (Normal Range: 0.01–0.08) | 0.04 ± 0.02 | 0.03 (0.00–0.06) | 0.04 ± 0.02 | 0.03 (0.01–0.06) | 0.951 |
| RBC [106/mm3] (Normal Range: 3.93–5.22) | 4.55 ± 0.31 | 4.58 (3.91–5.66) | 4.45 ± 0.49 | 4.47 (3.81–5.66) | 0.095 |
| HGB [g/dL] (Normal Range: 11.20–15.70) | 13.34 ± 0.91 | 13.30 (11.10–14.70) | 13.08 ± 0.96 | 13.20 (11.60–14.70) | 0.609 |
| PLT [103/mm3] (Normal Range: 150.00–400.00) | 291.86 ± 73.27 | 299.00 (128.00–387.00) | 261.55 ± 52.03 | 246.00 (182.00–387.00) | 0.126 |
| TSH [µL/mL] (Normal Range: 0.30–3.00) | 0.01 ± 0.01 | 0.01 (0.00–0.09) | 2.67 ± 0.80 | 2.71 (0.92–3.98) | 0.000 * |
| fT3 [pg/mL] (Normal Range: 2.00–4.40) | 11.75 ± 5.14 | 11.14 (6.90–31.79) | 4.98 ± 0.87 | 4.97 (3.60–6.40) | 0.000 * |
| fT4 [ng/dL] (Normal Range: 0.93–1.70) | 35.49 ± 16.02 | 30.95 (19.05–82.22) | 17.09 ± 2.39 | 17.04 (11.47–21.47) | 0.000 * |
| Parametrs | Patients with HD (n = 35) | Healthy Volunteers (n = 20) | p-Value | ||
|---|---|---|---|---|---|
| Mean ± SD | Median (Range) | Mean ± SD | Median (Range) | ||
| mDC BDCA-1+CD19− in PB [%] | 1.02 ± 0.36 | 1.02 (0.44–1.82) | 0.54 ± 0.29 | 0.53 (0.13–1.10) | 0.0000 * |
| pDC BDCA-2+CD123+ in PB [%] | 0.49 ± 0.36 | 0.40 (0.02–1.54) | 0.23 ± 0.24 | 0.15 (0.01–0.97) | 0.0000 * |
| mDC BDCA-1+CD19−TLR2+ in PB [%] | 20.12 ± 7.60 | 20.20 (7.50–35.40) | 5.04 ± 3.49 | 4.30 (1.30–13.70) | 0.0000 * |
| pDC BDCA-2+CD123+TLR2+ in PB [%] | 13.33 ± 6.12 | 12.60 (5.90–27.80) | 4.24 ± 2.93 | 3.60 (1.40–11.50) | 0.0004 * |
| Classical MONs CD14+CD16− in PB [%] | 85.80 ± 4.70 | 87.10 (75.50–94.50) | 93.10 ± 2.40 | 95.5 (88.30–95.40) | 0.028 * |
| Non-classical MONs CD14+CD16+ in PB [%] | 9.9 ± 4.9 | 8.5 (3.1–20.5) | 4.4 ± 2.2 | 3.6 (2.2–10.3) | 0.2200 |
| Classical MONs CD14+CD16−TLR2+ in PB [%] | 15.9 ± 12.9 | 11.9 (2.5–43.4) | 4.33 ± 1.96 | 4.00 (2.00–9.10) | 0.0003 * |
| Non-classical MONs CD14+CD16+TLR2+ in PB [%] | 18.42 ± 14.85 | 15.80 (1.10–50.80) | 5.34 ± 2.73 | 4.80 (1.80–9.90) | 0.0007 * |
| TLR2 plasma concentration [ng/mL] | 32.19 ± 13.34 | 30.4 (14.40–56.90) | 4.98 ± 3.87 | 4.6 (0.7–13.9) | 0.000 * |
| Parameter | Prognostic Value | Area under the ROC Curve (AUC) | 95% Confidence Interval (95%CI) |
|---|---|---|---|
| TSH serum concentration [µL/mL] | 0.02 | 0.81 | 0.66–0.96 |
| Frequencies of classical MONs CD14+CD16− in the PB [%] | 86.32 | 0.92 | 0.81–1.0 |
| Frequencies of classical MONs CD14+CD16−TLR2+ in the PB [%] | 20.02 | 1.0 | 1.0 |
| Frequencies of non-classical MONs CD14+CD16+ in the PB [%] | 8.3 | 0.89 | 0.79–0.998 |
| Frequencies of non-classical MONs CD14+CD16+TLR2+ in the PB [%] | 20.09 | 0.96 | 0.88–1.0 |
| Parametrs | Patients with HD (n = 35) | Healthy Volunteers (n = 20) | ||
|---|---|---|---|---|
| Mean ± SD | Median (Range) | Mean ± SD | Median (Range) | |
| Age (years) | 41.00 ± 16.21 | 36.00 (22.00–95.00) | 39.30 ± 11.30 | 36.00 (23.00–65.00) |
| TSH Receptor Antibodies (TRAb) [U/L] | 12.17 ± 9.69 | 9.75 (1.70–39.40) | 1.65 ± 0.69 | 1.45 (0.59–1.86) |
| anti-TPO antibodies [U/mL] | 1272.36 ± 551.83 | 1322.70 (13.70–1873.70) | 1.69 ± 0.31 | 1.54 (0.74–4.51) |
| anti-TG antibodies [U/mL] | 255.81 ± 292.77 | 160.77 (10.00–1360.00) | 1.31 ± 0.27 | 1.58 (0.89–3.11) |
| Thyroid gland volume [mL] | 20.47 ± 8.32 | 20.35 (13.30–47.60) | not applicable | |
| fT3 [pg/mL] | 11.75 ± 5.14 | 11.14 (6.90–31.79) | 4.98 ± 0.87 | 4.97 (3.60–6.40) |
| fT4 [ng/dL] | 35.49 ± 16.02 | 30.95 (19.05–82.22) | 17.09 ± 2.39 | 17.04 (11.47–21.47) |
| TSH [µL/mL] | 0.01 ± 0.01 | 0.01 (0.00–0.09) | 2.67 ± 0.80 | 2.71 (0.92–3.98) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klatka, M.; Polak, A.; Mertowska, P.; Mertowski, S.; Łyczba, J.; Hymos, A.; Korona-Głowniak, I.; Grywalska, E. The Role of Toll-like Receptor 2 (TLR2) in the Development and Progression of Hashimoto’s Disease (HD): A Case Study on Female Patients in Poland. Int. J. Mol. Sci. 2023, 24, 5344. https://doi.org/10.3390/ijms24065344
Klatka M, Polak A, Mertowska P, Mertowski S, Łyczba J, Hymos A, Korona-Głowniak I, Grywalska E. The Role of Toll-like Receptor 2 (TLR2) in the Development and Progression of Hashimoto’s Disease (HD): A Case Study on Female Patients in Poland. International Journal of Molecular Sciences. 2023; 24(6):5344. https://doi.org/10.3390/ijms24065344
Chicago/Turabian StyleKlatka, Maria, Agnieszka Polak, Paulina Mertowska, Sebastian Mertowski, Jakub Łyczba, Anna Hymos, Izabela Korona-Głowniak, and Ewelina Grywalska. 2023. "The Role of Toll-like Receptor 2 (TLR2) in the Development and Progression of Hashimoto’s Disease (HD): A Case Study on Female Patients in Poland" International Journal of Molecular Sciences 24, no. 6: 5344. https://doi.org/10.3390/ijms24065344





