Transcriptome Dynamics Underlying Planticine®-Induced Defense Responses of Tomato (Solanum lycopersicum L.) to Biotic Stresses
Abstract
:1. Introduction
2. Results
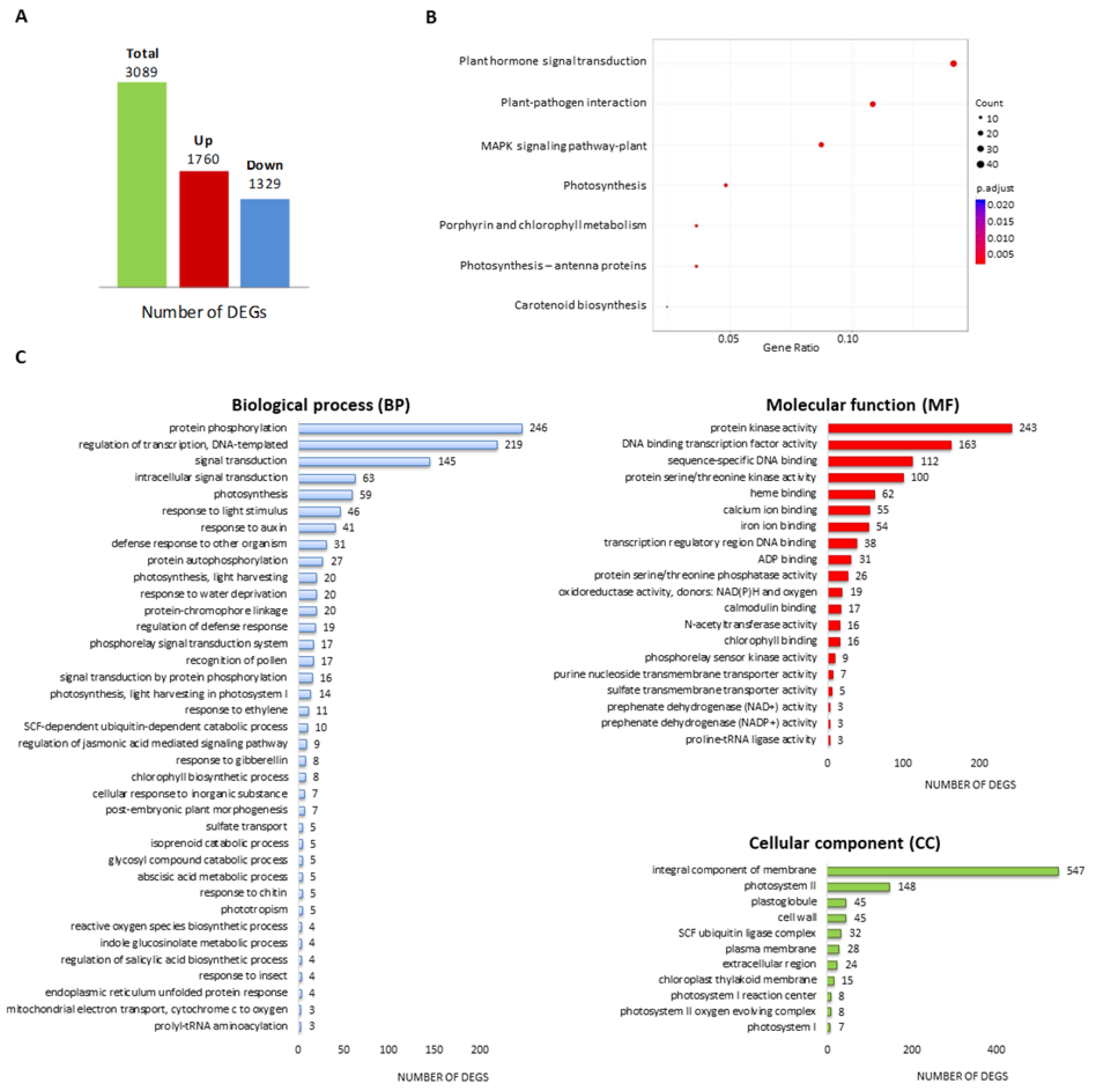
2.1. Genome-Wide Identification of Expressed Genes in Tomatoes Exposed to Planticine®
2.1.1. Genes Related to Plant-Pathogen Interaction
2.1.2. Genes Related to Plant Hormone Signal Transduction
2.2. The Content of Phytohormones
2.3. Powdery Mildew on Tomato Leaves
2.4. Thrips on Tomato Leaves
3. Discussion
4. Materials and Methods
4.1. Tomato Trail in the Climatic Chamber
4.1.1. RNA Extraction and RNA-Seq Analysis
4.1.2. SA, JA, and ABA Assays
4.2. Greenhouse Trials with Powdery Mildew and Thrips on Tomatoes
4.2.1. Trial with Powdery Mildew on Tomatoes
4.2.2. Trial with Thrips on Tomato
4.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Van Aubel, G.; Buonatesta, R.; Van Cutsem, P. COS-OGA: A novel oligosaccharidic elicitor that protects grapes and cucumbers against powdery mildew. Crop Prot. 2014, 65, 129–137. [Google Scholar] [CrossRef]
- Younus Wani, M.; Mehraj, S.; Rather, R.A.; Rani, S.; Hajam, O.A.; Ganie, N.A.; Mir, M.R.; Baqual, M.F.; Kamili, A.S. Systemic acquired resistance (SAR): A novel strategy for plant protection with reference to mulberry. Int. J. Chem. Stud. 2018, 6, 1184–1192. [Google Scholar]
- Kunkel, B.N.; Brooks, D.M. Cross talk between signaling pathways in pathogen defense. Curr. Opin. Plant Biol. 2002, 5, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef] [Green Version]
- Choi, H.W.; Klessig, D.F. DAMPs, MAMPs, and NAMPs in plant innate immunity. BMC Plant Biol. 2016, 16, 232. [Google Scholar] [CrossRef] [Green Version]
- Malik, N.A.A.; Kumar, I.S.; Nadarajah, K. Elicitor and receptor molecules: Orchestrators of plant defense and immunity. Int. J. Mol. Sci. 2020, 21, 963. [Google Scholar] [CrossRef] [Green Version]
- Vallad, G.E.; Goodman, R.M. Systemic acquired resistance and induced systemic resistance in conventional agricultur. Crop Sci. 2004, 44, 1920–1934. [Google Scholar] [CrossRef] [Green Version]
- Van Wees, S.C.M.; Van der Ent, S.; Pieterse, C.M.J. Plant immune responses triggered by beneficial microbes. Curr. Opin. Plant Biol. 2008, 11, 443–448. [Google Scholar] [CrossRef] [Green Version]
- Fu, Z.Q.; Dong, X. Systemic acquired resistance: Turning local infection into global defense. Annu. Rev. Plant Biol. 2013, 64, 839–863. [Google Scholar] [CrossRef] [Green Version]
- Muthamilarasan, M.; Prasad, M. Plant innate immunity: An updated insight into defense mechanism. J. Biosci. 2013, 38, 433–449. [Google Scholar] [CrossRef]
- Vorwerk, S.; Somerville, S.; Somerville, C. The role of plant cell wall polysaccharide composition in disease resistance. Trends Plant Sci. 2004, 9, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Tedman-Jones, J.D.; Lei, R.; Jay, F.; Fabro, G.; Li, X.; Reiter, W.-D.; Brearley, C.; Jones, J.D.G. Characterization of Arabidopsis mur3 mutations that result in constitutive activation of defence in petioles, but not leaves. Plant J. 2008, 56, 691–703. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, S.; Savatin, D.V.; Sicilia, F.; Gramegna, G.; Cervone, F.; De Lorenzo, G. Oligogalacturonides: Plant damage-associated molecular patterns and regulators of growth and development. Front. Plant Sci. 2013, 4, 49. [Google Scholar] [CrossRef] [Green Version]
- Boller, T.; Felix, G. A renaissance of elicitors: Perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annu. Rev. Plant Biol. 2009, 60, 379–407. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Van Der Does, D.; Zamioudis, C.; Leon-Reyes, A.; Van Wees, S.C. Hormonal modulation of plant immunity. Annu. Rev. Cell Dev. Biol. 2012, 28, 489–521. [Google Scholar] [CrossRef] [Green Version]
- Weinberger, F.; Friedlander, M. Endogenous and exogenous elicitors of a hypersensitive response in Gracilaria conferta (Rhodophyta). J. Appl. Phycol. 2000, 12, 139–145. [Google Scholar] [CrossRef]
- Howe, G.A.; Jander, G. Plant immunity to insect herbivores. Annu. Rev. Plant Biol. 2008, 59, 41–66. [Google Scholar] [CrossRef] [Green Version]
- Peng, Y.; Van Wersch, R.; Zhang, Y. Convergent and divergent signaling in PAMP-triggered immunity and effector-triggered immunity. Mol. Plant Microbe Interact. 2018, 31, 403–409. [Google Scholar] [CrossRef] [Green Version]
- Bigeard, J.; Colcombet, J.; Hirt, H. Signaling mechanisms in pattern-triggered immunity (PTI). Mol. Plant 2015, 8, 521–539. [Google Scholar] [CrossRef] [Green Version]
- Nadarajah, K.; Kassim, N.M. Effect of signal molecules and hormones on the expression of protein kinase gene OrMKK1 in rice. J. Plant Sci. 2009, 4, 32–42. [Google Scholar] [CrossRef] [Green Version]
- Chandra, S.; Chakraborty, N.; Dasgupta, A.; Sarkar, J.; Panda, K.; Acharya, K. Chitosan nanoparticles: A positive modulator of innate immune responses in plants. Sci. Rep. 2015, 5, 15195. [Google Scholar] [CrossRef] [Green Version]
- Iriti, M.; Varoni, E.M. Chitosan-induced antiviral activity and innate immunity in plants. Environ. Sci. Pollut. Res. 2015, 22, 2935–2944. [Google Scholar] [CrossRef] [PubMed]
- Sharif, R.; Mujtaba, M.; Ur Rahman, M.; Shalmani, A.; Ahmad, H.; Anwar, T.; Tianchan, D.; Wang, X. The multifunctional role of chitosan in horticultural crops; a review. Molecules 2018, 23, 872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fesel, P.H.; Zuccaro, A. Crucial component of the fungal cell wall and elusive MAMP in plants. Fungal Genet. Biol. 2016, 90, 53–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marfà, V.; David, J.; Gollin, D.J.; Eberhard, S.; Mohnen, D.; Dan/ill, A.; Albersheim, P. Oligogalacturonides are able to induce flowers to form on tobacco explants. Plant J. 1991, 1, 217–225. [Google Scholar] [CrossRef]
- Moscatiello, R.; Baldan, B.; Squartini, A.; Mariani, P.; Navazio, L. Oligogalacturonides: Novel signaling molecules in rhizobium-legume communications. Mol. Plant-Microbe Interact. 2012, 25, 1387–1395. [Google Scholar] [CrossRef] [Green Version]
- Camejo, D.; Martí, M.C.; Olmos, E.; Torres, W.; Sevilla, F.; Jiménez, A. Oligogalacturonides stimulate antioxidant system in alfalfa roots. Biol. Plant. 2012, 56, 537–544. [Google Scholar] [CrossRef]
- González-Pérez, L.; Vázquez-Glaría, A.; Perrotta, L.; Acosta, A.; Scriven, S.A.; Herbert, R.; Cabrera, J.C.; Francis, D.; Rogers, H.J. Oligosaccharins and Pectimorf® stimulate root elongation and shorten the cell cycle in higher plants. Plant Growth Regul. 2012, 68, 211–221. [Google Scholar] [CrossRef]
- Denoux, C.; Galletti, R.; Mammarella, N.; Gopalan, S.; Werck, D.; De Lorenzo, G.; Ferrari, S.; Ausubel, F.M.; Dewdney, J. Activation of defense response pathways by OGs and Flg22 elicitors in Arabidopsis seedlings. Mol. Plant 2008, 1, 423–445. [Google Scholar] [CrossRef] [Green Version]
- Davis, K.R.; Darvill, A.G.; Albersheim, P.; Dell, A. Host-Pathogen Interactions: XXIX. Oligogalacturonides released from sodium polypectate by endopolygalacturonic acid lyase are elicitors of phytoalexins in soybean. Plant Physiol. 1986, 80, 568–577. [Google Scholar] [CrossRef] [Green Version]
- Aziz, A.; Heyraud, A.; Lambert, B. Oligogalacturonide signal transduction, induction of defense-related responses and protection of grapevine against Botrytis cinerea. Planta 2004, 218, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Forrest, R.S.; Lyon, G.D. Substrate degradation patterns of polygalacturonic acid lyase from Erwinia carotovora and Bacillus polymyxa and release of phytoalexin-eliciting oligosaccharides from potato cell walls. J. Exp. Bot. 1990, 41, 481–488. [Google Scholar] [CrossRef]
- Selim, S.; Sanssené, J.; Rossard, S.; Courtois, J. Systemic induction of the defensin and phytoalexin pisatin pathways in pea (Pisum sativum) against aphanomyces euteiches by acetylated and nonacetylated oligogalacturonides. Molecules 2017, 22, 1017. [Google Scholar] [CrossRef] [Green Version]
- Moloshok, T.; Pearce, G.; Ryan, C.A. Oligouronide signaling of proteinase inhibitor genes in plants: Structure-activity relationships of di- and trigalacturonic acids and their derivatives. Arch. Biochem. Biophys. 1992, 294, 731–734. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.; Olsen, O.; Wegener, C.; Von Wettstein, D. Digalacturonates from pectin degradation induce tissue responses against potato soft rot. Physiol. Mol. Plant Pathol. 1996, 48, 389–401. [Google Scholar] [CrossRef]
- Montesano, M.; Kõiv, V.; Mäe, A.; Palva, E.T. Novel receptor-like protein kinases induced by Erwinia carotovora and short oligogalacturonides in potato. Mol. Plant Pathol. 2001, 2, 339–346. [Google Scholar] [CrossRef]
- Ramírez-Zavaleta, C.Y.; García-Barrera, L.J.; Rodríguez-Verástegui, L.L.; Arrieta-Flores, D.; Gregorio-Jorge, J. An overview of PRR- and NLR-mediated immunities: Conserved signaling components across the plant kingdom that communicate both pathways. Int. J. Mol. Sci. 2022, 23, 12974. [Google Scholar] [CrossRef]
- Gong, B.Q.; Guo, J.; Zhang, N.; Yao, X.; Wang, H.B.; Li, J.F. Cross-microbial protection via priming a conserved immune co-receptor through juxtamembrane phosphorylation in plants. Cell Host Microbe 2019, 26, 810–822. [Google Scholar] [CrossRef]
- Kushalappa, A.C.; Yogendra, K.N.; Karre, S. Plant innate immune response: Qualitative and quantitative resistance. CRC. Crit. Rev. Plant Sci. 2016, 35, 38–55. [Google Scholar] [CrossRef] [Green Version]
- Lai, Z.; Mengiste, T. Genetic and cellular mechanisms regulating plant responses to necrotrophic pathogens. Curr. Opin. Plant Biol. 2013, 16, 505–512. [Google Scholar] [CrossRef]
- Agrios, G.N. Chapter six—How plants defend themselves against pathogens. In Plant Pathology; Elsevier Academic Press: Amsterdam, The Netherlands, 2005; pp. 207–248. [Google Scholar]
- Nawrot, R.; Barylski, J.; Nowicki, G.; Broniarczyk, J.; Buchwald, W.; Goździcka-Józefiak, A. Plant antimicrobial peptides. Folia Microbiol. 2014, 59, 181–196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yogendra, K.N.; Pushpa, D.; Mosa, K.A.; Kushalappa, A.C.; Murphy, A.; Mosquera, T. Quantitative resistance in potato leaves to late blight associated with induced hydroxycinnamic acid amides. Funct. Integr. Genom. 2014, 14, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.H.; Meng, J.; Zhang, M.; Luan, Y.-S. Identification of tomato circular RNAs responsive to Phytophthora infesta identification of tomato circular RNAs responsive to Phytophthora infestans. Gene 2020, 746, 144652. [Google Scholar] [CrossRef]
- Backer, R.; Naidoo, S.; Van den Berg, N. The NONEXPRESSOR OF PATHOGENESIS-RELATED GENES 1 (NPR1) and related family: Mechanistic insights in plant disease resistance. Front. Plant Sci. 2019, 10, 102. [Google Scholar] [CrossRef] [Green Version]
- Zhou, J.M.; Zhang, Y. Plant immunity: Danger perception and signaling. Cell 2020, 181, 978–989. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.M.; Zhu, S.; Kachroo, P.; Kachroo, A. Signal regulators of systemic acquired resistance. Front. Plant Sci. 2015, 6, 228. [Google Scholar] [CrossRef] [Green Version]
- Hao, Q.; Wang, W.; Han, X.; Wu, X.; Lyu, B.; Chen, F.; Caplan, A.; Li, C.; Wu, J.; Wang, W.; et al. Isochorismate-based salicylic acid biosynthesis confers basal resistance to Fusarium graminearum in barley. Mol. Plant Pathol. 2018, 19, 1995–2010. [Google Scholar] [CrossRef] [Green Version]
- Pieterse, C.M.J.; Van Wees, S.C.M.; Van Pelt, J.A.; Knoester, M.; Laan, R.; Gerrits, H.; Weisbeek, P.J.; Van Loon, L.C. A novel signaling pathway controlling induced systemic resistance in Arabidopsis. Plant Cell 1998, 10, 1571–1580. [Google Scholar] [CrossRef] [Green Version]
- Niki, T.; Mitsuhara, I.; Seo, S.; Ohtsubo, N.; Ohashi, Y. Antagonistic effect of salicylic acid and jasmonic acid on the expression of pathogenesis-related (PR) protein genes in wounded mature tobacco leaves. Plant Cell Physiol. 1998, 39, 500–507. [Google Scholar] [CrossRef]
- Leon-Reyes, A.; Spoel, S.H.; De Lange, E.S.; Abe, H.; Kobayashi, M.; Tsuda, S.; Millenaar, F.F.; Welschen, R.A.M.; Ritsema, T.; Pieterse, C.M.J. Ethylene modulates the role of NONEXPRESSOR OF PATHOGENESIS-RELATED GENES1 in cross talk between salicylate and jasmonate signaling. Plant Physiol. 2009, 149, 1797–1809. [Google Scholar] [CrossRef] [Green Version]
- Abe, H.; Tomitaka, Y.; Shimoda, T.; Seo, S.; Sakurai, T.; Kugimiya, S.; Tsuda, S.; Kobayashi, M. Antagonistic plant defense system regulated by phytohormones assists interactions among vector insect, thrips and a tospovirus. Plant Cell Physiol. 2012, 53, 204–212. [Google Scholar] [CrossRef] [Green Version]
- Thomas, B.; Murray, B.G.; Murphy, D.J. Encyclopedia of Applied Plant Sciences; Academic Press: Cambridge, MA, USA, 2016; pp. 1–3. [Google Scholar]
- Piwowar, A. The use of pesticides in Polish agriculture after integrated pest management (IPM) implementation. Environ. Sci. Pollut. Res. 2021, 28, 26628–26642. [Google Scholar] [CrossRef]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq-A Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexa, A.; Rahnenfuhrer, J. TopGO: Enrichment Analysis for Gene Ontology. R Package Version 2.38.1 2019. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9630283/ (accessed on 20 January 2022).
- Mao, X.; Cai, T.; Olyarchuk, J.G.; Wei, L. Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 2005, 21, 3787–3793. [Google Scholar] [CrossRef]
- Halka, M.; Smoleń, S.; Czernicka, M.; Klimek Chodacka, M.; Pitala, J.; Tutaj, K. Iodine biofortification through expression of HMT, SAMT and S3H genes in Solanum lycopersicum L. Plant Physiol. Biochem. 2019, 144, 35–48. [Google Scholar] [CrossRef]
- Smoleń, S.; Kowalska, I.; Halka, M.; Ledwożyw-Smoleń, I.; Grzanka, M.; Skoczylas, Ł.; Czernicka, M.; Pitala, J. Selected Aspects of iodate and iodosalicylate metabolism in lettuce including the activity of vanadium dependent haloperoxidases as affected by exogenous vanadium. Agronomy 2020, 10, 1. [Google Scholar] [CrossRef] [Green Version]
- Luna, E.; Bruce, T.J.A.; Roberts, M.R.; Flors, V.; Ton, J. Next-generation systemic acquired resistance. Plant Physiol. 2012, 158, 844–853. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smiglak, M.; Kukawka, R.; Lewandowski, P.; Pospieszny, H. Cationic derivatives of the plant resistance inducer benzo[1,2,3]thiadiazole-7-carbothioic acid S-methyl ester (BTH) as bifunctional ionic liquids. Tetrahedron Lett. 2014, 55, 3565–3568. [Google Scholar] [CrossRef]

| Gene Name | Description | Gene ID | Chromosome | log2 Fold Change Planticine® vs. Control | False Discovery Rate (FDR) |
|---|---|---|---|---|---|
| Elicitor and effector recognition receptor genes (ELRR and ERR) | |||||
| EIX1 | Receptor-like protein EIX1 | Solyc07g008620.1 | 7 | 1.1 | 3 × 10−4 |
| EIX2 | Receptor-like protein EIX2 | Solyc12g005610.2 | 12 | 1.0 | 4 × 10−2 |
| ELF | Elongation factor Tu | Solyc03g112150.1 | 3 | −0.6 | 1 × 10−11 |
| CERK1 (LYK4) | Chitin elicitor receptor kinase 1, LysM domain receptor-like kinase 4 | Solyc02g089920.2 | 2 | 2.1 | 5 × 10−4 |
| CERK1 (LYK3) | Chitin elicitor receptor kinase 1, LysM domain receptor-like kinase 3 | Solyc06g075030.1 | 6 | −0.7 | 8 × 10−3 |
| CERK1 | Chitin elicitor receptor kinase 1 | Solyc07g049180.3 | 7 | 0.8 | 1 × 10−4 |
| RLK5 | Receptor-like protein kinase 5 | Solyc08g066310.2 | 8 | 1.0 | 1 × 10−9 |
| RLK7 | Receptor-like protein kinase 7 | Solyc01g106500.3 | 1 | 1.0 | 3 × 10−2 |
| RLK | Probable receptor-like protein kinase At1g33260 | Solyc12g005450.1 | 12 | 3.4 | 1 × 10−27 |
| Putative receptor-like protein kinase At1g72540 | Solyc06g062920.3 | 6 | 3.5 | 6 × 10−5 | |
| CNGC | Probable cyclic nucleotide-gated ion channel | Solyc05g050380.3 | 5 | 1.0 | 6 × 10−4 |
| Solyc03g098210.3 | 3 | 0.6 | 2 × 10−3 | ||
| FLII | Flagellum-specific ATP synthase | Solyc01g107740.3 | 1 | 2.0 | 8 × 10−21 |
| FLS2 | LRR receptor-like serine/threonine-protein kinase FLS2 | Solyc02g070920.3 | 2 | −0.6 | 3 × 10−6 |
| SIK1 | LRR receptor-like serine/threonine-protein kinase SIK1 | Solyc08g066320.3 | 8 | 3.2 | 6 × 10−16 |
| GSO2 | LRR receptor-like serine/threonine-protein kinase GSO2 | Solyc10g052880.1 | 10 | 2.0 | 3 × 10−7 |
| RFK1 | Probable LRR receptor-like serine/threonine-protein kinase RFK1 | Solyc02g071870.3 | 2 | 0.6 | 2 × 10−2 |
| FEI2 | LRR receptor-like serine/threonine-protein kinase FEI2 | Solyc03g059490.1 | 3 | 0.6 | 3 × 10−6 |
| WAK | Wall-associated receptor kinase-like | Solyc02g090110.3 | 2 | 1.2 | 5 × 10−9 |
| LRL | Leaf rust 10 disease-resistance locus receptor-like protein kinase-like | Solyc01g008500.3 | 1 | 1.2 | 10 × 10−5 |
| SERK1 (BAKIBKK1) | Somatic embryogenesis receptor kinase 1 (brassinosteroid insensitive 1-associated receptor kinase 1) | Solyc04g072560.3 | 4 | 0.5 | 9 × 10−4 |
| Mitogen-activated protein kinases (MAPKs) | |||||
| SlMAPKKK76 | Calcium-dependent protein kinase 76 | Solyc10g079130.2 | 10 | 1.3 | 7 × 10−8 |
| CDPK | Calcium-dependent protein kinase | Solyc02g083850.3 | 2 | 1.0 | 2 × 10−2 |
| CDPK2 | Calcium-dependent protein kinase 2 | Solyc03g033540.3 | 3 | 0.7 | 2 × 10−3 |
| CDPK4 | Calcium-dependent protein kinase 4 | Solyc06g065380.3 | 6 | −0.8 | 2 × 10−4 |
| CML | Calmodulin-like protein (putative calcium-binding protein CML19) | Solyc02g094000.1 | 2 | 3.4 | 6 × 10−4 |
| Calmodulin-like protein (probable calcium-binding protein CML45 | Solyc06g069740.1 | 6 | 2.1 | 2 × 10−10 | |
| Calcium-binding protein CML37 | Solyc11g071760.2 | 11 | 3.1 | 3 × 10−3 | |
| Calmodulin-like protein (probable calcium-binding protein CML) | Solyc03g005040.1 | 3 | 3.0 | 2 × 10−7 | |
| Calmodulin-like protein (probable calcium-binding protein CML) | Solyc06g073830.1 | 6 | 2.3 | 1 × 10−8 | |
| Probable calcium-binding protein CML10 | Solyc01g091465.1 | 1 | 1.8 | 3 × 10−13 | |
| Calmodulin (calcium-binding protein CML24) | Solyc02g091500.1 | 2 | 1.6 | 2 × 10−7 | |
| Calmodulin-like protein (probable calcium-binding protein CML46) | Solyc03g115930.2 | 3 | 1.4 | 5 × 10−7 | |
| Calmodulin (calcium-binding protein CML23) | Solyc02g063350.1 | 2 | 1.0 | 6 × 10−16 | |
| Calmodulin-like protein (probable calcium-binding protein CML45) | Solyc02g088090.1 | 2 | 1.0 | 2 × 10−3 | |
| Probable calcium-binding protein CML36 | Solyc10g079755.1 | 10 | −0.4 | 1 × 10−5 | |
| MPK3 | Mitogen-activated protein kinase 3 | Solyc06g005170.3 | 6 | 1.9 | 3 × 10−3 |
| MKS1 | MAP kinase substrate 1 | Solyc11g005720.1 | 11 | 1.4 | 4 × 10−5 |
| Transcription factor genes (TF) and other regulatory genes | |||||
| WRKY | Probable WRKY 53 | Solyc08g008280.3 | 8 | 3.4 | 9 × 10−5 |
| WRKY 22 | Solyc10g011910.3 | 10 | 3.2 | 5 × 10−4 | |
| Probable WRKY 41 | Solyc03g007380.2 | 3 | 3.1 | 6 × 10−12 | |
| Probable WRKY 40 | Solyc03g116890.3 | 3 | 3.1 | 7 × 10−5 | |
| Probable WRKY 60 | Solyc08g067360.3 | 8 | 2.6 | 4 × 10−9 | |
| WRKY 72A | Solyc03g113120.3 | 3 | 2.6 | 1 × 10−2 | |
| WRKY 6 | Solyc02g080890.3 | 2 | 2.5 | 3 × 10−9 | |
| Probable WRKY 33 | Solyc09g014990.3 | 9 | 2.4 | 2 × 10−2 | |
| WRKY 70 | Solyc03g095770.3 | 3 | 2.3 | 6 × 10−3 | |
| Probable WRKY 30 | Solyc10g009550.3 | 10 | 2.1 | 6 × 10−31 | |
| Probable WRKY 7 | Solyc04g078550.3 | 4 | 1.9 | 2 × 10−11 | |
| Probable WRKY 75 | Solyc05g015850.3 | 5 | 1.7 | 4 × 10−4 | |
| Probable WRKY 51 | Solyc04g051690.3 | 4 | 1.5 | 7 × 10−5 | |
| Probable WRKY 11 | Solyc12g096350.2 | 12 | 1.5 | 2 × 10−6 | |
| WRKY WRKY76 | Solyc06g068460.3 | 6 | 1.4 | 4 × 10−2 | |
| Probable WRKY 50 | Solyc08g062490.3 | 8 | 1.4 | 1 × 10−5 | |
| WRKY 22 | Solyc01g095100.3 | 1 | 1.2 | 3 × 10−11 | |
| Probable WRKY 19 | Solyc01g104910.3 | 1 | −0.3 | 3 × 10−3 | |
| Probable WRKY 21 | Solyc09g066010.3 | 9 | −0.4 | 4 × 10−2 | |
| ERF | Ethylene-responsive TF ERF22 | Solyc11g042560.1 | 11 | 5.4 | 2 × 10−7 |
| Ethylene-responsive TF ERF112 | Solyc02g090770.1 | 2 | 4.5 | 2 × 10−4 | |
| Ethylene-responsive TF ERF109 | Solyc10g050970.1 | 10 | 4.2 | 2 × 10−3 | |
| Ethylene-responsive TF ERF17 | Solyc06g054630.2 | 6 | 3.8 | 7 × 10−15 | |
| Ethylene-responsive TF ERF25 | Solyc06g035700.1 | 6 | 3.2 | 1 × 10−17 | |
| Ethylene-responsive TF ERF109 | Solyc01g108240.3 | 1 | 2.9 | 1 × 10−2 | |
| Ethylene-responsive TF ERF118 | Solyc04g009435.1 | 4 | 2.8 | 1 × 10−4 | |
| Ethylene-responsive TF ERF12 | Solyc11g012980.1 | 11 | 2.1 | 3 × 10−6 | |
| Ethylene-responsive TF ERF5 | Solyc08g078190.1 | 8 | 1.5 | 2 × 10−4 | |
| Ethylene-responsive TF ERF4 | Solyc07g053740.1 | 7 | 1.0 | 2 × 10−2 | |
| Ethylene-responsive TF ERFC3 | Solyc04g014530.1 | 4 | −1.4 | 4 × 10−4 | |
| Ethylene-responsive TF ERF13 | Solyc01g090340.2 | 1 | −2.2 | 1 × 10−16 | |
| MYB | Transcription factor MYB14 | Solyc07g054980.2 | 7 | 3.7 | 7 × 10−3 |
| Transcription factor MYB77 | Solyc04g079360.1 | 4 | 3.2 | 3 × 10−24 | |
| Transcription factor MYB14 | Solyc12g005640.2 | 12 | 2.0 | 2 × 10−3 | |
| Transcription factor MYB15 | Solyc07g053230.3 | 7 | 1.9 | 2 × 10−7 | |
| Transcription factor MYB102 | Solyc02g079280.3 | 2 | 1.6 | 2 × 10−2 | |
| Transcription factor MYB20 | Solyc11g011050.2 | 11 | 1.5 | 4 × 10−3 | |
| Transcription factor MYB73 | Solyc04g078420.1 | 4 | 0.9 | 1 × 10−9 | |
| Transcription factor MYB36 | Solyc09g008250.3 | 9 | −1.2 | 8 × 10−3 | |
| Transcription factor MYB16 | Solyc02g088190.3 | 2 | −1.1 | 2 × 10−8 | |
| Transcription factor MYB113 | Solyc10g086250.2 | 10 | −1.0 | 1 × 10−7 | |
| NAC | NAC domain-containing protein 90 | Solyc11g068620.2 | 11 | 2.4 | 4 × 10−3 |
| NAC domain-containing protein 22 | Solyc10g055760.2 | 10 | 1.2 | 1 × 10−5 | |
| NAC domain-containing protein 79 | Solyc03g115850.3 | 3 | 1.0 | 3 × 10−3 | |
| NAC domain-containing protein 17 | Solyc04g072220.3 | 4 | 0.4 | 4 × 10−2 | |
| NAC domain-containing protein 35 | Solyc01g102740.3 | 1 | −0.5 | 2 × 10−3 | |
| bZIP | BZIP TF family protein expressed | Solyc01g110480.3 | 1 | 1.2 | 4 × 10−9 |
| WFI1 | whitefly-induced gp91 (circRNA) | Solyc03g117980.3 | 3 | 2.1 | 2 × 10−4 |
| RBOHC | Respiratory burst oxidase homolog protein C | Solyc03g117980.3 | 3 | 2.1 | 2 × 10−4 |
| Resistance-related protein (RRP) biosynthetic genes | |||||
| A70 | Pathogen-associated molecular patterns-induced protein A70 | Solyc01g079660.2 | 1 | 3.2 | 1 × 10−3 |
| PTI5 | Pathogenesis-related genes transcriptional activator PTI5 | Solyc02g077370.1 | 2 | 2.1 | 9 × 10−7 |
| PTI6 | Pathogenesis-related genes transcriptional activator PTI6 | Solyc06g082590.1 | 6 | 0.9 | 1 × 10−2 |
| PR1 | Thaumatin, pathogenesis-related protein | Solyc12g056390.2 | 12 | 1.4 | 8 × 10−6 |
| PRS2 | Pathogenesis-related protein STH-2 | Solyc12g096960.2 | 12 | 1.2 | 2 × 10−3 |
| E70 | Exocyst complex protein EXO70 | Solyc09g005830.1 | 9 | 2.0 | 2 × 10−2 |
| Solyc06g075610.1 | 6 | 1.4 | 2 × 10−4 | ||
| Solyc11g073010.1 | 11 | 1.0 | 3 × 10−2 | ||
| RIN4 | RPM1-interacting protein 4 | Solyc09g059430.3 | 9 | 1.4 | 1 × 10−6 |
| Solyc06g083390.3 | 6 | 1.2 | 1 × 10−6 | ||
| RIN1 | RPM1-interacting protein 1 | Solyc11g010170.2 | 11 | 0.9 | 2 × 10−6 |
| R13L4 | Disease resistance RPP13-like protein 4 | Solyc02g084890.2 | 2 | 2.0 | 2 × 10−3 |
| PBS1 | Probable serine/threonine-protein kinase PBL7 | Solyc11g072660.2 | 11 | 1.4 | 1 × 10−5 |
| Probable serine/threonine-protein kinase PBL19 | Solyc08g077560.3 | 8 | 2.3 | 3 × 10−9 | |
| Probable serine/threonine-protein kinase PBL3 | Solyc01g010660.3 | 1 | 1.8 | 4 × 10−9 | |
| HSP71 | Heat shock cognate 70 kDa protein 1 | Solyc06g076020.3 | 6 | 2.4 | 3 × 10−2 |
| HSP12 | 18.2 kDa class I heat shock protein | Solyc10g086680.1 | 10 | 1.9 | 4 × 10−6 |
| HSF24 | Heat shock factor protein HSF24 | Solyc02g090820.3 | 2 | 1.5 | 2 × 10−8 |
| HS704 | Heat shock 70 kDa protein 4 | Solyc06g005435.1 | 6 | −1.8 | 1 × 10−3 |
| EDS1 | Protein EDS1 | Solyc06g071280.3 | 6 | 0.8 | 2 × 10−5 |
| KCS11 | 3-ketoacyl-CoA synthase 11 | Solyc06g065560.2 | 6 | 1.8 | 9 × 10−13 |
| Solyc09g065800.3 | 9 | −2.2 | 1 × 10−10 | ||
| HIR1 | Hypersensitive-induced reaction 1 protein | Solyc03g113220.3 | 3 | 0.4 | 3 × 10−2 |
| ROQ1 | Disease resistance protein Roq1 | Solyc01g102840.3 | 1 | 2.8 | 1 × 10−8 |
| RPV1 | Disease resistance protein RPV1 | Solyc01g102880.2 | 1 | 1.3 | 1 × 10−8 |
| RUN1 | Disease resistance protein RUN1 | Solyc04g007320.2 | 4 | 1.2 | 1 × 10−5 |
| NGR1 | Probable disease resistance protein | Solyc02g090380.3 | 2 | 1.1 | 4 × 10−7 |
| EDR4 | Protein enhanced disease resistance 4 | Solyc03g095610.3 | 3 | 0.6 | 3 × 10−2 |
| DGK1 | Diacylglycerol kinase 1 | Solyc03g115370.3 | 3 | 0.5 | 6 × 10−3 |
| MCA1 | Metacaspase-1 | Solyc03g094160.3 | 3 | 1.5 | 3 × 10−7 |
| Resistance-related metabolite (RRM) biosynthetic genes | |||||
| LOX31 | Linoleate 13S-lipoxygenase 3-1 | Solyc03g122340.3 | 3 | 2.8 | 1 × 10−18 |
| LOX21 | Linoleate 13S-lipoxygenase 2-1 | Solyc12g011030.2 | 12 | 2.3 | 3 × 10−26 |
| LOX15 | Probable linoleate 9S-lipoxygenase 5 | Solyc08g029000.3 | 8 | 1.7 | 2 × 10−2 |
| AOS | Allene oxide synthase 1 | Solyc04g079730.1 | 4 | 2.7 | 6 × 10−3 |
| TPS5 | (R)-linalool synthase TPS5 | Solyc01g105890.3 | 1 | 1.1 | 1 × 10−4 |
| ACS3 | 1-aminocyclopropane-1-carboxylate synthase 3 | Solyc02g063540.2 | 2 | 3.7 | 1 × 10−4 |
| Solyc02g091990.3 | 2 | 2.8 | 6 × 10−6 | ||
| ACS CMW33 | 1-aminocyclopropane-1-carboxylate synthase CMW33 | Solyc08g081555.1 | 8 | 1.8 | 5 × 10−9 |
| ACS | 1-aminocyclopropane-1-carboxylate synthase | Solyc08g081540.3 | 8 | 1.7 | 3 × 10−5 |
| Treatment | Dose L ha−1 | µg kg−1 d.w. | ||
|---|---|---|---|---|
| SA | JA | ABA | ||
| Control | - | 671.7 ± 52.8 | 8.39 ± 2.9 | 563.5 ± 24.2 |
| Planticine® | 2 | 885.8 ± 50.6 | 5.16 ± 1.16 | 585.8 ± 40.9 |
| Test t-student | - | * | * | NS |
| Treatment | Dose per ha (L ha−1) | Observation | |||||
|---|---|---|---|---|---|---|---|
| T3 + 10 | T4 + 10 | T5 + 10 | |||||
| Leaves Infestation (%)/Efficacy (%) | |||||||
| Control | - | 8.78 c | - | 30.2 d | - | 68.8 d | - |
| Planticine® | 2 | 0.78 b | 91% | 7.4 b | 76% | 36.6 b | 47% |
| Planticine® | 3 | 0.84 b | 90% | 10.4 c | 66% | 42.9 c | 38% |
| Scorpion 325 SC | 1 | 0.0 a | 100% | 0.0 a | 100% | 0.2 a | 99% |
| Treatment | Dose per ha (L/kg ha−1) | Observation | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T0 | T1 + 3 | T1 + 7 | T2 + 7 | T2 + 14 | T2 + 21 | |||||||
| Number of Adults/Efficacy (%) | ||||||||||||
| Control | - | 3.5 a | 1.75 a | - | 2.25 a | - | 2.75 ab | - | 2.75 ab | - | 5.0 a | - |
| Planticine® | 2 | 7.25 a | 2.0 a | 45 | 3.75 a | 20 | 2.25 a | 61 | 1.5 a | 74 | 3.25 a | 69 |
| Planticine® | 3 | 2.25 a | 3.25 a | - | 3.25 a | - | 3.25 ab | - | 6.0 c | - | 1.0 a | 69 |
| Mospilan 20 SP | 0.24 | 6.0 a | 6.5 b | - | 5.25 a | - | 5.25 b | - | 3.5 b | 26 | 2.0 a | 77 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rakoczy-Lelek, R.; Czernicka, M.; Ptaszek, M.; Jarecka-Boncela, A.; Furmanczyk, E.M.; Kęska-Izworska, K.; Grzanka, M.; Skoczylas, Ł.; Kuźnik, N.; Smoleń, S.; et al. Transcriptome Dynamics Underlying Planticine®-Induced Defense Responses of Tomato (Solanum lycopersicum L.) to Biotic Stresses. Int. J. Mol. Sci. 2023, 24, 6494. https://doi.org/10.3390/ijms24076494
Rakoczy-Lelek R, Czernicka M, Ptaszek M, Jarecka-Boncela A, Furmanczyk EM, Kęska-Izworska K, Grzanka M, Skoczylas Ł, Kuźnik N, Smoleń S, et al. Transcriptome Dynamics Underlying Planticine®-Induced Defense Responses of Tomato (Solanum lycopersicum L.) to Biotic Stresses. International Journal of Molecular Sciences. 2023; 24(7):6494. https://doi.org/10.3390/ijms24076494
Chicago/Turabian StyleRakoczy-Lelek, Roksana, Małgorzata Czernicka, Magdalena Ptaszek, Anna Jarecka-Boncela, Ewa M. Furmanczyk, Kinga Kęska-Izworska, Marlena Grzanka, Łukasz Skoczylas, Nikodem Kuźnik, Sylwester Smoleń, and et al. 2023. "Transcriptome Dynamics Underlying Planticine®-Induced Defense Responses of Tomato (Solanum lycopersicum L.) to Biotic Stresses" International Journal of Molecular Sciences 24, no. 7: 6494. https://doi.org/10.3390/ijms24076494
APA StyleRakoczy-Lelek, R., Czernicka, M., Ptaszek, M., Jarecka-Boncela, A., Furmanczyk, E. M., Kęska-Izworska, K., Grzanka, M., Skoczylas, Ł., Kuźnik, N., Smoleń, S., Macko-Podgórni, A., Gąska, K., Chałańska, A., Ambroziak, K., & Kardasz, H. (2023). Transcriptome Dynamics Underlying Planticine®-Induced Defense Responses of Tomato (Solanum lycopersicum L.) to Biotic Stresses. International Journal of Molecular Sciences, 24(7), 6494. https://doi.org/10.3390/ijms24076494








