Exploring the Conformational Equilibrium of Mefenamic Acid Released from Silica Aerogels via NMR Analysis
Abstract
:1. Introduction
2. Results and Discussion
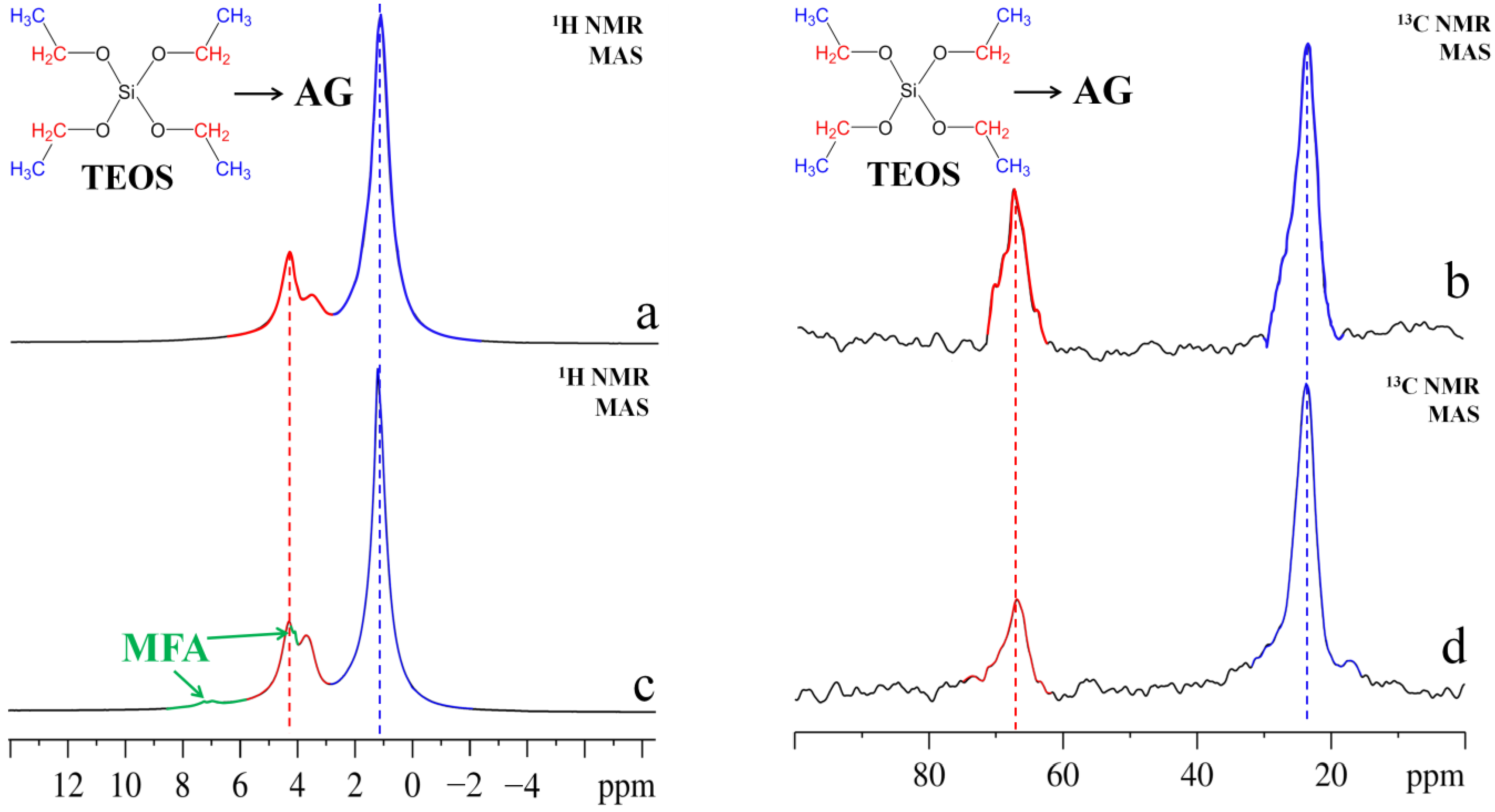
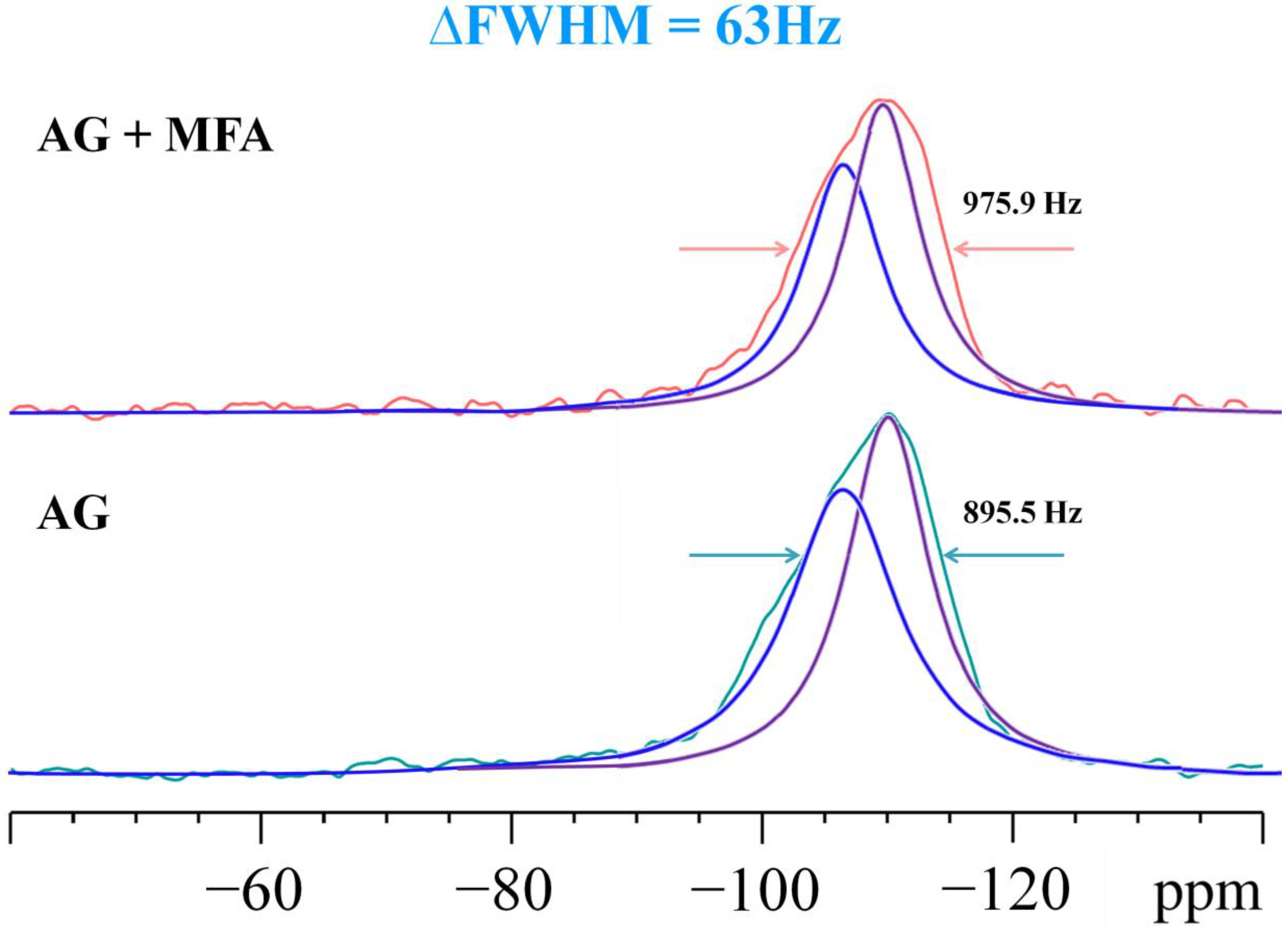
2.1. Solid State MAS NMR Analysis of Aerogel Composite Material
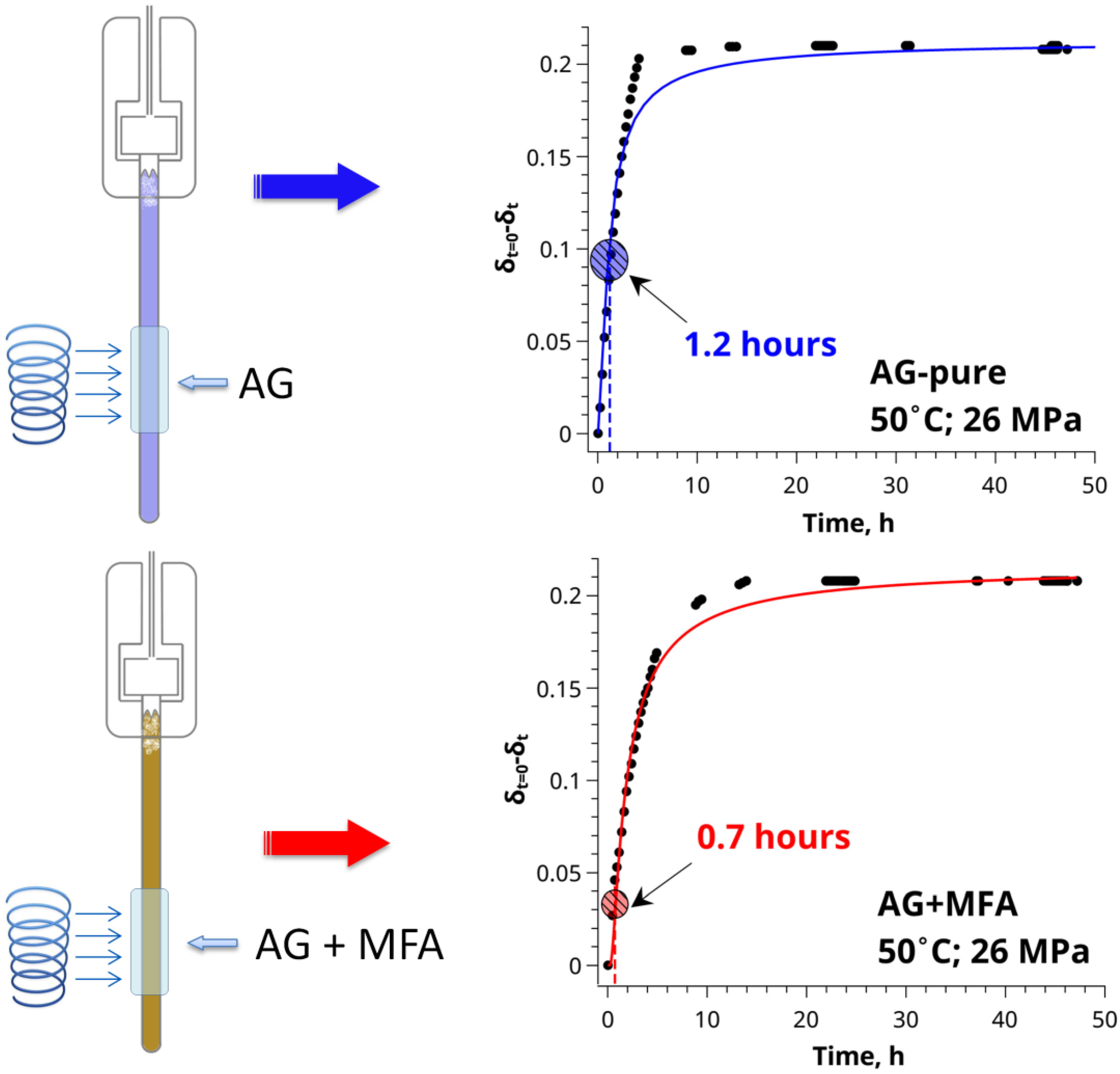
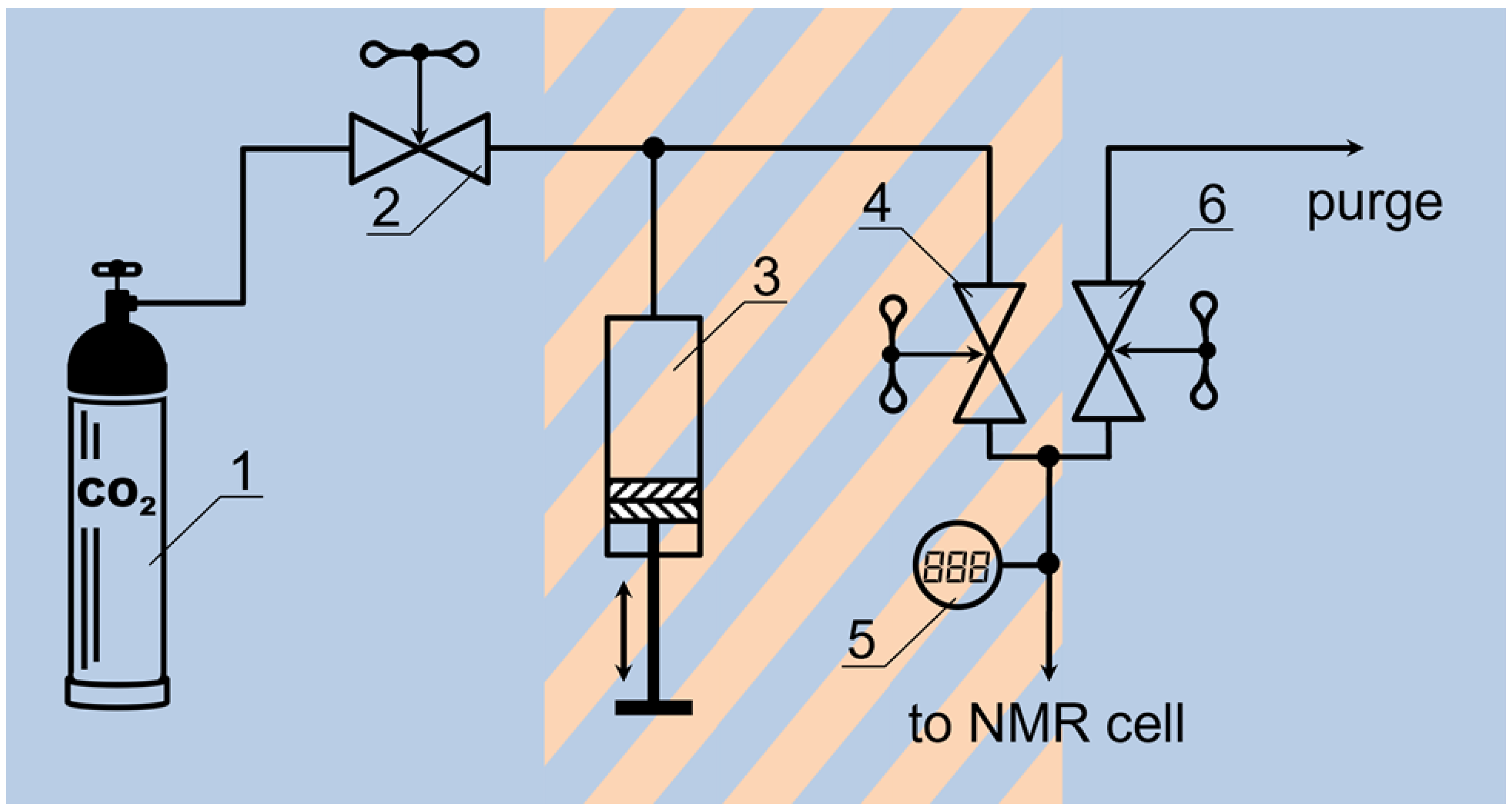
2.2. High-Pressure 13C NMR Kinetic Study of CO2 Sorption
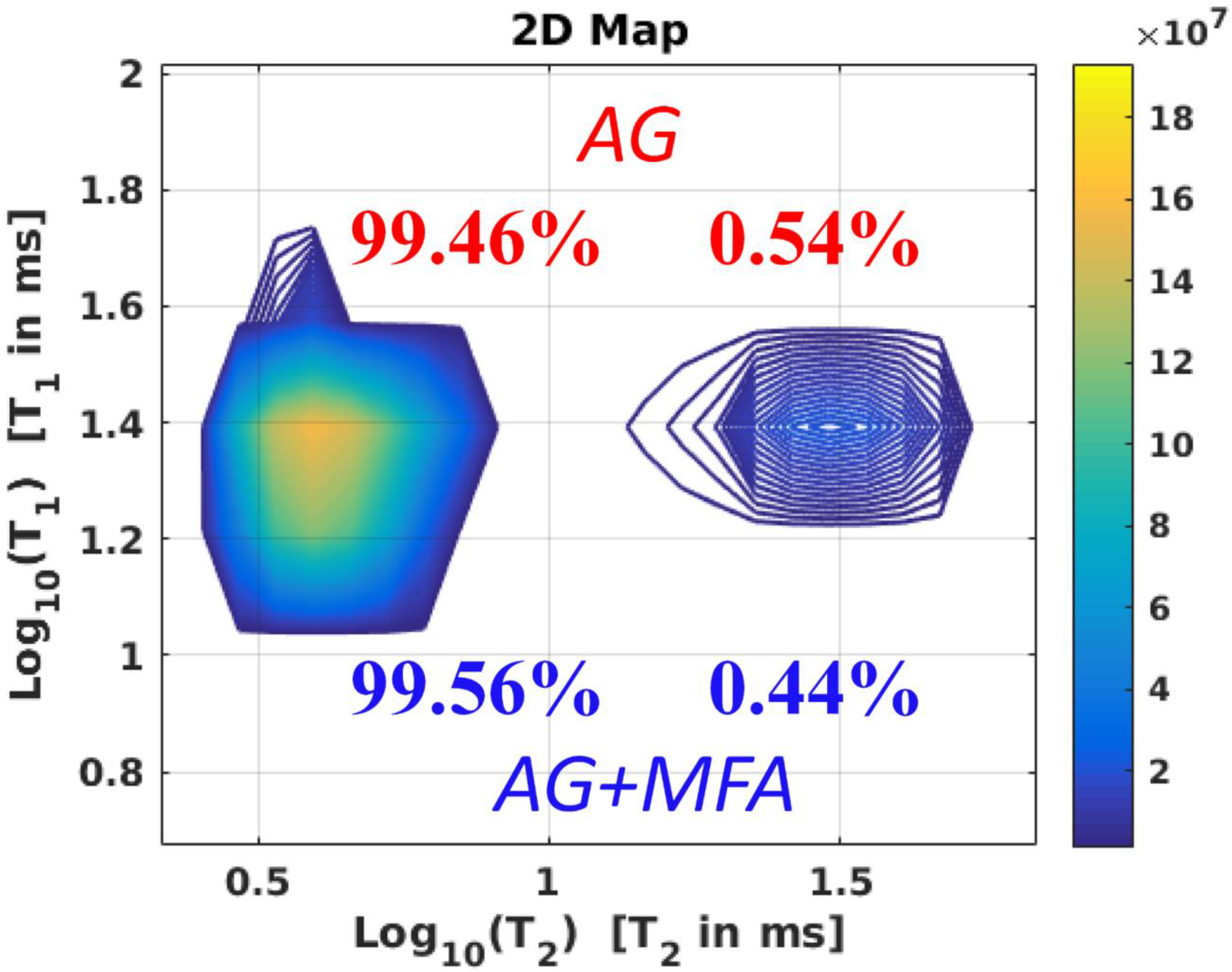
2.3. A High-Pressure T1–T2 RRCOSY Study
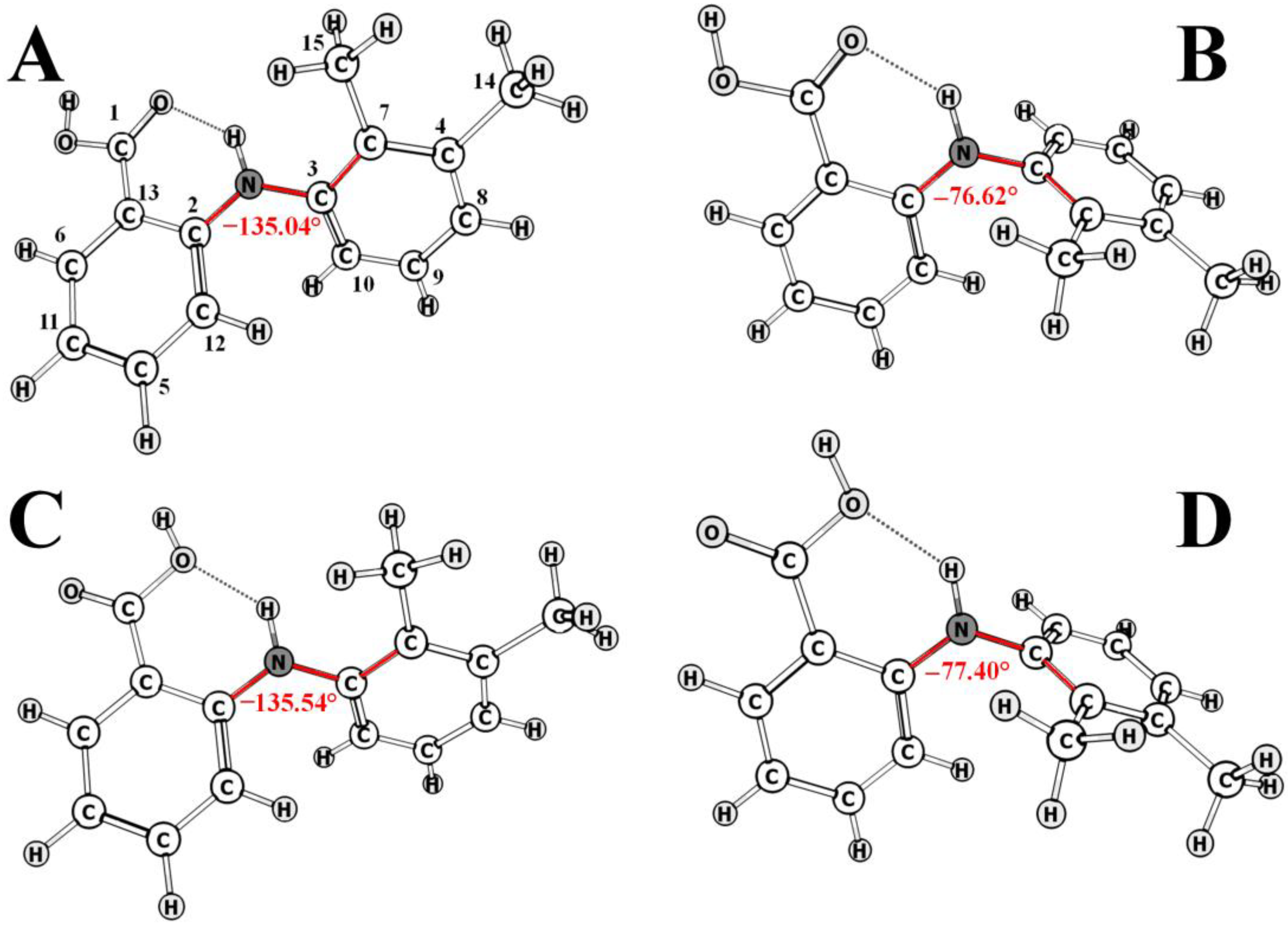
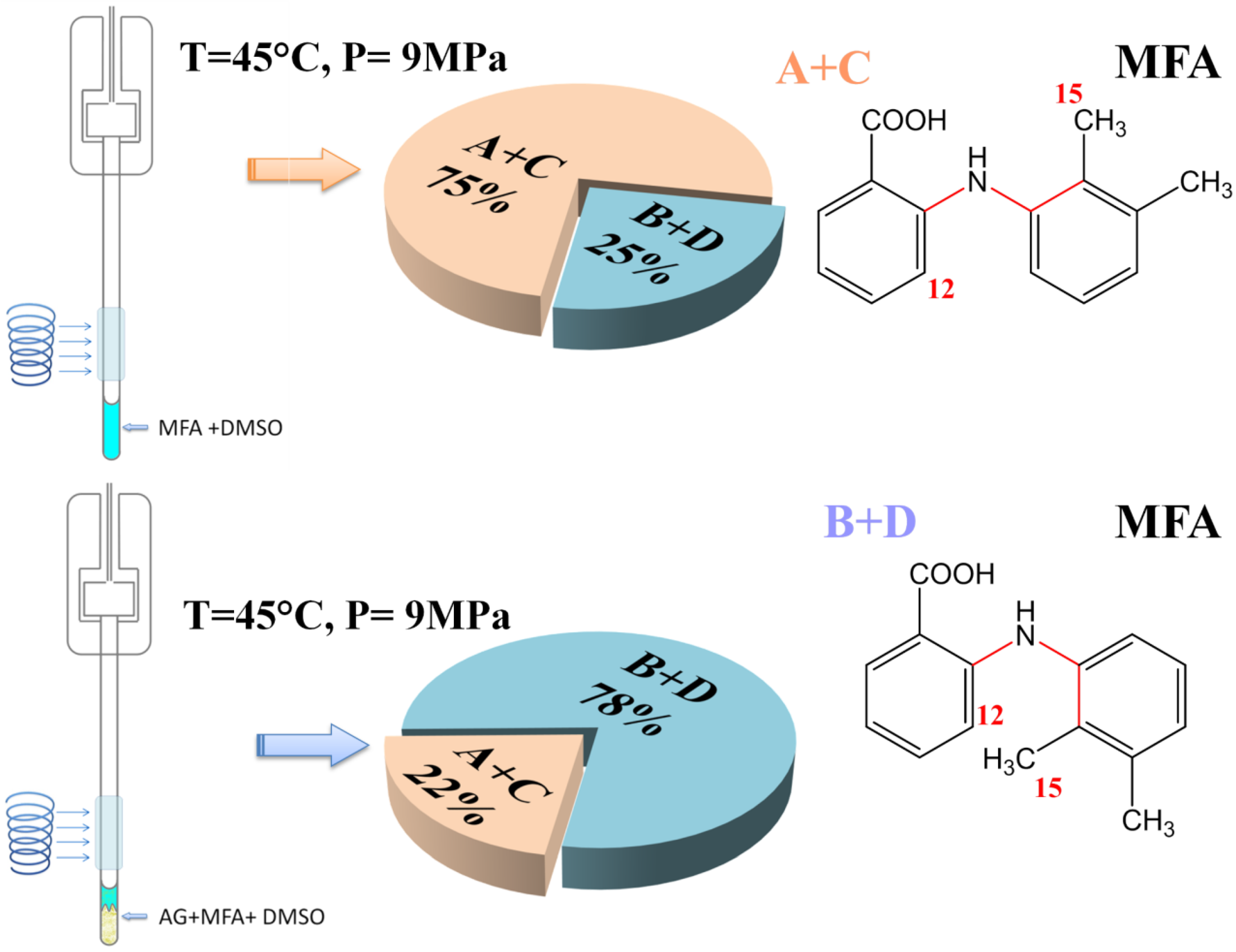
2.4. High-Pressure NOESY Study of the Conformational Preference of Mefenamic Acid Release
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Guo, W.; Yang, C.; Cui, L.; Lin, H.; Qu, F. An Enzyme-Responsive Controlled Release System of Mesoporous Silica Coated with Konjac Oligosaccharide. Langmuir 2014, 30, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Champeau, M.; Thomassin, J.M.; Tassaing, T.; Jérôme, C. Drug Loading of Polymer Implants by Supercritical CO2 Assisted Impregnation: A Review. J. Control. Release 2015, 209, 248–259. [Google Scholar] [CrossRef]
- Smirnova, I.; Suttiruengwong, S.; Arlt, W. Feasibility Study of Hydrophilic and Hydrophobic Silica Aerogels as Drug Delivery Systems. J. Non. Cryst. Solids 2004, 350, 54–60. [Google Scholar] [CrossRef]
- Alleso, M.; Chieng, N.; Rehder, S.; Rantanen, J.; Rades, T.; Aaltonen, J. Enhanced Dissolution Rate and Synchronized Release of Drugs in Binary Systems through Formulation: Amorphous Naproxen–cimetidine Mixtures Prepared by Mechanical Activation. J. Control. Release 2009, 136, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.C.; Ng, W.K.; Chia, L.; Dong, Y.C.; Tan, R.B.H. Stabilized Amorphous State of Ibuprofen by Co-Spray Drying With Mesoporous SBA-15 to Enhance Dissolution Properties. J. Pharm. Sci. 2010, 99, 1997–2007. [Google Scholar] [CrossRef] [PubMed]
- Blagden, N.; de Matas, M.; Gavan, P.T.; York, P. Crystal Engineering of Active Pharmaceutical Ingredients to Improve Solubility and Dissolution Rates. Adv. Drug Deliv. Rev. 2007, 59, 617–630. [Google Scholar] [CrossRef] [PubMed]
- Franco, P.; Marco, I. De Supercritical Antisolvent Process for Pharmaceutical Applications: A Review. Process 2020, 8, 938. [Google Scholar] [CrossRef]
- Singh, N.; Vinjamur, M.; Mukhopadhyay, M. Insights into Adsorptive Drug Loading on Silica Aerogels from Supercritical CO2. Langmuir 2022, 38, 13075–13083. [Google Scholar] [CrossRef]
- Akhter, F.; Jamali, A.R.; Khan, W. TEOS-Doped vs Non-TEOS Silica Aerogels: A Comparative Study of Synthesis, Characterization, Isotherm Studies and Performance Evaluation for Pb (II) Removal from Synthetic Wastewater. Water. Air. Soil Pollut. 2023, 234, 1–9. [Google Scholar] [CrossRef]
- Yar, A. Enhanced Output Performance of Tetraethyl Orthosilicate and Graphene Nanoplates-Decorated Nanofiber-Based Triboelectric Nanogenerators. Colloids Surf. A Physicochem. Eng. Asp. 2021, 631, 127670. [Google Scholar] [CrossRef]
- Giray, S.; Bal, T.; Kartal, A.M.; Kizilel, S.; Erkey, C. Controlled Drug Delivery through a Novel PEG Hydrogel Encapsulated Silica Aerogel System. J. Biomed. Mater. Res. Part A 2012, 100, 1307–1315. [Google Scholar] [CrossRef] [PubMed]
- Ulker, Z.; Erkey, C. An Emerging Platform for Drug Delivery: Aerogel Based Systems. J. Control. Release 2014, 177, 51–63. [Google Scholar] [CrossRef]
- Nakapraves, S.; Warzecha, M.; Mustoe, C.L.; Srirambhatla, V.; Florence, A.J. Prediction of Mefenamic Acid Crystal Shape by Random Forest Classification. Pharm. Res. 2022, 39, 3099–3111. [Google Scholar] [CrossRef] [PubMed]
- Charoenchaitrakool, M.; Roubroum, T.; Sudsakorn, K. Processing of a Novel Mefenamic Acid−paracetamol−nicotinamide Cocrystal Using Gas Anti-Solvent Process. J. CO2 Util. 2022, 62, 102080. [Google Scholar] [CrossRef]
- Díaz, J.d.l.A.C.; Bordín, S.F.; Mattea, F.; Romero, M.R. Loading of Drugs in Biodegradable Polymers Using Supercritical Fluid Technology. Drug Des. Using Mach. Learn. 2022, 1, 195–224. [Google Scholar] [CrossRef]
- Tan, F.; Yang, R.; Xu, X.; Chen, X.; Wang, Y.; Ma, H.; Liu, X.; Wu, X.; Chen, Y.; Liu, L.; et al. Drug Repositioning by Applying “expression Profiles” Generated by Integrating Chemical Structure Similarity and Gene Semantic Similarity. Mol. Biosyst. 2014, 10, 1126–1138. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.H.; Huang, J.Q.; Zheng, G.F.; Lam, S.K.; Karlberg, J.; Wong, B.C.Y. Non-Steroidal Anti-Inflammatory Drug Use and the Risk of Gastric Cancer: A Systematic Review and Meta-Analysis. JNCI J. Natl. Cancer Inst. 2003, 95, 1784–1791. [Google Scholar] [CrossRef] [Green Version]
- Tkalec, G.; Pantić, M.; Novak, Z.; Knez, Ž. Supercritical Impregnation of Drugs and Supercritical Fluid Deposition of Metals into Aerogels. J. Mater. Sci. 2015, 50, 1–12. [Google Scholar] [CrossRef]
- Rivas, M.V.; Arenas Muñetón, M.J.; Spagnuolo, C.C.; Lombardo, M.V.; Bordoni, A.V.V.; Wolosiuk, A. Revisiting Carboxylic Group Functionalization of Silica Sol–gel Materials. J. Mater. Chem. B 2023, 8. [Google Scholar] [CrossRef]
- Watanabe, T.; Wakiyama, N.; Usui, F.; Ikeda, M.; Isobe, T.; Senna, M. Stability of Amorphous Indomethacin Compounded with Silica. Int. J. Pharm. 2001, 226, 81–91. [Google Scholar] [CrossRef]
- Lovskaya, D.D.; Lebedev, A.E.; Menshutina, N.V. Aerogels as Drug Delivery Systems: In Vitro and in Vivo Evaluations. J. Supercrit. Fluids 2015, 106, 115–121. [Google Scholar] [CrossRef]
- Malfait, W.J.; Zhao, S.; Verel, R.; Iswar, S.; Rentsch, D.; Fener, R.; Zhang, Y.; Milow, B.; Koebel, M.M. Surface Chemistry of Hydrophobic Silica Aerogels. Chem. Mater. 2015, 27, 6737–6745. [Google Scholar] [CrossRef]
- Rao, V.; Nilsen, E.; Einarsrud, M.A. Effect of Precursors, Methylation Agents and Solvents on the Physicochemical Properties of Silica Aerogels Prepared by Atmospheric Pressure Drying Method. J. Non. Cryst. Solids 2001, 296, 165–171. [Google Scholar] [CrossRef]
- Rao, A.P.; Rao, A.V.; Pajonk, G.M. Hydrophobic and Physical Properties of the Two Step Processed Ambient Pressure Dried Silica Aerogels with Various Exchanging Solvents. J. Sol-Gel Sci. Technol. 2005, 36, 285–292. [Google Scholar] [CrossRef]
- Lee, J.H.; Park, J.; Yang, J.; Yang, K.H.; Ju, S.; Lim, T.; Jeong, S.M. Hydrophobic Halochromic Aerogel Capable of Reversibly Measuring Acidic and Basic Vapors. AIP Adv. 2021, 11, 115115. [Google Scholar] [CrossRef]
- Kristiansen, T.; Mathisen, K.; Einarsrud, M.A.; Bjorgen, M.; Nicholson, D.G. Single-Site Copper by Incorporation in Ambient Pressure Dried Silica Aerogel and Xerogel Systems: An X-Ray Absorption Spectroscopy Study. J. Phys. Chem. C 2011, 115, 19260–19268. [Google Scholar] [CrossRef]
- Zhao, W.; Chen, H.; Li, Y.; Li, L.; Lang, M.; Shi, J. Investigation of Templated Mesoporous Silicate Thin Films Using High Speed, Solid-State 1H MAS and Double Quantum NMR Spectroscopy. Macromol. Chem. Phys. 2003, 204, 2023–2030. [Google Scholar] [CrossRef]
- Mauder, D.; Akcakayiran, D.; Lesnichin, S.B.; Findenegg, G.H.; Shenderovich, I.G. Acidity of Sulfonic and Phosphonic Acid-Functionalized SBA-15 under Almost Water-Free Conditions. J. Phys. Chem. C 2009, 113, 19185–19192. [Google Scholar] [CrossRef]
- Nourbakhsh, S.; Talebian, A.; Valipour, P.; Ghaffar Ebadi, A. Study of Corona Discharge Treatment on Physical and Chemical Properties of Cotton Fabric. Asian J. Chem. 2008, 20, 211–216. [Google Scholar]
- Ma, Z.; Dunn, B.C.; Turpin, G.C.; Eyring, E.M.; Ernst, R.D.; Pugmire, R.J. Solid State NMR Investigation of Silica Aerogel Supported Fischer–Tropsch Catalysts. Fuel Process. Technol. 2007, 88, 29–33. [Google Scholar] [CrossRef]
- Malfait, W.J.; Verel, R.; Koebel, M.M. Hydrophobization of Silica Aerogels: Insights from Quantitative Solid-State NMR Spectroscopy. J. Phys. Chem. C 2014, 118, 25545–25554. [Google Scholar] [CrossRef]
- Kim, J.; Heo, I.; Luu, Q.S.; Nguyen, Q.T.; Do, U.T.; Whiting, N.; Yang, S.H.; Huh, Y.M.; Min, S.J.; Shim, J.H.; et al. Dynamic Nuclear Polarization of Selectively 29Si-Enriched Core@shell Silica Nanoparticles. Anal. Chem. 2023, 95, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Moretto, E.; Kachouri, O.; Biagi, J.L.; Thomann, J.S.; Kayser, F.; Dieden, R. Revealing the Dehydration/deuteration Processes at the Liquid-Solid Interface by Nuclear Magnetic Resonance Spectroscopy. Colloids Surf. A Physicochem. Eng. Asp. 2022, 637, 128260. [Google Scholar] [CrossRef]
- Viitala, R.; Jokinen, M.; Maunu, S.L.; Jalonen, H.; Rosenholm, J.B. Chemical Characterization of Bioresorbable Sol–gel Derived SiO2 Matrices Prepared at Protein-Compatible pH. J. Non. Cryst. Solids 2005, 351, 3225–3234. [Google Scholar] [CrossRef]
- Rim, G.; Marchese, A.K.; Stallworth, P.; Greenbaum, S.G.; Park, A.H.A. 29Si Solid State MAS NMR Study on Leaching Behaviors and Chemical Stability of Different Mg-Silicate Structures for CO2 Sequestration. Chem. Eng. J. 2020, 396, 125204. [Google Scholar] [CrossRef]
- Moribe, K.; Kinoshita, R.; Higashi, K.; Tozuka, Y.; Yamamoto, K. Coloration Phenomenon of Mefenamic Acid in Mesoporous Silica FSM-16. Chem. Pharm. Bull. 2010, 58, 214–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, W.; Chen, H.; Li, Y.; Li, A.; Lang, M.; Shi, J. Uniform Rattle-Type Hollow Magnetic Mesoporous Spheres as Drug Delivery Carriers and Their Sustained-Release Property. Adv. Funct. Mater. 2008, 18, 2780–2788. [Google Scholar] [CrossRef]
- Gómez-Cerezo, N.; Verron, E.; Montouillout, V.; Fayon, F.; Lagadec, P.; Bouler, J.M.; Bujoli, B.; Arcos, D.; Vallet-Regí, M. The Response of Pre-Osteoblasts and Osteoclasts to Gallium Containing Mesoporous Bioactive Glasses. Acta Biomater. 2018, 76, 333–343. [Google Scholar] [CrossRef]
- Graetsch, H.; Mosset, A.; Gies, H. XRD and 29Si MAS-NMR Study on Some Non-Crystalline Silica Minerals. J. Non. Cryst. Solids 1990, 119, 173–180. [Google Scholar] [CrossRef]
- Moreno, E.M.; Zayat, M.; Morales, M.P.; Serna, C.J.; Roig, A.; Levy, D. Preparation of Narrow Size Distribution Superparamagnetic γ-Fe2O3 Nanoparticles in a Sol-Gel Transparent SiO2 Matrix. Langmuir 2002, 18, 4972–4978. [Google Scholar] [CrossRef]
- Vinogradova, E.; Estrada, M.; Moreno, A. Colloidal Aggregation Phenomena: Spatial Structuring of TEOS-Derived Silica Aerogels. J. Colloid Interface Sci. 2006, 298, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.; Wu, Z.Y.; Huang, Y.H.; Xiong, X.W.; Zhang, R.Z.; Ma, Q.H.; He, Z.; Li, R.C.; Zhang, D.Y.; Zhou, X.Y. Synthesis and Properties of P2O5-SnO2-ZnO Penetrated into Silica Aerogel Matrix by Sol-Gel Technique. Adv. Mater. Res. 2013, 800, 447–453. [Google Scholar] [CrossRef]
- Paradowska, K.; Wawer, I. Solid-State NMR in the Analysis of Drugs and Naturally Occurring Materials. J. Pharm. Biomed. Anal. 2014, 93, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Liu, J.; Wang, Y.; Shi, F.; Shan, Z.; Zhou, J.; Liu, J. Adsorption of Antibiotics from Aqueous Solution by Different Aerogels. J. Non. Cryst. Solids 2019, 505, 72–78. [Google Scholar] [CrossRef]
- Editors, G.; Hodgkinson, P.; Wimperis, S.; Ashbrook, S.E.; Zhu, J.; Geris, A.J.; Wu, G.; Chem, P.; Stebbins, J.F.; Kelsey, K.E.; et al. Anomalous Resonances in 29Si and 27Al NMR Spectra of Pyrope ([Mg,Fe]3Al2Si3O12) Garnets: Effects of Paramagnetic Cations. Phys. Chem. Chem. Phys. 2009, 11, 6906–6917. [Google Scholar] [CrossRef]
- Barron, P.F.; Frost, R.L.; Skjemstad, J.O. 29Si Spin–lattice Relaxation in Aluminosilicates. J. Chem. Soc. Chem. Commun. 1983, 581–583. [Google Scholar] [CrossRef]
- Watanabe, T.; Shimizu, H.; Masuda, A.; Saitô, H. Studies of 29Si Spin-Lattice Relaxation Times and Paramagnetic Impurities in Clay Minerals by Magic-Angle Spinning 29Si-NMR and EPR. Chem. Latters 2006, 12, 1293–1296. [Google Scholar] [CrossRef]
- Grey, C.P.; Dobson, C.M.; Cheetham, A.K.; Jakeman, R.J.B. Studies of Rare-Earth Stannates by 119Sn MAS NMR. The Use of Paramagnetic Shift Probes in the Solid State. J. Am. Chem. Soc. 1989, 111, 505–511. [Google Scholar] [CrossRef]
- Dajda, N.; Dixon, J.M.; Smith, M.E.; Carthey, N.; Bishop, P.T. Atomic Site Preferences and Structural Evolution in Vanadium-Doped ZrSiO4 from Multinuclear Solid-State NMR. Phys. Rev. B 2003, 67, 024201. [Google Scholar] [CrossRef]
- Wang, C.; Okubayashi, S. Polyethyleneimine-Crosslinked Cellulose Aerogel for Combustion CO2 Capture. Carbohydr. Polym. 2019, 225, 115248. [Google Scholar] [CrossRef]
- Alhwaige, A.A.; Ishida, H.; Qutubuddin, S. Carbon Aerogels with Excellent CO2 Adsorption Capacity Synthesized from Clay-Reinforced Biobased Chitosan-Polybenzoxazine Nanocomposites. ACS Sustain. Chem. Eng. 2016, 4, 1286–1295. [Google Scholar] [CrossRef]
- Liu, Q.; Han, Y.; Qian, X.; He, P.; Fei, Z.; Chen, X.; Zhang, Z.; Tang, J.; Cui, M.; Qiao, X. CO2 Adsorption over Carbon Aerogels: The Effect of Pore and Surface Properties. ChemistrySelect 2019, 4, 3161–3168. [Google Scholar] [CrossRef]
- He, P.; Qian, X.; Fei, Z.; Liu, Q.; Zhang, Z.; Chen, X.; Tang, J.; Cui, M.; Qiao, X. Structure Manipulation of Carbon Aerogels by Managing Solution Concentration of Precursor and Its Application for CO2 Capture. Process 2018, 6, 35. [Google Scholar] [CrossRef] [Green Version]
- Anas, M.; Gönel, A.G.; Bozbag, S.E.; Erkey, C. Thermodynamics of Adsorption of Carbon Dioxide on Various Aerogels. J. CO2 Util. 2017, 21, 82–88. [Google Scholar] [CrossRef]
- Keshavarz, L.; Ghaani, M.R.; MacElroy, J.M.D.; English, N.J. A Comprehensive Review on the Application of Aerogels in CO2-Adsorption: Materials and Characterisation. Chem. Eng. J. 2021, 412, 128604. [Google Scholar] [CrossRef]
- Rother, G.; Vlcek, L.; Gruszkiewicz, M.S.; Chialvo, A.A.; Anovitz, L.M.; Bañuelos, J.L.; Wallacher, D.; Grimm, N.; Cole, D.R. Sorption Phase of Supercritical CO2 in Silica Aerogel: Experiments and Mesoscale Computer Simulations. J. Phys. Chem. C 2014, 118, 15525–15533. [Google Scholar] [CrossRef]
- Sobornova, V.V.; Belov, K.V.; Dyshin, A.A.; Gurina, D.L.; Khodov, I.A.; Kiselev, M.G. Molecular Dynamics and Nuclear Magnetic Resonance Studies of Supercritical CO2 Sorption in Poly(Methyl Methacrylate). Polymers 2022, 14, 5332. [Google Scholar] [CrossRef]
- English, A.E.; Whittall, K.P.; Joy, M.L.G.; Henkelman, R.M. Quantitative Two-Dimensional Time Correlation Relaxometry. Magn. Reson. Med. 1991, 22, 425–434. [Google Scholar] [CrossRef]
- Venkataramanan, L.; Song, Y.Q.; Hürlimann, M.D. Solving Fredholm Integrals of the First Kind with Tensor Product Structure in 2 and 2.5 Dimensions. IEEE Trans. Signal Process. 2002, 50, 1017–1026. [Google Scholar] [CrossRef]
- Song, Y.Q.; Venkataramanan, L.; Hürlimann, M.D.; Flaum, M.; Frulla, P.; Straley, C. T1-T2 Correlation Spectra Obtained Using a Fast Two-Dimensional Laplace Inversion. J. Magn. Reson. 2002, 154, 261–268. [Google Scholar] [CrossRef]
- McDonald, P.J.; Korb, J.P.; Mitchell, J.; Monteilhet, L. Surface Relaxation and Chemical Exchange in Hydrating Cement Pastes: A Two-Dimensional NMR Relaxation Study. Phys. Rev. E—Stat. Nonlinear, Soft Matter Phys. 2005, 72, 011409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monteilhet, L.; Korb, J.P.; Mitchell, J.; McDonald, P.J. Observation of Exchange of Micropore Water in Cement Pastes by Two-Dimensional T2–T2 Nuclear Magnetic Resonance Relaxometry. Phys. Rev. E—Stat. Nonlinear, Soft Matter Phys. 2006, 74, 061404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burcaw, L.M.; Callaghan, P.T. Correlation of the Inhomogeneous Field as a Measure of Sample Heterogeneity. J. Magn. Reson. 2012, 216, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Washburn, K.E.; Callaghan, P.T. Tracking Pore to Pore Exchange Using Relaxation Exchange Spectroscopy. Phys. Rev. Lett. 2006, 97, 175502. [Google Scholar] [CrossRef]
- Khodov, I.; Dyshin, A.; Efimov, S.; Ivlev, D.; Kiselev, M. High-Pressure NMR Spectroscopy in Studies of the Conformational Composition of Small Molecules in Supercritical Carbon Dioxide. J. Mol. Liq. 2020, 309, 113113. [Google Scholar] [CrossRef] [Green Version]
- Efimov, S.V.; Dubinin, M.V.; Kobchikova, P.P.; Zgadzay, Y.O.; Khodov, I.A.; Belosludtsev, K.N.; Klochkov, V.V. Comparison of Cyclosporin Variants B–E Based on Their Structural Properties and Activity in Mitochondrial Membranes. Biochem. Biophys. Res. Commun. 2020, 526, 1054–1060. [Google Scholar] [CrossRef]
- Kobchikova, P.P.; Efimov, S.V.; Khodov, I.A.; Klochkov, V.V. Features of Spatial Structures of Cyclosporins D, E and G Revealed by NMR and MD Simulations. J. Mol. Liq. 2021, 336, 116244. [Google Scholar] [CrossRef]
- Hu, H.; Krishnamurthy, K. Revisiting the Initial Rate Approximation in Kinetic NOE Measurements. J. Magn. Reson. 2006, 182, 173–177. [Google Scholar] [CrossRef]
- Belov, K.V.; Dyshin, A.A.; Krestyaninov, M.A.; Efimov, S.V.; Khodov, I.A.; Kiselev, M.G. Conformational Preferences of Tolfenamic Acid in DMSO-CO2 Solvent System by 2D NOESY. J. Mol. Liq. 2022, 367, 120481. [Google Scholar] [CrossRef]
- Khodov, I.A.; Belov, K.V.; Dyshin, A.A.; Krestyaninov, M.A.; Kiselev, M.G. Pressure Effect on Lidocaine Conformational Equilibria in scCO2: A Study by 2D NOESY. J. Mol. Liq. 2022, 367, 120525. [Google Scholar] [CrossRef]
- Efimov, S.V.; Khodov, I.A.; Ratkova, E.L.; Kiselev, M.G.; Berger, S.; Klochkov, V.V. Detailed NOESY/T-ROESY Analysis as an Effective Method for Eliminating Spin Diffusion from 2D NOE Spectra of Small Flexible Molecules. J. Mol. Struct. 2016, 1104, 63–69. [Google Scholar] [CrossRef]
- Khodov, I.A.; Nikiforov, M.Y.; Alper, G.A.; Blokhin, D.S.; Efimov, S.V.; Klochkov, V.V.; Georgi, N. Spatial Structure of Felodipine Dissolved in DMSO by 1D NOE and 2D NOESY NMR Spectroscopy. J. Mol. Struct. 2013, 1035, 358–362. [Google Scholar] [CrossRef]
- Eventova, V.A.; Belov, K.V.; Efimov, S.V.; Khodov, I.A. Conformational Screening of Arbidol Solvates: Investigation via 2D NOESY. Pharmaceutics 2023, 15, 226. [Google Scholar] [CrossRef] [PubMed]
- Khodov, I.A.; Belov, K.V.; Krestyaninov, M.A.; Dyshin, A.A.; Kiselev, M.G.; Krestov, G.A. Investigation of the Spatial Structure of Flufenamic Acid in Supercritical Carbon Dioxide Media via 2D NOESY. Materials 2023, 16, 1524. [Google Scholar] [CrossRef]
- Belov, K.V.V.; Dyshin, A.A.A.; Kiselev, M.G.G.; Krestyaninov, M.A.A.; Sobornova, V.V.; Khodov, I.A.A. Determination of the Spatial Structure of Lidocaine in SC-CO2 by the 2D NOESY Method. Russ. J. Phys. Chem. B 2021, 15, 1303–1309. [Google Scholar] [CrossRef]
- Khodov, I.A.; Belov, K.V.; Krestyaninov, M.A.; Kiselev, M.G. Conformational Equilibria of a Thiadiazole Derivative in Solvents of Different Polarities: An NMR Study. Russ. J. Phys. Chem. A 2022, 96, 765–772. [Google Scholar] [CrossRef]
- Belov, K.V.; Batista de Carvalho, L.A.E.; Dyshin, A.A.; Efimov, S.V.; Khodov, I.A. The Role of Hidden Conformers in Determination of Conformational Preferences of Mefenamic Acid by NOESY Spectroscopy. Pharmaceutics 2022, 14, 2276. [Google Scholar] [CrossRef]
- SeethaLekshmi, S.; Row, T.N.G. Conformational Polymorphism in a Non-Steroidal Anti-Inflammatory Drug, Mefenamic Acid. Cryst. Growth Des. 2012, 12, 4283–4289. [Google Scholar] [CrossRef]
- Khodov, I.A.; Belov, K.V.; Pogonin, A.E.; Savenkova, M.A.; Gamov, G.A. Spatial Structure and Conformations of Hydrazones Derived from Pyridoxal 5′-Phosphate and 2-, 3-Pyridinecarbohydrazide in the Light of NMR Study and Quantum Chemical Calculations. J. Mol. Liq. 2021, 342, 117372. [Google Scholar] [CrossRef]
- Khodov, I.A.; Kiselev, M.G.; Efimov, S.V.; Klochkov, V.V. Comment On “conformational Analysis of Small Organic Molecules Using NOE and RDC Data: A Discussion of Strychnine and α-Methylene-γ-Butyrolactone”. J. Magn. Reson. 2016, 266, 67–68. [Google Scholar] [CrossRef]
- Sternberg, U.; Witter, R. Structures Controlled by Entropy: The Flexibility of Strychnine as Example. Molecules 2022, 27, 7987. [Google Scholar] [CrossRef] [PubMed]
- Belov, K.V.; Batista de Carvalho, L.A.E.; Dyshin, A.A.; Kiselev, M.G.; Sobornova, V.V.; Khodov, I.A. Conformational Analysis of Mefenamic Acid in scCO2-DMSO by the 2D NOESY Method. Russ. J. Phys. Chem. B 2023, 16, 1191–1199. [Google Scholar] [CrossRef]
- Khodov, I.A.; Belov, K.V.; Krestyaninov, M.A.; Sobornova, V.V.; Dyshin, A.A.; Kiselev, M.G. Does DMSO Affect the Conformational Changes of Drug Molecules in Supercritical CO2 Media? J. Mol. Liq. 2023; in press. [Google Scholar]
- Khodov, I.A.; Musabirova, G.S.; Klochkov, V.V.; Karataeva, F.K.; Huster, D.; Scheidt, H.A. Structural Details on the Interaction of Fenamates with Lipid Membranes. J. Mol. Liq. 2022, 367, 120502. [Google Scholar] [CrossRef]
- Andreatta, A.E.; Florusse, L.J.; Bottini, S.B.; Peters, C.J. Phase Equilibria of Dimethyl Sulfoxide (DMSO) + Carbon Dioxide, and DMSO + Carbon Dioxide + Water Mixtures. J. Supercrit. Fluids 2007, 42, 60–68. [Google Scholar] [CrossRef]
- Danilyuk, A.F.; Kravchenko, E.A.; Okunev, A.G.; Onuchin, A.P.; Shaurman, S.A. Synthesis of Aerogel Tiles with High Light Scattering Length. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrom. Detect. Assoc. Equip. 1999, 433, 406–407. [Google Scholar] [CrossRef]
- Danilyuk, A.F.; Kononov, S.A.; Kravchenko, E.A.; Onuchin, A.P. Aerogel Cherenkov Detectors in Colliding Beam Experiments. Physics-Uspekhi 2015, 58, 503. [Google Scholar] [CrossRef]
- Oparin, R.D.; Belov, K.V.; Khodov, I.A.; Dyshin, A.A.; Kiselev, M.G. Impregnation of Polymethyl Methacrylate with Carbamazepine in Supercritical Carbon Dioxide. Russ. J. Phys. Chem. B 2021, 15, 1157–1165. [Google Scholar] [CrossRef]
- Mandal, C.; Donthula, S.; Rewatkar, P.M.; Sotiriou-Leventis, C.; Leventis, N. Experimental Deconvolution of Depressurization from Capillary Shrinkage during Drying of Silica Wet-Gels with SCF CO2 Why Aerogels Shrink? J. Sol-Gel Sci. Technol. 2019, 92, 662–680. [Google Scholar] [CrossRef]
- Chen, Y.X.; Sepahvand, S.; Gauvin, F.; Schollbach, K.; Brouwers, H.J.H.; Yu, Q. One-Pot Synthesis of Monolithic Silica-Cellulose Aerogel Applying a Sustainable Sodium Silicate Precursor. Constr. Build. Mater. 2021, 293, 123289. [Google Scholar] [CrossRef]
- Shenderovich, I.G.; Mauder, D.; Akcakayiran, D.; Buntkowsky, G.; Limbach, H.H.; Findenegg, G.H. NMR Provides Checklist of Generic Properties for Atomic-Scale Models of Periodic Mesoporous Silicas. J. Phys. Chem. B 2007, 111, 12088–12096. [Google Scholar] [CrossRef]
- Gutiérrez-Ortega, J.A.; Gómez-Salazar, S.; Shenderovich, I.G.; Manríquez-González, R. Efficiency and Lead Uptake Mechanism of a Phosphonate Functionalized Mesoporous Silica through P/Pb Association Ratio. Mater. Chem. Phys. 2020, 239, 122037. [Google Scholar] [CrossRef]
- MacKenzie, K.J.D.; Smith, M.E. Multinuclear Solid-State NMR of Inorganic Materials. PERGAMON Mater. Ser. 2002, 6, 748. [Google Scholar]
- Shieh, Y.-T.; Liu, K.-H. Solubility of CO2 in Glassy PMMA and PS over a Wide Pressure Range: The Effect of Carbonyl Groups. J. Polym. Res. 2002, 9, 107–113. [Google Scholar] [CrossRef]
- Sauceau, M.; Letourneau, J.-J.; Richon, D.; Fages, J. Enhanced Density-Based Models for Solid Compound Solubilities in Supercritical Carbon Dioxide with Cosolvents. Fluid Phase Equilib. 2003, 208, 99–113. [Google Scholar] [CrossRef] [Green Version]
- Jaxel, J.; Gusenbauer, C.; Böhmdorfer, S.; Liebner, F.; Hansmann, C. Improving Single-Step scCO2 Dyeing of Wood by DMSO-Induced Micro-Swelling. J. Supercrit. Fluids 2020, 165, 104978. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khodov, I.; Sobornova, V.; Mulloyarova, V.; Belov, K.; Dyshin, A.; de Carvalho, L.B.; Tolstoy, P.; Kiselev, M. Exploring the Conformational Equilibrium of Mefenamic Acid Released from Silica Aerogels via NMR Analysis. Int. J. Mol. Sci. 2023, 24, 6882. https://doi.org/10.3390/ijms24086882
Khodov I, Sobornova V, Mulloyarova V, Belov K, Dyshin A, de Carvalho LB, Tolstoy P, Kiselev M. Exploring the Conformational Equilibrium of Mefenamic Acid Released from Silica Aerogels via NMR Analysis. International Journal of Molecular Sciences. 2023; 24(8):6882. https://doi.org/10.3390/ijms24086882
Chicago/Turabian StyleKhodov, Ilya, Valentina Sobornova, Valeriya Mulloyarova, Konstantin Belov, Alexey Dyshin, Luís Batista de Carvalho, Peter Tolstoy, and Michael Kiselev. 2023. "Exploring the Conformational Equilibrium of Mefenamic Acid Released from Silica Aerogels via NMR Analysis" International Journal of Molecular Sciences 24, no. 8: 6882. https://doi.org/10.3390/ijms24086882










