Lovastatin, an Up-Regulator of Low-Density Lipoprotein Receptor, Enhances Follicular Development in Mouse Ovaries
Abstract
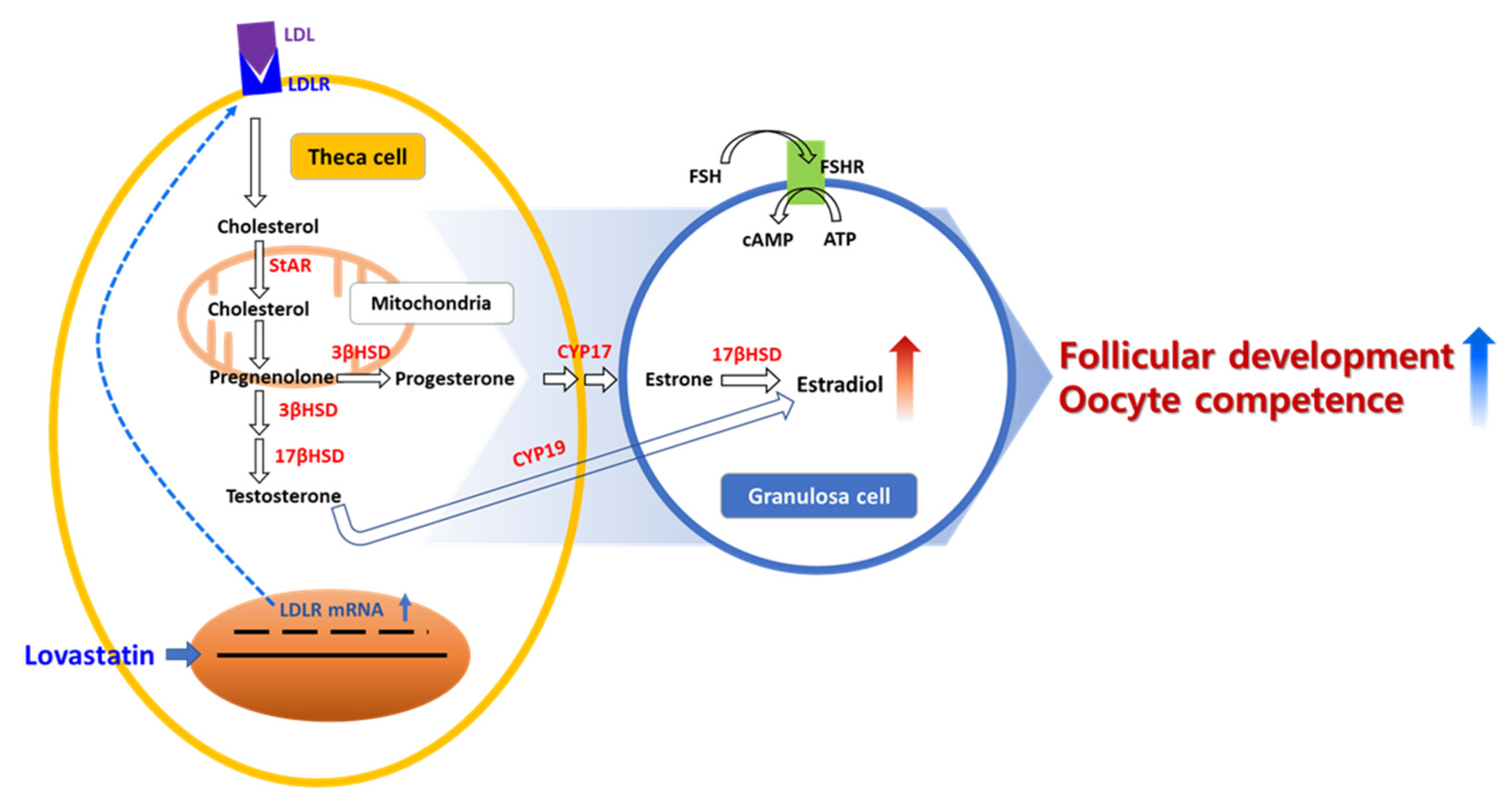
1. Introduction
2. Results
2.1. Viability of Lovastatin-Treated Cells
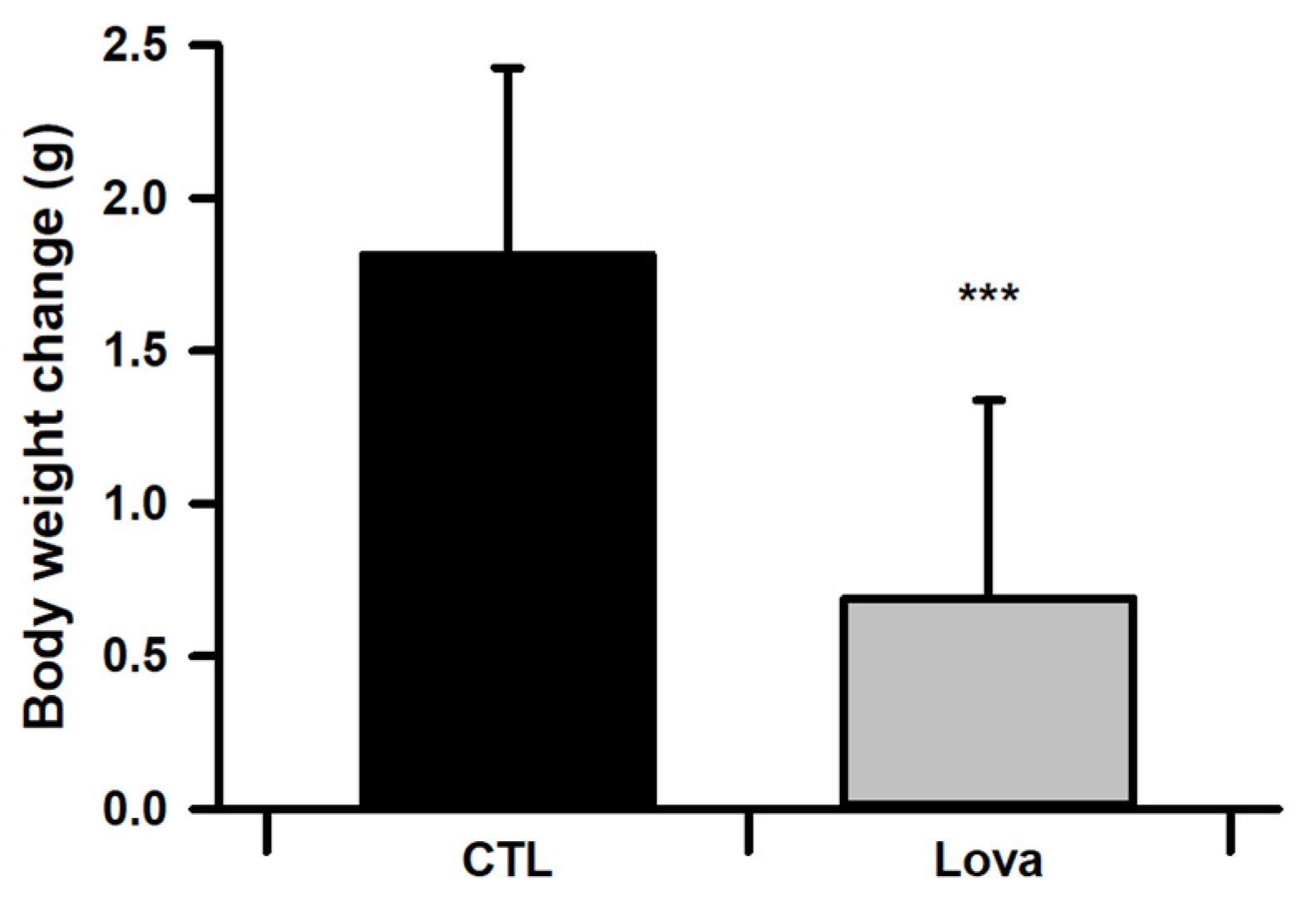
2.2. Body Weight Changes of Lovastatin-Treated Mice
2.3. Lovastatin Treatment Enhances Follicular Development in Hormone-Stimulated Mouse Ovaries
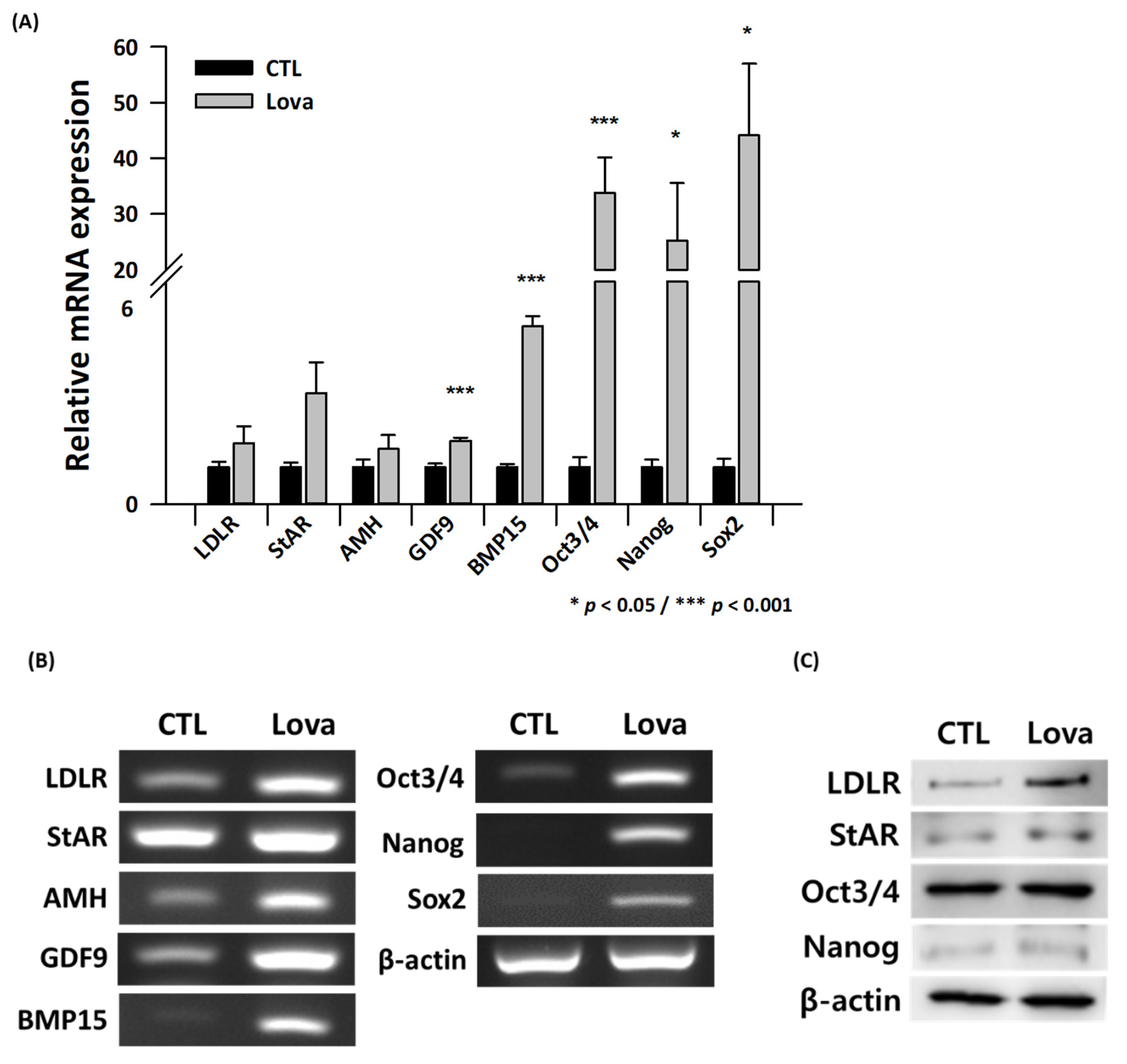
2.4. Expression of Primordial Oogenesis Factors and Ovarian Function Markers Is Increased in Lovastatin-Treated Mouse Ovaries
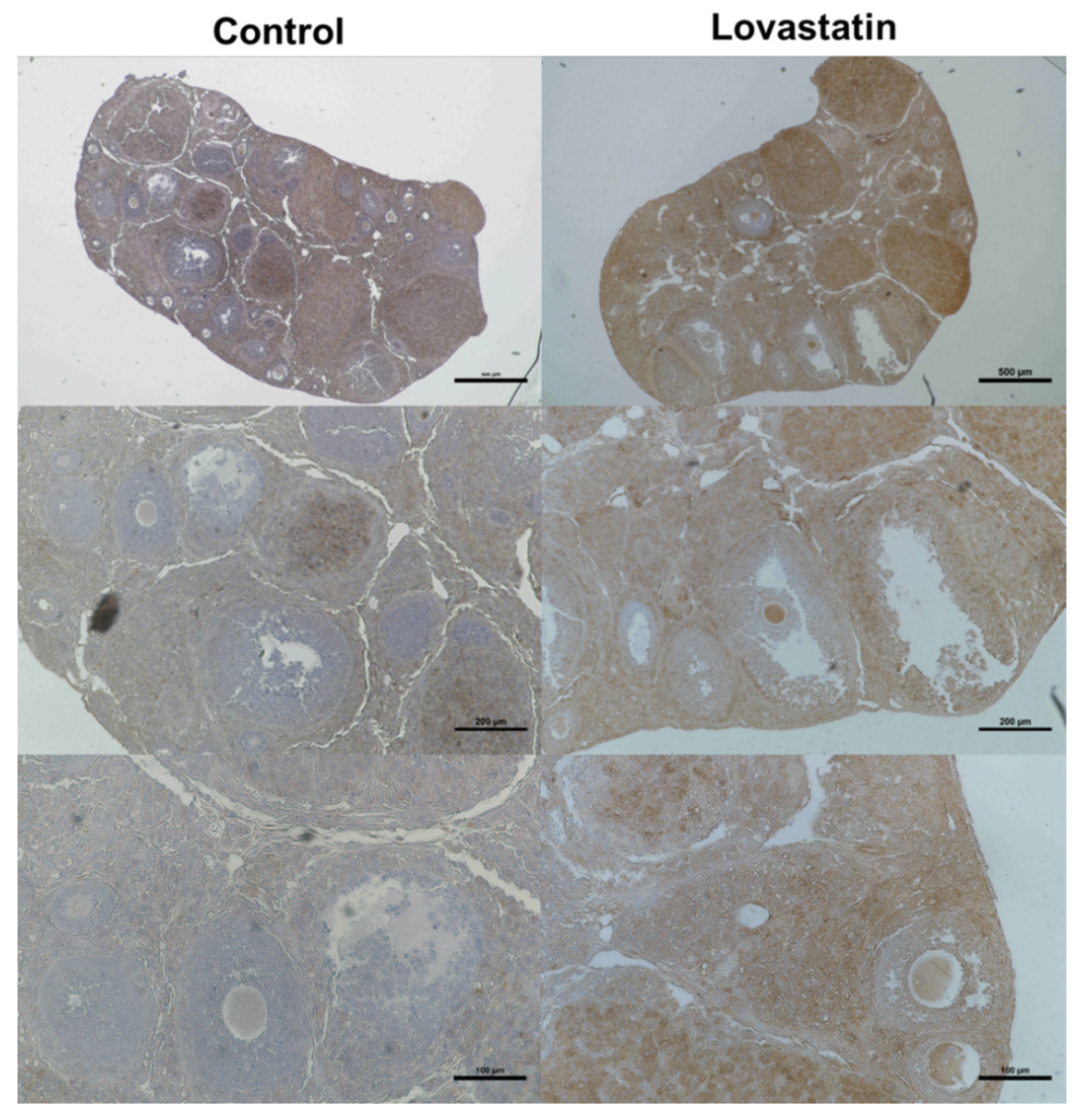
2.5. Immunohistological Analysis Using an Anti-LDLR Antibody
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Animals
4.3. Determination of the Effect of Lovastatin on Ovaries In Vivo
4.4. Histological Analysis and Immunohistochemistry
4.5. Gene Expression Analysis
4.6. Quantitative Real-Time PCR
4.7. Protein Expression Analysis
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Aflatoonian, A.; Eftekhar, M.; Mohammadian, F.; Yousefnejad, F. Outcome of assisted reproductive technology in women aged 40 years and older. Iran. J. Reprod. Med. 2011, 9, 281–284. [Google Scholar] [PubMed]
- Amini, P.; Ramezanali, F.; Parchehbaf-Kashani, M.; Maroufizadeh, S.; Omani-Samani, R.; Ghaheri, A. Factors Associated with In Vitro Fertilization Live Birth Outcome: A Comparison of Different Classification Methods. Int. J. Fertil. Steril. 2021, 15, 128–134. [Google Scholar] [CrossRef]
- Igarashi, H.; Takahashi, T.; Nagase, S. Oocyte aging underlies female reproductive aging: Biological mechanisms and therapeutic strategies. Reprod. Med. Biol. 2015, 14, 159–169. [Google Scholar] [CrossRef]
- Al-Edani, T.; Assou, S.; Ferrieres, A.; Bringer Deutsch, S.; Gala, A.; Lecellier, C.H.; Ait-Ahmed, O.; Hamamah, S. Female aging alters expression of human cumulus cells genes that are essential for oocyte quality. Biomed Res. Int. 2014, 2014, 964614. [Google Scholar] [CrossRef] [PubMed]
- Eichenlaub-Ritter, U. Oocyte ageing and its cellular basis. Int. J. Dev. Biol. 2012, 56, 841–852. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Zhang, J.; Li, H.; Wang, T.Y.; Zhang, C.X.; Luo, M.J.; Tan, J.H. Cumulus cells accelerate oocyte aging by releasing soluble Fas ligand in mice. Sci. Rep. 2015, 5, 8683. [Google Scholar] [CrossRef]
- Cimadomo, D.; Fabozzi, G.; Vaiarelli, A.; Ubaldi, N.; Ubaldi, F.M.; Rienzi, L. Impact of Maternal Age on Oocyte and Embryo Competence. Front. Endocrinol. 2018, 9, 327. [Google Scholar] [CrossRef]
- Sanchez, F.; Smitz, J. Molecular control of oogenesis. Biochim. Biophys. Acta 2012, 1822, 1896–1912. [Google Scholar] [CrossRef]
- Jagarlamudi, K.; Rajkovic, A. Oogenesis: Transcriptional regulators and mouse models. Mol. Cell. Endocrinol. 2012, 356, 31–39. [Google Scholar] [CrossRef]
- McReynolds, S.; Dzieciatkowska, M.; McCallie, B.R.; Mitchell, S.D.; Stevens, J.; Hansen, K.; Schoolcraft, W.B.; Katz-Jaffe, M.G. Impact of maternal aging on the molecular signature of human cumulus cells. Fertil. Steril. 2012, 98, 1574–1580.e1575. [Google Scholar] [CrossRef]
- Grummer, R.R.; Carroll, D.J. A review of lipoprotein cholesterol metabolism: Importance to ovarian function. J. Anim. Sci. 1988, 66, 3160–3173. [Google Scholar] [CrossRef]
- Hansen, K.K.; Knopp, R.H.; Soules, M.R. Lipoprotein-cholesterol levels in infertile women with luteal phase deficiency ** This research was supported by grants R01 HD18967 (M.R.S.) and DK-35816 (Clinical Nutrition Research Unit) (R.H.K.), from the National Institute of Health, Bethesda, Maryland. Fertil. Steril. 1991, 55, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Kim, Y.S.; Kim, Y.J.; Lee, H.R.; Choi, K.H.; Park, E.A.; Kang, K.Y.; Yoon, T.K.; Hwang, S.; Ko, J.J.; et al. Upregulation of Low-Density Lipoprotein Receptor of the Steroidogenesis Pathway in the Cumulus Cells is Associated with the Maturation of Oocytes and Achievement of Pregnancy. Cells 2021, 10, 2389. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Ito, Y.; Mizuta, H.; Massaki, K.; Hiramatsu, N.; Todo, T.; Reading, B.J.; Sullivan, C.V.; Hara, A. Molecular cloning and partial characterization of an ovarian receptor with seven ligand binding repeats, an orthologue of low-density lipoprotein receptor, in the cutthroat trout (Oncorhynchus clarki). Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2013, 166, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, T.; Fujimoto, S. The potential function of steroid sulphatase activity in steroid production and steroidogenic acute regulatory protein expression. Biochem. J. 2004, 380, 153–160. [Google Scholar] [CrossRef]
- Kehler, J.; Tolkunova, E.; Koschorz, B.; Pesce, M.; Gentile, L.; Boiani, M.; Lomeli, H.; Nagy, A.; McLaughlin, K.J.; Scholer, H.R.; et al. Oct4 is required for primordial germ cell survival. EMBO Rep. 2004, 5, 1078–1083. [Google Scholar] [CrossRef]
- Iwase, A.; Osuka, S.; Goto, M.; Murase, T.; Nakamura, T.; Takikawa, S.; Kikkawa, F. Clinical application of serum anti-Mullerian hormone as an ovarian reserve marker: A review of recent studies. J. Obstet. Gynaecol. Res. 2018, 44, 998–1006. [Google Scholar] [CrossRef]
- Su, Y.Q.; Sugiura, K.; Wigglesworth, K.; O’Brien, M.J.; Affourtit, J.P.; Pangas, S.A.; Matzuk, M.M.; Eppig, J.J. Oocyte regulation of metabolic cooperativity between mouse cumulus cells and oocytes: BMP15 and GDF9 control cholesterol biosynthesis in cumulus cells. Development 2008, 135, 111–121. [Google Scholar] [CrossRef]
- Sreerangaraja Urs, D.B.; Wu, W.H.; Komrskova, K.; Postlerova, P.; Lin, Y.F.; Tzeng, C.R.; Kao, S.H. Mitochondrial Function in Modulating Human Granulosa Cell Steroidogenesis and Female Fertility. Int. J. Mol. Sci. 2020, 21, 3592. [Google Scholar] [CrossRef]
- Regan, S.L.P.; Knight, P.G.; Yovich, J.L.; Leung, Y.; Arfuso, F.; Dharmarajan, A. Granulosa Cell Apoptosis in the Ovarian Follicle-A Changing View. Front. Endocrinol. 2018, 9, 61. [Google Scholar] [CrossRef]
- Kuokkanen, S.; Polotsky, A.J.; Chosich, J.; Bradford, A.P.; Jasinska, A.; Phang, T.; Santoro, N.; Appt, S.E. Corpus luteum as a novel target of weight changes that contribute to impaired female reproductive physiology and function. Syst. Biol. Reprod. Med. 2016, 62, 227–242. [Google Scholar] [CrossRef] [PubMed]
- Kordus, R.J.; LaVoie, H.A. Granulosa cell biomarkers to predict pregnancy in ART: Pieces to solve the puzzle. Reproduction 2017, 153, R69–R83. [Google Scholar] [CrossRef] [PubMed]
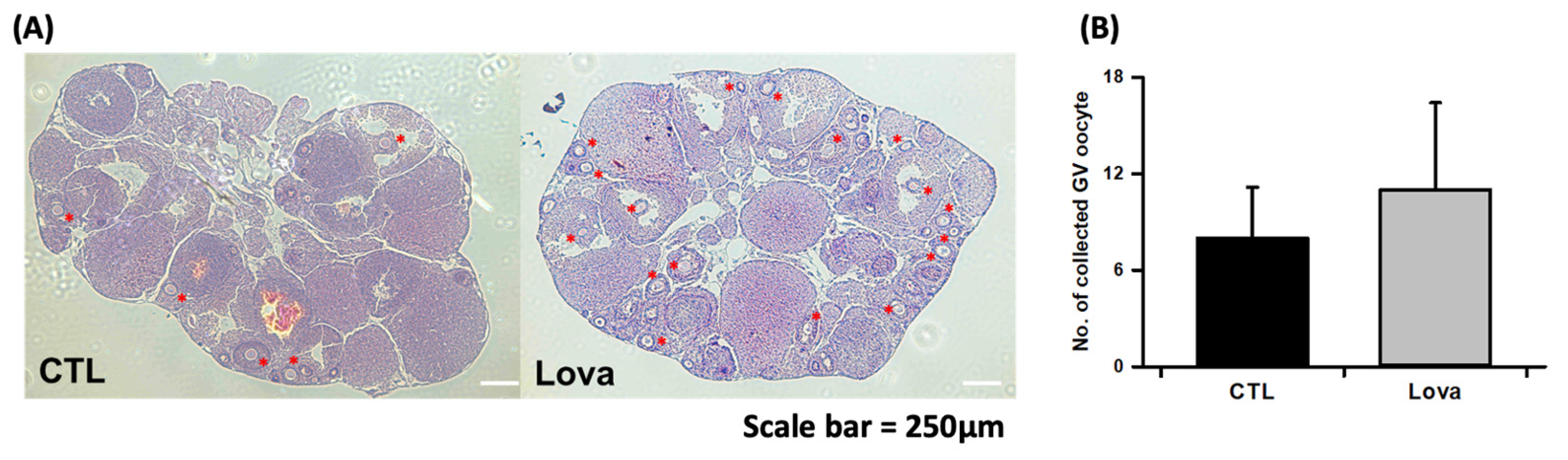
| Total Collected GV Oocytes | Total Collected Degradation GV | GVBD (%) | MII (%) | Not Mature (%) | |
|---|---|---|---|---|---|
| CTL | 32 | 27 | 14 (44%) | 13 (41%) | 5 (16%) |
| Lova | 44 | 21 | 12 (27%) | 24 (55%) | 7 (16%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.J.; Cho, Y.I.; Jang, J.; Koo, Y.D.; Park, S.W.; Lee, J.H. Lovastatin, an Up-Regulator of Low-Density Lipoprotein Receptor, Enhances Follicular Development in Mouse Ovaries. Int. J. Mol. Sci. 2023, 24, 7263. https://doi.org/10.3390/ijms24087263
Kim YJ, Cho YI, Jang J, Koo YD, Park SW, Lee JH. Lovastatin, an Up-Regulator of Low-Density Lipoprotein Receptor, Enhances Follicular Development in Mouse Ovaries. International Journal of Molecular Sciences. 2023; 24(8):7263. https://doi.org/10.3390/ijms24087263
Chicago/Turabian StyleKim, Yu Jin, Yong Il Cho, JuYi Jang, Yun Dong Koo, Sung Woon Park, and Jae Ho Lee. 2023. "Lovastatin, an Up-Regulator of Low-Density Lipoprotein Receptor, Enhances Follicular Development in Mouse Ovaries" International Journal of Molecular Sciences 24, no. 8: 7263. https://doi.org/10.3390/ijms24087263
APA StyleKim, Y. J., Cho, Y. I., Jang, J., Koo, Y. D., Park, S. W., & Lee, J. H. (2023). Lovastatin, an Up-Regulator of Low-Density Lipoprotein Receptor, Enhances Follicular Development in Mouse Ovaries. International Journal of Molecular Sciences, 24(8), 7263. https://doi.org/10.3390/ijms24087263







