T-Cells Subsets in Castleman Disease: Analysis of 28 Cases Including Unicentric, Multicentric and HHV8-Related Clinical Forms
Abstract
:1. Introduction
2. Results
2.1. Clinical Features
2.1.1. UCD
2.1.2. MCD
iMCD
HHV8+/HIV− MCD
HHV8+/HIV+ MCD
2.2. Histopathological Features
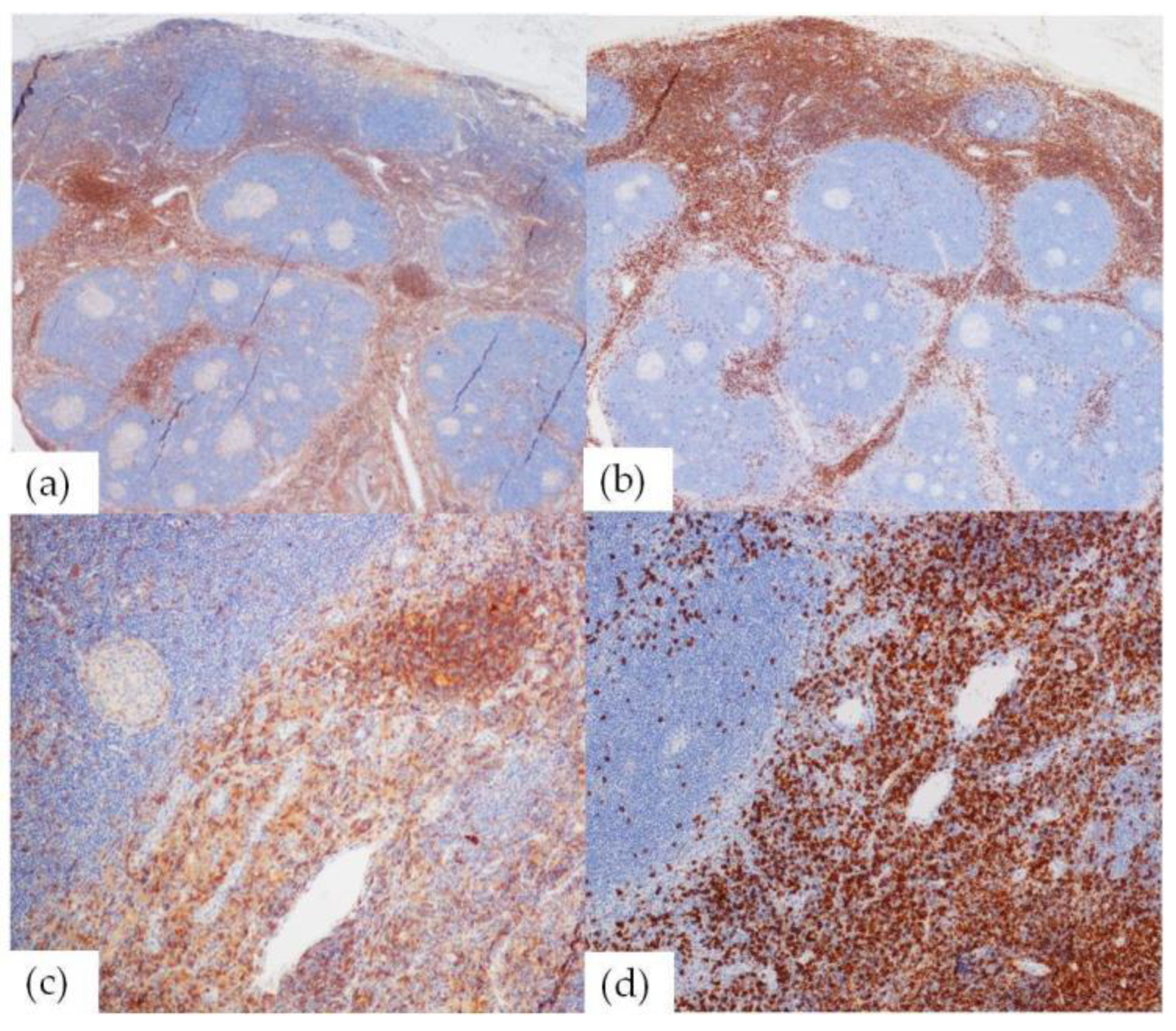
2.3. T-Cell Subset Analysis
2.3.1. CD4/CD8 Analysis
2.3.2. T-reg Analysis
3. Discussion
4. Materials and Methods
4.1. Patients Selection
4.2. Pathological Methods
4.3. T-Cell Subset Analysis
4.4. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A. Histopathological Features
| Case | UCD/iMCD | Histologic Subtype | Regressed GC | FDC Prominence | Vascularity | Hyperplastic GC | Plasmacytosis |
|---|---|---|---|---|---|---|---|
| 1 | UCD | HV | 3 | 3 | 2 | 1 | 0 |
| 2 | UCD | HV | 3 | 1 | 2 | 0 | 0 |
| 4 | UCD | mixed | 2 | 1 | 1 | 2 | 2 |
| 5 | UCD | HV | 2 | 2 | 1 | 2 | 0 |
| 6 | UCD | HV | 2 | 2 | 1 | 1 | 0 |
| 7 | iMCD | mixed | 2 | 1 | 2 | 1 | 2 |
| 9 | UCD | HV | 2 | 2 | 2 | 1 | 0 |
| 10 | UCD | HV | 3 | 2 | 0 | 0 | 0 |
| 11 | UCD | mixed | 1 | 3 | 0 | 3 | 2 |
| 13 | iMCD | plasma cell-type | 2 | 2 | 1 | 1 | 3 |
| 14 | iMCD | HyperV | 2 | 2 | 1 | 1 | 0 |
| 16 | iMCD | mixed | 2 | 2 | 1 | 2 | 2 |
| 18 | iMCD | mixed | 3 | 2 | 1 | 1 | 2 |
| 19 | UCD | HV | 2 | 3 | 1 | 2 | 0 |
| 22 | iMCD | mixed | 3 | 2 | 2 | 1 | 2 |
| 24 | iMCD | HyperV | 3 | 0 | 1 | 0 | 1 |
| 25 | iMCD | mixed | 2 | 1 | 2 | 1 | 3 |
| 26 | UCD | HV | 2 | 2 | 1 | 0 | 0 |
| 28 | UCD | HV | 2 | 3 | 3 | 1 | 2 |
| 30 | iMCD | plasma cell-type | 1 | 2 | 2 | 1 | 3 |
| 32 | UCD | HV | 2 | 2 | 3 | 2 | 1 |
| 33 | UCD | HV | 2 | 1 | 1 | 0 | 0 |


References
- Alaggio, R.; Amador, C.; Anagnostopoulos, I.; Attygalle, A.D.; Araujo, I.B.D.O.; Berti, E.; Bhagat, G.; Borges, A.M.; Boyer, D.; Calaminici, M.; et al. The 5th Edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia 2022, 36, 1720–1748. [Google Scholar] [CrossRef] [PubMed]
- Dispenzieri, A.; Fajgenbaum, D.C. Overview of Castleman Disease. Blood 2020, 135, 1353–1364. [Google Scholar] [CrossRef]
- Oksenhendler, E.; Boutboul, D.; Fajgenbaum, D.; Mirouse, A.; Fieschi, C.; Malphettes, M.; Vercellino, L.; Meignin, V.; Gérard, L.; Galicier, L. The Full Spectrum of Castleman Disease: 273 Patients Studied over 20 Years. Br. J. Haematol. 2018, 180, 206–216. [Google Scholar] [CrossRef]
- Pierson, S.K.; Kanhai, K.; Bagg, A.; Alapat, D.; Lim, M.S.; Lechowicz, M.J.; Srkalovic, G.; Uldrick, T.S.; van Rhee, F.; Fajgenbaum, D.C. Characterizing Mortality Associated with Idiopathic Multicentric Castleman Disease. Blood 2021, 138, 1623. [Google Scholar] [CrossRef]
- Fajgenbaum, D.C.; Uldrick, T.S.; Bagg, A.; Frank, D.; Wu, D.; Srkalovic, G.; Simpson, D.; Liu, A.Y.; Menke, D.; Chandrakasan, S.; et al. International, Evidence-Based Consensus Diagnostic Criteria for HHV-8–Negative/Idiopathic Multicentric Castleman Disease. Blood 2017, 129, 1646–1657. [Google Scholar] [CrossRef]
- van Rhee, F.; Oksenhendler, E.; Srkalovic, G.; Voorhees, P.; Lim, M.; Dispenzieri, A.; Ide, M.; Parente, S.; Schey, S.; Streetly, M.; et al. International Evidence-Based Consensus Diagnostic and Treatment Guidelines for Unicentric Castleman Disease. Blood Adv. 2020, 4, 6039–6050. [Google Scholar] [CrossRef]
- Keller, A.R.; Hochholzer, L.; Castleman, B. Hyaline-Vascular and Plasma-Cell Types of Giant Lymph Node Hyperplasia of the Mediastinum and Other Locations. Cancer 1972, 29, 670–683. [Google Scholar] [CrossRef] [PubMed]
- Medina, E.A.; Fuehrer, N.E.; Miller, F.R.; Kinney, M.C.; Higgins, R.A. Dysplastic Follicular Dendritic Cells in Hyaline-Vascular Castleman Disease: A Rare Occurrence Creating Diagnostic Difficulty. Pathol. Int. 2016, 66, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Lim, M.S.; Jaffe, E.S. Pathology of Castleman Disease. Hematol. Oncol. Clin. N. Am. 2018, 32, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, M.F.; Nishimura, Y.; Nishikori, A.; Yoshino, T.; Sato, Y. Historical and Pathological Overview of Castleman Disease. J. Clin. Exp. Hematop. 2022, 62, 60–72. [Google Scholar] [CrossRef]
- Carbone, A.; Borok, M.; Damania, B.; Gloghini, A.; Polizzotto, M.N.; Jayanthan, R.K.; Fajgenbaum, D.C.; Bower, M. Castleman Disease. Nat. Rev. Dis. Primer 2021, 7, 84. [Google Scholar] [CrossRef]
- Dupin, N.; Diss, T.L.; Kellam, P.; Tulliez, M.; Du, M.-Q.; Sicard, D.; Weiss, R.A.; Isaacson, P.G.; Boshoff, C. HHV-8 Is Associated with a Plasmablastic Variant of Castleman Disease That Is Linked to HHV-8–Positive Plasmablastic Lymphoma. Blood 2000, 95, 1406–1412. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, C.; Hentrich, M.; Tiemann, M.; Rosenwald, A.; Weber, F.; Willenbacher, W.; Hübel, K. Recent Advances in Castleman Disease. Oncol. Res. Treat. 2022, 45, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Wing, A.; Xu, J.; Meng, W.; Rosenfeld, A.M.; Li, E.Y.; Wertheim, G.; Paessler, M.; Bagg, A.; Frank, D.; Tan, K.; et al. Transcriptome and Unique Cytokine Microenvironment of Castleman Disease. Mod. Pathol. 2022, 35, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Horna, P.; King, R.L.; Jevremovic, D.; Fajgenbaum, D.C.; Dispenzieri, A. The Lymph Node Transcriptome of Unicentric and Idiopathic Multicentric Castleman Disease. Haematologica 2023, 108, 207–218. [Google Scholar] [CrossRef]
- Shilling, D.; Stadanlick, J.E.; Meng, W.; Rao, A.; Krymskaya, M.V.P.; Prak, E.T.L.; Eruslanov, E.B.; Fajgenbaum, M.D.C. Oligoclonal Expansion of Cd8+ T Cells during Idiopathic Multicentric Castleman Disease Flares Suggests an Antigen Driven Process. Blood 2018, 132, 2411. [Google Scholar] [CrossRef]
- Fajgenbaum, D.C.; Langan, R.-A.; Japp, A.S.; Partridge, H.L.; Pierson, S.K.; Singh, A.; Arenas, D.J.; Ruth, J.R.; Nabel, C.S.; Stone, K.; et al. Identifying and Targeting Pathogenic PI3K/AKT/MTOR Signaling in IL-6-Blockade-Refractory Idiopathic Multicentric Castleman Disease. J. Clin. Investig. 2019, 129, 4451–4463. [Google Scholar] [CrossRef] [PubMed]
- Kurose, N.; Guo, X.; Shioya, A.; Mizutani, K.-I.; Kumagai, M.; Fujimoto, S.; Kawabata, H.; Masaki, Y.; Takai, K.; Aoki, S.; et al. The Potential Role of Follicular Helper T Cells in Idiopathic Multicentric Castleman Disease with and without TAFRO Syndrome. Pathol. Res. Pract. 2019, 215, 152563. [Google Scholar] [CrossRef]
- Zou, W. Regulatory T Cells, Tumour Immunity and Immunotherapy. Nat. Rev. Immunol. 2006, 6, 295–307. [Google Scholar] [CrossRef]
- Maharaj, K.; Uriepero, A.; Sahakian, E.; Pinilla-Ibarz, J. Regulatory T Cells (Tregs) in Lymphoid Malignancies and the Impact of Novel Therapies. Front. Immunol. 2022, 13, 943354. [Google Scholar] [CrossRef]
- Takeuchi, K. Idiopathic Plasmacytic Lymphadenopathy: A Conceptual History along with a Translation of the Original Japanese Article Published in 1980. J. Clin. Exp. Hematop. 2022, 62, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Umehara, H.; Okazaki, K.; Kawa, S.; Takahashi, H.; Goto, H.; Matsui, S.; Ishizaka, N.; Akamizu, T.; Sato, Y.; Kawano, M.; et al. The 2020 Revised Comprehensive Diagnostic (RCD) Criteria for IgG4-RD. Mod. Rheumatol. 2021, 31, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Nishikori, A.; Nishimura, M.F.; Nishimura, Y.; Notohara, K.; Satou, A.; Moriyama, M.; Nakamura, S.; Sato, Y. Investigation of IgG4-Positive Cells in Idiopathic Multicentric Castleman Disease and Validation of the 2020 Exclusion Criteria for IgG4-Related Disease. Pathol. Int. 2022, 72, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Satou, A.; Notohara, K.; Zen, Y.; Nakamura, S.; Yoshino, T.; Okazaki, K.; Sato, Y. Clinicopathological Differential Diagnosis of IgG4-Related Disease: A Historical Overview and a Proposal of the Criteria for Excluding Mimickers of IgG4-Related Disease. Pathol. Int. 2020, 70, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Yoshizaki, K.; Murayama, S.; Ito, H.; Koga, T. The Role of Interleukin-6 in Castleman Disease. Hematol. Oncol. Clin. N. Am. 2018, 32, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Fajgenbaum, D.C.; Shilling, D. Castleman Disease Pathogenesis. Hematol. Oncol. Clin. N. Am. 2018, 32, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Pierson, S.K.; Stonestrom, A.J.; Shilling, D.; Ruth, J.; Nabel, C.S.; Singh, A.; Ren, Y.; Stone, K.; Li, H.; van Rhee, F.; et al. Plasma Proteomics Identifies a “chemokine Storm” in Idiopathic Multicentric Castleman Disease. Am. J. Hematol. 2018, 93, 902–912. [Google Scholar] [CrossRef]
- Yang, R.; Masters, A.R.; Fortner, K.A.; Champagne, D.P.; Yanguas-Casás, N.; Silberger, D.J.; Weaver, C.T.; Haynes, L.; Rincon, M. IL-6 Promotes the Differentiation of a Subset of Naive CD8+ T Cells into IL-21–Producing B Helper CD8+ T Cells. J. Exp. Med. 2016, 213, 2281–2291. [Google Scholar] [CrossRef]
- St. Paul, M.; Saibil, S.D.; Lien, S.C.; Han, S.; Sayad, A.; Mulder, D.T.; Garcia-Batres, C.R.; Elford, A.R.; Israni-Winger, K.; Robert-Tissot, C.; et al. IL6 Induces an IL22+ CD8+ T-Cell Subset with Potent Antitumor Function. Cancer Immunol. Res. 2020, 8, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Fisher, D.T.; Appenheimer, M.M.; Evans, S.S. The Two Faces of IL-6 in the Tumor Microenvironment. Semin. Immunol. 2014, 26, 38–47. [Google Scholar] [CrossRef]
- Li, Z.; Li, D.; Tsun, A.; Li, B. FOXP3+ Regulatory T Cells and Their Functional Regulation. Cell. Mol. Immunol. 2015, 12, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.-Z.; Novak, A.J.; Ziesmer, S.C.; Witzig, T.E.; Ansell, S.M. Attenuation of CD8+ T-Cell Function by CD4+CD25+ Regulatory T Cells in B-Cell Non-Hodgkin’s Lymphoma. Cancer Res. 2006, 66, 10145–10152. [Google Scholar] [CrossRef]
- Barnett, J.C.; Bean, S.M.; Whitaker, R.S.; Kondoh, E.; Baba, T.; Fujii, S.; Marks, J.R.; Dressman, H.K.; Murphy, S.K.; Berchuck, A. Ovarian Cancer Tumor Infiltrating T-Regulatory (T(Reg)) Cells Are Associated with a Metastatic Phenotype. Gynecol. Oncol. 2010, 116, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Viguier, M.; Lemaître, F.; Verola, O.; Cho, M.-S.; Gorochov, G.; Dubertret, L.; Bachelez, H.; Kourilsky, P.; Ferradini, L. Foxp3 Expressing CD4+ CD25 (High) Regulatory T Cells Are Overrepresented in Human Metastatic Melanoma Lymph Nodes and Inhibit the Function of Infiltrating T Cells. J. Immunol. 2004, 173, 1444–1453. [Google Scholar] [CrossRef] [PubMed]
- Barua, S.; Fang, P.; Sharma, A.; Fujimoto, J.; Wistuba, I.; Rao, A.U.K.; Lin, S.H. Spatial Interaction of Tumor Cells and Regulatory T Cells Correlates with Survival in Non-Small Cell Lung Cancer. Lung Cancer 2018, 117, 73–79. [Google Scholar] [CrossRef]
- Sakaguchi, S.; Ono, M.; Setoguchi, R.; Yagi, H.; Hori, S.; Fehervari, Z.; Shimizu, J.; Takahashi, T.; Nomura, T. Foxp3+ CD25+ CD4+ Natural Regulatory T Cells in Dominant Self-Tolerance and Autoimmune Disease. Immunol. Rev. 2006, 212, 8–27. [Google Scholar] [CrossRef]


| Clinical and Laboratory Features | All Patients (N = 28) | |||
|---|---|---|---|---|
| UCD | iMCD | HHV8+ HIV− MCD | HHV8+ HIV+ MCD | |
| B-symptoms * | 1/10 | 4/7 | 3/4 | 1/1 |
| Anemia (Hb < 13 g/dL M, <12 g/dL F) | 1/10 | 2/7 | 4/4 | 1/1 |
| Serum monoclonal component ** | 2/7 | 7/7 | 1/3 | 1/1 |
| Hepatosplenomegaly | 0/10 | 5/8 | 3/4 | 1/1 |
| Fluid effusions | 2/10 | 0/7 | 1/3 | 0/1 |
| Increased creatininemia (>1.2 mg/dL) | 0/9 | 0/7 | 3/4 | 0/1 |
| Proteinuria (>15 mg/dL) | 1/6 | 2/6 | 2/4 | 1/1 |
| Interstitial lymphocytic pneumonia (LIP) | 0/9 | 0/9 | 0/4 | 0/1 |
| Peripheral neuropathy | 0/13 | 2/9 | 0/4 | 0/1 |
| LDH > UNL | 4/10 | 4/6 | 1/4 | 1/1 |
| IL-6 > UNL | 2/5 | 3/5 | 1/1 | 1/1 |
| HHV8 (circulating DNA and/or IHC) + | 0/13 | 0/9 | 5/5 | 1/1 |
| EBV serology (IgG: EBNA, VCA) + | 3/4 | 4/6 | 2/2 | 1/1 |
| EBV-DNA+ | 0/4 | 0/5 | 1/2 | 0/1 |
| CMV serology (IgG) + | 3/5 | 4/6 | 2/3 | 1/1 |
| CMV-DNA+ | 0/5 | 0/6 | 2/3 | 0/1 |
| HIV serology + | 0/9 | 0/7 | 0/5 | 1/1 |
| HCV serology + | 0/9 | 0/7 | 0/5 | 0/1 |
| HBV serology (anti-HBc antibodies) + | 0/9 | 2/7 | 1/5 | 0/1 |
| Quantiferon TB test + | 0/5 | 0/4 | 0/2 | 0/1 |
| Cherry hemangiomas | 0/10 | 0/9 | 0/4 | 0/1 |
| Secondary amyloidosis | 2/13 | 0/9 | 0/5 | 0/1 |
| Lymphoproliferative clonal disorder | 0/13 | 2/9 | 0/4 | 0/1 |
| Hemophagocytic lymphohistiocytosis | 0/13 | 0/9 | 2/4 | 0/1 |
| Solid tumor | 1/13 | 2/9 | 2/4 | 1/1 |
| Autoimmune disease *** | 0/13 | 2/9 | 1/4 | 0/1 |
| POEMS syndrome | - | 0/9 | 0/4 | 0/1 |
| TAFRO syndrome | - | 0/9 | 0/4 | 0/1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fraticelli, S.; Lucioni, M.; Neri, G.; Marchiori, D.; Cristinelli, C.; Merli, M.; Monaco, R.; Borra, T.; Lazzaro, A.; Uccella, S.; et al. T-Cells Subsets in Castleman Disease: Analysis of 28 Cases Including Unicentric, Multicentric and HHV8-Related Clinical Forms. Int. J. Mol. Sci. 2023, 24, 7813. https://doi.org/10.3390/ijms24097813
Fraticelli S, Lucioni M, Neri G, Marchiori D, Cristinelli C, Merli M, Monaco R, Borra T, Lazzaro A, Uccella S, et al. T-Cells Subsets in Castleman Disease: Analysis of 28 Cases Including Unicentric, Multicentric and HHV8-Related Clinical Forms. International Journal of Molecular Sciences. 2023; 24(9):7813. https://doi.org/10.3390/ijms24097813
Chicago/Turabian StyleFraticelli, Sara, Marco Lucioni, Giuseppe Neri, Deborah Marchiori, Caterina Cristinelli, Michele Merli, Rodolfo Monaco, Tiziana Borra, Antonio Lazzaro, Silvia Uccella, and et al. 2023. "T-Cells Subsets in Castleman Disease: Analysis of 28 Cases Including Unicentric, Multicentric and HHV8-Related Clinical Forms" International Journal of Molecular Sciences 24, no. 9: 7813. https://doi.org/10.3390/ijms24097813







