Soybean (Glycine max L.) Lipoxygenase 1 (LOX 1) Is Modulated by Nitric Oxide and Hydrogen Sulfide: An In Vitro Approach
Abstract
:1. Introduction
2. Results and Discussion
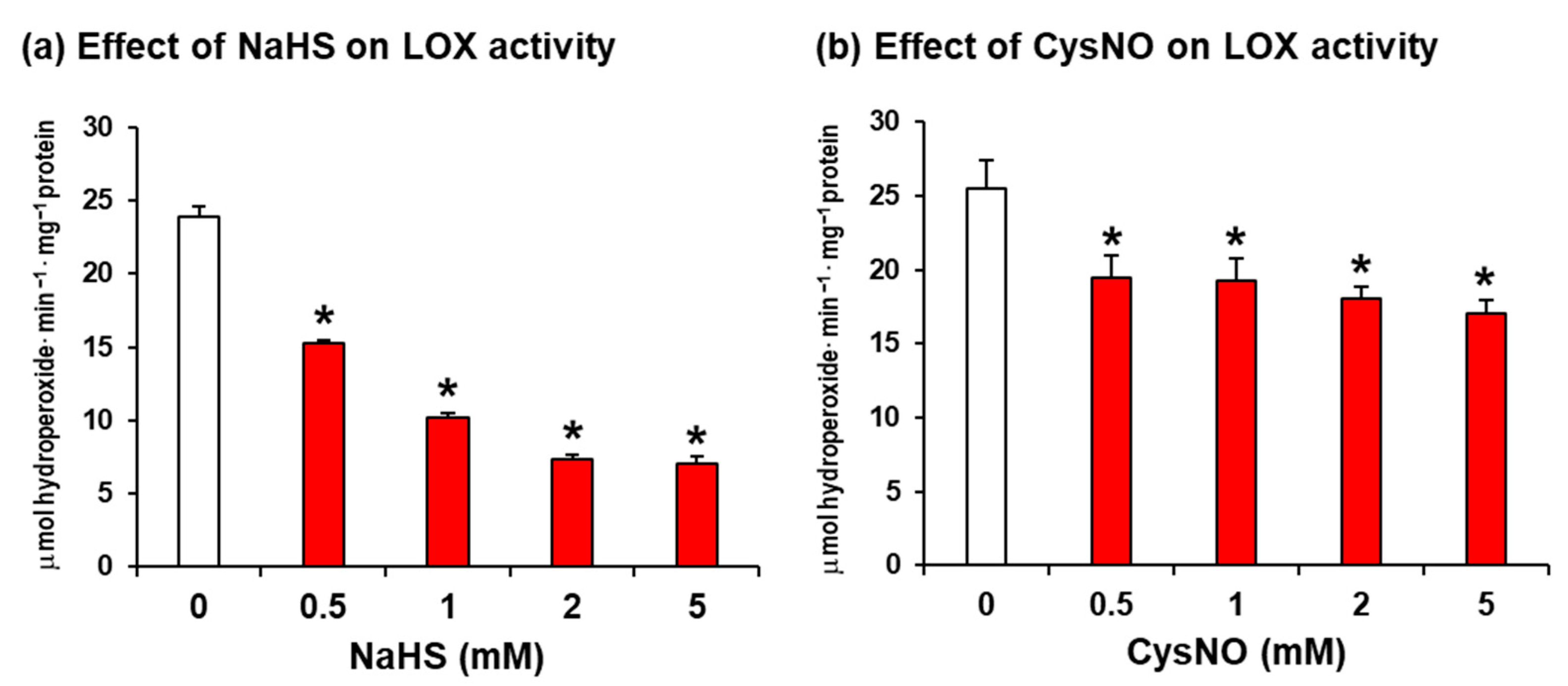
2.1. Soybean LOX 1 Activity Is Downregulated by H2S and NO Donors
2.2. Soybean LOX 1 Activity Is Also Negatively Modulated by Tyrosine Nitration
2.3. Analysis of the Structure of Soybean LOX 1
3. Materials and Methods
3.1. Plant Material
3.2. Enzymatic LOX Activity and Protein Assay
3.3. In Vitro Treatment with Hydrogen Sulfide (H2S) and Nitric Oxide (NO) Donors, and Peroxynitrite (ONOO−)
3.4. SDS-PAGE and Immunoblot Analyses
3.5. Identification of Nitrated Tyrosine in Soybean LOX 1 Using Liquid Chromatography with Tandem Mass Spectrometry (LC-MS/MS)
3.6. In Silico Analyses
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Feussner, I.; Wasternack, C. The Lipoxygenase Pathway. Annu. Rev. Plant Biol. 2002, 53, 275–297. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Borrego, E.J.; Gorman, Z.; Huang, P.-C.; Kolomiets, M.V. Relative Contribution of LOX10, Green Leaf Volatiles and JA to Wound-Induced Local and Systemic Oxylipin and Hormone Signature in Zea mays (Maize). Phytochemistry 2020, 174, 112334. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.; Li, R.; Zhang, C.; Li, W.; Li, L.; Hu, S.; Niu, J.; Cao, X.; Wang, D.; Wang, Z. Identification and Characterization of Jasmonic Acid Biosynthetic Genes in Salvia miltiorrhiza Bunge. Int. J. Mol. Sci. 2022, 23, 9384. [Google Scholar] [CrossRef] [PubMed]
- González-Gordo, S.; Cañas, A.; Muñoz-Vargas, M.A.; Palma, J.M.; Corpas, F.J. Lipoxygenase (LOX) in Sweet and Hot Pepper (Capsicum annuum L.) Fruits during Ripening and under an Enriched Nitric Oxide (NO) Gas Atmosphere. Int. J. Mol. Sci. 2022, 23, 15211. [Google Scholar] [CrossRef]
- Huang, D.; Ma, F.; Wu, B.; Lv, W.; Xu, Y.; Xing, W.; Chen, D.; Xu, B.; Song, S. Genome-Wide Association and Expression Analysis of the Lipoxygenase Gene Family in Passiflora edulis Revealing PeLOX4 Might Be Involved in Fruit Ripeness and Ester Formation. Int. J. Mol. Sci. 2022, 23, 12496. [Google Scholar] [CrossRef]
- Camargo, P.O.; Calzado, N.F.; Budzinski, I.G.F.; Domingues, D.S. Genome-Wide Analysis of Lipoxygenase (LOX) Genes in Angiosperms. Plants 2023, 12, 398. [Google Scholar] [CrossRef]
- Huang, F.-C.; Schwab, W. Cloning and Characterization of a 9-Lipoxygenase Gene Induced by Pathogen Attack from Nicotiana benthamiana for Biotechnological Application. BMC Biotechnol. 2011, 11, 30. [Google Scholar] [CrossRef]
- Babenko, L.M.; Shcherbatiuk, M.M.; Skaterna, T.D.; Kosakivska, I.V. Lipoxygenases and Their Metabolites in Formation of Plant Stress Tolerance. Ukr. Biochem. J. 2017, 89, 5–21. [Google Scholar] [CrossRef]
- Corpas, F.J.; González-Gordo, S.; Palma, J.M. Protein Nitration: A Connecting Bridge between Nitric Oxide (NO) and Plant Stress. Plant Stress 2021, 2, 100026. [Google Scholar] [CrossRef]
- González-Gordo, S.; Palma, J.M.; Corpas, F.J. Appraisal of H2S Metabolism in Arabidopsis thaliana: In Silico Analysis at the Subcellular Level. Plant Physiol. Biochem. 2020, 155, 579–588. [Google Scholar] [CrossRef]
- Papanatsiou, M.; Scuffi, D.; Blatt, M.R.; García-Mata, C. Hydrogen Sulfide Regulates Inward-Rectifying K+ Channels in Conjunction with Stomatal Closure. Plant Physiol. 2015, 168, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Zhang, J.; Zhou, M.; Zhou, H.; Cui, B.; Gotor, C.; Romero, L.C.; Fu, L.; Yang, J.; Foyer, C.H.; et al. Persulfidation-Based Modification of Cysteine Desulfhydrase and the NADPH Oxidase RBOHD Controls Guard Cell Abscisic Acid Signaling. Plant Cell 2020, 32, 1000–1017. [Google Scholar] [CrossRef] [PubMed]
- Mishra, V.; Singh, P.; Tripathi, D.K.; Corpas, F.J.; Singh, V.P. Nitric Oxide and Hydrogen Sulfide: An Indispensable Combination for Plant Functioning. Trends Plant Sci. 2021, 26, 1270–1285. [Google Scholar] [CrossRef] [PubMed]
- Ciacka, K.; Staszek, P.; Sobczynska, K.; Krasuska, U.; Gniazdowska, A. Nitric Oxide in Seed Biology. Int. J. Mol. Sci. 2022, 23, 14951. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Corpas, F.J. H2O2, NO, and H2S Networks during Root Development and Signalling under Physiological and Challenging Environments: Beneficial or Toxic? Plant Cell Environ. 2023, 46, 688–717. [Google Scholar] [CrossRef]
- Kharbech, O.; Houmani, H.; Chaoui, A.; Corpas, F.J. Alleviation of Cr(VI)-Induced Oxidative Stress in Maize (Zea mays L.) Seedlings by NO and H2S Donors through Differential Organ-Dependent Regulation of ROS and NADPH-Recycling Metabolisms. J. Plant Physiol. 2017, 219, 71–80. [Google Scholar] [CrossRef]
- Kharbech, O.; Sakouhi, L.; Ben Massoud, M.; Jose Mur, L.A.; Corpas, F.J.; Djebali, W.; Chaoui, A. Nitric Oxide and Hydrogen Sulfide Protect Plasma Membrane Integrity and Mitigate Chromium-Induced Methylglyoxal Toxicity in Maize Seedlings. Plant Physiol. Biochem. 2020, 157, 244–255. [Google Scholar] [CrossRef]
- Corpas, F.J.; González-Gordo, S.; Palma, J.M. Nitric Oxide: A Radical Molecule with Potential Biotechnological Applications in Fruit Ripening. J. Biotechnol. 2020, 324, 211–219. [Google Scholar] [CrossRef]
- Corpas, F.J.; González-Gordo, S.; Muñoz-Vargas, M.A.; Rodríguez-Ruiz, M.; Palma, J.M. The Modus Operandi of Hydrogen Sulfide(H2S)-Dependent Protein Persulfidation in Higher Plants. Antioxidants 2021, 10, 1686. [Google Scholar] [CrossRef]
- Rigo, A.A.; Dahmer, A.M.; Steffens, C.; Steffens, J.; Carrão-Panizzi, M.C. Characterization of Soybean Cultivars Genetically Improved for Human Consumption. ETP Int. J. Food Eng. 2015, 1, 1–7. [Google Scholar] [CrossRef]
- Nair, R.M.; Boddepalli, V.N.; Yan, M.-R.; Kumar, V.; Gill, B.; Pan, R.S.; Wang, C.; Hartman, G.L.; Silva, E.S.R.; Somta, P. Global Status of Vegetable Soybean. Plants 2023, 12, 609. [Google Scholar] [CrossRef]
- Axelrod, B.; Cheesbrough, T.M.; Laakso, S. [53] Lipoxygenase from Soybeans: EC 1.13.11.12 Linoleate:Oxygen Oxidoreductase. In Methods in Enzymology; Lipids Part C; Academic Press: Cambridge, MA, USA, 1981; Volume 71, pp. 441–451. [Google Scholar]
- Altchuler, M.; Scott Grayburn, W.; Collins, G.B.; Hildebrand, D.F. Developmental Expression of Lipoxygenases in Soybeans. Plant Sci. 1989, 63, 151–158. [Google Scholar] [CrossRef]
- Lenis, J.M.; Gillman, J.D.; Lee, J.D.; Shannon, J.G.; Bilyeu, K.D. Soybean Seed Lipoxygenase Genes: Molecular Characterization and Development of Molecular Marker Assays. Theor. Appl. Genet. 2010, 120, 1139–1149. [Google Scholar] [CrossRef]
- Loiseau, J.; Vu, B.L.; Macherel, M.-H.; Deunff, Y.L. Seed Lipoxygenases: Occurrence and Functions. Seed Sci. Res. 2001, 11, 199–211. [Google Scholar]
- Yuan, S.; Chang, S.K.-C. Selected Odor Compounds in Soymilk as Affected by Chemical Composition and Lipoxygenases in Five Soybean Materials. J. Agric. Food Chem. 2007, 55, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Stanojević, S.P.; Barac, M.B.; Pesic, M.B.; Vucelić-Radović, B.V. Distribution of β-Amylase and Lipoxygenase in Soy Protein Products Obtained during Tofu Production. Chem. Ind. 2017, 71, 119–126. [Google Scholar] [CrossRef]
- Ciabotti, S.; Juhász, A.C.P.; Mandarino, J.M.G.; Costa, L.L.; Corrêa, A.D.; Simão, A.A.; Santos, E.N.F. Chemical Composition and Lipoxygenase Activity of Soybean (Glycine max L. Merrill.) Genotypes, Specific for Human Consumption, with Different Tegument Colours. Braz. J. Food Technol. 2019, 22, e2018003. [Google Scholar] [CrossRef]
- Brash, A.R. Lipoxygenases: Occurrence, Functions, Catalysis, and Acquisition of Substrate. J. Biol. Chem. 1999, 274, 23679–23682. [Google Scholar] [CrossRef]
- Liavonchanka, A.; Feussner, I. Lipoxygenases: Occurrence, Functions and Catalysis. J. Plant. Physiol. 2006, 163, 348–357. [Google Scholar] [CrossRef]
- Liburdi, K.; Benucci, I.; Lombardelli, C.; Esti, M. Identification and Characterization of Lipoxygenase in Fresh Culinary Herbs. Int. J. Food Prop. 2017, 20, 1470–1478. [Google Scholar] [CrossRef]
- Prigge, S.T.; Boyington, J.C.; Gaffney, B.J.; Amzel, L.M. Structure Conservation in Lipoxygenases: Structural Analysis of Soybean Lipoxygenase-1 and Modeling of Human Lipoxygenases. Proteins 1996, 24, 275–291. [Google Scholar] [CrossRef]
- Dainese, E.; Sabatucci, A.; van Zadelhoff, G.; Angelucci, C.B.; Vachette, P.; Veldink, G.A.; Agrò, A.F.; Maccarrone, M. Structural Stability of Soybean Lipoxygenase-1 in Solution as Probed by Small Angle X-Ray Scattering. J. Mol. Biol. 2005, 349, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Prigge, S.T.; Boyington, J.C.; Faig, M.; Doctor, K.S.; Gaffney, B.J.; Amzel, L.M. Structure and Mechanism of Lipoxygenases. Biochimie 1997, 79, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Wood, I.; Trostchansky, A.; Rubbo, H. Structural Considerations on Lipoxygenase Function, Inhibition and Crosstalk with Nitric Oxide Pathways. Biochimie 2020, 178, 170–180. [Google Scholar] [CrossRef]
- Sun, Y.; Lv, D.; Wang, W.; Xu, W.; Wang, L.; Miao, C.; Lin, H.-H. Lipoxygenase 2 Functions in Exogenous Nitric Oxide-Induced Stomatal Closure in Arabidopsis thaliana. Funct. Plant Biol. 2015, 42, 1019–1025. [Google Scholar] [CrossRef]
- de Freitas-Silva, L.; Rodríguez-Ruiz, M.; Houmani, H.; da Silva, L.C.; Palma, J.M.; Corpas, F.J. Glyphosate-Induced Oxidative Stress in Arabidopsis thaliana Affecting Peroxisomal Metabolism and Triggers Activity in the Oxidative Phase of the Pentose Phosphate Pathway (OxPPP) Involved in NADPH Generation. J. Plant Physiol. 2017, 218, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Vargas, M.A.; González-Gordo, S.; Cañas, A.; López-Jaramillo, J.; Palma, J.M.; Corpas, F.J. Endogenous Hydrogen Sulfide (H2S) Is up-Regulated during Sweet Pepper (Capsicum annuum L.) Fruit Ripening. In Vitro Analysis Shows That NADP-Dependent Isocitrate Dehydrogenase (ICDH) Activity Is Inhibited by H2S and NO. Nitric Oxide 2018, 81, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Vargas, M.A.; González-Gordo, S.; Palma, J.M.; Corpas, F.J. Inhibition of NADP-Malic Enzyme Activity by H2S and NO in Sweet Pepper (Capsicum annuum L.) Fruits. Physiol. Plant 2020, 168, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.J. The Nitric Oxide Complex of Ferrous Soybean Lipoxygenase-1. Substrate, PH, and Ethanol Effects on the Active-Site Iron. J. Biol. Chem. 1987, 262, 12137–12142. [Google Scholar] [CrossRef]
- Wiesner, R.; Rathmann, J.; Holzhütter, H.G.; Stösser, R.; Mäder, K.; Nolting, H.; Kühn, H. Nitric Oxide Oxidises a Ferrous Mammalian Lipoxygenase to a Pre-Activated Ferric Species. FEBS Lett. 1996, 389, 229–232. [Google Scholar] [CrossRef]
- Holzhütter, H.G.; Wiesner, R.; Rathmann, J.; Stösser, R.; Kühn, H. A Kinetic Model for the Interaction of Nitric Oxide with a Mammalian Lipoxygenase. Eur. J. Biochem. 1997, 245, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Corpas, F.J.; del Río, L.A.; Barroso, J.B. Need of Biomarkers of Nitrosative Stress in Plants. Trends Plant Sci. 2007, 12, 436–438. [Google Scholar] [CrossRef] [PubMed]
- Corpas, F.J.; Leterrier, M.; Begara-Morales, J.C.; Valderrama, R.; Chaki, M.; López-Jaramillo, J.; Luque, F.; Palma, J.M.; Padilla, M.N.; Sánchez-Calvo, B.; et al. Inhibition of Peroxisomal Hydroxypyruvate Reductase (HPR1) by Tyrosine Nitration. Biochim. Biophys Acta 2013, 1830, 4981–4989. [Google Scholar] [CrossRef]
- Begara-Morales, J.C.; Chaki, M.; Sánchez-Calvo, B.; Mata-Pérez, C.; Leterrier, M.; Palma, J.M.; Barroso, J.B.; Corpas, F.J. Protein Tyrosine Nitration in Pea Roots during Development and Senescence. J. Exp. Bot. 2013, 64, 1121–1134. [Google Scholar] [CrossRef]
- Corpas, F.J.; Barroso, J.B. Peroxynitrite (ONOO-) Is Endogenously Produced in Arabidopsis Peroxisomes and Is Overproduced under Cadmium Stress. Ann. Bot. 2014, 113, 87–96. [Google Scholar] [CrossRef]
- Shibata, D.; Steczko, J.; Dixon, J.E.; Hermodson, M.; Yazdanparast, R.; Axelrod, B. Primary Structure of Soybean Lipoxygenase-1. J. Biol. Chem. 1987, 262, 10080–10085. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Takemoto, D.; Kawakita, K. Proteomic Analysis of S-Nitrosylated Proteins in Potato Plant. Physiol. Plant 2013, 148, 371–386. [Google Scholar] [CrossRef]
- Stourac, J.; Vavra, O.; Kokkonen, P.; Filipovic, J.; Pinto, G.; Brezovsky, J.; Damborsky, J.; Bednar, D. Caver Web 1.0: Identification of Tunnels and Channels in Proteins and Analysis of Ligand Transport. Nucleic Acids Res. 2019, 47, W414–W422. [Google Scholar] [CrossRef]
- Collazo, L.; Klinman, J.P. Control of the Position of Oxygen Delivery in Soybean Lipoxygenase-1 by Amino Acid Side Chains within a Gas Migration Channel. J. Biol. Chem. 2016, 291, 9052–9059. [Google Scholar] [CrossRef]
- Wu, H. Affecting the activity of soybean lipoxygenase-1. J. Mol. Graph. 1996, 14, 331–337. [Google Scholar] [CrossRef]
- Sudharshan, E.; Rao, A.G. Involvement of cysteine residues and domain interactions in the reversible unfolding of lipoxygenase-1. J. Biol. Chem. 1999, 274, 35351–35358. [Google Scholar] [CrossRef] [PubMed]
- Zaragoza, J.P.T.; Nguy, A.; Minnetian, N.; Deng, Z.; Iavarone, A.T.; Offenbacher, A.R.; Klinman, J.P. Detecting and Characterizing the Kinetic Activation of Thermal Networks in Proteins: Thermal Transfer from a Distal, Solvent-Exposed Loop to the Active Site in Soybean Lipoxygenase. J. Phys. Chem. B 2019, 123, 8662–8674. [Google Scholar] [CrossRef] [PubMed]
- Knapp, M.J.; Rickert, K.; Klinman, J.P. Temperature-Dependent Isotope Effects in Soybean Lipoxygenase-1: Correlating Hydrogen Tunneling with Protein Dynamics. J. Am. Chem. Soc. 2002, 124, 3865–3874. [Google Scholar] [CrossRef] [PubMed]
- Lua, R.C.; Wilson, S.J.; Konecki, D.M.; Wilkins, A.D.; Venner, E.; Morgan, D.H.; Lichtarge, O. UET: A Database of Evolutionarily-Predicted Functional Determinants of Protein Sequences That Cluster as Functional Sites in Protein Structures. Nucleic Acids Res. 2016, 44, D308–D312. [Google Scholar] [CrossRef] [PubMed]
- Mihalek, I.; Res, I.; Lichtarge, O. A Family of Evolution-Entropy Hybrid Methods for Ranking Protein Residues by Importance. J. Mol. Biol. 2004, 336, 1265–1282. [Google Scholar] [CrossRef]
- Chovancova, E.; Pavelka, A.; Benes, P.; Strnad, O.; Brezovsky, J.; Kozlikova, B.; Gora, A.; Sustr, V.; Klvana, M.; Medek, P.; et al. CAVER 3.0: A Tool for the Analysis of Transport Pathways in Dynamic Protein Structures. PLoS Comput. Biol. 2012, 8, e1002708. [Google Scholar] [CrossRef]
- Porta, H.; Rocha-Sosa, M. Plant Lipoxygenases. Physiological and Molecular Features. Plant Physiol. 2002, 130, 15–21. [Google Scholar] [CrossRef]
- Ogorodnikova, A.V.; Mukhitova, F.K.; Grechkin, A.N. Oxylipins in the Spikemoss Selaginella Martensii: Detection of Divinyl Ethers, 12-Oxophytodienoic Acid and Related Cyclopentenones. Phytochemistry 2015, 118, 42–50. [Google Scholar] [CrossRef]
- Lončarić, M.; Strelec, I.; Moslavac, T.; Šubarić, D.; Pavić, V.; Molnar, M. Lipoxygenase Inhibition by Plant Extracts. Biomolecules 2021, 11, 152. [Google Scholar] [CrossRef]
- Sanadhya, P.; Kumar, A.; Bucki, P.; Fitoussi, N.; Carmeli-Weissberg, M.; Borenstein, M.; Brown-Miyara, S. Tomato Divinyl Ether-Biosynthesis Pathway Is Implicated in Modulating of Root-Knot Nematode Meloidogyne Javanica’s Parasitic Ability. Front Plant Sci. 2021, 12, 670772. [Google Scholar] [CrossRef]
- Yang, A.; Smyth, H.; Chaliha, M.; James, A. Sensory Quality of Soymilk and Tofu from Soybeans Lacking Lipoxygenases. Food Sci. Nutr. 2016, 4, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, T.; Zhu, S.; Wang, D.; Sun, S.; Xin, L. Short-Term Hypobaric Treatment Alleviates Chilling Injury by Regulating Membrane Fatty Acids Metabolism in Peach Fruit. J. Food Biochem. 2022, 46, e14113. [Google Scholar] [CrossRef] [PubMed]


| # | Throughput | Length (Å) | Curvature 1 | Bottleneck (Å) 2 | Comments |
|---|---|---|---|---|---|
| #1 | 0.39 | 25.8 | 1.5 | 1.0 | |
| #2 | 0.33 | 25.9 | 1.3 | 1.1 | |
| #3 | 0.33 | 21.6 | 1.3 | 0.9 | |
| #4 | 0.27 | 30.2 | 1.5 | 1.0 | C492 (at bottleneck) |
| #5 | 0.26 | 41.3 | 2.0 | 1.0 | C127 |
| #6 | 0.22 | 46.3 | 2.1 | 1.0 | C127 |
| #7 | 0.16 | 41.2 | 1.5 | 1.0 | C492 |
| #8 | 0.17 | 60.6 | 1.9 | 1.0 | C492 |
| #9 | 0.16 | 42.3 | 1.7 | 1.0 | C127 |
| #10 | 0.13 | 60.6 | 1.7 | 0.9 | C127 |
| #11 | 0.12 | 41.5 | 1.8 | 0.9 | |
| #12 | 0.09 | 47.8 | 1.5 | 1.0 | C492 (at bottleneck) |
| #13 | 0.09 | 49.4 | 1.5 | 1.0 | C492 |
| #14 | 0.07 | 75.0 | 2.6 | 0.9 | C127 |
| #15 | 0.05 | 68.8 | 3.3 | 0.9 | C127 |
| #16 | 0.01 | 88.3 | 4.8 | 0.9 | C492 |
| #17 | 0.00 | 97.8 | 3.4 | 0.9 | C357C492 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Gordo, S.; López-Jaramillo, J.; Palma, J.M.; Corpas, F.J. Soybean (Glycine max L.) Lipoxygenase 1 (LOX 1) Is Modulated by Nitric Oxide and Hydrogen Sulfide: An In Vitro Approach. Int. J. Mol. Sci. 2023, 24, 8001. https://doi.org/10.3390/ijms24098001
González-Gordo S, López-Jaramillo J, Palma JM, Corpas FJ. Soybean (Glycine max L.) Lipoxygenase 1 (LOX 1) Is Modulated by Nitric Oxide and Hydrogen Sulfide: An In Vitro Approach. International Journal of Molecular Sciences. 2023; 24(9):8001. https://doi.org/10.3390/ijms24098001
Chicago/Turabian StyleGonzález-Gordo, Salvador, Javier López-Jaramillo, José M. Palma, and Francisco J. Corpas. 2023. "Soybean (Glycine max L.) Lipoxygenase 1 (LOX 1) Is Modulated by Nitric Oxide and Hydrogen Sulfide: An In Vitro Approach" International Journal of Molecular Sciences 24, no. 9: 8001. https://doi.org/10.3390/ijms24098001








