The State of the Art of Pediatric Multiple Sclerosis
Abstract
1. Introduction
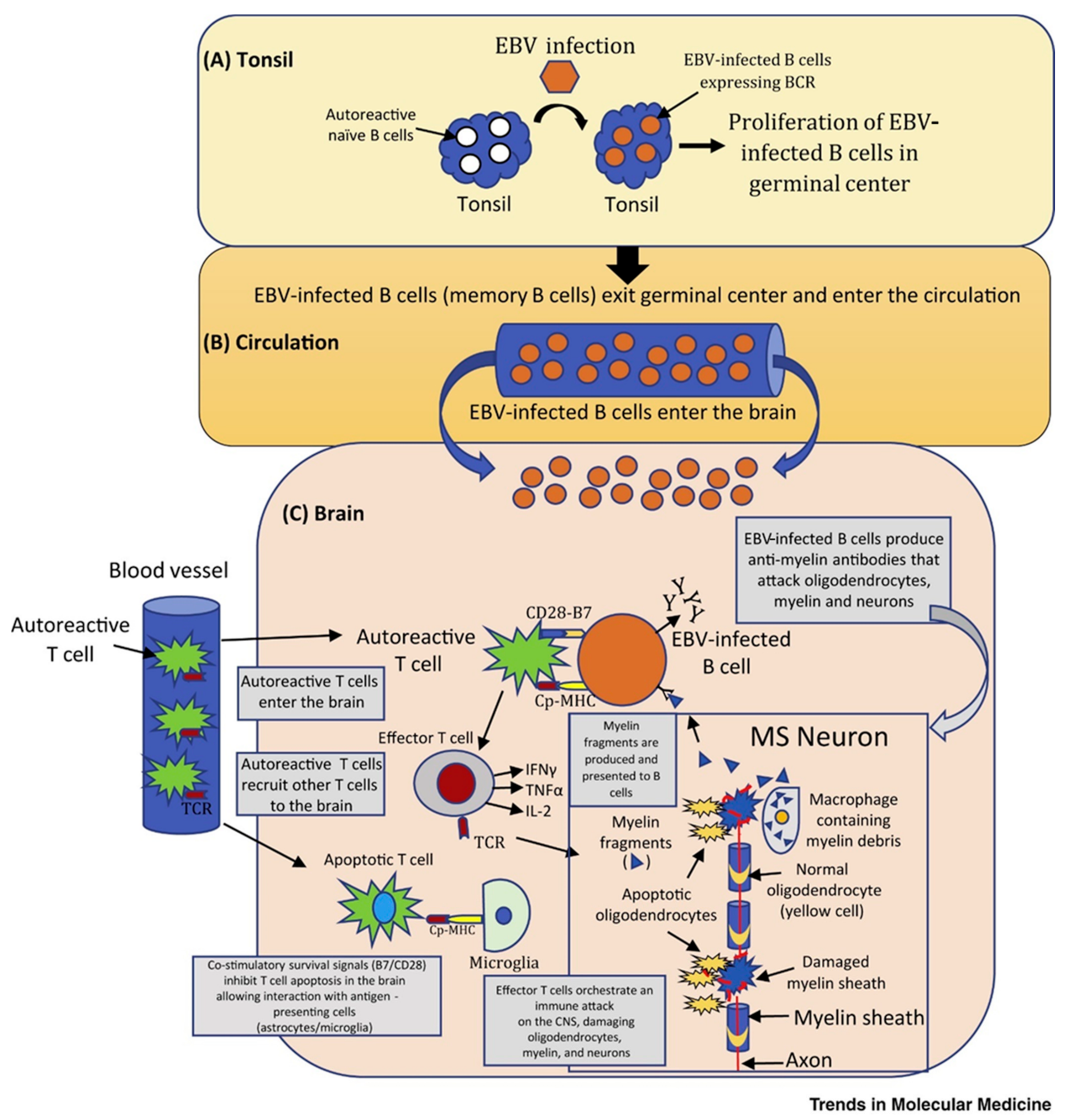
2. Etiology, Risk Factors, and Comorbidities
3. Diagnosis
4. Treatment
4.1. Conventional Approaches
4.2. Clinical Trials
4.3. Emerging Strategies
4.3.1. New Treatment Formulations
4.3.2. Stem Cell Therapies
4.3.3. Cognitive Training
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yan, K.; Balijepalli, C.; Desai, K.; Gullapalli, L.; Druyts, E. Epidemiology of pediatric multiple sclerosis: A systematic literature review and meta-analysis. Mult. Scler. Relat. Disord. 2020, 44, 102260. [Google Scholar] [CrossRef] [PubMed]
- Jancic, J.; Nikolic, B.; Ivancevic, N.; Djuric, V.; Zaletel, I.; Stevanovic, D.; Peric, S.; van den Anker, J.N.; Samardzic, J. Multiple Sclerosis in Pediatrics: Current Concepts and Treatment Options. Neurol. Ther. 2016, 5, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Abreu, C.M.; Soares-dos-Reis, R.; Melo, P.N.; Relvas, J.B.; Guimarães, J.; Sá, M.J.; Cruz, A.P.; Mendes Pinto, I. Emerging Biosensing Technologies for Neuroinflammatory and Neurodegenerative Disease Diagnostics. Front. Mol. Neurosci. 2018, 11, 164. [Google Scholar] [CrossRef] [PubMed]
- Dobson, R.; Giovannoni, G. Multiple sclerosis—A review. Eur. J. Neurol. 2019, 26, 27–40. [Google Scholar] [CrossRef] [PubMed]
- McGinley, M.P.; Goldschmidt, C.H.; Rae-Grant, A.D. Diagnosis and treatment of multiple sclerosis: A review. Jama 2021, 325, 765–779. [Google Scholar] [CrossRef]
- Immovilli, P.; De Mitri, P.; Bazzurri, V.; Vollaro, S.; Morelli, N.; Biasucci, G.; Magnifico, F.; Marchesi, E.; Lombardelli, M.L.; Gelati, L.; et al. The Impact of Highly Effective Treatment in Pediatric-Onset Multiple Sclerosis: A Case Series. Children 2022, 9, 1698. [Google Scholar] [CrossRef]
- Pilotto, S.; Gencarelli, J.; Bova, S.; Gerosa, L.; Baroncini, D.; Olivotto, S.; Alfei, E.; Zaffaroni, M.; Suppiej, A.; Cocco, E.; et al. Etiological research in pediatric multiple sclerosis: A tool to assess environmental exposures (PEDiatric Italian Genetic and enviRonment ExposurE Questionnaire). Mult. Scler. J. Exp. Transl. Clin. 2021, 7, 20552173211059048. [Google Scholar] [CrossRef]
- Padilha, I.G.; Fonseca, A.P.A.; Pettengill, A.L.M.; Fragoso, D.C.; Pacheco, F.T.; Nunes, R.H.; Maia, A.C.M.; da Rocha, A.J. Pediatric multiple sclerosis: From clinical basis to imaging spectrum and differential diagnosis. Pediatr. Radiol. 2020, 50, 776–792. [Google Scholar] [CrossRef]
- Huppke, P.; Huppke, B.; Ellenberger, D.; Rostasy, K.; Hummel, H.; Stark, W.; Brück, W.; Gärtner, J. Therapy of highly active pediatric multiple sclerosis. Mult. Scler. J. 2019, 25, 72–80. [Google Scholar] [CrossRef]
- Narula, S.; Hopkins, S.E.; Banwell, B. Treatment of Pediatric Multiple Sclerosis. Curr. Treat. Options Neurol. 2015, 17, 10. [Google Scholar] [CrossRef]
- Krupp, L.B.; Vieira, M.C.; Toledano, H.; Peneva, D.; Druyts, E.; Wu, P.; Boulos, F.C. A Review of Available Treatments, Clinical Evidence, and Guidelines for Diagnosis and Treatment of Pediatric Multiple Sclerosis in the United States. J. Child Neurol. 2019, 34, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Alroughani, R.; Boyko, A. Pediatric multiple sclerosis: A review. BMC Neurol. 2018, 18, 27. [Google Scholar] [CrossRef] [PubMed]
- Nikolic, B.; Zaletel, I.; Ivancevic, N.; Rovcanin, B.; Pepic, A.; Samardzic, J.; Jancic, J. The usefulness of visual evoked potentials in the assessment of the pediatric multiple sclerosis. Eur. J. Paediatr. Neurol. 2022, 36, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Macaron, G.; Feng, J.; Moodley, M.; Rensel, M. Newer Treatment Approaches in Pediatric-Onset Multiple Sclerosis. Curr. Treat. Options Neurol. 2019, 21, 50. [Google Scholar] [CrossRef] [PubMed]
- Langille, M.M.; Rutatangwa, A.; Francisco, C. Pediatric multiple sclerosis: A review. Adv. Pediatr. 2019, 66, 209–229. [Google Scholar] [CrossRef]
- Boiko, A.N.; Gusev, E.I.; Sudomoina, M.A.; Alekseenkov, A.D.; Kulakova, O.G.; Bikova, O.V.; Maslova, O.I.; Guseva, M.R.; Boiko, S.Y.; Guseva, M.E.; et al. Association and linkage of juvenile MS with HLA-DR2(15) in Russians. Neurology 2002, 58, 658. [Google Scholar] [CrossRef]
- Banwell, B.; Krupp, L.; Kennedy, J.; Tellier, R.; Tenembaum, S.; Ness, J.; Belman, A.; Boiko, A.; Bykova, O.; Waubant, E.; et al. Clinical features and viral serologies in children with multiple sclerosis: A multinational observational study. Lancet Neurol. 2007, 6, 773–781. [Google Scholar] [CrossRef]
- Balfour , H.H.; Schmeling, D.O.; Grimm-Geris, J.M. The promise of a prophylactic Epstein–Barr virus vaccine. Pediatr. Res. 2020, 87, 345–352. [Google Scholar] [CrossRef]
- Moreno, M.A.; Or-Geva, N.; Aftab, B.T.; Khanna, R.; Croze, E.; Steinman, L.; Han, M.H. Molecular signature of Epstein-Barr virus infection in MS brain lesions. Neurol.-Neuroimmunol. Neuroinflammation 2018, 5, e466. [Google Scholar] [CrossRef]
- Bar-Or, A.; Pender, M.P.; Khanna, R.; Steinman, L.; Hartung, H.-P.; Maniar, T.; Croze, E.; Aftab, B.T.; Giovannoni, G.; Joshi, M.A. Epstein–Barr virus in multiple sclerosis: Theory and emerging immunotherapies. Trends Mol. Med. 2020, 26, 296–310. [Google Scholar] [CrossRef]
- Thacker, E.L.; Mirzaei, F.; Ascherio, A. Infectious mononucleosis and risk for multiple sclerosis: A meta-analysis. Ann. Neurol. 2006, 59, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, T.R.; Rostgaard, K.; Nielsen, N.M.; Koch-Henriksen, N.; Haahr, S.; Sørensen, P.S.; Hjalgrim, H. Multiple sclerosis after infectious mononucleosis. Arch. Neurol. 2007, 64, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Pohl, D.; Krone, B.; Rostasy, K.; Kahler, E.; Brunner, E.; Lehnert, M.; Wagner, H.J.; Gärtner, J.; Hanefeld, F. High seroprevalence of Epstein–Barr virus in children with multiple sclerosis. Neurology 2006, 67, 2063–2065. [Google Scholar] [CrossRef]
- Alotaibi, S.; Kennedy, J.; Tellier, R.; Stephens, D.; Banwell, B. Epstein-Barr virus in pediatric multiple sclerosis. Jama 2004, 291, 1875–1879. [Google Scholar] [CrossRef] [PubMed]
- Munger, K.L.; Bentzen, J.; Laursen, B.; Stenager, E.; Koch-Henriksen, N.; Sørensen, T.I.A.; Baker, J.L. Childhood body mass index and multiple sclerosis risk: A long-term cohort study. Mult. Scler. J. 2013, 19, 1323–1329. [Google Scholar] [CrossRef] [PubMed]
- Hedström, A.K.; Olsson, T.; Alfredsson, L. Body mass index during adolescence, rather than childhood, is critical in determining MS risk. Mult. Scler. J. 2015, 22, 878–883. [Google Scholar] [CrossRef] [PubMed]
- Gianfrancesco, M.A.; Acuna, B.; Shen, L.; Briggs, F.B.S.; Quach, H.; Bellesis, K.H.; Bernstein, A.; Hedstrom, A.K.; Kockum, I.; Alfredsson, L.; et al. Obesity during childhood and adolescence increases susceptibility to multiple sclerosis after accounting for established genetic and environmental risk factors. Obes. Res. Clin. Pract. 2014, 8, e435–e447. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhang, T.T.; Yu, J.; Liu, Y.L.; Qi, S.F.; Zhao, J.J.; Liu, D.W.; Tian, Q.B. Excess Body Weight during Childhood and Adolescence Is Associated with the Risk of Multiple Sclerosis: A Meta-Analysis. Neuroepidemiology 2016, 47, 103–108. [Google Scholar] [CrossRef]
- Annette, L.-G.; Sonu, M.B.; Brandon, E.B.; Corinna, K. Childhood obesity and risk of pediatric multiple sclerosis and clinically isolated syndrome. Neurology 2013, 80, 548. [Google Scholar] [CrossRef]
- Hauer, L.; Perneczky, J.; Sellner, J. A global view of comorbidity in multiple sclerosis: A systematic review with a focus on regional differences, methodology, and clinical implications. J. Neurol. 2021, 268, 4066–4077. [Google Scholar] [CrossRef]
- Nociti, V.; Romozzi, M. Multiple Sclerosis and Autoimmune Comorbidities. J. Pers. Med. 2022, 12, 1828. [Google Scholar] [CrossRef] [PubMed]
- Magyari, M.; Sorensen, P.S. Comorbidity in Multiple Sclerosis. Front. Neurol. 2020, 11, 851. [Google Scholar] [CrossRef] [PubMed]
- Chavda, V.; Patel, V.; Patel, S. Investigation of neuroprotective potential of anti-diabetic agents in ischemic brain proteomics through in-silico molecular simulation studies. Biointerface Res. Appl. Chem. 2021, 12, 5347–5362. [Google Scholar]
- Bar-Or, A.; Hintzen, R.Q.; Dale, R.C.; Rostasy, K.; Brück, W.; Chitnis, T. Immunopathophysiology of pediatric CNS inflammatory demyelinating diseases. Neurology 2016, 87, S12. [Google Scholar] [CrossRef]
- Storm van’s Gravesande, K.; Blaschek, A.; Calabrese, P.; Rostásy, K.; Huppke, P.; Kessler, J.J.; Kalbe, E.; Mall, V. Fatigue and depression predict health-related quality of life in patients with pediatric-onset multiple sclerosis. Mult. Scler. Relat. Disord. 2019, 36, 101368. [Google Scholar] [CrossRef]
- Yeh, E.A. Real-world outcomes in pediatric MS: Psychiatric comorbidities and school performance. Mult. Scler. J. 2021, 27, 165–166. [Google Scholar] [CrossRef]
- Boesen, M.S.; Blinkenberg, M.; Thygesen, L.C.; Eriksson, F.; Magyari, M. School performance, psychiatric comorbidity, and healthcare utilization in pediatric multiple sclerosis: A nationwide population-based observational study. Mult. Scler. J. 2020, 27, 259–267. [Google Scholar] [CrossRef]
- Marchesi, O.; Vizzino, C.; Filippi, M.; Rocca, M.A. Current perspectives on the diagnosis and management of fatigue in multiple sclerosis. Expert Rev. Neurother. 2022, 22, 681–693. [Google Scholar] [CrossRef]
- Johnen, A.; Elpers, C.; Riepl, E.; Landmeyer, N.C.; Krämer, J.; Polzer, P.; Lohmann, H.; Omran, H.; Wiendl, H.; Göbel, K.; et al. Early effective treatment may protect from cognitive decline in paediatric multiple sclerosis. Eur. J. Paediatr. Neurol. 2019, 23, 783–791. [Google Scholar] [CrossRef]
- Simone, M.; Viterbo, R.G.; Margari, L.; Iaffaldano, P. A Randomized Computer-Assisted Rehabilitation Trial of Attention in Pediatric Multiple Sclerosis: A Post Hoc Analysis. Brain Sci. 2021, 11, 637. [Google Scholar] [CrossRef]
- Govindarajan, S.T.; Pan, R.; Krupp, L.; Charvet, L.; Duong, T.Q. Gray Matter Morphometry Correlates with Attentional Efficiency in Young-Adult Multiple Sclerosis. Brain Sci. 2021, 11, 80. [Google Scholar] [CrossRef] [PubMed]
- Till, C.; Ghassemi, R.; Aubert-Broche, B.; Kerbrat, A.; Collins, D.L.; Narayanan, S.; Arnold, D.L.; Desrocher, M.; Sled, J.G.; Banwell, B.L. MRI correlates of cognitive impairment in childhood-onset multiple sclerosis. Neuropsychology 2011, 25, 319–332. [Google Scholar] [CrossRef] [PubMed]
- Mesaros, S.; Rocca, M.A.; Absinta, M.; Ghezzi, A.; Milani, N.; Moiola, L.; Veggiotti, P.; Comi, G.; Filippi, M. Evidence of thalamic gray matter loss in pediatric multiple sclerosis. Neurology 2008, 70, 1107. [Google Scholar] [CrossRef] [PubMed]
- Kerbrat, A.; Aubert-Broche, B.; Fonov, V.; Narayanan, S.; Sled, J.G.; Arnold, D.A.; Banwell, B.; Collins, D.L. Reduced head and brain size for age and disproportionately smaller thalami in child-onset MS. Neurology 2012, 78, 194. [Google Scholar] [CrossRef] [PubMed]
- Bérengère, A.-B.; Vladimir, F.; Sridar, N.; Douglas, L.A.; David, A.; Dumitru, F.; Christine, T.; John, G.S.; Brenda, B.; Collins, D.L. Onset of multiple sclerosis before adulthood leads to failure of age-expected brain growth. Neurology 2014, 83, 2140. [Google Scholar] [CrossRef]
- Till, C.; Deotto, A.; Tipu, V.; Sled, J.G.; Bethune, A.; Narayanan, S.; Arnold, D.L.; Banwell, B.L. White matter integrity and math performance in pediatric multiple sclerosis: A diffusion tensor imaging study. Neuro Rep. 2011, 22, 1005–1009. [Google Scholar] [CrossRef]
- Rocca, M.A.; Morelli, M.E.; Amato, M.P.; Moiola, L.; Ghezzi, A.; Veggiotti, P.; Capra, R.; Pagani, E.; Portaccio, E.; Fiorino, A. Regional hippocampal involvement and cognitive impairment in pediatric multiple sclerosis. Mult. Scler. J. 2016, 22, 628–640. [Google Scholar] [CrossRef]
- Green, R.; Adler, A.; Banwell, B.L.; Fabri, T.L.; Yeh, E.A.; Collins, D.L.; Sled, J.G.; Narayanan, S.; Till, C. Involvement of the Amygdala in Memory and Psychosocial Functioning in Pediatric-Onset Multiple Sclerosis. Dev. Neuropsychol. 2018, 43, 524–534. [Google Scholar] [CrossRef]
- Maria, A.R.; Martina, A.; Maria Pia, A.; Lucia, M.; Angelo, G.; Pierangelo, V.; Ruggero, C.; Emilio, P.; Agnese, F.; Lorena, P.; et al. Posterior brain damage and cognitive impairment in pediatric multiple sclerosis. Neurology 2014, 82, 1314. [Google Scholar] [CrossRef]
- Amato, M.P.; Portaccio, E.; Goretti, B.; Zipoli, V.; Battaglini, M.; Bartolozzi, M.L.; Stromillo, M.L.; Guidi, L.; Siracusa, G.; Sorbi, S.; et al. Association of Neocortical Volume Changes With Cognitive Deterioration in Relapsing-Remitting Multiple Sclerosis. Arch. Neurol. 2007, 64, 1157–1161. [Google Scholar] [CrossRef]
- Calabrese, M.; Agosta, F.; Rinaldi, F.; Mattisi, I.; Grossi, P.; Favaretto, A.; Atzori, M.; Bernardi, V.; Barachino, L.; Rinaldi, L.; et al. Cortical Lesions and Atrophy Associated With Cognitive Impairment in Relapsing-Remitting Multiple Sclerosis. Arch. Neurol. 2009, 66, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Nelson, F.; Datta, S.; Garcia, N.; Rozario, N.L.; Perez, F.; Cutter, G.; Narayana, P.A.; Wolinsky, J.S. Intracortical lesions by 3T magnetic resonance imaging and correlation with cognitive impairment in multiple sclerosis. Mult. Scler. J. 2011, 17, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Geurts, J.J.G.; Pouwels, P.J.W.; Uitdehaag, B.M.J.; Polman, C.H.; Barkhof, F.; Castelijns, J.A. Intracortical lesions in multiple sclerosis: Improved detection with 3D double inversion-recovery MR imaging. Radiology 2005, 236, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Gaetani, L.; Salvadori, N.; Lisetti, V.; Eusebi, P.; Mancini, A.; Gentili, L.; Borrelli, A.; Portaccio, E.; Sarchielli, P.; Blennow, K.; et al. Cerebrospinal fluid neurofilament light chain tracks cognitive impairment in multiple sclerosis. J. Neurol. 2019, 266, 2157–2163. [Google Scholar] [CrossRef] [PubMed]
- Compas, B.E.; Jaser, S.S.; Reeslund, K.; Patel, N.; Yarboi, J. Neurocognitive deficits in children with chronic health conditions. Am. Psychol. 2017, 72, 326. [Google Scholar] [CrossRef]
- Galardi, M.M.; Gaudioso, C.; Ahmadi, S.; Evans, E.; Gilbert, L.; Mar, S. Differential Diagnosis of Pediatric Multiple Sclerosis. Children 2019, 6, 75. [Google Scholar] [CrossRef]
- Pérez, C.A. Pediatric Multiple Sclerosis. In Multiple Sclerosis: A Practical Manual for Hospital and Outpatient Care; Smith, A., Perez, C.A., Nelson, F., Eds.; Cambridge Manuals in Neurology; Cambridge University Press: Cambridge, UK, 2021; pp. 193–210. [Google Scholar]
- Krupp, L.B.; Tardieu, M.; Amato, M.P.; Banwell, B.; Chitnis, T.; Dale, R.C.; Ghezzi, A.; Hintzen, R.; Kornberg, A.; Pohl, D. International Pediatric Multiple Sclerosis Study Group criteria for pediatric multiple sclerosis and immune-mediated central nervous system demyelinating disorders: Revisions to the 2007 definitions. Mult. Scler. J. 2013, 19, 1261–1267. [Google Scholar] [CrossRef]
- Marignier, R.; Hacohen, Y.; Cobo-Calvo, A.; Pröbstel, A.-K.; Aktas, O.; Alexopoulos, H.; Amato, M.-P.; Asgari, N.; Banwell, B.; Bennett, J. Myelin-oligodendrocyte glycoprotein antibody-associated disease. Lancet Neurol. 2021, 20, 762–772. [Google Scholar] [CrossRef]
- Boesen, M.S.; Langkilde, A.R.; Ilginiene, J.; Magyari, M.; Blinkenberg, M. Oligoclonal bands, age 11–17 years, occipital lesion, and female sex differentiate pediatric multiple sclerosis from acute disseminated encephalomyelitis: A nationwide cohort study. Mult. Scler. Relat. Disord. 2022, 66, 104008. [Google Scholar] [CrossRef]
- Borisow, N.; Mori, M.; Kuwabara, S.; Scheel, M.; Paul, F. Diagnosis and Treatment of NMO Spectrum Disorder and MOG-Encephalomyelitis. Front Neurol 2018, 9, 888. [Google Scholar] [CrossRef]
- Kunchok, A.; Chen, J.J.; Saadeh, R.S.; Wingerchuk, D.M.; Weinshenker, B.G.; Flanagan, E.P.; Pittock, S.J. Application of 2015 Seronegative Neuromyelitis Optica Spectrum Disorder Diagnostic Criteria for Patients With Myelin Oligodendrocyte Glycoprotein IgG–Associated Disorders. JAMA Neurol. 2020, 77, 1572–1575. [Google Scholar] [CrossRef] [PubMed]
- Dean, M.W.; Brenda, B.; Jeffrey, L.B.; Philippe, C.; William, C.; Tanuja, C.; Jérôme de, S.; Kazuo, F.; Benjamin, G.; Anu, J.; et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 2015, 85, 177. [Google Scholar] [CrossRef]
- Thompson, A.J.; Banwell, B.L.; Barkhof, F.; Carroll, W.M.; Coetzee, T.; Comi, G.; Correale, J.; Fazekas, F.; Filippi, M.; Freedman, M.S.; et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018, 17, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Boesen, M.S.; Born, A.P.; Jensen, P.E.H.; Sellebjerg, F.; Blinkenberg, M.; Lydolph, M.C.; Jørgensen, M.K.; Rosenberg, L.; Thomassen, J.Q.; Børresen, M.L. Diagnostic Value of Oligoclonal Bands in Children: A Nationwide Population-Based Cohort Study. Pediatr. Neurol. 2019, 97, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Khaibullin, T.; Ivanova, V.; Martynova, E.; Cherepnev, G.; Khabirov, F.; Granatov, E.; Rizvanov, A.; Khaiboullina, S. Elevated levels of proinflammatory cytokines in cerebrospinal fluid of multiple sclerosis patients. Front. Immunol. 2017, 8, 531. [Google Scholar] [CrossRef]
- Fainardi, E.; Castellazzi, M.; Bellini, T.; Manfrinato, M.C.; Baldi, E.; Casetta, I.; Paolino, E.; Granieri, E.; Dallocchio, F. Cerebrospinal fluid and serum levels and intrathecal production of active matrix metalloproteinase-9 (MMP-9) as markers of disease activity in patients with multiple sclerosis. Mult. Scler. J. 2006, 12, 294–301. [Google Scholar] [CrossRef]
- Biela, A.; Watkinson, M.; Meier, U.C.; Baker, D.; Giovannoni, G.; Becer, C.R.; Krause, S. Disposable MMP-9 sensor based on the degradation of peptide cross-linked hydrogel films using electrochemical impedance spectroscopy. Biosens. Bioelectron. 2015, 68, 660–667. [Google Scholar] [CrossRef]
- Nuzziello, N.; Vilardo, L.; Pelucchi, P.; Consiglio, A.; Liuni, S.; Trojano, M.; Liguori, M. Investigating the Role of MicroRNA and Transcription Factor Co-regulatory Networks in Multiple Sclerosis Pathogenesis. Int. J. Mol. Sci. 2018, 19, 3652. [Google Scholar] [CrossRef]
- Hyun, J.-W.; Kim, Y.; Kim, G.; Kim, S.-H.; Kim, H.J. Longitudinal analysis of serum neurofilament light chain: A potential therapeutic monitoring biomarker for multiple sclerosis. Mult. Scler. J. 2020, 26, 659–667. [Google Scholar] [CrossRef]
- Reinert, M.-C.; Benkert, P.; Wuerfel, J.; Michalak, Z.; Ruberte, E.; Barro, C.; Huppke, P.; Stark, W.; Kropshofer, H.; Tomic, D. Serum neurofilament light chain is a useful biomarker in pediatric multiple sclerosis. Neurol. -Neuroimmunol. Neuroinflammation 2020, 7, e749. [Google Scholar] [CrossRef]
- Varhaug, K.N.; Torkildsen, Ø.; Myhr, K.-M.; Vedeler, C.A. Neurofilament light chain as a biomarker in multiple sclerosis. Front. Neurol. 2019, 10, 338. [Google Scholar] [CrossRef] [PubMed]
- Mitsuru, W.; Yuri, N.; Zuzanna, M.; Noriko, I.; Christian, B.; David, L.; Takuya, M.; Fumie, H.; Ryo, Y.; Jens, K.; et al. Serum GFAP and neurofilament light as biomarkers of disease activity and disability in NMOSD. Neurology 2019, 93, e1299. [Google Scholar] [CrossRef]
- Jančić, J.; Nikolić, B.; Ivančević, N.; Henčić, B.; Samardžić, J. Multiple sclerosis therapies in pediatric patients: Challenges and opportunities. In Multiple Sclerosis: Perspectives in Treatment and Pathogenesis; Zagon, I.S., McLaughlin, P.J., Eds.; Exon Publications: Brisbane, Australia, 2017; pp. 39–52. [Google Scholar]
- Goldenberg, M.M. Multiple sclerosis review. P T A Peer-Rev. J. Formul. Manag. 2012, 37, 175–184. [Google Scholar]
- Hauser, S.L.; Cree, B.A.C. Treatment of Multiple Sclerosis: A Review. Am. J. Med. 2020, 133, 1380–1390.e1382. [Google Scholar] [CrossRef] [PubMed]
- Rommer, P.S.; Berger, K.; Ellenberger, D.; Fneish, F.; Simbrich, A.; Stahmann, A.; Zettl, U.K. Management of MS Patients Treated With Daclizumab–a Case Series of 267 Patients. Front. Neurol. 2020, 11, 996. [Google Scholar] [CrossRef]
- Lancet, T. End of the road for daclizumab in multiple sclerosis. Lancet 2018, 391, 1000. [Google Scholar] [CrossRef]
- Haghikia, A.; Dendrou, C.A.; Schneider, R.; Grüter, T.; Postert, T.; Matzke, M.; Stephanik, H.; Fugger, L.; Gold, R. Severe B-cell-mediated CNS disease secondary to alemtuzumab therapy. Lancet Neurol. 2017, 16, 104–106. [Google Scholar] [CrossRef]
- Tzanetakos, D.; Breza, M.; Tzartos, J.S.; Bontzos, G.; Vakrakou, A.G.; Dermentzoglou, A.; Gkizis, I.; Smoustopoulos, G.; Evangelopoulos, M.-E.; Stefanis, L. Alemtuzumab-induced alopecia universalis and transient accommodation spasm in a patient with multiple sclerosis. Ther. Adv. Neurol. Disord. 2022, 15, 17562864221127476. [Google Scholar] [CrossRef]
- Health, N.L. Mobile Attentional Bias Modification Training in Pediatric MS. Available online: https://www.clinicaltrials.gov/ct2/show/NCT04441229?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=1 (accessed on 19 January 2023).
- Vural, P. Exercise Training in Pediatric-Onset Multiple Sclerosis Patients. Available online: https://www.clinicaltrials.gov/ct2/show/NCT04660227?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=2 (accessed on 19 January 2023).
- Yeh, E.A. ATOMIC (Active Teens Multiple Sclerosis) Physical Activity Research Program. Available online: https://www.clinicaltrials.gov/ct2/show/NCT04782466?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=3 (accessed on 19 January 2023).
- Yeh, E.A. ATOMIC (Active Teens with MultIple sClerosis) Teens: A Feasibility Study. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03137602?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=4 (accessed on 19 January 2023).
- BioPharma, I.R. A Study of NeuroVax™, a Novel Therapeutic TCR Peptide Vaccine for Pediatric Multiple Sclerosis. Available online: https://www.clinicaltrials.gov/ct2/show/NCT02200718?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=5 (accessed on 19 January 2023).
- Yeh, E.A. Adherence in Pediatric Multiple Sclerosis. Available online: https://www.clinicaltrials.gov/ct2/show/NCT02234713?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=6 (accessed on 19 January 2023).
- Health, N.L. Endeavor™ in Pediatric MS (Akili). Available online: https://www.clinicaltrials.gov/ct2/show/NCT04445116?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=7 (accessed on 19 January 2023).
- Iaffaldano, P. Cognitive Impairment in Pediatric Onset Multiple Sclerosis. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03190902?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=9 (accessed on 19 January 2023).
- Roche, H.-L. A Study To Evaluate Safety And Efficacy of Ocrelizumab in Comparison with Fingolimod in Children and Adolescents with Relapsing-Remitting Multiple Sclerosis (Operetta 2). Available online: https://www.clinicaltrials.gov/ct2/show/NCT05123703?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=10 (accessed on 19 January 2023).
- Muenster, U.H. DTI in Children with Multiple Sclerosis. Available online: https://www.clinicaltrials.gov/ct2/show/NCT02361697?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=12 (accessed on 19 January 2023).
- Biogen. PK and PD Study of Natalizumab in Pediatric Subjects with RRMS. Available online: https://www.clinicaltrials.gov/ct2/show/NCT01884935?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=13 (accessed on 19 January 2023).
- Pharmaceuticals, N. Safety and Efficacy of Fingolimod in Pediatric Patients with Multiple Sclerosis. Available online: https://www.clinicaltrials.gov/ct2/show/NCT01892722?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=14 (accessed on 19 January 2023).
- Pharmaceuticals, N. Efficacy and Safety of Ofatumumab and Siponimod Compared to Fingolimod in Pediatric Patients with Multiple Sclerosis (NEOS). Available online: https://www.clinicaltrials.gov/ct2/show/NCT04926818?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=15 (accessed on 19 January 2023).
- Sanofi. Efficacy, Safety and Pharmacokinetics of Teriflunomide in Pediatric Patients with Relapsing Forms of Multiple Sclerosis (TERIKIDS). Available online: https://www.clinicaltrials.gov/ct2/show/NCT02201108?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=16 (accessed on 19 January 2023).
- Biogen. Extension Study of BG00012 in Pediatric Subjects with Relapsing Remitting Multiple Sclerosis (RRMS). Available online: https://www.clinicaltrials.gov/ct2/show/NCT02555215?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=17 (accessed on 19 January 2023).
- Biogen. A Study to Evaluate the Safety, Tolerability, and Efficacy of BIIB017 (Peginterferon Beta-1a) in Pediatric Participants for the Treatment of Relapsing-Remitting Multiple Sclerosis. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03958877?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=19 (accessed on 19 January 2023).
- Biogen. Study of the Effect of BG00012 on MRI Lesions and Pharmacokinetics in Pediatric Subjects with RRMS (FOCUS). Available online: https://www.clinicaltrials.gov/ct2/show/NCT02410200?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=20 (accessed on 19 January 2023).
- Biogen. Phase 3 Efficacy and Safety Study of BG00012 in Pediatric Subjects with Relapsing-Remitting Multiple Sclerosis (RRMS) (CONNECT). Available online: https://www.clinicaltrials.gov/ct2/show/NCT02283853?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=21 (accessed on 19 January 2023).
- Sanofi. A Study to Evaluate Efficacy, Safety, and Tolerability of Alemtuzumab in Pediatric Patients with RRMS with Disease Activity on Prior DMT (LemKids). Available online: https://www.clinicaltrials.gov/ct2/show/NCT03368664?type=Intr&cond=Pediatric+Multiple+Sclerosis&draw=5&rank=22 (accessed on 19 January 2023).
- Yeh, E.A.; Chiang, N.; Darshan, B.; Nejati, N.; Grover, S.A.; Schwartz, C.E.; Slater, R.; Finlayson, M. Adherence in Youth with Multiple Sclerosis: A Qualitative Assessment of Habit Formation, Barriers, and Facilitators. Qual. Health Res. 2019, 29, 645–657. [Google Scholar] [CrossRef]
- Yeh, E.A.; Grover, S.A.; Powell, V.E.; Alper, G.; Banwell, B.L.; Edwards, K.; Gorman, M.; Graves, J.; Lotze, T.E.; Mah, J.K.; et al. Impact of an electronic monitoring device and behavioral feedback on adherence to multiple sclerosis therapies in youth: Results of a randomized trial. Qual. Life Res. Int. J. Qual. Life Asp. Treat. Care Rehabil. 2017, 26, 2333–2349. [Google Scholar] [CrossRef]
- Alroughani, R.; Das, R.; Penner, N.; Pultz, J.; Taylor, C.; Eraly, S. Safety and Efficacy of Delayed-Release Dimethyl Fumarate in Pediatric Patients With Relapsing Multiple Sclerosis (FOCUS). Pediatr. Neurol. 2018, 83, 19–24. [Google Scholar] [CrossRef]
- Cree, B.A.C.; Hartung, H.-P.; Barnett, M. New drugs for multiple sclerosis: New treatment algorithms. Curr. Opin. Neurol. 2022, 35, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Ioannides, Z.A.; Csurhes, P.A.; Douglas, N.L.; Mackenroth, G.; Swayne, A.; Thompson, K.M.; Hopkins, T.J.; Green, K.A.; Blum, S.; Hooper, K.D. Sustained clinical improvement in a subset of patients with progressive multiple sclerosis treated with Epstein–Barr virus-specific T cell therapy. Front. Neurol. 2021, 12, 652811. [Google Scholar] [CrossRef] [PubMed]
- Becic, A.; Leifeld, J.; Shaukat, J.; Hollmann, M. Tetraspanins as potential modulators of glutamatergic synaptic function. Front. Mol. Neurosci. 2022, 14, 801882. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C.; Thuru, X. Targeting of Tetraspanin CD81 with Monoclonal Antibodies and Small Molecules to Combat Cancers and Viral Diseases. Cancers 2023, 15, 2186. [Google Scholar] [CrossRef]
- Basile, M.S.; Mazzon, E.; Mangano, K.; Pennisi, M.; Petralia, M.C.; Lombardo, S.D.; Nicoletti, F.; Fagone, P.; Cavalli, E. Impaired Expression of Tetraspanin 32 (TSPAN32) in Memory T Cells of Patients with Multiple Sclerosis. Brain Sci. 2020, 10, 52. [Google Scholar] [CrossRef]
- Lombardo, S.D.; Mazzon, E.; Basile, M.S.; Campo, G.; Corsico, F.; Presti, M.; Bramanti, P.; Mangano, K.; Petralia, M.C.; Nicoletti, F.; et al. Modulation of Tetraspanin 32 (TSPAN32) Expression in T Cell-Mediated Immune Responses and in Multiple Sclerosis. Int. J. Mol. Sci. 2019, 20, 4323. [Google Scholar] [CrossRef] [PubMed]
- Milo, R. Therapies for multiple sclerosis targeting B cells. Croat. Med. J. 2019, 60, 87. [Google Scholar] [CrossRef] [PubMed]
- Kufukihara, K. Anti-B cell therapies in multiple sclerosis. Clin. Exp. Neuroimmunol. 2021, 12, 228–237. [Google Scholar] [CrossRef]
- Ancau, M.; Berthele, A.; Hemmer, B. CD20 monoclonal antibodies for the treatment of multiple sclerosis: Up-to-date. Expert Opin. Biol. Ther. 2019, 19, 829–843. [Google Scholar] [CrossRef]
- Margoni, M.; Preziosa, P.; Filippi, M.; Rocca, M.A. Anti-CD20 therapies for multiple sclerosis: Current status and future perspectives. J. Neurol. 2022, 269, 1316–1334. [Google Scholar] [CrossRef] [PubMed]
- Cadavid, D.; Mellion, M.; Hupperts, R.; Edwards, K.R.; Calabresi, P.A.; Drulović, J.; Giovannoni, G.; Hartung, H.-P.; Arnold, D.L.; Fisher, E.; et al. Safety and efficacy of opicinumab in patients with relapsing multiple sclerosis (SYNERGY): A randomised, placebo-controlled, phase 2 trial. Lancet Neurol. 2019, 18, 845–856. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, H.V.; Rosebraugh, M.R.; Misko, T.P.; Ziemann, A.; Liu, W.; Cree, B.A.C. Phase 1 Evaluation of Elezanumab (Anti–Repulsive Guidance Molecule A Monoclonal Antibody) in Healthy and Multiple Sclerosis Participants. Ann. Neurol. 2023, 93, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Eisen, A.; Greenberg, B.M.; Bowen, J.D.; Arnold, D.L.; Caggiano, A.O. A double-blind, placebo-controlled, single ascending-dose study of remyelinating antibody rHIgM22 in people with multiple sclerosis. Mult. Scler. J.–Exp. Transl. Clin. 2017, 3, 2055217317743097. [Google Scholar] [CrossRef] [PubMed]
- Cree, B.A.C.; Cutter, G.; Wolinsky, J.S.; Freedman, M.S.; Comi, G.; Giovannoni, G.; Hartung, H.P.; Arnold, D.; Kuhle, J.; Block, V.; et al. Safety and efficacy of MD1003 (high-dose biotin) in patients with progressive multiple sclerosis (SPI2): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. Neurol. 2020, 19, 988–997. [Google Scholar] [CrossRef]
- Brown, J.W.L.; Cunniffe, N.G.; Prados, F.; Kanber, B.; Jones, J.L.; Needham, E.; Georgieva, Z.; Rog, D.; Pearson, O.R.; Overell, J.; et al. Safety and efficacy of bexarotene in patients with relapsing-remitting multiple sclerosis (CCMR One): A randomised, double-blind, placebo-controlled, parallel-group, phase 2a study. Lancet. Neurol. 2021, 20, 709–720. [Google Scholar] [CrossRef]
- Koch, M.W.; Sage, K.; Kaur, S.; Kim, J.; Cerchiaro, G.; Yong, V.W.; Cutter, G.R.; Metz, L.M. Repurposing Domperidone in Secondary Progressive Multiple Sclerosis. Neurology 2021, 96, e2313. [Google Scholar] [CrossRef]
- Green, A.J.; Gelfand, J.M.; Cree, B.A.; Bevan, C.; Boscardin, W.J.; Mei, F.; Inman, J.; Arnow, S.; Devereux, M.; Abounasr, A.; et al. Clemastine fumarate as a remyelinating therapy for multiple sclerosis (ReBUILD): A randomised, controlled, double-blind, crossover trial. Lancet 2017, 390, 2481–2489. [Google Scholar] [CrossRef]
- Cao, G.; Edden, R.A.E.; Gao, F.; Li, H.; Gong, T.; Chen, W.; Liu, X.; Wang, G.; Zhao, B. Reduced GABA levels correlate with cognitive impairment in patients with relapsing-remitting multiple sclerosis. Eur. Radiol. 2018, 28, 1140–1148. [Google Scholar] [CrossRef]
- Teleanu, R.I.; Niculescu, A.-G.; Roza, E.; Vladâcenco, O.; Grumezescu, A.M.; Teleanu, D.M. Neurotransmitters-Key Factors in Neurological and Neurodegenerative Disorders of the Central Nervous System. Int. J. Mol. Sci. 2022, 23, 5954. [Google Scholar] [CrossRef]
- Syaifie, P.H.; Hemasita, A.W.; Nugroho, D.W.; Mardliyati, E.; Anshori, I. In silico investigation of propolis compounds as potential neuroprotective agent. Biointerface Res. Appl. Chem. 2022, 12, 8285–8306. [Google Scholar]
- Bahardoust, M. Role of adipose-derived mesenchymal stem cells in the regeneration of cardiac tissue and improvement of cardiac function: A narrative review. Biointerface Res. Appl. Chem. 2021, 11, 8446–8456. [Google Scholar] [CrossRef]
- Hassan, N.S.; Shaheen, N.E.M.; Haggag, N.Z.; Hassen, M.T. Mesenchymal Stem Cells and/or l-2-oxothiazolidine-4–Carboxylate Improve Hyperlipidemia and Lung Cell Proliferation in Chlorpyrifos-Treated Rats. Biointerface Res. Appl. Chem. 2022, 12, 7752–7774. [Google Scholar] [CrossRef]
- Genc, B.; Bozan, H.R.; Genc, S.; Genc, K. Stem Cell Therapy for Multiple Sclerosis. In Proceedings of the Tissue Engineering and Regenerative Medicine, Cham, Switzerland, 2019; pp. 145–174. [Google Scholar]
- European Society for Blood and Marrow Transplantation. Observational Study in Multiple Sclerosis Patients Treated with Autologous Hematopoietic Stem Cell Transplantation (OMST). Available online: https://clinicaltrials.gov/ct2/show/NCT04674280 (accessed on 22 January 2023).
- University, U. Hematopoietic Stem Cell Transplantation for Treatment of Multiple Sclerosis in Sweden (AutoMS-Swe). Available online: https://clinicaltrials.gov/ct2/show/NCT05029206 (accessed on 22 January 2023).
- Dasig, D.A. Reduced-intensity Immunoablation and Autologous Hematopoietic Stem Cell Transplantation (AHSCT) for Multiple Sclerosis. Available online: https://clinicaltrials.gov/ct2/show/NCT03113162 (accessed on 22 January 2023).
- Center, F.H.C. Autologous Peripheral Blood Stem Cell Transplant for Neurologic Autoimmune Diseases. Available online: https://clinicaltrials.gov/ct2/show/NCT00716066 (accessed on 22 January 2023).
- Moiseev, I.S. Fecal Microbiota Transplantation After Autologous HSCT in Patients with Multiple Sclerosis. Available online: https://clinicaltrials.gov/ct2/show/NCT04203017 (accessed on 22 January 2023).
- Hospital, H.U. RCT Comparing Autologous Hematopoietic Stem Cell Transplantation Versus Alemtuzumab, Cladribine or Ocrelizumab in MS (RAM-MS). Available online: https://clinicaltrials.gov/ct2/show/NCT03477500 (accessed on 3 March 2023).
- National Institute of Allergy and Infectious Diseases (NIAID). Best Available Therapy Versus Autologous Hematopoetic Stem Cell Transplant for Multiple Sclerosis (BEAT-MS) (BEAT-MS). Available online: https://clinicaltrials.gov/ct2/show/NCT04047628 (accessed on 3 March 2023).
- The Foundation for Orthopaedics and Regenerative Medicine. Safety of Cultured Allogeneic Adult Umbilical Cord Derived Mesenchymal Stem Cell Intravenous Infusion for MS. Available online: https://clinicaltrials.gov/ct2/show/NCT05003388 (accessed on 22 January 2023).
- Tisch Multiple Sclerosis Research Center of New York. Expanded Access to Intrathecal Administration of Autologous Mesenchymal Stem Cell-derived Neural Progenitors (MSC-NP) in Progressive Multiple Sclerosis. Available online: https://clinicaltrials.gov/ct2/show/NCT03822858 (accessed on 22 January 2023).
- Biotechnology, I. A Study to Evaluate the Safety, Tolerability, and Exploratory Efficacy of IMS001 in Subjects With Multiple Sclerosis. Available online: https://clinicaltrials.gov/ct2/show/NCT04956744 (accessed on 22 January 2023).
- Kurtzberg, J. Intrathecal Administration of DUOC-01 in Adults With Primary Progressive Multiple Sclerosis (DUOC for MS). Available online: https://clinicaltrials.gov/ct2/show/NCT04943289 (accessed on 22 January 2023).
- Hospital, H.U. Study of Mesenchymal Autologous Stem Cells as Regenerative Treatment for Multiple Sclerosis (SMART-MS). Available online: https://clinicaltrials.gov/ct2/show/NCT04749667 (accessed on 22 January 2023).
- Foundation, H.B.S.C.R. Randomized Double-Blind Phase 2 Efficacy and Safety of Autologous HB-MSCs vs Placebo for Treatment of Multiple Sclerosis (HBMS01). Available online: https://clinicaltrials.gov/ct2/show/NCT05116540 (accessed on 22 January 2023).
- Lampit, A.; Heine, J.; Finke, C.; Barnett, M.H.; Valenzuela, M.; Wolf, A.; Leung, I.H.K.; Hill, N.T.M. Computerized cognitive training in multiple sclerosis: A systematic review and meta-analysis. Neurorehabilit. Neural Repair 2019, 33, 695–706. [Google Scholar] [CrossRef] [PubMed]
- Simone, M.; Viterbo, R.G.; Margari, L.; Iaffaldano, P. Computer-assisted rehabilitation of attention in pediatric multiple sclerosis and ADHD patients: A pilot trial. BMC Neurol. 2018, 18, 82. [Google Scholar] [CrossRef]
- Tacchino, A.; Pedullà, L.; Bonzano, L.; Vassallo, C.; Battaglia, M.A.; Mancardi, G.; Bove, M.; Brichetto, G. A new app for at-home cognitive training: Description and pilot testing on patients with multiple sclerosis. JMIR Mhealth Uhealth 2015, 3, e4269. [Google Scholar] [CrossRef]
- Charvet, L.; Shaw, M.; Dobbs, B.; Frontario, A.; Sherman, K.; Bikson, M.; Datta, A.; Krupp, L.; Zeinapour, E.; Kasschau, M. Remotely Supervised Transcranial Direct Current Stimulation Increases the Benefit of At-Home Cognitive Training in Multiple Sclerosis. Neuromodulation Technol. Neural Interface 2018, 21, 383–389. [Google Scholar] [CrossRef]
- Alemayehu, D.; Hemmings, R.; Natarajan, K.; Roychoudhury, S. Perspectives on Virtual (Remote) Clinical Trials as the “New Normal” to Accelerate Drug Development. Clin. Pharmacol. Ther. 2022, 111, 373–381. [Google Scholar] [CrossRef]
- Inan, O.T.; Tenaerts, P.; Prindiville, S.A.; Reynolds, H.R.; Dizon, D.S.; Cooper-Arnold, K.; Turakhia, M.; Pletcher, M.J.; Preston, K.L.; Krumholz, H.M.; et al. Digitizing clinical trials. npj Digit. Med. 2020, 3, 101. [Google Scholar] [CrossRef]
- Rosa, C.; Marsch, L.A.; Winstanley, E.L.; Brunner, M.; Campbell, A.N.C. Using digital technologies in clinical trials: Current and future applications. Contemp. Clin. Trials 2021, 100, 106219. [Google Scholar] [CrossRef]
- Busnatu, Ș.S.; Niculescu, A.-G.; Bolocan, A.; Andronic, O.; Pantea Stoian, A.M.; Scafa-Udriște, A.; Stănescu, A.M.; Păduraru, D.N.; Nicolescu, M.I.; Grumezescu, A.M.; et al. A Review of Digital Health and Biotelemetry: Modern Approaches towards Personalized Medicine and Remote Health Assessment. J. Pers. Med. 2022, 12, 1656. [Google Scholar] [CrossRef] [PubMed]
- Weatherall, J.; Khan, F.M.; Patel, M.; Dearden, R.; Shameer, K.; Dennis, G.; Feldberg, G.; White, T.; Khosla, S. Chapter 10—Clinical trials, real-world evidence, and digital medicine. In The Era of Artificial Intelligence, Machine Learning, and Data Science in the Pharmaceutical Industry; Ashenden, S.K., Ed.; Academic Press: Cambridge, MA, USA, 2021; pp. 191–215. [Google Scholar]
- Parrish, J.B.; Fields, E. Cognitive Functioning in Patients with Pediatric-Onset Multiple Sclerosis, an Updated Review and Future Focus. Children 2019, 6, 21. [Google Scholar] [CrossRef] [PubMed]

| Immunomodulatory Therapy | Drug | Administration Route | Dose | Administration Frequency | Mechanism of Action | Side Effects |
|---|---|---|---|---|---|---|
| First-line | Interferon beta-1a | Intramuscular | 30 μg | Once a week | Inhibits lymphocyte trafficking in CNS Enhances suppressor T cell activity Reduces proinflammatory cytokine production | Flulike reactions, elevated transaminases, depression, injection site reactions |
| Subcutaneous | 22–44 μg | Three times a week | ||||
| Interferon beta-1b | Subcutaneous | 250 μg | Every other day | |||
| Glatiramer acetate | Subcutaneous | 20 mg | Once a day | Promotes Th2 cell activity Shifts toward an anti-inflammatory state | Injection site reactions | |
| Second-line | Natalizumab | Intravenous | 3–5 mg/kg | Once a month | mAb against alpha 4 integrin Prevents lymphocytes from crossing BBB | PML, infusion reaction, hepatotoxicity |
| Mitoxantrone | Intravenous | 10–20 mg/dose Up to a total of 200 mg | Once every three months | Reduces proliferation of lymphocytes | Cardiotoxicity, risk of cardiomyopathy, leukopenia, nausea, infections, alopecia, fatigue, and amenorrhea | |
| Fingolimod | Per os | 0.5 mg (>40 kg) 0.25 mg (<40kg) | Once a day | Sphingosine 1-phosphate receptor modulator Leads to downregulation in LN and prevents activated lymphocytes from leaving LN | Bradycardia, macular edema, infection, lymphopenia, increased LFT | |
| Teriflunomide | Per os | 7 and 14 mg | Once a day | Lymphocytopenia in T and B cells Disrupts pyridine synthesis | GI symptoms, alopecia, increased LFT, increase BP, peripheral neuropathy | |
| Azathioprine | Per os | 2.5–3 mg | Once a day | Antagonizes purine metabolism | Gastrointestinal disturbances, skin rashes, liver toxicity, and cytopenia | |
| Rituximab | Intravenous | 500–1000 mg | Every 6–12 months | mAb against CD 20 on B cells | Infusion reactions, PML (not in MS but has been seen in other conditions) | |
| Dimethyl fumarate | Per os | Initial dose: 120 mg Therapeutic dose: 240 mg | Twice daily | Nrf2 pathway Shift to Th2 or anti-inflammatory Cytokine profile Promotes antioxidant | Flushing, nausea, stomach upset, UTI, lymphopenia; PML has been reported | |
| Daclizumab | Subcutaneous | 150 mg | Once a month | Selectively binds to the IL-2 receptor alpha-chain | Serious infections, gastrointestinal disturbances, depression, liver toxicity with an elevation of liver enzymes, and serious cutaneous events |
| ClinicalTrials.gov Identifier | Official Title | Intervention/Treatment | Phase | Status | Actual/Estimated Study Completion Date | Ref. |
|---|---|---|---|---|---|---|
| NCT04441229 | A Prospective, Observational Study of Mobile Attentional Bias Modification Training (ABMT) in the Pediatric Multiple Sclerosis (MS) Population | Behavioral: ABMT mobile application | N/A | Completed | 26 March 2021 | [81] |
| NCT04660227 | Effectiveness of Exercise Training in Pediatric-Onset Multiple Sclerosis Patients | Procedure: Exercise Training Other: Control Group | N/A | Recruiting | January 2022 | [82] |
| NCT04782466 | Physical Activity, Quality of Life and Disease Outcomes in Youth With Multiple Sclerosis: the ATOMIC (Active Teens Multiple Sclerosis) Physical Activity Research Program | Behavioral: Physical Activity (PA) Intervention Behavioral: Waitlist attention-control | N/A | Recruiting | September 2023 | [83] |
| NCT03137602 | ATOMIC (Active Teens With MultIple sClerosis) Teens: A Feasibility Study | Device: ATOMIC mobile app | N/A | Completed | 30 September 2019 | [84] |
| NCT02200718 | A Phase I Study of NeuroVax™, a Novel Therapeutic TCR Peptide Vaccine for Pediatric Multiple Sclerosis | Biological: NeuroVax Biological: IFA Incomplete Freund’s Adjuvant | Phase 1 | Not yet recruiting | 9 November 2024 | [85] |
| NCT02234713 | Treatment Adherence in Pediatric Multiple Sclerosis | Behavioral: Motivational Interview Other: Video Attention Control | N/A | Completed | September 2016 | [86] |
| NCT04445116 | A Study of Endeavor™, a Video-Game Based Cognitive Remediation, in the Pediatric Multiple Sclerosis (MS) Population | Device: Action Video Game Treatment | N/A | Not yet recruiting | June 2024 | [87] |
| NCT03190902 | Cognitive Impairment in Pediatric Onset Multiple Sclerosis: Research of Biomarkers Predictive of Cognitive Impairment Progression | Behavioral: Attention Processing Training program (APT) Behavioral: nonspecific computer training (n-ST) | N/A | Completed | 30 April 2016 | [88] |
| NCT05123703 | A Phase III Multicenter, Randomized, Double-Blind, Double-Dummy Study To Evaluate Safety And Efficacy Of Ocrelizumab In Comparison With Fingolimod In Children And Adolescents With Relapsing-Remitting Multiple Sclerosis | Drug: Ocrelizumab Other: Ocrelizumab Placebo Drug: Fingolimod Other: Fingolimod Placebo | Phase 3 | Recruiting | 5 November 2025 | [89] |
| NCT02361697 | Monitoring of Neurodegenerative Processes in Children With Multiple Sclerosis by Diffusion-weighed Magnetic Resonance Imaging (DTI) | Other: DTI-MRI | N/A | Unknown | December 2017 | [90] |
| NCT01884935 | A Phase 1, Multicenter, Open-Label, Single-Arm, Multiple Dose Study to Evaluate the Pharmacokinetics and Pharmacodynamics of Natalizumab in Pediatric Subjects With Relapsing Remitting Multiple Sclerosis (RMS) | Biological: Natalizumab | Phase 1 | Completed | September 2014 | [91] |
| NCT01892722 | A 2 Year, Double-blind, Randomized, Multicenter, Active-controlled Core Phase to Evaluate Safety & Efficacy of Daily Fingolimod vs Weekly Interferon β-1a im in Pediatric Patients With Multiple Sclerosis and 5 Year Fingolimod Extension Phase | Drug: Interferon beta-1a Drug: Fingolimod Drug: Placebo capsule Drug: Placebo im injection | Phase 3 | Recruiting | 2 November 2028 | [92] |
| NCT04926818 | A 2-year Randomized, 3-arm, Double-blind, Non-inferiority Study Comparing the Efficacy and Safety of Ofatumumab and Siponimod Versus Fingolimod in Pediatric Patients With Multiple Sclerosis Followed by an Open-label Extension | Drug: Fingolimod Drug: Ofatumumab Drug: Siponimod Other: Fingolimod placebo Other: Siponimod placebo Other: Ofatumumab placebo | Phase 3 | Recruiting | 1 June 2029 | [93] |
| NCT02201108 | A Two Year, Multicenter, Randomized, Double-Blind, Placebo-Controlled, Parallel Group Trial to Evaluate Efficacy, Safety, Tolerability, and Pharmacokinetics of Teriflunomide Administered Orally Once Daily in Pediatric Patients With Relapsing Forms of Multiple Sclerosis Followed by an Open-Label Extension | Drug: Teriflunomide Drug: Placebo | Phase 3 | Active, not recruiting | 25 June 2025 | [94] |
| NCT02555215 | A Multicenter Extension Study to Determine the Long-Term Safety and Efficacy of BG00012 in Pediatric Subjects With Relapsing-Remitting Multiple Sclerosis | Drug: dimethyl fumarate | Phase 3 | Completed | 24 September 2018 | [95] |
| NCT03958877 | An Open-Label, Randomized, Multicenter, Active-Controlled, Parallel-Group Study to Evaluate the Safety, Tolerability, and Efficacy of BIIB017 in Pediatric Subjects Aged 10 to Less Than 18 Years for the Treatment of Relapsing-Remitting Multiple Sclerosis, With Optional Open-Label Extension | Drug: BIIB017 (peginterferon beta-1a) Drug: Interferon beta type 1a | Phase 3 | Recruiting | 5 November 2029 | [96] |
| NCT02410200 | Open-Label, Multicenter, Multiple-Dose Study of the Effect of BG00012 on MRI Lesions and Pharmacokinetics in Pediatric Subjects With Relapsing-Remitting Multiple Sclerosis Aged 10 to 17 Years | Drug: dimethyl fumarate | Phase 2 | Completed | 23 September 2016 | [97] |
| NCT02283853 | Open-Label, Randomized, Multicenter, Multiple-Dose, Active-Controlled, Parallel-Group, Efficacy and Safety Study of BG00012 in Children From 10 to Less Than 18 Years of Age With Relapsing-Remitting Multiple Sclerosis, With Optional Open-Label Extension | Drug: dimethyl fumarate Drug: Interferon β-1a | Phase 3 | Active, not recruiting | 8 September 2025 | [98] |
| NCT03368664 | A Multi-center, Open-label, Single-arm, Before and After Switch Study to Evaluate the Efficacy, Safety and Tolerability of Alemtuzumab in Paediatric Patients With Relapsing Remitting Multiple Sclerosis (RRMS) With Disease Activity on Prior Disease Modifying Therapy (DMT) | Drug: Alemtuzumab GZ402673 Drug: Glatiramer acetate Drug: Beta-Interferon Drug: Methylprednisolone Drug: Ranitidine Drug: Ceterizine Drug: Dexchlorpheniramine Drug: Paracetamol Drug: Acyclovir Drug: Prednisolone Drug: Diphenydramine Drug: Other H1 antagonist | Phase 3 | Active, not recruiting | December 2025 | [99] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teleanu, R.I.; Niculescu, A.-G.; Vladacenco, O.A.; Roza, E.; Perjoc, R.-S.; Teleanu, D.M. The State of the Art of Pediatric Multiple Sclerosis. Int. J. Mol. Sci. 2023, 24, 8251. https://doi.org/10.3390/ijms24098251
Teleanu RI, Niculescu A-G, Vladacenco OA, Roza E, Perjoc R-S, Teleanu DM. The State of the Art of Pediatric Multiple Sclerosis. International Journal of Molecular Sciences. 2023; 24(9):8251. https://doi.org/10.3390/ijms24098251
Chicago/Turabian StyleTeleanu, Raluca Ioana, Adelina-Gabriela Niculescu, Oana Aurelia Vladacenco, Eugenia Roza, Radu-Stefan Perjoc, and Daniel Mihai Teleanu. 2023. "The State of the Art of Pediatric Multiple Sclerosis" International Journal of Molecular Sciences 24, no. 9: 8251. https://doi.org/10.3390/ijms24098251
APA StyleTeleanu, R. I., Niculescu, A.-G., Vladacenco, O. A., Roza, E., Perjoc, R.-S., & Teleanu, D. M. (2023). The State of the Art of Pediatric Multiple Sclerosis. International Journal of Molecular Sciences, 24(9), 8251. https://doi.org/10.3390/ijms24098251







