1. Introduction
Prostate cancer (PCa) is the second most common cancer in men and ranks fifth among cancer-related mortality in men worldwide [
1,
2]. The incidence of PCa varies geographically and is more prevalent in developed countries, especially in North America, Europe, and Oceania. In China and other Asian countries, the incidence of this disease is rising, and cancers are first diagnosed in the middle/late stages [
1,
3]. Because the clinical manifestations of early-stage PCa are difficult to identify, an approximate majority of patients are diagnosed belatedly, leading to increased patient mortality [
4]. PCa is highly heterogeneous, complex in composition and pathogenesis, so it is difficult to find suitable and effective therapeutic targets. Current clinical treatment options for PCa include prostatectomy surgery, radiation therapy, and androgen deprivation therapy. These therapies can only temporarily limit the progression of PCa. Cancer can advance to the metastatic stage within 18–24 months, and these patients often have poor prognosis with an average survival of 12 months [
5,
6]. Immunotherapy is a hopeful approach that has demonstrated anti-tumor benefits in PCa [
7]. Tumor cells escape the immune system by binding to CTLA-4 or PD-1, promoting the differentiation and activity of Treg cells and suppressing the growth and activity of effector T cells [
8,
9]. But in PCa, many patients do not respond to monoclonal antibody therapy targeting PD1 or CTLA-4, possibly because tumor cells achieve immune escape through other pathways [
10,
11,
12]. Therefore, exploring new molecules with immunomodulatory functions to treat PCa is urgent and crucial. Recent studies have shown that tumor-infiltrating immune cells can affect the prognosis of PCa patients and the efficacy of anti-tumor immunotherapy [
13,
14,
15]. However, the molecular immune-related mechanisms of PCa remain ambiguous. Therefore, new molecular biomarkers and therapeutic approaches related to the immune infiltration in PCa are urgently needed to meet the challenging clinical needs.
GLIS1 is a GLI-related Krüppel-like zinc finger protein that acts as a transcription activator and repressor.
GLIS1 is known to play a key role in regulating many physiological processes and has also been widely reported to be linked to various pathologies, including oncogenesis [
16]. GLIS1, in conjunction with several other reprogramming factors, markedly increased the efficiency of generating induced pluripotent stem cells (iPSC) from somatic cells, suggesting their roles in cellular differentiation, proliferation, and stem cell renewal [
17,
18,
19]. Regarding tumors,
GLIS1 in cancer cells is involved in cell migration, invasion, and tumorigenesis [
20,
21]. A study by Charles et al. demonstrated that
GLIS1 is associated with the
WNT gene expression and epithelial-to-mesenchymal transition (EMT) features in breast cancer cells. Their study showed that co-transfection of
GLIS1 and
CUX1 synergistically stimulated
TCF/
β-catenin transcriptional activity, thereby increasing cell migration and invasion [
22]. A work conducted by Rong et al. demonstrated that
GLIS1 accelerates the process of CD8+ T cell exhaustion in hepatocellular carcinoma by modulating the
SGK1-
STAT3-
PD1 pathway, and exerts a synergistic effect with anti-PD1, providing a prospective method for cancer immunotherapy [
23].
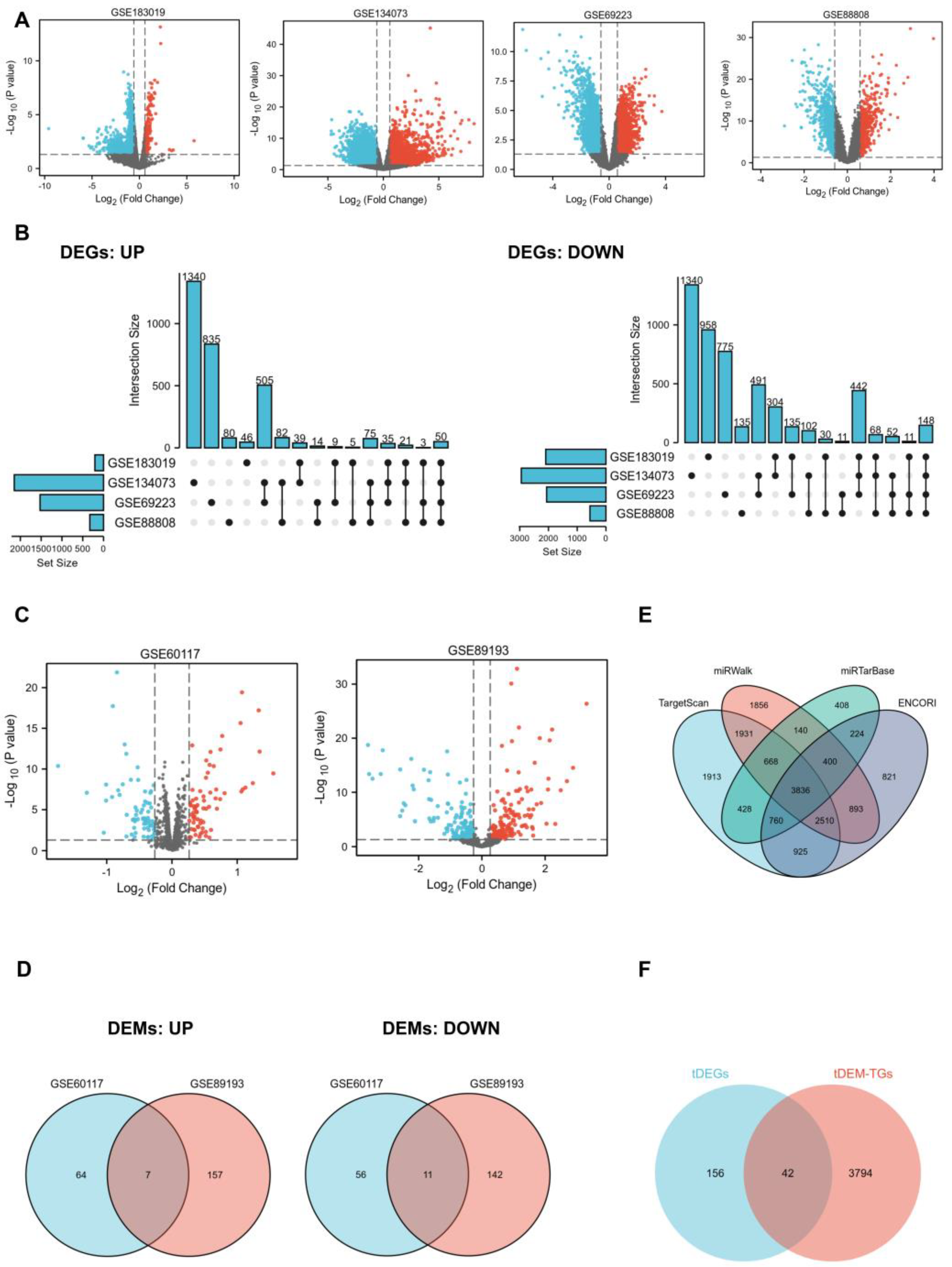
Many studies in recent years have revealed the presence of differentially expressed genes (DEGs) and differentially expressed microRNA (DEMs) in PCa [
24,
25,
26]. However, the analysis results obtained by different researchers vary. There are still unsolved concerns about the interaction of DEGs and DEMs during Pca development, and further analysis combining different studies is urgently needed to reveal the real targets.
In this work, we obtained DEGs and DEMs between normal and PCa tissue samples by applying four mRNA profiling datasets (GSE183019, GSE134073 [
27], GSE69223 [
28], and GSE88808 [
29]) and two miRNA profiling datasets (GSE89193 [
29] and GSE60117 [
30]) from the Gene Expression Omnibus (GEO) database. Subsequently, target genes of total DEMs (tDEM–TGs) were predicted, followed by overlapping analysis between tDEM–TGs and total DEGs (tDEGs) to filter potential key genes and miRNAs. We then comprehensively evaluated the differential expression of key gene
GLIS1 and its prognostic value in PCa through The Cancer Genome Atlas (TCGA) database. Additionally, we performed Gene Ontology (GO) analysis, Gene Set Enrichment Analysis (GSEA), and other methods to investigate the potential roles of
GLIS1 in tumor development and tumor immune microenvironment. This study is the first to comprehensively explore the relationship between
GLIS1 expression and its clinical, molecular, and immunotherapeutic aspects in PCa, shedding us new insights into the crucial role of
GLIS1 in PCa.
3. Discussion
It is well recognized that miRNA is a crucial molecular tool for non-invasive diagnosis and prognosis of cancer. MiRNAs can bind to the 3′ untranslated region (3′ UTR) of target mRNAs, leading to the degradation and translational repression of mRNAs, exerting regulatory roles in cancer progression or suppression [
31,
32]. Therefore, it is important to identify the most suitable DEGs that can be used as diagnostic markers or therapeutic targets by analyzing gene expression changes in combination with miRNAs. In this study, we first identified 198 tDEGs and 18 tDEMs between PCa tissues and normal tissues from GEO datasets. Through the utilization of GO and KEGG enrichment analysis, we acquired an in-depth understanding of these genes associated with the initiation and development of PCa. To learn the general patterns of molecular changes in PCa, we then uncovered
GLIS1 by aligning tDEM–TGs with tDEGs.
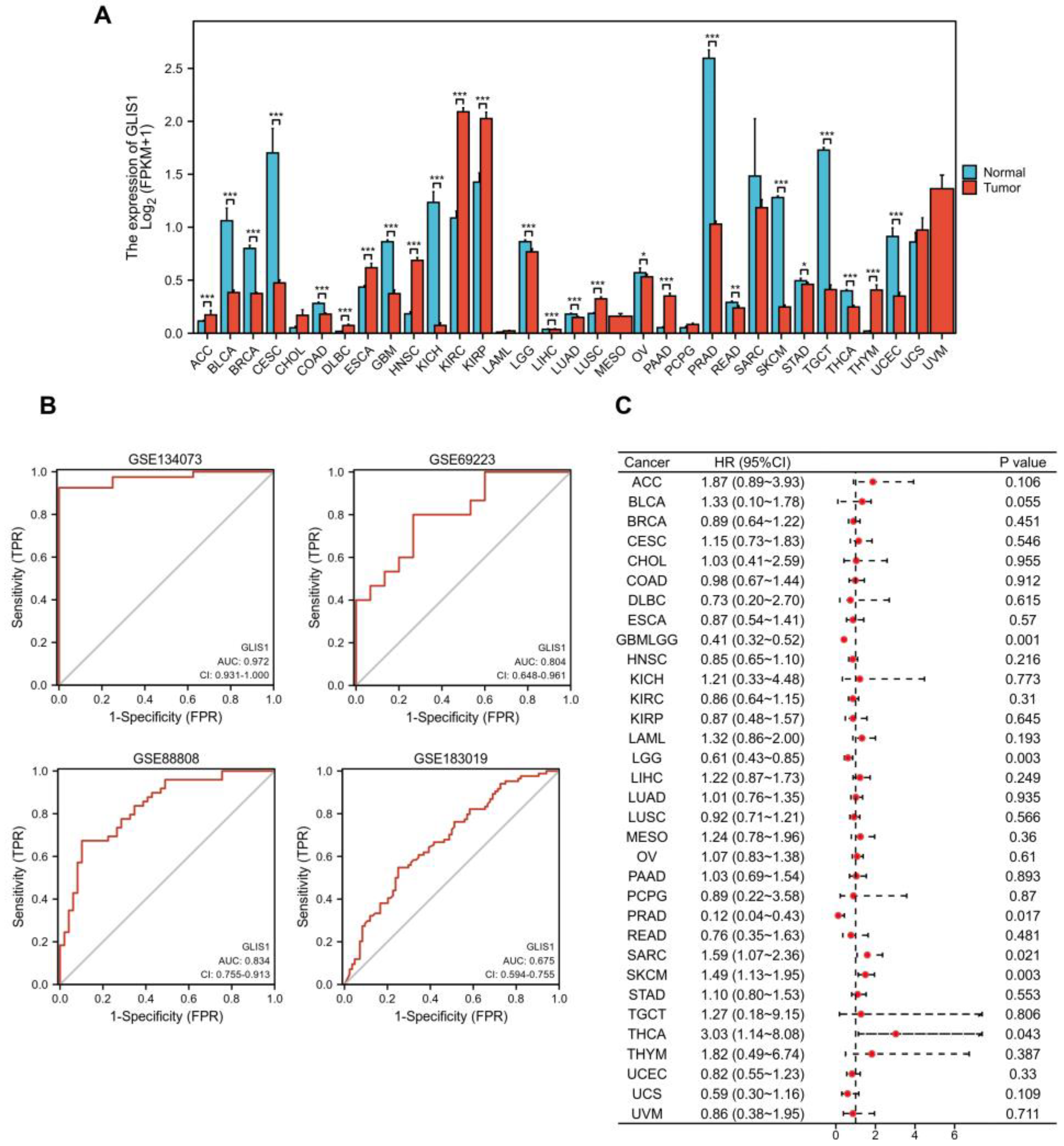
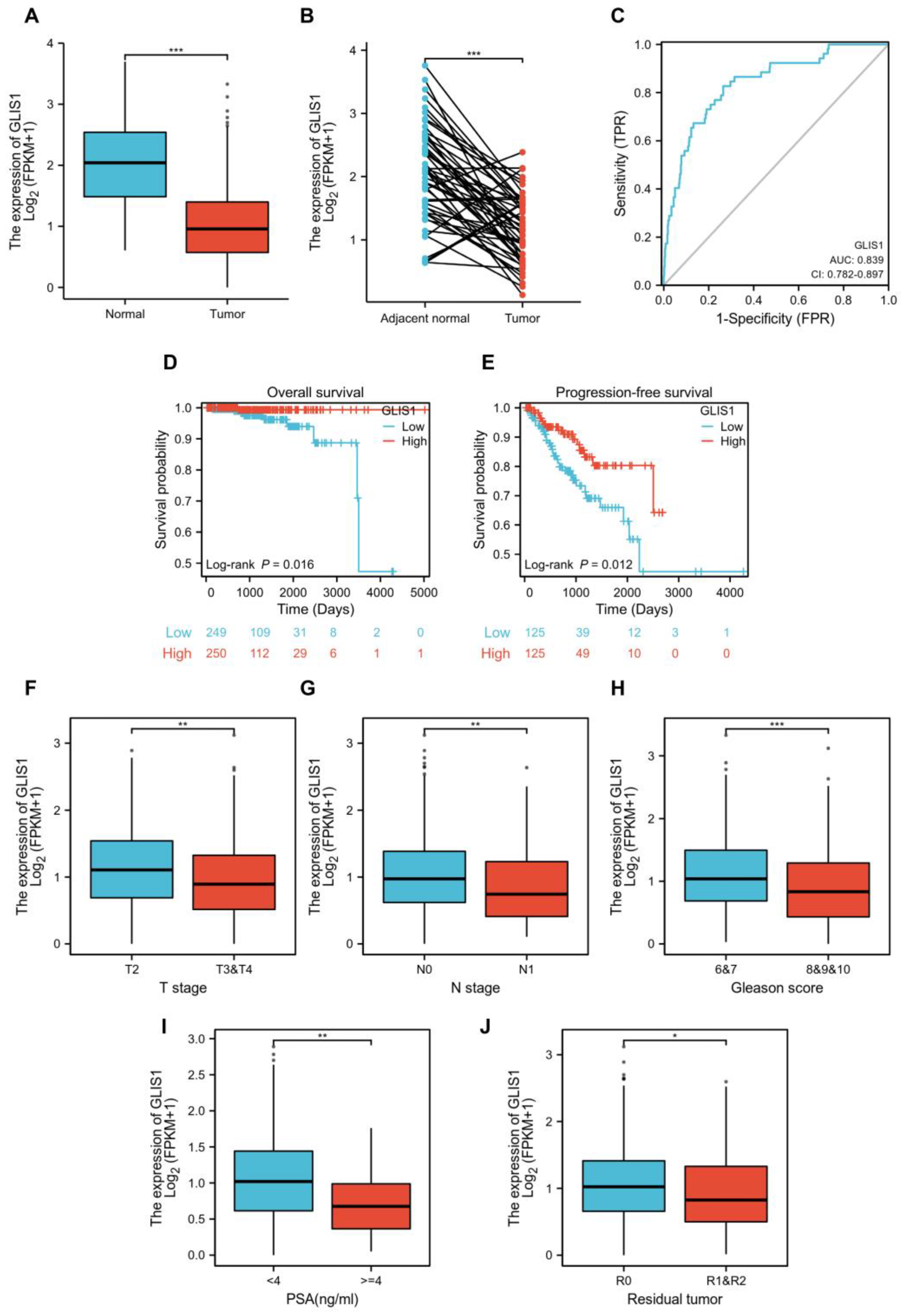
GLIS1 exhibited reduced expression levels in PCa and demonstrated a significant association with patient survival. The expression of
GLIS1 was found to be downregulated in a cohort of malignancies comparing tumor and normal tissues from independent datasets in the Genotype-Tissue Expression (GTEx) database and TCGA database. Additionally,
GLIS1 participated in the biological process of immune cells entering tumor tissues and improved the tumor microenvironment (TME) of patients, and thus may affect the development of PCa and patients’ prognosis. Therefore,
GLIS1 has great potential to serve as an immune-related biomarker in PCa. Our research provides a more comprehensive understanding of the mechanisms by which
GLIS1 involved in the development of PCa.
The study of tumor microenvironment, including various cell clusters such as immune cells, tumor cells, and fibroblasts, has gained significant attention in recent years and has a profound impact on the progression of cancers [
33,
34]. Rong et al. demonstrated that
GLIS1 contributes to CD8+ T cell exhaustion by transcriptionally regulating the
SGK1-
STAT3-
PD1 signaling pathway in HCC. Suppression of
GLIS1 in CD8+ T cells enhances the effectiveness of anti-PD1 treatment, presenting a promising strategy for HCC immunotherapy [
23]. These suggested that
GLIS1 might be involved in immune response. Therefore, how
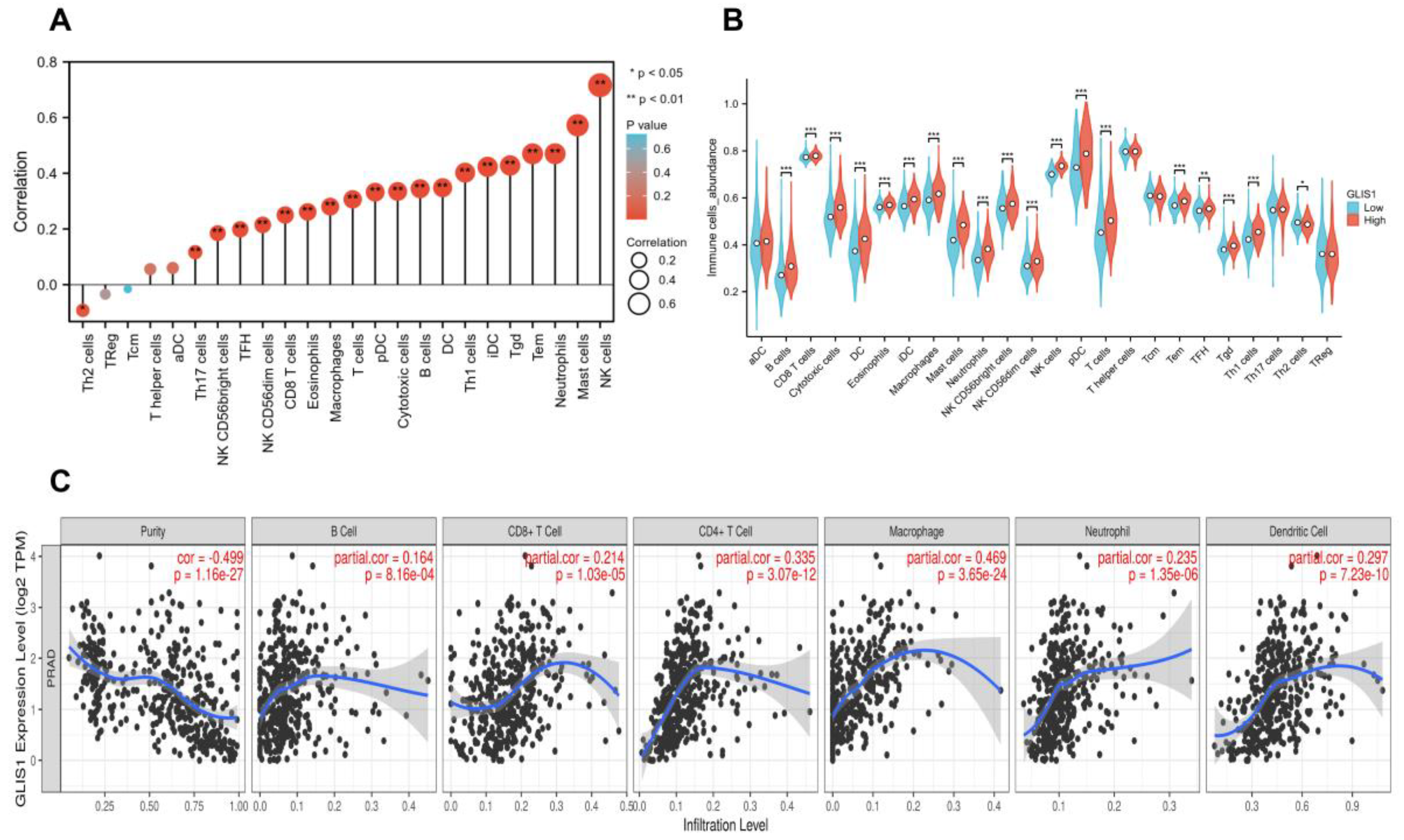
GLIS1 participates in the TME and influences tumor-infiltrating immune cells in PCa deserves exploration. Using the TIMER database, we identified that
GLIS1 exerted an influence on tumor-infiltrating immune cells in PCa. The expression of
GLIS1 exhibited a notable inverse association with tumor purity and was positively correlated with the infiltration of various immune cells in PCa, including B cells, DC, CD4+ T cells, CD8+ T cells, macrophages, and neutrophils. The ssGSEA methods further demonstrated a positive correlation between
GLIS1 expression and the infiltration of NK cells, NK CD56bright cells, NK CD56dim cells, DC, plasmacytoid DC, immature DC, several types of T cells (Tem, Tgd, Th1 cells, cytotoxic T cells, Th17 cells, CD8+ T cells, and TFH), B cells, mast cells, neutrophils, eosinophils, macrophages, and negatively correlated with Th2 cells. Increased NK cells and CD8+ T cells can strengthen the anti-tumor immune response through the secretion of diverse cytokines and the release of perforin and granzyme [
35]. NK cells, which are cytotoxic lymphocytes, consist of two primary subpopulations in humans: CD56bright and CD56dim cells [
36]. Both subsets of NK cells enhance their cytolytic ability by generating a diverse range of pro-inflammatory cytokines, including as IFN-γ and TNF-α, demonstrating increased cytotoxicity against malignancies [
37]. DC are the most powerful antigen-presenting cells, and their infiltration can facilitate the presentation of tumor-associated or tumor-specific antigens to T cells, thereby initiating primary immune responses [
38]. The functions of tumor-associated macrophages act as a “double-edged sword” in tumor development. M1 macrophages play an anti-tumor role by secreting pro-inflammatory factors and chemokines, participating in antigen presentation and immune surveillance, while M2 macrophages promote tumor growth and angiogenesis [
39,
40]. The increase in B cell infiltration facilitates tumor elimination through its involvement in antibody-dependent cytotoxicity. Th1 cells were reported to inhibit PCa growth by activating CD8+ T cells and NK cell-mediated cytolytic function via the manufacturing of Th1-type cytokines such as IFN-γ and IL-2 [
41]. However, Th2 cells are considered to promote tumor progression due to the immunosuppressive substances they release, including TGF-β, IL-4, IL-5, and IL-13 [
42]. Research has demonstrated that the infiltrating of Th2 cells is linked to weakened immune response and lower survival time across various cancer types [
43,
44]. In our study,
GLIS1 negatively regulated the infiltration of Th2 cells, which suggested that the decreased
GLIS1 expression in PCa may help mediate the tumor immune escape. As a result,
GLIS1 is involved in the regulation of TME in PCa by engaging in both cellular and humoral immunity and promoting anti-tumor activities. These findings are exciting, especially considering that there are still many PCa patients who do not respond to anti-PD1 or anti-CTLA-4 therapy, in part because PCa itself has low immune infiltration and in part because tumor cells may achieve immune escape through other pathways. Our study provides a new molecule with immunomodulatory functions to treat PCa. We may first use cells, tissues, and PCa patient-derived xenograft mice models to verify the immune regulatory function of
GLIS1 in PCa and further clarify its molecular mechanism involved in immune regulation. In vitro and in vivo experiments will help us detect the efficacy of
GLIS1 in enhancing PCa immunotherapy, such as the potential synergistic effect in combination with immune checkpoint inhibitors in PCa immunotherapy. The confirmation of in vitro and in vivo experiments will help us conduct human trials to explore the combined effect of GLIS1 monoclonal antibodies with anti-PD1 or anti-CTLA-4 for future PCa treatment.
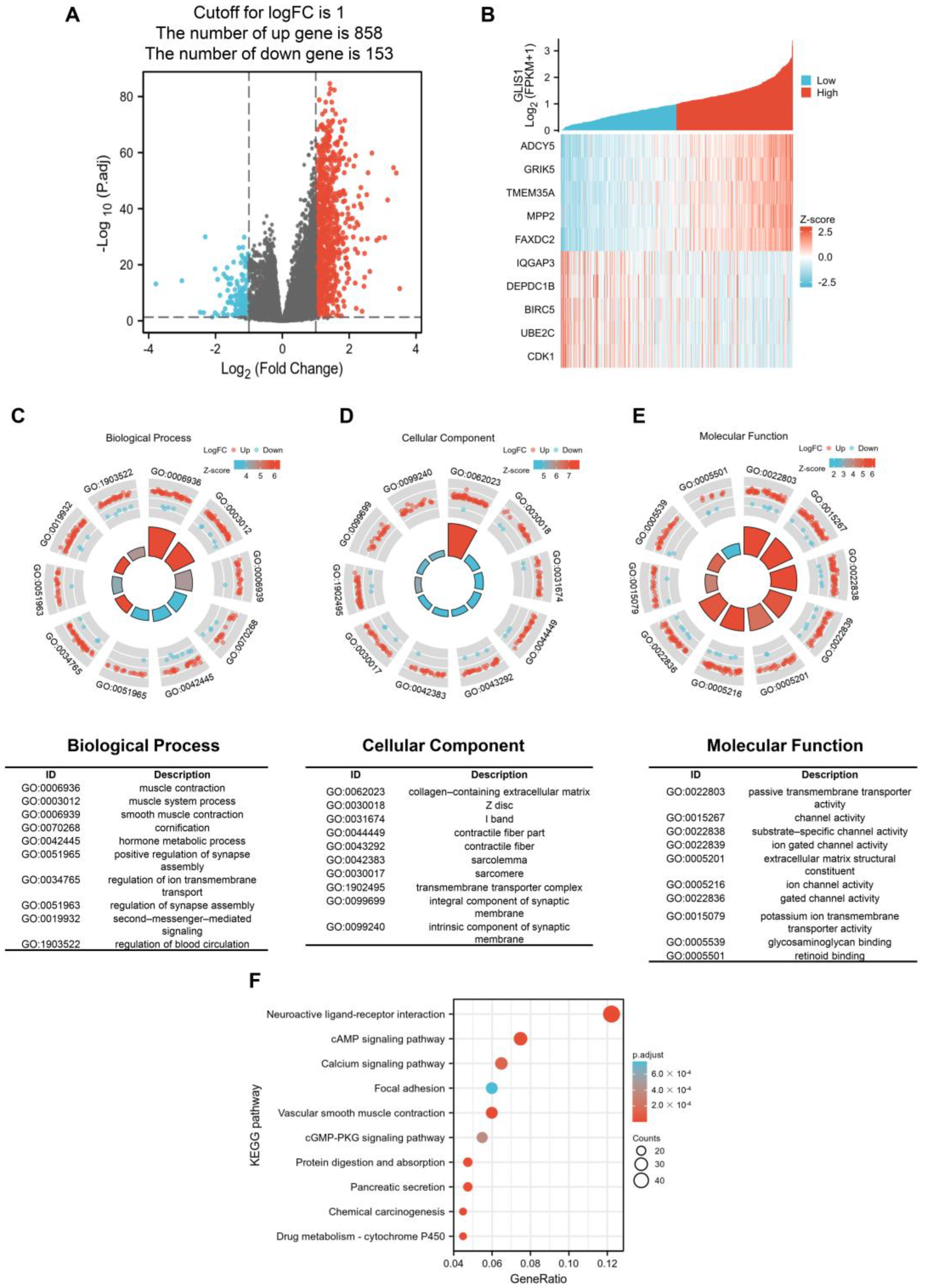
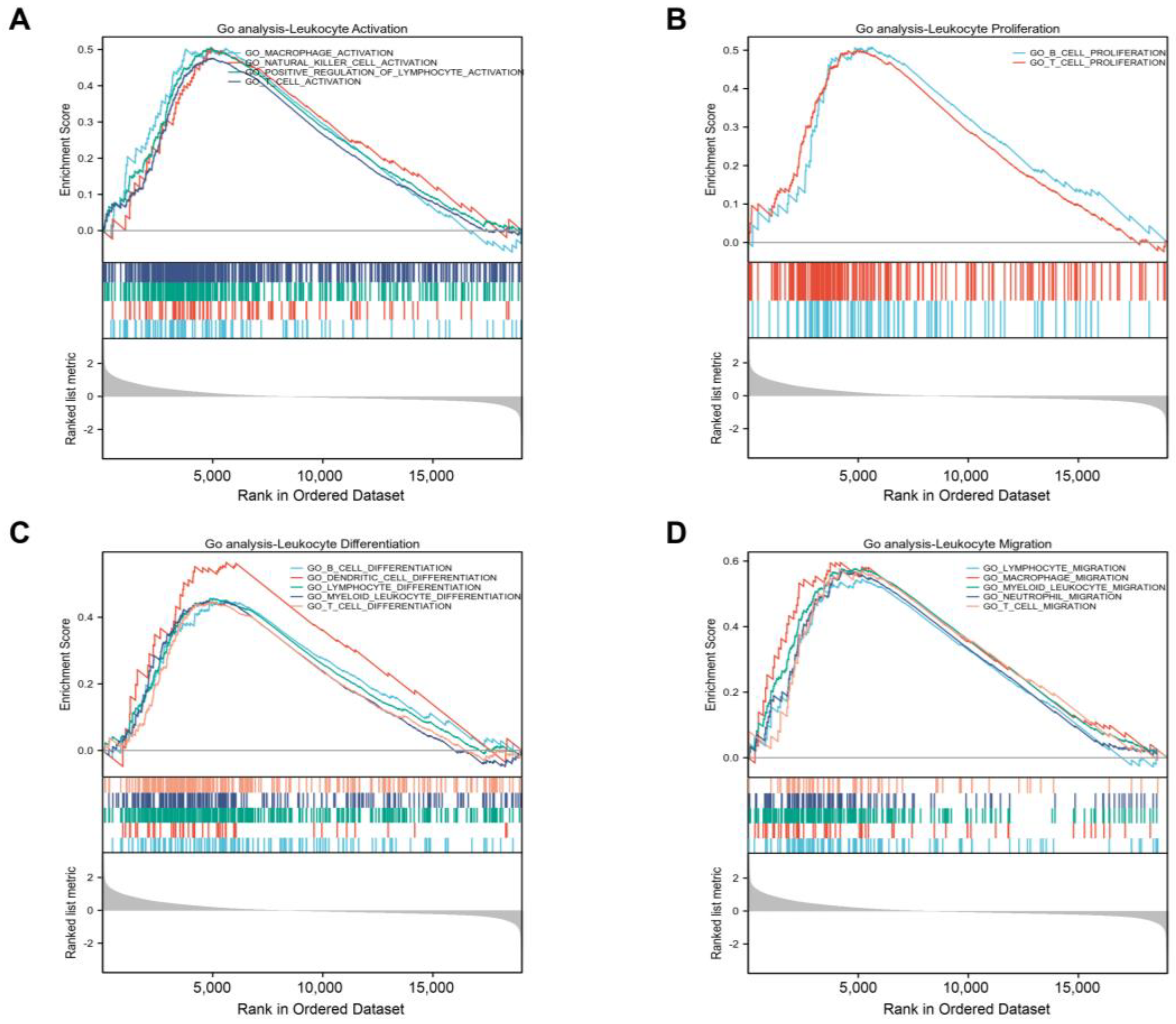
Concerning GO enrichment analysis,
GLIS1 was found to be involved in several signaling pathways in tumor cells, which is similar to the GO analysis results of tDEGs in PRAD (
Figure 4C–E and
Figure S1A), revealing a high degree of correlation regulation. For example, muscle contraction, and muscle system process for BP, collagen-containing extracellular matrix and contractile fiber for CC, and passive transmembrane transporter activity and channel activity for MF. Furthermore, the GSEA on GLIS-related immune function showed that
GLIS1 affected the activation, proliferation, differentiation, and migration of immune cells. It suggests that
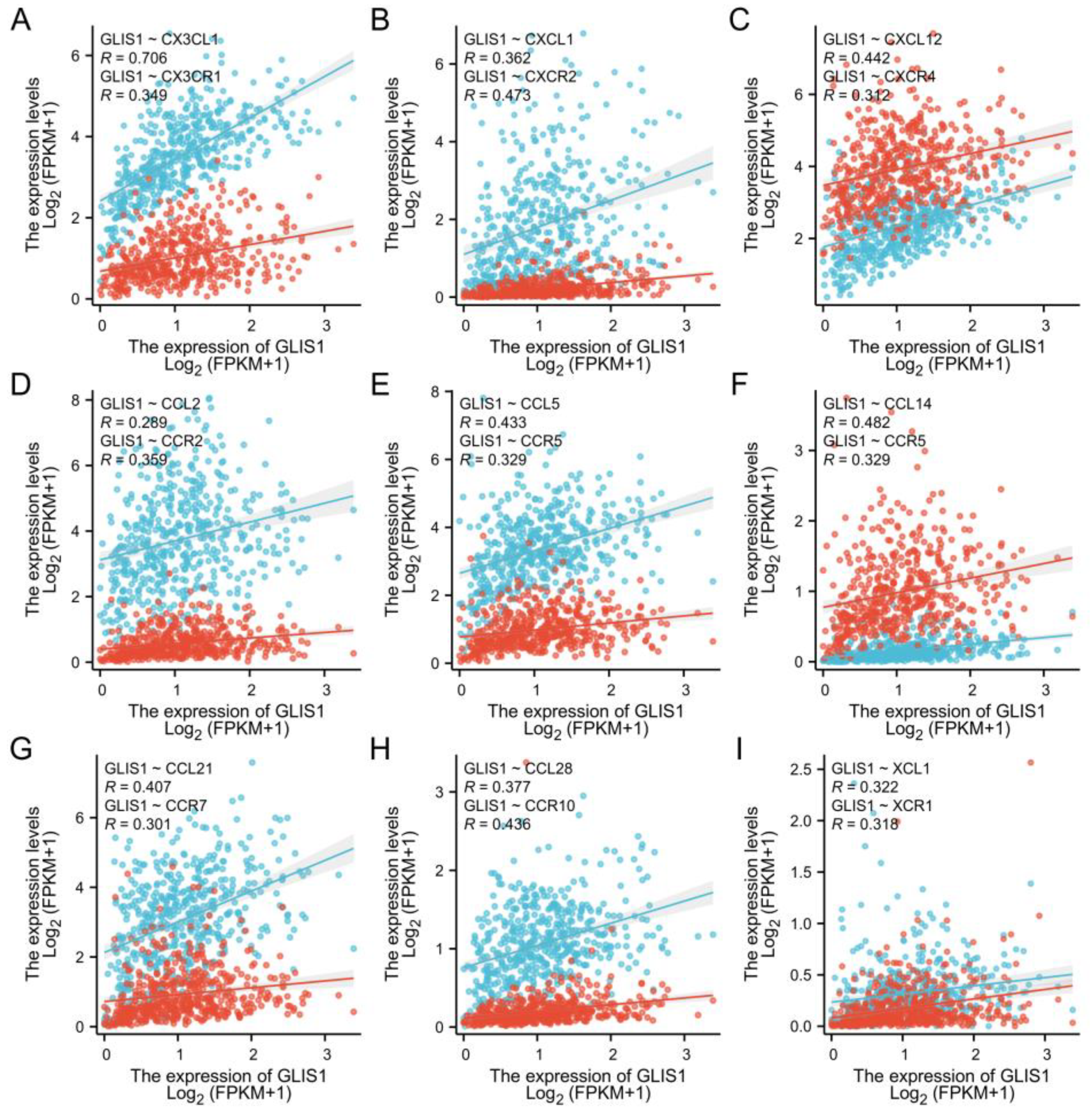
GLIS1 enhances the immunological response of PCa through multiple pathways. Chemokines regulate the positioning and motility patterns of immune cells and are essential for immune cell migration and TME homeostasis. Subsequently, we combined chemokines and chemokine receptors and examined the correlation between
GLIS1 and the migration of immune cells to investigate the potential immunological-related mechanism of
GLIS1 in PCa. The interaction between
CX3CL1 and
CX3CR1 is involved in the recruitment of NK cells, T cells, and monocytes, and is associated with the activation of cytotoxic T lymphocytes and NK cells [
45].
CCL5/
CCR5 plays a role in the migration of macrophage and NK cells, as well as in the interactions between T cells and DC cells [
46].
CXCL1/
CXCR2 and
CXCL12/
CXCR4 are related to neutrophil recruitment and NK cell migration, respectively [
47,
48].
CCL21/
CCR7 plays important roles in T cell recruitment to lymph nodes and in T cell migration [
49].
XCL1/
XCR1 interaction is associated with NK cell recruitment [
50]. In this study,
GLIS1 was positively associated with these chemokines/chemokine receptors, indicating that
GLIS1 may enhance tumor immune infiltration levels via modulating the migration of immune cells in PCa.
Though this study enhances our comprehension of the association between GLIS1 and PCa, it does have several limitations. Firstly, it is necessary to conduct molecular investigations to validate the mechanisms of GLIS1 and its influence on the clinical outcome of PCa. Furthermore, critical signaling pathways related to GLIS1 need to be validated. In addition, the mechanism by which GLIS1 regulates immune cell infiltration remains to be studied. It is also crucial to incorporate and clarify the connection between GLIS1 and chemokines/chemokine receptors, which can enhance our comprehension about TME, specifically the immune microenvironment in PCa.
In conclusion, this is the first comprehensive study to elaborate the association between the expression of GLIS1 and its clinical, molecular, and immunotherapeutic characteristics in PCa, providing us new insights into the important role of GLIS1 in PCa. Our work has found that the expression of GLIS1 varies between normal tissue and tumor tissues in various cancer types and is significantly downregulated in PCa, and decreased GLIS1 expression is associated with unfavorable prognosis in PCa. GLIS1 also plays a crucial role in the microenvironment of PCa by controlling the infiltration of immune cells into tumors, indicating GLIS1 as a potential therapeutic target to regulate immune responses against tumors. Consequently, GLIS1 shows considerable promise as a helpful biomarker and therapeutic target for PCa prognosis prediction and treatment.