Abstract
Intermediate filaments are one of three polymeric structures that form the cytoskeleton of epithelial cells. In the epithelium, these filaments are made up of a variety of keratin proteins. Intermediate filaments complete a wide range of functions in keratinocytes, including maintaining cell structure, cell growth, cell proliferation, cell migration, and more. Given that these functions are intimately associated with the carcinogenic process, and that hyperkeratinization is a quintessential feature of oral leukoplakias, the utility of keratins in oral leukoplakia is yet to be fully explored. This scoping review aims to outline the current knowledge founded on original studies on human tissues regarding the expression and utility of keratins as diagnostic, prognostic, and predictive biomarkers in oral leukoplakias. After using a search strategy developed for several scientific databases, namely, PubMed, Scopus, Web of Science, and OVID, 42 papers met the inclusion and exclusion criteria. One more article was added when it was identified through manually searching the list of references. The included papers were published between 1989 and 2024. Keratins 1–20 were investigated in the 43 included studies, and their expression was assessed in oral leukoplakia and dysplasia cases. Only five studies investigated the prognostic role of keratins in relation to malignant transformation. No studies evaluated keratins as a diagnostic adjunct or predictive tool. Evidence supports the idea that dysplasia disrupts the terminal differentiation pathway of primary keratins. Gain of keratin 17 expression and loss of keratin 13 were significantly observed in differentiated epithelial dysplasia. Also, the keratin 19 extension into suprabasal cells has been associated with the evolving features of dysplasia. The loss of keratin1/keratin 10 has been significantly associated with high-grade dysplasia. The prognostic value of cytokeratins has shown conflicting results, and further studies are required to ascertain their role in predicting the malignant transformation of oral leukoplakia.
1. Introduction
Intermediate filaments (IF) are one of three polymeric networks found in eukaryotic cells. They are 10 nm in diameter and composed of numerous proteins encoded by various genes [1]. Intermediate filaments can be categorized into several subtypes based on the proteins that constitute them [2], namely, Class I (keratins a), Class II (keratins b), Class III (vimentin, desmin, and more), Class IV (nestin and more), Class V (lamins), and Class VI (filensin and more) [3]. Each protein is expressed in various cell types. For example, vimentin is found in mesenchymal cells, and nestin is found in glial cells [3]. Class I and Class II IF proteins are found in epithelial cells and were originally catalogued and termed cytokeratins by Moll in 1982 [4]. Schweizer further refined this in 2006 [5]; whereby he largely appreciated Moll’s system [4], but reclassified cytokeratins into the now recommended nomenclature of keratin groups. For human epithelium, these were categorized as human type I epithelial keratins, which include K9–K28, and human type II epithelial keratins, which include K1–K8 and K71–K80 [5].
At least one member of type I or type II keratin must be present to form the heteropolymeric structure that makes up the IF. This pairing leads to a highly stabilized polymer compared to its monomer form, which is easily broken down. This keratin pairing has been found to be varied within the same cell family type, between different cell types, tissues, and more. Consequently, they fulfil a wide range of functions, including maintaining cell structure, cell integrity, cell adhesion, cell growth, cell proliferation, and cell migration [3]
Primary keratins that are produced consistently in normal oral epithelium include K4/K13, which are found suprabasally in the non-keratinizing oral epithelium and K1/K10, which are found suprabasally in the keratinizing oral epithelium [6,7,8,9,10,11]. K5/K14 are found in the basal layer of both th keratinizing and non-keratinizing oral epithelium [4,6,8]. Another notable keratin co-expressed suprabasally is K76 (previously K2p), which has been found in the gingiva and palate, both keratinized oral epithelium [9,12]. K8, K18, and K19 have been found localized to Merkel cells in the oral epithelium [4]. K19 is also found in the basal cells in non-keratinizing mucosa [6,13]. K20 has been found to be a marker of taste buds and Merkel cells as well [14].
Of particular interest in the oral context is oral leukoplakia (OLK). The World Health Organization (WHO) defines OLK as a predominantly white plaque of questionable risk, having excluded (other) known diseases or disorders that carry no increased risk of cancer [15]. It is classified as an potentially malignant oral disorder [16]. Prominent histological features of OLK include hyperplasia with hyperkeratosis (ortho or para), with or without dysplasia [16]. Notably, there have been multiple studies that have reported altered keratin expression in OLKs [17,18,19].
Given the above, in combination with keratins role in the carcinogenic process and its antigenic stability [20], keratins remain an untapped source of potential in the diagnosis and management of OLKs. This is notably seen in a variety of other cancers, such as renal cell carcinomas, breast adenocarcinomas [21], salivary gland malignancies [22], and metastatic cancers [23]. The utility of biomarkers in oncology can be diagnostic, prognostic, or predictive. A diagnostic biomarker may be used to identify the presence of disease and also subtyping (e.g., cancer type). A prognostic biomarker identifies patients’ disease progression or recurrence (e.g., cancer prognosis) with or without treatment. A predictive biomarker identifies patients who are more likely to benefit from a particular treatment [24].
In the context of keratins, the utility of keratins in OLKs may be diagnostic; for example, they may be used as an adjunct for the detection grading of dysplasia. Keratins may be used as a prognostic tool, for example, in predicting malignant transformation (MT). They may also be utilized as a predictive tool with which to identify the ideal treatment [25]. Hence, this manuscript aims to review the current knowledge of keratins expressed in OLK and their potential roles (e.g., diagnostic, prognostic, or predictive). This is the first scoping review to unravel the expression of keratins in oral leukoplakia and evaluate their diagnostic, prognostic, and predictive values.
2. Methods
The protocol was registered at the Open Science Framework, (registration https://doi.org/10.17605/OSF.IO/P2VXS) (accessed on 5 April 2024).
2.1. Eligibility Criteria
Eligibility criteria are outlined in Table 1 as guided by PECOS (participants, exposure, comparators, and study designs) based on the aims of this scoping review. As such, all relevant studies were filtered based on these predefined criteria, resulting in select studies being included in this scoping review.

Table 1.
PECOS criteria for inclusion and exclusion of studies.
2.2. Data Sources and Search Strategy I
Two authors (GO and JL) independently searched the following electronic databases: PubMed, OVID, SCOPUS, and Web of Science. No lower date limits were set, and the upper date limit was set to February 2024. The latest search was conducted on the 7th of February 2024. To increase the sensitivity of search results, searches in MeSH and free terms were utilized. The core search strategy included ‘Cytokeratin’ + ‘Location’ + ‘Pathology’, whereby location included the various sites in the oral cavity (buccal mucosa, tongue, palate, gingiva, etc.) that was searchable in the respective database, and pathology referred to leukoplakia or dysplasia or carcinoma in situ. Fields of search included ‘Title’, ‘Abstract’, ‘Keywords’, and ‘Author-specified keywords’, which were modified for each search database accordingly. A complete script of the search strategy in various databases is included in Supplementary Materials S1. Endnote X9.3.3 was utilized to manage references and duplicates. The retrieved records were also manually scanned by the authors after endnote removed duplicates to ensure all duplicates were removed.
2.3. Study Selection and Screening I
Two investigators (GO and JL) independently evaluated the articles retrieved from the databases. The first round of evaluation was performed by reviewing the title and abstract of the studies. The remaining studies were then considered suitable for the final round of eligibility assessment, which involved reading the full text, after which the final list of eligible studies remained (Figure 1). The reference list of eligible studies was also scanned for eligible studies. The included studies were cross-referenced between the authors at every stage, and any disagreements were resolved by further review and discussion among three reviewers (GO, JL, and OK).
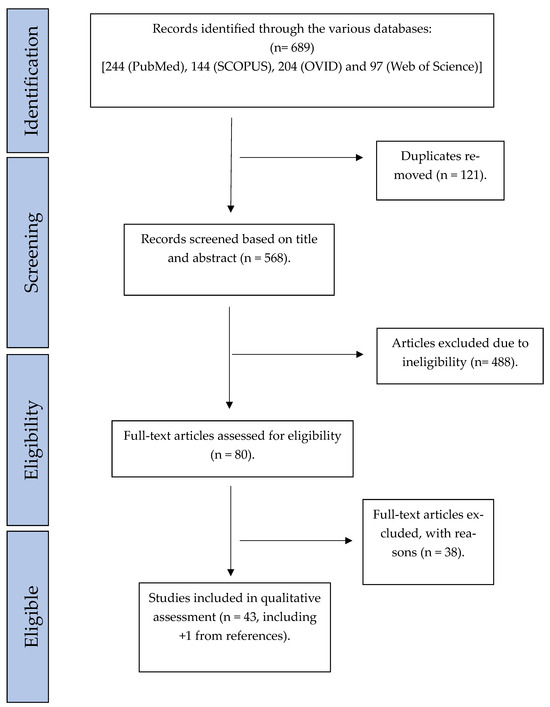
Figure 1.
Flowchart representing systematic literature search.
2.4. Data Extraction and Synthesis
Two reviewers (GO and JL) independently retrieved data from the included studies. A senior reviewer (OK) supervized the process and verified the extracted information. Data extracted were compiled using a standardized method using Microsoft Excel v. 365. Domains collected included authors, year, title, DOI, main keratin investigated, study’s aim, type of study, tissue site, storage of the sample, methodology for assessment of keratin, statistical analysis employed, dysplasia grading system, and study findings for 20 studies individually as a pilot round. After discussing with all 3 reviewers, data items collected included author, year, tissue of interest and site, controls, dysplasia grading system, if statistical analysis was completed, methodology of analysis of keratin expression, and study type (which included expression, diagnostic, prognostic, or predictive). Expression refers to the study being aimed to investigate the expression patterns and or changes in expression of keratin for that particular condition. This may have included increase/decrease in protein staining intensity, increase/decrease in number of cells stained. and increase/decrease in samples stained for that particular keratin. These were independently retrieved by 2 reviewers (GO and JL), and a senior reviewer (OK) verified the extracted information. Some studies included additional ‘non-keratin’ biomarkers, cell line studies, and non-oral sites. Only the relevant data consistent with the inclusion and exclusion criteria were extracted.
Results were synthesized and presented on a keratin pair (if applicable) or on a per-keratin basis. Overall, these were divided into keratin protein studies and mRNA studies. Within each keratin/keratin pair group, results were divided into the type of study (e.g., expression, prognosis). The general trend within each group was based on the highest number of studies supporting a particular pattern, for which studies against this trend were also discussed. Additional pertinent data for each trend were also discussed when required.
3. Results
A total of 689 studies were found, of which 121 duplicates were removed. Records were screened based on titles and abstracts, whereby 488 were removed. Eighty studies were assessed for eligibility (Figure 1). Of these, 42 studies fulfilled the inclusion and exclusion criteria, and 1 additional article was included, which was found by searching the reference list of the articles [26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68] (Table 2). The year of publication ranged from 1989 to 2024. A total of 2461 tissue samples from leukoplakia with or without dysplasia were compared to approximately 352 normal tissues/controls (some studies did not specify the number of normal specimens used as controls). Sample sizes varied from as low as 6 [31] to 200 [40] in the OLK/dysplasia group, with an average of 57 samples per study (median = 40). As many as 17 out of the 43 studies did not perform statistical testing. Forty-two studies utilized hematoxylin and eosin (H&E) and immunohistochemistry (IHC), and one study only used mRNA analysis without H&E [58]. In addition to H&E and IHC, seven studies [29,30,39,58,60,61,68] used in situ hybridization (ISH) to detect mRNA expression in their samples, and one study [68] used utilized reverse transcriptase polymerase chain reaction (RT-PCR) and electrophoresis to analyze mRNA expression levels between their groups. Additionally, three studies used gel electrophoresis and immunoblotting [33,57,63]. Seven different dysplasia grading systems were represented. Nine studies [31,32,33,38,41,42,43,52,56] used WHO 1978 [69], two studies [55,68] used WHO 1997 [70], seven studies [27,36,45,48,50,53,62] used WHO 2005 [71], five studies [28,40,54,65,66] used WHO 2017 [72], two studies [36,67] used SIN system [71], two studies [46,61] used Kramer 1980 [73], and one study [64] used Grassel-Pietrusky and Hornstein 1982 [74] (Table 2). Six studies [26,29,30,34,39,58] graded dysplasia without providing details of the criteria used, and seven studies [35,37,44,49,51,59,60] identified the presence of dysplasia without providing the details of the criteria used. Three studies did not assess for dysplasia at all [47,57,63]. Most studies utilized semi-quantitative assessment of staining intensity (Table 2). Two studies utilized automated systems to quantify IHC staining [37,52], and nine studies analyzed IHC concerning the various layers of the epithelium [27,31,34,42,43,46,51,62,64].

Table 2.
Overview of studies on keratin expression in oral leukoplakia.
All 43 studies explored various keratins and their expression in OLK and dysplasia, and 5 investigated their prognostic potential with regard to MT [26,28,65,66,67]. No studies evaluated keratins as a diagnostic adjunct or predictive tool. Keratins 1–20 are represented in the included studies (Table 2).
Regarding the prognostic studies, the first of which [51] investigated K4/K13 and had a follow up time ranging from 1 to 173 months (mean of 69 months and median of 61 months). The second prognostic study [67] investigated K13 and K17 and had a follow-up time ranging from 1 to 155 months (with a mean of 50.4 months and no median reported). The third study [65] investigated K13 and K17, with a follow up time of 11–183 months for patients who had MT of their OLK (median 51 months) and 109–258 months (median 148 months) for their non-progressors. The fourth study [66] investigated K13 and K17 as well, with a follow up time of 12–300 months (median 63 months). The fifth study [28] investigated K13 and K17, with a follow up time of 4–290 months (median 52 months).
4. Discussion
This scoping review explored the keratin expression in OLKs and their potential utility. Despite the heterogeneity of the studies, general trends of keratin expression can be observed. It must be emphasized that these are trends, and conflicting results of various studies are presented in Table 3, with direct comparisons being quite difficult. Nonetheless, these trends can be broadly summarized as follows (Table 3):

Table 3.
General trends of keratins protein in oral leukoplakia.
- Loss of expression of primary keratins normally found in oral epithelium. Specifically, K1/K10 and K4/K13 suprabasally in keratinized and non-keratinized epithelium. This is also true for K2p for keratinized mucosa. This suggests that dysplasia disrupts the terminal differentiation pathway of primary keratins.
- Suprabasal extension of primary keratins of the basal layer, which include K5/K14 and K19. However, K19 may also express atypically (suprabasal extension) in inflammatory lesions.
- The gain of K17 expression in OLK samples, which is the most studied keratin with respect to the increase in protein expression. Aside from the above, K8 and K18 have also shown increased expression, albeit in a smaller proportion of samples compared to K17.
None of the studies have managed to investigate the effectiveness of keratin as a diagnostic utility; that is, the improvement of inter- and intra-observer reliability with the utility of keratin is needed. This has been recently achieved in sites involving the upper aerodigestive tract, whereby the authors utilized the suprabasal extension of K19 to improve inter- and-intra observer reliability for the diagnosis of dysplasia [75]. The closest study in this review to achieving this was Becker [28], who reportedly used K13 and K17 to improve diagnosis of dysplasia. However, data on intra- or inter-observer variability improvements were not provided.
- Nonetheless, from this review, further studies on the following keratins may be helpful in the following scenarios (high-grade vs. low-grade dysplasia):
- LOE of K1/K10.
- There is a marked loss of K1/K10 expression in severe dysplasia (regardless of original site of OLK samples, e.g., keratinized or non-keratinized).
- Suprabasal extension of K19 to the most superficial layers of epithelium indicates severe or high-grade dysplasia.
- Prognostic utility:
- GOE of K17.
- Retention of K13 expression may indicate a lower risk of MT.
- Further studies investigating the significance of GOE for K6, K7, K8, K16, and K18 in OLK. Current evidence shows that not many studies stain for these keratins in their samples, and as such, data are not hampered by the quality but rather by quantity of studies.
Additionally, there were some notable exclusions from our review as they did not fit the inclusion and exclusion criteria. Five of these had samples which were taken adjacent to OSCC samples, which are at risk of exhibiting a different genetic signature and possibly phenotype to traditional OLKs [17,18,19,76,77]. One was non-specific for keratin antibodies [78], and for another two, the methodology was not consistent with the aims of the paper (e.g., aiming to prove a new entity or validate a new technique [79,80]). Most of these studies found results similar to the trends found. However, of particular interest was Khanom’s study [19], which found a K15 decreasing expression in basal cells of dysplastic samples as the grade of dysplasia worsened, similar to Sakamoto’s study [53]. However, they also had hyperplasia samples, for which K19 showed altered expression (suprabasal extension). In contrast, K15 largely retained its staining, prompting the authors to conclude that it was a more stable keratin to utilize as a diagnostic adjunct compared to K19.
Furthermore, it must be noted that most studies focused on the protein expression of keratins, likely due to their ease of staining. However, it may be beneficial to analyze mRNA expressions (Table 4) as the protein expression may be suppressed by post-transcriptional mechanisms. As such, the detection of mRNA may yield more accurate results.

Table 4.
General trends of keratin mRNA expression in oral leukoplakia.
It is interesting to note that with regards to gain of expression, the K17 has the most evidence in the context of OLK which is also reflected in the wider literature regarding OSCC. K17 has been extensively researched in a variety of cancers and has established oncogenic roles [81]. Squamous cell carcinomas of the oral cavity have been shown to have increased expression of K17, with some clinical studies finding prognostic implications for OSCC [82]. Several authors have attempted to investigate the mechanistic role of K17 in oncogenesis via in vitro OSCC cell line studies. Mikami [83] found that in ZK-1 cell lines with K17 knockout, the loss of keratin did not affect cell migration or invasion but decreased in cell size as compared to normal controls with retained K17. In Khanom’s in vitro study, they found that K17 stimulated Akt/mTOR pathway and glucose uptake, which supports its role in tumor growth. In Mikami’s 2017 study [84], knockout of K17 increased the number of cleaved caspase-3-positive HSC-2 cells, which are involved in apoptosis. As such, their conclusion advised that K17’s oncogenic role in OSCC tumor growth may not only be through the Akt/mTOR pathway but also the suppression of apoptosis.
The main limitation of this study is the lack of quantitative synthesis attributed to the significant heterogeneity between studies reviewed, even when assessing the state of knowledge for one keratin. Every study had a slightly varying methodology, comparison groups (or lack thereof), staining techniques, source of monoclonal antibodies, assessment protocols, populations, and more (Table 2). As such, the level of evidence was difficult to ascertain, hence the publication of a scoping review on this emerging topic. Ultimately, prospective cohort studies assessing keratin as a prognostic or predictive biomarker would yield the highest evidence. Inter- and intra-observer comparison studies would help with its diagnostic utility.
However, many questions still remain with respect to the utility of keratins in OLKs. In which context should the biomarker be used? Should it be used to distinguish between dysplasia and hyperkeratosis? Should it be used to aid in the differentiation of various grades of dysplasia? What instrument/tool/methodology will be used to assess keratins? Is keratin best assessed through the percentage of positive cells or through staining intensity and assessed by layers? The keratins and their suggested utility, as outlined in this review, require further research for validation.
5. Conclusions
The use of keratin as a biomarker in OLKs has untapped potential. Further research is necessary to fully elucidate the roles keratins may play as, to date, most studies have only focused on the expression of keratins in OLKs, with limited studies investigating the diagnostic, prognostic, and predictive utility of keratins. The current state of knowledge suggests that the loss of expression of primary keratins K4/K13, K1/K10, and K2p/K76 suprabasally can be investigated for their use as a diagnostic adjunct. The suprabasal spread of K19 and LOE of K15 basally may also have potential use as a diagnostic adjunct. The superficial spread of K19 may also be used to distinguish between high-grade and low-grade dysplasia. Furthermore, retention of K13 in OLK samples without dysplasia may have prognostic potential. There is evidence that the gain of expression of K17 may have some prognostic utility. The significance of protein expression of K8, K18, K6, and K16 in a small number of OLK samples should also be explored as there is a lack of studies investigating these keratins.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms25115597/s1, Table S1: Search Strategies.
Author Contributions
Conceptualization, O.K.; methodology, G.M.O.; validation, J.L. and O.K.; formal analysis, G.M.O. and J.L.; investigation, G.M.O., J.L. and O.K.; resources, R.B.; data curation, G.M.O; writing—original draft preparation, G.M.O.; writing—review and editing, R.B., J.L., A.M.F. and O.K.; supervision, A.M.F., R.B. and O.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Peter, A.; Stick, R. Evolutionary aspects in intermediate filament proteins. Curr. Opin. Cell Biol. 2015, 32, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Guzenko, D.; Chernyatina, A.A.; Strelkov, S.V. Crystallographic Studies of Intermediate Filament Proteins. Fibrous Proteins Struct. Mech. 2017, 82, 151–170. [Google Scholar] [CrossRef] [PubMed]
- Etienne-Manneville, S. Cytoplasmic Intermediate Filaments in Cell Biology. Annu. Rev. Cell Dev. Biol. 2018, 34, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Moll, R.; Franke, W.W.; Schiller, D.L.; Geiger, B.; Krepler, R. The catalog of human cytokeratins: Patterns of expression in normal epithelia, tumors and cultured cells. Cell 1982, 31, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, J.; Bowden, P.E.; Coulombe, P.A.; Langbein, L.; Lane, E.B.; Magin, T.M.; Maltais, L.; Omary, M.B.; Parry, D.A.; Rogers, M.A.; et al. New consensus nomenclature for mammalian keratins. J. Cell Biol. 2006, 174, 169–174. [Google Scholar] [CrossRef]
- Moll, R.; Divo, M.; Langbein, L. The human keratins: Biology and pathology. Histochem. Cell Biol. 2008, 129, 705–733. [Google Scholar] [CrossRef] [PubMed]
- Bragulla, H.H.; Homberger, D.G. Structure and functions of keratin proteins in simple, stratified, keratinized and cornified epithelia. J. Anat. 2009, 214, 516–559. [Google Scholar] [CrossRef] [PubMed]
- Morgan, P.R.; Shirlaw, P.J.; Johnson, N.W.; Leigh, I.M.; Lane, E.B. Potential applications of anti-keratin antibodies in oral diagnosis. J. Oral Pathol. 1987, 16, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Bosch, F.X.; Ouhayoun, J.P.; Bader, B.L.; Collin, C.; Grund, C.; Lee, I.; Franke, W.W. Extensive changes in cytokeratin expression patterns in pathologically affected human gingiva. Virchows Arch. B Cell Pathol. Incl. Mol. Pathol. 1989, 58, 59–77. [Google Scholar] [CrossRef]
- Ouhayoun, J.P.; Gosselin, F.; Forest, N.; Winter, S.; Franke, W.W. Cytokeratin patterns of human oral epithelia: Differences in cytokeratin synthesis in gingival epithelium and the adjacent alveolar mucosa. Differentiation 1985, 30, 123–129. [Google Scholar] [CrossRef]
- Clausen, H.; Moe, D.; Buschard, K.; Dabelsteen, E. Keratin proteins in human oral mucosa. J. Oral Pathol. 1986, 15, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Collin, C.; Ouhayoun, J.P.; Grund, C.; Franke, W.W. Suprabasal marker proteins distinguishing keratinizing squamous epithelia: Cytokeratin 2 polypeptides of oral masticatory epithelium and epidermis are different. Differentiation 1992, 51, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Bienengraber, V.; Jonas, L. Immunohistological detection of cytokeratins in oral epithelia by poly- and monoclonal antibodies. Z. Mikrosk. Anat. Forsch. Abt. 2 1988, 102, 708–710. [Google Scholar]
- Barrett, A.W.; Cort, E.M.; Patel, P.; Berkovitz, B.K.B. An immunohistological study of cytokeratin 20 in human and mammalian oral epithelium. Arch. Oral Biol. 2000, 45, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Warnakulasuriya, S.; Johnson, N.W.; van der Waal, I. Nomenclature and classification of potentially malignant disorders of the oral mucosa. J. Oral Pathol. Med. 2007, 36, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Warnakulasuriya, S.; Kujan, O.; Aguirre-Urizar, J.M.; Bagan, J.V.; González-Moles, M.; Kerr, A.R.; Lodi, G.; Mello, F.W.; Monteiro, L.; Ogden, G.R.; et al. Oral potentially malignant disorders: A consensus report from an international seminar on nomenclature and classification, convened by the WHO Collaborating Centre for Oral Cancer. Oral Dis. 2021, 27, 1862–1880. [Google Scholar] [CrossRef] [PubMed]
- Matsuhira, A.; Noguchi, S.; Sato, K.; Tanaka, Y.; Yamamoto, G.; Mishima, K.; Katakura, A. Cytokeratin 13, Cytokeratin 17, Ki-67 and p53 Expression in Upper Layers of Epithelial Dysplasia Surrounding Tongue Squamous Cell Carcinoma. Bull. Tokyo Dent. Coll. 2015, 56, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, S.; Sato, K.; Yamamoto, G.; Tonogi, M.; Tanaka, Y.; Tachikawa, T.; Yamane, G.-y. Expression of cytokeratin 13 and 17 in tongue squamous cell carcinoma and epithelial dysplasia. Asian J. Oral Maxillofac. Surg. 2011, 23, 53–58. [Google Scholar] [CrossRef]
- Khanom, R.; Sakamoto, K.; Pal, S.K.; Shimada, Y.; Morita, K.; Omura, K.; Miki, Y.; Yamaguchi, A. Expression of basal cell keratin 15 and keratin 19 in oral squamous neoplasms represents diverse pathophysiologies. Histol. Histopathol. 2012, 27, 949–959. [Google Scholar]
- Linder, S. Cytokeratin markers come of age. Tumour. Biol. 2007, 28, 189–195. [Google Scholar] [CrossRef]
- Karantza, V. Keratins in health and cancer: More than mere epithelial cell markers. Oncogene 2011, 30, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Swid, M.A.; Li, L.; Drahnak, E.M.; Idom, H.; Quinones, W. Updated Salivary Gland Immunohistochemistry: A Review. Arch. Pathol. Lab. Med. 2023, 147, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Selves, J.; Long-Mira, E.; Mathieu, M.C.; Rochaix, P.; Ilié, M. Immunohistochemistry for Diagnosis of Metastatic Carcinomas of Unknown Primary Site. Cancers 2018, 10, 108. [Google Scholar] [CrossRef] [PubMed]
- Califf, R.M. Biomarker definitions and their applications. Exp. Biol. Med. 2018, 243, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A.; Bradley, M.D.; Elyan, S.; Kurian, K.M. Tumour biomarkers: Diagnostic, prognostic, and predictive. BMJ Br. Med. J. 2015, 351, h3449. [Google Scholar] [CrossRef] [PubMed]
- Ambatipudi, S.; Bhosale, P.G.; Heath, E.; Pandey, M.; Kumar, G.; Kane, S.; Patil, A.; Maru, G.B.; Desai, R.S.; Watt, F.M.; et al. Downregulation of keratin 76 expression during oral carcinogenesis of human, hamster and mouse. PLoS ONE 2013, 8, e70688. [Google Scholar] [CrossRef] [PubMed]
- Barakat, S.M.; Siar, C.H. Differential expression of stem cell-like proteins in normal, hyperplastic and dysplastic oral epithelium. J. Appl. Oral Sci. 2015, 23, 79–86. [Google Scholar] [CrossRef][Green Version]
- Becker, A.S.; Holm, M.; Liese, J.; Engel, N.; Zimpfer, A.H. Diagnosis of differentiated dysplasia as a variant of oral epithelial dysplasia. Oral Dis. 2024. [Google Scholar] [CrossRef] [PubMed]
- Bloor, B.K.; Seddon, S.V.; Morgan, P.R. Gene expression of differentiation-specific keratins in oral epithelial dysplasia and squamous cell carcinoma. Oral Oncol. 2001, 37, 251–261. [Google Scholar] [CrossRef]
- Bloor, B.K.; Tidman, N.; Leigh, I.M.; Odell, E.; Dogan, B.; Wollina, U.; Ghali, L.; Waseem, A. Expression of keratin K2e in cutaneous and oral lesions: Association with keratinocyte activation, proliferation, and keratinization. Am. J. Pathol. 2003, 162, 963–975. [Google Scholar] [CrossRef]
- Cema, I.; Stengel, B.; Nizze, H. Expression of cytokeratins in oral dysplasias and squamous cell carcinomas. Proc. Latvian Acad. Sci. Sect. B Nat. Exact Appl. Sci. 1998, 52, 119–125. [Google Scholar]
- Cintorino, M.; Petracca, R.; Vindigni, C.; Tripodi, S.A.; Leoncini, P. Topography-related expression of individual cytokeratins in normal and pathological (non-neoplastic and neoplastic) human oral mucosa. Virchows Arch. A Pathol. Anat. Histopathol. 1990, 417, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Ermich, T.; Schulz, J.; Raabe, G.; Schumann, D. Pattern of oral cytokeratins. III. SDS-electrophoretic analysis and immunoblotting of cytokeratins in leukoplakias and squamous cell carcinoma of the oral mucosa. Biomed. Biochim. Acta 1989, 48, 393–401. [Google Scholar]
- Farrar, M.; Sandison, A.; Peston, D.; Gailani, M. Immunocytochemical analysis of AE1/AE3, CK 14, Ki-67 and p53 expression in benign, premalignant and malignant oral tissue to establish putative markers for progression of oral carcinoma. Br. J. Biomed. Sci. 2004, 61, 117–124. [Google Scholar] [CrossRef]
- Farrukh, S.; Syed, S.; Pervez, S. Differential Expression of Cytokeratin 13 in Non-Neoplastic, Dysplastic and Neoplastic Oral Mucosa in a High Risk Pakistani Population. Asian Pac. J. Cancer Prev. 2015, 16, 5489–5492. [Google Scholar] [CrossRef] [PubMed]
- Fillies, T.; Jogschies, M.; Kleinheinz, J.; Brandt, B.; Joos, U.; Buerger, H. Cytokeratin alteration in oral leukoplakia and oral squamous cell carcinoma. Oncol. Rep. 2007, 18, 639–643. [Google Scholar] [CrossRef][Green Version]
- Gires, O.; Mack, B.; Rauch, J.; Matthias, C. CK8 correlates with malignancy in leukoplakia and carcinomas of the head and neck. Biochem. Biophys. Res. Commun. 2006, 343, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Heyden, A.; Huitfeldt, H.S.; Koppang, H.S.; Thrane, P.S.; Bryne, M.; Brandtzaeg, P. Cytokeratins as epithelial differentiation markers in premalignant and malignant oral lesions. J. Oral Pathol. Med. 1992, 21, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Ida-Yonemochi, H.; Maruyama, S.; Kobayashi, T.; Yamazaki, M.; Cheng, J.; Saku, T. Loss of keratin 13 in oral carcinoma in situ: A comparative study of protein and gene expression levels using paraffin sections. Mod. Pathol. 2012, 25, 784–794. [Google Scholar] [CrossRef]
- Ikeda, M.; Shima, K.; Kondo, T.; Semba, I. Atypical immunohistochemical patterns can complement the histopathological diagnosis of oral premalignant lesions. J. Oral Biosci. 2020, 62, 93–98. [Google Scholar] [CrossRef]
- Kannan, S.; Balaram, P.; Chandran, G.J.; Pillai, M.R.; Mathew, B.; Nalinakumari, K.R.; Nair, M.K. Differential expression of cytokeratin proteins during tumour progression in oral mucosa. Epithel. Cell Biol. 1994, 3, 61–69. [Google Scholar]
- Kannan, S.; Balaram, P.; Chandran, G.J.; Pillai, M.R.; Mathew, B.; Nalinakumari, K.R.; Nair, M.K. Alterations in expression of terminal differentiation markers of keratinocytes during oral carcinogenesis. Pathobiology 1994, 62, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Kannan, S.; Chandran, G.J.; Balaram, P.; Chidambaram, S.; Nair, M.K. Potential biological markers for the staging of tumor progression in oral mucosa: A multivariate analysis. Int. J. Biol. Markers 1996, 11, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Kiani, M.N.; Asif, M.; Ansari, F.M.; Ara, N.; Ishaque, M.; Khan, A.R. Diagnostic utility of Cytokeratin 13 and Cytokeratin 17 in Oral Epithelial Dysplasia and Oral Squamous Cell Carcinoma. Asian Pac. J. Cancer Biol. 2020, 5, 153–158. [Google Scholar] [CrossRef]
- Kitamura, R.; Toyoshima, T.; Tanaka, H.; Kawano, S.; Kiyosue, T.; Matsubara, R.; Goto, Y.; Hirano, M.; Oobu, K.; Nakamura, S. Association of cytokeratin 17 expression with differentiation in oral squamous cell carcinoma. J. Cancer Res. Clin. Oncol. 2012, 138, 1299–1310. [Google Scholar] [CrossRef]
- Lindberg, K.; Rheinwald, J.G. Suprabasal 40 kd keratin (K19) expression as an immunohistologic marker of premalignancy in oral epithelium. Am. J. Pathol. 1989, 134, 89–98. [Google Scholar] [PubMed]
- Nanda, K.D.; Ranganathan, K.; Devi, U.; Joshua, E. Increased expression of CK8 and CK18 in leukoplakia, oral submucous fibrosis, and oral squamous cell carcinoma: An immunohistochemistry study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 113, 245–253. [Google Scholar] [CrossRef]
- Nobusawa, A.S.T.; Negishi, A.; Yokoo, S.; Oyama, T. Immunohistochemical staining patterns of cytokeratins 13, 14, and 17 in oral epithelial dysplasia including orthokeratotic dysplasia. Pathol. Int. 2014, 64, 20–27. [Google Scholar] [CrossRef]
- Okada, Y.; Moride, M. Immunohistochemical study of differential expressions of cytokeratin-13, -14,-17 and p53 in epithelial dysplasia and carcinoma of the tongue. J. Hard Tissue Biol. 2010, 19, 123–130. [Google Scholar] [CrossRef][Green Version]
- Rajeswari, P.; Janardhanan, M.; Suresh, R.; Savithri, V.; Aravind, T.; Raveendran, G. Expression of CK 19 as a biomarker in early detection of oral squamous cell carcinoma. J. Oral Maxillofac. Pathol. 2020, 24, 523–529. [Google Scholar] [CrossRef]
- Ram Prassad, V.V.; Nirmala, N.R.; Kotian, M.S. Immunohistochemical evaluation of expression of cytokeratin 19 in different histological grades of leukoplakia and oral squamous cell carcinoma. Indian J. Dent. Res. 2005, 16, 6–11. [Google Scholar] [PubMed]
- Safadi, R.A.; Musleh, A.S.; Al-Khateeb, T.H.; Hamasha, A.A.H. Analysis of Immunohistochemical Expression of K19 in Oral Epithelial Dysplasia and Oral Squamous Cell Carcinoma Using Color Deconvolution-Image Analysis Method. Head Neck Pathol. 2010, 4, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, K.; Aragaki, T.; Morita, K.; Kawachi, H.; Kayamori, K.; Nakanishi, S.; Omura, K.; Miki, Y.; Okada, N.; Katsube, K.; et al. Down-regulation of keratin 4 and keratin 13 expression in oral squamous cell carcinoma and epithelial dysplasia: A clue for histopathogenesis. Histopathology 2011, 58, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Sanguansin, S.; Kosanwat, T.; Juengsomjit, R.; Poomsawat, S. Diagnostic Value of Cytokeratin 17 during Oral Carcinogenesis: An Immunohistochemical Study. Int. J. Dent. 2021, 2021, 4089549. [Google Scholar] [CrossRef] [PubMed]
- Sawant, S.; Vaidya, M.; Chaukar, D.; Gangadaran, P.; Singh, A.K.; Rajadhyax, S.; Kannan, S.; Kane, S.; Pagare, S. Clinicopathological Features and Prognostic Implications of Loss of K5 and Gain of K1, K8 and K18 in Oral Potentially Malignant Lesions and Squamous Cell Carcinomas: An Immunohistochemical Analysis. Edorium J. Tumor Biol. 2014, 1, 1–22. [Google Scholar] [CrossRef]
- Schaaij-Visser, T.B.M.; Bremmer, J.F.; Braakhuis, B.J.M.; Heck, A.J.R.; Slijper, M.; van der Waal, I.; Brakenhoff, R.H. Evaluation of cornulin, keratin 4, keratin 13 expression and grade of dysplasia for predicting malignant progression of oral leukoplakia. Oral Oncol. 2010, 46, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Schulz, J.; Ermich, T.; Kasper, M.; Raabe, G.; Schumann, D. Cytokeratin pattern of clinically intact and pathologically changed oral mucosa. Int. J. Oral Maxillofac. Surg. 1992, 21, 35–39. [Google Scholar] [CrossRef]
- Shahabinejad, M.; Zare, R.; Mohajertehran, F.; Amouzad Mahdiraji, A. Cytokeratins (CK7 and CK20) Genes Expression Association with Clinicopathological Indices in Oral Squamous Cell Carcinoma and Dysplastic Oral Epithelium. Rep. Biochem. Mol. Biol. 2021, 10, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Sihmar, S.S.; Ramalingam, K.; Rathi, S.; Solkhe, M.; Monu, P. Immunohistochemical Expression of Ck8 and Ck18 in Oral Potentially Malignant Disorders and Oral Squamous Cell Carcinoma-A Retrospective Study On 70 Samples. J. Pharm. Negat. Results 2022, 13, 1153–1169. [Google Scholar] [CrossRef]
- Su, L.; Morgan, P.R.; Lane, E.B. Protein and mRNA expression of simple epithelial keratins in normal, dysplastic, and malignant oral epithelia. Am. J. Pathol. 1994, 145, 1349–1357. [Google Scholar]
- Su, L.; Morgan, P.R.; Lane, E.B. Keratin 14 and 19 expression in normal, dysplastic and malignant oral epithelia. A study using in situ hybridization and immunohistochemistry. J. Oral Pathol. Med. 1996, 25, 293–301. [Google Scholar] [CrossRef]
- Takeda, T.; Sugihara, K.; Hirayama, Y.; Hirano, M.; Tanuma, J.I.; Semba, I. Immunohistological evaluation of Ki-67, p63, CK19 and p53 expression in oral epithelial dysplasias. J. Oral Pathol. Med. 2006, 35, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, M.M.; Sawant, S.S.; Borges, A.M.; Ogale, S.B.; Bhisey, A.N. Cytokeratin expression in precancerous lesions of the human oral cavity. Oral Oncol. 1998, 34, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Vigneswaran, N.; Peters, K.P.; Hornstein, O.P.; Haneke, E. Comparison of cytokeratin, filaggrin and involucrin profiles in oral leukoplakias and squamous carcinomas. J. Oral Pathol. Med. 1989, 18, 377–390. [Google Scholar] [CrossRef]
- Wils, L.J.; Poell, J.B.; Evren, I.; Koopman, M.S.; Brouns, E.; de Visscher, J.; Brakenhoff, R.H.; Bloemena, E. Incorporation of differentiated dysplasia improves prediction of oral leukoplakia at increased risk of malignant progression. Mod. Pathol. 2020, 33, 1033–1040. [Google Scholar] [CrossRef]
- Wils, L.J.; Poell, J.B.; Peferoen, L.A.N.; Evren, I.; Brouns, E.R.; de Visscher, J.G.A.M.; van der Meij, E.H.; Brakenhoff, R.H.; Bloemena, E. The role of differentiated dysplasia in the prediction of malignant transformation of oral leukoplakia. J. Oral Pathol. Med. 2023, 52, 930–938. [Google Scholar] [CrossRef]
- Yagyuu, T.; Obayashi, C.; Ueyama, Y.; Takano, M.; Tanaka, Y.; Kawaguchi, M.; Takeda, M.; Kasai, T.; Kirita, T. Multivariate analyses of Ki-67, cytokeratin 13 and cytokeratin 17 in diagnosis and prognosis of oral precancerous lesions. J. Oral Pathol. Med. 2015, 44, 523–531. [Google Scholar] [CrossRef]
- Yoshida, K.; Sato, K.; Tonogi, M.; Tanaka, Y.; Yamane, G.Y.; Katakura, A. Expression of Cytokeratin 14 and 19 in Process of Oral Carcinogenesis. Bull. Tokyo Dent. Coll. 2015, 56, 105–111. [Google Scholar] [CrossRef][Green Version]
- Kramer, I.R.; Lucas, R.B.; Pindborg, J.J.; Sobin, L.H. Definition of leukoplakia and related lesions: An aid to studies on oral precancer. Oral Surg. Oral Med. Oral Pathol. 1978, 46, 518–539. [Google Scholar] [PubMed]
- Pindborg, J.J.; Reichart, P.A.; Smith, C.J.; van der Waal, I. Histological Classification of Cancer and Precancer of the Oral Mucosa. In Histological Typing of Cancer and Precancer of the Oral Mucosa; Pindborg, J.J., Reichart, P.A., Smith, C.J., van der Waal, I., Eds.; Springer: Berlin/Heidelberg, Germany, 1997; pp. 9–10. [Google Scholar]
- Barnes, L.; Eveson, J.; Reichart, P.; Sidransky, D. World Health Organization Classification of Tumours: Pathology and Genetics of Head and Neck Tumours, 3rd ed.; IARC Press: Lyon, France, 2005. [Google Scholar]
- El-Naggar, A.K.; Chan, J.K.C.; Grandis, J.R.; Takata, T.; Slootweg, P.J. WHO Classification of Head and Neck Tumours, 4th ed.; IARC Press: Lyon, France, 2017. [Google Scholar]
- Kramer, I. Basic histopathological features of oral premalignant lesions. Oral Premalig. Lesions 1980, 23–24, 23–24. [Google Scholar]
- Gräßel-Pietrusky, R.; Hornstein, O. Histologische Klassifikation oraler Präkanzerosen. Dtsch. Z. Mund. Kiefer GesichtsChir 1982, 6, 343–351. [Google Scholar]
- Safadi, R.A.; AlRomaizan, A.; Alshagroud, R.S.; Divakar, D.D.; Alshieban, S. Cytokeratin 19 Immunostain Reduces Variability in Grading Epithelial Dysplasia of the Non-Keratinized Upper Aerodigestive Tract Mucosa. Head Neck Pathol. 2020, 14, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Mikami, T.; Cheng, J.; Maruyama, S.; Kobayashi, T.; Funayama, A.; Yamazaki, M.; Adeola, H.A.; Wu, L.; Shingaki, S.; Saito, C.; et al. Emergence of keratin 17 vs. loss of keratin 13: Their reciprocal immunohistochemical profiles in oral carcinoma in situ. Oral Oncol. 2011, 47, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Kale, A.D.; Mane, D.R.; Babji, D.; Gupta, K. Establishment of field change by expression of cytokeratins 8/18, 19, and MMP-9 in an apparently normal oral mucosa adjacent to squamous cell carcinoma: A immunohistochemical study. J. Oral Maxillofac. Pathol. 2012, 16, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Batool, S.; Fahim, A.; Qureshi, A.; Jabeen, H.; Ali, S.N.; Khoso, M.Y. Role of Alteration of Ck5\6 Profile In Dysplastic Progression Of Oral Mucosa In Tobacco Users. JAMC 2020, 32, 527–530. [Google Scholar] [PubMed]
- Ohta, K.; Ogawa, I.; Ono, S.; Taki, M.; Mizuta, K.; Miyauchi, M.; Takechi, M.; Shigeishi, H.; Takata, T.; Kamata, N. Histopathological evaluation including cytokeratin 13 and Ki-67 in the border between Lugol-stained and -unstained areas. Oncol. Rep. 2010, 24, 9–14. [Google Scholar] [CrossRef]
- Kobayashi, T.; Maruyama, S.; Abé, T.; Cheng, J.; Takagi, R.; Saito, C.; Saku, T. Keratin 10-positive orthokeratotic dysplasia: A new leucoplakia-type precancerous entity of the oral mucosa. Histopathology 2012, 61, 910–920. [Google Scholar] [CrossRef]
- Baraks, G.; Tseng, R.; Pan, C.H.; Kasliwal, S.; Leiton, C.V.; Shroyer, K.R.; Escobar-Hoyos, L.F. Dissecting the Oncogenic Roles of Keratin 17 in the Hallmarks of Cancer. Cancer Res. 2022, 82, 1159–1166. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, Y.; Xia, T.; Lu, L.; Luo, M.; Chen, Y.; Liu, Y.; Li, Y. The Role of Keratin17 in Human Tumours. Front. Cell Dev. Biol. 2022, 10, 818416. [Google Scholar] [CrossRef]
- Mikami, T.; Maruyama, S.; Abé, T.; Kobayashi, T.; Yamazaki, M.; Funayama, A.; Shingaki, S.; Kobayashi, T.; Jun, C.; Saku, T. Keratin 17 is co-expressed with 14-3-3 sigma in oral carcinoma in situ and squamous cell carcinoma and modulates cell proliferation and size but not cell migration. Virchows Arch. 2015, 466, 559–569. [Google Scholar] [CrossRef]
- Mikami, Y.; Fujii, S.; Nagata, K.; Wada, H.; Hasegawa, K.; Abe, M.; Yoshimoto, R.U.; Kawano, S.; Nakamura, S.; Kiyoshima, T. GLI-mediated Keratin 17 expression promotes tumor cell growth through the anti-apoptotic function in oral squamous cell carcinomas. J. Cancer Res. Clin. Oncol. 2017, 143, 1381–1393. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).