Hematopoietic Stem Cells and Their Niche in Bone Marrow
Abstract
:1. Introduction
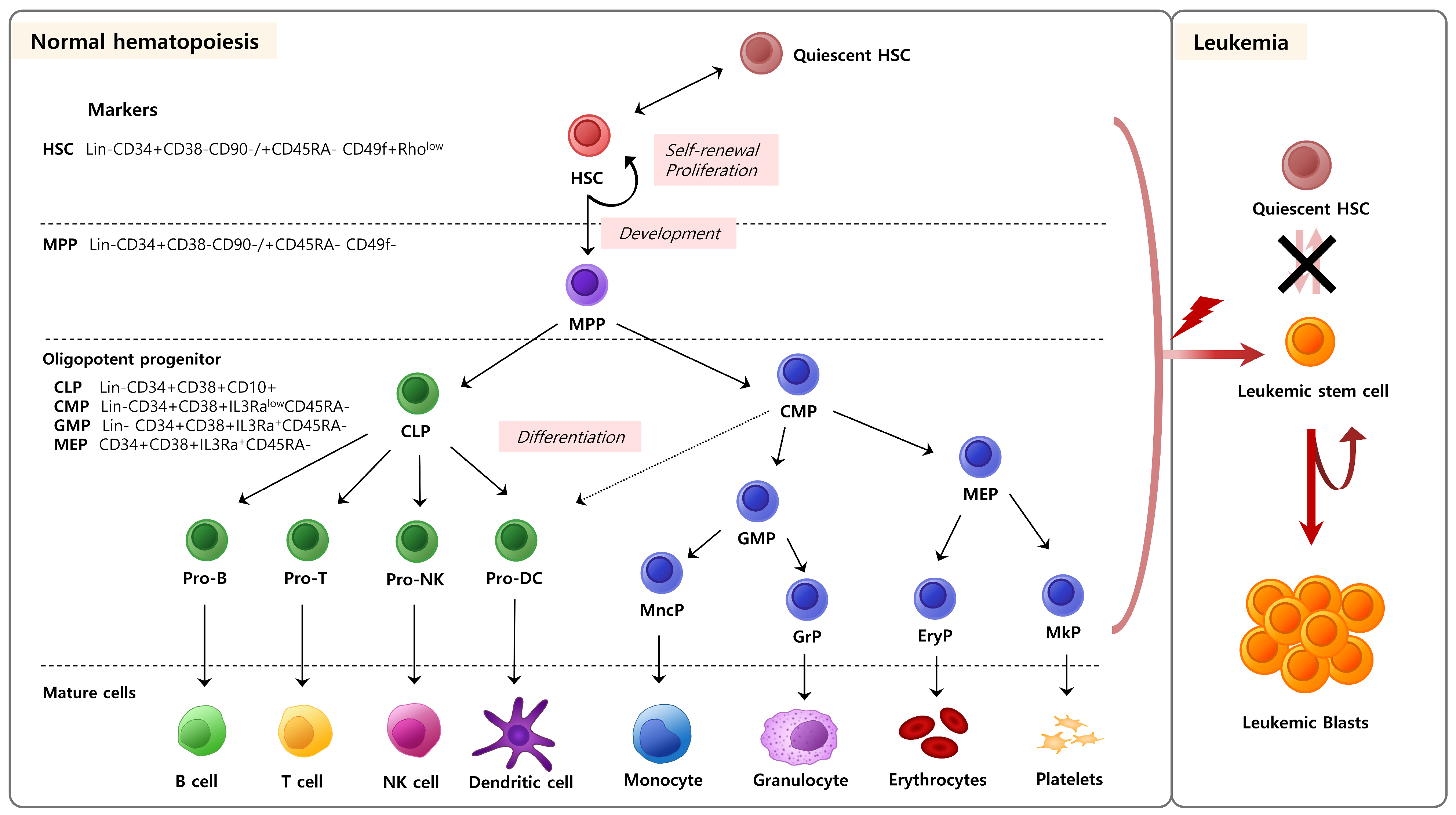
2. Hematopoietic Stem/Progenitor Cells
3. The Relationship between Hematopoietic Stem/Progenitor Cells and Aging
3.1. The Quantity and Role of Hematopoietic Stem/Progenitor Cells in Aging
3.2. Heterogeneity of Hematopoietic Stem/Progenitor Cell Aging
3.3. Regeneration of Aged/Mature Hematopoietic Stem or Progenitor Cells
4. Bone Marrow Microenvironment
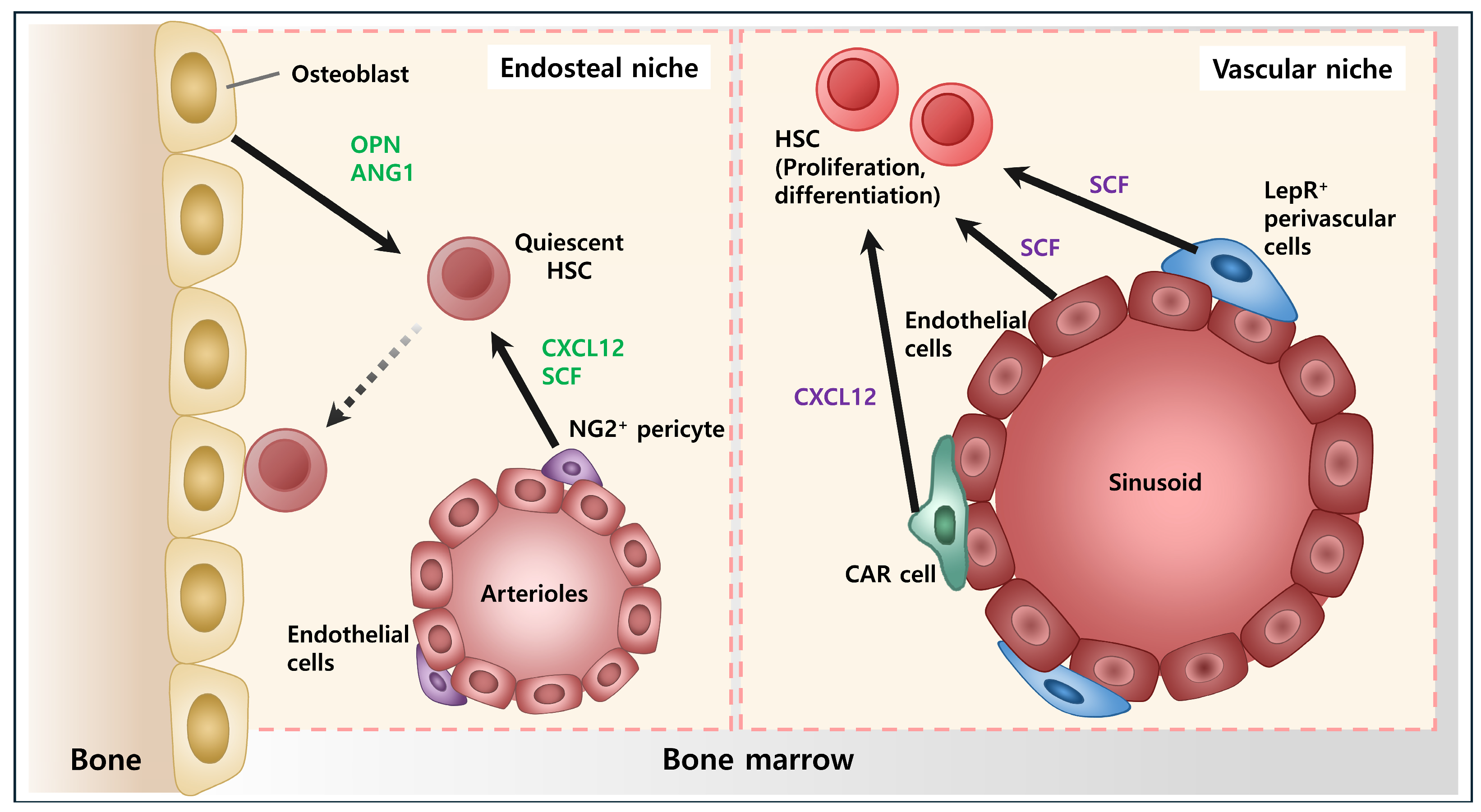
5. Vascular Niche
6. Osteoblastic Niche
7. Other Components of Niche
8. Stem Cell Therapy
8.1. HSC Transplantation
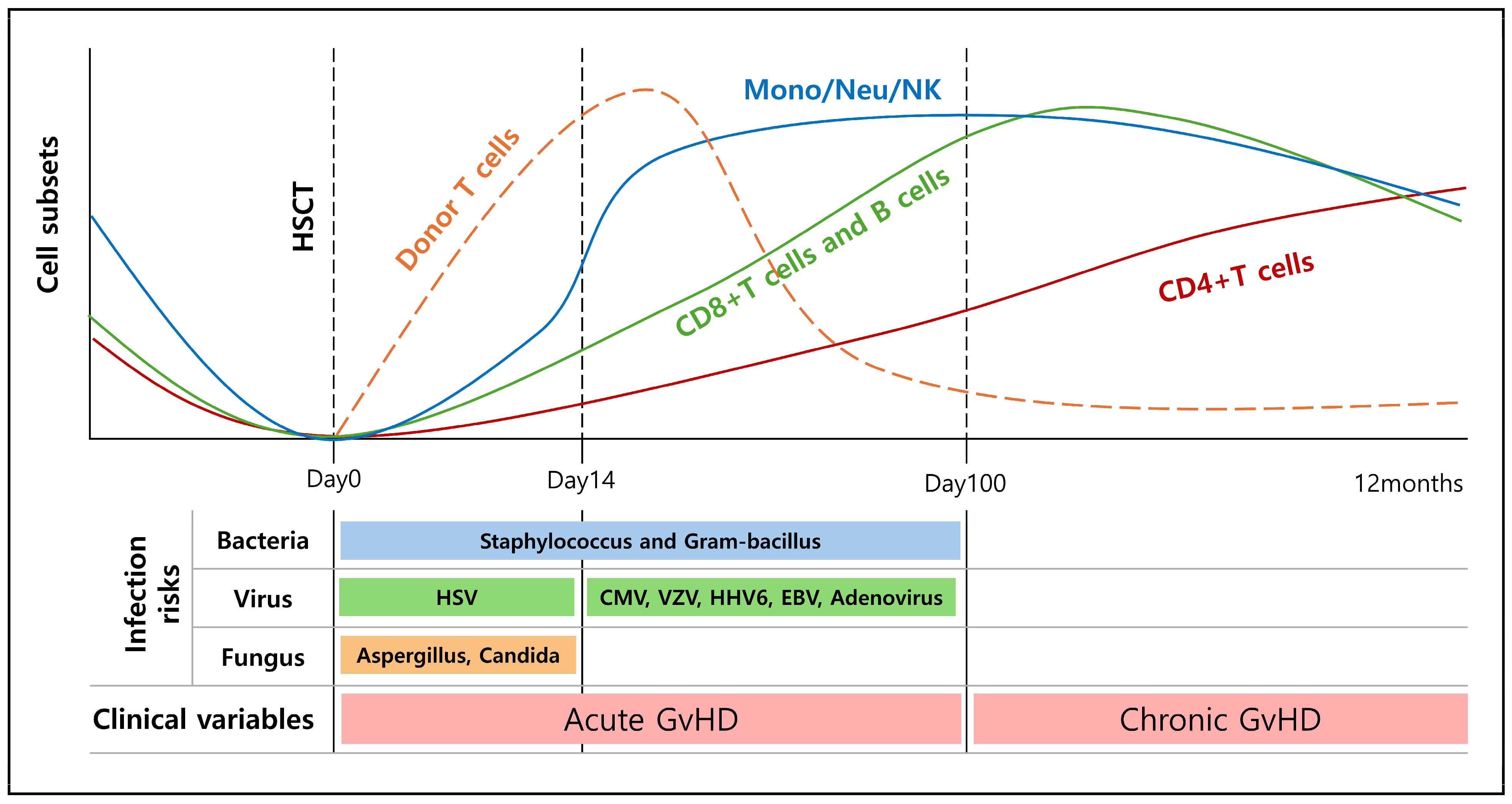
8.2. Immune Recovery after HSCT
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Adams, G.B.; Scadden, D.T. The hematopoietic stem cell in its place. Nat. Immunol. 2006, 7, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Scadden, D.T. Nice neighborhood: Emerging concepts of the stem cell niche. Cell 2014, 157, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Emerson, S.G. Hematopoietic cytokines, transcription factors and lineage commitment. Oncogene 2002, 21, 3295–3313. [Google Scholar] [CrossRef] [PubMed]
- Mendelson, A.; Frenette, P.S. Hematopoietic stem cell niche maintenance during homeostasis and regeneration. Nat. Med. 2014, 20, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Kunisaki, Y.; Frenette, P.S. The secrets of the bone marrow niche: Enigmatic niche brings challenge for HSC expansion. Nat. Med. 2012, 18, 864–865. [Google Scholar] [CrossRef] [PubMed]
- Schofield, R. The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells 1978, 4, 7–25. [Google Scholar] [PubMed]
- Morrison, S.J.; Scadden, D.T. The bone marrow niche for haematopoietic stem cells. Nature 2014, 505, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Krause, D.S.; Scadden, D.T. A hostel for the hostile: The bone marrow niche in hematologic neoplasms. Haematologica 2015, 100, 1376–1387. [Google Scholar] [CrossRef] [PubMed]
- Scadden, D.T. The stem-cell niche as an entity of action. Nature 2006, 441, 1075–1079. [Google Scholar] [CrossRef]
- Hoggatt, J.; Kfoury, Y.; Scadden, D.T. Hematopoietic Stem Cell Niche in Health and Disease. Annu. Rev. Pathol. 2016, 11, 555–581. [Google Scholar] [CrossRef]
- Gustafsson, K.; Scadden, D.T. Growing old in the age of heterogeneity: The perils of shifting clonality. Curr. Opin. Hematol. 2019, 26, 222–227. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Sharpless, N.E. Senescence in Health and Disease. Cell 2017, 169, 1000–1011. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Galluzzi, L.; Freije, J.M.P.; Madeo, F.; Kroemer, G. Metabolic Control of Longevity. Cell 2016, 166, 802–821. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [PubMed]
- Mejia-Ramirez, E.; Florian, M.C. Understanding intrinsic hematopoietic stem cell aging. Haematologica 2020, 105, 22–37. [Google Scholar] [CrossRef] [PubMed]
- Fox, R.G.; Park, F.D.; Koechlein, C.S.; Kritzik, M.; Reya, T. Musashi signaling in stem cells and cancer. Annu. Rev. Cell Dev. Biol. 2015, 31, 249–267. [Google Scholar] [CrossRef] [PubMed]
- Vetrie, D.; Helgason, G.V.; Copland, M. The leukaemia stem cell: Similarities, differences and clinical prospects in CML and AML. Nat. Rev. Cancer 2020, 20, 158–173. [Google Scholar] [CrossRef]
- Whiteley, A.E.; Price, T.T.; Cantelli, G.; Sipkins, D.A. Leukaemia: A model metastatic disease. Nat. Rev. Cancer 2021, 21, 461–475. [Google Scholar] [CrossRef] [PubMed]
- Orford, K.W.; Scadden, D.T. Deconstructing stem cell self-renewal: Genetic insights into cell-cycle regulation. Nat. Rev. Genet. 2008, 9, 115–128. [Google Scholar] [CrossRef]
- He, S.; Nakada, D.; Morrison, S.J. Mechanisms of stem cell self-renewal. Annu. Rev. Cell Dev. Biol. 2009, 25, 377–406. [Google Scholar] [CrossRef]
- Viswanathan, S.; Shi, Y.; Galipeau, J.; Krampera, M.; Leblanc, K.; Martin, I.; Nolta, J.; Phinney, D.G.; Sensebe, L. Mesenchymal stem versus stromal cells: International Society for Cell & Gene Therapy (ISCT®) Mesenchymal Stromal Cell committee position statement on nomenclature. Cytotherapy 2019, 21, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Abdellateif, M.S.; Zekri, A.N. Stem cell therapy for hepatocellular carcinoma and end-stage liver disease. J. Egypt. Natl. Cancer Inst. 2023, 35, 35. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.; Yao, J.; Xie, Y.; Jiang, P.; Yan, J.; Li, X. Bioreactor-based stem cell therapy for liver fibrosis. Biofabrication 2024, 16, 025028. [Google Scholar] [CrossRef] [PubMed]
- Xiang, X.N.; Zhu, S.Y.; He, H.C.; Yu, X.; Xu, Y.; He, C.Q. Mesenchymal stromal cell-based therapy for cartilage regeneration in knee osteoarthritis. Stem Cell Res. Ther. 2022, 13, 14. [Google Scholar] [CrossRef]
- Chen, H.X.; Liang, F.C.; Gu, P.; Xu, B.L.; Xu, H.J.; Wang, W.T.; Hou, J.Y.; Xie, D.X.; Chai, X.Q.; An, S.J. Exosomes derived from mesenchymal stem cells repair a Parkinson’s disease model by inducing autophagy. Cell Death Dis. 2020, 11, 288. [Google Scholar] [CrossRef] [PubMed]
- Palanisamy, C.P.; Pei, J.; Alugoju, P.; Anthikapalli, N.V.A.; Jayaraman, S.; Veeraraghavan, V.P.; Gopathy, S.; Roy, J.R.; Janaki, C.S.; Thalamati, D.; et al. New strategies of neurodegenerative disease treatment with extracellular vesicles (EVs) derived from mesenchymal stem cells (MSCs). Theranostics 2023, 13, 4138–4165. [Google Scholar] [CrossRef] [PubMed]
- Joyce, N.; Annett, G.; Wirthlin, L.; Olson, S.; Bauer, G.; Nolta, J.A. Mesenchymal stem cells for the treatment of neurodegenerative disease. Regen. Med. 2010, 5, 933–946. [Google Scholar] [CrossRef]
- Heris, R.M.; Shirvaliloo, M.; Abbaspour-Aghdam, S.; Hazrati, A.; Shariati, A.; Youshanlouei, H.R.; Niaragh, F.J.; Valizadeh, H.; Ahmadi, M. The potential use of mesenchymal stem cells and their exosomes in Parkinson’s disease treatment. Stem Cell Res. Ther. 2022, 13, 371. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef]
- Park, S.; Park, C.W.; Eom, J.H.; Jo, M.Y.; Hur, H.J.; Choi, S.K.; Lee, J.S.; Nam, S.T.; Jo, K.S.; Oh, Y.W.; et al. Preclinical and dose-ranging assessment of hESC-derived dopaminergic progenitors for a clinical trial on Parkinson’s disease. Cell Stem Cell 2024, 31, 25–38.e28. [Google Scholar] [CrossRef]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. Hallmarks of aging: An expanding universe. Cell 2023, 186, 243–278. [Google Scholar] [CrossRef]
- Beerman, I.; Maloney, W.J.; Weissmann, I.L.; Rossi, D.J. Stem cells and the aging hematopoietic system. Curr. Opin. Immunol. 2010, 22, 500–506. [Google Scholar] [CrossRef]
- de Haan, G.; Lazare, S.S. Aging of hematopoietic stem cells. Blood 2018, 131, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.Y.; Lee, H.; Park, S.M.; Choi, H.S.; Kim, J.; Kwon, M.; Sohn, J.; Nam, J.H.; Kim, H.S.; Song, P.; et al. Role of reactive oxygen species in regulating 27-hydroxycholesterol-induced apoptosis of hematopoietic progenitor cells and myeloid cell lines. Cell Death Dis. 2022, 13, 916. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, H.; Hong, C.M.; Nam, J.H.; Yeo, H.J.; Cho, W.H.; Kim, H.S.; Hong, C.; Kim, Y.H.; Lee, D. Novel endogenous endoplasmic reticulum transmembrane protein SURF4 suppresses cell death by negatively regulating the STING-STAT6 axis in myeloid leukemia. Cancer Commun. 2023, 43, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Melnick, A.M.; Westendorf, J.J.; Polinger, A.; Carlile, G.W.; Arai, S.; Ball, H.J.; Lutterbach, B.; Hiebert, S.W.; Licht, J.D. The ETO protein disrupted in t(8;21)-associated acute myeloid leukemia is a corepressor for the promyelocytic leukemia zinc finger protein. Mol. Cell. Biol. 2000, 20, 2075–2086. [Google Scholar] [CrossRef]
- Melnick, A.; Carlile, G.W.; McConnell, M.J.; Polinger, A.; Hiebert, S.W.; Licht, J.D. AML-1/ETO fusion protein is a dominant negative inhibitor of transcriptional repression by the promyelocytic leukemia zinc finger protein. Blood 2000, 96, 3939–3947. [Google Scholar] [CrossRef]
- Ponnusamy, K.; Chen-Wichmann, L.; Kuvardina, O.N.; Lausen, J.; Henschler, R.; Wichmann, C. The truncated RUNX1/ETO activates VLA-4-dependent adhesion and migration of hematopoietic progenitor cells. Haematologica 2014, 99, e253–e256. [Google Scholar] [CrossRef]
- Chaudhury, S.; O’Connor, C.; Cañete, A.; Bittencourt-Silvestre, J.; Sarrou, E.; Prendergast, Á.; Choi, J.; Johnston, P.; Wells, C.A.; Gibson, B.; et al. Age-specific biological and molecular profiling distinguishes paediatric from adult acute myeloid leukaemias. Nat. Commun. 2018, 9, 5280. [Google Scholar] [CrossRef]
- Matteini, F.; Mulaw, M.A.; Florian, M.C. Aging of the Hematopoietic Stem Cell Niche: New Tools to Answer an Old Question. Front. Immunol. 2021, 12, 738204. [Google Scholar] [CrossRef]
- Jaiswal, S.; Fontanillas, P.; Flannick, J.; Manning, A.; Grauman, P.V.; Mar, B.G.; Lindsley, R.C.; Mermel, C.H.; Burtt, N.; Chavez, A.; et al. Age-related clonal hematopoiesis associated with adverse outcomes. N. Engl. J. Med. 2014, 371, 2488–2498. [Google Scholar] [CrossRef] [PubMed]
- McKerrell, T.; Park, N.; Moreno, T.; Grove, C.S.; Ponstingl, H.; Stephens, J.; Crawley, C.; Craig, J.; Scott, M.A.; Hodkinson, C.; et al. Leukemia-associated somatic mutations drive distinct patterns of age-related clonal hemopoiesis. Cell Rep. 2015, 10, 1239–1245. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.; Lu, C.; Wang, J.; McLellan, M.D.; Johnson, K.J.; Wendl, M.C.; McMichael, J.F.; Schmidt, H.K.; Yellapantula, V.; Miller, C.A.; et al. Age-related mutations associated with clonal hematopoietic expansion and malignancies. Nat. Med. 2014, 20, 1472–1478. [Google Scholar] [CrossRef] [PubMed]
- Bowman, R.L.; Busque, L.; Levine, R.L. Clonal Hematopoiesis and Evolution to Hematopoietic Malignancies. Cell Stem Cell 2018, 22, 157–170. [Google Scholar] [CrossRef]
- Buscarlet, M.; Provost, S.; Zada, Y.F.; Barhdadi, A.; Bourgoin, V.; Lépine, G.; Mollica, L.; Szuber, N.; Dubé, M.P.; Busque, L. DNMT3A and TET2 dominate clonal hematopoiesis and demonstrate benign phenotypes and different genetic predispositions. Blood 2017, 130, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, K.D.; Helin, K. Role of TET enzymes in DNA methylation, development, and cancer. Genes Dev. 2016, 30, 733–750. [Google Scholar] [CrossRef] [PubMed]
- Sano, S.; Oshima, K.; Wang, Y.; MacLauchlan, S.; Katanasaka, Y.; Sano, M.; Zuriaga, M.A.; Yoshiyama, M.; Goukassian, D.; Cooper, M.A.; et al. Tet2-Mediated Clonal Hematopoiesis Accelerates Heart Failure Through a Mechanism Involving the IL-1β/NLRP3 Inflammasome. J. Am. Coll. Cardiol. 2018, 71, 875–886. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Wahab, O.; Adli, M.; LaFave, L.M.; Gao, J.; Hricik, T.; Shih, A.H.; Pandey, S.; Patel, J.P.; Chung, Y.R.; Koche, R.; et al. ASXL1 mutations promote myeloid transformation through loss of PRC2-mediated gene repression. Cancer Cell 2012, 22, 180–193. [Google Scholar] [CrossRef] [PubMed]
- Bick, A.G.; Weinstock, J.S.; Nandakumar, S.K.; Fulco, C.P.; Bao, E.L.; Zekavat, S.M.; Szeto, M.D.; Liao, X.; Leventhal, M.J.; Nasser, J.; et al. Inherited causes of clonal haematopoiesis in 97,691 whole genomes. Nature 2020, 586, 763–768. [Google Scholar] [CrossRef]
- Genovese, G.; Kähler, A.K.; Handsaker, R.E.; Lindberg, J.; Rose, S.A.; Bakhoum, S.F.; Chambert, K.; Mick, E.; Neale, B.M.; Fromer, M.; et al. Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N. Engl. J. Med. 2014, 371, 2477–2487. [Google Scholar] [CrossRef]
- Young, A.L.; Challen, G.A.; Birmann, B.M.; Druley, T.E. Clonal haematopoiesis harbouring AML-associated mutations is ubiquitous in healthy adults. Nat. Commun. 2016, 7, 12484. [Google Scholar] [CrossRef]
- Mosteo, L.; Storer, J.; Batta, K.; Searle, E.J.; Duarte, D.; Wiseman, D.H. The Dynamic Interface Between the Bone Marrow Vascular Niche and Hematopoietic Stem Cells in Myeloid Malignancy. Front. Cell Dev. Biol. 2021, 9, 635189. [Google Scholar] [CrossRef]
- Yamazaki, S.; Iwama, A.; Takayanagi, S.; Eto, K.; Ema, H.; Nakauchi, H. TGF-beta as a candidate bone marrow niche signal to induce hematopoietic stem cell hibernation. Blood 2009, 113, 1250–1256. [Google Scholar] [CrossRef]
- Zink, F.; Stacey, S.N.; Norddahl, G.L.; Frigge, M.L.; Magnusson, O.T.; Jonsdottir, I.; Thorgeirsson, T.E.; Sigurdsson, A.; Gudjonsson, S.A.; Gudmundsson, J.; et al. Clonal hematopoiesis, with and without candidate driver mutations, is common in the elderly. Blood 2017, 130, 742–752. [Google Scholar] [CrossRef]
- Morganti, C.; Ito, K. Mitochondrial Contributions to Hematopoietic Stem Cell Aging. Int. J. Mol. Sci. 2021, 22, 1117. [Google Scholar] [CrossRef]
- Tikhonova, A.N.; Dolgalev, I.; Hu, H.; Sivaraj, K.K.; Hoxha, E.; Cuesta-Domínguez, Á.; Pinho, S.; Akhmetzyanova, I.; Gao, J.; Witkowski, M.; et al. The bone marrow microenvironment at single-cell resolution. Nature 2019, 569, 222–228. [Google Scholar] [CrossRef]
- Baryawno, N.; Przybylski, D.; Kowalczyk, M.S.; Kfoury, Y.; Severe, N.; Gustafsson, K.; Kokkaliaris, K.D.; Mercier, F.; Tabaka, M.; Hofree, M.; et al. A Cellular Taxonomy of the Bone Marrow Stroma in Homeostasis and Leukemia. Cell 2019, 177, 1915–1932.e1916. [Google Scholar] [CrossRef]
- Baccin, C.; Al-Sabah, J.; Velten, L.; Helbling, P.M.; Grünschläger, F.; Hernández-Malmierca, P.; Nombela-Arrieta, C.; Steinmetz, L.M.; Trumpp, A.; Haas, S. Combined single-cell and spatial transcriptomics reveal the molecular, cellular and spatial bone marrow niche organization. Nat. Cell Biol. 2020, 22, 38–48. [Google Scholar] [CrossRef]
- Itkin, T.; Gur-Cohen, S.; Spencer, J.A.; Schajnovitz, A.; Ramasamy, S.K.; Kusumbe, A.P.; Ledergor, G.; Jung, Y.; Milo, I.; Poulos, M.G.; et al. Distinct bone marrow blood vessels differentially regulate haematopoiesis. Nature 2016, 532, 323–328. [Google Scholar] [CrossRef]
- Bergiers, I.; Andrews, T.; Vargel Bölükbaşı, Ö.; Buness, A.; Janosz, E.; Lopez-Anguita, N.; Ganter, K.; Kosim, K.; Celen, C.; Itır Perçin, G.; et al. Single-cell transcriptomics reveals a new dynamical function of transcription factors during embryonic hematopoiesis. eLife 2018, 7, e29312. [Google Scholar] [CrossRef]
- Zhang, X.; Han, P.; Qiu, J.; Huang, F.; Luo, Q.; Cheng, J.; Shan, K.; Yang, Y.; Zhang, C. Single-cell RNA sequencing reveals the complex cellular niche of pterygium. Ocul. Surf. 2024, 32, 91–103. [Google Scholar] [CrossRef]
- Bandyopadhyay, S.; Duffy, M.P.; Ahn, K.J.; Sussman, J.H.; Pang, M.; Smith, D.; Duncan, G.; Zhang, I.; Huang, J.; Lin, Y.; et al. Mapping the cellular biogeography of human bone marrow niches using single-cell transcriptomics and proteomic imaging. Cell 2024, 187, 3120–3140.e3129. [Google Scholar] [CrossRef]
- Kucinski, I.; Campos, J.; Barile, M.; Severi, F.; Bohin, N.; Moreira, P.N.; Allen, L.; Lawson, H.; Haltalli, M.L.R.; Kinston, S.J.; et al. A time- and single-cell-resolved model of murine bone marrow hematopoiesis. Cell Stem Cell 2024, 31, 244–259.e210. [Google Scholar] [CrossRef]
- Bergers, G.; Song, S. The role of pericytes in blood-vessel formation and maintenance. Neuro-Oncology 2005, 7, 452–464. [Google Scholar] [CrossRef]
- Majesky, M.W. Vascular Development. Arterioscler. Thromb. Vasc. Biol. 2018, 38, e17–e24. [Google Scholar] [CrossRef]
- Naito, H.; Iba, T.; Takakura, N. Mechanisms of new blood-vessel formation and proliferative heterogeneity of endothelial cells. Int. Immunol. 2020, 32, 295–305. [Google Scholar] [CrossRef]
- Comazzetto, S.; Shen, B.; Morrison, S.J. Niches that regulate stem cells and hematopoiesis in adult bone marrow. Dev. Cell 2021, 56, 1848–1860. [Google Scholar] [CrossRef]
- Doan, P.L.; Chute, J.P. The vascular niche: Home for normal and malignant hematopoietic stem cells. Leukemia 2012, 26, 54–62. [Google Scholar] [CrossRef]
- He, N.; Zhang, L.; Cui, J.; Li, Z. Bone marrow vascular niche: Home for hematopoietic stem cells. Bone Marrow Res. 2014, 2014, 128436. [Google Scholar] [CrossRef]
- Li, H.; Yue, R.; Wei, B.; Gao, G.; Du, J.; Pei, G. Lysophosphatidic acid acts as a nutrient-derived developmental cue to regulate early hematopoiesis. EMBO J. 2014, 33, 1383–1396. [Google Scholar] [CrossRef]
- Ottersbach, K.; Dzierzak, E. The murine placenta contains hematopoietic stem cells within the vascular labyrinth region. Dev. Cell 2005, 8, 377–387. [Google Scholar] [CrossRef]
- Rafii, S.; Lyden, D.; Benezra, R.; Hattori, K.; Heissig, B. Vascular and haematopoietic stem cells: Novel targets for anti-angiogenesis therapy? Nat. Rev. Cancer 2002, 2, 826–835. [Google Scholar] [CrossRef]
- Takakura, N.; Huang, X.L.; Naruse, T.; Hamaguchi, I.; Dumont, D.J.; Yancopoulos, G.D.; Suda, T. Critical role of the TIE2 endothelial cell receptor in the development of definitive hematopoiesis. Immunity 1998, 9, 677–686. [Google Scholar] [CrossRef]
- Gotohda, N.; Iwagaki, H.; Itano, S.; Horiki, S.; Fujiwara, T.; Saito, S.; Hizuta, A.; Isozaki, H.; Takakura, N.; Terada, N.; et al. Can POSSUM, a scoring system for perioperative surgical risk, predict postoperative clinical course? Acta Medica Okayama 1998, 52, 325–329. [Google Scholar] [CrossRef]
- Asahara, T.; Murohara, T.; Sullivan, A.; Silver, M.; van der Zee, R.; Li, T.; Witzenbichler, B.; Schatteman, G.; Isner, J.M. Isolation of putative progenitor endothelial cells for angiogenesis. Science 1997, 275, 964–967. [Google Scholar] [CrossRef]
- Heissig, B.; Hattori, K.; Dias, S.; Friedrich, M.; Ferris, B.; Hackett, N.R.; Crystal, R.G.; Besmer, P.; Lyden, D.; Moore, M.A.; et al. Recruitment of stem and progenitor cells from the bone marrow niche requires MMP-9 mediated release of kit-ligand. Cell 2002, 109, 625–637. [Google Scholar] [CrossRef]
- Hattori, K.; Dias, S.; Heissig, B.; Hackett, N.R.; Lyden, D.; Tateno, M.; Hicklin, D.J.; Zhu, Z.; Witte, L.; Crystal, R.G.; et al. Vascular endothelial growth factor and angiopoietin-1 stimulate postnatal hematopoiesis by recruitment of vasculogenic and hematopoietic stem cells. J. Exp. Med. 2001, 193, 1005–1014. [Google Scholar] [CrossRef]
- Gerber, H.P.; Malik, A.K.; Solar, G.P.; Sherman, D.; Liang, X.H.; Meng, G.; Hong, K.; Marsters, J.C.; Ferrara, N. VEGF regulates haematopoietic stem cell survival by an internal autocrine loop mechanism. Nature 2002, 417, 954–958. [Google Scholar] [CrossRef]
- Lévesque, J.P.; Helwani, F.M.; Winkler, I.G. The endosteal ‘osteoblastic’ niche and its role in hematopoietic stem cell homing and mobilization. Leukemia 2010, 24, 1979–1992. [Google Scholar] [CrossRef]
- Pinho, S.; Frenette, P.S. Haematopoietic stem cell activity and interactions with the niche. Nat. Rev. Mol. Cell Biol. 2019, 20, 303–320. [Google Scholar] [CrossRef]
- Whetton, A.D.; Graham, G.J. Homing and mobilization in the stem cell niche. Trends Cell Biol. 1999, 9, 233–238. [Google Scholar] [CrossRef]
- Acar, M.; Kocherlakota, K.S.; Murphy, M.M.; Peyer, J.G.; Oguro, H.; Inra, C.N.; Jaiyeola, C.; Zhao, Z.; Luby-Phelps, K.; Morrison, S.J. Deep imaging of bone marrow shows non-dividing stem cells are mainly perisinusoidal. Nature 2015, 526, 126–130. [Google Scholar] [CrossRef]
- Ding, L.; Saunders, T.L.; Enikolopov, G.; Morrison, S.J. Endothelial and perivascular cells maintain haematopoietic stem cells. Nature 2012, 481, 457–462. [Google Scholar] [CrossRef]
- Omatsu, Y.; Sugiyama, T.; Kohara, H.; Kondoh, G.; Fujii, N.; Kohno, K.; Nagasawa, T. The essential functions of adipo-osteogenic progenitors as the hematopoietic stem and progenitor cell niche. Immunity 2010, 33, 387–399. [Google Scholar] [CrossRef]
- Sugiyama, T.; Kohara, H.; Noda, M.; Nagasawa, T. Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity 2006, 25, 977–988. [Google Scholar] [CrossRef]
- Kunisaki, Y.; Bruns, I.; Scheiermann, C.; Ahmed, J.; Pinho, S.; Zhang, D.; Mizoguchi, T.; Wei, Q.; Lucas, D.; Ito, K.; et al. Arteriolar niches maintain haematopoietic stem cell quiescence. Nature 2013, 502, 637–643. [Google Scholar] [CrossRef]
- Ono, N.; Ono, W.; Mizoguchi, T.; Nagasawa, T.; Frenette, P.S.; Kronenberg, H.M. Vasculature-associated cells expressing nestin in developing bones encompass early cells in the osteoblast and endothelial lineage. Dev. Cell 2014, 29, 330–339. [Google Scholar] [CrossRef]
- Le, P.M.; Andreeff, M.; Battula, V.L. Osteogenic niche in the regulation of normal hematopoiesis and leukemogenesis. Haematologica 2018, 103, 1945–1955. [Google Scholar] [CrossRef]
- Kim, M.J.; Valderrábano, R.J.; Wu, J.Y. Osteoblast Lineage Support of Hematopoiesis in Health and Disease. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2022, 37, 1823–1842. [Google Scholar] [CrossRef]
- Nilsson, S.K.; Johnston, H.M.; Whitty, G.A.; Williams, B.; Webb, R.J.; Denhardt, D.T.; Bertoncello, I.; Bendall, L.J.; Simmons, P.J.; Haylock, D.N. Osteopontin, a key component of the hematopoietic stem cell niche and regulator of primitive hematopoietic progenitor cells. Blood 2005, 106, 1232–1239. [Google Scholar] [CrossRef]
- Zhou, B.O.; Ding, L.; Morrison, S.J. Hematopoietic stem and progenitor cells regulate the regeneration of their niche by secreting Angiopoietin-1. eLife 2015, 4, e05521. [Google Scholar] [CrossRef]
- Arai, F.; Hirao, A.; Ohmura, M.; Sato, H.; Matsuoka, S.; Takubo, K.; Ito, K.; Koh, G.Y.; Suda, T. Tie2/angiopoietin-1 signaling regulates hematopoietic stem cell quiescence in the bone marrow niche. Cell 2004, 118, 149–161. [Google Scholar] [CrossRef]
- Iwama, A.; Hamaguchi, I.; Hashiyama, M.; Murayama, Y.; Yasunaga, K.; Suda, T. Molecular cloning and characterization of mouse TIE and TEK receptor tyrosine kinase genes and their expression in hematopoietic stem cells. Biochem. Biophys. Res. Commun. 1993, 195, 301–309. [Google Scholar] [CrossRef]
- Zhang, J.; Niu, C.; Ye, L.; Huang, H.; He, X.; Tong, W.G.; Ross, J.; Haug, J.; Johnson, T.; Feng, J.Q.; et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 2003, 425, 836–841. [Google Scholar] [CrossRef]
- Visnjic, D.; Kalajzic, Z.; Rowe, D.W.; Katavic, V.; Lorenzo, J.; Aguila, H.L. Hematopoiesis is severely altered in mice with an induced osteoblast deficiency. Blood 2004, 103, 3258–3264. [Google Scholar] [CrossRef]
- Karanu, F.N.; Murdoch, B.; Gallacher, L.; Wu, D.M.; Koremoto, M.; Sakano, S.; Bhatia, M. The notch ligand jagged-1 represents a novel growth factor of human hematopoietic stem cells. J. Exp. Med. 2000, 192, 1365–1372. [Google Scholar] [CrossRef]
- Karanu, F.N.; Murdoch, B.; Miyabayashi, T.; Ohno, M.; Koremoto, M.; Gallacher, L.; Wu, D.; Itoh, A.; Sakano, S.; Bhatia, M. Human homologues of Delta-1 and Delta-4 function as mitogenic regulators of primitive human hematopoietic cells. Blood 2001, 97, 1960–1967. [Google Scholar] [CrossRef]
- Stier, S.; Cheng, T.; Dombkowski, D.; Carlesso, N.; Scadden, D.T. Notch1 activation increases hematopoietic stem cell self-renewal in vivo and favors lymphoid over myeloid lineage outcome. Blood 2002, 99, 2369–2378. [Google Scholar] [CrossRef]
- Varnum-Finney, B.; Brashem-Stein, C.; Bernstein, I.D. Combined effects of Notch signaling and cytokines induce a multiple log increase in precursors with lymphoid and myeloid reconstituting ability. Blood 2003, 101, 1784–1789. [Google Scholar] [CrossRef] [PubMed]
- Varnum-Finney, B.; Xu, L.; Brashem-Stein, C.; Nourigat, C.; Flowers, D.; Bakkour, S.; Pear, W.S.; Bernstein, I.D. Pluripotent, cytokine-dependent, hematopoietic stem cells are immortalized by constitutive Notch1 signaling. Nat. Med. 2000, 6, 1278–1281. [Google Scholar] [CrossRef] [PubMed]
- Calvi, L.M.; Adams, G.B.; Weibrecht, K.W.; Weber, J.M.; Olson, D.P.; Knight, M.C.; Martin, R.P.; Schipani, E.; Divieti, P.; Bringhurst, F.R.; et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 2003, 425, 841–846. [Google Scholar] [CrossRef]
- Ghosh, J.; Koussa, R.E.; Mohamad, S.F.; Liu, J.; Kacena, M.A.; Srour, E.F. Cellular components of the hematopoietic niche and their regulation of hematopoietic stem cell function. Curr. Opin. Hematol. 2021, 28, 243–250. [Google Scholar] [CrossRef]
- Pinho, S.; Marchand, T.; Yang, E.; Wei, Q.; Nerlov, C.; Frenette, P.S. Lineage-Biased Hematopoietic Stem Cells Are Regulated by Distinct Niches. Dev. Cell 2018, 44, 634–641.e634. [Google Scholar] [CrossRef]
- Wei, Q.; Frenette, P.S. Niches for Hematopoietic Stem Cells and Their Progeny. Immunity 2018, 48, 632–648. [Google Scholar] [CrossRef]
- Chow, A.; Lucas, D.; Hidalgo, A.; Méndez-Ferrer, S.; Hashimoto, D.; Scheiermann, C.; Battista, M.; Leboeuf, M.; Prophete, C.; van Rooijen, N.; et al. Bone marrow CD169+ macrophages promote the retention of hematopoietic stem and progenitor cells in the mesenchymal stem cell niche. J. Exp. Med. 2011, 208, 261–271. [Google Scholar] [CrossRef]
- Seyfried, A.N.; Maloney, J.M.; MacNamara, K.C. Macrophages Orchestrate Hematopoietic Programs and Regulate HSC Function During Inflammatory Stress. Front. Immunol. 2020, 11, 1499. [Google Scholar] [CrossRef]
- Chang, K.H.; Sengupta, A.; Nayak, R.C.; Duran, A.; Lee, S.J.; Pratt, R.G.; Wellendorf, A.M.; Hill, S.E.; Watkins, M.; Gonzalez-Nieto, D.; et al. p62 is required for stem cell/progenitor retention through inhibition of IKK/NF-κB/Ccl4 signaling at the bone marrow macrophage-osteoblast niche. Cell Rep. 2014, 9, 2084–2097. [Google Scholar] [CrossRef]
- Chang, M.K.; Raggatt, L.J.; Alexander, K.A.; Kuliwaba, J.S.; Fazzalari, N.L.; Schroder, K.; Maylin, E.R.; Ripoll, V.M.; Hume, D.A.; Pettit, A.R. Osteal tissue macrophages are intercalated throughout human and mouse bone lining tissues and regulate osteoblast function in vitro and in vivo. J. Immunol. 2008, 181, 1232–1244. [Google Scholar] [CrossRef]
- Mohamad, S.F.; Xu, L.; Ghosh, J.; Childress, P.J.; Abeysekera, I.; Himes, E.R.; Wu, H.; Alvarez, M.B.; Davis, K.M.; Aguilar-Perez, A.; et al. Osteomacs interact with megakaryocytes and osteoblasts to regulate murine hematopoietic stem cell function. Blood Adv. 2017, 1, 2520–2528. [Google Scholar] [CrossRef]
- Winkler, I.G.; Sims, N.A.; Pettit, A.R.; Barbier, V.; Nowlan, B.; Helwani, F.; Poulton, I.J.; van Rooijen, N.; Alexander, K.A.; Raggatt, L.J.; et al. Bone marrow macrophages maintain hematopoietic stem cell (HSC) niches and their depletion mobilizes HSCs. Blood 2010, 116, 4815–4828. [Google Scholar] [CrossRef]
- Wysoczynski, M.; Moore, J.B.T.; Uchida, S. A novel macrophage subtype directs hematopoietic stem cell homing and retention. Ann. Transl. Med. 2019, 7 (Suppl. S3), S79. [Google Scholar] [CrossRef]
- Bruns, I.; Lucas, D.; Pinho, S.; Ahmed, J.; Lambert, M.P.; Kunisaki, Y.; Scheiermann, C.; Schiff, L.; Poncz, M.; Bergman, A.; et al. Megakaryocytes regulate hematopoietic stem cell quiescence through CXCL4 secretion. Nat. Med. 2014, 20, 1315–1320. [Google Scholar] [CrossRef]
- Cheshier, S.H.; Prohaska, S.S.; Weissman, I.L. The effect of bleeding on hematopoietic stem cell cycling and self-renewal. Stem Cells Dev. 2007, 16, 707–717. [Google Scholar] [CrossRef]
- Asquith, N.L.; Carminita, E.; Camacho, V.; Rodriguez-Romera, A.; Stegner, D.; Freire, D.; Becker, I.C.; Machlus, K.R.; Khan, A.O.; Italiano, J.E. The bone marrow is the primary site of thrombopoiesis. Blood 2024, 143, 272–278. [Google Scholar] [CrossRef]
- Decker, M.; Leslie, J.; Liu, Q.; Ding, L. Hepatic thrombopoietin is required for bone marrow hematopoietic stem cell maintenance. Science 2018, 360, 106–110. [Google Scholar] [CrossRef]
- Nakamura-Ishizu, A.; Takubo, K.; Fujioka, M.; Suda, T. Megakaryocytes are essential for HSC quiescence through the production of thrombopoietin. Biochem. Biophys. Res. Commun. 2014, 454, 353–357. [Google Scholar] [CrossRef]
- Qian, H.; Buza-Vidas, N.; Hyland, C.D.; Jensen, C.T.; Antonchuk, J.; Månsson, R.; Thoren, L.A.; Ekblom, M.; Alexander, W.S.; Jacobsen, S.E. Critical role of thrombopoietin in maintaining adult quiescent hematopoietic stem cells. Cell Stem Cell 2007, 1, 671–684. [Google Scholar] [CrossRef]
- Zhang, Y.; Lin, C.H.S.; Kaushansky, K.; Zhan, H. JAK2V617F Megakaryocytes Promote Hematopoietic Stem/Progenitor Cell Expansion in Mice through Thrombopoietin/MPL Signaling. Stem Cells 2018, 36, 1676–1684. [Google Scholar] [CrossRef]
- Broxmeyer, H.E. Chemokines in hematopoiesis. Curr. Opin. Hematol. 2008, 15, 49–58. [Google Scholar] [CrossRef]
- Nagasawa, T.; Hirota, S.; Tachibana, K.; Takakura, N.; Nishikawa, S.; Kitamura, Y.; Yoshida, N.; Kikutani, H.; Kishimoto, T. Defects of B-cell lymphopoiesis and bone-marrow myelopoiesis in mice lacking the CXC chemokine PBSF/SDF-1. Nature 1996, 382, 635–638. [Google Scholar] [CrossRef]
- Tokoyoda, K.; Egawa, T.; Sugiyama, T.; Choi, B.I.; Nagasawa, T. Cellular niches controlling B lymphocyte behavior within bone marrow during development. Immunity 2004, 20, 707–718. [Google Scholar] [CrossRef]
- Peled, A.; Petit, I.; Kollet, O.; Magid, M.; Ponomaryov, T.; Byk, T.; Nagler, A.; Ben-Hur, H.; Many, A.; Shultz, L.; et al. Dependence of human stem cell engraftment and repopulation of NOD/SCID mice on CXCR4. Science 1999, 283, 845–848. [Google Scholar] [CrossRef]
- Arai, F.; Ohneda, O.; Miyamoto, T.; Zhang, X.Q.; Suda, T. Mesenchymal stem cells in perichondrium express activated leukocyte cell adhesion molecule and participate in bone marrow formation. J. Exp. Med. 2002, 195, 1549–1563. [Google Scholar] [CrossRef]
- Greenbaum, A.; Hsu, Y.M.; Day, R.B.; Schuettpelz, L.G.; Christopher, M.J.; Borgerding, J.N.; Nagasawa, T.; Link, D.C. CXCL12 in early mesenchymal progenitors is required for haematopoietic stem-cell maintenance. Nature 2013, 495, 227–230. [Google Scholar] [CrossRef]
- Wu, J.Y.; Purton, L.E.; Rodda, S.J.; Chen, M.; Weinstein, L.S.; McMahon, A.P.; Scadden, D.T.; Kronenberg, H.M. Osteoblastic regulation of B lymphopoiesis is mediated by Gs{alpha}-dependent signaling pathways. Proc. Natl. Acad. Sci. USA 2008, 105, 16976–16981. [Google Scholar] [CrossRef]
- Terashima, A.; Okamoto, K.; Nakashima, T.; Akira, S.; Ikuta, K.; Takayanagi, H. Sepsis-Induced Osteoblast Ablation Causes Immunodeficiency. Immunity 2016, 44, 1434–1443. [Google Scholar] [CrossRef]
- Terashima, A.; Takayanagi, H. The role of bone cells in immune regulation during the course of infection. Semin. Immunopathol. 2019, 41, 619–626. [Google Scholar] [CrossRef]
- Copelan, E.A. Hematopoietic stem-cell transplantation. N. Engl. J. Med. 2006, 354, 1813–1826. [Google Scholar] [CrossRef]
- de Witte, T.; Bowen, D.; Robin, M.; Malcovati, L.; Niederwieser, D.; Yakoub-Agha, I.; Mufti, G.J.; Fenaux, P.; Sanz, G.; Martino, R.; et al. Allogeneic hematopoietic stem cell transplantation for MDS and CMML: Recommendations from an international expert panel. Blood 2017, 129, 1753–1762. [Google Scholar] [CrossRef]
- Duarte, R.F.; Labopin, M.; Bader, P.; Basak, G.W.; Bonini, C.; Chabannon, C.; Corbacioglu, S.; Dreger, P.; Dufour, C.; Gennery, A.R.; et al. Indications for haematopoietic stem cell transplantation for haematological diseases, solid tumours and immune disorders: Current practice in Europe, 2019. Bone Marrow Transpl. 2019, 54, 1525–1552. [Google Scholar] [CrossRef]
- Sauerer, T.; Velázquez, G.F.; Schmid, C. Relapse of acute myeloid leukemia after allogeneic stem cell transplantation: Immune escape mechanisms and current implications for therapy. Mol. Cancer 2023, 22, 180. [Google Scholar] [CrossRef] [PubMed]
- Thol, F.; Döhner, H.; Ganser, A. How I treat refractory and relapsed acute myeloid leukemia. Blood 2024, 143, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Felipe Rico, J.; Hassane, D.C.; Guzman, M.L. Acute Myelogenous Leukemia Stem Cells: From Bench to Bedside. Cancer Lett. 2013, 338, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Ferrer, S.; Bonnet, D.; Steensma, D.P.; Hasserjian, R.P.; Ghobrial, I.M.; Gribben, J.G.; Andreeff, M.; Krause, D.S. Bone marrow niches in haematological malignancies. Nat. Rev. Cancer 2020, 20, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Duarte, D.; Hawkins, E.D.; Akinduro, O.; Ang, H.; De Filippo, K.; Kong, I.Y.; Haltalli, M.; Ruivo, N.; Straszkowski, L.; Vervoort, S.J.; et al. Inhibition of Endosteal Vascular Niche Remodeling Rescues Hematopoietic Stem Cell Loss in AML. Cell Stem Cell 2018, 22, 64–77.e66. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.B.; Myers, L.M.; Noh, J.J.; Collins, M.M.; Carmody, A.B.; Messer, R.J.; Dhuey, E.; Hasenkrug, K.J.; Weissman, I.L. Depleting myeloid-biased haematopoietic stem cells rejuvenates aged immunity. Nature 2024, 628, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Kekre, N.; Antin, J.H. Hematopoietic stem cell transplantation donor sources in the 21st century: Choosing the ideal donor when a perfect match does not exist. Blood 2014, 124, 334–343. [Google Scholar] [CrossRef] [PubMed]
- van Lier, Y.F.; Vos, J.; Blom, B.; Hazenberg, M.D. Allogeneic hematopoietic cell transplantation, the microbiome, and graft-versus-host disease. Gut Microbes 2023, 15, 2178805. [Google Scholar] [CrossRef]
- Hu, Y.; Zhang, M.; Yang, T.; Mo, Z.; Wei, G.; Jing, R.; Zhao, H.; Chen, R.; Zu, C.; Gu, T.; et al. Sequential CD7 CAR T-Cell Therapy and Allogeneic HSCT without GVHD Prophylaxis. N. Engl. J. Med. 2024, 390, 1467–1480. [Google Scholar] [CrossRef]
- Zhao, C.; Bartock, M.; Jia, B.; Shah, N.; Claxton, D.F.; Wirk, B.; Rakszawski, K.L.; Nickolich, M.S.; Naik, S.G.; Rybka, W.B.; et al. Post-transplant cyclophosphamide alters immune signatures and leads to impaired T cell reconstitution in allogeneic hematopoietic stem cell transplant. J. Hematol. Oncol. 2022, 15, 64. [Google Scholar] [CrossRef]
- Hossain, M.S.; Kunter, G.M.; El-Najjar, V.F.; Jaye, D.L.; Al-Kadhimi, Z.; Taofeek, O.K.; Li, J.M.; Waller, E.K. PD-1 and CTLA-4 up regulation on donor T cells is insufficient to prevent GvHD in allo-HSCT recipients. PLoS ONE 2017, 12, e0184254. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Hasegawa, Y.; Hashimoto, D.; Senjo, H.; Kikuchi, R.; Chen, X.; Yoneda, K.; Sekiguchi, T.; Kawase, T.; Tsuzuki, H.; et al. Gilteritinib enhances graft-versus-leukemia effects against FLT3-ITD mutant leukemia after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transpl. 2022, 57, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Senjo, H.; Harada, S.; Kubota, S.I.; Tanaka, Y.; Tateno, T.; Zhang, Z.; Okada, S.; Chen, X.; Kikuchi, R.; Miyashita, N.; et al. Calcineurin inhibitor inhibits tolerance induction by suppressing terminal exhaustion of donor T cells after allo-HCT. Blood 2023, 142, 477–492. [Google Scholar] [CrossRef] [PubMed]
- Cassady, K.; Martin, P.J.; Zeng, D. Regulation of GVHD and GVL Activity via PD-L1 Interaction With PD-1 and CD80. Front. Immunol. 2018, 9, 3061. [Google Scholar] [CrossRef] [PubMed]
- Pan, B.; Shang, L.; Liu, C.; Gao, J.; Zhang, F.; Xu, M.; Li, L.; Sun, Z.; Li, Z.; Xu, K. PD-1 antibody and ruxolitinib enhances graft-versus-lymphoma effect without increasing acute graft-versus-host disease in mice. Am. J. Transpl. Off. J. Am. Soc. Transpl. Am. Soc. Transpl. Surg. 2021, 21, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Asano, T.; Meguri, Y.; Yoshioka, T.; Kishi, Y.; Iwamoto, M.; Nakamura, M.; Sando, Y.; Yagita, H.; Koreth, J.; Kim, H.T.; et al. PD-1 modulates regulatory T-cell homeostasis during low-dose interleukin-2 therapy. Blood 2017, 129, 2186–2197. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.P.; Cepika, A.M.; Agarwal-Hashmi, R.; Saini, G.; Uyeda, M.J.; Louis, D.M.; Cieniewicz, B.; Narula, M.; Amaya Hernandez, L.C.; Harre, N.; et al. Alloantigen-specific type 1 regulatory T cells suppress through CTLA-4 and PD-1 pathways and persist long-term in patients. Sci. Transl. Med. 2021, 13, eabf5264. [Google Scholar] [CrossRef] [PubMed]
- Leotta, S.; Sapienza, G.; Camuglia, M.G.; Avola, G.; Marco, A.D.; Moschetti, G.; Curto Pelle, A.; Markovic, U.; Milone, G.A.; Cupri, A.; et al. Preliminary Results of a Combined Score Based on sIL2-Rα and TIM-3 Levels Assayed Early after Hematopoietic Transplantation. Front. Immunol. 2019, 10, 3158. [Google Scholar] [CrossRef] [PubMed]
- Veenstra, R.G.; Taylor, P.A.; Zhou, Q.; Panoskaltsis-Mortari, A.; Hirashima, M.; Flynn, R.; Liu, D.; Anderson, A.C.; Strom, T.B.; Kuchroo, V.K.; et al. Contrasting acute graft-versus-host disease effects of Tim-3/galectin-9 pathway blockade dependent upon the presence of donor regulatory T cells. Blood 2012, 120, 682–690. [Google Scholar] [CrossRef]
- Stelljes, M.; Krug, U.; Beelen, D.W.; Braess, J.; Sauerland, M.C.; Heinecke, A.; Ligges, S.; Sauer, T.; Tschanter, P.; Thoennissen, G.B.; et al. Allogeneic transplantation versus chemotherapy as postremission therapy for acute myeloid leukemia: A prospective matched pairs analysis. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2014, 32, 288–296. [Google Scholar] [CrossRef]
- Fleischhauer, K.; Tran, T.H.; Meisel, R.; Mytilineos, J.; Dreger, P.; Kröger, N. Donor Selection for Allogeneic Hematopoietic Cell Transplantation. Dtsch. Arztebl. Int. 2023, 120, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Gooley, T.A.; Chien, J.W.; Pergam, S.A.; Hingorani, S.; Sorror, M.L.; Boeckh, M.; Martin, P.J.; Sandmaier, B.M.; Marr, K.A.; Appelbaum, F.R.; et al. Reduced mortality after allogeneic hematopoietic-cell transplantation. N. Engl. J. Med. 2010, 363, 2091–2101. [Google Scholar] [CrossRef] [PubMed]
- Sabloff, M.; Tisseverasinghe, S.; Babadagli, M.E.; Samant, R. Total Body Irradiation for Hematopoietic Stem Cell Transplantation: What Can We Agree on? Curr. Oncol. 2021, 28, 903–917. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, P.V.; Jones, R.J. The development of post-transplant cyclophosphamide: Half a century of translational team science. Blood Rev. 2023, 62, 101034. [Google Scholar] [CrossRef] [PubMed]
- Beelen, D.W.; Trenschel, R.; Stelljes, M.; Groth, C.; Masszi, T.; Reményi, P.; Wagner-Drouet, E.M.; Hauptrock, B.; Dreger, P.; Luft, T.; et al. Treosulfan or busulfan plus fludarabine as conditioning treatment before allogeneic haemopoietic stem cell transplantation for older patients with acute myeloid leukaemia or myelodysplastic syndrome (MC-FludT.14/L): A randomised, non-inferiority, phase 3 trial. Lancet. Haematol. 2020, 7, e28–e39. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Jaber, M.M.; Brown, S.J.; Al-Kofahi, M. Population Pharmacokinetic Model of Intravenous Busulfan in Hematopoietic Cell Transplantation: Systematic Review and Comparative Simulations. Clin. Pharmacokinet. 2023, 62, 955–968. [Google Scholar] [CrossRef] [PubMed]
- Epperla, N.; Ahn, K.W.; Armand, P.; Jaglowski, S.; Ahmed, S.; Kenkre, V.P.; Savani, B.; Jagasia, M.; Shah, N.N.; Fenske, T.S.; et al. Fludarabine and Busulfan versus Fludarabine, Cyclophosphamide, and Rituximab as Reduced-Intensity Conditioning for Allogeneic Transplantation in Follicular Lymphoma. Biol. Blood Marrow Transpl. J. Am. Soc. Blood Marrow Transpl. 2018, 24, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Mehta, R.S.; Rezvani, K. Immune reconstitution post allogeneic transplant and the impact of immune recovery on the risk of infection. Virulence 2016, 7, 901–916. [Google Scholar] [CrossRef] [PubMed]
- de Koning, C.; Nierkens, S.; Boelens, J.J. Strategies before, during, and after hematopoietic cell transplantation to improve T-cell immune reconstitution. Blood 2016, 128, 2607–2615. [Google Scholar] [CrossRef] [PubMed]
- Lucarelli, B.; Merli, P.; Bertaina, V.; Locatelli, F. Strategies to accelerate immune recovery after allogeneic hematopoietic stem cell transplantation. Expert Rev. Clin. Immunol. 2016, 12, 343–358. [Google Scholar] [CrossRef]
- Velardi, E.; Tsai, J.J.; van den Brink, M.R.M. T cell regeneration after immunological injury. Nat. Rev. Immunol. 2021, 21, 277–291. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.M.; Schlussel, L.; Zhao, L.; Himburg, H.A. Dickkopf-1 Treatment Stimulates Hematopoietic Regenerative Function in Infused Endothelial Progenitor Cells. Radiat. Res. 2019, 192, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Poulos, M.G.; Ramalingam, P.; Gutkin, M.C.; Llanos, P.; Gilleran, K.; Rabbany, S.Y.; Butler, J.M. Endothelial transplantation rejuvenates aged hematopoietic stem cell function. J. Clin. Investig. 2017, 127, 4163–4178. [Google Scholar] [CrossRef] [PubMed]
- Eissner, G.; Multhoff, G.; Gerbitz, A.; Kirchner, S.; Bauer, S.; Haffner, S.; Sondermann, D.; Andreesen, R.; Holler, E. Fludarabine induces apoptosis, activation, and allogenicity in human endothelial and epithelial cells: Protective effect of defibrotide. Blood 2002, 100, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Newell, L.F.; Deans, R.J.; Maziarz, R.T. Adult adherent stromal cells in the management of graft-versus-host disease. Expert. Opin. Biol. Ther. 2014, 14, 231–246. [Google Scholar] [CrossRef] [PubMed]
- Hess, N.J.; Kink, J.A.; Hematti, P. Exosomes, MDSCs and Tregs: A new frontier for GVHD prevention and treatment. Front. Immunol. 2023, 14, 1143381. [Google Scholar] [CrossRef]
- Nicolay, N.H.; Lopez Perez, R.; Saffrich, R.; Huber, P.E. Radio-resistant mesenchymal stem cells: Mechanisms of resistance and potential implications for the clinic. Oncotarget 2015, 6, 19366–19380. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, M.; Kim, B.S.; Yoon, S.; Oh, S.-O.; Lee, D. Hematopoietic Stem Cells and Their Niche in Bone Marrow. Int. J. Mol. Sci. 2024, 25, 6837. https://doi.org/10.3390/ijms25136837
Kwon M, Kim BS, Yoon S, Oh S-O, Lee D. Hematopoietic Stem Cells and Their Niche in Bone Marrow. International Journal of Molecular Sciences. 2024; 25(13):6837. https://doi.org/10.3390/ijms25136837
Chicago/Turabian StyleKwon, Munju, Byoung Soo Kim, Sik Yoon, Sae-Ock Oh, and Dongjun Lee. 2024. "Hematopoietic Stem Cells and Their Niche in Bone Marrow" International Journal of Molecular Sciences 25, no. 13: 6837. https://doi.org/10.3390/ijms25136837





