Effect of Diets with the Addition of Edible Insects on the Development of Atherosclerotic Lesions in ApoE/LDLR−/− Mice
Abstract
:1. Introduction
2. Results
2.1. Nutritive Value and Fatty Acid Composition of Insects
2.2. Total Polyphenols Content and Antioxidant Activity of Insects’ Powder
2.3. Composition of Experimental Diets
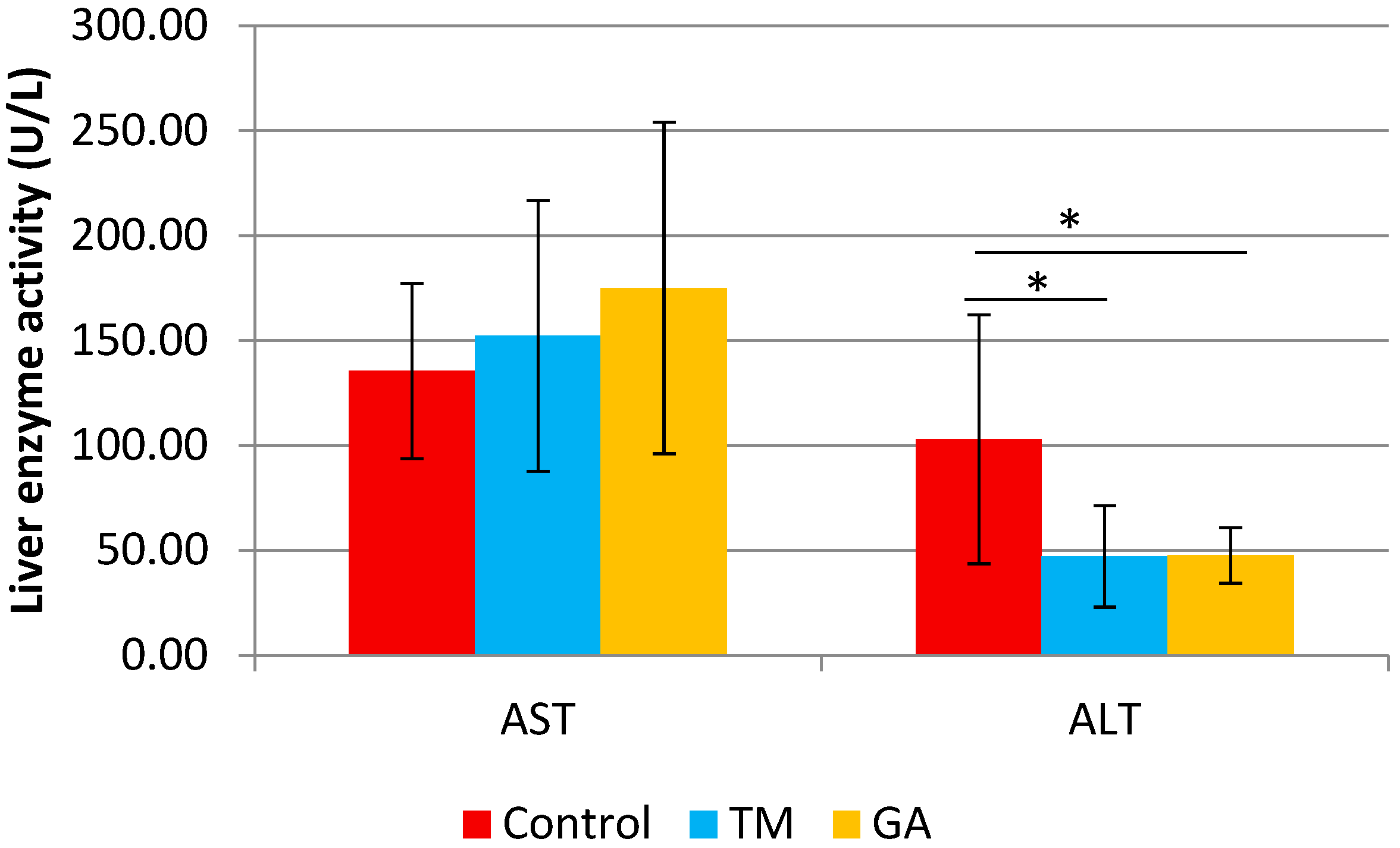
2.4. Effect of Edible Insects on Body and Liver Weight and Biochemical Parameters in ApoE/LDLR−/− Mice
2.5. Effect of Edible Insects on Fatty Acids Composition of Adipose Tissue and Liver in ApoE/LDLR−/− Mice
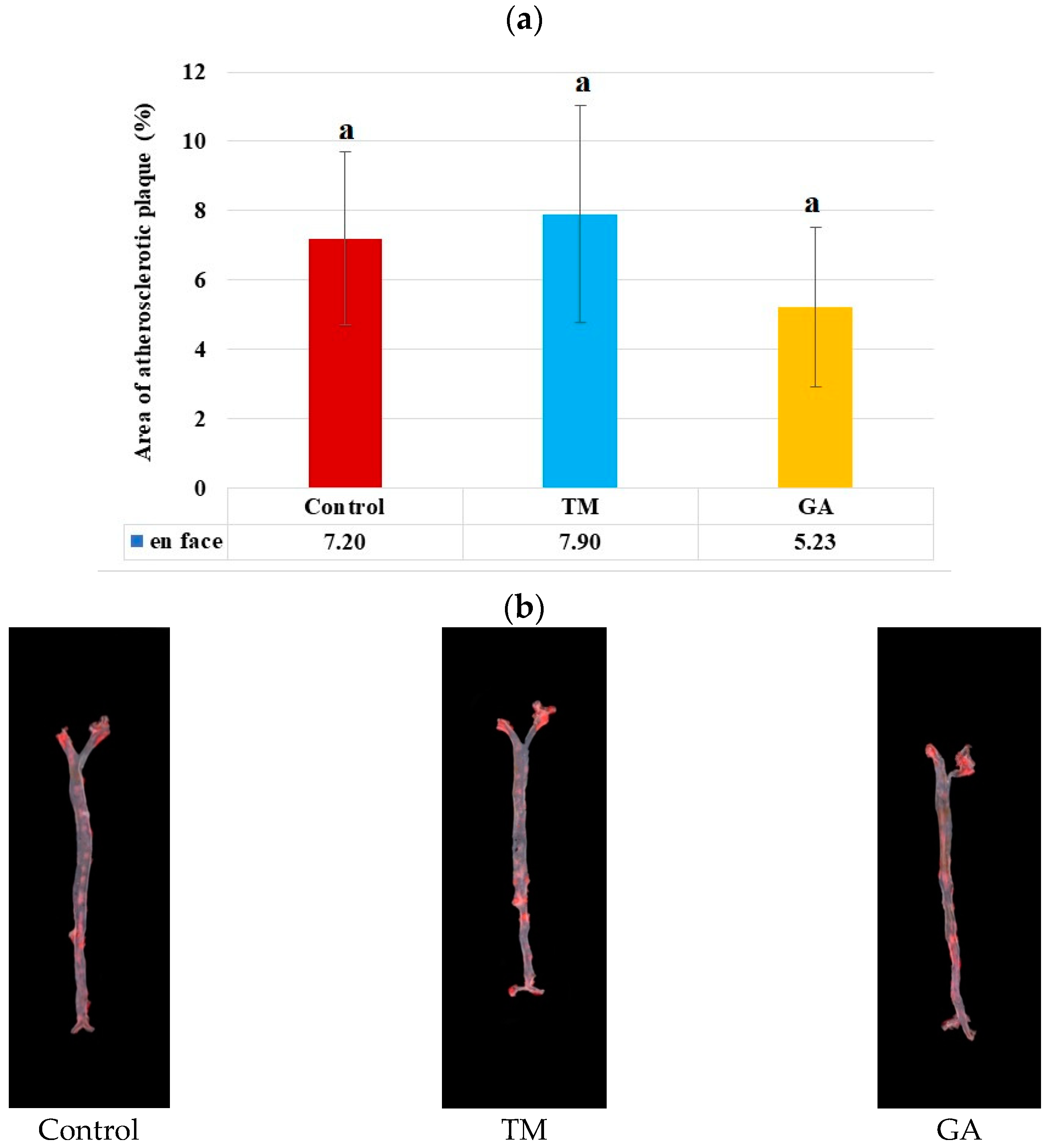
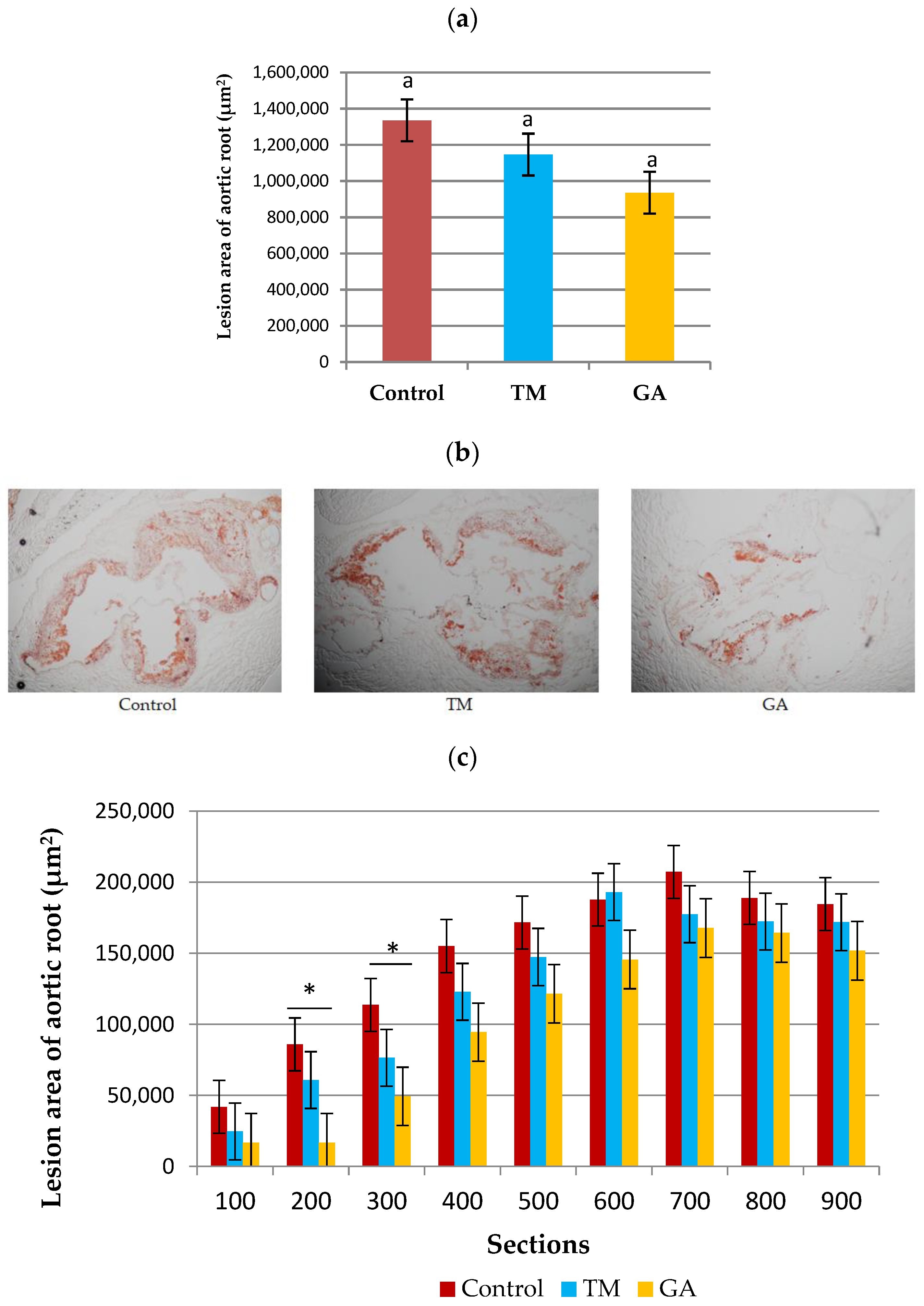
2.6. Effect of Edible Insects on the Development of Atherosclerosis in ApoE/LDLR−/− Mice
3. Discussion
4. Materials and Methods
4.1. Insects
4.1.1. Nutritive Value and Fatty Acid Composition of Insects
4.1.2. Determination of Total Polyphenols Content of Insects
4.1.3. Determination of Antioxidant Activity of Insects
ABTS•+ Radical Scavenging
DPPH• Radical Scavenging
Ferric Reducing Antioxidant Power (FRAP)
4.2. Animals and Housing
4.3. Diets and Feeding
4.4. Blood Sampling, Glucose, and Biochemical Analyses of Plasma
4.5. Fatty Acids Profile of Adipose Tissue and Liver
4.6. Quantification of Atherosclerosis in Aorta (En Face Method)
4.7. Quantification of Atherosclerosis in Aortic Roots (Cross-Section Method)
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roth, G.A.; Mensah, G.A.; Fuster, V. The global burden of cardiovascular diseases and risks: A compass for global action. J. Am. Coll. Cardiol. 2020, 76, 2980–2981. [Google Scholar] [CrossRef] [PubMed]
- Kobiyama, K.; Ley, K. Atherosclerosis: A chronic inflammatory disease with an autoimmune component. Circ. Res. 2018, 123, 1118–1120. [Google Scholar] [CrossRef] [PubMed]
- Boren, J.; Chapman, M.J.; Krauss, R.M.; Packard, C.J.; Bentzon, J.F.; Binder, C.J.; Daemen, M.J.; Demer, L.L.; Hegele, R.A.; Nicholls, S.J.; et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease: Pathophysiological, genetic, and therapeutic insights: A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur. Heart J. 2020, 41, 2313–2330. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.L.; Brown, M.S. A century of cholesterol and coronaries: From plaques to genes to statins. Cell 2015, 161, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Greenow, K.; Pearce, N.J.; Ramji, D.P. The key role of apolipoprotein E in atherosclerosis. J. Mol. Med. 2005, 83, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Knowles, J.W.; Maeda, N. Genetic modifiers of atherosclerosis in mice. Atheroscler. Thromb. Vasc. Biol. 2000, 20, 2336–2345. [Google Scholar] [CrossRef] [PubMed]
- Frostegård, J. Immunity, atherosclerosis and cardiovascular disease. BMC Med. 2013, 11, 117. [Google Scholar] [CrossRef]
- Kaminsky, L.A.; German, C.; Imboden, M.; Ozemek, C.; Peterman, J.E.; Brubaker, P.H. The importance of healthy lifestyle behaviors in the prevention of cardiovascular disease. Prog. Cardiovasc. Dis. 2022, 70, 8–15. [Google Scholar] [CrossRef]
- D’Antonio, V.; Battista, N.; Sacchetti, G.; Di Mattia, C.; Serafini, M. Functional properties of edible insects: A systematic review. Nutr. Res. Rev. 2023, 36, 98–119. [Google Scholar] [CrossRef]
- Payne, C.L.; Scarborough, P.; Rayner, M.; Nonaka, K. A systematic review of nutrient composition data available for twelve commercially available edible insects, and comparison with reference values. Trends Food Sci. Technol. 2016, 47, 69–77. [Google Scholar] [CrossRef]
- Nowakowski, A.C.; Miller, A.C.; Miller, M.E.; Xiao, H.; Wu, X. Potential health benefits of edible insects. Crit. Rev. Food Sci. Nutr. 2022, 62, 3499–3508. [Google Scholar] [CrossRef]
- Stull, V.J.; Finer, E.; Bergmans, R.S.; Febvre, H.P.; Longhurst, C.; Manter, D.K.; Patz, J.A.; Weir, T.L. Impact of edible cricket consumption on gut microbiota in healthy adults, a double-blind, randomized crossover trial. Sci. Rep. 2018, 8, 10762. [Google Scholar] [CrossRef] [PubMed]
- Chu, W.M. Tumor necrosis factor. Cancer Lett. 2013, 328, 222–225. [Google Scholar] [CrossRef]
- Rehman, K.U.; Hollah, C.; Wiesotzki, K.; Heinz, V.; Aganovic, K.; Rehman, R.U.; Petrusan, J.I.; Zheng, L.; Zhang, J.; Sohail, S.; et al. Insect-derived chitin and chitosan: A still unexploited resource for the edible insect sector. Sustainability 2023, 15, 4864. [Google Scholar] [CrossRef]
- Xia, W.; Liu, P.; Zhang, J.; Chen, J. Biological activities of chitosan and chitooligosaccharides. Food Hydrocoll. 2011, 25, 170–179. [Google Scholar] [CrossRef]
- Ahn, M.Y.; Hwang, J.S.; Kim, M.J.; Park, K.K. Antilipidemic effects and gene expression profiling of the glycosaminoglycans from cricket in rats on a high fat diet. Arch. Pharm. Res. 2016, 39, 926–936. [Google Scholar] [CrossRef] [PubMed]
- Ahn, M.Y.; Han, J.W.; Hwang, J.S.; Yun, E.Y.; Lee, B.M. Anti-inflammatory effect of glycosaminoglycan derived from Gryllus bimaculatus (a type of cricket, insect) on adjuvant-treated chronic arthritis rat model. Part A J. Toxicol. Environ. Health A 2014, 77, 1332–1345. [Google Scholar] [CrossRef]
- Ferrante, A.W., Jr. Obesity-induced inflammation: A metabolic dialogue in the language of inflammation. J. Intern. Med. 2007, 262, 408–414. [Google Scholar] [CrossRef]
- Quah, Y.; Tong, S.R.; Bojarska, J.; Giller, K.; Tan, S.A.; Ziora, Z.M.; Esatbeyoglu, T.; Chai, T.T. Bioactive peptide discovery from edible insects for potential applications in human health and agriculture. Molecules 2023, 28, 1233. [Google Scholar] [CrossRef]
- EFSA. European Parliament and Council. Regulation (EU) 2015/2283 of the European Parliament and of the Council of 25 November 2015 on novel foods, amending Regulation (EU) No 1169/2011 of the European Parliament and of the Council and repealing Regulation (EC) No 258/97 of the European Parliament and of the Council and Commission Regulation (EC) No 1852/2001. OJEU 2015. Available online: https://eurlex.europa.eu/eli/reg/2015/2283/oj (accessed on 11 May 2024).
- Raheem, D.; Carrascosa, C.; Oluwole, O.B.; Nieuwland, M.; Saraiva, A.; Millán, R.; Raposo, A. Traditional consumption of and rearing edible insects in Africa, Asia and Europe. Crit. Rev. Food Sci. Nutr. 2019, 59, 2169–2188. [Google Scholar] [CrossRef]
- Jongema, Y. List of Edible Insects of the World (April 1, 2017)—WUR. Available online: https://www.wur.nl/en/research-results/chair-groups/plant-sciences/laboratory-of-entomology/edible-insects/worldwide-species-list.htm (accessed on 10 May 2024).
- Oliveira, L.A.; Pereira, S.M.S.; Dias, K.A.; da Silva Paes, S.; Grancieri, M.; Jimenez, L.G.S.; Della, L.C.M. Nutritional content, amino acid profile, and protein properties of edible insects (Tenebrio molitor and Gryllus assimilis) powders at different stages of development. J. Food Compos. Anal. 2024, 125, 105804. [Google Scholar] [CrossRef]
- Gonzalez-de la Rosa, T.; Montserrat-de la Paz, S.; Rivero-Pino, F. Production, characterisation, and biological properties of Tenebrio molitor-derived oligopeptides. Food Chem. 2024, 450, 139400. [Google Scholar] [CrossRef] [PubMed]
- Khatun, H.; Claes, J.; Smets, R.; De Winne, A.; Akhtaruzzaman, M.; Van Der Borght, M. Characterization of freeze-dried, oven-dried and blanched house crickets (Acheta domesticus) and Jamaican field crickets (Gryllus assimilis) by means of their physicochemical properties and volatile compounds. Eur. Food Res. Technol. 2021, 247, 1291–1305. [Google Scholar] [CrossRef]
- Oonincx, D.G.A.B.; Finke, M.D. Nutritional value of insects and ways to manipulate their composition. JIFF 2021, 7, 639–659. [Google Scholar] [CrossRef]
- Adámková, A.; Mlček, J.; Adámek, M.; Borkovcová, M.; Bednářová, M.; Hlobilová, V.; Knížková, I.; Juríková, T. Tenebrio molitor (Coleoptera: Tenebrionidae)—Optimization of rearing conditions to obtain desired nutritional values. J. Insect Sci. 2020, 20, 24. [Google Scholar] [CrossRef] [PubMed]
- da Rosa Machado, C.; Thys, R.C.S. Cricket powder (Gryllus assimilis) as a new alternative protein source for gluten-free breads. Innov. Food Sci. Emerg. Technol. 2019, 56, 102180. [Google Scholar] [CrossRef]
- Araújo, R.R.S.; dos Santos Benfica, T.A.R.; Ferraz, V.P.; Santos, E.M. Nutritional composition of insects Gryllus assimilis and Zophobas morio: Potential foods harvested in Brazil. J. Food Compos. Anal. 2019, 76, 22–26. [Google Scholar] [CrossRef]
- Kulma, M.; Petříčková, D.; Kurečka, M.; Kotíková, Z.; Táborský, J.; Michlová, T.; Kouřimská, L. Effect of carrot supplementation on nutritional value of insects: A case study with Jamaican field cricket (Gryllus assimilis). JIFF 2022, 8, 621–629. [Google Scholar] [CrossRef]
- Syahrulawal, L.; Torske, M.O.; Sapkota, R.; Næss, G.; Khanal, P. Improving the nutritional values of yellow mealworm Tenebrio molitor (Coleoptera: Tenebrionidae) larvae as an animal feed ingredient: A review. J. Anim. Sci. Biotechnol. 2023, 14, 146. [Google Scholar] [CrossRef]
- Wu, R.A.; Ding, Q.; Yin, L.; Chi, X.; Sun, N.; He, R.; Luo, L.; Ma, H.; Li, Z. Comparison of the nutritional value of mysore thorn borer (Anoplophora chinensis) and mealworm larva (Tenebrio molitor): Amino acid, fatty acid, and element profiles. Food Chem. 2020, 323, 126818. [Google Scholar] [CrossRef]
- Santa-María, C.; López-Enríquez, S.; Montserrat-de la Paz, S.; Geniz, I.; Reyes-Quiroz, M.E.; Moreno, M.; Palomares, F.; Sobrino, F.; Alba, G. Update on anti-inflammatory molecular mechanisms induced by oleic acid. Nutrients 2023, 15, 224. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Babio, N.; Martínez-González, M.A.; Corella, D.; Ros, E.; Martin-Pelaez, S.; Estruch, R.; Arós, F.; Gómez-Gracia, E.; Fiol, M.; et al. Dietary fat intake and risk of cardiovascular disease and all-cause mortality in a population at high risk of cardiovascular disease. Am. J. Clin. Nutr. 2015, 102, 1563–1573. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, E.; Baraniak, B.; Karaś, M. Antioxidant and anti-inflammatory activities of hydrolysates and peptide fractions obtained by enzymatic hydrolysis of selected heat-treated edible insects. Nutrients 2017, 9, 970. [Google Scholar] [CrossRef] [PubMed]
- Messina, C.M.; Gaglio, R.; Morghese, M.; Tolone, M.; Arena, R.; Moschetti, G.; Santulli, A.; Francesca, N.; Settanni, L. Microbiological profile and bioactive properties of insect powders used in food and feed formulations. Foods 2019, 8, 400. [Google Scholar] [CrossRef] [PubMed]
- Pino, F.R.; Gálvez, R.P.; Carpio, F.J.E.; Guadix, E.M. Evaluation of Tenebrio molitor protein as a source of peptides for modulating physiological processes. Food Funct. 2020, 11, 4376–4386. [Google Scholar] [CrossRef]
- Mancini, S.; Fratini, F.; Turchi, B.; Mattioli, S.; Dal Bosco, A.; Tuccinardi, T.; Nozic, S.; Paci, G. Former food stuff products in Tenebrio molitor rearing: Effects on growth, chemical composition, microbiological load, and antioxidant status. Animals 2019, 9, 484. [Google Scholar] [CrossRef]
- Murugu, D.K.; Onyango, A.N.; Ndiritu, A.K.; Osuga, I.M.; Xavier, C.; Nakimbugwe, D.; Tanga, C.M. From farm to fork: Crickets as alternative source of protein, minerals, and vitamins. Front. Nutr. 2021, 8, 704002. [Google Scholar] [CrossRef]
- Kopecká, A.; Kouřimská, L.; Škvorová, P.; Kurečka, M.; Kulma, M. Effect of Temperature on the Nutritional Quality and Growth Parameters of Yellow Mealworm (Tenebrio molitor L.): A Preliminary Study. Appl. Sci. 2024, 14, 2610. [Google Scholar] [CrossRef]
- Di Mattia, C.; Battista, N.; Sacchetti, G.; Serafini, M. Antioxidant activities in vitro of water and liposoluble extracts obtained by different species of edible insects and invertebrates. Front. Nutr. 2019, 6, 106. [Google Scholar] [CrossRef]
- Oonincx, D.G.; Van Broekhoven, S.; Van Huis, A.; Van Loon, J.J. Feed conversion, survival and development, and composition of four insect species on diets composed of food by-products. PLoS ONE 2015, 10, e0144601. [Google Scholar] [CrossRef]
- Dreassi, E.; Cito, A.; Zanfini, A.; Materozzi, L.; Botta, M.; Francardi, V. Dietary fatty acids influence the growth and fatty acid composition of the yellow mealworm Tenebrio molitor (Coleoptera: Tenebrionidae). Lipids 2017, 52, 285–294. [Google Scholar] [CrossRef]
- Ruschioni, S.; Loreto, N.; Foligni, R.; Mannozzi, C.; Raffaelli, N.; Zamporlini, F.; Pasquini, M.; Roncolini, A.; Cardinali, F.; Osimani, A.; et al. Addition of olive pomace to feeding substrate affects growth performance and nutritional value of mealworm (Tenebrio molitor L.) larvae. Foods 2020, 9, 317. [Google Scholar] [CrossRef] [PubMed]
- Baek, M.; Kim, M.A.; Kwon, Y.S.; Hwang, J.S.; Goo, T.W.; Jun, M.; Yun, E.Y. Effects of processing methods on nutritional composition and antioxidant activity of mealworm (Tenebrio molitor) larvae. Entomol. Res. 2019, 49, 284–293. [Google Scholar] [CrossRef]
- Jozefiak, A.; Engberg, R.M. Insect proteins as a potential source of antimicrobial peptides in livestock production. A review. J. Anim. Feed Sci. 2017, 26, 87–99. [Google Scholar] [CrossRef]
- de Carvalho, N.M.; Madureira, A.R.; Pintado, M.E. The potential of insects as food sources–a review. Crit. Rev. Food Sci. Nutr. 2020, 60, 3642–3652. [Google Scholar] [CrossRef] [PubMed]
- Raubenheimer, D.; Rothman, J.M. Nutritional ecology of entomophagy in humans and other primates. Annu. Rev. Entomol. 2013, 58, 141–160. [Google Scholar] [CrossRef] [PubMed]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C. AIN-93 Purified Diets for Laboratory Rodents: Final Report of the American Institute of Nutrition Ad Hoc Writing Committee on the Reformulation of the AIN-76A Rodent Diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.; Goo, T.W.; Chung, M.Y.; Baek, M.; Hwang, J.S.; Kim, M.A.; Yun, E.Y. Tenebrio molitor larvae inhibit adipogenesis through AMPK and MAPKs signaling in 3T3-L1 adipocytes and obesity in high-fat diet-induced obese mice. Int. J. Mol. Sci. 2017, 18, 518. [Google Scholar] [CrossRef]
- Oibiokpa, F.I.; Akanya, H.O.; Jigam, A.A.; Saidu, A.N.; Egwim, E.C. Protein quality of four indigenous edible insect species in Nigeria. Food Sci. Hum. Wellness 2018, 7, 175–183. [Google Scholar] [CrossRef]
- Weru, J.; Chege, P.; Kinyuru, J. Nutritional potential of edible insects: A systematic review of published data. Int. J. Trop. Insect Sci. 2021, 41, 2015–2037. [Google Scholar] [CrossRef]
- Escobar-Ortiz, A.; Hernández-Saavedra, D.; Lizardi-Mendoza, J.; Pérez-Ramírez, I.F.; Mora-Izaguirre, O.; Ramos-Gómez, M.; Reynoso-Camacho, R. Consumption of cricket (Acheta domesticus) flour decreases insulin resistance and fat accumulation in rats fed with high-fat and-fructose diet. J. Food Biochem. 2022, 46, e14269. [Google Scholar] [CrossRef]
- Choi, R.Y.; Ham, J.R.; Ryu, H.S.; Lee, S.S.; Miguel, M.A.; Paik, M.J.; Ji, M.; Park, K.W.; Kang, K.Y.; Lee, H.I.; et al. Defatted Tenebrio molitor larva fermentation extract modifies steatosis, inflammation and intestinal microflora in chronic alcohol-fed rats. Nutrients 2020, 12, 1426. [Google Scholar] [CrossRef] [PubMed]
- Stocker, R.; Keaney, J.F., Jr. Role of oxidative modifications in atherosclerosis. Physiol. Rev. 2004, 84, 1381–1478. [Google Scholar] [CrossRef] [PubMed]
- Yamada, J.; Tomiyama, H.; Yambe, M.; Koji, Y.; Motobe, K.; Shiina, K.; Yamamoto, Y.; Yamashina, A. Elevated serum levels of alanine aminotransferase and gamma glutamyltransferase are markers of inflammation and oxidative stress independent of the metabolic syndrome. Atherosclerosis 2006, 189, 198–205. [Google Scholar] [CrossRef]
- Ahn, M.Y.; Kim, B.J.; Kim, H.J.; Jin, J.M.; Yoon, H.J.; Hwang, J.S.; Lee, B.M. Anti-diabetic activity of field cricket glycosaminoglycan by ameliorating oxidative stress. BMC Complement. Med. Ther. 2020, 20, 232. [Google Scholar] [CrossRef]
- Glick, N.R.; Fischer, M.H. The role of essential fatty acids in human health. Evid. Based Complement. Altern. Med. 2013, 18, 268–289. [Google Scholar] [CrossRef]
- Wang, D.; Bai, Y.Y.; Li, J.H.; Zhang, C.X. Nutritional value of the field cricket (Gryllus testaceus Walker). J. Insect Sci. 2004, 11, 275–283. [Google Scholar] [CrossRef]
- Paul, A.; Frederich, M.; Megido, R.C.; Alabi, T.; Malik, P.; Uyttenbroeck, R.; Francis, F.; Blecker, C.; Haubruge, E.; Lognay, G.; et al. Insect fatty acids: A comparison of lipids from three Orthopterans and Tenebrio molitor L. larvae. J. Asia-Pac. Entomol. 2017, 20, 337–340. [Google Scholar] [CrossRef]
- Green, C.J.; Hodson, L. The influence of dietary fat on liver fat accumulation. Nutrients 2014, 6, 5018–5033. [Google Scholar] [CrossRef]
- Rosqvist, F.; Kullberg, J.; Ståhlman, M.; Cedernaes, J.; Heurling, K.; Johansson, H.E.; Iggman, D.; Wilking, H.; Larsson, A.; Eriksson, O.; et al. Overeating saturated fat promotes fatty liver and ceramides compared with polyunsaturated fat: A randomized trial. J. Clin. Endocrinol. Metab. 2019, 104, 6207–6219. [Google Scholar] [CrossRef]
- Ullah, R.; Rauf, N.; Nabi, G.; Ullah, H.; Shen, Y.; Zhou, Y.D.; Fu, J. Role of nutrition in the pathogenesis and prevention of non-alcoholic fatty liver disease: Recent updates. Int. J. Biol. Sci. 2019, 15, 265. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Eguchi, K.; Kono, N.; Fujiu, K.; Matsumoto, S.; Shibata, M.; Oishi-Tanaka, Y.; Komuro, I.; Arai, H.; Nagai, R.; et al. Saturated fatty acid palmitate aggravates neointima formation by promoting smooth muscle phenotypic modulation. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 2596–2607. [Google Scholar] [CrossRef]
- Staiger, K.; Staiger, H.; Weigert, C.; Haas, C.; Häring, H.U.; Kellerer, M. Saturated, but not unsaturated, fatty acids induce apoptosis of human coronary artery endothelial cells via nuclear factor-κB activation. Diabetes 2006, 55, 3121–3126. [Google Scholar] [CrossRef] [PubMed]
- Kinyuru, J.N.; Mogendi, J.B.; Riwa, C.A.; Ndung’u, N.W. Edible insects—A novel source of essential nutrients for human diet: Learning from traditional knowledge. Anim. Front. 2015, 5, 14–19. [Google Scholar]
- Khosravi, M.; Poursaleh, A.; Ghasempour, G.; Farhad, S.; Najafi, M. The effects of oxidative stress on the development of atherosclerosis. J. Biol. Chem. 2019, 400, 711–732. [Google Scholar] [CrossRef]
- D’Antonio, V.; Serafini, M.; Battista, N. Dietary modulation of oxidative stress from edible insects: A mini-review. Front. Nutr. 2021, 8, 642551. [Google Scholar] [CrossRef]
- Kurdi, P.; Chaowiwat, P.; Weston, J.; Hansawasdi, C. Studies on microbial quality, protein yield, and antioxidant properties of some frozen edible insects. Int. J. Food Sci. Technol. 2021, 2021, 5580976. [Google Scholar] [CrossRef]
- Gumul, D.; Oracz, J.; Kowalski, S.; Mikulec, A.; Skotnicka, M.; Karwowska, K.; Areczuk, A. Bioactive compounds and antioxidant composition of nut bars with addition of various edible insect flours. Molecules 2023, 28, 3556. [Google Scholar] [CrossRef]
- Malekmohammad, K.; Sewell, R.D.; Rafieian-Kopaei, M. Antioxidants and atherosclerosis: Mechanistic aspects. Biomolecules 2019, 9, 301. [Google Scholar] [CrossRef]
- Muscolo, A.; Mariateresa, O.; Giulio, T.; Mariateresa, R. Oxidative stress: The role of antioxidant phytochemicals in the prevention and treatment of diseases. Int. J. Mol. Sci. 2024, 25, 3264. [Google Scholar] [CrossRef]
- Kulma, M.; Kouřimská, L.; Plachý, V.; Božik, M.; Adámková, A.; Vrabec, V. Effect of sex on the nutritional value of house cricket, Acheta domestica L. Food Chem. 2019, 272, 267–272. [Google Scholar] [CrossRef]
- Franczyk-Żarów, M.; Szymczyk, B.; Kostogrys, R.B. Effects of dietary conjugated linoleic acid and selected vegetable oils or vitamin E on fatty acid composition of hen egg yolks. Ann. Anim. Sci. 2019, 19, 173–188. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C.L.W.T. Use of a free radical method to evaluate antioxidant activity. LWT—Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Bonthu, S.; Heistad, D.D.; Chappell, D.A.; Lamping, K.G.; Faraci, F.M. Atherosclerosis, vascular remodeling, and impairment of endothelium-dependent relaxation in genetically altered hyperlipidemic mice. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 2333–2340. [Google Scholar] [CrossRef] [PubMed]
- Jawien, J.; Nastalek, P.; Korbut, R. Mouse models of experimental atherosclerosis. J. Physiol. Pharmacol. 2004, 55, 503–517. [Google Scholar] [PubMed]
- Yamada, N.; Shimano, H.; Yazaki, Y. Role of apolipoprotein E in lipoprotein metabolism and in the process of atherosclerosis. J. Atheroscler. Thromb. 1995, 2 (Suppl. S1), S29–S33. [Google Scholar] [CrossRef]
- Man, J.J.; Beckman, J.A.; Jaffe, I.Z. Sex as a biological variable in atherosclerosis. Circ. Res. 2020, 126, 1297–1319. [Google Scholar] [CrossRef]
- Franczyk-Żarów, M.; Kostogrys, R.B.; Szymczyk, B.; Jawień, J.; Gajda, M.; Cichocki, T.; Wojnar, L.; Chlopicki, S.; Pisulewski, P.M. Functional effects of eggs, naturally enriched with conjugated linoleic acid, on the blood lipid profile, development of atherosclerosis and composition of atherosclerotic plaque in apolipoprotein E and low-density lipoprotein receptor double-knockout mice (ApoE/LDLR−/−). Br. J. Nutr. 2008, 99, 49–58. [Google Scholar] [CrossRef]
- Centa, M.; Ketelhuth, D.F.; Malin, S.; Gisterå, A. Quantification of atherosclerosis in mice. J. Vis. Exp. 2019, 148, e59828. [Google Scholar] [CrossRef]
| Insect | DM (g/100 g Insects’ Powder) | Ash (g/100 g DM) | Crude Protein (g/100 g DM) | Fat (g/100 g DM) | Chitin (g/100 g DM) |
|---|---|---|---|---|---|
| TM | 96.19 ± 0.11 | 3.39 a ± 0.06 | 57.06 a ± 0.14 | 28.67 a ± 0.02 | 9.36 a ± 0.07 |
| GA | 96.10 ± 0.14 | 3.90 b ± 0.08 | 67.04 b ± 0.35 | 22.33 b ± 0.28 | 7.03 b ± 0.02 |
| Systematic Name (IUPAC) | Number of Carbon Atoms: Number π Bonds | Insect | |
|---|---|---|---|
| TM | GA | ||
| Dodecanoic acid | C12:0 | 0.46 a ± 0.02 | 0.19 b ± 0.08 |
| Tetradecanoic acid | C14:0 | 5.58 a ± 0.27 | 1.81 b ± 0.89 |
| Pentadecanoic acid | C15:0 | 0.21 ± 0.02 | 0.23 ± 0.05 |
| Hexadecanoic acid | C16:0 | 18.84 a ± 0.30 | 22.16 b ± 1.53 |
| cis-9-Hexadecenoic acid | C16:1 | 5.06 ± 0.22 | 4.65 ± 0.34 |
| Heptadecanoic acid | C17:0 | 0.3 ± 0.05 | 0.27 ± 0.05 |
| cis,cis-9,12-Hexadecadienoic acid | C16:2 | 0.89 a ± 0.02 | 0.41 b ± 0.25 |
| Octadecanoic acid | C18:0 | 8.45 a ± 0.14 | 11.98 b ± 0.81 |
| cis-9-Octadecenoic acid | C18:1 | 36.13 a ± 0.71 | 30.61 b ± 1.26 |
| Octadecadienoic acid | C18:2 | 20.44 a ± 0.57 | 23.43 b ± 0.76 |
| all cis-9,12,15-Octadecatrienoic acid | C18:3 (n − 3) | 2.58 a ± 0.05 | 3.27 b ± 0.19 |
| Other fatty acids | 1.07 ± 0.17 | 0.98 ± 0.53 | |
| MUFA | 41.19 a ± 0.92 | 35.27 b ± 1.51 | |
| PUFA | 23.91 a ± 0.52 | 27.11 b ± 0.40 | |
| SFA | 33.84 a ± 0.49 | 36.64 b ± 0.87 |
| Parameters | Insect | |
|---|---|---|
| TM | GA | |
| Polyphenols (mg GAE/100 g DM) | 567.92 ± 34.05 | 568.85 ± 31.15 |
| TEAC ABTS •+ (μΜTrolox/1 g DM) | 47.3 a ± 0.42 | 89.94 b ± 0.7 |
| RSA ABTS •+ (%) | 77.3 ± 0.42 | 71.2 ± 0.28 |
| TEAC DPPH• (μΜTrolox/1 g DM) | 81.52 a ± 0.92 | 178.63 b ± 1.46 |
| RSA DPPH• (%) | 76.15 ± 0.49 | 75.75 ± 0.35 |
| TEAC TPTZ (FRAP) (μmolTrolox/1 g DM) | 65.27 ± 10.28 | 54.36 ± 7.18 |
| Ingredient (g/kg) | AIN-93G (Control) | AIN-93G + 10% TM (TM) | AIN-93G + 10% GA (GA) |
|---|---|---|---|
| Casein | 200 | 146 | 144 |
| Protein from insect | - | 54 | 56 |
| Cornstarch | 533 | 533 | 533 |
| Sucrose | 100 | 100 | 100 |
| Cellulose | 50 | 44 | 43 |
| Chitin from insect | - | 6 | 7 |
| Soybean oil | 70 | 34 | 53 |
| Fat from insect | - | 36 | 17 |
| Vitamin mixture | 10 | 10 | 10 |
| Mineral mixture | 35 | 35 | 35 |
| Choline bitartrate | 2.5 | 2.5 | 2.5 |
| t-butylhydroquinone | 0.014 | 0.014 | 0.014 |
| TM | - | 100 | - |
| GA | - | - | 100 |
| Parameters | Experimental Groups | ||
|---|---|---|---|
| Control | TM | GA | |
| Body weight (g) | 20.96 ± 1.37 | 20.39 ± 1.24 | 19.87 ± 1.99 |
| Liver weight (g/100 g) | 4.18 ab ± 0.32 | 3.86 a ± 0.44 | 4.39 b ± 0.48 |
| Blood glucose (mg/dL) | 132.30 ± 14.31 | 118.50 ± 22.01 | 132.82 ± 22.54 |
| TC (mmol/L) | 20.58 ± 2.20 | 22.75 ± 2.51 | 21.92 ± 2.74 |
| LDL (mmol/L) | 8.86 ± 1.01 | 9.14 ± 1.42 | 8.01 ± 2.21 |
| HDL (mmol/L) | 1.00 ± 0.13 | 0.85 ± 0.45 | 0.85 ± 0.18 |
| TAG (mmol/L) | 2.34 ± 0.58 | 1.85 ± 0.39 | 2.13 ± 0.47 |
| Systematic Name (IUPAC) | Number of Carbon Atoms: Number π Bonds | Experimental Groups | ||
|---|---|---|---|---|
| Control | TM | GA | ||
| Dodecanoic acid | C12:0 | 0.15 a ± 0.02 | 0.20 b ± 0.01 | 0.14 a ± 0.03 |
| Tetradecanoic acid | C14:0 | 2.45 b ± 0.21 | 2.85 c ± 0.20 | 2.16 a ± 0.26 |
| Pentadecanoic acid | C15:0 | 0.15 a ± 0.03 | 0.22 b ± 0.05 | 0.13 a ± 0.02 |
| 14-Methylpentadecanoic acid | C16:0 iso | 0.15 a ± 0.03 | 0.15 a ± 0.02 | 0.13 a ± 0.02 |
| Hexadecanoic acid | C16:0 | 21.48 a ± 1.59 | 19.80 b ± 0.46 | 20.58 a,b ± 0.77 |
| cis-9-Hexadecenoic acid | C16:1 | 3.30 a ± 0.68 | 4.43 b ± 0.61 | 3.21 a ± 0.56 |
| Heptadecanoic acid | C17:0 | 0.37 a ± 0.08 | 0.51 b ± 0.07 | 0.44 a,b ± 0.08 |
| cis-10-Heptadecanoic acid | C17:1 | 0.42 a ± 0.07 | 0.62 b ± 0.05 | 0.55 a ± 0.06 |
| Octadecanoic acid | C18:0 | 12.42 a ± 1.60 | 11.70 a ± 1.61 | 13.25 a ± 1.67 |
| cis-9-Octadecenoic acid | C18:1 | 29.03 a ± 0.84 | 32.96 c ± 0.46 | 30.41 b ± 1.09 |
| Nonadecanoic acid | C19:0 | 0.13 a ± 0.09 | 0.20 a ± 0.06 | 0.21 a ± 0.08 |
| cis,cis-9,12-Octadecadienoic acid | C18:2 (n − 6) | 23.96 a ± 1.09 | 21.07 b ± 1.20 | 23.26 a ± 1.33 |
| all cis-6,9,12-Octadecatrienoic acid | C18:3 (n − 6) | 0.36 a ± 0.12 | 0.29 a ± 0.06 | 0.36 a ± 0.11 |
| all cis-9,12,15-Octadecatrienoic acid | C18:3 (n−3) | 4.72 a ± 1.46 | 4.32 a ± 0.43 | 4.53 a ± 0.56 |
| cis,cis-11,14-Eicosadienoic acid | C20:2 (n − 6) | 0.32 b ± 0.32 | 0.15 a,b ± 0.05 | 0.12 a ± 0.03 |
| all cis-5,8,11,14-Eicosatetraenoic acid | C20:4 (n − 6) | 0.13 a ± 0.06 | 0.14 a ± 0.03 | 0.11 a ± 0.03 |
| Other fatty acids | 0.33 a ± 0.17 | 0.29 a ± 0.17 | 0.37 a ± 0.08 | |
| Total SFA | 37.30 a ± 1.42 | 35.64 a ± 1.41 | 37.04 a ± 1.75 | |
| Total MUFA | 32.74 a ± 1.08 | 38.01 b ± 0.51 | 34.17 a ± 1.55 | |
| Total PUFA | 29.63 a ± 1.37 | 26.06 b ± 1.17 | 28.43 a ±1.56 | |
| 18:2/18:3 (n−6/n−3) | 5.66 a ± 2.13 | 4.93 a ± 0.65 | 5.20 a ± 0.65 | |
| 18:1/18:0 | 2.37 a ± 0.30 | 2.86 b ± 0.39 | 2.33 a ± 0.35 | |
| 16:1/16:0 | 0.15 a ± 0.03 | 0.22 b ± 0.03 | 0.16 a ± 0.03 | |
| 16:0/14:0 | 8.84 a ± 0.96 | 6.97 b ± 0.51 | 9.69 a ± 1.45 | |
| 18:0/16:0 | 0.58 a ± 0.11 | 0.59 a ± 0.09 | 0.64 a ± 0.09 | |
| 16:0/18:2 | 0.90 a,b ± 0.05 | 0.94 b ± 0.05 | 0.89 a ± 0.05 | |
| Systematic Name (IUPAC) | Number of Carbon Atoms: Number π Bonds | Experimental Groups | ||
|---|---|---|---|---|
| Control | TM | GA | ||
| Dodecanoic acid | C12:0 | 0.06 a ± 0.03 | 0.05 a ± 0.03 | 0.06 a ± 0.03 |
| Tetradecanoic acid | C14:0 | 1.05 a ± 0.43 | 0.90 a ± 0.22 | 0.91 a ± 0.13 |
| Pentadecanoic acid | C15:0 | 0.15 a ± 0.03 | 0.17 a ± 0.06 | 0.15 a ± 0.03 |
| 12-Methylpentadecanoic acid | C16:0 iso | 0.05 a ± 0.02 | 0.06 a ± 0.02 | 0.05 a ± 0.01 |
| Hexadecanoic acid | C16:0 | 21.81 a ± 0.86 | 23.17 a ± 2.55 | 22.73 a ± 1.05 |
| cis-9-Hexadecenoic acid | C16:1 | 3.84 b ± 1.33 | 3.19 a,b ± 1.03 | 2.69 a ± 0.60 |
| cis,cis-9,12-Hexadecadienoic acid | C16:2 | 0.32 a ± 0.11 | 0.36 a ± 0.10 | 0.41 a ± 0.13 |
| Heptadecanoic acid | C17:0 | 0.36 a ± 0.05 | 0.65 a ± 0.73 | 0.48 a ± 0.08 |
| Octadecanoic acid | C18:0 | 9.81 a,b ± 0.89 | 9.42 a ± 1.25 | 10.77 b ± 1.02 |
| cis-9-Octadecenoic acid | C18:1 | 28.80 a ± 1.16 | 31.76 b ± 0.77 | 28.58 a ± 1.05 |
| cis,cis-9,12-Octadecadienoic acid | C18:2(n − 6) | 23.16 b ± 1.99 | 21.28 a ± 1.13 | 22.92 a,b ± 1.40 |
| all cis-6,9,12-Octadecatrienoic acid | C18:3 (n − 6) | 1.07 a ± 0.28 | 0.84 a ± 0.25 | 0.95 a ± 0.18 |
| all cis-9,12,15-Octadecatrienoic acid | C18:3 (n−3) | 3.22 b ± 0.47 | 2.55 a ± 0.79 | 3.09 a,b ± 0.45 |
| cis,cis-11,14-Eicosadienoic acid | C20:2 (n − 6) | 0.74 a ±0.26 | 0.60 a ± 0.17 | 0.66 a ± 0.16 |
| all cis-5,8,11,14-Eicosatetraenoic acid | C20:4 (n − 6) | 3.07 a ± 0.64 | 3.00 a ± 0.62 | 3.18 a ± 0.94 |
| all cis-4,7,10,13,16-Docosapentaenoic acid (DPA) | C22:5 (n − 6) | 0.21 b ± 0.10 | 0.15 a,b ± 0.08 | 0.10 a ± 0.04 |
| all cis-4,7,10,13,16,19-Docosahexaenoic acid (DHA) | C22:6 (n − 3) | 1.48 a ± 0.33 | 1.28 a ± 0.35 | 1.49 a ± 0.54 |
| Other fatty acids | 0.49 a,b ± 0.14 | 0.37 a ± 0.15 | 0.55 b ± 0.17 | |
| Total SFA | 33.29 a ± 1.62 | 34.40 a,b ± 1.77 | 35.14 b ± 1.28 | |
| Total MUFA | 32.64 a ± 1.89 | 34.95 b ± 1.37 | 31.27 a ± 1.18 | |
| Total PUFA | 33.58 a ± 2.27 | 30.28 b ± 1.70 | 33.04 a ± 1.92 | |
| 18:2/18:3 (n-6/n-3) | 7.35 a ± 1.41 | 11.44 a ± 11.91 | 7.55 a ± 1.14 | |
| 18:1/18:0 | 2.96 a ± 0.33 | 3.44 b ± 0.56 | 2.68 a ± 0.31 | |
| 16:1/16:0 | 0.18 b ± 0.06 | 0.14 a,b ± 0.05 | 0.12 a ± 0.02 | |
| 16:0/14:0 | 23.98 a ± 10.11 | 28.68 a ± 15.16 | 25.28 a ± 2.43 | |
| 18:0/16:0 | 0.45 a ± 0.04 | 0.41 a ± 0.09 | 0.48 a ± 0.05 | |
| 16:0/18:2 | 0.95 a ± 0.09 | 1.09 b ± 0.14 | 0.99 a,b ± 0.07 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hassen, H.; Škvorová, P.; Pokhrel, K.; Kulma, M.; Piątkowska, E.; Kostogrys, R.B.; Kouřimská, L.; Tarko, T.; Franczyk-Żarów, M. Effect of Diets with the Addition of Edible Insects on the Development of Atherosclerotic Lesions in ApoE/LDLR−/− Mice. Int. J. Mol. Sci. 2024, 25, 7256. https://doi.org/10.3390/ijms25137256
Hassen H, Škvorová P, Pokhrel K, Kulma M, Piątkowska E, Kostogrys RB, Kouřimská L, Tarko T, Franczyk-Żarów M. Effect of Diets with the Addition of Edible Insects on the Development of Atherosclerotic Lesions in ApoE/LDLR−/− Mice. International Journal of Molecular Sciences. 2024; 25(13):7256. https://doi.org/10.3390/ijms25137256
Chicago/Turabian StyleHassen, Hayat, Petra Škvorová, Kshitiz Pokhrel, Martin Kulma, Ewa Piątkowska, Renata B. Kostogrys, Lenka Kouřimská, Tomasz Tarko, and Magdalena Franczyk-Żarów. 2024. "Effect of Diets with the Addition of Edible Insects on the Development of Atherosclerotic Lesions in ApoE/LDLR−/− Mice" International Journal of Molecular Sciences 25, no. 13: 7256. https://doi.org/10.3390/ijms25137256





