Potential for Drug Repositioning of Midazolam as an Inhibitor of Inflammatory Bone Resorption
Abstract
:1. Introduction
2. Results
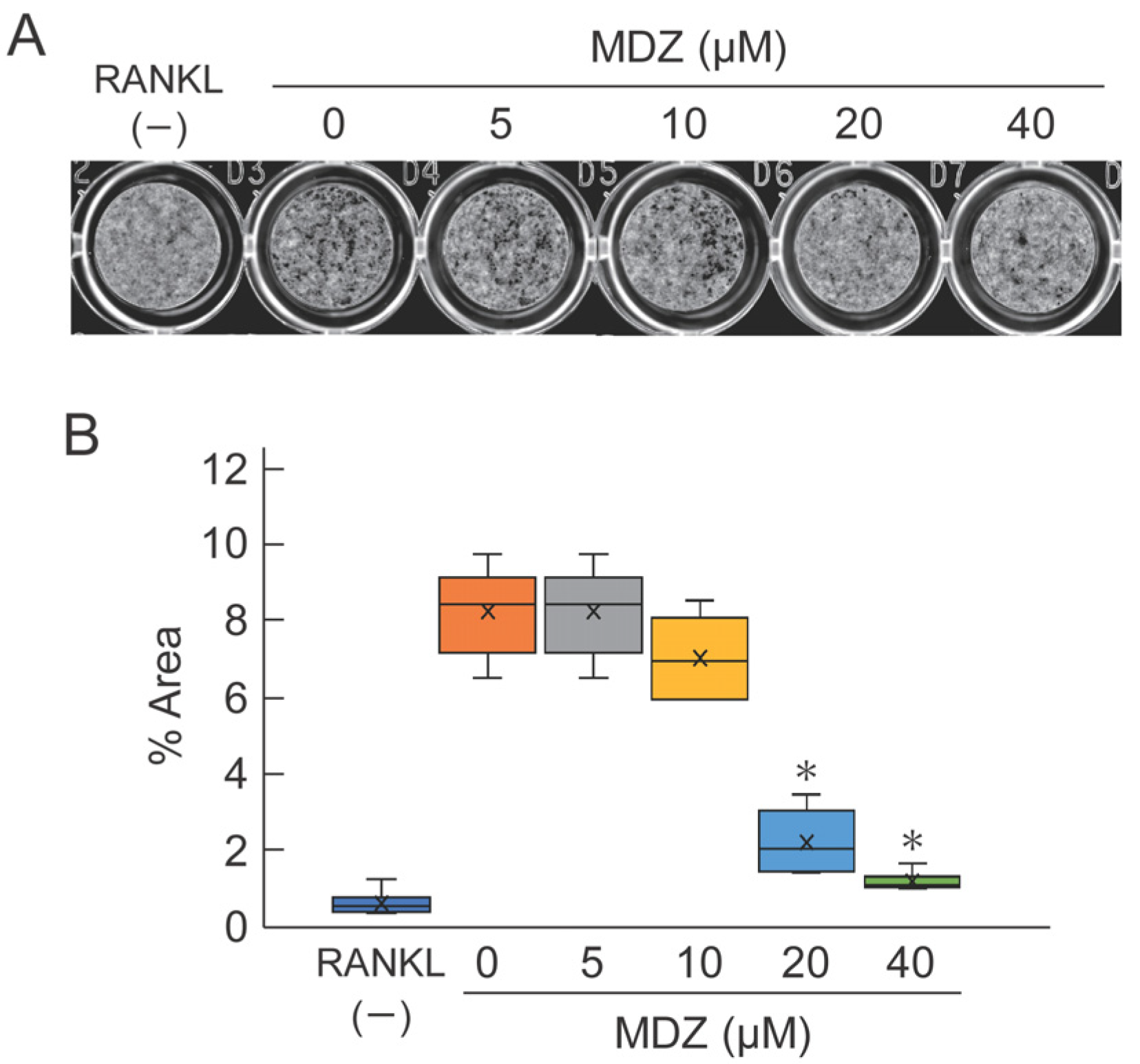
2.1. Effect of MDZ on the Differentiation of RAW264 Cells into Osteoclasts
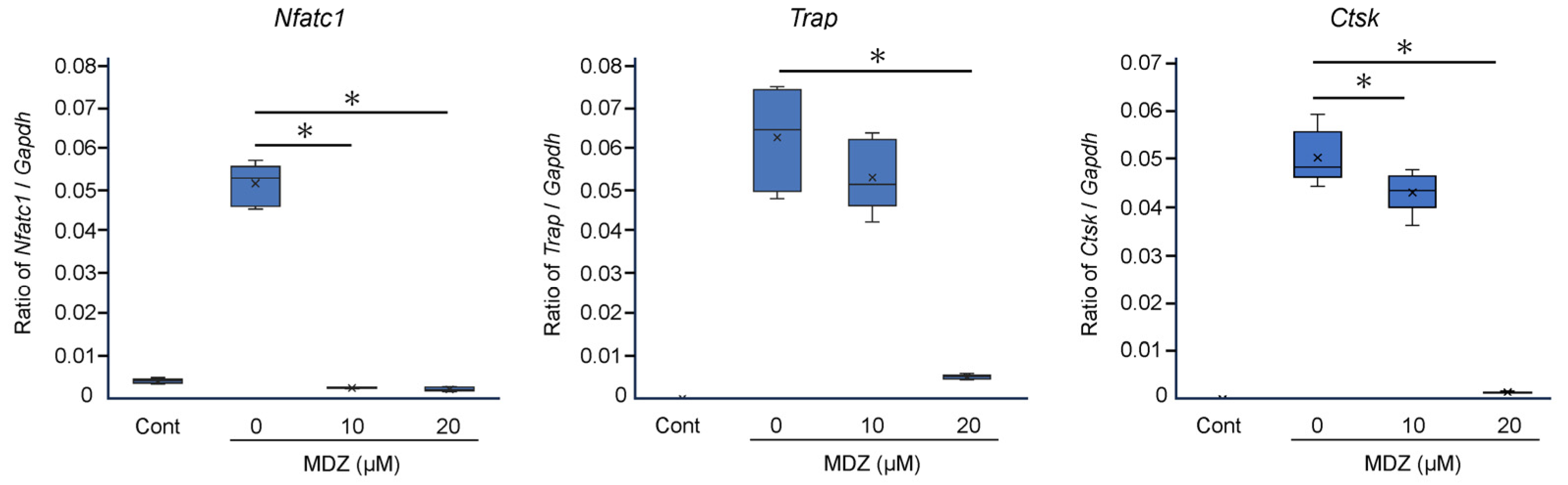
2.2. Effect of MDZ on the Expression of Osteoclast Differentiation Marker Genes
2.3. Effect of MDZ on Osteoclast-Mediated Bone Resorption
2.4. Animal Experiments
2.4.1. Morphological Assessment of the Effect of MDZ on Lipopolysaccharide (LPS)-Induced Calvarial Mouse
2.4.2. Validation of Suture Width and Bone Matrix Volume on Bone Destruction in LPS-Induced Calvarial Mice
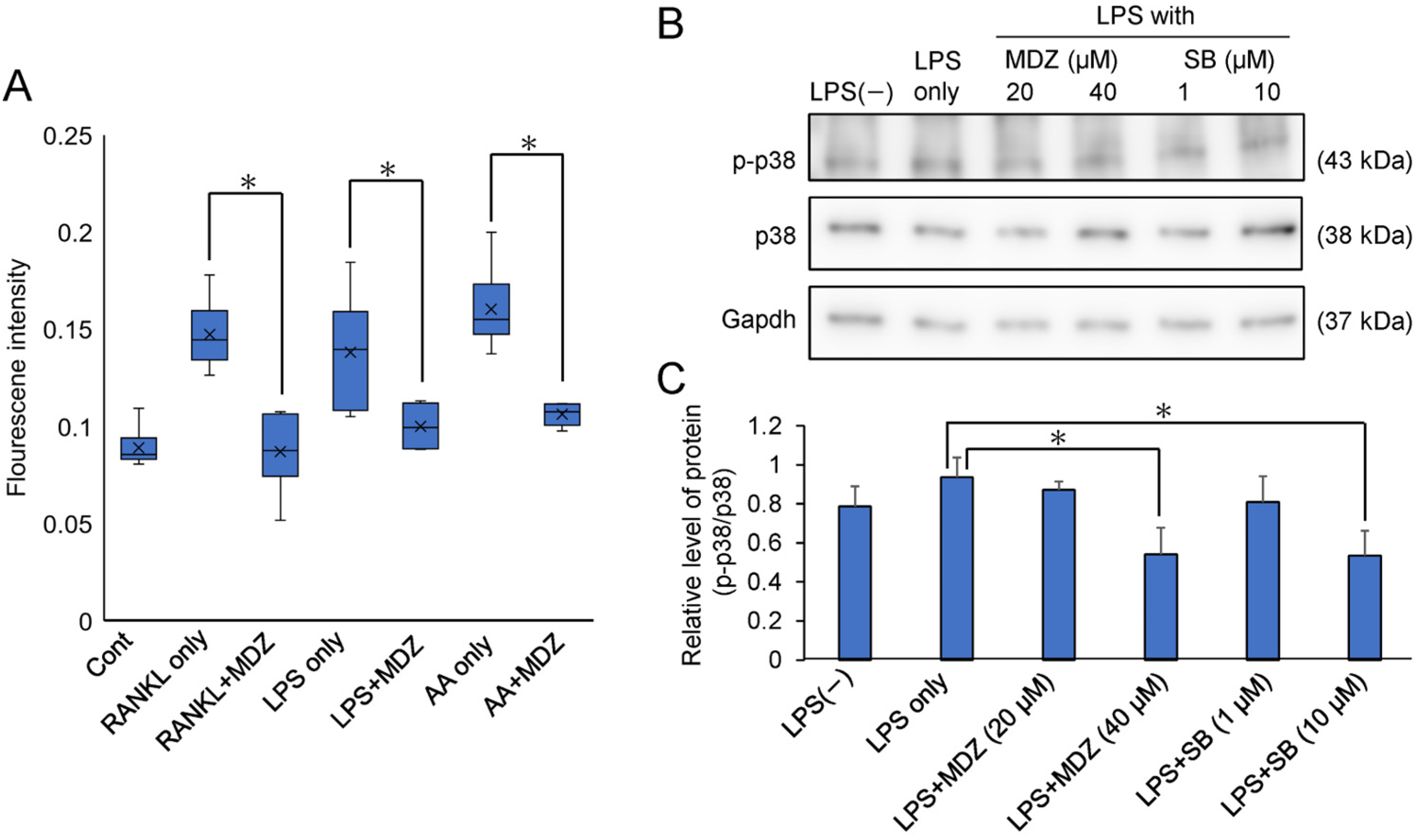
2.5. Elucidation of the Mechanism of MDZ-Induced Inhibition of Osteoclast Differentiation
3. Discussion
4. Materials and Methods
4.1. Biochemical and Cell Biology Experiments
4.1.1. Preparation of Mouse Macrophage-like Cell Line (RAW264 Cells)
4.1.2. TRAP Assay
4.1.3. TRAP Staining of RAW264 Cells
4.1.4. Pit Formation Assay
4.1.5. Quantitative Polymerase Chain Reaction Analysis
4.2. Animal Experiments
4.2.1. Establishment of Lipopolysaccharide-Induced Calvarial Model Mouse and TRAP Staining
4.2.2. Micro-Computed Tomography (Micro-CT) and Quantitative Analysis of Suture
4.3. Measurement of Reactive Oxygen Species
4.4. Western Blot Analysis
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EC50 | Half-maximal (50%) effective concentration |
| PBS | Phosphate-buffered saline |
| mRNA | Messenger ribonucleic acid |
| qPCR | Quantitative polymerase chain reaction |
| WHO | World Health Organization |
| NADPH | Nicotinamide adenine dinucleotide phosphate reduced form |
| Na2HPO4 | Disodium hydrogen phosphate |
| RIPA | Radioimmunoprecipitation assay |
| HRP | Horseradish peroxidase |
| ECL | Enhanced chemiluminescence |
References
- Hadjidakis, D.J.; Androulakis, I.I. Bone Remodeling. Ann. N. Y. Acad. Sci. 2006, 1092, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Omi, M.; Mishina, Y. Roles of osteoclasts in alveolar bone remodeling. Genesis 2022, 60, e23490. [Google Scholar] [CrossRef] [PubMed]
- Brochado Martins, J.F.; Rodrigues, C.F.D.; Diogo, P.; Paulo, S.; Palma, P.J.; do Vale, F.F. Remodelling compartment in root cementum. Folia Morphol. 2021, 80, 972–979. [Google Scholar] [CrossRef] [PubMed]
- Davide, G.; Nicolò, S.; Diana, T.; Fabiola, R.; Sara, B.; Serena, B.; Maurizio, P.; Giuseppe, V. Regenerative Potential of Platelet—Rich Fibrin in Maxillary Sinus Floor Lift Techniques: A Systematic Review. J. Biol. Regul. Homeost. Agents 2023, 37, 2357–2369. [Google Scholar] [CrossRef]
- Michaud, D.S.; Fu, Z.; Shi, J.; Chung, M. Periodontal Disease, Tooth Loss, and Cancer Risk. Epidemiol. Rev. 2017, 39, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Chambrone, L.; Wang, H.L.; Romanos, G.E. Antimicrobial photodynamic therapy for the treatment of periodontitis and peri-implantitis: An American Academy of Periodontology best evidence review. J. Periodontol. 2018, 89, 783–803. [Google Scholar]
- Shirakata, Y.; Nakamura, T.; Setoguchi, F.; Imafuji, T.; Shinohara, Y.; Matsumura, S.; Iwata, M.; Noguchi, K.; Ramanauskaite, E.; Sculean, A. Histological evaluation of nonsurgical periodontal treatment with and without the use of sodium hypochlorite/amino acids and cross-linked hyaluronic acid gels in dogs. Clin. Oral Investig. 2024, 28, 281. [Google Scholar] [CrossRef] [PubMed]
- Hammarström, L. The role of enamel matrix proteins in the development of cementum and periodontal tissues. Ciba Found. Symp. 1997, 205, 246–255; discussion 255–260. [Google Scholar] [CrossRef] [PubMed]
- Gestrelius, S.; Lyngstadaas, S.P.; Hammarstrom, L. Emdogain-periodontal regeneration based on biomimicry. Clin. Oral Investig. 2000, 4, 120–125. [Google Scholar] [CrossRef]
- Kitamura, M.; Yamashita, M.; Miki, K.; Ikegami, K.; Takedachi, M.; Kashiwagi, Y.; Nozaki, T.; Yamanaka, K.; Masuda, H.; Ishihara, Y.; et al. An exploratory clinical trial to evaluate the safety and efficacy of combination therapy of REGROTH® and Cytrans® granules for severe periodontitis with intrabony defects. Regen. Ther. 2022, 21, 104–113. [Google Scholar] [CrossRef]
- Keil, C.; Gollmer, B.; Zeidler-Rentzsch, I.; Gredes, T.; Heinemann, F. Histological evaluation of extraction sites grafted with Bio-Oss Collagen: Randomized controlled trial. Ann. Anat. 2021, 237, 151722. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.; Schou, S.; Stavropoulos, A.; Terheyden, H.; Holmstrup, P. Maxillary sinus floor augmentation with Bio-Oss or Bio-Oss mixed with autogenous bone as graft: A systematic review. Clin. Oral. Implant. Res. 2012, 23, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Oortgiesen, D.A.; Plachokova, A.S.; Geenen, C.; Meijer, G.J.; Walboomers, X.F.; van den Beucken, J.J.; Jansen, J.A. Alkaline phosphatase immobilization onto Bio-Gide® and Bio-Oss® for periodontal and bone regeneration. J. Clin. Periodontol. 2012, 39, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, G.; Mao, J.; Yang, G.; Wang, H. Influence of different incision designs on bone increment of guided bone regeneration (Bio-Gide collagen membrane + Bio-OSS bone powder) during the same period of maxillary anterior tooth implantation. Bioengineered 2021, 12, 2155–2163. [Google Scholar] [CrossRef] [PubMed]
- Olkkola, K.T.; Ahonen, J. Midazolam and Other Benzodiazepines. In Modern Anesthetics. Handbook of Experimental Pharmacology; Schüttler, J., Schwilden, H., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; Volume 182, pp. 335–360. [Google Scholar]
- Karakida, T.; Onuma, K.; Saito, M.M.; Yamamoto, R.; Chiba, T.; Chiba, R.; Hidaka, Y.; Fujii-Abe, K.; Kawahara, H.; Yamakoshi, Y. Potential for Drug Repositioning of Midazolam for Dentin Regeneration. Int. J. Mol. Sci. 2019, 20, 670. [Google Scholar] [CrossRef]
- Hidaka, Y.; Chiba-Ohkuma, R.; Karakida, T.; Onuma, K.; Yamamoto, R.; Fujii-Abe, K.; Saito, M.M.; Yamakoshi, Y.; Kawahara, H. Combined Effect of Midazolam and Bone Morphogenetic Protein-2 for Differentiation Induction from C2C12 Myoblast Cells to Osteoblasts. Pharmaceutics 2020, 12, 218. [Google Scholar] [CrossRef] [PubMed]
- Miyawaki, T.; Sogawa, N.; Maeda, S.; Kohjitani, A.; Shimada, M. Effect of Midazolam on Interleukin-6 Mrna Expression in Human Peripheral Blood Mononuclear Cells in the Absence of Lipopolysaccharide. Cytokine 2001, 15, 320–327. [Google Scholar] [CrossRef]
- Park, W.H.; Han, Y.W.; Kim, S.H.; Kim, S.Z. An ROS generator, antimycin A, inhibits the growth of HeLa cells via apoptosis. J. Cell. Biochem. 2007, 102, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Cuetara, B.L.; Crotti, T.N.; O’Donoghue, A.J.; McHugh, K.P. Cloning and characterization of osteoclast precursors from the raw264.7 cell line. Vitr. Cell. Dev. Biol. Anim. 2006, 42, 182–188. [Google Scholar] [CrossRef]
- Kong, L.; Smith, W.; Hao, D. Overview of RAW264.7 for osteoclastogensis study: Phenotype and stimuli. J. Cell Mol. Med. 2019, 23, 3077–3087. [Google Scholar] [CrossRef]
- Matsumoto, M.; Sudo, T.; Saito, T.; Osada, H.; Tsujimoto, M. Involvement of p38 Mitogen-activated Protein Kinase Signaling Pathway in Osteoclastogenesis Mediated by Receptor Activator of NF-κ B Ligand (RANKL). J. Biol. Chem. 2000, 275, 31155–31161. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Tao, L.; Zhu, F.; Wang, Z.; Zhuang, Q.; Li, Y.; Yang, Y.; Feng, C.; Shi, H.; Shi, J.; et al. BushenHuoxue decoction suppresses M1 macrophage polarization and prevents LPS induced inflammatory bone loss by activating AMPK pathway. Heliyon 2023, 9, e15583. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Yang, D.; Okamura, H.; Teramachi, J.; Ochiai, K.; Qiu, L.; Haneji, T. Calcium Hydroxide Suppresses Porphyromonas endodontalis Lipopolysaccharide-induced Bone Destruction. J. Dent. Res. 2014, 93, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Fu, X.; Liu, Y.; Ji, Y.; Shang, Z. Sulforaphane Inhibits Osteoclastogenesis via Suppression of the Autophagic Pathway. Molecules 2021, 26, 347. [Google Scholar] [CrossRef] [PubMed]
- Raka, R.N.; Zhiqian, D.; Yue, Y.; Luchang, Q.; Suyeon, P.; Junsong, X.; Hua, W. Pingyin rose essential oil alleviates LPS-Induced inflammation in RAW 264.7 cells via the NF-κB pathway: An integrated in vitro and network pharmacology analysis. BMC Complement. Med. Ther. 2022, 22, 272. [Google Scholar] [CrossRef] [PubMed]
- Vermot, A.; Petit-Härtlein, I.; Smith, S.M.E.; Fieschi, F. NADPH Oxidases (NOX): An Overview from Discovery, Molecular Mechanisms to Physiology and Pathology. Antioxidants 2021, 10, 890. [Google Scholar] [CrossRef] [PubMed]
- Ambasta, R.K.; Kumar, P.; Griendling, K.K.; Schmidt, H.H.; Busse, R.; Brandes, R.P. Direct Interaction of the Novel Nox Proteins with p22phox Is Required for the Formation of a Functionally Active NADPH Oxidase. J. Biol. Chem. 2004, 279, 45935–45941. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, J.; Nakagawa, K.; Yamasaki, T.; Nakamura, K.; Takeya, R.; Kuribayashi, F.; Imajoh-Ohmi, S.; Igarashi, K.; Shibata, Y.; Sueishi, K.; et al. The superoxide-producing NAD(P)H oxidase Nox4 in the nucleus of human vascular endothelial cells. Genes Cells 2005, 10, 1139–1151. [Google Scholar] [CrossRef]
- Ago, T.; Kuroda, J.; Pain, J.; Fu, C.; Li, H.; Sadoshima, J. Upregulation of Nox4 by Hypertrophic Stimuli Promotes Apoptosis and Mitochondrial Dysfunction in Cardiac Myocytes. Circ. Res. 2010, 106, 1253–1264. [Google Scholar] [CrossRef]
- Zhao, H.; Zhang, M.; Zhou, F.; Cao, W.; Bi, L.; Xie, Y.; Yang, Q.; Wang, S. Cinnamaldehyde ameliorates LPS-induced cardiac dysfunction via TLR4-NOX4 pathway: The regulation of autophagy and ROS production. J. Mol. Cell. Cardiol. 2016, 101, 11–24. [Google Scholar] [CrossRef]
- Liang, P.; Wang, L.; Yang, S.; Pan, X.; Li, J.; Zhang, Y.; Liang, Y.; Li, J.; Zhou, B. 5-Methoxyflavone alleviates LPS-mediated lung injury by promoting Nrf2-mediated the suppression of NOX4/TLR4 axis in bronchial epithelial cells and M1 polarization in macrophages. J. Inflamm. 2022, 19, 24. [Google Scholar] [CrossRef]
- Kanzaki, H.; Shinohara, F.; Kanako, I.; Yamaguchi, Y.; Fukaya, S.; Miyamoto, Y.; Wada, S.; Nakamura, Y. Molecular regulatory mechanisms of osteoclastogenesis through cytoprotective enzymes. Redox Biol. 2016, 8, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Kanzaki, H.; Shinohara, F.; Itohiya, K.; Yamaguchi, Y.; Katsumata, Y.; Matsuzawa, M.; Fukaya, S.; Miyamoto, Y.; Wada, S.; Nakamura, Y. RANKL induces Bach1 nuclear import and attenuates Nrf2-mediated antioxidant enzymes, thereby augmenting intracellular reactive oxygen species signaling and osteoclastogenesis in mice. FASEB J. 2017, 31, 781–792. [Google Scholar] [CrossRef] [PubMed]
- Gavish, M.; Bachman, I.; Shoukrun, R.; Katz, Y.; Veenman, L.; Weisinger, G.; Weizman, A. Enigma of the Peripheral Benzodiazepine Receptor. Pharmacol. Rev. 1999, 51, 629–650. [Google Scholar]
- Feng, H.; Liu, Y.; Zhang, R.; Liang, Y.; Sun, L.; Lan, N.; Ma, B. TSPO Ligands PK11195 and Midazolam Reduce NLRP3 Inflammasome Activation and Proinflammatory Cytokine Release in BV-2 Cells. Front. Cell Neurosci. 2020, 14, 544431. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Sudo, T.; Maruyama, M.; Osada, H.; Tsujimoto, M. Activation of p38 mitogen-activated protein kinase is crucial in osteoclastogenesis induced by tumor necrosis factor. FEBS Lett. 2000, 486, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, R.; Hayakawa, T.; Takeda, K.; Ichijo, H. Therapeutic targets in the ASK1-dependent stress signaling pathways. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2012, 88, 434–453. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Xu, R.; Feng, L.; Guo, W.; Cao, N.; Qian, C.; Teng, P.; Wang, L.; Wu, X.; Sun, Y.; et al. A Novel Chromone Derivative with Anti-Inflammatory Property via Inhibition of ROS-Dependent Activation of TRAF6-ASK1-p38 Pathway. PLoS ONE 2012, 7, e37168. [Google Scholar] [CrossRef]
- Miyazaki, T.; Miyauchi, S.; Tawada, A.; Anada, T.; Suzuki, O. Effect of chondroitin sulfate-E on the osteoclastic differentiation of RAW264 cells. Dent. Mater. J. 2010, 29, 403–410. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harigaya, H.; Chiba-Ohkuma, R.; Karakida, T.; Yamamoto, R.; Fujii-Abe, K.; Kawahara, H.; Yamakoshi, Y. Potential for Drug Repositioning of Midazolam as an Inhibitor of Inflammatory Bone Resorption. Int. J. Mol. Sci. 2024, 25, 7651. https://doi.org/10.3390/ijms25147651
Harigaya H, Chiba-Ohkuma R, Karakida T, Yamamoto R, Fujii-Abe K, Kawahara H, Yamakoshi Y. Potential for Drug Repositioning of Midazolam as an Inhibitor of Inflammatory Bone Resorption. International Journal of Molecular Sciences. 2024; 25(14):7651. https://doi.org/10.3390/ijms25147651
Chicago/Turabian StyleHarigaya, Hiroko, Risako Chiba-Ohkuma, Takeo Karakida, Ryuji Yamamoto, Keiko Fujii-Abe, Hiroshi Kawahara, and Yasuo Yamakoshi. 2024. "Potential for Drug Repositioning of Midazolam as an Inhibitor of Inflammatory Bone Resorption" International Journal of Molecular Sciences 25, no. 14: 7651. https://doi.org/10.3390/ijms25147651






