Comparative Proteomic Analysis of Floral Buds before and after Opening in Walnut (Juglans regia L.)
Abstract
1. Introduction
2. Results
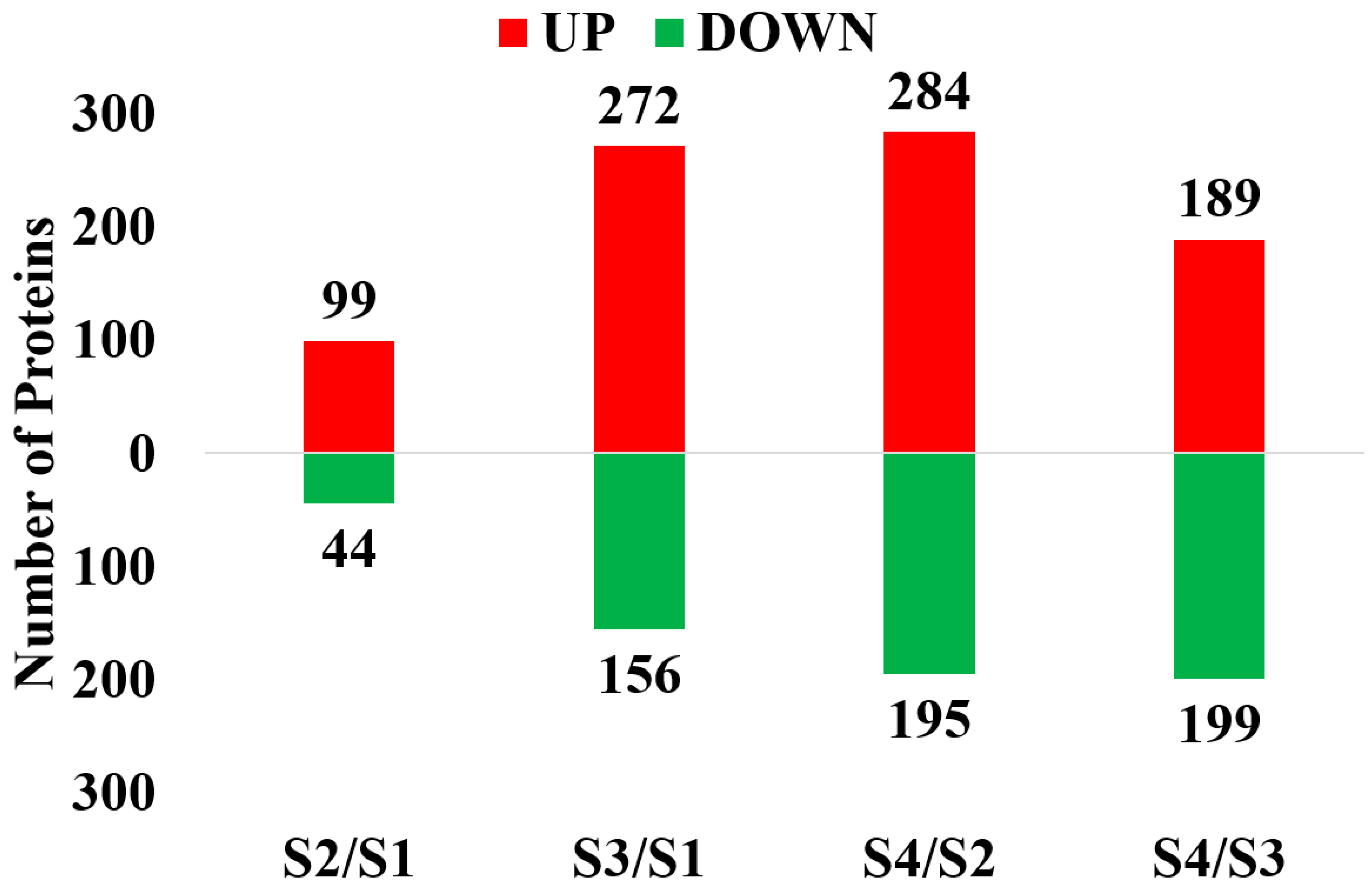
2.1. Proteomic Identification of Floral Buds before and after Opening in Walnut
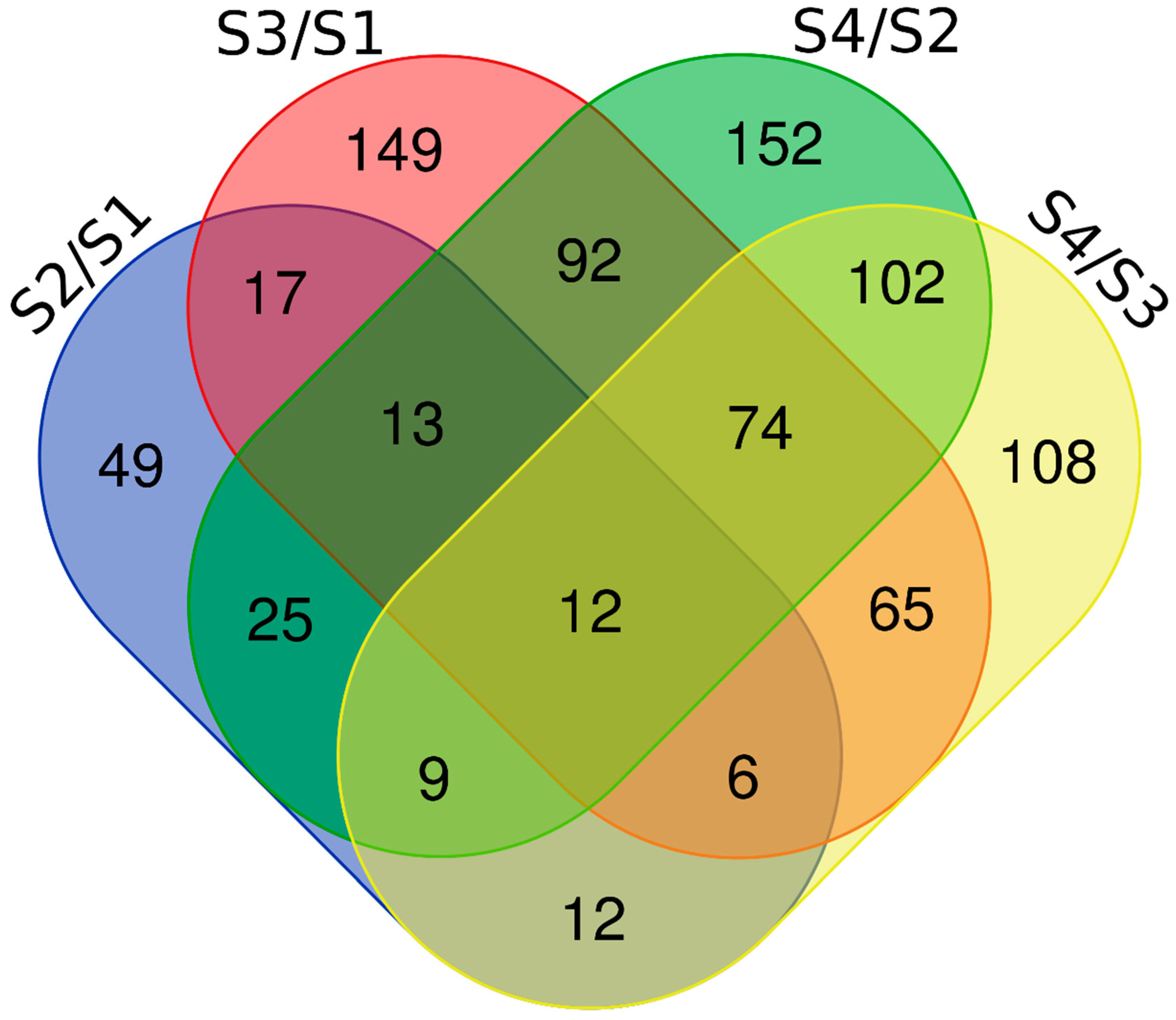
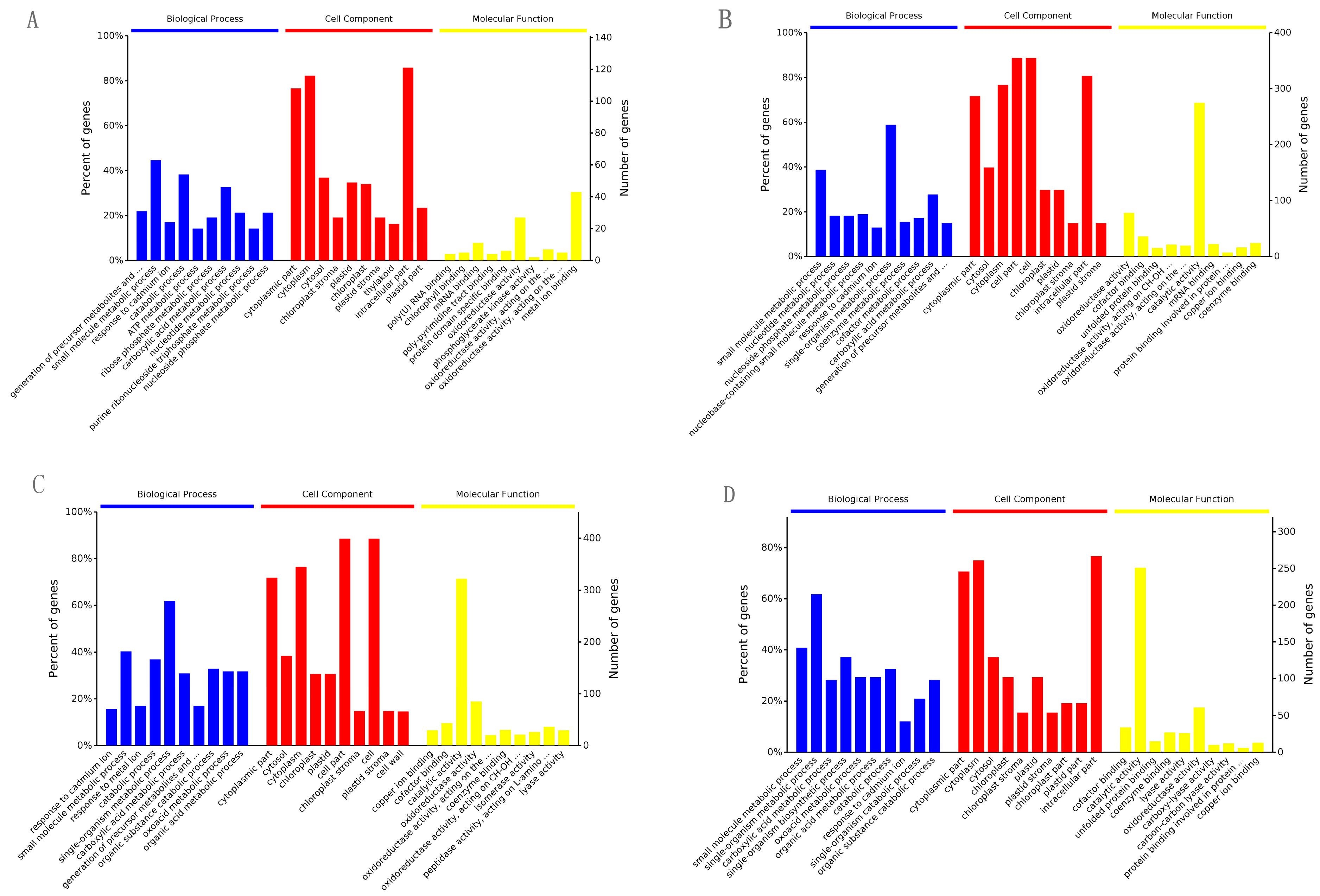
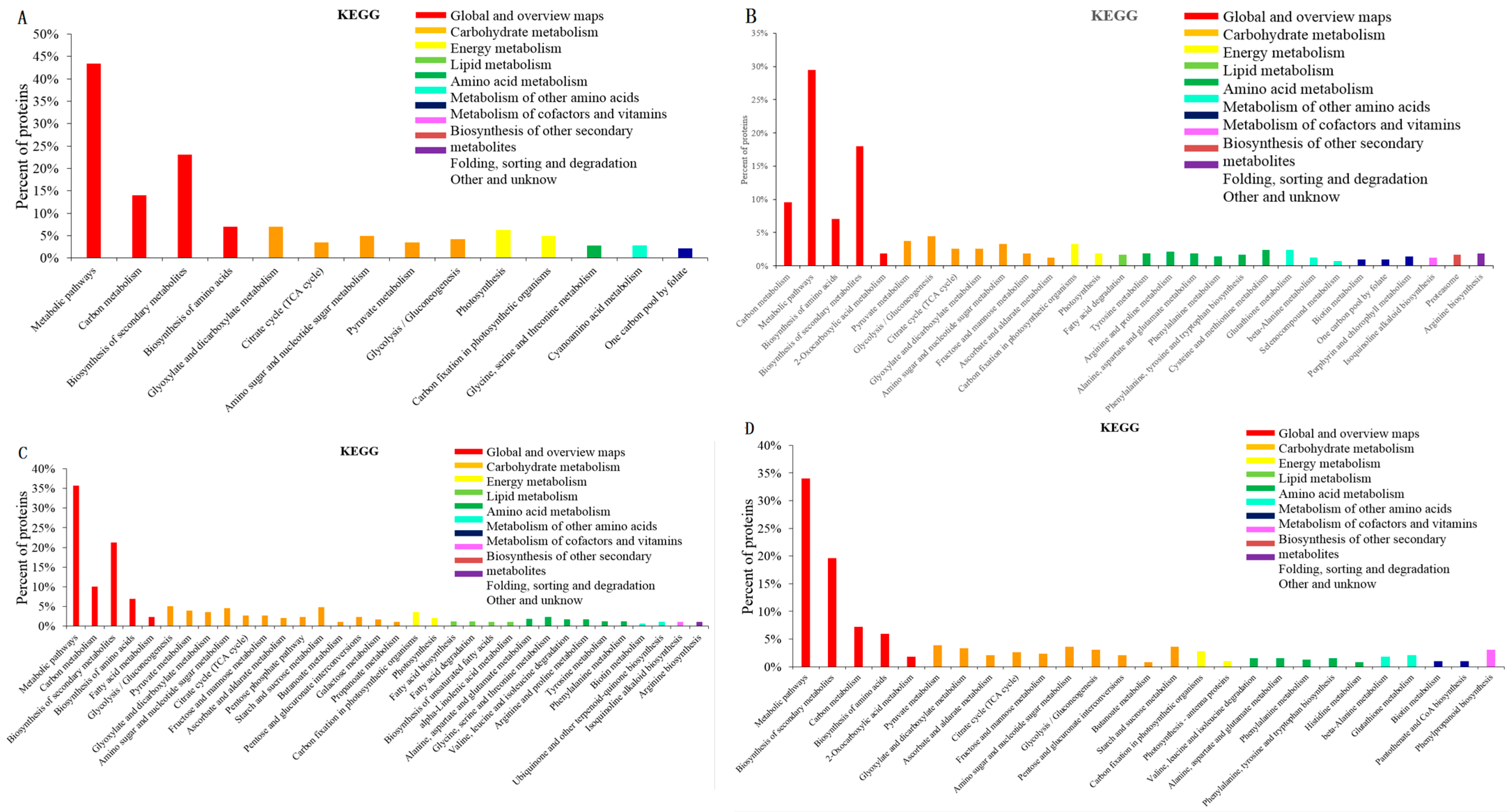
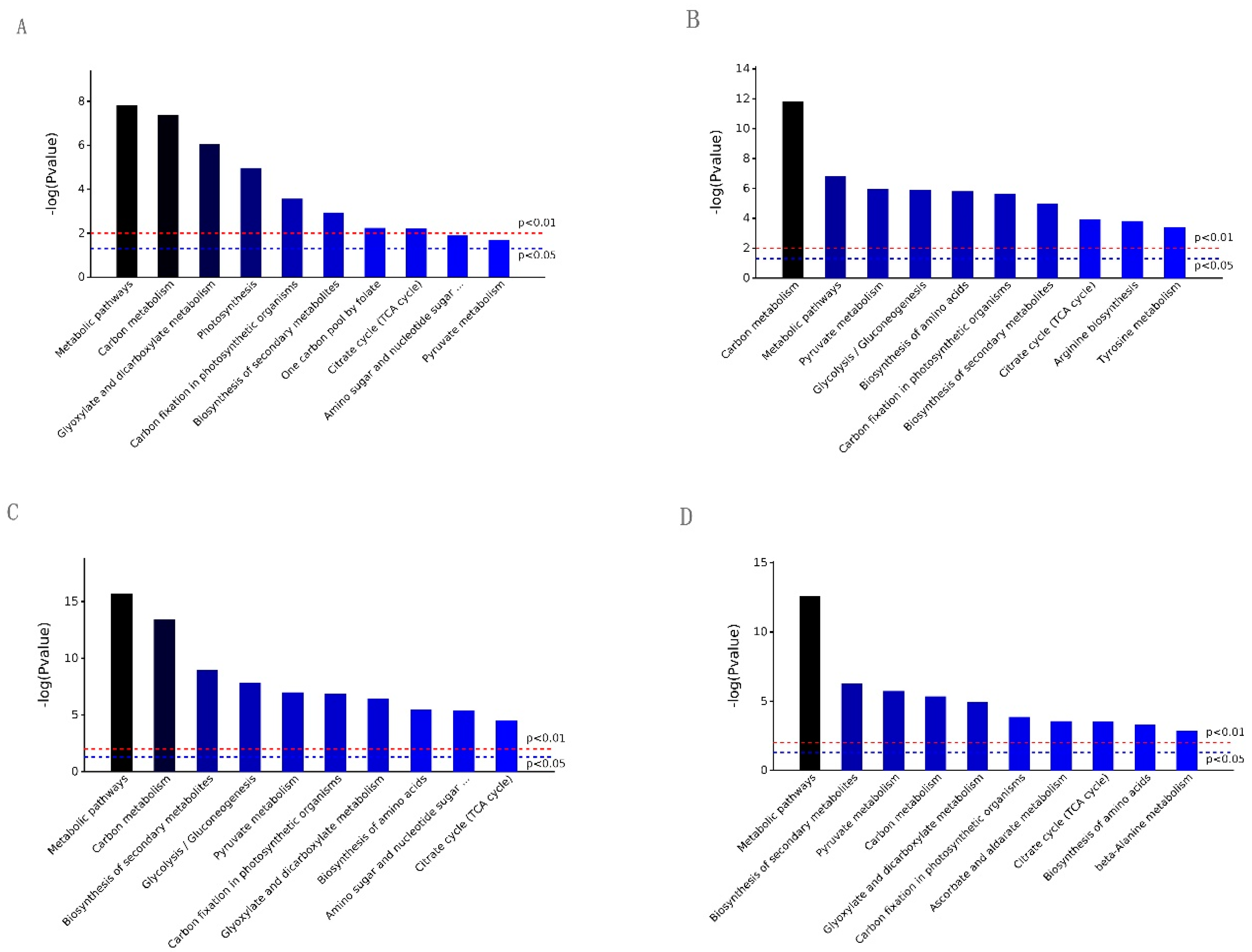
2.2. GO and KEGG Analysis of the Differential Expression Proteins in the Four Contrasts
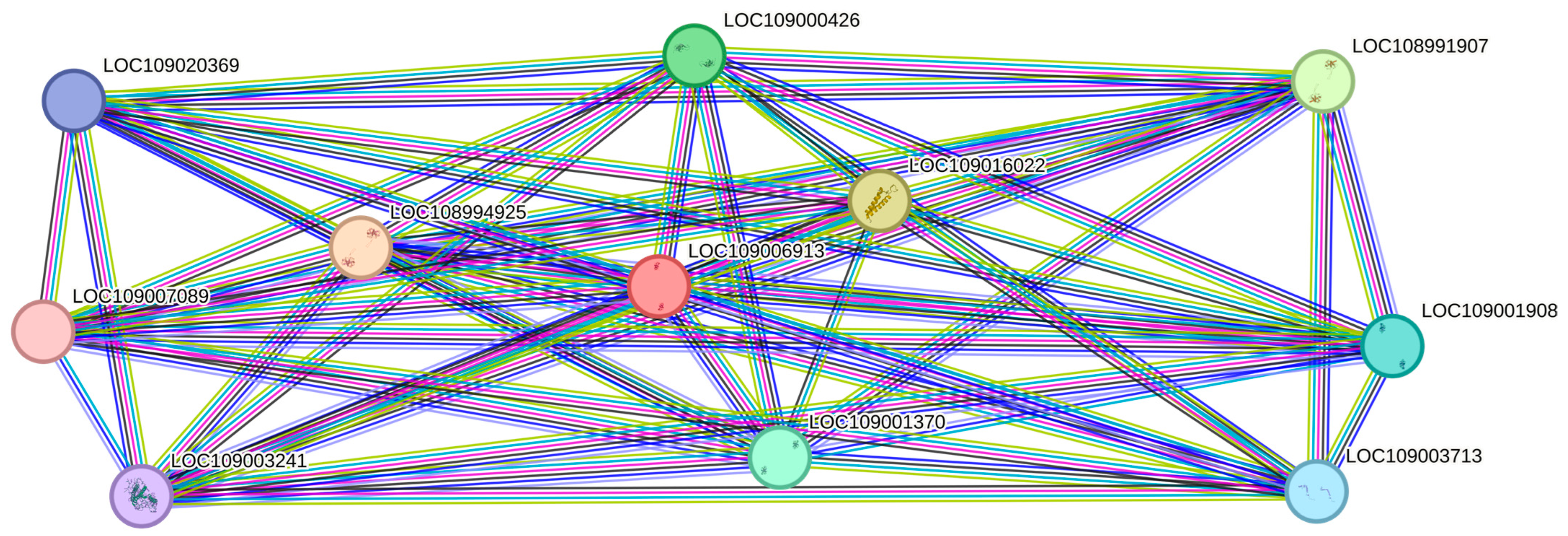
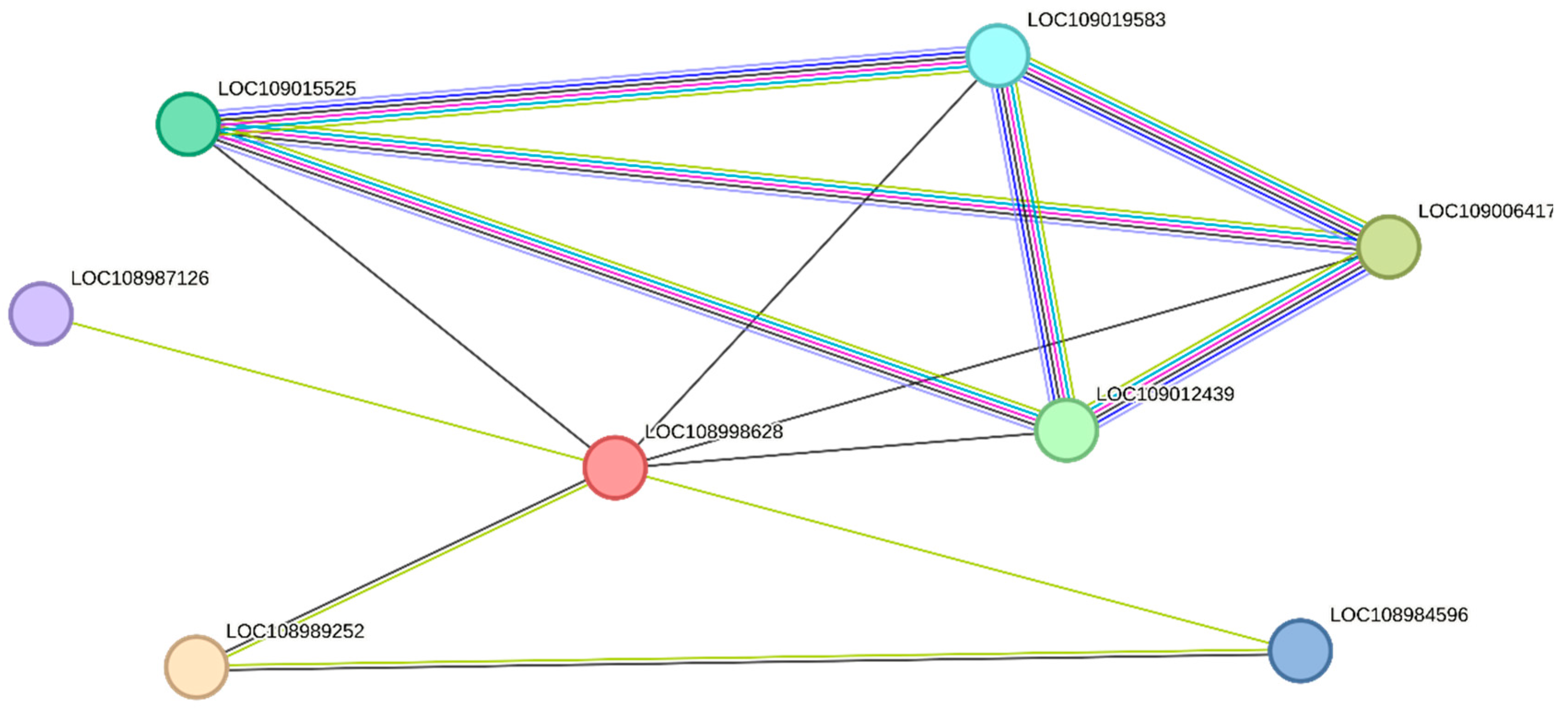
2.3. Protein–Protein Interaction (PPI)
3. Discussion
3.1. Changes in Floral Bud Characteristics before and after Opening in Walnut
3.2. Common DEPs Play Promotional Roles in All Four Contrasts
3.3. Protein Species Expressed Specifically Related to Photosynthesis in Floral Bud Development
3.4. Protein Species Expressed Specifically Related to Morphogenesis in Floral Bud Development
3.5. Protein Species Expressed Specifically Related to Metabolism in Floral Bud Development
3.6. Protein Species Expressed Specifically Related to Stress Response in Floral Buds’ Development
4. Materials and Methods
4.1. Plant Materials
4.2. Protein Extraction and Quantization
4.3. Protein Hydrolysis and iTRAQ Labeling
4.4. HPLC Separation of Peptide Mixture
4.5. LC-MS/MS Analysis
4.6. Data Analysis and Reliable Protein Screening
4.7. Bioinformatics Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gleeson, S.K. Heterodichogamy in walnuts: Inheritance and stable ratios. Evolution 1982, 36, 892–902. [Google Scholar] [CrossRef] [PubMed]
- Robert, I.B.; Christian, M.N. Dichogamy in angiosperms. Bot. Rev. 1993, 59, 112–152. [Google Scholar]
- Sargent, R.D.; Mandegar, M.A.; Otto, S.P. A model of the evolution of dichogamy incorporating sex-ratio selection, anther-stigma interference, and inbreeding depression. Evolution 2006, 60, 934–944. [Google Scholar] [PubMed]
- Dellaporta, S.L.; Calderon-Urrea, A. Sex Determination in Flowering Plants. Plant Cell 1993, 5, 1241–1251. [Google Scholar] [PubMed]
- Charlesworth, D. Plant sex chromosomes. Annu. Rev. Plant Biol. 2016, 67, 397–420. [Google Scholar] [CrossRef] [PubMed]
- Scott, R.; Pereira, A. The Arabidopsis MALE STERILITY 2 protein shares similarity with reductases in elongation/condensation complexes. Plant J. 1997, 12, 615–623. [Google Scholar]
- Kubo, K.-I.; Entani, T.; Takara, A.; Wang, N.; Fields, A.M.; Hua, Z.; Toyoda, M.; Kawashima, S.-I.; Ando, T.; Isogai, A.; et al. Collaborative non-self recognition system in S-RNase-based self-incompatibility. Science 2010, 330, 796–799. [Google Scholar] [CrossRef]
- Pollegioni, P.; Olimpieri, I.; Woeste, K.E.; Simoni, G.D.; Gras, M.; Malvolti, M.E. Barriers to interspecific hybridization between Juglans nigra L. and J. regia L species. Tree Genet. Genomes 2013, 9, 291–305. [Google Scholar] [CrossRef]
- Janković, S.; Stanković, J.; Janković, D.; Milatović, D. Morphology and morphogenesis of female reproductive organs in some walnut (Juglans regia L.) genotypes. Sci. Hortic. 2021, 289, 110471. [Google Scholar] [CrossRef]
- Fu, X.X.; Feng, L.; Shang, X.L.; Yang, W.X.; Fang, S.Z. Observation of morphological and anatomical characters on staminate and pistillate flower differentiation in Cyclocarya paliurus. J. Nanjing For. Univ. (Nat. Sci. Ed.) 2011, 35, 17–22. [Google Scholar]
- Hansche, P.E.; Beres, V.; Forde, H.I. Estimates of quantitative generic properties of walnut and their implications for cultivar improvement. J. Am. Soc. Hortic. Sci. 1972, 97, 279–285. [Google Scholar] [CrossRef]
- Zhang, L.J.; Guo, C.; Qin, B.T.; Lu, X.J.; Yang, C.Y.; Qi, Y.H.; Shen, H.L. Phonological characteristics of flowering and pollen viability of Juglans mandshurica. J. Northeast For. Univ. 2019, 47, 4–8. [Google Scholar]
- Tanurdzic, M.; Banks, J.A. Sex-Determining Mechanisms in Land Plants. Plant Cell 2004, 16, S61–S71. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.M.; Jia, H.J.; Cai, Q.L.; Wang, Y.; Zhao, H.B.; Wang, G.J.; Li, Y.H.; Zhan, D.L.; Shen, Y.T.; Niu, Q.F.; et al. The red bayberry genome and genetic basis of sex determination. Plant Biotechnol. J. 2019, 17, 397–409. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.S.; Shen, D.; Zhang, W.; Zhang, X.H.; Qiu, Y.; Wang, H.P.; Dou, X.X.; Li, S.G.; Wu, Y.Q.; Song, J.P.; et al. Temperature and photoperiod changes affect cucumber sex expression by different epigenetic regulations. BMC Plant Biol. 2018, 18, 268. [Google Scholar] [CrossRef]
- Zhou, Y.; Ahammed, G.J.; Wang, Q.; Wu, C.Q.; Wan, C.P.; Yang, Y.X. Transcriptomic insights into the blue light-induced female floral sex expression in cucumber (Cucumis sativus L.). Sci. Rep. 2018, 8, 14261. [Google Scholar] [CrossRef] [PubMed]
- Ming, R.; Bendahmane, A.; Renner, S.S. Sex chromosomes in land plants. Annu. Rev. Plant Biol. 2011, 62, 485–514. [Google Scholar] [CrossRef] [PubMed]
- Akagi, T.; Kajita, K.; Kibe, T.; Morimura, H.; Tsujimoto, T.; Nishiyama, S.; Kawai, T.; Yamane, H.; Tao, R. Development of molecular markers associated with sexuality in Diospyros lotus L. and their application in D. kaki Thunb. J. Jpn. Soc. Hortic. Sci. 2013, 83, 214–221. [Google Scholar] [CrossRef]
- Aryal, R.; Ming, R. Sex determination in flowering plants: Papaya as a model system. Plant Sci. 2014, 217–218, 56–62. [Google Scholar] [CrossRef]
- Antcliff, A.J. Inheritance of sex in Vitis. Ann Amelior Plantes 1980, 30, 113–122. [Google Scholar]
- Akagi, T.; Henry, I.M.; Tao, R.; Comai, L. A Y-chromosomeencoded small RNA acts as a sex determinant in persimmons. Science 2014, 346, 646–650. [Google Scholar] [CrossRef] [PubMed]
- Akagi, T.; Henry, I.M.; Ohtani, H.; Morimoto, T.; Beppu, K.; Kataoka, I.; Tao, R. A Y-encoded suppressor of feminization arose via lineage-specific duplication of a cytokinin response regulator in kiwifruit. Plant Cell 2018, 30, 780–795. [Google Scholar] [CrossRef] [PubMed]
- Akagi, T.; Pilkington, S.M.; Varkonyi-Gasic, E.; Firl, A.; McNeilage, M.A.; Douglas, M.J.; Wang, T.; Rebstock, R.; Voogd, C.; Datson, P.; et al. Two Y-chromosome-encoded genes determine sex in kiwifruit. Nat. Plants 2019, 5, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Urasaki, N.; Tarora, K.; Shudo, A.; Ueno, H.; Tamaki, M.; Miyagi, N.; Adaniya, S.; Matsumura, H. Digital transcriptome analysis of putative sex-determination genes in papaya (Carica papaya). PLoS ONE 2012, 7, e40904. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y.; Lin, H.J.; Viswanath, K.K.; Lin, C.P.; Chang, B.C.H.; Chiu, P.H.; Chiu, C.T.; Wang, R.H.; Chin, S.W.; Chen, F.C. The development of functional mapping by three sex-related loci on the third whorl of different sex types of Carica papaya L. PLoS ONE 2018, 13, e0194605. [Google Scholar]
- Moghaddam, M.R.B.; Van den End, W. Sugars, the clock and transition to flowering. Front. Plant Sci. 2013, 4, 22. [Google Scholar] [CrossRef]
- Weigel, D.; Alvarez, J.; Smyth, D.R. LEAFY controls floral meristem identity in Arabidopsis. Cell 1992, 69, 843–859. [Google Scholar] [CrossRef]
- Mandel, M.A.; Gustafson-Brown, C.; Savidge, B.; Yanofsky, M.F. Molecular characterization of the Arabidopsis floral homeotic gene APETALA1. Nature 1992, 360, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Koornneef, M.; Blankestijn-de Vries, H.; Hanhart, C.J.; Soppe, W.; Peeters, T. The phenotype of some late-flowering mutants is enhanced by a locus on chromosome 5 that is not effective in the Landsberg erecta wild-type. Plant J. 1994, 6, 911–919. [Google Scholar] [CrossRef]
- Gu, Q.; Ferrándiz, C.; Yanofsky, M.F.; Martienssen, R. The FRUITFULL MADS-box gene mediates cell differentiation during Arabidopsis fruit development. Development 1998, 125, 1509–1517. [Google Scholar] [CrossRef]
- Kardailsky, I.; Shukla, V.K.; Ahn, J.H.; Dagenais, N.; Christensen, S.K.; Nguyen, J.T.; Chory, J.; Harrison, M.J.; Weigel, D. Activation tagging of the floral inducer FT. Science 1999, 286, 1962–1965. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, C.C.; Burn, J.E.; Perez, P.P.; Metzger, J.; Dennis, E.S. The FLF MADS box gene: A repressor of flowering in Arabidopsis regulated by vernalization and methylation. Plant Cell 1999, 11, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Samach, A.; Onouchi, H.; Gold, S.E. Distinct roles of CONSTANS target genes in reproductive development of Arabidopsis. Science 2000, 288, 1613–1616. [Google Scholar] [CrossRef] [PubMed]
- Teper-Bamnolker, P.; Samach, A. The flowering integrator FT regulates SEPALLATA3 and FRUITFULL accumulation in Arabidopsis leaves. Plant Cell 2005, 17, 2661–2675. [Google Scholar] [CrossRef]
- Yan, F.; Zhou, H.J.; Yue, M.; Yang, G.; Zhao, P. Genome-Wide Identification and Transcriptional Expression Profiles of the F-box Gene Family in Common Walnut (Juglans regia L.). Forests 2019, 10, 275. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, L.N.; Xia, X.C.; Jing, D.; Li, H.X.; Luo, X.; Cao, S.Y. Cloning and expression analysis of walnut FRUITFULL (JrFUL) gene. J. Fruit Sci. 2019, 36, 992–1004. [Google Scholar]
- He, F.Q.; Wang, H.X.; Zhang, Z.H. Molecular cloning and sequence analysis of an LFY homologous gene from Juglans regia L. Front. Agric. China 2011, 5, 366–371. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, L.N.; Xia, X.C.; Jing, D.; Li, H.X.; Luo, X.; Cao, S.Y. Cloning and expression analysis of FLOWERING LOCUS T (FT) gene in walnut (Juglans regia). Acta Agric. Jiangxi 2019, 31, 13–20. [Google Scholar]
- Davis, S.J. Integrating hormones into the floral-transition pathway of Arabidopsis thaliana. Plant Cell Environ. 2009, 32, 1201–1210. [Google Scholar] [CrossRef]
- Noir, S.; Brautigam, A.; Colby, T.; Schmidt, J.; Panstruga, R. A reference map of the Arabidopsis thaliana mature pollen proteome. Biochem. Biophys. Res. Commun. 2005, 337, 1257–1266. [Google Scholar] [CrossRef]
- Yang, B.X.; Zhong, Z.H.; Wang, T.T.; Zhang, L. Integrative omics of Lonicera japonica Thunb. flower development unravels molecular changes regulating secondary metabolites. J. Proteom. 2019, 208, 103470–103482. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zeng, L.T.; Zhao, H.Y.; Ye, Q.S. Proteomic analysis of the early development of the Phalaenopsis amabilis flower bud under low temperature induction using the iTRAQ/MRM approach. Molecules 2020, 25, 1244. [Google Scholar] [CrossRef] [PubMed]
- Nazemof, N.; Couroux, P.; Xing, T.; Robert, L.S. Proteomic analysis of the mature Brassica stigma reveals proteins with diverse roles in vegetative and reproductive development. Plant Sci. 2016, 250, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Dafny-Yelin, M.; Guterman, I.; Menda, N.; Ovadis, M.; Shalit, M.; Pichersky, E.; Zamir, D.; Lewinsohn, E.; Adam, Z.; Weiss, D.; et al. Flower proteome: Changes in protein spectrum during the advanced stages of rose petal development. Planta 2005, 222, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Xiong, E.H.; Wu, X.L.; Shi, J.; Wang, X.Y.; Wang, W. Proteomic identification of differentially expressed proteins between male and female plants in Pistacia chinensis. PLoS ONE 2013, 8, e064276. [Google Scholar] [CrossRef] [PubMed]
- Gómez, J.F.; Talle, B.; Wilson, Z.A. Anther and pollen development: A conserved developmental pathway. J. Integr. Plant Biol. 2015, 57, 876–891. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.; Liu, B. Tapetum-dependent male meiosis progression in plants: Increasing evidence emerges. Front. Plant Sci. 2019, 10, 1667. [Google Scholar] [CrossRef]
- Shi, J.; Cui, M.; Li, Y.; Kim, Y.J.; Zhang, D. Genetic and biochemical mechanisms of pollen wall development. Trends Plant Sci. 2015, 20, 741–753. [Google Scholar] [CrossRef] [PubMed]
- Wilson, Z.A.; Zhang, D.B. From Arabidopsis to rice: Pathways in pollen development. J. Exp. Bot. 2009, 60, 1479–1492. [Google Scholar] [CrossRef]
- Devani, R.S.; Chirmade, T.; Sinha, S.; Bendahmane, A.; Dholakia, B.B.; Banerjee, A.K.; Banerjee, J. Flower bud proteome reveals modulation of sex-biased proteins potentially associated with sex expression and modification in dioecious Coccinia grandis. BMC Plant Biol. 2019, 19, 330–344. [Google Scholar] [CrossRef]
- Holmes-davis, R.; Tanaka, C.K.; Vensel, W.H.; Hurkman, W.J.; McCormick, S. Proteome mapping of mature pollen of Arabidopsis thaliana. Proteomics 2005, 5, 4864–4884. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.B.; Zhang, X.X.; Liu, G.R.; Gong, W.; Yang, Y.J.; Tian, Z.Y.; Yang, Z.W. Proteomic characteristics of male and female flower buds development in Zanthoxylum planispinum var. dintanensis. J. Sichuan Agric. Univ. 2022, 40, 758–774. [Google Scholar]
- Wu, Z.X.; Jiang, F.L.; Liu, M.; Xing, X.D.; Fu, C.N.; Wu, Z. The flower bud differentiation process of Allium sativum and its anatomical structure and morphological characteristics changes. J. Plant Resour. Environ. 2019, 28, 25–33. [Google Scholar]
- Zhao, Y.F.; Dai, J.L.; Gao, S.P.; Lei, T.; Jiang, Y.L.; Zhang, X.Z.; Zhu, Y.T. Flower bud differentiation process and its morphology and anatomical structure in twotypes of stigma of Plumbago auriculata. J. Nanjing For. Univ. (Nat. Sci. Ed.) 2018, 42, 203–208. [Google Scholar]
- Yu, D.; Zhao, Y.; Han, L.Q.; Guo, C.H.; Kang, C.; Ma, K.; Niu, J.X. Comparative study on flower bud differentiation of female and male early blooming type walnut varieties. J. Fruit Sci. 2022, 39, 1054–1062. [Google Scholar]
- Amsino, R. Seasonal and developmental timing of flowering. Plant J. 2010, 61, 1001–1003. [Google Scholar] [CrossRef]
- Burgarell, C.; Chantret, N.; Gay, L.; Prosperi, J.M.; Bonhomme, M.; Tiffin, P.; Young, N.D.; Ronfort, J. Adaptation to climate through flowering phenology: A case study in Medicago truncatula. Mol. Ecol. 2016, 25, 3397–3415. [Google Scholar] [CrossRef]
- Mei, Y.; Chen, L.M. Research progresses on the plant formate dehydrogenase. Biotechnol. Bull. 2010, 5, 23–33. [Google Scholar]
- Shin, Y.K.; Yum, H.; Kim, E.S.; Cho, H.; Gothandam, K.M.; Hyun, J.; Chung, Y.Y. BcXTH1, a brassica campestris homologue of Arabidopsis XTH9, is associated with cell expansion. Planta 2006, 224, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Kurasawa, K.; Matsui, A.; Yokoyama, R.; Kuriyama, T.; Yoshizumi, T.; Matsui, M.; Suwabe, K.; Watanabe, M.; Nishitani, K. The AtXTH28 gene, a xyloglucan endotransglucosylase/hydr-olase is involved in automatic self-pollination in Arabidopsis thaliana. Plant Cell Physiol. 2009, 50, 413–422. [Google Scholar] [CrossRef]
- Li, X.B.; Xu, W.W.; Chowdhury, M.R.; Jin, F. Comparative proteomic analysis of labellum and inner lateral petals in Cymbidium ensifolium flowers. Int. J. Mol. Sci. 2014, 15, 19877–19897. [Google Scholar] [CrossRef]
- Harrison, E.P.; Willingham, N.M.; Lloyd, J.C.; Raines, C.A. Reduced sedoheptulose-1,7-bisphosphatase levels in transgenic tobacco lead to decreased photosynthetic capacity and altered carbohydrate accumulation. Planta 1997, 204, 27–36. [Google Scholar] [CrossRef]
- Ding, F.; Wang, M.L.; Zhang, S.X.; Ai, X.Z. Changes in SBPase activity influence photosynthetic capacity, growth, and tolerance to chilling stress in transgenic tomato plants. Sci. Rep. 2016, 6, 32741. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.X.; Zhou, Y.Y.; Han, X.; An, Y.; Guo, H.H.; Xia, X.L.; Yin, W.L.; Liu, C. Overexpression of Populus SBPase gene promoting photosynthesis and vegetative growth in Arabidopsis thaliana. J. Beijing For. Univ. 2018, 40, 26–33. [Google Scholar]
- Maoni, C.; Genhai, H.; Jie, D.; Chen, Y.; Fu, Y.Z.; Zhang, J.B.; Wang, Q.L. Sequence Characteristics and Expression Analysis of the Gene Encoding Sedoheptulose-1,7-Bisphosphatase, an important Calvin cycle enzyme in upland cotton (Gossypium hirsutum L.). Int. J. Mol. Sci. 2023, 24, 6648. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yu, C.M.; Xiang, B.C.; Niu, J.X.; Zheng, Y.Y. Processing tomato chlorophyll a/b-binding protein 1C interacts with CMV 2b protein. Physiol. Mol. Plant Pathol. 2022, 120, 101857. [Google Scholar] [CrossRef]
- Zhang, J.; Xiao, J.W.; Li, Y.Q.; Su, B.D.; Xu, H.M.; Shan, X.Y.; Song, C.W.; Xie, J.B.; Li, R.L. PDM3, a pentatricopeptide repeat-containing protein, affects chloroplast development. J. Exp. Bot. 2017, 68, 5615–5627. [Google Scholar] [CrossRef] [PubMed]
- Mayfield, S.P.; Rahire, M.; Frank, G.; Zuber, H.; Rochaix, J.D. Expression of the nuclear gene encoding oxygen-evolving enhancer protein 2 is required for high levels of photosynthetic oxygen evolution in Chlamydomonas reinhardtii. Proc. Natl. Acad. Sci. USA 1987, 84, 749–753. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, L.; Shu, L.F.; Zhuang, X.S.; Liu, Y.M.; Chen, J.; Hu, Z.L. Sustainable photosynthetic H2-production mediated by artificial miRNA silencing of OEE2 gene in green alga Chlamydomonas reinhardtii. Int. J. Hydrog. Energy 2015, 16, 5609–5616. [Google Scholar] [CrossRef]
- Yang, E.J.; Oh, Y.A.; Lee, E.S.; Park, A.R.; Cho, S.K.; Yoo, Y.J.; Park, O.K. Oxygen-evolving enhancer protein 2 is phosphorylated by glycine-rich protein 3/wall-associated kinase 1 in Arabidopsis. Biochem. Biophys. Res. Commun. 2003, 305, 862–868. [Google Scholar] [CrossRef]
- Chen, H.M.; Chen, Y.; Wang, D.; Jiang, D.A. The role of ribulose-1,5-bisphosphate carboxylase/oxygenase activase in resistance of plant to abiotic stresses. Plant Physiol. J. 2016, 52, 1637–1648. [Google Scholar]
- Feiz, L.; Williams-Carrier, R.; Wostrikoff, K.; Belcher, S.; Barkan, A.; Stern, D.B. Ribulose-1,5-bis-phosphate carboxylase/oxygenase accumulation factor1 is required for holoenzyme assembly in maize. Plant Cell 2012, 24, 3435–3446. [Google Scholar] [CrossRef]
- Vaughn, K.C.; Lax, A.R.; Duke, S.O. Polyphenol oxidase: The chloroplast enzyme with no established function. Physiol. Plant. 1988, 72, 659–665. [Google Scholar] [CrossRef]
- van Dorssen, R.J.; Breton, J.; Plijter, J.J.; Satoh, K.; van Gorkom, H.J.; Amesz, J. Spectroscopic properties of the reaction center and of the 47 kDa chlorophyll protein of Photosystem II. Biochim. Biophys. Acta (BBA)—Bioenerg. 1987, 893, 267–274. [Google Scholar] [CrossRef]
- Wu, H.Y.; Shen, Y.; Hu, Y.L.; Tan, S.J.; Lin, Z.P. A phytocyanin-related early nodulin-like gene, BcBCP1, cloned from Boea crassifolia enhances osmotic tolerance in transgenic tobacco. J. Plant Physiol. 2011, 168, 935–943. [Google Scholar] [CrossRef]
- Gounaris, I.; Sherwood, R.T.; Gustine, D.L. Stamen-specific proteins of buffelgrass (Cenchrus ciliaris). J. Plant Physiol. 1991, 137, 437–440. [Google Scholar] [CrossRef]
- Torii, K.U.; Hanson, L.A.; Josefsson, C.A.B.; Shpak, E.D. Regulation of inflorescence architecture and organ shape by the ERECTA gene in Arabidopsis. Morphog. Pattern Form. Biol. Syst. 2003, 153–164. [Google Scholar]
- Liu, T.; Jiang, G.Q.; Yao, X.F.; Liu, C.M. The leucine-rich repeat receptor-like kinase OsERL plays a critical role in anther lobe formation in rice. Biochem. Biophys. Res. Commun. 2021, 563, 85–91. [Google Scholar] [CrossRef]
- Dobritsa, A.; Shrestha, J.; Morant, M.; Pinot, F.; Matsuno, M.; Swanson, R.; Møller, B.L.; Preuss, D. CYP704B1 is a long-chain fatty acid ω-hydroxylase essential for sporopollenin synthesis in pollen of Arabidopsis. Plant Physiol. 2009, 151, 574–589. [Google Scholar] [CrossRef]
- Ji, J.L.; Cao, W.X.; Yang, L.M.; Fang, Z.Y.; Zhang, Y.Y.; Zhuang, M.; Lv, H.H.; Wang, Y.; Liu, Y.M.; Li, Z.S.; et al. Genome-wide analysis of cabbage cytochrome P450 genes and characterization of BoCYP704B1, a gene responsible for cabbage anther development. Sci. Hortic. 2021, 283, 110096. [Google Scholar] [CrossRef]
- An, X.; Dong, Z.; Tian, Y.; Xie, K.; Wu, S.; Zhu, T.; Zhang, D.; Zhou, Y.; Niu, C.; Ma, B.; et al. ZmMs30 Encoding a Novel GDSL Lipase Is Essential for Male Fertility and Valuable for Hybrid Breeding in Maize. Mol. Plant 2019, 12, 343–359. [Google Scholar] [CrossRef] [PubMed]
- Kundu, S.; Sundd, M.; Jagannadham, M.V. Purification and Characterization of a Stable Cysteine Protease Ervatamin B, with Two Disulfide Bridges, from the Latex of Ervatamia coronaria. J. Agric. Food Chem. 2000, 48, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.D.; Liu, D.; Lv, X.M.; Wang, Y.; Xun, Z.L.; Liu, Z.X.; Li, F.L.; Lu, H. The cysteine protease CEP1, a key executor involved in tapetal programmed cell death, regulates pollen development in Arabidopsis. Plant Cell 2014, 26, 2939–2961. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Zhou, Z.; Tang, S.; Zhang, Z.; Xia, S.; Qin, M.; Li, B.; Wen, J.; Yi, B.; Shen, J.; et al. Ectopic expression of BnaC. CP20. 1 results in premature tapetal programmed cell death in Arabidopsis. Plant Cell Physiol 2016, 57, 1972–1984. [Google Scholar] [CrossRef] [PubMed]
- Niu, R.H.; Chen, Y.Y.; Chen, C.; Zhang, B.P.; Tan, J.Z. Analysis of differential expression proteins from different parts of pistillate flower in Mulberry (Morus alba). Adv. Mater. Res. 2013, 796, 62–66. [Google Scholar] [CrossRef]
- van Loon, L.C.; Rep, M.; Pieterse, C.M.J. Significance of inducible defense-related proteins in infected plants. Annu. Rev. Phytopathol. 2006, 44, 135–162. [Google Scholar] [CrossRef]
- Worrall, D.; Hird, D.L.; Hodge, R.; Paul, W.; Draper, J.; Scott, R. Premature dissolution of the microsporocyte callose wall causes male sterility in transgenic tobacco. Plant Cell 1992, 4, 759–771. [Google Scholar] [PubMed]
- Tsugama, D.; Fujino, K.; Liu, S.K.; Takano, T. A GDSL-type esterase/lipase gene, GELP77, is necessary for pollen dissociation and fertility in Arabidopsis. Biochem. Biophys. Res. Commun. 2020, 526, 1036–1041. [Google Scholar] [CrossRef] [PubMed]
- Yuan, G.; Zou, T.; Zhang, X.; Liu, M.; Luo, T.; He, Z.; Tao, Y.; Zhou, D.; Zhao, H.; Liu, S.; et al. A rice GDSL esterase/lipase protein (GELP) is required for anther and pollen development. Mol. Breed. 2020, 40, 90. [Google Scholar] [CrossRef]
- Chen, W.W.; Yu, X.H.; Zhang, K.S.; Shi, J.X.; De Oliveira, S.; Schreiber, L.; Shanklin, J.; Zhang, D.B. Male Sterile2 encodes a plastid-localized fatty acyl carrier protein reductase required for pollen exine development in Arabidopsis. Plant Physiol. 2011, 157, 842–853. [Google Scholar] [CrossRef]
- Doan, T.T.P.; Domergue, F.; Fournier, A.E.; Vishwanath, S.J.; Rowland, O.; Moreau, P.; Wood, C.C.; Carlsson, A.S.; Hamberg, M.; Hofvander, P. Biochemical characterization of a chloroplast localized fatty acid reductase from Arabidopsis thaliana. Biochim. Biophys. Acta (BBA)—Mol. Cell Biol. Lipids 2012, 1821, 1244–1255. [Google Scholar] [CrossRef]
- Aarts, M.G.; Dirkse, W.G.; Stiekema, W.J.; Pereira, A. Transposon tagging of a male sterility gene in Arabidopsis. Nature 1993, 363, 715–717. [Google Scholar] [CrossRef]
- Shi, J.; Tan, H.; Yu, X.-H.; Liu, Y.; Liang, W.; Ranathunge, K.; Franke, R.B.; Schreiber, L.; Wang, Y.; Kai, G.; et al. Defective pollen wall is required for anther and microspore development in rice and encodes a fatty acyl carrier protein reductase. Plant Cell 2011, 23, 2225–2246. [Google Scholar] [CrossRef]
- Huang, C.Y.; Wen, W.; Li, Q.Q.; Wang, M.F.; Xiao, S.J.; Zhang, X.K.; Huang, Q.; Qian, G.; Lin, L.; Xu, D.L. Identification, characterization and expression analysis of the 4-coumarate-coA ligase gene family in Bletilla striata. Gene Rep. 2023, 32, 101785. [Google Scholar] [CrossRef]
- Sun, H.; Li, Y.; Feng, S.; Zou, W.H.; Guo, K.; Fan, C.F.; Si, S.L.; Peng, L.C. Analysis of five rice 4-coumarate: Coenzyme A ligase enzyme activity and stress response for potential roles in lignin and flavonoid biosynthesis in rice. Biochem. Biophys. Res. Commun. 2013, 430, 1151–1156. [Google Scholar] [CrossRef]
- Ye, M.; Liu, C.S.; Liu, Y.T.; Zhang, M.; Su, K.; Zhang, C.G.; Li, X.-Y.; Wang, H.-J.; Xiao, X.; Zhang, L.-B.; et al. Transcriptomic analysis reveals the involvement of PpCYP703A2, Pp4CL, and PpABCG26 in regulating peach (Prunus persica) Pollen Fertility. J. Agric. Biotechnol. 2023, 31, 1405–1418. [Google Scholar]
- Lairson, L.L.; Henrissat, B.; Davies, G.J.; Withers, S.G. Glycosyltransferases: Structures, functions, and mechanisms. Annu. Rev. Biochem 2008, 77, 521–555. [Google Scholar] [CrossRef]
- Yonekura-Sakakibara, K.; Hanada, K. An evolutionary view of functional diversity in family 1 glycosyltransferases. Plant J. 2011, 66, 182–193. [Google Scholar] [CrossRef]
- Markovič, O.; Janeček, Š. Pectin degrading glycoside hydrolases of family 28: Sequence-structural features, specificities and evolution. Protein Eng. 2001, 14, 615–631. [Google Scholar] [CrossRef]
- Hayama, H.; Tatsuki, M.; Ito, A.; Kashimura, Y. Ethylene and fruit softening in the stony hard mutation in peach. Postharvest Biol. Technol. 2006, 41, 16–21. [Google Scholar] [CrossRef]
- He, Y.; Song, S.; Li, M.Y.; Bo, W.H.; Li, Y.Y.; Cao, M.; Wang, A.; Li, G.Q.; Liu, X.Y.; Pang, X.M. Identification and Expression Analysis of the Polygalacturonase Gene Family in Chinese Jujube (Ziziphus jujuba Mill.). Mol. Plant Breed. 2021, 19, 1442–1450. [Google Scholar]
- Hadfield, K.A.; Bennett, A.B. Polygalacturonases: Many genes in search of a function. Plant Physiol. 1998, 117, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Mohnen, D. Pectin structure and biosynthesis. Curr. Opin. Plant Biol. 2008, 11, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, M.; Kay, P.; Wilson, S.; Swain, S. ARABIDOPSIS DEHISCENCE ZONE POLYGALACTURONASE1 (ADPG1), ADPG2, and QUARTET2 are polygalacturonases required for cell separation during reproductive development in Arabidopsis. Plant Cell 2009, 21, 216–233. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.Z.; Sun, M.T.; Wu, Z.; Yu, L.; Yu, Q.H.; Tang, Y.P.; Jiang, F.L. LncRNA regulates tomato fruit cracking by coordinating gene expression via a hormone-redox-cell wall network. Res. Sq. 2020, 20, 162. [Google Scholar] [CrossRef] [PubMed]
- Western, T.L.; Cheng, Y.; Liu, J.; Chen, X. HUA ENHANCER2, a putative DExH-box RNA helicase, maintains homeotic B and C gene expression in Arabidopsis. Development 2002, 129, 1569–1581. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.S.; Tso, Y.S.; Zheng, Y.L.; Zhang, F.D. Cloning and Characterization of DEAD-box RNA Helicases Gene from Maize (Zea mays). Chin. J. Biochem. Mol. Biol. 2006, 22, 640–646. [Google Scholar]
- Breitenbach, H.H.; Wenig, M.; Wittek, F.; Jordá, L.; Maldonado-Alconada, A.M.; Sarioglu, H.; Colby, T.; Knappe, C.; Bichlmeier, M.; Pabst, E.; et al. Contrasting roles of the apoplastic aspartyl protease APOPLASTIC, ENHANCED DISEASE SUSCEPTIBILITY1-DEPENDENT1 and LEGUME LECTIN-LIKE PROTEIN1 in Arabidopsis systemic acquired resistance. Plant Physiol. 2014, 165, 791–809. [Google Scholar] [CrossRef]
- Byrne, M.E. A role for the ribosome in development. Trends Plant Sci. 2009, 14, 512–519. [Google Scholar] [CrossRef]
- Tsai, Y.C.C.; Mueller-Cajar, O.; Saschenbrecker, S.; Hartl, F.U.; Hayer-Hartl, M. Chaperonin Cofactors, Cpn10 and Cpn20, of Green Algae and Plants Function as Hetero-oligomeric Ring Complexes. J. Biol. Chem. 2012, 287, 20471–20481. [Google Scholar] [CrossRef]
- Williams, M.E.; Sussex, I.M. Developmental regulation of ribosomal protein L16 genes in Arabidopsis thaliana. Plant J. 1995, 8, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Barakat, A.; Szick-Miranda, K.; Chang, I.F.; Guyot, R.; Blanc, G.; Cooke, R.; Delseny, M.; Bailey-Serres, J. The organization of cytoplasmic ribosomal protein genes in the Arabidopsis genome. Plant Physiol. 2001, 127, 398–415. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Hofius, D.; Hajirezaei, M.R.; Fernie, A.; Börnke, F.; Sonnewald, U. Functional analysis of the essential bifunctional tobacco enzyme 3-dehydroquinate dehydratase/shikimate dehydrogenase in transgenic tobacco plants. J. Exp. Bot. 2007, 58, 2053–2067. [Google Scholar] [CrossRef] [PubMed]
- Close, T.J.; Kortt, A.A.; Chandler, P.M. A cDNA-based comparison of dehydration-induced proteins (dehydrins) in barley and corn. Plant Mol. Biol. 1989, 13, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Close, T.J. Dehydrins: A commonality in the response of plants to dehydration and low temperature. Physiol. Plant. 1997, 100, 291–296. [Google Scholar] [CrossRef]
- Wiśniewski, K.; Zagdańska, B. Genotype-dependent proteolytic response of spring wheat to water deficiency. J. Exp. Bot. 2001, 52, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Rizhsky, L.; Liang, H.J.; Mittler, R. The Combined Effect of Drought Stress and Heat Shock on Gene Expression in Tobacco. Plant Physiol. 2002, 130, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.D.; Wang, F.; Smith, A.G. A flower-specific gene encoding an osmotin-like protein from Lycopersicon esculentum. Gene 1996, 179, 301–302. [Google Scholar] [CrossRef]
- Wisniewski, J.R.; Zougman, A.; Nagaraj, N.; Mann, M. Universal sample preparation method for proteome analysis. Nat. Methods 2009, 6, 359–362. [Google Scholar] [CrossRef]

| Biological Function | Accession Number | Protein Description | p Value | Fold Change | |||
|---|---|---|---|---|---|---|---|
| S2/S1 | S3/S1 | S4/S2 | S4/S3 | ||||
| Single-organism process/post-embryonic development | XP_018834821.1 | Stress-induced protein KIN2-like [Juglans regia] | 4.78 × 10−13/2.38 × 10−3 | 2.0669 | 23.8047 | 47.2468 | 4.8394 |
| Carbohydrate metabolic process | XP_018860371.1 | Probable xyloglucan endotransglucosylase/hydrolase protein 6 [Juglans regia] | 8.43 × 10−21 | 2.899 | 5.8383 | 7.8709 | 3.1480 |
| Generation of precursor metabolites and energy/photosynthesis | XP_018841899.1 | Chlorophyll a-b binding protein of LHC II type 1-like [Juglans regia] | 7.25 × 10−20/1.17 × 10−8 | 2.3899 | 2.2833 | 2.0106 | 2.1159 |
| Response to cadmium ions | XP_018823000.1 | Formate dehydrogenase, mitochondrial [Juglans regia] | 2.59 × 10−21 | 2.2702 | 3.0172 | 2.7081 | 2.1303 |
| Small-molecule metabolic process/photosynthesis/post-embryonic development | XP_018821822.1 | Sedoheptulose-1,7-bisphosphatase, chloroplastic [Juglans regia] | 1.38 × 10−36/1.17 × 10−8/2.38 × 10−3 | 2.0007 | 3.2978 | 2.8434 | 1.6835 |
| Catabolic process | XP_018852509.1 | Puromycin-sensitive aminopeptidase isoform X1 [Juglans regia] | 3.90 × 10−25 | 1.5417 | 1.8803 | 2.3313 | 1.9589 |
| Metabolic process | XP_018811660.1 | Putative 4-hydroxy-4-methyl-2-oxoglutarate aldolase 2 [Juglans regia] | 2.56 × 10−10 | 2.9671 | 2.2085 | 0.3743 | 0.4143 |
| Small-molecule metabolic process | XP_018837921.1 | 1,4-Dihydroxy-2-naphthoyl-CoA synthase, peroxisomal-like isoform X1 [Juglans regia] | 1.38 × 10−36 | 0.3629 | 6.7074 | 2.8236 | 0.1578 |
| Single-organism cellular process | XP_018809241.1 | COBRA-like protein 7 [Juglans regia] | 7.94 × 10−14 | 0.4089 | 2.2769 | 13.4576 | 3.7633 |
| Small-molecule metabolic process/post-embryonic development | XP_018845399.1 | Inositol-3-phosphate synthase-like [Juglans regia] | 1.38 × 10−36/2.38 × 10−3 | 0.4059 | 2.9501 | 3.6717 | 0.5321 |
| ― | XP_018845416.1 | Uncharacterized protein LOC109009413 [Juglans regia] | ― | 0.4834 | 2.9261 | 1.7107 | 0.3114 |
| Single-organism cellular process | XP_018831299.1 | Tubulin-folding cofactor A-like [Juglans regia] | 7.94 × 10−14 | 0.4295 | 0.3698 | 2.2558 | 2.4488 |
| Contrast | Accession Number | Protein Description | Fold Change |
|---|---|---|---|
| DEPs (S2/S1) | XP_018832838.1 | oxygen-evolving enhancer protein 2, chloroplastic [Juglans regia] | 4.9200 |
| XP_018846385.1 | probable nucleoredoxin 1 [Juglans regia] | 4.2982 | |
| XP_018814447.1 | ribulose bisphosphate carboxylase/oxygenase activase, chloroplastic-like isoform X1 [Juglans regia] | 4.0772 | |
| XP_018835529.1 | endochitinase-like [Juglans regia] | 4.0325 | |
| XP_018843857.1 | beta-glucosidase 12-like [Juglans regia] | 4.0188 | |
| XP_018858848.1 | polyphenol oxidase, chloroplastic-like [Juglans regia] | 3.7727 | |
| YP_009186194.1 | photosystem II 47 kDa protein (chloroplast) [Juglans regia] | 3.6141 | |
| XP_018847910.1 | phosphoglycerate kinase, chloroplastic [Juglans regia] | 3.3870 | |
| XP_018834056.1 | probable alpha-mannosidase At5g13980 isoform X4 [Juglans regia] | 3.3830 | |
| XP_018843283.1 | profilin [Juglans regia] | 3.3010 | |
| XP_018831299.1 | tubulin-folding cofactor A-like [Juglans regia] | 0.4295 | |
| XP_018807936.1 | DEAD-box ATP-dependent RNA helicase 3, chloroplastic-like [Juglans regia] | 0.4122 | |
| XP_018809241.1 | COBRA-like protein 7 [Juglans regia] | 0.4089 | |
| XP_018845399.1 | inositol-3-phosphate synthase-like [Juglans regia] | 0.4059 | |
| XP_018842878.1 | 10 kDa chaperonin-like [Juglans regia] | 0.4022 | |
| XP_018850608.1 | caffeic acid 3-O-methyltransferase [Juglans regia] | 0.3738 | |
| XP_018828035.1 | enhancer of mRNA-decapping protein 4-like [Juglans regia] | 0.3632 | |
| XP_018837921.1 | 1,4-dihydroxy-2-naphthoyl-CoA synthase, peroxisomal-like isoform X1 [Juglans regia] | 0.3630 | |
| XP_018845332.1 | aldehyde oxidase GLOX [Juglans regia] | 0.3007 | |
| XP_018849734.1 | UBP1-associated protein 2C-like isoform X3 [Juglans regia] | 0.2916 | |
| DEPs (S3/S1) | XP_018836778.1 | 4-coumarate-CoA ligase-like 1 [Juglans regia] | 43.2116 |
| XP_018852740.1 | pentatricopeptide repeat-containing protein At1g02060, chloroplastic-like [Juglans regia] | 39.9188 | |
| XP_018830780.1 | stamen-specific protein FIL1-like [Juglans regia] | 34.8341 | |
| XP_018848917.1 | probable LRR receptor-like serine/threonine-protein kinase At4g36180 [Juglans regia] | 34.7458 | |
| XP_018822513.1 | putative leucine-rich repeat receptor-like serine/threonine-protein kinase At2g24130 [Juglans regia] | 32.0020 | |
| XP_018848853.1 | fatty acyl-CoA reductase 2 [Juglans regia] | 29.4214 | |
| XP_018844051.1 | probable glucan endo-1,3-beta-glucosidase A6 [Juglans regia] | 28.6322 | |
| XP_018835774.1 | pathogenesis-related protein 5-like [Juglans regia] | 27.6657 | |
| XP_018838485.1 | UDP-glycosyltransferase 79B30 isoform X1 [Juglans regia] | 26.5040 | |
| XP_018833146.1 | GDSL esterase/lipase At5g22810-like [Juglans regia] | 26.3574 | |
| XP_018818895.1 | interactor of constitutive active ROPs 3 [Juglans regia] | 0.2238 | |
| XP_018815447.1 | 14-3-3-like protein A [Juglans regia] | 0.2179 | |
| XP_018847629.1 | bifunctional 3-dehydroquinate dehydratase/shikimate dehydrogenase, chloroplastic-like [Juglans regia] | 0.2094 | |
| XP_018821706.1 | probably inactive leucine-rich repeat receptor-like protein kinase IMK2 [Juglans regia] | 0.2087 | |
| XP_018824157.1 | rubisco accumulation factor 1, chloroplastic [Juglans regia] | 0.2060 | |
| XP_018824485.1 | DNA replication licensing factor MCM6 [Juglans regia] | 0.1852 | |
| XP_018824515.1 | 40S ribosomal protein S4-1-like [Juglans regia] | 0.1628 | |
| XP_018852330.1 | 60S ribosomal protein L5 [Juglans regia] | 0.1592 | |
| XP_018812412.1 | 10 kDa chaperonin-like [Juglans regia] | 0.1138 | |
| XP_018833667.1 | early nodulin-like protein 1 [Juglans regia] | 0.1126 | |
| DEPs (S4/S2) | XP_018849923.1 | exopolygalacturonase-like [Juglans regia] | 53.2114 |
| XP_018834136.1 | late embryogenesis abundant protein 2-like [Juglans regia] | 48.7676 | |
| XP_018834821.1 | stress-induced protein KIN2-like [Juglans regia] | 47.2469 | |
| XP_018834135.1 | late embryogenesis abundant protein 2-like [Juglans regia] | 43.2973 | |
| XP_018847735.1 | xyloglucan endotransglucosylase/hydrolase protein 2-like [Juglans regia] | 40.0863 | |
| XP_018838067.1 | late embryogenesis abundant protein 2-like [Juglans regia] | 39.2749 | |
| XP_018843119.1 | mannan endo-1,4-beta-mannosidase 4-like [Juglans regia] | 37.5167 | |
| XP_018824181.1 | ervatamin-B-like [Juglans regia] | 37.0114 | |
| XP_018848556.1 | cysteine proteinase inhibitor B-like [Juglans regia] | 36.5067 | |
| XP_018814951.1 | senescence-specific cysteine protease SAG39-like [Juglans regia] | 35.1314 | |
| XP_018818948.1 | protein argonaute 4-like isoform X2 [Juglans regia] | 0.1346 | |
| XP_018840279.1 | protein fluG [Juglans regia] | 0.1293 | |
| XP_018837465.1 | uncharacterized protein At5g48480-like [Juglans regia] | 0.1278 | |
| XP_018837432.1 | uncharacterized protein LOC109003650 [Juglans regia] | 0.1175 | |
| XP_018831451.1 | metalloendoproteinase 2-MMP-like [Juglans regia] | 0.1023 | |
| XP_018847629.1 | bifunctional 3-dehydroquinate dehydratase/shikimate dehydrogenase, chloroplastic-like [Juglans regia] | 0.0802 | |
| XP_018857107.1 | 60S ribosomal protein L11 [Juglans regia] | 0.0728 | |
| XP_018818184.1 | aspartyl protease AED3-like [Juglans regia] | 0.0630 | |
| XP_018823302.1 | osmotin-like protein isoform X1 [Juglans regia] | 0.0625 | |
| XP_018833667.1 | early nodulin-like protein 1 [Juglans regia] | 0.0384 | |
| DEPs (S4/S3) | XP_018845149.1 | uncharacterized protein LOC109009208 [Juglans regia] | 7.4335 |
| XP_018842192.1 | aspartic proteinase nepenthesin-1-like, partial [Juglans regia] | 7.2816 | |
| XP_018819962.1 | basic form of pathogenesis-related protein 1-like [Juglans regia] | 7.1306 | |
| XP_018824181.1 | ervatamin-B-like [Juglans regia] | 6.6394 | |
| XP_018817239.1 | serine carboxypeptidase-like 40 [Juglans regia] | 6.5505 | |
| XP_018845045.1 | probable receptor-like protein kinase At5g24010 [Juglans regia] | 6.4490 | |
| XP_018807966.1 | putative pectinesterase 63 [Juglans regia] | 6.3742 | |
| XP_018849923.1 | exopolygalacturonase-like [Juglans regia] | 6.2691 | |
| XP_018860192.1 | putative germin-like protein 2-1 [Juglans regia] | 6.1890 | |
| XP_018825423.1 | protein MEN-8-like [Juglans regia] | 6.0855 | |
| XP_018839956.1 | LRR receptor-like serine/threonine-protein kinase GSO1 [Juglans regia] | 0.0909 | |
| XP_018818184.1 | aspartyl protease AED3-like [Juglans regia] | 0.0896 | |
| XP_018844051.1 | probable glucan endo-1,3-beta-glucosidase A6 [Juglans regia] | 0.0891 | |
| XP_018851272.1 | leucine-rich repeat receptor protein kinase EMS1-like [Juglans regia] | 0.0837 | |
| XP_018845266.1 | cytochrome P450 704B1-like isoform X2 [Juglans regia] | 0.0696 | |
| XP_018848917.1 | probable LRR receptor-like serine/threonine-protein kinase At4g36180 [Juglans regia] | 0.0670 | |
| XP_018841433.1 | type III polyketide synthase B [Juglans regia] | 0.0628 | |
| XP_018814962.1 | aldehyde oxidase GLOX-like [Juglans regia] | 0.0609 | |
| XP_018822513.1 | putative leucine-rich repeat receptor-like serine/threonine-protein kinase At2g24130 [Juglans regia] | 0.0558 | |
| XP_018848853.1 | fatty acyl-CoA reductase 2 [Juglans regia] | 0.0480 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Chen, L.; Liu, R.; Cao, S.; Lu, Z. Comparative Proteomic Analysis of Floral Buds before and after Opening in Walnut (Juglans regia L.). Int. J. Mol. Sci. 2024, 25, 7878. https://doi.org/10.3390/ijms25147878
Li H, Chen L, Liu R, Cao S, Lu Z. Comparative Proteomic Analysis of Floral Buds before and after Opening in Walnut (Juglans regia L.). International Journal of Molecular Sciences. 2024; 25(14):7878. https://doi.org/10.3390/ijms25147878
Chicago/Turabian StyleLi, Haoxian, Lina Chen, Ruitao Liu, Shangyin Cao, and Zhenhua Lu. 2024. "Comparative Proteomic Analysis of Floral Buds before and after Opening in Walnut (Juglans regia L.)" International Journal of Molecular Sciences 25, no. 14: 7878. https://doi.org/10.3390/ijms25147878
APA StyleLi, H., Chen, L., Liu, R., Cao, S., & Lu, Z. (2024). Comparative Proteomic Analysis of Floral Buds before and after Opening in Walnut (Juglans regia L.). International Journal of Molecular Sciences, 25(14), 7878. https://doi.org/10.3390/ijms25147878






