The Impact of a Non-Pathogenic Strain of Fusarium Oxysporum on Structural and Biochemical Properties of Flax Suspension Cultures
Abstract
1. Introduction
2. Results and Discussion
2.1. Determination of Biocontrol Properties of F. Oxysporum Fo47
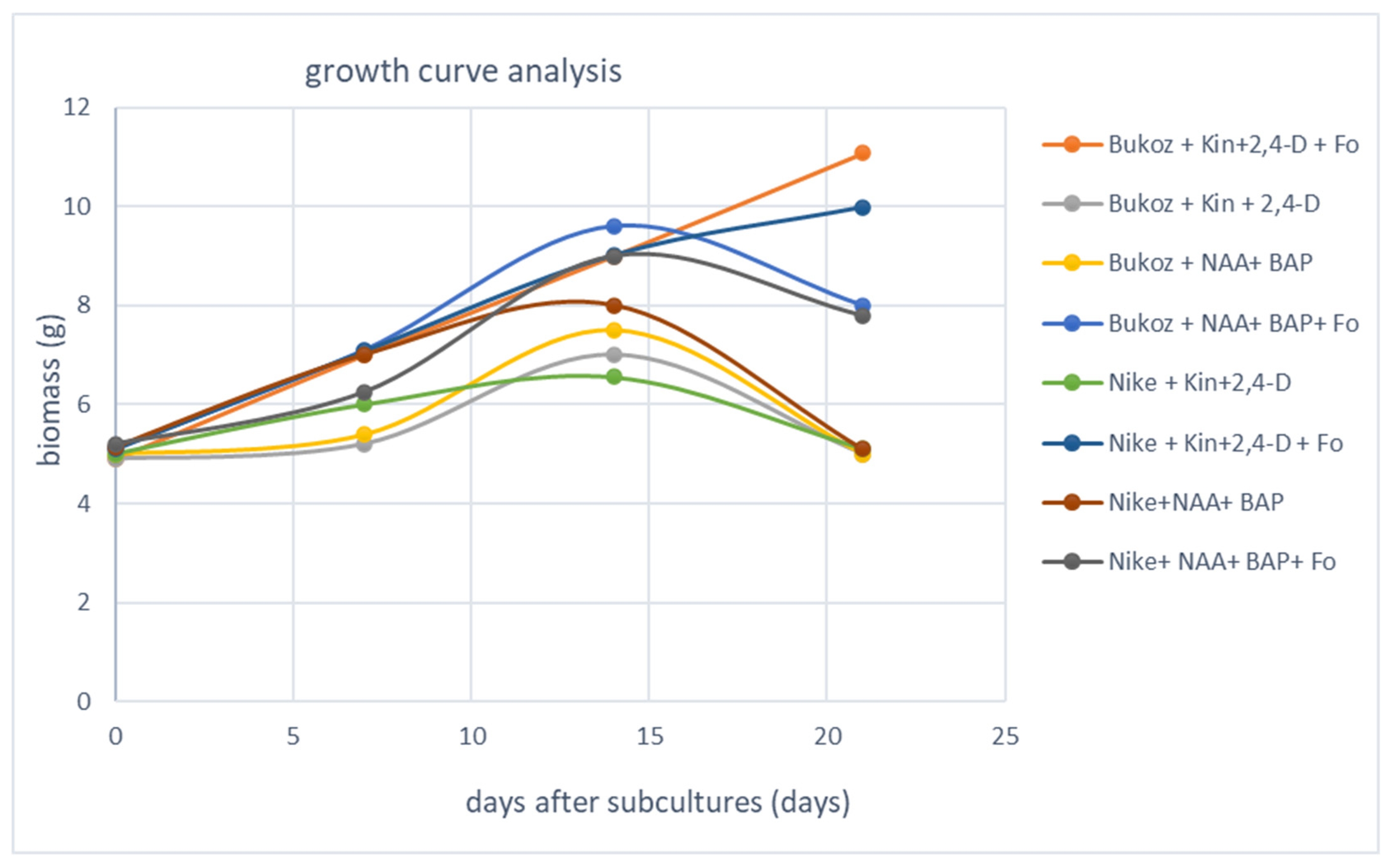
2.2. Determination of Biomass in Callus Suspension Cultures
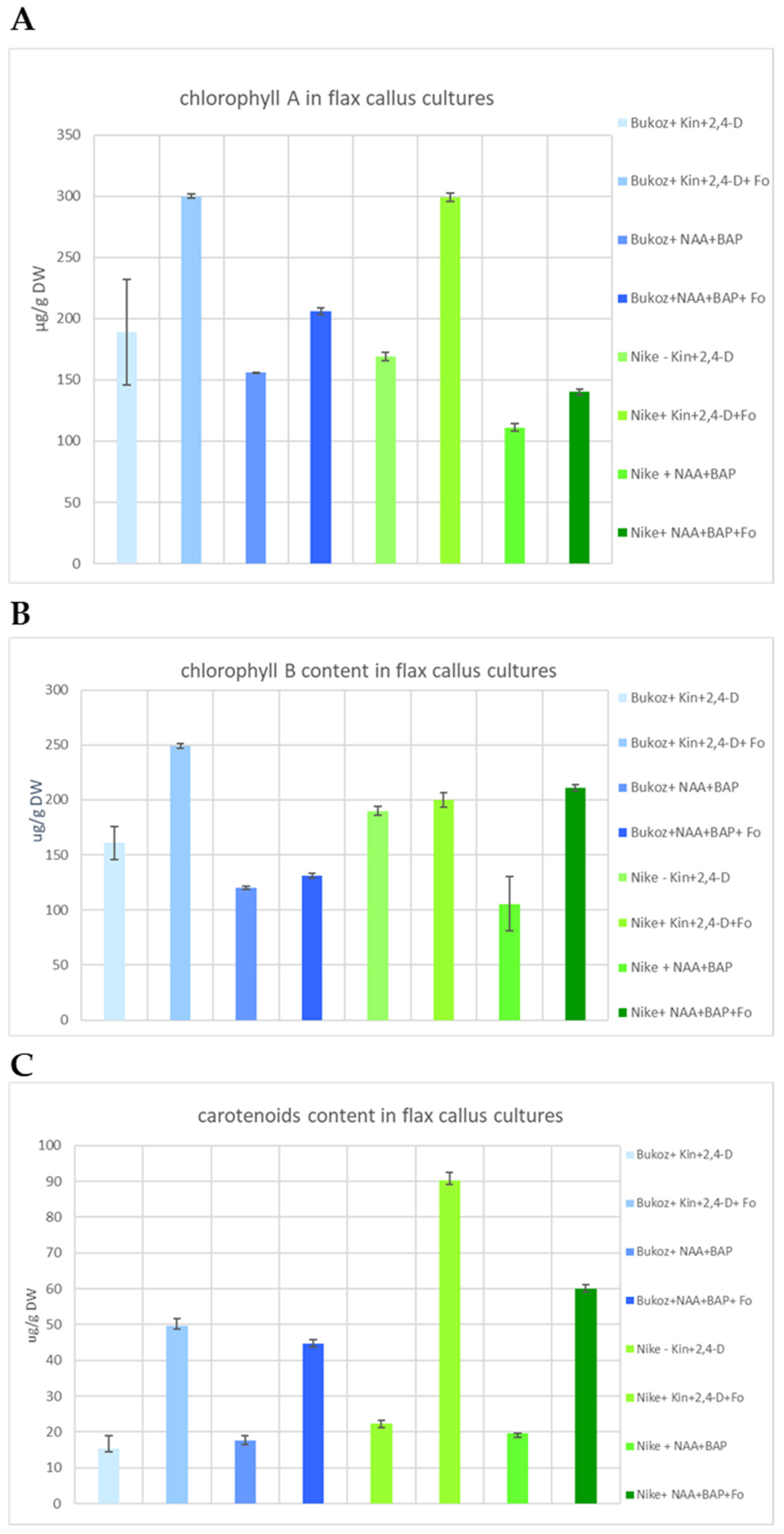
2.3. Content of Photosynthetically Active Pigments in Flax Suspension Cultures
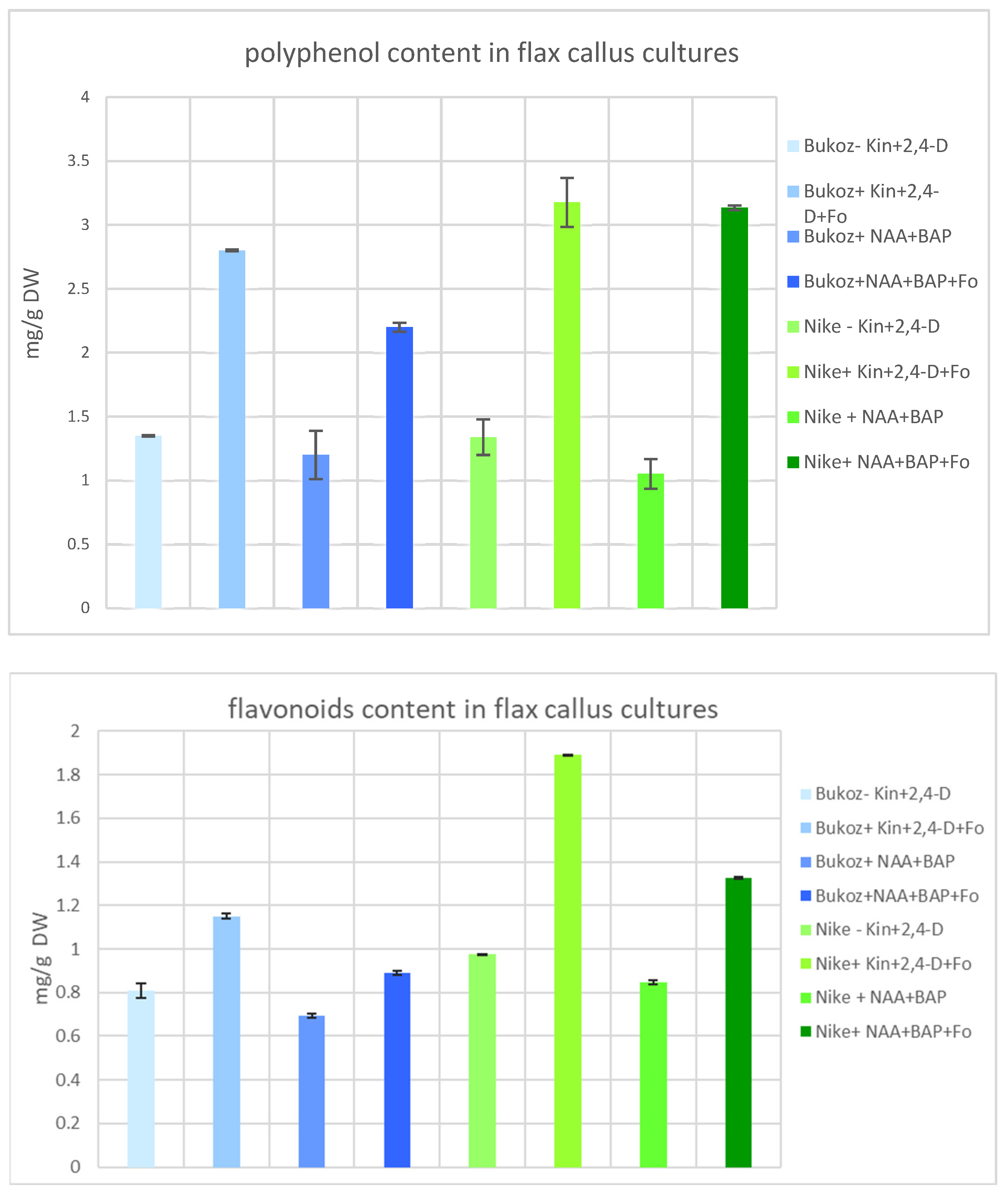
2.4. Content of Polyphenols and Flavonoids in Flax Suspension Cultures
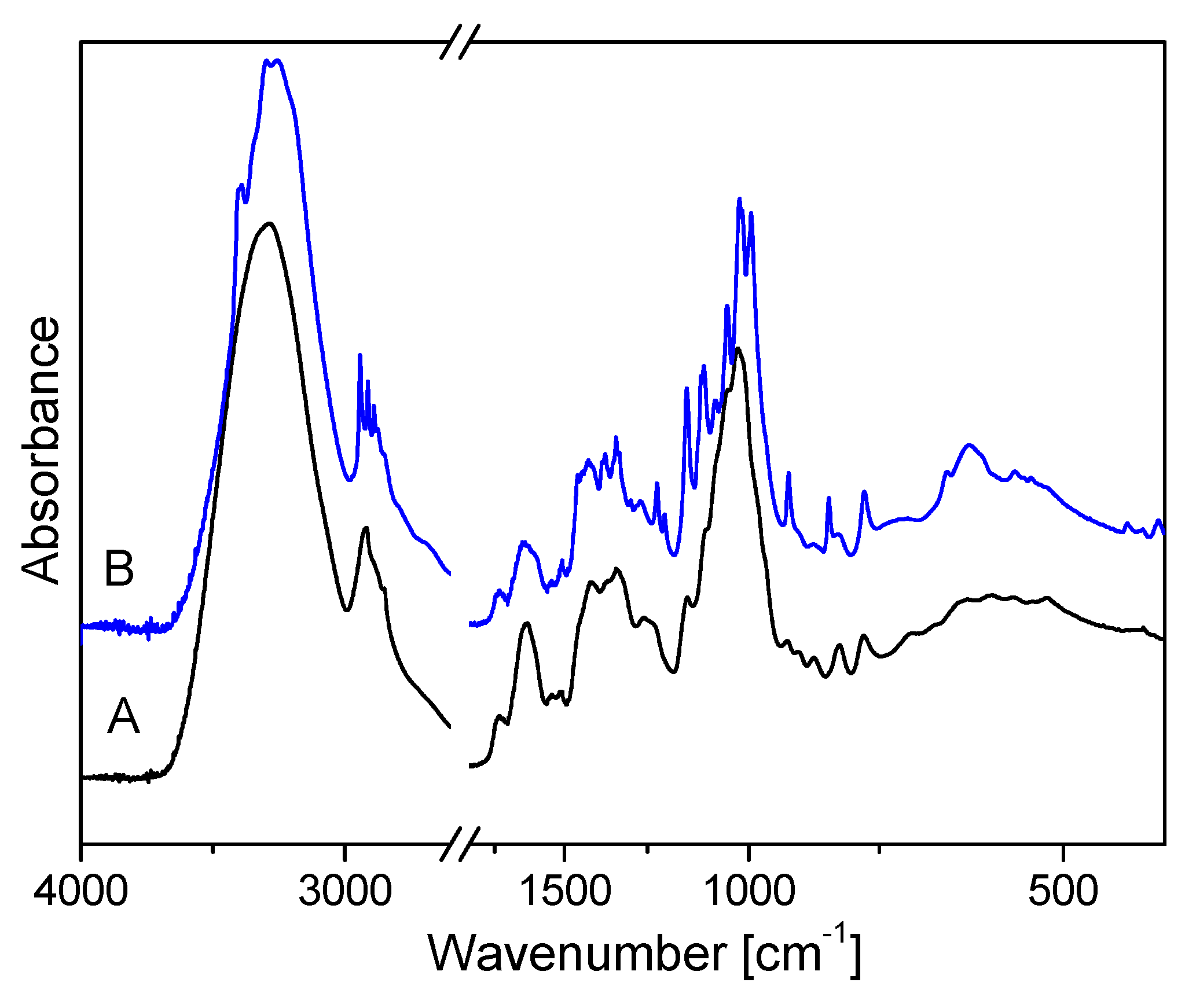
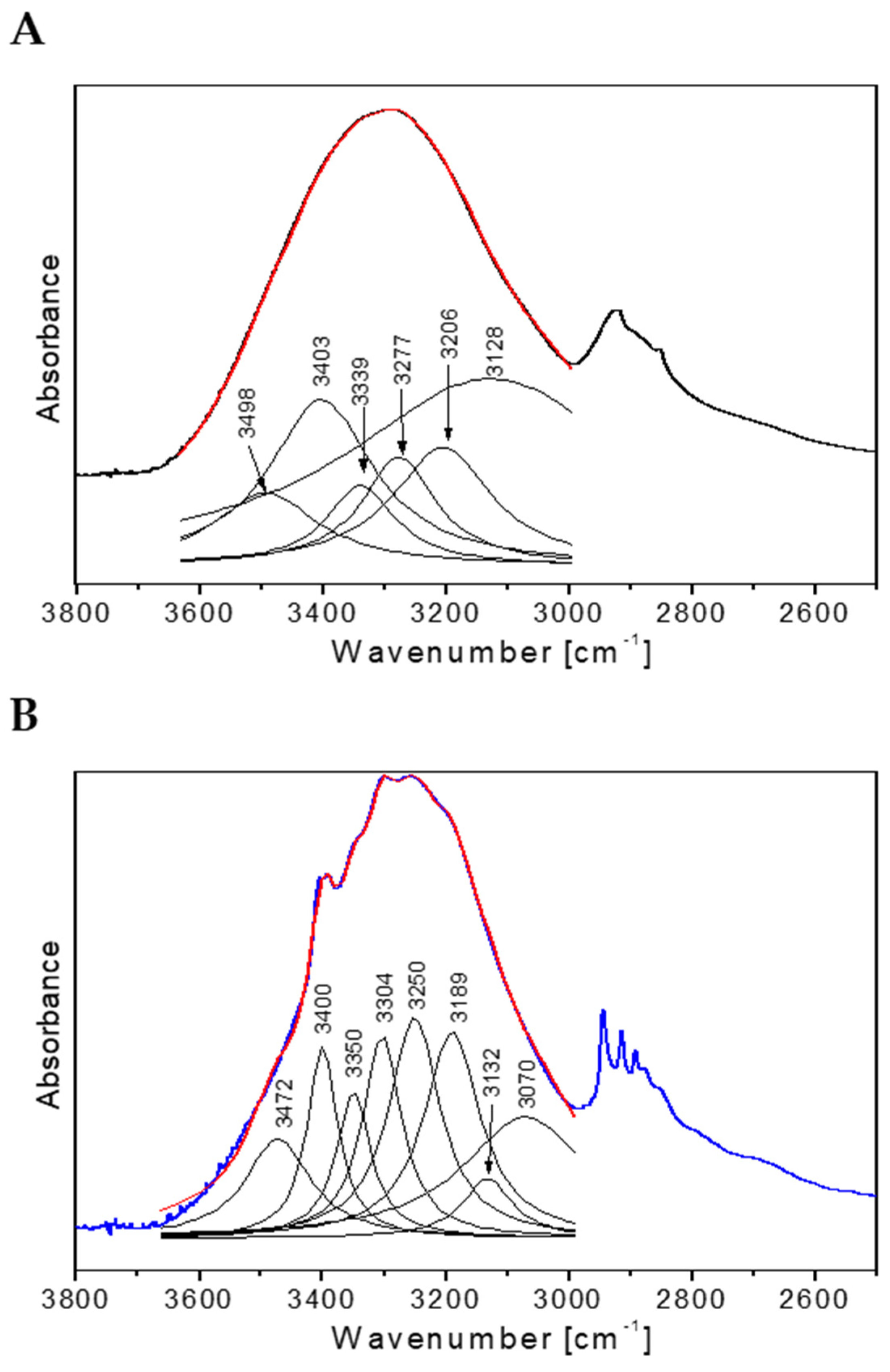
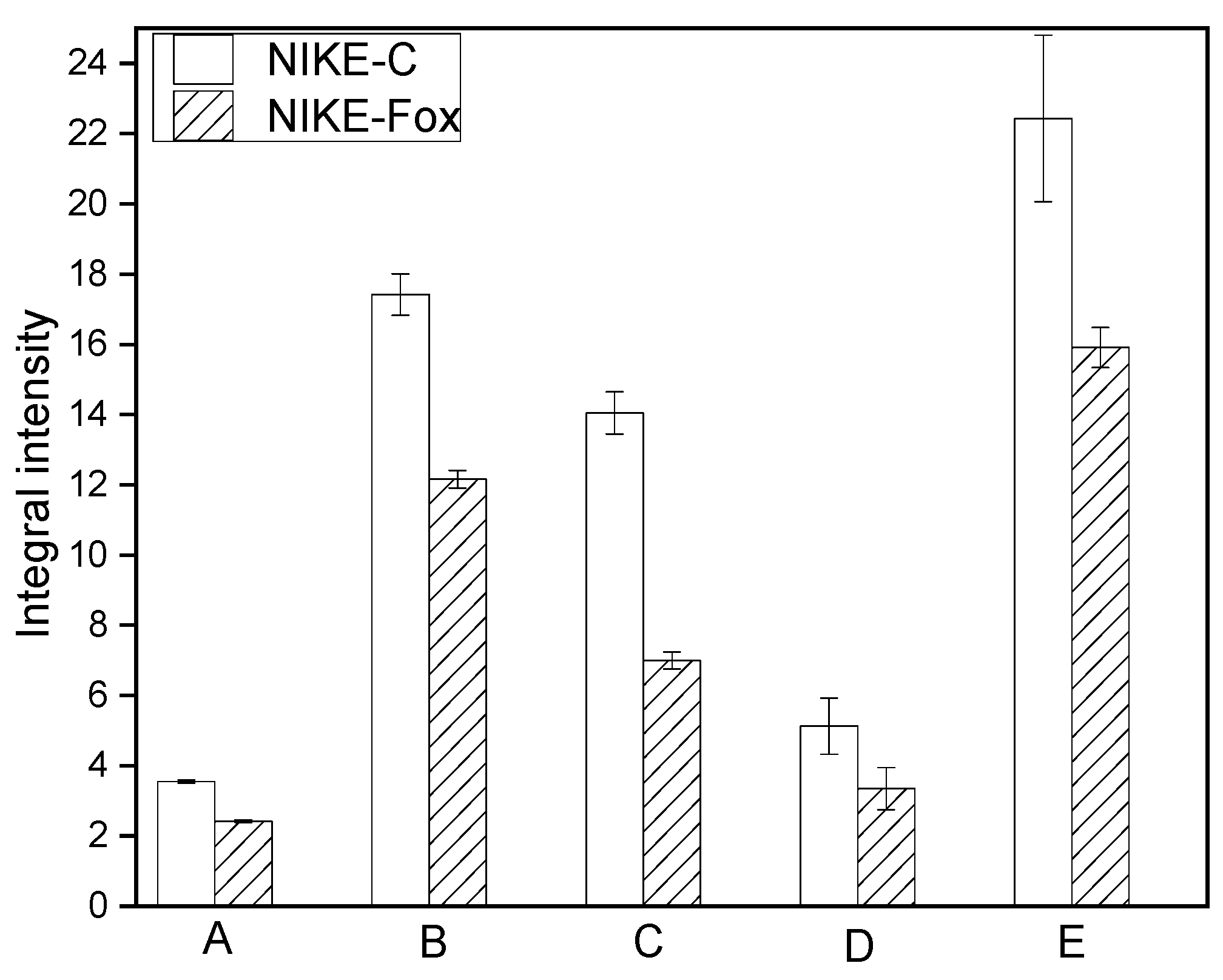
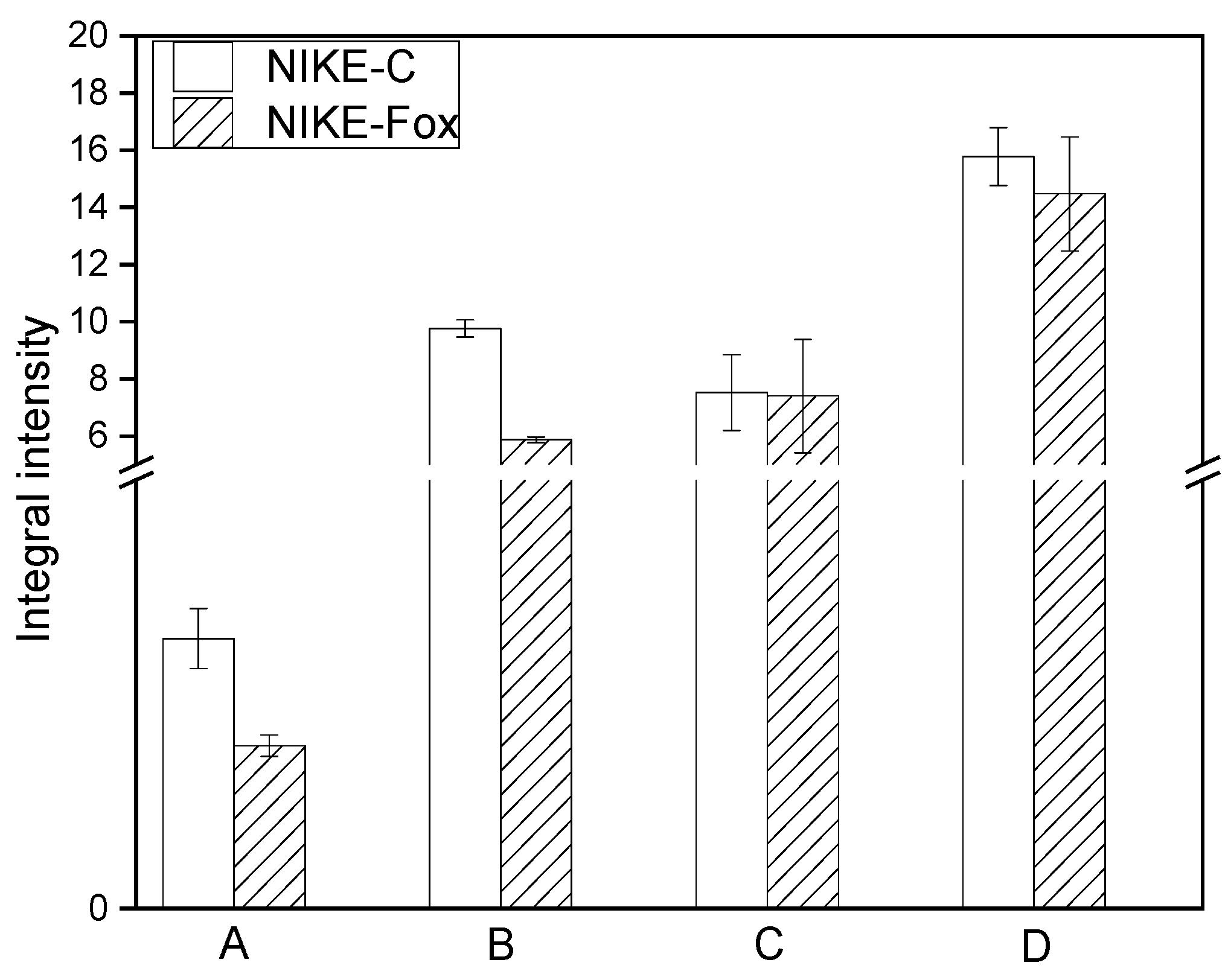
2.5. Spectroscopic Analysis of Flax Suspension Cultures Elicited with F. Oxysporum Fo47
3. Materials and Methods
3.1. Plant Material
3.2. Induction of Callus
3.3. Fungal Material
3.4. Growth Inhibition Tests of Plant Pathogens
3.5. Callus Suspension Cultures
3.6. Plant Treatment with F. Oxysporum Strain Fo47
3.7. Analysis of FTIR Spectra
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chandran, H.; Meena, M.; Barupal, T.; Sharma, K. Plant tissue culture as a perpetual source for production of industrially important bioactive compounds. Biotechnol. Rep. 2020, 26, e00450. [Google Scholar] [CrossRef]
- Hasnain, A.; Naqvi, S.A.H.; Ayesha, S.I.; Khalid, F.; Ellahi, M.; Iqbal, S.; Hassan, M.Z.; Abbas, A.; Adamski, R.; Markowska, D.; et al. Corrigendum: Plants in vitro propagation with its applications in food, pharmaceuticals and cosmetic industries; current scenario and future approaches. Front. Plant Sci. 2023, 14. [Google Scholar] [CrossRef]
- Selwal, N.; Rahayu, F.; Herwati, A.; Latifah, E.; Supriyono; Suhara, C.; Suastika, I.B.K.; Mahayu, W.M.; Wani, A.K. Enhancing secondary metabolite production in plants: Exploring traditional and modern strategies. J. Agric. Food Res. 2023, 14. [Google Scholar] [CrossRef]
- Grzegorczyk-Karolak, I.; Tabaka, P.; Weremczuk-Jeżyna, I. Enhancing polyphenol yield in Salvia viridis L. shoot culture through liquid medium optimization and light spectrum manipulation. Plant Cell Tissue Organ Cult. (PCTOC) 2024, 156, 1–13. [Google Scholar] [CrossRef]
- Talebi, S.M. Changes in Plant Secondary Metabolite Profiles in Response to Environmental Stresses. In Plant Stress Mitigators; Elsevier: Amsterdam, The Netherlands, 2023; pp. 325–339. [Google Scholar]
- de Lamo, F.J.; Spijkers, S.B.; Takken, F.L.W. Protection to Tomato Wilt Disease Conferred by the Nonpathogen Fusarium oxysporum Fo47 is More Effective Than that Conferred by Avirulent Strains. Phytopathology 2021, 111, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Olivain, C.; Trouvelot, S.; Binet, M.N.; Cordier, C.; Pugin, A.; Alabouvette, C. Colonization of flax roots and early physio-logical responses of flax cells inoculated with pathogenic and nonpathogenic strains of Fusarium oxysporum. Appl. Environ. Microbiol. 2003, 69, 5453–5462. [Google Scholar] [CrossRef] [PubMed]
- Chitnis, V.R.; Suryanarayanan, T.S.; Nataraja, K.N.; Prasad, S.R.; Oelmüller, R.; Shaanker, R.U. Fungal Endophyte-Mediated Crop Improvement: The Way Ahead. Front. Plant Sci. 2020, 11, 561007. [Google Scholar] [CrossRef]
- Rodriguez, R.J.; White, J.F., Jr.; Arnold, A.E.; Redman, R.S. Fungal endophytes: Diversity and functional roles. New Phytol. 2009, 182, 314–330. [Google Scholar] [CrossRef]
- Benhamou, N.; Garand, C.; Goulet, A. Ability of Nonpathogenic Fusarium oxysporum Strain Fo47 To Induce Resistance against Pythium ultimum Infection in Cucumber. Appl. Environ. Microbiol. 2002, 68, 4044–4060. [Google Scholar] [CrossRef]
- Olivain, C.; Humbert, C.; Nahalkova, J.; Fatehi, J.; L’Haridon, F.; Alabouvette, C. Colonization of Tomato Root by Pathogenic and Nonpathogenic Fusarium oxysporum Strains Inoculated Together and Separately into the Soil. Appl. Environ. Microbiol. 2006, 72, 1523–1531. [Google Scholar] [CrossRef]
- Constantin, M.E.; de Lamo, F.J.; Vlieger, B.V.; Rep, M.; Takken, F.L.W. Endophyte-Mediated Resistance in Tomato to Fusarium oxysporum Is Independent of ET, JA, and SA. Front. Plant Sci. 2019, 10, 979. [Google Scholar] [CrossRef] [PubMed]
- Olivain, C.; Alabouvette, C. Colonization of tomato root by a non-pathogenic strain of Fusarium oxysporum. New Phytol. 1997, 137, 481–494. [Google Scholar] [CrossRef]
- Wojtasik, W.; Kostyn, K.; Preisner, M.; Czuj, T.; Zimniewska, M.; Szopa, J.; Wróbel-Kwiatkowska, M. Cottonization of De-corticated and Degummed Flax Fiber–A Novel Approach to Improving the Quality of Flax Fiber and its Biomedical Appli-cations. J. Nat. Fibers 2024, 21, 1–24. [Google Scholar] [CrossRef]
- Lanubile, A.; Muppirala, U.K.; Severin, A.J.; Marocco, A.; Munkvold, G.P. Transcriptome profiling of soybean (Glycine max) roots challenged with pathogenic and non-pathogenic isolates of Fusarium oxysporum. BMC Genom. 2015, 16, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, M.; Abbasi, B.H.; Garros, L.; Drouet, S.; Zahir, A.; Ahmad, W.; Giglioli-Guivarc’h, N.; Hano, C. Yeast-extract improved biosynthesis of lignans and neolignans in cell suspension cultures of Linum usitatissimum L. Plant Cell Tissue Organ Cult. (PCTOC) 2018, 135, 347–355. [Google Scholar] [CrossRef]
- Wróbel-Kwiatkowska, M.; Turski, W.; Silska, G.; Rakicka-Pustułka, M.; Dymińska, L.; Rymowicz, W. Determination of Bio-active Compound Kynurenic Acid in Linum usitatissimum L. Molecules 2024, 29, 1702. [Google Scholar] [CrossRef]
- Czemplik, M.; Boba, A.; Kostyn, K.; Kulma, A.; Mituła, A.; Sztajnert, M.; Wróbel-Kwiatkowska, M.; Żuk, M.; Szopa, J.; Skórkowska-Telichowska, K. Flax Engineering for Biomedical Application. InTech 2011, 407–434. [Google Scholar]
- Zhang, J.; Chen, J.; Jia, R.; Ma, Q.; Zong, Z.; Wang, Y. Suppression of Plant Wilt Diseases by Nonpathogenic Fusarium ox-ysporum Fo47 Combined with Actinomycete Strains. Biocontrol Sci. Technol. 2018, 28, 562–573. [Google Scholar] [CrossRef]
- Guo, L.; Yu, H.; Wang, B.; Vescio, K.; De Iulio, G.A.; Yang, H.; Berg, A.; Zhang, L.; Edel-Hermann, V.; Steinberg, C.; et al. Metatranscriptomic Comparison of Endophytic and Pathogenic Fusarium–Arabidopsis Interactions Reveals Plant Tran-scriptional Plasticity. Mol. Plant-Microbe Interact. 2021, 34, 1071–1083. [Google Scholar] [CrossRef]
- Sun, T.; Rao, S.; Zhou, X.; Li, L. Plant carotenoids: Recent advances and future perspectives. Mol. Hortic. 2022, 2, 1–21. [Google Scholar] [CrossRef]
- Dhami, N.; Cazzonelli, C.I. Environmental impacts on carotenoid metabolism in leaves. Plant Growth Regul. 2020, 92, 455–477. [Google Scholar] [CrossRef]
- Veloso, J.; Alabouvette, C.; Olivain, C.; Flors, V.; Pastor, V.; García, T.; Díaz, J. Modes of action of the protective strain Fo47 in controlling verticillium wilt of pepper. Plant Pathol. 2015, 65, 997–1007. [Google Scholar] [CrossRef]
- Olivares-García, C.A.; Mata-Rosas, M.; Peña-Montes, C.; Quiroz-Figueroa, F.; Segura-Cabrera, A.; Shannon, L.M.; Loyo-la-Vargas, V.M.; Monribot-Villanueva, J.L.; Elizalde-Contreras, J.M.; Ibarra-Laclette, E.; et al. Phenylpropanoids Are Con-nected to Cell Wall Fortification and Stress Tolerance in Avocado Somatic Embryogenesis. Int. J. Mol. Sci. 2020, 21, 5679. [Google Scholar] [CrossRef] [PubMed]
- Ramaroson, M.-L.; Koutouan, C.; Helesbeux, J.-J.; Le Clerc, V.; Hamama, L.; Geoffriau, E.; Briard, M. Role of Phenylpropanoids and Flavonoids in Plant Resistance to Pests and Diseases. Molecules 2022, 27, 8371. [Google Scholar] [CrossRef]
- Lorenc-Kukuła, K.; Wróbel-Kwiatkowska, M.; Starzycki, M.; Szopa, J. Engineering flax with increased flavonoid content and thus Fusarium resistance. Physiol. Mol. Plant Pathol. 2007, 70, 38–48. [Google Scholar] [CrossRef]
- Carrillo, F.; Colom, X.; Sunol, J.J.; Saurina, J. Structural FTIR Analysis and Thermal Characterisation of Lyocell and Vis-cose-Type Fibres. Eur. Polym. J. 2004, 40, 2229–2234. [Google Scholar] [CrossRef]
- Colom, X.; Carrillo, F. Crystallinity changes in lyocell and viscose-type fibres by caustic treatment. Eur. Polym. J. 2002, 38, 2225–2230. [Google Scholar] [CrossRef]
- Dai, D.; Fan, M. Investigation of the dislocation of natural fibres by Fourier-transform infrared spectroscopy. Vib. Spectrosc. 2011, 55, 300–306. [Google Scholar] [CrossRef]
- Schwanninger, M.; Rodrigues, J.; Pereira, H.; Hinterstoisser, B. Effects of short-time vibratory ball milling on the shape of FT-IR spectra of wood and cellulose. Vib. Spectrosc. 2004, 36, 23–40. [Google Scholar] [CrossRef]
- Schulz, H.; Baranska, M. Identification and quantification of valuable plant substances by IR and Raman spectroscopy. Vib. Spectrosc. 2006, 43, 13–25. [Google Scholar] [CrossRef]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies, 3rd ed.; J. Wiley & Sons: Chichester, UK, 2001; Chap. 2; p. 50. [Google Scholar]
- Wojtkowiak, B.; Chabanel, M. Spectrochimie Moleculaire, Technique et Documentation; PWN: Warszawa, Poland, 1984; Chapter 4; p. 114. [Google Scholar]
- Boeriu, C.G.; Bravo, D.; Gosselink, R.J.; van Dam, J.E. Characterisation of structure-dependent functional properties of lignin with infrared spectroscopy. Ind. Crop. Prod. 2004, 20, 205–218. [Google Scholar] [CrossRef]
- Košťálová, Z.; Hromádková, Z.; Ebringerová, A.; Polovka, M.; Michaelsen, T.E.; Paulsen, B.S. Polysaccharides from the Styrian oil-pumpkin with antioxidant and complement-fixing activity. Ind. Crop. Prod. 2013, 41, 127–133. [Google Scholar] [CrossRef]
- Kruer-Zerhusen, N.; Cantero-Tubilla, B.; Wilson, D.B. Characterization of cellulose crystallinity after enzymatic treatment using Fourier transform infrared spectroscopy (FTIR). Cellulose 2017, 25, 37–48. [Google Scholar] [CrossRef]
- Wróbel-Kwiatkowska, M.; Kostyn, K.; Dymińska, L.; Hanuza, J.; Kurzawa, A.; Żuk, M.; Rymowicz, W. Spectroscopic and biochemical characteristics of flax transgenic callus cultures producing PHB. Plant Cell Tissue Organ Cult. (PCTOC) 2020, 141, 489–497. [Google Scholar] [CrossRef]
- Thongkamngam, T.; Jaenaksorn, T. Fusarium oxysporum (F221-B) as biocontrol agent against plant pathogenic fungi in vitro and in hydroponics. Plant Prot. Sci. 2017, 53, 85–95. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wróbel-Kwiatkowska, M.; Osika, A.; Liszka, J.; Lipiński, M.; Dymińska, L.; Piegza, M.; Rymowicz, W. The Impact of a Non-Pathogenic Strain of Fusarium Oxysporum on Structural and Biochemical Properties of Flax Suspension Cultures. Int. J. Mol. Sci. 2024, 25, 9616. https://doi.org/10.3390/ijms25179616
Wróbel-Kwiatkowska M, Osika A, Liszka J, Lipiński M, Dymińska L, Piegza M, Rymowicz W. The Impact of a Non-Pathogenic Strain of Fusarium Oxysporum on Structural and Biochemical Properties of Flax Suspension Cultures. International Journal of Molecular Sciences. 2024; 25(17):9616. https://doi.org/10.3390/ijms25179616
Chicago/Turabian StyleWróbel-Kwiatkowska, Magdalena, Aleksandra Osika, Justyna Liszka, Mateusz Lipiński, Lucyna Dymińska, Michał Piegza, and Waldemar Rymowicz. 2024. "The Impact of a Non-Pathogenic Strain of Fusarium Oxysporum on Structural and Biochemical Properties of Flax Suspension Cultures" International Journal of Molecular Sciences 25, no. 17: 9616. https://doi.org/10.3390/ijms25179616
APA StyleWróbel-Kwiatkowska, M., Osika, A., Liszka, J., Lipiński, M., Dymińska, L., Piegza, M., & Rymowicz, W. (2024). The Impact of a Non-Pathogenic Strain of Fusarium Oxysporum on Structural and Biochemical Properties of Flax Suspension Cultures. International Journal of Molecular Sciences, 25(17), 9616. https://doi.org/10.3390/ijms25179616





