Synthesis, Biological Activity, and Molecular-Docking Studies of New Brassinosteroid Analogs
Abstract
1. Introduction
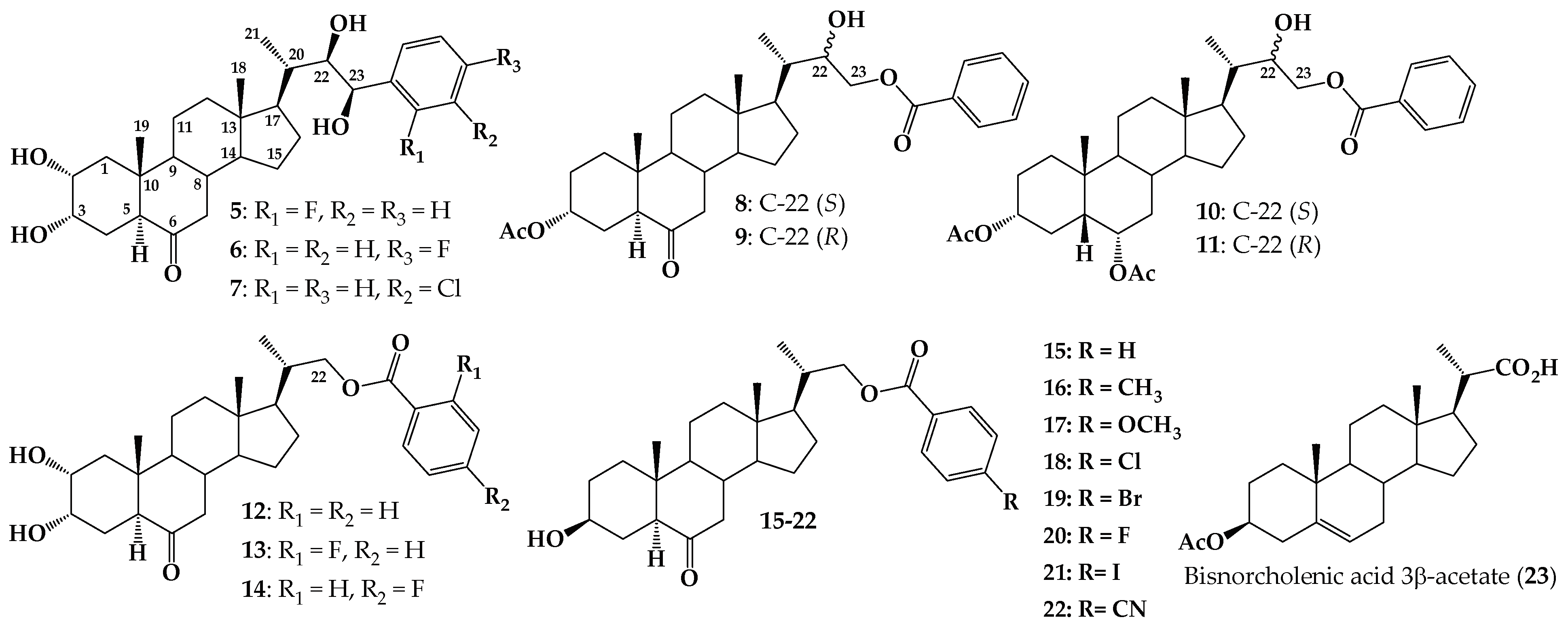
2. Results and Discussion
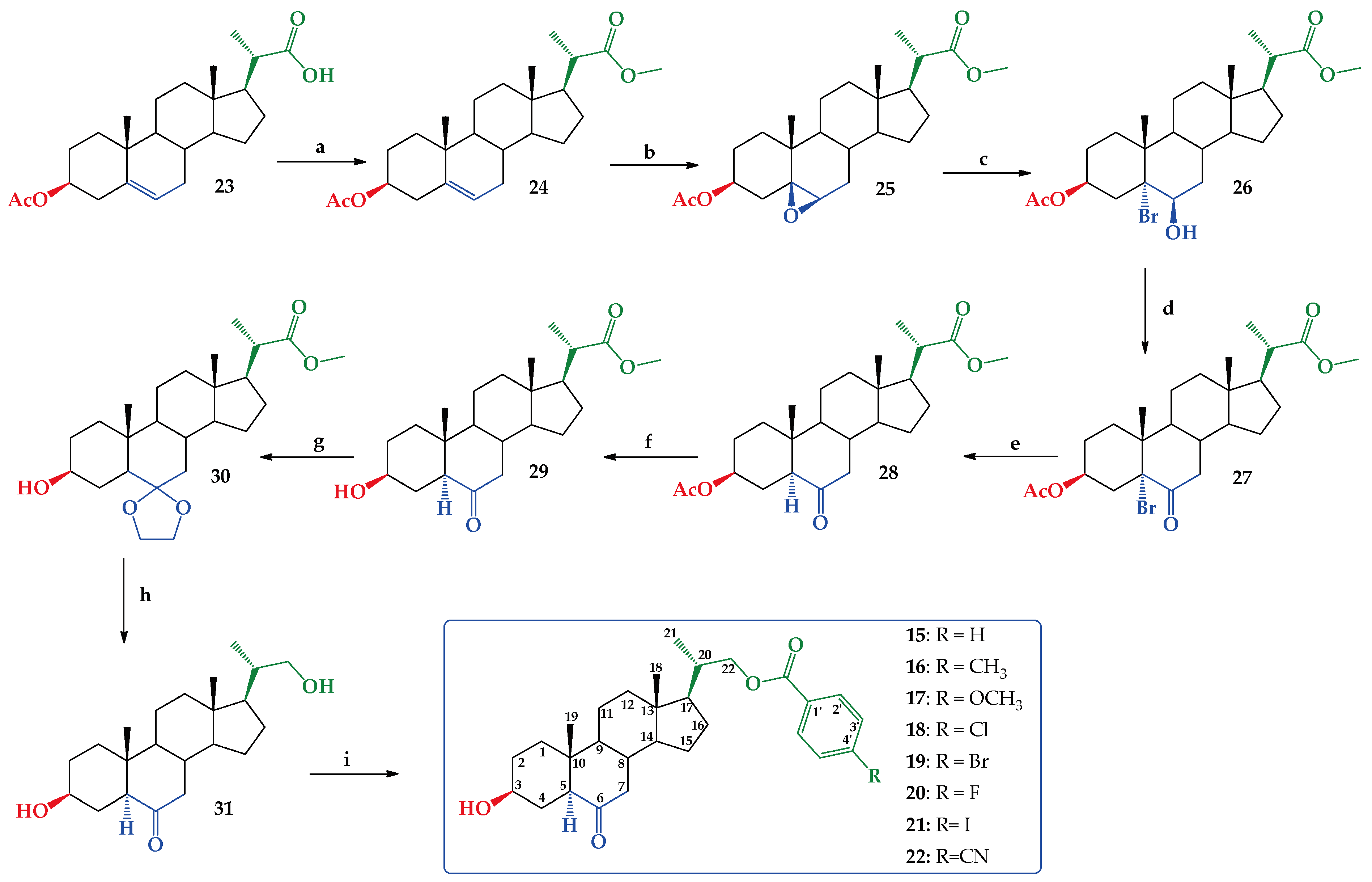
2.1. Chemical Synthesis
2.2. Biological Activity
2.2.1. Rice Lamina Inclination Test
2.2.2. Inhibition of Root and Hypocotyl Elongation in Arabidopsis thaliana Seedlings Assays
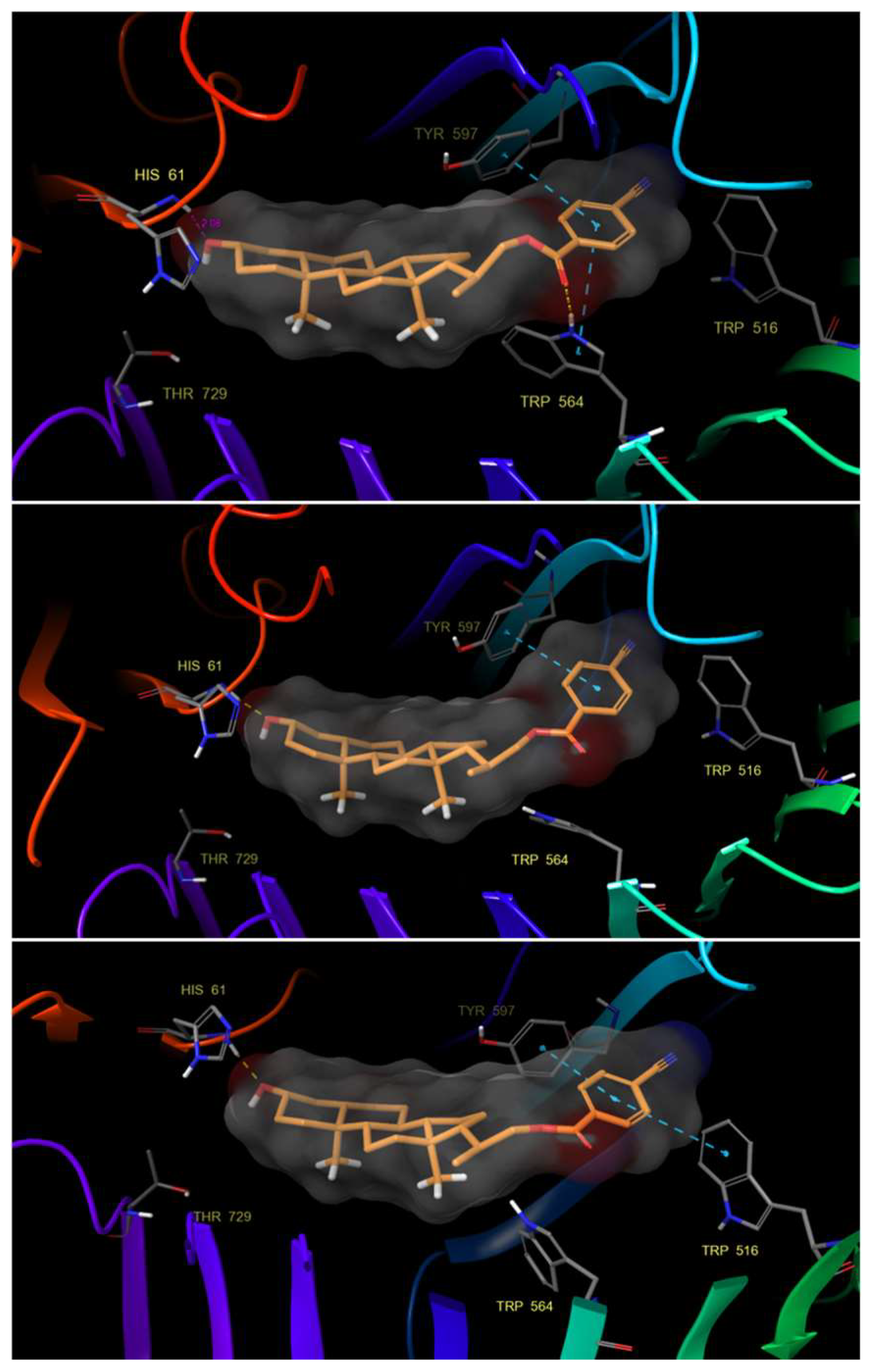
2.3. Molecular Docking and Induced Fit (IFD)
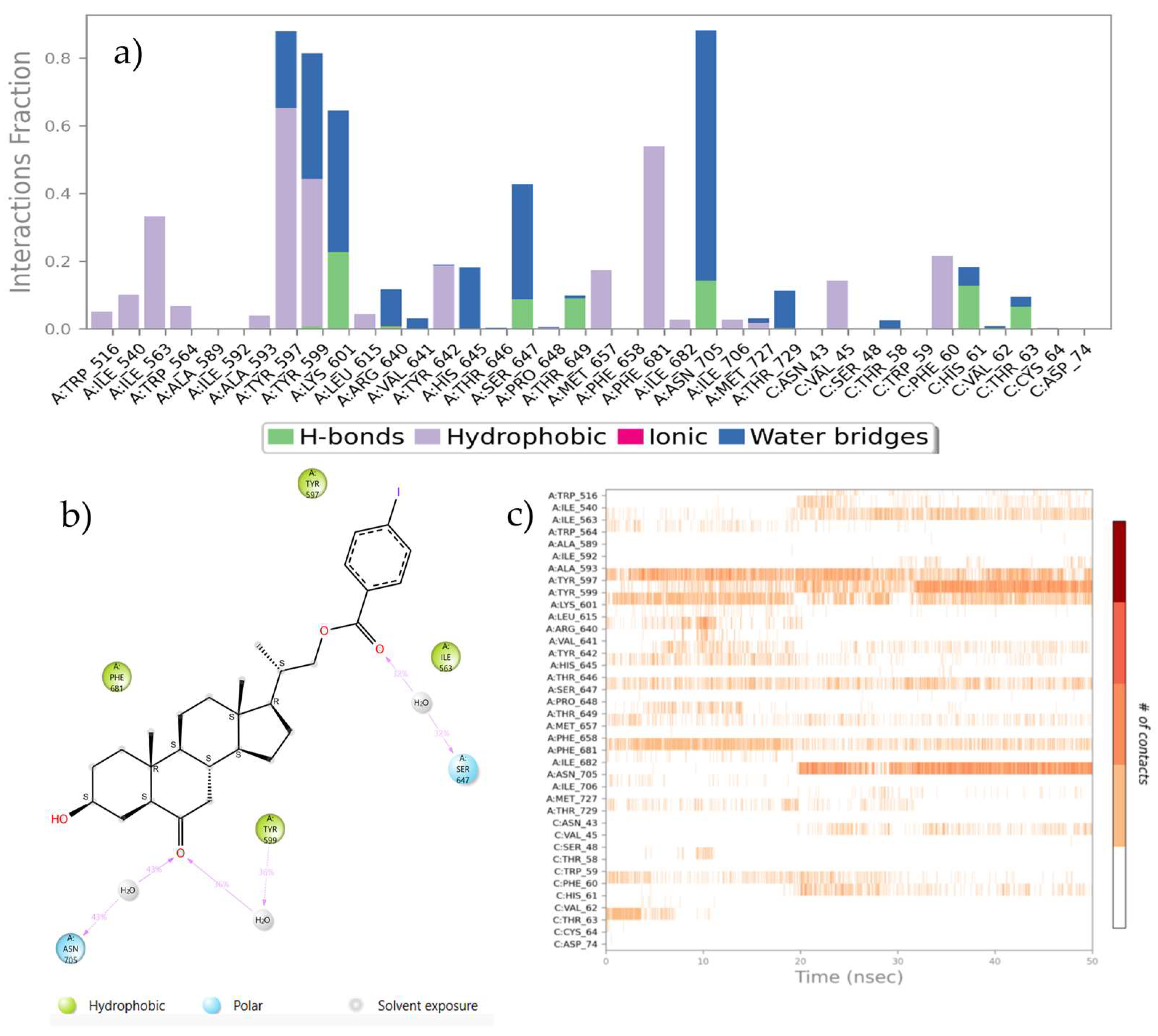
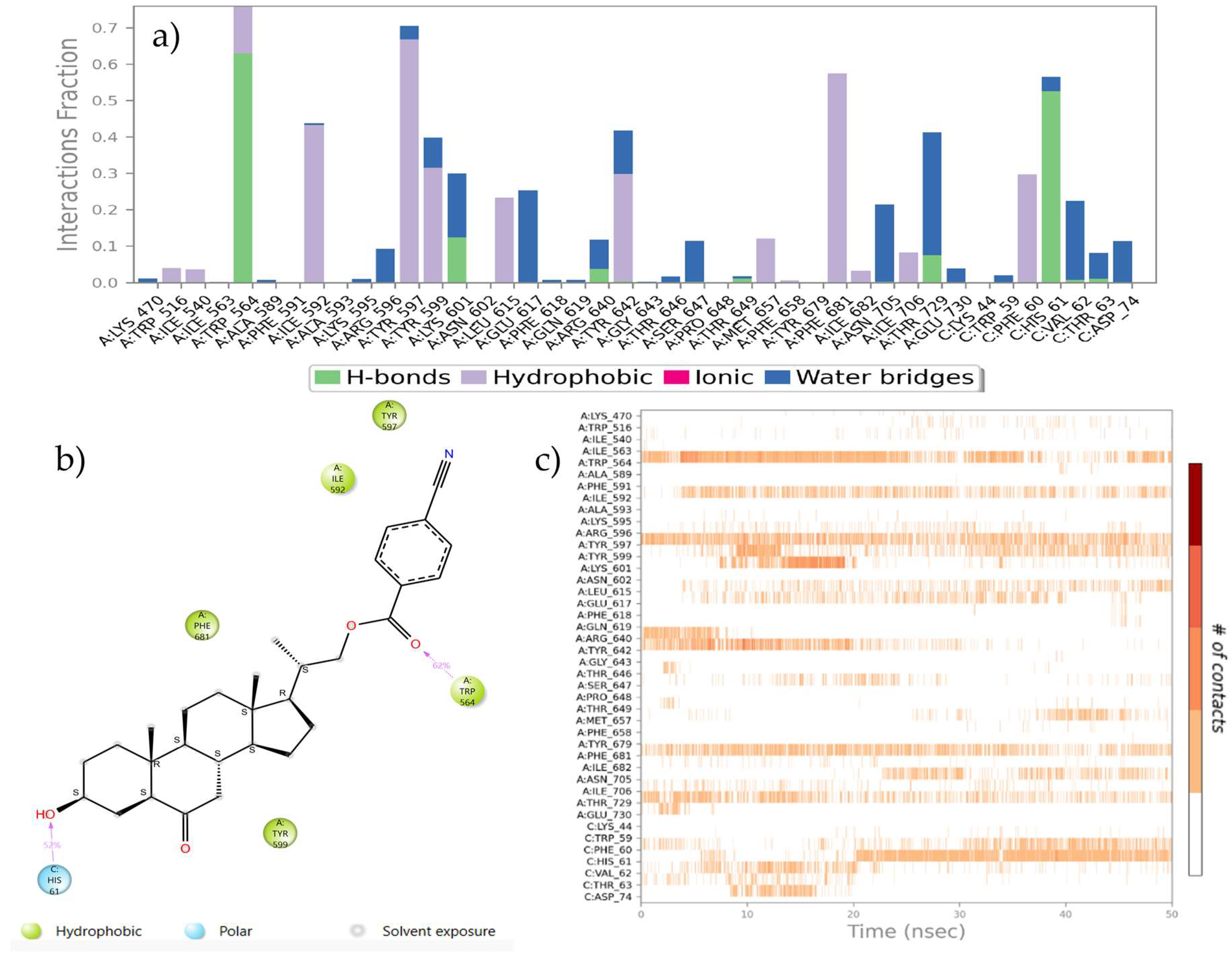
2.4. Molecular Dynamic Simulations
3. Materials and Methods
3.1. General
3.2. Synthesis
3.2.1. Methyl (20S)-3β-acetoxypregn-5-ene-20-carboxylate (24)
3.2.2. Methyl (20S)-3β-acetoxypregn-5β,6β-epoxy-20-carboxylate (25)
3.2.3. Methyl (20S)-3β-acetoxypregn-5α-bromo-6β-hydroxy-20-carboxylate (26)
3.2.4. Methyl (20S)-3β-acetoxypregn-5α-bromo-6-oxo-20-carboxylate (27)
3.2.5. Methyl (20S)-3β-acetoxy-5α-pregn-6-oxo-20-carboxylate (28)
3.2.6. Methyl (20S)-3β-hydroxy-5α-pregn-6-oxo-20-carboxylate (29)
3.2.7. Methyl (20S)-3β-hydroxy-5α-pregn-6-dioxolan-20-carboxylate (30)
3.2.8. 3β-22-dihydroxy-5α-cholan-23,24-dinor-6-oxa (31)
3.2.9. 3β-hydroxy-5α-cholan-6-oxo-23,24-dinor-22-(4-substituted)benzoate-22-yl (15–22)
3.3. Biological Activity
3.3.1. Rice Lamina Inclination Test (RLIT)
3.3.2. Inhibition of Root and Hypocotyl Elongation in Arabidopsis thaliana Seedlings
3.4. Computational Studies
3.4.1. Protein Preparation and Molecular Docking
3.4.2. Induced-Fit Docking
3.4.3. Molecular Dynamics Simulation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bajguz, A. Brassinosteroids—Occurence and chemical structures in plants. In Brassinosteroids: A Class of Plant Hormone; Hayat, S., Ahmad, A., Eds.; Springer: Dordrecht, The Netherlands, 2016; pp. 1–27. [Google Scholar]
- Oklestkova, J.; Rarova, L.; Kvasnica, M.; Strnad, M. Brassinosteroids: Synthesis and biological activities. Phytochem. Rev. 2015, 14, 1053–1072. [Google Scholar] [CrossRef]
- Bajguz, A.; Hayat, S. Effects of brassinosteroids on the plant responses to environmental stresses. Plant Physiol. Biochem. 2009, 47, 1–8. [Google Scholar] [CrossRef]
- Manghwar, H.; Hussain, A.; Ali, Q.; Liu, F. Brassinosteroids (BRs) Role in Plant Development and Coping with Different Stresses. Int. J. Mol. Sci. 2022, 23, 1012. [Google Scholar] [CrossRef]
- Temmem, O.; Uguen, D.; De Cian, A.; Gruber, N. Toward a total synthesis of brassinosteroids; structure assessment of the Ireland–Claisen products of geranyl and neryl esters. Tetrahedron Lett. 2002, 43, 3169–3173. [Google Scholar] [CrossRef]
- Temmem, O.; Zoller, T.; Uguen, D. Toward a total synthesis of brassinosteroids; stereoselective generation of the hydrindane ring system. Tetrahedron Lett. 2002, 43, 3181–3184. [Google Scholar] [CrossRef]
- Aburatani, M.; Takeuchi, T.; Mori, K. Facile Syntheses of Brassinosteroids: Brassinolide, Castasterone, Teasterone and Typhasterol. Agric. Biol. Chem. 1987, 51, 1909–1913. [Google Scholar] [CrossRef]
- Back, T.G.; Blazecka, P.G.; Krishna, M.V. A new synthesis of castasterone and brassinolide from stigmasterol—A concise and stereoselective elaboration of the side-chain from a C-22 aldehyde. Can. J. Chem. 1993, 71, 156–163. [Google Scholar] [CrossRef]
- Khripach, V.A.; Zhabinskii, V.N.; de Groot, A.E. Chapter VII—Syntheses of natural BS. In Brassinosteroids; Khripach, V.A., Zhabinskii, V.N., de Groot, A.E., Eds.; Academic Press: San Diego, CA, USA, 1999; pp. 137–184. [Google Scholar]
- Back, T.; Pharis, R. Structure-Activity Studies of Brassinosteroids and the Search for Novel Analogues and Mimetics with Improved Bioactivity. J. Plant Growth Regul. 2004, 22, 350–361. [Google Scholar] [CrossRef]
- Baron, D.L.; Luo, W.; Janzen, L.; Pharis, R.P.; Back, T.G. Structure–activity studies of brassinolide B-ring analogues. Phytochem. 1998, 49, 1849–1858. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, D.; Sun, X.; Ding, T.; Lei, B.; Zhang, C. Structure-activity relationship of brassinosteroids and their agricultural practical usages. Steroids 2017, 124, 1–17. [Google Scholar] [CrossRef]
- Tian, W.S.; Zhou, W.; Jiang, B.; Pan, X.F. Studies on steroidal plant-growth regulator IX: The preparation of 22R- and 22S-24, 25, 26, 27, 28-penta-nor-brassinolides. Acta Chim. Sinica 1989, 47, 1017–1021. [Google Scholar]
- Carvajal, R.; Gonzalez, C.; Olea, A.F.; Fuentealba, M.; Espinoza, L. Synthesis of 2-Deoxybrassinosteroids Analogs with 24-nor, 22(S)-23-Dihydroxy-Type Side Chains from Hyodeoxycholic Acid. Molecules 2018, 23, 1306. [Google Scholar] [CrossRef]
- Oyarce, J.; Aitken, V.; Gonzalez, C.; Ferrer, K.; Olea, A.F.; Parella, T.; Espinoza, L. Synthesis and structural determination of new brassinosteroid 24-nor-5α-cholane type analogs. Molecules 2019, 24, 4612. [Google Scholar] [CrossRef]
- Diaz, K.; Espinoza, L.; Carvajal, R.; Conde-Gonzalez, M.; Niebla, V.; Olea, A.F.; Coll, Y. Biological Activities and Molecular Docking of Brassinosteroids 24-Norcholane Type Analogs. Int. J. Mol. Sci. 2020, 21, 1832. [Google Scholar] [CrossRef]
- Soto, N.; Ferrer, K.; Díaz, K.; González, C.; Taborga, L.; Olea, A.F.; Carrasco, H.; Espinoza, L. Synthesis and Biological Activity of New Brassinosteroid Analogs of Type 24-Nor-5β-Cholane and 23-Benzoate Function in the Side Chain. Int. J. Mol. Sci. 2021, 22, 4808. [Google Scholar] [CrossRef]
- Ferrer, K.; Díaz, K.; Kvasnica, M.; Olea, A.F.; Cuellar, M.; Espinoza, L. Synthesis of New Brassinosteroid 24-Norcholane Type Analogs Conjugated in C-3 with Benzoate Groups. Molecules 2021, 26, 1173. [Google Scholar] [CrossRef]
- Voigt, B.; Schmidt, J.; Adam, G. Synthesis of 24-epiteasterone, 24-epityphasterol and their B-homo-6a-oxalactones from ergosterol. Tetrahedron 1996, 52, 1997–2004. [Google Scholar] [CrossRef]
- Brosa, C.; Capdevila, J.M.; Zamora, I. Brassinosteroids: A new way to define the structural requirements. Tetrahedron 1996, 52, 2435–2448. [Google Scholar] [CrossRef]
- Voigt, B.; Porzel, A.; Bruhn, C.; Wagner, C.; Merzweiler, K.; Adam, G. Synthesis of 24-epicathasterone and related brassinosteroids with modified side chain. Tetrahedron 1997, 53, 17039–17054. [Google Scholar] [CrossRef]
- Seto, H.; Fujioka, S.; Koshino, H.; Yoshida, S.; Tsubuki, M.; Honda, T. Synthesis and biological evaluation of extra-hydroxylated brassinolide analogs. Tetrahedron 1999, 55, 8341–8352. [Google Scholar] [CrossRef]
- Iglesias-Arteaga, M.; Gil, R.P.; Leliebre-Lara, V.; Martinez, C.S.P.; Manchado, F. Synthesis and biological activity of (22R,25R)-5 alpha-furostan-2 alpha,3 alpha,26-triol. J. Chem. Res. 1996, 28, 504–505. [Google Scholar]
- Iglesias-Arteaga, M.; Gil, R.; Leliebre-Lara, V.; Coll-Manchado, F.; Pérez, C.S.; Rosado, A. Synthesis of (25R)-5α-Spirostan-2α,3α,6β-triol Triacetate. Synth. Commun. 1998, 28, 75–81. [Google Scholar] [CrossRef]
- Iglesias-Arteaga, M.; Gil, R.P.; Leliebre-Lara, V.; Martinez, C.S.P.; Manchado, F. Synthesis of (22R,25R)-2 alpha,3 alpha,26-trihydroxy-5 alpha-furostanaone-6-one. Synth. Commun. 1998, 28, 1779–1784. [Google Scholar] [CrossRef]
- Iglesias-Arteaga, M.; Gil, R.P.; Leliebre-Lara, V.; Martinez, C.S.P.; Manchado, F.; Perez, A.R.; Rios, L.P. Synthesis of (22R,25R)-3 beta,26-dihydroxy-5 alpha-furostan-6-one. Synth. Commun. 1998, 28, 1381–1386. [Google Scholar] [CrossRef]
- Iglesias-Arteaga, M.A.; PérezGil, R.; LeliebreLara, V.; CollManchado, F.; PérezMartínez, C.S. Synthesis of (25R)-2α,3α-Epoxy-5α-Spirostan-6,23-Dione. Synth. Commun. 1998, 28, 4387–4392. [Google Scholar] [CrossRef]
- Iglesias-Arteaga, M.A.; Martinez, C.S.P.; Manchado, F.C. Synthesis and characterization of (25R)-2 alpha,3 alpha-epoxy-5 alpha-spirostan-12,23-dione. Synth. Commun. 1999, 29, 1811–1818. [Google Scholar] [CrossRef]
- Iglesias-Arteaga, M.A.; Gil, R.-P.; Pérez-Martínez, C.S.; Coll-Manchado, F. Synthetic Steroidal Sapogenins. Part III 23-Ketohecogenin and 23-Ketoisochiapagenin. Synth. Commun. 2000, 30, 163–170. [Google Scholar] [CrossRef]
- Zhou, W.; Jiang, B.; Shen, J. Synthesis of cholesteric lactones and analogs as plant growth regulators. Patent CN 1184113 A, 10 June 1998. [Google Scholar]
- Zhou, W.S.; Tian, W.S. The Synthesis of Steroids Containing Structural Unit of A, B Ring of Brassinolide and Ecdysone from Hyodeoxycholic Acid. Acta Chim. Sinica 1984, 42, 1173–1177. [Google Scholar]
- Kvasnica, M.; Oklestkova, J.; Bazgier, V.; Rarova, L.; Berka, K.; Strnad, M. Biological activities of new monohydroxylated brassinosteroid analogues with a carboxylic group in the side chain. Steroids 2014, 85, 58–64. [Google Scholar] [CrossRef]
- Duran, M.I.; Gonzalez, C.; Acosta, A.; Olea, A.F.; Diaz, K.; Espinoza, L. Synthesis of Five Known Brassinosteroid Analogs from Hyodeoxycholic Acid and Their Activities as Plant-Growth Regulators. Int. J. Mol. Sci. 2017, 18, 516. [Google Scholar] [CrossRef]
- Diachkov, M.V.; Ferrer, K.; Oklestkova, J.; Rarova, L.; Bazgier, V.; Kvasnica, M. Synthesis and Biological Activity of Brassinosteroid Analogues with a Nitrogen-Containing Side Chain. Int. J. Mol. Sci. 2021, 22, 155. [Google Scholar] [CrossRef]
- Huang, L.F.; Zhou, W.S. Studies on Steroidal Plant-Growth Regulators. Part 33. Novel Method for Construction of the Side-Chain of 23-Arylbrassinosteroids Via Heck Arylation and Asymmetric Dihydroxylation As Key Steps. J. Chem. Soc. Perkin Trans. 1 1994, 24, 3579–3585. [Google Scholar] [CrossRef]
- Korinkova, P.; Bazgier, V.; Oklestkova, J.; Rarova, L.; Strnad, M.; Kvasnica, M. Synthesis of novel aryl brassinosteroids through alkene cross-metathesis and preliminary biological study. Steroids 2017, 127, 46–55. [Google Scholar] [CrossRef]
- Aitken, V.; Diaz, K.; Soto, M.; Olea, A.F.; Cuellar, M.A.; Nuñez, M.; Espinoza-Catalán, L. New Brassinosteroid Analogs with 23,24-Dinorcholan Side Chain, and Benzoate Function at C-22: Synthesis, Assessment of Bioactivity on Plant Growth, and Molecular Docking Study. Int. J. Mol. Sci. 2024, 25, 419. [Google Scholar] [CrossRef]
- Kvasnica, M.; Oklestkova, J.; Bazgier, V.; Rárová, L.; Korinkova, P.; Mikulík, J.; Budesinsky, M.; Béres, T.; Berka, K.; Lu, Q.; et al. Design, synthesis and biological activities of new brassinosteroid analogues with a phenyl group in the side chain. Org. Biomol. Chem. 2016, 14, 8691–8701. [Google Scholar] [CrossRef]
- Kohout, L.J.; Chodounská, H.; Macek, T.; Strnad, M. Synthesis of (20S)-2α,3α-Dihydroxy-6-oxo-7-oxa-7a-homo-5α-pregnane-20-carboxylic Acid as a Brassinosteroid Part of Ligands for Binding to Affinity Chromatography Carriers. Collect. Czech. Chem. Commun. 2000, 65, 1754–1761. [Google Scholar] [CrossRef]
- Iglesias-Arteaga, M.A.; Símuta-Lopez, E.M.; Xochihua-Moreno, S.; Viñas-Bravo, O.; Montiel Smith, S.; Meza Reyes, S.; Sandoval-Ramírez, J. A Convenient Procedure for the Synthesis of 3β-Hydroxy-6-oxo-5α-steroids: Application to the Synthesis of Laxogenin. J. Mex. Chem. Soc. 2005, 49, 134–142. [Google Scholar]
- Rosado-Abon, A.; Romero-Avila, M.; Iglesias-Arteaga, M.A. An unexpected and useful E-ring oxidative cleavage in furostanes. ARKIVOC 2008, 2008, 274–281. [Google Scholar] [CrossRef]
- Hunter, A.C.; Priest, S.-M. An efficient one-pot synthesis generating 4-ene-3,6-dione functionalised steroids from steroidal 5-en-3β-ols using a modified Jones oxidation methodology. Steroids 2006, 71, 30–33. [Google Scholar] [CrossRef]
- Cimino, F.P.; Núñez, M.G.; Rosado-Abón, A.; Amesty, Á.; Estévez-Braun, A.; Díaz, K.; Espinoza, L.C.; Iglesias-Arteaga, M.A. Methyl esters of 23,24-Dinor-5α-cholan-22-oic acids as brassinosteroid Analogues. Synthesis, evaluation of plant growth promoting activity and Molecular docking. Steroids 2023, 196, 109248. [Google Scholar] [CrossRef]
- Antonchick, A.P.; Schneider, B.; Zhabinskii, V.N.; Khripach, V.A. Synthesis of [26,27-2H6]brassinosteroids from 23,24-bisnorcholenic acid methyl ester. Steroids 2004, 69, 617–628. [Google Scholar] [CrossRef]
- Yu, G.; Clive, D.L. Conversion of cycloalk-2-enones into 2-methylcycloalkane-1,3-diones--assessment of various Tamao-Fleming procedures and mechanistic insight into the use of the Me3SiMe2Si unit. Org. Biomol. Chem. 2016, 14, 1653–1664. [Google Scholar] [CrossRef]
- Wada, K.; Marumo, S.; Abe, H.; Morishita, T.; Nakamura, K.; Uchiyama, M.; Mori, K. A Rice Lamina Inclination Test—A Micro-Quantitative Bioassay for Brassinosteroids. Agric. Biol. Chem. 1984, 48, 719–726. [Google Scholar]
- Li, H.; Wang, H.; Jang, S. Rice Lamina Joint Inclination Assay. Bio-Protocol 2017, 7, e2409. [Google Scholar] [CrossRef]
- Wang, Y.-Q.; Luo, W.-H.; Xu, R.-J.; Zhao, Y.-J.; Zhou, W.-S.; Huang, L.-F.; Shen, J.-M. Biological Activity of Brassinosteroids and Relationship of Structure to Plant Growth Promoting Effects. Chin. Sci. Bull. 1994, 39, 1573–1577. [Google Scholar]
- Müssig, C.; Shin, G.-H.; Altmann, T. Brassinosteroids Promote Root Growth in Arabidopsis. Plant Physiol. 2003, 133, 1261–1271. [Google Scholar] [CrossRef]
- Planas-Riverola, A.; Gupta, A.; Betegón-Putze, I.; Bosch, N.; Ibañes, M.; Caño-Delgado, A.I. Brassinosteroid signaling in plant development and adaptation to stress. Development 2019, 146, dev151894. [Google Scholar] [CrossRef]
- Lv, B.; Tian, H.; Zhang, F.; Liu, J.; Lu, S.; Bai, M.; Li, C.; Ding, Z. Brassinosteroids regulate root growth by controlling reactive oxygen species homeostasis and dual effect on ethylene synthesis in Arabidopsis. PLoS Genet. 2018, 14, e1007144. [Google Scholar] [CrossRef]
- González-García, M.-P.; Vilarrasa-Blasi, J.; Zhiponova, M.; Divol, F.; Mora-García, S.; Russinova, E.; Caño-Delgado, A.I. Brassinosteroids control meristem size by promoting cell cycle progression in Arabidopsis roots. Development 2011, 138, 849. [Google Scholar] [CrossRef]
- Vukašinović, N.; Russinova, E. BRexit: Possible Brassinosteroid Export and Transport Routes. Trends Plant Sci. 2018, 23, 285–292. [Google Scholar] [CrossRef]
- Hola, D. Role of Brassinosteroids in the Plant Response to Drought: Do We Know Anything for Certain? In Brassinosteroids: Plant Growth and Development; Hayat, S., Yusuf, M., Bhardwaj, R., Bajguz, A., Eds.; Springer: Singapore, 2019; pp. 101–168. [Google Scholar]
- Yin, Y.; Wang, Z.-Y.; Mora-Garcia, S.; Li, J.; Yoshida, S.; Asami, T.; Chory, J. BES1 Accumulates in the Nucleus in Response to Brassinosteroids to Regulate Gene Expression and Promote Stem Elongation. Cell 2002, 109, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.; Kim, K.; Cho, H.; Hwang, I. Predominant Actions of Cytosolic BSU1 and Nuclear BIN2 Regulate Subcellular Localization of BES1 in Brassinosteroid Signaling. Mol. Cells 2010, 29, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Nam, K.H.; Li, J. BRI1/BAK1, a receptor kinase pair mediating brassinosteroid signaling. Cell 2002, 110, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Goshe, M.B.; Soderblom, E.J.; Phinney, B.S.; Kuchar, J.A.; Li, J.; Asami, T.; Yoshida, S.; Huber, S.C.; Clouse, S.D. Identification and functional analysis of in vivo phosphorylation sites of the Arabidopsis BRASSINOSTEROID-INSENSITIVE1 receptor kinase. Plant Cell 2005, 17, 1685–1703. [Google Scholar] [CrossRef]
- Clouse, S.D. Brassinosteroid signal transduction: From receptor kinase activation to transcriptional networks regulating plant development. Plant Cell 2011, 23, 1219–1230. [Google Scholar] [CrossRef]
- Kinoshita, T.; Caño-Delgado, A.; Seto, H.; Hiranuma, S.; Fujioka, S.; Yoshida, S.; Chory, J. Binding of brassinosteroids to the extracellular domain of plant receptor kinase BRI1. Nature 2005, 433, 167–171. [Google Scholar] [CrossRef]
- She, J.; Han, Z.; Kim, T.-W.; Wang, J.; Cheng, W.; Chang, J.; Shi, S.; Wang, J.; Yang, M.; Wang, Z.-Y.; et al. Structural insight into brassinosteroid perception by BRI1. Nature 2011, 474, 472. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jiang, J.; Wang, J.; Chen, L.; Fan, S.-L.; Wu, J.-W.; Wang, X.; Wang, Z.-X. Structural insights into the negative regulation of BRI1 signaling by BRI1-interacting protein BKI1. Cell Res. 2014, 24, 1328–1341. [Google Scholar] [CrossRef]
- Yang, C.-J.; Zhang, C.; Lu, Y.-N.; Jin, J.-Q.; Wang, X.-L. The Mechanisms of Brassinosteroids’ Action: From Signal Transduction to Plant Development. Mol. Plant 2011, 4, 588–600. [Google Scholar] [CrossRef]
- Hothorn, M.; Belkhadir, Y.; Dreux, M.; Dabi, T.; Noel, J.P.; Wilson, I.A.; Chory, J. Structural basis of steroid hormone perception by the receptor kinase BRI1. Nature 2011, 474, 467–471. [Google Scholar] [CrossRef]
- Sun, Y.; Han, Z.; Tang, J.; Hu, Z.; Chai, C.; Zhou, B.; Chai, J. Structure reveals that BAK1 as a co-receptor recognizes the BRI1-bound brassinolide. Cell Res. 2013, 23, 1326–1329. [Google Scholar] [CrossRef]
- Lei, B.; Liu, J.; Yao, X. Unveiling the molecular mechanism of brassinosteroids: Insights from structure-based molecular modeling studies. Steroids 2015, 104, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Fleming, F.F.; Yao, L.; Ravikumar, P.C.; Funk, L.; Shook, B.C. Nitrile-Containing Pharmaceuticals: Efficacious Roles of the Nitrile Pharmacophore. J. Med. Chem. 2010, 53, 7902–7917. [Google Scholar] [CrossRef] [PubMed]
- Friesner, R.A.; Murphy, R.B.; Repasky, M.P.; Frye, L.L.; Greenwood, J.R.; Halgren, T.A.; Sanschagrin, P.C.; Mainz, D.T. Extra Precision Glide: Docking and Scoring Incorporating a Model of Hydrophobic Enclosure for Protein−Ligand Complexes. J. Med. Chem. 2006, 49, 6177–6196. [Google Scholar] [CrossRef]
- Glide. Glide Software; Schrodinger, LLC: New York, NY, USA, 2022. [Google Scholar]
- Friesner, R.A.; Banks, J.L.; Murphy, R.B.; Halgren, T.A.; Klicic, J.J.; Mainz, D.T.; Repasky, M.P.; Knoll, E.H.; Shelley, M.; Perry, J.K.; et al. Glide: A New Approach for Rapid, Accurate Docking and Scoring. 1. Method and Assessment of Docking Accuracy. J. Med. Chem. 2004, 47, 1739–1749. [Google Scholar] [CrossRef]
- Sherman, W.; Beard, H.S.; Farid, R. Use of an induced fit receptor structure in virtual screening. Chem. Biol. Drug Des. 2006, 67, 83–84. [Google Scholar] [CrossRef]
- Lu, C.; Wu, C.; Ghoreishi, D.; Chen, W.; Wang, L.; Damm, W.; Ross, G.A.; Dahlgren, M.K.; Russell, E.; Von Bargen, C.D.; et al. OPLS4: Improving Force Field Accuracy on Challenging Regimes of Chemical Space. J. Chem. Theory Comput. 2021, 17, 4291–4300. [Google Scholar] [CrossRef]
- Jorgensen, W.L.; Chandrasekhar, J.; Madura, J.D.; Impey, R.W.; Klein, M.L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 1983, 79, 926–935. [Google Scholar] [CrossRef]
- Bowers, K.J.; Dror, R.O.; Shaw, D.E. The midpoint method for parallelization of particle simulations. J. Chem. Phys. 2006, 124, 184109. [Google Scholar] [CrossRef]
- Gibson, D.A.; Carter, E.A. Time-reversible multiple time scale ab initio molecular dynamics. J. Phys. Chem. 1993, 97, 13429–13434. [Google Scholar] [CrossRef]




| Bending Angle between Laminae and Sheaths (°) (Degrees ± Standard Error) 1 | |||
|---|---|---|---|
| Compounds | 1 × 10−8 M | 1 × 10−7 M | 1 × 10−6 M |
| 1 | 22 ± 3.5 b | 31 ± 2.6 a | 71 ± 4.5 a |
| 15 | 21 ± 1.0 b | 18 ± 2.7 b | 24 ± 1.2 b |
| 16 | 23 ± 2.7 b | 10 ± 1.2 c | 18 ± 2.6 c |
| 17 | 33 ± 2.6 a | 15 ± 2.0 b | 5 ± 2.0 e |
| 18 | 12 ± 2.0 d | 19 ± 2.6 b | 9 ± 2.1 d |
| 19 | 15 ± 2.0 c | 19 ± 2.6 b | 3 ± 2.7 e |
| 20 | 16 ± 2.0 c | 21 ± 3.2 b | 20 ± 2.0 c |
| 21 | 38 ± 2.5 a | 24 ± 2.0 b | 6 ± 1.8 e |
| 22 | 37 ± 2.0 a | 9 ± 2.0 c | 18 ± 1.0 c |
| 31 | 21 ± 1.2 b | 14 ± 2.8 b | 4 ± 2.4 e |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nuñez, M.; Wang, Y.; Russinova, E.; Estévez-Braun, A.; Amesty, A.; Olea, A.F.; Mellado, M.; Díaz, K.; Espinoza Catalán, L. Synthesis, Biological Activity, and Molecular-Docking Studies of New Brassinosteroid Analogs. Int. J. Mol. Sci. 2024, 25, 10158. https://doi.org/10.3390/ijms251810158
Nuñez M, Wang Y, Russinova E, Estévez-Braun A, Amesty A, Olea AF, Mellado M, Díaz K, Espinoza Catalán L. Synthesis, Biological Activity, and Molecular-Docking Studies of New Brassinosteroid Analogs. International Journal of Molecular Sciences. 2024; 25(18):10158. https://doi.org/10.3390/ijms251810158
Chicago/Turabian StyleNuñez, María, Yaowei Wang, Eugenia Russinova, Ana Estévez-Braun, Angel Amesty, Andrés F. Olea, Marco Mellado, Katy Díaz, and Luis Espinoza Catalán. 2024. "Synthesis, Biological Activity, and Molecular-Docking Studies of New Brassinosteroid Analogs" International Journal of Molecular Sciences 25, no. 18: 10158. https://doi.org/10.3390/ijms251810158
APA StyleNuñez, M., Wang, Y., Russinova, E., Estévez-Braun, A., Amesty, A., Olea, A. F., Mellado, M., Díaz, K., & Espinoza Catalán, L. (2024). Synthesis, Biological Activity, and Molecular-Docking Studies of New Brassinosteroid Analogs. International Journal of Molecular Sciences, 25(18), 10158. https://doi.org/10.3390/ijms251810158








