Possible Combinatorial Utilization of Phytochemicals and Extracellular Vesicles for Wound Healing and Regeneration
Abstract
1. Introduction
2. Wound Healing after Traumatic Injury, Surgery as a Controlled Injury, and Injury Arising from Infectious Diseases
2.1. Wound Healing after Traumatic Injury and Surgery
2.2. Damage by Infectious Diseases—An Example of Olfactory Dysfunction by COVID-19
3. Phytochemicals with Bioactive Properties: Suppression of Inflammation and Enhancement of Wound Healing
3.1. Beta-Caryophyllene
3.2. Other Phytochemicals
| Action | Targets: Receptors/Channels/Other Targets | Phytochemical Compound | Triggers/Function/Reference |
|---|---|---|---|
| Activation | Cannabinoid receptor 2 (CB2) | cannabigerol, β-caryophyllene, citral (those that activate CB1 as well are excluded) | Activation of CB2 has anti-inflammatory, analgesic, and anxiolytic effects (in vivo, in vitro, and reviews [36,122,123,124]) |
| Transient receptor potential channel ankyrin 1 (TRPA1) | Allicin, allyl isothiocyanate, bradykinin, cannabigerol, carvacrol, cinnamaldehyde, citral, curcumin, diallyldisulfide, eugenol, ligustilide, D-limonene, linalool, linalyl acetate, osthole, methylsalicylate, paclitaxel, THC, and others | Thermosensation, mechanosensation, chemosensation, pain, inflammation [125]; activated by multiple different mechanisms [126]; activation by phytochemicals induces changes in hormone secretion, neuropeptide/neurotransmitter release [127] (reviews) | |
| Transient receptor potential channel melastatin 8 (TRPM8) | L-Carvone, 1,8-cineole (eucalyptol), citral (geranial), eugenol, geraniol, hydroxyl-citronellal, icilin, isopulegol, linalool, 1-menthol, menthyl lactate, and others | Activated by cold [126], voltage, pressure, cooling compounds (menthol, icilin), hyperosmolarity [127,128] (reviews) | |
| Transient receptor potential channel vanilloid 1 (TRPV1) | Allicin, β-caryophyllene, camphor, cannabidiol, cannabigerol, capsaicin, carvacrol, citral, eugenol, evodiamine, gingerol, 1-menthol, piperine, resiniferatoxin, thymol, vanillin, and others | Activated by heat, suppresses pain [127] (review) | |
| Transient receptor potential channel vanilloid 3 (TRPV3) | Borneol, camphor, carvacrol, carveol, eugenol, thymol, and others | Warnth sensor, skin sensitization [127]; analgesic (review) | |
| Peroxisome proliferator-activated receptor (PPAR) | Apigenin, berberine, β-caryophyllene, cannabigerol, carnosic acid, carvacrol, catechins, curcumin, eugenol, hesperetin, isorhamnetin, kaempferol, naringenin, phloroglucinol, phlorotannins, procyanidin, quercetin, resveratrol, rutin, shogaol, terpinen-4-ol, and others | Regulation of energy homeostasis, insulin sensitization, glucose metabolism, and fatty acid metabolism [129,130]. Antioxidant, anti-inflammatory, anti-obesity, and other effects (reviews) | |
| GABA/5-HT | Apigenin, carvacrol, citral, geraniol, kaempferol, d-limonene, linalool, linalyl acetate, luteolin, methyl eugenol, obovatol, oleanolic acid, querecetin, rosmarinic acid, rutin, santin, saponins, tannins, terpinen-4-ol | Acts as inhibitory neurotransmitter and reduces excitability. Phytochemicals in jujuba seeds regulate GABA and 5-HT receptors to exert their anxiolytic effects [131]. A review [132] | |
| ERα | Apigenin, coumestrol, genistein, liquiritigenin, resveratrol | Phytochemicals with binding affinity with ERα receptor (phytoestrogen) produce estrogenic effects [133]. A review [134] | |
| Adenosine A2A | D-limonene [102] | Adenosine A2A regulates, for example, immune responses cardiovascular function, sleep regulation, and others [135]. A review [102] | |
| 67 kDa laminin receptor (67LR) | EGCG [91,136,137] | Involved in cell adhesion, migration, proliferation, and survival. A review [136] | |
| Aryl hydrocarbon receptor (AhR) | Curcumin potential ligand [70] | Detoxication of xenobiotic compounds [138]. In vitro sudy using rat astrocytes [70] | |
| GPR55 | Curcumin [87] | Increases intracellular calcium. In vitro study [87] | |
| Suppression | Kv (potassium channels) (removed the specific type) | Citronellol, citral, EGCG, geraniol, linalool, loureirin B, luteolin, naringenin, quercetin, resveratrol | Involved in cell proliferation, hormone secretion, neurotransmitter release, and others (a review and in vitro study [139,140]) |
| Cav (calcium channels) (removed the specific type) | α-besabolol, betulinic acid, camphene, cannabidiol, curcumin, eugenol, gingerol, shogaol, linalool, quercetin | Review [141] | |
| Nav (sodium channels) (removed the specific type) | Gingerol, imperatorin, lappaconitine, methyl eugenol, narirutin, peimine, shogaol | Activation of neuronal signaling related to the perception of pain (reviews and an in vivo study [141,142,143]) | |
| Nuclear factor kappa-light-chain-enhancer of activated B (NF-kB) | Apigenin, caffeic acid, curcumin [88], EGCG, gallic acid, genistein, 6-gingerol, quercetin, resveratrol | Reviews [144,145] | |
| Toll-like receptors (TLR) | Curcumin, EGCG, helenalin, cinnamaldehyde, sulforaphane, but not resveratrol | Detect pathogens and activate proinflammatory pathways to eliminate pathogens; overactivation is involved in inflammatory diseases (reviews and an in vitro study [144,146,147,148,149,150]) | |
| Tank binding kinaze 1 (TBK1) | Resveratrol, EGCG, luteolin, quercetin, chrysin, eriodictyol [146] | Inducer of type 1 interferons and involved in innate immunity signaling (a review [151]) | |
| Nucleotide-binding oligomerization domain proteins (NOD) 2 | Curcumin, parthenolide, helanalin, but not resveratrol and EGCG | Detect pathogens and activate proinflammatory pathways (a review [146]) | |
| Tumor necrosis factor α (TNFα) | Capsaicin, curcumin, EGCG, kaempferol, naringenin, piperine, quercetin resveratrol, retinoic acid, rosmarinic acid, rutin, 6-shogaol, theaflavin [152] | Reviews [152,153] | |
| Rat sarcoma (Ras) | Oridonin, perillyl alcohol | GTP-binding protein which stimulates various vital cellular processes such as cell proliferation and survival, differentiation, and others Activation triggers a pathway of RAS -> RAF -> MEK -> ERK-1/2 (review [154]) | |
| Mitogen-activated protein kinase (MAPK) | Caffeic acid, EGCG, kaempferol, magnolol, perillyl alcohol, quercetin, resveratrol, ursolic acid | Review [155] | |
| Adenosine A2A | Caffeine [156], flavonoids such as galangin [157] |
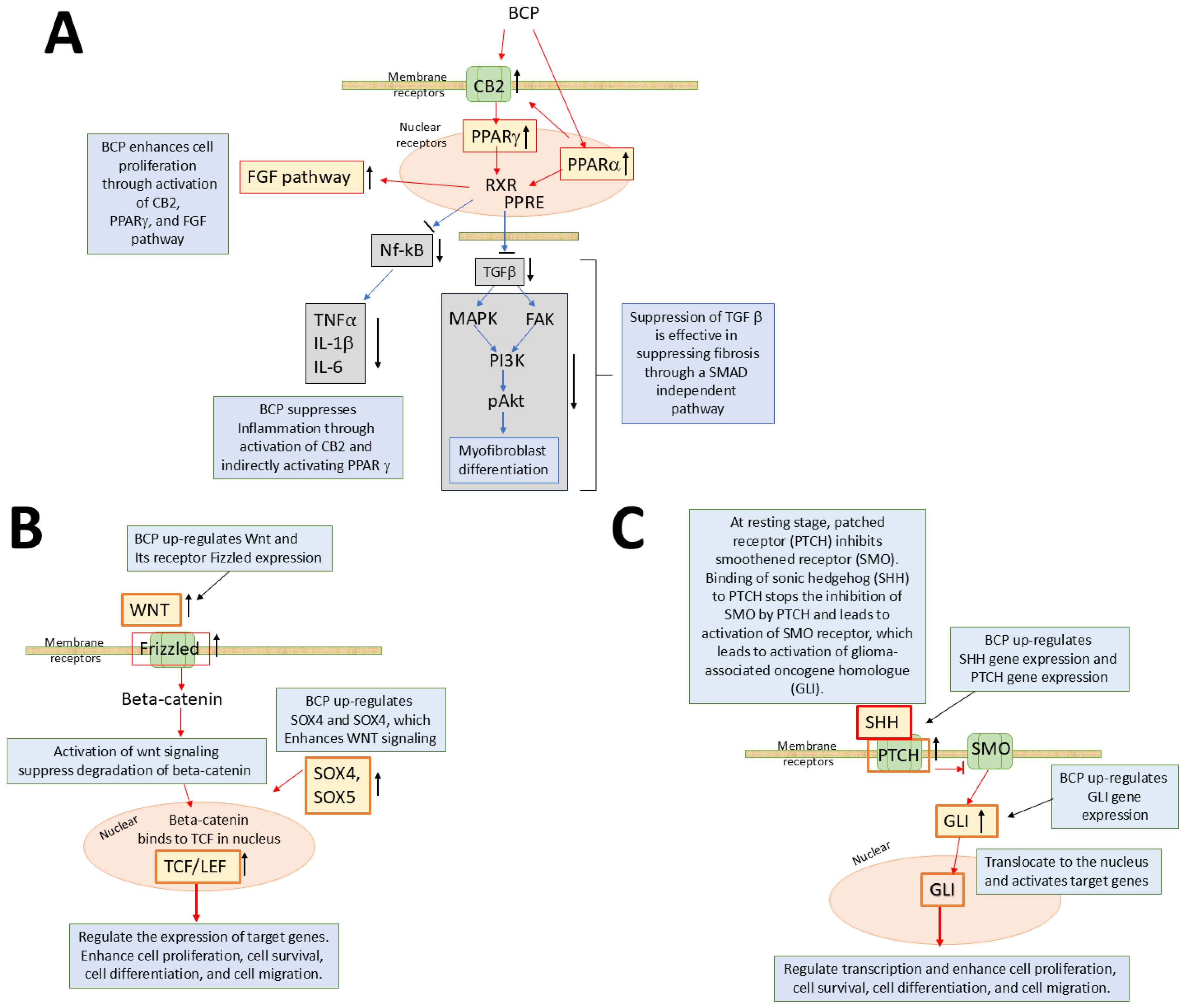
3.3. Combinatorial Use of Phytochemicals
4. Extracellular Vesicles (EVs) for Wound Healing
4.1. Biology of EVs
| Source | Effects Found | References | |
|---|---|---|---|
| Mammal/Plant | Cell/Tissue/Organ | ||
| Mammal | Cell (MSC) | Promoted corneal epithelial cell proliferation, migration in vitro, and wound healing in vivo; umbilical cord MSC | [179] |
| Cell fragments (Platelet) | Corneal endothelial cells showed increased viability and enhanced wound healing, adhering rate, and proliferation markers when exposed to EV from platelets | [178] | |
| Cell (MSC) | EV enhanced in vivo cutaneous wound healing and suppressed apoptosis; adipose-derived MSC | [180] | |
| Cell (MSC) | EV enhanced cell proliferation and migration of human corneal epithelial cells, p44/42 MAPK pathway was activated; they suppressed inflammation and suppressed upregulation of α-SMA (fibrosis) | [10] | |
| Cell (MSC) | Review; accelerated wound-healing process; suppressed inflammation; promoted vascularization, cell proliferation, and migration | [208] | |
| Cell (MSC) | Review; improved axon extension, apoptosis inhibition, Schwann cell proliferation, and neuroregeneration in nerve regeneration | [185] | |
| Cell (MSC) | Review; improved axon extension, promoted axon regeneration by delivering neurotrophic factors, suppressed neuroinflammation, mediated vascular regeneration | [184] | |
| Cell (ASC) | ASC-derived EV loaded in thermosensitive hydrogel enhanced Schwann cell migration and proliferation, as well as axon extension in in vitro and nerve repair in vivo (rat) | [187] | |
| Cell (ASC) | ASC-derived EV loaded on collagen sheet suppressed early inflammatory response and post-repair tendon gap and facilitated collagen formation | [188] | |
| Cell (Sch) | EV from Schwann cells that received mechanical stimulation by exposure to magnetic field had stronger influences of enhancing axonal growth in vitro and nerve regeneration in vivo than EV from Schwann cells without mechanical stimulation | [190] | |
| Cell (Muscle) | EV from muscle cell line enhanced neurite growth and survival | [209] | |
| Tissue (Muscle) | EV from denervated muscle improved the motor neuron’s ability to correctly project back to the original terminal muscle branch, showing enhanced regeneration accuracy | [191] | |
| Plant | Tea leaf | Suppressed breast tumor cell cycle by increasing ROS, triggering mitochondria damage and causing apoptosis in tumor cells | [199] |
| Review | Review on the effects of plant-derived vesicles on cancer and inflammation | [196] | |
| Lemon | EV from lemon juice suppressed tumor cell proliferation | [201] | |
| Ginseng | EV from ginseng increased apoptosis in mouse melanoma cells | [202] | |
| Cannabis | EV from cannabis suppressed cell proliferation and increased apoptosis, suppressing tumor growth | [200] | |
| Ginger | EV from ginger suppressed acute colitis, facilitated intestinal recovery, and suppressed colitis-related cancer. EV contained a large amount of 6-gingerol and 6-shogaol | [203] | |
| Grapefruit | EV from grapefruit suppressed dextran-sulfate sodium (DSS)-induced colitis in mice | [204] | |
4.2. The Role of EVs as Drug Carrier
4.3. The Possibility of Utilizing EVs in Drug Delivery
5. Limitations
| Pros | Cons | |
|---|---|---|
| Phytochemicals |
|
|
| EVs |
|
|
6. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Granholm, A.-C. Long-Term Effects of SARS-CoV-2 in the Brain: Clinical Consequences and Molecular Mechanisms. J. Clin. Med. 2023, 12, 3190. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Garcia, P.; Fiorillo Moreno, O.; Zarate Peñata, E.; Calderon-Villalba, A.; Pacheco Lugo, L.; Acosta Hoyos, A.; Villarreal Camacho, J.L.; Navarro Quiroz, R.; Pacheco Londoño, L.; Aroca Martinez, G.; et al. From Cell to Symptoms: The Role of SARS-CoV-2 Cytopathic Effects in the Pathogenesis of COVID-19 and Long COVID. Int. J. Mol. Sci. 2023, 24, 8290. [Google Scholar] [CrossRef] [PubMed]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding Light on the Cell Biology of Extracellular Vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Raposo, G.; Théry, C. Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu. Rev. Cell Dev. Biol. 2014, 30, 255–289. [Google Scholar] [CrossRef] [PubMed]
- Avalos, P.N.; Forsthoefel, D.J. An Emerging Frontier in Intercellular Communication: Extracellular Vesicles in Regeneration. Front. Cell Dev. Biol. 2022, 10, 849905. [Google Scholar] [CrossRef]
- Bazzan, E.; Tinè, M.; Casara, A.; Biondini, D.; Semenzato, U.; Cocconcelli, E.; Balestro, E.; Damin, M.; Radu, C.M.; Turato, G.; et al. Critical Review of the Evolution of Extracellular Vesicles’ Knowledge: From 1946 to Today. Int. J. Mol. Sci. 2021, 22, 6417. [Google Scholar] [CrossRef]
- Kalra, H.; Drummen, G.P.C.; Mathivanan, S. Focus on Extracellular Vesicles: Introducing the Next Small Big Thing. Int. J. Mol. Sci. 2016, 17, 170. [Google Scholar] [CrossRef]
- Miron, R.J.; Zhang, Y. Understanding Exosomes: Part 1—Characterization, Quantification and Isolation Techniques. Periodontology 2000 2024, 94, 231–256. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, Z.; Li, S.; Xie, X.; Qin, L.; Zhang, Q.; Yang, Y.; Wang, T.; Zhang, Y. Exosomes: Compositions, Biogenesis, and Mechanisms in Diabetic Wound Healing. J. Nanobiotechnology 2024, 22, 398. [Google Scholar] [CrossRef]
- Zhou, J.; Ding, Y.; Zhang, Y.; Zheng, D.; Yan, L.; Guo, M.; Mao, Y.; Yang, L. Exosomes from Bone Marrow-Derived Mesenchymal Stem Cells Facilitate Corneal Wound Healing via Regulating the P44/42 MAPK Pathway. Graefes Arch. Clin. Exp. Ophthalmol. 2023, 261, 723–734. [Google Scholar] [CrossRef]
- Hercher, D.; Nguyen, M.Q.; Dworak, H. Extracellular Vesicles and Their Role in Peripheral Nerve Regeneration. Exp. Neurol. 2022, 350, 113968. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Li, Z.; Wang, Y.; Zhou, K.; Li, H.; Bi, S.; Wang, Y.; Wu, W.; Huang, Y.; Peng, B.; et al. Bioengineered MSC-Derived Exosomes in Skin Wound Repair and Regeneration. Front. Cell Dev. Biol. 2023, 11, 1029671. [Google Scholar] [CrossRef]
- Li, K.; Yan, G.; Huang, H.; Zheng, M.; Ma, K.; Cui, X.; Lu, D.; Zheng, L.; Zhu, B.; Cheng, J.; et al. Anti-Inflammatory and Immunomodulatory Effects of the Extracellular Vesicles Derived from Human Umbilical Cord Mesenchymal Stem Cells on Osteoarthritis via M2 Macrophages. J. Nanobiotechnology 2022, 20, 38. [Google Scholar] [CrossRef]
- Miron, R.J.; Estrin, N.E.; Sculean, A.; Zhang, Y. Understanding Exosomes: Part 2—Emerging Leaders in Regenerative Medicine. Periodontology 2000 2024, 94, 257–414. [Google Scholar] [CrossRef] [PubMed]
- Valentino, A.; Di Cristo, F.; Bosetti, M.; Amaghnouje, A.; Bousta, D.; Conte, R.; Calarco, A. Bioactivity and Delivery Strategies of Phytochemical Compounds in Bone Tissue Regeneration. Appl. Sci. 2021, 11, 5122. [Google Scholar] [CrossRef]
- Kumar, G.P.; Khanum, F. Neuroprotective Potential of Phytochemicals. Pharmacogn. Rev. 2012, 6, 81–90. [Google Scholar] [CrossRef]
- Koyama, S.; Purk, A.; Kaur, M.; Soini, H.A.; Novotny, M.V.; Davis, K.; Kao, C.C.; Matsunami, H.; Mescher, A. Beta-Caryophyllene Enhances Wound Healing through Multiple Routes. PLoS ONE 2019, 14, e0216104. [Google Scholar] [CrossRef]
- Koyama, S.; Kondo, K.; Ueha, R.; Kashiwadani, H.; Heinbockel, T. Possible Use of Phytochemicals for Recovery from COVID-19-Induced Anosmia and Ageusia. Int. J. Mol. Sci. 2021, 22, 8912. [Google Scholar] [CrossRef]
- Koyama, S.; Heinbockel, T. The Effects of Essential Oils and Terpenes in Relation to Their Routes of Intake and Application. Int. J. Mol. Sci. 2020, 21, 1558. [Google Scholar] [CrossRef]
- Nag, A.; Banerjee, R.; Chowdhury, R.R.; Krishnapura Venkatesh, C. Phytochemicals as Potential Drug Candidates for Targeting SARS CoV 2 Proteins, an in Silico Study. Virusdisease 2021, 32, 98–107. [Google Scholar] [CrossRef]
- Chinnadurai, R.K.; Ponne, S.; Chitra, L.; Kumar, R.; Thayumanavan, P.; Subramanian, B. Pharmacoinformatic Approach to Identify Potential Phytochemicals against SARS-CoV-2 Spike Receptor-Binding Domain in Native and Variants of Concern. Mol. Divers. 2022, 27, 2741–2766. [Google Scholar] [CrossRef] [PubMed]
- Moretti, L.; Stalfort, J.; Barker, T.H.; Abebayehu, D. The Interplay of Fibroblasts, the Extracellular Matrix, and Inflammation in Scar Formation. J. Biol. Chem. 2022, 298, 101530. [Google Scholar] [CrossRef] [PubMed]
- Witzel, C.; Rohde, C.; Brushart, T.M. Pathway Sampling by Regenerating Peripheral Axons. J. Comp. Neurol. 2005, 485, 183–190. [Google Scholar] [CrossRef]
- Czarnecki, P.; Górecki, M.; Romanowski, L. Factors Affecting the Final Outcomes after Reconstruction of the Median and Ulnar Nerve at the Level of the Forearm: Analysis of 41 Patients. Injury 2020, 51, 2910–2915. [Google Scholar] [CrossRef]
- Koyama, S.; Mori, E.; Ueha, R. Insight into the Mechanisms of Olfactory Dysfunction by COVID-19. Auris Nasus Larynx 2023, 50, 490–498. [Google Scholar] [CrossRef]
- Frere, J.J.; Serafini, R.A.; Pryce, K.D.; Zazhytska, M.; Oishi, K.; Golynker, I.; Panis, M.; Zimering, J.; Horiuchi, S.; Hoagland, D.A.; et al. SARS-CoV-2 Infection in Hamsters and Humans Results in Lasting and Unique Systemic Perturbations Post Recovery. Sci. Transl. Med. 2022, 14, eabq3059. [Google Scholar] [CrossRef]
- Brann, D.H.; Tsukahara, T.; Wenreb, C.; Lipovsek, M.; van den Berge, K.; Gong, B.; Chance, R.; Macaulay, I.C.; Chou, H.-J.; Fletcher, R.B.; et al. Non-Neuronal Expression of SARS-CoV-2 Entry Genes in the Olfactory System Suggests Mechanisms Underlying COVID-19-Associated Anosmia. Sci. Adv. 2020, 6, eabc5801. [Google Scholar] [CrossRef]
- Chen, M.; Shen, W.; Rowan, N.R.; Kulaga, H.; Hillel, A.; Ramanathan, M.; Lane, A.P. Elevated ACE2 Expression in the Olfactory Neuroepithelium: Implications for Anosmia and Upper Respiratory SARS-CoV-2 Entry and Replication. Eur. Respir. J. 2020, 56, 2001948. [Google Scholar] [CrossRef]
- Ueha, R.; Kondo, K.; Kagoya, R.; Shichino, S.; Ueha, S.; Yamasoba, T. ACE2, TMPRSS2, and Furin Expression in the Nose and Olfactory Bulb in Mice and Human. Rhinol. J. 2020, 59, 105–109. [Google Scholar] [CrossRef]
- de Melo, G.D.; Lazarini, F.; Levallois, S.; Hautefort, C.; Michel, V.; Larrous, F.; Verillaud, B.; Aparicio, C.; Wagner, S.; Gheusi, G.; et al. COVID-19-Related Anosmia Is Associated with Viral Persistence and Inflammation in Human Olfactory Epithelium and Brain Infection in Hamsters. Sci. Transl. Med. 2021, 13, eabf8396. [Google Scholar] [CrossRef]
- Song, E.; Zhang, C.; Israelow, B.; Lu-Culligan, A.; Prado, A.V.; Skriabine, S.; Lu, P.; Weizman, O.-E.; Liu, F.; Dai, Y.; et al. Neuroinvasion of SARS-CoV-2 in Human and Mouse Brain. J. Exp. Med. 2021, 218, e20202135. [Google Scholar] [CrossRef] [PubMed]
- Buchrieser, J.; Dufloo, J.; Hubert, M.; Monel, B.; Planas, D.; Rajah, M.M.; Planchais, C.; Porrot, F.; Guivel-Benhassine, F.; Van der Werf, S.; et al. Syncytia Formation by SARS-CoV-2-infected Cells. EMBO J. 2020, 39, e106267. [Google Scholar] [CrossRef]
- Hernandez-Clavijo, A.; Gonzalez-Velandia, K.Y.; Rangaswamy, U.; Guarneri, G.; Boscolo-Rizzo, P.; Tofanelli, M.; Gardenal, N.; Sanges, R.; Dibattista, M.; Tirelli, G.; et al. Supporting Cells of the Human Olfactory Epithelium Co-Express the Lipid Scramblase TMEM16F and ACE2 and May Cause Smell Loss by SARS-CoV-2 Spike-Induced Syncytia. Cell Physiol. Biochem. 2022, 56, 254–269. [Google Scholar] [CrossRef] [PubMed]
- Shelton, J.F.; Shastri, A.J.; Fletez-Brant, K.; 23andMe COVID-19 Team; Aslibekyan, S.; Auton, A. The UGT2A1/UGT2A2 Locus Is Associated with COVID-19-Related Loss of Smell or Taste. Nat. Genet. 2022, 54, 121–124. [Google Scholar] [CrossRef]
- Chelu, M.; Musuc, A.M.; Popa, M.; Calderon Moreno, J. Aloe Vera-Based Hydrogels for Wound Healing: Properties and Therapeutic Effects. Gels 2023, 9, 539. [Google Scholar] [CrossRef]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Chen, J.-Z.; Xie, X.-Q.; Altmann, K.-H.; Karsak, M.; Zimmer, A. Beta-Caryophyllene Is a Dietary Cannabinoid. Proc. Natl. Acad. Sci. USA 2008, 105, 9099–9104. [Google Scholar] [CrossRef]
- Graham, E.S.; Ashton, J.C.; Glass, M. Cannabinoid Receptors: A Brief History and What Not. Front. Biosci. Landmark Ed. 2009, 14, 944–957. [Google Scholar] [CrossRef]
- Mackie, K.; Stella, N. Cannabinoid Receptors and Endocannabinoids: Evidence for New Players. AAPS J. 2006, 8, E298–E306. [Google Scholar] [CrossRef]
- Guindon, J.; Hohmann, A.G. Cannabinoid CB2 Receptors: A Therapeutic Target for the Treatment of Inflammatory and Neuropathic Pain. Br. J. Pharmacol. 2008, 153, 319–334. [Google Scholar] [CrossRef]
- Klauke, A.-L.; Racz, I.; Pradier, B.; Markert, A.; Zimmer, A.M.; Gertsch, J.; Zimmer, A. The Cannabinoid CB2 Receptor-Selective Phytocannabinoid Beta-Caryophyllene Exerts Analgesic Effects in Mouse Models of Inflammatory and Neuropathic Pain. Eur. Neuropsychopharmacol. 2014, 24, 608–620. [Google Scholar] [CrossRef]
- Bahi, A.; Al Mansouri, S.; Al Memari, E.; Al Ameri, M.; Nurulain, S.M.; Ojha, S. β-Caryophyllene, a CB2 Receptor Agonist Produces Multiple Behavioral Changes Relevant to Anxiety and Depression in Mice. Physiol. Behav. 2014, 135, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Calleja, M.A.; Vieites, J.M.; Montero-Meléndez, T.; Torres, M.I.; Faus, M.J.; Gil, A.; Suárez, A. The Antioxidant Effect of β-Caryophyllene Protects Rat Liver from Carbon Tetrachloride-Induced Fibrosis by Inhibiting Hepatic Stellate Cell Activation. Br. J. Nutr. 2013, 109, 394–401. [Google Scholar] [CrossRef]
- Pant, A.; Mishra, V.; Saikia, S.K.; Shukla, V.; Asthana, J.; Akhoon, B.A.; Pandey, R. Beta-Caryophyllene Modulates Expression of Stress Response Genes and Mediates Longevity in Caenorhabditis Elegans. Exp. Gerontol. 2014, 57, 81–95. [Google Scholar] [CrossRef]
- Yang, M.; Lv, Y.; Tian, X.; Lou, J.; An, R.; Zhang, Q.; Li, M.; Xu, L.; Dong, Z. Neuroprotective Effect of β-Caryophyllene on Cerebral Ischemia-Reperfusion Injury via Regulation of Necroptotic Neuronal Death and Inflammation: In Vivo and In Vitro. Front. Neurosci. 2017, 11, 583. [Google Scholar] [CrossRef]
- Mahmoud, M.F.; Swefy, S.E.; Hasan, R.A.; Ibrahim, A. Role of Cannabinoid Receptors in Hepatic Fibrosis and Apoptosis Associated with Bile Duct Ligation in Rats. Eur. J. Pharmacol. 2014, 742, 118–124. [Google Scholar] [CrossRef]
- Wu, C.; Jia, Y.; Lee, J.H.; Jun, H.; Lee, H.-S.; Hwang, K.-Y.; Lee, S.-J. Trans-Caryophyllene Is a Natural Agonistic Ligand for Peroxisome Proliferator-Activated Receptor-α. Bioorg. Med. Chem. Lett. 2014, 24, 3168–3174. [Google Scholar] [CrossRef] [PubMed]
- Irrera, N.; D’Ascola, A.; Pallio, G.; Bitto, A.; Mazzon, E.; Mannino, F.; Squadrito, V.; Arcoraci, V.; Minutoli, L.; Campo, G.M.; et al. β-Caryophyllene Mitigates Collagen Antibody Induced Arthritis (CAIA) in Mice Through a Cross-Talk between CB2 and PPAR-γ Receptors. Biomolecules 2019, 9, 326. [Google Scholar] [CrossRef] [PubMed]
- Youssef, D.A.; El-Fayoumi, H.M.; Mahmoud, M.F. Beta-Caryophyllene Alleviates Diet-Induced Neurobehavioral Changes in Rats: The Role of CB2 and PPAR-γ Receptors. Biomed. Pharmacother. 2019, 110, 145–154. [Google Scholar] [CrossRef]
- Lago-Fernandez, A.; Zarzo-Arias, S.; Jagerovic, N.; Morales, P. Relevance of Peroxisome Proliferator Activated Receptors in Multitarget Paradigm Associated with the Endocannabinoid System. Int. J. Mol. Sci. 2021, 22, 1001. [Google Scholar] [CrossRef]
- Hasankhani, A.; Bahrami, A.; Tavakoli-Far, B.; Iranshahi, S.; Ghaemi, F.; Akbarizadeh, M.R.; Amin, A.H.; Abedi Kiasari, B.; Mohammadzadeh Shabestari, A. The Role of Peroxisome Proliferator-Activated Receptors in the Modulation of Hyperinflammation Induced by SARS-CoV-2 Infection: A Perspective for COVID-19 Therapy. Front. Immunol. 2023, 14, 1127358. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB Signaling in Inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed]
- Barnabei, L.; Laplantine, E.; Mbongo, W.; Rieux-Laucat, F.; Weil, R. NF-κB: At the Borders of Autoimmunity and Inflammation. Front. Immunol. 2021, 12, 716469. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.; Wang, H.; Zhang, F.; Li, X.; Xiang, L.; Aiguo, S. PPARγ Agonist Pioglitazone Inhibits Microglia Inflammation by Blocking P38 Mitogen-Activated Protein Kinase Signaling Pathways. Inflamm. Res. 2010, 59, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Hommes, D.W. Mitogen Activated Protein (MAP) Kinase Signal Transduction Pathways and Novel Anti-Inflammatory Targets. Gut 2003, 52, 144–151. [Google Scholar] [CrossRef]
- Kulkarni, S.A.; Nagarajan, S.K.; Ramesh, V.; Palaniyandi, V.; Selvam, S.P.; Madhavan, T. Computational Evaluation of Major Components from Plant Essential Oils as Potent Inhibitors of SARS-CoV-2 Spike Protein. J. Mol. Struct. 2020, 1221, 128823. [Google Scholar] [CrossRef]
- Kumar, S.; Kashyap, P.; Chowdhury, S.; Kumar, S.; Panwar, A.; Kumar, A. Identification of Phytochemicals as Potential Therapeutic Agents That Binds to Nsp15 Protein Target of Coronavirus (SARS-CoV-2) That Are Capable of Inhibiting Virus Replication. Phytomedicine 2020, 85, 153317. [Google Scholar] [CrossRef]
- Gupta, S.; Singh, V.; Varadwaj, P.K.; Chakravartty, N.; Katta, A.V.S.K.M.; Lekkala, S.P.; Thomas, G.; Narasimhan, S.; Reddy, A.R.; Reddy Lachagari, V.B. Secondary Metabolites from Spice and Herbs as Potential Multitarget Inhibitors of SARS-CoV-2 Proteins. J. Biomol. Struct. Dyn. 2022, 40, 2264–2283. [Google Scholar] [CrossRef]
- Jang, M.; Park, R.; Park, Y.-I.; Cha, Y.-E.; Yamamoto, A.; Lee, J.I.; Park, J. EGCG, a Green Tea Polyphenol, Inhibits Human Coronavirus Replication in Vitro. Biochem. Biophys. Res. Commun. 2021, 547, 23–28. [Google Scholar] [CrossRef]
- Iorio, R.; Celenza, G.; Petricca, S. Multi-Target Effects of ß-Caryophyllene and Carnosic Acid at the Crossroads of Mitochondrial Dysfunction and Neurodegeneration: From Oxidative Stress to Microglia-Mediated Neuroinflammation. Antioxidants 2022, 11, 1199. [Google Scholar] [CrossRef]
- Wang, L.-C.; Wei, W.-H.; Zhang, X.-W.; Liu, D.; Zeng, K.-W.; Tu, P.-F. An Integrated Proteomics and Bioinformatics Approach Reveals the Anti-Inflammatory Mechanism of Carnosic Acid. Front. Pharmacol. 2018, 9, 370. [Google Scholar] [CrossRef]
- de Oliveira, M.R.; de Souza, I.C.C.; Fürstenau, C.R. Carnosic Acid Induces Anti-Inflammatory Effects in Paraquat-Treated SH-SY5Y Cells Through a Mechanism Involving a Crosstalk Between the Nrf2/HO-1 Axis and NF-κB. Mol. Neurobiol. 2018, 55, 890–897. [Google Scholar] [CrossRef]
- Habtemariam, S. Anti-Inflammatory Therapeutic Mechanisms of Natural Products: Insight from Rosemary Diterpenes, Carnosic Acid and Carnosol. Biomedicines 2023, 11, 545. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T.; Kosaka, K.; Itoh, K.; Kobayashi, A.; Yamamoto, M.; Shimojo, Y.; Kitajima, C.; Cui, J.; Kamins, J.; Okamoto, S.; et al. Carnosic Acid, a Catechol-Type Electrophilic Compound, Protects Neurons Both in Vitro and in Vivo through Activation of the Keap1/Nrf2 Pathway via S- Alkylation of Targeted Cysteines on Keap1. J. Neurochem. 2008, 104, 1116–1131. [Google Scholar] [CrossRef] [PubMed]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.B.; Rahu, N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxid. Med. Cell. Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Amini-Nik, S. The Role of Phytochemicals in the Inflammatory Phase of Wound Healing. Int. J. Mol. Sci. 2017, 18, 1068. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Ding, Y.; Pu, C.; Wang, Z.; Deng, W.; Jin, X. Curcumin Inhibits Pancreatic Cancer Cell Proliferation by Regulating Beclin1 Expression and Inhibiting the Hypoxia-Inducible Factor-1α-Mediated Glycolytic Pathway. J. Gastrointest. Oncol. 2022, 13, 3254–3262. [Google Scholar] [CrossRef]
- Bolat, Z.B.; Islek, Z.; Sahin, F.; Ucisik, M.H. Delivery of Curcumin within Emulsome Nanoparticles Enhances the Anti-Cancer Activity in Androgen-Dependent Prostate Cancer Cell. Mol. Biol. Rep. 2023, 50, 2531–2543. [Google Scholar] [CrossRef]
- Aydın, B.; Nazıroğlu, M. Involvement of TRPM7 Channel on the Induction of Diabetic Neuropathic Pain in Mice: Protective Role of Selenium and Curcumin. Biol. Trace Elem. Res. 2023, 201, 2377–2395. [Google Scholar] [CrossRef]
- Jacob, A.; Wu, R.; Zhou, M.; Wang, P. Mechanism of the Anti-Inflammatory Effect of Curcumin: PPAR-Gamma Activation. PPAR Res. 2007, 2007, 89369. [Google Scholar] [CrossRef]
- Lin, C.-H.; Chou, C.-C.; Lee, Y.-H.; Hung, C.-C. Curcumin Facilitates Aryl Hydrocarbon Receptor Activation to Ameliorate Inflammatory Astrogliosis. Molecules 2022, 27, 2507. [Google Scholar] [CrossRef]
- Nalli, M.; Ortar, G.; Schiano Moriello, A.; Di Marzo, V.; De Petrocellis, L. Effects of Curcumin and Curcumin Analogues on TRP Channels. Fitoterapia 2017, 122, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Beevers, C.S.; Huang, S. The Targets of Curcumin. Curr. Drug Targets 2011, 12, 332–347. [Google Scholar] [CrossRef] [PubMed]
- Rau, O.; Wurglics, M.; Paulke, A.; Zitzkowski, J.; Meindl, N.; Bock, A.; Dingermann, T.; Abdel-Tawab, M.; Schubert-Zsilavecz, M. Carnosic Acid and Carnosol, Phenolic Diterpene Compounds of the Labiate Herbs Rosemary and Sage, Are Activators of the Human Peroxisome Proliferator-Activated Receptor Gamma. Planta Med. 2006, 72, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Mirza, F.J.; Zahid, S.; Holsinger, R.M.D. Neuroprotective Effects of Carnosic Acid: Insight into Its Mechanisms of Action. Molecules 2023, 28, 2306. [Google Scholar] [CrossRef]
- Satoh, T.; Trudler, D.; Oh, C.-K.; Lipton, S.A. Potential Therapeutic Use of the Rosemary Diterpene Carnosic Acid for Alzheimer’s Disease, Parkinson’s Disease, and Long-COVID through NRF2 Activation to Counteract the NLRP3 Inflammasome. Antioxidants 2022, 11, 124. [Google Scholar] [CrossRef]
- Earley, S.; Gonzales, A.L.; Garcia, Z.I. A Dietary Agonist of Transient Receptor Potential Cation Channel V3 Elicits Endothelium-Dependent Vasodilation. Mol. Pharmacol. 2010, 77, 612–620. [Google Scholar] [CrossRef]
- Hotta, M.; Nakata, R.; Katsukawa, M.; Hori, K.; Takahashi, S.; Inoue, H. Carvacrol, a Component of Thyme Oil, Activates PPARα and γ and Suppresses COX-2 Expression. J. Lipid Res. 2010, 51, 132–139. [Google Scholar] [CrossRef]
- Melo, F.H.C.; Venâncio, E.T.; de Sousa, D.P.; de França Fonteles, M.M.; de Vasconcelos, S.M.M.; Viana, G.S.B.; de Sousa, F.C.F. Anxiolytic-like Effect of Carvacrol (5-Isopropyl-2-Methylphenol) in Mice: Involvement with GABAergic Transmission. Fundam. Clin. Pharmacol. 2010, 24, 437–443. [Google Scholar] [CrossRef]
- Marjanović, D.S.; Zdravković, N.; Milovanović, M.; Trailović, J.N.; Robertson, A.P.; Todorović, Z.; Trailović, S.M. Carvacrol Acts as a Potent Selective Antagonist of Different Types of Nicotinic Acetylcholine Receptors and Enhances the Effect of Monepantel in the Parasitic Nematode Ascaris Suum. Vet. Parasitol. 2020, 278, 109031. [Google Scholar] [CrossRef]
- Lee, M.; Lee, S.H.; Choi, S.; Choi, B.Y.; Suh, S.W. Carvacrol Inhibits Expression of Transient Receptor Potential Melastatin 7 Channels and Alleviates Zinc Neurotoxicity Induced by Traumatic Brain Injury. Int. J. Mol. Sci. 2022, 23, 13840. [Google Scholar] [CrossRef]
- Park, C.S.; Lee, J.Y.; Choi, H.Y.; Yune, T.Y. Suppression of Transient Receptor Potential Melastatin 7 by Carvacrol Protects against Injured Spinal Cord by Inhibiting Blood–Spinal Cord Barrier Disruption. J. Neurotrauma 2022, 39, 735–749. [Google Scholar] [CrossRef] [PubMed]
- Maffei, M.E. Plant Natural Sources of the Endocannabinoid (E)-β-Caryophyllene: A Systematic Quantitative Analysis of Published Literature. Int. J. Mol. Sci. 2020, 21, 6540. [Google Scholar] [CrossRef] [PubMed]
- Fidyt, K.; Fiedorowicz, A.; Strządała, L.; Szumny, A. β-Caryophyllene and β-Caryophyllene Oxide-Natural Compounds of Anticancer and Analgesic Properties. Cancer Med. 2016, 5, 3007–3017. [Google Scholar] [CrossRef]
- Aly, E.; Khajah, M.A.; Masocha, W. β-Caryophyllene, a CB2-Receptor-Selective Phytocannabinoid, Suppresses Mechanical Allodynia in a Mouse Model of Antiretroviral-Induced Neuropathic Pain. Molecules 2019, 25, 106. [Google Scholar] [CrossRef] [PubMed]
- Hajizadeh Moghaddam, A.; Mashayekhpour, M.A.; Tabari, M.A. Anxiolytic-like Effects of Citral in the Mouse Elevated plus Maze: Involvement of GABAergic and Serotonergic Transmissions. Naunyn. Schmiedebergs Arch. Pharmacol. 2023, 396, 301–309. [Google Scholar] [CrossRef]
- Alves Rodrigues Santos, S.A.; de Barros Mamede Vidal Damasceno, M.; Alves Magalhães, F.E.; Sessle, B.J.; Amaro de Oliveira, B.; Alves Batista, F.L.; Vieira-Neto, A.E.; Rolim Campos, A. Transient Receptor Potential Channel Involvement in Antinociceptive Effect of Citral in Orofacial Acute and Chronic Pain Models. EXCLI J. 2022, 21, 869–887. [Google Scholar] [CrossRef]
- Harada, N.; Okuyama, M.; Teraoka, Y.; Arahori, Y.; Shinmori, Y.; Horiuchi, H.; Luis, P.B.; Joseph, A.I.; Kitakaze, T.; Matsumura, S.; et al. Identification of G Protein-Coupled Receptor 55 (GPR55) as a Target of Curcumin. NPJ Sci. Food 2022, 6, 4. [Google Scholar] [CrossRef]
- Enayati, A.; Ghojoghnejad, M.; Roufogalis, B.D.; Maollem, S.A.; Sahebkar, A. Impact of Phytochemicals on PPAR Receptors: Implications for Disease Treatments. PPAR Res. 2022, 2022, 4714914. [Google Scholar] [CrossRef] [PubMed]
- Leamy, A.W.; Shukla, P.; McAlexander, M.A.; Carr, M.J.; Ghatta, S. Curcumin ((E,E)-1,7-Bis(4-Hydroxy-3-Methoxyphenyl)-1,6-Heptadiene-3,5-Dione) Activates and Desensitizes the Nociceptor Ion Channel TRPA1. Neurosci. Lett. 2011, 503, 157–162. [Google Scholar] [CrossRef]
- Negri, A.; Naponelli, V.; Rizzi, F.; Bettuzzi, S. Molecular Targets of Epigallocatechin-Gallate (EGCG): A Special Focus on Signal Transduction and Cancer. Nutrients 2018, 10, 1936. [Google Scholar] [CrossRef]
- Tachibana, H.; Koga, K.; Fujimura, Y.; Yamada, K. A Receptor for Green Tea Polyphenol EGCG. Nat. Struct. Mol. Biol. 2004, 11, 380–381. [Google Scholar] [CrossRef] [PubMed]
- Ohgitani, E.; Shin-Ya, M.; Ichitani, M.; Kobayashi, M.; Takihara, T.; Kawamoto, M.; Kinugasa, H.; Mazda, O. Significant Inactivation of SARS-CoV-2 In Vitro by a Green Tea Catechin, a Catechin-Derivative, and Black Tea Galloylated Theaflavins. Molecules 2021, 26, 3572. [Google Scholar] [CrossRef] [PubMed]
- Henss, L.; Auste, A.; Schürmann, C.; Schmidt, C.; von Rhein, C.; Mühlebach, M.D.; Schnierle, B.S. The Green Tea Catechin Epigallocatechin Gallate Inhibits SARS-CoV-2 Infection. J. Gen. Virol. 2021, 102, 001574. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Ma, R.; Sun, W.; Ji, Z. Antibacterial Activity of Epigallocatechin Gallate (EGCG) against Shigella Flexneri. Int. J. Environ. Res. Public. Health 2023, 20, 4676. [Google Scholar] [CrossRef]
- Tachibana, H. Molecular Basis for Cancer Chemoprevention by Green Tea Polyphenol EGCG. Forum Nutr. 2009, 61, 156–169. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Mao, B.; Cui, S.; Zhang, Q.; Zhao, J.; Tang, X.; Chen, W. Absorption, Metabolism, Bioactivity, and Biotransformation of Epigallocatechin Gallate. Crit. Rev. Food Sci. Nutr. 2023, 64, 6546–6566. [Google Scholar] [CrossRef]
- Nkambeu, B.; Salem, J.B.; Beaudry, F. Antinociceptive Activity of Vanilloids in Caenorhabditis Elegans Is Mediated by the Desensitization of the TRPV Channel OCR-2 and Specific Signal Transduction Pathways. Neurochem. Res. 2023, 48, 1900–1911. [Google Scholar] [CrossRef]
- Anjum, N.F.; Shanmugarajan, D.; Shivaraju, V.K.; Faizan, S.; Naishima, N.L.; Prashantha Kumar, B.R.; Javid, S.; Purohit, M.N. Novel Derivatives of Eugenol as Potent Anti-Inflammatory Agents via PPARγ Agonism: Rational Design, Synthesis, Analysis, PPARγ Protein Binding Assay and Computational Studies. RSC Adv. 2022, 12, 16966–16978. [Google Scholar] [CrossRef]
- Gao, Y.; Lu, Y.; Zhang, N.; Udenigwe, C.C.; Zhang, Y.; Fu, Y. Preparation, Pungency and Bioactivity of Gingerols from Ginger (Zingiber Officinale Roscoe): A Review. Crit. Rev. Food Sci. Nutr. 2022, 64, 2708–2733. [Google Scholar] [CrossRef]
- Mao, Q.-Q.; Xu, X.-Y.; Cao, S.-Y.; Gan, R.-Y.; Corke, H.; Beta, T.; Li, H.-B. Bioactive Compounds and Bioactivities of Ginger (Zingiber Officinale Roscoe). Foods 2019, 8, 185. [Google Scholar] [CrossRef]
- Semwal, R.B.; Semwal, D.K.; Combrinck, S.; Viljoen, A.M. Gingerols and Shogaols: Important Nutraceutical Principles from Ginger. Phytochemistry 2015, 117, 554–568. [Google Scholar] [CrossRef] [PubMed]
- Park, H.M.; Lee, J.H.; Yaoyao, J.; Jun, H.J.; Lee, S.J. Limonene, a Natural Cyclic Terpene, Is an Agonistic Ligand for Adenosine A(2A) Receptors. Biochem. Biophys. Res. Commun. 2011, 404, 345–348. [Google Scholar] [CrossRef]
- Patel, M.; Narke, D.; Kurade, M.; Frey, K.M.; Rajalingam, S.; Siddiquee, A.; Mustafa, S.J.; Ledent, C.; Ponnoth, D.S. Limonene-Induced Activation of A2A Adenosine Receptors Reduces Airway Inflammation and Reactivity in a Mouse Model of Asthma. Purinergic Signal. 2020, 16, 415–426. [Google Scholar] [CrossRef]
- Kaimoto, T.; Hatakeyama, Y.; Takahashi, K.; Imagawa, T.; Tominaga, M.; Ohta, T. Involvement of Transient Receptor Potential A1 Channel in Algesic and Analgesic Actions of the Organic Compound Limonene. Eur. J. Pain 2016, 20, 1155–1165. [Google Scholar] [CrossRef]
- Song, Y.; Seo, S.; Lamichhane, S.; Seo, J.; Hong, J.T.; Cha, H.J.; Yun, J. Limonene Has Anti-Anxiety Activity via Adenosine A2A Receptor-Mediated Regulation of Dopaminergic and GABAergic Neuronal Function in the Striatum. Phytomedicine Int. J. 2021, 83, 153474. [Google Scholar] [CrossRef] [PubMed]
- Harada, H.; Kashiwadani, H.; Kanmura, Y.; Kuwaki, T. Linalool Odor-Induced Anxiolytic Effects in Mice. Front. Behav. Neurosci. 2018, 12, 241. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, S.; Yamaguchi, R.; Ishikawa, S.; Sakurai, T.; Kajiya, K.; Kanmura, Y.; Kuwaki, T.; Kashiwadani, H. Odour-Induced Analgesia Mediated by Hypothalamic Orexin Neurons in Mice. Sci. Rep. 2016, 6, 37129. [Google Scholar] [CrossRef]
- Higa, Y.; Kashiwadani, H.; Sugimura, M.; Kuwaki, T. Orexinergic Descending Inhibitory Pathway Mediates Linalool Odor-Induced Analgesia in Mice. Sci. Rep. 2021, 11, 9224. [Google Scholar] [CrossRef] [PubMed]
- da Fonseca, C.O.; Linden, R.; Futuro, D.; Gattass, C.R.; Quirico-Santos, T. Ras Pathway Activation in Gliomas: A Strategic Target for Intranasal Administration of Perillyl Alcohol. Arch. Immunol. Ther. Exp. 2008, 56, 267–276. [Google Scholar] [CrossRef]
- Cho, H.-Y.; Wang, W.; Jhaveri, N.; Torres, S.; Tseng, J.; Leong, M.N.; Lee, D.J.; Goldkorn, A.; Xu, T.; Petasis, N.A.; et al. Perillyl Alcohol for the Treatment of Temozolomide-Resistant Gliomas. Mol. Cancer Ther. 2012, 11, 2462–2472. [Google Scholar] [CrossRef]
- Chen, T.C.; da Fonseca, C.O.; Schönthal, A.H. Intranasal Perillyl Alcohol for Glioma Therapy: Molecular Mechanisms and Clinical Development. Int. J. Mol. Sci. 2018, 19, 3905. [Google Scholar] [CrossRef] [PubMed]
- Tomaz-Morais, J.F.; Braga, R.M.; de Sousa, F.B.; de Sousa, D.P.; deM Pordeus, L.C.; de Almeida, R.N.; de Castro, R.D. Orofacial Antinociceptive Activity of (S)-(-)-Perillyl Alcohol in Mice: A Randomized, Controlled and Triple-Blind Study. Int. J. Oral Maxillofac. Surg. 2017, 46, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Gehm, B.D.; McAndrews, J.M.; Chien, P.-Y.; Jameson, J.L. Resveratrol, a Polyphenolic Compound Found in Grapes and Wine, Is an Agonist for the Estrogen Receptor. Proc. Natl. Acad. Sci. USA 1997, 94, 14138–14143. [Google Scholar] [CrossRef]
- Nwachukwu, J.C.; Srinivasan, S.; Bruno, N.E.; Parent, A.A.; Hughes, T.S.; Pollock, J.A.; Gjyshi, O.; Cavett, V.; Nowak, J.; Garcia-Ordonez, R.D.; et al. Resveratrol Modulates the Inflammatory Response via an Estrogen Receptor-Signal Integration Network. eLife 2014, 3, e02057. [Google Scholar] [CrossRef]
- De Amicis, F.; Chimento, A.; Montalto, F.; Casaburi, I.; Sirianni, R.; Pezzi, V. Steroid Receptor Signallings as Targets for Resveratrol Actions in Breast and Prostate Cancer. Int. J. Mol. Sci. 2019, 20, 1087. [Google Scholar] [CrossRef] [PubMed]
- Tiroch, J.; Sterneder, S.; Di Pizio, A.; Lieder, B.; Hoelz, K.; Holik, A.-K.; Pignitter, M.; Behrens, M.; Somoza, M.; Ley, J.P.; et al. Bitter Sensing TAS2R50 Mediates the Trans -Resveratrol-Induced Anti-Inflammatory Effect on Interleukin 6 Release in HGF-1 Cells in Culture. J. Agric. Food Chem. 2021, 69, 13339–13349. [Google Scholar] [CrossRef]
- Calleri, E.; Pochetti, G.; Dossou, K.S.S.; Laghezza, A.; Montanari, R.; Capelli, D.; Prada, E.; Loiodice, F.; Massolini, G.; Bernier, M.; et al. Resveratrol and Its Metabolites Bind to PPARs. Chembiochem 2014, 15, 1154–1160. [Google Scholar] [CrossRef]
- Wu, L.; Guo, C.; Wu, J. Therapeutic Potential of PPARγ Natural Agonists in Liver Diseases. J. Cell. Mol. Med. 2020, 24, 2736–2748. [Google Scholar] [CrossRef]
- Yamazaki, T.; Sagisaka, M.; Ikeda, R.; Nakamura, T.; Matsuda, N.; Ishii, T.; Nakayama, T.; Watanabe, T. The Human Bitter Taste Receptor hTAS2R39 Is the Primary Receptor for the Bitterness of Theaflavins. Biosci. Biotechnol. Biochem. 2014, 78, 1753–1756. [Google Scholar] [CrossRef]
- O’Neill, E.J.; Termini, D.; Albano, A.; Tsiani, E. Anti-Cancer Properties of Theaflavins. Molecules 2021, 26, 987. [Google Scholar] [CrossRef]
- Luo, T.; Jiang, J.J. Anticancer Effects and Molecular Target of Theaflavins from Black Tea Fermentation in Vitro and in Vivo. J. Agric. Food Chem. 2021, 69, 15052–15065. [Google Scholar] [CrossRef] [PubMed]
- Kibret, B.G.; Ishiguro, H.; Horiuchi, Y.; Onaivi, E.S. New Insights and Potential Therapeutic Targeting of CB2 Cannabinoid Receptors in CNS Disorders. Int. J. Mol. Sci. 2022, 23, 975. [Google Scholar] [CrossRef] [PubMed]
- Bie, B.; Wu, J.; Foss, J.F.; Naguib, M. An Overview of the Cannabinoid Type 2 Receptor System and Its Therapeutic Potential. Curr. Opin. Anaesthesiol. 2018, 31, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, E.C.D.; Assis, P.M.; Junqueira, L.A.; Cola, M.; Santos, A.R.S.; Raposo, N.R.B.; Dutra, R.C. Citral Inhibits the Inflammatory Response and Hyperalgesia in Mice: The Role of TLR4, TLR2/Dectin-1, and CB2 Cannabinoid Receptor/ATP-Sensitive K+ Channel Pathways. J. Nat. Prod. 2020, 83, 1190–1200. [Google Scholar] [CrossRef]
- Talavera, K.; Startek, J.B.; Alvarez-Collazo, J.; Boonen, B.; Alpizar, Y.A.; Sanchez, A.; Naert, R.; Nilius, B. Mammalian Transient Receptor Potential TRPA1 Channels: From Structure to Disease. Physiol. Rev. 2020, 100, 725–803. [Google Scholar] [CrossRef]
- Premkumar, L.S. Transient Receptor Potential Channels as Targets for Phytochemicals. ACS Chem. Neurosci. 2014, 5, 1117–1130. [Google Scholar] [CrossRef]
- Vriens, J.; Nilius, B.; Vennekens, R. Herbal Compounds and Toxins Modulating TRP Channels. Curr. Neuropharmacol. 2008, 6, 79–96. [Google Scholar] [CrossRef]
- Izquierdo, C.; Martín-Martínez, M.; Gómez-Monterrey, I.; González-Muñiz, R. TRPM8 Channels: Advances in Structural Studies and Pharmacological Modulation. Int. J. Mol. Sci. 2021, 22, 8502. [Google Scholar] [CrossRef]
- Tyagi, S.; Gupta, P.; Saini, A.S.; Kaushal, C.; Sharma, S. The Peroxisome Proliferator-Activated Receptor: A Family of Nuclear Receptors Role in Various Diseases. J. Adv. Pharm. Technol. Res. 2011, 2, 236–240. [Google Scholar] [CrossRef]
- Grygiel-Górniak, B. Peroxisome Proliferator-Activated Receptors and Their Ligands: Nutritional and Clinical Implications—A Review. Nutr. J. 2014, 13, 17. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, X.; Hu, C.; Zhang, Y.; Zhang, L.; Kan, J.; Li, B.; Du, J. Regulation of GABAA and 5-HT Receptors Involved in Anxiolytic Mechanisms of Jujube Seed: A System Biology Study Assisted by UPLC-Q-TOF/MS and RT-qPCR Method. Front. Pharmacol. 2020, 11, 01320. [Google Scholar] [CrossRef] [PubMed]
- Phootha, N.; Yongparnichkul, N.; Fang, Z.; Gan, R.-Y.; Zhang, P. Plants and Phytochemicals Potentials in Tackling Anxiety: A Systematic Review. Phytomedicine Plus 2022, 2, 100375. [Google Scholar] [CrossRef]
- Li, J.; Yu, J.; Zou, H.; Zhang, J.; Ren, L. Estrogen Receptor-Mediated Health Benefits of Phytochemicals: A Review. Food Funct. 2023, 14, 10681–10699. [Google Scholar] [CrossRef]
- Lecomte, S.; Demay, F.; Ferrière, F.; Pakdel, F. Phytochemicals Targeting Estrogen Receptors: Beneficial Rather Than Adverse Effects? Int. J. Mol. Sci. 2017, 18, 1381. [Google Scholar] [CrossRef]
- Carpenter, B.; Lebon, G. Human Adenosine A2A Receptor: Molecular Mechanism of Ligand Binding and Activation. Front. Pharmacol. 2017, 8, 898. [Google Scholar] [CrossRef]
- Umeda, D.; Yano, S.; Yamada, K.; Tachibana, H. Green Tea Polyphenol Epigallocatechin-3-Gallate Signaling Pathway through 67-kDa Laminin Receptor. J. Biol. Chem. 2008, 283, 3050–3058. [Google Scholar] [CrossRef]
- Fujimura, Y.; Kumazoe, M.; Tachibana, H. 67-kDa Laminin Receptor-Mediated Cellular Sensing System of Green Tea Polyphenol EGCG and Functional Food Pairing. Molecules 2022, 27, 5130. [Google Scholar] [CrossRef]
- Kou, Z.; Dai, W. Aryl Hydrocarbon Receptor: Its Roles in Physiology. Biochem. Pharmacol. 2021, 185, 114428. [Google Scholar] [CrossRef] [PubMed]
- Richter-Laskowska, M.; Trybek, P.; Delfino, D.V.; Wawrzkiewicz-Jałowiecka, A. Flavonoids as Modulators of Potassium Channels. Int. J. Mol. Sci. 2023, 24, 1311. [Google Scholar] [CrossRef]
- Shi, S.; Zhao, Q.; Ke, C.; Long, S.; Zhang, F.; Zhang, X.; Li, Y.; Liu, X.; Hu, H.; Yin, S. Loureirin B Exerts Its Immunosuppressive Effects by Inhibiting STIM1/Orai1 and KV1.3 Channels. Front. Pharmacol. 2021, 12, 685092. [Google Scholar] [CrossRef]
- Calderon-Rivera, A.; Loya-Lopez, S.; Gomez, K.; Khanna, R. Plant and Fungi Derived Analgesic Natural Products Targeting Voltage-Gated Sodium and Calcium Channels. Channels 2022, 16, 198–215. [Google Scholar] [CrossRef] [PubMed]
- Hameed, S. Nav1.7 and Nav1.8: Role in the Pathophysiology of Pain. Mol. Pain 2019, 15, 1744806919858801. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Shan, Z.; Guo, W.; Wang, Y.; Cai, S.; Li, F.; Huang, Q.; Liu, J.A.; Cheung, C.W.; Cai, S. Reversal of Peripheral Neuropathic Pain by the Small-Molecule Natural Product Narirutin via Block of Nav1.7 Voltage-Gated Sodium Channel. Int. J. Mol. Sci. 2022, 23, 14842. [Google Scholar] [CrossRef] [PubMed]
- Saleh, H.A.; Yousef, M.H.; Abdelnaser, A. The Anti-Inflammatory Properties of Phytochemicals and Their Effects on Epigenetic Mechanisms Involved in TLR4/NF-κB-Mediated Inflammation. Front. Immunol. 2021, 12, 606069. [Google Scholar] [CrossRef]
- Chauhan, A.; Islam, A.U.; Prakash, H.; Singh, S. Phytochemicals Targeting NF-κB Signaling: Potential Anti-Cancer Interventions. J. Pharm. Anal. 2022, 12, 394–405. [Google Scholar] [CrossRef]
- Zhao, L.; Lee, J.Y.; Hwang, D.H. Inhibition of Pattern Recognition Receptor-Mediated Inflammation by Bioactive Phytochemicals. Nutr. Rev. 2011, 69, 310–320. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Kao, C.-L.; Liu, C.-M. The Cancer Prevention, Anti-Inflammatory and Anti-Oxidation of Bioactive Phytochemicals Targeting the TLR4 Signaling Pathway. Int. J. Mol. Sci. 2018, 19, 2729. [Google Scholar] [CrossRef]
- Lee, J.Y.; Youn, H.S.; Hwang, D.H. The Novel Anti-inflammatory Mechanisms for Phytochemicals Mediated through Modulation of Toll-like Receptor Signaling. FASEB J. 2007, 21, A365. [Google Scholar] [CrossRef]
- Gao, W.; Xiong, Y.; Li, Q.; Yang, H. Inhibition of Toll-Like Receptor Signaling as a Promising Therapy for Inflammatory Diseases: A Journey from Molecular to Nano Therapeutics. Front. Physiol. 2017, 8, 508. [Google Scholar] [CrossRef]
- Hong Byun, E.; Fujimura, Y.; Yamada, K.; Tachibana, H. TLR4 Signaling Inhibitory Pathway Induced by Green Tea Polyphenol Epigallocatechin-3-Gallate through 67-kDa Laminin Receptor. J. Immunol. 2010, 185, 33–45. [Google Scholar] [CrossRef]
- Oakes, J.A.; Davies, M.C.; Collins, M.O. TBK1: A New Player in ALS Linking Autophagy and Neuroinflammation. Mol. Brain 2017, 10, 5. [Google Scholar] [CrossRef] [PubMed]
- Subedi, L.; Lee, S.E.; Madiha, S.; Gaire, B.P.; Jin, M.; Yumnam, S.; Kim, S.Y. Phytochemicals against TNFα-Mediated Neuroinflammatory Diseases. Int. J. Mol. Sci. 2020, 21, 764. [Google Scholar] [CrossRef] [PubMed]
- Zahedipour, F.; Hosseini, S.A.; Henney, N.C.; Barreto, G.E.; Sahebkar, A. Phytochemicals as Inhibitors of Tumor Necrosis Factor Alpha and Neuroinflammatory Responses in Neurodegenerative Diseases. Neural Regen. Res. 2022, 17, 1675–1684. [Google Scholar] [CrossRef] [PubMed]
- Ali, E.S.; Akter, S.; Ramproshad, S.; Mondal, B.; Riaz, T.A.; Islam, M.T.; Khan, I.N.; Docea, A.O.; Calina, D.; Sharifi-Rad, J.; et al. Targeting Ras-ERK Cascade by Bioactive Natural Products for Potential Treatment of Cancer: An Updated Overview. Cancer Cell Int. 2022, 22, 246. [Google Scholar] [CrossRef]
- Chen, H.; Liu, R.H. Potential Mechanisms of Action of Dietary Phytochemicals for Cancer Prevention by Targeting Cellular Signaling Transduction Pathways. J. Agric. Food Chem. 2018, 66, 3260–3276. [Google Scholar] [CrossRef]
- Do, H.N.; Akhter, S.; Miao, Y. Pathways and Mechanism of Caffeine Binding to Human Adenosine A2A Receptor. Front. Mol. Biosci. 2021, 8, 673170. [Google Scholar] [CrossRef]
- Jacobson, K.A.; Moro, S.; Manthey, J.A.; West, P.L.; Ji, X.-D. Interactions of Flavones and Other Phytochemicals with Adenosine Receptors. Adv. Exp. Med. Biol. 2002, 505, 163–171. [Google Scholar] [CrossRef]
- Iannotti, F.A.; Vitale, R.M. The Endocannabinoid System and PPARs: Focus on Their Signalling Crosstalk, Action and Transcriptional Regulation. Cells 2021, 10, 586. [Google Scholar] [CrossRef]
- Bento, A.F.; Marcon, R.; Dutra, R.C.; Claudino, R.F.; Cola, M.; Leite, D.F.P.; Calixto, J.B. β-Caryophyllene Inhibits Dextran Sulfate Sodium-Induced Colitis in Mice through CB2 Receptor Activation and PPARγ Pathway. Am. J. Pathol. 2011, 178, 1153–1166. [Google Scholar] [CrossRef]
- Dubrac, S.; Schmuth, M. PPAR-Alpha in Cutaneous Inflammation. Dermato-Endocrinology 2011, 3, 23–26. [Google Scholar] [CrossRef]
- Scirpo, R.; Fiorotto, R.; Villani, A.; Amenduni, M.; Spirli, C.; Strazzabosco, M. Stimulation of Nuclear Receptor Peroxisome Proliferator-Activated Receptor-γ Limits NF-κB-Dependent Inflammation in Mouse Cystic Fibrosis Biliary Epithelium. Hepatology 2015, 62, 1551–1562. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, A.A.; Thatcher, T.H.; Olsen, K.C.; Maggirwar, S.B.; Phipps, R.P.; Sime, P.J. PPAR-γ Ligands Repress TGFβ-Induced Myofibroblast Differentiation by Targeting the PI3K/Akt Pathway: Implications for Therapy of Fibrosis. PLoS ONE 2011, 6, e15909. [Google Scholar] [CrossRef]
- Hosseini, H.; Ghavidel, F.; Panahi, G.; Majeed, M.; Sahebkar, A. A Systematic Review and Meta-Analysis of Randomized Controlled Trials Investigating the Effect of the Curcumin and Piperine Combination on Lipid Profile in Patients with Metabolic Syndrome and Related Disorders. Phytother. Res. 2023, 37, 1212–1224. [Google Scholar] [CrossRef]
- Ghobadi, N.; Asoodeh, A. Co-Administration of Curcumin with Other Phytochemicals Improves Anticancer Activity by Regulating Multiple Molecular Targets. Phytother. Res. 2023, 37, 1688–1702. [Google Scholar] [CrossRef] [PubMed]
- Heidari, H.; Bagherniya, M.; Majeed, M.; Sathyapalan, T.; Jamialahmadi, T.; Sahebkar, A. Curcumin-Piperine Co-Supplementation and Human Health: A Comprehensive Review of Preclinical and Clinical Studies. Phytother. Res. 2023, 37, 1462–1487. [Google Scholar] [CrossRef]
- Wang, S.; Chen, R.; Zhong, Z.; Shi, Z.; Chen, M.; Wang, Y. Epigallocatechin-3-Gallate Potentiates the Effect of Curcumin in Inducing Growth Inhibition and Apoptosis of Resistant Breast Cancer Cells. Am. J. Chin. Med. 2014, 42, 1279–1300. [Google Scholar] [CrossRef]
- Yuan, J.; Zou, M.; Xiang, X.; Zhu, H.; Chu, W.; Liu, W.; Chen, F.; Lin, J. Curcumin Improves Neural Function after Spinal Cord Injury by the Joint Inhibition of the Intracellular and Extracellular Components of Glial Scar. J. Surg. Res. 2015, 195, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Dent, E.L.; Taylor, E.B.; Turbeville, H.R.; Ryan, M.J. Curcumin Attenuates Autoimmunity and Renal Injury in an Experimental Model of Systemic Lupus Erythematosus. Physiol. Rep. 2020, 8, e14501. [Google Scholar] [CrossRef]
- Coelho, A.M.; Queiroz, I.F.; Perucci, L.O.; Souza, M.O.D.; Lima, W.G.; Talvani, A.; Costa, D.C. Piperine as Therapeutic Agent in Paracetamol-Induced Hepatotoxicity in Mice. Pharmaceutics 2022, 14, 1800. [Google Scholar] [CrossRef]
- Abdelhamid, A.M.; Selim, A.; Zaafan, M.A. The Hepatoprotective Effect of Piperine Against Thioacetamide-Induced Liver Fibrosis in Mice: The Involvement of miR-17 and TGF-β/Smads Pathways. Front. Mol. Biosci. 2021, 8, 754098. [Google Scholar] [CrossRef]
- Choi, J.-W.; Lee, S.-K.; Kim, M.-J.; Kim, D.-G.; Shin, J.-Y.; Zhou, Z.; Jo, I.-J.; Song, H.-J.; Bae, G.-S.; Park, S.-J. Piperine Ameliorates the Severity of Fibrosis via Inhibition of TGF-β/SMAD Signaling in a Mouse Model of Chronic Pancreatitis. Mol. Med. Rep. 2019, 20, 3709–3718. [Google Scholar] [CrossRef] [PubMed]
- Stojanović-Radić, Z.; Pejčić, M.; Dimitrijević, M.; Aleksić, A.; Anil Kumar, N.V.; Salehi, B.; Cho, W.C.; Sharifi-Rad, J. Piperine-A Major Principle of Black Pepper: A Review of Its Bioactivity and Studies. Appl. Sci. 2019, 9, 4270. [Google Scholar] [CrossRef]
- El-Sayed, S.A.M.; Fouad, G.I.; Rizk, M.Z.; Beherei, H.H.; Mabrouk, M. Comparative Neuroprotective Potential of Nanoformulated and Free Resveratrol Against Cuprizone-Induced Demyelination in Rats. Mol. Neurobiol. 2024. [Google Scholar] [CrossRef]
- Takeda, M.; Sashide, Y.; Toyota, R.; Ito, H. The Phytochemical, Quercetin, Attenuates Nociceptive and Pathological Pain: Neurophysiological Mechanisms and Therapeutic Potential. Molecules 2024, 29, 3957. [Google Scholar] [CrossRef]
- Lee, G.B.; Kim, Y.; Lee, K.E.; Vinayagam, R.; Singh, M.; Kang, S.G. Anti-Inflammatory Effects of Quercetin, Rutin, and Troxerutin Result From the Inhibition of NO Production and the Reduction of COX-2 Levels in RAW 264.7 Cells Treated with LPS. Appl. Biochem. Biotechnol. 2024. [Google Scholar] [CrossRef]
- Zhou, D.-H.; Wang, X.; Yang, M.; Shi, X.; Huang, W.; Feng, Q. Combination of Low Concentration of (-)-Epigallocatechin Gallate (EGCG) and Curcumin Strongly Suppresses the Growth of Non-Small Cell Lung Cancer in Vitro and in Vivo through Causing Cell Cycle Arrest. Int. J. Mol. Sci. 2013, 14, 12023–12036. [Google Scholar] [CrossRef] [PubMed]
- Niedzwiecki, A.; Roomi, M.W.; Kalinovsky, T.; Rath, M. Anticancer Efficacy of Polyphenols and Their Combinations. Nutrients 2016, 8, 552. [Google Scholar] [CrossRef]
- Widyaningrum, R.; Wu, Y.-W.; Delila, L.; Lee, D.-Y.; Wang, T.-J.; Burnouf, T. In Vitro Evaluation of Platelet Extracellular Vesicles (PEVs) for Corneal Endothelial Regeneration. Platelets 2022, 33, 1237–1250. [Google Scholar] [CrossRef]
- Liu, X.; Li, X.; Wu, G.; Qi, P.; Zhang, Y.; Liu, Z.; Li, X.; Yu, Y.; Ye, X.; Li, Y.; et al. Umbilical Cord Mesenchymal Stem Cell-Derived Small Extracellular Vesicles Deliver miR-21 to Promote Corneal Epithelial Wound Healing through PTEN/PI3K/Akt Pathway. Stem Cells Int. 2022, 2022, 1252557. [Google Scholar] [CrossRef]
- Liao, X.; Yan, F.; Hu, S.; Mu, J.; Li, S.; He, Y.; Tang, M.; Chen, J.; Yu, L.; Sun, J. Adipose Mesenchymal Stem Cell Sheets-Derived Extracellular Vesicles-microRNA-10b Promote Skin Wound Healing by Elevating Expression of CDK6. Biomater. Adv. 2022, 136, 212781. [Google Scholar] [CrossRef]
- Wang, M.; Wu, P.; Huang, J.; Liu, W.; Qian, H.; Sun, Y.; Shi, H. Skin Cell-Derived Extracellular Vesicles: A Promising Therapeutic Strategy for Cutaneous Injury. Burns Trauma 2022, 10, tkac037. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.-J.; Bi, Q.-C.; Gan, L.-J.; Zhang, L.-L.; Wei, M.-J.; Hong, T.; Liu, R.; Qiu, C.-L.; Han, X.-J.; Jiang, L.-P. Exosomes Derived from Glioma Cells under Hypoxia Promote Angiogenesis through Up-Regulated Exosomal Connexin 43. Int. J. Med. Sci. 2022, 19, 1205–1215. [Google Scholar] [CrossRef] [PubMed]
- Moeinabadi-Bidgoli, K.; Rezaee, M.; Hossein-Khannazer, N.; Babajani, A.; Aghdaei, H.A.; Arki, M.K.; Afaghi, S.; Niknejad, H.; Vosough, M. Exosomes for Angiogenesis Induction in Ischemic Disorders. J. Cell. Mol. Med. 2023, 27, 763–787. [Google Scholar] [CrossRef]
- Dong, R.; Liu, Y.; Yang, Y.; Wang, H.; Xu, Y.; Zhang, Z. MSC-Derived Exosomes-Based Therapy for Peripheral Nerve Injury: A Novel Therapeutic Strategy. BioMed Res. Int. 2019, 2019, 6458237. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.-J.; Jung, G.N.; Park, W.-T.; Seo, M.-S.; Lee, G.W. Therapeutic Potential of Small Extracellular Vesicles Derived from Mesenchymal Stem Cells for Spinal Cord and Nerve Injury. Front. Cell Dev. Biol. 2023, 11, 1151357. [Google Scholar] [CrossRef]
- Gimble, J.M.; Katz, A.J.; Bunnell, B.A. Adipose-Derived Stem Cells for Regenerative Medicine. Circ. Res. 2007, 100, 1249–1260. [Google Scholar] [CrossRef]
- Chen, S.-H.; Kao, H.-K.; Wun, J.-R.; Chou, P.-Y.; Chen, Z.-Y.; Chen, S.-H.; Hsieh, S.-T.; Fang, H.-W.; Lin, F.-H. Thermosensitive Hydrogel Carrying Extracellular Vesicles from Adipose-Derived Stem Cells Promotes Peripheral Nerve Regeneration after Microsurgical Repair. APL Bioeng. 2022, 6, 046103. [Google Scholar] [CrossRef]
- Shen, H.; Yoneda, S.; Abu-Amer, Y.; Guilak, F.; Gelberman, R.H. Stem Cell-Derived Extracellular Vesicles Attenuate the Early Inflammatory Response after Tendon Injury and Repair. J. Orthop. Res. 2020, 38, 117–127. [Google Scholar] [CrossRef]
- Chen, J.; Li, X.; Liu, H.; Zhong, D.; Yin, K.; Li, Y.; Zhu, L.; Xu, C.; Li, M.; Wang, C. Bone Marrow Stromal Cell-Derived Exosomal Circular RNA Improves Diabetic Foot Ulcer Wound Healing by Activating the Nuclear Factor Erythroid 2-Related Factor 2 Pathway and Inhibiting Ferroptosis. Diabet. Med. 2022, 40, e15031. [Google Scholar] [CrossRef]
- Xia, B.; Gao, J.; Li, S.; Huang, L.; Zhu, L.; Ma, T.; Zhao, L.; Yang, Y.; Luo, K.; Shi, X.; et al. Mechanical Stimulation of Schwann Cells Promote Peripheral Nerve Regeneration via Extracellular Vesicle-Mediated Transfer of microRNA 23b-3p. Theranostics 2020, 10, 8974–8995. [Google Scholar] [CrossRef]
- Madison, R.D.; Robinson, G.A. Muscle-Derived Extracellular Vesicles Influence Motor Neuron Regeneration Accuracy. Neuroscience 2019, 419, 46–59. [Google Scholar] [CrossRef] [PubMed]
- Yeo, R.W.Y.; Lai, R.C.; Zhang, B.; Tan, S.S.; Yin, Y.; Teh, B.J.; Lim, S.K. Mesenchymal Stem Cell: An Efficient Mass Producer of Exosomes for Drug Delivery. Adv. Drug Deliv. Rev. 2013, 65, 336–341. [Google Scholar] [CrossRef]
- Gudbergsson, J.M.; Johnsen, K.B.; Skov, M.N.; Duroux, M. Systematic Review of Factors Influencing Extracellular Vesicle Yield from Cell Cultures. Cytotechnology 2016, 68, 579–592. [Google Scholar] [CrossRef] [PubMed]
- Grange, C.; Bussolati, B. Extracellular Vesicles in Kidney Disease. Nat. Rev. Nephrol. 2022, 18, 499–513. [Google Scholar] [CrossRef] [PubMed]
- Urzì, O.; Gasparro, R.; Ganji, N.R.; Alessandro, R.; Raimondo, S. Plant-RNA in Extracellular Vesicles: The Secret of Cross-Kingdom Communication. Membranes 2022, 12, 352. [Google Scholar] [CrossRef] [PubMed]
- Urzì, O.; Raimondo, S.; Alessandro, R. Extracellular Vesicles from Plants: Current Knowledge and Open Questions. Int. J. Mol. Sci. 2021, 22, 5366. [Google Scholar] [CrossRef]
- Karamanidou, T.; Tsouknidas, A. Plant-Derived Extracellular Vesicles as Therapeutic Nanocarriers. Int. J. Mol. Sci. 2021, 23, 191. [Google Scholar] [CrossRef]
- Barathan, M.; Zulpa, A.K.; Ng, S.L.; Lokanathan, Y.; Ng, M.H.; Law, J.X. Innovative Strategies to Combat 5-Fluorouracil Resistance in Colorectal Cancer: The Role of Phytochemicals and Extracellular Vesicles. Int. J. Mol. Sci. 2024, 25, 7470. [Google Scholar] [CrossRef]
- Chen, Q.; Zu, M.; Gong, H.; Ma, Y.; Sun, J.; Ran, S.; Shi, X.; Zhang, J.; Xiao, B. Tea Leaf-Derived Exosome-like Nanotherapeutics Retard Breast Tumor Growth by pro-Apoptosis and Microbiota Modulation. J. Nanobiotechnology 2023, 21, 6. [Google Scholar] [CrossRef]
- Tajik, T.; Baghaei, K.; Moghadam, V.E.; Farrokhi, N.; Salami, S.A. Extracellular Vesicles of Cannabis with High CBD Content Induce Anticancer Signaling in Human Hepatocellular Carcinoma. Biomed. Pharmacother. 2022, 152, 113209. [Google Scholar] [CrossRef]
- Raimondo, S.; Naselli, F.; Fontana, S.; Monteleone, F.; Lo Dico, A.; Saieva, L.; Zito, G.; Flugy, A.; Manno, M.; Di Bella, M.A.; et al. Citrus Limon-Derived Nanovesicles Inhibit Cancer Cell Proliferation and Suppress CML Xenograft Growth by Inducing TRAIL-Mediated Cell Death. Oncotarget 2015, 6, 19514–19527. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Yan, H.; Han, X.; Weng, L.; Wei, Q.; Sun, X.; Lu, W.; Wei, Q.; Ye, J.; Cai, X.; et al. Ginseng-Derived Nanoparticles Alter Macrophage Polarization to Inhibit Melanoma Growth. J. Immunother. Cancer 2019, 7, 326. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Viennois, E.; Prasad, M.; Zhang, Y.; Wang, L.; Zhang, Z.; Han, M.K.; Xiao, B.; Xu, C.; Srinivasan, S.; et al. Edible Ginger-Derived Nanoparticles: A Novel Therapeutic Approach for the Prevention and Treatment of Inflammatory Bowel Disease and Colitis-Associated Cancer. Biomaterials 2016, 101, 321–340. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Zhuang, X.; Deng, Z.-B.; Jiang, H.; Mu, J.; Wang, Q.; Xiang, X.; Guo, H.; Zhang, L.; Dryden, G.; et al. Targeted Drug Delivery to Intestinal Macrophages by Bioactive Nanovesicles Released from Grapefruit. Mol. Ther. 2014, 22, 522–534. [Google Scholar] [CrossRef]
- Li, N.; Wang, M.; Lyu, Z.; Shan, K.; Chen, Z.; Chen, B.; Chen, Y.; Hu, X.; Dou, B.; Zhang, J.; et al. Medicinal Plant-Based Drug Delivery System for Inflammatory Bowel Disease. Front. Pharmacol. 2023, 14, 1158945. [Google Scholar] [CrossRef]
- Chung, T.-W.; Li, S.; Lin, C.-C.; Tsai, S.-W. Antinociceptive and Anti-Inflammatory Effects of the Citrus Flavanone Naringenin. Ci Ji Yi Xue Za Zhi 2019, 31, 81–85. [Google Scholar] [CrossRef]
- Manchope, M.F.; Casagrande, R.; Verri, W.A. Naringenin: An Analgesic and Anti-Inflammatory Citrus Flavanone. Oncotarget 2017, 8, 3766–3767. [Google Scholar] [CrossRef]
- Hu, J.-C.; Zheng, C.-X.; Sui, B.-D.; Liu, W.-J.; Jin, Y. Mesenchymal Stem Cell-Derived Exosomes: A Novel and Potential Remedy for Cutaneous Wound Healing and Regeneration. World J. Stem Cells 2022, 14, 318–329. [Google Scholar] [CrossRef]
- Madison, R.D.; Robinson, G.A. Accuracy of Regenerating Motor Neurons: Influence of Diffusion in Denervated Nerve. Neuroscience 2014, 273, 128–140. [Google Scholar] [CrossRef]
- He, L.; Zhu, C.; Jia, J.; Hao, X.-Y.; Yu, X.-Y.; Liu, X.-Y.; Shu, M.-G. ADSC-Exos Containing MALAT1 Promotes Wound Healing by Targeting miR-124 through Activating Wnt/β-Catenin Pathway. Biosci. Rep. 2020, 40, BSR20192549. [Google Scholar] [CrossRef]
- Patel, N.A.; Moss, L.D.; Lee, J.-Y.; Tajiri, N.; Acosta, S.; Hudson, C.; Parag, S.; Cooper, D.R.; Borlongan, C.V.; Bickford, P.C. Long Noncoding RNA MALAT1 in Exosomes Drives Regenerative Function and Modulates Inflammation-Linked Networks Following Traumatic Brain Injury. J. Neuroinflammation 2018, 15, 204. [Google Scholar] [CrossRef]
- Kuang, L.; Zhang, C.; Li, B.; Deng, H.; Chen, R.; Li, G. Human Keratinocyte-Derived Exosomal MALAT1 Promotes Diabetic Wound Healing by Upregulating MFGE8 via microRNA-1914-3p. Int. J. Nanomedicine 2023, 18, 949–970. [Google Scholar] [CrossRef] [PubMed]
- Ross, K. MiR Equal than Others: MicroRNA Enhancement for Cutaneous Wound Healing. J. Cell. Physiol. 2021, 236, 8050–8059. [Google Scholar] [CrossRef] [PubMed]
- Mizbani, A.; Luca, E.; Rushing, E.J.; Krützfeldt, J. MicroRNA Deep Sequencing in Two Adult Stem Cell Populations Identifies miR-501 as a Novel Regulator of Myosin Heavy Chain during Muscle Regeneration. Development 2016, 143, 4137–4148. [Google Scholar] [CrossRef] [PubMed]
- Luca, E.; Turcekova, K.; Hartung, A.; Mathes, S.; Rehrauer, H.; Krützfeldt, J. Genetic Deletion of microRNA Biogenesis in Muscle Cells Reveals a Hierarchical Non-Clustered Network That Controls Focal Adhesion Signaling during Muscle Regeneration. Mol. Metab. 2020, 36, 100967. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, J.; Liu, S.; Yang, P.; Liang, Y.; Ma, J.; Mao, S.; Sun, C.; Yang, Y. Fibroblast Exosomal TFAP2C Induced by Chitosan Oligosaccharides Promotes Peripheral Axon Regeneration via the miR-132-5p/CAMKK1 Axis. Bioact. Mater. 2023, 26, 249–263. [Google Scholar] [CrossRef]
- Yang, J.; Wang, B.; Wang, Y.; Feng, C.; Chen, L.; Liu, Y.; Chen, X.; Dong, P. Exosomes Derived from Adipose Mesenchymal Stem Cells Carrying miRNA-22-3p Promote Schwann Cells Proliferation and Migration through Downregulation of PTEN. Dis. Markers 2022, 2022, 7071877. [Google Scholar] [CrossRef]
- Liu, W.; Wang, Y.; Gong, F.; Rong, Y.; Luo, Y.; Tang, P.; Zhou, Z.; Zhou, Z.; Xu, T.; Jiang, T.; et al. Exosomes Derived from Bone Mesenchymal Stem Cells Repair Traumatic Spinal Cord Injury by Suppressing the Activation of A1 Neurotoxic Reactive Astrocytes. J. Neurotrauma 2019, 36, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.; Li, G.; Li, D.; Huang, W.; Zhang, R.; Zhang, H.; Duan, Y.; Wang, B. hucMSC Derived Exosomes Promote Functional Recovery in Spinal Cord Injury Mice via Attenuating Inflammation. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 89, 194–204. [Google Scholar] [CrossRef]
- Butreddy, A.; Kommineni, N.; Dudhipala, N. Exosomes as Naturally Occurring Vehicles for Delivery of Biopharmaceuticals: Insights from Drug Delivery to Clinical Perspectives. Nanomaterials 2021, 11, 1481. [Google Scholar] [CrossRef]
- Rajput, A.; Varshney, A.; Bajaj, R.; Pokharkar, V. Exosomes as New Generation Vehicles for Drug Delivery: Biomedical Applications and Future Perspectives. Molecules 2022, 27, 7289. [Google Scholar] [CrossRef] [PubMed]
- Tenchov, R.; Sasso, J.M.; Wang, X.; Liaw, W.-S.; Chen, C.-A.; Zhou, Q.A. Exosomes—Nature’s Lipid Nanoparticles, a Rising Star in Drug Delivery and Diagnostics. ACS Nano 2022, 16, 17802–17846. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.; Xavier, J.; Kumar, N.; Ahmad, M.Z.; Ranjan, O.P. Exosomes as Natural Nanocarrier-Based Drug Delivery System: Recent Insights and Future Perspectives. 3 Biotech 2023, 13, 101. [Google Scholar] [CrossRef] [PubMed]
- De Jong, O.G.; Kooijmans, S.A.A.; Murphy, D.E.; Jiang, L.; Evers, M.J.W.; Sluijter, J.P.G.; Vader, P.; Schiffelers, R.M. Drug Delivery with Extracellular Vesicles: From Imagination to Innovation. Acc. Chem. Res. 2019, 52, 1761–1770. [Google Scholar] [CrossRef]
- Herrmann, I.K.; Wood, M.J.A.; Fuhrmann, G. Extracellular Vesicles as a Next-Generation Drug Delivery Platform. Nat. Nanotechnol. 2021, 16, 748–759. [Google Scholar] [CrossRef]
- Chen, C.; Wang, J.; Sun, M.; Li, J.; Wang, H.-M.D. Toward the Next-Generation Phyto-Nanomedicines: Cell-Derived Nanovesicles (CDNs) for Natural Product Delivery. Biomed. Pharmacother. 2022, 145, 112416. [Google Scholar] [CrossRef]
- Sun, D.; Zhuang, X.; Xiang, X.; Liu, Y.; Zhang, S.; Liu, C.; Barnes, S.; Grizzle, W.; Miller, D.; Zhang, H.-G. A Novel Nanoparticle Drug Delivery System: The Anti-Inflammatory Activity of Curcumin Is Enhanced When Encapsulated in Exosomes. Mol. Ther. 2010, 18, 1606–1614. [Google Scholar] [CrossRef] [PubMed]
- Pomatto, M.A.C.; Bussolati, B.; D’Antico, S.; Ghiotto, S.; Tetta, C.; Brizzi, M.F.; Camussi, G. Improved Loading of Plasma-Derived Extracellular Vesicles to Encapsulate Antitumor miRNAs. Mol. Ther. Methods Clin. Dev. 2019, 13, 133–144. [Google Scholar] [CrossRef]
- Li, J.; Xu, F.; Dai, Y.; Zhang, J.; Shi, Y.; Lai, D.; Sriboonvorakul, N.; Hu, J. A Review of Cyclodextrin Encapsulation and Intelligent Response for the Release of Curcumin. Polymers 2022, 14, 5421. [Google Scholar] [CrossRef]
- Santos, P.S.; Oliveira, T.C.; Júnior, L.M.R.; Figueiras, A.; Nunes, L.C.C. β-Caryophyllene Delivery Systems: Enhancing the Oral Pharmacokinetic and Stability. Curr. Pharm. Des. 2018, 24, 3440–3453. [Google Scholar] [CrossRef]
- Donthi, M.R.; Munnangi, S.R.; Krishna, K.V.; Saha, R.N.; Singhvi, G.; Dubey, S.K. Nanoemulgel: A Novel Nano Carrier as a Tool for Topical Drug Delivery. Pharmaceutics 2023, 15, 164. [Google Scholar] [CrossRef] [PubMed]
- Salem, H.F.; Kharshoum, R.M.; Abou-Taleb, H.A.; Naguib, D.M. Nanosized Nasal Emulgel of Resveratrol: Preparation, Optimization, in Vitro Evaluation and in Vivo Pharmacokinetic Study. Drug Dev. Ind. Pharm. 2019, 45, 1624–1634. [Google Scholar] [CrossRef] [PubMed]
- Vaz, G.R.; Carrasco, M.C.F.; Batista, M.M.; Barros, P.A.B.; Oliveira, M.d.C.; Muccillo-Baisch, A.L.; Yurgel, V.C.; Buttini, F.; Soares, F.A.A.; Cordeiro, L.M.; et al. Curcumin and Quercetin-Loaded Lipid Nanocarriers: Development of Omega-3 Mucoadhesive Nanoemulsions for Intranasal Administration. Nanomaterials 2022, 12, 1073. [Google Scholar] [CrossRef]
- Sköld, M.; Karlberg, A.-T.; Matura, M.; Börje, A. The Fragrance Chemical β-Caryophyllene—Air Oxidation and Skin Sensitization. Food Chem. Toxicol. 2006, 44, 538–545. [Google Scholar] [CrossRef]
- de Groot, A.C. Fragrances: Contact Allergy and Other Adverse Effects. Dermatitis 2020, 31, 13–35. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Li, Y.-M.; Wang, Z. Preserving Extracellular Vesicles for Biomedical Applications: Consideration of Storage Stability before and after Isolation. Drug Deliv. 2021, 28, 1501–1509. [Google Scholar] [CrossRef]
- Görgens, A.; Corso, G.; Hagey, D.W.; Jawad Wiklander, R.; Gustafsson, M.O.; Felldin, U.; Lee, Y.; Bostancioglu, R.B.; Sork, H.; Liang, X.; et al. Identification of Storage Conditions Stabilizing Extracellular Vesicles Preparations. J. Extracell. Vesicles 2022, 11, e12238. [Google Scholar] [CrossRef]
- Hovhannisyan, L.; Czechowska, E.; Gutowska-Owsiak, D. The Role of Non-Immune Cell-Derived Extracellular Vesicles in Allergy. Front. Immunol. 2021, 12, 702381. [Google Scholar] [CrossRef]
| Name CID CAS | Receptors/Channels and Signaling Pathways | Plants | Effects Other than Anti-Inflammation | Reference |
|---|---|---|---|---|
| Carnosic acid CID: 65126 CAS: 3650-09-7 | PPAR γ [73] | Salvia, rosemary | Neuroprotective (review [74]); Alzheimer’s disease, Parkinson’s disease, long-COVID (review [75]) | [73,74,75] |
| Carvacrol CID: 10364 CAS: 499-75-2 | TRPA1, TRPV3 [76], PPARα and PPARγ [77], GABAA (in mice [78]) Antagonist to nicotinic acetylcholine receptors (nAChRs) [79], antagonist to TRPM7 [80,81] | Oregano, thyme | [76,77,78,79,80,81] | |
| β-caryophyllene CID: 5281515 CAS: 87-44-5 | CB2 [36]; PPARα directly [46] and PPARγ indirectly [47,48,49] | Copaiba, black pepper, rosemary and others (review [82]) | Anti-carcinogenesis [83], analgesic [40,84], facilitates wound healing, cell proliferation/migration (in vivo in mice and in vitro [17]) | [17,36,40,82,83,84] |
| Citral (geranial) CID: 638011 CAS: 5392-40-5 | GABAA, 5-HT1A [85]; TRPV1, TRPM8 [86] | Lemon grass (Cymbopogon citratus), lemon | Anti-nociceptive (in mouse and rat model) [86]; anxiolytic (in mouse model) [85] | [85,86] |
| Curcumin CID: 969516 CAS: 458-37-7 | Potential ligand of Aryl hydrocarbon (AhR) [70], GPR55 [87], PPARγ [69,88], TRPA1 [71,89], and others | Turmeric (Curcuma longa) | Anti-carcinogenesis, anti-viral, antiarthritic, anti-amyloid, antioxidant [72] | Review, in vitro, and in vivo animal studies [69,70,71,72,87,88,89] |
| Epigallocatechin-3-gallate (EGCG) CID: 65064 CAS: 989-51-5 | 67LR (67-kDa laminin receptor) [90,91] | Tea plant (Camellia sinensis) | Antiviral [92,93], antibacterial [94], anti-oxidant, anti-cancer [95,96] | In vitro studies and reviews [90,91,92,93,94,95,96] |
| Eugenol CID: 3314 CAS: 97-53-0 | TRPV1 [97]; PPARγ [98] | Clove | Antidiabetic, antioxidant, anti-cancer [98] | In vitro study and a study using C. elegans [97,98] |
| Gingerol = group of phytochemicals such as 6-gingerol (CID 442793; CAS 23513-14-6), 10-gingerol (CID 168115, CAS 23513-15-7), and others | TRPV1 [99] | Ginger | Anti-oxidant, antibacterial, anti-cancer [99]; immunomodulatory, neuroprotective, respiratory protective, antiobesity, antidiabetic, antinausea [100]; antiallergic [101] | Reviews [99,100,101] |
| D-Limonene CID: 440917 CAS: 5989-27-5 | A2A adenosine receptor [102,103]; TRPA1 [104] | Bitter orange | Anti-carcinogenesis; anxiolytic [105]; topical application cause pain (algesic) through TRPA1 | In vivo and in vitro studies [102,103,104,105] |
| Linalool CID: 6549 CAS: 78-70-6 | GABAA [106]; hypothalamic orexin neurons involved [107,108] | Mint, lavender | Anxiolytic effects [106]; analgesic [108]; antinociceptive | In vivo studies using mice [106,107,108] |
| Perillyl alcohol CID: 10819 CAS: 536-59-4 | Ras/MAPK pathway inhibitor [109] | Lavender, citrus fruits, peppermint, spearmint, cherries | Anti-carcinogenesis [110,111], anti-nociceptive [112] | In vivo, in vitro, and review [109,110,111,112] |
| Resveratrol CID: 445154 CAS: 501-36-0 | Estrogen receptor α [113,114,115], TAS2R50 (bitter taste-sensing receptor) [116], PPARα and PPARγ antagonist [117]; but another study reports PPARγ agonist [118] | Grapes, peanuts, blueberries, cranberries | Amti-carcinogenesis [115], antiviral, antioxidant | In vitro studies and reviews [113,114,115,116,117,118] |
| Theaflavin CID: 135403798 CAS: 4670-05-7 | Bitter taste receptor hTAS2R39 hTAS2R14 [119] | Tea plant (Camellia sinensis) | Anti-viral [92], antibacterial, anti-diabetic, anti-cancer, [120,121] | In vitro and reviews [92,119,120,121] |
| Phytochemicals | Effects Found | References |
|---|---|---|
| Curcumin | Administration of curcumin suppressed expression of proinflammatory cytokines TNFα, IL-1β, and NF-kb and the expression of glial fibrillary acidic protein, suggesting reduced glial scar formation, in a rat model of spinal cord injury [167]. Transgenic mice with autoimmune desease (NZBWF1) administered with curcumin showed less renal injury [168] | [167,168] |
| Piperine | Piperine possesses anti-inflammatory effects, neuroprotective effects, anti-microbial effects, and anti-cancer effects (review). It also has bioavailability-enhancing effects (review). Piperine also suppressed cytochrome P450 in a mouse model [169] and had hepatoprotective effects on thioacetamide-induced liver fibrosis in mice [170]. Piperine administration suppressed the experimentally induced pancreatitis by administration of cerulein in mice [171] | [169,170,171,172] |
| Resveratrol | Anti-inflammatory effects and neuroprotective effects in rats with cuprizone-induced demyelination [173] | [173] |
| EGCG | Anti-viral effects on SARS-CoV-2 [58,92], anti-bacterial effects [94], anti-cancer effects [95] | [58,92,94,95] |
| Quercetin | Anti-inflammatory effects, antioxidant effects, suppression of nociceptive and pathological pain (review [174]). Quercetin suppressed COX-2 expression in RAW264.7 cells inflamed with lipopolysaccharides (LPS) [175] | [174,175] |
| Curcumin + piperine | Co-administration improved the lipid profile of metabolic syndrome [163]; suppressed cancer cell proliferation through inhibitory effects on Wnt signaling mTORC1 signaling [164] | [163,164,165] |
| Curcumin + EGCG | Suppresses cancer cell proliferation | [164,166,176,177] |
| Curcumin + resveratrol | Anti-cancer; suppresses cancer cell survival | [164] |
| Curcumin + quercetin | Suppresses cancer cell survival | [164] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koyama, S.; Weber, E.L.; Heinbockel, T. Possible Combinatorial Utilization of Phytochemicals and Extracellular Vesicles for Wound Healing and Regeneration. Int. J. Mol. Sci. 2024, 25, 10353. https://doi.org/10.3390/ijms251910353
Koyama S, Weber EL, Heinbockel T. Possible Combinatorial Utilization of Phytochemicals and Extracellular Vesicles for Wound Healing and Regeneration. International Journal of Molecular Sciences. 2024; 25(19):10353. https://doi.org/10.3390/ijms251910353
Chicago/Turabian StyleKoyama, Sachiko, Erin L. Weber, and Thomas Heinbockel. 2024. "Possible Combinatorial Utilization of Phytochemicals and Extracellular Vesicles for Wound Healing and Regeneration" International Journal of Molecular Sciences 25, no. 19: 10353. https://doi.org/10.3390/ijms251910353
APA StyleKoyama, S., Weber, E. L., & Heinbockel, T. (2024). Possible Combinatorial Utilization of Phytochemicals and Extracellular Vesicles for Wound Healing and Regeneration. International Journal of Molecular Sciences, 25(19), 10353. https://doi.org/10.3390/ijms251910353







