Insights into the Hormone-Regulating Mechanism of Adventitious Root Formation in Softwood Cuttings of Cyclocarya paliurus and Optimization of the Hormone-Based Formula for Promoting Rooting
Abstract
:1. Introduction
2. Results
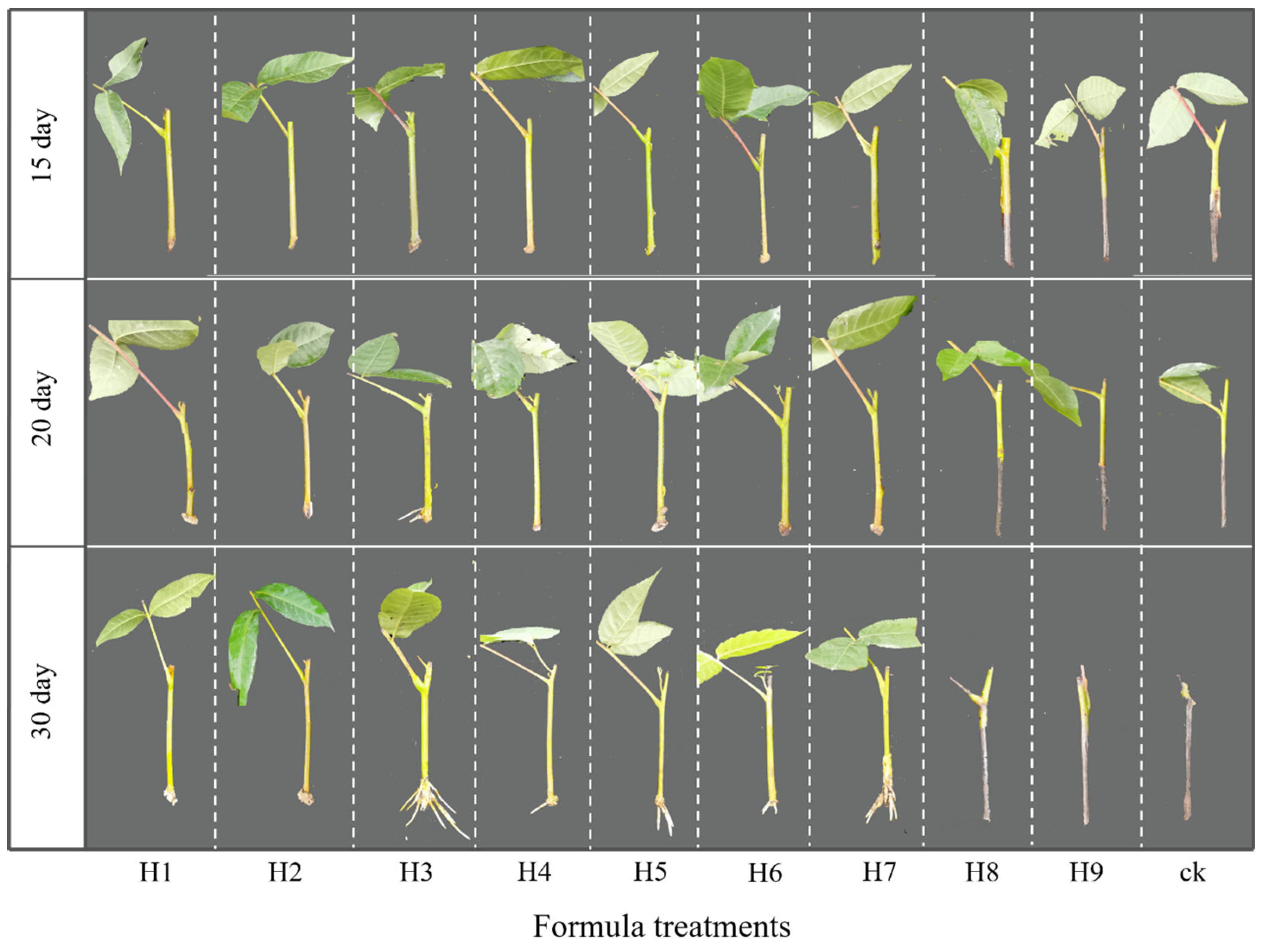
2.1. Variations in the Rooting Process and Related Rooting Parameters
2.2. Dynamics of Endogenous Hormones during the AR Formation Process
2.3. Overall Assessment of Transcriptome Profiles during the AR Formation Process
2.4. DEGs Involved in Phytohormone Signal Transduction Pathways
2.5. Co-Expression Network Analysis of Weight Genes
2.6. qRT-PCR Verification for Selected Genes
3. Discussion
3.1. Effects of Different Hormone-Based Formulas on Rooting Efficiency
3.2. Relationship between Endogenous Hormones and AR Formation
3.3. Regulatory Mechanisms of Endogenous Hormones during the AR Formation Process
4. Materials and Methods
4.1. Plant Materials
4.2. Experimental Design and Treatment Description
4.3. Rooting Process Observation and Sampling
4.4. Assessment of the Rooting Rate and Rooting Quality Indexes
4.5. Measurement of Endogenous Hormones
4.6. RNA Extraction and Transcriptomics Analysis
4.7. Quantitative Real-Time (qRT-PCR) Validation
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhang, S.; He, J.; Li, J.; He, H.; He, Y.; Wang, X.; Shu, H.; Zhang, J.; Xu, D.; Zou, K. Triterpenoid compounds from Cyclocarya paliurus: A review of their phytochemistry, quality control, pharmacology, and structure–activity relationship. Am. J. Chin. Med. 2023, 51, 2041. [Google Scholar] [CrossRef] [PubMed]
- Manchester, S.R.; Chen, Z.; Lu, A.; Uemura, K. Eastern Asian endemic seed plant genera and their paleogeographic history throughout the Northern Hemisphere. J. Syst. Evol. 2009, 47, 1–42. [Google Scholar] [CrossRef]
- Qin, J.; Yue, X.; Fang, S.; Qian, M.; Zhou, S.; Shang, X.; Yang, W. Responses of nitrogen metabolism, photosynthetic parameter and growth to nitrogen fertilization in Cyalocarya paliurus. For. Ecol. 2021, 502, 119715. [Google Scholar]
- Zhou, M.; Chen, P.; Lin, Y.; Fang, S.; Shang, X. A comprehensive assessment of bioactive metabolites, antioxidant and antiproliferative activities of Cyclocarya paliurus (Batal.) Iljinskaja Leaves. Forests 2019, 10, 625. [Google Scholar] [CrossRef]
- Fang, S.; Sun, D.; Shang, X.; Fu, X.; Yang, W. Variation in radial growth and wood density of Cyclocarya paliurus across its natural distribution. New For. 2020, 51, 453–467. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Z.; Fang, S.; Liu, Y.; Shang, X. Integrative analysis of metabolome and transcriptome reveals molecular regulatory mechanism of flavonoid biosynthesis in Cyclocarya paliurus under salt stress. Ind. Crops Prod. 2021, 170, 113823. [Google Scholar] [CrossRef]
- Zhou, M.; Chen, P.; Shang, X.; Yang, W.; Fang, S. Genotype–environment interactions for tree growth and leaf phytochemical content of Cyclocarya paliurus (Batal.) Iljinskaja. Forests 2021, 12, 735. [Google Scholar] [CrossRef]
- Kakar, M.; Naveed, M.; Saeed, M.; Zhao, S.; Rasheed, M.; Firdoos, S.; Manzoor, R.; Deng, Y.; Dai, R. A review on structure, extraction, and biological activities of polysaccharides isolated from Cyclocarya paliurus (Batalin) Iljinskaja. Int. J. Biol. Macromol. 2020, 156, 420–429. [Google Scholar] [CrossRef]
- Lan, L.; Xu, Z.; Sun, C.; Fang, S. Evaluation on germplasm resources of Cyclocarya paliurus and its oriented selection of superior families and trees. For. Res. 2022, 35, 42–51. (In Chinese) [Google Scholar]
- Thomas, A.; Brauer, D.; Sauer, T.; Coggeshall, M.V.; Ellersieck, M. Cultivar influences early rootstock and scion survival of grafted black walnut. J. Amer. Pomolog. Soc. 2008, 62, 3–12. [Google Scholar]
- Stevens, M.E.; Pijut, P.M. Origin of adventitious roots in black walnut (Juglans nigra) softwood cuttings rooted under optimized conditions in a fog chamber. New For. Tree Physiol. 2017, 48, 685–697. [Google Scholar] [CrossRef]
- Li, N.; Zhu, P.; Feng, C.; Wen, M.; Fang, S.; Shang, X. Variation in physiological characteristics of rootstock-scion and its relationship to graft compatibility during the grafting union process of Cyclocarya paliurus. J. Nanjing For. Univ. 2021, 45, 13–20. (In Chinese) [Google Scholar]
- Yang, W.; Zhuang, J.; Ding, S.; Zhang, M.; Tian, Y.; Wan, S.; Fang, S. Study on cutting cultivation technology and rooting mechanism of Cyclocarya paliurus. Ecol. Chem. Eng. S 2022, 29, 379–389. [Google Scholar] [CrossRef]
- Wan, S.Y.; Tian, Y.; Yang, W.; Fang, S. Effects of different hormone formulas on nutrient substance, enzyme activity and rooting in hardwood cutting of Cyclocarya paliurus. Non-Wood. For. Res. 2023, 41, 217–224, 300. (In Chinese) [Google Scholar]
- Druege, U.; Hilo, A.; Pérez-Pérez, J.M.; Klopotek, Y.; Acosta, M.; Shahinnia, F.; Zerche, S.; Franken, P.; Hajirezaei, M.R. Molecular and physiological control of adventitious rooting in cuttings: Phytohormone action meets resource allocation. Ann. Bot. 2019, 123, 929–949. [Google Scholar] [CrossRef]
- Villacorta-Martín, C.; Belén Sánchez-García, A.; Villanova, J.; Cano, A.; Van De Rhee, M.; De Haan, J.; Acosta, M.; Passarinho, P.; Manuel Pérez-Pérez, J. Gene expression profiling during adventitious root formation in carnation stem cuttings. BMC Genom. 2015, 16, 789. [Google Scholar] [CrossRef] [PubMed]
- Druege, U.; Franken, P.; Hajirezaei, M.R. Plant hormone homeostasis, signaling, and function during adventitious root formation in cuttings. Front. Plant Sci. 2016, 7, 381. [Google Scholar] [CrossRef]
- Ren, H.; Hu, H.; Luo, X.; Zhang, C.; Li, X.; Li, P.; Li, W.; Khawar, A.; Sun, X.; Ren, Z.; et al. Dynamic changes of phytohormone signaling in the base of Taxus media stem cuttings during adventitious root formation. Sci. Hortic. 2019, 246, 338–346. [Google Scholar] [CrossRef]
- Wang, Y.; Pang, D.; Ruan, L.; Liang, J.; Zhang, Q.; Qian, Y.; Zhang, Y.; Bai, P.; Wu, L.; Cheng, H.; et al. Integrated transcriptome and hormonal analysis of naphthalene acetic acid-induced adventitious root formation of tea cuttings (Camellia sinensis). BMC Plant Biol. 2022, 22, 319. [Google Scholar] [CrossRef]
- Bustillo-Avendaño, E.; Ibáñez, S.; Sanz, O.; Barros, J.A.S.; Gude, I.; Perianez-Rodriguez, J.; Micol, J.L.; del Pozo, J.C.; Moreno-Risueno, M.A.; Pérez-Pérez, J.M. Regulation of hormonal control, cell reprogramming, and patterning during de novo root organogenesis. Plant Physiol. 2018, 176, 1709–1727. [Google Scholar] [CrossRef]
- Pacurar, D.I.; Perrone, I.; Bellini, C. Auxin is a central player in the hormone cross-talks that control adventitious rooting. Physiol. Plant. 2014, 151, 83–96. [Google Scholar] [CrossRef]
- da Costa, C.T.; de Almeida, M.R.; Ruedell, C.M.; Schwambach, J.; Maraschin, F.S.; Fett-Neto, A.G. When stress and development go hand in hand: Main hormonal controls of adventitious rooting in cuttings. Front. Plant Sci. 2013, 4, 133. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Zhang, F.; Friml, J.; Ding, Z. Auxin signaling: Research advances over the past 30 years. J. Integr. Plant Biol. 2022, 64, 371–392. [Google Scholar] [CrossRef] [PubMed]
- Ahkami, A.H.; Melzer, M.; Ghaffari, M.R.; Pollmann, S.; Ghorbani Javid, M.; Shahinnia, F.; Hajirezaei, M.R.; Druege, U. Distribution of indole-3-acetic acid in Petunia hybrida shoot tip cuttings and relationship between auxin transport, carbohydrate metabolism and adventitious root formation. Planta 2013, 238, 499–517. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, L.; Mongelard, G.; Floková, K.; Pǎcurar, D.I.; Novák, O.; Staswick, P.; Kowalczyk, M.; Păcurar, M.; Demailly, H.; Geiss, G.; et al. Auxin controls Arabidopsis adventitious root initiation by regulating jasmonic acid homeostasis. Plant Cell 2012, 24, 2515–2527. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, Y.; Liu, X. Study on young branch cutting seedling technology of Cyclocarya paliurus. Prot. For. Sci. Technol. 2022, 3, 24–26. [Google Scholar]
- Liu, J.; Moore, S.; Chen, C.; Lindsey, K. Crosstalk complexities between auxin, cytokinin, and ethylene in Arabidopsis root development: From experiments to systems modeling, and back again. Mol. Plant 2017, 10, 1480–1496. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Opio, P.; Wang, S.; Ohkawa, K.; Kondo, S.; Maejima, T.; Ohara, H. Association of auxin, cytokinin, abscisic acid, and plant peptide response genes during adventitious root formation in Marubakaido apple rootstock (Malus prunifolia Borkh. var. ringo Asami). Acta. Physiol. Plant 2019, 41, 41. [Google Scholar] [CrossRef]
- Lakehal, A.; Bellini, C. Control of adventitious root formation: Insights into synergistic and antagonistic hormonal interactions. Physiol. Plant. 2019, 165, 90–100. [Google Scholar] [CrossRef]
- Ptošková, K.; Szecówka, M.; Jaworek, P.; Tarkowská, D.; Petřík, I.; Pavlović, I.; Novák, O.; Thomas, S.G.; Phillips, A.L.; Hedden, P. Changes in the concentrations and transcripts for gibberellins and other hormones in a growing leaf and roots of wheat seedlings in response to water restriction. BMC Plant Biol. 2022, 22, 284. [Google Scholar] [CrossRef]
- Hernández-García, J.; Briones-Moreno, A.; Blázquez, M.A. Origin and evolution of gibberellin signaling and metabolism in plants. Semin. Cell. Dev. Biol. 2021, 109, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Jing, W.; Zhang, S.; Fan, Y.; Deng, Y.; Wang, C.; Lu, J.; Sun, X.; Ma, N.; Shahid, M.O.; Li, Y.; et al. Molecular Evidences for the Interactions of Auxin, Gibberellin, and Cytokinin in Bent Peduncle Phenomenon in Rose (Rosa sp.). Int. J. Mol. Sci. 2020, 21, 1360. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Kong, W.; Wang, Q.; Fu, X. Genome-wide identification MIKC-Type MADS-Box gene family and their roles during development of floral buds in wheel wingnut (Cyclocarya paliurus). Int. J. Mol. Sci. 2021, 22, 10128. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Israeli, A.; Ori, N.; Sun, T.P. The interaction between DELLA and ARF/IAA mediates crosstalk between gibberellin and auxin signaling to control fruit initiation in tomato. Plant Cell 2018, 30, 1710–1728. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Guo, Z.; Zhang, R.; Li, Y.; Cao, L.; Liang, Y.; Huang, L. Auxin type, auxin concentration, and air and substrate temperature difference play key roles in the rooting of juvenile hardwood pecan cuttings. Hort. Technol. 2015, 25, 209–213. [Google Scholar] [CrossRef]
- Guo, C.; Shangguan, X.; Jiang, Y.; Yang, W.; Zhang, J.W. A study on the effects of growth regulators on cutting techniques of Cyclocarya paliurus. Acta. Agri. Univ. Jiangxiensis 2008, 30, 275–278. (In Chinese) [Google Scholar]
- Zhang, G. A study on soft cutting propagation technique of Cyclocarya paliurus in the southern region of Fujian province. Anhui Agri. Sci. Bull. 2016, 22, 78–80. [Google Scholar]
- Du, W.; Cheng, J. Morphological and anatomical observation on cortex rooting process of mulberry (Morus L.) greenwood cuttings. Sci. Seri. 2014, 40, 13–17. [Google Scholar]
- Liu, G.; Zhao, J.; Liao, T.; Wang, Y.; Guo, L.; Yao, Y.; Cao, J. Histological dissection of cutting-inducible adventitious rooting in Platycladus orientalis reveals developmental endogenous hormonal homeostasis. Ind. Crops. Prod. 2021, 170, 113817. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, J.; Xia, G.; Huang, Y.; Huang, J.; Wang, Z. Research of lignified cuttings and rooting mechanism of Carya illinoensis. J. Anhui Agric. Univ. 2014, 41, 203–208. (In Chinese) [Google Scholar]
- Otiende, M.A.; Fricke, K.; Nyabundi, J.O.; Ngamau, K.; Hajirezaei, M.R.; Druege, U. Involvement of the auxin-cytokinin homeostasis in adventitious root formation of rose cuttings as affected by their nodal position in the stock plant. Planta 2021, 254, 65. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, O. Research status of rooting mechanism of peach hardwood cutting. Plant Physiol. J. 2019, 55, 1595–1606. [Google Scholar]
- Quan, J.; Ni, R.; Wang, Y.; Sun, J.; Ma, M.; Bi, H. Effects of different growth regulators on the rooting of Catalpa bignonioides softwood cuttings. Life 2022, 12, 1231. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhou, T.; Zhang, C.; Zheng, W.; Li, J.; Jiang, W.; Xiao, C.; Wei, D.; Yang, C.; Xu, R.; et al. Gibberellin disturbs the balance of endogenesis hormones and inhibits adventitious root development of Pseudostellaria heterophylla through regulating gene expression related to hormone synthesis. Saudi. J. Bio. Sci. 2021, 28, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Busov, V.; Meilan, R.; Pearce, D.W.; Rood, S.B.; Ma, C.; Tschaplinski, T.J.; Strauss, S.H. Transgenic modification of gai or rgl1 causes dwarfing and alters gibberellins, root growth, and metabolite profiles in Populus. Planta 2006, 224, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Steffens, B.; Wang, J.; Sauter, M. Interactions between ethylene, gibberellin and abscisic acid regulate emergence and growth rate of adventitious roots in deepwater rice. Planta 2006, 223, 604–612. [Google Scholar] [CrossRef]
- Chen, R.; Zhao, D.; Huang, X. Transcriptome analysis of easy- and hard-to-root tea plants uncovers roles for CsGH3.2 and CsGH3.3 in adventitious root formation. Plant Cell Tissue Organ Cult. 2022, 150, 385–398. [Google Scholar] [CrossRef]
- Yuan, H.; Zhao, K.; Lei, H.; Shen, X.; Liu, Y.; Liao, X.; Li, T. Genome-wide analysis of the GH3 family in apple (Malus × domestica). BMC Genom. 2013, 14, 297. [Google Scholar] [CrossRef]
- Hu, Y.; Omary, M.; Hu, Y.; Doron, O.; Hoermayer, L.; Chen, Q.; Megides, O.; Chekli, O.; Ding, Z.; Friml, J.; et al. Cell kinetics of auxin transport and activity in Arabidopsis root growth and skewing. Nat. Commun. 2021, 12, 1657. [Google Scholar] [CrossRef]
- Luo, J.; Zhou, J.; Zhang, J. Aux/IAA gene family in plants: Molecular structure, regulation, and function, Int. J. Mol. Sci. 2018, 19, 259. [Google Scholar] [CrossRef]
- Chandler, J.W.; Werr, W. Cytokinin-auxin crosstalk in cell type specification. Trends Plant Sci. 2015, 20, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Kang, X.; Lei, W.; Yao, X.; Zou, L.; Zhang, D.; Lin, H. SHY2 as a node in the regulation of root meristem development by auxin, brassinosteroids, and cytokinin. J. Integr. Plant Biol. 2020, 6, 1500–1517. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xu, P.; Zhang, B.; Song, Y.; Wen, S.; Bai, Y.; Ji, L.; Lai, Y.; He, G.; Zhang, D. Paclobutrazol promotes root development of difficult-to-root plants by coordinating auxin and abscisic acid signaling pathways in Phoebe bournei. Int. J. Mol. Sci. 2023, 24, 3753. [Google Scholar] [CrossRef]
- Wong, J.; Spartz, A.K.; Park, M.; Du, M.; Gray, W. Mutation of a conserved motif of PP2C.D phosphatases confers SAUR immunity and constitutive activity. Plant Physiol. 2019, 181, 353–366. [Google Scholar] [CrossRef] [PubMed]
- Emenecker, R.J.; Strader, L.C. Auxin-Abscisic acid interactions in plant growth and development. Biomolecules 2008, 10, 281. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Welti, R.; Wang, X. Quantitative analysis of major plant hormones in crude plant extracts by high-performance liquid chromatography-mass spectrometry. Nat. Protoc. 2010, 5, 986–992. [Google Scholar] [CrossRef]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. Fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, 884–890. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, J.; Wang, S.; Zhang, H.; Liu, Y.; Yang, M. Integrative transcriptomic and metabolomic analyses reveal the flavonoid biosynthesis of Pyrus hopeiensis flowers under cold stress. Hortic. Plant J. 2023, 9, 395–413. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real time quantitative PCR and the 2−ΔΔCT. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]








Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, Y.; Yang, W.; Wan, S.; Fang, S. Insights into the Hormone-Regulating Mechanism of Adventitious Root Formation in Softwood Cuttings of Cyclocarya paliurus and Optimization of the Hormone-Based Formula for Promoting Rooting. Int. J. Mol. Sci. 2024, 25, 1343. https://doi.org/10.3390/ijms25021343
Tian Y, Yang W, Wan S, Fang S. Insights into the Hormone-Regulating Mechanism of Adventitious Root Formation in Softwood Cuttings of Cyclocarya paliurus and Optimization of the Hormone-Based Formula for Promoting Rooting. International Journal of Molecular Sciences. 2024; 25(2):1343. https://doi.org/10.3390/ijms25021343
Chicago/Turabian StyleTian, Yuan, Wanxia Yang, Shiying Wan, and Shengzuo Fang. 2024. "Insights into the Hormone-Regulating Mechanism of Adventitious Root Formation in Softwood Cuttings of Cyclocarya paliurus and Optimization of the Hormone-Based Formula for Promoting Rooting" International Journal of Molecular Sciences 25, no. 2: 1343. https://doi.org/10.3390/ijms25021343





