Exploring the Combined Action of Adding Pertuzumab to Branded Trastuzumab versus Trastuzumab Biosimilars for Treating HER2+ Breast Cancer
Abstract
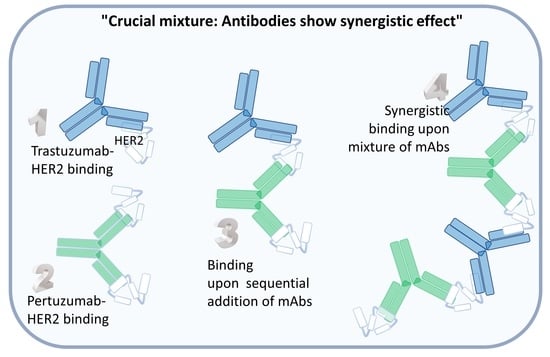
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Swain, S.M.; Shastry, M.; Hamilton, E. Targeting HER2-Positive Breast Cancer: Advances and Future Directions. Nat. Rev. Drug Discov. 2023, 22, 101–126. [Google Scholar] [CrossRef]
- Giordano, S.H.; Temin, S.; Davidson, N.E. Systemic Therapy for Patients with Advanced Human Epidermal Growth Factor Receptor 2-Positive Breast Cancer: ASCO Clinical Practice Guideline Update Summary. J. Oncol. Pract. 2018, 14, 501–504. [Google Scholar] [CrossRef] [PubMed]
- Hudis, C.A. Trastuzumab—Mechanism of Action and Use in Clinical Practice. N. Engl. J. Med. 2007, 357, 39–51. [Google Scholar] [CrossRef] [PubMed]
- De Mattos-Arruda, L.; Cortes, J. Use of Pertuzumab for the Treatment of HER2-Positive Metastatic Breast Cancer. Adv. Ther. 2013, 30, 645–658. [Google Scholar] [CrossRef] [PubMed]
- Von Minckwitz, G.; Procter, M.; De Azambuja, E.; Zardavas, D.; Benyunes, M.; Viale, G.; Suter, T.; Arahmani, A.; Rouchet, N.; Clark, E.; et al. Adjuvant Pertuzumab and Trastuzumab in Early Her2-Positive Breast Cancer. N. Engl. J. Med. 2017, 377, 122–131. [Google Scholar] [CrossRef]
- Jagosky, M.; Tan, A.R. Combination of Pertuzumab and Trastuzumab in the Treatment of Her2-Positive Early Breast Cancer: A Review of the Emerging Clinical Data. Breast Cancer Targets Ther. 2021, 13, 393–407. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Fang, Y.; Li, Y.; Li, Y.; Qi, L.; Wang, X. Pertuzumab Combined with Trastuzumab Compared to Trastuzumab in the Treatment of HER2-Positive Breast Cancer: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Front. Oncol. 2022, 12, 894861. [Google Scholar] [CrossRef] [PubMed]
- Baselga, J.; Cortés, J.; Kim, S.-B.; Im, S.-A.; Hegg, R.; Im, Y.-H.; Roman, L.; Pedrini, J.L.; Pienkowski, T.; Knott, A.; et al. Pertuzumab plus Trastuzumab plus Docetaxel for Metastatic Breast Cancer. N. Engl. J. Med. 2012, 366, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Swain, S.M.; Baselga, J.; Kim, S.-B.; Ro, J.; Semiglazov, V.; Campone, M.; Ciruelos, E.; Ferrero, J.-M.; Schneeweiss, A.; Heeson, S.; et al. Pertuzumab, Trastuzumab, and Docetaxel in HER2-Positive Metastatic Breast Cancer. N. Engl. J. Med. 2015, 372, 724–734. [Google Scholar] [CrossRef]
- Krop, I.E.; Modi, S.; LoRusso, P.M.; Pegram, M.; Guardino, E.; Althaus, B.; Lu, D.; Strasak, A.; Elias, A. Phase 1b/2a Study of Trastuzumab Emtansine (T-DM1), Paclitaxel, and Pertuzumab in HER2-Positive Metastatic Breast Cancer. Breast Cancer Res. 2016, 18, 34. [Google Scholar] [CrossRef]
- Miller, K.D.; Diéras, V.; Harbeck, N.; Andre, F.; Mahtani, R.L.; Gianni, L.; Albain, K.S.; Crivellari, D.; Fang, L.; Michelson, G.; et al. Phase IIa Trial of Trastuzumab Emtansine with Pertuzumab for Patients with Human Epidermal Growth Factor Receptor 2-Positive, Locally Advanced, or Metastatic Breast Cancer. J. Clin. Oncol. 2014, 32, 1437–1444. [Google Scholar] [CrossRef]
- Urruticoechea, A.; Rizwanullah, M.; Im, S.-A.; Sánchez Ruiz, A.C.; Láng, I.; Tomasello, G.; Douthwaite, H.; Crnjevic, T.B.; Heeson, S.; Eng-Wong, J.; et al. Randomized Phase III Trial of Trastuzumab plus Capecitabine with or without Pertuzumab in Patients with Human Epidermal Growth Factor Receptor 2-Positive Metastatic Breast Cancer Who Experienced Disease Progression during or after Trastuzumab-Based Therapy. J. Clin. Oncol. 2017, 35, 3030–3038. [Google Scholar] [CrossRef] [PubMed]
- Rimawi, M.; Ferrero, J.-M.; De La Haba-Rodriguez, J.; Poole, C.; De Placido, S.; Osborne, C.K.; Hegg, R.; Easton, V.; Wohlfarth, C.; Arpino, G. First-Line Trastuzumab plus an Aromatase Inhibitor, with or without Pertuzumab, in Human Epidermal Growth Factor Receptor 2-Positive and Hormone Receptor-Positive Metastatic or Locally Advanced Breast Cancer (PERTAIN): A Randomized, Open-Label Phase II Trial. J. Clin. Oncol. 2018, 36, 2826–2835. [Google Scholar] [CrossRef] [PubMed]
- Perez, E.A.; Barrios, C.; Eiermann, W.; Toi, M.; Im, Y.-H.; Conte, P.; Martin, M.; Pienkowski, T.; Pivot, X.B.; Burris, H.A.; et al. Trastuzumab Emtansine with or without Pertuzumab versus Trastuzumab with Taxane for Human Epidermal Growth Factor Receptor 2–Positive Advanced Breast Cancer: Final Results from MARIANNE. Cancer 2019, 125, 3974–3984. [Google Scholar] [CrossRef] [PubMed]
- Patel, T.A.; Ensor, J.E.; Creamer, S.L.; Boone, T.; Rodriguez, A.A.; Niravath, P.A.; Darcourt, J.G.; Meisel, J.L.; Li, X.; Zhao, J.; et al. A Randomized, Controlled Phase II Trial of Neoadjuvant Ado-Trastuzumab Emtansine, Lapatinib, and Nab-Paclitaxel versus Trastuzumab, Pertuzumab, and Paclitaxel in HER2-Positive Breast Cancer (TEAL Study). Breast Cancer Res. 2019, 21, 100. [Google Scholar] [CrossRef] [PubMed]
- Swain, S.M.; Miles, D.; Kim, S.-B.; Im, Y.-H.; Im, S.-A.; Semiglazov, V.; Ciruelos, E.; Schneeweiss, A.; Loi, S.; Monturus, E.; et al. Pertuzumab, Trastuzumab, and Docetaxel for HER2-Positive Metastatic Breast Cancer (CLEOPATRA): End-of-Study Results from a Double-Blind, Randomised, Placebo-Controlled, Phase 3 Study. Lancet Oncol. 2020, 21, 519–530. [Google Scholar] [CrossRef]
- Xu, B.; Li, W.; Zhang, Q.; Li, Q.; Wang, X.; Li, H.; Sun, T.; Yin, Y.; Zheng, H.; Feng, J.; et al. Pertuzumab, Trastuzumab, and Docetaxel for Chinese Patients with Previously Untreated HER2-Positive Locally Recurrent or Metastatic Breast Cancer (PUFFIN): Final Analysis of a Phase III, Randomized, Double-Blind, Placebo-Controlled Study. Breast Cancer Res. Treat. 2023, 197, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Phillips, G.D.L.; Fields, C.T.; Li, G.; Dowbenko, D.; Schaefer, G.; Miller, K.; Andre, F.; Burris III, H.A.; Albain, K.S.; Harbeck, N.; et al. Dual Targeting of HER2-Positive Cancer with Trastuzumab Emtansine and Pertuzumab: Critical Role for Neuregulin Blockade in Antitumor Response to Combination Therapy. Clin. Cancer Res. 2014, 20, 456–468. [Google Scholar] [CrossRef]
- Wang, C.; Chen, J.; Xu, X.; Hu, X.; Kong, D.; Liang, G.; Wang, X. Dual HER2 Blockade in Neoadjuvant Treatment of HER2+ Breast Cancer: A Meta-Analysis and Review. Technol. Cancer Res. Treat. 2020, 19, 1–10. [Google Scholar] [CrossRef]
- Triantafyllidi, E.; Triantafillidis, J.K. Systematic Review on the Use of Biosimilars of Trastuzumab in HER2+ Breast Cancer. Biomedicines 2022, 10, 2045. [Google Scholar] [CrossRef]
- Waller, C.F.; Möbius, J.; Fuentes-Alburo, A. Intravenous and Subcutaneous Formulations of Trastuzumab, and Trastuzumab Biosimilars: Implications for Clinical Practice. Br. J. Cancer 2021, 124, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Janjigian, Y.Y.; Bissig, M.; Curigliano, G.; Coppola, J.; Latymer, M. Talking to Patients about Biosimilars. Future Oncol. 2018, 14, 2403–2414. [Google Scholar] [CrossRef]
- Miller, E.M.; Schwartzberg, L.S. Biosimilars for Breast Cancer: A Review of HER2-Targeted Antibodies in the United States. Ther. Adv. Med. Oncol. 2019, 11, 1–9. [Google Scholar] [CrossRef]
- Lamb, Y.N. SB3 (Ontruzant®): A Trastuzumab Biosimilar. BioDrugs 2018, 32, 293–296. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Yeo, J.H.; Kim, Y.S.; Park, I.; Ahn, H.K.; Cho, E.K.; Shin, D.B.; Yang, J.Y.; Kim, H.S.; Lee, W.K.; et al. Efficacy and Safety of Trastuzumab Biosimilar (CT-P6) Compared With Reference Trastuzumab in Patients With HER2-Positive Advanced Gastric Cancer A Retrospective Analysis. Am. J. Clin. Oncol. Cancer Clin. Trials 2022, 45, 61–65. [Google Scholar] [CrossRef]
- Oda, M.; Uchiyama, S.; Noda, M.; Nishi, Y.; Koga, M.; Mayanagi, K.; Robinson, C.V.; Fukui, K.; Kobayashi, Y.; Morikawa, K.; et al. Effects of Antibody Affinity and Antigen Valence on Molecular Forms of Immune Complexes. Mol. Immunol. 2009, 47, 357–364. [Google Scholar] [CrossRef]
- Wen, J.; Arakawa, T.; Wypych, J.; Langley, K.E.; Schwartz, M.G.; Philo, J.S. Chromatographic Determination of Extinction Coefficients of Non-Glycosylated Proteins Using Refractive Index (RI) and UV Absorbance (UV) Detectors: Applications for Studying Protein Interactions by Size Exclusion Chromatography with Light-Scattering, UV, and RI Detectors. Tech. Protein Chem. 1997, 8, 113–119. [Google Scholar] [CrossRef]
- Arakawa, T.; Wen, J. Size-Exclusion Chromatography with on-Line Light Scattering. Curr. Protoc. Protein Sci. 2001, 25, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Mayer, C.L.; Snyder, W.K.; Swietlicka, M.A.; Vanschoiack, A.D.; Austin, C.R.; McFarland, B.J. Size-Exclusion Chromatography Can Identify Faster-Associating Protein Complexes and Evaluate Design Strategies. BMC Res. Notes 2009, 2, 135. [Google Scholar] [CrossRef]
- Bai, Y. Detecting Protein-Protein Interactions by Gel Filtration Chromatography. In Protein-Protein Interactions: Methods and Applications, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 223–232. [Google Scholar]
- Goyon, A.; Fekete, S.; Beck, A.; Veuthey, J.-L.; Guillarme, D. Unraveling the Mysteries of Modern Size Exclusion Chromatography—The Way to Achieve Confident Characterization of Therapeutic Proteins. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2018, 1092, 368–378. [Google Scholar] [CrossRef]
- Vega, J.F.; Ramos, J.; Cruz, V.L.; Vicente-Alique, E.; Sánchez-Sánchez, E.; Sánchez-Fernández, A.; Wang, Y.; Hu, P.; Cortés, J.; Martínez-Salazar, J. Molecular and Hydrodynamic Properties of Human Epidermal Growth Factor Receptor HER2 Extracellular Domain and Its Homodimer: Experiments and Multi-Scale Simulations. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 2406–2416. [Google Scholar] [CrossRef] [PubMed]
- Ramos, J.; Vega, J.F.; Cruz, V.; Sanchez-Sanchez, E.; Cortes, J.; Martinez-Salazar, J. Hydrodynamic and Electrophoretic Properties of Trastuzumab/HER2 Extracellular Domain Complexes as Revealed by Experimental Techniques and Computational Simulations. Int. J. Mol. Sci. 2019, 20, 1076. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.C.; von Hippel, P.H. Calculation of Protein Extinction Coefficients from Amino Acid Sequence Data. Anal. Biochem. 1989, 182, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Pace, C.N.; Vajdos, F.; Fee, L.; Grimsley, G.; Gray, T. How to Measure and Predict the Molar Absorption Coefficient of a Protein. Protein Sci. 1995, 4, 2411–2423. [Google Scholar] [CrossRef]
- Cruz, V.L.; Souza-Egipsy, V.; Gion, M.; Pérez-García, J.; Cortes, J.; Ramos, J.; Vega, J.F. Binding Affinity of Trastuzumab and Pertuzumab Monoclonal Antibodies to Extracellular HER2 Domain. Int. J. Mol. Sci. 2023, 24, 12031. [Google Scholar] [CrossRef]
- Hughes-Jones, N.C.; Gorick, B.D.; Howard, J.C. The Mechanism of Synergistic Complement-Mediated Lysis of Rat Red Cells by Monoclonal IgG Antibodies. Eur. J. Immunol. 1983, 13, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Robak, T. The Emerging Therapeutic Role of Antibody Mixtures. Expert Opin. Biol. Ther. 2013, 13, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Raju, T.S.; Strohl, W.R. Potential Therapeutic Roles for Antibody Mixtures. Expert Opin. Biol. Ther. 2013, 13, 1347–1352. [Google Scholar] [CrossRef] [PubMed]
- Larbouret, C.; Gros, L.; Pèlegrin, A.; Chardès, T. Improving Biologics’ Effectiveness in Clinical Oncology: From the Combination of Two Monoclonal Antibodies to Oligoclonal Antibody Mixtures. Cancers 2021, 13, 4620. [Google Scholar] [CrossRef]
- Skartved, N.J.Ø.; Jacobsen, H.J.; Pedersen, M.W.; Jensen, P.F.; Sen, J.W.; Jørgensen, T.K.; Hey, A.; Kragh, M. Preclinical Pharmacokinetics and Safety of Sym004: A Synergistic Antibody Mixture Directed against Epidermal Growth Factor Receptor. Clin. Cancer Res. 2011, 17, 5962–5972. [Google Scholar] [CrossRef]
- Meng, Q.; Garcia-Rodriguez, C.; Manzanarez, G.; Silberg, M.A.; Conrad, F.; Bettencourt, J.; Pan, X.; Breece, T.; To, R.; Li, M.; et al. Engineered Domain-Based Assays to Identify Individual Antibodies in Oligoclonal Combinations Targeting the Same Protein. Anal. Biochem. 2012, 430, 141–150. [Google Scholar] [CrossRef]
- Singh, P.; Roche, A.; Van Der Walle, C.F.; Uddin, S.; Du, J.; Warwicker, J.; Pluen, A.; Curtis, R. Determination of Protein-Protein Interactions in a Mixture of Two Monoclonal Antibodies. Mol. Pharm. 2019, 16, 4775–4786. [Google Scholar] [CrossRef]
- Wang, B.; Deng, R.; Hennig, S.; Badovinac Crnjevic, T.; Kaewphluk, M.; Kågedal, M.; Quartino, A.L.; Girish, S.; Li, C.; Kirschbrown, W.P. Population Pharmacokinetic and Exploratory Exposure–Response Analysis of the Fixed-Dose Combination of Pertuzumab and Trastuzumab for Subcutaneous Injection in Patients with HER2-Positive Early Breast Cancer in the FeDeriCa Study. Cancer Chemother. Pharmacol. 2021, 88, 499–512. [Google Scholar] [CrossRef]
- Yadav, S.; Liu, J.; Shire, S.J.; Kalonia, D.S. Specific interactions in high concentration antibody solutions resulting in high viscosity. J. Pharm. Sci. 2010, 99, 1152–1168. [Google Scholar] [CrossRef]
- Saito, S.; Hasegawa, J.; Kobayashi, N.; Kishi, N.; Uchiyama, S.; Fukui, K. Behavior of Monoclonal Antibodies: Relation between the Second Virial Coefficient (B2) at Low Concentrations and Aggregation Propensity and Viscosity at High Concentrations. Pharm. Res. 2012, 29, 397–410. [Google Scholar] [CrossRef] [PubMed]
- Barnett, G.V.; Qi, W.; Amin, S.; Lewis, E.N.; Razinkov, V.I.; Kerwin, B.A.; Liu, Y.; Roberts, C.J. Structural Changes and Aggregation Mechanisms for AntiStreptavidin IgG1 at Elevated Concentration. J. Phys. Chem. B 2015, 119, 15150–15163. [Google Scholar] [CrossRef] [PubMed]
- Jaccoulet, E.; Boccard, J.; Taverna, M.; Azevedos, A.S.; Rudaz, S.; Smadja, C. High-throughput identification of monoclonal antibodies after compounding by UV spectroscopy coupled to chemometrics analysis. Anal. Bioanal. Chem. 2016, 408, 5915–5924. [Google Scholar] [CrossRef] [PubMed]
- Vermeer, A.W.P.; Norde, W. The Thermal Stability of Immunoglobulin: Unfolding and Aggregation of a Multi-Domain Protein. Biophys. J. 2000, 78, 394–404. [Google Scholar] [CrossRef]
- Le Basle, Y.; Chenell, P.; Tokhadze, N.; Astier, A.; Sautou, V. Physicochemical Stability of Monoclonal Antibodies: A Review. J. Pharm. Sci. 2020, 109, 169–190. [Google Scholar] [CrossRef]
- Lehermayr, C.; Mahler, H.-C.; Mäder, K.; Fischer, S. Assessment of Net Charge and Protein–Protein Interactions of Different Monoclonal Antibodies. J. Pharm. Sci. 2011, 100, 2551–2562. [Google Scholar] [CrossRef]
- Roberts, D.; Keeling, R.; Tracka, M.; van der Walle, C.F.; Uddin, S.; Warwicker, J.; Curtis, R. The Role of Electrostatics in Protein–Protein Interactions of a Monoclonal Antibody. Mol. Pharm. 2014, 11, 2475–2489. [Google Scholar] [CrossRef] [PubMed]
- Kiraga, J.; Mackiewicz, P.; Mackiewicz, D.; Kowalczuk, M.; Biecek, P.; Polak, N.; Smolarczyk, K.; Dudek, M.R.; Cebrat, S. The relationships between the isoelectric point and: Length of proteins, taxonomy and ecology of organisms. BMC Genom. 2007, 8, 163. [Google Scholar] [CrossRef] [PubMed]
- Knight, C.G.; Kassen, R.; Hebestreit, H.; Rainey, P.B. Global analysis of predicted proteomes: Functional adaptation of physical properties. Proc. Natl. Acad. Sci. USA 2004, 101, 8390–8395. [Google Scholar] [CrossRef]
- Wang, M.; Zhu, D.; Zhu, J.; Nussinov, R.; Ma, B. Local and Global Anatomy of Antibody-Protein Antigen Recognition. J. Mol. Recognit. 2018, 31, e2693. [Google Scholar] [CrossRef] [PubMed]






| Sample | Mw (kDa) | [η] 102 (cm3·g−1) s.d. ± 0.2 | rh (nm) s.d. ± 0.1 | dA/dc (g−1·mL·cm−1) s.d. ± 0.02 |
|---|---|---|---|---|
| Perjeta | 147.1 ± 0.8 | 6.4 | 5.5 | 1.33 |
| Herceptin | 147.0 ± 1.6 | 6.5 | 5.5 | 1.38 |
| Herzuma | 147.9 ± 1.4 | 6.3 | 5.5 | 1.38 |
| Ontruzant | 147.7 ± 1.3 | 6.4 | 5.5 | 1.37 |
| g-eHER2 | 86.3 ± 1.0 | 6.5 | 4.4 | 0.90 |
| Sample C1/C2 | Mw (kDa) | [η] 102 (cm3·g−1) s.d. ± 0.2 | rh (nm) s.d. ± 0.1 | dA/dc (g−1·mL·cm−1) s.d. ± 0.02 |
|---|---|---|---|---|
| HRC/HER2 | 235.7/310.8 | 6.7/7.9 | 6.2/7.1 | 1.22/1.15 |
| ONT/HER2 | 237.5/313.6 | 6.9/7.7 | 6.2/7.2 | 1.23/1.14 |
| HZM/HER2 | 238.2/315.7 | 6.8/7.8 | 6.2/7.2 | 1.22/1.15 |
| C3 | Case 1: TZM/HER2/PZM | |||
| HRC/HER2/PJT | 482.5 | 8.1 | 8.5 | 1.22 |
| ONT/HER2/PJT | 483.2 | 8.2 | 8.6 | 1.22 |
| Sample C2 | Mw (kDa) | [η] 102 (cm3·g−1) s.d. ± 0.2 | rh (nm) s.d. ± 0.1 | dA/dc (g−1·mL·cm−1) s.d. ± 0.02 |
|---|---|---|---|---|
| PJT/HER2 | 310.0 | 8.0 | 7.4 | 1.12 |
| C3 | Case 2: PZM/HER2/TZM | |||
| PJT/HER2/HRC | 484.6 | 8.1 | 8.5 | 1.22 |
| PJT/HER2/ONT | 481.8 | 8.2 | 8.5 | 1.23 |
| PJT/HER2/HZM | 491.6 | 8.1 | 8.6 | 1.22 |
| Sample | Mw (kDa) | [η] 102 (cm3·g−1) s.d. ± 0.2 | rh (nm) s.d. ± 0.1 | dA/dc (g−1·mL·cm−1) s.d. ± 0.02 |
| C3 | Case 3: TZM/PZM/HER2 | |||
| PJT-HRC/HER2 | 932.0/489.0 | 9.8/8.0 | 9.5/8.7 | 1.23/1.23 |
| PJT-ONT/HER2 | 945.0/489.0 | n.d./8.2 | n.d./8.8 | 1.25/1.25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franco-Mateos, E.; Souza-Egipsy, V.; García-Estévez, L.; Pérez-García, J.; Gion, M.; Garrigós, L.; Cortez, P.; Saavedra, C.; Gómez, P.; Ortiz, C.; et al. Exploring the Combined Action of Adding Pertuzumab to Branded Trastuzumab versus Trastuzumab Biosimilars for Treating HER2+ Breast Cancer. Int. J. Mol. Sci. 2024, 25, 3940. https://doi.org/10.3390/ijms25073940
Franco-Mateos E, Souza-Egipsy V, García-Estévez L, Pérez-García J, Gion M, Garrigós L, Cortez P, Saavedra C, Gómez P, Ortiz C, et al. Exploring the Combined Action of Adding Pertuzumab to Branded Trastuzumab versus Trastuzumab Biosimilars for Treating HER2+ Breast Cancer. International Journal of Molecular Sciences. 2024; 25(7):3940. https://doi.org/10.3390/ijms25073940
Chicago/Turabian StyleFranco-Mateos, Emma, Virginia Souza-Egipsy, Laura García-Estévez, José Pérez-García, María Gion, Laia Garrigós, Patricia Cortez, Cristina Saavedra, Patricia Gómez, Carolina Ortiz, and et al. 2024. "Exploring the Combined Action of Adding Pertuzumab to Branded Trastuzumab versus Trastuzumab Biosimilars for Treating HER2+ Breast Cancer" International Journal of Molecular Sciences 25, no. 7: 3940. https://doi.org/10.3390/ijms25073940