Differential Occupation of Available Coral Hosts by Coral-Dwelling Damselfish (Pomacentridae) on Australia’s Great Barrier Reef
Abstract
:1. Introduction
2. Materials and Methods
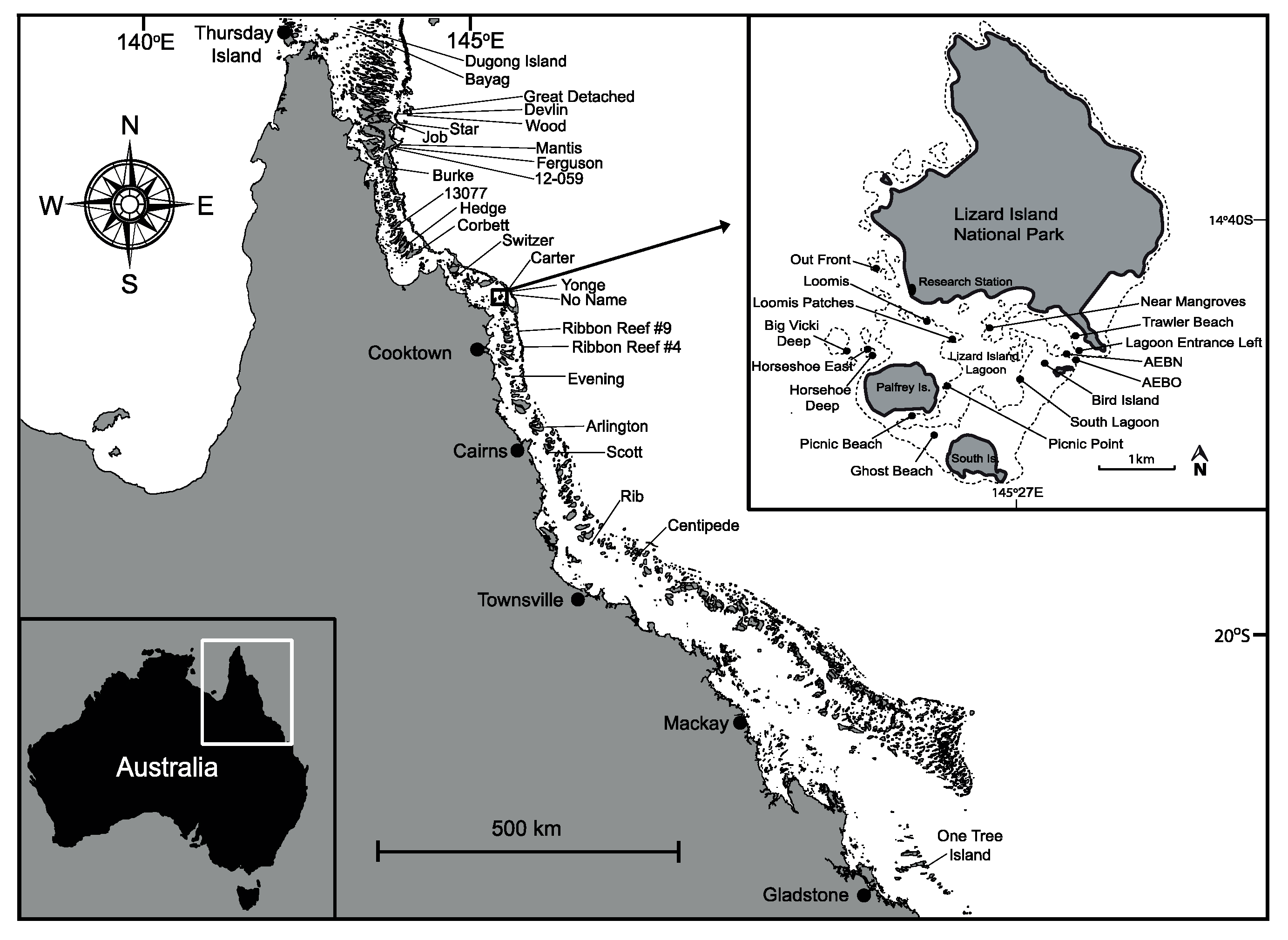
2.1. Study Sites and Surveys
2.2. Data Analysis
2.2.1. Reefscape Prevalence of Fish–Coral Interactions
2.2.2. Effects of Colony Position and Structure on Damselfish’s Occupancy
2.2.3. Effects of Colony Position and Structure on the Biomass of Damselfish
3. Results
3.1. Range of Damselfish’s Occupations across the Great Barrier Reef (GBR)
3.2. Patterns of Damselfish Biomass across Reefs on Occupied Colonies
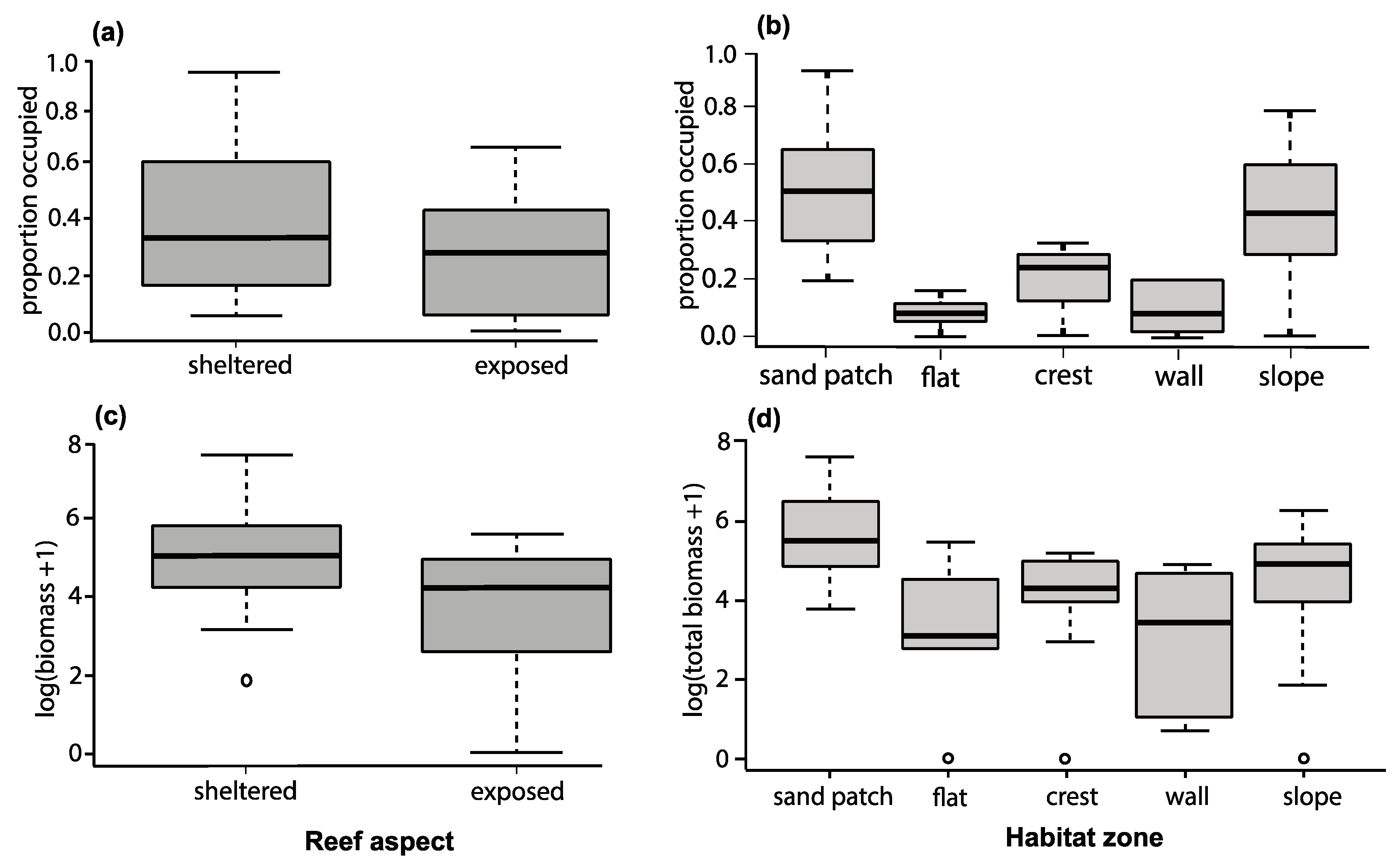
3.3. Colony Orientation as a Determinant of Damselfish’s Occupation and Group Biomass
3.4. Colony Structure as a Determinant of Damselfish’s Occupation and Biomass
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Graham, N.A.J.; Nash, K.L. The importance of structural complexity in coral reef ecosystems. Coral Reefs 2013, 32, 315–326. [Google Scholar] [CrossRef]
- Bell, J.; Galzin, R. Influence of live coral cover on coral-reef fish communities. Mar. Ecol. Prog. Ser. 1984, 15, 265–274. [Google Scholar] [CrossRef]
- Roberts, C.; Ormond, R. Habitat complexity and coral reef fish diversity and abundance on Red Sea fringing reefs. Mar. Ecol. Prog. Ser. 1987, 41, 1–8. [Google Scholar] [CrossRef]
- Friedlander, A.M.; Parrish, J.D. Habitat characteristics affecting fish assemblages on a Hawaiian coral reef. J. Exp. Mar. Bio. Ecol. 1998, 224, 1–30. [Google Scholar] [CrossRef]
- Sale, P. Extremely Limited Home Range in a Coral Reef Fish, Dascyllus aruanus (Pisces; Pomacentridae). Copeia 1971, 1971, 324–327. [Google Scholar] [CrossRef]
- Munday, P.L.; Jones, G.P.; Caley, M.J. Habitat specialisation and the distribution and abundance of coral-dwelling gobies. Mar. Ecol. Prog. Ser. 1997, 152, 227–239. [Google Scholar] [CrossRef]
- Wilson, S.; Burgess, S.; Cheal, A.; Emslie, M.; Fisher, R.; Miller, I.; Polunin, N. Habitat utilization by coral reef fish: implications for specialists vs. generalists in a changing environment. J. Anim. Ecol. 2007, 77, 220–228. [Google Scholar] [CrossRef]
- Feary, D.A. The influence of resource specialization on the response of reef fish to coral disturbance. Mar. Biol. 2007, 153, 153–161. [Google Scholar] [CrossRef]
- Komyakova, V.; Munday, P.L.; Jones, G.P. Relative importance of coral cover, habitat complexity and diversity in determining the structure of reef fish communities. PLoS ONE 2013, 8, e83178. [Google Scholar] [CrossRef]
- Graham, N.A.J.; Wilson, S.K.; Jennings, S.; Polunin, N.V.C.; Bijoux, J.P.; Robinson, J. Dynamic fragility of oceanic coral reef ecosystems. Proc. Natl. Acad. Sci. USA 2006, 103, 8425–8429. [Google Scholar] [CrossRef]
- Wilson, S.K.; Graham, N.A.J.; Pratchett, M.S.; Jones, G.P.; Polunin, N.V.C. Multiple disturbances and the global degradation of coral reefs: Are reef fishes at risk or resilient? Glob. Chang. Biol. 2006, 12, 2220–2234. [Google Scholar] [CrossRef]
- Kramer, M.J.; Bellwood, D.R.; Taylor, R.B.; Bellwood, O. Benthic crustacea from tropical and temperate reef locations: Differences in assemblages and their relationship with habitat structure. Coral Reefs 2017, 36, 971–980. [Google Scholar] [CrossRef]
- Messmer, V.; Jones, G.P.; Munday, P.L.; Holbrook, S.J.; Schmitt, R.J.; Brooks, A.J. Habitat biodiversity as a determinant of fish community structure on coral reefs. Ecology 2011, 92, 2285–2298. [Google Scholar] [CrossRef] [PubMed]
- Beukers, J.; Jones, J. Habitat complexity modifies the impact of piscivores on a coral reef fish population. Oecologia 1997, 114, 50–59. [Google Scholar] [CrossRef]
- Almany, G.R. Differential effects of habitat complexity, predators and competitors on abundance of juvenile and adult coral reef fishes. Oecologia 2004, 141, 105–113. [Google Scholar] [CrossRef]
- Munday, P.; Jones, G.; Caley, J. Interspecific competition and coexistence in a guild of coral-dwelling fishes. Ecology 2001, 82, 2177–2189. [Google Scholar] [CrossRef]
- Cole, A.J.; Pratchett, M.S.; Jones, G.P. Diversity and functional importance of coral-feeding fishes on tropical coral reefs. Fish Fish 2008, 9, 286–307. [Google Scholar] [CrossRef]
- Emslie, M.J.; Logan, M.; Cheal, A.J. The Distribution of planktivorous damselfishes (Pomacentridae) on the Great Barrier Reef and the relative influences of habitat and predation. Diversity 2019, 11, 33. [Google Scholar] [CrossRef]
- Jones, G.P.; McCormick, M.I.; Srinivasan, M.; Eagle, J.V. Coral decline threatens fish biodiversity in marine reserves. Proc. Natl. Acad. Sci. USA 2004, 101, 8251–8253. [Google Scholar] [CrossRef]
- Booth, D.J.; Wellington, G. Settlement preferences in coral-reef fishes: Effects on patterns of adult and juvenile distributions, individual fitness and population structure. Aust. J. Ecol. 1998, 23, 274–279. [Google Scholar] [CrossRef]
- Holbrook, S.J.; Forrester, G.E.; Schmitt, R.J. Spatial patterns in abundance of damselfish reflect availability of suitable habitat. Oecologia 2000, 122, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Pratchett, M.S.; Coker, D.J.; Jones, G.P.; Munday, P.L. Specialization in habitat use by coral reef damselfishes and their susceptibility to habitat loss. Ecol. Evol. 2012, 2, 2168–2180. [Google Scholar] [CrossRef] [PubMed]
- Munday, P.L. Does habitat availability determine geographical-scale abundances of coral-dwelling fishes? Coral Reefs 2002, 21, 105–116. [Google Scholar] [CrossRef]
- Mellin, C.; Bradshaw, C.; Meekan, M.; Caley, M. Environmental and spatial predictors of species richness and abundance in coral reef fishes. Glob. Ecol. Biogeogr. 2019, 19, 212–222. [Google Scholar] [CrossRef]
- Darling, E.S.; Graham, N.A.J.; Januchowski-Hartley, F.A.; Nash, K.L.; Pratchett, M.S.; Wilson, S.K. Relationships between structural complexity, coral traits, and reef fish assemblages. Coral Reefs 2017, 36, 561–575. [Google Scholar] [CrossRef]
- Holbrook, S.J.; Brooks, A.J.; Schmitt, R.J.; Stewart, H.L. Effects of sheltering fish on growth of their host corals. Mar. Biol. 2008, 155, 521–530. [Google Scholar] [CrossRef]
- Chase, T.; Pratchett, M.; Frank, G.; Hoogenboom, M. Coral-dwelling fish moderate bleaching susceptibility of coral hosts. PLoS ONE 2018, 13, e0208545. [Google Scholar] [CrossRef]
- Meyer, J.; Schultz, E.; Helfman, G. Fish Schools: An Asset to Corals. Science 1983, 220, 1047–1049. [Google Scholar] [CrossRef]
- Kane, C.N.; Brooks, A.J.; Holbrook, S.J.; Schmitt, R.J. The role of microhabitat preference and social organization in determining the spatial distribution of a coral reef fish. Environ. Biol. Fishes 2009, 84, 1–10. [Google Scholar] [CrossRef]
- Nash, K.L.; Graham, N.A.J.; Wilson, S.K.; Bellwood, D.R. Cross-scale habitat structure drives fish body size distributions on coral reefs. Ecosystems 2013, 16, 478–490. [Google Scholar] [CrossRef]
- Epstein, H.E.; Kingsford, M.J. Are soft coral habitats unfavourable? A closer look at the association between reef fishes and their habitat. Environ. Biol. Fishes 2019, 102, 479–497. [Google Scholar] [CrossRef]
- Bay, L.; Jones, G.; McCormick, M. Habitat selection and aggression as determinants of spatial segregation among damselifsh on a coral reef. Coral Reefs 2001, 20, 289–298. [Google Scholar]
- Sale, P.F. Appropriate spatial scales for studies of reef-fish ecology. Austral. Ecol. 1998, 23, 202–208. [Google Scholar] [CrossRef]
- Frédérich, B.; Fabri, G.; Lepoint, G.; Vandewalle, P.; Parmentier, E. Trophic niches of thirteen damselfishes (Pomacentridae) at the Grand Récif of Toliara, Madagascar. Ichthyol. Res. 2009, 56, 10–17. [Google Scholar] [CrossRef]
- Gajdzik, L.; Parmentier, E.; Michel, L.N.; Sturaro, N.; Soong, K.; Lepoint, G.; Frédérich, B. Similar levels of trophic and functional diversity within damselfish assemblages across Indo-Pacific coral reefs. Funct. Ecol. 2018, 32, 1358–1369. [Google Scholar] [CrossRef]
- McMahon, K.W.; Thorrold, S.R.; Houghton, L.A.; Berumen, M.L. Tracing carbon flow through coral reef food webs using a compound-specific stable isotope approach. Oecologia 2016, 180, 809–821. [Google Scholar] [CrossRef]
- Wyatt, A.S.J.; Waite, A.M.; Humphries, S. Stable isotope analysis reveals community-level variation in fish trophodynamics across a fringing coral reef. Coral Reefs 2012, 31, 1029–1044. [Google Scholar] [CrossRef]
- Gajdzik, L.; Parmentier, E.; Sturaro, N.; Frédérich, B. Trophic specializations of damselfishes are tightly associated with reef habitats and social behaviours. Mar. Biol. 2016, 163, 1–15. [Google Scholar] [CrossRef]
- Nadler, L.E.; McNeill, D.C.; Alwany, M.A.; Bailey, D.M. Effect of habitat characteristics on the distribution and abundance of damselfish within a Red Sea reef. Environ. Biol. Fishes 2014, 97, 1265–1277. [Google Scholar] [CrossRef]
- Fulton, C.J.; Bellwood, D.R.; Wainwright, P.C. Wave energy and swimming performance shape coral reef fish assemblages. Proc. R. Soc. B Biol. Sci. 2005, 272, 827–832. [Google Scholar] [CrossRef] [Green Version]
- Johansen, J.L.; Bellwood, D.R.; Fulton, C.J. Coral reef fishes exploit flow refuges in high-flow habitats. Mar. Ecol. Prog. Ser. 2008, 360, 219–226. [Google Scholar] [CrossRef] [Green Version]
- Fulton, C.J.; Bellwood, D.R. Wave-induced water motion and the functional implications for coral reef fish assemblages. Limnol. Oceanogr. 2005, 50, 255–264. [Google Scholar] [CrossRef] [Green Version]
- Sambrook, K.; Jones, G.P.; Bonin, M.C. Life on the edge: Coral reef fishes exhibit strong responses to a habitat boundary. Mar. Ecol. Prog. Ser. 2016, 561, 203–215. [Google Scholar] [CrossRef]
- Kerry, J.T.; Bellwood, D.R. The effect of coral morphology on shelter selection by coral reef fishes. Coral Reefs 2012, 31, 415–424. [Google Scholar] [CrossRef]
- Pratchett, M.S.; Baird, A.H.; McCowan, D.M.; Coker, D.J.; Cole, A.J.; Wilson, S.K. Protracted declines in coral cover and fish abundance following climate-induced coral bleaching on the Great Barrier Reef. In Proceedings of the 11th International Coral Reef Symposium, Fort Lauderdale, FL, USA, 7–11 July 2008; pp. 1309–1313. [Google Scholar]
- Coker, D.; Wilson, S.; Pratchett, M. Importance of live coral habitat for reef fishes. Rev. Fish Biol. Fish. 2014, 24, 89–126. [Google Scholar] [CrossRef]
- Chase, T.; Pratchett, M.; Walker, S.; Hoogenboom, M. Small-scale environmental variation influences whether coral-dwelling fish promote or impede coral growth. Oecologia 2014, 176, 1009–1022. [Google Scholar] [CrossRef]
- Randall, J.; Allen, G.; Steene, R. Fishes of the Great Barrier Reef and Coral Sea; University of Hawai’i Press: Honolulu, HI, USA, 1997. [Google Scholar]
- Allen, G.; Steene, R.; Humann, P.; DeLoach, N. Reef Fish Identification—Tropical Pacific; New World Pulications, Inc.: Jacksonville, FL, USA, 2003. [Google Scholar]
- Chase, T.; Pratchett, M.; Hoogenboom, M. Behavioural trade-offs and habitat associations of coral-dwelling damselfishes (family Pomacentridae). Mar. Ecol. Prog. Ser. 2019. [Google Scholar] [CrossRef]
- Hughes, T.P.; Kerry, J.T.; Álvarez-Noriega, M.; Álvarez-Romero, J.G.; Anderson, K.D.; Baird, A.H.; Babcock, R.C.; Beger, M.; Bellwood, D.R.; Berkelmans, R.; et al. Global warming and recurrent mass bleaching of corals. Nature 2017, 543, 373–377. [Google Scholar] [CrossRef]
- Hoogenboom, M.O.; Frank, G.E.; Chase, T.J.; Jurriaans, S.; Álvarez-Noriega, M.; Peterson, K.; Critchell, K.; Berry, K.L.E.; Nicolet, K.J.; Ramsby, B.; et al. Environmental drivers of variation in bleaching severity of Acropora species during an extreme thermal anomaly. Front. Mar. Sci. 2017, 4, 376. [Google Scholar] [CrossRef] [Green Version]
- Graham, N.A.J.; Chong-Seng, K.M.; Huchery, C.; Januchowski-Hartley, F.A.; Nash, K.L. Coral reef community composition in the context of disturbance history on the Great Barrier Reef, Australia. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Hopley, D.; Smithers, S.; Parnell, K. The Geomorphology of the Great Barrier Reef: Development, Diversity and Change; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Ménard, A.; Turgeon, K.; Roche, D.G.; Binning, S.A.; Kramer, D.L. Shelters and their use by fishes on fringing coral reefs. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [Green Version]
- Veron, J.; Stafford-Smith, M. Corals of the World; Australian Institute of Marine Science: Townsville, Australia, 2004.
- Hill, J.; Wilkinson, C. Methods for Ecological Monitoring of Coral Reefs; Australian Institute of Marine Science: Townsville, Australia, 2004; 117p.
- Wehrberger, F.; Herler, J. Microhabitat characteristics influence shape and size of coral-associated fishes. Mar. Ecol. Prog. Ser. 2014, 500, 203–214. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, I.M.; Bastos, Y.F.; Barreto, D.S.; Lourenço, L.S.; Penha, J.M. The efficacy of clove oil as an anaesthetic and in euthanasia procedure for small-sized tropical fishes. Braz. J. Biol. 2016, 77, 444–450. [Google Scholar] [CrossRef] [Green Version]
- Javahery, S.; Nekoubin, H.; Moradlu, A.H. Effect of anaesthesia with clove oil in fish (review). Fish Physiol. Biochem. 2012, 38, 1545–1552. [Google Scholar] [CrossRef]
- Burkepile, D.E.; Allgeier, J.E.; Shantz, A.A.; Pritchard, C.E.; Lemoine, N.P.; Bhatti, L.H.; Layman, C.A. Nutrient supply from fishes facilitates macroalgae and suppresses corals in a Caribbean coral reef ecosystem. Sci. Rep. 2014, 3, 19–21. [Google Scholar] [CrossRef] [Green Version]
- Cribari-Neto, F.; Zeileis, A. Journal of Statistical Software Beta Regression in R. J. Stat. Softw. 2010, 34, 1–24. [Google Scholar] [CrossRef] [Green Version]
- Warton, D.I.; Hui, F. The arcsine is asinine: The analysis of proportions in ecology. Ecology 2011, 92, 3–10. [Google Scholar] [CrossRef] [Green Version]
- Affleck, D.L.R. Additivity and maximum likelihood estimation of nonlinear component biomass models. In Proceedings of the New Directions in Inventory Techinques & Applicaitons Forest Inventory & Analysis (FIA) Symposium 2015, 8–12 December 2015; U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station: Portland, OR, USA; pp. 13–17. [Google Scholar]
- Zuur, A.; Ieno, E.; Walker, N.; Saveliev, A.; Smith, G. Mixed Effects Models and Extension in Ecology with R; Springer: New York, NY, USA, 2009. [Google Scholar]
- Zuur, A.F.; Ieno, E.N. A protocol for conducting and presenting results of regression-type analyses. Methods Ecol. Evol. 2016, 7, 636–645. [Google Scholar] [CrossRef]
- Bartón, K. MuMIN: Multi-model Interference R Package Version 1.9.12; The R Foundation: Vienna, Austria, 2013. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Multimodel Inference: Understanding AIC and BIC in Model Selection, 2nd ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Benjamini, Y.; Hochber, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Venables, W.; Ripley, B. Modern Applied Statistics with S, 4th ed.; Springer: Berlin, Germany, 2002. [Google Scholar]
- Mardia, K.; Kent, J.; Bibby, J. Multivariate Analysis; Academic Press: London, UK, 1979. [Google Scholar]
- Clarke, K.R. Non-parametric multivariate analyses of changes in community structure. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Warton, D.I.; Wright, S.T.; Wang, Y. Distance-based multivariate analyses confound location and dispersion effects. Methods Ecol. Evol. 2012, 3, 89–101. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; The R Foundation: Vienna, Austria, 2018. [Google Scholar]
- Hothorn, T.; Bretz, F.; Westfall, P. Simulatnous Inference in Generall Parametric Models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lenth, R.V. Least-Squares Means: The R Package lsmeans. J. Stat. Softw. 2016, 69, 1–33. [Google Scholar] [CrossRef] [Green Version]
- Chase, T.; Hoogenboom, M. Occupation of Damselfishes across Reef Seascape and Colony Scale, GBR 2016 Data. James Cook University (Dataset). Available online: http://dx.doi.org/10.25903/5dcb4c44aa86a (accessed on 19 November 2019).
- Noonan, S.H.C.; Jones, G.P.; Pratchett, M.S. Coral size, health and structural complexity: Effects on the ecology of a coral reef damselfish. Mar. Ecol. Prog. Ser. 2012, 456, 127–137. [Google Scholar] [CrossRef] [Green Version]
- Pereira, P.H.C.; Munday, P.L. Coral colony size and structure as determinants of habitat use and fitness of coral-dwelling fishes. Mar. Ecol. Prog. Ser. 2016, 553, 163–172. [Google Scholar] [CrossRef]
- Waldner, R.E.; Robertson, D.R. Patterns of habitat partitioning by eight species of territorial caribbean damselfishes (Pisces: Pomacentridae). Bull. Mar. Sci. 1980, 30, 171–186. [Google Scholar]
- Morais, R.A.; Bellwood, D.R. Pelagic Subsidies Underpin Fish Productivity on a Degraded Coral Reef. Curr. Biol. 2019, 29, 1521–1527.e6. [Google Scholar] [CrossRef]
- Meekan, M.G.; Steven, A.D.L.; Fortin, M.J. Spatial patterns in the distribution of damselfishes on a fringing coral reef. Coral Reefs 1995, 14, 151–161. [Google Scholar] [CrossRef]
- Waldock, C.; Stuart-Smith, R.D.; Edgar, G.J.; Bird, T.J.; Bates, A.E. The shape of abundance distributions across temperature gradients in reef fishes. Ecol. Lett. 2019, 22, 685–696. [Google Scholar] [CrossRef]
- Ault, T.; Johnson, C. Spatial variation in fish species richness on coral reefs: Habitat fragmentation and stochastic structuring processes. Okios 1998, 82, 354–364. [Google Scholar] [CrossRef] [Green Version]
- Bergman, K.C.; Öhman, M.C.; Svensson, S. Influence of habitat structure on Pomacentrus sulfureus, a western Indian Ocean reef fish. Environ. Biol. Fishes 2000, 59, 243–252. [Google Scholar] [CrossRef]
- Maire, E.; Villeger, S.; Graham, N.A.J.; Hoey, A.S.; Cinner, J.; Ferse, S.C.A.; Aliaume, C.; Booth, D.J.; Feary, D.A.; Kulbicki, M.; et al. Community-wide scan identifies fish species associated with coral reef services across the Indo-Pacific. Proc. R. Soc. B Biol. Sci. 2018, 285, 1621–1629. [Google Scholar] [CrossRef] [Green Version]
- Doherty, P.; Fowler, T. An empirical test of recruitment limitation in a coral reef fish. Science 1994, 263, 935–939. [Google Scholar] [CrossRef]
- Forrester, G. Strong density-dependent survival and recruitment regulate the abundance of a coral reef fish. Oecologia 1995, 103, 275–282. [Google Scholar] [CrossRef]
- Holbrook, S.J.; Schmitt, R.J. Spatial and temporal variation in mortality of newly settled damselfish: Patterns, causes and co-variation with settlement. Oecologia 2003, 135, 532–541. [Google Scholar] [CrossRef]
- Motro, R.; Ayalon, I.; Genin, A. Near-bottom depletion of zooplankton over coral reefs: III: Vertical gradient of predation pressure. Coral Reefs 2005, 24, 95–98. [Google Scholar] [CrossRef]
- Zikova, A.V.; Britaev, T.A.; Ivanenko, V.N.; Mikheev, V.N. Planktonic and symbiotic organisms in nutrition of coralobiont fish. J. Ichthyol. 2011, 51, 769–775. [Google Scholar] [CrossRef]
- Chamberlain, S.A.; Bronstein, J.L.; Rudgers, J.A. How context dependent are species interactions? Ecol. Lett. 2014, 17, 881–890. [Google Scholar] [CrossRef]
- Nanami, A.; Nishihira, M. Effects of habitat connectivity on the abundance and species richness of coral reef fishes: Comparison of an experimental habitat established at a rocky reef flat and at a sandy sea bottom. Environ. Biol. Fishes 2003, 68, 183–196. [Google Scholar] [CrossRef]
- Shpigel, M.; Fishelson, L. Behavior and physiology of coexistence in two species of Dascyllus (Pomacentridae, Teleostei). Environ. Biol. Fishes 1986, 17, 253–265. [Google Scholar] [CrossRef]
- Sale, P.F. Effect of cover on agonistic behavior of a reef fish: A possible spacing mechanism. Ecolgoy 1972, 53, 753–758. [Google Scholar] [CrossRef]
- Sale, P.F. Influence of corals in the dispersion of the Pomacentrid fish, Dascyllus aruanus. Ecology 1972, 53, 741–744. [Google Scholar] [CrossRef]
- Pratchett, M.; Hoey, A.; Wilson, S.; Hobbs, J.; Allen, G. Habitat-use and specialisation among coral reef damselfishes. In Biology of Damselfishes; Frédérich, B., Parmentier, E., Eds.; CRC Press: Boca Raton, FL, USA, 2016; pp. 84–121. [Google Scholar]
- Eurich, J.; McCormick, M.; Jones, G. Habitat selection and aggression as determinants of fine-scale partitioning of coral reef zones in a guild of territorial damselfishes. Mar. Ecol. Prog. Ser. 2018, 587, 201–215. [Google Scholar] [CrossRef]
- Almany, G.R. Does increased habitat complexity reduce predation and competition in coral reef fish assemblages? Oikos 2004, 106, 275–284. [Google Scholar] [CrossRef]
- Wen, C.K.C.; Pratchett, M.S.; Almany, G.R.; Jones, G.P. Patterns of recruitment and microhabitat associations for three predatory coral reef fishes on the southern Great Barrier Reef, Australia. Coral Reefs 2013, 32, 389–398. [Google Scholar] [CrossRef]
- Chase, T. Effects of Coral-Dwelling Damselfishes’ Abundances and Diversity on Host Coral Dynamics; James Cook University: Townsvile, Australia, 2019. [Google Scholar]
- Shantz, A.A.; Ladd, M.C.; Schrack, E.; Burkepile, D.E. Fish-derived nutrient hotspots shape coral reef benthic communities. Ecol. Appl. 2015, 25, 2142–2152. [Google Scholar] [CrossRef]
- Garcia-Herrera, N.; Ferse, S.C.A.; Kunzmann, A.; Genin, A. Mutualistic damselfish induce higher photosynthetic rates in their host coral. J. Exp. Biol. 2017, 220, 1803–1811. [Google Scholar] [CrossRef] [Green Version]
- Meyer, J.L.; Schultz, E.T. Migrating haemulid fishes as a source of nutrients and organic matter on coral reefs. Limnol. Oceanogr. 1985, 30, 146–156. [Google Scholar] [CrossRef] [Green Version]
- Meyer, J.; Schultz, E. Tissue condition and growth rate of corals associated with schooling fish. Limnol. Oceanogr. 2019, 30, 157–166. [Google Scholar] [CrossRef] [Green Version]
- Hoegh-Guldberg, O.; Smith, G.J. The effect of sudden changes in temperature, light and salinity on the population density and export of zooxanthellae from the reef corals Stylophora pistillata Exper and Seriatopora hystrix Dana. J. Exp. Mar. Biol. Ecol. 1989, 129, 279–303. [Google Scholar] [CrossRef]

| (a) Reef Seascape Surveys: | Description of Data |
|---|---|
| Sampling: 50 × 5 m belt transects n =3097 colonies on 51 sites spread among 22 reefs, including sheltered (n = 28 sites) and exposed (n = 23 sites) sites, and sand patches (n = 11 sites), reef flat (n = 5 sites), reef crest (n = 12 sites), reef wall (n = 5 sites), and reef slope (n =18 sites) habitats. | |
| Latitude | Sites grouped by latitude: Far north (12.3° S to 10.5° S), North (12.3° S), Central (14.7° S to 18.9° S) and South (One Tree Island, 23.5° S) |
| Aspect | Exposed locations (high water flow, mainly eastern side of GBR reefs) vs sheltered locations (low water flow, mainly western side of GBR reefs) |
| Habitat Zone | Lagoon sand patch, reef flat, reef crest, reef wall, and reef slope/base |
| Benthic cover [51] | Percentage cover measured on four 10 m line intercept transects at each site using the following categories: Isopora, Montipora, tabular Acropora, staghorn Acropora, other Acropora, Pocillopora damicornis, Seriatopora, Stylophora, other pocilloporids, Mussidae, Faviidae, Poritidae, other scleractinians, soft corals, and other sessile fauna |
| Coral Species | Acropora intermedia, Acropora spathulata, Pocillopora damicornis, Seriatopora hystrix, and Stylophora pistillata |
| (b) Colony orientation:Sampling: Digital photos of n = 226 colonies on 15 sites on 11 exposed and sheltered reefs | |
| Colony Orientation [52] (Position of Coral on Substratum) | Crevice—colony grew within a crack in the reef matrix; Open—colony is on flat reef benthos without any obvious shading by competitors; Sand—colony grew above a sand patch; Underhang—colony was shaded by reef matrix or other colonies |
| (c) Colony structure:Sampling: Digital photos of n = 226 colonies on 15 sites on 11 exposed and sheltered reefs | |
| Colony Diameter | Average of the longest colony diameter dimension and the diameter perpendicular to that dimension |
| Planar Area | Digitally traced along colony perimeter |
| Colony Height | Distance from top of coral colony to substrate |
| Branch Width | Average width of branches (n = 5 branches colony−1) located throughout the colony |
| Branch Spacing | Average distance between branches (n = 5 branches colony−1) located throughout the colony |
| Isolation | Distance to nearest branching, columnar, tabular, or foliaceous colony |
| Comparison | p-Value |
|---|---|
| A. intermedia – A. spathulata | 0.5089 |
| A. intermedia – P. damicornis | 0.0050 |
| A. intermedia – S. hystrix | 0.9996 |
| A. intermedia – S. pistillata | 0.0131 |
| A. spathulata – P. damicornis | 0.8963 |
| A. spathulata – S. hystrix | 0.4492 |
| A. spathulata – S. pistillata | 0.9588 |
| P. damicornis – S. hystrix | <0.001 |
| Aspect | Coral Species | n | Average Biomass (g) per Coral Species per Site Aspect | ||||
|---|---|---|---|---|---|---|---|
 |  |  |  |  | |||
| C. viridis | D. aruanus | D. reticulatus | P. amboinensis | P. moluccensis | |||
| Sheltered | A. intermedia | 38 | 0.92 ± 0.79 | 2.21 ± 0.96 | 0.94 ± 0.89 | 1.06 ± 0.31 | 2.80 ± 0.54 |
| A. spathulata | 30 | 4.66 ± 2.03 | 0.60 ± 0.48 | 0.17 ± 0.17 | 0.09 ± 0.06 | 5.70 ± 0.96 | |
| P. damicornis | 233 | 0.52 ± 0.22 | 5.00 ± 0.79 | 0.26 ± 0.14 | 0.84 ± 0.12 | 3.89 ± 0.34 | |
| S. hystrix | 147 | 2.63 ± 1.10 | 8.49 ± 1.15 | 0.00 ± 0.00 | 0.63 ± 0.11 | 2.47 ± 0.34 | |
| S. pistillata | 179 | 0.22 ± 0.02 | 4.72 ± 0.64 | 0.36 ± 0.12 | 0.50 ± 0.99 | 4.74 ± 0.51 | |
| Exposed | A. intermedia | 16 | 0.19 ± 0.19 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.24 ± 0.13 | 3.91 ± 1.06 |
| A. spathulata | 6 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 9.18 ± 3.31 | |
| P. damicornis | 86 | 0.24 ± 0.23 | 0.00 ± 0.00 | 1.52 ± 1.03 | 0.48 ± 0.16 | 6.45 ± 1.00 | |
| S. hystrix | 42 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.04 ± 0.04 | 0.84 ± 0.22 | 5.38 ± 0.89 | |
| S. pistillata | 121 | 0.00 ± 0.00 | 0.18 ± 0.12 | 0.22 ± 0.11 | 0.75 ± 0.16 | 5.79 ± 0.50 | |
| Coral Species | n | Isolation | Branch Spacing | Colony Diameter | Planar Area | Colony Height | Branch Width |
|---|---|---|---|---|---|---|---|
| A. intermedia | 22 | <0.001 | 0.527 | 0.019 | <0.001 | 0.068 | 0.185 |
| A. spathulata | 32 | 0.099 | 0.357 | 0.020 | <0.001 | 0.008 | 0.416 |
| P. damicornis | 66 | 0.014 | 0.002 | <0.001 | <0.001 | <0.001 | 0.423 |
| S. hystrix | 44 | 0.002 | <0.001 | 0.304 | 0.809 | 0.018 | 0.198 |
| S. pistillata | 62 | <0.001 | <0.001 | 0.734 | 0.357 | 0.015 | <0.001 |
| All colonies | 226 | <0.001 | 0.007 | <0.001 | <0.001 | <0.001 | 0.176 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chase, T.J.; Hoogenboom, M.O. Differential Occupation of Available Coral Hosts by Coral-Dwelling Damselfish (Pomacentridae) on Australia’s Great Barrier Reef. Diversity 2019, 11, 219. https://doi.org/10.3390/d11110219
Chase TJ, Hoogenboom MO. Differential Occupation of Available Coral Hosts by Coral-Dwelling Damselfish (Pomacentridae) on Australia’s Great Barrier Reef. Diversity. 2019; 11(11):219. https://doi.org/10.3390/d11110219
Chicago/Turabian StyleChase, Tory J, and Mia O Hoogenboom. 2019. "Differential Occupation of Available Coral Hosts by Coral-Dwelling Damselfish (Pomacentridae) on Australia’s Great Barrier Reef" Diversity 11, no. 11: 219. https://doi.org/10.3390/d11110219





