Diversity and Structure of an Arid Woodland in Southwest Angola, with Comparison to the Wider Miombo Ecoregion
Abstract
:1. Introduction
- Describe the tree species diversity and structure of woodlands in Bicuar National Park, and compare this composition with other woodlands across the miombo eco-region.
- Explore the role of environmental factors in driving changes in tree species composition across the miombo ecoregion.
- Describe variation in tree species composition and woodland structure between disturbed and undisturbed woodland patches within Bicuar National Park.
2. Materials and Methods
2.1. Study Area
2.2. Plot Data Collection
2.3. Climatic Data
2.4. Data Analysis
3. Results
3.1. Alpha Diversity
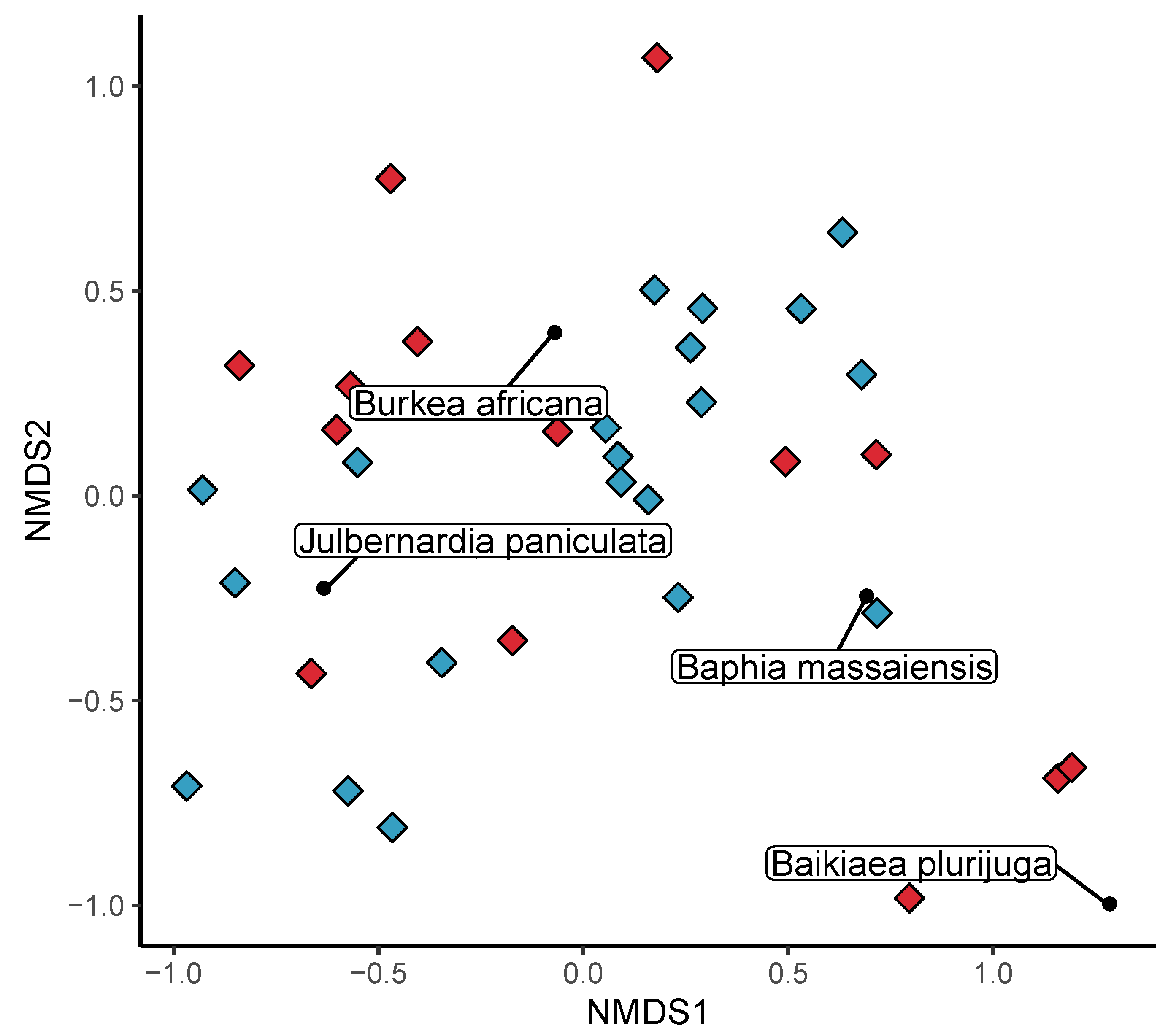
3.2. Beta Diversity
3.3. Woodland Structure
3.4. Effect of Disturbance via Shifting Cultivation on Diversity within Bicuar National Park
4. Discussion
4.1. Comparison of Bicuar National Park with Other Woodlands across the Miombo Ecoregion
4.2. Delineation of Woodland Types within Bicuar National Park
4.3. Comparison of Disturbed and Undisturbed Woodland Plots
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of Variance |
| DD | Decimal Degrees |
| MAP | Mean Annual Precipitation |
| MAT | Mean Annual Temperature |
| MAT SD | Standard Deviation of Mean Annual Temperature (Seasonality) |
| NMDS | Non-metric Multidimensional Scaling |
| NP | National Park |
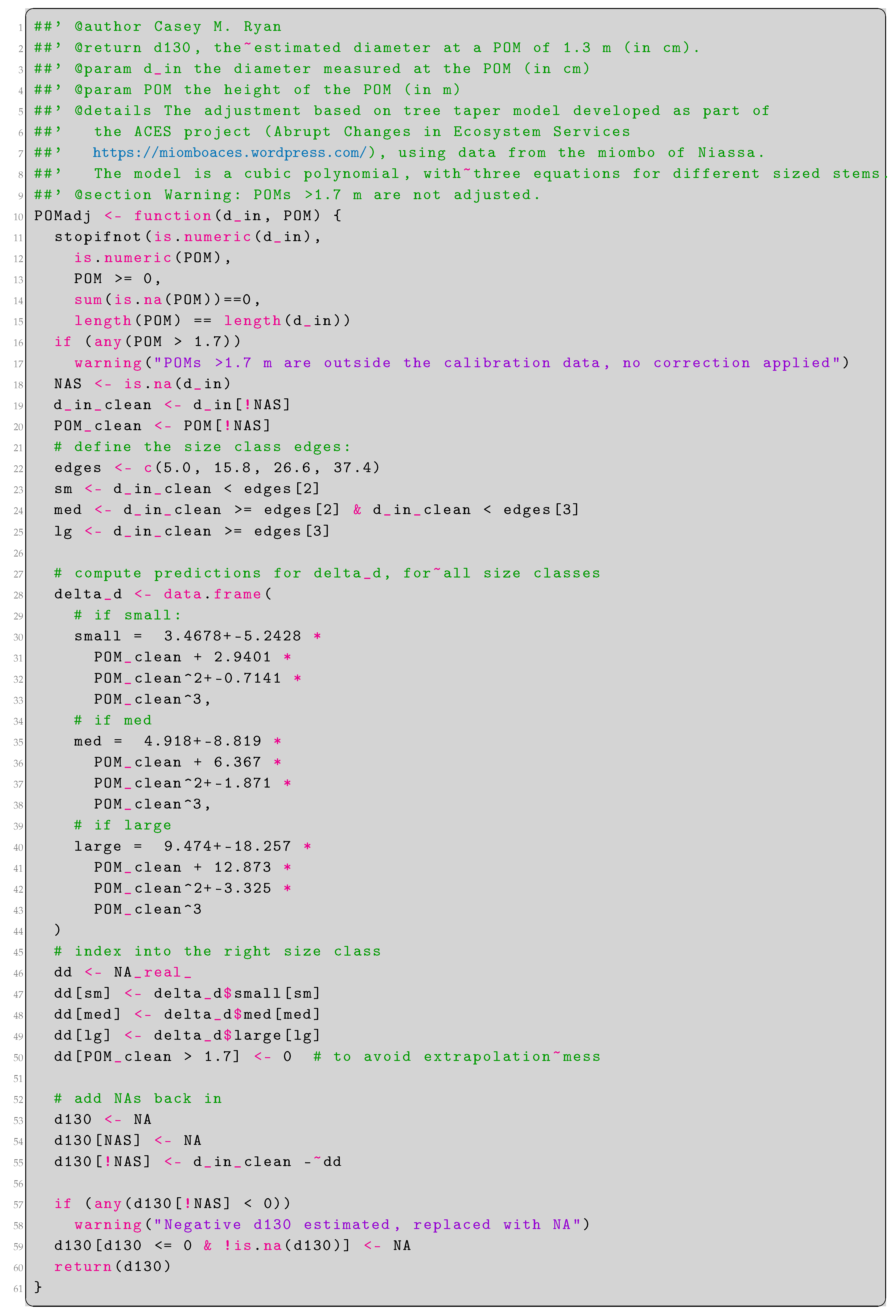
Appendix A. Estimation of Stem Diameter at 1.3 m via Tree Taper
References
- White, F. The Vegetation of Africa: A Descriptive Memoir to Accompany the UNESCO/AETFAT/UNSO Vegetation Map of Africa; The United Nations Educational, Scientific and Cultural Organization (UNESCO): Paris, France, 1983. [Google Scholar] [CrossRef]
- Mayaux, P.; Bartholomé, E.; Fritz, S.; Belward, A. A new land-cover map of Africa for the year 2000. J. Biogeogr. 2004, 31, 861–877. [Google Scholar] [CrossRef]
- Arino, O.; Perez, J.R.; Kalogirou, V.; Defourny, P.; Achard, F. Globcover 2009. In Proceedings of the ESA Living Planet Symposium, Bergen, Norway, 28 June–2 July 2010; pp. 1–3. [Google Scholar]
- Chidumayo, E. Miombo Ecology and Management: An Introduction; Intermediate Technology Publications: London, UK, 1997. [Google Scholar]
- Campbell, B.M.; Jeffrey, S.; Kozanayi, W.; Luckert, M.; Mutamba, M. Household Livelihoods in Semi-Arid Regions: Options and Constraints; Center for International Forestry Research: Bogor, Indonesia, 2002; p. 153. [Google Scholar]
- Azani, N.; Babineau, M.; Bailey, C.D.; Banks, H.; Barbosa, A.R.; Pinto, R.B.; Boatwright, J.S.; Borges, L.M.; Brown, G.K.; Bruneau, A.; et al. A new subfamily classification of the Leguminosae based on a taxonomically comprehensive phylogeny: The Legume Phylogeny Working Group (LPWG). Taxon 2017, 66, 44–77. [Google Scholar] [CrossRef] [Green Version]
- Privette, J.L.; Tian, Y.; Roberts, G.; Scholes, R.J.; Wang, Y.; Caylor, K.K.; Frost, P.; Mukelabai, M. Vegetation structure characteristics and relationships of Kalahari woodlands and savannas. Glob. Chang. Biol. 2004, 10, 281–291. [Google Scholar] [CrossRef] [Green Version]
- Caylor, K.K.; Dowty, P.R.; Shugart, H.H.; Ringrose, S. Relationship between small-scale structural variability and simulated vegetation productivity across a regional moisture gradient in southern Africa. Glob. Chang. Biol. 2004, 10, 374–382. [Google Scholar] [CrossRef]
- Chidumayo, E.N. Changes in miombo woodland structure under different land tenure and use systems in central Zambia. J. Biogeogr. 2002, 29, 1619–1626. [Google Scholar] [CrossRef]
- Ratnam, J.; Bond, W.J.; Fensham, R.J.; Hoffmann, W.A.; Archibald, S.; Lehmann, C.E.R.; Anderson, M.T.; Higgins, S.I.; Sankaran, M. When is a ‘forest’ a savanna, and why does it matter? Glob. Ecol. Biogeogr. 2011, 20, 653–660. [Google Scholar] [CrossRef]
- Dexter, K.G.; Smart, B.; Baldauf, C.; Baker, T.R.; Balinga, M.P.B.; Brienen, R.J.W.; Fauset, S.; Feldpausch, T.R.; Ferreira-da Silva, L.; Muledi, J.I.; et al. Floristics and biogeography of vegetation in seasonally dry tropical regions. Int. For. Rev. 2015, 17, 10–32. [Google Scholar] [CrossRef]
- Oliveras, I.; Malhi, Y. Many shades of green: The dynamic tropical forest-savannah transition zones. Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Dantas, V.L.; Hirota, M.; Oliveira, R.S.; Pausas, J.G. Disturbance maintains alternative biome states. Ecol. Lett. 2016, 19, 12–19. [Google Scholar] [CrossRef]
- Banda-R, K.; Delgado-Salinas, A.; Dexter, K.G.; Linares-Palomino, R.; Oliveira-Filho, A.; Prado, D.; Pullan, M.; Quintana, C.; Riina, R.; Rodríguez, G.M.; et al. Plant diversity patterns in neotropical dry forests and their conservation implications. Science 2016, 353, 1383–1387. [Google Scholar] [CrossRef] [Green Version]
- Torello-Raventos, M.; Feldpausch, T.R.; Veenendaal, E.; Schrodt, F.; Saiz, G.; Domingues, T.F.; Djagbletey, G.; Ford, A.; Kemp, J.; Marimon, B.S.; et al. On the delineation of tropical vegetation types with an emphasis on forest/savanna transitions. Plant Ecol. Divers. 2013, 6, 101–137. [Google Scholar] [CrossRef]
- Frost, P. The ecology of miombo woodlands. In The Miombo in Transition: Woodlands and Welfare in Africa; Campbell, B., Ed.; Center for International Forestry Research: Bogor, Indonesia, 1996; pp. 11–55. [Google Scholar]
- Ryan, C.M.; Pritchard, R.; McNicol, I.; Owen, M.; Fisher, J.A.; Lehmann, C. Ecosystem services from southern African woodlands and their future under global change. Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Mayaux, P.; Eva, H.; Brink, A.; Achard, F.; Belward, A. Remote sensing of land-cover and land-use dynamics. In Earth Observation of Global Change: The Role of Satellite Remote Sensing in Monitoring the Global Environment; Springer: Berlin, Germany, 2008; pp. 85–108. [Google Scholar] [CrossRef]
- Clarke, D.A.; York, P.H.; Rasheed, M.A.; Northfield, T.D. Does biodiversity-ecosystem function literature neglect tropical ecosystems. Trends Ecol. Evol. 2017, 32, 320–323. [Google Scholar] [CrossRef] [PubMed]
- Huntley, B.J.; Lages, F.; Russo, V.; Ferrand, N. (Eds.) Biodiversity of Angola: Science & Conservation: A Modern Synthesis; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef] [Green Version]
- Soares de Oliveira, R. Magnificent and Beggar Land: Angola since the Civil War; Hurst Publishers: London, UK, 2015. [Google Scholar]
- Figueiredo, E.; Smith, G.F.; César, J. The flora of Angola: First record of diversity and endemism. Taxon 2009, 58, 233–236. [Google Scholar] [CrossRef] [Green Version]
- Chisingui, A.V.; Gonçalves, F.M.P.; Tchamba, J.J.; Camôngua, L.J.; Rafael, M.F.F.; Alexandre, J.L.M. Vegetation Survey of the Woodlands of Huíla Province; Klaus Hess Publishers: Gottingen, Germany; Windhoek, Namibia, 2018. [Google Scholar] [CrossRef] [Green Version]
- Soares, M.; Abreu, J.; Nunes, H.; Silveira, P.; Schrire, B.; Figueiredo, E. The leguminosae of Angola: Diversity and endemism. Syst. Geogr. Plants 2007, 77, 141–212. [Google Scholar] [CrossRef]
- Figueiredo, E.; Smith, G.F. Plants of Angola/Plantas de Angola. Strelitzia 2008, 22, 1–279. [Google Scholar]
- Linder, H.P. Plant diversity and endemism in sub-Saharan tropical Africa. J. Biogeogr. 2001, 28, 169–182. [Google Scholar] [CrossRef]
- Droissart, V.; Dauby, G.; Hardy, O.J.; Deblauwe, V.; Harris, D.J.; Janssens, S.; Mackinder, B.A.; Blach-Overgaard, A.; Sonké, B.; Sosef, M.S.M.; et al. Beyond trees: Biogeographical regionalization of tropical Africa. J. Biogeogr. 2018, 45, 1153–1167. [Google Scholar] [CrossRef]
- Schneibel, A.; Stellmes, M.; Revermann, R.; Finckh, M.; Röder, A.; Hill, J. Agricultural expansion during the post-civil war period in southern Angola based on bi-temportal Landsat data. Biodivers. Ecol. 2013, 5, 311–319. [Google Scholar] [CrossRef] [Green Version]
- McNicol, I.M.; Ryan, C.M.; Williams, M. How resilient are African woodlands to disturbance from shifting cultivation? Ecol. Appl. 2015, 25, 2320–2336. [Google Scholar] [CrossRef]
- Gonçalves, F.M.P.; Revermann, R.; Gomes, A.L.; Aidar, M.P.M.; Finckh, M.; Juergens, N. Tree species diversity and composition of Miombo woodlands in South-Central Angola: A chronosequence of forest recovery after shifting cultivation. Int. J. For. Res. 2017, 2017, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Ryan, C.M.; Williams, M.; Grace, J. Above- and belowground carbon stocks in a miombo woodland landscape of Mozambique. Biotropica 2011, 43, 423–432. [Google Scholar] [CrossRef] [Green Version]
- McNicol, I.M.; Ryan, C.M.; Dexter, K.G.; Ball, S.M.J.; Williams, M. Aboveground carbon storage and its links to stand structure, tree diversity and floristic composition in south-eastern Tanzania. Ecosystems 2018, 21, 740–754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muledi, J.I.; Bauman, D.; Drouet, T.; Vleminckx, J.; Jacobs, A.; Lejoly, J.; Meerts, P.; Shutcha, M.N. Fine-scale habitats influence tree species assemblage in a miombo forest. J. Plant Ecol. 2017, 10, 958–969. [Google Scholar] [CrossRef] [Green Version]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Conservatoire et Jardin Botaniques de la Ville de Genève and South African National Biodiversity Institute. African Plant Database (Version 3.4.0). 2020. Available online: https://www.ville-ge.ch/musinfo/bd/cjb/africa/recherche.php (accessed on 5 November 2019).
- Palgrave, K.C. Trees of Southern Africa; Struik Publications: Cape Town, South Africa, 2003. [Google Scholar]
- Kershaw, J.A.; Ducey, M.J.; Beers, T.W.; Husch, B. Forest Mensuration; John Wiley & Sons: Chichester, UK, 2017. [Google Scholar]
- UNEP-WCMC and IUCN. Protected Planet: The World Database on Protected Areas (WDPA). 2019. Available online: https://www.protectedplanet.net/ (accessed on 15 January 2019).
- Kahle, D.; Wickham, H. ggmap: Spatial visualization with ggplot2. R J. 2013, 5, 144–161. [Google Scholar] [CrossRef] [Green Version]
- Chave, J.; Réjou-Méchain, M.; Búrquez, A.; Chidumayo, E.; Colgan, M.S.; Delitti, W.B.C.; Duque, A.; Eid, T.; Fearnside, P.M.; Goodman, R.C.; et al. Improved allometric models to estimate the aboveground biomass of tropical trees. Glob. Chang. Biol. 2014, 20, 3177–3190. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. vegan: Community Ecology Package; R Package Version 2.5-5; 2019; Available online: https://CRAN.R-project.org/package=vegan/ (accessed on 15 January 2019).
- Koleff, P.; Gaston, K.J.; Lennon, J.J. Measuring beta diversity for presence-absence data. J. Anim. Ecol. 2003, 72, 367–382. [Google Scholar] [CrossRef] [Green Version]
- Smith, B.; Wilson, J.B. A consumer’s guide to evenness indices. Oikos 1996, 76, 70–82. [Google Scholar] [CrossRef]
- Legendre, P.; De Cáceres, M. Beta diversity as the variance of community data: Dissimilarity coefficients and partitioning. Ecol. Lett. 2013, 16, 951–963. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Terra, M.C.N.S.; Santos, R.M.D.; Prado Júnior, J.A.; de Mello, J.M.; Scolforo, J.R.S.; Fontes, M.A.L.; ter Steege, H. Water availability drives gradients of tree diversity, structure and functional traits in the Atlantic-Cerrado-Caatinga transition, Brazil. J. Plant Ecol. 2018, 11, 803–814. [Google Scholar] [CrossRef] [Green Version]
- Strickland, C.; Liedloff, A.C.; Cook, G.D.; Dangelmayr, G.; Shipman, P.D. The role of water and fire in driving tree dynamics in Australian savannas. J. Ecol. 2016, 104, 828–840. [Google Scholar] [CrossRef]
- Timberlake, J.; Chidumayo, E.; Sawadogo, L. Distribution and characteristics of African dry forests and woodlands. In The Dry Forests and Woodlands of Africa: Managing for Products and Services; EarthScan: London, UK, 2010; pp. 11–42. [Google Scholar]
- Ng’andwe, P.; Chungu, D.; Shakacite, O.; Vesa, L. Abundance and distribution of top five most valuable hardwood timber species in Zambia and their implications on sustainable supply. In Proceedings of the 6th International Conference on Hardwood Processing, Lahti, Finland, 25–28 September 2017; Mottonen, V., Heinonen, E., Eds.; Natural Resources Institute Finland: Helsinki, Finland, 2017; pp. 18–27. [Google Scholar]
- Wallenfang, J.; Finckh, M.; Oldeland, J.; Revermann, R. Impact of shifting cultivation on dense tropical woodlands in southeast Angola. Trop. Conserv. Sci. 2015, 8, 863–892. [Google Scholar] [CrossRef] [Green Version]
- Sianga, K.; Fynn, R. The vegetation and wildlife habitats of the Savuti-Mababe-Linyati ecosystem, northern Botswana. KOEDOE 2017, 59, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Mukwashi, K.; Gandiwa, E.; Kativu, S. Impact of African elephants on Baikiaea Plurijuga woodland around natural and artificial watering points in northern Hwange National Park, Zimbabwe. Int. J. Environ. Sci. 2012, 2, 1355–1368. [Google Scholar] [CrossRef] [Green Version]
- Smith, P.; Allen, Q. Field Guide to the Trees and Shrubs of the Miombo Woodlands; Royal Botanic Gardens, Kew: London, UK, 2004. [Google Scholar]
- Caro, T.M. Species richness and abundance of small mammals inside and outside and African national park. Biol. Conserv. 2001, 98, 251–257. [Google Scholar] [CrossRef]
- McNicol, I.M.; Ryan, C.M.; Mitchard, E.T.A. Carbon losses from deforestation and widespread degradation offset by extensive growth in African woodlands. Nat. Commun. 2018, 9, 1–11. [Google Scholar] [CrossRef]
- Shackleton, C.M. Comparison of plant diversity in protected and communal lands in the Bushbuckridge lowveld savanna, South Africa. Biol. Conserv. 2000, 94, 273–285. [Google Scholar] [CrossRef]
- Kalaba, F.K.; Quinn, C.H.; Dougill, A.J.; Vinya, R. Floristic composition, species diversity and carbon storage in charcoal and agriculture fallows and management implications in Miombo woodlands of Zambia. For. Ecol. Manag. 2013, 304, 99–109. [Google Scholar] [CrossRef] [Green Version]
- Luoga, E.J.; Witkowski, E.T.F.; Balkwill, K. Regeneration by coppicing (resprouting) of miombo (African savanna) trees in relation to land use. For. Ecol. Manag. 2004, 189, 23–25. [Google Scholar] [CrossRef]
- Van Wyk, B.; van Wyk, P. Field Guide to Trees of Southern Africa; Struik Nature: Cape Town, South Africa, 2014. [Google Scholar]
- Muvengwi, J.; Chisango, T.; Mpakairi, K.; Mbiba, M.; Witkowski, E.T.F. Structure, composition and regeneration of miombo woodlands within harvested and unharvested areas. For. Ecol. Manag. 2020, 458, 1–10. [Google Scholar] [CrossRef]
- Chidumayo, E.N. Forest degradation and recovery in a miombo woodland landscape in Zambia: 22 years of observations on permanent sample plots. For. Ecol. Manag. 2013, 291, 154–161. [Google Scholar] [CrossRef]







| Plot Group | MAT (C) | MAP (mm y−1) | CWD (mm y−1) | Latitude (DD) | Longitude (DD) | N Plots | N Species |
|---|---|---|---|---|---|---|---|
| Bicuar NP | 20.5 | 864 | −815 | −15.12 | 14.81 | 15 | 49 |
| DRC | 20.4 | 1115 | −762 | −11.49 | 27.67 | 12 | 89 |
| Mozambique | 24.4 | 1029 | −662 | −18.95 | 34.16 | 15 | 162 |
| Tanzania | 25.8 | 956 | −754 | −9.05 | 39.05 | 22 | 248 |
| Family | Species | Stem Diam. (cm) | Basal Area (m2 ha−1) | N Stems | N Stems ha−1 |
|---|---|---|---|---|---|
| Fabaceae | Albizia antunesiana | 9.1(2.03) | 0.07(0.040) | 40 | 8(4.81) |
| Fabaceae | ⁎Baikiaea plurijuga | 28.9(0.75) | 1.72(0.570) | 331 | 55.2(17.83) |
| Fabaceae | ⁎Baphia bequaertii | 7.4(0.36) | 0.08(0.050) | 127 | 31.8(18.14) |
| Fabaceae | ⁎Baphia massaiensis | 6.6(0.17) | 0.05(0.020) | 303 | 30.3(11.20) |
| Fabaceae | Bobgunnia madagascariensis | 7.8(0.91) | 0.04(0.020) | 32 | 10.7(9.67) |
| Fabaceae | ⁎Brachystegia longifolia | 12.9(0.48) | 1.14(0.430) | 576 | 115.2(72.67) |
| Fabaceae | Brachystegia spiciformis | 11.4(0.52) | 0.74(0.430) | 326 | 81.5(46.56) |
| Phyllanthaceae | ⁎Bridelia mollis | 5.7(0.31) | 0.02(NA) | 23 | 23(NA) |
| Fabaceae | Burkea africana | 8.5(0.33) | 0.39(0.120) | 863 | 71.9(19.11) |
| Combretaceae | Combretum apiculatum | 7.6(0.45) | 0.06(0.040) | 60 | 30(15.00) |
| Combretaceae | Combretum celastroides | 5.6(0.34) | <0.01(0.000) | 7 | 3.5(2.50) |
| Combretaceae | Combretum collinum | 6.3(0.09) | 0.07(0.020) | 609 | 50.8(20.48) |
| Combretaceae | ⁎Combretum hereroense | 6.7(0.26) | 0.02(0.010) | 73 | 12.2(5.69) |
| Combretaceae | ⁎Combretum psidioides | 7.4(0.43) | 0.01(0.010) | 33 | 6.6(4.17) |
| Combretaceae | Combretum zeyheri | 6.3(0.35) | 0.01(0.000) | 61 | 10.2(3.03) |
| Euphorbiaceae | ⁎Croton gratissimus | 6.1(1.55) | <0.01(NA) | 4 | 4(NA) |
| Ebenaceae | ⁎Diospyros batocana | 8.4(2.14) | <0.01(0.000) | 2 | 1(0.00) |
| Ebenaceae | ⁎Diospyros kirkii | 9.3(1.64) | 0.03(NA) | 11 | 11(NA) |
| Apocynaceae | Diplorhynchus condylocarpon | 8.2(0.52) | 0.08(0.060) | 174 | 19.3(7.57) |
| Malvaceae | ⁎Dombeya rotundifolia | 5.5(0.19) | <0.01(NA) | 2 | 2(NA) |
| Celastraceae | ⁎Elachyptera parvifolia | 7.3(NA) | <0.01(NA) | 1 | 1(NA) |
| Meliaceae | ⁎Entandrophragma spicatum | 14.6(NA) | <0.01(NA) | 1 | 1(NA) |
| Fabaceae | Erythrophleum africanum | 9.0(0.84) | 0.10(0.040) | 128 | 18.3(6.82) |
| Rubiaceae | ⁎Gardenia volkensii | 5.6(1.15) | <0.01(0.000) | 5 | 2.5(1.50) |
| Fabaceae | ⁎Guibourtia coleosperma | 7.2(1.00) | 0.02(0.010) | 31 | 6.2(3.54) |
| Phyllanthaceae | Hymenocardia acida | 5.9(1.25) | <0.01(NA) | 6 | 6(NA) |
| Fabaceae | Julbernardia paniculata | 10.1(0.21) | 0.92(0.200) | 1624 | 162.4(50.60) |
| Fabaceae | ⁎Lonchocarpus nelsii | 13.4(0.88) | 0.15(0.030) | 165 | 15(2.77) |
| Dipterocarpaceae | ⁎Monotes angolensis | 7.4(0.83) | <0.01(0.000) | 2 | 1(0.00) |
| Ochnaceae | ⁎Ochna pulchra | 6.5(0.80) | 0.01(0.000) | 26 | 8.7(3.76) |
| Picrodendraceae | ⁎Oldfieldia dactylophylla | 8.5(NA) | <0.01(NA) | 1 | 1(NA) |
| Fabaceae | ⁎Peltophorum africanum | 11.5(NA) | <0.01(NA) | 1 | 1(NA) |
| Fabaceae | Pericopsis angolensis | 8.4(0.61) | 0.06(0.020) | 97 | 12.1(5.08) |
| Phyllanthaceae | Pseudolachnostylis maprouneifolia | 6.7(0.45) | 0.03(0.010) | 84 | 9.3(3.00) |
| Combretaceae | ⁎Pteleopsis anisoptera | 6.8(0.46) | 0.07(0.020) | 81 | 20.2(15.11) |
| Fabaceae | Pterocarpus angolensis | 13.0(0.61) | 0.15(0.100) | 102 | 17(8.65) |
| Fabaceae | ⁎Pterocarpus lucens | 6.9(0.94) | <0.01(NA) | 4 | 4(NA) |
| Rubiaceae | ⁎Rothmannia engleriana | 6.8(0.66) | <0.01(0.000) | 5 | 1.7(0.67) |
| Euphorbiaceae | ⁎Schinziophyton rautanenii | 8.0(2.82) | <0.01(NA) | 3 | 3(NA) |
| Polygalaceae | Securidaca longepedunculata | 7.3(1.12) | <0.01(0.010) | 4 | 2(1.00) |
| Loganiaceae | Strychnos cocculoides | 10.4(1.17) | 0.03(0.020) | 19 | 6.3(3.53) |
| Loganiaceae | ⁎Strychnos pungens | 6.1(0.48) | <0.01(0.000) | 18 | 3.6(0.93) |
| Loganiaceae | Strychnos spinosa | 6.8(0.36) | 0.02(0.010) | 97 | 9.7(4.07) |
| Combretaceae | ⁎Terminalia brachystemma | 6.5(0.21) | 0.04(0.020) | 174 | 29(12.04) |
| Combretaceae | Terminalia sericea | 7.1(0.28) | 0.06(0.030) | 214 | 23.8(12.18) |
| Ximeniaceae | Ximenia americana | 6.1(0.53) | <0.01(0.000) | 7 | 1.8(0.25) |
| Sapindaceae | Zanha africana | 9.4(1.12) | 0.01(NA) | 6 | 6(NA) |
| Rhamnaceae | ⁎Ziziphus abyssinica | 5.9(1.13) | <0.01(NA) | 2 | 2(NA) |
| Dependent Variable: | ||||
|---|---|---|---|---|
| Species Richness | Basal Area | Shannon () | Shannon Equit. () | |
| (1) | (2) | (3) | (4) | |
| DRC | 27.920 *** | 4.175 *** | 1.055 *** | 0.080 |
| (5.538) | (0.452) | (0.236) | (0.053) | |
| Tanzania | 12.440 ** | −0.721 * | 0.605 *** | 0.064 |
| (4.788) | (0.391) | (0.204) | (0.046) | |
| Mozambique | 27.930 *** | 0.653 | 0.792 *** | 0.028 |
| (5.221) | (0.427) | (0.223) | (0.050) | |
| Constant | 14.330 *** | 2.778 *** | 1.617 *** | 0.631 *** |
| (3.692) | (0.302) | (0.158) | (0.035) | |
| Observations | 64 | 64 | 64 | 64 |
| Adjusted R | 0.363 | 0.691 | 0.237 | 0.003 |
| Residual Std. Error (df = 60) | 14.300 | 1.168 | 0.611 | 0.137 |
| F Statistic (df = 3; 60) | 12.980 *** | 48.040 *** | 7.537 *** | 1.000 |
| Site 1 | Site 2 | Shared Species | |
|---|---|---|---|
| Bicuar NP (34) | DRC (74) | 20.6 | 14 |
| Bicuar NP (34) | Tanzania (147) | 13.4 | 14 |
| Bicuar NP (37) | Mozambique (236) | 7.5 | 11 |
| DRC (64) | Tanzania (137) | 19.3 | 24 |
| DRC (69) | Mozambique (228) | 11.3 | 19 |
| Tanzania (139) | Mozambique (225) | 10.8 | 22 |
| Dependent Variable: | ||||
|---|---|---|---|---|
| Species Richness | Basal Area | Shannon () | Shannon Equit. () | |
| Disturbed | 2.450 *** | 0.098 | 0.372 ** | 0.035 |
| (0.859) | (0.122) | (0.140) | (0.045) | |
| Constant | 6.200 *** | 0.416 *** | 1.311 *** | 0.756 *** |
| (0.650) | (0.092) | (0.106) | (0.034) | |
| Observations | 35 | 35 | 35 | 35 |
| R | 0.198 | 0.019 | 0.176 | 0.018 |
| Residual Std. Error (df = 33) | 2.516 | 0.357 | 0.410 | 0.131 |
| F Statistic (df = 1; 33) | 8.126 *** | 0.639 | 7.040 ** | 0.617 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Godlee, J.L.; Gonçalves, F.M.; Tchamba, J.J.; Chisingui, A.V.; Muledi, J.I.; Shutcha, M.N.; Ryan, C.M.; Brade, T.K.; Dexter, K.G. Diversity and Structure of an Arid Woodland in Southwest Angola, with Comparison to the Wider Miombo Ecoregion. Diversity 2020, 12, 140. https://doi.org/10.3390/d12040140
Godlee JL, Gonçalves FM, Tchamba JJ, Chisingui AV, Muledi JI, Shutcha MN, Ryan CM, Brade TK, Dexter KG. Diversity and Structure of an Arid Woodland in Southwest Angola, with Comparison to the Wider Miombo Ecoregion. Diversity. 2020; 12(4):140. https://doi.org/10.3390/d12040140
Chicago/Turabian StyleGodlee, John L., Francisco Maiato Gonçalves, José João Tchamba, Antonio Valter Chisingui, Jonathan Ilunga Muledi, Mylor Ngoy Shutcha, Casey M. Ryan, Thom K. Brade, and Kyle G. Dexter. 2020. "Diversity and Structure of an Arid Woodland in Southwest Angola, with Comparison to the Wider Miombo Ecoregion" Diversity 12, no. 4: 140. https://doi.org/10.3390/d12040140
APA StyleGodlee, J. L., Gonçalves, F. M., Tchamba, J. J., Chisingui, A. V., Muledi, J. I., Shutcha, M. N., Ryan, C. M., Brade, T. K., & Dexter, K. G. (2020). Diversity and Structure of an Arid Woodland in Southwest Angola, with Comparison to the Wider Miombo Ecoregion. Diversity, 12(4), 140. https://doi.org/10.3390/d12040140





