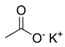
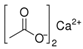
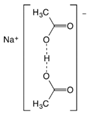
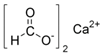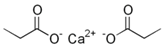Abstract
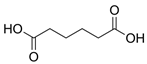
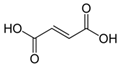
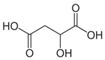
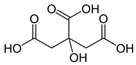
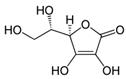
Organic food additives are popular in various spheres of human activity: the food industry, agriculture, veterinary and human medicine, the chemical industry, and other sectors. Due to the relatively short time of their breakdown in the environment, their non-toxicity to various organisms in certain concentrations, and their large annual amounts of production, application, and influx into the environment, it is an important task to study their influence on ecology. To help reduce toxic pressure on the environment, we studied the influence of 32 organic food additives on larvae of the nematodes Strongyloides papillosus, Haemonchus contortus, Muellerius capillaris, which are parasites of the digestive tracts of ruminants. This article presents the results of in vitro studies of organic food additives (acetic, propionic, lactic, sorbic, adipic, fumaric, malic, citric, ascorbic, and dehydroacetic acids, sodium formate, calcium formate, potassium acetate, calcium acetate, sodium diacetate, calcium propanoate, sodium lactate, potassium sorbate, sodium sorbate, calcium sorbate, trisodium citrate, monosodium glutamate, sodium dehydroacetate, sodium benzoate, potassium benzoate, calcium benzoate, biphenyl, dibutylhydroxytoluene, hexamethylenetetramine, 2-phenylphenol, natamycin, and nisin) on nematode larvae at various stages of their development. Propionic acid and 2-phenylphenol, used in the food industry, exerted notable nematicidal effects on larvae of S. papillosus, M. capillaris, and H. contortus: 1% concentrations of the substances killed 85% of these larvae, except H. contortus, which were tolerant to 2-phenylphenol in 52% of cases. Other tested food additives at 1% concentrations could not kill more than 70% of invasive nematode larvae in 24 h. The research determined various degrees of toxicity of the studied substances to invasive (third age) and non-invasive (first and second age) larvae, which are related to a decrease in the feeding intensity of parasitic nematodes on the stage of third age larvae. Thus, some organic food additives, introduced into the environment with food wastes, are able to affect the vitality of certain nematode species to a varying extent.
1. Introduction
Food additives play a significant role in the diet of modern people because most ready-to-use foods available in stores (products of advanced and highly advanced processing) contain food additives [1]. The greater the processing of the product, and the higher the temperature at which it is stored, the more its content of preservatives, stabilizers, emulsifiers, and other substances that prolong its lifetime on the market. Therefore, many products contain 3–5% rather than 1% of food additives, in relation to their mass [1,2]. The world’s population consumes food additives in varying amounts: people with a high income consume fewer additives, because they are aware of the harm many of those substances cause to health; the poorest portion of the Earth’s population and people living in rural area consume relatively small quantities of additives. The greatest quantities of additives are consumed by city residents in countries having low and average levels of income per capita.
Produced food additives, often in unused products that are thrown away, end up in landfills of solid municipal wastes: in rich countries, food waste accounts for 20–25% of the total amount of food; in poor countries, it accounts for 10% of food. Thus, at least 10% of additives (i.e., 2.81 M T per year) are introduced into the environment unchanged. In addition, some additives consumed by people, after passing through the intestine, enter sewage systems unaltered (and, in some of the poorest countries, directly into the environment without any filtrating system). These food additives create a constant source of contamination in territories around settlements [2,3,4,5,6]. The greater portion of organic substances is broken down quickly by soil microorganisms [7], but some food additives are able to influence fauna of natural ecosystems [8]. Therefore, the effects of various concentrations of organic food additives on different groups of soil invertebrates have become a subject of interest.
The influence of food additives has only been studied for separate groups of invertebrates: Acari [9,10], insects [11,12,13,14,15], and nematodes [16,17,18,19]. In some gradients of their concentration, food additives are able to cause migration of certain invertebrates [20], alter the intensity of metabolic processes in living organisms [21], or cause their death [14,15].
A significant proportion of food additives are totally safe for natural ecosystems, because their molecules enter biochemical processes in cytoplasm and mitochondria of cells: for example, acetic acids, lactic acid, citric acid, ascorbic acid, potassium acetate, calcium acetate, and sodium diacetate. Other additives, for example, biphenyl, 2-phenylphenol, hexamethylenetetramine, are absent inside cells of living organisms and the environment, and therefore their influence on invertebrates is of great interest from an ecological perspective, and in the study of possibilities for regulating certain groups of organisms that are dangerous for humans and agriculture [2]. To date, the scientific literature has accumulated a large amount of data on the properties of organic food additives and their application in the food industry. However, we are faced with a lack of information about the effect of these substances on different groups of living organisms. Such studies are innovative in the field of pest control of farm animals. Experiments on the effects of food additives on nematodes provide an opportunity to create alternative methods in relation to classical therapeutic and prophylactic measures in veterinary medicine. The objectives of this article were to examine the effects of broadly used organic food additives on the vitality of nematode larvae, which are parasites of farm mammals, in laboratory conditions.
2. Materials and Methods
Feces of ruminants were collected in the territory of the clinical-diagnostic center of the Dnipro National Agrarian-Economic University (Dnipropetrovsk Oblast, Ukraine). Goats were naturally infected (while grazing in meadows) with Strongyloides papillosus (Wedl, 1856), Haemonchus contortus (Rudolphi, 1803) and Muellerius capillaris (Muller, 1889). To find eggs of helminths, the samples were studied using the McMaster technique [22]. Larvae of S. papillosus and H. contortus were cultivated in feces for 10 days at the temperature of 18–22 °C. To discover larvae of S. papillosus and H. contortus and M. capillaris, we used the Baermann test [22]. The experiment was carried out on first and second age larvae (L1, L2) and third age larvae (L3) of S. papillosus, third age (L3) H. contortus, and first age (L1) M. capillaris, living in the environment. The larvae were identified based on morphological parameters [23,24]. We took into account the internal structure of the larvae: presence of intestinal cells, their number, number of rows in which the cells were arranged, shape of cells, and form of the esophagus.
After finding and identification, the larvae were placed in water (4 mL) in test tubes of 10 mL and centrifuged for 4 min at 1500 rpm. After removing the supernatant, the larvae in the vessel were uniformly stirred and placed into 1.5 mL plastic test tubes, with 0.1 mL in each. Then, solutions (1%, 0.1%, 0.01%) of the studied substances were alternately added to cultivated larvae. Distilled water was used as a control. The exposure lasted 24 h at the temperature of 22 °C. In the process of the experiment, after exposure to the tested food additives, we determined the numbers of live and dead individuals. When confirming death of the larvae, we took into account the combination of two factors: the mobility of larvae and the breakdown of the intestinal tissue.
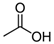
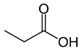
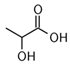
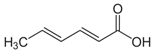
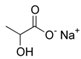
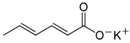
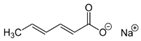
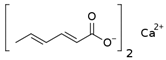
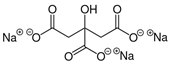
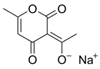
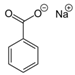
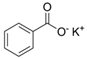
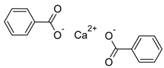
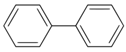
Larvae were subjected to the influence of organic food additives (Table 1 and Table 2) in five replications for each of the variants of the experiment. For the experiment, we used acetic, propionic, lactic, sorbic, adipic, fumaric, malic, citric, ascorbic, and dehydroacetic acids, sodium formate, calcium formate, potassium acetate, calcium acetate, sodium diacetate, calcium propanoate, sodium lactate, potassium sorbate, sodium sorbate, calcium sorbate, trisodium citrate, monosodium glutamate, sodium dehydroacetate, sodium benzoate, potassium benzoate, calcium benzoate, biphenyl, dibutylhydroxytoluene, hexamethylenetetramine, 2-phenylphenol, natamycin, and nisin (Alma-Veko Food, Kyiv, Ukraine).

Table 1.
Use of 32 organic food additives utilized for determining level of survivability of nematodes in the laboratory experiment.

Table 2.
Maximum level (mg/kg) of food additives permitted for use by the Codex Alimentarius in basic human foods.
The statistical analysis of the results was performed using Statistica 12 (StatSoft Inc., Tulsa, OK, USA). Tables present average value (x) ± standard deviation (SD). Differences between the values of the control and experimental groups were determined using the Tukey test, where the differences were considered significant at p < 0.05.
3. Results
The results of the research on the influence of organic acids used in the food industry on the vitality of nematodes of ruminants (Table 3) revealed notable nematicidal properties of propionic acid. At 24 h, its 1% solution killed over 85% of larvae of the tested species of nematodes at various stages of development. At the same time, a high proportion of mortality was observed for non-invasive larvae, in addition to more-tolerant invasive larvae. The larvae that were most tolerant to 1% concentration of this acid were invasive H. contortus. Nonetheless, over 87.5% of these died in the 24 h experiment.

Table 3.
Mortality of larvae of S. papillosus, H. contortus, and M. capillaris (%) during 24 h laboratory experiment under the influence of organic acids used as food additives (x ± SD, n = 5).
Other acids exerted various effects on the vitality of larvae of nematode species at different stages of their development. Therefore, acetic, adipic, lactic, fumaric, citric, and ascorbic acids had the strongest effects on S. papillosus larvae of the first and second stages and M. capillaris of the first stage. Invasive larvae of S. papillosus and H. contortus were most tolerant to those acids. Sorbic and malic acids displayed actions only toward non-invasive stages of S. papillosus.
In general, the study of the influence of salts of organic acids on the vitality of nematode larvae of ruminants revealed a smaller effect compared with the acids (Table 4). Third-stage larvae of H. contortus remained vital for 24 h or longer in more than 85% of cases. At the same time, no damaged intestinal cells were found in nematodes. Invasive larvae of S. papillosus were also affected by salts of organic acids, but no more often than in 40% cases (calcium propanoate). During the 24 h period, most tested salts of organic acids in 1% concentration, and also their weaker solutions, had no significant effect on the vitality of invasive nematode larvae.

Table 4.
Mortality of larvae of S. papillosus, H. contortus, and M. capillaris (%) during 24 h laboratory experiment under the influence of salts of organic acids, used as food additives (x ± SD, n = 5).
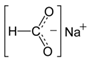
Less tolerant to the exposure to salts of organic salts were non-invasive stages of nematode larvae. Therefore, under 24 h of the influence of 1% solution of calcium propanoate, no more than 10% of first-stage larvae of M. capillaris remained vital. Sodium formate also had a significant effect on M. capillaris. Over 50% of first-stage larvae of this nematode species died in 1% solution of this food additive. Moreover, sodium formate had an effect on non-invasive stages of S. papillosus larvae. S. papillosus of this age were also affected by potassium acetate and sodium diacetate (more than 50% of them died during 24 h exposure to 1% solutions).
Research on the influence of salts of organic food additives of other groups on nematode larvae of ruminants revealed a significant effect of 2-phenylphenol (Table 5). Twenty-four-hour exposure to its 1% solution was lethal for all larvae of S. papillosus and M. capillaris. The most tolerant larvae to this substance were invasive H. contortus. Despite the fact that the proportion of vital third-stage larvae of this species was 51%, 0.1% concentration of 2-phenylphenol also significantly affected the first- and second-stage S. papillosus, and also first-stage M. capillaris. Over 55% of these larvae died in 24 h under the influence of 0.1% concentration of 2-phenylphenol. However, third-stage larvae of S. papillosus and H. contortus were more tolerant to this concentration of 2-phenylphenol. Biphenyl and hexamethylenetetramine in 1% concentrations caused death to more than 50% of first- and second-stage larvae of S. papillosus. The remaining tested food additives in Table 4 had no significant effects on either invasive or non-invasive nematode larvae.

Table 5.
Mortality of larvae of S. papillosus, H. contortus, and M. capillaris (%) during 24 h laboratory experiment under the influence of salts of organic food additives of various groups (x ± SD, n = 5).
4. Discussion
Studies into combating parasites of agricultural animals deal not only with influence of nematicidal drugs, but also substances used in the food industry, and plants that may be used as food additives for agricultural animals. Our previous studies [27,28] were focused on effects on medicinal plants on the vitality of nematodes in the environment.
Organic food additives are used not only in the food industry, they are also universal ingredients used in pharmaceutics and perfumery as solvents, and in the oil and textile industries. These substances are constituents of detergents, plastics, rubbers, colorings, and glues [29], and they are broadly used in the production of chemical substances in the automobile and construction industries. Over the last few years, there have been many experimental studies of the use of organic food additives in agriculture for fighting pests. This direction is especially popular in organic arable farming and animal husbandry, where drugs and other chemically synthesized substances are prohibited. Many farms are currently reducing the amounts of pesticides they use in order to decrease toxic pressure on natural ecosystems. Their negative impact on ecosystems manifests through accumulation in the environment, because they are do not break down easily and can remain in the soil for a long time. Thus, pesticides are harmful to the health of animals and people, and also have negative effects on soil microorganisms. The term “biopesticides”, referring to substances present in nature, is being used increasingly often. These are also called natural pesticides. This category includes components of some plants, bacteria, fungi, and other living organisms. They may also be substances obtained from natural sources [30,31]. Santos et al. [32] described the properties of Bacillus thuringiensis, which is used in the compound of bioinsecticides. This species of bacteria produces toxins and spores, which are toxic for some species of insects. The efficacy of those bioinsecticides depends on the quality of their application. Good results have been shown by the combined use of bioinsecticides and some propionic acid-based adjuvants.
Some organic food substances are also biopesticides. The Ukrainian Certifying Body “Organic Standard” is the leader in organic certification in Ukraine, and some other international certifying bodies (bio.inspecta, CERES, Control Union and others) give certificates in Ukraine, based on the law of the European Union on Organic Agriculture. The list of substances that are allowed for use as food additives for animals contains such organic acids as sorbic, formic, acetic, proprionic, and citric acids. Some of these acids (formic, acetic, and oxalic) are allowed as products for disinfection. There are numerous data about the use of formic acid as a biocaricide to combat the honey bee parasite Varroa destructor [33,34,35]. Laboratory experiments aimed at evaluation of the efficiency of various concentrations of formic acid against Acari in different temperature conditions were conducted by Underwood and Currie [33,34]. Our previous studies of this food additive also indicated nematicidal properties of formic acid against nematode larvae, which are parasites of ruminants. The lowest LD50 values were observed for formic acid: this parameter equaled 4.7 g/kg on average for third-stage larvae of S. papillosus, 0.076 g/kg for first- and second-stage larvae of S. papillosus, and 4.1 g/kg for third-stage larvae of H. contortus [36].
Beyhan et al. [37] evaluated the influence of acetic acid on eggs of Ascaris lumbricoides, which are nematodes of the digestive tract of humans: 1% solution of acetic acid caused no death to the ascarids; however, after increasing the concentration to 3%, 95% of the nematodes died. The influence of 5% acetic acid resulted in the death of all the eggs of this nematode species. One of the studies [38] was focused on the influence of short-chained and long-chained fatty acids on the vitality of eggs of Ascaris suum, which are parasites of the digestive tract of swine. The main objective of these studies was to determine a safe and effective method of inactivation of eggs of parasites in wastewater, without causing a negative impact on the environment. Complete inactivation (100%) of eggs of A. suum by acetic acid was observed for mixtures of acetic acid (288 mM), butyric acid (240 mM), and hexanoic acid (16 mM); and acetic acid (288 mM), butyric acid (240 mM), valeric acid (16 mM), and hexanoic acid (16 mM) [38]. Acetic acid is also used to dehydrate stained helminths [39]. Rojas-Oropeza et al. [40] studied the influence of organic acids used in the food industry on the vitality of eggs of A. suum, finding a mixture of acetic, propionic-butyric, valeric, and isovaleric acids to be effective. We also observed the effect of acetic and propionic acids on the vitality of nematode larvae of ruminants. The larvae most susceptible to acetic acids were non-invasive S. papillosus and M. capillaris. These acids may be more effective against invasive larvae in mixtures rather than the pure substances analyzed in the article. We obtained these results after exposing nematodes to lactic, ascorbic, adipic, and citric acids. However, the greatest impact on the vitality of nematode larvae of various stages was caused by 1% solution of propionic acid.
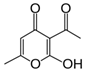
As an insecticide and fungicide, Dunkel [41] proposed using sorbic acid. It has notable insecticidal and fungicidal properties. This acid was registered in the Agency of Environmental Protection (USA) as a safe substance for maintenance of grain and maize. The concentration needed to effectively combat fungi was equal to 0.3%, whereas against some insects it was 0.5%. Furthermore, an important factor is the insect species, because a difference in concentration of even 0.1% may decrease insecticidal influence. No notable nematocidal properties of sorbic acid against nematode larvae of ruminants were found. Even in 1% concentration, over 95% of invasive larvae remained vital at 24 h. This concentration of sorbic acid only produced 100% mortality of non-invasive larvae of S. papillosus.
The scientific literature often contains data about natamycin, which is the food additive preservative E235. This substance is obtained using Streptomyces natalensis bacteria. It has notable fungicidal properties [42,43]. However, according to the results of studies, natamycin is not active against nematode larvae, which are parasites of agricultural animals. Over 80% of first- and third-stage larvae of the studied species of nematodes remained vital and mobile after 24 h of exposure.
Polychlorinated biphenyls are chemical substances that are stable in the environment. They can exert various toxic effects on living organisms and have high tolerance to bioaccumulation [44]. As our experiment on nematode larvae in the environment demonstrated, biphenyl, unbound to chlorine and used as a food additive, varies in effect on different species of parasites. The lowest tolerance to biphenyl was observed for third-stage larvae of S. papillosus, whereas less than 30% of invasive larvae died. Non-invasive stages of larvae of this species of nematodes were found to be less tolerant; we observed death of over 50% of larvae.
5. Conclusions
The food industry is not the only sphere in which the organic substances are valuable. Some of these substances are now used to combat agricultural pests. According to the results of our studies, the food additives propionic acid and 2-phenylphenol also have nematocidal properties against first- and second-stage larvae of S. papillosus, first-stage M. capillaris, and invasive larvae of S. papillosus and H. contortus. Invasive larvae of S. papillosus were also found to be affected by sodium formate, sodium diacetate, potassium benzoate, sodium dehydroacetate, and calcium propanoate, but no more often than in 45% of cases. Non-invasive larvae, with the exception of the substances indicated above, were also susceptible to acetic, adipic, lactic, fumaric, citric, and ascorbic acids, calcium propanoate, and potassium acetate.
Food additives that are introduced into the environment in relatively small concentrations can have local negative effects on nematodes living in the soil (including free-living larval stages of nematodes, which are parasites of agricultural animals and humans). By comparison, the relatively well-studied safety of food additives for humans suggests that the potentials of these substances are not fully realized against parasitic nematodes in farms, in the disinfection of places where animals are maintained, in addition to tools and equipment used in farming.
Author Contributions
Conceptualization, O.B. and V.B.; methodology, O.B.; validation, V.B.; formal analysis, V.B.; investigation, O.B.; resources, O.B. and V.B.; data curation, O.B. and V.B.; writing–original draft preparation, O.B. and V.B.; writing–review and editing, O.B. and V.B.; visualization, O.B. and V.B.; supervision, O.B. and V.B.; project administration, O.B.; funding acquisition, O.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Ministry of Education and Science of Ukraine, grant number 0120U102384 “Ecological evaluation of use of food and fodder additives in animal husbandry”.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Carocho, M.; Barreiro, M.F.; Morales, P.; Ferreira, I.C.F.R. Adding molecules to food, pros and cons: A review on synthetic and natural food additives. Compr. Rev. Food Sci. Food Saf. 2014, 13, 377–399. [Google Scholar] [CrossRef] [PubMed]
- Skovgaard, N. Safety evaluation of certain food additives and contaminants. Int. J. Food Microbiol. 2004, 90, 115–116. [Google Scholar] [CrossRef]
- Birch, G.F.; Drage, D.S.; Thompson, K.; Eaglesham, G.; Mueller, J.F. Emerging contaminants (pharmaceuticals, personal care products, a food additive and pesticides) in waters of Sydney estuary, Australia. Mar. Pollut. Bull. 2015, 97, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Dorier, M.; Béal, D.; Tisseyre, C.; Marie-Desvergne, C.; Dubosson, M.; Barreau, F.; Houdeau, E.; Herlin-Boime, N.; Rabilloud, T.; Carriere, M. The food additive E171 and titanium dioxide nanoparticles indirectly alter the homeostasis of human intestinal epithelial cells in vitro. Environ. Sci. Nano 2019, 6, 1549–1561. [Google Scholar] [CrossRef]
- Anim, A.K.; Thompson, K.; Duodu, G.O.; Tscharke, B.; Birch, G.; Goonetilleke, A.; Ayoko, G.A.; Mueller, J.F. Pharmaceuticals, personal care products, food additive and pesticides in surface waters from three Australian east coast estuaries (Sydney, Yarra and Brisbane). Mar. Pollut. Bull. 2020, 153, 111014. [Google Scholar] [CrossRef] [PubMed]
- Bellani, L.; Muccifora, S.; Barbieri, F.; Tassi, E.; Castiglione, M.R.; Giorgetti, L. Genotoxicity of the food additive E171, titanium dioxide, in the plants Lens culinaris L. and Allium cepa L. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2020, 849, 503142. [Google Scholar] [CrossRef] [PubMed]
- Van Veen, J.A.; Kuikman, P.J. Soil structural aspects of decomposition of organic matter by microorganisms. Biogeochemistry 1990, 11, 213–233. [Google Scholar] [CrossRef]
- Tkaczyk, A.; Mitrowska, K.; Posyniak, A. Synthetic organic dyes as contaminants of the aquatic environment and their implications for ecosystems: A review. Sci. Total Environ. 2020, 717, 137222. [Google Scholar] [CrossRef]
- Lee, H.S. P-anisaldegydae: Acaricidal component of Pimpinella anisum seed oil against the house dust mites Dermatophagoides farina and Dermatophagoides pteronyssinus. Planta Med. 2004, 70, 279–281. [Google Scholar] [CrossRef]
- Shen, F.; Xing, M.; Liu, L.; Tang, X.; Wang, W.; Wang, X.; Wu, X.; Wang, X.; Wang, X.; Wang, G.; et al. Efficacy of trans-cinnamaldehyde against Psoroptes cuniculi in vitro. Parasitol. Res. 2012, 110, 1321–1326. [Google Scholar] [CrossRef]
- Lee, E.J.; Kim, J.R.; Choi, D.R.; Ahn, Y.J. Toxicity of cassia and cinnamon oil compounds and cinnamaldehyde-related compounds to Sitophilus oryzae (Coleoptera: Curculionidae). J. Econ. Entomol. 2008, 101, 1960–1966. [Google Scholar] [CrossRef] [PubMed]
- Knoblauch, C.; Fry, K. Non-Toxic Insecticide. U.S. Patent 7956092, 7 June 2011. [Google Scholar]
- Ullah, I.; Latif, A.K.; Ali, L.; Khan, A.R.; Waqas, M.; Hussain, J.; Lee, I.J.; Shin, J.H. Benzaldehyde as an insecticidal, antimicrobial, and antioxidant compound produced by Photorhabdus temperata M1021. J. Microbiol. 2015, 53, 127–133. [Google Scholar] [CrossRef]
- Martynov, V.O.; Brygadyrenko, V.V. The influence of synthetic food additives and surfactants on the body weight of larvae of Tenebrio molitor (Coleoptera, Tenebrionidae). Biosyst. Divers. 2017, 25, 236–242. [Google Scholar] [CrossRef]
- Martynov, V.O.; Brygadyrenko, V.V. The influence of the synthetic food colourings tartrazine, allura red and indigo carmine on the body weight of Tenebrio molitor (Coleoptera, Tenebrionidae) larvae. Regul. Mech. Biosyst. 2018, 9, 479–484. [Google Scholar] [CrossRef]
- Katiki, L.M.; Barbieri, A.M.E.; Araujo, R.C.; Veríssimo, C.J.; Louvandini, H.; Ferreira, J.F.S. Synergistic interaction of ten essential oils against Haemonchus contortus in vitro. Vet. Parasitol. 2017, 243, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Boyko, O.O.; Brygadyrenko, V.V. The impact of certain flavourings and preservatives on the survivability of larvae of nematodes of Ruminantia. Regul. Mech. Biosyst. 2018, 9, 118–123. [Google Scholar] [CrossRef]
- Boyko, O.O.; Brygadyrenko, V.V. The impact of acids approved for use in foods on the vitality of Haemonchus contortus and Strongyloides papillosus (Nematoda) larvae. Helminthologia 2019, 56, 202–210. [Google Scholar] [CrossRef]
- Boyko, O.O.; Brygadyrenko, V.V. The impact of certain flavourings and preservatives on the survivability of eggs of Ascaris suum and Trichuris suis. Regul. Mech. Biosyst. 2020, 11, 344–348. [Google Scholar] [CrossRef]
- Martynov, V.O.; Titov, O.G.; Kolombar, T.M.; Brygadyrenko, V.V. Influence of essential oils of plants on the migration activity of Tribolium confusum (Coleoptera, Tenebrionidae). Biosyst. Divers. 2019, 27, 177–185. [Google Scholar] [CrossRef]
- Kolombar, T.M.; Gugosyan, Y.A.; Brygadyrenko, V.V. Impact of mineral fertilizers, growth stimulators, pH regulators, vitamins and pigment supplements on the vitality of entomopathogenic nematodes of Steinernematidae and Heterorhabditidae families. Regul. Mech. Biosyst. 2020, 11, 323–329. [Google Scholar] [CrossRef]
- Zajac, A.M.; Conboy, G.A. Veterinary Clinical Parasitology, 8th ed.; Willey-Blackwell: New York, NY, USA, 2011; 354p. [Google Scholar]
- Van Wyk, A.; Cabaret, J.; Michael, L.M. Morphological identifcation of nematode larvae of small ruminants and cattle simplifed. Vet. Parasitol. 2004, 119, 277–306. [Google Scholar] [CrossRef]
- Van Wyk, J.A.; Mayhew, E. Morphological identifcation of parasitic nematode infective larvae of small ruminants and cattle: A practical lab guide. Onderstepoort J. Vet. Res. 2013, 80, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Branen, A.L.; Davidson, P.M.; Salminen, S.; Thorngate, J. Food Additives; Marcel Dekker Inc.: New York, NY, USA; Basel, Switzerland, 2002; 938p. [Google Scholar]
- Singh, R.; Kumar, M.; Mittal, A.; Mehta, P.K. Microbial metabolites in nutrition, healthcare and agriculture. 3 Biotech 2017, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Boyko, O.O.; Brygadyrenko, V.V. Nematocidial activity of aqueous solutions of plants of the families Cupressaceae, Rosaceae, Asteraceae, Fabaceae, Cannabaceae and Apiaceae. Biosyst. Divers. 2019, 27, 227–232. [Google Scholar] [CrossRef]
- Boyko, O.O.; Kabar, A.M.; Brygadyrenko, V.V. Nematicidal activity of aqueous tinctures of medicinal plants against larvae of the nematodes Strongyloides papillosus and Haemonchus contortus. Biosyst. Divers. 2020, 28, 119–123. [Google Scholar] [CrossRef]
- Sun, X.; Shen, X.; Jain, R.; Lin, Y.; Wang, J.; Sun, J.; Wang, J.; Yan, Y.; Yuan, O. Synthesis of chemicals by metabolic engineering of microbes. Chem. Soc. Rev. 2015, 44, 3760–3785. [Google Scholar] [CrossRef]
- Oguh, C.E.; Okpaka, C.O.; Ubani, C.S.; Okekeaji, U.; Joseph, P.S.; Amadi, E.U. Natural pesticides (biopesticides) and uses in pest management-a critical review. Asian J. Biotechnol. Genet. Eng. 2019, 2, 1–18. [Google Scholar]
- Baweja, P.; Kumar, S.; Kumar, G. Fertilizers and pesticides: Their impact on soil health and environment. In Soil Health. Soil Biology; Giri, B., Varma, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2020; Volume 59, pp. 265–285. [Google Scholar] [CrossRef]
- Santos, C.A.M.D.; Santos, R.T.D.S.; Della’Vechia, J.F.; Griesang, F.; Polanczyk, R.A.; Ferreira, M.D.C. Effect of addition of adjuvants on physical and chemical characteristics of Bt bioinsecticide mixture. Sci. Rep. 2019, 9, 12525. [Google Scholar] [CrossRef]
- Underwood, R.M.; Currie, R.W. The effects of temperature and dose of formic acid on treatment efficacy against Varroa destructor (Acari: Varroidae), a parasite of Apis mellifera (Hymenoptera: Apidae). Exp. Appl. Acarol. 2003, 29, 303. [Google Scholar] [CrossRef]
- Underwood, R.M.; Currie, R.W. Effects of release pattern and room ventilation on survival of Varroa mites and queens during indoor winter fumigation of honey bee colonies with formic acid. Can. Entomol. 2007, 139, 881–893. [Google Scholar] [CrossRef]
- Ostermann, D.J.; Currie, R.W. Effect of formic acid formulations on honey bee (Hymenoptera: Apidae) colonies and influence of colony and ambient conditions on formic acid concentration in the hive. J. Econ. Entomol. 2004, 97, 1500–1508. [Google Scholar] [CrossRef]
- Boyko, O.O.; Brygadyrenko, V.V. The viability of Haemonchus contortus (Nematoda, Strongylida) and Strongyloides papillosus (Nematoda, Rhabditida) larvae exposed to various flavourings and source materials used in food production. Vestn. Zool. 2019, 53, 433–442. [Google Scholar] [CrossRef]
- Beyhan, Y.E.; Yilmaz, H.; Hokelek, M. Effects of acetic acid on the viability of Ascaris lumbricoides eggs. Is vinegar reliable enough to clean the vegetables? Saudi Med. J. 2016, 37, 288–292. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D. Inactivation of Helminth Eggs with Short- and Medium-Chain Fatty Acids Alone and in Combinations at Naturally Occurring Concentrations. Master’s Thesis, Cornell University, Ithaca, NY, USA, 2014. [Google Scholar]
- Chubb, J.C. Acetic acid as a diluent and dehydrant in the preparation of whole, stained helminths. Biotech. Histochem. 1962, 37, 179–182. [Google Scholar] [CrossRef]
- Rojas-Oropeza, M.; Hernández-Uresti, A.S.; Ortega-Charleston, L.S.; Cabirol, N. Effect of volatile fatty acids in anaerobic conditions on viability of helminth ova (Ascaris suum) in sanitization of municipal sludge. Environ. Technol. 2016, 38, 2202–2208. [Google Scholar] [CrossRef] [PubMed]
- Dunkel, F.V. The relationship of insects to the deterioration of stored grain by fungi. Int. J. Food Microbiol. 1988, 7, 227–244. [Google Scholar] [CrossRef]
- Roberts, D.W.; Leger, R.J. Metarhizium spp., cosmopolitan insect-pathogenic fungi: Mycological aspects. Adv. Appl. Microbiol. 2004, 54, 1–70. [Google Scholar] [CrossRef]
- Knottenbelt, D.C. Skin disorders of the donkey and mule. Vet. Clin. North Am. Equine Pract. 2019, 35, 493–514. [Google Scholar] [CrossRef] [PubMed]
- Maurya, P.K.; Malik, D.S. Bioaccumulation of xenobiotics compound of pesticides in riverine system and its control technique: A critical review. J. Ind. Pollut. Control 2016, 32, 580–594. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).