1. Introduction
Nematodes are the most abundant and diverse group among meiofauna animals and play important roles as various trophic guilds. The estimated number of nematode species ranges from half a million to ten million, and only 1% to 5% of these species have been identified [
1]. Phylogenetic relationships among nematodes are still not well understood due to the scarcity of available molecular sequences deposited in GenBank. Linhomoeidae Filipjev, 1922, and Xyalidae Chitwood, 1951, are the two most complex families in the order Monhysterida. Genera among them are morphological similar, and genus relationships between
Metadesmolaimus Schuurmans Stekhoven, 1935 and
Daptonema Cobb, 1920, as well as between
Metadesmolaimus and
Theristus Bastian, 1865, have been a source of confusion for a long time [
2,
3,
4].
The genus
Desmolaimus was established in 1880 by de Man, based on the following morphological characteristics of the type species,
D. zeelandicus de Man, 1880: non-striated cuticle, an undistinguished head, circular amphids, a small, cup-shaped buccal cavity with cuticularized ridges, a pharynx with a posterior bulb, and a symmetrical, elongated female reproductive system [
5]. Kreis revised the genus characteristics based on head sensilla, amphidial fovea shape, cardia, and tail shape, and described
D. longicaudatus Kreis, 1929 [
6]. After Kreis,
D. zosterae Allgén, 1933,
D. calvus Gerlach, 1956,
D. bibulbosus Allgén, 1959,
D. conicaudatus Allgén, 1959,
D. greenpatchi Allgén, 1959,
D. macrocirculus Allgén, 1959, and
D. propinquus Allgén, 1959 were described. Schneider transferred
D. brachystoma (Hofmänner & Menzel, 1914) Micoletzky, 1925 and
D. thienemanni Micoletzky, 1922 to the genus
Hofmaenneria Gerlach & Meyl, 1957 [
7]. Gerlach discussed the position of
Desmolaimus within the family Linhomoeidae Filipjev, 1922 based on cephalic setae, cervical setae, amphidial fovea, and buccal cavity. He revised the genus’s character, mainly based on head sensilla and subcephalic setae. He described
D. brasiliensis Gerlach, 1963, synonymized
D. demani Schulz, 1932 and
D. fennicus (Schneider, 1926) Gerlach, 1953 as
D. zeelandicus, and considered
D. mirabilis Allgén, 1935,
D. tristis Allgén, 1935,
D. bibulbosus, and
D. greenpatchi as species inquirenda [
8]. After Gerlach,
D. bulbulus Lorenzen, 1969,
D. courti Leduc & Gwyther, 2008 and
D. minor Gagarin, 2019 were described. The genus relationship between
Desmolaimus and
Metadesmolaimus Schuurmans Stekhoven, 1935 was discussed by Leduc & Gwyther, and transverse cuticularized rings in the buccal cavity were considered a reliable distinguishing character from
Metadesmolaimus [
9]. Gagarin considered
D. conicaudatus,
D. macrocirculus, and
D. propinquus as species inquirenda because they were not fully described, especially description the buccal cavity structure absent, and the detailed illustrations not provided [
10]. As of now, eight species have been considered as valid.
The genus
Metadesmolaimus was first erected by Schuurmans Stekhoven in 1935 based on
M. labiosetosus (Schuurmans Stekhoven, 1935) with the genus character of labial sensilla in three circles, a circular amphidial fovea, a buccal cavity similar to
Sphaerolaimus Bastian, 1865 and
Desmolaimus, and a tail elongated and conical [
11]. But
M. labiosetosus was based on a single juvenile specimen and considered as species inquirenda. Later, Gerlach described
M. aversivulva and differentiated the genus from
Theristus mainly based on the buccal cavity structure [
12]. Lorenzen described
M. aduncus Lorenzen, 1971,
M. pandus Lorenzen, 1971,
M. heteroclitus Lorenzen, 1971,
M. varians Lorenzen, 1971 and transferred
Paramonhystera canicula (Wieser & Hopper, 1967),
Theristus hamatus (Gerlach 1956) to genus
Metadesmolaimus [
2]. Gerlach & Riemann transferred
Paramonhystera canicula to
Metadesmolaimus [
13]. Lorenzen systematically reviewed
Metadesmolaimus considering cuticle, cephalic setae, gonads, and tail shape. Lorenzen also redescribed and transferred
Daptonema gelana (Warwick & Platt, 1973) to
Metadesmolaimus based on the presence of two bristle-shaped structures [
14]. Later,
M. gaelicus Platt, 1983,
M. similis Tchesunov, 1990,
M. psammophilus Tchesunov, 1990,
M. communis Gagarin, 2013, and
M. elegans Gagarin, 2013 were described. Tchesunov & Miljutin discussed the relationship between
Metadesmolaimus and
Daptonema based on the structure of the copulatory apparatus and noted that morphological characters between these two genera are overlapping and loosely defined [
3]. Guo et al. recently described
M. zhanggi Guo Chen & Liu, 2016 and further discussed the genus characteristics, including the buccal cavity with an extended cylindrical anterior section, the existence of ventrolateral soft setiform structure between the labial and cephalic setae, and the brownish cuticle colour [
15]. As of now, 15 species have been considered as valid.
During our study of marine nematode diversity in the Rizhao coast, Yellow Sea, China, two new species, Desmolaimus magnus sp. nov. and Metadesmolaimus robustus sp. nov., are described, and sequences of SSU and the D2–D3 fragment of LSU rDNA are acquired. Phylogenetic analyses within the family Linhomoeidae and Xyalidae are conducted to clarify their classification at the generic level.
3. Results and Discussion
3.1. Taxonomy of Desmolaimus magnus sp. nov.
Order Monhysterida Filipjev, 1929
Family Linhomoeidae Filipjev, 1922
Genus Desmolaimus de Man, 1880
Desmolaimus magnus sp. nov.
LSIDurn:lsid:zoobank.org:act:D5D54F33-2679-49D5-A3A4-F38E499BA3ED
Type material: Five males and four females were measured and studied. Holotype: m#1 on slide 22LJW3–2–4; paratypes: m#2, m#3 on 22LJW3–2–4, m#4, m#5 on 22LJW3–2–8; f#1, f#2, f#3 on 22LJW3–2–3 and f#4 on 22LJW3–1–9.
Type locality and habitat: Rizhao coast, Shandong Province, China. 35°18′ N, 119°31′ E (LJW), 0–2 cm sediment depth, sandy sediment.
Etymology: The term “magnus” refers to the large body size.
Measurements: All measurement data can be found in
Table 1.
3.1.1. Description
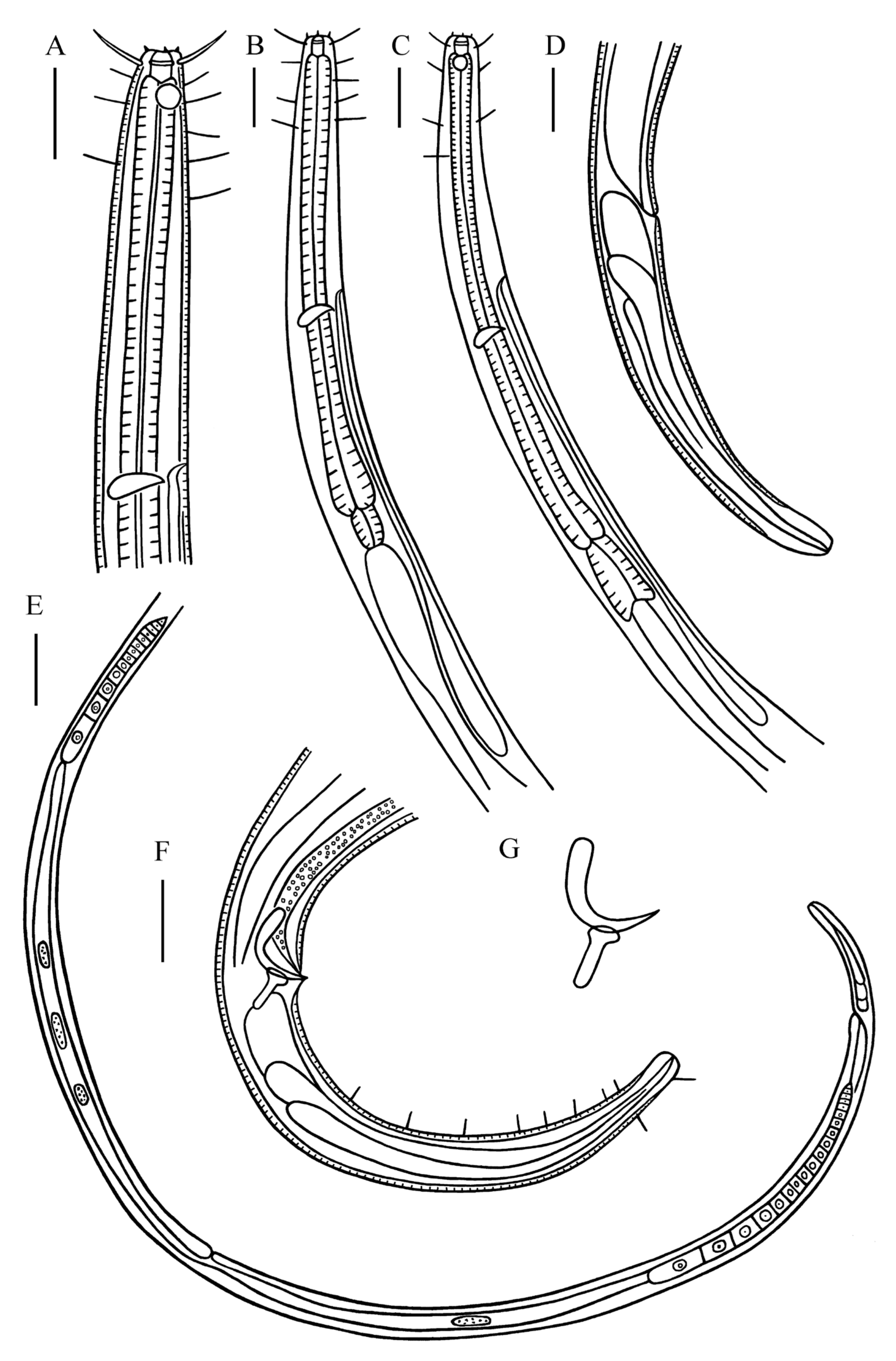
Males. Body cylindrical and relatively large, anterior, and posterior body ends narrowed. Cuticle finely striated. Six inner labial sensilla papilliform and six outer labial sensilla setiform, 3 μm in length (0.20–0.21 head diameter) in two separate circles. Four cephalic setae, 0.87–0.93 head diameter in length, situated at the level of cuticularized ring. Four subcephalic setae, 11 µm in length, situated at the level of amphidial fovea, somatic setae only present in anterior pharynx (11–12 μm in length) and at tail region (5 μm in length). Head blunt. Buccal cavity relatively large, 10–12 µm in depth with cuticularized walls. Pharyngostoma relatively extensive, with cuticularized transverse rings. Amphidial fovea circular (44.4–47.1% corresponding body diameter–c.b.d.), situated at junction of buccal cavity and pharynx, 0.60–0.79 head diameter from anterior end. Pharynx cylindrical, posterior region muscular and swollen but without forming basal bulb. Nerve ring slightly posterior to middle pharynx region (54.2–56.3% of pharynx length). Cardia muscular, 22–23 μm in length. Secretory–excretory system present, renette cell situated slightly posterior to cardia, excretory pore located posterior to nerve ring.
Reproductive system diorchic, testes opposed and outstretched. Spicules paired and strongly curved, 1.05–1.21 cloacal body diameters along arc, with slightly swollen proximal end and pointed distal end. Gubernaculum short with dorso–caudally directed apophysis. Precloacal supplements not observed. Tail elongated conical with a truncated tip, 6.34–7.31 cloacal body diameters in length. Tail with two longitudinal rows of subventral setae, 6–9 per row, 1–2 short caudal setae on dorsal side and two setae at terminal end (6 μm in length). Three caudal glands present.
Females. Similar to males in most characteristics. Reproductive system didelphic, with opposed and outstretched ovaries. Anterior ovary to the right and posterior ovary to the left of intestine. Vulva slightly at the posterior of the total body. Vagina short and surrounded by constrictor muscle. Tail elongated conical without caudal setae.
3.1.2. Differential Diagnosis and Discussion
Desmolaimus magnus sp. nov. is characterized by its large body size, finely striated cuticle, a large buccal cavity with cuticularized transverse rings, six outer labial sensilla setiform, and four long cephalic setae. It also features an amphidial fovea located at junction of the buccal cavity and pharynx. The pharynx lacks a bulb, and the spicules are curved with a slightly swollen proximal end. Additionally, there is a gubernaculum with a dorso–caudal apophysis, and the tail is elongated and conical with short caudal setae.
Desmolaimus magnus sp. nov. differs from other species of the genus
Desmolaimus in body length, the location of the amphidial fovea at the junction of the buccal cavity and pharynx, and the absence of a pharyngeal bulb.
D. magnus sp. nov. is similar to
D. brasiliensis,
D. courti, and
D. zosterae in spicules, gubernaculum, and tail shape. But it differs from
D. brasiliensis in amphidial fovea diameter (44.4–47.1% c.b.d. vs. 35% c.b.d.), spicules shape, and length (proximal end slightly widened, 39–41 μm vs. proximal end with ventral interruption, 20 μm), and gubernaculum apophysis shape (straight dorsal caudal apophysis vs. wave–shaped dorsal apophysis) [
8]; differs from
D. courti in subcephalic setae (4 vs. 8), higher de Man’s ratio a (114.2–157.1 vs. 67.0–81.9), buccal cavity shape (one cuticularized rings vs. two cuticularized rings), amphidial fovea width (44.4–47.1% c.b.d. vs. 31–35% c.b.d.), spicules shape and length (proximal end slightly widened, 39–41 μm vs. proximal end not widened, 22–27 μm), and tail length (6.34–7.31 cloacal body diameters vs. 4.50–5.10 cloacal body diameters) [
9]; differs from
D. zosterae in higher de Man’s ratio a (114.2–157.1 vs. 30.0–43.5), longer cephalic setae (11–13 μm vs. 7 μm), amphidial fovea width (44.4–47.1% c.b.d. vs. 28.5% c.b.d.), spicules shape (slightly swollen at the proximal end vs. proximal end widened in the form of button), and tail length (7.13–8.26 cloacal body diameters vs. 4.5 cloacal body diameters) [
20].
3.1.3. Molecular Phylogenetic Analysis
The ML topology based on SSU and LSU rDNA D2–D3 fragment sequences are in accordance with the BI topology, and only the BI trees are shown in
Figure 3 and
Figure 4.
Five genera,
Eleutherolaimus Filipjev, 1922,
Desmolaimus,
Linhomoeus Bastian, 1865,
Metalinhomoeus de Man, 1907 and
Terschellingia de Man, 1888 are included in the SSU analysis and only the genus
Terschellingia forms a well–supported monophyletic clade (100% posterior probability and 81% bootstrap value), constituting a sister clade to the other genera within Lihomoedae. This is in accordance with Leduc & Zhao [
21] probably for its minute or conical shaped buccal cavity in
Terschellingia distinguishing from cup–shaped buccal cavity of the other genera. Sequences of
Terschellingia longicaudata were not shown as monophyletic clade in accordance with Leduc & Zhao and Sahraen for the high intraspecific variation [
21,
22].
Metalinhomoeus sp. (MK626797.1) and
Terschellingia lutosa (MK626784.1) constitute a highly supported clade (100% posterior probability and 100% bootstrap value) and are identical to each other. These two sequences were submitted by the same author and should be retained for further examination.
Four species of Desmolaimus have been identified to the species level: D. brasiliensis, D. longicaudatus, D. magnus sp. nov., and D. zeelandicus. They are shown as a paraphyletic group. D. magnus sp. nov. forms a highly supported clade with Eleutherolaimus paraschneideri Leduc & Zhao, 2023 in the topology trees based on both SSU and LSU sequences (100% posterior probability and 97% bootstrap value, 100% posterior probability and 100% bootstrap value, respectively). These two species share similarities in amphidial fovea position, absence of a pharynx bulb, gubernaculum shape, and a conical tail with truncated end. However, they can be easily distinguished by the presence or absence of a cuticularized ring in the buccal cavity and the arrangement of cephalic setae.
3.2. Taxonomy of Metadesmolaimus robustus sp. nov.
Order Monhysterida Filipjev, 1929
Family Xyalidae Chitwood, 1951
Genus Metadesmolaimus Schuurmans Stekhoven, 1935
Metadesmolaimus robustus sp. nov.
LSIDurn:lsid:zoobank.org:act:901DC71D-C907-441A-A4B7-4E146D699F27
Type material: Four males and one female were measured and studied. Holotype: m#1 on slide 22SHT5–2–11; paratypes: m#2 on 22RZDT4–2–8, m#3 on 22RZDT4–1–9, m#4 on 22SHT5–1–28; f#1 on 22RZDT4–3–6.
Type locality and habitat: Rizhao cost, Shandong Province, China. 35°26′ N, 119°34′ E (SHT); 35°23′ N, 119°33′ E (RZDT), 0–2 cm sediment depth, sandy sediment.
Etymology: The term “robustus” refers to the strongly muscularized pharynx region.
Measurements: All measurement data can be found in
Table 2.
3.2.1. Description
Males. Body cylindrical and long. Cuticle coarsely striated in 2 μm, not conspicuously brown in color. Anterior region slightly setoff, lips high. Six inner labial sensilla setiform, 6–7 μm in length (0.26–0.29 head diameter). Six outer labial setae, 20–23 µm in length (0.91–0.95 head diameter) and four cephalic setae, 11–15 µm in length (0.48–0.60 head diameter) in a same circle. Two lateral setae present next to outer labial setae, 0.48–0.60 head diameter in length, form 6 + 12 arrangement. Ventrolateral setiform structure exists between labial and cephalic setae. Entire pharynx region with numerous somatic setae of various lengths, 15–60 μm in length. Buccal cavity relatively spacious with cuticularized walls, 22–27 µm in depth, 19 μm in width, divided into two parts by cuticularized rings. Amphidial fovea not observed. Pharynx cylindrical, strongly muscularized and divided into 16–20 parts (15–19 µm) without pharynx bulb. Nerve ring anterior to the pharynx region (27.5–33.8% of pharynx length). Cardia short. Excretory pore not observed.
Reproductive system with single outstretched anterior testis to right of intestine. Spicules paired and equal, straight with proximal end slightly swollen, distal portion widened and jointed with hooked distal end, 1.49–1.58 cloacal body diameters long. Gubernaculum brownish, comma shaped. Cloacal setae in one circle, 9–10 µm in length. Precloacal supplement not observed. Tail conico–cylindrical, 4.52–5.15 cloacal body diameters in length, conical part short (35.8–40.2% of tail length). Two terminal setae present, 15–20 µm in length. Caudal glands not observed.
Female. Similar to males in most characteristics. Reproductive system with single outstretched ovary to left of intestine. Vulva at posterior of total body. Vagina short. Three caudal glands in line.
3.2.2. Differential Diagnosis and Discussion
Metadesmolaimus robustus sp. nov. is characterized by its large body size, coarsely striated cuticle, spacious buccal cavity with cuticularized rings, six inner labial sensilla setiform, six outer labial setae, and four long cephalic setae. The pharynx is muscular and lacks a bulb. Spicules are straight with a slightly swollen proximal end and a jointed distal portion with a hooked distal end. The gubernaculum has a comma shape, and the tail is conico–cylindrical with a short conical part.
Metadesmolaimus robustus sp. nov. differs from other species of the genus
Metadesmolaimus by having jointed spicules and a comma–shaped gubernaculum.
M. robustus sp. nov. is similar to
M. communis,
M. hamatus, and
M. zhanggi in having straight spicules. However, it differs from
M. communis in body length (1471–2084 µm vs. 827–932 µm), higher de Man’s ratio a (46.9–49.2 vs. 22–28), longer inner labial sensilla (6–7 µm vs. 1.0–1.5 µm), longer outer labial sensilla (20–23 µm vs. 7–9 µm), longer spicules (39–52 µm vs. 27–29 µm), and a different tail shape (conico–cylindrical vs. elongated conical) [
23]. It differs from
M. hamatus in inner labial setae length (6–7 µm vs. papilliform), outer labial sensilla length (20–23 µm vs. 10–11 µm), spicules length (39–52 µm vs. 28 µm), and tail shape (conico–cylindrical vs. elongated conical) [
24]. It also differs from
M. zhanggi in body length (1471–2084 µm vs. 930–980 µm), the shape of inner and outer labial sensilla (unjointed vs. jointed), and spicules length (39–52 µm vs. 24–29 µm) [
15].
3.2.3. Molecular Phylogenetic Relationships and Analysis
The ML topology based on SSU and LSU rDNA D2–D3 fragment sequences closely aligns with the BI topology. As a result, only the BI trees are presented in
Figure 7 and
Figure 8. In the SSU analysis, only sequences identified to the species level are retained, with the exception of the genus
Metadesmolaimus, which includes
M. robustus sp. nov. This selection is made due to the high morphological similarity among genera.
Eight genera,
Cobbia de Man, 1907,
Daptonema,
Metadesmolaimus,
Paramonohystera Steiner, 1916,
Promonhystera Wieser, 1956,
Steineria Micoletzky, 1922,
Theristus, and
Zygonemella Cobb, 1920 are included in the SSU analysis. Notably,
Steineria vietnamica Gagarin, 2013 (MG670034.1) stands apart from the main Xyalidae clade. This distinction can be attributed to the unique cuticle shape of
S. vietnamica, characterized by cuticle annules in resolvable dots, setting it apart from other Xyalid species [
25]. In the topology tree,
S. vietnamica does not cluster with
Steineria sterreri, and its precise position may require further discussion, especially with the availability of more molecular sequences from the other four
Steineria species exhibiting the same cuticle feature.
Genera
Daptonema and
Theristus are the most complex genera within the family Xyalidae, with minor morphological differences that primarily revolve around tail form and the terminal setae at the tail tip [
4]. In the SSU analysis, ten species of
Daptonema have been identified to the species level and are shown as paraphyletic groups. Notably,
Daptonema amphorum Leduc, 2015 (MW175369.1) and
D. normandicum split from the main clade of the other eight
Daptonema species (99% posterior probability and 83% bootstrap value, 86% posterior probability, respectively) in accordance with Leduc [
26]. Meanwhile,
Daptonema setosum (Bütschli, 1874) Lorenzen, 1977 and
D. hirsutum (Vitiello, 1967) Lorenzen, 1977 show a close relationship in SSU molecular analyses. Neres et al. considered
D. setosum as a junior synonym of
D. hirsutum based on molecular and morphological analyses, this is also supported in our SSU phylogenetic tree [
27]. In the LSU phylogenetic tree,
Daptonema sequences also form a well–supported clade (95% posterior probability and 85% bootstrap value). In the SSU topology tree, six species of genus
Theristus form a moderately supported monophyletic group (100% posterior probability and 42% bootstrap value), although this is not supported in the LSU tree. The SSU topology tree for
Daptonema and
Theristus largely aligns with Leduc [
26], but it depicts
Theristus as a monophylectic clade within the family Xyalidae. Conversely, the LSU topology tree indicates that the genus
Daptonema forms a monophylectic group within the family Xyalidae.
In the SSU analysis, five sequences of the genus Metadesmolaimus are included with only M. aduncus and M. robustus sp. nov. identified to the species level. These two species are depicted as paraphyletic clades. In the SSU topology tree, M. robustus sp. nov. is also shown as a sister clade to genera Daptonema, Zygonemella, Cobbia and Paramonohystera. In the LSU topology tree, M. robustus sp. nov. diverges early from the main Xyalidae clade, confirming its status as a new species.
The genus
Zygonemella exhibits morphological similarities to the genus
Daptonema, with
Z. striata Cobb, 1920 being the sole species within
Zygonemella.
Z. striata clusters with
Daptonema oxycerca, forming a moderately supported clade (98% posterior probability and 65% bootstrap value). This lends support to the conclusion that
Z. striata is synonymous with the genus
Daptonema [
28].
4. Conclusions
Desmolaimus magnus sp. nov. can be distinguished from other species in the genus Desmolaimus by its larger body size, possession of six outer labial sensilla setiform, and four long cephalic setae, an amphidial fovea located at the junction of the buccal cavity and pharynx, a pharynx without a bulb, curved spicules with a slightly swollen proximal end, a gubernaculum with a dorso–caudal apophysis, and an elongated conical tail with short caudal setae. Phylogenetic analysis confirms D. magnus sp. nov. as a new species, while it also reveals that the genus Desmolaimus forms a paraphyletic group and the genus Terschellingia constitutes a monophyletic group. With the description of D. magnus sp. nov., a total of nine species of Desmolaimus have been identified.
Metadesmolaimus robustus sp. nov. sets itself apart from other species in the genus Metadesmolaimus with its large body size, possession of six inner labial sensilla setiform, six long outer labial setae, four long cephalic setae, a muscularized pharynx without a bulb, straight and jointed spicules, brownish, and a comma–shaped gubernaculum. Phylogenetic analysis confirms the status of M. robustus sp. nov. as a new species. It also reveals that the genus Metadesmolaimus forms a paraphyletic group, while the genus Theristus is a monophyletic group based on SSU sequence data, and the genus Daptonema is a monophyletic group based on LSU sequence data. Furthermore, the analysis supports the conclusion that Z. striata is synonymous with the genus Daptonema. With the description of M. robustus sp. nov., a total of 16 species of Metadesmolaimus have been identified.