Waldsteinia within Geum s.l. (Rosaceae): Main Aspects of Phylogeny and Speciation History
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material Collection
2.2. Counting Chromosome Numbers
2.3. DNA Isolation and PCR
2.4. Cloning and Sequencing
2.5. Sequence Alignment and Phylogenetic Analysis
3. Results
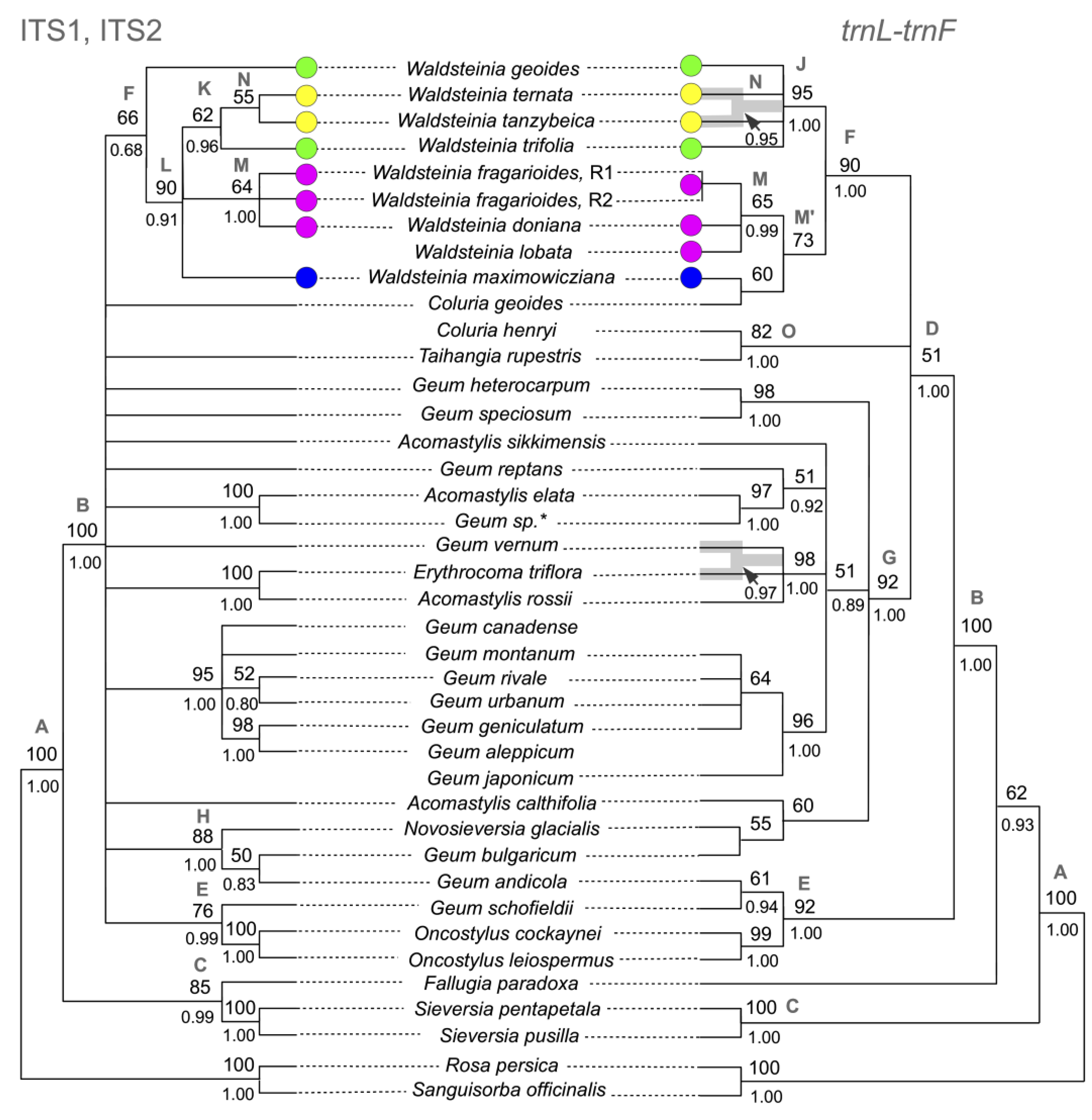
3.1. Phylogenetic Analysis Based on Nuclear DNA
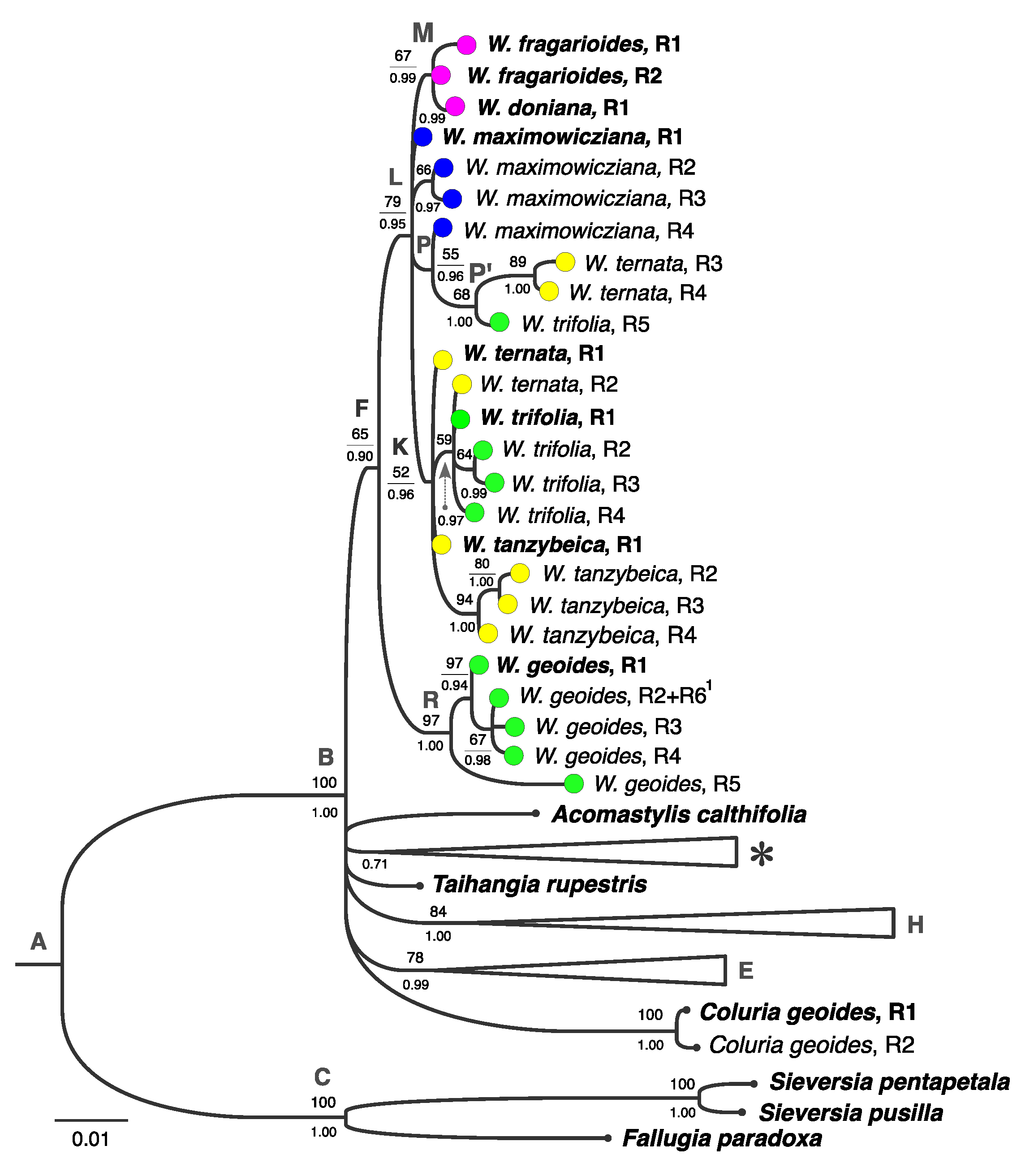
3.2. Phylogenetic Analysis Based on Plastid DNA
3.3. Combined Phylogenetic Analysis
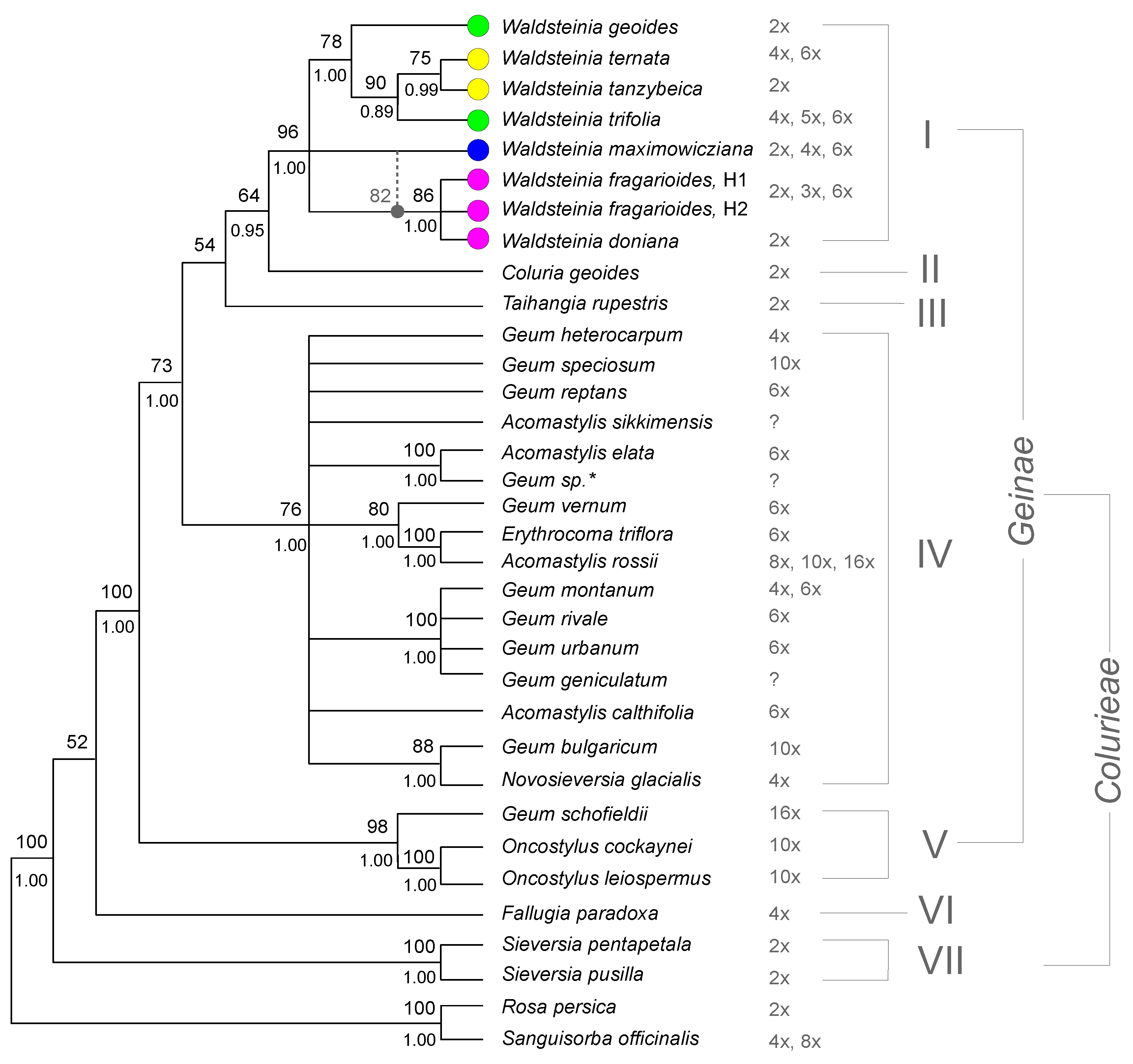
3.4. Chromosome Numbers
3.5. Geographical Patterns of Plastotype Distribution
4. Discussion
4.1. The Updated Phylogenetic Reconstruction of Colurieae
4.2. Relationships between Waldsteinia and Coluria
4.3. Phylogenetic Structure of Waldsteinia
4.4. Possible Scenarios of Waldsteinia’s History
4.5. Geum Sensu Lato vs. Geum Sensu Stricto
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gajewski, W. A Cytogenetic Study on the Genus Geum L. Monogr. Bot. 1957, 4, 3–420. [Google Scholar] [CrossRef]
- Smedmark, J.E.E. Recircumscription of Geum L. (Colurieae: Rosaceae). Bot. Jahrbücher Für Syst. Pflanzengesch. Pflanzengeogr. 2006, 126, 409–417. [Google Scholar] [CrossRef]
- Gajewski, W. Evolution in the Genus Geum. Evolution 1959, 13, 378–388. [Google Scholar] [CrossRef]
- Teppner, H. Waldsteinia ternata (Rosaceae) Und Ihre Vorkommen in Den Südöstlichen Alpen. Phyton Austria 1974, 16, 281–299. [Google Scholar]
- Yakubov, V.V. Waldsteinia Willd. In Vascular Plants of the Soviet Far East, Vol. 8; Kharkevich, S.S., Ed.; Nauka: St.-Petersburg, Russia, 1996; pp. 209–210. (In Russian) [Google Scholar]
- Li, C.; Ikeda, H.; Ohba, H. Waldsteinia Willd. In Flora of China, Vol. 9; Wu, Z.-Y., Raven, P.H., Deyuan, H., Eds.; Science Press and Missouri Botanical Garden Press: Beijing, China; St. Louis, MI, USA, 2003; p. 291. [Google Scholar]
- Hill, S.R. Conservation Assessment for Barren Strawberry (Waldsteinia fragarioides (Michx.) Tratt. Ssp. Fragarioides); Illinois Natural History Survey: Champaign, IL, USA, 2003. [Google Scholar]
- Probatova, N.S. Waldsteinia Willd. In Flora of the Russian Far East. Addenda and Corrigenda to “Vascular Plants of the Soviet Far East”, Vol. 1–8 (1985–1996); Kozhevnikova, A.E., Probatova, N.S., Eds.; Dalnauka: Vladivostok, Russia, 2006; p. 161. (In Russian) [Google Scholar]
- Chepinoga, V.V.; Mishina, A.V.; Protopopova, M.V.; Pavlichenko, V.V.; Bystrov, S.O.; Vilor, M.A. New Data on Distribution of Several Nemoral Relict Plant Species on the Foothills of the Khamar-Daban Ridge (Southern Baikal). Bot. Zhurnal 2015, 100, 478–489. (In Russian) [Google Scholar]
- Chepinoga, V.V.; Stepanov, N.V.; Protopopova, M.V.; Pavlichenko, V.V. The Distribution of Waldsteinia ternata (Rosaceae) in the West Sayan Mts. Bot. Zhurnal 2019, 104, 1203–1210. (In Russian) [Google Scholar] [CrossRef]
- Hassler, M. Synonymic Checklists of the Vascular Plants of the World. In Catalogue of Life Checklist (Version 15.0, Mar 2023). 2023. Available online: https://www.catalogueoflife.org/data/dataset/1141 (accessed on 17 March 2023).
- Yang, J.-C.; Hwang, H.-S.; Lee, H.-J.; Jung, S.-Y.; Ji, S.-J.; Oh, S.-H.; Lee, Y.-M. Distribution of Vascular Plants along the Altitudinal Gradient of Gyebangsan (Mt.) in Korea. J. Asia-Pac. Biodivers. 2014, 7, e40–e71. [Google Scholar] [CrossRef]
- Teppner, H. Zur Kenntnis Der Gattung Waldsteinia. Dissertation, Universität Graz, Graz, Austria, 1968. [Google Scholar]
- Gajewski, W. Waldsteinia Willd. In Flora Europaea, Vol. 2, Rosaceae to Umbelliferae; Tutin, T.G., Heywood, V.H., Burges, N.A., Moore, D.M., Valentine, D.H., Walters, S.M., Webb, D.A., Eds.; Cambridge University Press: Cambridge, UK, 1968; p. 36. [Google Scholar]
- Kamelin, R.V. Waldsteinia Willd. In Flora Europae Orientalis, Vol. 10, Magnoliophyta (=Angiospermae), Magnoliopsida (=Dicotyledones); Tzvelev, N.N., Ed.; Academia Chemico-Pharmaceutica Petropolitana: St. Petersburg, Russia, 2001; p. 267. (In Russian) [Google Scholar]
- Stepanov, N.V. A New Species of the Genus Waldsteinia (Rosaceae) from the West Sayan. Bot. Zhurnal 1994, 79, 109–114. (In Russian) [Google Scholar]
- Teppner, H.; Schuehly, W.; Weakley, A.S. The Chromosome Numbers of Waldsteinia (Rosaceae-Colurieae) in North America. Phyton 2009, 48, 225–238. [Google Scholar]
- Weakley, A.S.; Gandhi, K.N. Recognition of Three Taxa of Eastern North American “Waldsteinia” and Their Appropriate Names When Incroporated into Geum (Colurieae: Rosaceae). J. Bot. Res. Inst. Tex. 2008, 2, 415–418. [Google Scholar]
- Phipps, J.B. Waldsteinia. In Flora of North America North of Mexico, Vol. 9, Magnoliophyta: Picramniaceae to Rosaceae; Flora of North America Editorial Committee, Ed.; Oxford University Press: Oxford, UK; New York, NY, USA, 2014; pp. 71–72. ISBN 9780195340297. [Google Scholar]
- Polozii, A.V.; Krapivkina, E.D. Relikty Tretichnyh Shirokolistvennyh Lesov vo Flore Sibiri [Relics of Tertiary Deciduous Forests in the Flora of Siberia]; Tomsk University Press: Tomsk, USSR, 1985. (In Russian) [Google Scholar]
- Belov, A.V.; Bezrukova, E.V.; Sokolova, L.P.; Abzayeva, A.A.; Letunova, P.P.; Fisher, E.E.; Orlova, L.A. Vegetation of the Baikal Region as an Indicator of Global and Regional Changes in Natural Conditions of North Asia in the Late Cainozoic. Geogr. Nat. Resour. 2006, 6, 5–18. (In Russian) [Google Scholar]
- Košanin, N. Waldsteinia ternata (Stepn.) Fritsch im Bestande von Picea omorica. Öesterreichische Bot. Z. 1921, 70, 299. [Google Scholar] [CrossRef]
- Wood, C.E. Morphology and Phytogeography: The Classical Approach to the Study of Disjunctions. Ann. Mo. Bot. Gard. 1972, 59, 107–124. [Google Scholar] [CrossRef]
- Baikov, K.S. Conspectus Florae Rossiae Asiaticae: Plantae Vasculares; Publishing house of the Siberian Branch of the Russian Academy of Sciences: Novosibirsk, Russia, 2012; ISBN 9785769212130. (In Russian) [Google Scholar]
- Steyermark, J.A. Flora of Missouri; The Iowa State University Press: Ames, IA, USA, 1963. [Google Scholar]
- Potter, D.; Eriksson, T.; Evans, R.C.; Oh, S.; Smedmark, J.E.E.; Morgan, D.R.; Kerr, M.; Robertson, K.R.; Arsenault, M.; Dickinson, T.A.; et al. Phylogeny and Classification of Rosaceae. Plant Syst. Evol. 2007, 266, 5–43. [Google Scholar] [CrossRef]
- Smedmark, J.E.E.; Eriksson, T.; Evans, R.C.; Campbell, C.S. Ancient Allopolyploid Speciation in Geinae (Rosaceae): Evidence from Nuclear Granule-Bound Starch Synthase (GBSSI) Gene Sequences. Syst. Biol. 2003, 52, 374–385. [Google Scholar] [CrossRef]
- Smedmark, J.E.E.; Eriksson, T.; Bremer, B. Allopolyploid Evolution in Geinae (Colurieae: Rosaceae)—Building Reticulate Species Trees from Bifurcating Gene Trees. Org. Divers. Evol. 2005, 5, 275–283. [Google Scholar] [CrossRef]
- Christenhusz, M.J.M.; Fay, M.F.; Chase, M.W. Plants of the World: An Illustrated Encyclopedia of Vascular Plants; Royal Botanic Gardens: Kew, Richmond, UK; University of Chicago Press: Chicago, MI, USA, 2017; p. 816. ISBN 9781842466346. [Google Scholar]
- Smedmark, J.E.E.; Eriksson, T. Phylogenetic Relationships of Geum (Rosaceae) and Relatives Inferred from the NrITS and TrnL-TrnF Regions. Syst. Bot. 2002, 27, 303–317. [Google Scholar]
- Navashin, M.S. Metodika Citologicheskogo Issledovaniya Dlya Selektsionnykh Tselei [the Technique of Cytological Studies in Breeding]; OGIZ-Selhozgiz: Moscow, USSR, 1936; Volume 85. (In Russian) [Google Scholar]
- Abramova, L.I.; Orlova., I.N. Citologicheskaya I Citoembriologicheskaya Tekhnika (Dlya Issledovaniya Kulturnykh Rasteniy): Metodicheskiye Ukazaniya [Cytoembryological and Cytological Techniques (for Cultivated Plants): The Guidelines]; VIR: Leningrad, Russia, 1982. (In Russian) [Google Scholar]
- Doyle, J.J.; Doyle, J.L. A Rapid DNA Isolation Procedure for Small Quantities of Fresh Leaf Tissue. Phytochem. Bull. 1987, 19, 11–15. [Google Scholar]
- Protopopova, M.V.; Pavlichenko, V.V. Eranthis Salisb. (Ranunculaceae) in South Siberia: Insights into Phylogeography and Taxonomy. Diversity 2022, 14, 779. [Google Scholar] [CrossRef]
- Utelli, A.; Roy, B.; Baltisberger, M. Molecular and Morphological Analyses of European Aconitum Species (Ranunculaceae). Plant Syst. Evol. 2000, 224, 195–212. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Lahr, D.J.G.; Katz, L.A. Reducing the Impact of PCR-Mediated Recombination in Molecular Evolution and Environmental Studies Using a New-Generation High-Fidelity DNA Polymerase. Biotechniques 2009, 47, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Taberlet, P.; Gielly, L.; Pautou, G.; Bouvet, J. Universal Primers for Amplification of Three Non-Coding Regions of Chloroplast DNA. Plant Mol. Biol. 1991, 17, 1105–1109. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-D.; Jin, J.-J.; Chen, S.-Y.; Chase, M.W.; Soltis, D.E.; Li, H.-T.; Yang, J.-B.; Li, D.-Z.; Yi, T.-S. Diversification of Rosaceae since the Late Cretaceous Based on Plastid Phylogenomics. New Phytol. 2017, 214, 1355–1367. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-X.; Zhang, M.-L.; Sanderson, S.C. Spatial Genetic Structure of Forest and Xerophytic Plant Species in Arid Eastern Central Asia: Insights from Comparative Phylogeography and Ecological Niche Modelling. Biol. J. Linn. Soc. 2017, 120, 612–625. [Google Scholar] [CrossRef]
- Kress, W.J.; Wurdack, K.J.; Zimmer, E.A.; Weigt, L.A.; Janzen, D.H. Use of DNA Barcodes to Identify Flowering Plants. Proc. Natl. Acad. Sci. USA 2005, 102, 8369–8374. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Pak, J.-H. Phylogeny of Korean Rubus (Rosaceae) Based on Its (NrDNA) and TrnL/F Intergenic Region (CpDNA). J. Plant Biol. 2006, 49, 44–54. [Google Scholar] [CrossRef]
- Kerr, M.S. A Phylogenetic and Biogeographic Analysis of Sanguisorbeae (Rosaceae), with Emphasis on the Pleistocene Radiation of the High Andean Genus Polylepis. Ph.D. Dissertation, University of Maryland, College Park, MD, USA, 2004. [Google Scholar]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian Phylogenetic Inference and Model Choice across a Large Model Space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Hasegawa, M.; Kishino, H.; Yano, T. Dating of the Human-Ape Splitting by a Molecular Clock of Mitochondrial DNA. J. Mol. Evol. 1985, 22, 160–174. [Google Scholar] [CrossRef]
- Felsenstein, J. Evolutionary Trees from DNA Sequences: A Maximum Likelihood Approach. J. Mol. Evol. 1981, 17, 368–376. [Google Scholar] [CrossRef]
- Jukes, T.H.; Cantor, C.R. Evolution of Protein Molecules; Academic Press: New York, NY, USA, 1969; Volume 3, pp. 21–132. [Google Scholar]
- Rambaut, A. FigTree: Tree Figure Drawing Tool, Version 1.4.3. Available online: http://tree.bio.ed.ac.uk/software/figtree/ (accessed on 4 October 2016).
- Kimura, M. A Simple Method for Estimating Evolutionary Rates of Base Substitutions through Comparative Studies of Nucleotide Sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K. Estimation of the Number of Nucleotide Substitutions When There Are Strong Transition-Transversion and G+C-Content Biases. Mol. Biol. Evol. 1992, 9, 678–687. [Google Scholar] [CrossRef]
- Leigh, J.W.; Bryant, D. PopART: Full-Feature Software for Haplotype Network Construction. Methods Ecol. Evol. 2015, 6, 1110–1116. [Google Scholar] [CrossRef]
- Chepinoga, V.V.; Gnutikov, A.A.; Lubogoschinsky, P.I. Chromosome Numbers of Some Vascular Plant Species from the South of Baikal Siberia. Bot. Pac. 2012, 1, 127–132. [Google Scholar] [CrossRef]
- Teppner, H. Erfahrungen Mit Ex-Situ-Erhaltung Im Botanishen Garten Des Institutes Für Botanik Universität Graz. Fritschiana Graz 2003, 39, 1–22. [Google Scholar]
- Stepanov, N.V. Chromosome Numbers in Some Nemoral Species of the West Sayan (Krasnoyarsk Region). Bot. Zhurnal 1994, 79, 125–128. (In Russian) [Google Scholar]
- Probatova, N.S.; Rudyka, E.G.; Shatokhina, A.V.; Barkalov, V.Y.; Krjukova, M.V.; Tsyrenova, D.Y. Chromosome Numbers of Some Plant Species of the Primkorsky Territory and the Amur River Basin. Bot. Zhurnal 2006, 91, 785–804. (In Russian) [Google Scholar]
- Probatova, N.S.; Kozhevnikova, Z.V.; Rudyka, E.G.; Shatokhina, A.V.; Kozhevnikov, A.E.; Barkalov, V.Y.; Seledets, V.P. Chromosome Numbers for the Flora of the Russian Far East and the East Siberia. Bot. Zhurnal 2009, 94, 764–780. (In Russian) [Google Scholar]
- Probatova, N.S.; Kozhevnikova, Z.V.; Rudyka, E.G.; Kozhevnikov, A.E.; Nechaev, V.A. Chromosome Numbers of Some Vascular Plants from the Far East of Russia. Bot. Zhurnal 2010, 95, 1008–1020. (In Russian) [Google Scholar]
- Probatova, N.S.; Seledets, V.P.; Rudyka, E.G.; Kudryavtseva, E.P. Chromosome Numbers of Some Vascular Plant Species from the Islands of Peter the Great Bay (Sea of Japan). Bot. Pac. 2013, 2, 73–78. [Google Scholar] [CrossRef]
- Sokolovskaya, A.P.; Probatova, N.S.; Rudyka, E.G. Chromosome Numbers in the Species of the Families Asteraceae, Poaceae, Rosaceae from the Primorye Region, Kamchatka and Sakhalin. Bot. Zhurnal SSSR 1985, 70, 126–128. (In Russian) [Google Scholar]
- Probatova, N.S.; Barkalov, V.Y.; Nechaev, V.A. Further Chromosome Studies on Vascular Plants in Primorsky Krai. Uchenye Zap. Zabaikalskogo Gos. Univ. Sch. Notes Transbaikal State Univ. 2016, 11, 27–37. (In Russian) [Google Scholar]
- Probatova, N.S.; Andriyanova, E.A.; Chernyagina, O.A.; Konotop, N.K.; Kozhevnikova, Z.V.; Krivenko, D.A.; Vinogradova, J.S. Botanica Pacifica Plant Chromosome Data 2. Bot. Pac. 2022, 11, 163–172. [Google Scholar] [CrossRef]
- Probatova, N.S.; Barkalov, V.Y.; Rudyka, E.G.; Pavlova, N.S. Further Chromosome Studies on Vascular Plant Species from Sakhalin, Moneran and Kurile Islands. Biodivers. Biogeogr. Kuril Isl. Sakhalin 2006, 2, 93–110. [Google Scholar]
- Nishikawa, T. Chromosome Counts of Flowedng Plants of Hokkaido (22). J. Hokkaido Univ. Educ. Nat. Sci. 2003, 53, 27–37. [Google Scholar]
- Májovksý, J. Index of Chromosome Numbers of Slovakian Flora (Part 6). Acta Fac. Rerum Nat. Univ. Comen. Bot. 1978, 26, 1–42. [Google Scholar]
- Májovský, J.; Murín, A.; Feráková, V.; Hindáková, M.; Schwarzová, T.; Uhríková, A.; Váchová, M.; Záborský, J. Karyotaxonomický Prehľad Flóry Slovenska; Veda: Bratislava, Czechoslovakia, 1987. [Google Scholar]
- Marhold, K.; Martonfi, P.; Mered’a jnr, P.; Mraz, P. Chromosome Number Survey of the Ferns and Flowering Plants of Slovakia; Veda: Bratislava, Slovakia, 2007; ISBN 9788022409803. [Google Scholar]
- Maurer, W.; Pagitz, W.; Teppner, H. Ein Weiterer Fund von Waldsteinia ternata (Rosaceae) in Kärnten. Carinth. II 1974, 164, 245–247. [Google Scholar]
- Lovka, M.; Susnik, F. IOPB Chromosome Numbers Reports. XL. Taxon 1973, 22, 285–289. [Google Scholar] [CrossRef]
- Pólya, L. Magyarországi Növényfajok Kromoszómaszámai I Chromosome Numbers of Some Hungarian Plants. Acta Geobot. Hung. 1949, 6, 124–137. [Google Scholar]
- Reese, G. Ergänzende Mitteilungen Über Die Chromosomenzahlen Mitteleuropäischer Gefäßpflanzen. I. Ber. Der Dtsch. Bot. Ges. 1952, 64, 240–255. [Google Scholar]
- Pagitz, W. Zur Karylogie von Coluria und Waldsteinia. Diploma Thesis, Universität Graz, Graz, Austria, 1974. [Google Scholar]
- Czapik, R. Apomictic Embryo Sacs in Diploid Waldsteinia geoides Willd. (Rosaceae). Acta Biol. Crac. Ser. Bot. 1985, 27, 29–37. [Google Scholar]
- Rice, A.; Glick, L.; Abadi, S.; Einhorn, M.; Kopelman, N.M.; Salman-Minkov, A.; Mayzel, J.; Chay, O.; Mayrose, I. The Chromosome Counts Database (CCDB)—A Community Resource of Plant Chromosome Numbers. New Phytol. 2015, 206, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, G.A.; Baum, B.R. IOPB Chromosome Number Reports XXVIII. Taxon 1970, 19, 609. [Google Scholar] [CrossRef]
- Baillon, H. Histoire Des Plantes; Librairie Hachette: Paris, France, 1867. [Google Scholar]
- Yuzepchuk, S.V.; Coluria, R. BR. In Flora of the USSR, Vol. 10; Komarov, V.L., Ed.; Academy of Sciences of the USSR: Leningrad, USSR, 1941; pp. 241–242. (In Russian) [Google Scholar]
- Yuzepchuk, S.V. Waldsteinia Willd. In Flora of USSR, Vol. 10; Komarov, V.L., Ed.; Academy of Sciences of the USSR: Leningrad, USSR, 1941; pp. 239–241. (In Russian) [Google Scholar]
- Vydryna, S.N.; Kurbatskii, V.I.; Polozhij, A.V. Flora Sibibiae. Rosaceae; Nauka: Novosibirsk, USSR, 1988; ISBN 5-02-028878-0. (In Russian) [Google Scholar]
- Li, C.; Ikeda, H.; Ohba, H.; Coluria, R. Brown. In Flora of China, Vol. 9; Wu, Z.-Y., Raven, P.H., Deyuan, H., Eds.; Science Press and Missouri Botanical Garden Press: Beijing, China; St. Louis, MI, USA, 2003; pp. 289–290. [Google Scholar]
- Protopopova, M.V.; Pavlichenko, V.V.; Stepanov, N.V. Phylogeographical Patterns Based on trnH-psbA Plastid DNA Shed Light on Evolution within Waldsteinia (Rosaceae). IOP Conf. Ser. Earth Environ. Sci. 2021, 908, 012027. [Google Scholar] [CrossRef]
- Nishimoto, Y.; Ohnishi, O.; Hasegawa, M. Topological Incongruence between Nuclear and Chloroplast DNA Trees Suggesting Hybridization in the Urophyllum Group of the Genus Fagopyrum (Polygonaceae). Genes Genet. Syst. 2003, 78, 139–153. [Google Scholar] [CrossRef]
- Barber, J.; Finch, C.; Francisco-Ortega, J.; Santos-Guerra, A.; Jansen, R. Hybridization in Macaronesian Sideritis (Lamiaceae): Evidence from Incongruence of Multiple Independent Nuclear and Chloroplast Sequence Datasets. Taxon 2007, 56, 74–88. [Google Scholar]
- Campbell, C.S.; Evans, R.C.; Morgan, D.R.; Dickinson, T.A.; Arsenault, M.P. Phylogeny of Subtribe Pyrinae (Formerly the Maloideae, Rosaceae): Limited Resolution of a Complex Evolutionary History. Plant Syst. Evol. 2007, 266, 119–145. [Google Scholar] [CrossRef]
- Friar, E.A.; Prince, L.M.; Cruse-Sanders, J.M.; McGlaughlin, M.E.; Butterworth, C.A.; Baldwin, B.G. Hybrid Origin and Genomic Mosaicism of Dubautia scabra (Hawaiian Silversword Alliance; Asteraceae, Madiinae). Syst. Bot. 2008, 33, 589–597. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Q.; Chen, T.; Tang, H.; Liu, L.; Wang, X. Phylogenetic Insights into Chinese Rubus (Rosaceae) from Multiple Chloroplast and Nuclear DNAs. Front. Plant Sci. 2016, 29, 968. [Google Scholar] [CrossRef]
- Hughes, M.; Peng, C.I.; Lin, C.W.; Rubite, R.R.; Blanc, P.; Chung, K.F. Chloroplast and Nuclear DNA Exchanges among Begonia Sect. Baryandra Species (Begoniaceae) from Palawan Island, Philippines, and Descriptions of Five New Species. PLoS ONE 2018, 13, e0194877. [Google Scholar] [CrossRef]
- Barke, B.H.; Daubert, M.; Hörandl, E. Establishment of Apomixis in Diploid F2 Hybrids and Inheritance of Apospory from F1 to F2 Hybrids of the Ranunculus auricomus Complex. Front. Plant Sci. 2018, 9, 1111. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.L. Natural Hybridization and Evolution; Oxford University Press: New York, NY, USA, 1997; ISBN 9780195099751. [Google Scholar]
- Feliner, G.N.; Álvarez, I.; Fuertes-Aguilar, J.; Heuertz, M.; Marques, I.; Moharrek, F.; Piñeiro, R.; Riina, R.; Rosselló, J.A.; Soltis, P.S.; et al. Is Homoploid Hybrid Speciation That Rare? An Empiricist’s View. Heredity 2017, 118, 513–516. [Google Scholar] [CrossRef] [PubMed]
- Goulet, B.E.; Roda, F.; Hopkins, R. Hybridization in Plants: Old Ideas, New Techniques. Plant Physiol. 2017, 173, 65–78. [Google Scholar] [CrossRef]
- Nogler, G.A. Gametophytic Apomixis. In Embryology of Angiosperms; Johri, B.M., Ed.; Springer: Berlin/Heidelberg, Germany, 1984; pp. 475–518. [Google Scholar]
- Grossniklaus, U.; Nogler, G.A.; van Dijk, P.J. How to Avoid Sex: The Genetic Control of Gametophytic Apomixis. Plant Cell 2001, 13, 1491–1498. [Google Scholar] [CrossRef] [PubMed]
- Schranz, M.E.; Dobes, C.; Koch, M.A.; Mitchell-Olds, T. Sexual Reproduction, Hybridization, Apomixis, and Polyploidization in the Genus Boechera (Brassicaceae). Am. J. Bot. 2005, 92, 1797–1810. [Google Scholar] [CrossRef] [PubMed]
- Zarrei, M.; Stefanović, S.; Dickinson, T.A. Reticulate Evolution in North American Black-Fruited Hawthorns (Crataegus Section Douglasia; Rosaceae): Evidence from Nuclear ITS2 and Plastid Sequences. Ann. Bot. 2014, 114, 253–269. [Google Scholar] [CrossRef]
- Xu, B.; Zeng, X.M.; Gao, X.F.; Jin, D.P.; Zhang, L.B. ITS Non-Concerted Evolution and Rampant Hybridization in the Legume Genus Lespedeza (Fabaceae). Sci. Rep. 2017, 7, 40057. [Google Scholar] [CrossRef] [PubMed]
- Hegarty, M.J.; Hiscock, S.J. Hybrid Speciation in Plants: New Insights from Molecular Studies. New Phytol. 2005, 165, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Probatova, N.S. Validation of the Combination Waldsteinia maximowicziana (Teppner) Prob. (Rosaceae). Novit. Syst. Plant. Vasc. 2015, 46, 237. [Google Scholar]
- Tiffney, B.H. Perspectives on the Origin of the Floristic Similarity between Eastern Asia and Eastern North America. J. Arnold Arbor. 1985, 66, 73–94. [Google Scholar] [CrossRef]
- Wolfe, J.A. An Analysis of Neogene Climates in Beringia. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1994, 108, 207–216. [Google Scholar] [CrossRef]
- Krestov, P.V.; Barkalov, V.Y.; Omelko, A.M.; Yakubov, V.V.; Nakamura, Y.; Sato, K. Relic Vegetation Complexes in the Modern Refugia of Northeast Asia. Komar. Chtenia V. L. Komar. Meml. Lect. 2009, 56, 5–63. (In Russian) [Google Scholar]
- Wen, J. Evolution of Eastern Asian and Eastern North American Disjunct Distributions in Flowering Plants. Annu. Rev. Ecol. Syst. 1999, 30, 421–455. [Google Scholar] [CrossRef]
- Tiffney, B.H.; Manchester, S.R. The Use of Geological and Paleontological Evidence in Evaluating Plant Phylogeographic Hypotheses in the Northern Hemisphere Tertiary. Int. J. Plant Sci. 2001, 162, S3–S17. [Google Scholar] [CrossRef]
- Xiang, Q.Y.; Soltis, D.E. Dispersal-Vicariance Analyses of Intercontinental Disjuncts: Historical Biogeographical Implications for Angiosperms in the Northern Hemisphere. Int. J. Plant Sci. 2001, 162, S29–S39. [Google Scholar] [CrossRef]
- Donoghue, M.J.; Bell, C.D.; Li, J. Phylogenetic Patterns in Northern Hemisphere Plant Geography. Int. J. Plant Sci. 2001, 162, 41–52. [Google Scholar] [CrossRef]
- Yi, T.-S.; Jin, G.-H.; Wen, J. Chloroplast Capture and Intra- and Inter-Continental Biogeographic Diversification in the Asian—New World Disjunct Plant Genus Osmorhiza (Apiaceae). Mol. Phylogenetics Evol. 2015, 85, 10–21. [Google Scholar] [CrossRef]
- Zuo, Y.-J.; Wen, J.; Zhou, S.-L. Intercontinental and Intracontinental Biogeography of the Eastern Asian—Eastern North American Disjunct Panax (the Ginseng Genus, Araliaceae), Emphasizing Its Diversification Processes in Eastern Asia. Mol. Phylogenetics Evol. 2017, 117, 60–74. [Google Scholar] [CrossRef]
- Yang, Z.; Zhao, T.-T.; Ma, Q.-H.; Liang, L.-S.; Wang, G.-X. Resolving the Speciation Patterns and Evolutionary History of the Intercontinental Disjunct Genus Corylus (Betulaceae) Using Genome-Wide SNPs. Front. Plant Sci. 2018, 9, 1386. [Google Scholar] [CrossRef]
- Bell, C.D.; Gonzalez, L.A. Historical Biogeography and Temporal Diversification in Symphoricarpos (Caprifolieae, Caprifoliaceae, Dipsacales). Syst. Bot. 2019, 44, 83–89. [Google Scholar] [CrossRef]
- Gladenkov, A.Y.; Oleinik, A.E.; Marincovich, L.; Barinov, K.B. A Refined Age for the Earliest Opening of Bering Strait. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2002, 183, 321–328. [Google Scholar] [CrossRef]
- Milne, R.I. Northern Hemisphere Plant Disjunctions: A Window on Tertiary Land Bridges and Climate Change? Ann. Bot. 2006, 98, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Milne, R.I.; Abbott, R.J. The Origin and Evolution of Tertiary Relict Floras. Adv. Bot. Res. 2002, 38, 281–314. [Google Scholar] [CrossRef]
- Krestov, P.V.; Omelko, A.M.; Nakamura, Y. Phytogeography of Higher Units of Forests and Krummholz in North Asia and Formation of Vegetation Complex in the Holocene. Phytocoenologia 2010, 40, 41–56. [Google Scholar] [CrossRef]
- Chepinoga, V.V.; Protopopova, M.V.; Pavlichenko, V.V. Detection of the Most Probable Pleistocene Microrefugia on the Northern Macroslope of the Khamar-Daban Ridge (Southern Prebaikalia). Contemp. Probl. Ecol. 2017, 10, 38–42. [Google Scholar] [CrossRef]
- Protopopova, M.; Pavlichenko, V.; Gnutikov, A.; Chepinoga, V. DNA Barcoding of Waldsteinia Willd. (Rosaceae) Species Based on ITS and trnH-psbA Nucleotide Sequences. In Information Technologies in the Research of Biodiversity; Springer: Cham, Switzerland, 2019; pp. 107–115. [Google Scholar] [CrossRef]
- Rieseberg, L.H.; Brouillet, L. Are Many Plant Species Paraphyletic? Taxon 1994, 43, 21–32. [Google Scholar] [CrossRef]
- Lo, E.Y.Y.; Stefanović, S.; Dickinson, T.A. Reconstructing Reticulation History in a Phylogenetic Framework and the Potential of Allopatric Speciation Driven by Polyploidy in an Agamic Complex in Crataegus (Rosaceae). Evolution 2010, 64, 3593–3608. [Google Scholar] [CrossRef]
- Lo, E.Y.; Donoghue, M.J. Expanded Phylogenetic and Dating Analyses of the Apples and Their Relatives (Pyreae, Rosaceae). Mol. Phylogenetics Evol. 2012, 63, 230–243. [Google Scholar] [CrossRef]
- Dickinson, T.A. Sex and Rosaceae Apomicts. Taxon 2018, 67, 1093–1107. [Google Scholar] [CrossRef]


| Waldsteinia Species | Species Name According to ‘Geum’ Concept | Distribution [3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19] |
|---|---|---|
| subgenus Waldsteinia | ||
| ++W. geoides Willd. | Geum waldsteinia Baill. =G. waldsteiniae Smedmark | C and E, incl. SE Europe (Bulgaria, Slovakia, Hungary, Macedonia, Serbia, Kosovo, Croatia, Romania, SW Ukraine) |
| subgenus Comaropsis (Rich. ex Nestl.) Teppner | ||
| +W. fragarioides (Michx.) Tratt. ≡W. fragarioides subsp. fragarioides | G. fragarioides (Michx.) Smedmark | E of North America (from SE Canada: New Brunswick, Ontario and Quebec, and NE USA: from Maine west to Minnesota, to SE USA: Tennessee, North Carolina, and Arkansas) |
| +W. doniana Tratt. ≡W. fragarioides subsp. doniana (Tratt.) Teppner =W. parviflora Small =W. fragarioides var. parviflora (Small) Fernald | G. donianum (Tratt.) Weakley & Gandhi | SE of North America (USA: Alabama, Georgia, Kentucky, N Carolina, S Carolina, Pennsylvania (?), Tennessee, and Virginia) |
| W. idahoensis Piper | G. idahoense (Piper) Smedmark | W of North America (USA: Idaho, Montana) |
| +W. lobata (Baldwin) Torr. & A. Gray | G. lobatum (Baldwin) Smedmark | SE of North America (USA: Georgia, S Carolina, and N Carolina) |
| +W. tanzybeica Stepanov | G. tanzybeicum (Stepanov) Smedmark | South Siberia (the Western Sayan Mts.) |
| +W. ternata (Stephan) Fritsch | G. ternatum (Stephan) Smedmark | South Siberia (the Western Sayan Mts., the Eastern Sayan Mts., and the Khamar-Daban Ridge) |
| +W. trifolia Rochel ex W.D.J.Koch ≡W. ternata subsp. trifolia (Rochel ex W.D.J.Koch) Teppner | G. ternatum (Stephan) Smedmark | C and SE Europe (SE Austria, Slovenia, Slovakia, Romania, and Serbia) |
| +W. maximowicziana (Teppner) Prob. ≡W. ternata subsp. maximowicziana Teppner | G. ternatum (Stephan) Smedmark | East Asia (NE China: S Jilin; Japan: Hokkaido, Honshu; North Korea, South Korea), SE of Russian Far East (the Low Amur: Khabarovsk and Primorsky regions; Sakhalin, and the Kuril Islands (?)) |
| Taxon | Locality, Voucher, and Isolate Information | Field Specimen/ Life Collection/ Herbarium | Coordinates, Altitude 1 |
|---|---|---|---|
| Coluria geoides (Pall.) Bunge | Russia, South Siberia, the Altai Mts., the Katun Riv., 12 June 2022, V. Chepinoga and N. Lashchinskiy (NSK0092604), isolate A1 | Field specimen | N 50.392198°, E 86.672328° 756 m alt. |
| Coluria henryi Batalin | China, Chongqing, 15 April 1938, K.L. Chu (PE01274689), isolate PE-Ch1 | PE (China) | Unknown |
| Waldsteinia geoides Willd. | Europe, unknown locality, 5 August 2022, M. Protopopova and V. Pavlichenko (IRKU084896), isolate BGI1 | Botanic garden of Irkutsk State University (Russia), life collection | Unknown |
| Waldsteinia doniana Tratt. | USA, Alabama, Winston County, the Sipsey Riv. near Addison, 10 April 1953, J.W. Hardin and W.H. Duncan (LE01182710), isolate LE-Wp1 | LE (Russia) | Unknown |
| Waldsteinia fragarioides (Michx.) Tratt. | Canada, Ontario, Peterborough Country, near Douro-Dummer Township, 11 June 1948, J. H. Soper and H. M. Dale (LE01182708), isolate LE-Wf1 | LE (Russia) | Unknown |
| Waldsteinia fragarioides (Michx.) Tratt. | USA, New York, Oatka Creek Park near Rochester, 20 May 1965, H. Ernst (LE01182709), isolate LE-Wf4 | LE (Russia) | Unknown |
| Waldsteinia lobata (Baldwin) Torr. & A.Gray | USA, South Carolina, Oconee County, Brasstown Creek area, 11 May 1989, S.R. Hill and C.N. Horn (PE01683697), isolate PE-Wlo1 | PE (China) | Unknown |
| Waldsteinia maximowicziana (Teppner) Prob. | Russia, the Far East, Lower Amur region, Sirenevka settlement, 11 September 2015, E.A. Pimenova (IRKU084897), isolate PK8 | Field specimen | N 43°26′4.31″, E 131°58′59.02″ |
| Waldsteinia tanzybeica Stepanov | Russia, South Siberia, the Western Sayan Mts., the Bolshoy Kebezh Riv., 11 June 2018, V. Pavlichenko, V. Chepinoga, M. Protopopova (IRKU084855), isolate BK1 | Field specimen | N 53.071575°, E 093.132594° 406 m alt. |
| Waldsteinia ternata (Stephan) Fritsch | Russia, South Siberia, the Khamar-Daban Ridge, the Bezymyannaya Riv., 31 May 2014, V. Chepinoga, V. Pavlichenko, M. Protopopova, and S. Bystrov (IRKU058136), isolate Bz1 | Field specimen | N 51.59398°, E 103.90883° 496 m alt. |
| Waldsteinia ternata (Stephan) Fritsch | Russia, South Siberia, the Khamar-Daban Ridge, the Snezhnaya Riv., 2 June 2022, V. Pavlichenko, M. Protopopova (IRKU084895), isolate S1 | Field specimen | N 51.418623°, E 104.631946° 476 m alt. |
| Waldsteinia ternata (Stephan) Fritsch | Russia, South Siberia, the Eastern Sayan Mts., the Zima Riv., 15 June 2015, M. Protopopova, V. Chepinoga (IRKU058083), isolate Z1 | Field specimen | N 53.664800°, E 100.662747° 613 m alt. |
| Waldsteinia trifolia Rochel ex W.D.J.Koch | Europe, unknown locality, 28 September 2018, V. Pavlichenko (MSKH33328), isolate MSKH2 | The Central Botanical Garden of the National Academy of Sciences of Belarus (Republic of Belarus), life collection | Unknown |
| Taxon Name 1 | Synonym by ‘Geum’ Concept | Locality 2 | GenBank Accession Numbers | Ref. 4 | |||
|---|---|---|---|---|---|---|---|
| Ribotype (R) 3 | Plastotype (P) 3 | ||||||
| ITS | trnL-trnF | ||||||
| Acomastylis calthifolia (Sm.) F.Bolle | Geum calthifolium Sm. | – | – | AJ302338.1 | – | AJ297324.1 | [30] |
| A. elata (Wall.) F.Bolle | G. elatum Wall. | – | – | AJ302339.1 | – | KY419976.1 | [30,39] |
| A. rossii (R.Br.) Greene | G. rossii (R.Br.) Ser. | – | – | AJ302340.1 | – | AJ297326.1 | [30] |
| A. sikkimensis (Prain) F.Bolle | G. sikkimense Prain | – | – | AJ302341.1 | – | AJ297327.1 | [30] |
| Coluria geoides (Pall.) Bunge. | G. geoides (Pall.) Smedmark | A1 | R1 | MN478378 | P1 | MN478380 | curr. |
| C. geoides (Pall.) Bunge. | G. geoides (Pall.) Smedmark | – | R2 | AJ302343.1 | – | – | [30] |
| C. henryi Batalin | G. henryi (Batalin) Smedmark | PE-Ch1 | – | – | P1 | MN478381 | curr. |
| Erythrocoma triflorum (Pursh) Greene | G. triflorum Pursh | – | – | AJ302344.1 | – | AJ297330.1 | [30] |
| Fallugia paradoxa (D.Don) Endl. ex Torr. | Fallugia paradoxa (D.Don) Endl. ex Torr. | – | – | AJ302345.1 | – | AJ297331.1 | [30] |
| Geum aleppicum Jacq. | G. aleppicum Jacq. | – | – | KX645654.1 | – | – | [40] |
| G. andicola (Phil.) Reiche | G. andicola (Phil.) Reiche | – | – | AJ302346.1 | – | AJ297332.1 | [30] |
| G. bulgaricum Pančić | G. bulgaricum Pančić | – | – | AJ302347.1 | – | AJ297333.1 | [30] |
| G. canadense Jacq. | G. canadense Jacq. | – | – | DQ006033.1 | – | – | [41] |
| G. geniculatum Michx. | G. geniculatum Michx. | – | – | AJ302348.1 | – | AJ297334.1 | [30] |
| G. heterocarpum Boiss. | G. heterocarpum Boiss. | – | – | AJ302349.1 | – | AJ297335.1 | [30] |
| G. japonicum Thunb. | G. japonicum Thunb. | – | – | – | – | AY818238.1 | [42] |
| G. montanum L. | G. montanum L. | – | – | AJ302350.1 | – | AJ297336.1 | [30] |
| G. reptans L. | G. reptans L. | – | – | AJ302351.1 | – | AJ297337.1 | [30] |
| G. rivale L. | G. rivale L. | – | – | AJ302352.1 | – | AJ297338.1 | [30] |
| G. schofieldii Calder & Roy L.Taylor | Geum schofieldii Calder & Roy L.Taylor | – | – | AJ302353.1 | – | AJ297339.1 | [30] |
| Geum sp. 5 | Geum sp. | – | – | AJ302342.1 | – | AJ297328.1 | [30] |
| G. speciosum Albov | G. speciosum Albov | – | – | AJ302354.1 | – | AJ297340.1 | [30] |
| G. urbanum L. | G. urbanum L. | – | – | AJ302337.1 | – | AJ297323.1 | [30] |
| G. vernum (Raf.) Torr. & A.Gray | G. vernum (Raf.) Torr. & A.Gray | – | – | AJ302355.1 | – | AJ297341.1 | [30] |
| Novosieversia glacialis (Adams ex Fisch.) F.Bolle | G. glaciale Adams ex Fisch. | – | – | AJ302356.1 | – | AJ297342.1 | [30] |
| Oncostylus cockaynei F.Bolle | G. cockaynei (F.Bolle) Molloy & C.J.Webb | – | – | AJ302357.1 | – | AJ297343.1 | [30] |
| O. leiospermus (Petrie) F.Bolle | G. leiospermum Petrie | – | – | AJ302358.1 | – | AJ297344.1 | [30] |
| Rosa persica J.F.Gmel. | Rosa persica J.F.Gmel. | – | – | AJ416468.1 | – | AJ416466.1 | [30] |
| Sanguisorba officinalis L. | Sanguisorba officinalis L. | – | – | AY635041.1 | – | AY634774.1 | [43] |
| Sieversia pentapetala (L.) Greene | G. pentapetalum (L.) Makino | – | – | AJ302359.1 | – | AJ297345.1 | [30] |
| S. pusilla (Gaertn.) Hultén | Geum selinifolium (Fisch. ex F. Schmidt) Hultén | – | – | AJ302360.1 | – | AJ297346.1 | [30] |
| Taihangia rupestris T.T.Yu & C.L.Li | G. rupestre (T.T.Yu & C.L.Li) Smedmark | – | – | AJ302361.1 | – | AJ297347.1 | [30] |
| Waldsteinia doniana Tratt. | G. donianum (Tratt.) Weakley & Gandhi | LE-Wp1 | R1 | MK616360 | P1 | MK616367 | curr. |
| W. fragarioides (Michx.) Tratt. | G. fragarioides (Michx.) Smedmark | LE-Wf1 | R1 | MK616358 | P1 | MK616366 | curr. |
| W. fragarioides (Michx.) Tratt. | G. fragarioides (Michx.) Smedmark | LE-Wf4 | R2 | MK616359 | P1 | OQ632997 | curr. |
| W. geoides Willd. | G. waldsteinia Baill. | BGI1 | R1 | MK616352 | P1 | MK616361 | curr. |
| W. geoides Willd. | G. waldsteinia Baill. | BGI1 | R2 | OQ625814 | – | – | curr. |
| W. geoides Willd. | G. waldsteinia Baill. | BGI1 | R3 | OQ625815 | – | – | curr. |
| W. geoides Willd. | G. waldsteinia Baill. | BGI1 | R4 | OQ625816 | – | – | curr. |
| W. geoides Willd. | G. waldsteinia Baill. | BGI1 | R5 | OQ625817 | – | – | curr. |
| W. geoides Willd. | G. waldsteinia Baill. | BGI1 | R6 | OQ629840 | – | – | curr. |
| W. lobata (Baldwin) Torr. & A.Gray | G. lobatum (Baldwin) Smedmark | PE-Wlo1 | – | P1 | MN478379 | curr. | |
| W. maximowicziana (Teppner) Prob. | G. ternatum (Stephan) Smedmark | PK8 | R1 | MK616357 | P1 | MK616365 | curr. |
| W. maximowicziana (Teppner) Prob. | G. ternatum (Stephan) Smedmark | PK8 | R2 | OQ625818 | – | – | curr. |
| W. maximowicziana (Teppner) Prob. | G. ternatum (Stephan) Smedmark | PK8 | R3 | OQ625819 | – | – | curr. |
| W. maximowicziana (Teppner) Prob. | G. ternatum (Stephan) Smedmark | PK8 | R4 | OQ625820 | – | – | curr. |
| W. tanzybeica Stepanov | G. tanzybeicum (Stepanov) Smedmark | BK1 | R1 | MK616354, MK616355 | P1 | MK616363 | curr. |
| W. tanzybeica Stepanov | G. tanzybeicum (Stepanov) Smedmark | BK1 | R2 | OQ625821 | – | – | curr. |
| W. tanzybeica Stepanov | G. tanzybeicum (Stepanov) Smedmark | BK1 | R3 | OQ625822 | – | – | curr. |
| W. tanzybeica Stepanov | G. tanzybeicum (Stepanov) Smedmark | BK1 | R4 | OQ625823 | – | – | curr. |
| W. ternata (Stephan) Fritsch | G. ternatum (Stephan) Smedmark | BZ1 | R1 | OQ625824 | P1 | OQ632998 | curr. |
| W. ternata (Stephan) Fritsch | G. ternatum (Stephan) Smedmark | S1 | R1 | MK616353 | P1 | MK616362 | curr. |
| W. ternata (Stephan) Fritsch | G. ternatum (Stephan) Smedmark | Z1 | R1 | OQ625825 | P1 | OQ632999 | curr. |
| W. ternata (Stephan) Fritsch | G. ternatum (Stephan) Smedmark | BZ1 | R2 | OQ625826 | – | – | curr. |
| W. ternata (Stephan) Fritsch | G. ternatum (Stephan) Smedmark | BZ1 | R3 | OQ625827 | – | – | curr. |
| W. ternata (Stephan) Fritsch | G. ternatum (Stephan) Smedmark | BZ1 | R4 | OQ625828 | – | – | curr. |
| W. trifolia Rochel ex W.D.J.Koch | G. ternatum (Stephan) Smedmark | MSKH2 | R1 | MK616356 | P1 | MK616364 | curr. |
| W. trifolia Rochel ex W.D.J.Koch | G. ternatum (Stephan) Smedmark | MSKH2 | R2 | OQ625829 | – | – | curr. |
| W. trifolia Rochel ex W.D.J.Koch | G. ternatum (Stephan) Smedmark | MSKH2 | R3 | OQ625830 | – | – | curr. |
| W. trifolia Rochel ex W.D.J.Koch | G. ternatum (Stephan) Smedmark | MSKH2 | R4 | OQ625831 | – | – | curr. |
| W. trifolia Rochel ex W.D.J.Koch | G. ternatum (Stephan) Smedmark | MSKH2 | R5 | OQ625832 | – | – | curr. |
| The Part of Range | Locality | Region 1 | Coordinates (If Known) | Voucher 2 | 2n | Ref. |
|---|---|---|---|---|---|---|
| I. Waldsteinia ternata | ||||||
| The Khamar-Daban Ridge (Southern Siberia) | the Bezymannaya riv. | Irk | N 51.59373°, E 103.90829°, 461 m alt. | C1533, IRKU | 28 | curr. |
| the Utulik riv. | Irk | N 51.54594°, E 104.04675°, 453 m alt. | C1549, IRKU | 28 | curr. | |
| the Khara-Murin riv. | Irk | N 51.45202°, E 104.41242°, 468 m alt. | C1540, IRKU | 28 | curr. | |
| the Snezhnaya riv., # 1 | Irk | N 51.43906°, E 104.63385°, 474 m alt. | C1521, IRKU | 28 | curr. | |
| the Snezhnaya riv., # 2 | Irk | N 51.3833°, E 104.6333°, 492 m alt. | C0958, IRKU | 42 | [53] | |
| the Bolshoi Mamai riv., # 1 | Bur | N 51.44864°, E 104.77549°, 472 m alt. | C1546, IRKU | 28 | curr. | |
| the Bolshoi Mamai riv., # 2 | Bur | N 51.45546°, E 104.78033°, 456 m alt. | C1518, IRKU | 28 | curr. | |
| the Vydrinaya riv. | Bur | N 51.48181°, E 104.85162°, 457 m alt. | C1510, IRKU | 28 | curr. | |
| the Anosovka riv. | Bur | N 51.5167°, E 104.9501°, 470 m alt. | C1067, IRKU | 28 | [53] | |
| the Dulikha riv. | Bur | N 51.53376°, E 105.02878°, 474 m alt. | C1514, IRKU | 28 | curr. | |
| Unknown | – | – | – | 42 | [54] | |
| The Eastern Sayan Mts. (Southern Siberia) | the Zima riv. | Irk | N 53.66476°, E 100.66254°, 613 m alt. | C1561, IRKU | 28 | curr. |
| The Western Sayan Mts. (Southern Siberia) | the Kaldar riv. | Krs | N 53.02776°, E 092.39216°, 379 m alt. | C1683, IRKU | 28 | curr. |
| II. Waldsteinia tanzibeica | ||||||
| The Western Sayan Mts. | the Bolshoy Kebezh riv., # 1 | Krs | N 53°04′, E 93°08′ | KRSU | 14 | [55] |
| the Bolshoy Kebezh riv., # 2 | Krs | N 53.071575°, E 93.132594°, 406 m alt. | C1677, IRKU | 14 | curr. | |
| III. Waldsteinia maximowicziana | ||||||
| The Russian Far East | Akademgorodok # 1 | Prk (Vla) | – | 07607, VLA | 14 | [56] |
| The Mal. Sedanka riv. | Prk (Vla) | – | 09514, VLA | 14 | [56] | |
| The Bogataya Griva | Prk (Vla) | – | 10968, VLA | 14 | [57] | |
| Taiozhny settl. | Prk | – | 07266, VLA | 14 | [56] | |
| Akademgorodok # 2 | Prk (Vla) | – | 11387, VLA | 28 | [58] | |
| Russky island | Prk (Vla) | – | 12242, VLA | 28 | [59] | |
| Vtoraya rechka, # 1 | Prk (Vla) | – | 05697, VLA | 28 | [60] | |
| Vtoraya rechka, # 2 | Prk (Vla) | – | 10548, VLA | 28 | [57] | |
| Chernaya rechka | Prk (Vla) | – | 10985, VLA | 28 | [58] | |
| Partizan settl. | Prk | – | 10177, VLA | 28 | [57] | |
| Razdolnoe settl. | Prk | – | 11314, VLA | 28 | [58] | |
| Razdolnoe settl. | Prk | – | 12759, VLA | 28 | [61] | |
| Tigrovy settl. | Prk | – | 09515, VLA | 28 | [56] | |
| The Tigrovaya riv. | Prk | 13637, VLA | 14 | [62] | ||
| Komsomolsk-on-Amur | Khk | – | 08808, VLA | 28 | [56] | |
| Palevo settl. | Sakh | – | 08885, VLA | 28 | [63] | |
| – | – | – | – | 42 | [13,54] | |
| The Japanese archipelago | Arasmyama, near Asahikawa | Hokk | – | – | 28 | [64] |
| IV. Waldsteinia trifolia | ||||||
| The South-Eastern Alps and the Carpatians (Central and Eastern Europe) | Bleiburg, northern (Carinthia) | Aus | – | – | 28 | [4] |
| Meža (Ranve) | Sln | – | – | 28 | [4] | |
| Paka riv. (Valenje) | Sln | – | – | 28 | [54] | |
| Tisovec | Slk | – | SLO | 28 | [65] | |
| Revúca | Slk | – | SLO | 28 | [65] | |
| Strelníky | Slk | – | SLO | 28 | [66,67] | |
| Frantschach (Carinthia) | Aus | – | – | 35 | [4] | |
| Wolfsberg (Carinthia) | Aus | – | – | 35 | [4] | |
| Lavamünd (Carinthia) | Aus | – | – | 35 | [4] | |
| Bleiburg, southern (Carinthia) | Aus | – | – | 35 | [4] | |
| Lippitzbach (Carintia) | Aus | – | – | 35 | [68] | |
| Nevljica riv. (Kamnic) | Sln | – | – | 35 | [69] | |
| Hliník nad Hronom | Slk | – | GZU | 35 | [4] | |
| Transylvania | Rom | – | – | 42 | [1,4] | |
| V. Waldsteinia geoides | ||||||
| The Carpathians (Central and Eastern Europe) | Unknown | Hun | – | – | 14 | [70] |
| Unknown (cult., Kiel) | – | – | – | 14 | [71] | |
| Unknown | – | – | – | 14 | [17,72] | |
| Jablonov nad Turňou | Slk | – | SLO | 14 | [65] | |
| Unknown (cult.) | Pol | – | – | 14 | [73,74] | |
| VI. Waldsteinia fragarioides | ||||||
| The Appalachian Mts. and the Great Lakes region (North America) | Greater Napanee | Ont | – | – | 14 | [13] |
| Ottawa | Ont | – | 3552, DAO | 14 | [75] | |
| Fitzroy Provincial Park # 1 | Ont | – | 3553, DAO | 14 | [17,75] | |
| Fitzroy Provincial Park # 2 | Ont | – | 3554, DAO | 14 | [17,75] | |
| Kutztown | Penn | – | 3556, DAO | 14 | [17,75] | |
| Otter Lake Sanctuary | Ont | – | 3550, DAO | 21 | [75] | |
| Gatineau Park | Que | – | 3551, DAO | 21 | [75] | |
| Ottsville | Penn | – | 3555, DAO | 21 | [17,75] | |
| George Landis Arboretum | NY | – | – | 21 | [13] | |
| Smart View, Blue Ridge parkw. | Va | – | – | 42 | [54] | |
| VII. Waldsteinia doniana | ||||||
| The Piedmont of the Appalachian Mts. (North America) | Uwharrie National Forest | NC | – | GZU | 14 | [17] |
| VIII. Waldsteinia idahoensis | ||||||
| The Bitterroot Mts. region (North America) | Lochsa River | Ida | – | GZU | 28 | [17] |
| IX. Waldsteinia lobata | ||||||
| The Southern Appalachian Mts. (North America) | Brasstown Creek | SC | – | GZU | 14 | [17] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Protopopova, M.; Pavlichenko, V.; Chepinoga, V.; Gnutikov, A.; Adelshin, R. Waldsteinia within Geum s.l. (Rosaceae): Main Aspects of Phylogeny and Speciation History. Diversity 2023, 15, 479. https://doi.org/10.3390/d15040479
Protopopova M, Pavlichenko V, Chepinoga V, Gnutikov A, Adelshin R. Waldsteinia within Geum s.l. (Rosaceae): Main Aspects of Phylogeny and Speciation History. Diversity. 2023; 15(4):479. https://doi.org/10.3390/d15040479
Chicago/Turabian StyleProtopopova, Marina, Vasiliy Pavlichenko, Victor Chepinoga, Alexander Gnutikov, and Renat Adelshin. 2023. "Waldsteinia within Geum s.l. (Rosaceae): Main Aspects of Phylogeny and Speciation History" Diversity 15, no. 4: 479. https://doi.org/10.3390/d15040479
APA StyleProtopopova, M., Pavlichenko, V., Chepinoga, V., Gnutikov, A., & Adelshin, R. (2023). Waldsteinia within Geum s.l. (Rosaceae): Main Aspects of Phylogeny and Speciation History. Diversity, 15(4), 479. https://doi.org/10.3390/d15040479










