Abstract
The dinoflagellate genus Karlodinium J. Larsen is well known to form harmful algal blooms (HABs), some of which can produce karlotoxins or other ichthyotoxins and thus cause fish-killing events. Among the 16 currently accepted species of Karlodinium (about half of which are reported to be toxic), six species (K. australe, K. decipiens, K. digitatum, K. elegans, K. veneficum, and K. zhouanum) have been reported or described in the coastal waters of China. However, a fine morphological and molecular characterization of the seldom-observed species K. decipiens has not been conducted; moreover, the negative effects of this species on aquatic animals have not been investigated. This work reports the morphological and phylogenetic characterization of a strain of K. decipiens isolated from Jiaozhou Bay, China, in 2019. The characterization of the strain was conducted using light and scanning electron microscopy, LSU, SSU rDNA, and ITS sequences-based systematic analyses, pigment analysis, and a detailed investigation of its potential toxic/harmful activity on aquatic animals. We observed the typical diagnostic features of K. decipiens, including its relatively large size, ellipsoidal or ovoid cell shape, ventral pore, ventral ridge connecting the two displaced ends of the cingulum, cingulum with a displacement of about one-third of the cell length, numerous polyhedral or slightly elongated chloroplasts distributed peripherally, and large nucleus located centrally. However, we also observed a large amphiesmal vesicle at the dorsal end of the ASC at the dorsal epicone, which is a novel feature that has never been reported from any species of the genus. Based on the results of this study, it is not clear whether this feature is a specific structure of the species or a common characteristic of the genus; therefore, this novel feature is worthy of further examination. Fucoxanthin was the most abundant pigment among all the carotenoids detected. The phylogenies inferred using Bayesian inference (BI) and maximum likelihood (ML) techniques confirmed the conspecificity of our isolate with the holotype K. decipiens (accession no. EF469236). In molecular trees, K. decipiens and K. antarcticum form a separate clade from other species of Karlodinium, and it should be examined whether a large amphiesma vesicle may be a characteristic of this clade. The exposure bioassays using brine shrimp (Artemia salina) indicated that K. decipiens exhibited toxicity to zooplankton, with 100% and 68% mortality observed in brine shrimp using live cell cultures and cell culture lysates over 120 h, respectively. Our work provides a detailed morphological and molecular characterization of K. decipiens from China. The results of this study broaden the known geographical distribution of this species and demonstrate it to be a harmful dinoflagellate.
1. Introduction
Dinoflagellates are protists with considerable diversity in their lifestyles, habitats, and trophic modes [1,2,3]. Dinoflagellates have traditionally been divided into armored and unarmored groups; the two groups are distinguishable by the number and arrangement of their amphiesmal plates and the location and displacement of their cingulum [4,5,6]. Increasingly, researchers have realized that the apical structure complex (ASC; syn. apical groove, or acrobase) is one of the most significant discernable characteristics of the unarmored species [7,8,9,10,11]; this now is widely accepted and applied for dinoflagellate identification and classification [12,13,14,15,16,17]. More than 2800 dinoflagellate species belonging to more than 600 genera have been described or reported [1,18,19]; moreover, many new species are described every year, and environmental DNA surveys conducted globally reveal that numerous dinoflagellates have not yet been described [20,21,22].
The Kareniaceae family includes the genera Karlodinium J.Larsen, Karenia Gert Hansen & Moestrup, Takayama M.F.Salas, Bolch, Botes & Hallegraeff, Asterodinium Sournia, Gertia K.Takahashi, G.Benico, Wai Mun Lum & M.Iwataki, and Shimiella Ok, H.J.Jeong, S.Y.Lee & Noh [8,9,23,24,25,26], and about 38 species have been described [27,28,29]. Karlodinium is one of the most important genera, as several Karlodinium species are well known to form harmful blooms, some of which can produce karlotoxins or other potentially toxic chemical compounds, and thus cause fish-killing events [30,31,32,33,34]. Thus far, the genus Karlodinium contains 16 accepted species, about half of which have been reported to be toxic [29]. Therefore, the accurate identification of Karlodinium species is important for harmful algal bloom (HAB) monitoring. Based on previous studies, the genus Karlodinium is polyphyletic and consists of the following three subclades: subclade I contains K. decipiens Salas & Laza-Martinez, K. antarcticum Salas, K. digitatum sensu Luo et al. [35], and an unidentified Karenia species; subclade II contains K. digitatum sensu Cen et al. [34], K. australe Salas, Bolch & Hallegraeff, and K. armiger Bergholtz, Daugbjerg & Moestrup; and subclade III contains K. veneficum (D.Ballantine) J.Larsen, K. conicum Salas, K. corrugatum Salas, K. elegans J.Y.Cen, S.H.Lu & J.Y.Wang, K. zhouanum Z.Luo & H.Gu, K. jejuense Zh.Li & H.H.Shin, K. ballantinum Salas, K. gentienii E.Nézan, N.Chomérat & R.Siano, and several unidentified Karlodinium species [35,36,37,38]. However, although subclade I contains K. decipiens, K. antarcticum, K. digitatum sensu Luo et al. [35], and an unidentified Karenia species, forming a sister clade of the Takayama species, these Karlodinium species are morphologically different from the Takayama species [34,35,36,37,38]. This contradiction could be resolved depending on the characteristics of the ultrastructure or novel species.
Reports of Karlodinium species date back to the 2000s in China. Zhou et al. [39] isolated a strain of K. veneficum (reported as K. micrum) from the East China Sea in 2005, which is probably the first report of the Karlodinium species in China. Moreover, Liu et al. [40] found that this species is distributed along the coast of China. In 2012 and 2016, Luo et al. [35] isolated K. australe and K. zhouanum from the East China Sea and the South China Sea, respectively. Cen et al. [34,37] reported K. digitatum, K. decipiens, and K. elegans from bloom water in the Pingtan coastal areas of Fujian province, China. Thus far, six Karlodinium species have been described or reported in the coastal waters of China [29], and most of them (K. veneficum, K. australe, K. zhouanum, and K. elegans) have been intensively investigated. Karlodinium decipiens has been reported by Cen et al. [34,37]; however, the knowledge of this species is still very limited.
Based on previous work, the species’ identity, its toxicity, and its position between Takayama and Karlodinium are worth determining. In this study, we established a clonal culture of the Karlodinium species from the coastal waters of Jiaozhou Bay, China, in 2019, which led us to further characterization of its morphology, phylogenetic relationships with all other species of the genus, and pigment composition. We then conducted a detailed investigation of its potential toxic/harmful activity on an aquatic animal. The identity of the isolate was confirmed as Karlodinium decipiens and, here, we present the details of our characterization.
2. Materials and Methods
2.1. Sampling and Culture Establishment
A culture of K. decipiens strain KDJZBB29 was isolated from a water sample collected at the coast of Qingdao, Shandong province, China (36.10° N, 120.25° E) in 2019. The culture was maintained in natural seawater (with a salinity of 31) supplemented with f/2 medium without silicon [41] at 21 °C, in 12 h light/dark conditions, with approximately 100 μmol quanta m−2·s−1 supplied by white fluorescent lights. A final concentration of 200 I.U. penicillin and 200 μg mL−1 streptomycin (Solarbio, Beijing, China) was added into the medium immediately before inoculation to inhibit bacterial growth.
2.2. Light Microscopy (LM)
Live cells were observed and photographed with light microscopes (BX53, Olympus, Tokyo, Japan; AXIO Vert.A1, Zeiss, Oberkochen, Germany) equipped with a digital camera (DP74, Olympus, Tokyo, Japan; Axiocam 705 color, Zeiss, Oberkochen, Germany). For epifluorescence microscopy, cells were stained with SYBR Green I (Solarbio, Beijing, China), viewed, and photographed for chlorophyll-induced red autofluorescence and SYBR Green I-induced green nuclear fluorescence. Cell sizes of K. decipiens for 50 live cells at the mid-exponential growth phase were measured at × 200 or × 400 magnification using a DP74 digital camera (Olympus, Tokyo, Japan).
2.3. Scanning Electron Microscopy (SEM)
For SEM observations, specimens were prepared according to Hu et al. [12,14]. Specifically, cultures at the mid-exponential growth stage were fixed in a final concentration of 2% OsO4 solution made up of 0.2 µm of filtered natural seawater (with a salinity of 31) for about 40–50 min at room temperature. The fixed cells were then gently filtered through an 11 μm pore-size Millipore nylon membrane, and then dehydrated in an acetone series (10%, 30%, 50%, 70%, 90%, and three times in 100%; 15 min for each step). Next, the cells were dried using a critical point dryer with liquid CO2 (EM CPD300, Leica, Vienna, Austria), and coated with platinum–palladium (EM ACE200, Leica, Vienna, Austria). The cells were observed using an S-3400N SEM (Hitachi, Hitachinaka, Japan) at a 5 kV accelerating voltage.
2.4. DNA Extraction, PCR Amplification, and Sequencing
The genomic DNA of K. decipiens strain KDJZBB29 was extracted using a Tiangen plant extraction kit (Beijing, China), following the manufacturer’s protocol. Approximately 1500 bp of 28S (large subunit [LSU]) rDNA (D1–D6) was amplified using the primer pair D1R (forward, 5′-ACCCGCTGAATTTAAGCATA-3′) [42] and 28-1483R (reverse, 5′-GCTACTACCACCAAGATCTGC-3′) [8]; approximately 660 bp of ITS was amplified using the primers ITS1 (forward, 5′-TCCGTAGGTGAACCTGCGG-3′) and ITS4 (reverse, 5′-GCATATCAATAAGCGGAGGA-3′) [43], and approximately 1800 bp of 18S (small subunit [SSU]) rDNA) was amplified using the primers EukA (forward, 5′-ACCTGGTTGATCCTGCCAGT-3′) and EukB (reverse, 5′-TCACCTACGGAAACCTTGT-3′) [44]. Polymerase chain reaction (PCR) reactions were conducted using a G1000 PCR system (Bioer Technology, Hangzhou, China) and performed with a final volume of 25 μL, containing 9.5 μL of ddH2O, 12.5 μL of 2 × Taq PCR MasterMix, 1 μL of each PCR primer (10 mM), and 1 μL of the DNA template. The thermal conditions of the PCR were as follows: an initial denaturation at 94 °C for 5 min; 35 cycles at 94 °C for 20 s, 55 °C for 30 s, and 72 °C for 2 min; and a final elongation step of 10 min at 72 °C. The amplification was confirmed via 1% agarose gel electrophoresis and visualized with ultraviolet light. The PCR product was purified using an agarose gel DNA fragment purification kit (GENEray Biotechnology, Shanghai, China) following the protocol, ligated with pMD18-T cloning vector (TaKaRa, Tokyo, Japan), and then sequenced (Genewiz, Guangzhou, China). New sequences were deposited in GenBank with accession nos. PP758579 (LSU rDNA), PP758588 (ITS), and PP758578 (SSU rDNA).
2.5. Phylogenetic Analyses
The newly obtained LSU, ITS, and SSU rDNA sequences were incorporated into those of closely related species available in the GenBank, and those of outgroup taxa were first aligned using the MAFFT v7.511 [45,46] online program (http://mafft.cbrc.jp/alignment/server/ (accessed on 2 May 2024)) with default settings, and alignments were manually checked using BioEdit v7.2.5 [47]. The final alignments of the LSU, ITS, and SSU rDNA sequences of K. decipiens consisted of 45, 31, and 19 taxa and contained 1000, 750, and 2210 positions (including gaps introduced from alignment), respectively, and the sequences of Gymnodinium catenatum (accession nos. AF200672 and DQ779990) were used as the outgroup. The program jModelTest 2.1.4 was used to select the most appropriate model of molecular evolution with the Akaike information criterion (AICc) [48]. As a result, GTR + I + G, TrN + G, and TIM1 + G were selected as the best-fit models for the LSU, ITS, and SSU rDNA datasets of K. decipiens. Phylogenetic trees were constructed using Bayesian inference (BI) and maximum likelihood (ML) analyses. Bayesian inference (BI) was performed with MrBayes 3.2.6 [49] with the best-fitting substitution model (GTR + I + G for LSU, TrN + G for ITS, and TIM1 + G for SSU). Four independent Markov chain Monte Carlo simulations were run simultaneously for 10,000,000 generations and trees were sampled every 1000 generations. The first 10% of the trees were discarded as burn-in. The convergence was assessed based on the average standard deviation of the split frequencies (all less than 0.01). The remaining trees were used to generate a consensus tree and calculate the posterior probabilities of all branches using a majority rule consensus approach. Maximum likelihood (ML) analyses were conducted with raxmlGUI v2.0.10 [50,51,52] using the models GTR + G + I (LSU), GTR + G (ITS; the model GTR + G ranked second, and the score of this model was closest to the model TrN + G), and GTR + G (SSU; the model GTR + G ranked third, and the score of this model was closest to the model TIM1 + G). Node support was assessed with 1000 bootstrap replicates. FigTree (v1.4.4) was used to view and edit trees for publication.
2.6. Pigment Analyses
For pigment analysis, a 30 mL culture of K. decipiens strain KDJZBB29 (cell density: 2.23 × 106 cells·L−1) at the exponential growth stage was filtered onto a 25 mm-diameter GF/F filter (Whatman, Maidstone, UK). The sample was immediately stored frozen at −80 °C immediately until analysis. Pigment extraction followed the methods of Zapata et al. [53] and Wang et al. [54]. Specifically, the frozen filter was cut into very small pieces and immediately placed into a 1.5 mL brown vial with 1.4 mL of 95% methanol and 0.1 mL of apo-carotenal solution (750 μg L−1 in 90% acetone; apo-carotenal 8′-apo-β, ψ-carotaldehyde was purchased from Sigma and used as the internal standard (IS)). Subsequently, the mixture was sonicated in an ultrasonic ice bath for 5 min and then filtered through a 0.22 μm polytetrafluoroethylene (PTEE) syringe filter. The pigments were analyzed using an Alliance HPLC (e2695, Waters, Milford, Massachusetts, USA) with a 100 μL sample injection, as according to Kong et al. [55] and Hu et al. [14,15]. The pigments were identified and quantified using Shimadzu Class-VP software (EMPOWER2, Pleasanton, CA, USA) and by comparing pigment spectra and retention times with the standard pigments manufactured by the Danish Hydraulic Institute Water and Environment, Hørsholm, Denmark.
2.7. Bioassays for Possible Effects on Aquatic Organisms
To test whether K. decipiens strain KDJZBB29 has toxic effects on aquatic animals, exposure experiments of a widely distributed marine zooplankton (brine shrimp, Artemia salina) were conducted using the live cell culture and cell lysates of K. decipiens. The resting eggs of brine shrimp Ar. salina were purchased from Boaifeng Biological Products Co., Ltd. (Urumqi, China), and two-day-old nauplii were used in this study. Cell lysates were prepared by sonicating the live cell culture on ice at 250 W for 60 × 10 s with a high-energy sonicator (JY92-II, Scientz Biotechnology Co., Ltd., Ningbo, China). Microscopic examination (AXIO Vert.A1, Zeiss, Germany) confirmed that there were no intact cells present. Test animals were exposed to a series of densities of live cell cultures and cell lysates (4.8 × 103, 3.6 × 103, 2.4 × 103, 1.2 × 103, and 0.5 × 103 cells·mL−1, respectively), which were obtained from diluting the initial culture (4.8 × 103 cells·mL−1, at the exponential growth stage) using f/2-Si medium [41]. A total volume of 2.5 mL of culture was gently transferred to a well of sterile 24-well culture plates, with 2–3 nauplii in each well (n = 50). The f/2-Si medium [41] and Isochrysis galbana (strain T-ISO, non-toxic prey) were used as negative controls. The activity of Ar. salina were observed at time points of 0, 0.5, 1, 2, 4, 8, 12, 24, 48, 72, 96, and 120 h, and the death of an animal was recorded when a lack of movement was observed. Dissolved oxygen (DO) levels of the culture medium were measured at the beginning of the experiment and immediately after animal death, or at the end of the experiment.
3. Results
3.1. Morphology
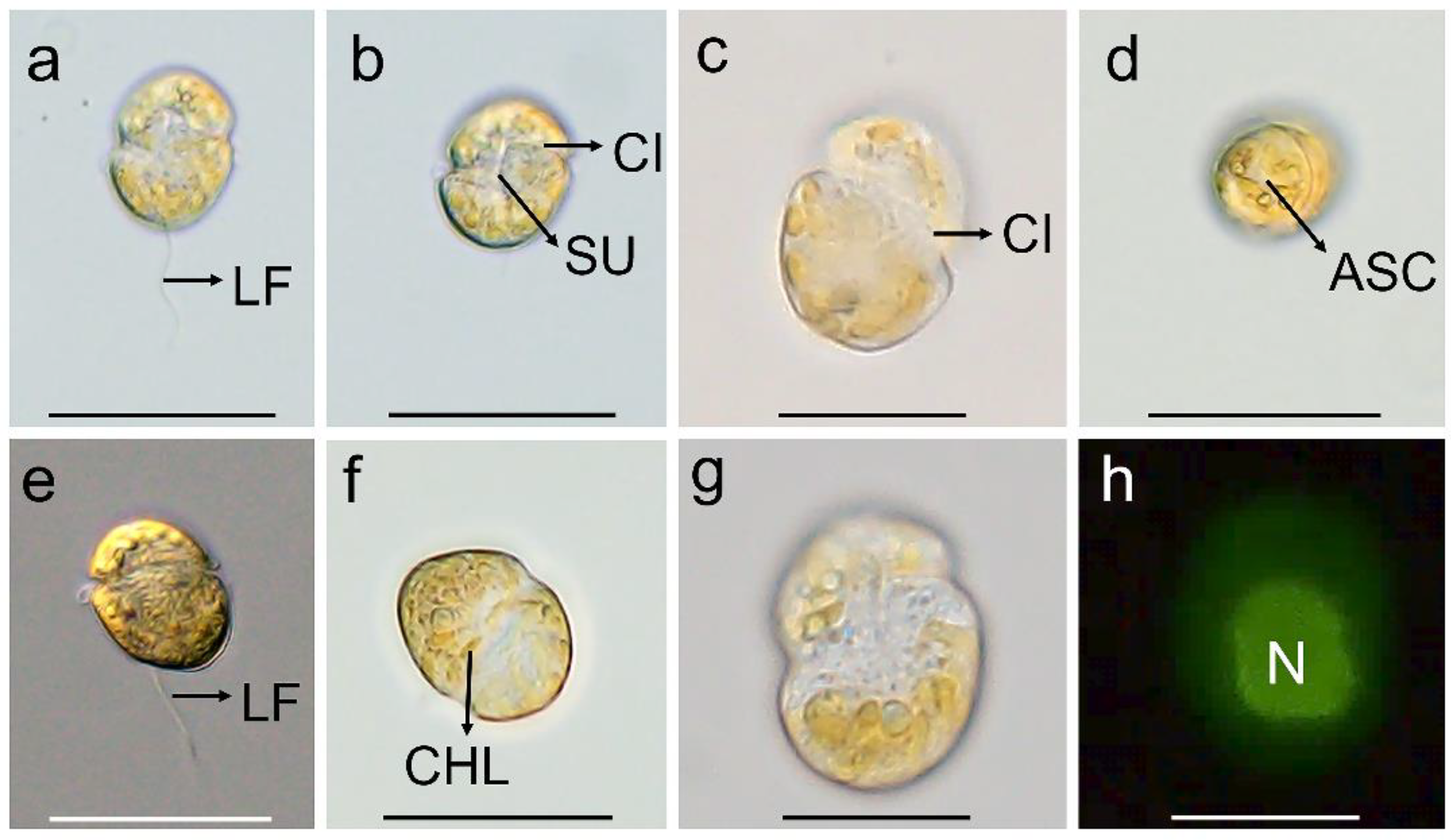
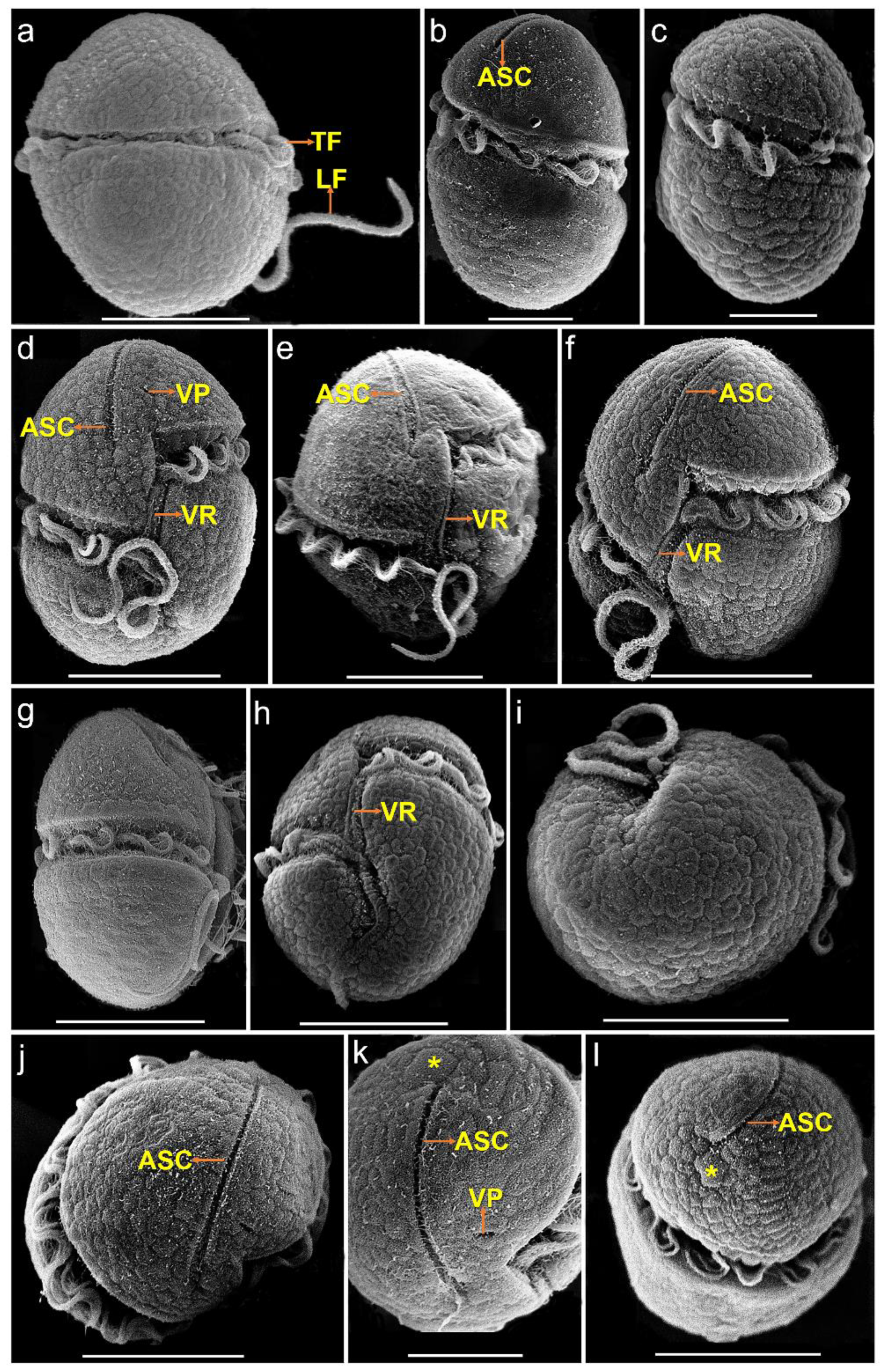
The vegetative cells of K. decipiens were ellipsoidal or ovoid, 12.4–22.9 µm (16.2 ± 2.6 µm, n = 50) in length, and 8.1–17.1 µm (11.5 ± 2.3 µm, n = 50) in width (Figure 1 and Figure 2). Numerous amphiesmal vesicles (AVs) of different shapes (polygonal or ellipsoidal) covered the cell surface (Figure 2). The epicone and hypocone were sub-hemispherical to hemispherical, and the hypocone was slightly smaller than the epicone (Figure 1 and Figure 2). A ventral pore (VP) was positioned directly above the start of the cingulum (Figure 2d,k). A long and linear apical structure complex (ASC) originated from the side of the start of the cingulum parallel (Figure 1d and Figure 2d–f,j,k), and its end connected with a large amphiesmal vesicle (Figure 2k,l). The cingulum was displaced by about 1/3 of the cell length (Figure 2d–f,h). The sulcus was short and connected with the start and end of the cingulum, and a ventral ridge (VR) was clearly visible along the sulcus (Figure 2d–f,h). Many polyhedral or slightly elongated chloroplasts were distributed peripherally (Figure 1a–g). The nucleus was large and located centrally (Figure 1h).
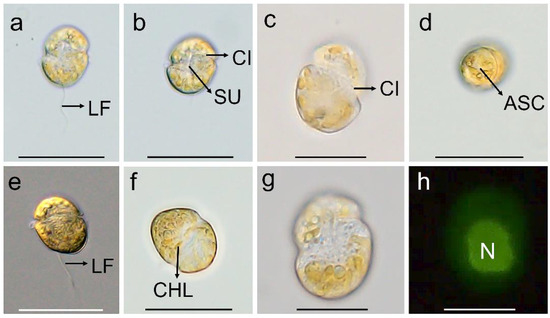
Figure 1.
Micrographs (LM) of Karlodinium decipiens strain KDJZBB29. (a,b) The surface of the ventral view showing the longitudinal flagellum (LF), cingulum (CI), and sulcus (SU); (c) dorsal view showing the cingulum (CI); (d) apical view showing the apical structure complex (ASC); (e) lateral–dorsal view showing the cingulum and longitudinal flagellum (LF); (f) surface view showing numerous chloroplasts (CHLs); and (g,h) bright and epifluorescence light microscopy observation of the same cell showing a large nucleus (N) located centrally. Scale bars for (c,g,h) = 10 μm; (a,b,d–f) = 20 μm.
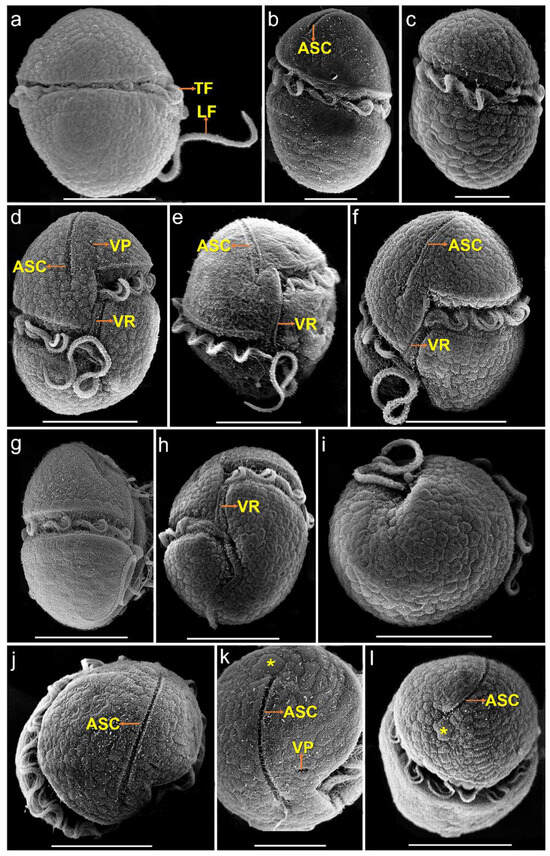
Figure 2.
SEM micrographs of Karlodinium decipiens strain KDJZBB29. (a) Dorsal view showing the longitudinal (LF) and transverse flagella (TF) in the sulcus and cingulum, respectively; (b) lateral–dorsal view showing the transverse flagella (TF) in the cingulum and the apical structure complex (ASC); (c) lateral–dorsal view showing the transverse flagellum (TF) in the cingulum; (d,e) ventral view showing the apical structure complex (ASC), ventral pore (VP), and ventral ridge (VR); (f) lateral–ventral view showing the apical structure complex (ASC) and ventral ridge (VR); (g) lateral–ventral view showing the transverse flagellum (TF) in the cingulum; (h) antapical–ventral view showing the sulcus, cingulum, and ventral ridge (VR); (i) antapical view showing amphiesmal vesicles on the cell surface; (j) apical view showing the apical structure complex (ASC); (k) apical view showing a ventral pore (VP), the apical structure complex (ASC), and a large amphiesmal vesicle (yellow asterisk) connecting the end of the ASC; and (l) apical view showing the apical structure complex (ASC) and a large amphiesmal vesicle (yellow asterisk) connecting the end of the ASC. Scale bars for (a,d,e–j,l) = 10 μm; (b,c,k) = 5 μm.
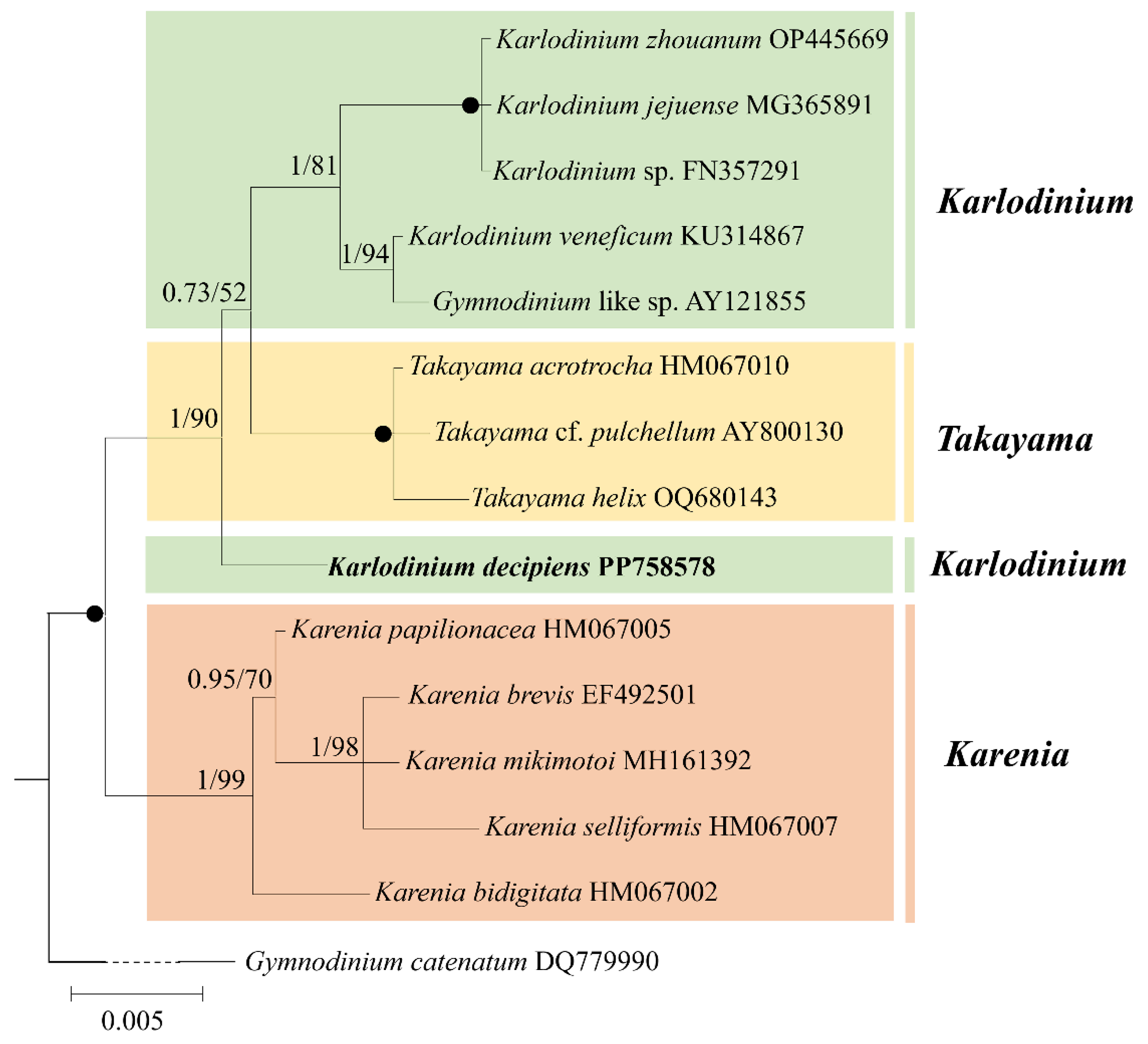
3.2. Phylogeny
We compared our 1509 bp LSU rDNA sequence (accession no. PP758579) of K. decipiens with other Gymnodiniales species and found that it was 99.9% (1456/1458) identical to the holotype sequence of K. decipiens (accession no. EF469236), while it was 99.1–100% identical to other strains of K. decipiens, indicating that our isolate is conspecific to K. decipiens (accession no. EF469236) (Table S1). Our isolate was 91.8–100% identical to other Karlodinium species, 95.0–96.9% identical to Takayama species, 85.4–91.9% identical to Karenia species, and 81.0% identical to G. catenatum (accession no. AF200672) (Table S1). ITS sequence comparison showed that our 656 bp ITS sequence (accession no. PP758588) of K. decipiens was 98.3–99.4% identical to other strains of K. decipiens, 77.4–99.5% identical to other Karlodinium species, 82.3–83.9% identical to Takayama species, 77.2–99.5% identical to Karenia species, and 89.8% identical to G. catenatum (accession no. DQ779990) (Table S2). The SSU sequence showed that our 1778 bp SSU rDNA sequence (accession no. PP758578) of K. decipiens was 98.2–98.8% identical to other Karlodinium species, 98.5–98.7% identical to Takayama species, 97.7–98.2% identical to Karenia species, and 96.1% identical to G. catenatum (accession no. DQ779990) (Table S3).
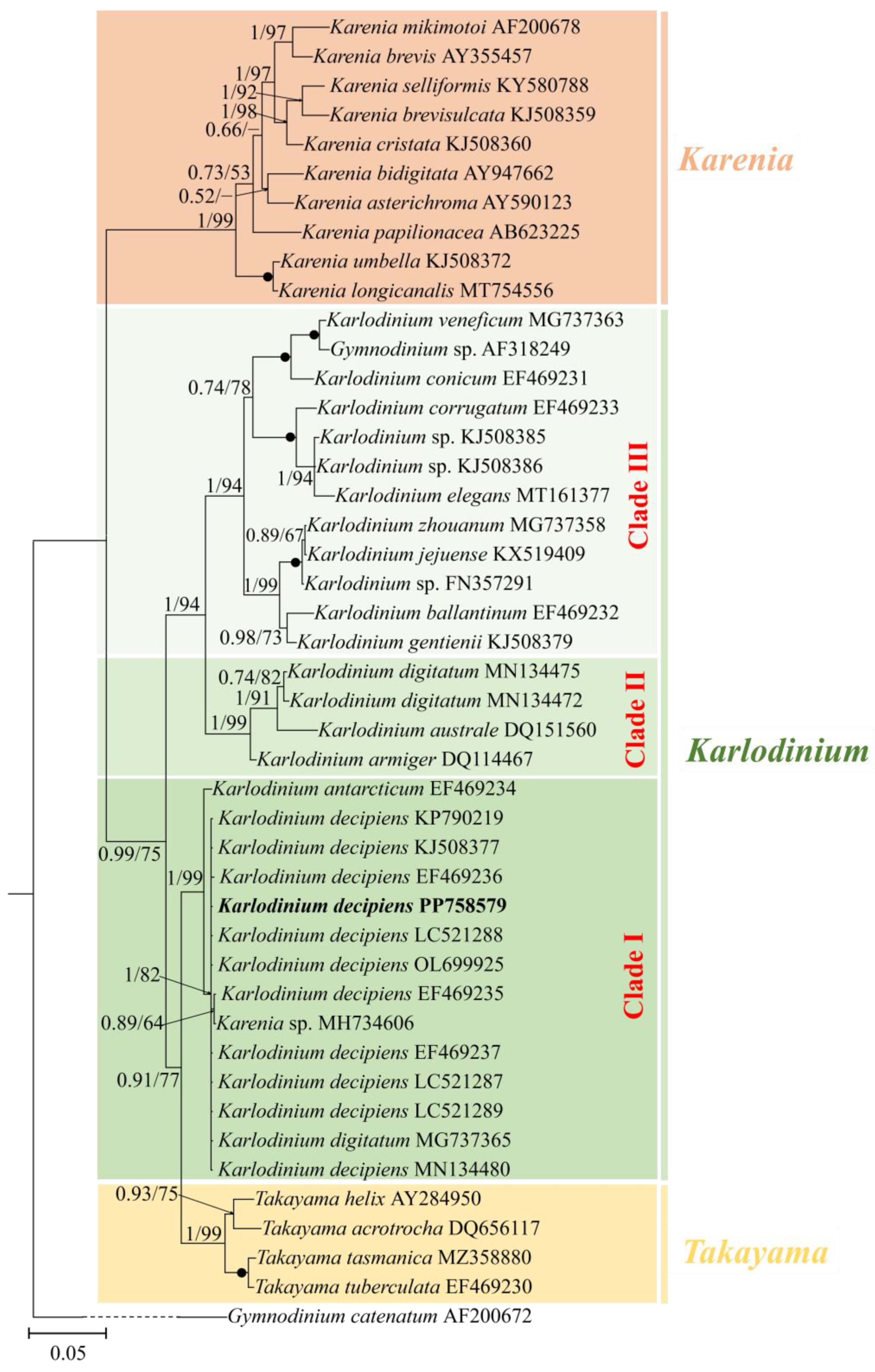
The phylogenetic position of K. decipiens was inferred from the partial LSU, ITS, and SSU rDNA sequences obtained in this study (Figure 3, Figure 4 and Figure 5). Phylogenetic analyses using Bayesian inference (BI) and maximum likelihood (ML) generated similar trees based on LSU rDNA sequences (Figure 3). The two genera Takayama and Karenia were monophyletic, with a very high support (1.00 PP/99 BS). The genus Karlodinium was polyphyletic and consisted of the following three subclades: subclade I contained 11 strains of K. decipiens, K. antarcticum, an entity annotated as K. digitatum, and an unidentified Karenia species; subclade II contained K. digitatum, K. australe, and K. armiger; and subclade III contained the rest of the Karlodinium species (Figure 3). The 11 strains of K. decipiens, K. digitatum (accession no. MG737365), and Karlodinium sp. formed a coherent clade with high support (1.00 PP/82 BS), and this clade formed a sister group with K. antarcticum with high support (1.00 PP/99 BS) (Figure 3).
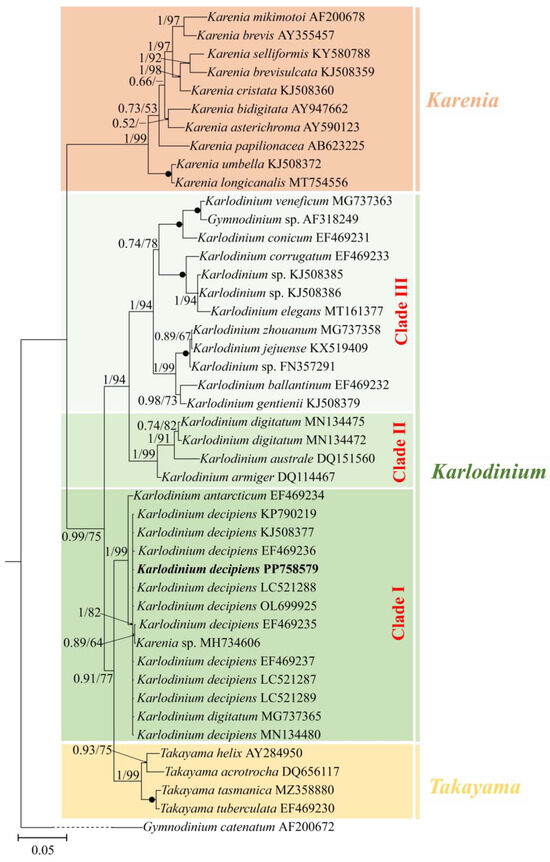
Figure 3.
Phylogeny of Karlodinium decipiens and other Gymnodiniales species inferred from large subunit rDNA (LSU rDNA) sequences using Bayesian inference (BI) with Gymnodinium catenatum (AF200672) as an outgroup. The new sequence of K. decipiens (PP758579) is indicated in bold. Numbers on branches are statistical support values to clusters to their right (Bayesian posterior probability/ML bootstrap support). Posterior probabilities of >0.5 and bootstrap values of >50% are shown. Solid dots indicate maximal support (Bayesian posterior probability: 1.00/ML bootstrap support: 100).
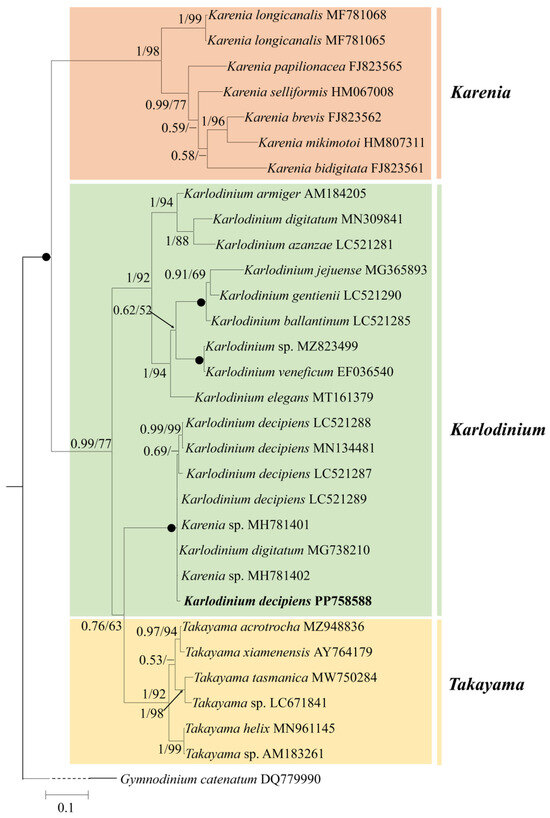
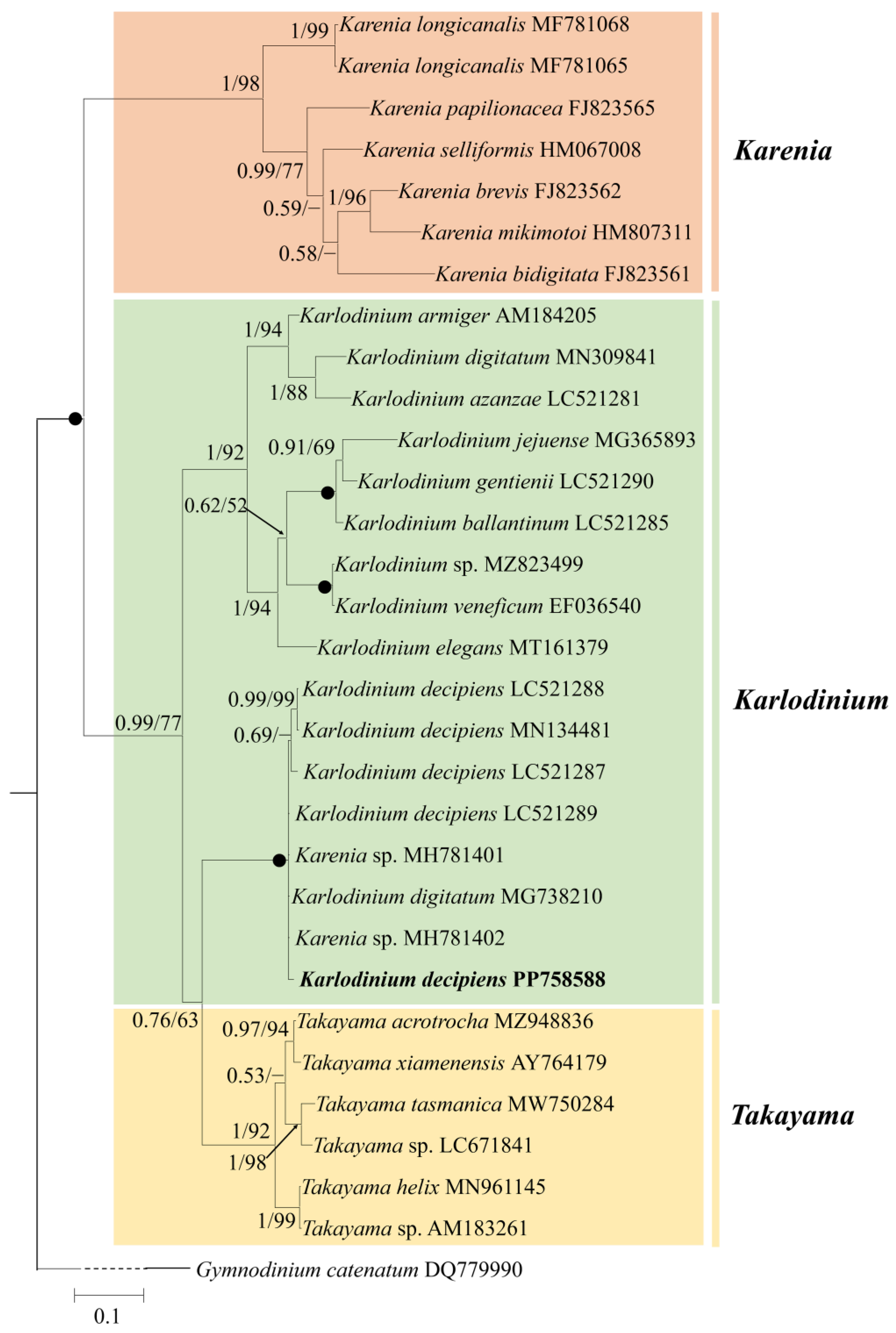
Figure 4.
Phylogeny of Karlodinium decipiens and other Gymnodiniales species inferred from ITS sequences using Bayesian inference (BI) with Gymnodinium catenatum (DQ779990) as an outgroup. The new sequence of K. decipiens (PP758588) is indicated in bold. Numbers on branches are statistical support values to clusters to their right (Bayesian posterior probability/ML bootstrap support). Posterior probabilities of >0.5 and bootstrap values of >50% are shown. Solid dots indicate maximal support (Bayesian posterior probability: 1.00/ML bootstrap support: 100).
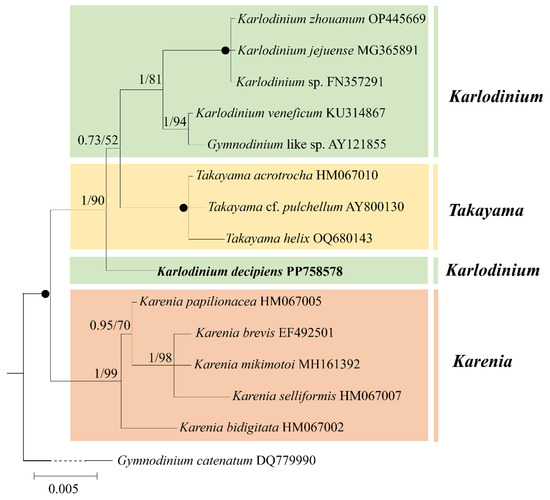
Figure 5.
Phylogeny of Karlodinium decipiens and other Gymnodiniales species inferred from small subunit rDNA (SSU rDNA) sequences using Bayesian inference (BI) with Gymnodinium catenatum (DQ779990) as an outgroup. The new sequence of K. decipiens (PP758578) is indicated in bold. Numbers on branches are statistical support values to clusters to their right (Bayesian posterior probability/ML bootstrap support). Posterior probabilities of >0.5 and bootstrap values of >50% are shown. Solid dots indicate maximal support (Bayesian posterior probability: 1.00/ML bootstrap support: 100).
Figure 4 shows the best phylogenetic tree constructed using BI using ITS sequences, and ML analysis generated a similar topology that differed in only a few internal nodes. The phylogeny inferred from the ITS dataset was very similar to that from the LSU rDNA dataset, in which the genus Karlodinium consisted of three subclades (Figure 4). The five strains of K. decipiens, K. digitatum (accession no. MG738210), and two unspecified Karenia species formed a coherent clade with maximum support (1.00 PP/100 BS), and this clade formed a sister group with Takayama species with high support (0.76 PP/63 BS) (Figure 4).
Phylogenetic analyses using Bayesian inference (BI) and maximum likelihood (ML) generated similar trees based on the SSU rDNA sequences (Figure 5). The genus Karenia formed a sister group with the genera Karlodinium and Takayama (1.00 PP/100 BS) (Figure 5). Karlodinium decipiens formed a sister group with the other Karlodinium species and Takayama with high support (1.00 PP/90 BS) (Figure 5).
3.3. Pigment
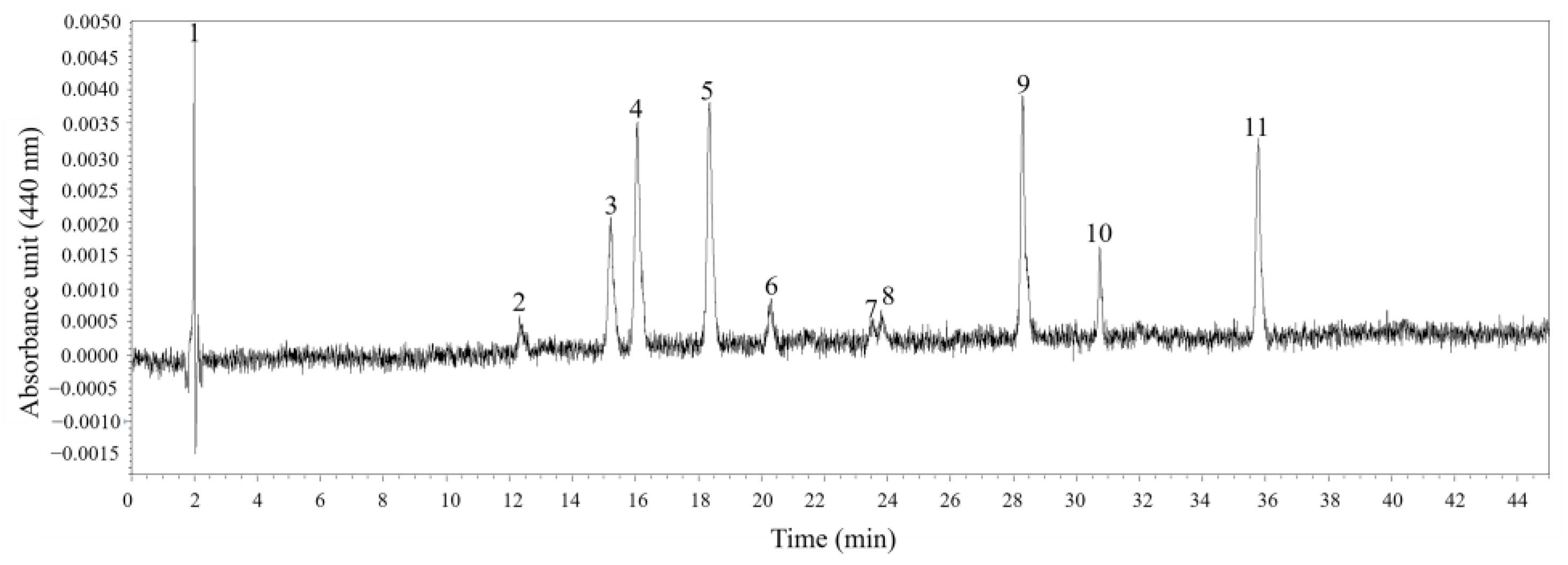
Six different pigments in K. decipiens strain KDJZBB29 were detected and confirmed according to the standards—specifically, one chlorophyll, Chl a, and five carotenoids: 19-butanoyloxyfucoxanthin, fucoxanthin, 19′-Hexanoyloxyfucoxanthin, diadinoxanthin, and diatoxanthin (Figure 6). The content of Chl a was 3.16 pg·cell−1 (Figure 6). For the identified pigments, fucoxanthin was the most abundant pigment among all carotenoids and pigments detected (1.99 pg·cell−1), higher than that of 19-butanoyloxyfucoxanthin (0.93 pg·cell−1), 19′-Hexanoyloxyfucoxanthin (1.73 pg·cell−1), diadinoxanthin (0.28 pg·cell−1), and diatoxanthin (0.14 pg·cell−1) (Figure 6). There were four unidentified peaks, which might be novel pigments or pigments without standards used in this study (Figure 6). Pigment ratios to Chl a based on concentration are shown in Table 1.
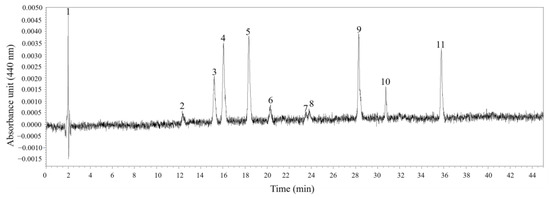
Figure 6.
Absorption chromatograms (440 nm) of the pigment extracts of Karlodinium decipiens strain KDJZBB29 from Jiaozhou Bay, China. The peak numbers and corresponding pigments are as follows: 1, 3, 7, and 10, unknown; 2, 19-butanoyloxyfucoxanthin (But-fuco); 4, fucoxanthin (Fuco); 5, 19′-Hexanoyloxyfucoxanthin (Hex-fuco); 6, diadinoxanthin (Diadino); 8, diatoxanthin (Diato); 9, internal standard (IS); and 11, chlorophyll a (Chl a).

Table 1.
Pigment composition and ratios to Chl a of Karlodinium decipiens (strain KDJZBB29).
3.4. Toxicity of Karlodinium decipiens to Brine Shrimp
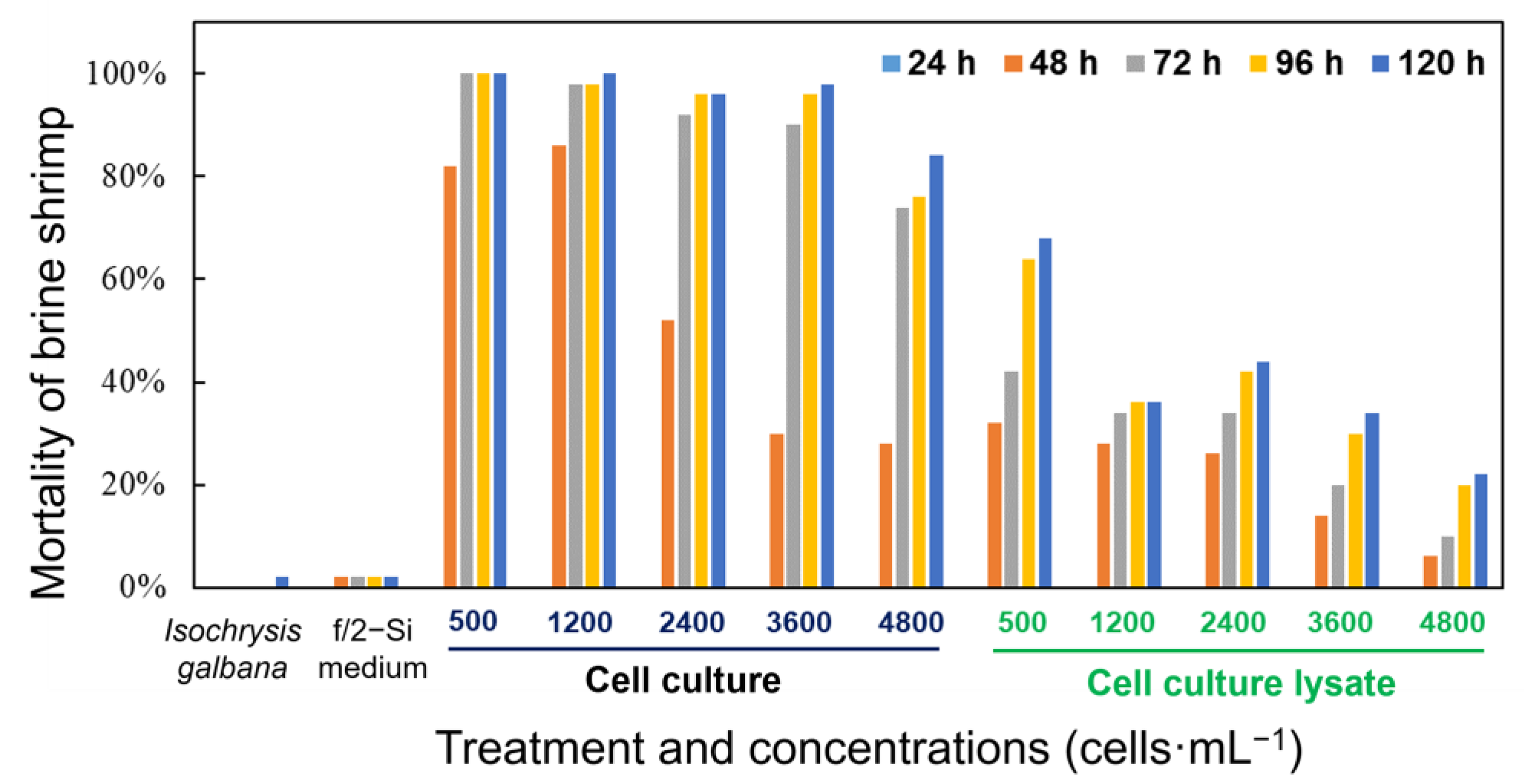
Both the whole cell culture and cell lysate of K. decipiens exhibited strong toxicity to brine shrimp (Figure 7). In the bioassays, the brine shrimp exposed to the live cell culture showed significantly higher mortality rates compared with those exposed to the sonicated cell lysate at the same cell concentration, with 28–100% mortality observed in the live cell cultures at the cell density of 500–4800 cells·mL−1 within 120 h, but with only 6–68% brine shrimp-kills observed in the treatment of cell lysates with equivalent cell densities (Figure 7). There were no mortalities among the brine shrimp exposed to the whole cell cultures and the sonicated cell lysate in the initial 24 h. The live cell cultures of K. decipiens at all cell densities (4.8 × 103, 3.6 × 103, 2.4 × 103, 1.2 × 103, and 0.5 × 103 cells·mL−1) caused 28–86% mortality within the first 48 h of exposure, while reaching up to 84–100% by the end of experiment (120 h) (Figure 7). However, the mortality rates among brine shrimp exposed to the cell lysates (4.8 × 103, 3.6 × 103, 2.4 × 103, 1.2 × 103, and 0.5 × 103 cells·mL−1) were 6–32% in the first 48 h, while reaching up to 22–68% by the end of experiment (120 h) (Figure 7). The dissolved oxygen (DO) levels were 80–90% of the saturated DO concentrations at 21 °C, and only one brine shrimp died in the negative controls within 120 h (Figure 7).
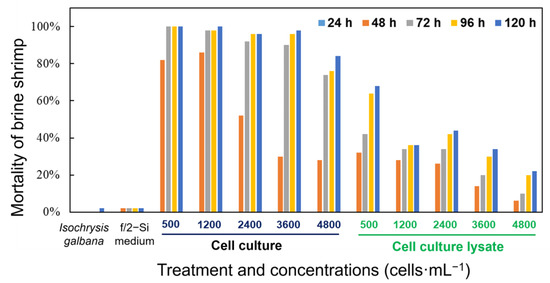
Figure 7.
Mortality of the brine shrimp Artemia salina treated with a density range of live cells and cell lysates of K. decipiens strain KDJZBB29 within 120 h. F/2-Si medium and Isochrysis galbana (T-ISO, non-toxic) were used as negative controls.
4. Discussion
The genus Karlodinium was characterized based on the following morphological traits: unarmoured dinoflagellates with chloroplasts containing internal, lenticular pyrenoids and fucoxanthin or fucoxanthin derivatives as main accessory pigments, with a straight apical groove [8,35]. Our isolate has the typical characteristics of Karlodinium: a lack of a theca, many polyhedral or slightly elongated chloroplasts, fucoxanthin as the richest pigments besides Chl a, and a straight ASC. We also observed that the general morphology of our isolate was ellipsoidal or ovoid, with a ventral pore and a ventral ridge (VR) present, and the cingulum was displaced by about 1/3 of the cell length. Our isolate was 99.9% identical to the entity of K. decipiens (accession no. EF469236). Karlodinium decipiens was described by de Salas et al. [56], who distinguished it from other Karlodinium species by its general cell morphology, its large number of polyhedral to lenticular chloroplasts, the length of its apical groove, the position of its ventral pore, and the size, morphology, and location of its nucleus [29,35,36,38,56]. Based on the abovementioned morphological and molecular characterization, we can conclude that our isolate is conspecific with the entity annotated as K. decipiens. In addition, a large amphiesmal vesicle at the dorsal epicone connected with the end of the ASC was first observed in K. decipiens, which has not been reported in K. vitiligo [57], K. veneficum [8,26], K. australe [58], K. armiger [26], K. antarcticum [56], K. ballantinum [56], K. conicum [56], K. corrugatum [56], K. corsicum [59], K. gentienii [60], K. zhouanum [35], K. jejuense [36], K. digitatum [34], K. azanzae [38], and K. elegans [37].
A large amphiesmal vesicle at the dorsal epicone connected with the end of the ASC was not observed in the type species of the genus Karlodinium, K. veneficum, or another species, K. australe (Figure S1), whose identities were confirmed in our previous works [32,40]. The current absence of this feature in all other described species of Karlodinium is due to the different qualities of SEM micrographs in the original descriptions of those species. The characteristic of amphiesmal vesicles covering the cell of K. antarcticum was not observed in de Salas et al.’s study, which suggests that this species does not have this feature or that the cells had not been well-fixed [56]. Furthermore, since the description of K. antarcticum, this species has not been reported anywhere, which makes it difficult for us to compare K. decipiens with K. antarcticum. The presence of a large amphiesmal vesicle at the dorsal epicone connecting the end of the ASC could be used as a diagnostic feature to distinguish K. decipiens from other Karlodinium species, as it is a distinct and “qualitative” characteristic; however, confirming this novel feature requires further study of the presence or absence of this characteristic in all Karlodinium species.
Molecular phylogenetic analyses revealed that 11 strains of K. decipiens, K. digitatum sensu Luo et al. [35], and an unidentified Karenia species (accession no. MH734606) form a clade with high support (refer to Figure 3). The 11 strains of K. decipiens differed from each other by 0–0.9%, showing only a very small difference (0.1%, one base pair) with Karenia sp. (Accession no. MH734606) and no difference with K. digitatum sensu Luo et al. [35] in the partial LSU rDNA sequence. In the ITS sequence phylogenetic tree (refer to Figure 4), five strains of K. decipiens, K. digitatum sensu Luo et al. [35] and two unidentified Karenia species (accession nos. MH781401 and MH781402) formed a clade with maximum support. The five strains of K. decipiens differed from each other by 0.6–1.7%, but there was a smaller difference (0.5%, three base pairs) between the two Karenia species and K. digitatum sensu Luo et al. [35]. Cen et al. [34] considered that K. digitatum sensu Luo et al. [35] is more likely a strain of K. decipiens, and our phylogeny supports this theory, suggesting that the two Karenia entities were likely misannotated.
It is noteworthy that in the LSU rDNA sequence phylogenetic tree (refer to Figure 3), K. decipiens and K. antarcticum formed a monophyletic clade with high statistical support (1.00 PP/99 BS), and they differed from each other in eight bases only (0.5% difference), which is even lower than the maximal difference among strains of K. decipiens (0.9% difference). As previously mentioned, K. decipiens and K. antarcticum formed a clade, which was closer to the Takayama clade with high support (0.91 PP/77 BS in LSU; 0.76 PP/63 BS in ITS) than to the other two clades of Karlodinium species (Figure 3 and Figure 4). Our results are in accordance with the the authors of [34,35,36,37,56] and Cen et al. [37], in that K. decipiens and K. antarcticum may have a common ancestor with Takayama. However, de Salas et al. [56] found, based on the phylogenetic relationship between K. decipiens, K. antarcticum, other Karlodinium species, and Takayama species, that K. decipiens and K. antarcticum were genetically distinct from both Karlodinium and Takayama, and thus proposed that the two species could be separated as a novel genus. However, there has been no observation of any distinct morphological feature to support this separation. However, depending on further observations on the presence or absence of the abovementioned large amphiesmal vesicle at the dorsal epicone in all Karlodinium species, there is a possibility that this structure may be an adequate feature for the establishment of this new genus.
Approximately half of Karlodinium species produce various toxins, form harmful algal blooms, or have detrimental impacts on aquatic animals [29]. Thus far, K. veneficum, K. australe, K. zhouanum, K. digitatum, K. decipiens, and K. elegans have been described or reported in the coastal waters of China [29]. Among them, K. veneficum, K. australe, and K. digitatum have been reported to impact aquatic animals and the aquaculture industry [61], while K. zhouanum and K. elegans are not harmful to aquatic aminals [37,62]. Karlodinium decipiens was recently discovered in the coastal waters of China. Identifying whether Karlodinium decipiens is a toxic/non-toxic Karlodinium species in China is important to fisheries and marine ecosystems. We found that the whole cell culture and cell lysate of K. decipiens exhibited strong toxicity to brine shrimp; in addition, the brine shrimp exposed to the live cell culture showed significantly higher mortality compared with those exposed to the sonicated cell lysate at the same cell concentration. This result indicated that the toxicity of K. decipiens is dependent on live cells, which is similar to that of K. veneficum [33]. However, the chemical characteristics of the species and its toxins, as well as whether K. decipiens is toxic to other aquatic animals (e.g., zooplankton and fish), were not elucidated in this study. Therefore, further investigations of the toxin(s) and toxicity of K. decipiens are required.
5. Conclusions
Our morphological observations and phylogenetic analyses together confirmed the presence of K. decipiens in Jiaozhou Bay, China. The results of our analyses broaden the known geographical distribution of K. decipiens in the northern marine ecosystem of China. We also observed a novel morphological feature consisting of a large amphiesmal vesicle at the dorsal epicone that connects to the end of the ASC in K. decipiens. The significance of this feature requires further detailed investigation in all Karlodinium species. Finally, exposure bioassays using brine shrimp (Artemia salina) demonstrated that K. decipiens is a harmful dinoflagellate.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/d16080449/s1, Figure S1: SEM micrographs of Karlodinium veneficum strain KVBDH-1 (a) and Karlodinium australe strain KaJb05 (b). (a, b) Apical view showing the apical structure complex (ASC) connecting an amphiesmal vesicle (yellow asterisk) at the end of the ASC. Scale bars = 5 μm; Table S1: Comparisons in the LSU rDNA sequences between Karlodinium decipiens (1509 bp, GenBank accession no. PP758579) and that of the type specimen (1458 bp, GenBank accession no. EF469236) and other species in Gymnodiniales in NCBI database; Table S2: Comparisons in the ITS sequences between Karlodinium decipiens (656 bp, GenBank accession no. PP758588) and other species in Gymnodiniales in NCBI database; Table S3: Comparisons in the SSU rDNA sequences between Karlodinium decipiens (1778 bp, GenBank accession no. PP758578) and other species in Gymnodiniales in NCBI database.
Author Contributions
Conceptualization, Z.H. and Y.Z.T.; methodology, Z.H., J.W. and Y.S.; software, J.Y., Y.F. and Z.H.; validation, J.Y., Y.F., Z.H., Y.D. and Y.Z.T.; formal analysis, J.Y., Y.F., Q.C., X.C. and Z.H.; investigation, J.Y., Y.F., Q.C., X.C., Z.H., J.W., Y.D., Y.S. and Y.Z.T.; resources, Z.H. and Y.Z.T.; data curation, J.Y., Y.F., Q.C., X.C., Z.H., J.W., Y.D., Y.S. and Y.Z.T.; writing—original draft preparation, J.Y., Y.F., Q.C. and Z.H.; writing—review and editing, J.Y., Y.F., Q.C., X.C., Z.H., Y.D., J.W., Y.S. and Y.Z.T.; supervision, Z.H. and Y.Z.T.; project administration, Z.H. and Y.Z.T.; funding acquisition, Z.H. and Y.Z.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Science & Technology Basic Resources Investigation Program of China, grant number 2018FY100200, the Program for Scientific Research Start-up Funds of Guangdong Ocean University, grant number 060302022201, the Undergraduate Student Innovation and Entrepreneurship Training Program Project (S202210566010), and the Undergraduate Innovation Team of Guangdong Ocean University (CXTD2023002).
Institutional Review Board Statement
Not applicable.
Data Availability Statement
Data are contained within the article and Supplementary Materials.
Acknowledgments
The authors greatly appreciate the constructive advice of the anonymous reviewers.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Gómez, F. Diversity and Classification of Dinoflagellates. In Dinoflagellates: Classification, Evolution, Physiology, and Ecological Significance; Rao, D.V.S., Ed.; Nova Science Publisher: New York, NY, USA, 2020; p. 38. [Google Scholar]
- Taylor, F.J.R.; Hoppenrath, M.; Saldarriaga, J.F. Dinoflagellate diversity and distribution. Biodivers. Conserv. 2008, 17, 407–418. [Google Scholar] [CrossRef]
- Morse, D.; Tse, S.P.K.; Lo, S.C.L. Exploring dinoflagellate biology with high-throughput proteomics. Harmful Algae 2018, 75, 16–26. [Google Scholar] [CrossRef]
- Taylor, F.J.R. Dinoflagellate Morphology. In The Biology of Dinoflagellates; Taylor, F.J.R., Ed.; Blackwell Scientific Publications: Oxford, UK, 1987; pp. 24–91. [Google Scholar]
- Fensome, R.A.; Taylor, F.J.R.; Norris, G.; Sargeant, W.A.S.; Wharton, D.I.; Williams, G.L. A Classification of Living and Fossil Dinoflagellates; Sheridan Press: Hanover, PA, USA, 1993; Volume 7, pp. 1–351. [Google Scholar]
- Kofoid, C.A.; Swezy, O. The Free-Living Unarmored Dinoflagellata; University of California Press: Berkeley, CA, USA, 1921; Volume 5, p. 559. [Google Scholar]
- Takayama, H. Apical grooves of unarmored dinoflagellates. Bull. Plankton Soc. Jpn. 1985, 32, 129–140. [Google Scholar]
- Daugbjerg, N.; Hansen, G.; Larsen, J.; Moestrup, Ø. Phylogeny of some of the major genera of dinoflagellates based on ultrastructure and partial LSU rDNA sequence data, including the erection of three new genera of unarmoured dinoflagellates. Phycologia 2000, 39, 302–317. [Google Scholar] [CrossRef]
- de Salas, M.F.; Bolch, C.J.S.; Botes, L.; Nash, G.; Wright, S.W.; Hallegraeff, G.M. Takayama gen. nov. (Gymnodiniales, Dinophyceae), A new genus of unarmored dinoflagellates with sigmoid apical grooves, including the description of two new species. J. Phycol. 2003, 39, 1233–1246. [Google Scholar] [CrossRef]
- Moestrup, Ø.; Hakanen, P.; Hansen, G.; Daugbjerg, N.; Ellegaard, M. On Levanderina fissa gen. & comb. nov. (Dinophyceae) (syn. Gymnodinium fissum, Gyrodinium instriatum, Gyr. uncatenum), a dinoflagellate with a very unusual sulcus. Phycologia 2014, 53, 265–292. [Google Scholar] [CrossRef]
- Hoppenrath, M. Dinoflagellate taxonomy—A review and proposal of a revised classification. Mar. Biodivers. 2017, 47, 381–403. [Google Scholar] [CrossRef]
- Gao, M.; Hu, Z.; Luo, Z.; Deng, Y.; Shang, L.; Sun, Y.; Tang, Y. Morphological and molecular characterization of the unarmored dinoflagellate Gymnodinium trapeziforme (Dinophyceae) from Jiaozhou Bay, China. Diversity 2023, 15, 1186. [Google Scholar] [CrossRef]
- Hu, Z.; Deng, Y.; Li, Y.; Tang, Y.Z. The morphological and phylogenetic characterization for the dinoflagellate Margalefidinium fulvescens (=Cochlodinium fulvescens) isolated from the Jiaozhou Bay, China. Acta Oceanol. Sin. 2018, 37, 11–17. [Google Scholar] [CrossRef]
- Hu, Z.; Deng, Y.; Luo, Z.; Shang, L.; Kong, F.; Gu, H.; Zhao, Z.; Tang, Y.Z. Characterization of the unarmored dinoflagellate Pseliodinium pirum (Ceratoperidiniaceae) from Jiaozhou Bay, China. Phycol. Res. 2020, 68, 3–13. [Google Scholar] [CrossRef]
- Hu, Z.; Li, Z.; Deng, Y.; Iwataki, M.; Luo, Z.; Wang, J.; Sun, Y.; Zhao, Z.; Gu, H.; Shin, H.H.; et al. Morphology, ultrastructure, and molecular phylogeny of the unarmoured dinoflagellate Kirithra sigma sp. nov. (Ceratoperidiniaceae, Dinophyceae). Phycologia 2020, 59, 385–396. [Google Scholar] [CrossRef]
- Hu, Z.; Xu, N.; Gu, H.; Chai, Z.; Takahashi, K.; Li, Z.; Deng, Y.; Iwataki, M.; Matsuoka, K.; Tang, Y.Z. Morpho-molecular description of a new HAB species, Pseudocochlodinium profundisulcus gen. et sp. nov., and its LSU rRNA gene based genetic diversity and geographical distribution. Harmful Algae 2021, 108, 102098. [Google Scholar] [CrossRef] [PubMed]
- Boutrup, P.V.; Moestrup, Ø.; Tillmann, U.; Daugbjerg, N. Ultrastructure and phylogeny of Kirithra asteri gen. et sp. nov. (Ceratoperidiniaceae, Dinophyceae)—A free-living, thin-walled marine photosynthetic dinoflagellate from Argentina. Protist 2017, 168, 586–611. [Google Scholar] [CrossRef] [PubMed]
- Gómez, F. A checklist and classification of living dinoflagellates (Dinoflagellata, Alveolata). CICIMAR Ocean. 2012, 27, 65–140. [Google Scholar] [CrossRef]
- Guiry, M.D. How many species of algae are there? A reprise. Four kingdoms, 14 phyla, 63 classes and still growing. J. Phycol. 2024, 60, 214–228. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, Y.; Chai, Z.; Hu, Z.; Tang, Y.Z. A combined approach detected novel species diversity and distribution of dinoflagellate cysts in the Yellow Sea, China. Mar. Pollut. Bull. 2023, 187, 114567. [Google Scholar] [CrossRef] [PubMed]
- Djurhuus, A.; Closek, C.J.; Kelly, R.P.; Pitz, K.J.; Michisaki, R.P.; Starks, H.A.; Walz, K.R.; Andruszkiewicz, E.A.; Olesin, E.; Hubbard, K.; et al. Environmental DNA reveals seasonal shifts and potential interactions in a marine community. Nat. Commun. 2020, 11, 254. [Google Scholar] [CrossRef] [PubMed]
- Stern, R.F.; Horak, A.; Andrew, R.L.; Coffroth, M.-A.; Andersen, R.A.; Küpper, F.C.; Jameson, I.; Hoppenrath, M.; Véron, B.; Kasai, F.; et al. Environmental barcoding reveals massive dinoflagellate diversity in marine environments. PLoS ONE 2010, 5, e13991. [Google Scholar] [CrossRef]
- Sournia, A. Quatre nouveaux dinoflagellés du plancton marin. Phycologia 1972, 11, 71–74. [Google Scholar] [CrossRef]
- Takahashi, K.; Benico, G.; Lum, W.M.; Iwataki, M. Gertia stigmatica gen. et sp. nov. (Kareniaceae, Dinophyceae), a new marine unarmored dinoflagellate possessing the peridinin-type chloroplast with an eyespot. Protist 2019, 170, 125680. [Google Scholar] [CrossRef]
- Ok, J.H.; Jeong, H.J.; Lee, S.Y.; Park, S.A.; Noh, J.H. Shimiella gen. nov. and Shimiella gracilenta sp. nov. (Dinophyceae, Kareniaceae), a kleptoplastidic dinoflagellate from Korean waters and its survival under starvation. J. Phycol. 2021, 57, 70–91. [Google Scholar] [CrossRef]
- Bergholtz, T.; Daugbjerg, N.; Moestrup, Ø.; Fernández-Tejedor, M. On the identity of Karlodinium veneficum and description of Karlodinium armiger sp. nov. (Dinophyceae), based on light and electron microscopy, nuclear-encoded LSU rDNA, and pigment composition. J. Phycol. 2006, 42, 170–193. [Google Scholar] [CrossRef]
- Cen, J.; Lu, S.; Moestrup, Ø.; Jiang, T.; Ho, K.C.; Li, S.; Li, M.; Huan, Q.; Wang, J. Five Karenia species along the Chinese coast: With the description of a new species, Karenia hui sp. nov. (Kareniaceae, Dinophyta). Harmful Algae 2024, 137, 102645. [Google Scholar] [CrossRef]
- Guiry, M.D.; Guiry, G.M. AlgaeBase. Available online: https://www.algaebase.org/browse/taxonomy/#91756 (accessed on 21 May 2024).
- Yang, H.; Hu, Z.; Tang, Y.Z. Plasticity and multiplicity of trophic modes in the dinoflagellate Karlodinium and their pertinence to population maintenance and bloom dynamics. J. Mar. Sci. Eng. 2021, 9, 51. [Google Scholar] [CrossRef]
- Place, A.R.; Bowers, H.A.; Bachvaroff, T.R.; Adolf, J.E.; Deeds, J.R.; Sheng, J. Karlodinium veneficum—The little dinoflagellate with a big bite. Harmful Algae 2012, 14, 179–195. [Google Scholar] [CrossRef]
- Lim, H.C.; Leaw, C.P.; Tan, T.H.; Kon, N.F.; Yek, L.H.; Hii, K.S.; Teng, S.T.; Razali, R.M.; Usup, G.; Iwataki, M.; et al. A bloom of Karlodinium australe (Gymnodiniales, Dinophyceae) associated with mass mortality of cage-cultured fishes in West Johor Strait, Malaysia. Harmful Algae 2014, 40, 51–62. [Google Scholar] [CrossRef]
- Song, X.; Hu, Z.; Shang, L.; Leaw, C.P.; Lim, P.T.; Tang, Y.Z. Contact micropredation may play a more important role than exotoxicity does in the lethal effects of Karlodinium australe blooms: Evidence from laboratory bioassays. Harmful Algae 2020, 99, 101926. [Google Scholar] [CrossRef]
- Yang, H.; Hu, Z.; Shang, L.; Deng, Y.; Tang, Y.Z. A strain of the toxic dinoflagellate Karlodinium veneficum isolated from the East China Sea is an omnivorous phagotroph. Harmful Algae 2020, 93, 101775. [Google Scholar] [CrossRef]
- Cen, J.; Wang, J.; Huang, L.; Ding, G.; Qi, Y.; Cao, R.; Cui, L.; Lü, S. Who is the “murderer” of the bloom in coastal waters of Fujian, China, in 2019? J. Oceanol. Limnol. 2020, 38, 722–732. [Google Scholar] [CrossRef]
- Luo, Z.; Wang, L.; Chan, L.; Lu, S.; Gu, H. Karlodinium zhouanum, a new dinoflagellate species from China, and molecular phylogeny of Karenia digitata and Karenia longicanalis (Gymnodiniales, Dinophyceae). Phycologia 2018, 57, 401–412. [Google Scholar] [CrossRef]
- Li, Z.; Shin, H.H. Morphology and phylogeny of an unarmored dinoflagellate, Karlodinium jejuense sp. nov. (Gymnodiniales), isolated from the northern East China Sea. Phycol. Res. 2018, 66, 318–328. [Google Scholar] [CrossRef]
- Cen, J.; Wang, J.; Huang, L.; Lin, Y.; Ding, G.; Qi, Y.; Lü, S. Karlodinium elegans sp. nov. (Gymnodiniales, Dinophyceae), a novel species isolated from the East China Sea in a dinoflagellate bloom. J. Oceanol. Limnol. 2021, 39, 242–258. [Google Scholar] [CrossRef]
- Benico, G.; Takahashi, K.; Lum, W.M.; Yñiguez, A.T.; Iwataki, M. The harmful unarmored dinoflagellate Karlodinium in Japan and Philippines, with reference to ultrastructure and micropredation of Karlodinium azanzae sp. nov. (Kareniaceae, Dinophyceae). J. Phycol. 2020, 56, 1264–1282. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Sun, X.; Feng, J.; Yan, X. Microscopic observations and molecular identification of toxic unarmoured dinoflagellates Karlodinium micrum (Dinophyceae) from the East China Sea (ECS). Mar. Sci. Bull. 2008, 27, 32–37. [Google Scholar]
- Liu, Y.; Hu, Z.; Deng, Y.; Tang, Y.Z. Evidence for resting cyst production in the cosmopolitan toxic dinoflagellate Karlodinium veneficum and the cyst distribution in the China seas. Harmful Algae 2020, 93, 101788. [Google Scholar] [CrossRef] [PubMed]
- Guillard, R.R.L. Culture of Phytoplankton for Feeding Marine Invertebrates. In Culture of Marine Invertebrate Animals: Proceedings—1st Conference on Culture of Marine Invertebrate Animals Greenport; Smith, W.L., Chanley, M.H., Eds.; Springer: Boston, MA, USA, 1975; pp. 29–60. [Google Scholar]
- Scholin, C.A.; Herzog, M.; Sogin, M.; Anderson, D.M. Identification of group- and strain-specific genetic markers for globally distributed Alexandrium (Dinophyceae). II. Sequence analysis of a fragment of the LSU rRNA gene. J. Phycol. 1994, 30, 999–1011. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Medlin, L.K.; Elwood, H.J.; Stickel, S.K.; Sogin, M.L. The characterization of enzymatically amplified eukaryotic 16S-like rRNA-coding regions. Gene 1988, 71, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Misawa, K.; Kuma, K.i.; Miyata, T. MAFFT: A novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 2002, 30, 3059–3066. [Google Scholar] [CrossRef]
- Nakamura, T.; Yamada, K.D.; Tomii, K.; Katoh, K. Parallelization of MAFFT for large-scale multiple sequence alignments. Bioinformatics 2018, 34, 2490–2492. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Posada, D. jModelTest: Phylogenetic Model Averaging. Mol. Biol. Evol. 2008, 25, 1253–1256. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Huelsenbeck, J.P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar] [CrossRef]
- Silvestro, D.; Michalak, I. raxmlGUI: A graphical front-end for RAxML. Org. Divers. Evol. 2012, 12, 335–337. [Google Scholar] [CrossRef]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef]
- Edler, D.; Klein, J.; Antonelli, A.; Silvestro, D. raxmlGUI 2.0: A graphical interface and toolkit for phylogenetic analyses using RAxML. Methods Ecol. Evol. 2021, 12, 373–377. [Google Scholar] [CrossRef]
- Zapata, M.; Rodríguez, F.; Garrido, J.L. Separation of chlorophylls and carotenoids from marine phytoplankton: A new HPLC method using a reversed phase C8 column and pyridine-containing mobile phases. Mar. Ecol. Prog. Ser. 2000, 195, 29–45. [Google Scholar] [CrossRef]
- Wang, J.; Kong, F.; Geng, H.; Zhao, Y.; Guan, W.; He, C.; Kang, Z.; Guo, W.; Zhou, Z.; Zhang, Q.; et al. Pigment characterization of the giant-colony-forming Haptophyte Phaeocystis globosa in the Beibu Gulf reveals blooms of different origins. Appl. Environ. Microbiol. 2022, 88, e01654-21. [Google Scholar] [CrossRef]
- Kong, F.; Yu, R.; Zhang, Q.; Yan, T.; Zhou, M. Pigment characterization for the 2011 bloom in Qinhuangdao implicated “brown tide” events in China. Chin. J. Oceanol. Limnol. 2012, 30, 361–370. [Google Scholar] [CrossRef]
- de Salas, M.F.; Laza-Martínez, A.; Hallegraeff, G.M. Novel unarmored dinoflagellates from the toxigenic family Kareniaceae (Gymnodiniales): Five new species of Karlodinium and one new Takayama from the australian sector of the Southern Ocean. J. Phycol. 2008, 44, 241–257. [Google Scholar] [CrossRef]
- Ballantine, D. Two new marine species of Gymnodinium isolated from the Plymouth area. J. Mar. Biol. Assoc. UK 1956, 35, 467–474. [Google Scholar] [CrossRef]
- de Salas, M.F.; Bolch, C.J.S.; Hallegraeff, G.M. Karlodinium australe sp. nov. (Gymnodiniales, Dinophyceae), a new potentially ichthyotoxic unarmoured dinoflagellate from lagoonal habitats of south-eastern Australia. Phycologia 2005, 44, 640–650. [Google Scholar] [CrossRef]
- Siano, R.; Kooistra, W.H.C.F.; Montresor, M.; Zingone, A. Unarmoured and thin-walled dinoflagellates from the Gulf of Naples, with the description of Woloszynskia cincta sp. nov. (Dinophyceae, Suessiales). Phycologia 2009, 48, 44–65. [Google Scholar] [CrossRef]
- Nézan, E.; Siano, R.; Boulben, S.; Six, C.; Bilien, G.; Chèze, K.; Duval, A.; Le Panse, S.; Quéré, J.; Chomérat, N. Genetic diversity of the harmful family Kareniaceae (Gymnodiniales, Dinophyceae) in France, with the description of Karlodinium gentienii sp. nov.: A new potentially toxic dinoflagellate. Harmful Algae 2014, 40, 75–91. [Google Scholar] [CrossRef]
- Gu, H.; Wu, Y.; Lü, S.; Lu, D.; Tang, Y.Z.; Qi, Y. Emerging harmful algal bloom species over the last four decades in China. Harmful Algae 2021, 102059. [Google Scholar] [CrossRef]
- Pan, Y.; Meng, R.; Li, Y.; Yang, L.; Mei, L.; Wu, Y.; Xu, J.; Zhou, C.; Yan, X. Changes in biochemical metabolites in manila clam after a temporary culture with high-quality microalgal feed mixed with the dinoflagellate species Karlodinium veneficum and K. zhouanum. Harmful Algae 2023, 125, 102422. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).