The Effects of Termites and Beetle Larvae on the Decomposition of Pinus densiflora and Quercus acutissima in South Korea over a 6-Year Period
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Data Analysis
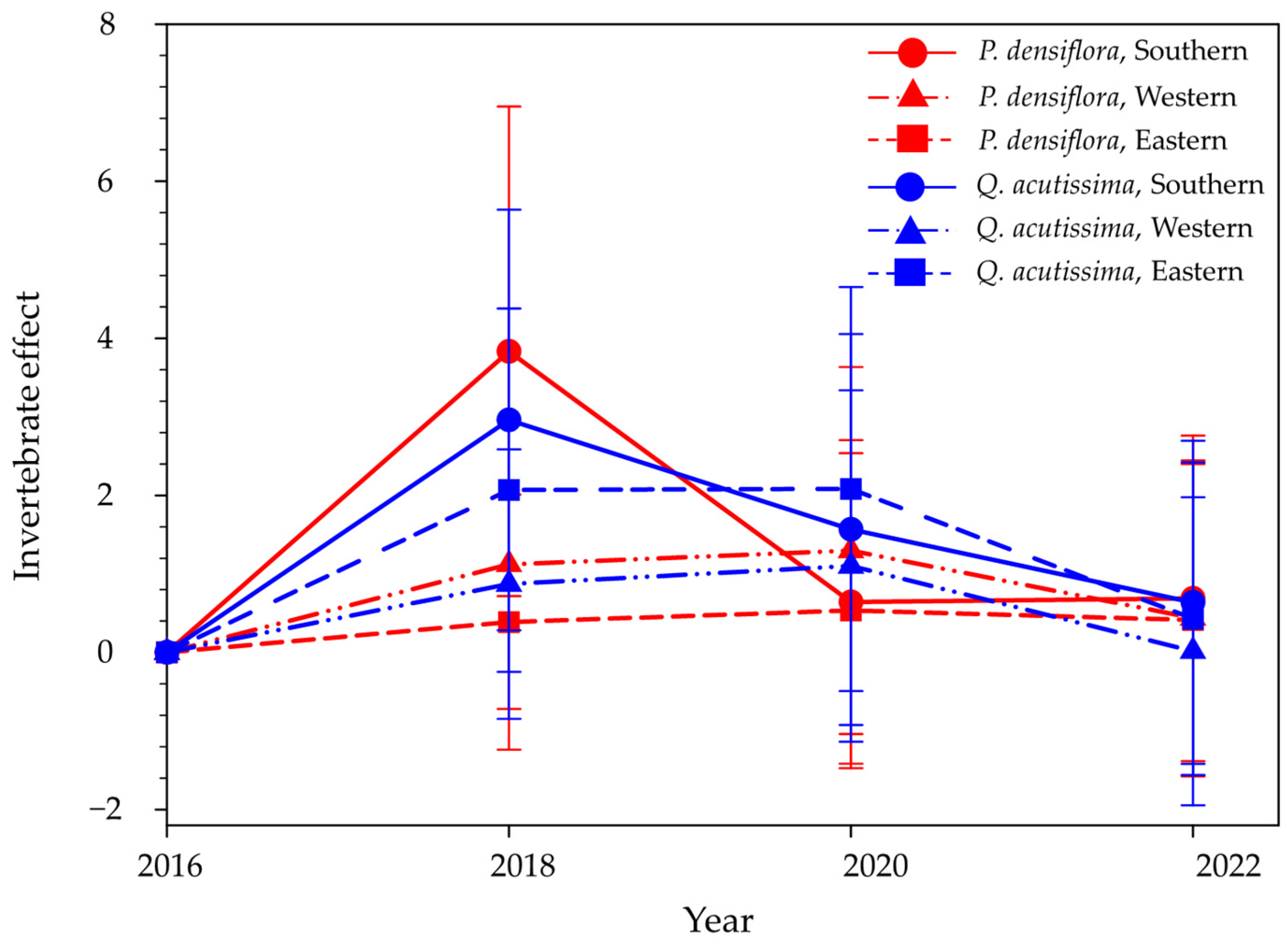
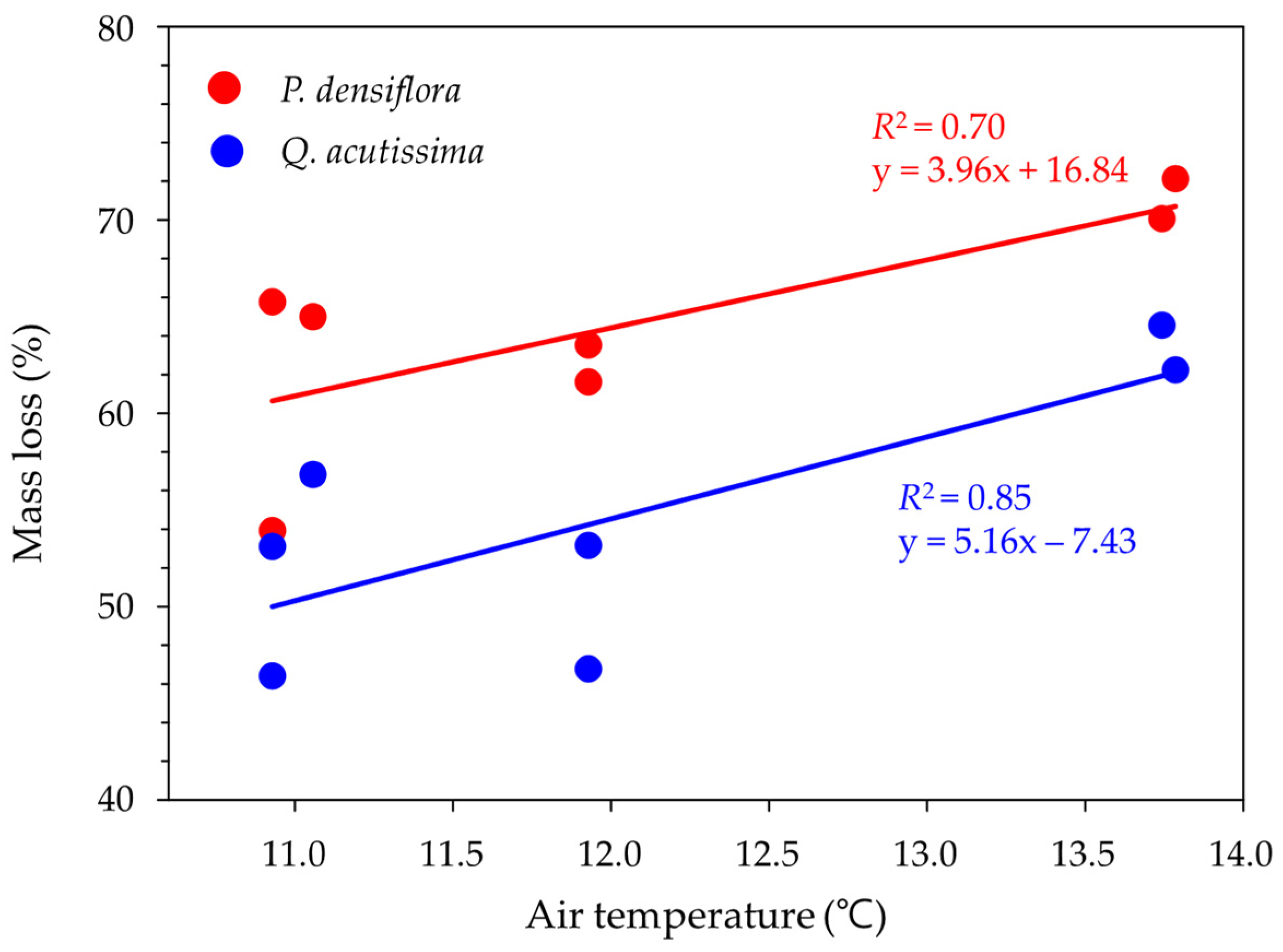
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cornwell, W.K.; Cornelissen, J.H.; Allison, S.D.; Bauhus, J.; Eggleton, P.; Preston, C.M.; Scarff, F.; Weedon, J.T.; Wirth, C.; Zanne, A.E. Plant traits and wood fates across the globe: Rotted, burned, or consumed? Glob. Chang. Biol. 2009, 15, 2431–2449. [Google Scholar] [CrossRef]
- Takamura, K. Effects of termite exclusion on decay of heavy and light hardwood in a tropical rain forest of Peninsular Malaysia. J. Trop. Ecol. 2001, 17, 541–548. [Google Scholar] [CrossRef]
- Peralta, R.C.G.; Menezes, E.B.; Carvalho, A.G.; Aguiar-Menezes, E.D.L. Wood consumption rates of forest species by subterranean termites (Isoptera) under field conditions. Rev. Árvore 2004, 28, 283–289. [Google Scholar] [CrossRef]
- Weedon, J.T.; Cornwell, W.K.; Cornelissen, J.H.; Zanne, A.E.; Wirth, C.; Coomes, D.A. Global meta-analysis of wood decomposition rates: A role for trait variation among tree species? Ecol. Lett. 2009, 12, 45–56. [Google Scholar] [CrossRef]
- Seibold, S.; Rammer, W.; Hothorn, T.; Seidl, R.; Ulyshen, M.D.; Lorz, J.; Cadotte, M.W.; Lindenmayer, D.B.; Adhikari, Y.P.; Aragón, R.; et al. The contribution of insects to global forest deadwood decomposition. Nature 2021, 597, 77–81. [Google Scholar] [CrossRef]
- Gossner, M.M.; Wende, B.; Levick, S.; Schall, P.; Floren, A.; Linsenmair, K.E.; Steffan-Dewenter, I.; Schulze, E.D.; Weisser, W.W. Deadwood enrichment in European forests–Which tree species should be used to promote saproxylic beetle diversity? Biol. Conserv. 2016, 201, 92–102. [Google Scholar] [CrossRef]
- Horák, J. Niche partitioning among dead wood-dependent beetles. Sci. Rep. 2021, 11, 15178. [Google Scholar] [CrossRef]
- Milberg, P.; Bergman, K.O.; Johansson, H.; Jansson, N. Low host-tree preferences among saproxylic beetles: A comparison of four deciduous species. Insect Conserv. Diver. 2014, 7, 508–522. [Google Scholar] [CrossRef]
- Ulyshen, M.D.; Wagner, T.L. Quantifying arthropod contributions to wood decay. Methods Ecol. Evol. 2013, 4, 345–352. [Google Scholar] [CrossRef]
- Kim, S.H.; Chung, Y.J. Analysis of factors affecting termite damage to wooden architectural heritage buildings in Korea. Forests 2022, 13, 465. [Google Scholar] [CrossRef]
- Lee, H.B.; Seo, M.S.; Lee, S.B.; Lee, W. New distribution of Reticulitermes speratus speratus (Blattodea: Rhinotermitidae) in Korea. J. Econ. Entomol. 2023, 116, 2027–2034. [Google Scholar] [CrossRef]
- Kwon, T.S.; Lee, C.M.; Kim, S.S. Prediction of abundance of beetles according to climate warming in South Korea. J. Asia-Pac. Biodivers. 2015, 8, 7–30. [Google Scholar] [CrossRef]
- Ulyshen, M.D. Wood decomposition as influenced by invertebrates. Biol. Rev. 2016, 91, 70–85. [Google Scholar] [CrossRef]
- Noh, N.J.; Yoon, T.K.; Kim, R.H.; Bolton, N.W.; Kim, C.; Son, Y. Carbon and nitrogen accumulation and decomposition from coarse woody debris in a naturally regenerated Korean red pine (Pinus densiflora S. et Z.) forest. Forests 2017, 8, 214. [Google Scholar] [CrossRef]
- Warren, R.J.; Bradford, M.A. Ant colonization and coarse woody debris decomposition in temperate forests. Insectes Soc. 2012, 59, 215–221. [Google Scholar] [CrossRef]
- Ulyshen, M.D. Interacting effects of insects and flooding on wood decomposition. PLoS ONE 2014, 9, e101867. [Google Scholar] [CrossRef]
- Zuo, J.; Fonck, M.; van Hal, J.; Cornelissen, J.H.C.; Berg, M.P. Diversity of macro-detritivores in dead wood is influenced by tree species, decay stage and environment. Soil Biol. Biochem. 2014, 78, 288–297. [Google Scholar] [CrossRef]
- Parisi, F.; Pioli, S.; Lombardi, F.; Fravolini, G.; Marchetti, M.; Tognetti, R. Linking deadwood traits with saproxylic invertebrates and fungi in European forests—A review. iForest 2018, 11, 423–436. [Google Scholar] [CrossRef]
- Kim, S.; Han, S.H.; Li, G.; Roh, Y.; Kim, H.J.; Son, Y. The initial effects of microclimate and invertebrate exclusion on multi-site variation in the mass loss of temperate pine and oak deadwoods. Sci. Rep. 2021, 11, 14840. [Google Scholar] [CrossRef]
- Tuma, J.; Eggleton, P.; Fayle, T.M. Ant-termite interactions: An important but under-explored ecological linkage. Biol. Rev. 2020, 95, 555–572. [Google Scholar] [CrossRef]
- Wong, N.; Lee, C.-Y. Effects of disturbance and the presence of termite and other invertebrate carcasses at feeding sites on the behavior of the subterranean termite Microceratermes crassus (Blattodea: Termitidae). Sociobiology 2010, 55, 353–367. [Google Scholar]
- Viana-Junior, A.B.; Côrtes, M.O.; Cornelissen, T.G.; Neves, F.D.S. Interactions between wood-inhabiting fungi and termites: A meta-analytical review. Arth.-Plant Int. 2018, 12, 229–235. [Google Scholar] [CrossRef]
- Bani, A.; Pioli, S.; Ventura, M.; Panzacchi, P.; Borruso, L.; Tognetti, R.; Tonon, G.; Brusetti, L. The role of microbial community in the decomposition of leaf litter and deadwood. Appl. Soil Ecol. 2018, 126, 75–84. [Google Scholar] [CrossRef]
- Jacobsen, R.M.; Kauserud, H.; Sverdrup-Thygeson, A.; Bjorbækmo, M.M.; Birkemoe, T. Wood-inhabiting insects can function as targeted vectors for decomposer fungi. Fungal Ecol. 2017, 29, 76–84. [Google Scholar] [CrossRef]
- Gautam, B.K.; Henderson, G. Wood consumption by Formosan subterranean termites (Isoptera: Rhinotermitidae) as affected by wood moisture content and temperature. Ann. Entomol. Soc. Am. 2011, 104, 459–464. [Google Scholar] [CrossRef]
- Cao, R.; Su, N.Y. Temperature preferences of four subterranean termite species (Isoptera: Rhinotermitidae) and temperature-dependent survivorship and wood-consumption rate. Ann. Entomol. Soc. Am. 2016, 109, 64–71. [Google Scholar] [CrossRef]
- Zanne, A.E.; Flores-Moreno, H.; Powell, J.R.; Cornwell, W.K.; Dalling, J.W.; Austin, A.T.; Classen, A.T.; Eggleton, P.; Okada, K.I.; Parr, C.L.; et al. Termite sensitivity to temperature affects global wood decay rates. Science 2022, 377, 1440–1444. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.A.; Warren II, R.J.; Baldrian, P.; Crowther, T.W.; Maynard, D.S.; Oldfield, E.E.; Wieder, W.R.; Wood, S.A.; King, J.R. Climate fails to predict wood decomposition at regional scales. Nat. Clim. Chang. 2014, 4, 625–630. [Google Scholar] [CrossRef]
- Palviainen, M.; Laiho, R.; Mäkinen, H.; Finer, L. Do decomposing Scots pine, Norway spruce, and silver birch stems retain nitrogen? Can. J. For. Res. 2008, 38, 3047–3055. [Google Scholar] [CrossRef]
- Köster, K.; Metslaid, M.; Engelhart, J.; Köster, E. Dead wood basic density, and the concentration of carbon and nitrogen for main tree species in managed hemiboreal forests. For. Ecol. Manag. 2015, 354, 35–42. [Google Scholar] [CrossRef]
- Purahong, W.; Wubet, T.; Lentendu, G.; Hoppe, B.; Jariyavidyanont, K.; Arnstadt, T.; Baber, K.; Otto, P.; Kellner, H.; Hofrichter, M.; et al. Determinants of deadwood-inhabiting fungal communities in temperate forests: Molecular evidence from a large scale deadwood decomposition experiment. Front. Microbiol. 2018, 9, 2120. [Google Scholar] [CrossRef] [PubMed]





| Region | Site | Coordinate | Air Temperature (°C) 1 | Precipitation (mm) 1 | Humidity (%) 1 | Altitude (m) 2 | Tree Density (Trees ha−1) 2 | Dominant Tree Species 2 |
|---|---|---|---|---|---|---|---|---|
| Southern | site 1 | 35°21′ N, 128°10′ E | 13.79 ± 0.56 | 1274.63 ± 18.09 | 69.53 ± 1.78 | 430 | 1200 | Pinus densiflora, Quercus variabilis, Acer pictum, Toxicodendron vernicifluum |
| site 2 | 35°12′ N, 128°10′ E | 13.74 ± 0.47 | 1394.60 ± 19.04 | 67.54 ± 1.91 | 180 | 1560 | P. densiflora, Q. dentata, Q. acutissima, T. vernicifluum | |
| Western | site 3 | 37°47′ N, 127°10′ E | 11.93 ± 0.57 | 1315.01 ± 18.55 | 66.83 ± 2.31 | 470 | 650 | P. densiflora, Q. aliena, Q. variabilis, A. pictum |
| site 4 | 37°46′ N, 127°10′ E | 11.93 ± 0.57 | 1315.01 ± 18.55 | 66.83 ± 2.31 | 470 | 1140 | P. densiflora, Q. mongolica. Carpinus cordata | |
| Eastern | site 5 | 37°30′ N, 128°56′ E | 11.06 ± 0.66 | 1165.23 ± 14.53 | 66.21 ± 1.33 | 690 | 610 | P. densiflora, Q. mongolica. C. Cordata |
| site 6 | 38°02′ N, 128°22′ E | 10.93 ± 0.66 | 1176.89 ± 15.38 | 66.81 ± 1.62 | 610 | 1050 | P. densiflora, Q. mongolica, Q. serrata, A. pseudosieboldianum | |
| site 7 | 38°03′ N, 128°22′ E | 10.93 ± 0.66 | 1176.89 ± 15.38 | 66.81 ± 1.62 | 590 | 900 | P. densiflora, Q. mongolica, Q. serrata, A. pseudosieboldianum |
| Species | Invertebrate Exclusion | Region | The Number of Samples Analyzed (n) | ||
|---|---|---|---|---|---|
| Mass Loss | C and N Concentrations | Subtotal | |||
| P. densiflora | Control 1 | Southern | 25 | 16 | 41 |
| Western | 72 | 22 | 94 | ||
| Eastern | 88 | 19 | 107 | ||
| Treatment 2 | Southern | 52 | 13 | 65 | |
| Western | 72 | 18 | 90 | ||
| Eastern | 70 | 21 | 91 | ||
| Q. acutissima | Control | Southern | 47 | 16 | 63 |
| Western | 55 | 16 | 71 | ||
| Eastern | 73 | 22 | 95 | ||
| Treatment | Southern | 55 | 20 | 75 | |
| Western | 59 | 20 | 79 | ||
| Eastern | 72 | 21 | 93 | ||
| Total | 740 | 224 | 964 | ||
| Species | Region | Site | Year | ||
|---|---|---|---|---|---|
| 2018 | 2020 | 2022 | |||
| P. densiflora | Southern | Site 1 | 3 (termite) | 0 | 0 |
| Site 2 | 6 (termite) | 0 | 0 | ||
| Eastern | Site 3 | 0 | 0 | 3 (ant) | |
| Site 4 | 0 | 0 | 0 | ||
| Site 5 | 0 | 0 | 0 | ||
| Western | Site 6 | 0 | 0 | 0 | |
| Site 7 | 1 (termite) | 0 | 0 | ||
| Q. acutissima | Southern | Site 1 | 3 (termite) | 0 | 0 |
| Site 2 | 3 (termite) | 0 | 1 (ant) | ||
| Eastern | Site 3 | 0 | 0 | 2 (ant) | |
| Site 4 | 0 | 0 | 2 (ant) | ||
| Site 5 | 0 | 0 | 0 | ||
| Western | Site 6 | 0 | 0 | 1 (ant) | |
| Site 7 | 0 | 0 | 0 | ||
| Effect | DF | F | p |
|---|---|---|---|
| Species (S) | 1 | 619.63 | *** |
| Exclusion (E) | 1 | 381.98 | *** |
| Region (R) | 2 | 120.84 | *** |
| S × E | 1 | 4.48 | * |
| S × R | 2 | 23.56 | *** |
| E × R | 2 | 1.67 | NS |
| S × E × R | 2 | 15.69 | *** |
| Time (T) | 3 | 2,222,563.10 | *** |
| T × S | 3 | 17,585.35 | *** |
| T × E | 3 | 14,236.55 | *** |
| T× R | 6 | 11,818.37 | *** |
| T × S × E | 3 | 1102.52 | *** |
| T × S × R | 6 | 1186.93 | *** |
| T × E × R | 6 | 764.54 | *** |
| T × S × E × R | 6 | 1625.09 | *** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roh, D.; Kim, S.; Kim, H.-S.; Han, S.H.; Kim, G.; Son, Y. The Effects of Termites and Beetle Larvae on the Decomposition of Pinus densiflora and Quercus acutissima in South Korea over a 6-Year Period. Diversity 2024, 16, 452. https://doi.org/10.3390/d16080452
Roh D, Kim S, Kim H-S, Han SH, Kim G, Son Y. The Effects of Termites and Beetle Larvae on the Decomposition of Pinus densiflora and Quercus acutissima in South Korea over a 6-Year Period. Diversity. 2024; 16(8):452. https://doi.org/10.3390/d16080452
Chicago/Turabian StyleRoh, Dakyum, Seongjun Kim, Hyung-Sub Kim, Seung Hyun Han, Gaeun Kim, and Yowhan Son. 2024. "The Effects of Termites and Beetle Larvae on the Decomposition of Pinus densiflora and Quercus acutissima in South Korea over a 6-Year Period" Diversity 16, no. 8: 452. https://doi.org/10.3390/d16080452





