Sex Ratio Distortion of Aedes aegypti (L.) in El Salvador: Biocontrol Implications for Seasonally Dry Urban Neotropical Environments
Abstract
1. Introduction
2. Materials and Methods
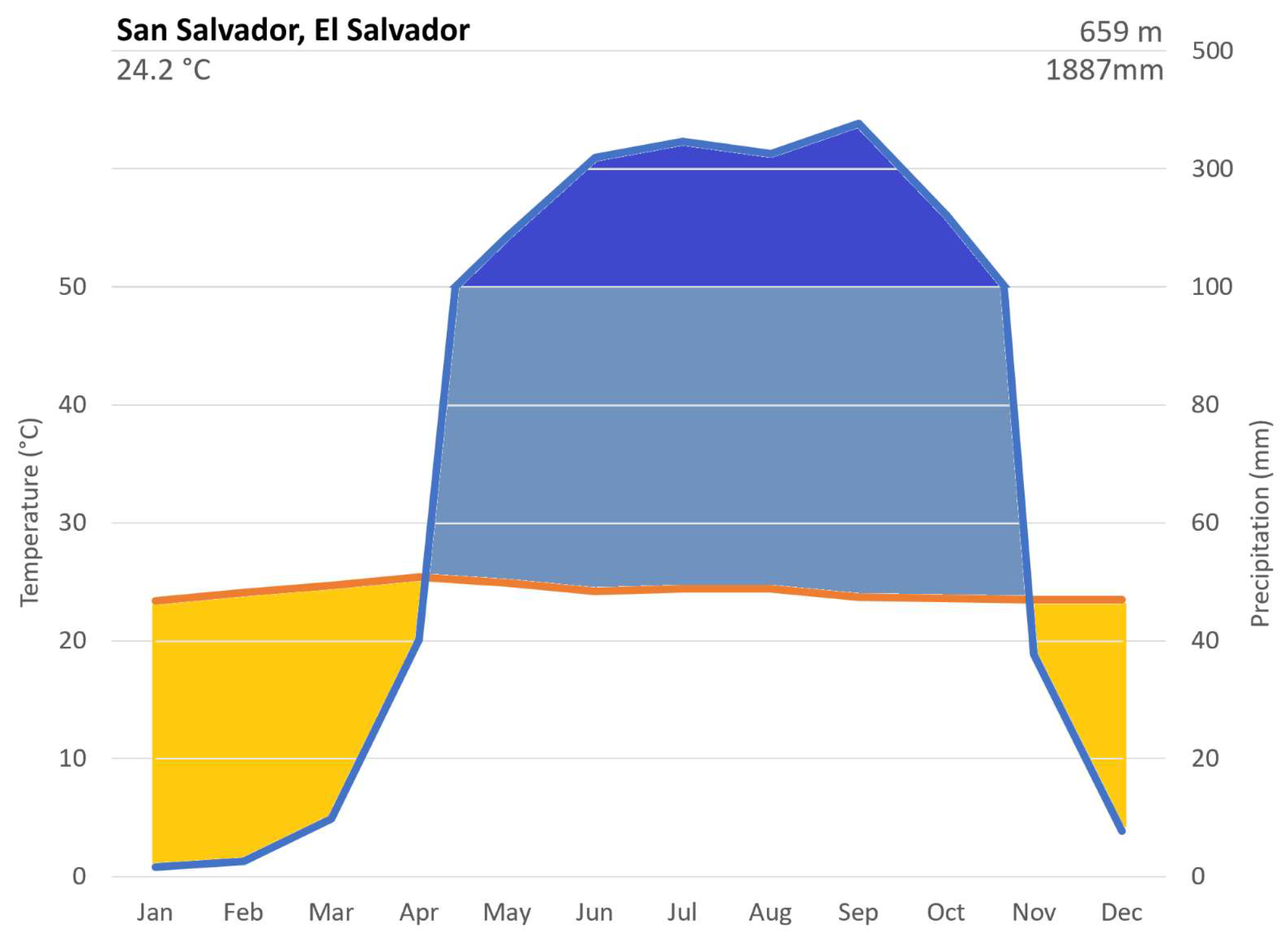
2.1. Climate Data
2.2. Site Description
2.3. Collection of Mosquito Larvae
2.4. Determination of Sex
2.5. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Vector-Borne Diseases. Available online: https://www.who.int/news-room/fact-sheets/detail/vector-borne-diseases (accessed on 28 November 2023).
- Brown, J.E.; McBride, C.S.; Johnson, P.; Ritchie, S.; Paupy, C.; Bossin, H.; Lutomiah, J.; Fernandez-Salas, I.; Ponlawat, A.; Cornel, A.J.; et al. Worldwide patterns of genetic differentiation imply multiple ‘domestications’ of Aedes aegypti, a major vector of human diseases. Proc. Biol. Sci. 2011, 278, 2446–2454. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.E.; Evans, B.R.; Zheng, W.; Obas, V.; Barrera-Martinez, L.; Egizi, A.; Zhao, H.; Caccone, A.; Powell, J.R. Human impacts have shaped historical and recent evolution in Aedes aegypti, the dengue and yellow fever mosquito. Evolution 2014, 68, 514–525. [Google Scholar] [CrossRef] [PubMed]
- Facchinelli, L.; Badolo, A.; McCall, P.J. Biology and Behaviour of Aedes aegypti in the Human Environment: Opportunities for Vector Control of Arbovirus Transmission. Viruses 2023, 15, 636. [Google Scholar] [CrossRef] [PubMed]
- Guzman, M.G.; Gubler, D.J.; Izquierdo, A.; Martinez, E.; Halstead, S.B. Dengue infection. Nat. Rev. Dis. Primers 2016, 2, 16055. [Google Scholar] [CrossRef]
- Wilson, A.L.; Courtenay, O.; Kelly-Hope, L.A.; Scott, T.W.; Takken, W.; Torr, S.J.; Lindsay, S.W. The importance of vector control for the control and elimination of vector-borne diseases. PLoS Negl. Trop. Dis. 2020, 14, e0007831. [Google Scholar] [CrossRef]
- Gubler, D.J.; Ooi, E.E.; Vasudevan, S.; Farrar, J. Dengue and Dengue Hemorrhagic Fever, 2nd ed.; CABI: Wallingford, UK, 2014. [Google Scholar]
- Pimenta, P.F.; Touray, M.; Miller, L. The journey of malaria sporozoites in the mosquito salivary gland. J. Eukaryot. Microbiol. 1994, 41, 608–624. [Google Scholar] [CrossRef]
- Ngugi, H.N.; Mutuku, F.M.; Ndenga, B.A.; Musunzaji, P.S.; Mbakaya, J.O.; Aswani, P.; Irungu, L.W.; Mukoko, D.; Vulule, J.; Kitron, U.; et al. Characterization and productivity profiles of Aedes aegypti (L.) breeding habitats across rural and urban landscapes in western and coastal Kenya. Parasites Vectors 2017, 10, 331. [Google Scholar] [CrossRef]
- Wilke, A.B.B.; Vasquez, C.; Carvajal, A.; Moreno, M.; Fuller, D.O.; Cardenas, G.; Petrie, W.D.; Beier, J.C. Urbanization favors the proliferation of Aedes aegypti and Culex quinquefasciatus in urban areas of Miami-Dade County, Florida. Sci. Rep. 2021, 11, 22989. [Google Scholar] [CrossRef]
- Powell, J.R.; Tabachnick, W.J. History of domestication and spread of Aedes aegypti—A review. Mem. Inst. Oswaldo Cruz 2013, 108, 11–17. [Google Scholar] [CrossRef]
- Slosek, J. Aedes aegypti mosquitoes in the Americas: A review of their interactions with the human population. Soc. Sci. Med. 1986, 23, 249–257. [Google Scholar] [CrossRef]
- Joyce, A.L.; Torres, M.M.; Torres, R.; Moreno, M. Genetic variability of the Aedes aegypti (Diptera: Culicidae) mosquito in El Salvador, vector of dengue, yellow fever, chikungunya and Zika. Parasites Vectors 2018, 11, 1–14. [Google Scholar]
- Ocampo, C.B.; Wesson, D.M. Population dynamics of Aedes aegypti from a dengue hyperendemic urban setting in Colombia. Am. J. Trop. Med. Hyg. 2004, 71, 506–513. [Google Scholar] [PubMed]
- Charnov, E.L. The Theory of Sex Allocation; Princeton University Press: Princeton, NJ, USA, 1982. [Google Scholar]
- Fisher, R.A. The Genetical Theory of Natural Selection: A Complete Variorum Edition; Oxford University Press: Oxford, UK, 1999. [Google Scholar]
- Edwards, A.W.F. Natural Selection and the Sex Ratio: Fisher’s Sources. Am. Nat. 1998, 151, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Barrett, S.C.; Yakimowski, S.B.; Field, D.L.; Pickup, M. Ecological genetics of sex ratios in plant populations. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 2549–2557. [Google Scholar] [CrossRef]
- Vergés, A.; Paul, N.A.; Steinberg, P.D. Sex and life-history stage alter herbivore responses to a chemically defended red alga. Ecology 2008, 89, 1334–1343. [Google Scholar] [CrossRef]
- Hamilton, W.D. Extraordinary sex ratios. A sex-ratio theory for sex linkage and inbreeding has new implications in cytogenetics and entomology. Science 1967, 156, 477–488. [Google Scholar] [CrossRef]
- Hawley, W. Population Dynamics of Aedes sierrensis. In Ecology of Mosquitoes: Proceedings of a Workshop; Lounibos, L.P., Rey, J.R., Frank, J.H., Eds.; Florida Medical Entomology Laboratory: Vero Beach, FL, USA, 1985; pp. 167–184. [Google Scholar]
- Lounibos, L.P.; Escher, R.L. Sex ratios of mosquitoes from long-term censuses of Florida tree holes. J. Am. Mosq. Control Assoc. 2008, 24, 11–15. [Google Scholar] [CrossRef]
- Unlu, I.; Farajollahi, A.; Rochlin, I.; Crepeau, T.N.; Strickman, D.; Gaugler, R. Differences in male-female ratios of Aedes albopictus (Diptera: Culicidae) following ultra-low volume adulticide applications. Acta Trop. 2014, 137, 201–205. [Google Scholar] [CrossRef]
- Yates, M. The biology of the tree-hole breeding mosquito Aedes geniculatus (Olivier)(Diptera: Culicidae) in southern England. Bull. Entomol. Res. 1979, 69, 611–628. [Google Scholar]
- Groat-Carmona, A.M.; Orozco, S.; Friebe, P.; Payne, A.; Kramer, L.; Harris, E. A novel coding-region RNA element modulates infectious dengue virus particle production in both mammalian and mosquito cells and regulates viral replication in Aedes aegypti mosquitoes. Virology 2012, 432, 511–526. [Google Scholar] [CrossRef]
- Ross, P.A.; Callahan, A.G.; Yang, Q.; Jasper, M.; Arif, M.A.K.; Afizah, A.N.; Nazni, W.A.; Hoffmann, A.A. An elusive endosymbiont: Does Wolbachia occur naturally in Aedes aegypti? Ecol. Evol. 2020, 10, 1581–1591. [Google Scholar] [CrossRef] [PubMed]
- Simberloff, D. Risks of biological control for conservation purposes. BioControl 2012, 57, 263–276. [Google Scholar] [CrossRef]
- Werren, J.H.; Baldo, L.; Clark, M.E. Wolbachia: Master manipulators of invertebrate biology. Nat. Rev. Microbiol. 2008, 6, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Jaenike, J. Sex Chromosome Meiotic Drive. Annu. Rev. Ecol. Evol. Syst. 2001, 32, 25–49. [Google Scholar] [CrossRef]
- Hurst, G.D.; Jiggins, F.M. Male-killing bacteria in insects: Mechanisms, incidence, and implications. Emerg. Infect. Dis. 2000, 6, 329–336. [Google Scholar] [CrossRef]
- Beck, H.E.; Zimmermann, N.E.; McVicar, T.R.; Vergopolan, N.; Berg, A.; Wood, E.F. Present and future Köppen-Geiger climate classification maps at 1-km resolution. Sci. Data 2018, 5, 180214. [Google Scholar] [CrossRef]
- Holdridge, L.R. Determination of World Plant Formations from Simple Climatic Data. Science 1947, 105, 367–368. [Google Scholar] [CrossRef]
- Bullock, S.H.; Mooney, H.A.; Medina, E. Seasonally Dry Tropical Forests; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Dexter, K.G.; Pennington, R.T.; Oliveira-Filho, A.T.; Bueno, M.L.; Silva de Miranda, P.L.; Neves, D.M. Inserting Tropical Dry Forests into the Discussion on Biome Transitions in the Tropics. Front. Ecol. Evol. 2018, 6, 104. [Google Scholar] [CrossRef]
- Pablo-Cea, J.D.; Velado-Cano, M.A.; Noriega, J.A. A first step to evaluate the impact of ecotourism on biodiversity in El Salvador: A case study using dung beetles in a National Park. J. Ecotour. 2021, 20, 51–69. [Google Scholar]
- Trewin, B.J.; Kay, B.H.; Darbro, J.M.; Hurst, T.P. Increased container-breeding mosquito risk owing to drought-induced changes in water harvesting and storage in Brisbane, Australia. Int. Health 2013, 5, 251–258. [Google Scholar] [CrossRef]
- Choi, J.; Carmona-Galindo, V.; Paredes, G.R.; Marín Recinos, M.F.; de Abrego, V.C. An exploration of land use and poverty as an integrative model for mitigating Chagas disease in El Salvador. BIOS 2020, 91, 125–132. [Google Scholar] [CrossRef]
- Karger, D.N.; Conrad, O.; Böhner, J.; Kawohl, T.; Kreft, H.; Soria-Auza, R.W.; Zimmermann, N.E.; Linder, H.P.; Kessler, M. Climatologies at high resolution for the earth’s land surface areas. Sci. Data 2017, 4, 170122. [Google Scholar] [CrossRef] [PubMed]
- Rueda, L.M. Pictorial keys for the identification of mosquitoes (Diptera: Culicidae) associated with dengue virus transmission. Zootaxa 2004, 589, 1–60. [Google Scholar]
- de Araújo, H.R.C.; Kojin, B.B.; Capurro, M.L. Sex determination and Aedes population control. Parasites Vectors 2018, 11, 644. [Google Scholar] [CrossRef]
- Shettima, A.; Joseph, S.; Ishak, I.H.; Abdul Raiz, S.H.; Abu Hasan, H.; Othman, N. Evaluation of Total Female and Male Aedes aegypti Proteomes Reveals Significant Predictive Protein–Protein Interactions, Functional Ontologies, and Differentially Abundant Proteins. Insects 2021, 12, 752. [Google Scholar] [CrossRef]
- Robinson, A.S. Sex-ratio manipulation in relation to insect pest control. Annu. Rev. Genet. 1983, 17, 191–214. [Google Scholar] [CrossRef]
- Benelli, G.; Jeffries, C.L.; Walker, T. Biological Control of Mosquito Vectors: Past, Present, and Future. Insects 2016, 7, 52. [Google Scholar] [CrossRef]
- Gubler, D.J. The partnership for dengue control—A new global alliance for the prevention and control of dengue. Vaccine 2015, 33, 1233. [Google Scholar] [CrossRef]
- Benedict, M.Q.; Robinson, A.S. The first releases of transgenic mosquitoes: An argument for the sterile insect technique. Trends Parasitol. 2003, 19, 349–355. [Google Scholar] [CrossRef]
- Hendrichs, J.; Robinson, A. Sterile insect technique. In Encyclopedia of Insects; Academic Press: Amsterdam, The Netherlands, 2009; pp. 953–957. [Google Scholar]
- Lance, D.; McInnis, D. Biological basis of the sterile insect technique. In Sterile Insect Technique; CRC Press: Boca Raton, FL, USA, 2021; pp. 113–142. [Google Scholar]
- Moreira, L.A.; Iturbe-Ormaetxe, I.; Jeffery, J.A.; Lu, G.; Pyke, A.T.; Hedges, L.M.; Rocha, B.C.; Hall-Mendelin, S.; Day, A.; Riegler, M. A Wolbachia symbiont in Aedes aegypti limits infection with dengue, Chikungunya, and Plasmodium. Cell 2009, 139, 1268–1278. [Google Scholar] [CrossRef]
- Walker, T.; Moreira, L.A. Can Wolbachia be used to control malaria? Mem. Inst. Oswaldo Cruz 2011, 106, 212–217. [Google Scholar] [PubMed]
- Zhang, D.; Lees, R.S.; Xi, Z.; Bourtzis, K.; Gilles, J.R. Combining the sterile insect technique with the incompatible insect technique: III-robust mating competitiveness of irradiated triple Wolbachia-infected Aedes albopictus males under semi-field conditions. PLoS ONE 2016, 11, e0151864. [Google Scholar]
- Garcia, G.d.A.; Sylvestre, G.; Aguiar, R.; da Costa, G.B.; Martins, A.J.; Lima, J.B.P.; Petersen, M.T.; Lourenço-de-Oliveira, R.; Shadbolt, M.F.; Rašić, G. Matching the genetics of released and local Aedes aegypti populations is critical to assure Wolbachia invasion. PLoS Negl. Trop. Dis. 2019, 13, e0007023. [Google Scholar]
- Lindholm, A.K.; Dyer, K.A.; Firman, R.C.; Fishman, L.; Forstmeier, W.; Holman, L.; Johannesson, H.; Knief, U.; Kokko, H.; Larracuente, A.M.; et al. The Ecology and Evolutionary Dynamics of Meiotic Drive. Trends Ecol. Evol. 2016, 31, 315–326. [Google Scholar] [CrossRef]
- Pen, I.; Weissing, F.J. Sex-ratio optimization with helpers at the nest. Proc. Biol. Sci. 2000, 267, 539–543. [Google Scholar] [CrossRef]
- Carmona-Galindo, V.; Carmona, T.; Carmona-Valdivieso, D. El papel del pez chimbolo (Poeciliopsis gracilis Heckel 1848) en el biocontrol de zancudos y la recuperación de servicios ecosistémicos en los ríos urbanos de El Salvador. El Salv. Cienc. Tecnol. 2018, 23, 23–27. [Google Scholar]
- Cea, M. Concluyen las liberaciones de zancudos con Wolbachia en Santa Ana. La Prensa Gráfica, 9 June 2024. [Google Scholar]
- Alemán, M. Decretan Alerta Roja por el Aumento de Casos de Dengue en El Salvador; Associated Press: New York, NY, USA, 2024. [Google Scholar]
- Padmanaban, D. Can mass-produced mosquitoes slow dengue’s spread? Harvard Public Health, 15 August 2023. [Google Scholar]
- Cea, M. Alabi: Liberaciones de zancudos con Wolbachia no tienen relación con alza de dengue. La Prensa Gráfica, 25 July 2024. [Google Scholar]
- Fernández, D.; Montañez, A.; Sarmanto, N. Diagnóstico de la Prestación de los Servicios de Agua Potable y Alcantarillado en El Salvador; Comisión Económica para América Latina y el Caribe (CEPAL): Santiago, Chile, 2023; 25p. [Google Scholar]
- Benelli, G. Managing mosquitoes and ticks in a rapidly changing world—Facts and trends. Saudi J. Biol. Sci. 2019, 26, 921–929. [Google Scholar] [CrossRef]
- Ferraguti, M.; Martínez-de La Puente, J.; Roiz, D.; Ruiz, S.; Soriguer, R.; Figuerola, J. Effects of landscape anthropization on mosquito community composition and abundance. Sci. Rep. 2016, 6, 29002. [Google Scholar]
- Kache, P.A.; Santos-Vega, M.; Stewart-Ibarra, A.M.; Cook, E.M.; Seto, K.C.; Diuk-Wasser, M.A. Bridging landscape ecology and urban science to respond to the rising threat of mosquito-borne diseases. Nat. Ecol. Evol. 2022, 6, 1601–1616. [Google Scholar] [CrossRef]


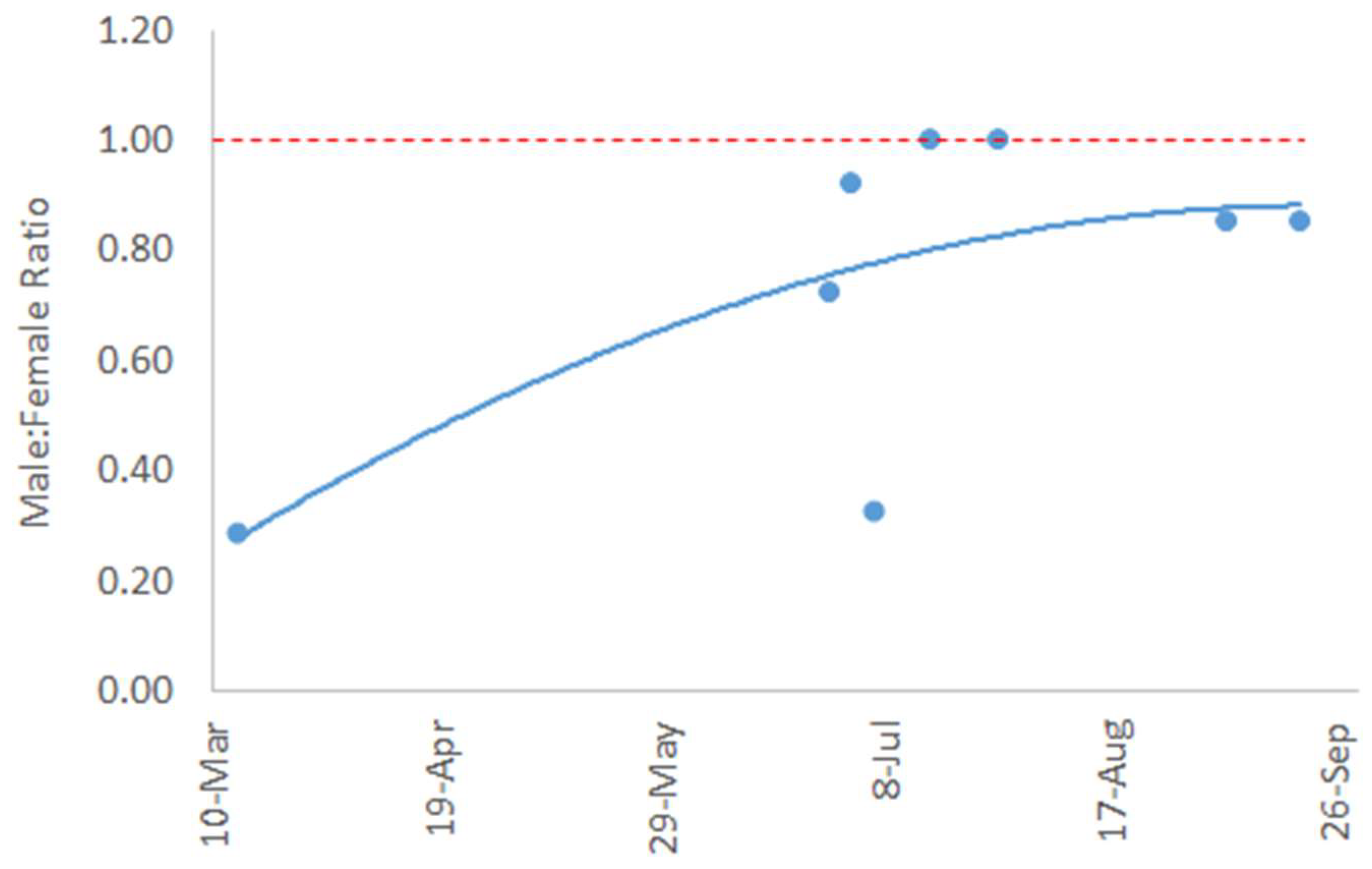
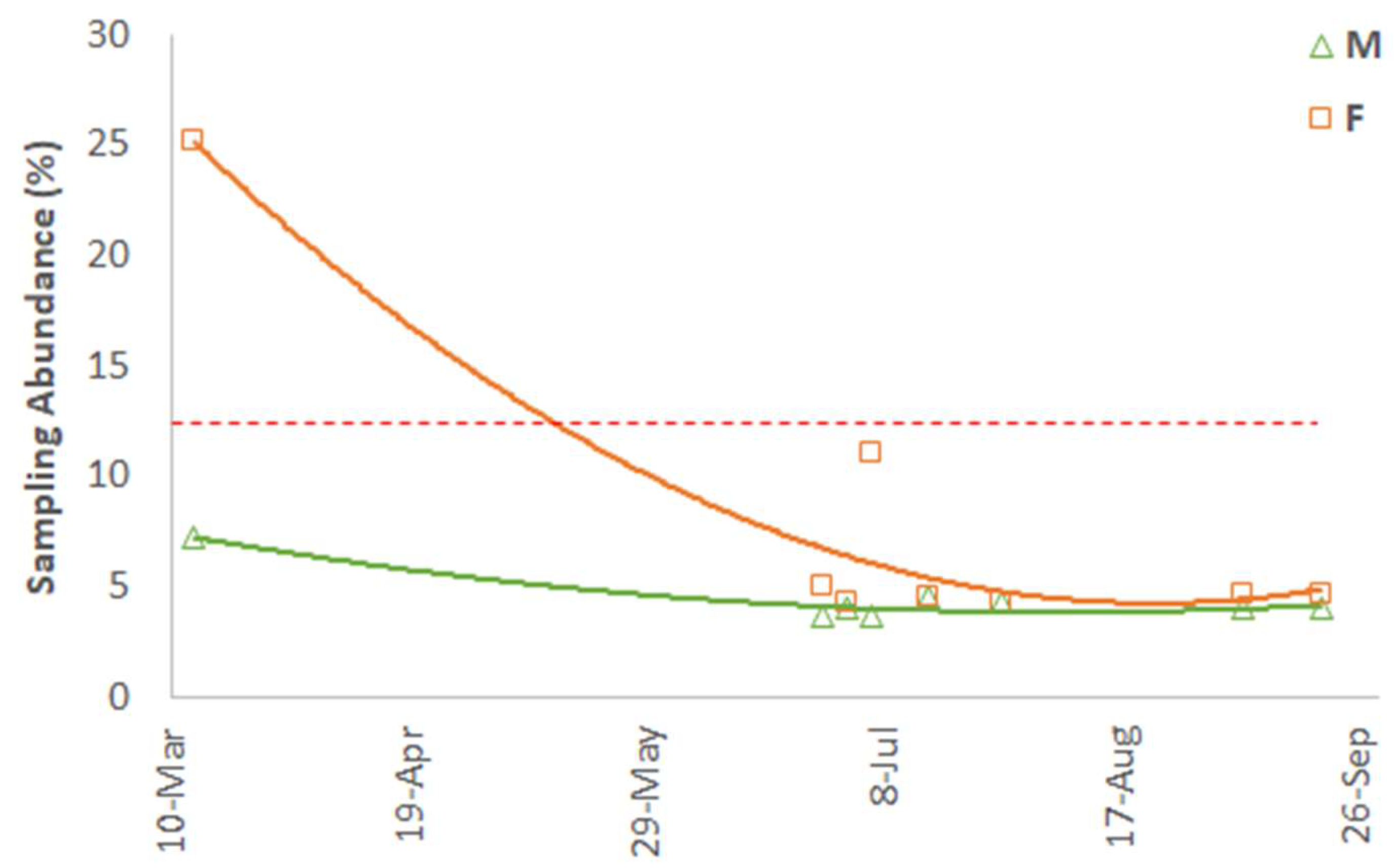
| Date | Department | Municipality | Hamlet | Location (Latitude and Longitude) | No. Samples | M:F Ratio |
|---|---|---|---|---|---|---|
| 14 March 2021 | Usulutan | Ereguayquin | Unnamed | 13°20.5880′ N 88°23.3880′ W | 187 | 29:100 |
| 28 June 2022 | Morazan | San Carlos | Barrio El Centro | 13°38′48.5″ N 88°05′47.4″ W | 50 | 18:25 |
| 2 July 2022 | Morazan | Jocoro | Barrio San Sebastian | 13°36′56.3″ N 88°01′21.3″ W | 48 | 23:25 |
| 6 July 2021 | San Miguel | San Rafael Oriente | Barrio San Benito | 13°22.8290′ N 88°21.2050′ W | 89 | 33:100 |
| 16 July 2022 | Morazan | Jocoro | Las Marias Centro | 13°37′08.1″ N 87°58′56.1″ W | 52 | 1:1 |
| 28 July 2022 | La Union | Santa Rosa de Lima | El Limón | 13°36′25.8″ N 87°55′47.3″ W | 50 | 1:1 |
| 7 September 2022 | La Union | Pasaquina | Santa Clara | 13°35′12.4″ N 87°46′47.2″ W | 50 | 17:20 |
| 20 September 2022 | La Union | San José la Fuente | Barrio El Calvario | 13°33′33.2″ N 87°54′16.4″ W | 50 | 17:20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Groat-Carmona, A.M.; Velado Cano, M.A.; González Pérez, A.M.; Carmona-Galindo, V.D. Sex Ratio Distortion of Aedes aegypti (L.) in El Salvador: Biocontrol Implications for Seasonally Dry Urban Neotropical Environments. Diversity 2025, 17, 257. https://doi.org/10.3390/d17040257
Groat-Carmona AM, Velado Cano MA, González Pérez AM, Carmona-Galindo VD. Sex Ratio Distortion of Aedes aegypti (L.) in El Salvador: Biocontrol Implications for Seasonally Dry Urban Neotropical Environments. Diversity. 2025; 17(4):257. https://doi.org/10.3390/d17040257
Chicago/Turabian StyleGroat-Carmona, Anna M., Maryory A. Velado Cano, Ana M. González Pérez, and Víctor D. Carmona-Galindo. 2025. "Sex Ratio Distortion of Aedes aegypti (L.) in El Salvador: Biocontrol Implications for Seasonally Dry Urban Neotropical Environments" Diversity 17, no. 4: 257. https://doi.org/10.3390/d17040257
APA StyleGroat-Carmona, A. M., Velado Cano, M. A., González Pérez, A. M., & Carmona-Galindo, V. D. (2025). Sex Ratio Distortion of Aedes aegypti (L.) in El Salvador: Biocontrol Implications for Seasonally Dry Urban Neotropical Environments. Diversity, 17(4), 257. https://doi.org/10.3390/d17040257








