A High-Throughput Phenotypic Screen of the ‘Pandemic Response Box’ Identifies a Quinoline Derivative with Significant Anthelmintic Activity
Abstract
:1. Introduction
2. Results
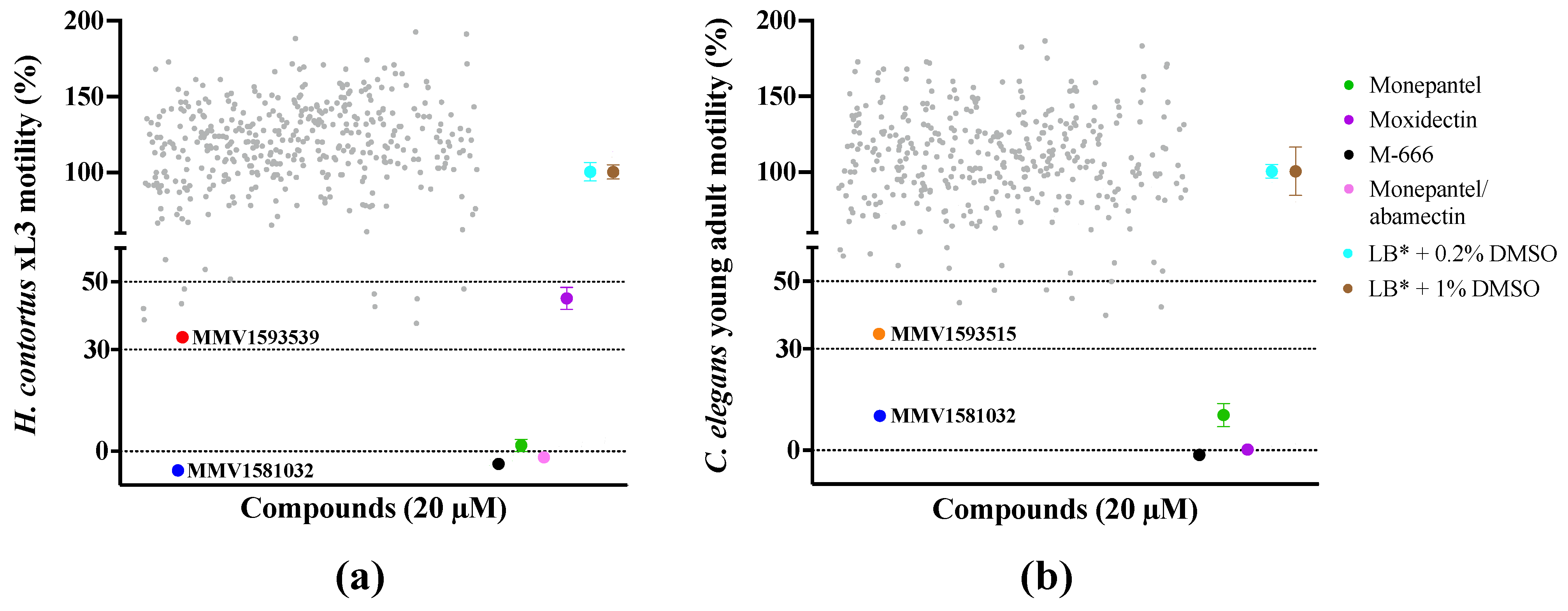
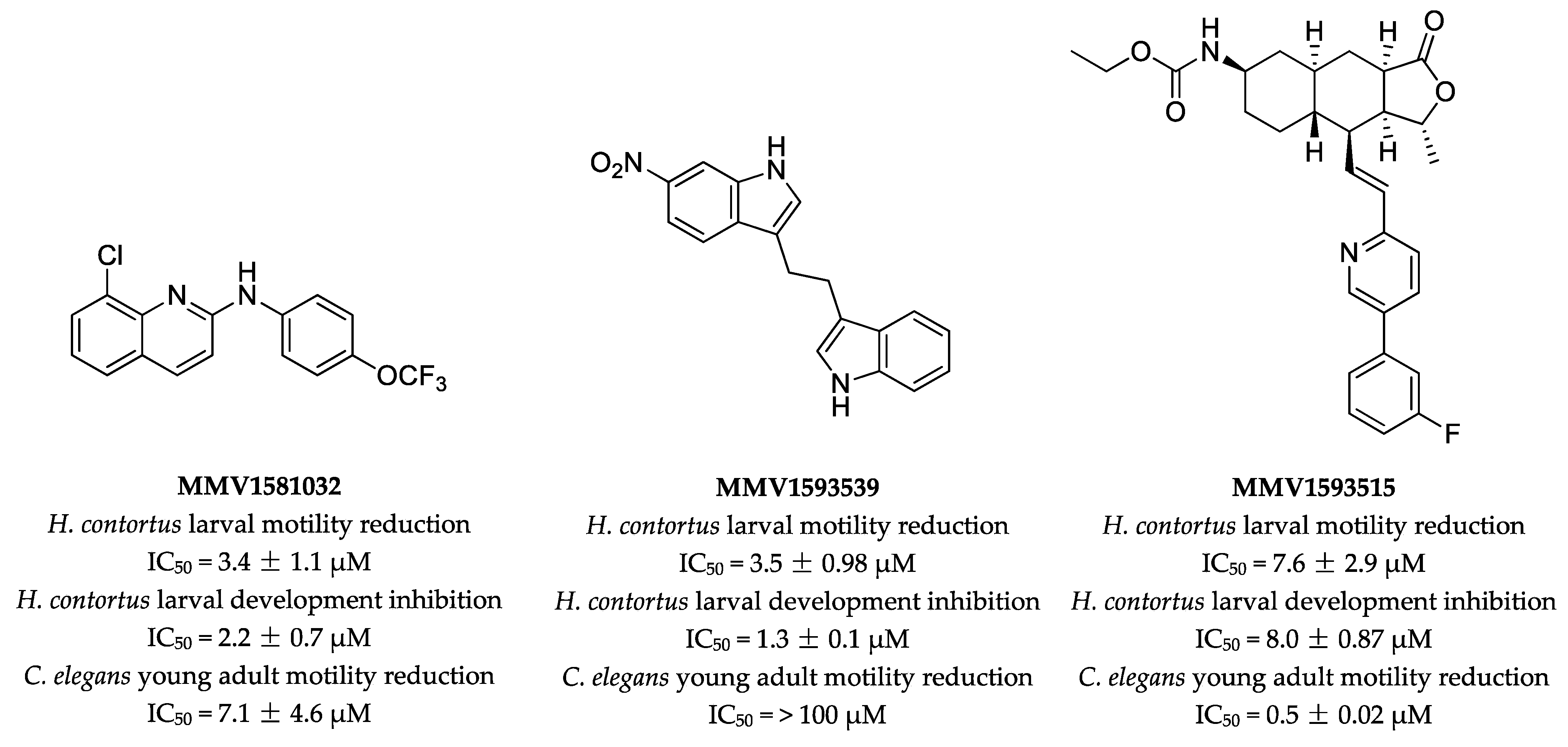
2.1. Primary Screen Identifies Three Compounds with Anthelmintic Activity
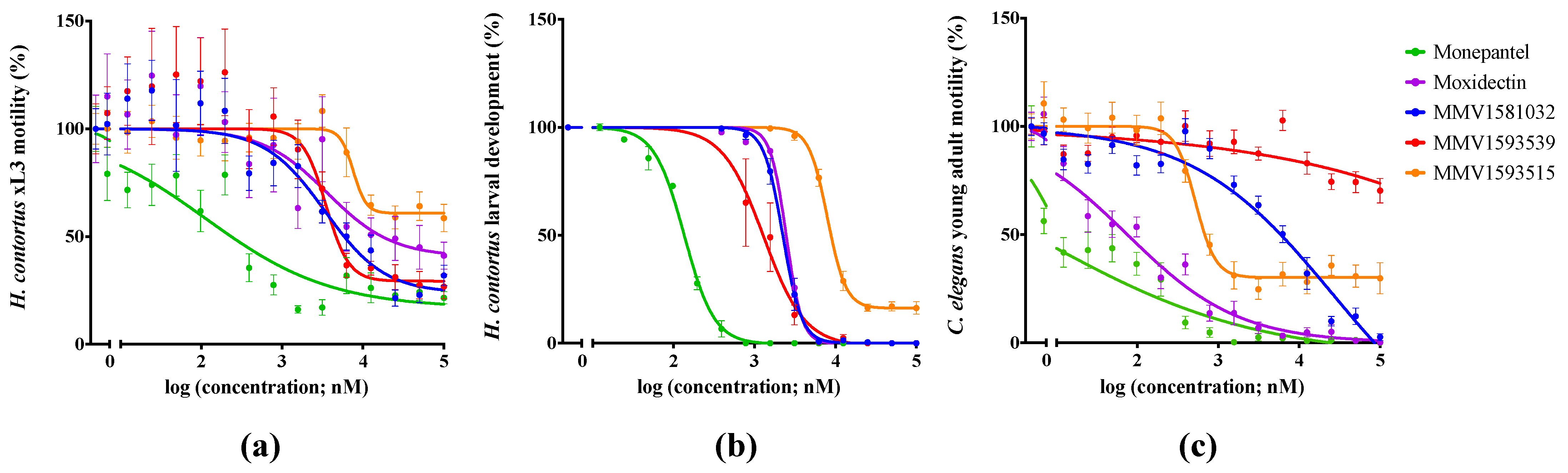
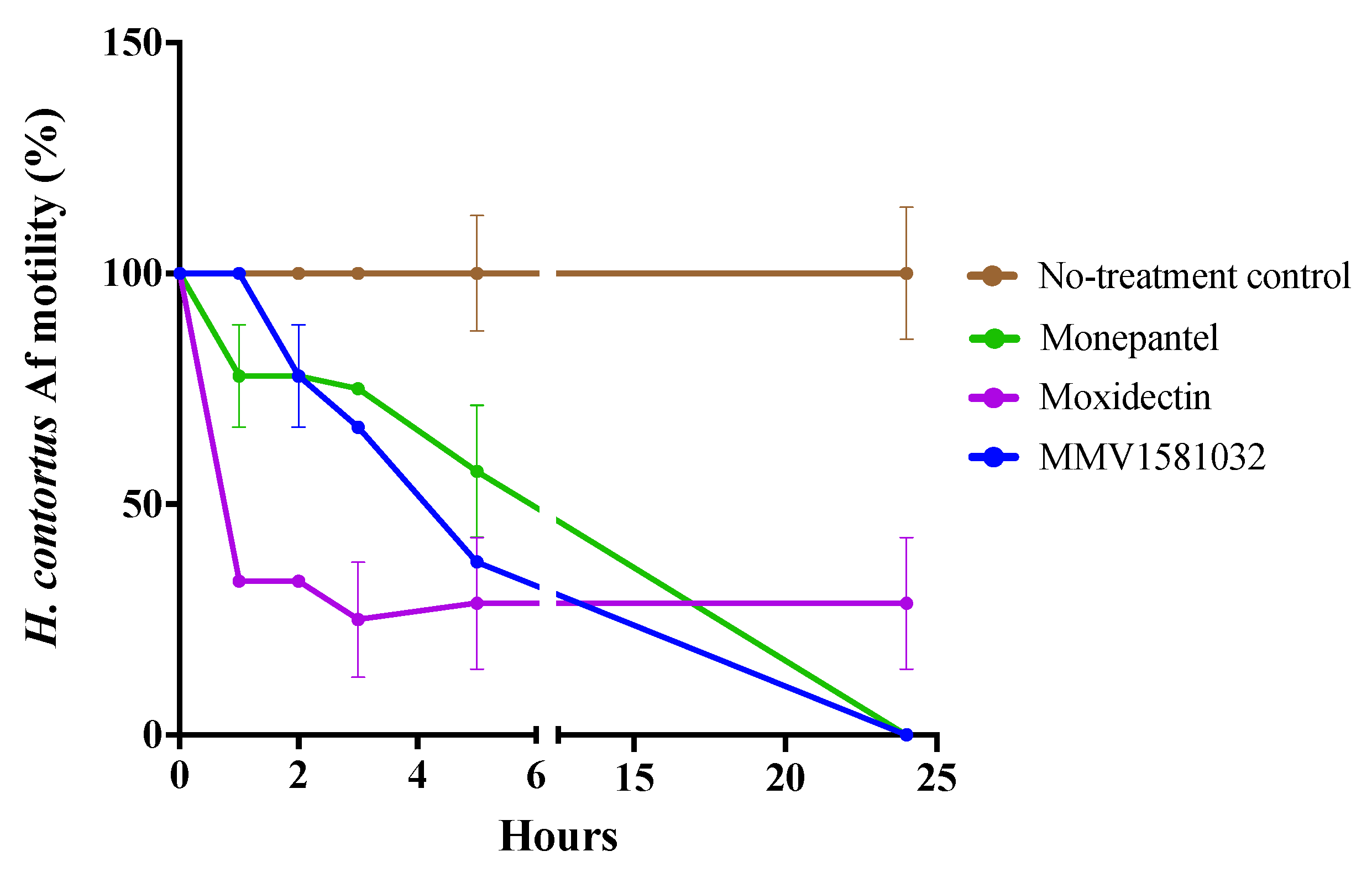
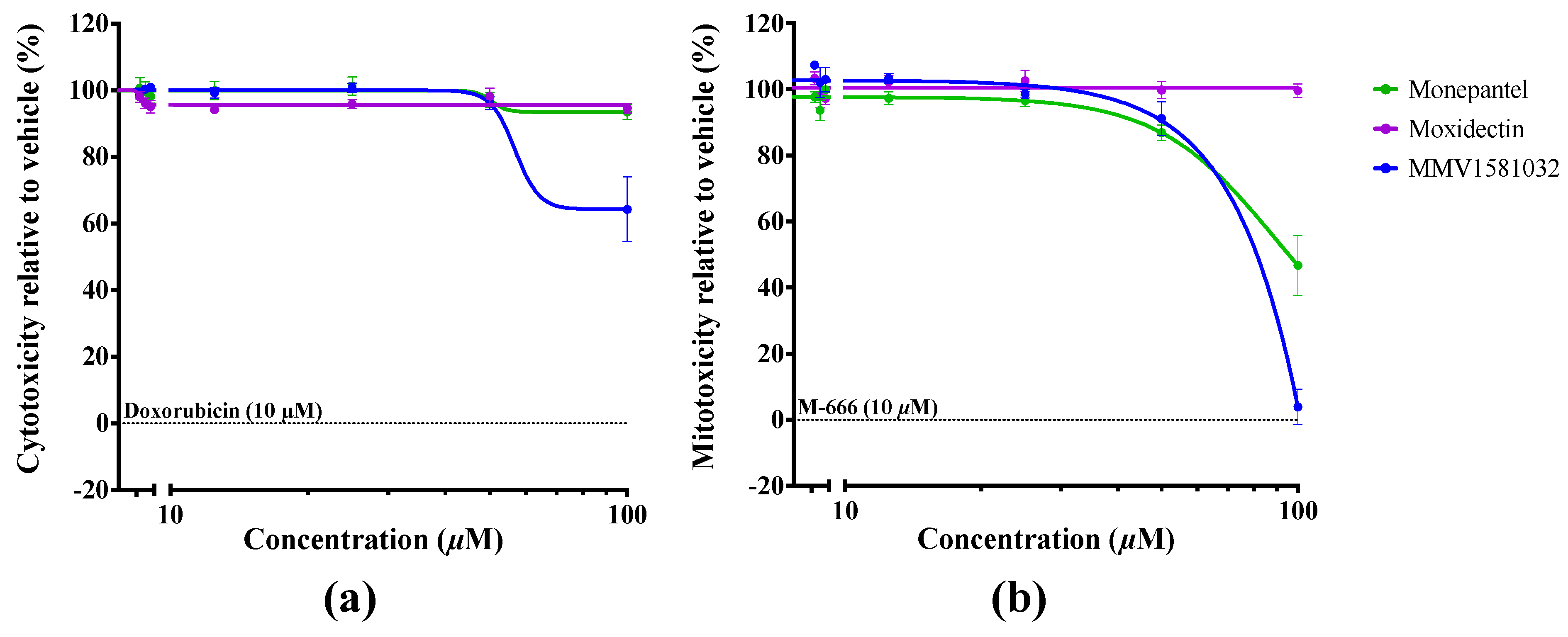
2.2. Potency and Toxicity Assessments Reveal One Promising Anthelmintic Candidate
3. Discussion
4. Materials and Methods
4.1. Preparation of Compounds for Screening
4.2. Production, Storage and Preparation of H. contortus
4.3. Production, Storage and Preparation of C. elegans
4.4. Screening for Anthelmintic Activity against H. contortus
4.5. Screening for Anthelmintic Activity against C. elegans
4.6. Dose-Response Assay Using H. contortus
4.7. Dose-Response Assay Using C. elegans
4.8. Motility Assay against Adult Female H. contortus
4.9. Evaluation of Cellular and Mitochondrial Toxicities Using HepG2 Cells
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fenwick, A. The global burden of neglected tropical diseases. Public Health 2012, 126, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, J.L. Global food security: The impact of veterinary parasites and parasitologists. Vet. Parasitol. 2013, 195, 233–248. [Google Scholar] [CrossRef] [PubMed]
- Casulli, A. New global targets for NTDs in the WHO roadmap 2021–2030. PLoS Negl. Trop. Dis. 2021, 15, e0009373. [Google Scholar] [CrossRef]
- Roeber, F.; Jex, A.R.; Gasser, R.B. Impact of gastrointestinal parasitic nematodes of sheep, and the role of advanced molecular tools for exploring epidemiology and drug resistance—An Australian perspective. Parasit. Vectors 2013, 6, 153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le Jambre, L.F. Relationship of blood loss to worm numbers, biomass and egg production in Haemonchus infected sheep. Int. J. Parasitol. 1995, 25, 269–273. [Google Scholar] [CrossRef]
- Besier, R.B.; Kahn, L.P.; Sargison, N.D.; Van Wyk, J.A. Diagnosis, treatment and management of Haemonchus contortus in small ruminants. Adv. Parasitol. 2016, 93, 181–238. [Google Scholar] [CrossRef] [PubMed]
- Veglia, F. The Anatomy and Life-History of Haemonchus contortus (Rud). Rep. Dir. Vet. Res. 1915, 3–4, 347–500. [Google Scholar]
- Kearney, P.E.; Murray, P.J.; Hoy, J.M.; Hohenhaus, M.; Kotze, A. The “toolbox” of strategies for managing Haemonchus contortus in goats: What’s in and what’s out. Vet. Parasitol. 2016, 220, 93–107. [Google Scholar] [CrossRef]
- Lanusse, C.E.; Alvarez, L.I.; Lifschitz, A.L. Gaining insights into the pharmacology of anthelmintics using Haemonchus contortus as a model nematode. Adv. Parasitol. 2016, 93, 465–518. [Google Scholar] [CrossRef]
- Sargison, N.D. Pharmaceutical treatments of gastrointestinal nematode infections of sheep—future of anthelmintic drugs. Vet. Parasitol. 2012, 189, 79–84. [Google Scholar] [CrossRef]
- Kotze, A.C.; Prichard, R.K. Anthelmintic resistance in Haemonchus contortus: History, mechanisms and diagnosis. Adv. Parasitol. 2016, 93, 397–428. [Google Scholar] [CrossRef]
- Waller, P.J. Global perspectives on nematode parasite control in ruminant livestock: The need to adopt alternatives to chemotherapy, with emphasis on biological control. Anim. Health Res. Rev. 2003, 4, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Sargison, N.D. Keys to solving health problems in small ruminants: Anthelmintic resistance as a threat to sustainable nematode control. Small Rumin. Res. 2016, 142, 11–15. [Google Scholar] [CrossRef] [Green Version]
- Papadopoulos, E. Anthelmintic resistance in sheep nematodes. Small Rumin. Res. 2008, 76, 99–103. [Google Scholar] [CrossRef]
- Kaminsky, R.; Gauvry, N.; Schorderet Weber, S.; Skripsky, T.; Bouvier, J.; Wenger, A.; Schroeder, F.; Desaules, Y.; Hotz, R.; Goebel, T.; et al. identification of the amino-acetonitrile derivative monepantel (AAD 1566) as a new anthelmintic drug development candidate. Parasitol. Res. 2008, 103, 931–939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, B.H.; Clothier, M.F.; Dutton, F.E.; Nelson, S.J.; Johnson, S.S.; Thompson, D.P.; Geary, T.G.; Whaley, H.D.; Haber, C.L.; Marshall, V.P.; et al. Marcfortine and paraherquamide class of anthelmintics: Discovery of PNU-141962. Curr. Top. Med. Chem. 2002, 2, 779–793. [Google Scholar] [CrossRef] [PubMed]
- Little, P.R.; Hodges, A.; Watson, T.G.; Seed, J.A.; Maeder, S.J. Field efficacy and safety of an oral formulation of the novel combination anthelmintic, derquantel-abamectin, in sheep in New Zealand. N. Z. Vet. J. 2010, 58, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.; Preston, S.; Hofmann, A.; Taki, A.; Baell, J.; Chang, B.C.H.; Jabbar, A.; Gasser, R.B. A perspective on the discovery of selected compounds with anthelmintic activity against the barber’s pole worm-where to from here? Adv. Parasitol. 2020, 108, 1–45. [Google Scholar] [CrossRef]
- Herath, H.M.P.D.; Taki, A.C.; Sleebs, B.E.; Hofmann, A.; Nguyen, N.; Preston, S.; Davis, R.A.; Jabbar, A.; Gasser, R.B. Advances in the discovery and development of anthelmintics by harnessing natural product scaffolds. Adv. Parasitol. 2021, 111, 203–251. [Google Scholar] [CrossRef]
- Camp, D.; Newman, S.; Pham, N.B.; Quinn, R.J. Nature Bank and the Queensland compound library: Unique international resources at the Eskitis Institute for Drug Discovery. Comb. Chem. High Throughput Screen. 2014, 17, 201–209. [Google Scholar] [CrossRef] [Green Version]
- Preston, S.; Jiao, Y.; Jabbar, A.; McGee, S.L.; Laleu, B.; Willis, P.; Wells, T.N.C.; Gasser, R.B. Screening of the “Pathogen Box” identifies an approved pesticide with major anthelmintic activity against the barber’s pole worm. Int. J. Parasitol. Drugs Drug Resist. 2016, 6, 329–334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herath, H.M.P.D.; Preston, S.; Hofmann, A.; Davis, R.A.; Koehler, A.V.; Chang, B.C.H.; Jabbar, A.; Gasser, R.B. Screening of a small, well-curated natural product-based library identifies two rotenoids with potent nematocidal activity against Haemonchus contortus. Vet. Parasitol. 2017, 244, 172–175. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.; Preston, S.; Song, H.; Jabbar, A.; Liu, Y.; Baell, J.; Hofmann, A.; Hutchinson, D.; Wang, T.; Koehler, A.V.; et al. Assessing the anthelmintic activity of pyrazole-5-carboxamide derivatives against Haemonchus contortus. Parasit. Vectors 2017, 10, 272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiao, Y.; Preston, S.; Koehler, A.V.; Stroehlein, A.J.; Chang, B.C.H.; Simpson, K.J.; Cowley, K.J.; Palmer, M.J.; Laleu, B.; Wells, T.N.C.; et al. Screening of the ‘Stasis Box’ identifies two kinase inhibitors under pharmaceutical development with activity against Haemonchus contortus. Parasit. Vectors 2017, 10, 323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Preston, S.; Jiao, Y.; Baell, J.B.; Keiser, J.; Crawford, S.; Koehler, A.V.; Wang, T.; Simpson, M.M.; Kaplan, R.M.; Cowley, K.J.; et al. Screening of the ‘Open Scaffolds’ collection from compounds Australia identifies a new chemical entity with anthelmintic activities against different developmental stages of the barber’s pole worm and other parasitic nematodes. Int. J. Parasitol. Drugs Drug Resist. 2017, 7, 286–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herath, H.M.P.D.; Song, H.; Preston, S.; Jabbar, A.; Wang, T.; McGee, S.L.; Hofmann, A.; Garcia-Bustos, J.; Chang, B.C.H.; Koehler, A.V.; et al. Arylpyrrole and fipronil analogues that inhibit the motility and/or development of Haemonchus contortus in vitro. Int. J. Parasitol. Drugs Drug Resist. 2018, 8, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.; Preston, S.; Garcia-Bustos, J.F.; Baell, J.B.; Ventura, S.; Le, T.; McNamara, N.; Nguyen, N.; Botteon, A.; Skinner, C.; et al. Tetrahydroquinoxalines induce a lethal evisceration phenotype in Haemonchus contortus in vitro. Int. J. Parasitol. Drugs Drug Resist. 2019, 9, 59–71. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Kurz, T.; Preston, S.; Brueckmann, H.; Lungerich, B.; Herath, H.M.P.D.; Koehler, A.V.; Wang, T.; Skálová, L.; Jabbar, A.; et al. Phenotypic screening of the ‘Kurz-box’ of chemicals identifies two compounds (BLK127 and HBK4) with anthelmintic activity in vitro against parasitic larval stages of Haemonchus contortus. Parasit. Vectors 2019, 12, 191. [Google Scholar] [CrossRef]
- Taki, A.C.; Brkljača, R.; Wang, T.; Koehler, A.V.; Ma, G.; Danne, J.; Ellis, S.; Hofmann, A.; Chang, B.C.H.; Jabbar, A.; et al. Natural compounds from the marine brown alga Caulocystis cephalornithos with potent in vitro-activity against the parasitic nematode Haemonchus contortus. Pathogens 2020, 9, 550. [Google Scholar] [CrossRef]
- Taki, A.C.; Jabbar, A.; Kurz, T.; Lungerich, B.; Ma, G.; Byrne, J.J.; Pflieger, M.; Asfaha, Y.; Fischer, F.; Chang, B.C.H.; et al. Three small molecule entities (MPK18, MPK334 and YAK308) with activity against Haemonchus contortus in vitro. Molecules 2021, 26, 2819. [Google Scholar] [CrossRef]
- Taki, A.C.; Byrne, J.J.; Wang, T.; Sleebs, B.E.; Nguyen, N.; Hall, R.S.; Korhonen, P.K.; Chang, B.C.H.; Jackson, P.; Jabbar, A.; et al. High-throughput phenotypic assay to screen for anthelmintic activity on Haemonchus contortus. Pharmaceuticals 2021, 14, 616. [Google Scholar] [CrossRef] [PubMed]
- Le, T.G.; Kundu, A.; Ghoshal, A.; Nguyen, N.H.; Preston, S.; Jiao, Y.; Ruan, B.; Xue, L.; Huang, F.; Keiser, J.; et al. Structure-activity relationship studies of tolfenpyrad reveal subnanomolar inhibitors of Haemonchus contortus development. J. Med. Chem. 2019, 62, 1036–1053. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le, T.G.; Kundu, A.; Ghoshal, A.; Nguyen, N.H.; Preston, S.; Jiao, Y.; Ruan, B.; Xue, L.; Huang, F.; Keiser, J.; et al. Novel 1-methyl-1 h-pyrazole-5-carboxamide derivatives with potent anthelmintic activity. J. Med. Chem. 2019, 62, 3367–3380. [Google Scholar] [CrossRef] [PubMed]
- Laing, R.; Kikuchi, T.; Martinelli, A.; Tsai, I.J.; Beech, R.N.; Redman, E.; Holroyd, N.; Bartley, D.J.; Beasley, H.; Britton, C.; et al. The genome and transcriptome of Haemonchus contortus, a key model parasite for drug and vaccine discovery. Genome Biol. 2013, 14, R88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwarz, E.M.; Korhonen, P.K.; Campbell, B.E.; Young, N.D.; Jex, A.R.; Jabbar, A.; Hall, R.S.; Mondal, A.; Howe, A.C.; Pell, J.; et al. The genome and developmental transcriptome of the strongylid nematode Haemonchus contortus. Genome Biol. 2013, 14, R89. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Nie, S.; Ma, G.; Korhonen, P.K.; Koehler, A.V.; Ang, C.-S.; Reid, G.E.; Williamson, N.A.; Gasser, R.B. The developmental lipidome of Haemonchus contortus. Int. J. Parasitol. 2018, 48, 887–895. [Google Scholar] [CrossRef]
- Wang, T.; Ma, G.; Ang, C.-S.; Korhonen, P.K.; Koehler, A.V.; Young, N.D.; Nie, S.; Williamson, N.A.; Gasser, R.B. High throughput LC-MS/MS-based proteomic analysis of excretory-secretory products from short-term in vitro culture of Haemonchus contortus. J. Proteom. 2019, 204, 103375. [Google Scholar] [CrossRef]
- Wang, T.; Ma, G.; Ang, C.-S.; Korhonen, P.K.; Xu, R.; Nie, S.; Koehler, A.V.; Simpson, R.J.; Greening, D.W.; Reid, G.E.; et al. Somatic proteome of Haemonchus contortus. Int. J. Parasitol. 2019, 49, 311–320. [Google Scholar] [CrossRef]
- Doyle, S.R.; Tracey, A.; Laing, R.; Holroyd, N.; Bartley, D.; Bazant, W.; Beasley, H.; Beech, R.; Britton, C.; Brooks, K.; et al. Genomic and transcriptomic variation defines the chromosome-scale assembly of Haemonchus contortus, a model gastrointestinal worm. Commun. Biol. 2020, 3, 1–16. [Google Scholar] [CrossRef]
- Ma, G.; Gasser, R.B.; Wang, T.; Korhonen, P.K.; Young, N.D. Toward integrative ’omics of the barber’s pole worm and related parasitic nematodes. Infect. Genet. Evol. 2020, 85, 104500. [Google Scholar] [CrossRef]
- Ma, G.; Wang, T.; Korhonen, P.K.; Hofmann, A.; Sternberg, P.W.; Young, N.D.; Gasser, R.B. Elucidating the molecular and developmental biology of parasitic nematodes: Moving to a multiomics paradigm. Adv. Parasitol. 2020, 108, 175–229. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Gasser, R.B. Prospects of using high-throughput proteomics to underpin the discovery of animal host–nematode interactions. Pathogens 2021, 10, 825. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Ma, G.; Ang, C.-S.; Korhonen, P.K.; Stroehlein, A.J.; Young, N.D.; Hofmann, A.; Chang, B.C.H.; Williamson, N.A.; Gasser, R.B. The developmental phosphoproteome of Haemonchus contortus. J. Proteom. 2020, 213, 103615. [Google Scholar] [CrossRef] [PubMed]
- Holden-Dye, L.; Walker, R.J. Anthelmintic drugs and nematicides: Studies in Caenorhabditis elegans. In WormBook: The Online Review of C. elegans Biology; The C. elegans Research Community: Pasadena, CA, USA, 2014; pp. 1–29. [Google Scholar]
- Hahnel, S.R.; Dilks, C.M.; Heisler, I.; Andersen, E.C.; Kulke, D. Caenorhabditis elegans in anthelmintic research—Old model, new perspectives. Int. J. Parasitol. Drugs Drug Resist. 2020, 14, 237–248. [Google Scholar] [CrossRef]
- Sepúlveda-Crespo, D.; Reguera, R.M.; Rojo-Vázquez, F.; Balaña-Fouce, R.; Martínez-Valladares, M. drug discovery technologies: Caenorhabditis elegans as a model for anthelmintic therapeutics. Med. Res. Rev. 2020, 40, 1715–1753. [Google Scholar] [CrossRef] [PubMed]
- Harris, T.W.; Arnaboldi, V.; Cain, S.; Chan, J.; Chen, W.J.; Cho, J.; Davis, P.; Gao, S.; Grove, C.A.; Kishore, R.; et al. WormBase: A modern model organism information resource. Nucleic Acids Res. 2020, 48, D762–D767. [Google Scholar] [CrossRef]
- Gasser, R.B.; Schwarz, E.M.; Korhonen, P.K.; Young, N.D. Understanding Haemonchus contortus better through genomics and transcriptomics. Adv. Parasitol. 2016, 93, 519–567. [Google Scholar] [CrossRef]
- Herath, H.M.P.D.; Preston, S.; Jabbar, A.; Garcia-Bustos, J.; Addison, R.S.; Hayes, S.; Rali, T.; Wang, T.; Koehler, A.V.; Chang, B.C.H.; et al. Selected α-pyrones from the plants Cryptocarya novoguineensis (Lauraceae) and Piper methysticum (Piperaceae) with activity against Haemonchus contortus in vitro. Int. J. Parasitol. Drugs Drug Resist. 2019, 9, 72–79. [Google Scholar] [CrossRef]
- Zoraghi, R.; Campbell, S.; Kim, C.; Dullaghan, E.M.; Blair, L.M.; Gillard, R.M.; Reiner, N.E.; Sperry, J. Discovery of a 1,2-bis(3-indolyl)ethane that selectively inhibits the pyruvate kinase of methicillin-resistant Staphylococcus aureus over human isoforms. Bioorg. Med. Chem. Lett. 2014, 24, 5059–5062. [Google Scholar] [CrossRef]
- Palaima, E.; Leymarie, N.; Stroud, D.; Mizanur, R.M.; Hodgkin, J.; Gravato-Nobre, M.J.; Costello, C.E.; Cipollo, J.F. The Caenorhabditis elegans bus-2 mutant reveals a new class of O-glycans affecting bacterial resistance. J. Biol. Chem. 2010, 285, 17662–17672. [Google Scholar] [CrossRef] [Green Version]
- Hubbard, T.D.; Murray, I.A.; Perdew, G.H. Indole and tryptophan metabolism: Endogenous and dietary routes to Ah receptor activation. Drug Metab. Dispos. 2015, 43, 1522–1535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nepali, K.; Lee, H.-Y.; Liou, J.-P. Nitro-group-containing drugs. J. Med. Chem. 2019, 62, 2851–2893. [Google Scholar] [CrossRef] [PubMed]
- Chackalamannil, S.; Wang, Y.; Greenlee, W.J.; Hu, Z.; Xia, Y.; Ahn, H.-S.; Boykow, G.; Hsieh, Y.; Palamanda, J.; Agans-Fantuzzi, J.; et al. Discovery of a novel, orally active himbacine-based thrombin receptor antagonist (sCH 530348) with potent antiplatelet activity. J. Med. Chem. 2008, 51, 3061–3064. [Google Scholar] [CrossRef] [PubMed]
- Gryka, R.J.; Buckley, L.F.; Anderson, S.M. Vorapaxar: The current role and future directions of a novel protease-activated receptor antagonist for risk reduction in atherosclerotic disease. Drugs R&D 2017, 17, 65–72. [Google Scholar] [CrossRef] [Green Version]
- Malaska, M.J.; Fauq, A.H.; Kozikowski, A.P.; Aagaard, P.J.; McKinney, M. Chemical modification of ring C of himbacine: Discovery of a pharmacophoric element for M2-selectivity. Bioorg. Med. Chem. Lett. 1995, 5, 61–66. [Google Scholar] [CrossRef]
- Chackalamannil, S.; Doller, D.; McQuade, R.; Ruperto, V. Himbacine analogs as muscarinic receptor antagonists—Effects of tether and heterocyclic variations. Bioorg. Med. Chem. Lett. 2004, 14, 3967–3970. [Google Scholar] [CrossRef]
- Steger, K.A.; Avery, L. The GAR-3 muscarinic receptor cooperates with calcium signals to regulate muscle contraction in the Caenorhabditis elegans pharynx. Genetics 2004, 167, 633–643. [Google Scholar] [CrossRef] [Green Version]
- Blaxter, M.L.; De Ley, P.; Garey, J.R.; Liu, L.X.; Scheldeman, P.; Vierstraete, A.; Vanfleteren, J.R.; Mackey, L.Y.; Dorris, M.; Frisse, L.M.; et al. A molecular evolutionary framework for the phylum Nematoda. Nature 1998, 392, 71–75. [Google Scholar] [CrossRef]
- Smythe, A.B.; Holovachov, O.; Kocot, K.M. Improved phylogenomic sampling of free-living nematodes enhances resolution of higher-level nematode phylogeny. BMC Evol. Biol. 2019, 19, 121. [Google Scholar] [CrossRef] [Green Version]
- Chackalamannil, S.; Davies, R.J.; Asberom, T.; Doller, D.; Leone, D. A highly efficient total synthesis of (+)-himbacine. J. Am. Chem. Soc. 1996, 118, 9812–9813. [Google Scholar] [CrossRef]
- Chackalamannil, S.; Davies, R.J.; Wang, Y.; Asberom, T.; Doller, D.; Wong, J.; Leone, D.; McPhail, A.T. Total synthesis of (+)-himbacine and (+)-himbeline. J. Org. Chem. 1999, 64, 1932–1940. [Google Scholar] [CrossRef] [PubMed]
- Clasby, M.C.; Chackalamannil, S.; Czarniecki, M.; Doller, D.; Eagen, K.; Greenlee, W.; Kao, G.; Lin, Y.; Tsai, H.; Xia, Y.; et al. Metabolism-identified identification of a potent thrombin receptor antagonist. J. Med. Chem. 2007, 50, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Campos, N.; Myburgh, R.; Garcel, A.; Vautrin, A.; Lapasset, L.; Nadal, E.S.; Mahuteau-Betzer, F.; Najman, R.; Fornarelli, P.; Tantale, K.; et al. Long lasting control of viral rebound with a new drug ABX464 targeting Rev—Mediated viral RNA biogenesis. Retrovirology 2015, 12, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vautrin, A.; Manchon, L.; Garcel, A.; Campos, N.; Lapasset, L.; Laaref, A.M.; Bruno, R.; Gislard, M.; Dubois, E.; Scherrer, D.; et al. Both anti-inflammatory and antiviral properties of novel drug candidate ABX464 are mediated by modulation of RNA splicing. Sci. Rep. 2019, 9, 792. [Google Scholar] [CrossRef] [PubMed]
- Besier, R.B.; Kahn, L.P.; Sargison, N.D.; Van Wyk, J.A. The pathophysiology, ecology and epidemiology of Haemonchus contortus infection in small ruminants. Adv. Parasitol. 2016, 93, 95–143. [Google Scholar] [CrossRef] [PubMed]
- Senerovic, L.; Opsenica, D.; Moric, I.; Aleksic, I.; Spasić, M.; Vasiljevic, B. Quinolines and quinolones as antibacterial, antifungal, anti-virulence, antiviral and anti-parasitic agents. In Advances in Microbiology, Infectious Diseases and Public Health, Advances in Experimental Medicine and Biology; Donelli, G., Ed.; Springer International Publishing: Cham, Switzerland, 2019; Volume 1282, pp. 37–69. ISBN 978-3-030-53647-3. [Google Scholar]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997, 23, 3–25. [Google Scholar] [CrossRef]
- Gonatopoulos-Pournatzis, T.; Cowling, V.H. Cap-binding complex (CBC). Biochem. J. 2014, 457, 231–242. [Google Scholar] [CrossRef] [Green Version]
- Tazi, J.; Begon-Pescia, C.; Campos, N.; Apolit, C.; Garcel, A.; Scherrer, D. Specific and selective induction of miR-124 in immune cells by the quinoline ABX464: A transformative therapy for inflammatory diseases. Drug Discov. Today 2020, 26, 1030–1039. [Google Scholar] [CrossRef]
- Kaminsky, R.; Ducray, P.; Jung, M.; Clover, R.; Rufener, L.; Bouvier, J.; Weber, S.S.; Wenger, A.; Wieland-Berghausen, S.; Goebel, T.; et al. A new class of anthelmintics effective against drug-resistant nematodes. Nature 2008, 452, 176–180. [Google Scholar] [CrossRef]
- Rufener, L.; Mäser, P.; Roditi, I.; Kaminsky, R. Haemonchus contortus acetylcholine receptors of the DEG-3 subfamily and their role in sensitivity to monepantel. PLoS Pathog. 2009, 5, e1000380. [Google Scholar] [CrossRef] [Green Version]
- Gilleard, J.S. Haemonchus contortus as a paradigm and model to study anthelmintic drug resistance. Parasitology 2013, 140, 1506–1522. [Google Scholar] [CrossRef]
- Friman, T. Mass spectrometry-based cellular thermal shift assay (CETSA®) for target deconvolution in phenotypic drug discovery. Bioorg. Med. Chem. 2020, 28, 115174. [Google Scholar] [CrossRef] [PubMed]
- Bertani, G. Studies on lysogenesis. I. The mode of phage liberation by lysogenic Escherichia coli. J. Bacteriol. 1951, 62, 293–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Preston, S.; Jabbar, A.; Nowell, C.; Joachim, A.; Ruttkowski, B.; Baell, J.; Cardno, T.; Korhonen, P.K.; Piedrafita, D.; Ansell, B.R.E.; et al. Low cost whole-organism screening of compounds for anthelmintic activity. Int. J. Parasitol. 2015, 45, 333–343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fleming, M.W. Size of inoculum dose regulates in part worm burdens, fecundity, and lengths in ovine Haemonchus contortus infections. J. Parasitol. 1988, 74, 975–978. [Google Scholar] [CrossRef] [PubMed]
- Coyne, M.J.; Smith, G. The mortality and fecundity of Haemonchus contortus in parasite-naive and parasite-exposed sheep following single experimental infections. Int. J. Parasitol. 1992, 22, 315–325. [Google Scholar] [CrossRef]
- Stiernagle, T. Maintenance of C. elegans. In WormBook: The Online Review of C. elegans Biology; The C. elegans Research Community: Pasadena, CA, USA, 2006; pp. 1–11. [Google Scholar]
- Porta-de-la-Riva, M.; Fontrodona, L.; Villanueva, A.; Cerón, J. Basic Caenorhabditis elegans methods: Synchronization and observation. J. Vis. Exp. 2012, 64, e4019. [Google Scholar] [CrossRef] [Green Version]
- Le, T.G.; Kundu, A.; Ghoshal, A.; Nguyen, N.H.; Preston, S.; Jiao, Y.; Ruan, B.; Xue, L.; Huang, F.; Keiser, J.; et al. Optimization of novel 1-methyl-1 h-pyrazole-5-carboxamides leads to high potency larval development inhibitors of the barber’s pole worm. J. Med. Chem. 2018, 61, 10875–10894. [Google Scholar] [CrossRef]
- Zhang, J.-H.; Chung, T.D.Y.; Oldenburg, K.R. A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J. Biomol. Screen. 1999, 4, 67–73. [Google Scholar] [CrossRef]
- Taki, A.C.; Byrne, J.J.; Boag, P.R.; Jabbar, A.; Gasser, R.B. Practical high-throughput method to screen compounds for anthelmintic activity against Caenorhabditis elegans. Molecules 2021, 26, 4156. [Google Scholar] [CrossRef]
- Swiss, R.L.; Will, Y. Assessment of mitochondrial toxicity in HepG2 cells cultured in high-glucose- or galactose-containing media. Curr. Protoc. Toxicol. 2011, 49, 2.20.1–2.20.14. [Google Scholar]
- Kamalian, L.; Chadwick, A.E.; Bayliss, M.; French, N.S.; Monshouwer, M.; Snoeys, J.; Park, B.K. The utility of HepG2 cells to identify direct mitochondrial dysfunction in the absence of cell death. Toxicol. In Vitro 2015, 29, 732–740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Śliwka, L.; Wiktorska, K.; Suchocki, P.; Milczarek, M.; Mielczarek, S.; Lubelska, K.; Cierpiał, T.; Łyżwa, P.; Kiełbasiński, P.; Jaromin, A.; et al. The comparison of MTT and CVS assays for the assessment of anticancer agent interactions. PLoS ONE 2016, 11, e0155772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Compound. | H. contortus | C. elegans | ||
|---|---|---|---|---|
| Larval Motility (90 h) IC50 ± SD (μM) | Larval Development (168 h) IC50 ± SD (μM) | Abnormal Phenotype (168 h) | Young Adult Motility (40 h) IC50 ± SD (μM) | |
| MMV1581032 | 3.4 ± 1.1 (76) | 2.2 ± 0.7 | Str | 7.1 ± 4.6 (96) |
| MMV1593539 | 3.5 ± 0.98 (71) | 1.3 ± 0.1 | Str | >100 |
| MMV1593515 | 7.6 ± 2.9 (38) | 8.0 ± 0.87 | – | 0.5 ± 0.02 (75) |
| Monepantel | 0.11 ± 0.003 (84) | 0.013 ± 0.002 | Coi | 0.01 ± 0.002 (100) |
| Moxidectin | 7.4 ± 4.2 (70) | 2.4 ± 0.01 | Cur | 0.04 ± 0.0004 (100) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shanley, H.T.; Taki, A.C.; Byrne, J.J.; Jabbar, A.; Wells, T.N.C.; Samby, K.; Boag, P.R.; Nguyen, N.; Sleebs, B.E.; Gasser, R.B. A High-Throughput Phenotypic Screen of the ‘Pandemic Response Box’ Identifies a Quinoline Derivative with Significant Anthelmintic Activity. Pharmaceuticals 2022, 15, 257. https://doi.org/10.3390/ph15020257
Shanley HT, Taki AC, Byrne JJ, Jabbar A, Wells TNC, Samby K, Boag PR, Nguyen N, Sleebs BE, Gasser RB. A High-Throughput Phenotypic Screen of the ‘Pandemic Response Box’ Identifies a Quinoline Derivative with Significant Anthelmintic Activity. Pharmaceuticals. 2022; 15(2):257. https://doi.org/10.3390/ph15020257
Chicago/Turabian StyleShanley, Harrison T., Aya C. Taki, Joseph J. Byrne, Abdul Jabbar, Tim N. C. Wells, Kirandeep Samby, Peter R. Boag, Nghi Nguyen, Brad E. Sleebs, and Robin B. Gasser. 2022. "A High-Throughput Phenotypic Screen of the ‘Pandemic Response Box’ Identifies a Quinoline Derivative with Significant Anthelmintic Activity" Pharmaceuticals 15, no. 2: 257. https://doi.org/10.3390/ph15020257









