Abstract
Psychotropic effect of Crocus sativus L. (family Iridaceae) biologically active chemical compounds are quite well documented and they can therefore be used in addition to the conventional pharmacological treatment of depression. This systematic review on antidepressant compounds in saffron crocus and their mechanisms of action and side effects is based on publications released between 1995–2022 and data indexed in 15 databases under the following search terms: antidepressant effect, central nervous system, Crocus sativus, cognitive impairement, crocin, crocetin, depression, dopamine, dopaminergic and serotonergic systems, picrocrocin, phytotherapy, neurotransmitters, safranal, saffron, serotonin, and biologically active compounds. The comparative analysis of the publications was based on 414 original research papers. The investigated literature indicates the effectiveness and safety of aqueous and alcoholic extracts and biologically active chemical compounds (alkaloids, anthocyanins, carotenoids, flavonoid, phenolic, saponins, and terpenoids) isolated from various organs (corms, leaves, flower petal, and stigmas) in adjuvant treatment of depression and anxiety. Monoamine reuptake inhibition, N-methyl-d-aspartate (NMDA) receptor antagonism, and gamma-aminobutyric acid (GABA)-α agonism are the main proposed mechanism of the antidepressant action. The antidepressant and neuroprotective effect of extract components is associated with their anti-inflammatory and antioxidant activity. The mechanism of their action, interactions with conventional drugs and other herbal preparations and the safety of use are not fully understood; therefore, further detailed research in this field is necessary. The presented results regarding the application of C. sativus in phytotherapy are promising in terms of the use of herbal preparations to support the treatment of depression. This is particularly important given the steady increase in the incidence of this disease worldwide and social effects.
1. Introduction
1.1. Symptoms of Depression
Major depressive disorder (MDD), known also as depression (lat. depressio ‘deepness’ from deprimere ‘overwhelm’), is a chronic, recurrent, and potentially life-threatening mental disorder characterised by at least two weeks of omnipresent low mood. It is usually accompanied with persistent feeling of sadness, anhedonia, pain without a clear cause, difficulties in thinking and concentration, loss of interest in doing anything, psychomotor retardation, fatigue, spending time sleeping, feelings of worthlessness or inappropriate guilt, and recurrent thoughts of death. These symptoms cause distress or impairment in social life and are not an effect of the influence of other medical conditions [1,2,3,4,5,6,7,8]. The spectrum of symptoms in individual patients depends on the type of depression and ranges from excessive consumption of chocolate during episodes of seasonal affective disorder to nihilistic delusions of extensive and absurd content characteristic of Cotard syndrome, which is a rare mental disorder in which the affected person holds the delusional belief that they are dead, do not exist, are putrefying, or have lost their blood or internal organs [9,10]. Depression can affect people at any age (children, adolescents, adults and old individuals) and is characterised by high mortality rates throughout one’s lifetime. Depression very often affects women during the menopausal transition, pregnant women, and both parents after childbirth. “Secondary depression” may be a result of a chronic or terminal medical condition, such as asthma, Lyme disease, cancer, COVID-19, or HIV/AIDS [11,12,13,14,15,16,17,18,19,20,21,22].
1.2. Pathogenesis of Depression
The major depressive disorder has a neuroprogressive nature [23,24] with accelerated cellular aging [25,26,27,28,29], and a higher risk of co-morbid somatic age-related diseases [25,26,27,28,29,30,31,32,33]. Neuroprogression recognised at the clinical, structural, and biochemical levels in the major depressive disorder includes stage-related neurodegeneration, cell death, reduced neurogenesis, reduced neuronal plasticity, and increased autoimmune responses [33,34,35,36,37,38,39]. Depression in its endogenous form accompanies many organic diseases (including infectious diseases), as well as being related to disease processes or treatment (e.g., pharmacologically induced immune-related depression). It can also be a result of stressful events, chronic lifestyle diseases, pollution (e.g., cadmium), and a reduced ability to adapt to the environment or cultural accommodation [40,41,42,43,44,45,46,47,48,49].
The theories of depression pathophysiology are mainly based on: (i) the monoaminergic hypothesis which indicates insufficient activity of monoamine neurotransmitters, (ii) abnormalities analysed in the limbic cortical model and cortico-striatal model, (iii) hypothalamic–pituitary–adrenal axis dysfunction, (iv) overactivation of proinflammatory cytokines [50,51,52,53,54,55,56,57,58].
Results of genetic and neuroradiological studies suggest that changes in specific genes influencing some parts of the brain (e.g., prefrontal brain regions, hippocampus and white matter tracts) may cause major depressive disorder. Many genes related to this disease have been found and epigenetic factor analyses contribute to a deepening of this research [59,60,61,62,63]. According to Wray et al. [64] all humans carry lesser or greater numbers of genetic risk factors for major depression. It should be added that genetic relationships between depression and other diseases, including Crohn’s disease, are also still studied [62].
1.3. Economic and Social Cost of Depression
Depression is one of the most common and still increasing global multidimensional mental health problems, affecting all areas of human life, with high economic and social costs. In 2017, major depressive disorder affected approximately 163 million people (2% of the global population). Now it is estimated that 40 million people suffer from depression across Europe and over 260 million people worldwide. By 2030, depression is supposed to be the leading cause of disease burden in high-income countries. The total direct healthcare cost of depression, depending on the jurisdiction where the analysis was run and the range of cost items included, ranges between €508 and €24,069, whilst indirect costs range between €1963 and €27,364. The economic impact of depression in the European Economic Area (EEA) is thought to be up to €92 billion annually. Decreased productivity is linked to unemployment, poor housing and poverty and therefore many are trapped in a circle of deprivation and illness [65,66,67].
2. Phytotherapy for Depression
In addition to psychotherapy and electroconvulsive therapy, pharmacotherapy is one of the methods for treatment of depression. Currently, increasing attention is being paid to the application of phytochemicals and their derivatives as preventive and therapeutic compounds in supportive therapy of patients treated for neuropsychiatric diseases, including neurodegenerative disorders and depression.
2.1. Taxonomic Diversity of Plants Used in Depression Therapy
Given the numerous undesirable effects of antidepressants and electroconvulsive therapy, effective and safer therapeutic options are being explored [68,69]. It is reasonable to draw attention to the potential of the application of drugs based on phytochemicals with lower toxicity and effective action [70,71,72]. Currently, phytotherapy supporting the treatment of depression and alleviating its symptoms is based on various active chemical compounds obtained from many plant taxa from different families of monocotyledons: Cyperaceae [73], Iridaceae [71,74,75], Xanthorrhoeaceae [74,76] as well as dicotyledons, e.g., Apiaceae [77], Aquifoliaceae [78,79], Asteraceae [80], Capparaceae [81,82], Caprifoliaceae [83,84], Fabaceae [85], Hypericaceae [86,87,88,89,90,91], Lamiaceae [92,93,94], Lauraceae [95], Passifloraceae [96,97,98], Polygalaceae [99], Rutaceae [100,101], Thymelaeaceae [73], and Solanaceae [102] (Table 1).

Table 1.
Raw material of selected plant species from various families with antidepressant properties.
The biologically active chemical compounds present in these plants have antidepressant activity comparable to that of standard anxiolytics and antidepressants [81,103,104,105,106]. Crocus sativus from the Iridaceae family is one of many such plant species. The rationale behind the choice of this plant is not only its well-known medicinal properties and wide use in folk medicine to alleviate symptoms of many diseases, but also its medicinal applications. Especially in the pandemic times, a new search for safe phytochemicals from Crocus sativus with antidepressant effects is the focus of clinical trials. Hence, this species was analysed in detail with reference to the current phytotherapeutic and clinical knowledge.
2.2. Crocus sativus
2.2.1. C. sativus—Characteristics of Pharmacopoeial Raw Material
Crocus sativus L. (family Iridaceae), commonly known as saffron crocus, is a therapeutic plant native to Asia Minor and Southern Europe [133,134,135,136,137,138,139]. The plant is cultivated in Iran, India, Afghanistan, Greece, Morocco, and Italy [133,134,135,136,137,138,139,140,141,142]. It propagates vegetatively. The plant produces an underground tuber and basal, stiff, lanceolate leaves. Its lilac–purple flowers are composed of six tepals, three stamens, and a pistil with a long style and a tripartite dark orange stigma [143,144,145,146,147]. Stigmas, commonly referred to as saffron, are hand-picked during the flowering period and dried immediately after harvesting. Approximately 110,000 to 200,000 flowers are needed to collect 1 kg of stigmas [148,149,150]. Croci sativi stigmas (Stigma Croci) are a pharmacopoeial raw material [151,152,153]. They have high economic importance and are the most expensive raw material in the world. Currently, saffron retail prices reach up to $11,000 per kilogram, while the petals are much cheaper [142,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158,159,160,161].
2.2.2. Biologically Active Chemical Compounds in Various C. sativus Organs
Dried C. sativus stigmas contain over 150 volatile compounds, mainly terpenes and their esters [162,163,164,165,166]. Detailed information about the C. sativus biologically active compounds and their pharmacological activities was compiled by Mykhailenko et al. [167]. Various organs of C. sativus, i.e., the corm, leaf, tepal, stigma, and whole flowers, contain bioactive compounds representing different classes, e.g., anthocyanins, carotenoids, phenolic compounds, flavonoids, carotenoids, saponins, and terpenoids (Table 2).

Table 2.
Total content of some classes of phytotherapeutic bioactive chemical compounds contained in the dry matter (dry weight d.w.) of different organs of C. sativus.
2.2.3. Application of C. sativus in Herbal Medicine and Industry and Therapeutic Activity
Crocus sativus is used in Asian and, in particular, Indian (Ayurveda) and Persian (Islamic) traditional medicine (ITM) as a sedative agent to strengthen the body against such stresses as trauma and anxiety, an anticonvulsant and memory enhancer, and a remedy for alleviation of chronic fatigue, depression, and inflammation [71,135]. This therapeutic activity of Crocus, known since the 6th century BC, has been confirmed in the most recent basic research conducted on animals (rodents) and in human clinical studies [134,175,176,177,178,179,180,181,182,183].
Currently, there is a search for new methods of treatment based on the use of phytochemicals contained in herbal raw materials with significant efficacy in relieving the symptoms of depression confirmed by meta-analyses and clinical trials [75,90,111]. The numerous side effects of antidepressants as well as the attitudes of many patients preferring herbal rather than conventional drugs support the assessment of the impact of saffron crocus stigmas on depression patients [70,71].
Bioactive compounds of C. sativus have a wide range of applications due to their valuable health-enhancing properties [184,185,186]. They are used in many branches of industry, including the pharmaceutical [187,188,189,190,191,192] cosmetic [193,194,195] dairy [196,197], and food [198,199,200,201], industries. These phytochemicals are also used in in the production of nutraceuticals [201,202,203,204,205] and in nanotechnology [206,207,208,209,210], e.g., nanomedicine [211,212] and nanocosmetics [213]. Furthermore, they are applied in therapeutic practice [163,214,215], adjuvant therapy [216,217], and chemopreventive treatment [218,219,220] and have great importance in cosmetic marketing [221], genetic studies, and transgenic plant production [222,223,224,225].
Currently, numerous experiments, cell line studies conducted in various biological models, and clinical trials are ongoing in an attempt to assess the pharmacological effectiveness of biologically active chemical compounds from various organs of saffron crocus in the treatment of some diseases (Table 3, Table 4 and Table 5). These compounds exert a wide spectrum of important healing effects, including antidepressant [175,226,227,228,229], anxiolytic [230,231,232,233,234], and anti-inflammatory [189,204,235,236,237,238,239,240,241,242], activities. Biologically active chemical compounds of saffron crocus have also been shown to have a few other kinds of activity resulting in antimicrobial [243], anticancer [244,245,246,247,248,249,250,251,252], analgesic [176,253,254,255], anticonvulsant [256,257], antitussive [258], antigenotoxic and anticytotoxic [245,259,260,261], relaxant [262,263], antihypertensive [264,265], and antioxidative [171,266,267,268,269,270,271,272,273,274] effects.
In vitro studies have confirmed the antigenotoxic and anticytotoxic effects of active substances isolated from C. sativus [245]. This should be emphasised, as other aspects of the pleiotropic activity of some cytokines and a wide spectrum of the impact of the transcription factor called the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) is present in almost all animal type cells [275,276,277,278].
As reported by Wang et al. [226], the antidepressant properties of stigma aqueous extracts are related to the presence of crocin 1, but further studies regarding the precise site and mechanism of the anti-depressive action of chemical compounds isolated from petroleum ether and dichloromethane fractions of C. sativus corms are required. Karimi et al. [175] have found that flavonoids and anthocyanins are the main constituents involved in the antidepressant action of C. sativus extracts.
Considering the multidirectional phytotherapeutic effect of C. sativus, this paper is a review of available literature data and presents the current information about the effectiveness of bioactive chemical compounds contained in this species and the mechanisms of their action in the supportive therapy of depression, with emphasis on the safety of application of these substances. The thesis of the antidepressant effectiveness was verified by an analysis of the results of the latest basic research conducted in animal models, and human clinical trials. Additionally, the study highlights the difficulties and limitations in laboratory analyses and clinical studies of the antidepressant effects of the phytochemicals and indicates further perspectives of research on their use and potential methods for control of treatment in relation to the disease pathogenesis.
Since depression is a serious growing global health problem with social and economic consequences, intensified investigations are being carried out to search for biologically active chemical compounds of plant origin, which will prove effective in supporting the treatment of this disease. Crocus sativus L. is a species known for its healing properties and widely used in folk medicine to alleviate the symptoms of many diseases. The thesis on antidepressant effectiveness was verified by analysis of the results of the latest basic research in cell cultures, animal models and human clinical trials.




Table 3.
Therapeutic effects of selected biologically active chemical compounds from the classes of anthocyanins, terpenoids, and saponins extracted from C. sativus corms and flowers.
Table 3.
Therapeutic effects of selected biologically active chemical compounds from the classes of anthocyanins, terpenoids, and saponins extracted from C. sativus corms and flowers.
| Classes of Biologically Active Chemical Compounds | Biologically Active Chemical Compounds | Organ | Therapeutic Effects | Reference |
|---|---|---|---|---|
| Anthocyanins | delphinidin, | tepals | antioxidant | [279,280] |
| malvidin | ||||
| petunidin, | ||||
| 3,5-di-O-β-glucosides | ||||
| Terpenoids | monoterpenoids | corm | antibacterial anticancer | [174] |
| sesquiterpenoids | ||||
| Saponins | oleanane type:azafrine 1 azafrine 2 | antitumor, increase immune responses to protein-based vaccines | [281] | |
| bidesmosidic type | ||||
| 3-O-d-glucopyranosiduronic acid echinocystic acid 28-O-d-galactopyranosyl-(1→2)-l-arabinopyranosyl-(1→2)-[-dxylopyranosyl-(1→4)]-d-rhamnopyranosyl-(1→2)-[4-O-di-L-rhamnopyranosyl-3,16-dihydroxy-10-oxo-hexadecanoyl]-d-fucopyranoside | antitumor against HeLa cells | [282] |

Table 4.
Therapeutic effects of selected C. sativus biologically active chemical compounds from the class of phenolic compounds and essential oils.
Table 4.
Therapeutic effects of selected C. sativus biologically active chemical compounds from the class of phenolic compounds and essential oils.
| Classes of Biologically Active Chemical Compounds | Biologically Active Chemical Compounds | Organ | Therapeutic Effects | Reference | |
|---|---|---|---|---|---|
| Polyphenol | pyrogallol | stigma | antioxidant | [171] | |
| Phenolic acid | benzoic acid derivatives | gallic acid | |||
| p-hydroxybenzoic acid | corm | [283] | |||
| salicylic acid | |||||
| gentisic acid | |||||
| syringic acid | |||||
| cinnamic acid derivatives | caffeic acid | ||||
| p-coumaric acid | |||||
| t-ferulic acid | |||||
| cinnamic acid | |||||
| Polyphenols | catechol | ||||
| Phenolic aldehyde | vanillin | ||||
| Essential oils | β-isophorone | stigma | has not been presented | [284] | |
| β-Linalool | |||||
| α-Isophorone | |||||
| palmitic acid methyl ester | |||||
| α, β-dihydro-β-ionone | |||||

Table 5.
Therapeutic effects of selected C. sativus biologically active chemical compounds from the classes of carotenoids extracted from stigmas.
Table 5.
Therapeutic effects of selected C. sativus biologically active chemical compounds from the classes of carotenoids extracted from stigmas.
| Carotenoids | Therapeutic Effects | Reference |
|---|---|---|
| Crocin | inhibited xylene-induced swelling of mouse ear and increased capillary permeability and writhing induced by acetic acid in mice; at 50 mg/kg, it inhibited carrageenan- and fresh egg white-induced oedema of the hind paw in rats. It inhibited sheep red blood cells (SRBC)-induced footpad reaction and inhibited picryl chloride-induced contact dermatitis | [285] |
| cytotoxic effect on human and animal adenocarcinoma cells (HT-29 and DHD/K12-PROb cells) | [286] | |
| a prolonged blood coagulation time in mice and markedly inhibited dose-dependent thrombin- and ADP-induced blood platelet aggregation in rabbits (in vivo); an inhibitory effect on thrombus formation in rats with arteriovenous shunt and relieved respiratory distress due to pulmonary thrombosis in mice induced by ADP and AA | [287] | |
| cardiovascular protective effects; the cardioprotective effects of crocin may be attributed to the attenuation of [Ca2+] through inhibition of ICa-L in rat cardiomyocytes as well as negative inotropic effects on myocardial contractility | [288] | |
| it affected tubulin polymerisation and structure, increased the microtubule nucleation rate, induced conformational changes in tubulin, and affected several cell processes through interaction with tubulin proteins or microtubules | [289] | |
| Crocetin | vasomodulatory effects in hypertension, improvement of endothelium-dependent acetylcholine relaxations via endothelial nitric oxide, improvement of acetylcholine-induced vascular relaxation in hypertension | [290] |
| Crocetin | interaction of carotenoids with topoisomerase II, an enzyme involved in cellular DNA–protein interaction, immunomodulatory activity on T Helper Cell Type 1 (Th)1 and Th2, anticancer properties | [219] |
| Crocin | ||
| Carotene | source of vitamin A, preventive agents against cancer and heart disease, antioxidant and memory effect enhancer | [291] |
| Crocetin | ||
| Licopene | ||
| β-zeaxanthin |
3. Methodology
This review is a presentation of possible treatment methods available across the range of herbal medicines that are relevant to the pathogenesis of depression, with the indication of ways of treatment control with clinical tests used by authors of the cited papers and medical imaging of brain functions for the future scientific purposes. This publication is based on a search in scientific databases of literature reports covering the contemporary research on antidepressant bioactive substances from Crocus sativus L.
3.1. Bibliographic Databases and Searched Phrases
The original scientific publications were found in 15 multidisciplinary specialised scientific databases: Web of Knowledge, EBSCO, Google Scholar, ISI Web of Science, Medline, ProQuest Central, ProQuest SciTech Collection, PubMed, ScienceDirect, Scopus, Springer, Taylor & Francis, Web of Knowledge, Web of Science, and Wiley Online Library. The search engines of these databases provided access to original scientific publications mainly in the fields of medical, preclinical, biological, chemical, and social sciences and sociology. The search was performed using the following phrases: antidepressant effect, central nervous system, Crocus sativus, crocin, crocetin, depression, dopamine, dopaminergic and serotonergic systems, picrocrocin, phytotherapy, melatonin, neurotransmitters, safranal, saffron, serotonin, and biologically active compounds, safety of saffron treatment, and saffron in depression add-on/adjuvant therapy.
3.2. Number and Methods of Analysis of Resources
In total, 414 thematically coherent scientific reports (cited in this review) were selected, including 408 original publications and 7 other sources, e.g., chapters from monographs and books. The analysis was focused on original scientific publications on C. sativus addressing the following issues: (i) biological activity of chemical compounds in various organs, (ii) therapeutic activity, (iii) antidepressant effect of extracts and their components, (iv) mechanisms of antidepressant action, (v) possible future ways for the therapy and its control to proceed in practice, and (vi) challenges for further research. The results of the studies were arranged and presented in the tables according to scheme: (a) animal studies, and (b) human trials.
4. Antidepressant Activity of C. sativus
4.1. Biologically Active Chemical Compounds with Antidepressant Effects
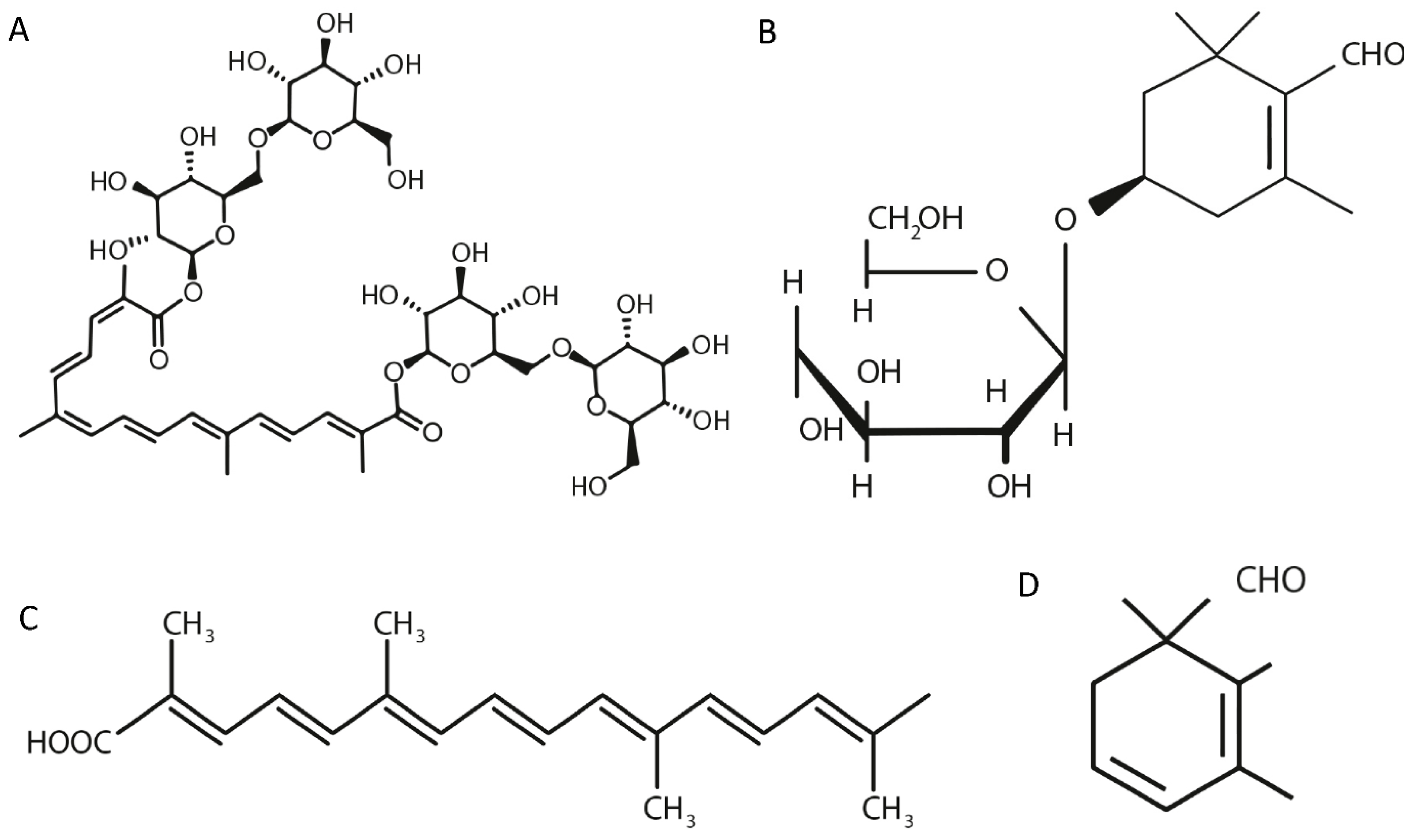
Among the biologically active chemical compounds identified in various C. sativus organs, the antidepressant effects have mainly been ascribed to safranal, crocin, crocetin, and picrocrocin [216,292,293,294,295,296]. The structural formulas of these phytochemicals are shown in Figure 1. The contents of picrocrocin, crocin, crocetin gentiobiose glucose ester and crocetin di-glucose ester in ethanol extracts of Crocus sativus L. are about 40, 20, 10 and 2–3%, respectively [297].
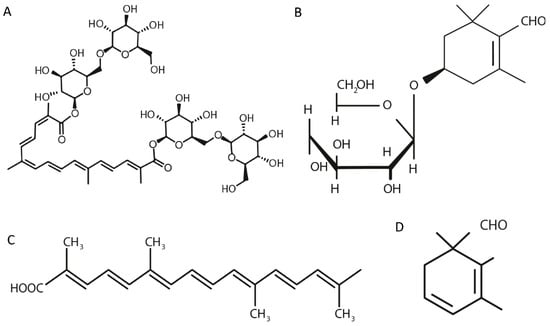
Figure 1.
Structural formula of biologically active chemical compounds: crocin (A), crocetin (B), picrocrocin (C), and safranal (D) present in C. sativus [298,299,300,301].
The content of safranal in saffron crocus stigmas is in the range of 0.1–0.6% d.w. [302,303,304], however, other authors have reported that the content of this compound ranges from 1.07 to 6.15% d.w. [305]. In turn, there are also reports showing that the content of safranal in red stigma samples were 49.64 and 50.29%, while in threads with yellow styles it was 50.42%, 57.02% and 61.31% [306]. The concentration of crocin was estimated at 20–30% of the total dry matter of the spice [154,303,307], but some study results revealed a much wider range of this compound, i.e., 0.85–32.4% [305]. As reported by Zhang and co-workers [308], the content of crocin varied significantly among saffron populations from seven different production areas, i.e., Nepal, Greece, Morocco, Spain, Iran, and China (Jiande, Chongming), and ranged from 80.59 to 230.36 mg/g. Zeka et al. [309] reported that dried petals contained 0.6% of crocin. As suggested by Acar et al. [302] the crocin content of commercial saffron dried in a freeze dryer and dried naturally under the sun was 900 and 600 mg/g, respectively. Azarabadi and Özdemir [306] found that crocin amount was higher in red stigmas samples (66.67 mg/g) than in yellow stigmas samples (51.66 mg/g). The content of crocetin esters represents 16–30% of saffron stigma [310] Crocin is largely absent from petal extracts [311].
The picrocrocin content found in dried stigmas ranged from 0.8 to 26.6% [312,313]. Some authors propose a slightly narrower range of limits for the content of this compound i.e., 0.79–12.94% [305,314], 7–20% [315] and 5–7 mg/g d.w. [303]. The reasons for such a large discrepancy of limits in the content of safranal, crocin, crocin, crocetin and picrocorocin should be sought for in the different drying methods, and storage and extraction conditions of saffron, which degrade these compounds significantly; the degree of degradation depends on temperature, humidity, light irradiation and other compounds in the environment [305].
Othman and co-workers [316] found markedly various crocin and crocetin content in saffron crocus stigmas from different geographical origins. Iranian saffron was characterised by substantially higher amount of crocin content than Turkish and Kashmiri saffron (11,414.67 and 311.63 µg/g d.m. of crocin, respectively). In turn crocetin was detectable in Iranian and Turkish (1054.73 and 186.64 µg/g d.m. of crocetin, respectively) but not in Kashmiri saffron. These differences were suggested to be related to various environmental factors, e.g., climatic conditions, agricultural practices, and stigma separation, as well as storing and drying processes [316].
The information about physicochemical properties of the saffron crocus bioactive compounds, which are important in the preparation of medicinal formulations were presented in the Table 6. The most important stigma constituents include antioxidative carotenoids (with the water-soluble crocin and its derivatives responsible for the colour: zeaxanthin β-carotenes, lycopene), anthocyanins (delphinidin), terpenes (fat-soluble safranal responsible for the odour and aroma and its monoterpene glycoside precursor picrocrocin responsible for the special bitter flavour), polysaccharides, amino acids, proteins, starch, mineral matter, gums, and other chemical compounds [191,201,214,228,317,318].
α-Crocin (systematic IUPAC name: 8, 8-diapo-8, 8-carotenoic acid), which is primarily responsible for the golden yellow-orange colour of the stigma, is a trans-crocetin di-(β-d-) ester. Crocin, underlying the aroma of saffron, is a digentiobiose ester of crocetin. Crocins are hydrophilic carotenoids that are either monoglycosyl or di-glycosyl polyene esters of crocetin. In contrast, crocetin is a hydrophobic and thus oil-soluble conjugated polyene dicarboxylic acid. However, the product of esterification of crocetin with two water-soluble gentiobioses (sugars) is soluble in water [300,317,319].
It is believed that the glycolysed carotenoid crocetin—a natural apocarotenoid dicarboxylic acid—is the most pharmacologically active constituent of stigma extracts, besides the carboxylic carotenoid crocin. Saffron extracts and crocetin had a clear binding capacity at the phencyclidine (PCP) binding side of the N-methyl-d-aspartate receptor (NMDA receptor; NMDAR) and at the σ1 (sigma-1) receptor, while the crocins and picrocrocin were not effective, which give the biochemical support for the pharmacological effect of saffron including depression treatment [188,191,294,295,296,320].
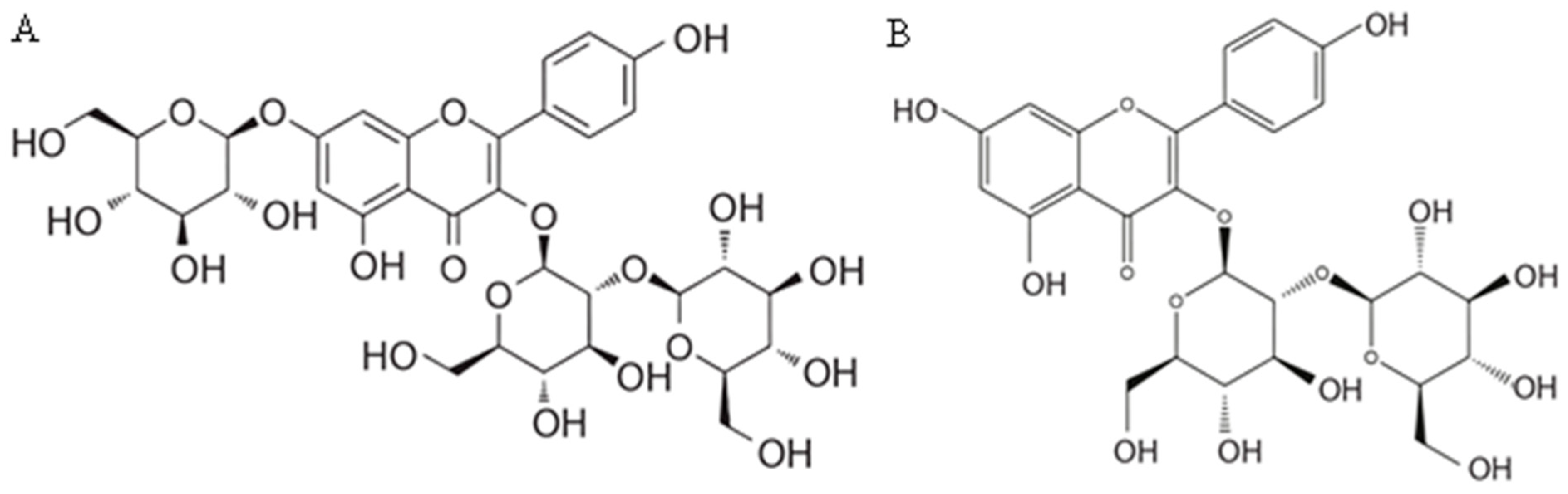
Hosseinzadeh et al. [321] postulated that the flavonol kaempferol was responsible for the antidepressant effect of C. sativus petals. Kaempferol 3-O-β-sophoroside-7-O-β-glucoside is the most important flavonol in saffron. Its relative content ranges from 37% to 63% of total flavonoids, and its absolute content values vary between 1.47 and 2.58 equivalent milligrams of rutin g−1. Kaempferol 3-O-β-sophoroside is the next major flavonol in order of importance, related to the concentration in saffon. Its relative content ranges from 16% to 47% of total flavonoids with absolute content values ranging from 0.61 to 3.12 equivalent milligrams of rutin g−1 [166]. This flavonol was extracted from saffron floral bioresidues that were mainly made up of tepals, and an extract yield of 2.3 mg g−1 dry weight was obtained. Its content in tepals ranges from 0.69 to 12.60 equivalent milligrams of kaempferol 3-O-β-glucoside g1 dry weight [169,309]. According to other literature data, the content of kaempferol-3-O-sophoroside in saffron crocus tepals was 62.19–99.48 mg/g [322]. Another flavonol found in saffron is kaempferol 3,7,4′-tri-O-β-glucoside. Its relative content ranges from 16% to 22% of total flavonoids, and its absolute content values ranges from 0.59 to 1.09 equivalent milligrams of rutin g−1 [166]. In the stamen, the number of flavonoids was lower than in the tepal. The amount of kaempferol-3-O-glucoside, as the most abundant compound, ranged between 1.72–7.44 mg/g [322]. Structures of saffron crocus kaempferol 3-O-β-sophoroside-7-O-β-glucoside and kaempferol 3-O-β-sophoroside are presented in Figure 2.


Table 6.
General characteristics of some biologically active chemical compounds of C. sativus showing antidepressant action.
Table 6.
General characteristics of some biologically active chemical compounds of C. sativus showing antidepressant action.
| Biologically Active Chemical Compounds | Chemical Formula | Molecular Weight [g/mol] | Physical Description | Melting Point [°C] | Solubility | Reference | |
|---|---|---|---|---|---|---|---|
| Traditional Name | Classes | ||||||
| Safranal | monoterpene aldehyde | C10H14O | 150.22 | pale yellowish oily liquid, tobacco-herbaceous odour | <25 | insoluble in water, soluble in oils, miscible in ethanol | [293,323,324,325,326] |
| Crocin | diterpenoid | C26H34O9 * C32H44O14 ** C32H44O14 *** C44H64O24 † C50H24O2 †† | 976.96 | solid | 186 | freely soluble in hot water, sparingly soluble in alcohol, ether and other organic solvents | [251,325,327,328,329,330,331] |
| Crocetin | tetraterpenoid | C20H24O4 | 328.40 | reddish crystals | 186 | slightly soluble in aqueous solution, soluble in organic bases | [331] |
| Picrocrocin | monoterpene glycoside | C16H26O7 | 330.37 | ns | 164–156 | Soluble in water | [251,305,325] |
Explanations: no—not specified, * crocin-1, ** crocin-2, *** crocin-3, † crocin-4, †† crocin-5.
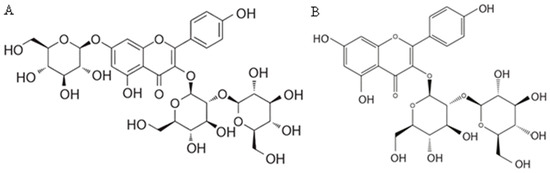
Figure 2.
Structural formula of kaempferol 3-O-β-sophoroside-7-O-β-glucoside (A) and kaempferol 3-O-β-sophoroside (B) from C. sativus [166].
4.2. Antidepressant Effect of C. sativus L.
Extracts of C. sativus and their active biologically chemical substances have been shown to exert beneficial effects on the activity of the central nervous system. Therefore, they can potentially be used as adjuvant agents in treatment of mental disorders, including depression [204,233,311,332,333,334,335,336]. Literature data have demonstrated in a number of in vitro, in vivo, basic and clinical trials that dried C. sativus stigmas and petals as well as their active ingredients exhibit strong antidepressant properties similar to those of the current conventional antidepressant medications from the class of the selective serotonin re-uptake inhibitors (SSRIs), including citalopram [337], fluoxetine (Prozac) [338,339,340,341,342], and sertraline [343], as well as the tricyclic antidepressant imipramine [176,344] and the benzodiazepine diazepam [345,346]. Table 7, Table 8, Table 9 and Table 10 summarise the results of preclinical studies, conducted in animal models and human clinical trials, on the antidepressant effect of extracts and bioactive chemical compounds from the saffron crocus.

Table 7.
Results of the basic (preclinical) studies of the anti-depressive effects of saffron (Crocus sativus L.) stigma extracts or its derivative compounds (crocin, crocetin and safranal) using animal model of depression.

Table 8.
Results of the basic (preclinical) studies of the antidepressive effects of Crocus sativus L. of corms and petals, apart from stigma extracts or kaempferol using animal model of depression.

Table 9.
Results of clinical studies on the antidepressant effect of bioactive chemical compounds contained in C. sativus L. stigmas administered as capsules, tablets, or extracts.

Table 10.
Results of clinical studies on the antidepressant effect of bioactive chemical compounds contained in C. sativus L. tepals and stigmas administered to patients as capsules and as a combination with saffron capsules.
Although the results of clinical trials clearly suggest that saffron reduces the severity of depression based on Hamilton Depression Rating Scale (HAM-D) and Beck’s Depression Inventory (BDI) scores, the optimum dose and duration of treatment is still unclear [75].
Saffron and its bioactive constituents (crocetin esters, picrocrocin, and safranal) may be considered as a potential adjuvant in the form of anti-depressants in the future drug formulations. Recently, they seem to be a suitable candidate for the management of anxiety, depression, neuropsychiatric disorders and the other long-term effects including subacute and chronic abnormalities of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection such as fatigue, dyspnoea, cognitive problems, sleep abnormalities, and deterioration in the quality of life. Detailed research on dosage, methods of administration and others needs to be undertaken to explore the potential of saffron in managing the health issues arising due to the COVID-19 pandemic [397,398]. Moreover, crocin appears to reduce the COVID-19-related cytokine cascade and downregulate angiotensin-converting enzyme 2 (ACE2) gene expression. Lastly, in silico studies suggest that saffron’s astragalin and crocin could have inhibitory actions on the SARS-CoV-2 protease and spike protein, respectively. However, future appropriate randomised clinical trials using biomarkers as surrogates to assess inflammatory status should be designed in order to assess the clinical efficacy of saffron and allow its use as an adjunct treatment modality, particularly in resource-poor settings where access to drugs may be limited [399]. Soheilipur and co-workers claim [400] that the oral use of a single-dose of 40 mg saffron extract is effective in alleviating anxiety among the candidates for coronary angiography (CA), while lippia extract (capsule 40 mg; Lippia citriodora Kunth) and saffron–lippia (20 mg:20 mg) extract combination had no significant effects on their anxiety.
Saffron application is recognised as a promising natural and safe nutritional strategy to improve sleep duration and quality. The investigations carried out by Shahdadi et al. [401] revealed that daily (between 12 noon and 2 pm) intake of a 300 mg saffron capsule after lunch for a week was effective in reducing anxiety and improving the quality of sleep among diabetic patients. Six weeks of saffron extract supplementation (15 mg/day) to the subjects presenting with mild-to-moderate sleep disorders associated with anxiety led to an increased time in bed assessed by actigraphy, to an improved ease of getting to sleep as evaluated by the LSEQ (the Leeds sleep evaluation) questionnaire, and to an improved sleep quality, sleep latency, sleep duration, and global scores evaluated by the PSQI questionnaire (Pittsburgh Sleep Quality Index) [402]. Standardised saffron extract (affron®; 14 mg twice daily per 28-days) improved sleep quality in adults with self-reported sleep problems. The beneficial effect of saffron was manifested by improvements in ISI total score (The Insomnia Severity Index), RSQ total score (the Restorative Sleep Questionnaire), and PSD (Pittsburgh Sleep Diary) sleep quality ratings [403]. Further investigations concerning four weeks of treatment with affron® (14 mg, or 28 mg 1 h before bed) revealed improvements in sleep quality ratings assessed with Pittsburgh Sleep Diary, mood ratings after awakening (Profile of Mood States), the ISQ total score (Insomnia Symptom Questionnaire), and ISQ insomnia classifications without affecting the score of the Restorative Sleep Questionnaire and the Functional Outcomes of Sleep Questionnaire. Moreover, saffron supplementation was associated with increases in evening melatonin concentrations but did not affect evening cortisol. Sleep improvements were similar for the two saffron doses with no reported significant adverse effects. [404]. Results of the studies on the effect of crocetin on on sleep quality in healthy adult participants with mild sleep complaint assessed showed that supplementation with this bioactive compound contributes to sleep maintenance, leading to improved subjective sleep quality. This beneficial effect of two intervention periods of 14 days each, separated by a 14 day wash-out period, was manifested with an increase in an objective sleep parameters (delta power) measured using single-channel electroencephalography and improvements in the subjective sleep parameters sleepiness on rising and feeling refreshed assessed with using the Oguri–Shirakawa–Azumi Sleep Inventory, Middle-age and Aged version (OSA-MA).There were no significant differences in the other sleep parameters, including sleep latency, sleep efficiency, total sleep time, and wake after sleep onset [405].
A single study indicated that saffron odor was effective in treating menstrual distress by relieving the symptoms of premenstrual syndrome (PMS) and alleviating dysmenorrhea (menstrual pain) as well as helping to control irregular menstruation. As Fukui and co-workers claim [406], healthy woman with a normal sense of smell exposed to saffron aroma for 20 min experienced a decrease in salivatory cortisol and increase in 17-β estradiol level in both the follicular and luteal phases, which was accompanied with a decrease in anxiety measured using the State-Trait Anxiety Inventory (STAI). It was the first evidence of beneficial psychological and neuroendocrinological effects of saffron odour.
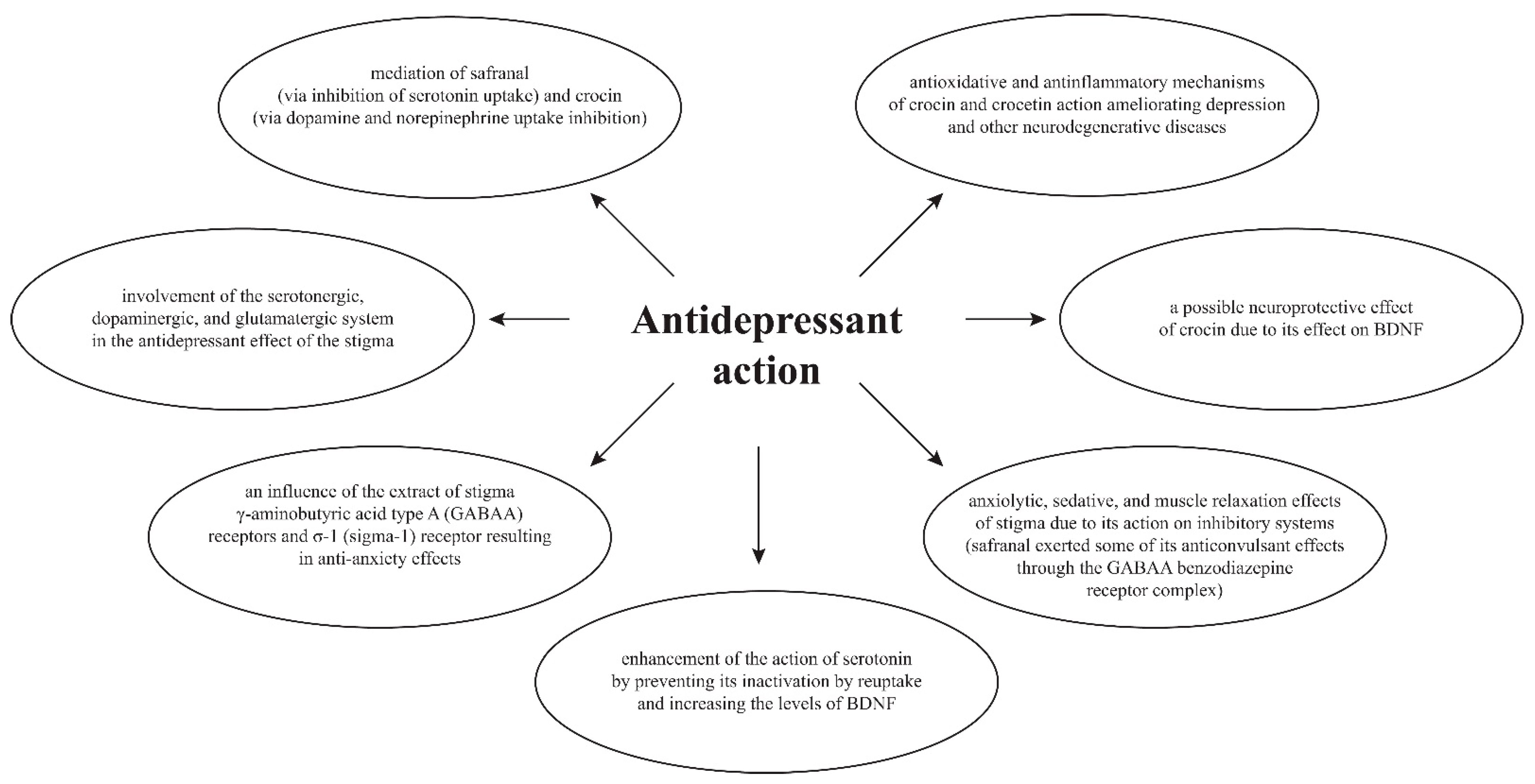
4.3. Mechanism of Antidepressant Action
The mechanism of the in vitro and in vivo antidepressant action of C. sativus stigmas is attributed to e.g., crocin, which inhibits monoamine (noradrenaline and dopamine) reuptake, and safranal, which inhibits serotonin reuptake, and to their action towards GABAergic (gamma-aminobutyric acid) receptors and neurotrophic effects, e.g., through activation of BDNF (brain-derived neurotrophic factor). Stigmas of C. sativus (called saffron) have been demonstrated to contain an antagonist of postsynaptic NMDA (N-methyl-d-aspartate) receptors [71,407,408]. It has been proven that C. sativus modulates the levels of neurotransmitters, especially serotonin, in the brain by inhibiting serotonin reuptake, thereby retaining serotonin in the brain longer [409] (Figure 3).
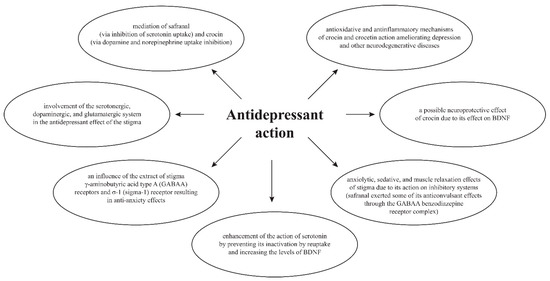
Figure 3.
Mechanism of antidepressant action [216,228,231,293,345,364,374,375,410].
In other studies, increased levels of CREB, BDNF, and VGF in the hippocampus were found [229,294]. There is strong evidence that VGF and BDNF are involved in depressive disorders and transcription thereof is CREB-dependent. The neuropeptide VGF enhances hippocampal synaptic activity and is involved in energy balance and homeostasis regulation. In turn, BDNF, which is widely expressed in the mammalian brain, is implicated in the survival of neurons during hippocampal development, neural regeneration, synaptic transmission, synaptic plasticity, and neurogenesis [269,270]. As reported by Asrari et al. [353], there is mediation of the P-CREB protein in cerebellum, which is consistent with the increased expression of this protein in the cerebellum described by Ghasemi et al. [229]. There is evidence that the cerebellum not only plays a role in motor function and coordination of movement, but also contributes to an important role in emotion and cognition processing. To sum up among many different proposed mechanisms explaining C. sativus stigma and petals effectiveness in the depression treatment, the most important is the one involving the anti-inflammatory and antioxidant effects, followed by the action on neurotransmitters in favour of the hypothesis of their deficiency in depression. Neurotrophic factors, particularly BDNF, are also of interest since they are involved, even if indirectly, in the regulation of neurotransmitters such as 5- HT, DA, glutamate and GABA and in various types of signalling such as CREB [411,412].
Studies of multiple genes have yielded some positive results regarding the usefulness of genotyping cytochrome P450 enzymes (CYP450) in the treatment of depression in groups of patients, but the choice of medications for a specific patient is not still established [413,414]. The results of investigations conducted on male Wistar albino rats receiving safranal (4, 20, and 100 mg/kg/day) or intraperitoneal injections of crocin (4, 20, and 100 mg/kg/day) indicate that both these compounds increase the total protein content and determine the metabolic activity of liver microsomal CYP450 isoforms (CYP3A, CYP2C11, CYP2B, and CYP2A) [410]. It was found that, in general, crocin markedly reduced and safranal significantly enhanced the metabolic activity of all the CYP enzymes mentioned above, except for changes in CYP2A activity induced by safranal. Therefore, the authors claim that crocin and safranal could increase the risk of interactions with co-administered substances metabolised by cytochrome P450 enzymes.
5. Challenges for Further Research
The scientific knowledge of the beneficial or negative impact of herbal treatment of depression is incomplete. Further investigations should focus on: (1) adequate methods of extraction of selected biologically active compounds and practical pharmaceutical applications thereof; (2) promotion of trust in phytotherapy and the use of biotechnological procedures to ensure the biodiversity of the product; (3) the use of genetic technologies to obtain good quality and high concentrations of effective phytochemicals that can be used in the future to support treatment of depression as progress in herbal psychopharmacology; (4) standard medical therapies based on herbal products, including changes in the regulations, standardisation, and financing of research on selected phytochemicals with anti-depressant effects; (5) insightful and more detailed analyses of natural compounds in terms of the basic mechanisms involved in the anti-depressant actions and justifying the application of selected plant species in the therapeutic practice of depression, taking into account antidepressant properties of these plants that have already been confirmed by scientists; (6) thorough clinical trials of selected phytochemicals—effective substances in depression treatment facilitating production of antidepressant drugs and antioxidants from these substances; (7) confirmation of the safety and efficacy of action in the treatment of depression, which will support the decision to use these compounds (as in the case of pharmaceutical drugs).
6. Conclusions
Crocus sativus, commonly known as saffron crocus, is native to the Western and Eastern Asia and Southern Europe. For centuries, it has been used in traditional Asian medicine as an agent for healing various health problems, including infections, pain, inflammation, chronic fatigue, insomnia, memory impairment, mood and personality disorders (anxiety, depression), and other mental illnesses. The medicinal activity of C. sativus extracts in alleviation of inflammation and central nervous system disorders, including depression, has been confirmed in the most recent basic animal (rodent) studies and human clinical trials. A number of in vitro, in vivo, and clinical trials have demonstrated that both dried stigmas and petals of C. sativus (water and alcohol extracts) as well as their ingredients are safe and effective antidepressants. Saffron stigma, bulbs and petals and its bioactive compounds may be considered as a potential adjuvant in the form of anti-depressant in future drug formulations. Their efficacy is similar to current antidepressant medications such as fluoxetine, imipramine, and citalopram, but fewer side effects are reported. The active compounds of aqueous and alcoholic crocus extracts exhibiting antidepressant activity include unique hydrophilic crocin carotenoids, i.e., monoglycosyl or di-glycosyl esters of crocetin, hydrophobic crocetin, and terpenoid safranal. The following mechanisms of the antidepressant action of C. sativus components are proposed: (1) inhibition of monoamine (dopamine, norepinephrine, serotonin) reuptake, (2) N-methyl-d-aspartate (NMDA) receptor antagonism, and (3) gamma-aminobutyric acid (GABA)-α agonism. Crocin acts via inhibition of dopamine and norepinephrine uptake, while safranal acts via serotonin. The antidepressant and neuroprotective effect of C. sativus extracts and their components is associated with anti-inflammatory and antioxidant activity. This activity is manifested by e.g., mood improvement, alleviation of anxiety symptoms, beneficial effects on learning and remembering, and a positive influence on the emotional sphere. However, due to many limitations presented in the papers cited in this protocol, there is a need for conducting further experiments to confirm the current results on the effectiveness of the antidepressant activity of C. sativus extract and its components and to elucidate the mechanisms of their action fully. Research reported by many authors has documented the application of herbal formulations in treatment of depression, insomnia, and anxiety, but detailed research on dosage, methods of administration and others needs to be undertaken to explore their potential in managing the health issues Although phytochemicals are natural substances and should therefore be safe, side effects have been noted due to contamination of preparation or drug interactions.
Author Contributions
R.M.-G. supervision; M.C. and R.M.-G. conceptualisation; methodology; collection and analysis of literature reports; compilation of tables with modern research results; writing—original draft preparation; writing—review and editing; K.T. and M.M.S. compilation of tables, original draft preparation and preparation of graphic abstract, collection and analysis of literature reports, writing and editing the paper. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Fried, E.I.; Nesse, R.M. Depression is not a consistent syndrome: An investigation of unique symptom patterns in the STAR* D study. J. Affect. Disord. 2015, 172, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Fried, E.I.; Nesse, R.M. Depression sum-scores don’t add up: Why analyzing specific depression symptoms is essential. BMC Med. 2015, 13, 72. [Google Scholar] [CrossRef] [PubMed]
- Sadock, B.J.; Sadock, V.A.; Ruiz, P. Mood disorders. In Kaplan and Sadock’s Synopsis of Psychiatry. Behavioral Sciences/Clinical Psychiatry, 11th ed.; Sadock, B.J., Ed.; Wolters Kluwer: Philadelphia, PA, USA, 2015; pp. 347–386. [Google Scholar]
- Otte, C.; Gold, S.M.; Penninx, B.W.; Pariante, C.M.; Etkin, A.; Fava, M.; Mohr, D.C.; Schatzberg, A.F. Major depressive disorder. Nat. Rev. Dis. Primers 2016, 2, 16065. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.; Seedat, S. The diagnosis and management of depression in the era of the DSM-5. S. Afr. Fam. Pract. 2018, 22, 22–28. [Google Scholar] [CrossRef]
- Clack, S.; Ward, T. The classification and explanation of depression. Behav. Chang. 2019, 36, 41–55. [Google Scholar] [CrossRef]
- Giannelli, F.R. Major depressive disorder. JAAPA 2020, 33, 19–20. [Google Scholar] [CrossRef]
- Nussbaum, A.M. Questionable agreement: The experience of depression and DSM-5 major depressive disorder criteria. J. Med. Philos. 2020, 45, 623–643. [Google Scholar] [CrossRef]
- Lindhardt, A. Psykiske sydgomme. In Almen Praksis, 3rd ed.; Hippe, E., Ed.; ForlagsGruppen ApS: Copenhaga, Denmark, 2007; Volume 794, pp. 771–776. (In Danish) [Google Scholar]
- Huarcaya-Victoria, J.; Bojórquez-De la Torre, J.; De la Cruz-Oré, J. Factor structure of cotard’s syndrome: Systematic review of case reports. Rev. Colomb. Psiquiatr. 2020, 49, 187–193. [Google Scholar] [CrossRef]
- Makara-Studzińska, M.; Rolla-Szczepańska, R.; Urbańska, A.; Nowakowska-Domagała, K.; Stecz, P. Anxiety and depression in patients infected with Borrelia burgdorferi. Eur. J. Psychiatry 2017, 31, 165–171. [Google Scholar] [CrossRef]
- Tran, B.X.; Ho, R.; Ho, C.S.; Latkin, C.A.; Phan, H.T.; Ha, G.H.; Vu, G.T.; Ying, J.; Zhang, M.W. Depression among patients with HIV/AIDS: Research development and effective interventions (GAPRESEARCH). Int. J. Environ. Res. Public Health. 2019, 16, 1772. [Google Scholar] [CrossRef]
- Zhu, Q.Y.; Huang, D.S.; Lv, J.D.; Guan, P.; Bai, X.H. Prevalence of perinatal depression among HIV-positive women: A systematic review and meta-analysis. BMC Psychiatry 2019, 19, 330. [Google Scholar] [CrossRef]
- Banasiewicz, J.; Zaręba, K.; Bińkowska, M.; Rozenek, H.; Wójtowicz, S.; Jakiel, G. Perinatal predictors of postpartum depression: Results of a retrospective comparative study. J. Clin. Med. 2020, 9, 2952. [Google Scholar] [CrossRef]
- Fiske, A.; Wetherell, J.L.; Gatz, M. Depression in older adults. Annu. Rev. Clin. Psychol. 2009, 5, 363–389. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Yang, D.; Lei, B.; Yan, C.; Tian, Y.; Huang, X.; Lei, J. The short-and long-term effectiveness of mother–infant psychotherapy on postpartum depression: A systematic review and meta-analysis. J. Affect. Disord. 2020, 260, 670–679. [Google Scholar] [CrossRef]
- Kong, X.; Zheng, K.; Tang, M.; Kong, F.; Zhou, J.; Diao, L.; Wu, S.; Jiao, P.; Su, T.; Dong, Y. Prevalence and factors associated with depression and anxiety of hospitalized patients with COVID-19. MedRxiv 2020. [Google Scholar] [CrossRef]
- Rao, W.W.; Zhu, X.M.; Zong, Q.Q.; Zhang, Q.; Hall, B.J.; Ungvari, G.S.; Xiang, Y.T. Prevalence of prenatal and postpartum depression in fathers: A comprehensive meta-analysis of observational surveys. J. Affect. Disord. 2020, 15, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Serfaty, M.; King, M.; Nazareth, I.; Moorey, S.; Aspden, T.; Mannix, K.; Jones, L. Effectiveness of cognitive–behavioural therapy for depression in advanced cancer: CanTalk randomised controlled trial. Br. J. Psychiatry 2020, 216, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Zaręba, K.; Banasiewicz, J.; Rozenek, H.; Wójtowicz, S.; Jakiel, G. Peripartum predictors of the risk of postpartum depressive disorder: Results of a case-control study. Int. J. Environ. Res. Public Health 2020, 17, 8726. [Google Scholar] [CrossRef]
- Cooley, C.; Park, Y.; Ajilore, O.; Leow, A.; Nyenhuis, S.M. Impact of interventions targeting anxiety and depression in adults with asthma. J. Asthma 2022, 59, 273–287. [Google Scholar] [CrossRef]
- Santomauro, D.F.; Mantilla Herrera, A.M.; Shadid, J.; Zheng, P.; Ashbaugh, C.; Pigott, D.M.; Abbafati, C.; Adolph, C.; Amlag, J.O.; Aravkin, A.Y.; et al. Global prevalence and burden of depressive and anxiety disorders in 204 countries and territories in 2020 due to the COVID-19 pandemic. Lancet 2021, 398, 1700–1712. [Google Scholar] [CrossRef]
- Oriolo, G.; Grande, I.; Martin-Santos, R.; Vieta, E.; Carvalho, A.F. Pathways driving neuroprogression in depression: The role of immune activation. In Inflammation and Immunity in Depression: Basic Science and Clinical Applications; Baune, B.T., Ed.; Academic Press Elsevier: London, UK, 2018; pp. 173–198. [Google Scholar]
- Ruiz, N.A.L.; Del Ángel, D.S.; Olguín, H.J.; Silva, M.L. Neuroprogression: The hidden mechanism of depression. Neuropsychiatr. Dis. Treat. 2018, 14, 2837–2845. [Google Scholar] [CrossRef] [PubMed]
- Hegeman, J.M.; De Waal, M.W.M.; Comijs, H.C.; Kok, R.M.; Van Der Mast, R.C. Depression in later life: A more somatic presentation? J. Affect. Disord. 2015, 170, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Hovatta, I. Genetics: Dynamic cellular aging markers associated with major depression. Curr. Biol. 2015, 25, R409–R411. [Google Scholar] [CrossRef] [PubMed]
- Verhoeven, J.; Révész, D.; Van Oppen, P.; Epel, E.; Wolkowitz, O.; Penninx, B. Anxiety disorders and accelerated cellular ageing. Br. J. Psychiatr. 2015, 206, 371–378. [Google Scholar] [CrossRef]
- Yu, R.; Woo, J.W. Exploring the link between depression and accelerated cellular aging: Telomeres hold the key. Res. Rep. Biochem. 2015, 6, 1–12. [Google Scholar] [CrossRef][Green Version]
- Agnafors, S.; Kjellström, A.N.; Torgerson, J.; Rusner, M. Somatic comorbidity in children and adolescents with psychiatric disorders. Eur. Child Adolesc. Psychiatry 2019, 28, 1517–1525. [Google Scholar] [CrossRef]
- González, A.C.T.; Ignácio, Z.M.; Jornada, L.K.; Réus, G.Z.; Abelaira, H.M.; Santos, M.; Ceretta, L.B.; Quevedo, J.L.D. Depressive disorders and comorbidities among the elderly: A population-based study. Rev. Bras. Geriatr. Gerontol. 2016, 19, 95–103. [Google Scholar] [CrossRef]
- Schaakxs, R.; Comijs, H.C.; Lamers, F.; Beekman, A.T.F.; Penninx, B. Age-related variability in the presentation of symptoms of major depressive disorder. Psychol. Med. 2017, 47, 543–552. [Google Scholar] [CrossRef]
- Steffen, A.; Nübel, J.; Jacobi, F.; Bätzing, J.; Holstiege, J. Mental and somatic comorbidity of depression: A comprehensive cross-sectional analysis of 202 diagnosis groups using German nationwide ambulatory claims data. BMC Psychiatry 2020, 20, 2–15. [Google Scholar] [CrossRef]
- Gassen, N.C.; Rein, T. Is there a role of autophagy in depression and antidepressant action? Front. Psychiatry 2019, 10, 337. [Google Scholar] [CrossRef]
- Solek, P.; Koszla, O.; Mytych, J.; Badura, J.; Chelminiak, Z.; Cuprys, M.; Fraczek, J.; Tabecka-Lonczynska, A.; Koziorowski, M. Neuronal life or death linked to depression treatment: The interplay between drugs and their stress-related outcomes relate to single or combined drug therapies. Apoptosis 2019, 24, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Collo, G.; Merlo Pich, E. A human translational model based on neuroplasticity for pharmacological agents potentially effective in treatment-resistant depression: Focus on dopaminergic system. Neural Regen Res. 2020, 15, 1027–1029. [Google Scholar] [CrossRef] [PubMed]
- Endres, D.; Rauer, S.; Venhoff, N.; Süß, P.; Dersch, R.; Runge, K.; Fiebich, B.L.; Nickel, K.; Matysik, M.; Maier, S.; et al. Probable autoimmune depression in a patient with multiple sclerosis and antineuronal antibodies. Front. Psychiatry 2020, 11, 745. [Google Scholar] [CrossRef] [PubMed]
- Hidayat, R.; Saleh, M.I.; Parisa, N. Neuronal cell death induces depressive disorder in rats depression-like behaviors caused by chronic stress. Sci. Psychiatr. 2020, 13, 16–24. [Google Scholar] [CrossRef]
- Li, Y.; Jia, Y.; Wang, D.; Zhuang, X.; Li, Y.; Guo, C.; Chu, H.; Zhu, F.; Wang, J.; Wang, X.; et al. Programmed cell death 4 as an endogenous suppressor of BDNF translation is involved in stress-induced depression. Mol. Psychiatry 2021, 26, 2316–2333. [Google Scholar] [CrossRef] [PubMed]
- Price, R.B.; Duman, R. Neuroplasticity in cognitive and psychological mechanisms of depression: An integrative model. Mol. Psych. 2020, 25, 530–543. [Google Scholar] [CrossRef]
- Kostrubiak, D.E.; Vacchi-Suzzi, C.; Smith, D.M.; Meliker, J.R. Blood cadmium and depressive symptoms: Confounded by cigarette smoking. Psychiatry Res. 2017, 256, 444–447. [Google Scholar] [CrossRef]
- Baranyi, A.; Meinitzer, A.; Rothenhäusler, H.B.; Amouzadeh-Ghadikolai, O.; Lewinski, D.V.; Breitenecker, R.J.; Herrmann, M. Metabolomics approach in the investigation of depression biomarkers in pharmacologically induced immune-related depression. PLoS ONE 2018, 29, e0208238. [Google Scholar] [CrossRef]
- Clarke, T.K.; Zeng, Y.; Navrady, L.; Xia, C.; Haley, C.; Campbell, A.; Navarro, P.; Amador, C.; Adams, M.J.; Howard, D.M.; et al. Genetic and environmental determinants of stressful life events and their overlap with depression and neuroticism. Wellcome Open Res. 2018, 3, 11. [Google Scholar] [CrossRef]
- Gładka, A.; Rymaszewska, J.; Zatoński, T. Impact of air pollution on depression and suicide. Int. J. Occup. Med. Environ. Health. 2018, 31, 711–721. [Google Scholar] [CrossRef]
- Jurczak, A.; Brodowska, A.; Szkup, M.; Prokopowicz, A.; Karakiewicz, B.; Łój, B.; Kotwas, A.; Brodowska, A.; Grochans, E. Influence of Pb and Cd levels in whole blood of postmenopausal women on the incidence of anxiety and depressive symptoms. Ann. Agric. Environ. Med. 2018, 25, 219–223. [Google Scholar] [CrossRef]
- Losiak, W.; Blaut, A.; Kłosowska, J.; Losiak-Pilch, J. Stressful life events, cognitive biases, and symptoms of depression in young adults. Front. Psychol. 2019, 10, 2165. [Google Scholar] [CrossRef] [PubMed]
- Warne, N.; Collishaw, S.; Rice, F. 2019. Examining the relationship between stressful life events and overgeneral autobiographical memory in adolescents at high familial risk of depression. Memory 2019, 27, 314–327. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, X.; Tu, J.; Rong, H.; Werz, O.; Xinchun Chen, X. The interplay between depression and tuberculosis. J. Leukoc. Biol. 2019, 106, 749–757. [Google Scholar] [CrossRef]
- Brietzke, E.; Magee, T.; Freire, R.C.R.; Gomes, F.A.; Milev, R. Three insights on psychoneuroimmunology of mood disorders to be taken from the COVID-19 pandemic. Brain Behav. Immun. Health. 2020, 5, 100076. [Google Scholar] [CrossRef]
- Chibowska, K.; Chlubek, D.; Baranowska-Bosiacka, I. Exposure to lead in the pre- and neonatal periods may result in brain inflammation. Pomeranian J. Life Sci. 2020, 66, 29–38. [Google Scholar]
- Yang, L.; Zhao, Y.; Wang, Y.; Liu, L.; Zhang, X.; Li, B.; Cui, R. The Effects of Psychological Stress on Depression. Curr. Neuropharmacol. 2015, 13, 494–504. [Google Scholar] [CrossRef]
- Dunlop, B.W.; Nemeroff, C.B. The role of dopamine in the pathophysiology of depression. Arch. Gen. Psychiat. 2007, 64, 327–337. [Google Scholar] [CrossRef]
- Belzung, C.; Willner, P.; Philippot, P. Depression: From psychopathology to pathophysiology. Curr. Opin. Neurobiol. 2015, 30, 24–30. [Google Scholar] [CrossRef]
- Verduijn, J.; Milaneschi, Y.; Schoevers, R.A.; van Hemert, A.V.; Beekman, A.T.F.; Penninx, B. Pathophysiology of major depressive disorder: Mechanisms involved in etiology are not associated with clinical progression. Transl. Psychiatry 2015, 5, e649. [Google Scholar] [CrossRef]
- Ménard, C.; Hodes, G.E.; Russo, S.J. Pathogenesis of depression: Insights from human and rodent studies. Neuroscience 2016, 321, 138–162. [Google Scholar] [CrossRef] [PubMed]
- Dmitrzak-Weglarz, M.; Reszka, E. Pathophysiology of depression: Molecular regulation of melatonin homeostasis–current status. Neuropsychobiology 2017, 76, 117–129. [Google Scholar] [CrossRef]
- Jesulola, E.; Micalos, P.; Baguley, I.J. Understanding the pathophysiology of depression: From monoamines to the neurogenesis hypothesis model-are we there yet? Behav. Brain Res. 2018, 341, 79–90. [Google Scholar] [CrossRef]
- Bakalov, D.; Hadjiolova, R.; Pechlivanova, D. Pathophysiology of depression and novel sources of phytochemicals for its treatment–a systematic review. Acta Med. Bulg. 2020, 47, 69–74. [Google Scholar] [CrossRef]
- Spellman, T.; Liston, C. Toward circuit mechanisms of pathophysiology in depression. Am. J. Psychiatry 2020, 177, 381–390. [Google Scholar] [CrossRef]
- Chiriţă, A.L.; Gheorman, V.; Bondari, D.; Rogoveanu, I. Current understanding of the neurobiology of major depressive disorder. Rom. J. Morphol. Embryol. 2015, 56 (Suppl. S2), 651–658. [Google Scholar]
- Qi, S.; Yang, X.; Zhao, L.; Calhoun, V.D.; Perrone-Bizzozero, N.; Liu, S.; Jiang, R.; Jiang, T.; Sui, J.; Ma, X. MicroRNA132 associated multimodal neuroimaging patterns in unmedicated major depressive disorder. Brain 2018, 141, 916–926. [Google Scholar] [CrossRef]
- Zhang, H.F.; Mellor, D.; Peng, D.H. Neuroimaging genomic studies in major depressive disorder: A systematic review. CNS Neurosci. Ther. 2018, 24, 1020–1036. [Google Scholar] [CrossRef]
- Howard, D.M.; Adams, M.J.; Clarke, T.K.; Hafferty, J.D.; Gibson, J.; Shirali, M.; Coleman, J.R.; Hagenaars, S.P.; Ward, J.; Wigmore, E.M.; et al. Genome-wide meta-analysis of depression identifies 102 independent variants and highlights the importance of the prefrontal brain regions. Nat. Neurosci. 2019, 22, 343–352. [Google Scholar] [CrossRef]
- Barbu, M.C.; Shen, X.; Walker, R.M.; Howard, D.M.; Evans, K.L.; Whalley, H.C.; Porteous, D.J.; Morris, S.W.; Deary, I.J.; Zeng, Y.; et al. Epigenetic prediction of major depressive disorder. Mol. Psychiatry 2021, 26, 5112–5123. [Google Scholar] [CrossRef]
- Wray, N.R.; Ripke, S.; Mattheisen, M.; Trzaskowski, M.; Byrne, E.M.; Abdellaoui, A.; Bacanu, S.A. Genome-wide association analyses identify 44 risk variants and refine the genetic architecture of major depression. Nat. Genet. 2018, 50, 668–681. [Google Scholar] [CrossRef] [PubMed]
- McKeever, A.; Agius, M.; Mohr, P. A review of the epidemiology of major depressive disorder and of its consequences for society and the individual. Psychiatr. Danub. 2017, 1, 222–231. [Google Scholar]
- Coretti, S.; Rumi, F.; Cicchetti, A. The social cost of major depression: A systematic review. Rev. Eur. Stud. 2019, 11, 73–83. [Google Scholar] [CrossRef]
- König, H.; König, H.; Konnopka, A. The excess costs of depression: A systematic review and meta-analysis. Epidem. Psychiat. Sci. 2020, 29, E30. [Google Scholar] [CrossRef] [PubMed]
- Taciak, P.P.; Lysenko, N.; Mazurek, A.P. Drugs which influence serotonin transporter and serotonergic receptors: Pharmacological and clinical properties in the treatment of depression. Pharmacol. Rep. 2018, 70, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Rong, H.; Xu, S.X.; Zeng, J.; Yang, Y.J.; Zhao, J.; Lai, W.T.; Chen, L.C.; Deng, W.F.; Zhang, X.; Zhang, Y.L.; et al. Study protocol for a parallel-group, double-blinded, randomized, controlled, noninferiority trial: The effect and safety of hybrid electroconvulsive therapy (Hybrid-ECT) compared with routine electroconvulsive therapy in patients with depression. BMC Psychiatry 2019, 19, 344. [Google Scholar] [CrossRef]
- Tabeshpour, J.; Sobhani, F.; Sadjadi, S.A.; Hosseinzadeh, H.; Mohajeri, S.A.; Rajabi, O.; Taherzadeh, Z.; Eslami, S. A double-blind, randomized, placebo-controlled trial of saffron stigma (Crocus sativus L.) in mothers suffering from mild-to-moderate postpartum depression. Phytomedicine 2017, 36, 145–152. [Google Scholar] [CrossRef]
- Shafiee, M.; Arekhi, S.; Omranzadeh, A.; Sahebkar, A.; Shafiee, M.; Arekhi, S.; Omranzadeh, A.; Sahebkar, A. Saffron in the treatment of depression, anxiety and other mental disorders: Current evidence and potential mechanisms of action. J. Affect. Disord. 2018, 227, 330–337. [Google Scholar] [CrossRef]
- Ha, A.; Mehdi, S.; Kl, K. Medicinal herbs and phytochemicals used in the treatment of depression: A review. Asian J. Pharm. Clin. Res. 2019, 12, 8–14. [Google Scholar]
- Choi, J.E.; Park, D.M.; Chun, E.; Choi, J.J.; Seo, J.H.; Kim, S.; Park, Y.C.; Jung, I.C. Control of stress-induced depressive disorders by So-ochim-tang-gamibang, a Korean herbal medicine. J. Ethnopharmacol. 2017, 196, 141–150. [Google Scholar]
- Matraszek-Gawron, R.; Chwil, M.; Terlecka, P.; Skoczylas, M.M. Recent studies on anti-depressant bioactive substances in selected species from the genera Hemerocallis and Gladiolus: A systematic review. Pharmaceuticals 2019, 12, 172. [Google Scholar] [CrossRef]
- Tóth, B.; Hegyi, P.; Lantos, T.; Szakacs, Z.; Keremi, B.; Varga, G.; Tenk, J.; Petervari, E.; Balasko, M.; Rumbus, Z.; et al. The efficacy of saffron in the treatment of mild to moderate depression: A meta-analysis. Planta Med. 2019, 85, 24–31. [Google Scholar] [CrossRef]
- Lin, H.Y.; Tsai, J.C.; Wu, L.Y.; Peng, W.H. Reveals of new candidate active components in Hemerocallis radix and its anti-depression action of mechanism based on network pharmacology approach. Int. J. Mol. Sci. 2020, 21, 1868. [Google Scholar] [CrossRef]
- Lee, W.H.; Kim, D.H.; Lee, T.H. Effect of mixture extracted from Bupleuri Radix and Physalidis Herba on the LPS-induced depression in rats. Korea J. Herbol. 2015, 30, 69–76. [Google Scholar]
- Lim, D.W.; Kim, J.G.; Han, T.; Jung, S.K.; Lim, E.Y.; Han, D.; Kim, Y.T. Analgesic effect of Ilex paraguariensis extract on postoperative and neuropathic pain in rats. Biol. Pharm. Bull. 2015, 38, 1573–1579. [Google Scholar] [CrossRef][Green Version]
- Lutomski, P.; Goździewska, M.; Florek-Łuszczki, M. Health properties of yerba mate. Ann. Agric. Environ. Med. 2020, 27, 310–313. [Google Scholar] [CrossRef]
- Keefe, J.R.; Mao, J.J.; Soeller, I.; Li, Q.S.; Amsterdam, J.D. Short-term open-label chamomile (Matricaria chamomilla L.) therapy of moderate to severe generalized anxiety disorder. Phytomedicine 2016, 23, 1699–1705. [Google Scholar] [CrossRef]
- Benneh, C.K.; Biney, R.P.; Adongo, D.W.; Mante, P.K.; Ampadu, F.A.; Tandoh, A.; Jato, J.; Woode, E. Anxiolytic and antidepressant effects of Maerua angolensis DC. Stem bark extract in mice. Depress. Res. Treat. 2018, 17, 2018. [Google Scholar]
- Shehu, A.; Anyip, B.; Magaji, M.G. Antidepressant effect of methanol root bark extract of Acacia seyal Del. (Fabaceae): Possible involvement of the inflammatory pathway. Trop. J. Pharm. Res. 2020, 19, 1459–1464. [Google Scholar] [CrossRef]
- Choi, J.H.; Lee, M.J.; Chang, Y.; Lee, S.; Kim, H.J.; Lee, S.W.; Kim, Y.O.; Cho, I.H. Valeriana fauriei exerts antidepressant-like effects through anti-inflammatory and antioxidant activities by inhibiting brain-derived neurotrophic factor associated with chronic restraint stress. Rejuvenat. Res. 2020, 23, 245–255. [Google Scholar] [CrossRef]
- Wang, L.; Sun, Y.; Zhao, T.; Li, Y.; Zhao, X.; Zhang, L.; Wu, L.; Zhang, L.; Zhang, T.; Wei, G.; et al. Antidepressant effects and mechanisms of the total iridoids of Valeriana jatamansi on the brain-gut Axis. Planta Med. 2020, 86, 172–179. [Google Scholar] [PubMed]
- Chwil, M.; Matraszek-Gawron, R.; Terlecka, P.; Kostryco, M. Plant antidepressants in selected species from the family Fabaceae—A review. Ann. Hortic. 2017, 27, 58–68. [Google Scholar] [CrossRef]
- Wurglics, M.; Schubert-Zsilavecz, M. Hypericum perforatum: A ‘modern’ herbal antidepressant. Clin. Pharmacokinet. 2006, 45, 449–468. [Google Scholar] [CrossRef]
- Lazzara, S.; Napoli, E.; Carrubba, A. Hypericum spp.: A resource from wild Mediterranean flora for the treatment of mild depression. Bioact. Phytochem. Perspect. Biol. Med. 2015, 3, 337–354. [Google Scholar]
- Zhai, X.J.; Chen, F.; Chen, C.; Zhu, C.R.; Lu, Y.N. LC–MS/MS based studies on the anti-depressant effect of hypericin in the chronic unpredictable mild stress rat model. J. Ethnopharmacol. 2015, 169, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Maher, A.R.; Hempel, S.; Apaydin, E.; Shanman, R.M.; Booth, M.; Miles, J.N.; Sorbero, M.E. St. John’s Wort for major depressive disorder: A systematic review. Rand. Health Q. 2016, 5, 12. [Google Scholar] [PubMed]
- Ng, Q.X.; Venkatanarayanan, N.; Ho, C.Y. Clinical use of Hypericum perforatum (St. John’s wort) in depression: A meta-analysis. J. Affect. Disord. 2017, 210, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Zirak, N.; Shafiee, M.; Soltani, G.; Mirzaei, M.; Sahebkar, A. Hypericum perforatum in the treatment of psychiatric and neurodegenerative disorders: Current evidence and potential mechanisms of action. J. Cell. Physiol. 2019, 234, 8496–8508. [Google Scholar] [CrossRef]
- Abdelhalim, A.; Karim, N.; Chebib, M.; Aburjai, T.; Khan, I.; Johnston, G.A.; Hanrahan, J. Antidepressant, anxiolytic and antinociceptive activities of constituents from Rosmarinus officinalis. J. Pharm. Pharm. Sci. 2015, 18, 448–459. [Google Scholar] [CrossRef]
- Guo, Y.; Xie, J.; Li, X.; Yuan, Y.; Zhang, L.; Hu, W.; Luo, H.; Yu, H.; Zhang, R. Antidepressant effects of rosemary extracts associate with anti-inflammatory effect and rebalance of gut microbiota. Front. Pharmacol. 2018, 9, 1126. [Google Scholar] [CrossRef]
- Ali, S.S.; Abd El Wahab, M.G.; Ayuob, N.N.; Suliaman, M. The antidepressant-like effect of Ocimum basilicum in an animal model of depression. Biotech. Histochem. 2017, 92, 390–401. [Google Scholar] [CrossRef]
- Guzmán-Gutiérrez, S.L.; Gómez-Cansino, R.; García-Zebadúa, J.C.; Jiménez-Pérez, N.C.; Reyes-Chilpa, R. Antidepressant activity of Litsea glaucescens essential oil: Identification of β-pinene and linalool as active principles. J. Ethnopharmacol. 2012, 143, 673–679. [Google Scholar] [CrossRef] [PubMed]
- El Hamdaoui, Y.; Zheng, F.; Fritz, N.; Ye, L.; Tran, M.A.; Schwickert, K.; Schirmeister, T.; Braeuning, A.; Lichtenstein, D.; Hellmich, U.A.; et al. Analysis of hyperforin (St. John’s wort) action at TRPC6 channel leads to the development of a new class of antidepressant drugs. Mol. Psychiatry 2022, 27, 5070–5085. [Google Scholar] [CrossRef]
- Ayres, A.S.; Santos, W.B.; Junqueira-Ayres, D.D.; Costa, G.M.; Ramos, F.A.; Castellanos, L.; Alves, J.S.; Asth, L.; de Medeiros, I.U.; Zucolotto, S.M.; et al. Monoaminergic neurotransmission is mediating the antidepressant-like effects of Passiflora edulis Sims fo. edulis. Neurosci. Lett. 2017, 660, 79–85. [Google Scholar] [CrossRef] [PubMed]
- da Silva, J.A.; da Costa, M.; da Alves, M.; da Braga, J.; da Lima, C.; da Pordeus, L. Effects of the single supplementation and multiple doses of Passiflora incarnata L. on human anxiety: A clinical trial, double-blind, placebo-controlled, randomized. Int. Arch. Med. 2017, 10. [Google Scholar] [CrossRef]
- Shahmohammadi, A.; Ramezanpour, N.; Mahdavi Siuki, M.; Dizavandi, F.; Ghazanfarpour, M.; Rahmani, Y.; Tahajjodi, R.; Babakhanian, M. The efficacy of herbal medicines on anxiety and depression in peri- and postmenopausal women: A systematic review and meta-analysis. Post Reprod Health. 2019, 25, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.; Pereira, J.A. On the effect of aromatherapy with Citrus fragrance in the therapy of major depressive disorder. J. Psych. Psychother. 2015, 5, 1000169. [Google Scholar]
- Sawamoto, A.; Okuyama, S.; Yamamoto, K.; Amakura, Y.; Yoshimura, M.; Nakajima, M.; Furukawa, Y. 3, 5, 6, 7, 8, 3′, 4′-Heptamethoxyflavone, a citrus flavonoid, ameliorates corticosterone-induced depression-like behavior and restores brain-derived neurotrophic factor expression, neurogenesis, and neuroplasticity in the hippocampus. Molecules 2016, 21, 541. [Google Scholar] [CrossRef]
- Petreanu, M.; Maia, P.; da Rocha Pittarello, J.L.; Loch, L.C.; Monache, F.D.; Perez, A.L.; Solano-Arias, G.; Filho, V.C.; de Souza, M.M.; Niero, R. Antidepressant-like effect and toxicological parameters of extract and withanolides isolated from aerial parts of Solanum capsicoides All. (Solanaceae). Naunyn-Schmiedeb. Arch. Pharmacol. 2019, 392, 979–990. [Google Scholar] [CrossRef]
- Dhingra, D.; Valecha, R. Evaluation of antidepressant-like activity of aqueous and ethanolic extracts of Terminalia bellirica Roxb. fruits in mice. Indian J. Exp. Biol. 2007, 45, 610–616. [Google Scholar]
- Mathew, S.J.; Shah, A.; Lapidus, K.; Clark, C.; Jarun, N.; Ostermeyer, B.; Murrough, J.W. Ketamine for treatment-resistant unipolar depression. CNS Drugs 2012, 26, 189–204. [Google Scholar] [CrossRef]
- Simplice, H.F.; Emery, D.; Hervé, N.A. Enhancing spatial memory: Anxiolytic and antidepressant effects of Tapinanthus dodoneifolius (DC) Danser in mice. Neurol. Res. Int. 2014, 2014, 974308. [Google Scholar] [CrossRef]
- Shahamat, Z.; Abbasi-Maleki, S.; Mohammadi, M.S. Evaluation of antidepressant-like effects of aqueous and ethanolic extracts of Pimpinella anisum fruit in mice. Avicenna J. Phytomed. 2016, 6, 322–328. [Google Scholar] [PubMed]
- Ngoupaye, G.T.; Bum, E.N.; Daniels, W.M. Antidepressant-like effects of the aqueous macerate of the bulb of Gladiolus dalenii Van Geel (Iridaceae) in a rat model of epilepsy-associated depression. BMC Complement. Altern. Med. 2013, 13, 272. [Google Scholar] [CrossRef] [PubMed]
- Ngoupaye, G.T.; Bum, E.N.; Ngah, E.; Talla, E.; Taiwe, G.S.; Rakotonirina, A.; Rakotonirina, S.V. The anticonvulsant and sedative effects of Gladiolus dalenii extracts in mice. Epilepsy Behav. 2013, 28, 450–456. [Google Scholar] [CrossRef]
- Xu, P.; Wang, K.Z.; Lu, C.; Dong, L.M.; Le, Z.J.; Liao, Y.H.; Liu, X.M. Antidepressant-like effects and cognitive enhancement of the total phenols extract of Hemerocallis citrina Baroni in chronic unpredictable mild stress rats and its related mechanism. J. Ethnopharmacol. 2016, 194, 819–826. [Google Scholar] [CrossRef]
- Marchyshyn, S.M.; Zarichanska, O.V.; Cholach, S.Y. Investigation of acute toxicity and neurotropic properties of the flowers’dense extracts of tawny daylily (Hemerocallis fulva L.) and hybrid daylily (Hemerocallis hybrida var. Stella De Oro). Farm. čas. 2016, 1, 79–84. [Google Scholar]
- Yang, L.; Shergis, J.L.; Di, Y.M.; Zhang, A.L.; Lu, C.; Guo, X.; Fang, Z.; Xue, C.C.; Li, Y. Managing depression with Bupleurum chinense herbal formula: A systematic review and meta-analysis of randomized controlled trials. J. Alter. Compl. Med. 2020, 26, 8–24. [Google Scholar] [CrossRef] [PubMed]
- Bagci, E.; Aydin, E.; Mihasan, M.; Maniu, C.; Hritcu, L. Anxiolytic and antidepressant-like effects of Ferulago angulata essential oil in the scopolamine rat model of Alzheimer’s disease. Flavour Fragr. J. 2016, 31, 70–80. [Google Scholar] [CrossRef]
- Dereli, F.T.G.; Ilhan, M.; Akkol, E.K. Identification of the main active antidepressant constituents in a traditional Turkish medicinal plant, Centaurea kurdica Reichardt. J. Ethnopharmacol. 2020, 249, 112373. [Google Scholar]
- Ioniță, R.; Postu, P.A.; Cioancă, O.; Mircea, C.; Hăncianu, M.; Hrițcu, L.; Mircea, C. Anxiolytic and antidepressant effects of Matricaria chamomilla hydroalcoholic extract in a rat model of scopolamine. Farmacia 2019, 67, 68–72. [Google Scholar] [CrossRef]
- Cárdenas, J.; Reyes-Pérez, V.; Hernández-Navarro, M.D.; Dorantes-Barrón, A.M.; Almazán, S.; Estrada-Reyes, R. Anxiolytic- and antidepressant-like effects of an aqueous extract of Tanacetum parthenium L. Schultz-Bip (Asteraceae) in mice. J. Ethnopharmacol. 2017, 200, 22–30. [Google Scholar] [CrossRef]
- Cornara, L.; Ambu, G.; Trombetta, D.; Denaro, M.; Alloisio, S.; Frigerio, J.; Labra, M.; Ghimire, G.; Valussi, M.; Smeriglio, A. Comparative and functional screening of three species traditionally used as antidepressants: Valeriana officinalis L., Valeriana jatamansi Jones ex Roxb. and Nardostachys jatamansi (D. Don) DC. Plants 2020, 9, 994. [Google Scholar] [CrossRef] [PubMed]
- Gill, M.; Kinra, M.; Rai, A.; Chamallamudi, M.R.; Kumar, N. Evaluation of antidepressant activity of methanolic extract of Saraca asoca bark in a chronic unpredictable mild stress model. Neuroreport 2018, 29, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Mateo, C.C.; Prado, B.; Rabanal, R.M. Antidepressant effects of the methanol extract of several Hypericum species from the Canary Islands. J. Ethnopharmacol. 2002, 79, 119–127. [Google Scholar] [CrossRef]
- Rahmati, B.; Kiasalari, Z.; Roghani, M.; Khalili, M.; Ansari, F. Antidepressant and anxiolytic activity of Lavandula officinalis aerial parts hydroalcoholic extract in scopolamine-treated rats. Pharm. Biol. 2017, 55, 958–965. [Google Scholar] [CrossRef]
- Nematolahi, P.; Mehrabani, M.; Karami-Mohajeri, S.; Dabaghzadeh, F. Effects of Rosmarinus officinalis L. on memory performance, anxiety, depression, and sleep quality in university students: A randomized clinical trial. Complem. Ther. Clin. Pract. 2018, 30, 24–28. [Google Scholar] [CrossRef]
- Choukairi, Z.; Hazzaz, T.; Lkhider, M.; Ferrandez, J.M.; Fechtali, T. Effect of Salvia officinalis L. and Rosmarinus officinalis L. leaves extracts on anxiety and neural activity. Bioinformation 2019, 5, 172–178. [Google Scholar] [CrossRef]
- Adel, A.; Ikram, R.; Wasi, N. Salvia hispanica (White chia): A new window for its antidepressant and memory boosting activity. Pak. J. Pharm. Sci. 2019, 32, 1005–1009. [Google Scholar]
- Sarkoohi, P.; Fathalipour, M.; Ghasemi, F.; Javidnia, K.; Emamghoreishi, M. Antidepressant effects of the aqueous and hydroalcoholic extracts of Salvia mirzayanii and Salvia macrosiphon in male mice. Shiraz. E-Med. J. 2020, 21, e91276. [Google Scholar] [CrossRef]
- El Gabbas, Z.; Bezza, K.; Laadraoui, J.; Makbal, R.; Aboufatima, R.; Chait, A. Salvia officinalis induces antidepressant-like effect, anxiolytic activity and learning improvement in hippocampal lesioned and intact adult rats. Bangladesh J. Pharmacol. 2018, 13, 367–378. [Google Scholar] [CrossRef]
- Hamann, F.R.; Zago, A.M.; Rossato, M.F.; Beck, V.R.; Mello, C.F.; de Brum, T.F.; de Carvalho, L.M.; Faccin, H.; Oliveira, S.M.; Rubin, M.A. Antinociceptive and antidepressant-like effects of the crude extract of Vitex megapotamica in rats. J. Ethnopharmacol. 2016, 192, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Devika, M.; Nalini, M.S. Evaluation of antidepressant activity of Litsea floribunda (Bl.) Gamble-Lauraceae using animal models. Int. J. Pharm. Sci. Res. 2018, 9, 3427–3432. [Google Scholar]
- Fiebich, B.L.; Knörle, R.; Appel, K.; Kammler, T.; Weiss, G. Pharmacological studies in an herbal drug combination of St. John’s Wort (Hypericum perforatum) and passion flower (Passiflora incarnata): In vitro and in vivo evidence of synergy between Hypericum and Passiflora in antidepressant pharmacological models. Fitoterapia 2011, 82, 474–480. [Google Scholar] [CrossRef]
- Alves, J.; Silva, A.; da Silva, R.M.; Tiago, P.; de Carvalho, T.G.; de Araújo, R.F., Jr.; de Azevedo, E.P.; Lopes, N.P.; Ferreira, L.S.; Gavioli, E.C.; et al. In Vivo antidepressant effect of Passiflora edulis f. flavicarpa into cationic nanoparticles: Improving bioactivity and safety. Pharmaceutics 2020, 12, 383. [Google Scholar] [CrossRef]
- Jafarpoor, N.; Abbasi-Maleki, S.; Asadi-Samani, M.; Khayatnouri, M.H. Evaluation of antidepressant-like effect of hydroalcoholic extract of Passiflora incarnata in animal models of depression in male mice. J. Herb.Med. Pharmacol. 2014, 3, 41–45. [Google Scholar]
- Bettio, L.E.; Machado, D.G.; Cunha, M.P.; Capra, J.C.; Missau, F.C.; Santos, A.R.; Pizzolatti, M.G.; Rodrigues, A.L. Antidepressant-like effect of extract from Polygala paniculata: Involvement of the monoaminergic systems. Pharm. Biol. 2011, 49, 1277–1285. [Google Scholar] [CrossRef]
- Ibrahim, M.; Amin, M.N.; Millat, M.S.; Raju, J.A.; Hussain, M.S.; Sultana, F.; Islam, M.M.; Hasan, M.M. Methanolic extract of peel of Citrus maxima fruits exhibit analgesic, CNS depressant and anti-inflammatory activities in swiss albino mice. BEMS Rep. 2018, 4, 7–11. [Google Scholar] [CrossRef]
- Barua, C.C.; Haloi, P.; Saikia, B.; Sulakhiya, K.; Pathak, D.C.; Tamuli, S.; Rizavi, H.; Ren, X. Zanthoxylum alatum abrogates lipopolysaccharide-induced depression-like behaviours in mice by modulating neuroinflammation and monoamine neurotransmitters in the hippocampus. Pharm. Biol. 2018, 56, 1744–5116. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, V.; Devi, K.; Sharma, M.; Singh, M.K.; Ahuja, P.S. State of art of saffron (Crocus sativus L.) agronomy: A comprehensive review. Food Rev. Int. 2008, 25, 44–85. [Google Scholar] [CrossRef]
- Hosseinzadeh, H.; Nassiri-Asl, M. Avicenna’s (Ibn Sina) the canon of medicine and saffron (Crocus sativus): A review. Phytother. Res. 2013, 27, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Javadi, B.; Sahebkar, A.; Emami, S.A. A survey on saffron in major islamic traditional medicine books. Iran. J. Basic. Med. Sci. 2013, 16, 1–11. [Google Scholar]
- Jan, S.; Wani, A.A.; Kamili, A.N.; Kashtwari, M. Distribution, chemical composition and medicinal importance of saffron (Crocus sativus L.). Afr. J. Plant. 2014, 8, 537–545. [Google Scholar]
- Ngoupaye, G.T.; Bum, E.N.; Taiwe, G.S.; Moto, F.C.; Talla, E. Antidepressant properties of aqueous macerate from Gladiolus dalenii corms. Afr. J. Tradit. Complement. Altern. Med. 2013, 11, 53–61. [Google Scholar] [PubMed]
- Nemati, Z.; Harpke, D.; Gemicioglu, A.; Kerndorff, H.; Blattner, F.R. Saffron (Crocus sativus) is an autotriploid that evolved in Attica (Greece) from wild Crocus cartwrightianus. Mol. Phylogenet. Evol. 2019, 136, 14–20. [Google Scholar] [CrossRef]
- Shokrpour, M. Saffron (Crocus sativus L.) breeding: Opportunities and challenges. In Advances in Plant Breeding Strategies: Industrial and Food Crops, 1st ed.; Al-Khayri, J.M., Jain, S.M., Johnson, D.V., Eds.; Springer: Cham, Switzerland, 2019; Volume 6, pp. 675–706. [Google Scholar]
- El Midaoui, A.; Ghzaiel, I.; Vervandier-Fasseur, D.; Ksila, M.; Zarrouk, A.; Nury, T.; Khallouki, F.; El Hessni, A.; Ibrahimi, S.O.; Latruffe, N.; et al. Saffron (Crocus sativus L.): A source of nutrients for health and for the treatment of neuropsychiatric and age-related diseases. Nutrients 2022, 14, 597. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Devi, M.; Kumar, R.; Kumar, S. Introduction of high-value Crocus sativus (saffron) cultivation in non-traditional regions of India through ecological modelling. Sci. Rep. 2022, 12, 11925. [Google Scholar] [CrossRef]
- Fujii, S.; Morita, Y.; Ohta, T.; Uto, T.; Shoyama, Y. Saffron (Crocus sativus L.) as a valuable spice and food product: A narrative review. Longhua Chin. Med. 2022, 5, 18. [Google Scholar] [CrossRef]
- Gresta, F.; Lombardo, G.M.; Siracusa, L.; Ruberto, G. Saffron, an alternative crop for sustainable agricultural systems. a review. agronomy for sustainable development. Agron. Sustain. Dev. 2008, 28, 95–112. [Google Scholar] [CrossRef]
- Grilli Caiola, M.; Canini, A. Looking for saffron’s (Crocus sativus L.) parents. Funct. Plant Sci. Biotechnol. 2010, 4, 1–14. [Google Scholar]
- Ahmad, M.; Zaffar, G.; Habib, M.; Arshid, A.; Dar, N.A.; Dar, Z.A. Saffron (Crocus sativus L.) in the light of biotechnological approaches: A review. Sci. Res. Essays 2014, 9, 13–18. [Google Scholar]
- Dar, R.A.; Shahnawaz, M.; Malik, S.B.; Sangale, M.K.; Ade, A.B.; Qazi, P.H. Cultivation, distribution, taxonomy, chemical composition and medical importance of Crocus sativus. J. Phytopharmacol. 2017, 6, 356–358. [Google Scholar] [CrossRef]
- Torricelli, R.; Yousefi, J.; Albertini, E.; Venanzoni, R.; Hosseinzadeh, Y. Morphological and molecular characterization of Italian, Iranian and Spanish saffron (Crocus sativus L.) accessions. Appl. Ecol. Environ. Res. 2019, 17, 1875–1887. [Google Scholar] [CrossRef]
- Ulbricht, C.; Conquer, J.; Costa, D.; Hollands, W.; Iannuzzi, C.; Isaac, R.; Jordan, J.K.; Ledesma, N.; Ostroff, C.; Serrano, J.M.; et al. An evidence-based systematic review of saffron (Crocus sativus) by the Natural Standard Research Collaboration. J. Diet. Suppl. 2011, 8, 58–114. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.; Sun, Y.; Zhou, G.; Yuan, Y.; Li, J.; Huang, H.; Xu, L.; Li, L. Single-molecule real-time transcript sequencing identified flowering regulatory genes in Crocus sativus. BMC Genom. 2019, 20, 857. [Google Scholar] [CrossRef]
- Pandita, D. Saffron (Crocus sativus L.): Phytochemistry, therapeutic significance and omics-based biology. In Medicinal and Aromatic Plants, 1st ed.; Aftab, T., Hakeem, K.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 325–396. [Google Scholar]
- He, F.; Dai, Z.; He, Y.; Zhang, Y.M.; Lu, J. Study on crocins reference extract and application on assay of Croci Stigma. Chin. J. Chin. Mat. Med. 2015, 40, 2378–2382. [Google Scholar]
- Al-Snafi, A.E. The pharmacology of Crocus sativus-a review. IOSR J. Pharm. 2016, 6, 8–38. [Google Scholar]
- Zhou, G.F.; Liu, Y.Y.; Qian, X.D.; Yao, C.; Chen, S.H. Improvement of quality evaluation of Croci Stigma in the process of producing and distributing based on Chinese pharmacopoeia and ISO*. Chin. J. Pharm. Anal. 2016, 36, 835–841. [Google Scholar]
- Melnyk, J.P.; Wang, S.; Marcone, M.F. Chemical and biological properties of the world’s most expensive spice: Saffron. Food. Res. Int. 2010, 43, 1981–1989. [Google Scholar] [CrossRef]
- Kyriakoudi, A.; Ordoudi, A.; Roldán-Medina, M.; Tsimidou, Z. Saffron, a functional spice. Austin J. Nutri. Food Sci. 2015, 3, 1059. [Google Scholar]
- Kafi, M.; Kamili, A.N.; Husaini, A.M.; Ozturk, M.; Altay, V. An expensive spice saffron (Crocus sativus L.): A case study from Kashmir, Iran, and Turkey. In Global Perspectives on Underutilized Crops; Ozturk, M., Hakeem, K.R., Ashraf, M., Ahmad, M.S.A., Eds.; Springer: Cham, Germany, 2018; pp. 109–149. [Google Scholar]
- Hosseini, A.; Razavi, B.M.; Hosseinzadeh, H. Saffron (Crocus sativus) petal as a new pharmacological target: A review. Iran. J. Basic Med. Sci. 2018, 21, 1091. [Google Scholar] [PubMed]
- Cardone, L.; Castronuovo, D.; Perniola, M.; Cicco, N.; Candido, V. Saffron (Crocus sativus L.) the king of spices: An overview. Sci. Hort. 2020, 272, 109560. [Google Scholar] [CrossRef]
- Kothari, D.; Thakur, R.; Kumar, R. Saffron (Crocus sativus L.): Gold of the spices—A comprehensive review. Hortic. Environ. Biotechnol. 2021, 62, 661–677. [Google Scholar] [CrossRef]
- Xing, B.; Li, S.; Yang, J.; Lin, D.; Feng, Y.; Lu, J.; Shao, Q. Phytochemistry, pharmacology, and potential clinical applications of saffron: A review. J. Ethnopharmacol. 2021, 281, 114555. [Google Scholar] [CrossRef]
- Chen, J.; Zhou, G.; Dong, Y.; Qian, X.; Li, J.; Xu, X.; Huang, H.; Xu, L.; Li, L. Screening of key proteins affecting floral initiation of saffron under cold stress using iTRAQ-based proteomics. Front. Plant Sci. 2021, 12, 644934. [Google Scholar] [CrossRef] [PubMed]
- Gohari, A.R.; Saeidnia, S.; Mahmoodabadi, M.K. An overview on saffron, phytochemicals, and medicinal properties. Pharmacog. Rev. 2013, 7, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Malathi, M.; Devi, D.R.; Hari, B.V. Crocus sativus Linn–a potential source for diverse therapeutic applications. Int. J. Pharm. Sci. Rev. Res. 2014, 26, 299–305. [Google Scholar]
- Rahimi, M. Chemical and medicinal properties of saffron. Bull. Environ. Pharmacol. Life Sci. 2015, 4, 69–81. [Google Scholar]
- Kosar, M.; Demirci, B.; Goger, F.; Kara, I.; Baser, K.H.C. Volatile composition, antioxidant activity, and antioxidant components in saffron cultivated in Turkey. Int. J. Food Prop. 2017, 20 (Suppl. 1), S746–S754. [Google Scholar] [CrossRef]
- Moratalla-López, N.; Bagur, M.J.; Lorenzo, C.; Martínez-Navarro, M.E.; Salinas, M.R.; Alonso, G.L. Bioactivity and bioavailability of the major metabolites of Crocus sativus L. flower. Molecules 2019, 24, 2827. [Google Scholar] [CrossRef]
- Mykhailenko, O.; Kovalyov, V.; Goryacha, O.; Ivanauskas, L.; Georgiyants, V. Biologically active compounds and pharmacological activities of species of the genus Crocus: A review. Phytochemistry 2019, 162, 56–89. [Google Scholar] [CrossRef]
- Jafari, S.M.; Mahdavee Khazaei, K.; Assadpour, E. Production of a natural color through microwave-assisted extraction of saffron tepal’s anthocyanins. Food Sci. Nutr. 2019, 7, 1438–1445. [Google Scholar] [CrossRef] [PubMed]
- Goupy, P.; Vian, M.A.; Chemat, F.; Caris-Veyrat, C. Identification and quantification of flavonols, anthocyanins and lutein diesters in tepals of Crocus sativus by ultra performance liquid chromatography coupled to diode array and ion trap mass spectrometry detections. Ind. Crop. Prod. 2013, 44, 496–510. [Google Scholar] [CrossRef]
- Baba, S.A.; Malik, A.H.; Wani, Z.A.; Mohiuddin, T.; Shah, Z.; Abbas, N.; Ashraf, N. Phytochemical analysis and antioxidant activity of different tissue types of Crocus sativus and oxidative stress alleviating potential of saffron extract in plants, bacteria, and yeast. S. Afr. J. Bot. 2015, 99, 80–87. [Google Scholar] [CrossRef]
- Karimi, E.; Oskoueian, E.; Hendra, R.; Jaafar, H.Z. Evaluation of Crocus sativus L. stigma phenolic and flavonoid compounds and its antioxidant activity. Molecules 2010, 15, 6244–6256. [Google Scholar] [CrossRef] [PubMed]
- Mir, M.A.; Parihar, K.; Tabasum, U.; Kumari, E. Estimation of alkaloid, saponin and flavonoid, content in various extracts of Crocus sativa. J. Med. Plants Stud. 2016, 4, 171–174. [Google Scholar]
- Goli, S.A.H.; Mokhtari, F.; Rahimmalek, M. Phenolic compounds and antioxidant activity from saffron (Crocus sativus L) petal. J. Agric. Sci. 2012, 4, 175–181. [Google Scholar]
- Masuda, A.; Mori, K.; Oda, Y.; Miyazawa, M. Volatile oil compounds from corms and flowers of Crocus vernus L. Hill and corms of C. sativus L. Libyan Agric. Res. Cen. J. Int. 2010, 1, 244–249. [Google Scholar]
- Karimi, G.R.; Hosseinzadeh, H.; Khaleghpanah, P. Study of antidepressant effect of aqueous and ethanolic extract of Crocus sativus in mice. Iran J. Basic Med. Sci. 2001, 4, 11–15. [Google Scholar]
- Hosseinzadeh, H.; Younesi, H.M. Antinociceptive and anti-inflammatory effects of Crocus sativus L. stigma and petal extracts in mice. BMC Pharmacol. 2002, 2, 7. [Google Scholar]
- Akhondzadeh, S.; Fallah-Pour, H.; Afkham, K.; Jamshidi, A.H.; Khalighi-Cigaroudi, F. Comparison of Crocus sativus L. and imipramine in the treatment of mild to moderate depression: A pilot double-blind randomized trial [ISRCTN45683816]. BMC Complement. Altern. Med. 2004, 4, 12. [Google Scholar] [CrossRef] [PubMed]
- Akhondzadeh, S.; Tahmacebi-Pour, N.; Noorbala, A.A.; Amini, H.; Fallah Pour, H.; Jamshidi, A.H.; Khani, M. Crocus sativus L. in the treatment of mild to moderate depression: A double-blind, randomized and placebo controlled trial. Phytother. Res. 2005, 19, 148–151. [Google Scholar] [CrossRef] [PubMed]
- Kamalipour, M.; Jamshidi, A.H.; Akhondzadeh, S. Antidepressant effect of Crocus sativus: An evidence based review. J. Med. Plants. 2010, 9, 35–38. [Google Scholar]
- Mousavi, S.Z.; Bathaie, S.Z. Historical uses of saffron: Identifying potential new avenues for modern research. Avicenna J. Phytomed. 2011, 1, 57–66. [Google Scholar]
- Moshiri, M.; Vahabzadeh, M.; Hosseinzadeh, H. Clinical applications of saffron (Crocus sativus) and its constituents: A review. Drug Res. Stuttg. 2015, 65, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Selamoglu, Z.; Ozgen, S. Therapeutic potential of saffron crocus (Crocus sativus L.). Turkish JAF Sci. Tech. 2016, 4, 1240–1245. [Google Scholar] [CrossRef][Green Version]
- Feldman, E. Saffron and depression: What do we know and where do we go? Integr. Med. Alert. 2017, 20, 8. [Google Scholar]
- Loizzo, M.R.; Marrelli, M.; Pugliese, A.; Conforti, F.; Nadjafi, F.; Menichini, F.; Tundis, R. Crocus cancellatus subsp. Damascenus stigmas: Chemical profile, and inhibition of α-amylase, α-glucosidase and lipase, key enzymes related to type 2 diabetes and obesity. J. Enzyme Inhib. Med. Chem. 2016, 31, 212–218. [Google Scholar] [CrossRef]
- Ashktorab, H.; Soleimani, A.; Singh, G.; Amin, A.; Tabtabaei, S.; Latella, G.; Stein, U.; Akhondzadeh, S.; Solanki, N.; Gondré-Lewis, M.C.; et al. Saffron: The golden spice with therapeutic properties on digestive diseases. Nutrients 2019, 11, 943. [Google Scholar] [CrossRef] [PubMed]
- Ghaderi, A.; Asbaghi, O.; Reiner, Ž.; Kolahdooz, F.; Amirani, E.; Mirzaei, H.; Banafshe, H.R.; Dana, P.M.; Asemi, Z. The effects of saffron (Crocus sativus L.) on mental health parameters and C-reactive protein: A meta-analysis of randomized clinical trials. Complement. Ther. Med. 2020, 1, 102250. [Google Scholar] [CrossRef]
- Abdullaev, F. Biological properties and medicinal use of saffron (Crocus sativus L.). Acta Hortic. 2007, 739, 339–345. [Google Scholar] [CrossRef]
- Bhargava, B.K. Medicinal uses and pharmacological properties of Crocus sativus Linn. (saffron). Int. J. Pharm. Pharm. Sci. 2011, 3, 22–26. [Google Scholar]
- Poma, A.; Fontecchio, G.; Carlucci, G.; Chichiricco, G. Anti-inflammatory properties of drugs from saffron crocus. Antiinflamm. Antiallergy Agents Med. Chem. 2012, 11, 37–51. [Google Scholar] [CrossRef] [PubMed]
- Bahramsoltani, R.; Farzaei, M.H.; Rahimi, R. Medicinal plants and their natural components as future drugs for the treatment of burn wounds: An integrative review. Arch. Dermatol. Res. 2014, 306, 601–617. [Google Scholar] [CrossRef] [PubMed]
- Christodoulou, E.; Kadoglou, N.P.; Kostomitsopoulos, N.; Valsami, G. Saffron: A natural product with potential pharmaceutical applications. J. Pharm. Pharmacol. 2015, 67, 1634–1649. [Google Scholar] [CrossRef]
- Khorasany, A.R.; Hosseinzadeh, H. Therapeutic effects of saffron (Crocus sativus L.) in digestive disorders: A review. Iran J. Basic. Med. Sci. 2016, 19, 455–469. [Google Scholar]
- Vyas, L.K.; Tapar, K.K.; Nema, R.K. Study of Crocus sativus as complexion promoter in skin care. Int. J. Pharm. Clin. Res. 2010, 2, 76–79. [Google Scholar]
- Bathaie, S.Z.; Farajzade, A.; Hoshyar, R. A review of the chemistry and uses of crocins and crocetin, the carotenoid natural dyes in saffron, with particular emphasis on applications as colorants including their use as biological stains. Biotech. Histochem. 2014, 89, 401–411. [Google Scholar] [CrossRef]
- Mzabri, I.; Addi, M.; Berrichi, A. Traditional and modern uses of saffron (Crocus sativus). Cosmetics 2019, 6, 63. [Google Scholar] [CrossRef]
- Licón, C.C.; Carmona, M.; Rubio, R.; Molina, A.; Berruga, M.I. Preliminary study of saffron (Crocus sativus L. stigmas) color extraction in a dairy matrix. Dyes Pigm. 2012, 92, 1355–1360. [Google Scholar] [CrossRef]
- Çelik, S.Y. Purification of protease enzyme from çiğdem (Crocus biflorus) tubers and investigation of usability of the purified protease enzyme in coagulation of casein. GIDA 2018, 43, 231–239. [Google Scholar] [CrossRef]
- Shariati-Moghaddam, A. New saffron products and food technology. Acta Hortic. 2004, 650, 369–371. [Google Scholar] [CrossRef]
- Serrano-Díaz, J.; Sánchez, A.M.; Martínez-Tomé, M.; Winterhalter, P.; Alonso, G.L. A contribution to nutritional studies on Crocus sativus flowers and their value as food. J. Food Compos. Anal. 2013, 31, 101–108. [Google Scholar] [CrossRef]
- Almodóvar, P.; Prodanov, M.; Arruñada, O.; Inarejos-García, A.M.; Almodóvar, P.; Prodanov, M.; Arruñada, O.; Inarejos-García, A.M. Saffron eye, a natural extract of saffron (Crocus sativus L.) with colorant properties as novel replacer of saffron stigmas in culinary and food applications. Int. J. Gastron. Food Sci. 2018, 12, 1–5. [Google Scholar] [CrossRef]
- Bagur, M.; Alonso Salinas, G.L.; Jiménez-Monreal, A.M.; Chaouqi, S.; Llorens, S.; Martínez-Tomé, M.; Alonso, G.L. Saffron: An old medicinal plant and a potential novel functional food. Molecules 2018, 23, 30. [Google Scholar] [CrossRef]
- Takur, M.; Sharma, N. Saffron: A golden condiment and a respository of nutraceutical potential. Food Sci. 2014, 5, 59–67. [Google Scholar]
- Sarfarazi, M.; Jafari, S.M.; Rajabzadeh, G. Extraction optimization of saffron nutraceuticals through response surface methodology. Food Anal. Methods 2015, 8, 2273–2285. [Google Scholar] [CrossRef]
- Bathaie, S.Z. Saffron as a functional food and a nutraceutical using saffron and its constituents as the nutraceutics to protect against chronic diseases. Acta Hortic. 2018, 1200, 201–204. [Google Scholar] [CrossRef]
- Khan, A.; Muhamad, N.A.; Ismail, H.; Nasir, A.; Khalil, A.A.K.; Anwar, Y.; Khan, Z.; Ali, A.; Taha, R.M.; Al-Shara, B.; et al. Potential nutraceutical benefits of in vivo grown saffron (Crocus sativus L.) as analgesic, anti-inflammatory, anticoagulant, and antidepressant in mice. Plants 2020, 9, 1414. [Google Scholar] [CrossRef]
- Merrell, J.G.; McLaughlin, S.W.; Tie, L.; Laurencin, C.T.; Chen, A.F.; Nair, L.S. Curcumin-loaded poly (ε-caprolactone) nanofibres: Diabetic wound dressing with anti-oxidant and anti-inflammatory properties. Clin. Exp. Pharmacol. Physiol. 2009, 36, 1149–1156. [Google Scholar] [CrossRef]
- Thamer, N.A.; AL-Mashhady, L.A. Acute toxicity of green synthesis of silver nanoparticles using Crocus sativus L. on white albino rats. Int. J. Phytopharm. 2016, 7, 13–16. [Google Scholar]
- Yildiztekin, M.; Nadeem, S.; Yildiztekin, F.; Varol, O.; Ozler, M.A.; Tuna, A.L. Green synthesis and characterization of silver nanoparticles from Crocus mathewii; a disremembered Turkish flowering plant. Indian J. Pharm. Sci. 2017, 79, 536–543. [Google Scholar] [CrossRef]
- Bagherzade, G.; Tavakoli, M.M.; Namaei, M.H. Green synthesis of silver nanoparticles using aqueous extract of saffron (Crocus sativus L.) wastages and its antibacterial activity against six bacteria. Asian Pac. J. Trop. Biomed. 2017, 7, 227–233. [Google Scholar] [CrossRef]
- Mirhadi, E.; Nassirli, H.; Malaekeh-Nikouei, B. An updated review on therapeutic effects of nanoparticle-based formulations of saffron components (safranal, crocin, and crocetin). J. Pharm. Investig. 2020, 50, 47–58. [Google Scholar] [CrossRef]
- Hafezi Ghahestani, Z.; Alebooye Langroodi, F.; Mokhtarzadeh, A.; Ramezani, M.; Hashemi, M. Evaluation of anti-cancer activity of PLGA nanoparticles containing crocetin. Artif. Cells Nanomed. Biotechnol. 2017, 45, 955–960. [Google Scholar] [CrossRef]
- Azizian-Shermeh, O.; Valizadeh, M.; Taherizadeh, M.; Beigomi, M. Phytochemical investigation and phytosynthesis of eco-friendly stable bioactive gold and silver nanoparticles using petal extract of saffron (Crocus sativus L.) and study of their antimicrobial activities. Appl. Nanosci. 2020, 10, 2907–2920. [Google Scholar] [CrossRef]
- Zeka, K.; Ruparelia, K.C.; Sansone, C.; Macchiarelli, G.; Continenza, M.A.; Arroo, R.R. New hydrogels enriched with antioxidants from saffron crocus can find applications in wound treatment and/or beautification. Skin Pharmacol. Physiol. 2018, 31, 95–98. [Google Scholar] [CrossRef]
- Schmidt, M.; Betti, G.; Hensel, A. Saffron in phytotherapy: Pharmacology and clinical uses. Wien Med. Wochenschr. 2007, 157, 315–319. [Google Scholar] [CrossRef]
- Gutheil, G.W.; Reed, G.; Ray, A.; Anant, S.; Dhar, A. Crocetin: An agent derived from saffron for prevention and therapy for cancer derived from saffron for prevention and therapy for cancer. Curr. Pharm. Biotechnol. 2012, 13, 173–179. [Google Scholar] [CrossRef]
- Talaei, A.; Hassanpour Moghadam, M.; Sajadi Tabassi, S.A.; Mohajeri, S.A. Crocin, the main active saffron constituent, as an adjunctive treatment in major depressive disorder: A randomized, double-blind, placebo-controlled, pilot clinical trial. J. Affect. Disord. 2015, 174, 51–56. [Google Scholar] [CrossRef]
- Jafarnia, N.; Ghorbani, Z.; Nokhostin, M.; Manayi, A.; Nourimajd, S.; Jahromi, S.R. Effect of saffron (Crocus sativus L.) as an add-on therapy to sertraline in mild to moderate generalized anxiety disorder: A double blind randomized controlled trial. Arch. Neurosci. 2017, 4, e14332. [Google Scholar]
- Zhang, Z.; Wang, C.Z.; Wen, X.D.; Shoyama, Y.; Yuan, C.S. Role of saffron and its constituents on cancer chemoprevention. Pharm. Biol. 2013, 5, 920–924. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, P.R. Crocus sativus L. (saffron) for cancer chemoprevention: A mini review. J. Tradit. Complement. Med. 2015, 5, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Naeimi, M.; Shafiee, M.; Kermanshahi, F.; Khorasanchi, Z.; Khazaei, M.; Ryzhikov, M.; Avan, A.; Gorji, N.; Hassanian, S.M. Saffron (Crocus sativus) in the treatment of gastrointestinal cancers: Current findings and potential mechanisms of action. J. Cell. Biochem. 2019, 120, 16330–16339. [Google Scholar] [CrossRef]
- Dawalbhakta, M.; Telang, M. Patents on therapeutic and cosmetic applications of bioactives of Crocus sativus L. and their production through synthetic biology methods: A review. Ther. Pat. Eotpeg. 2017, 11, 3–19. [Google Scholar]
- Alsayied, N.F.; Fernández, J.A.; Schwarzacher, T.; Heslop-Harrison, J.S. Diversity and relationships of Crocus sativus and its relatives analysed by inter-retroelement amplified polymorphism (IRAP). Ann. Bot. 2015, 116, 359–368. [Google Scholar] [CrossRef]
- Gedik, A.; Duygu, A.; Erdogmus, S.; Comertpay, G.; Tanyolac, M.B.; Ozkan, H. Genetic diversity of Crocus sativus and its close relative species analyzed by iPBS-retrotransposons. Turk. J. Field Crop. 2017, 22, 243–252. [Google Scholar] [CrossRef]
- Bayat, M.; Amirnia, R.; Özkan, H.; Gedik, A.; Ates, D.; Tanyulac, B.; Rahimi, M. Diversity and phylogeny of saffron (Crocus sativus L.) accessions based on IPBS markers. Genetica 2018, 50, 33–44. [Google Scholar] [CrossRef]
- Yousefi Javan, I.; Gharari, F. Genetic diversity in saffron (Crocus sativus L.) cultivars grown in Iran using SSR and SNP markers. J. Agric. Sci. Technol. 2018, 20, 1213–1226. [Google Scholar]
- Wang, Y.; Han, T.; Zhu, Y.; Zheng, C.J.; Ming, Q.L.; Rahman, K.; Qin, L.P. Antidepressant properties of bioactive fractions from the extract of Crocus sativus L. J. Nat. Med. 2010, 64, 24–30. [Google Scholar] [CrossRef]
- Lopresti, A.L.; Maker, G.L.; Hood, S.D.; Drummond, P.D. A review of peripheral biomarkers in major depression: The potential of inflammatory and oxidative stress biomarkers. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014, 48, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Lopresti, A.L.; Drummond, P.D. Saffron (Crocus sativus) for depression: A systematic review of clinical studies and examination of underlying antidepressant mechanisms of action. Hum. Psychopharmacol. Clin. Exp. 2014, 29, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, T.; Abnous, K.; Vahdati, F.; Mehri, S.; Razavi, B.; Hosseinzadeh, H. Antidepressant effect of Crocus sativus aqueous extract and its effect on CREB, BDNF, and VGF transcript and protein levels in rat hippocampus. Drug Res. 2015, 65, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Pitsikas, N.; Boultadakis, A.; Georgiadou, G.; Tarantilis, P.A.; Sakellaridis, N. Effects of the active constituents of Crocus sativus L., crocins, in an animal model of anxiety. Phytomedicine 2008, 15, 1135–1139. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, H.; Noraei, N.B. Anxiolytic and hypnotic effect of Crocus sativus aqueous extract and its constituents, crocin and safranal, in mice. Phytother Res. 2009, 23, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Mazidi, M.; Shemshian, M.; Mousavi, S.H.; Norouzy, A.; Kermani, T.; Moghiman, T.; Sadeghi, A.; Mokhber, N.; Ghayour-Mobarhan, M.; Ferns, G.A. A double-blind, randomized and placebo-controlled trial of saffron (Crocus sativus L.) in the treatment of anxiety and depression. J. Complement. Integr. Med. 2016, 13, 195–199. [Google Scholar] [CrossRef]
- Pitsikas, N. Constituents of saffron (Crocus sativus L.) as potential candidates for the treatment of anxiety disorders and schizophrenia. Molecules 2016, 21, 303. [Google Scholar] [CrossRef]
- Lopresti, A.L.; Drummond, P.D.; Inarejos-García, A.M.; Prodanov, M. Affron, a standardised extract from saffron (Crocus sativus L.) for the treatment of youth anxiety and depressive symptoms: A randomised, double-blind, placebo-controlled study. J. Affect. Disord. 2018, 232, 349–357. [Google Scholar] [CrossRef]
- Nam, K.N.; Park, Y.M.; Jung, H.J.; Lee, J.Y.; Min, B.D.; Park, S.U.; Jung, W.S.; Cho, K.H.; Park, J.H.; Kang, I.; et al. Anti-inflammatory effects of crocin and crocetin in rat brain microglial cells. Eur. J. Pharmacol. 2010, 648, 110–116. [Google Scholar] [CrossRef]
- Kumar, V.; Bhat, Z.A.; Kumar, D.; Khan, N.A.; Chashoo, I.A.; Shah, M.Y. Evaluation of anti-inflammatory potential of petal extracts of Crocus sativus ‘Cashmerianus’. Intl. J. Pjournal. Phytopharmacol. 2012, 3, 27–31. [Google Scholar]
- Rathore, B.; Jaggi, K.; Thakur, S.K.; Mathur, A.; Mahdi, F. Anti-inflammatory activity of Crocus sativus extract in experimental arthritis. Int. J. Pharm. Sci. Res. 2015, 6, 1473–1478. [Google Scholar]
- Zhang, C.; Ma, J.; Fan, L.; Zou, Y.; Dang, X.; Wang, K.; Song, J. Neuroprotective effects of safranal in a rat model of traumatic injury to the spinal cord by anti-apoptotic, anti-inflammatory and edema-attenuating. Tissue Cell. 2015, 47, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Boskabady, M.H.; Farkhondeh, T. Anti-inflammatory, antioxidant, and immunomodulatory effects of Crocus sativus L. and its main constituents. Phytother. Res. 2016, 30, 1072–1094. [Google Scholar] [CrossRef]
- Kermani, T.; Zebarjadi, M.; Mehrad-Majd, H.; Mirhafez, S.R.; Shemshian, M.; Ghasemi, F.; Mohammadzadeh, E.; Mousavi, S.H.; Norouzy, A.; Moghiman, T.; et al. Anti-inflammatory effect of Crocus sativus on serum cytokine levels in subjects with metabolic syndrome: A randomized, double-blind, placebo-controlled trial. Curr. Clin. Pharmacol. 2017, 12, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Yarijani, Z.M.; Pourmotabbed, A.; Pourmotabbed, T.; Najafi, H. Crocin has anti-inflammatory and protective effects in ischemia-reperfusion induced renal injuries. Iran J. Basic Med. Sci. 2017, 20, 753–759. [Google Scholar] [PubMed]
- Zeinali, M.; Zirak, M.R.; Rezaee, S.A.; Karimi, G.; Hosseinzadeh, H. Immunoregulatory and anti-inflammatory properties of Crocus sativus (Saffron) and its main active constituents: A review. Iran J. Basic Med. Sci. 2019, 22, 334–344. [Google Scholar] [PubMed]
- Liu, M.; Amini, A.; Ahmad, Z. Safranal and its analogs inhibit Escherichia coli ATP synthase and cell growth. Int. J. Biol. Macromol. 2017, 95, 145–152. [Google Scholar] [CrossRef]
- Abduallev, F.I. Cancer chemopreventive and tumoricidal properties of saffron (Crocus sativus L.). Exp. Biol. Med. 2002, 227, 20–25. [Google Scholar] [CrossRef]
- Abdullaev, F.I.; Riveron-Negrete, L.; Caballero-Ortega, H.; Hernández, J.M.; Perez-Lopez, I.; Pereda-Miranda, R.; Espinosa-Aguirre, J.J. Use of in vitro assays to assess the potential antigenotoxic and cytotoxic effects of saffron (Crocus sativus L.). Toxicol. In Vitr. 2003, 17, 731–736. [Google Scholar] [CrossRef]
- Abdullaev, F.I. Antitumor effect of saffron (Crocus sativus L.): Overview and perspectives. Acta Hortic. 2004, 650, 491–499. [Google Scholar] [CrossRef]
- Fernández, J.A. Anticancer properties of saffron, Crocus sativus Linn. Adv. Phytomed. 2006, 2, 316–330. [Google Scholar]
- Premkumar, K.; Thirunavukkarasu, C.; Abraham, S.K.; Santhiya, S.T.; Ramesh, A. Protective effect of saffron (Crocus sativus L.) aqueous extract against genetic damage induced by anti-tumor agents in mice. Hum. Exp. Toxicol. 2006, 25, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Chryssanthi, D.G.; Lamari, F.N.; Iatrou, G.; Pylara, A.; Karamanos, N.K.; Cordopatis, P. Inhibition of breast cancer cell proliferation by style constituents of different Crocus species. Anticancer Res. 2007, 27, 357–362. [Google Scholar]
- Abdullaev, F.I.; Riveron-Negrete, L.; Caballero-Ortega, H.; Hernández, J.M.; Perez-Lopez, I.; Pereda-Miranda, R.; Espinosa-Aguirre, J.J. Use of in vitro assays to assess the potential antiproliferative and cytotoxic effects of saffron (Crocus sativus L.) in human lung cancer cell line. Pharmacogn. Mag. 2010, 6, 309–314. [Google Scholar]
- Samarghandian, S.; Borji, A. Anticarcinogenic effect of saffron (Crocus sativus L.) and its ingredients. Pharmacogn. Res. 2014, 6, 99–107. [Google Scholar] [CrossRef]
- Gezici, S. Comparative anticancer activity analysis of saffron extracts and a principle component, crocetin for prevention and treatment of human malignancies. J. Food Sci. Technol. 2019, 56, 5435–5443. [Google Scholar] [CrossRef]
- Hosseinzadeh, H.; Shariaty, V.M. Anti-nociceptive effect of safranal, a constituent of Crocus sativus (saffron), in mice. Pharmacologyonline 2007, 2, 498–503. [Google Scholar]
- Amin, B.; Hosseini, S.; Hosseinzadeh, H. Enhancement of antinociceptive effect by co-administration of amitriptyline and Crocus sativus in a rat model of neuropathic pain. Iran. J. Pharm. Res. 2017, 16, 187–200. [Google Scholar]
- Li Puma, S.; Landini, L.; Macedo, S.J., Jr.; Seravalli, V.; Marone, I.M.; Coppi, E.; Patacchini, R.; Geppetti, P.; Materazzi, S.; Nassini, R.; et al. TRPA 1 mediates the antinociceptive properties of the constituent of Crocus sativus L., safranal. J. Cell Mol. Med. 2019, 23, 1976–1986. [Google Scholar] [CrossRef]
- Hosseinzadeh, H.; Khosravan, V. Anticonvulsant effect of Crocus sativus L. stigmas aqueous and ethanolic extracts in mice. Arch. Iran Med. 2002, 5, 44–47. [Google Scholar]
- Hosseinzadeh, H.; Talebzadeh, F. Anticonvulsant evaluation of safranal and crocin from Crocus sativus in mice. Fitoterapia 2005, 76, 722–724. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, H.; Ghenaati, J. Evaluation of the antitussive effect of stigma and petals of saffron (Crocus sativus) and its components, safranal and crocin in guinea pigs. Fitoterapia 2006, 77, 446–448. [Google Scholar] [CrossRef] [PubMed]
- Tavakkol-Afshari, J.; Brook, A.; Mousavi, S.H. Study of cytotoxic and apoptogenic properties of saffron extract in human cancer cell lines. Food Chem. Toxicol. 2008, 46, 3443–3447. [Google Scholar] [CrossRef] [PubMed]
- Vazifedan, V.; Mousavi, S.H.; Sargolzaei, J.; Soleymanifard, S.; Pakdel, A.F. Study of crocin radiotherapy-induced cytotoxicity and apoptosis in the head and neck cancer (HN-5) cell line. Iran J. Pharm. Res. 2017, 16, 230–237. [Google Scholar]
- Koul, A.; Abraham, S.K. Efficacy of crocin and safranal as protective agents against genotoxic stress induced by gamma radiation, urethane and procarbazine in mice. Hum. Exp. Toxicol. 2018, 37, 13–20. [Google Scholar] [CrossRef]
- Boskabady, M.H.; Aslani, M.R. Relaxant effect of Crocus sativus (saffron) on guinea-pig tracheal chains and its possible mechanisms. J. Pharm. Pharmacol. 2006, 58, 1385–1390. [Google Scholar] [CrossRef]
- Mokhtari-Zaer, A.; Khazdair, M.R.; Boskabady, M.H. Smooth muscle relaxant activity of Crocus sativus (saffron) and its constituents: Possible mechanisms. Avicenna J. Phytomed. 2015, 5, 365–375. [Google Scholar]
- Fatehi, M.; Rashidabady, T.; Fatehi-Hassanabad, Z. Effects of Crocus sativus petals’ extract on rat blood pressure and on response induced by electrical field stimulation in the rat isolated vas deferens and guinea-pig ileum. J. Ethnopharmacol. 2003, 84, 199–203. [Google Scholar] [CrossRef]
- Imenshahidi, M.; Razavi, B.M.; Faal, A.; Gholampoor, A.; Mehran, S. The effect of chronic administration of saffron (Crocus sativus) stigma aqueous extract on systolic blood pressure in rats. Jundishapur J. Nat. Pharm. Prod. 2013, 8, 175–179. [Google Scholar] [CrossRef]
- Papandreou, M.A.; Kanakis, C.D.; Polissiou, M.G.; Efthimiopoulos, S.; Cordopatis, P.; Margarity, M.; Lamari, F.N. Inhibitory activity on amyloid-β aggregation and antioxidant properties of Crocus sativus stigmas extract and its crocin constituents. J. Agric. Food Chem. 2006, 54, 8762–8768. [Google Scholar] [CrossRef]
- Termentzi, A.; Kokkalou, E. LC-DAD-MS (ESI+) analysis and antioxidant capacity of Crocus sativus petal extracts. Planta Med. 2008, 74, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, H.; Shamsaie, F.; Mehri, S. Antioxidant activity of aqueous and ethanolic extracts of Crocus sativus L. stigma and its bioactive constituents, crocin and safranal. Pharmacogn. Mag. 2009, 5, 419–424. [Google Scholar]
- Acar, G.; Dogan, N.M.; Duru, M.E.; Kıvrak, I. Phenolic profiles, antimicrobial and antioxidant activity of the various extracts of Crocus species in Anatolia. Afr. J. Microbiol. Res. 2010, 4, 1154–1161. [Google Scholar]
- Vatankhah, E.; Niknam, V.; Ebrahimzadeh, H. Activity of antioxidant enzyme during in vitro organogenesis in Crocus sativus. Biol. Plant. 2010, 54, 509–514. [Google Scholar] [CrossRef]
- Gismondi, A.; Serio, M.; Canuti, L.; Canini, A. Biochemical, antioxidant and antineoplastic properties of Italian saffron (Crocus sativus L.). Am. J. Plant Sci. 2012, 3, 1573–1580. [Google Scholar] [CrossRef]
- Sachdeva, J.; Tanwar, V.; Golechha, M.; Siddiqui, K.M.; Nag, T.C.; Ray, R.; Kumari, S.; Arya, D.S. Crocus sativus L. (saffron) attenuates isoproterenol-induced myocardial injury via preserving cardiac functions and strengthening antioxidant defense system. Exp. Toxicol. Pathol. 2012, 64, 557–564. [Google Scholar] [CrossRef]
- Rahaiee, S.; Moini, S.; Hashemi, M.; Shojaosadati, S.A. Evaluation of antioxidant activities of bioactive compounds and various extracts obtained from saffron (Crocus sativus L.): A review. J. Food Sci. Tech. 2015, 52, 1881–1888. [Google Scholar] [CrossRef]
- Wang, C.; Cai, X.; Hu, W.; Li, Z.; Kong, F.; Chen, X.; Wang, D. Investigation of the neuroprotective effects of crocin via antioxidant activities in HT22 cells and in mice with Alzheimer’s disease. Internat. J. Mol. Med. 2019, 43, 956–966. [Google Scholar] [CrossRef]
- Kim, Y.K.; Na, K.S.; Shin, K.H.; Jung, H.Y.; Choi, S.H.; Kim, J.B. Cytokine imbalance in the pathophysiology of major depressive disorder. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2007, 31, 1044–1053. [Google Scholar] [CrossRef]
- Kim, Y.K.; Na, K.S.; Myint, A.M.; Leonard, B.E. The role of pro-inflammatory cytokines in neuroinflammation, neurogenesis and the neuroendocrine system in major depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2016, 64, 277–284. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target Ther. 2017, 2, e17023. [Google Scholar] [CrossRef] [PubMed]
- Petralia, M.C.; Mazzon, E.; Fagone, P.; Basile, M.S.; Lenzo, V.; Quattropani, M.C.; di Nuovo, S.; Bendtzen, K.; Nicoletti, F. The cytokine network in the pathogenesis of major depressive disorder. Close to translation? Autoimmun. Rev. 2020, 19, 102504. [Google Scholar] [CrossRef] [PubMed]
- Nørbæk, R.; Kondo, T. Anthocyanins from flowers of Crocus (Iridaceae). Phytochemistry 1998, 47, 861–864. [Google Scholar] [CrossRef]
- Nørbæk, R.; Kondo, T. Further anthocyanins from flowers of Crocus antalyensis (Iridaceae). Phytochemistry 1999, 50, 325–328. [Google Scholar] [CrossRef]
- Castro-Díaz, N.; Salaun, B.; Perret, R.; Sierro, S.; Romero, J.F.; Fernández, J.A.; Rubio-Moraga, A.; Romero, P. Saponins from the Spanish saffron Crocus sativus are efficient adjuvants for protein-based vaccines. Vaccine 2012, 30, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Moraga, Á.; Gerwig, G.J.; Castro-Díaz, N.; Jimeno, M.L.; Escribano, J.; Fernández, J.A.; Kamerling, J.P. Triterpenoid saponins from corms of Crocus sativus: Localization, extraction and characterization. Ind. Crop. Prod. 2011, 34, 1401–1409. [Google Scholar] [CrossRef]
- Esmaeili, N.; Ebrahimzadeh, H.; Abdi, K.; Safarian, S. Determination of some phenolic compounds in Crocus sativus L. corms and its antioxidant activities study. Pharmacogn. Mag. 2011, 7, 74–80. [Google Scholar]
- Zhu, Y.; Han, T.; Hou, T.T.; Hu, Y.; Zhang, Q.Y.; Rahman, K.; Qin, L.P. Comparative study of composition of essential oil from stigmas and of extract from corms of Crocus sativus. Chem. Nat. Compd. 2008, 44, 666–667. [Google Scholar] [CrossRef]
- Ma, S.; Zhou, S.; Bing, S. Pharmacological studies Crocus glycosides I. Effects on antiinflammatory and immune function. Chin. Tradit. Herbal Drugs 1998, 129, 536–539. [Google Scholar]
- García-Olmo, D.C.; Riese, H.H.; Escribano, J.; Ontañón, J.; Fernandez, J.A.; Atiénzar, M.; García-Olmo, D. Effects of long-term treatment of colon adenocarcinoma with crocin, a carotenoid from saffron (Crocus sativus L.): An experimental study in the rat. Nutr. Cancer. 1999, 35, 120–126. [Google Scholar] [CrossRef]
- Arjmand, M.H.; Hashemzehi, M.; Soleimani, A.; Asgharzadeh, F.; Avan, A.; Mehraban, S.; Fakhraei, M.; Ferns, G.A.; Ryzhikov, M.; Gharib, M.; et al. Therapeutic potential of active components of saffron in post-surgical adhesion band formation. J. Tradit. Complement. Med. 2021, 11, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Liu, B.; Zhou, S.; Xu, X.; Yang, Q.; Zhou, J. Pharmacological studies of glycosides of saffron crocus (Crocus sativus). II. Effects on blood coagulation, platelet aggregation and thromobosis. Chin. Tradit. Herbal Drugs 1999, 30, 196–198. [Google Scholar]
- Liu, T.; Chu, X.; Wang, H.; Zhang, X.; Zhang, Y.; Guo, H.; Liu, Z.; Dong, Y.; Liu, H.; Liu, Y.; et al. Crocin, a carotenoid component of Crocus sativus, exerts inhibitory effects on L-type Ca2+ current, Ca2+ transient, and contractility in rat ventricular myocytes. Can. J. Physiol. Pharmacol. 2016, 94, 302–308. [Google Scholar] [CrossRef]
- Jaliani, H.Z.; Riazi, G.H.; Ghaffari, S.M.; Karima, O.; Rahmani, A. The effect of the Crocus sativus L. carotenoid, crocin, on the polymerization of microtubules, in vitro. Iran. J. Basic. Med. Sci. 2013, 16, 101–107. [Google Scholar]
- Mancini, A.; Serrano-Díaz, J.; Nava, E.; D’alessandro, A.M.; Alonso, G.L.; Carmona, M.; Llorens, S. Crocetin, a carotenoid derived from saffron (Crocus sativus L.), improves acetylcholine-induced vascular relaxation in hypertension. J. Vasc. Res. 2014, 51, 393–404. [Google Scholar] [CrossRef]
- Siracusa, L.; Gresta, F.; Ruberto, G. Saffron (Crocus sativus L.) apocarotenoids: A review of their biomolecular features and biological activity perspectives. In Carotenoids: Properties, Effects and Diseases, 2nd ed.; Yamaguchi, M., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2011; pp. 145–178. [Google Scholar]
- Hosseinzadeh, H.; Karimi, G.; Niapoor, M. Antidepressant effect of Crocus sativus L. stigma extracts and their constituents, crocin and safranal, in mice. Acta Hort. 2004, 650, 435–445. [Google Scholar] [CrossRef]
- Vahdati Hassani, F.; Naseri, V.; Razavi, B.M.; Mehri, S.; Abnous, K.; Hosseinzadeh, H. Antidepressant effects of crocin and its effects on transcript and protein levels of CREB, BDNF, and VGF in rat hippocampus. DARU J. Pharm. Sci. 2014, 22, 16. [Google Scholar] [CrossRef]
- Amin, B.; Nakhsaz, A.; Hosseinzadeh, H. Evaluation of the antidepressant-like effects of acute and sub-acute administration of crocin and crocetin in mice. Avicenna J. Phytomed. 2015, 5, 458–468. [Google Scholar]
- Jam, I.N.; Sahebkar, A.H.; Eslami, S.; Mokhber, N.; Nosrati, M.; Khademi, M.; Foroutan-Tanha, M.; Ghayour-Mobarhan, M.; Hadizadeh, F.; Ferns, G.; et al. The effects of crocin on the symptoms of depression in subjects with metabolic syndrome. Adv. Clin. Exp. Med. 2017, 26, 925–930. [Google Scholar]
- Sugiura, M.; Shoyama, Y.; Saito, H.; Nishiyama, N. Crocin improves the ethanol-induced impairment of learning behaviors of mice in passive avoidance tasks. Proc. Jpn. Acad. B Phys. Biol. Sci. 1995, 71, 319–324. [Google Scholar] [CrossRef]
- Hoshyar, R.; Bathaie, S.Z.; Ashrafi, M. Interaction of safranal and picrocrocin with ctDNA and their preferential mechanisms of binding to GC- and AT-rich oligonucleotides. DNA Cell Biol. 2008, 27, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Kang, C.; Sun, Y.; Huang, W.; Liu, W.; Qian, Z. Crocetin inhibits lipopolysaccharide-induced inflammatory response in human umbilical vein endothelial cells. Cell. Physiol. Biochem. 2016, 40, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Rameshrad, M.; Razavi, B.M.; Hosseinzadeh, H. Saffron and its derivatives, crocin, crocetin and safranal: A patent review. Expert Opin. Ther. Pat. 2018, 28, 147–165. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; He, P.; Zhao, D.; Ye, L.; Dai, L.; Zhang, X.; Sun, Y.; Zheng, J.; Bi, C. Construction of Escherichia coli cell factories for crocin biosynthesis. Microb. Cell Factories 2019, 18, 120. [Google Scholar] [CrossRef] [PubMed]
- Acar, B.; Sadikoglu, H.; Ozkaymak, M. Freeze drying of saffron (Crocus sativus L.). Dry Technol. 2011, 29, 1622–1627. [Google Scholar] [CrossRef]
- Zarinkamar, F.; Tajik, S.; Soleimanpour, S. Effects of altitude on anatomy and concentration of crocin, picrocrocin and safranal in Crocus sativus L. Aust. J. Crop Sci. 2011, 5, 831–838. [Google Scholar]
- García-Rodríguez, M.V.; López-Córcoles, H.; Alonso, G.L.; Pappas, C.S.; Polissiou, M.G.; Tarantilis, P.A. Comparative evaluation of an ISO 3632 method and an HPLC-DAD method for safranal quantity determination in saffron. Food Chem. 2017, 221, 838–843. [Google Scholar] [CrossRef]
- Lage, M.; Cantrell, C.L. Quantification of saffron (Crocus sativus L.) metabolites crocins, picrocrocin and safranal for quality determination of the spice grown under different environmental Moroccan conditions. Sci. Hortic. 2009, 121, 366–373. [Google Scholar] [CrossRef]
- Azarabadi, N.; Özdemir, F. Determination of crocin content and volatile components in different qualities of Iranian saffron. Gıda 2018, 43, 476–489. [Google Scholar] [CrossRef]
- Kyriakoudi, A.; Tsimidou, M.Z.; O’Callaghan, Y.C.; Galvin, K.; O’Brien, N.M. Changes in total and individual crocetin esters upon in vitro gastrointestinal digestion of saffron aqueous extracts. J. Agric. Food Chem. 2013, 61, 5318–5327. [Google Scholar] [CrossRef]
- Zhang, A.; Shen, Y.; Cen, M.; Hong, X.; Shao, Q.; Chen, Y.; Zheng, B. Polysaccharide and crocin contents, and antioxidant activity of saffron from different origins. Ind. Crop. Prod. 2019, 133, 111–117. [Google Scholar] [CrossRef]
- Zeka, K.; Ruparelia, K.C.; Continenza, M.A.; Stagos, D.; Vegliò, F.; Arroo, R.R.J. Petals of Crocus sativus L. as a potential source of the antioxidants crocin and kaempferol. Fitoterapia 2015, 107, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Moratalla-López, N.; Zalacain, A.; Bagur, M.J.; Salinas, M.R.; Alonso, G.L. Saffron. In Food Integrity Handbook: A Guide to Food Authenticity Issues and Analytical Solutions; Morin, J.F., Lees, M., Eds.; Eurofins Analytics France: Nantes, France, 2018; pp. 193–204. [Google Scholar]
- Moshiri, E.; Basti, A.A.; Noorbala, A.A.; Jamshidi, A.H.; Hesameddin Abbasi, S.; Akhondzadeh, S. Crocus sativus L. (petal) in the treatment of mild-to-moderate depression: A double-blind, randomized and placebo-controlled trial. Phytomedicine 2006, 13, 607–611. [Google Scholar] [CrossRef] [PubMed]
- Iborra, J.L.; Castellar, M.R.; Cánovas, M.; Manjón, A. Picrocrocin hydrolysis by immobilized β-glucosidase. Biotechnol. Lett. 1992, 14, 475–480. [Google Scholar] [CrossRef]
- Sánchez, A.M.; Carmona, M.; Jarén-Galán, M.; Mínguez Mosquera, M.I.; Alonso, G.L. Picrocrocin kinetics in aqueous saffron spice extracts (Crocus sativus L.) upon thermal treatment. J. Agric. Food Chem. 2011, 59, 249–255. [Google Scholar] [CrossRef]
- Alonso, G.L.; Salinas, M.R.; Garijo, J.; Sánchez-Fernández, M.A. Composition of crocins and picrocrocin from Spanish saffron (Crocus sativus L.). J. Food Qual. 2001, 24, 219–233. [Google Scholar] [CrossRef]
- del Campo, C.P.; Carmona, M.; Maggi, L.; Kanakis, C.D.; Anastasaki, E.G.; Tarantilis, P.A.; Polissiou, M.G.; Alonso, G.L. Picrocrocin content and quality categories in different (345) worldwide samples of sffron (Crocus sativus L.). J. Agric. Food Chem. 2010, 58, 1305–1312. [Google Scholar] [CrossRef]
- Othman, R.; Hatta, F.A.M.; Hassan, N.M.; Kamoona, S. Characterization of carotenoids content and composition of saffron from different localities. J. Pharm. Nutr. Sci. 2020, 10, 34–40. [Google Scholar] [CrossRef]
- Liakopoulou-Kyriakides, M.; Kyriakidi, D. Croscus sativus-biological active constitutents. Stud. Nat. Prod. Chem. 2002, 26, 293–312. [Google Scholar]
- Mashmoul, M.; Azlan, A.; Khaza’ai, H.; Yusof, B.N.M.; Noor, S.M. Saffron: A natural potent antioxidant as a promising anti-obesity drug. Antioxidants 2013, 2, 293–308. [Google Scholar] [CrossRef]
- Yeats, T.H.; Nagel, R. Subcellular spice trade routes: Crocin biosynthesis in the saffron crocus (Crocus sativus). Plant Physiol. 2018, 177, 869–870. [Google Scholar] [CrossRef]
- Bathaie, S.Z.; Mousavi, S.Z. New applications and mechanisms of action of saffron and its important ingredients. Crit. Rev. Food Sci. Nutr. 2010, 50, 761–786. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, H.; Motamedshariaty, V.; Hadizadeh, F. Antidepressant effect of kaempferol, a constituent of saffron (Crocus sativus) petal, in mice and rats. Pharmacologyonline 2007, 2, 367–370. [Google Scholar]
- Mottaghipisheh, J.; Sourestani, M.M.; Kiss, T.; Horváth, A.; Tóth, B.; Ayanmanesh, M.; Ayanmanesh, M.; Khamushi, A.; Csupor, D. Comprehensive chemotaxonomic analysis of saffron crocus tepal and stamen samples, as raw materials with potential antidepressant activity. J. Pharm. Biomed. Anal. 2020, 184, 113183. [Google Scholar] [CrossRef]
- Maggi, L.; Carmona, M.; Zalacain, A.; Kanakis, C.D.; Anastasaki, E.; Tarantilis, P.A.; Polissiou, M.G.; Alonso, G.L. Changes in saffron volatile profile according to its storage time. Food Res. Int. 2010, 43, 1329–1334. [Google Scholar] [CrossRef]
- Srivastava, R.; Ahmed, H.; Dixit, R.K. Crocus sativus L.: A comprehensive review. Pharmacogn. Rev. 2010, 4, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Nyeem, M.A.B.; Alam, M.K.; Mizanur, M.; Khan, R.; Alam, M.S.; Ahammed, M.M. Pharmacological effects of Crocus sativus (zaffran) and its chemical constituents: A review. Int. J. Physiol. Nutr. Phys. Educ. 2018, 3, 593–597. [Google Scholar]
- Feyzi, S.; Varidi, M.; Housaindokht, M.R.; Es’haghi, Z. Innovative method for analysis of safranal under static and dynamic conditions through combination of HS-SPME-GC technique with mathematical modelling. Phytochem. Anal. 2020, 31, 564–574. [Google Scholar] [CrossRef]
- Singla, R.K.; Bhat, G.V. Crocin: An overview. Indo Glob. Res. J. Pharm. Sci. 2011, 1, 281–286. [Google Scholar] [CrossRef]
- Shahi, T.; Assadpour, E.; Jafari, S.M. Main chemical compounds and pharmacological activities of stigmas and tepals of ‘red gold’; saffron. Trends Food Sci. Technol. 2016, 58, 69–78. [Google Scholar] [CrossRef]
- Rocchi, R.; Mascini, M.; Sergi, M.; Compagnone, D.; Mastrocola, D.; Pittia, P. Crocins pattern in saffron detected by UHPLC-MS/MS as marker of quality, process and traceability. Food Chem. 2018, 264, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Hoshyar, R.; Mollaei, H. A comprehensive review on anticancer mechanisms of the main carotenoid of saffron, crocin. J. Pharm. Pharmacol. 2017, 69, 1419–1427. [Google Scholar] [CrossRef] [PubMed]
- Finley, J.W.; Gao, S. A perspective on Crocus sativus L. (Saffron) constituent crocin: A potent water-soluble antioxidant and potential therapy for Alzheimer’s disease. J. Agric. Food Chem. 2017, 65, 1005–1020. [Google Scholar] [CrossRef] [PubMed]
- Khazdair, M.R.; Boskabady, M.H.; Hosseini, M.; Rezaee, R.; Tsatsakis, A.M. The effects of Crocus sativus (saffron) and its constituents on nervous system: A review. Avicenna J. Phytomed. 2015, 5, 376–391. [Google Scholar] [PubMed]
- Milajerdi, A.; Bitarafan, V.; Mahmoudi, M. A review on the effects of saffron extract and its constituents on factors related to neurologic, cardiovascular and gastrointestinal diseases. J. Med. Plants 2015, 3, 9–28. [Google Scholar]
- Farkhondeh, T.; Samarghandian, S.; Shaterzadeh Yazdi, H.; Samini, F. The protective effects of crocin in the management of neurodegenerative diseases: A review. Am. J. Neurodegener. Dis. 2018, 7, 1–10. [Google Scholar]
- Sarris, J. Herbal medicines in the treatment of psychiatric disorders: 10-year updated review. Phytother. Res. 2018, 32, 1147–1162. [Google Scholar] [CrossRef]
- Bian, Y.; Zhao, C.; Lee, S.M. Neuroprotective potency of saffron against neuropsychiatric diseases, neurodegenerative diseases, and other brain disorders: From bench to bedside. Front. Pharmacol. 2020, 6, 579052. [Google Scholar] [CrossRef]
- Ghajar, A.; Neishabouri, S.M.; Velayati, N.; Jahangard, L.; Matinnia, N.; Haghighi, M.; Ghaleiha, A.; Afarideh, M.; Salimi, S.; Meysamie, A.; et al. Crocus sativus L. versus citalopram in the treatment of major depressive disorder with anxious distress: A double-blind, controlled clinical trial. Pharmacopsychiatry 2017, 50, 152–160. [Google Scholar] [CrossRef]
- Noorbala, A.A.; Akhondzadeh, S.; Tamacebi-Pour, N.; Jamshidi, A.H. Hydro-alcoholic extract of Crocus sativus L. versus fluoxetine in the treatment of mild to moderate depression: A double-blind, randomized pilot trial. J. Ethnopharmacol. 2005, 97, 281–284. [Google Scholar] [CrossRef]
- Akhondzadeh Basti, A.; Moshri, E.; Noorbala, A.A.; Jamshidi, A.H.; Abassi, S.H.; Akhondzadeh, S. Comparison of petal of Crocus sativus L. and fluoxetine in the treatment of depressed outpatients: A pilot double-blind randomized trial. Prog. Neuro-Psychopharmocol. Biol. Psychiatry 2007, 31, 439–442. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Chen, L.; Wang, W. Safety and efficacy of saffron (Crocus sativus L.) for treating mild to moderate depression: A systematic review and meta-analysis. J. Nerv. Ment. Dis. 2020, 208, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Kashani, L.; Eslatmanesh, S.; Saedi, N.; Niroomand, N.; Ebrahimi, M.; Hosseinian, M.; Foroughifar, T.; Salimi, S.; Akhondzadeh, S. Comparison of saffron versus fluoxetine in treatment of mild to moderate postpartum depression: A double-blind, randomized clinical trial. Pharmacopsychiatry 2017, 50, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Khaksarian, M.; Behzadifar, M.; Behzadifar, M.; Alipour, M.; Jahanpanah, F.; Re, T.S.; Bragazzi, N.L. The efficacy of Crocus sativus (Saffron) versus placebo and fluoxetine in treating depression: A systematic review and meta-analysis. Psychol. Res. Behav. Manag. 2019, 12, 297–305. [Google Scholar] [CrossRef]
- Ahmadpanah, M.; Ramezanshams, F.; Ghaleiha, A.; Akhondzadeh, S.; Sadeghi Bahmani, D.; Brand, S. Crocus sativus L. (saffron) versus sertraline on symptoms of depression among older people with major depressive disorders-a double-blind, randomized intervention study. Psychiatry Res. 2019, 282, 112613. [Google Scholar] [CrossRef]
- Dorri, S.A.; Hosseinzadeh, H.; Abnous, K.; Hasani, F.V.; Robati, R.Y.; Razavi, B.M. Involvement of brain-derived neurotrophic factor (BDNF) on malathion induced depressive-like behavior in subacute exposure and protective effects of crocin. Iran. J. Basic Med. Sci. 2015, 18, 958–966. [Google Scholar]
- Basiri-Moghadam, M.; Hamzei, A.; Moslem, A.R.; Pasban-Noghabi, S.; Ghorbani, N.; Ghenaati, J. Comparison of the anxiolytic effects of saffron (Crocus sativus. L) and diazepam before herniorrhaphy surgery: A double blind randomized clinical trial. Zahedan J. Res. Med. Sci. 2016, 18, 6248. [Google Scholar] [CrossRef]
- Sahraian, A.; Jelodar, S.; Javid, Z.; Mowla, A.; Ahmadzadeh, L. Study the effects of saffron on depression and lipid profiles: A double blind comparative study. Asian J. Psychiatr. 2016, 22, 174–176. [Google Scholar] [CrossRef]
- Hooshmandi, Z.; Rohani, A.H.; Eidi, A.; Fatahi, Z.; Golmanesh, L.; Sahraei, H. Reduction of metabolic and behavioral signs of acute stress in male Wistar rats by saffron water extract and its constituent safranal. Pharm. Biol. 2011, 49, 947–954. [Google Scholar] [CrossRef][Green Version]
- Georgiadou, G.; Tarantilis, P.; Pitsikas, N. Effects of the active constituents of Crocus sativus L., crocins, in an animal model of obsessive–compulsive disorder. Neurosci. Lett. 2012, 528, 27–30. [Google Scholar] [CrossRef]
- Ettehadi, H.; Mojabi, S.; Ranjbaran, M.; Shams, J.; Sahraei, H.; Hedayati, M.; Asefi, F. Aqueous extract of saffron (Crocus sativus) increases brain dopamine and glutamate concentrations in rats. J. Behav. Brain. Sci. 2013, 3, 315–319. [Google Scholar] [CrossRef]
- Reddy, H.S.G.; Rajashekharappa, R.S.; Jayaram, K.S.; Jyothi, C.H. Evaluation of antidepressant-like activity of Crocus sativus Linn stigmas in mice. Int. J. Pharm. Sci. Rev. Res. 2013, 23, 133–136. [Google Scholar]
- Sunanda, B.P.V.; Rammohan, B.; Kumar, A. The effective study of aqueous extract of Crocus sativus Linn. (Saffron) in depressed mice. Int. J. PharmTech Res. 2014, 6, 1143–1152. [Google Scholar]
- Razavi, B.M.; Sadeghi, M.; Abnous, K.; Vahdati Hasani, F.; Hosseinzadeh, H. Study of the role of CREB, BDNF, and VGF neuropeptide in long term antidepressant activity of crocin in the rat cerebellum. Iran. J. Pharm. Sci. 2017, 16, 1452–1462. [Google Scholar]
- Asrari, N.; Yazdian-Robati, R.; Abnous, K.; Razavi, B.M.; Rashednia, M.; Hasani, F.V.; Hosseinzadeh, H. Antidepressant effects of aqueous extract of saffron and its effects on CREB, P-CREB, BDNF, and VGF proteins in rat cerebellum. J. Pharmacopunct. 2018, 21, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Ceremuga, T.E.; Ayala, M.P.; Chicoine, C.R.W.; Chun, C.S.M.; De Groot, C.J.M.; Henson, D.T.; Randall, C.S.A.; Stanley, C.L.R.; Beaumont, C.D.M. Investigation of the anxiolytic and antidepressant effects of crocin, a compound from saffron (Crocus sativus L.), in the male sprague-dawley rat. AANA J. 2018, 86, 225–233. [Google Scholar]
- Farkhondeh, T.; Samarghandian, S.; Samini, F.; Sanati, A.R. Protective effects of crocetin on depression-like behavior induced by immobilization in rat. CNS Neurol. Disord. Drug Targets 2018, 17, 361–369. [Google Scholar] [CrossRef]
- Zhang, L.; Previn, R.; Lu, L.; Liao, R.F.; Jin, Y.; Wang, R.K. Crocin, a natural product attenuates lipopolysaccharide-induced anxiety and depressive-like behaviors through suppressing NF-kB and NLRP3 signaling pathway. Brain Res. Bull. 2018, 142, 352–359. [Google Scholar] [CrossRef]
- Ghalandari-Shamami, M.; Nourizade, S.; Yousefi, B.; Vafaei, A.A.; Pakdel, R.; Rashidy-Pour, A. Beneficial effects of physical activity and crocin against adolescent stress induced anxiety or depressive-like symptoms and dendritic morphology remodeling in prefrontal cortex in adult male rats. Neurochem. Res. 2019, 44, 917–929. [Google Scholar] [CrossRef]
- Lu, L.; Wu, D.; Wang, K.; Tang, J.; Chen, G. Beneficial effects of crocin against depression via pituitary adenylate cyclase-activating polypeptide. BioMed Res. Int. 2020, 2020, 3903125. [Google Scholar] [CrossRef]
- Xiao, Q.; Xiong, Z.; Yu, C.; Zhou, J.; Shen, Q.; Wang, L.; Xie, X.; Fu, Z. Antidepressant activity of crocin-I is associated with amelioration of neuroinflammation and attenuates oxidative damage induced by corticosterone in mice. Physiol. Behav. 2019, 212, 112699. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; He, Q.; Chen, H.; Lin, Z.; Xu, Y.; Yang, C. Crocin ameliorates chronic obstructive pulmonary disease-induced depression via PI3K/Akt mediated suppression of inflammation. Eur. J. Pharmacol. 2019, 862, 172640. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Wang, W.; Peng, Y.; Deng, Z. Antidepressive effects of kaempferol mediated by reduction of oxidative stress, proinflammatory cytokines and up-regulation of AKT/β-catenin cascade. Metab. Brain Dis. 2019, 34, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Shu, R.; Wu, C.; Tong, Y.; Xiong, Z.; Zhou, J.; Yu, C.; Xie, X.; Fu, Z. Crocin-I alleviates the depression-like behaviors probably via modulating “microbiota-gut-brain” axis in mice exposed to chronic restraint stress. J. Affect. Disord. 2020, 276, 476–486. [Google Scholar] [CrossRef]
- Tang, J.; Lu, L.; Wang, Q.; Liu, H.; Xue, W.; Zhou, T.; Xu, L.; Wang, K.; Wu, D.; Wei, F.; et al. Crocin reverses depression-like behavior in Parkinson disease mice via VTA-mPFC pathway. Mol. Neurobiol. 2020, 57, 3158–3170. [Google Scholar] [CrossRef]
- Orio, L.; Alen, F.; Ballesta, A.; Martin, R.; Gomez de Heras, R. Antianhedonic and antidepressant effects of affron, a standardized saffron (Crocus sativus L.) extract. Molecules 2020, 25, 3207. [Google Scholar] [CrossRef]
- Wu, R.; Xiao, D.; Shan, X.; Dong, Y.; Tao, W.W. Rapid and prolonged antidepressant-like effect of crocin is associated with GHSR mediated hippocampal plasticity-related proteins in mice exposed to prenatal stress. Acs. Chem. Neurosci. 2020, 11, 1159–1170. [Google Scholar] [CrossRef]
- Lin, S.; Li, Q.; Jiang, S.; Xu, Z.; Jiang, Y.; Liu, L.; Jiang, J.; Tong, Y.; Wang, P. Crocetin ameliorates chronic restraint stress-induced depression-like behaviors in mice by regulating MEK/ERK pathways and gut microbiota. J. Ethnopharmacol. 2021, 268, 113608. [Google Scholar] [CrossRef]
- Kiashemshaki, B.; Safakhah, H.A.; Ghanbari, A.; Khaleghian, A.; Miladi-Gorji, H. Saffron (Crocus sativus L.) stigma reduces symptoms of morphine-induced dependence and spontaneous withdrawal in rats. Am. J. Drug Alcohol Abuse. 2021, 47, 170–181. [Google Scholar] [CrossRef]
- Monchaux De Oliveira, C.; Pourtau, L.; Vancassel, S.; Pouchieu, C.; Capuron, L.; Gaudout, D.; Castanon, N. Saffron extract-induced improvement of depressive-like behavior in mice is associated with modulation of monoaminergic neurotransmission. Nutrients 2021, 13, 904. [Google Scholar] [CrossRef]
- Agha-Hosseini, M.; Kashani, L.; Aleyaseen, A.; Ghoreishi, A.; Rahmanpour, H.; Zarrinara, A.R.; Akhondzadeh, S. Crocus sativus L. (saffron) in the treatment of premenstrual syndrome: A double-blind, randomised and placebo-controlled trial. BJOG Int. J. Obstet. 2008, 115, 515–519. [Google Scholar] [CrossRef]
- Mansoori, P.; Akhondzadeh, S.; Raisi, F.; Ghaeli, P.; Jamshidi, A.H.; Nasehi, A.A.; Sohrabi, H.; Saroukhani, S. A randomized, double blind, placebo—Controlled study of safety of the adjunctive saffron on sexual dysfunction induced by a selective serotonin reuptake inhibitor. J. Med. Plants 2011, 10, 121–130. [Google Scholar]
- Moosavi, S.M.; Ahmadi, M.; Amini, M.; Vazirzadeh, B. The effects of 40 and 80 mg hydro-alcoholic extract of Crocus sativus in the treatment of mild to moderate depression. J. Mazandaran. Univ. Med. Sci. 2014, 24, 48–53. [Google Scholar]
- Shahmansouri, N.; Farokhnia, M.; Abbasi, S.H.; Kassaian, S.E.; Noorbala Tafti, A.A.; Gougol, A.; Yekehtaz, H.; Forghani, S.; Mahmoodian, M.; Saroukhani, S.; et al. A randomized, double-blind, clinical trial comparing the efficacy and safety of Crocus sativus L. with fluoxetine for improving mild to moderate depression in post percutaneous coronary intervention patients. J. Affect. Disord. 2014, 155, 216–222. [Google Scholar] [CrossRef]
- Esalatmanesh, S.; Biuseh, M.; Noorbala, A.A.; Mostafavi, S.A.; Rezaei, F.; Mesgarpour, B.; Mohammadinejad, P.; Akhondzadeh, S. Comparison of saffron and fluvoxamine in the treatment of mild to moderate obsessive-compulsive disorder: A double blind randomized clinical trial. Iran. J. Psychiatry 2017, 12, 154–162. [Google Scholar]
- Modabbernia, A.; Sohrabi, H.; Nasehi, A.A.; Raisi, F.; Saroukhani, S.; Jamshidi, A.; Tabrizi, M.; Ashrafi, M.; Akhondzadeh, S. Effect of saffron on fluoxetine-induced sexual impairment in men: Randomized double-blind placebo-controlled trial. Psychopharmacology 2012, 223, 381–388. [Google Scholar] [CrossRef]
- Kashani, L.; Raisi, F.; Saroukhani, S.; Sohrabi, H.; Modabbernia, A.; Nasehi, A.A.; Jamshidi, A.; Ashrafi, M.; Mansouri, P.; Ghaeli, P.; et al. Saffron for treatment of fluoxetine-induced sexual dysfunction in women: Randomized double-blind placebo-controlled study. Hum. Psychopharmacol. Clin. Exp. 2013, 28, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Lopresti, A.L.; Drummond, P.D. Efficacy of curcumin, and a saffron/curcumin combination for the treatment of major depression: A randomised, double-blind, placebo-controlled study. J. Affect. Disord. 2017, 207, 188–196. [Google Scholar] [CrossRef]
- Abedimanesh, N.; Ostadrahimi, A.; Bathaie, S.Z.; Abedimanesh, S.; Motlagh, B.; Jafarabadi, M.A.; Sadeghi, M.T. Effects of saffron aqueous extract and its main constituent, crocin, on health-related quality of life, depression, and sexual desire in coronary artery disease patients: A double-blind, placebo-controlled, randomized clinical trial. Iran. Red Crescent Med. J. 2017, 19, e13676. [Google Scholar] [CrossRef]
- Bangratz, M.; Ait Abdellah, S.; Berlin, A.; Blondeau, C.; Guilbot, A.; Dubourdeaux, M.; Lemoine, P. A preliminary assessment of a combination of rhodiola and saffron in the management of mild-moderate depression. Neuropsychiatr. Dis. Treat. 2018, 2018, 1821–1829. [Google Scholar] [CrossRef]
- Jelodar, G.; Javid, Z.; Sahraian, A.; Jelodar, S. Saffron improved depression and reduced homocysteine level in patients with major depression: A randomized, double-blind study. Avicenna J. Phytomed. 2018, 8, 43–50. [Google Scholar] [PubMed]
- Shakiba, M.; Moazen-Zadeh, E.; Noorbala, A.A.; Jafarinia, M.; Divsalar, P.; Kashani, L.; Shahmansouri, N.; Tafakhori, A.; Bayat, H.; Akhondzadeh, S. Saffron (Crocus sativus) versus duloxetine for treatment of patients with fibromyalgia: A randomized double-blind clinical trial. Avicenna J. Phytomed. 2018, 8, 513–523. [Google Scholar]
- Kashani, L.; Esalatmanesh, S.; Eftekhari, F.; Salimi, S.; Foroughifar, T.; Etesam, F.; Safiaghdam, H.; Moazen-Zadeh, E.; Akhondzadeh, S. Efficacy of Crocus sativus (saffron) in treatment of major depressive disorder associated with post-menopausal hot flashes: A double-blind, randomized, placebo-controlled trial. Arch. Gynecol. Obstet. 2018, 297, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Kell, G.; Rao, A.; Beccaria, G.; Clayton, P.; Inarejos-García, A.M.; Prodanov, M. Saffron a novel saffron extract (Crocus sativus L.) improves mood in healthy adults over 4 weeks in a double-blind, parallel, randomized, placebo-controlled clinical trial. Complement. Ther. Med. 2017, 33, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Lopresti, A.L.; Smith, S.J.; Hood, S.D.; Drummond, P.D. Efficacy of a standardised saffron extract saffron as an add-on to antidepressant medication for the treatment of persistent depressive symptoms in adults: A randomised, double-blind, placebo-controlled study. J. Psychopharmacol. 2019, 33, 1415–1427. [Google Scholar] [CrossRef]
- Ghaderi, A. Clinical and metabolic responses to crocin in patients under methadone maintenance treatment, a randomized clinical trial. Phytother. Res. 2019, 33, 2714–2725. [Google Scholar] [CrossRef]
- Lopresti, A.L.; Smith, S.J. The effects of a saffron extract saffron on menopausal symptoms in women during perimenopause: A randomised, double-blind, placebo-controlled study. J. Menopausal. Med. 2021, 2, 66–78. [Google Scholar] [CrossRef]
- Jalali, F.; Hashemi, S. The effect of saffron on depression among recovered consumers of methamphetamine living with HIV/AIDS. Subst. Use Misuse. 2018, 53, 1951–1957. [Google Scholar] [CrossRef]
- Milajerdi, A.; Jazayeri, S.; Shirzadi, E.; Hashemzadeh, N.; Azizgol, A.; Djazayery, A.; Esmaillzadeh, A.; Akhondzadeh, S. The effects of alcoholic extract of saffron (Crocus satious L.) on mild to moderate comorbid depression-anxiety, sleep quality, and life satisfaction in type 2 diabetes mellitus: A double-blind, randomized and placebo-controlled clinical trial. Complement. Ther. Med. 2018, 41, 196–202. [Google Scholar] [CrossRef]
- Moazen-Zadeh, E.; Abbasi, S.H.; Safi-Aghdam, H.; Shahmansouri, N.; Arjmandi-Beglar, A.; Hajhosseinn Talasaz, A.; Salehiomran, A.; Forghani, S.; Akhondzadeh, S. Effects of saffron on cognition, anxiety, and depression in patients undergoing coronary artery bypass grafting: A randomized double-blind placebo-controlled trial. J. Altern. Complement. Med. 2018, 24, 361–368. [Google Scholar] [CrossRef]
- Khalatbari-Mohseni, A.; Banafshe, H.R.; Mirhosseini, N.; Asemi, Z.; Ghaderi, A.; Omidi, A. The effects of crocin on psychological parameters in patients under methadone maintenance treatment: A randomized clinical trial. Subst. Abuse Treat. Prev. Policy 2019, 14, 9. [Google Scholar] [CrossRef] [PubMed]
- Akhondzadeh, S.; Mostafavi, S.A.; Keshavarz, S.A.; Mohammadi, M.R.; Hosseini, S.; Eshraghian, M.R. A placebo controlled randomized clinical trial of Crocus sativus L. (saffron) on depression and food craving among overweight women with mild to moderate depression. J. Clin. Pharm. Ther. 2020, 45, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Nemat-Shahi, M.; Asadi, A.; Nemat-Shahi, M.; Soroosh, D.; Mozari, S.; Bahrami-Taghanaki, H.; Mehrpour, M. Comparison of saffron versus fluoxetine in treatment of women with premenstrual syndrome: A randomized clinical trial study. Indian J. Forensic Med. Toxicol. 2020, 14, 1760–1765. [Google Scholar]
- Jackson, P.A.; Forster, J.; Khan, J.; Pouchieu, C.; Dubreuil, S.; Gaudout, D.; Moras, B.; Pourtau, L.; Joffre, F.; Vaysse, C.; et al. Effects of saffron extract supplementation on mood, well-being, and response to a psychosocial stressor in healthy adults: A randomized, double-blind, parallel group, clinical trial. Front. Nutr. 2021, 7, 606124. [Google Scholar] [CrossRef] [PubMed]
- Moghadam, B.H.; Bagheri, R.; Roozbeh, B.; Ashtary-Larky, D.; Gaeini, A.A.; Dutheil, F.; Wong, A. Impact of saffron (Crocus Sativus Linn) supplementation and resistance training on markers implicated in depression and happiness levels in untrained young males. Physiol. Behav. 2021, 233, 113352. [Google Scholar] [CrossRef] [PubMed]
- Salek, R.; Dehghani, M.; Mohajeri, S.A.; Talaei, A.; Fanipakdel, A.; Javadinia, S.A. Amelioration of anxiety, depression, and chemotherapy-related toxicity after crocin administration during chemotherapy of breast cancer: A double-blind, randomized clinical trial. Phytother. Res. 2021, 35, 5143–5153. [Google Scholar] [CrossRef]
- Tajaddini, A.; Roshanravan, N.; Mobasseri, M.; Aeinehchi, A.; Sefid-Mooye Azar, P.; Hadi, A.; Ostadrahimi, A. Saffron improves life and sleep quality, glycaemic status, lipid profile and liver function in diabetic patients: A double-blind, placebo-controlled, randomised clinical trial. Int. J. Clin. Pract. 2021, 75, e14334. [Google Scholar] [CrossRef]
- Jomehpour, H.; Aghayan, S.; Khosravi, A.; Afzaljavan, F. The effect of krocina on decreasing substance user withdrawal syndrome, craving, depression and stress: A double-blind randomized parallel clinical trial. Subst. Use Misuse 2022, 57, 613–620. [Google Scholar] [CrossRef]
- Bashir, H.; Kumhar, S.H.; Qadir, W. Effect of Crocus sativus (saffron) on corona virus. World J. Pharm. Pharm. Sci. 2021, 10, 860–876. [Google Scholar]
- Husaini, A.M.; Jan, K.N.; Wani, G.A. Saffron: A potential drug-supplement for severe acute respiratory syndrome coronavirus (COVID) management. Heliyon 2021, 7, e07068. [Google Scholar] [CrossRef]
- Mentis, A.F.A.; Dalamaga, M.; Lu, C.; Polissiou, M.G. Saffron for “toning down” COVID-19-related cytokine storm: Hype or hope? A mini-review of current evidence. Metab. Open 2021, 11, 100111. [Google Scholar] [CrossRef]
- Soheilipur, K.; Khazdair, M.R.; Moezi, S.A.; Mahmoudirad, G. Comparing the effects of saffron, lippia, and saffron-lippia combination on anxiety among candidates for coronary angiography. Avicenna J. Phytomed. 2021, 11, 515–526. [Google Scholar]
- Shahdadi, H.; Balouchi, A.; Dehghanmehr, S. Effect of saffron oral capsule on anxiety and quality of sleep of diabetic patients in a tertiary healthcare facility in southeastern Iran: A quasi-experimental study. Trop. J. Pharm. Res. 2017, 16, 2749–2753. [Google Scholar] [CrossRef]
- Pachikian, B.D.; Copine, S.; Suchareau, M.; Deldicque, L. Effects of saffron extract on sleep quality: A randomized double-blind controlled clinical trial. Nutrients 2021, 13, 1473. [Google Scholar] [CrossRef]
- Lopresti, A.L.; Smith, S.J.; Metse, A.P.; Drummond, P.D. Effects of saffron on sleep quality in healthy adults with self-reported poor sleep: A randomized, double-blind, placebo-controlled trial. J. Clin. Sleep. Med. 2020, 16, 937–947. [Google Scholar] [CrossRef] [PubMed]
- Lopresti, A.L.; Smith, S.J.; Drummond, P.D. An investigation into an evening intake of a saffron extract (Saffron®) on sleep quality, cortisol, and melatonin concentrations in adults with poor sleep: A randomised, double-blind, placebo-controlled, multi-dose study. Sleep Med. 2021, 86, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Umigai, N.; Takeda, R.; Mori, A. Effect of crocetin on quality of sleep: A randomized, double-blind, placebo-controlled, crossover study. Complement. Ther. Clin. Pract. 2018, 41, 47–51. [Google Scholar] [CrossRef]
- Fukui, H.; Toyoshima, K.; Komaki, R. Psychological and neuroendocrinological effects of odor of saffron (Crocus sativus). Phytomedicine 2011, 18, 726–730. [Google Scholar] [CrossRef]
- Lechtenberg, M.; Schepmann, D.; Niehues, M.; Hellenbrand, N.; Wünsch, B.; Hensel, A. Quality and functionality of saffron: Quality control, species assortment and affinity of extract and isolated saffron compounds to NMDA and sigma1 (sigma-1) receptors. Planta Med. 2008, 74, 764–772. [Google Scholar] [CrossRef]
- Abe, K.; Sugiura, M.; Shoyama, Y.; Saito, H. Crocin antagonizes ethanol inhibition of NMDA receptor-mediated responses in rat hippocampal neurons. Brain Res. 1998, 787, 132–138. [Google Scholar] [CrossRef]
- Hausenblas, H.A.; Saha, D.; Dubyak, P.J.; Anton, S.D. Saffron (Crocus sativus L.) and major depressive disorder: A meta-analysis of randomized clinical trials. J. Integr. Med. 2013, 1, 377–383. [Google Scholar] [CrossRef]
- Dovrtělová, G.; Nosková, K.; Juřica, J.; Turjap, M.; Zendulka, O. Can bioactive compounds of Crocus sativus L. influence the metabolic activity of selected CYP enzymes in the rat? Physiol. Res. 2015, 64 (Suppl. 4), 453–458. [Google Scholar] [CrossRef]
- Dahchour, A. The effects of saffron (Crocus sativus L.) and its components on depression: From basic to clinical studies. Moroccan J. Biol. 2021, 18, 10–33. [Google Scholar]
- Siddiqui, S.A.; Ali Redha, A.; Snoeck, E.R.; Singh, S.; Simal-Gandara, J.; Ibrahim, S.A.; Jafari, S.M. Anti-depressant properties of crocin molecules in saffron. Molecules 2022, 27, 2076. [Google Scholar] [CrossRef] [PubMed]
- Perlis, R.H. Cytochrome P450 genotyping and antidepressants. Br. Med. J. 2007, 334, 7597. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dubovsky, S.L. The usefulness of genotyping cytochrome P450 enzymes in the treatment of depression. Expert. Opin. Drug. Metab. Toxicol. 2015, 11, 369–379. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).