Quercetin and Kaempferol as Multi-Targeting Antidiabetic Agents against Mouse Model of Chemically Induced Type 2 Diabetes
Abstract
:1. Introduction
2. Results
2.1. In Silico Studies
2.1.1. Drug Scan and ADMET Profiling
2.1.2. Molecular Docking
2.2. In Vitro Anti-Hyperglycemic Activity Using α-Amylase Inhibition Assay
2.3. In Vivo Antidiabetic Activities
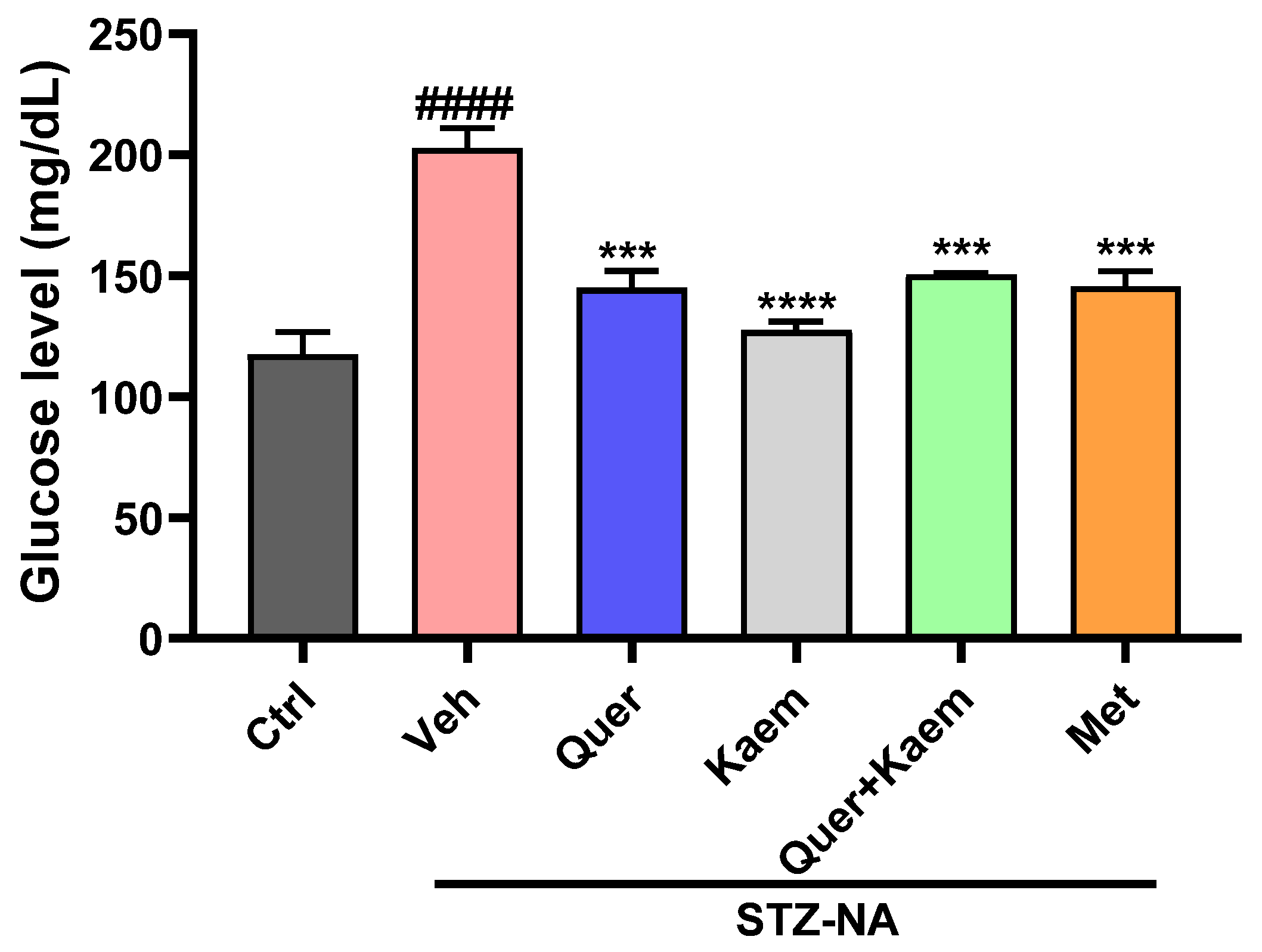
2.3.1. Effect of Quercetin, Kaempferol, and Their Combination on Blood Glucose Levels of Diabetic Mice
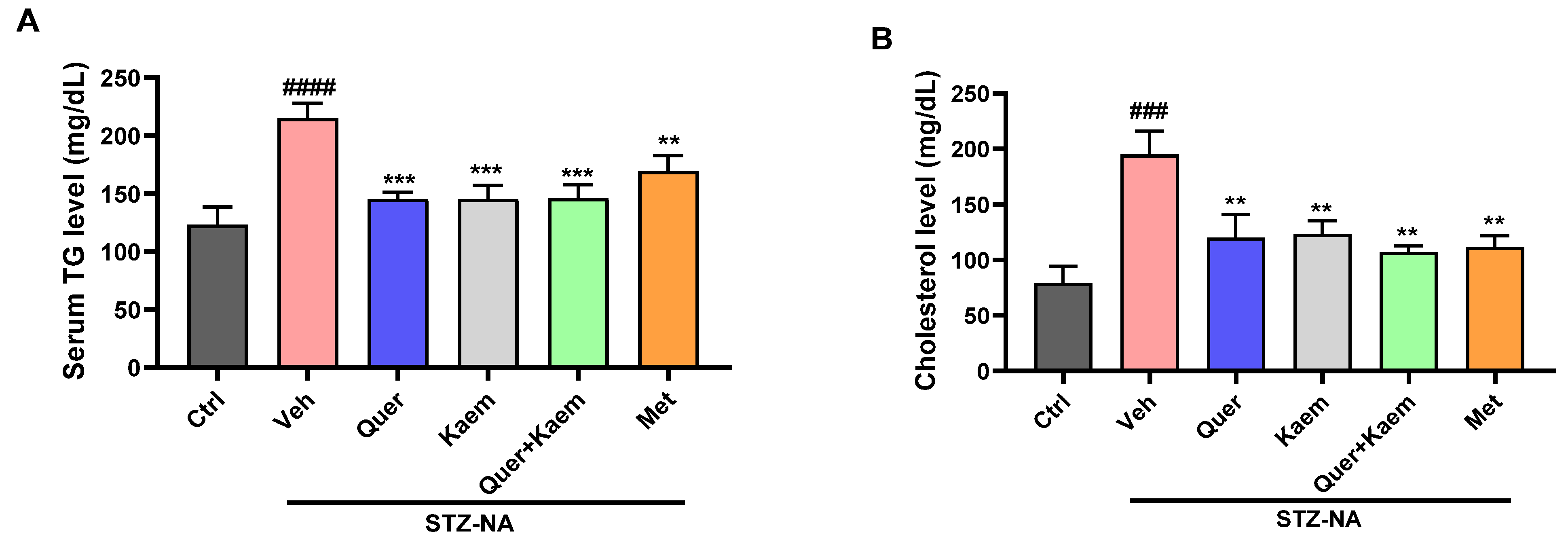
2.3.2. Effect of Quercetin, Kaempferol, and Their Combination on Lipid Profile of Diabetic Mice
2.3.3. Effect of Quercetin, Kaempferol, and Their Combination on Hepatic Markers of Diabetic Mice
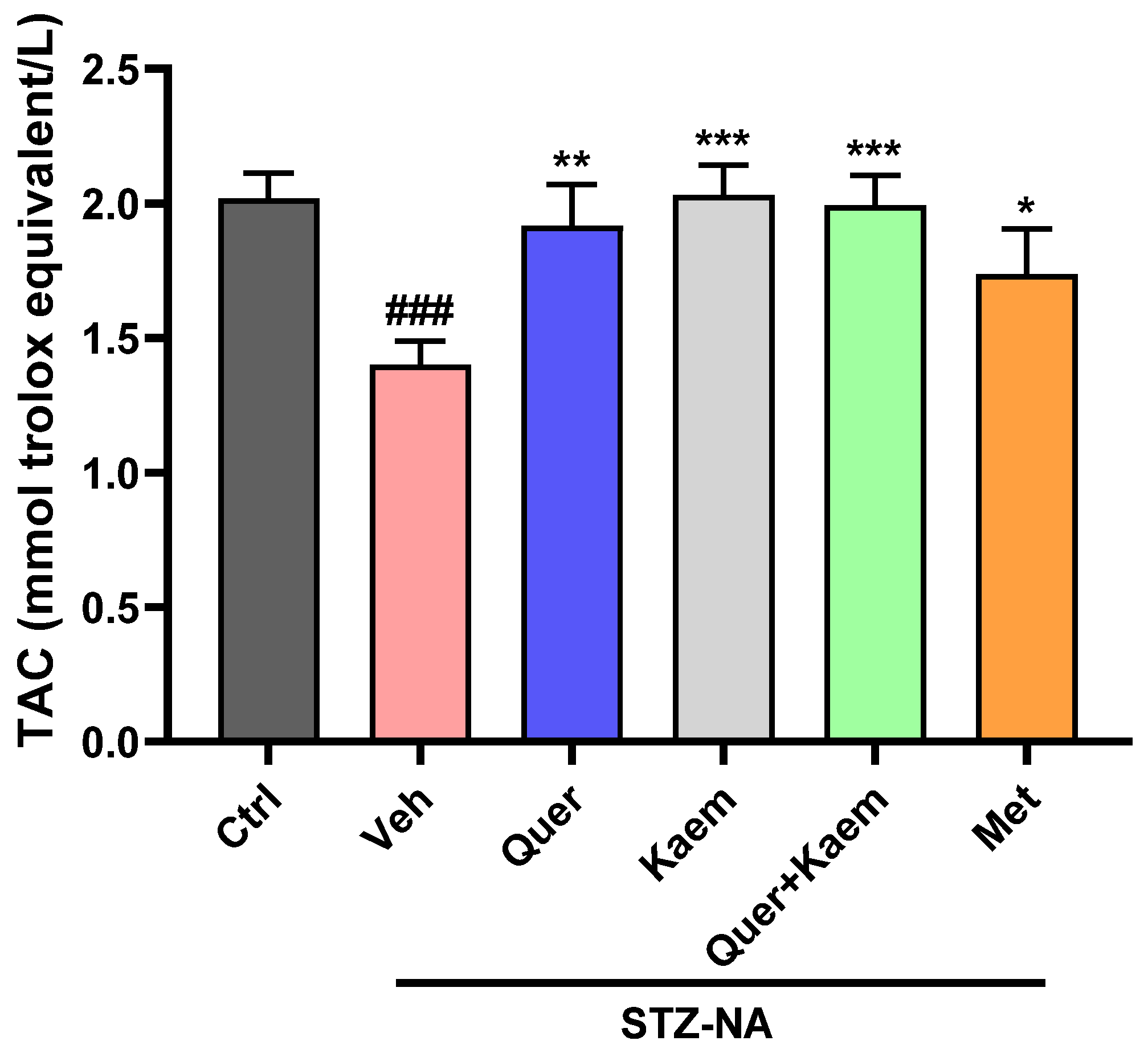
2.3.4. Effect of Quercetin, Kaempferol, and Their Combination on Total Antioxidant Status (TAC) of Hepatic Tissues
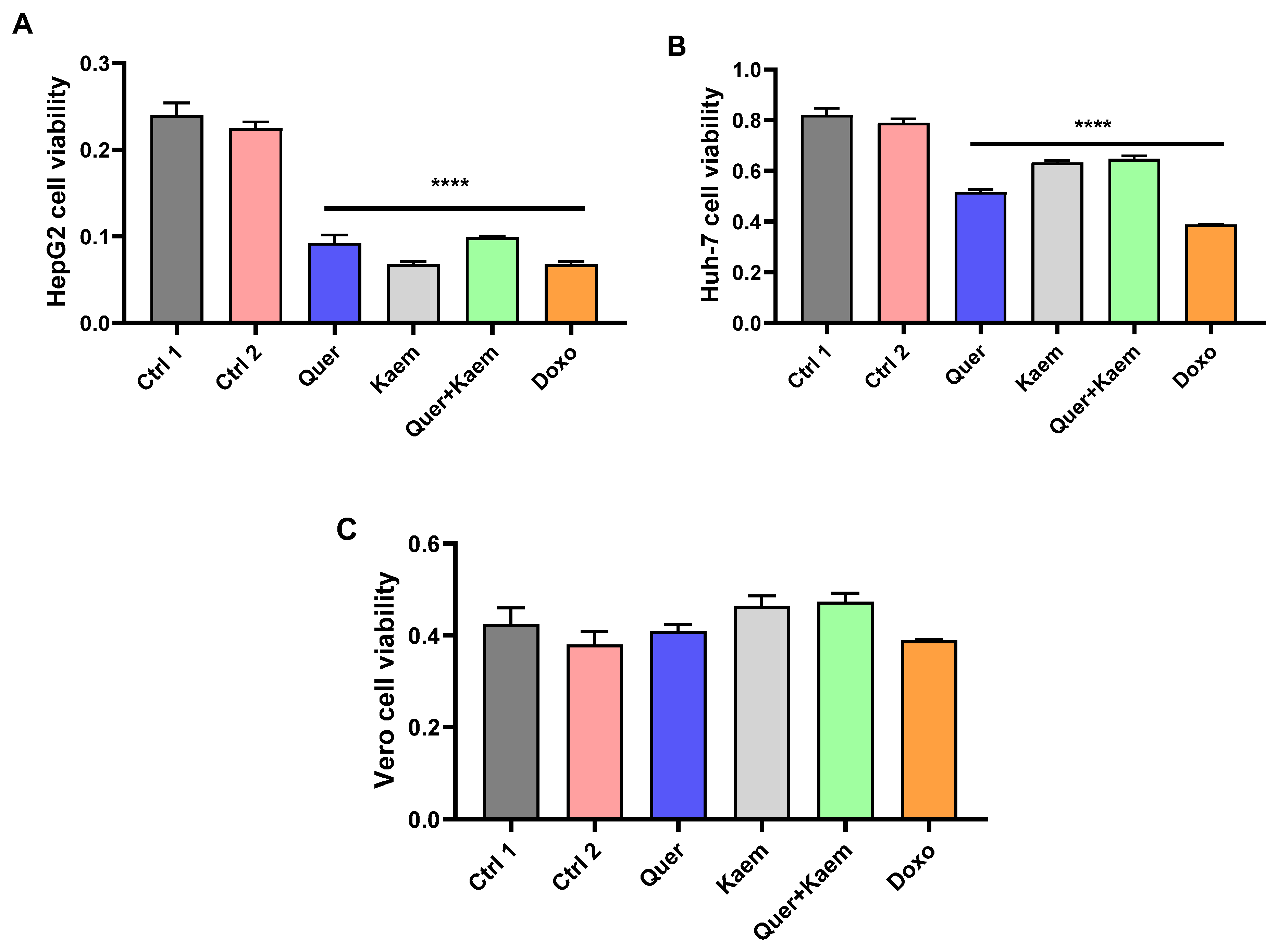
2.4. Cell Viability Assay
3. Discussion
4. Materials and Methods
4.1. Reagents and Chemicals
4.2. Cell Lines
4.3. In Silico Studies
4.3.1. Drug Scan and ADMET Profiling
4.3.2. Protein and Ligand Preparation
4.3.3. Docking Analysis
4.4. In Vitro Alpha-Amylase Inhibition Assay
4.5. In Vivo Studies
4.5.1. Experimental Animals
4.5.2. Induction of Diabetes
4.5.3. Animal Grouping
4.5.4. Blood Sampling and Glucose Level Analysis
4.5.5. Lipid Profile
4.5.6. Levels of Bilirubin, ALT, and ALP
4.5.7. Total Antioxidant Capacity (TAC)
4.6. Cell Viability Assay
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alam, U.; Asghar, O.; Azmi, S.; Malik, R.A. General aspects of diabetes mellitus. Handb. Clin. Neurol. 2014, 126, 211–222. [Google Scholar] [PubMed]
- Abdalrada, A.S.; Abawajy, J.; Al-Quraishi, T.; Islam, S.M.S. Machine learning models for prediction of co-occurrence of diabetes and cardiovascular diseases: A retrospective cohort study. J. Diabetes Metab. Disord. 2022, 21, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Lachin, T. Effect of antioxidant extract from cherries on diabetes. Recent Pat. Endocr. Metab. Immune Drug Discov. 2014, 8, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Al-Goblan, A.S.; Al-Alfi, M.A.; Khan, M.Z. Mechanism linking diabetes mellitus and obesity. Diabetes Metab. Syndr. Obes. Targets Ther. 2014, 7, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, G.; Kaliamurthi, S.; Thirugnasambandan, R. Effect of Glycosin alkaloid from Rhizophora apiculata in non-insulin dependent diabetic rats and its mechanism of action: In vivo and in silico studies. Phytomedicine 2016, 23, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Asmat, U.; Abad, K.; Ismail, K. Diabetes mellitus and oxidative stress—A concise review. Saudi Pharm. J. 2016, 24, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Shikata, K.; Ninomiya, T.; Kiyohara, Y. Diabetes mellitus and cancer risk: Review of the epidemiological evidence. Cancer Sci. 2013, 104, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Kaul, K.; Tarr, J.M.; Ahmad, S.I.; Kohner, E.M.; Chibber, R. Introduction to diabetes mellitus. In Diabetes: An Old Disease, a New Insight; Springer: New York, NY, USA, 2013; pp. 1–11. [Google Scholar]
- Engler, C.; Leo, M.; Pfeifer, B.; Juchum, M.; Chen-Koenig, D.; Poelzl, K.; Schoenherr, H.; Vill, D.; Oberdanner, J.; Eisendle, E. Long-term trends in the prescription of antidiabetic drugs: Real-world evidence from the Diabetes Registry Tyrol 2012–2018. BMJ Open Diabetes Res. Care 2020, 8, e001279. [Google Scholar] [CrossRef] [PubMed]
- Garber, A. Liraglutide in oral antidiabetic drug combination therapy. Diabetes Obes. Metab. 2012, 14, 13–19. [Google Scholar] [CrossRef]
- Rashied, R.M.; Abdelfattah, M.A.; El-Beshbishy, H.A.; ElShazly, A.M.; Mahmoud, M.F.; Sobeh, M. Syzygium samarangense leaf extract exhibits distinct antidiabetic activities: Evidences from in silico and in vivo studies. Arab. J. Chem. 2022, 15, 103822. [Google Scholar] [CrossRef]
- Li, S.; Zhang, B.; Zhang, N. Network target for screening synergistic drug combinations with application to traditional Chinese medicine. BMC Syst. Biol. 2011, 5, S10. [Google Scholar] [CrossRef]
- Tassopoulou, V.P.; Tzara, A.; Kourounakis, A.P. Design of Improved Antidiabetic Drugs: A Journey from Single to Multitarget Agents. ChemMedChem 2022, 23, e202200320. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, K.C.; Haixiao, J.; Naman, C.B.; Sajeevan, T. Prospects of multitarget drug designing strategies by linking molecular docking and molecular dynamics to explore the protein–ligand recognition process. Drug Dev. Res. 2020, 81, 685–699. [Google Scholar] [CrossRef] [PubMed]
- Brogi, S.; Ramalho, T.C.; Kuca, K.; Medina-Franco, J.L.; Valko, M. In silico methods for drug design and discovery. Front. Chem. 2020, 8, 612. [Google Scholar] [CrossRef] [PubMed]
- Adelusi, T.I.; Oyedele, A.-Q.K.; Boyenle, I.D.; Ogunlana, A.T.; Adeyemi, R.O.; Ukachi, C.D.; Idris, M.O.; Olaoba, O.T.; Adedotun, I.O.; Kolawole, O.E. Molecular modeling in drug discovery. Inform. Med. Unlocked 2022, 29, 100880. [Google Scholar] [CrossRef]
- Oselusi, S.O.; Egieyeh, S.A.; Christoffels, A. Cheminformatic profiling and hit prioritization of natural products with activities against methicillin-resistant Staphylococcus aureus (MRSA). Molecules 2021, 26, 3674. [Google Scholar] [CrossRef] [PubMed]
- Oselusi, S.O.; Christoffels, A.; Egieyeh, S.A. Cheminformatic characterization of natural antimicrobial products for the development of new lead compounds. Molecules 2021, 26, 3970. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhu, W. Molecular docking for drug discovery and development: A widely used approach but far from perfect. Future Med. Chem. 2016, 8, 1707–1710. [Google Scholar] [CrossRef] [PubMed]
- Opara, E.C. Role of oxidative stress in the etiology of type 2 diabetes and the effect of antioxidant supplementation on glycemic control. J. Invest. Med. 2004, 52, 19. [Google Scholar] [CrossRef]
- Barwal, I.; Sood, A.; Sharma, M.; Singh, B.; Yadav, S.C. Development of stevioside Pluronic-F-68 copolymer based PLA-nanoparticles as an antidiabetic nanomedicine. Colloids Surf. B Biointerfaces 2013, 101, 510–516. [Google Scholar] [CrossRef]
- Van, L.V.; Pham, E.C.; Nguyen, C.V.; Duong, N.T.N.; Le Thi, T.V.; Truong, T.N. In vitro and in vivo antidiabetic activity, isolation of flavonoids, and in silico molecular docking of stem extract of Merremia tridentata (L.). Biomed. Pharmacother. 2022, 146, 112611. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.J. Metformin: Historical overview. Diabetologia 2017, 60, 1566–1576. [Google Scholar] [CrossRef] [PubMed]
- Jan, R.; Khan, M.; Asaf, S.; Lubna; Asif, S.; Kim, K.-M. Bioactivity and therapeutic potential of kaempferol and quercetin: New insights for plant and human health. Plants 2022, 11, 2623. [Google Scholar] [CrossRef] [PubMed]
- Suganthy, N.; Devi, K.P.; Nabavi, S.F.; Braidy, N.; Nabavi, S.M. Bioactive effects of quercetin in the central nervous system: Focusing on the mechanisms of actions. Biomed. Pharmacother. 2016, 84, 892–908. [Google Scholar] [CrossRef] [PubMed]
- Minaei, A.; Sabzichi, M.; Ramezani, F.; Hamishehkar, H.; Samadi, N. Co-delivery with nano-quercetin enhances doxorubicin-mediated cytotoxicity against MCF-7 cells. Mol. Biol. Rep. 2016, 43, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Dewi, B.; Desti, H.; Ratningpoeti, E.; Sudiro, M.; Angelina, M. Effectivity of quercetin as antiviral to dengue virus-2 strain New Guinea C in Huh 7-it 1 cell line. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Tangerang, Indonesia, 23–24 October 2019; p. 012033. [Google Scholar]
- Lee, H.N.; Shin, S.A.; Choo, G.S.; Kim, H.J.; Park, Y.S.; Kim, B.S.; Kim, S.K.; Cho, S.D.; Nam, J.S.; Choi, C.S. Anti-inflammatory effect of quercetin and galangin in LPS-stimulated RAW264. 7 macrophages and DNCB-induced atopic dermatitis animal models. Int. J. Mol. Med. 2018, 41, 888–898. [Google Scholar] [PubMed]
- Pei, Y.; Otieno, D.; Gu, I.; Lee, S.-O.; Parks, J.S.; Schimmel, K.; Kang, H.W. Effect of quercetin on nonshivering thermogenesis of brown adipose tissue in high-fat diet-induced obese mice. J. Nutr. Biochem. 2021, 88, 108532. [Google Scholar] [CrossRef] [PubMed]
- Aghababaei, F.; Hadidi, M. Recent advances in potential health benefits of quercetin. Pharmaceuticals 2023, 16, 1020. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Salehi, B.; Sharifi-Rad, J.; Aslam Gondal, T.; Saeed, F.; Imran, A.; Shahbaz, M.; Tsouh Fokou, P.V.; Umair Arshad, M.; Khan, H. Kaempferol: A key emphasis to its anticancer potential. Molecules 2019, 24, 2277. [Google Scholar] [CrossRef]
- Yu, L.; Chen, C.; Wang, L.-F.; Kuang, X.; Liu, K.; Zhang, H.; Du, J.-R. Neuroprotective effect of kaempferol glycosides against brain injury and neuroinflammation by inhibiting the activation of NF-κB and STAT3 in transient focal stroke. PLoS ONE 2013, 8, e55839. [Google Scholar] [CrossRef]
- Shih, T.-Y.; Young, T.-H.; Lee, H.-S.; Hsieh, C.-B.; Hu, O.Y.-P. Protective effects of kaempferol on isoniazid-and rifampicin-induced hepatotoxicity. AAPS J. 2013, 15, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Li, W.; Zhou, Y.; Shen, J.; Wu, Z.; Liu, G.; Lee, P.W.; Tang, Y. admetSAR: A Comprehensive Source and Free Tool for Assessment of Chemical ADMET Properties; ACS Publications: Washington, DC, USA, 2012. [Google Scholar]
- Mahrosh, H.S.; Tanveer, M.; Arif, R.; Mustafa, G. Computer-aided prediction and identification of phytochemicals as potential drug candidates against MERS-CoV. BioMed Res. Int. 2021, 2021, 5578689. [Google Scholar] [CrossRef]
- Butt, S.S.; Badshah, Y.; Shabbir, M.; Rafiq, M. Molecular docking using chimera and autodock vina software for nonbioinformaticians. JMIR Bioinform. Biotechnol. 2020, 1, e14232. [Google Scholar] [CrossRef]
- Gery, I.; Gershon, R.K.; Waksman, B.H. Potentiation of the T-lymphocyte response to mitogens: I. The responding cell. J. Exp. Med. 1972, 136, 128–142. [Google Scholar] [CrossRef]
- D’Alessandris, C.; Lauro, R.; Presta, I.; Sesti, G. C-reactive protein induces phosphorylation of insulin receptor substrate-1 on Ser307 and Ser612 in L6 myocytes, thereby impairing the insulin signalling pathway that promotes glucose transport. Diabetologia 2007, 50, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Shree, P.; Tripathi, Y.B. Active phytochemicals of Pueraria tuberosa for DPP-IV inhibition: In silico and experimental approach. J. Diabetes Metab. Disord. 2017, 16, 46. [Google Scholar] [CrossRef]
- Verma, S.K.; Thareja, S. Molecular docking assisted 3D-QSAR study of benzylidene-2, 4-thiazolidinedione derivatives as PTP-1B inhibitors for the management of Type-2 diabetes mellitus. RSC Adv. 2016, 6, 33857–33867. [Google Scholar] [CrossRef]
- Thiyagarajan, G.; Muthukumaran, P.; Sarath Kumar, B.; Muthusamy, V.S.; Lakshmi, B.S. Selective Inhibition of PTP 1B by Vitalboside A from Syzygium cumini Enhances Insulin Sensitivity and Attenuates Lipid Accumulation Via Partial Agonism to PPAR γ: In Vitro and In Silico Investigation. Chem. Biol. Drug Des. 2016, 88, 302–312. [Google Scholar] [CrossRef]
- Guo, Y.; Ran, Z.; Zhang, Y.; Song, Z.; Wang, L.; Yao, L.; Zhang, M.; Xin, J.; Mao, X. Marein ameliorates diabetic nephropathy by inhibiting renal sodium glucose transporter 2 and activating the AMPK signaling pathway in db/db mice and high glucose–treated HK-2 cells. Biomed. Pharmacother. 2020, 131, 110684. [Google Scholar] [CrossRef]
- Mechchate, H.; Es-Safi, I.; Bourhia, M.; Kyrylchuk, A.; El Moussaoui, A.; Conte, R.; Ullah, R.; Ezzeldin, E.; Mostafa, G.A.; Grafov, A. In-vivo antidiabetic activity and in-silico mode of action of LC/MS-MS identified flavonoids in oleaster leaves. Molecules 2020, 25, 5073. [Google Scholar] [CrossRef]
- Saravanan, S.; Parimelazhagan, T. In vitro antioxidant, antimicrobial and anti-diabetic properties of polyphenols of Passiflora ligularis Juss. fruit pulp. Food Sci. Hum. Wellness 2014, 3, 56–64. [Google Scholar] [CrossRef]
- Franz, M.J. Protein: Metabolism and effect on blood glucose levels. Diabetes Educ. 1997, 23, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Hokanson, J.E.; Austin, M.A. Plasma triglyceride level is a risk factor for cardiovascular disease independent of high-density lipoprotein cholesterol level: A metaanalysis of population-based prospective studies. Eur. J. Cardiovasc. Prev. Rehabil. 1996, 3, 213–219. [Google Scholar] [CrossRef]
- Lippi, G.; Schena, F.; Montagnana, M.; Salvagno, G.L.; Banfi, G.; Guidi, G.C. Significant variation of traditional markers of liver injury after a half-marathon run. Eur. J. Intern. Med. 2011, 22, e36–e38. [Google Scholar] [CrossRef]
- Opara, E.C.; Abdel-Rahman, E.; Soliman, S.; Kamel, W.A.; Souka, S.; Lowe, J.E.; Abdel-Aleem, S. Depletion of total antioxidant capacity in type 2 diabetes. Metabolism 1999, 48, 1414–1417. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Forte, V.; Abdallah, M.; Alickaj, A.; Mahmud, S.; Asad, S.; McFarlane, S. Diabetes mellitus and the risk of cancer. Minerva Endocrinol. 2011, 36, 187–209. [Google Scholar]
- González-Álvarez, H.; Bravo-Jiménez, A.; Martínez-Arellanes, M.; Gamboa-Osorio, G.O.; Chávez-Gutiérrez, E.; González-Hernández, L.A.; Gallardo-Ignacio, K.; Quintana-Romero, O.J.; Ariza-Castolo, A.; Guerra-Araiza, C. In silico-based design and in vivo evaluation of an anthranilic acid derivative as a multitarget drug in a diet-induced metabolic syndrome model. Pharmaceuticals 2021, 14, 914. [Google Scholar] [CrossRef]
- Akinyede, K.A.; Oyewusi, H.A.; Hughes, G.D.; Ekpo, O.E.; Oguntibeju, O.O. In vitro evaluation of the anti-diabetic potential of aqueous acetone Helichrysum petiolare Extract (AAHPE) with molecular docking relevance in diabetes mellitus. Molecules 2021, 27, 155. [Google Scholar] [CrossRef]
- Banerjee, M.; Saxena, M. Interleukin-1 (IL-1) family of cytokines: Role in type 2 diabetes. Clin. Chim. Acta 2012, 413, 1163–1170. [Google Scholar] [CrossRef]
- Bharti, S.K.; Krishnan, S.; Kumar, A.; Rajak, K.K.; Murari, K.; Bharti, B.K.; Gupta, A.K. Antihyperglycemic activity with DPP-IV inhibition of alkaloids from seed extract of Castanospermum australe: Investigation by experimental validation and molecular docking. Phytomedicine 2012, 20, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Patil, V.S.; Biradar, P.R.; Attar, V.; Khanal, P. In silico Docking Analysis of Active Biomolecules from Cissus quadrangularis L. against PPAR-γ. Indian J. Pharm. Educ. 2019, 53, S332–S337. [Google Scholar] [CrossRef]
- Elhady, S.S.; Youssef, F.S.; Alahdal, A.M.; Almasri, D.M.; Ashour, M.L. Anti-hyperglycaemic evaluation of Buddleia indica leaves using in vitro, in vivo and in silico studies and its correlation with the major phytoconstituents. Plants 2021, 10, 2351. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Yang, J.; Wang, C.; Wu, L.; Liu, Y. Screening bifunctional flavonoids of anti-cholinesterase and anti-glucosidase by in vitro and in silico studies: Quercetin, kaempferol and myricetin. Food Biosci. 2023, 51, 102312. [Google Scholar] [CrossRef]
- Kazemi, T.; Hajihosseini, M.; Moossavi, M.; Hemmati, M.; Ziaee, M. Cardiovascular risk factors and atherogenic indices in an Iranian population: Birjand East of Iran. Clin. Med. Insights Cardiol. 2018, 12, 1179546818759286. [Google Scholar] [CrossRef] [PubMed]
- Soliman, G.Z. Anti-diabetic activity of dried Moringa oleifera leaves in normal and streptozotocin (STZ)-induced diabetic male rats. Indian J. Appl. Res 2013, 3, 18–23. [Google Scholar] [CrossRef]
- Jeong, S.-M.; Kang, M.-J.; Choi, H.-N.; Kim, J.-H.; Kim, J.-I. Quercetin ameliorates hyperglycemia and dyslipidemia and improves antioxidant status in type 2 diabetic db/db mice. Nutr. Res. Pract. 2012, 6, 201–207. [Google Scholar] [CrossRef]
- Widyawati, T.; Syahputra, R.A.; Syarifah, S.; Sumantri, I.B. Analysis of Antidiabetic Activity of Squalene via In Silico and In Vivo Assay. Molecules 2023, 28, 3783. [Google Scholar] [CrossRef] [PubMed]
- Nyblom, H.; Björnsson, E.; Simrén, M.; Aldenborg, F.; Almer, S.; Olsson, R. The AST/ALT ratio as an indicator of cirrhosis in patients with PBC. Liver Int. 2006, 26, 840–845. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Khan, I.A.; ur-Rehman, M.; Gilani, S.A.; Mehmood, Z.; Mubarak, M.S. Anticancer potential of quercetin: A comprehensive review. Phytother. Res. 2018, 32, 2109–2130. [Google Scholar] [CrossRef]
- Srivastava, A.; Ahmad, R.; Khan, M.A. Evaluation and comparison of the in vitro cytotoxic activity of Withania somnifera Methanolic and ethanolic extracts against MDA-MB-231 and Vero cell lines. Sci. Pharm. 2016, 84, 41–59. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.A. Lead-and drug-like compounds: The rule-of-five revolution. Drug Discov. Today Technol. 2004, 1, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Oyedemi, S.O.; Oyedemi, B.O.; Ijeh, I.I.; Ohanyerem, P.E.; Coopoosamy, R.M.; Aiyegoro, O.A. Alpha-amylase inhibition and antioxidative capacity of some antidiabetic plants used by the traditional healers in Southeastern Nigeria. Sci. World J. 2017, 2017, 3592491. [Google Scholar] [CrossRef] [PubMed]
- Gumuslu, E.; Cine, N.; Ertan, M.; Mutlu, O.; Komsuoglu Celikyurt, I.; Ulak, G. Exenatide upregulates gene expression of glucagon—Like peptide—1 receptor and nerve growth factor in streptozotocin/nicotinamide-induced diabetic mice. Fundam. Clin. Pharmacol. 2018, 32, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Erel, O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin. Biochem. 2004, 37, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Ogbole, O.O.; Segun, P.A.; Adeniji, A.J. In vitro cytotoxic activity of medicinal plants from Nigeria ethnomedicine on Rhabdomyosarcoma cancer cell line and HPLC analysis of active extracts. BMC Complement. Altern. Med. 2017, 17, 494. [Google Scholar] [CrossRef]
- Wang, J.; Tang, L.; White, J.; Fang, J. Inhibitory effect of gallic acid on CCl 4-mediated liver fibrosis in mice. Cell Biochem. Biophys. 2014, 69, 21–26. [Google Scholar] [CrossRef]
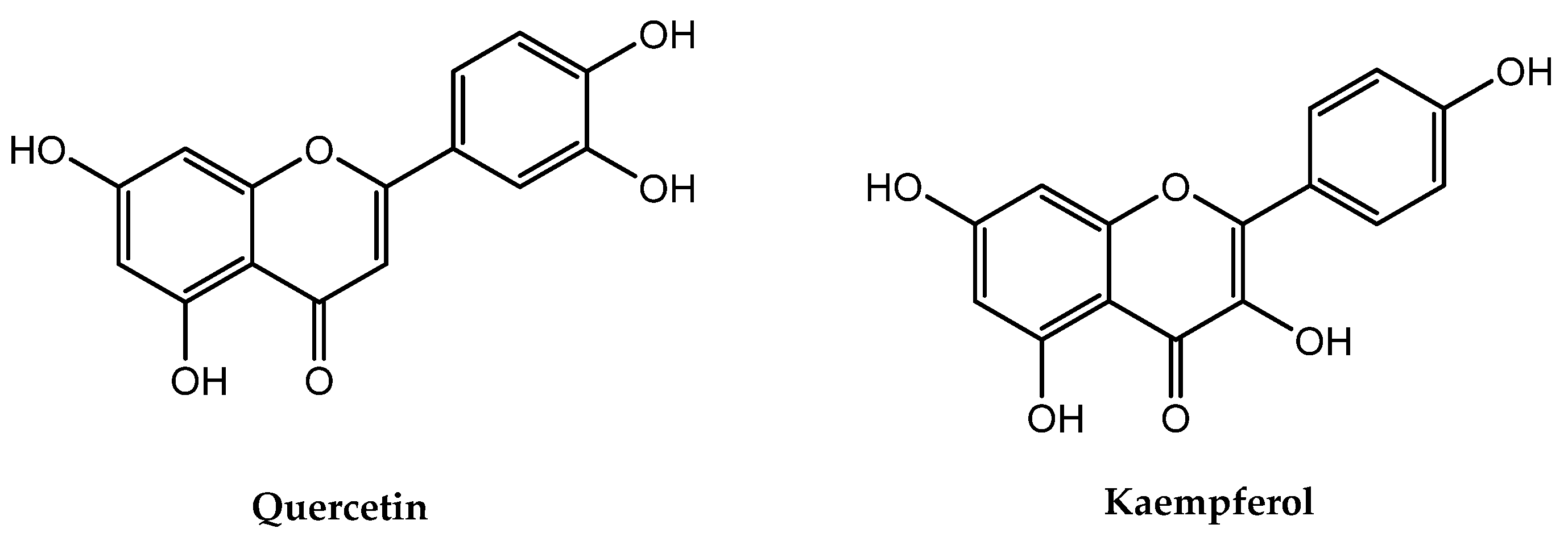
| Compounds | Mol. WT (g/mol) | HBD | HBA | RBN | LogP | A | Violation |
|---|---|---|---|---|---|---|---|
| Quercetin | 302.24 | 5 | 7 | 1 | 1.23 | 78.03 | 0 |
| Kaempferol | 286.24 | 4 | 6 | 1 | 1.58 | 76.01 | 0 |
| Metformin | 126.12 | 3 | 6 | 0 | −1.38 | 37 | 0 |
| Quercetin | Kaempferol | Metformin | |
|---|---|---|---|
| Absorption | |||
| BBB | − | − | − |
| HIA | + | + | + |
| CaCo2 permeability | − | − | − |
| PGS | − | − | − |
| PGI | − | − | − |
| ROCT | − | − | − |
| Metabolism | |||
| CYP3A4 Substrate | + | + | − |
| CYP2C9 Substrate | − | − | − |
| CYP2D6 Substrate | − | − | − |
| CYP3A4 Inhibition | + | + | − |
| CYP2C9 Inhibition | − | − | − |
| CYP2C19 Inhibition | − | + | − |
| CYP2D6 Inhibition | − | − | − |
| CYP1A2 Inhibition | + | + | − |
| Toxicity | |||
| Ames toxicity | NAT | NAT | NAT |
| Carcinogens | Non-carcinogenic | Non-carcinogenic | Non-carcinogenic |
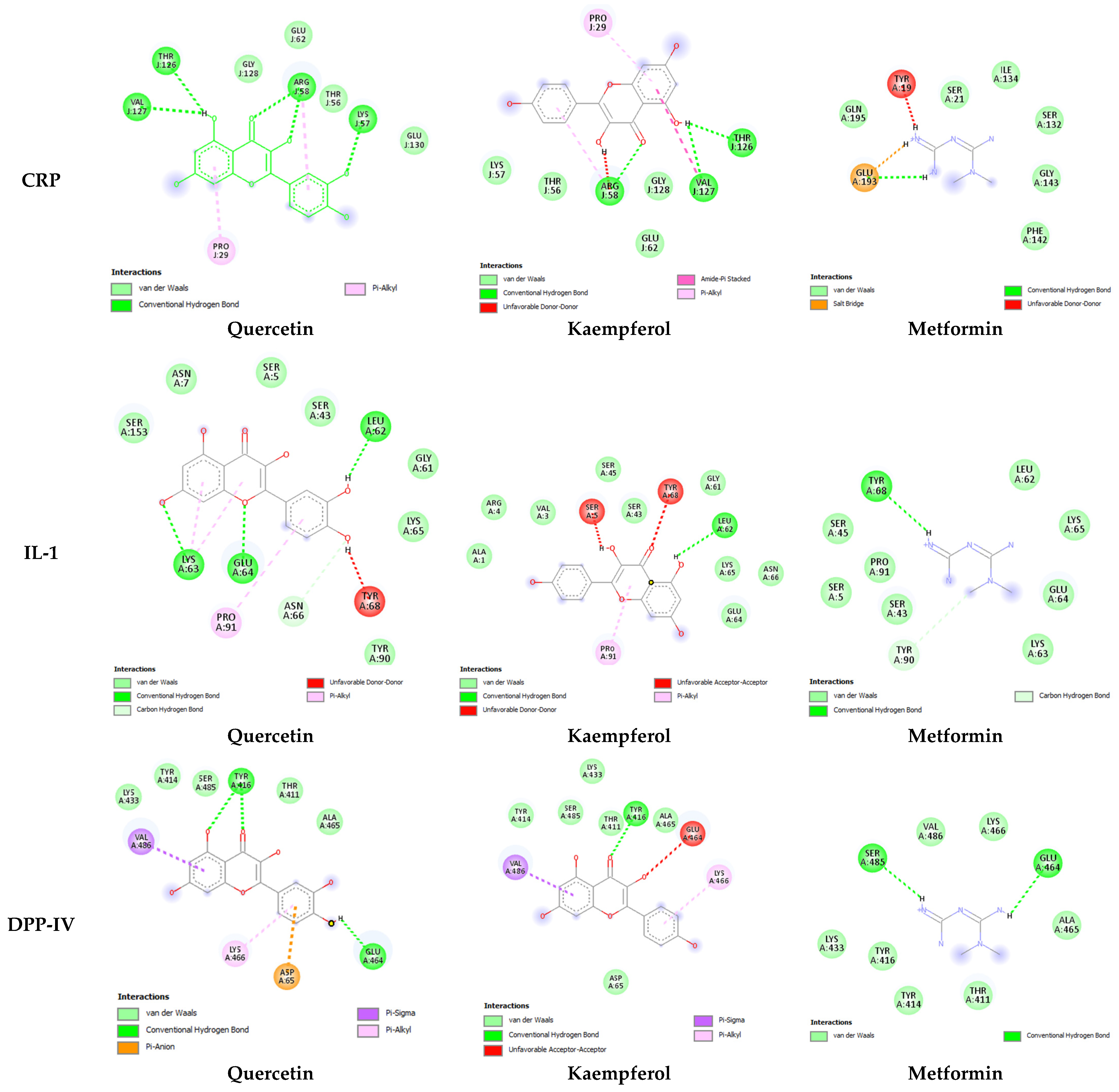
| Target Receptor | Ligand | Binding H-Bond Residues | No. of H-Bonds | |
|---|---|---|---|---|
| Name | Binding Energy (Kcal/mol) | |||
| CRP | Quercetin | −5.8 | Thr126, Arg58, Lys57, Val127, Glu62, Gly128, Glu130, Pro29 | 5 |
| Kaempferol | −5.6 | Arg58, Val127, Thr126, Lys57, Thr56, Gly128, Glu62, Pro29, Arg58 | 3 | |
| Metformin | −4.2 | Glu293, Gln145, Ser21, Ile134, Ser132 Gly143, Phe142 | 1 | |
| IL-1 | Quercetin | −7.4 | Leu62, Lys63, Glu64, Ser153, Asn7, Ser5, Ser43, Gly61, Lys65 Typ90, Pro91,Lys63 | 3 |
| Kaempferol | 6.8 | Leu62, Ala1, Arg4, Val3, Ser45, Ser,43 Gly61, Lys65, Asn66, Glu64, Ser5, Thre68 | 1 | |
| Metformin | −5 | Tyr68, Lu62, Lys65, Glu64, Lys63, Ser43, Pro91, Ser5, Ser54, Val486 | 1 | |
| DDPIV | Quercetin | −6.6 | Thr516, Glu464, Lys 433, Tyr414, Ser484, Thr411, Ala465 Glu464, Val486 | 2 |
| Kaempferol | −6.5 | Tyr416, Tyr414, Ser485, Lys433, Thr411, Ala465, Asp65, Lys566 | 1 | |
| Metformin | −4.3 | Ser485, Glu464, Lys433, Tyr416, Tyr414, Thr411, Ala465, Lys466, Val486 | 2 | |
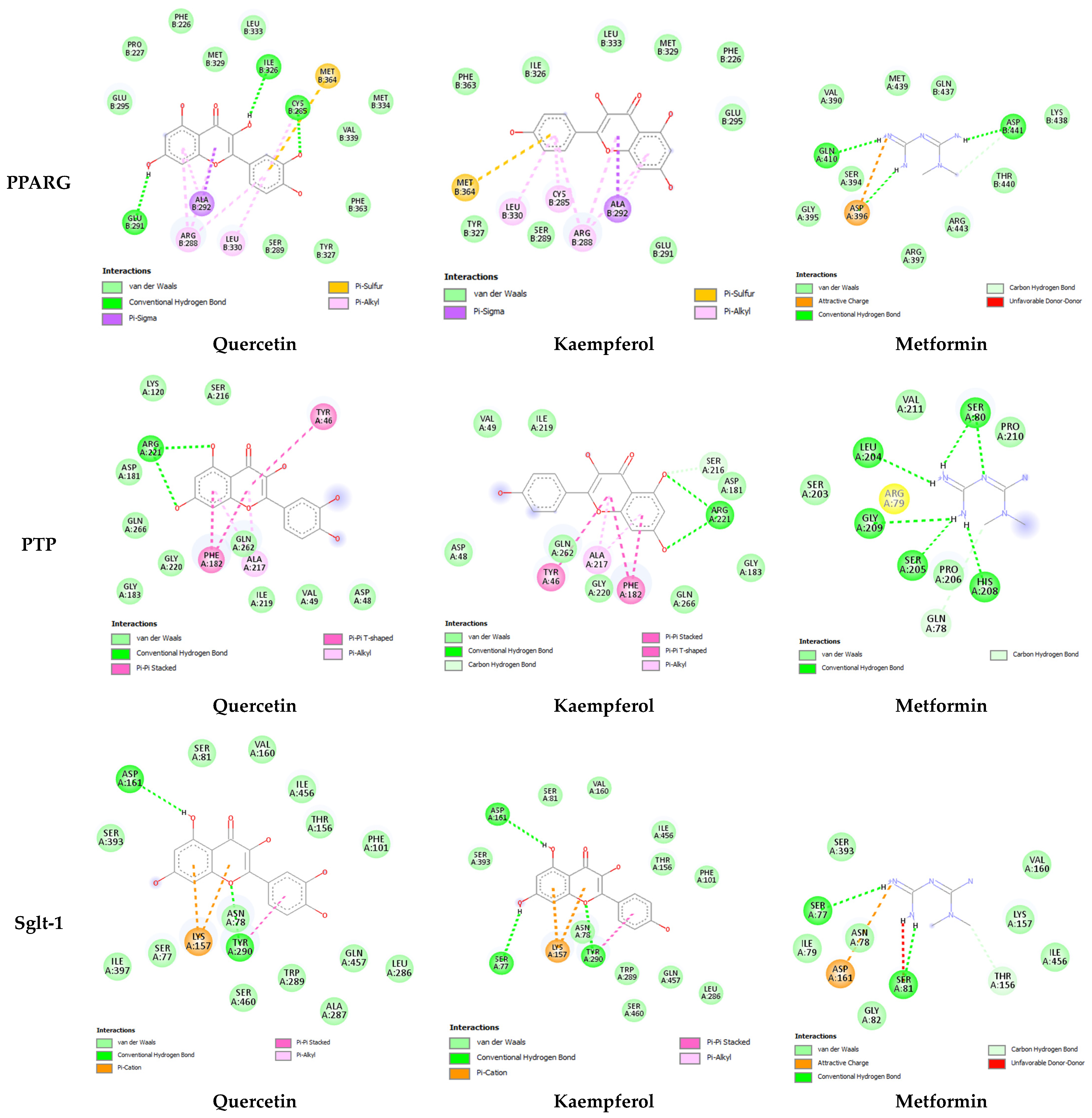
| PPRG | Quercetin | −8.4 | Ile326, Cys285, Glue291, Glu295, Pro227, Phe226, Met329, Lue333, Met334, Val339, Phe363, Tyr321, Ser289, Arg288, Leu330, Ala292 | 3 |
| Kaempferol | −8.3 | Phe363, Ile326, Lue333, Met329, Phe226, Glu295, Glu291, Ser284, Tyr327, Arg288, Cys285, Leu330, Ala292 | -- | |
| Metformin | −5.3 | Gln410, Asp441, Asp396, Val390, Met439, Gln437 | 3 | |
| PTP | Quercetin | 8.2 | Arg221, Lys120, Ser216, Asp181, Gln266, Gly220, Gln262, Gly183, Ile219, Val64, Asp48, Phe182, Tyr46, Ala217 | 2 |
| Kaempferol | −8.3 | Arg221, Val49, Ile219, Ser216, Asp181, Gly183, Gln226, Gly220, Gln261, Asp48 | 2 | |
| Metformin | 4.8 | Ser80, Leu204, Gly209, Ser205, His208, Val221, pro210, Ser203, Arg79, Pro206, Gln78 | 6 | |
| SGLT-1 | Quercetin | −8.4 | Asp161, Tyr290, Ser81, Val160, Ile456, Thr156, Phe101, Gln457, Leu286, Trp289, Ala287, Ser72, Ile397, Ser393 | 2 |
| Kaempferol | −8.4 | Asp161, Ser71, Tyr290, Ser81, Val160, Ile456, Thr156, Phe101, Ser393, Asn78, Trp289, Gln457, Leu286, Ser460 | 3 | |
| Metformin | −5.3 | Ser77, Ser81, Ser393, Val160, Lys157, Ile456, Thr156, Gly82, Asn78, Ile79 | 2 | |
| Treatments | ALT (IU/L) | ALP (IU/L) | Bilirubin (mg/dL) |
|---|---|---|---|
| Control | 69.0 ± 11 | 87.3 ± 6.3 | 0.55 ± 0.03 |
| STZ-NA + Veh | 80.5 ± 17 | 89 ± 07 | 0.64 ± 0.04 |
| STZ-NA + Quer | 68.0 ± 07 | 79.5 ± 2 | 0.605 ± 0.02 |
| STZ-NA + Kaem | 78.5 ± 08 | 78.5 ± 12 | 0.565 ± 0.11 |
| STZ-NA + Quer + Kaem | 85 ± 6.8 | 93 ± 15 | 0.625 ± 0.01 |
| STZ-NA + Met | 80.5 ± 18.9 | 92 ± 14 | 0.47 ± 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, M.; Hassan, M.; Ansari, S.A.; Alkahtani, H.M.; Al-Rasheed, L.S.; Ansari, S.A. Quercetin and Kaempferol as Multi-Targeting Antidiabetic Agents against Mouse Model of Chemically Induced Type 2 Diabetes. Pharmaceuticals 2024, 17, 757. https://doi.org/10.3390/ph17060757
Ali M, Hassan M, Ansari SA, Alkahtani HM, Al-Rasheed LS, Ansari SA. Quercetin and Kaempferol as Multi-Targeting Antidiabetic Agents against Mouse Model of Chemically Induced Type 2 Diabetes. Pharmaceuticals. 2024; 17(6):757. https://doi.org/10.3390/ph17060757
Chicago/Turabian StyleAli, Muhammad, Mudassir Hassan, Siddique Akber Ansari, Hamad M. Alkahtani, Lamees S. Al-Rasheed, and Shoeb Anwar Ansari. 2024. "Quercetin and Kaempferol as Multi-Targeting Antidiabetic Agents against Mouse Model of Chemically Induced Type 2 Diabetes" Pharmaceuticals 17, no. 6: 757. https://doi.org/10.3390/ph17060757






